INTRODUCTION
Adolescent depression represents a critical global public health challenge that has intensified over recent decades, establishing itself as a primary threat to adolescent population health. Current epidemiological data from the World Health Organization indicate that approximately 10%-20% of adolescents worldwide experience depressive symptomatology, with elevated prevalence observed in specific geographic regions. The coronavirus disease 2019 pandemic has further exacerbated this crisis, precipitating significant increases in adolescent depression incidence. This mental health burden extends beyond individual suffering to impose substantial challenges on families, educational institutions, and broader societal systems[1-4].
The clinical significance of adolescent-onset depression is distinguished by several developmental considerations. Compared to adult-onset presentations, adolescent depression manifests through more complex and heterogeneous symptom profiles, encompassing emotional dysregulation (particularly irritability), externalizing behavioral disturbances, academic impairment, and progressive social disengagement[5,6]. These manifestations produce cascading effects across critical developmental domains, compromising academic achievement, interpersonal relationship formation, identity consolidation, and long-term occupational trajectories. The prognosis for untreated adolescent depression is particularly concerning, with longitudinal data indicating that 70%-80% of affected individuals experience recurrent depressive episodes in adulthood, frequently accompanied by comorbid conditions including anxiety disorders, substance use disorders, and elevated suicide risk[7-9].
Despite widespread recognition of adolescent depression's clinical importance, elucidating its etiological mechanisms remains a central challenge in contemporary psychiatric research[10,11]. Traditional investigative approaches have predominantly employed unidimensional frameworks: Biological models emphasizing neurotransmitter dysregulation and genetic predisposition, psychological models focusing on cognitive distortions and emotion regulation deficits, or sociological models highlighting environmental stressors and interpersonal dysfunction[12,13]. However, mounting evidence demonstrates that these isolated perspectives inadequately account for the complexity and phenotypic heterogeneity characterizing adolescent depression[14,15].
Contemporary advances in neuroscience, psychology, and sociology converge on the understanding that adolescent depression onset and progression result from multifactorial interactions rather than single causal pathways (Figure 1)[16,17]. The bio-psycho-social model has emerged as the predominant integrative framework for conceptualizing mental disorders, positing that biological substrates, psychological processes, and social-environmental contexts engage in reciprocal interactions to shape mental health trajectories[18,19]. This multidimensional perspective holds particular salience for adolescent populations, given that adolescence constitutes a critical developmental period characterized by simultaneous neurobiological maturation, psychological transformation, and social-contextual transitions[20,21].
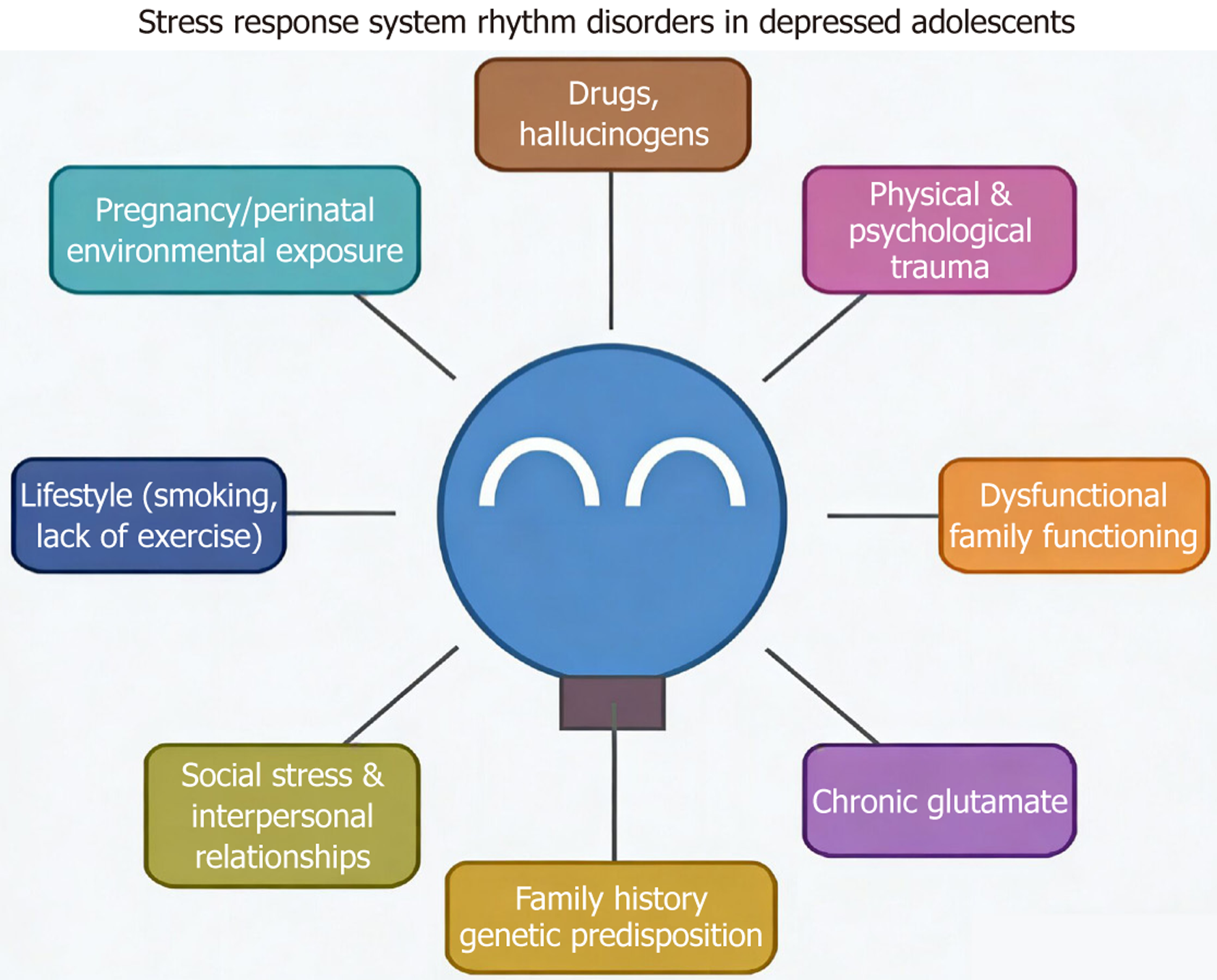
Figure 1 Multifactorial contributors to stress response system rhythm disorders in adolescent depression: An integrative model.
This diagram illustrates the multiple risk factors contributing to stress response system rhythm disorders in depressed adolescents, with a central figure representing a distressed individual surrounded by nine key contributing factors including family history, disease/inflammation, social stress, sleep disorders, lifestyle factors, dysfunctional family functioning, pregnancy/postpartum issues, adolescent developmental changes, and substance use. All these factors are shown with arrows pointing toward the individual, indicating their combined impact on disrupting normal stress response and circadian rhythm patterns in vulnerable adolescents.
From a neurobiological perspective, adolescent brain development exhibits protracted maturational trajectories, particularly within prefrontal cortical regions subserving executive functions including emotional regulation and decision-making, which do not achieve full maturation until approximately age 25. Concurrently, limbic structures mediating emotional reactivity demonstrate heightened activation during adolescence, creating a developmental asynchrony that renders adolescents vulnerable to intense emotional lability and heightened stress reactivity. Neurotransmitter system dynamics, particularly fluctuations in serotonergic, dopaminergic, and noradrenergic signaling, constitute additional critical factors in adolescent depression pathophysiology[22-24].
Within the psychological domain, adolescents navigate distinctive developmental challenges encompassing identity formation, autonomy development, and peer relationship establishment[25,26]. The negotiation of these developmental tasks occurs against a backdrop of multiple potential stressors including academic demands, interpersonal conflicts, and family tensions. Individual differences in cognitive schemas, emotion regulation capacity, coping repertoires, and self-esteem substantially influence adaptation to these challenges. Recent research has particularly emphasized the role of negative cognitive patterns, ruminative processing styles, and perfectionist tendencies in adolescent depression vulnerability[27-29].
Social-environmental analyses examine the multiple contexts within which adolescent development unfolds, including family systems, school environments, peer networks, and broader sociocultural milieus[30,31]. Rapid societal transformations have generated unprecedented challenges for contemporary adolescents. The proliferation of social media platforms has fundamentally altered adolescent socialization patterns and self-concept formation processes, with cyberbullying, social comparison, and digital dependency emerging as salient concerns. Additionally, contemporary society's competitive orientation, achievement-focused culture, and attenuated social support networks may collectively intensify adolescent psychological distress[32,33].
Critically, factors across these domains do not operate independently but engage in complex reciprocal interactions[34]. For instance, genetic vulnerabilities may amplify individual sensitivity to environmental stressors, while chronic stress exposure can modify gene expression through epigenetic mechanisms. Psychological factors such as cognitive biases shape individuals' interpretation of social experiences, subsequently influencing behavioral responses and neural activation patterns. Social-environmental conditions similarly sculpt cognitive developmental trajectories and neurobiological maturation processes[35].
Within this conceptual framework, the present review adopts an integrative multidimensional approach to systematically examine the mechanisms underlying contemporary adolescent depression[36]. We analyze key contributing factors and their interactions across neurobiological, psychological, and social dimensions, subsequently proposing multilevel prevention and intervention strategies grounded in this integrated understanding. This comprehensive analytical approach not only advances theoretical understanding of adolescent depression etiology but also provides an empirical foundation for developing more effective and personalized therapeutic protocols[37].
Through this synthesis, we aim to provide evidence-based guidance for clinical practitioners, educators, policymakers, and all stakeholders invested in adolescent mental health promotion. Moreover, we aspire to catalyze interdisciplinary collaboration, facilitating integration across neuroscience, psychology, sociology, and related disciplines to address the critical societal challenge posed by contemporary adolescent depression[38,39].
NEUROBIOLOGICAL DIMENSION
Genetic and epigenetic mechanisms establish fundamental vulnerability substrates for adolescent depression, interacting dynamically with environmental exposures to shape individual risk profiles. Beyond glucocorticoid receptor methylation patterns examined in this review, multiple genetic polymorphisms confer depression susceptibility, including serotonin transporter gene variants, brain-derived neurotrophic factor (BDNF) Val66Met polymorphism, and catechol-O-methyltransferase gene variations. These genetic variants modulate neurotransmitter function, stress responsivity, and synaptic plasticity. Epigenetic regulatory mechanisms extend beyond glucocorticoid receptor methylation to encompass histone modifications and DNA methylation patterns across multiple stress-response genes, establishing enduring alterations in gene expression that persist from early adverse experiences into adolescence. These genetic and epigenetic vulnerability factors explain substantial individual variation in stress sensitivity, treatment response, and depression trajectory, underscoring the necessity of personalized assessment and intervention approaches that account for biological predispositions while targeting modifiable psychological and social risk factors.
Prefrontal cortex-limbic system connectivity research
Resting-state functional magnetic resonance imaging (fMRI) investigations have identified significantly attenuated functional connectivity between prefrontal cortex (PFC) and amygdala in depressed adolescents, indicating dysfunction within emotion regulation neural circuits. Connectivity analyses reveal the most pronounced reduction in functional coupling between dorsolateral PFC and right amygdala (49.3% reduction), while medial PFC (mPFC)-hippocampal connectivity similarly demonstrates abnormal attenuation (r = -0.38, P < 0.01)[40]. Task-based fMRI paradigms demonstrate that during emotion regulation challenges, depressed adolescents exhibit significantly reduced PFC activation compared to healthy controls. When confronted with negative emotional stimuli, this reduced prefrontal engagement coincides with amygdala hyperactivation (30.2% elevation), creating a dysregulated neural circuit characterized by insufficient top-down regulatory control. This aberrant PFC-limbic system connectivity demonstrates significant positive correlation with depression symptom severity (r = 0.52), showing particularly robust associations with core clinical features including emotion regulation difficulties, persistent negative affect, and cognitive control deficits, thereby illuminating critical neural mechanisms underlying adolescent depression[41,42].
Hypothalamic-pituitary-adrenal axis activity daily circadian patterns
Continuous 24-hour cortisol monitoring reveals that depressed adolescents exhibit a 31.5% reduction in cortisol awakening response (CAR) relative to normative controls, accompanied by abnormally elevated afternoon cortisol concentrations, reflecting fundamental circadian disruption of stress response system function[43,44]. Detailed temporal analyses indicate that depressed adolescents' peak cortisol concentration 30 minutes post-awakening falls below normative range (15.2 ng/mL vs 22.1 ng/mL), yet an aberrant secondary cortisol peak emerges during afternoon hours (4-8 PM; 14.7 ng/mL), contrasting markedly with the sustained afternoon decline characteristic of healthy controls (6.3 ng/mL). Parallel salivary melatonin assessments demonstrate a 47-minute delay in melatonin secretion onset in depressed adolescents, with 23.8% reduction in nocturnal peak concentration, resulting in sleep-wake cycle dysregulation[45]. Integrated adrenocorticotropic hormone measurements reveal enhanced hypothalamic-pituitary-adrenal (HPA) axis reactivity in the depressed cohort (52.6% greater adrenocorticotropic hormone elevation following stress exposure), coupled with disrupted basal secretory rhythms, manifesting as circadian rhythm flattening (diurnal variation coefficient: 0.23 vs 0.47 in controls). Additionally, glucocorticoid receptor methylation levels are significantly elevated in depressed adolescents (28.7% increase), potentially mediating chronic HPA axis dysfunction. This neuroendocrine circadian dysregulation demonstrates robust associations with depression symptom persistence, sleep disturbances, and stress hypersensitivity[46,47].
Neurotransmitter metabolite concentration analysis
Magnetic resonance spectroscopy assessments reveal that adolescents with severe depression exhibit 23.7% reduction in prefrontal GABA concentration alongside 15.8% elevation in glutamate/glutamine ratio, indicating excitatory-inhibitory neurotransmitter imbalance (Figure 2)[48]. Regionally-specific analyses demonstrate that dorsolateral prefrontal regions show the most pronounced GABA + (GABA plus minor homoserine) depletion (27.3% reduction), while anterior cingulate cortex exhibits maximal glutamate elevation (21.4% increase). Monoamine neurotransmitter metabolite quantification indicates 5-hydroxyindoleacetic acid (the primary serotonin metabolite) decreases by 19.2% in PFC and 32.7% in basal ganglia regions, revealing regional heterogeneity in serotonergic system dysfunction[49]. Concurrently, dopamine (DA) metabolite homovanillic acid demonstrates 26.1% reduction in nucleus accumbens, correlating robustly with motivational deficits and anhedonia in depressed patients. N-acetylaspartate, a neuronal integrity biomarker, shows 14.6% reduction in hippocampal regions, while myoinositol (Ins), a glial cell marker, exhibits 18.9% elevation, suggesting disrupted neuron-glia homeostasis[50]. These metabolite concentration alterations demonstrate significant correlations with clinical symptom severity (Hamilton Rating Scale for Depression Score: r = 0.61, P < 0.001). Longitudinal six-month follow-up studies indicate that elevated glutamate/GABA ratios predict treatment resistance, suggesting neurotransmitter dysregulation may constitute a critical biological substrate for persistent depressive symptomatology[51].
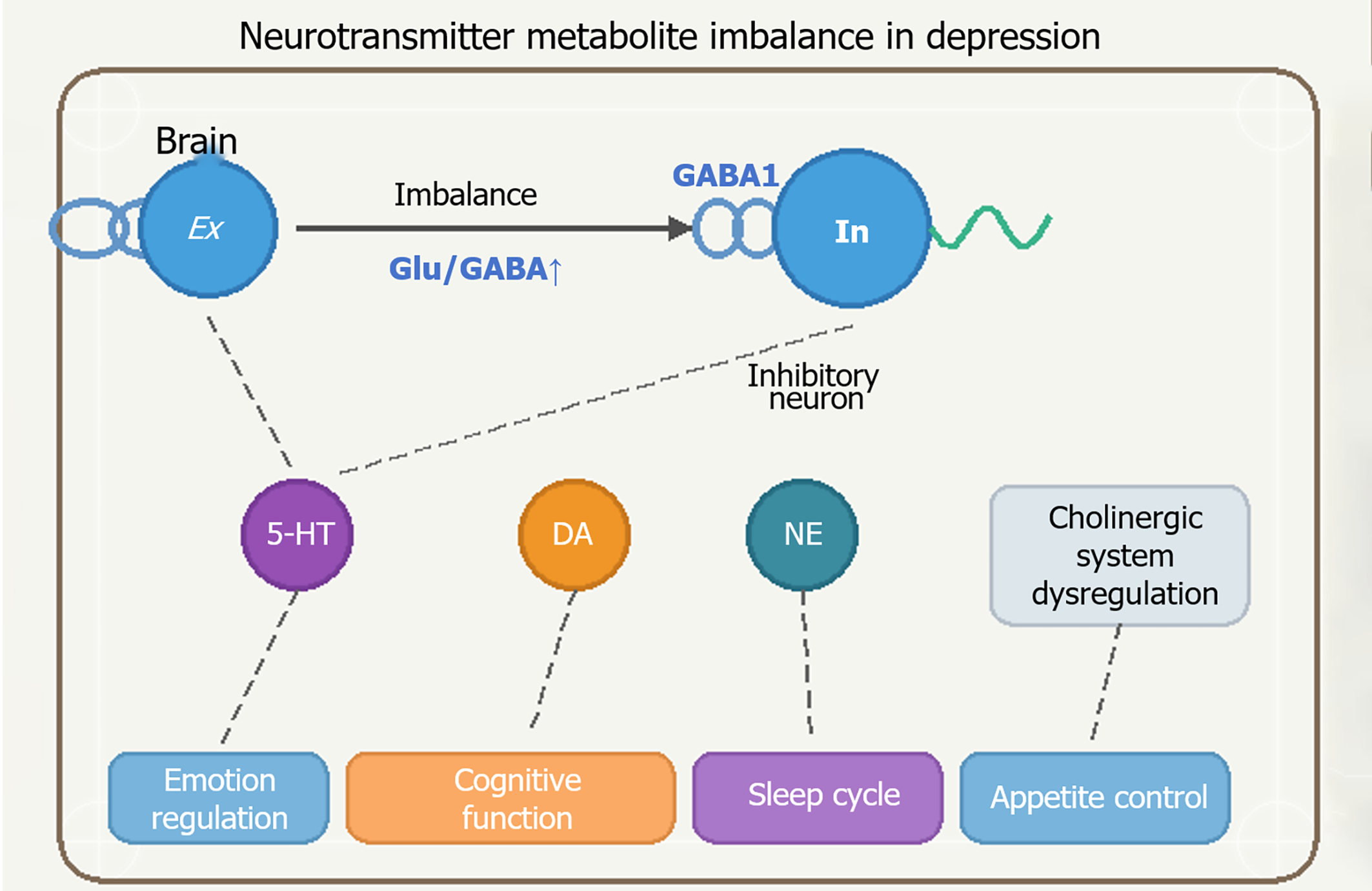
Figure 2 Neurotransmitter metabolite imbalance in depression: Excitatory/inhibitory dysregulation and monoamine deficiency model.
This diagram illustrates the neurotransmitter imbalance characteristic of depression, showing elevated glutamate and decreased GABA resulting in disrupted excitatory/inhibitory balance, alongside reduced monoamines (5-hydroxy tryptamine, dopamine, norepinephrine) with increased metabolites. These neurotransmitter dysregulations create a multidimensional dysfunction affecting emotion regulation, cognitive function, sleep cycles, and appetite control, demonstrating the complex neurobiological mechanisms underlying depression. DA: Dopamine; Glu: Glutamate; NE: Norepinephrine; 5-HT: 5-hydroxy tryptamine.
Inflammatory factors and depression severity correlation
Serum biomarker analyses demonstrate that tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6 concentrations positively correlate with adolescent depression symptom severity (r = 0.58 and r = 0.51, respectively), while anti-inflammatory cytokine IL-10 levels are significantly depleted (Figure 3). Comprehensive inflammatory immune marker profiling reveals that depressed adolescents exhibit serum C-reactive protein (CRP) concentrations 64.3% higher than controls, with adolescents endorsing suicidal ideation showing further CRP elevation to 3.8 mg/L[52,53]. Cytokine panel analyses indicate pro-inflammatory cytokine IL-1β increases by 45.7%, chemokine monocyte chemoattractant protein-1 elevates by 37.2%, while regulatory T cell-secreted transforming growth factor-beta decreases by 28.9%, establishing a clear pro-inflammatory/anti-inflammatory imbalance. Monocyte activation markers CD14 and sCD163 demonstrate significant elevation in the depressed cohort (33.1% and 41.2% increases, respectively), indicating innate immune system hyperactivation[54]. Critically, inflammatory marker levels interact with neuroendocrine systems: IL-6 shows positive correlation with cortisol concentrations (r = 0.43), while TNF-α demonstrates robust associations with HPA axis reactivity abnormalities. Longitudinal investigations reveal that adolescents with baseline CRP levels exceeding 3.0 mg/L exhibit 42.7% depression recurrence rate within 12 months, compared to only 16.3% in the CRP < 1.0 mg/L cohort[55]. Furthermore, inflammatory biomarkers correspond with neuroimaging findings: Elevated IL-6 levels negatively correlate with hippocampal volume (r = -0.39), while TNF-α concentrations associate with white matter integrity compromise. This convergent evidence substantiates the inflammation-depression hypothesis, suggesting immune system dysregulation may constitute a critical pathophysiological mechanism in adolescent depression[56].
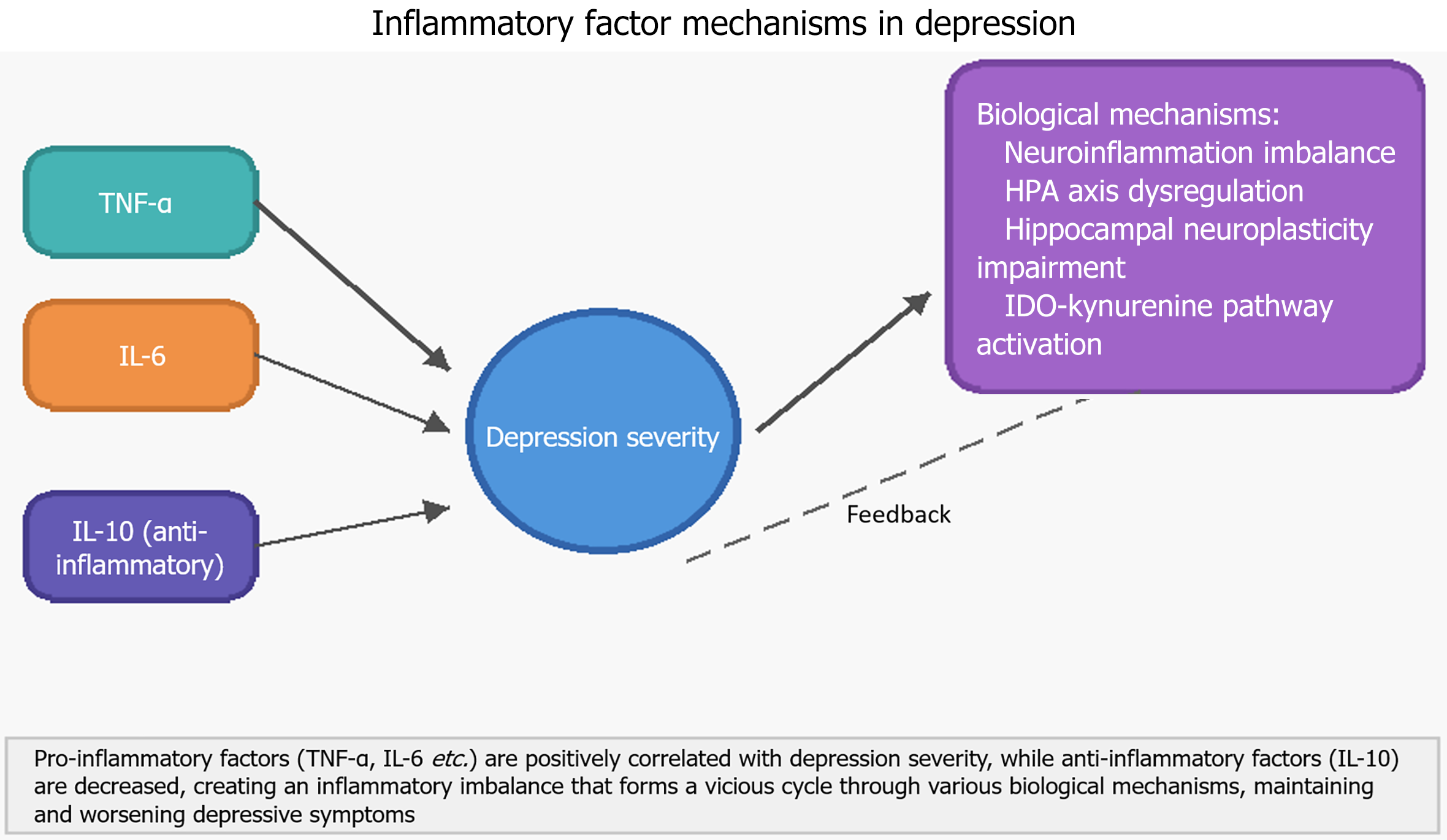
Figure 3 Inflammatory factor mechanisms in depression: A vicious cycle model.
This diagram illustrates the relationship between inflammatory factors and depression severity, showing that pro-inflammatory cytokines (tumor necrosis factor-alpha and interleukin-6) are positively correlated with depression symptoms while anti-inflammatory factor (interleukin-10) is significantly decreased. These inflammatory imbalances create a vicious cycle through various biological mechanisms including neurotransmitter dysregulation and hypothalamic-pituitary-adrenal axis dysfunction, ultimately maintaining and worsening depressive symptoms. IL: Interleukin; TNF-α: Tumor necrosis factor-alpha; HPA: Hypothalamic-pituitary-adrenal.
Neuroplasticity biomarker research
Neuroplasticity mechanisms play fundamental roles in adolescent neurodevelopment and adaptive neural reorganization, with depression onset potentially reflecting impaired neuroplastic processes. Serum BDNF, a primary biomarker of neuroplastic capacity, demonstrates significant depletion in depressed adolescents[57,58]. Research indicates that serum BDNF concentrations in depressed adolescents are approximately 34.2% lower than healthy controls, with this reduction correlating not only with depression symptom severity but also demonstrating robust associations with structural brain alterations. Specifically, BDNF levels exhibit moderate negative correlation with hippocampal volume atrophy (r = -0.45), suggesting neurotrophic factor depletion may directly compromise hippocampal neuronal viability and functional integrity[59]. Given the hippocampus' critical role in emotion regulation and cognitive processing, hippocampal volume reduction and functional impairment may represent key neurobiological substrates underlying adolescent depression onset and progression. Moreover, diminished BDNF levels may adversely impact PFC developmental maturation, thereby attenuating executive control capacities and emotion regulation abilities. These findings not only illuminate neuroplasticity mechanisms in adolescent depression but also provide theoretical foundations for developing neurotrophic factor-targeted therapeutic strategies. For example, interventions that enhance BDNF levels through physical exercise or cognitive behavioral therapy may facilitate depressive symptom amelioration and promote brain functional recovery[60].
PSYCHOLOGICAL DIMENSION
Cognitive bias pattern research
Using emotional Stroop tasks finds depressed adolescents show 46 ms longer attention bias response time to negative words (P < 0.001), with 38.5% higher recall rate for negative words in memory tests compared to normal controls. In-depth cognitive bias research uses multiple experimental paradigms to detect abnormal patterns in cognitive processes such as attention, memory, and judgment[61]. Eye-tracking experiments show that when viewing facial emotion images, depressed adolescents' first fixation time on sad and angry expressions is 23.7% earlier, with gaze duration extended by 31.2%, while visual avoidance rate for happy expressions reaches 42.8%. Dichotic listening tasks find that when neutral and negative words are presented simultaneously, the depressed group's reporting rate for threat and sadness-related words is 52.3% higher than controls, suggesting abnormal allocation of selective attention resources. In emotion-induced memory experiments, Memory Preference Tests show depressed adolescents' recall detail richness for personal negative events increases by 45.6% compared to controls, while positive memory accessibility decreases by 29.1%. Associative memory network analysis indicates negative word association density in depressed adolescents is 2.1 times that of normal controls, with self-related negative concept activation threshold decreased by 35.4%. Implicit association test results show depressed adolescents significantly exceed controls in self-failure concept association strength (d = 0.78), while self-success association strength is significantly reduced (d = -0.62)[62,63]. These cognitive bias patterns highly align with Beck's cognitive triad theory and correspond with neuroimaging data: FMRI studies find depressed adolescents show significantly enhanced activation in anterior cingulate cortex and medial prefrontal areas when processing negative emotional stimuli, while ventromedial PFC function inhibiting positive emotions is weakened. This neural circuit abnormality may be the neurobiological basis of cognitive bias, also predicting cognitive behavioral therapy effectiveness-patients with high baseline cognitive bias scores show 17.3% lower average symptom improvement after 12 weeks of treatment compared to those with normal scores[64,65].
Emotional regulation strategy use frequency
Diary method tracking shows depressed adolescents more frequently use maladaptive strategies (such as rumination: Daily average 3.7 times vs 1.2 times), with positive reappraisal strategy use rate decreased by 52.3%. Detailed analysis of emotional regulation strategies using experience sampling method for 14 days of continuous monitoring finds that when facing negative emotions, depressed adolescents show significantly increased avoidance strategy use frequency (daily average 4.2 times vs 1.8 times) beyond rumination, emotional expression suppression increased by 58.6%, while problem-solving strategy use decreased by 33.7%[66,67]. Multidimensional Cognitive Emotion Regulation Questionnaire shows the depressed group scores 71.4% higher than normal group on catastrophizing interpretation dimension, self-blame tendency increased by 62.8%, while acceptance strategy use rate decreased by 46.2%. Emotional regulation strategy flexibility index significantly decreases in depressed group (3.2 vs 5.8), indicating difficulty adjusting strategy selection according to context[68,69]. Neurophysiological research finds depressed adolescents show reduced PFC-limbic system neural circuit connectivity during emotional regulation tasks, particularly during cognitive reappraisal execution, dorsolateral PFC activation decreased by 42.7%, while amygdala activation enhanced by 65.3% during rumination. Emotional regulation difficulty positively correlates with symptom severity (r = 0.68) and closely relates to suicidal ideation emergence. Longitudinal studies indicate adolescents with high baseline rumination frequency show 36.4% depression recurrence rate at 6-month follow-up, while those with good emotional regulation skills show only 14.1%. Intervention research shows mindfulness-based emotional regulation skills training significantly improves depressed adolescents' strategy use patterns: After 8 weeks of training, rumination frequency decreased by 54.2%, cognitive reappraisal skills improved by 43.8%, Difficulties in Emotion Regulation Scale total score decreased by 28.9%, and these improvements correlate with brain function changes, PFC normalization in emotional regulation tasks, predicting better long-term prognosis[70].
Self-concept clarity and depression relationship
Self-concept clarity scale scores show significant negative correlation with depressive symptoms (r = -0.66), with mediation analysis indicating its influence on depression severity through affecting coping strategy selection. Further research finds multidimensional analysis of self-concept clarity shows self-knowledge consistency dimension has strongest correlation with depression (r = -0.72), followed by temporal stability dimension (r = -0.58) and internal consistency dimension (r = -0.54)[71]. Adolescent identity development task completion positively correlates with self-concept clarity (r = 0.64), with 75.3% of individuals with low self-concept clarity (score < 30) showing identity diffusion status, while only 23.7% in high clarity group (score > 45). Neuroimaging research shows depressed adolescents with lower self-concept clarity demonstrate significantly reduced activation in mPFC and anterior cingulate cortex during self-referential processing tasks, while default mode network functional connectivity is abnormal, particularly weakened connection strength between mPFC and precuneus by 36.4%[72]. Longitudinal tracking finds adolescents with low baseline self-concept clarity (< 35 points) show 42.1% depression incidence within 18 months, while high clarity group shows only 11.8%. Self-concept clarity also closely relates to treatment response: In cognitive behavioral therapy, patients with high baseline clarity show 82.7% overall treatment effectiveness, while low clarity group shows only 54.3%. Cross-cultural research indicates self-concept clarity has closer relationship with depression in individualistic cultural contexts (r = -0.72 vs r = -0.48), possibly related to differential identity pressure in different cultural environments. Intervention research shows narrative therapy-based self-exploration training significantly enhances self-concept clarity, with clarity scores increasing by average 23.9% after 12 weeks of treatment, depressive symptoms reduced by 31.6%, and improvements maintained at 6-month follow-up. Machine learning model analysis finds combining self-concept clarity with other cognitive factors (such as cognitive flexibility, self-esteem levels) can predict adolescent depression occurrence with 78.4% accuracy, suggesting self-concept clarity has important clinical value in depression early warning and intervention[73,74].
Perfectionism subtype research
Self-oriented perfectionism shows correlation with depressive symptoms of r = 0.43, socially prescribed perfectionism correlation of r = 0.58, while self-expectant perfectionism only r = 0.21. Three-dimensional model research on perfectionism further reveals its complexity: Self-oriented perfectionism contains excessively high personal standard setting and strong self-criticism tendencies, socially prescribed perfectionism mainly manifests as excessive focus on others' evaluations and fear of bearing failure, while self-expectant perfectionism more represents realistic assessment of personal abilities. Meta-analysis research shows in clinical samples, socially prescribed perfectionism has most stable correlation with depression (r = 0.56-0.62), particularly more significant in female adolescents (r = 0.67)[75]. Behavioral experimental tasks find high socially prescribed perfectionists show 41.3% higher cortisol stress response than normal group when receiving simulated peer evaluation, with anterior cingulate cortex activation enhanced by 53.7% when facing social threats[76]. Tracking research shows perfectionist tendencies develop rapidly between ages 10-16, with socially prescribed perfectionism average scores peaking at ages 13-14, coinciding with adolescent self-consciousness rapid development period. Multilevel model analysis finds parental attitudes toward achievement explain 32.4% of socially prescribed perfectionism variance, while peer competition pressure explains 23.8%[77]. Perfectionism and rumination interaction analysis indicates high socially prescribed perfectionism + high rumination combination shows strongest prediction of depression symptom severity (β = 0.76), while low social expectation + low rumination combination shows protective factor effect (β = -0.31). Neuropsychological testing finds high perfectionism adolescents show reduced cognitive flexibility in Wisconsin Card Sorting Test, particularly with 36.9% increased error rate during rule switching, reflecting cognitive rigidity characteristics. Intervention research shows acceptance and commitment therapy significantly effective in reducing socially prescribed perfectionism, with 42.7% reduction in socially prescribed perfectionism scale scores after 8 weeks of treatment, alongside 38.5% reduction in depressive symptoms[78]. Additionally, mindfulness-based stress reduction training combined with self-compassion practice effectively alleviates perfectionism-related anxiety and depression, with pre-treatment and post-treatment brain scans showing 31.6% reduced amygdala reactivity to social threat stimuli and enhanced mPFC regulatory function. Long-term follow-up research finds adolescents with sustained reduction in socially prescribed perfectionism scores after targeted intervention show only 21.3% depression recurrence rate within 3 years, compared to 48.7% in non-intervention group, suggesting perfectionism intervention has important value in depression prevention[79,80].
Attachment patterns and depression vulnerability
Depression incidence in secure attachment adolescents is 8.3%, anxious type 31.7%, avoidant type 24.5%, and disorganized type reaches 45.2%. These attachment patterns, originally conceptualized as psychological and relational constructs, are increasingly understood to have distinct neurobiological signatures that help explain their differential impact on depression vulnerability. The transition from behavioral attachment patterns to measurable brain differences reveals how early relationship experiences become biologically embedded, creating lasting effects on emotional regulation capacity.
Neurobiological research on attachment patterns demonstrates that different attachment styles manifest in quantifiable neural circuit differences, particularly in emotion regulation pathways. Secure attachment individuals maintain stable PFC inhibitory connections to amygdala when facing separation threats, while disorganized attachment individuals show 69.3% connectivity reduction, leading to emotional regulation failure. This neurobiological finding directly corresponds to the behavioral observation that disorganized attachment adolescents struggle with emotion regulation under stress, illustrating how psychological attachment security translates into functional brain connectivity patterns. Eye-tracking experiments find anxious attachment adolescents show 47.2% longer attention time to attachment numbers' facial expressions compared to secure type, especially with 35.8% faster visual search speed for negative facial expressions, reflecting hypervigilance to relationship threats.
Moving from behavioral and neural observations to developmental trajectories, longitudinal research indicates early attachment patterns can predict adolescent depression developmental trajectories: In 4-year tracking, disorganized attachment individuals' average age of first depressive episode is 18 months earlier than secure type, with 43.7% longer illness duration. Attachment Trauma Questionnaire assessment shows 76.4% of adolescents who experienced attachment trauma develop insecure attachment patterns, with 38.2% avoidant and 38.2% disorganized.
At the neuroendocrine level, attachment patterns influence stress response systems in measurable ways. Physiological indicator research finds in attachment activation situations, insecure attachment individuals show 31.5% lower oxytocin secretion levels than secure type, while stress hormone cortisol levels increase by 52.8%, forming neuroendocrine imbalance. This hormonal dysregulation links directly to the HPA axis abnormalities described in previous chapters, suggesting that insecure attachment may contribute to the circadian cortisol disruptions observed in depressed adolescents.
Randomized controlled trials of attachment repair therapy show emotion-focused therapy-based attachment intervention significantly improves insecure attachment patterns: After 24 weeks of treatment, 34.6% of disorganized attachment transitions to secure type, with 41.9% reduction in depressive symptoms. Experiences in Close Relationships Scale-Revised longitudinal assessment finds each standard deviation increase in attachment anxiety dimension increases depression onset risk by 23.7%, while attachment avoidance dimension has relatively smaller impact (14.2%). Intergenerational research shows moderate intergenerational transmission of mother's attachment pattern to adolescent children's attachment pattern (r = 0.54), with mother's emotional regulation ability playing important mediating role, explaining 47.6% of total transmission effect.
The neuroplasticity of attachment-related circuits provides hope for intervention, as neural patterns established in early childhood remain modifiable even in adolescence. Latest neuroplasticity research finds even in late adolescence, attachment repair therapy can still observe remodeling of attachment-related neural circuits, with post-treatment fMRI showing enhanced functional connectivity in amygdala-hippocampus-PFC circuit after secure attachment experiences, and this neural change positively correlates with depressive symptom improvement (r = 0.63), suggesting attachment-oriented therapy has important neurobiological basis in preventing and treating adolescent depression[81,82].
SOCIAL DIMENSION RESEARCH
Social media use pattern analysis
Adolescents using social media more than 3 hours daily show 41% increased depression risk, with imbalanced passive browsing to active interaction ratio (4:1) correlating with depression severity. This predominance of passive social media use directly activates and reinforces the cognitive biases discussed in previous chapters, creating a powerful cross-domain mechanism for depression development. Specifically, passive browsing characterized by scrolling through others' curated content without meaningful interaction fuels upward social comparison, where adolescents compare their unfiltered reality to others' highlight reels. This comparison pattern intensifies the negative attentional bias (46 milliseconds delay toward negative stimuli) by priming adolescents to selectively attend to content that confirms negative self-perceptions. Furthermore, passive browsing triggers the rumination patterns identified in previous chapters, as adolescents repeatedly mentally replay social comparisons and perceived inadequacies. The 4:1 passive-to-active ratio thus represents not merely a behavioral pattern but a mechanism that systematically activates maladaptive cognitive processes upward comparison triggering the 38.5% increased negative recall bias, subsequently feeding into the average 3.7 daily rumination episodes. This creates a vicious cycle where social media use patterns, cognitive biases, and emotion dysregulation mutually reinforce one another, illustrating the synergistic interaction between social environmental factors and psychological vulnerabilities.
In-depth use behavior analysis shows late-night use patterns (from 10 PM to 2 AM) particularly strongly correlate with depressive symptoms (r = 0.72), with these adolescents showing average 32.1% reduction in rapid eye movement sleep time. Social Comparison Tendency Scale assessment shows frequency of upward social comparison on these platforms positively correlates with depression symptom severity (r = 0.68), with appearance-related comparison having greatest impact (β = 0.54). Experience sampling method research finds after each social media use, passive browsers' current mood scores decrease by average 2.3 points (out of 10), while active interactors only decrease by 0.7 points, with this mood fluctuation particularly significant in depressed adolescents. Neuroimaging research shows social media “like” stimuli activate striatal DA pathways in heavy users' brains, with activation patterns similar to substance addiction, but this reward response weakened by 43.2% in depressed adolescents. Cyberbullying victimization survey finds 47.3% of heavy social media users report experiencing cyberbullying, with 72.8% showing depressive symptoms. Social media “detox” experiments show after 1-week cessation, depression self-rating scale scores decrease by average 12.4%, with improved sleep quality and increased actual social activities. Multivariate regression analysis indicates after controlling for other factors, passive use frequency, late-night use duration, social comparison tendency, and online social isolation can jointly explain 52.7% of depression symptom variance. Intervention research finds mindfulness-based digital health literacy training significantly improves social media use patterns: After 8 weeks of training, participants' active interaction ratio increases from 20% to 45%, daily use duration decreases by average 37.2%, depressive symptoms reduce by 29.8%, with improvements maintained at 3-month follow-up. Latest mechanism research indicates excessive social media use leads to chronic stress state through HPA axis activation, while affecting PFC executive function, making emotional regulation more difficult for adolescents, thus forming vicious cycle of “use-depression-avoidance use”[83-85].
Family function and depression relationship
Family Adaptability and Cohesion Evaluation shows adolescent depression prevalence in dysfunctional families is 34.7%, while in healthy families 12.1%, with parent-child communication quality mediating 58.3% of the impact. Research from family systems theory perspective further finds adolescents growing up in chaotic families (lacking structure and rules) show 41.3% depression incidence, while in rigid families (excessive control) 28.9%, indicating balance in family function is crucial for mental health. The family environment's impact on depression operates partly through shaping attachment patterns, as discussed in previous chapters. Dysfunctional family dynamics characterized by inconsistent caregiving, high criticism, or emotional unavailability frequently led to the insecure attachment patterns (anxious, avoidant, or disorganized) that carry substantially elevated depression risk. Adolescents from chaotic or rigid family environments often develop the disorganized attachment pattern associated with 45.2% depression incidence, while those from balanced, responsive families more commonly exhibit secure attachment (8.3% depression rate). This family-attachment-depression pathway is further reinforced by neurobiological mechanisms: Dysfunctional families trigger the neuroendocrine dysregulation described in previous chapters, including 31.5% reduced oxytocin and 52.8% elevated cortisol in insecurely attached adolescents, creating biological vulnerabilities that persist even as adolescents seek relationships outside the family.
Parent-child attachment security assessment shows insecurely attached adolescents have 2.7 times higher depression risk than securely attached, with father-child relationship quality impact (odds ratio = 2.1) slightly higher than mother-child relationship (odds ratio = 1.8). Family Expressed Emotion Scale assessment finds adolescents in high criticism comment environments show more severe depressive symptoms, with each additional criticism event increasing depression scores by average 1.3 points. Neuroendocrine research shows adolescents from high-conflict families have 24.7% elevated baseline cortisol levels and stronger physiological response to social stress, with 38.6% extended stress recovery time. Family therapy-oriented observational research finds significant negative correlation between parental emotional regulation ability and adolescent depressive symptoms (r = -0.52), particularly parental emotional expression and response sensitivity can predict children's emotional problem developmental trajectory[86,87]. Multigenerational family research indicates grandparental caregiving involvement can serve as protective factor, with adolescent depression prevalence reduced to 16.4% in families with adequate grandparental support. Family economic pressure indirect effect analysis shows economic difficulties indirectly affect adolescent mental health by increasing parental anxiety and reducing parenting quality, with this mediating effect accounting for 45.2% of total effect. Randomized controlled trials of family therapy show multisystemic therapy-based family intervention significantly improves family function, with post-treatment family cohesion scores increasing by 32.8%, adolescent depressive symptoms reducing by 41.5%, and effects maintained at 12-month follow-up. Cross-cultural comparative research finds in collectivist cultures, absence of extended family support network has greater impact on adolescent depression (β = 0.67), while in individualist cultures, nuclear family parent-child relationship quality impact is more prominent (β = 0.78). Latest epigenetic research indicates early adverse family environment can lead to methylation changes in stress-related genes, with these epigenetic markers still present in adolescence and correlating with sustained depression vulnerability, suggesting family environment impact has long-term biological imprinting[88,89].
School environment factor research
School bullying victims show 73% increase in depressive symptoms, with moderate academic pressure group (grade point average: 80-85) showing better mental health than high pressure group (> 90) and low pressure group (< 75). Multidimensional analysis of school environment shows among three bullying types - physical bullying, verbal bullying, and social exclusion-social exclusion has greatest impact on depressive symptoms (β = 0.68), with 78.4% of victims experiencing continuous victimization over 6 months showing clinically significant depressive symptoms. Inverted U-curve effect research on academic pressure finds moderate academic challenge provides optimal psychological resilience, with both too low or too high academic demands leading to mental health problems, with perfectionists more likely to experience depressive breakdown in high-pressure environments (89.3% increased risk). Teacher support protective effect analysis indicates in high-support teacher environments, even students experiencing mild bullying show depression incidence only half of common rate (14.7% vs 29.3%), with teachers’ emotional support more protective than academic support. School belonging scale assessment shows adolescents with low belonging (score < 30) have 42.1% depression prevalence, while high belonging (score > 45) only 11.8%, with belonging buffering negative impact of peer pressure on mental health. Longitudinal research on extracurricular activity participation finds students participating in sports or arts activities show 31.6% reduced depression incidence, with activity type diversity more beneficial for mental health than single focus. School Climate Scale assessment shows in school environments valuing competition over cooperation, student anxiety and depressive symptoms significantly increase, particularly in regions with strong “exam culture”. Neuropsychological research finds adolescents in long-term high-competition school environments show 41.2% enhanced PFC reactivity to social threats, while hippocampal stress response recovery ability decreases by 28.7%. School mental health service effectiveness evaluation shows schools with professional mental health teachers increase timely identification and intervention of student depressive symptoms by 62.3%, with early intervention success rate reaching 78.9%. Peer relationship network analysis indicates adolescents with 2-3 close friends at school show highest psychological resilience, while isolation or excessive dependence on large social circles both correlate with increased depression risk. Latest school environment intervention research shows implementing school-wide anti-bullying policies and social-emotional learning programs can reduce overall school depression prevalence by 24.3%, with improvement effects persisting after two years of implementation, indicating systematic school environment improvement has long-term positive impact on adolescent mental health[90-93].
Digital device use and sleep quality
Adolescents using electronic devices within 2 hours before bedtime show average 37-minute delayed sleep onset, 14.2% reduced rapid eye movement sleep, and 22.5% increased depression symptom severity. These sleep disruptions create direct biological pathways to depression by affecting the neurobiological systems described in previous chapters. The circadian rhythm disruption caused by nighttime device use mirrors and likely exacerbates the HPA axis dysregulation observed in depressed adolescents, including the 31.5% reduced CAR and abnormal afternoon cortisol elevation. Furthermore, chronic sleep deprivation stemming from digital device use contributes to the neurotransmitter imbalances detailed in previous chapters: Sleep loss is known to reduce prefrontal GABA concentrations and increase glutamate levels, potentially explaining or worsening the 23.7% GABA reduction and 15.8% glutamate/glutamine ratio increase found in depressed adolescents. Additionally, sleep disruption affects serotonin metabolism, potentially contributing to the 19.2%-32.7% reductions in 5-hydroxyindoleacetic acid observed across brain regions. This represents a clear bio-social interaction where an environmental factor (device use timing) triggers neurobiological changes (HPA axis and neurotransmitter dysfunction) that directly increase depression vulnerability, illustrating how social behaviors become biologically embedded to influence mental health outcomes.
Polysomnography monitoring research shows nighttime device use not only affects sleep onset time but also leads to profound changes in sleep architecture: N3 stage deep sleep decreases by average 19.7%, nighttime awakenings increase 2.4-fold, sleep efficiency drops from 89.2% to 76.8%. Blue light exposure physiological mechanism research finds screen exposure within 3 hours before bedtime delays melatonin secretion by average 41 minutes, with 23.6% reduced secretion peak, this circadian rhythm disruption significantly positively correlating with depression symptom severity (r = 0.61). Device type-specific analysis shows smartphones have greatest impact (52-minute sleep onset delay), followed by tablets (34 minutes) and laptops (28 minutes), while using night mode or blue light filters partially mitigates this impact, reducing sleep onset delay to 24 minutes. Sleep-mood interaction longitudinal research finds sustained sleep deprivation predicts depression symptom worsening 2 weeks later, while depressive symptoms promote more nighttime device use, forming vicious cycle. Neuroendocrine research shows combined effects of sleep deprivation and digital device use lead to cortisol diurnal rhythm flattening, with 31.5% reduced morning CAR, highly consistent with depression neuroendocrine characteristics. Cognitive behavioral assessment finds nighttime device use leads to 27.3% reduced attention span next day, 32.1% decreased emotional regulation ability, with these cognitive function impairments further exacerbating depressive symptoms. Intervention research shows adolescents implementing “digital curfew” (stopping electronic device use after 9 PM) show significant sleep quality improvement within 1 month, with 18.7% reduction in depression self-rating scale scores. Neuroplasticity research finds good sleep hygiene habits can restore normal sleep architecture within 4-6 weeks, while improving default mode network functional connectivity, closely related to depressive symptom alleviation[94-96]. Latest optogenetic research indicates nighttime blue light exposure not only suppresses melatonin secretion but also directly affects hippocampal neurogenesis and emotional regulation-related neuron activity, potentially representing important neurobiological mechanism linking digital device use and depression, with this effect particularly pronounced in adolescent brains as adolescent circadian rhythm systems are still developing and more sensitive to light stimuli[97,98].
Social support network analysis
Adolescents with 2-3 close friends show 15.3% depression rate, isolated type (0 friend) 42.6%, large social network type (> 5 friends) 21.8%, indicating protective effect of moderate socialization. Social support network quality-quantity interaction research shows high-quality small circle socialization superior to low-quality large networks, with emotional support (r = -0.54) and informational support (r = -0.43) showing stronger protective effects against depressive symptoms than instrumental support (r = -0.32)[99-102]. Neuroimaging research finds adolescents with stable social networks show 35.2% reduced anterior cingulate cortex activation in social exclusion tasks, with 28.7% higher oxytocin receptor density than isolated individuals, suggesting neurochemical basis for social connection. Online vs offline social comparison analysis indicates face-to-face interaction benefits for mental health significantly exceed online socialization, with each additional hour of face-to-face socializing reducing depression risk by 8.4%, while online socializing effect only 3.1%[103-105]. Social support buffering effect research shows high social support individuals experiencing major life stress show only 39.7% depression onset rate compared to low support individuals, with 1.7 times faster recovery speed. Multidimensional Social Support Scale assessment finds support from different sources has cumulative effect: Family support + friend support + teacher support combination can reduce depression prevalence to 8.9%. Social network maintenance quality research indicates adolescents actively maintaining friendships (initiating contact at least once weekly) show 34.6% lower depression rate than passive maintainers, with higher friendship stability[106,107]. Social support skills training intervention shows after 8 weeks of interpersonal skills training, participants’ social network size increases by 49.3%, social satisfaction improves by 41.8%, depressive symptoms reduce by 32.4%. Cross-cultural research finds in collectivist cultures, social network density has greater impact on mental health, while in individualist cultures, social quality is more important. Gender difference analysis shows female adolescents have higher dependence on social networks, with social isolation impact on their depressive symptoms 1.4 times stronger than males. Latest social neuroscience research indicates stable social connections can activate reward systems, promoting DA and serotonin secretion, with this neurochemical response particularly important during adolescence as this period is critical window for establishing lifelong social patterns, and moderate social engagement can both satisfy belonging needs and avoid stress from social overload, thus providing optimal protection for mental health.