Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.114009
Revised: October 14, 2025
Accepted: December 12, 2025
Published online: March 19, 2026
Processing time: 172 Days and 7 Hours
This editorial examines the constraints and novel approaches to management of cervical spondylosis patients with generalized anxiety disorder. Cervical spon
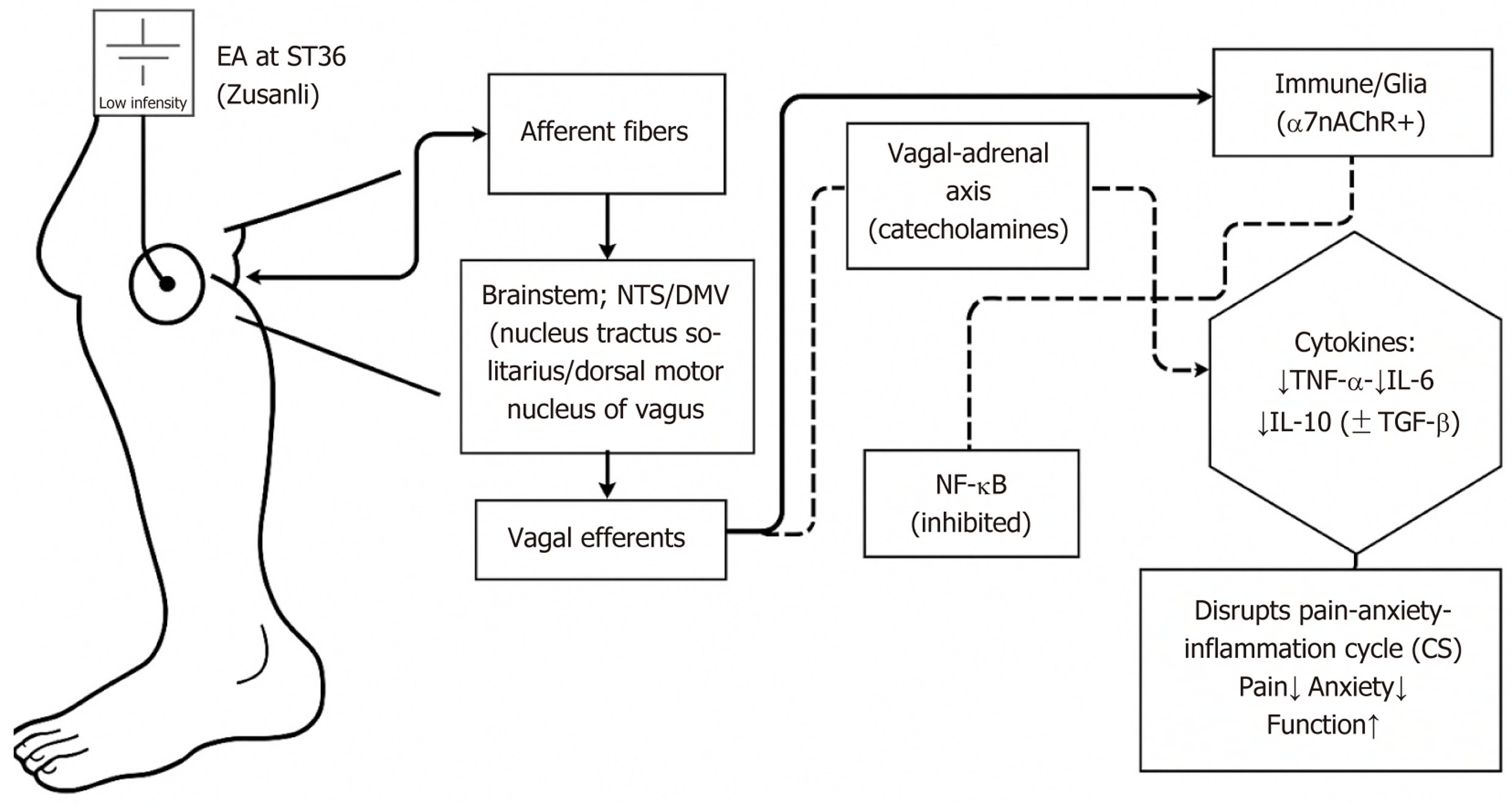
Core Tip: Cervical spondylosis is frequently comorbid with generalized anxiety disorder, creating a loop of pain-anxiety-inflammation that complicates therapy. This editorial proposes electroacupuncture (EA) as a possible treatment that can reduce pain, anxiety, and functional impairment in chronic stress patients with generalized anxiety disorder by influencing neural inflammation and the release of endogenous opioids. EA aids in regulating inflammatory signals and restoring brain function, complementing conventional therapy. Due to its superior safety profile and potential cost savings, EA offers a novel, evidence-based approach to enhance quality of life, decrease medication usage, and alleviate both physical and mental symptoms in these patients.
- Citation: Karmakar R, Kandalkar A, Wang HC, Mukundan A. Redefining pain and mental health management in cervical spondylosis: Electroacupuncture as a neuroinflammatory modulator and multimodal therapeutic innovation. World J Psychiatry 2026; 16(3): 114009
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/114009.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.114009
The study by Li et al[1], evaluated electroacupuncture (EA) in a retrospective cohort of 83 patients with cervical spondylosis (CS)-related pain and comorbid generalized anxiety disorder (GAD). After 8 weeks of EA treatment, patients showed significant reductions in pain, anxiety symptoms, and key neuroinflammatory markers such as interleukin (IL)-6 and tumor necrosis factor-alpha (TNF-α). Improvements in pain were strongly correlated with decreases in anxiety and inflammation. These findings suggest that EA may be an effective integrative therapy that simultaneously targets pain, anxiety, and neuroinflammatory processes in this population. CS is a common degenerative condition of the cervical spine, marked by substantial degeneration of intervertebral discs, facet joints, and surrounding ligamentous structures. The overall frequency rates vary from 30% to 50% within middle-aged and older demographics, contributing to a signi
Li et al’s work[1] demonstrates significant clinical implications for managing CS-related discomfort in patients with concurrent GAD. The shown efficacy of EA in simultaneously alleviating physical pain and psychological symptoms signifies a paradigm change towards integrated, multimodal treatment strategies that acknowledge the complex bidirectional link between chronic pain and anxiety disorders. The substantial reduction in pain intensity and functional dysfunction, symptomatic of alleviation and enhancement of patients’ daily functioning, is achievable through EA and its compromise in CS. The concurrent alleviation of anxiety symptoms, without altering pharmacological anxiolytic regimens, demonstrates EA’s efficacy as a beneficial treatment for the psychological distress commonly associated with chronic pain. Conventional treatment is inadequate for simultaneously addressing both anxiety and pain, often failing to manage them concurrently. Additionally, pharmacological interactions can impact and restrict prescription usage, com
Comorbid GAD with CS imposes significant socioeconomic difficulties due to its prevalence, resulting in long-term incapacity and health-related expenses. Indirect losses may occur for both individuals and society, often manifesting as decreased productivity, absenteeism, and worse quality of life. EA has demonstrated therapeutic efficacy and offers socioeconomic advantages by reducing reliance on costly medications and invasive procedures that predominantly carry risks and side effects. EA has reduced health care utilization associated with managing medication reactions or surgical complications due to its robust safety profile and few adverse effects. EA has significantly enhanced disability associated with chronic pain, demonstrating therapeutic advantages that last beyond the present therapy period, underscoring its clinical significance[9]. By alleviating both pain and anxiety symptoms, EA enhances functional capacity, facilitates an earlier return to work, and increases engagement in society. This may also aid in reducing chronicity, diminishing long-term disability, and reducing the larger socioeconomic load when administered early, particularly in patients with a shorter symptom duration. The extensive accessibility and integration of EA into collaborative care may reduce dis
For clinical translation, EA protocols must explicitly define acupoint selection, include at least one point with dem
This study primarily examines the clinical efficacy of EA for CS with GAD, while also emphasizing the broader socio
This study identifies EA as an effective method for reducing chronic pain associated with CS and concurrent GAD by addressing the neuroinflammatory process. The results demonstrate substantial enhancement in physical function, psychological distress, and inflammatory markers, corroborating the multiple therapeutic effects of EA. This approach not only improves daily functioning but also reduces reliance on medicine and its attendant hazards. Clinically, EA represents a more promising multimodal intervention. Economical and accessible therapy that reduces productivity losses and enhances healthcare equity, resulting in a substantial impact, is achievable using EA. Early EA integration can considerably reduce chronicity and long-term handicap, alleviating the social load. Integrating EA into multidisciplinary care may improve patient adherence, satisfaction, and sustainable healthcare delivery[1]. However, the limitations include the necessity for extensive investigations and the examination of effects beyond the 8-week intervention period. Future research must identify patient benefits, optimize therapies, and evaluate cost-effectiveness across various heal
| 1. | Li Z, Gao Y, Yan D, Feng YH. Efficacy of electroacupuncture for cervical spondylosis-related pain in patients with generalized anxiety disorder. World J Psychiatry. 2025;15:108288. [PubMed] [DOI] [Full Text] |
| 2. | Echeverria-Villalobos M, Tortorici V, Brito BE, Ryskamp D, Uribe A, Weaver T. The role of neuroinflammation in the transition of acute to chronic pain and the opioid-induced hyperalgesia and tolerance. Front Pharmacol. 2023;14:1297931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 3. | Gagnon CM, Scholten P, Atchison J, Jabakhanji R, Wakaizumi K, Baliki M. Structural MRI Analysis of Chronic Pain Patients Following Interdisciplinary Treatment Shows Changes in Brain Volume and Opiate-Dependent Reorganization of the Amygdala and Hippocampus. Pain Med. 2020;21:2765-2776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 4. | Bhagar R, Le-Niculescu H, Corey SC, Gettelfinger AS, Schmitz M, Ebushi A, Matei E, Woods C, Mullen J, Kurian SM, Shekhar A, White FA, Niculescu AB. Next-generation precision medicine for pain. Mol Psychiatry. 2026;31:869-894. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 5. | Zhang Q, Zhou M, Huo M, Si Y, Zhang Y, Fang Y, Zhang D. Mechanisms of acupuncture-electroacupuncture on inflammatory pain. Mol Pain. 2023;19:17448069231202882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 6. | Zhou Y, Wang W, Tian K, Huang H, Jia M. Efficacy and safety of electroacupuncture in treatment of cervical spondylosis: A protocol of randomized controlled trial. Medicine (Baltimore). 2021;100:e25570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Li W, Liu X, Lin J, Huang J, Li S, Xu N, Fu W, Liu J. Clinical Efficacy of Electroacupuncture in the Treatment of Chronic Neck Pain: A Randomized Clinical Trial. J Pain Res. 2025;18:2909-2922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | Wang C, Liu Y, Li L, Zhang H, Ye Z, Zhao L. The efficacy of electroacupuncture for cervical nerve edema and movement disorder caused by the brachial plexus injury: a case report. Front Neurol. 2024;15:1342844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Kong JT, Puetz C, Tian L, Haynes I, Lee E, Stafford RS, Manber R, Mackey S. Effect of Electroacupuncture vs Sham Treatment on Change in Pain Severity Among Adults With Chronic Low Back Pain: A Randomized Clinical Trial. JAMA Netw Open. 2020;3:e2022787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 10. | Lu M, Sharmin S, Tao Y, Xia X, Yang G, Cong Y, Yang G, Razavilar N, Aziz R, Jiang J, Xiao Y, Peng L, Xu B. Economic evaluation of acupuncture in treating patients with pain and mental health concerns: the results of the Alberta Complementary Health Integration Project. Front Public Health. 2024;12:1362751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Davies BM, Phillips R, Clarke D, Furlan JC, Demetriades AK, Milligan J, Witiw CD, Harrop JS, Aarabi B, Kurpad SN, Guest JD, Wilson JR, Kwon BK, Vaccaro AR, Fehlings MG, Rahimi-Movaghar V, Kotter MRN. Establishing the Socio-Economic Impact of Degenerative Cervical Myelopathy Is Fundamental to Improving Outcomes [AO Spine RECODE-DCM Research Priority Number 8]. Global Spine J. 2022;12:122S-129S. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/