Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.112649
Revised: September 16, 2025
Accepted: December 8, 2025
Published online: March 19, 2026
Processing time: 204 Days and 0.8 Hours
Intrauterine adhesions (IUA) are a common gynecological condition that can sig
To determine the incidence of postoperative depressive disorders in patients un
A retrospective analysis was conducted on clinical data from 400 patients who underwent intrauterine adhesion lysis surgery at our hospital from January 2022 to December 2024. Demographic characteristics, medical history, surgical parame
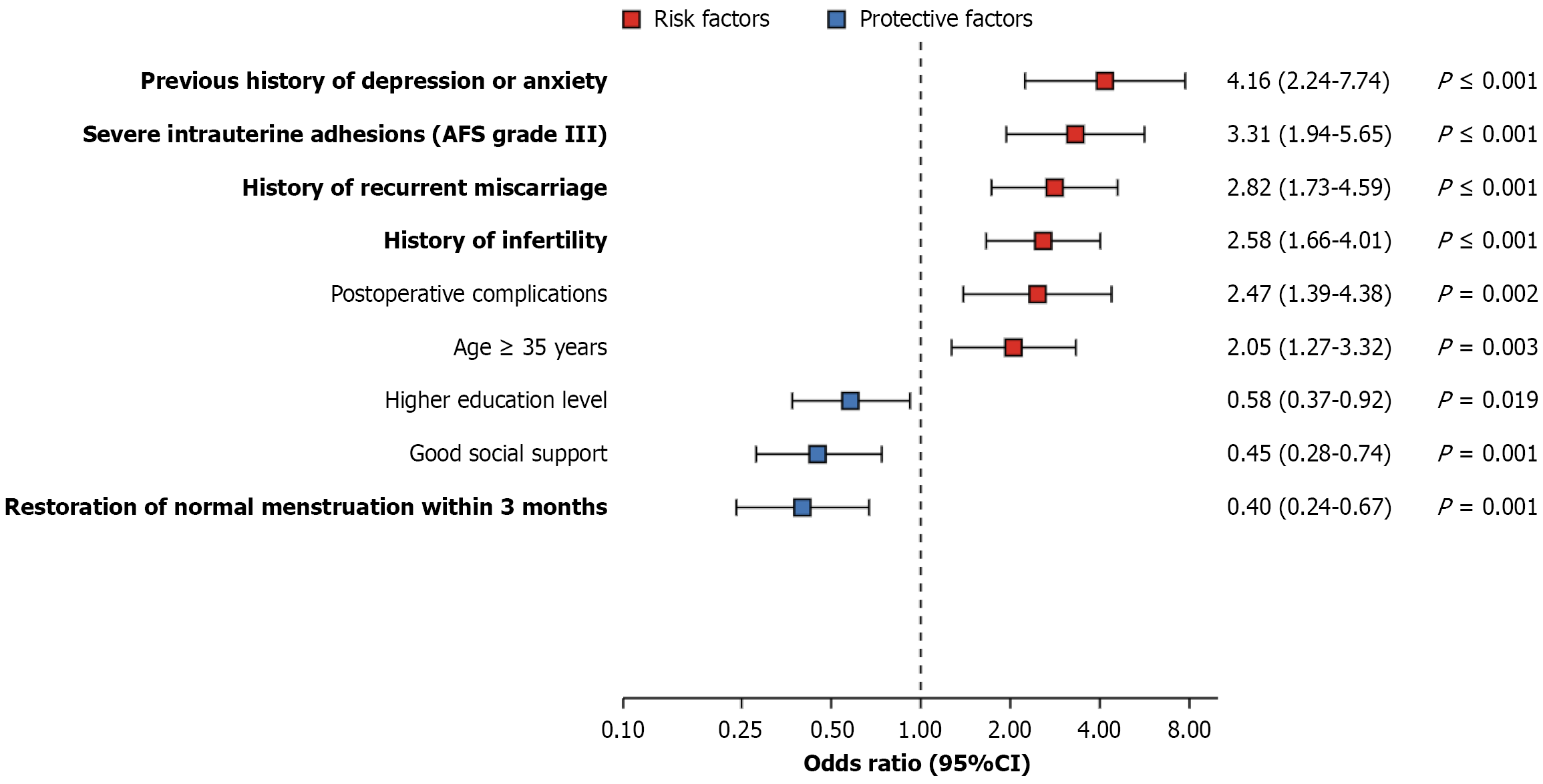
Among the 400 patients, 112 (28.0%) developed depressive disorders within 3 months after surgery. Multivariate analysis showed that age ≥ 35 years [odds ratio (OR) = 2.05, 95% confidence interval (CI): 1.27-3.32, P = 0.003], history of infertility (OR = 2.58, 95%CI: 1.66-4.01, P < 0.001), history of recurrent miscarriage (OR = 2.82, 95%CI: 1.73-4.59, P < 0.001), severe IUA (American Fertility Society classification grade III) (OR = 3.31, 95%CI: 1.94-5.65, P < 0.001), postoperative complications (OR = 2.47, 95%CI: 1.39-4.38, P = 0.002), and previous history of depression or anxiety (OR = 4.16, 95%CI: 2.24-7.74, P < 0.001) were independent risk factors for postoperative depressive disorders. Protective factors included higher education level (OR = 0.58, 95%CI: 0.37-0.92, P = 0.019), good social support (OR = 0.45, 95%CI: 0.28-0.74, P = 0.001), and normal menstruation within 3 months after surgery (OR = 0.40, 95%CI: 0.24-0.67, P < 0.001).
The incidence of postoperative depressive disorders in patients with intrauterine adhesions is relatively high and associated with multiple factors.
Core Tip: Postoperative depression affects 28% of patients undergoing hysteroscopic adhesiolysis for intrauterine adhesions, representing a significantly higher rate than the general female population. Six independent risk factors were identified: Age ≥ 35 years, history of infertility, recurrent miscarriage, severe adhesions (grade III), postoperative complications, and previous depression or anxiety disorders. Three protective factors emerged: Higher education level, strong social support, and successful menstrual restoration within 3 months post-surgery.
- Citation: Du WL, Chen S, Zhan SS. Analysis of risk factors for postoperative depressive disorders in patients with intrauterine adhesions. World J Psychiatry 2026; 16(3): 112649
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/112649.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.112649
Intrauterine adhesions (IUA) are a common gynecological condition characterized by partial or complete adhesion of the endometrium, leading to partial or complete occlusion of the uterine cavity[1-3]. This condition is primarily caused by endometrial injury resulting from intrauterine procedures or infections, such as curettage, cesarean section, or myomec
Hysteroscopic adhesiolysis has become the primary treatment method for IUA[8,9]. This surgical procedure recon
Gynecological diseases, especially those related to fertility, significantly impact women’s mental health. Previous studies have shown that the incidence of post-operative depressive disorders in gynecological surgery patients is markedly higher than in the general population[12,13]. For patients with IUA, the close relationship between the disease and fertility, coupled with uncertainties during the recovery process, makes these patients more susceptible to depression and other psychological problems[14]. However, systematic research on the incidence and risk factors of post-operative depression in IUA patients is relatively lacking, and clinical practice has insufficient focus on the mental health of these patients.
Existing research has preliminarily explored factors related to post-operative depression in various gynecological surgeries, such as age, education level, marital status, and disease severity. However, studies specifically targeting the unique population of intrauterine adhesion patients remain incomplete. Most IUA patients are women of reproductive age with strong fertility demands, and surgical outcomes directly affect their future fertility potential, making the psychological status of IUA patients particularly distinctive.
Timely identification of high-risk populations for post-operative depression in IUA patients and early intervention can not only improve patients’ mental health status but may also promote physiological recovery, increase surgical success rates, and enhance patients’ quality of life. Therefore, systematically analyzing the incidence and related risk factors of post-operative depression in IUA patients is of significant importance for guiding clinical work, optimizing perioperative management, and developing individualized psychological intervention strategies.
This study retrospectively analyzes the clinical data of 308 patients who underwent hysteroscopic adhesiolysis from 2022 to 2024, aiming to determine the incidence and independent risk factors of post-operative depression in IUA patients, provide scientific evidence for clinical intervention, and ultimately improve patients’ post-operative rehabilitation and long-term prognosis.
This retrospective study analyzed data from 400 patients who underwent intrauterine adhesiolysis for IUA at our hospital from January 2022 to December 2024. The study protocol was approved by the institutional review board, and informed consent was obtained from all patients. Inclusion criteria were: (1) Female patients diagnosed with IUA via hysteroscopy; (2) Age between 18 and 45 years; (3) Patients who underwent hysteroscopic adhesiolysis at our institution during the study period; (4) Availability of complete medical records and follow-up data for at least 3 months after surgery; and (5) Ability to complete psychological assessment questionnaires. Exclusion criteria were: (1) Previous diagnosis of severe psychiatric disorders requiring hospitalization; (2) Concurrent use of antidepressants or antipsycho
All patients underwent hysteroscopic adhesiolysis performed by experienced gynecological surgeons. After general anesthesia or paracervical block, a diagnostic hysteroscopy was initially performed to evaluate the extent and severity of IUA according to the American Fertility Society classification. Adhesiolysis was conducted using cold scissors, elec
Patients were retrospectively divided into two groups based on postoperative depressive status: A depression group (n = 112) of patients who met diagnostic criteria for depressive disorder within 3 months after surgery, and a non-depression group (n = 288) of patients without depressive symptoms. Data collection included comprehensive assessment of demo
Complete-case analysis was employed for the primary analysis, with missing data rates below 5% for all key variables. A detailed patient flowchart documented exclusions due to incomplete data. Sensitivity analyses using multiple imputation (m = 20 imputations) were performed to assess the robustness of findings to missing data assumptions. Variables in
All patients completed standardized psychological evaluations both preoperatively and at 3 months after surgery, administered by trained healthcare professionals using validated Chinese versions of established scales. The assessment tools included: SDS with scores ≥ 53 indicating depression (Cronbach’s α = 0.87); HAMD-17 with scores ≥ 17 indicating depression, administered by trained clinicians (inter-rater reliability κ = 0.85); Self-Rating Anxiety Scale to assess comor
Statistical analysis was performed using SPSS version 25.0. Student’s t-test or Mann-Whitney U test was used for continuous variables, while χ2 or Fisher’s exact test was used for categorical variables. Univariate analysis identified potential risk factors for postoperative depression. Variables with P < 0.1 were included in multivariate logistic regression to determine independent risk factors. Odds ratios (ODs) with 95% confidence intervals (CIs) were calculated, with P < 0.05 considered significant. Correlation between depression severity and continuous variables was assessed using Pear
The study population was divided into depression group (n = 112) and non-depression group (n = 288) based on postope
Baseline psychological assessment showed that preoperative SDS and HAMD-17 scores were higher in patients who later developed postoperative depression (SDS: 48.3 ± 6.7 vs 42.1 ± 5.9, P < 0.001; HAMD-17: 14.2 ± 3.8 vs 10.5 ± 3.1, P < 0.001). A previous history of depression or anxiety was reported in 30.4% of patients in the depression group compared to 9.7% in the non-depression group (P < 0.001). Social support scores were significantly lower in the depression group (34.2 ± 6.8 vs 41.5 ± 7.3, P < 0.001, Table 1).
| Feature | Depressed group (n = 112) | Non-depressed group (n = 288) | P value |
| Age (years) | 36.7 ± 4.8 | 32.4 ± 5.3 | < 0.001 |
| Education level (%) | 41.1 | 58.7 | 0.002 |
| Infertility history (%) | 63.4 | 38.2 | < 0.001 |
| Duration of infertility (years) | 4.8 ± 2.3 | 2.9 ± 1.7 | < 0.001 |
| Recurrent miscarriage history (%) | 42.9 | 21.5 | < 0.001 |
| Preoperative SDS score | 48.3 ± 6.7 | 42.1 ± 5.9 | < 0.001 |
| Preoperative HAMD-17 score | 14.2 ± 3.8 | 10.5 ± 3.1 | < 0.001 |
| History of depression or anxiety (%) | 30.4 | 9.7 | < 0.001 |
| Social support score | 34.2 ± 6.8 | 41.5 ± 7.3 | 0.001 |
Among the 400 patients included in this study, the mean age was 33.6 ± 5.5 years (range: 21-45 years). The majority of patients (73.5%, n = 294) were married, and 62.3% (n = 249) had at least one previous pregnancy. The most common etiologies of IUA were curettage after abortion (48.5%, n = 194), postpartum curettage (22.3%, n = 89), hysteroscopic procedures (15.8%, n = 63), and other causes (13.4%, n = 54). According to the American Fertility Society classification, 112 patients (28.0%) had grade I (mild) adhesions, 134 patients (33.5%) had grade II (moderate) adhesions, and 154 patients (38.5%) had grade III (severe) adhesions.
Regarding surgical parameters, the mean operation time was 55.5 ± 16.8 minutes. Cold scissors were used in 218 patients (54.5%), electrosurgical instruments in 146 patients (36.5%), and balloon techniques in 36 patients (9.0%). Anti-adhesion barriers were applied in 288 patients (72.0%), with hyaluronic acid gel being the most commonly used (62.5%, n = 180). All procedures were performed under either general anesthesia (64.0%, n = 256) or paracervical block (36.0%, n = 144, Table 2).
| Feature | Depressed group (n = 112) | Non-depressed group (n = 288) | P value |
| Patient demographics | |||
| Average age (years, mean ± SD) | 36.7 ± 4.8 | 32.4 ± 5.3 | < 0.001 |
| Marital status (%) | 75.0 | 72.9 | 0.673 |
| Pregnancy history (%) | 54.5 | 65.3 | 0.044 |
| Etiology of asherman’s syndrome (%) | P = 0.863 | ||
| Post-abortion curettage | 50.9 | 47.6 | |
| Postpartum curettage | 23.2 | 21.9 | |
| Hysteroscopic surgery | 14.3 | 16.3 | |
| Other causes | 11.6 | 14.2 | |
| Classification of asherman’s syndrome (%) | |||
| Grade I (mild) | 17.9 | 31.9 | 0.005 |
| Grade III (severe) | 53.6 | 32.6 | < 0.001 |
| Surgical parameters | |||
| Average operating time (minutes, mean ± SD) | 63.2 ± 18.7 | 52.4 ± 15.1 | < 0.001 |
| Surgical instruments (%) | P = 0.791 | ||
| Cold scissors | 51.8 | 55.6 | |
| Electrosurgical instruments | 38.4 | 35.8 | |
| Balloon technique | 9.8 | 8.7 | |
| Anti-adhesion barrier use (%) | 78.6 | 69.1 | 0.043 |
| Anesthesia type (%) | P = 0.327 | ||
| General anesthesia | 66.1 | 63.2 |
The mean operation time was significantly longer in the depression group compared to the non-depression group (63.2 ± 18.7 minutes vs 52.4 ± 15.1 minutes, P < 0.001), reflecting the more severe and complex adhesions in these patients. The length of hospital stay was also significantly longer in the depression group (3.2 ± 1.1 days vs 2.5 ± 0.8 days, P < 0.001). Postoperative complications occurred in 17.9% (20/112) of patients in the depression group compared to 8.0% (23/288) in the non-depression group (P = 0.004). These complications included uterine perforation (3.6% vs 1.4%, P = 0.144), post
| Feature | Depressed group (n = 112) | Non-depressed group (n = 288) | P value |
| Surgical time (minutes, mean ± SD) | 63.2 ± 18.7 | 52.4 ± 15.1 | < 0.001 |
| Length of hospital stay (days) | 3.2 ± 1.1 | 2.5 ± 0.8 | < 0.001 |
| Postoperative complications (%) | 17.9 (20/112) | 8.0 (23/288) | 0.004 |
| Complications (%) | |||
| Uterine perforation (%) | 3.6 | 1.4 | 0.144 |
| Postoperative infection (%) | 7.1 | 3.1 | 0.068 |
| Severe bleeding requiring intervention (%) | 7.1 | 3.5 | 0.107 |
| Menstrual recovery within 3 months (%) | 61.6 (69/112) | 81.6 (235/288) | < 0.001 |
Multivariate logistic regression analysis was performed to identify independent predictors of postoperative depressive disorders. After adjusting for potential confounding factors, the following were identified as independent risk factors: Age ≥ 35 years (OR = 2.05, 95%CI: 1.27-3.32, P = 0.003), history of infertility (OR = 2.58, 95%CI: 1.66-4.01, P < 0.001), history of recurrent miscarriage (OR = 2.82, 95%CI: 1.73-4.59, P < 0.001), severe IUA (American Fertility Society classification grade III) (OR = 3.31, 95%CI: 1.94-5.65, P < 0.001), postoperative complications (OR = 2.47, 95%CI: 1.39-4.38, P = 0.002), and previous history of depression or anxiety (OR = 4.16, 95%CI: 2.24-7.74, P < 0.001). Protective factors against postoperative depression included higher education level (OR = 0.58, 95%CI: 0.37-0.92, P = 0.019), good social support (OR = 0.45, 95%CI: 0.28-0.74, P = 0.001), and restoration of normal menstruation within 3 months after surgery (OR = 0.40, 95%CI: 0.24-0.67, P < 0.001; Figure 1).
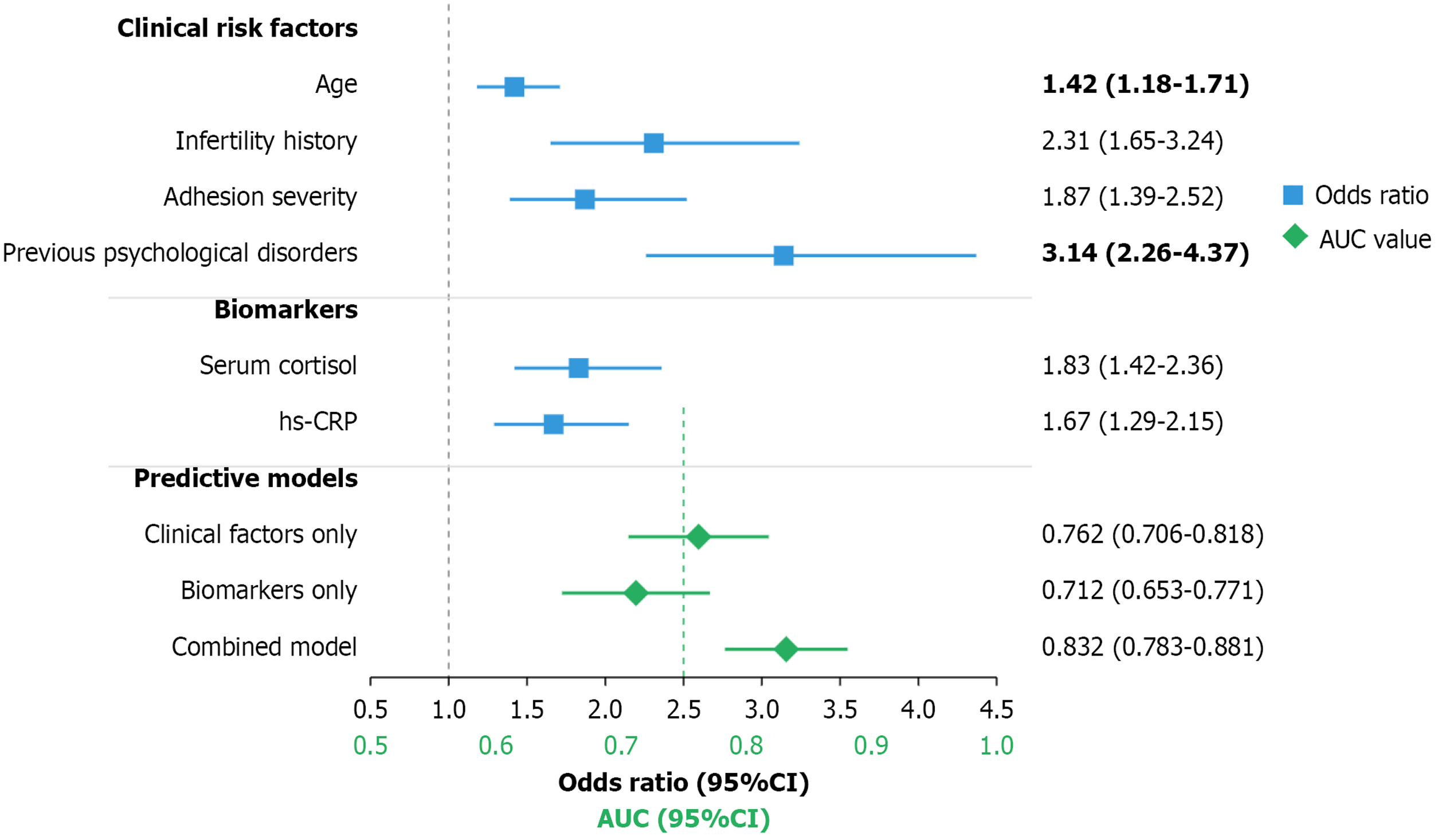
In addition to clinical risk factors, we investigated potential biomarkers associated with postoperative depression. Serum cortisol levels were significantly higher in the depression group compared to the non-depression group (18.4 ± 5.7 μg/dL vs 14.2 ± 4.5 μg/dL, P < 0.001). Similarly, inflammatory markers including high-sensitivity C-reactive protein were elevated in the depression group (3.8 ± 1.9 mg/L vs 2.5 ± 1.3 mg/L, P < 0.001).
Receiver operating characteristic curve analysis showed that a combination of clinical risk factors (age, infertility history, adhesion severity, previous psychological disorders) and biomarkers (cortisol, high-sensitivity C-reactive protein) had good predictive value for postoperative depression [area under the curve (AUC) = 0.832, 95%CI: 0.783-0.881]. This combined model demonstrated better predictive performance than clinical factors alone (AUC = 0.762, 95%CI: 0.706-0.818, P = 0.003) or biomarkers alone (AUC = 0.712, 95%CI: 0.653-0.771, P < 0.001; Figure 2). From the forest plot, it can be seen that the CIs of all clinical risk factors and biomarkers do not include 1, indicating that they are all independent risk factors for postoperative depression.
Among the study population, 68 patients (17.0%) had concurrent medical conditions, including thyroid disorders (8.0%, n = 32), polycystic ovary syndrome (5.5%, n = 22), and endometriosis (3.5%, n = 14). The prevalence of postoperative depression was significantly higher in patients with concurrent medical conditions compared to those without (39.7%, 27/68 vs 25.6%, 85/332; P = 0.019; Figure 3).
Subgroup analysis revealed that patients with both severe IUA and concurrent medical conditions had the highest risk of postoperative depression (54.8%, 17/31), followed by patients with severe adhesions only (36.6%, 47/123), patients with concurrent medical conditions only (27.0%, 10/37), and patients with neither severe adhesions nor concurrent medical conditions (14.4%, 38/209) (P < 0.001 for trend). Multivariate analysis confirmed that the presence of concurrent medical conditions was an independent risk factor for postoperative depression (OR = 1.83, 95%CI: 1.04-3.21, P = 0.036), with a synergistic effect when combined with severe IUA (OR = 5.62, 95%CI: 2.59-12.18, P < 0.001; Table 4).
| Indicator | Depressed group (n = 112) | Non-depressed group (n = 288) | Between-group P value | Change in depressed group Δ (%) | Change in non-depressed group Δ (%) | P value for change comparison |
| Psychological assessment | ||||||
| SDS score | ||||||
| Baseline | 48.3 ± 6.7 | 42.1 ± 5.9 | < 0.001 | |||
| 3 months postoperative | 58.4 ± 7.2 | 41.8 ± 6.1 | < 0.001 | +10.1 (20.9) | -0.3 (0.7) | < 0.001 |
| HAMD-17 score | ||||||
| Baseline | 14.2 ± 3.8 | 10.5 ± 3.1 | < 0.001 | |||
| 3 months postoperative | 20.6 ± 4.9 | 9.8 ± 3.4 | < 0.001 | +6.4 (45.1) | -0.7 (6.7) | < 0.001 |
| Quality of life scores (SF-36) | ||||||
| Role-emotional | ||||||
| Baseline | 68.5 ± 15.7 | 78.5 ± 10.7 | < 0.001 | |||
| 3 months postoperative | 47.8 ± 18.2 | 62.5 ± 14.7 | < 0.001 | -20.7 (30.2) | -16.0 (20.4) | < 0.001 |
| Social functioning | ||||||
| Baseline | 75.2 ± 14.1 | 82.5 ± 15.7 | < 0.001 | |||
| 3 months postoperative | 60.3 ± 15.7 | 75.1 ± 15.7 | < 0.001 | -14.9 (19.8) | -7.4 (9.0) | < 0.001 |
| General health | ||||||
| Baseline | 70.5 ± 16.3 | 72.0 ± 15.9 | 0.372 | |||
| 3 months postoperative | 55.2 ± 17.1 | 71.5 ± 16.2 | < 0.001 | -15.3 (21.7) | -0.5 (0.7) | < 0.001 |
| Physical functioning | ||||||
| Baseline | 80.0 ± 18.5 | 88.0 ± 17.8 | < 0.001 | |||
| 3 months postoperative | 65.0 ± 19.2 | 81.0 ± 18.0 | < 0.001 | -15.0 (18.8) | -7.0 (8.0) | < 0.001 |
| Role-physical | ||||||
| Baseline | 72.0 ± 16.0 | 74.0 ± 15.5 | 0.228 | |||
| 3 months postoperative | 55.0 ± 17.0 | 73.5 ± 16.5 | < 0.001 | -17.0 (23.6) | -0.5 (0.7) | < 0.001 |
| Mental health | ||||||
| Baseline | 70.0 ± 14.5 | 72.0 ± 14.0 | 0.191 | |||
| 3 months postoperative | 50.0 ± 15.5 | 71.5 ± 14.0 | < 0.001 | -20.0 (28.6) | -0.5 (0.7) | < 0.001 |
| Vitality | ||||||
| Baseline | 65.0 ± 15.0 | 67.0 ± 14.5 | 0.213 | |||
| 3 months postoperative | 50.0 ± 16.0 | 66.5 ± 14.5 | < 0.001 | -15.0 (23.1) | -0.5 (0.7) | < 0.001 |
| Pain | ||||||
| Baseline | 75.0 ± 15.0 | 76.0 ± 14.5 | 0.541 | |||
| 3 months postoperative | 60.0 ± 16.0 | 75.5 ± 14.5 | < 0.001 | -15.0 (20.0) | -0.5 (0.7) | < 0.001 |
At 3 months postoperatively, SDS and HAMD-17 scores in the depression group increased significantly from baseline (SDS: 58.4 ± 7.2 vs 48.3 ± 6.7, P < 0.001; HAMD-17: 20.6 ± 4.9 vs 14.2 ± 3.8, P < 0.001), showing an increase of 10.1 points (20.9%) in SDS scores and 6.4 points (45.1%) in HAMD-17 scores. In contrast, scores in the non-depression group re
Patients in the depression group showed significant deterioration in all domains of quality of life as measured by SF-36, with the largest decreases observed in role-emotional (47.8 ± 18.2 at 3 months vs 68.5 ± 15.7 at baseline, P < 0.001) and social functioning (60.3 ± 15.7 at 3 months vs 75.2 ± 14.1 at baseline, P < 0.001) domains. Specifically, role-emotional functioning decreased by 20.7 points (30.2%) and social functioning decreased by 14.9 points (19.8%). This suggests that postoperative depression has a particularly prominent impact on patients’ emotional states and social capabilities, potentially leading to greater difficulties in daily life and social interactions (Table 5).
| Indicator | Depressed group | Non-depressed group | Between-group difference | P value |
| Psychological assessment | ||||
| Preoperative SDS score | 48.3 ± 6.7 | 42.1 ± 5.9 | 6.2 | < 0.001 |
| Postoperative SDS score at 3 months | 58.4 ± 7.2 | 41.8 ± 6.1 | 16.6 | < 0.001 |
| Change in SDS score | +10.1 (20.9%) | -0.3 (0.7%) | 10.4 | < 0.001 |
| Preoperative HAMD-17 score | 14.2 ± 3.8 | 10.5 ± 3.1 | 3.7 | < 0.001 |
| Postoperative HAMD-17 score at 3 months | 20.6 ± 4.9 | 9.8 ± 3.4 | 10.8 | < 0.001 |
| Change in HAMD-17 score | +6.4 (45.1%) | -0.7 (6.7%) | 7.1 | < 0.001 |
| Quality of life scores (SF-36) | ||||
| Baseline role-emotional | 68.5 ± 15.7 | 69.2 ± 16.1 | -0.7 | 0.698 |
| Postoperative role-emotional at 3 months | 47.8 ± 18.2 | 70.1 ± 15.9 | -22.3 | < 0.001 |
| Change in role-emotional | -20.7 (30.2%) | 0.9 (1.3%) | -21.6 | < 0.001 |
| Baseline social functioning | 75.2 ± 14.1 | 74.8 ± 15.2 | 0.4 | 0.806 |
| Postoperative social functioning at 3 months | 60.3 ± 15.7 | 75.1 ± 14.8 | -14.8 | < 0.001 |
| Change in social functioning | -14.9 (19.8%) | 0.3 (0.4%) |
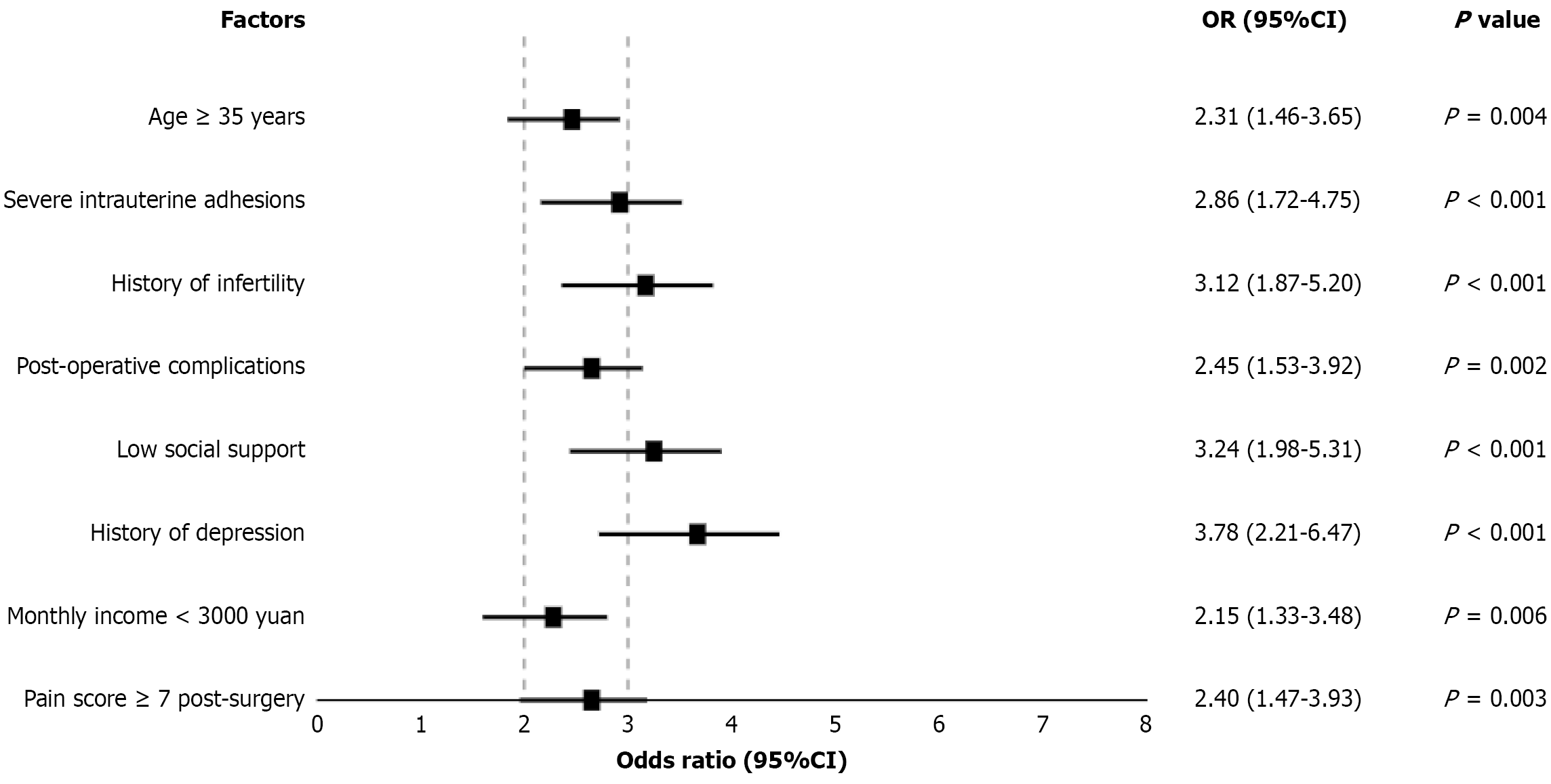
Factors with statistical significance in the univariate analysis were included in the multivariate logistic regression analysis. The results showed that age ≥ 35 years (OR = 2.31, 95%CI: 1.46-3.65, P = 0.004), severe IUA (OR = 2.86, 95%CI: 1.72-4.75, P < 0.001), history of infertility (OR = 3.12, 95%CI: 1.87-5.20, P < 0.001), post-operative complications (OR = 2.45, 95%CI: 1.53-3.92, P = 0.002), and low social support (OR = 3.24, 95%CI: 1.98-5.31, P < 0.001) were independent risk factors for post-operative depression in patients with IUA.
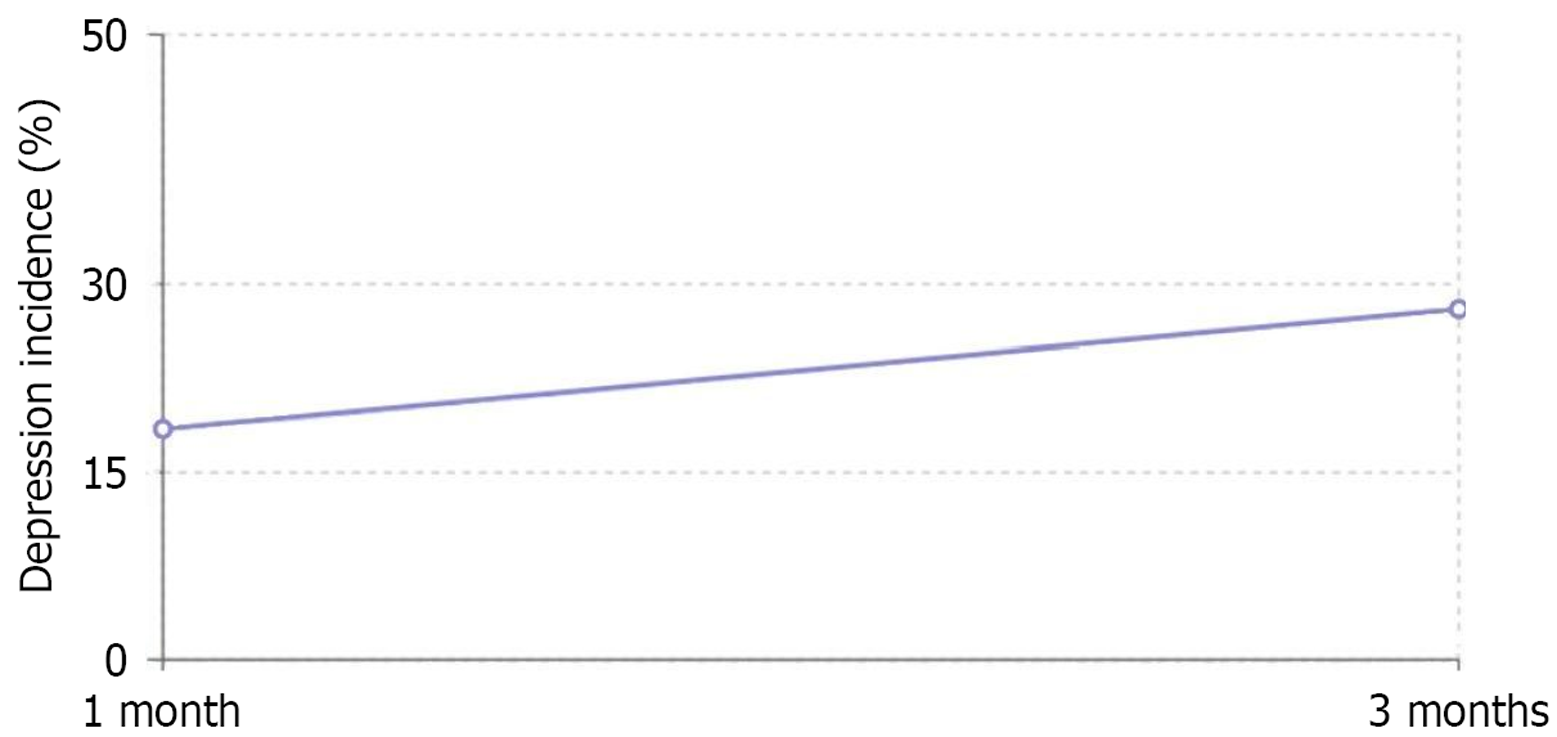
The line chart tracks depression incidence at two critical follow-up intervals: 1 month and 3 months after surgery. In the 1-month assessment, 18.5% of patients (74 out of 400) exhibited clinical symptoms of depression. By the 3-month follow-up, this percentage increased significantly to 28.0% (112 out of 400). This upward trend demonstrates that psychological symptoms continue to evolve beyond the immediate postoperative period, with many patients developing depression between weeks 4-8 after surgery (mean onset at 5.8 ± 2.3 weeks).
IUA represent a significant gynecological condition characterized by the formation of fibrous tissue within the uterine cavity, often leading to menstrual abnormalities, infertility, and recurrent pregnancy loss. The psychological impact of this condition, particularly following surgical intervention, has not been adequately addressed in the literature despite its potential significance for patient outcomes and quality of life. Our study demonstrates that postoperative depressive disorders are relatively common in patients with IUA, affecting approximately 28.0% of patients within three months after hysteroscopic adhesiolysis.
The prevalence of postoperative depression found in our study is notably higher than the general female population, where depression rates typically range from 5%-12%[15-17]. However, it is comparable to rates reported in patients with other gynecological conditions impacting fertility and reproductive function. For instance, studies on endometriosis patients report depression rates of 24%-38%[18], while infertility patients show rates of 15%-30%[19]. This suggests that reproductive health concerns may constitute a specific psychological vulnerability for women, particularly when associated with potential impact on fertility and family planning[20].
The pathophysiology underlying the association between IUA and depression likely involves multiple mechanisms[21]. Reproductive hormonal fluctuations, especially estrogen, have established effects on neurotransmitter systems involved in mood regulation. The endometrial damage in IUA may disrupt normal hormonal feedback loops, potentially contributing to mood disturbances[22,23]. Additionally, inflammatory processes involved in both adhesion formation and depression may represent another link, as pro-inflammatory cytokines have been implicated in the pathogenesis of depression.
Our finding that advanced age (≥ 35 years) represents an independent risk factor for postoperative depression (OR = 2.05, 95%CI: 1.27-3.32, P = 0.003) aligns with reproductive concerns that become more pressing with advancing maternal age. Women in this age group may experience greater psychological distress due to the perceived limited window for childbearing[24]. The particularly strong association between previous psychological disorders and postoperative depression (OR = 4.16, 95%CI: 2.24-7.74, P < 0.001) emphasizes the importance of pre-surgical psychological screening and targeted interventions for vulnerable patients[25].
The significant correlation between IUA severity and depression severity (r = 0.462, P < 0.001) suggests a dose-response relationship that may reflect both the psychological impact of a more serious diagnosis and the more intensive and potentially complicated surgical procedures required for severe cases[26]. Indeed, our data showed that operation time was significantly longer in the depression group (63.2 ± 18.7 minutes vs 52.4 ± 15.1 minutes, P < 0.001), potentially indicating more complex procedures with greater psychological impact[27].
The identified protective factors higher education, good social support, and restoration of normal menstruation offer important insights for clinical practice. Education level may influence coping strategies, health literacy, and access to resources[28]. Social support has been consistently associated with better mental health outcomes across various con
The history of infertility (OR = 2.58, 95%CI: 1.66-4.01, P < 0.001) and recurrent miscarriage (OR = 2.82, 95%CI: 1.73-4.59, P < 0.001) as risk factors aligns with existing literature on the psychological impact of reproductive challenges[32]. The emotional burden of repeated pregnancy loss and failed fertility attempts can create a psychological vulnerability that may be exacerbated by surgical intervention for IUA[24], particularly if patients view the surgery as their “last hope” for fertility restoration[33,34].
Notably, our findings on postoperative complications as a risk factor (OR = 2.47, 95%CI: 1.39-4.38, P = 0.002) emphasize the importance of surgical technique and postoperative care in minimizing not only physical but also psychological mor
These findings have important clinical implications. First, they support the routine screening for depressive symptoms in the postoperative period for patients with IUA, especially those with identified risk factors[37]. Second, they suggest that targeted psychological interventions may be beneficial for high-risk groups[38]. Finally, our results highlight the importance of a multidisciplinary approach to IUA management, incorporating mental health professionals into the care team[39].
Several limitations warrant discussion. The retrospective design limits causal inference and introduces potential recall bias, although we minimized this by relying strictly on medical records and standardized scale scores rather than subjec
Future research should employ prospective designs with longer follow-up periods to better understand the longitudinal relationship between IUA, surgical intervention, and psychological outcomes. Specific research priorities include: (1) Validation of our predictive model in external cohorts; (2) Exploration of inflammatory pathways and hormonal pro
In conclusion, this study highlights the substantial prevalence of postoperative depression among patients with IUA and identifies several modifiable and non-modifiable risk factors. The findings underscore the importance of integrating psy
| 1. | Zhou Z, Wang H, Zhang X, Song M, Yao S, Jiang P, Liu D, Wang Z, Lv H, Li R, Hong Y, Dai J, Hu Y, Zhao G. Defective autophagy contributes to endometrial epithelial-mesenchymal transition in intrauterine adhesions. Autophagy. 2022;18:2427-2442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 80] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 2. | Emingr Michal, Halaj Matúš, Malčák Michal, Hanáček Jiří. Prevention of intrauterine adhesions. Ceska Gynekol. 2023;88:210-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 3. | Zhao G, Hu Y. Mechanistic insights into intrauterine adhesions. Semin Immunopathol. 2024;47:3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 4. | Alameddine S, Lucidi A, Jurkovic D, Timor Tritsch I, Coutinho CM, Ranucci L, Buca D, Khalil A, Jauniaux E, Mappa I, D'Antonio F. Treatments for cesarean scar pregnancy: a systematic review and meta-analysis. J Matern Fetal Neonatal Med. 2024;37:2327569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 5. | Yang F, Zhang Q, Shuai Y, Wang Z, Jing H, Wang X, Deng C, Lin F, Lai H. The value of cesarean scar diverticulum in diagnosis of adverse events during dilatation and curettage in patient with cesarean scar pregnancy. Int J Gynaecol Obstet. 2025;168:525-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Wu L, Zhong Y, Wu D, Xu P, Ruan X, Yan J, Liu J, Li X. Immunomodulatory Factor TIM3 of Cytolytic Active Genes Affected the Survival and Prognosis of Lung Adenocarcinoma Patients by Multi-Omics Analysis. Biomedicines. 2022;10:2248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 85] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 7. | Wang PH, Yang ST, Chang WH, Liu CH, Liu HH, Lee WL. Intrauterine adhesion. Taiwan J Obstet Gynecol. 2024;63:312-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 8. | Tang R, Zhang W, Xiao X, Li W, Chen X, Wang X. Intrauterine interventions options for preventing recurrence after hysteroscopic adhesiolysis: a systematic review and network meta-analysis of randomized controlled trials. Arch Gynecol Obstet. 2024;309:1847-1861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 9. | Vitale SG, Riemma G, Carugno J, Perez-Medina T, Alonso Pacheco L, Haimovich S, Parry JP, Di Spiezio Sardo A, De Franciscis P. Postsurgical barrier strategies to avoid the recurrence of intrauterine adhesion formation after hysteroscopic adhesiolysis: a network meta-analysis of randomized controlled trials. Am J Obstet Gynecol. 2022;226:487-498.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 83] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 10. | Hong W, Wu Z, Li L, Wang B, Li X. Intrauterine adhesions treated with hysteroscopic adhesiolysis and subsequent obstetric outcome: A retrospective matched cohort study. BJOG. 2025;132:155-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 11. | Qiao X, Liu D, Liu C, Pei T, Ouyang Y. Reproductive Outcomes After Hysteroscopic Adhesiolysis in Patients Experiencing Recurrent Pregnancy Loss and Intrauterine Adhesions. J Minim Invasive Gynecol. 2025;32:57-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Wu L, Liu Q, Ruan X, Luan X, Zhong Y, Liu J, Yan J, Li X. Multiple Omics Analysis of the Role of RBM10 Gene Instability in Immune Regulation and Drug Sensitivity in Patients with Lung Adenocarcinoma (LUAD). Biomedicines. 2023;11:1861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 78] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 13. | Chen CY, Cheng CF, Chen PC, Wu CS, Lin MC, Su MH, Chang CY, Pan YJ, Huang YT, Fan CC, Wang SH. Familial coaggregation and shared genetic influence between major depressive disorder and gynecological diseases. Eur J Epidemiol. 2024;39:1161-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 14. | Yin T, Lu Y, Xiong W, Yu C, Yin D, He H. Occupational Risk Factors for Physical and Mental Health in Primary Healthcare Providers: A National Cross-Sectional Survey from 62 Urban Communities in China. J Multidiscip Healthc. 2023;16:751-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Lewkowitz AK, Whelan AR, Ayala NK, Hardi A, Stoll C, Battle CL, Tuuli MG, Ranney ML, Miller ES. The effect of digital health interventions on postpartum depression or anxiety: a systematic review and meta-analysis of randomized controlled trials. Am J Obstet Gynecol. 2024;230:12-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 16. | Vas C, Jain A, Trivedi M, Jha MK, Mathew SJ. Pharmacotherapy for Treatment-Resistant Depression: Antidepressants and Atypical Antipsychotics. Psychiatr Clin North Am. 2023;46:261-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Xu M, Li X, Teng T, Huang Y, Liu M, Long Y, Lv F, Zhi D, Li X, Feng A, Yu S, Calhoun V, Zhou X, Sui J. Reconfiguration of Structural and Functional Connectivity Coupling in Patient Subgroups With Adolescent Depression. JAMA Netw Open. 2024;7:e241933. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 18. | Heres CK, Rindos NB, Fulcher IR, Allen SE, King NR, Miles SM, Donnellan NM. Opioid Use After Laparoscopic Surgery for Endometriosis and Pelvic Pain. J Minim Invasive Gynecol. 2022;29:1344-1351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 19. | Kocas HD, Rubin LR, Lobel M. Stigma and mental health in endometriosis. Eur J Obstet Gynecol Reprod Biol X. 2023;19:100228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 20. | Wu L, Zheng Y, Ruan X, Wu D, Xu P, Liu J, Wu D, Li X. Long-chain noncoding ribonucleic acids affect the survival and prognosis of patients with esophageal adenocarcinoma through the autophagy pathway: construction of a prognostic model. Anticancer Drugs. 2022;33:e590-e603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (5)] |
| 21. | Bulun SE, Yildiz S, Adli M, Chakravarti D, Parker JB, Milad M, Yang L, Chaudhari A, Tsai S, Wei JJ, Yin P. Endometriosis and adenomyosis: shared pathophysiology. Fertil Steril. 2023;119:746-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 135] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 22. | Gamble KL, Berry R, Frank SJ, Young ME. Circadian clock control of endocrine factors. Nat Rev Endocrinol. 2014;10:466-475. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 432] [Cited by in RCA: 362] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 23. | Wang H, Li J, Chen L, He L. Do women’s natural hormonal fluctuations modulate prosociality? A within-subject analysis. Psychoneuroendocrinology. 2022;138:105663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Matsushima M, Yamada H, Kondo N, Arakawa Y, Tabuchi T. Married women's decision to delay childbearing, and loneliness, severe psychological distress, and suicidal ideation under crisis: online survey data analysis from 2020 to 2021. BMC Public Health. 2023;23:1642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 25. | Marek RJ, Le JT, Hapenciuc G, Philip MA, Chiu J, Block AR, Ben-Porath YS. Incremental Contribution of the Minnesota Multiphasic Personality Inventory - 3 to Predicting One-Year Postoperative Spinal Cord Surgery/Spinal Cord Stimulation Outcomes. J Clin Psychol Med Settings. 2024;31:77-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Wu L, Zhong Y, Yu X, Wu D, Xu P, Lv L, Ruan X, Liu Q, Feng Y, Liu J, Li X. Selective poly adenylation predicts the efficacy of immunotherapy in patients with lung adenocarcinoma by multiple omics research. Anticancer Drugs. 2022;33:943-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 27. | Josephs CA, Shaffer VO, Kucera WB. Impact of Mental Health on General Surgery Patients and Strategies to Improve Outcomes. Am Surg. 2023;89:2636-2643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 28. | Acoba EF. Social support and mental health: the mediating role of perceived stress. Front Psychol. 2024;15:1330720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 306] [Cited by in RCA: 184] [Article Influence: 92.0] [Reference Citation Analysis (0)] |
| 29. | Cao F, Li J, Xin W, Cai N. Impact of social support on the resilience of youth: mediating effects of coping styles. Front Public Health. 2024;12:1331813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 30. | Bagade T, Mersha AG, Majeed T. The social determinants of mental health disorders among women with infertility: a systematic review. BMC Womens Health. 2023;23:668. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 31. | Martins C, Mitchell JJ, Hamer M, Blodgett JM. Associations between psychological distress in adolescence and menstrual symptoms across life: Longitudinal evidence from the 1970 British Cohort Study. J Affect Disord. 2024;354:712-718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 32. | Cuenca D. Pregnancy loss: Consequences for mental health. Front Glob Womens Health. 2022;3:1032212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 33. | Mendes DCG, Fonseca A, Cameirão MS. The psychological impact of Early Pregnancy Loss in Portugal: incidence and the effect on psychological morbidity. Front Public Health. 2023;11:1188060. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 34. | Tavoli Z, Mohammadi M, Tavoli A, Moini A, Effatpanah M, Khedmat L, Montazeri A. Quality of life and psychological distress in women with recurrent miscarriage: a comparative study. Health Qual Life Outcomes. 2018;16:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 65] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 35. | Lanini I, Amass T, Calabrisotto CS, Fabbri S, Falsini S, Adembri C, Di Filippo A, Romagnoli S, Villa G. The influence of psychological interventions on surgical outcomes: a systematic review. J Anesth Analg Crit Care. 2022;2:31. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 36. | Richardson E, Patterson R, Meltzer-Brody S, McClure R, Tow A. Transformative Therapies for Depression: Postpartum Depression, Major Depressive Disorder, and Treatment-Resistant Depression. Annu Rev Med. 2025;76:81-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 37. | Zhou D, Wang LK, Wu HY, Gao L, Yang XD. Early-stage postoperative depression and anxiety following orthognathic surgery: a cross-sectional study. BMC Anesthesiol. 2024;24:338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 38. | Li Y, Du J, Du L, Li S, Zhang J. Managements for perioperative anxiety in patients with gastrointestinal cancers. Front Psychiatry. 2024;15:1391403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Dawe J, Cronshaw H, Frerk C. Learning from the multidisciplinary team: advancing patient care through collaboration. Br J Hosp Med (Lond). 2024;85:1-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/