Published online Nov 19, 2025. doi: 10.5498/wjp.v15.i11.110699
Revised: July 9, 2025
Accepted: September 18, 2025
Published online: November 19, 2025
Processing time: 144 Days and 20 Hours
Patients with chronic kidney disease (CKD), particularly those undergoing maintenance hemodialysis (MHD), often experience mental health issues. Recent studies have indicated a significantly elevated prevalence of anxiety and de
Core Tip: Studies have revealed that patients with chronic kidney disease (CKD) on long-term hemodialysis may experience mental health impairment. Multiple factors can exacerbate anxiety and depression; it interacts with the physiological processes of CKD, forming a pathological psychophysiological interaction mechanism that affects their quality of life and prognosis. A comprehensive management approach for patients with CKD should include a multidisciplinary medical team, personalized non-pharmacological intervention, and integrated pharmacological therapy. Assessing the risk-benefit ratio of antidepressants and hypnotics and drug intervention combined with non-drug intervention may help alleviate anxiety and depression, improve quality of life, break the psychophysiological vicious cycle, and optimize long-term outcomes.
- Citation: Li JB, Ye XB, Tu XJ. Interaction between anxiety, depression, and low resilience predicts poor prognosis in patients with chronic kidney disease. World J Psychiatry 2025; 15(11): 110699
- URL: https://www.wjgnet.com/2220-3206/full/v15/i11/110699.htm
- DOI: https://dx.doi.org/10.5498/wjp.v15.i11.110699
Patients with chronic kidney disease (CKD) experience severe nephron damage, metabolite retention, and water-electrolyte and acid-base status imbalances, with complications affecting multiple organ systems. Hemodialysis is a crucial approach for delaying CKD progression[1]. However, even regular maintenance hemodialysis (MHD) involves various complications and may cause significant patient distress. Notably, the prevalences of anxiety and depression are quite high in this population, severely compromising their quality of life (QoL)[2-4]. Recently, research results by Ye et al[5] demonstrated that approximately half of the patients with CKD exhibited varying degrees of anxiety, depression, and elevated self-perceived burden (negative self-evaluation arising from symptom-related guilt), with all having significant negative correlations with resilience. Their study suggests that low resilience could exacerbate anxiety-depression and self-perceived burden in CKD patients, it provides robust evidence for the association between psychological resilience and negative emotions. However, the mechanism through which these factors interact with CKD progression remains unclear. In this editorial, we explore the psychophysiological interplay in CKD outcomes, evaluating current evidence and hypothesizing mechanistic pathways. This has significant practical implications for developing intervention strategies to improve CKD prognosis, enhance patients’ QoL, and achieve better clinical outcomes.
Depression and anxiety in patients undergoing MHD show significant associations with higher cognitive distortions, lower resilience, and poorer QoL. Lower resilience is an independent risk factor for reduced QoL[6]. Depressive symptoms correlate with decreased self-efficacy and resilience, impairing patients’ activities of daily living and overall QoL[7]. In patients undergoing dialysis for CKD, diminished QoL is significantly associated with multiple factors, including depressive symptoms, complications (e.g., recurrent infections and anemia), chronic pain, and post-dialysis fatigue[8]. Studies reveal that treatment adherence is positively correlated with resilience[9] and sleep quality scores in patients with CKD[10]. These findings suggest that patients with lower resilience tend to have poorer treatment compliance and more frequent sleep disturbances. Resilience showed significant positive correlations with disease awareness, self-efficacy, and self-esteem and a negative correlation with age in patients undergoing MHD. This implies that older patients undergoing MHD may have particularly impaired resilience[11]. Notably, in patients undergoing MHD, anxiety is a significant independent determinant and depression is a crucial independent influencing factor for lower QoL[12]. A study involving 707 patients undergoing MHD and with depressive symptoms reported that de
Studies show that patients with CKD have significantly higher depression, anxiety, and fatigue scores than healthy controls, along with elevated serum markers of neuronal injury (neurofilament light chain, myelin basic protein, and nestin) and inflammation [C-reactive protein (CRP) and interleukin-10 (IL-10)]. These neuronal damage markers strongly correlate with neuropsychiatric symptom severity[20]. CKD-related neuroinflammation may exacerbate anxiety, depression, and cognitive dysfunction via pro-inflammatory cytokines (IL-1β, tumor necrosis factor-α, and C-C motif chemokine ligand 2) crossing the blood-brain barrier[21].
Meta-analyses reveal that patients with CKD with comorbid depression exhibit higher pro-inflammatory cytokines (CRP, IL-6, tumor necrosis factor-α) and lower anti-inflammatory IL-10 than non-depressed counterparts[22]. Serum endotoxin and IL-6 Levels closely correlate with depressive and fatigue symptoms, with endotoxin and depression independently predicting fatigue[23]. CKD patients frequently exhibit insulin resistance, muscle wasting, malnutrition, and chronic low-grade inflammation, along with dysregulated brain-derived neurotrophic factor (BDNF). BDNF abnormalities are implicated in insulin resistance, muscle atrophy, depression, oxidative stress, and inflammation, highlighting its central role in CKD’s mind-body interface[24]. Interventional and autonomic evidence further supports this link. A 10-day noise management intervention improved sleep quality, anxiety, blood pressure, heart rate, cortisol levels, and QoL in patients with CKD[25]. These patients also show reduced heart rate variability (HRV), which correlates with disease progression[26] and independently predicts 8-year mortality in MHD patients[27]. Notably, HRV abnormalities are associated with chronic stress, sleep disturbance, and depression[28]. Thus, depression and anxiety in CKD may worsen outcomes via neuroimmune dysregulation (pro-/anti-inflammatory imbalance), hypothalamic-pituitary-adrenal axis dysfunction (cortical alterations), autonomic dysfunction (reduced HRV), and neuro-immune-endocrine network disruption (BDNF changes). This bidirectional psychophysiological interaction may create a vicious cycle in which poor health exacerbates mood disorders, further deteriorating QoL and prognosis (Figure 1). These mechanisms likely contribute to CKD’s adverse clinical trajectory.
The degree of acceptance of the need for long-term MHD is positively correlated with QoL. Accepting the reality of the disease is an important independent determinant of higher QoL[29]. Therefore, resisting the reality of illness negatively impacts QoL. In contrast, accepting it and adapting to the unchangeable fact of severe kidney disease are proactive psychological strategies for facing these health challenges. For example, life skills training interventions (focusing on goal setting, cognitive restructuring, acceptance/gratitude/mindfulness, and personal strengths) can improve anxiety, depression, QoL, illness perception, self-efficacy, self-management skills, benefit finding, and resilience in patients undergoing high-flux hemodialysis (HFHD)[30]. Psychoeducational interventions incorporating coping method, goal setting, and relaxation techniques demonstrated benefits such as improved QoL and reduced anxiety/depression in patients undergoing MHD in short- (< 1 month) and medium-term (1-3 months)[31]. Narrative nursing care with enhanced psychological elements improves treatment safety, reduces anxiety/depression, and enhances QoL in patients with CKD undergoing HFHD[32]. Evidence-based psychological therapies such as cognitive behavioral therapy (CBT) are effective for treating depression in patients with CKD[33,34]. CBT combined with resilience training proved superior to CBT alone in enhancing resilience, alleviating anxiety/depression symptoms, and improving QoL in patients undergoing MHD[35]. A controlled study on mindfulness meditation combined with progressive muscle relaxation training in patients undergoing MHD showed that the intervention group demonstrated significant improvements in exercise capacity, physical function, general health, energy, symptom/problem checklist, sleep, pain, cognitive function, emotional well-being, and QoL scores after 12 weeks[36,37]. After 12 weeks of mindfulness-based CBT, patients undergoing HFHD had significantly improved QoL, depression, anxiety, perceived stress, and serum creatinine levels[38]. Psychosocial interventions (including counseling, support groups, CBT, relaxation techniques, education, and social support) effectively reduce depression severity and improve QoL in patients with CKD[39]. Music therapy effectively alleviates depressive symptoms and enhances QoL in patients undergoing MHD, particularly in terms of functional capacity, pain management, mental health, vitality, and overall health status[40]. These findings suggest that psychological interventions positively influence mental health and physical outcomes, in patients undergoing MHD.
Studies have shown that fatigue tends to be more severe on dialysis days and negatively correlates with physical activity levels on non-dialysis days[41]. Depression severity is positively associated with fatigue, suggesting that physical inactivity and depression may exacerbate fatigue symptoms[41]. Moderate physical exercise is beneficial for patients with CKD. Breathing exercises performed thrice daily (each session lasts 5 minutes: Inhale slowly through the nose for 4 seconds, hold the breath for 7 seconds, then exhale for 8 seconds to release the air) for 30 days significantly reduced depression scores and improved QoL and symptom control in patients undergoing MHD[42]. Clinical benefits of structured exercise programs such as aerobic and resistance training include improvement in depressive symptoms, restless legs syndrome, muscle cramps, and fatigue. Exercise improves inflammation, oxidative stress, vascular function, immune response, and macromolecular metabolism across all stages of CKD, while significantly enhancing patients’ QoL[43-45] and muscle strength and reducing frailty. Personalized exercise plans may lower all-cause and infection-related mortalities, thereby improving long-term prognosis[46-48]. Notable clinical trials, such as a randomized controlled study combining resistance training (training is administered 4 times per week for 24 weeks; during the first week, it begins at 50% of the initial one-repetition maximum, with an increase in the load by 5% monthly) with religious music (Quran recitation), demonstrated significantly greater improvements in 6-minute walk test performance, anxiety, depression, and QoL than exercise alone[49]. Participants completed 36 supervised sessions over 12 weeks (frequency: 3 ×/week). Session structure: 5-minute warm-up (dynamic stretching), 20-minute endurance phase (magnetic-resistance mini-cycle ergometry), and 5-minute cool-down (static stretching). This exercise program caused a measurable QoL improvement and depression reduction in the intervention group, whereas no significant changes were observed in the control group[50]. Comprehensive lifestyle approaches such as nature-based physical activities promote physical and mental well-being by reducing anxiety, fatigue, and stress while enhancing social skills[51]. Collectively, these findings suggest that appropriately personalized physical activity regimens may significantly benefit the overall physical and mental health of patients undergoing MHD.
Depressive symptoms are significantly correlated with low resilience and can impair patients’ activities of daily living and overall QoL[52]. Clinical studies on sertraline [a selective serotonin reuptake inhibitor (SSRI)] demonstrate the following benefits in patients with end-stage renal disease undergoing dialysis: Improved depressive mood, reduced intradialytic refractory syncope and pruritus, lowered hypotensive episode risk, modulated neurotransmitters by enhancing serotonergic transmission and downregulating postsynaptic receptors (attenuating hypovolemic stress responses), and maintained a good safety profile without affecting platelet function[53]. SSRIs also exhibit the following anti-inflammatory effects: Decreased pro-inflammatory cytokines (CRP, IL-1β, IL-6, and IL-12) and increased anti-inflammatory factors (IL-4, transforming growth factor-β1)[54-56]. The European Renal Best Practice guidelines recommend an 8-12-week SSRI trial for moderate-to-severe depression in patients undergoing dialysis and efficacy evaluation at 12 weeks to avoid ineffective medication[57].
Thus, the following safety considerations are recommended. Cardiac risks: Citalopram and escitalopram carry higher QT prolongation risk, potentially increasing sudden cardiac death risks in patients undergoing MHD[58]; comparative safety: Fluoxetine, fluvoxamine, paroxetine, and sertraline show lower cardiac risk, no significant association with 5-year all-cause mortality, cardiovascular events, or CKD progression, and potential 1-year increased risk of hip fractures (post-fall) and upper gastrointestinal bleeding. Long-term adverse outcomes - including all-cause mortality, major adverse cardiovascular events, CKD progression, and suicidal behavior - showed no significant association with SSRI use[59]. In CKD patients with comorbid depression and anxiety, agomelatine and paroxetine significantly improved symptoms and daily living capacity. Notably, agomelatine exhibited superior efficacy in alleviating anxiety and depression, as well as improving sleep quality[60,61]. Comparative studies found melatonin more effective than alprazolam in enhancing sleep quality and reducing insomnia severity[62]. A multinational survey of 112 medical institutions across three countries identified melatonin as the most frequently prescribed first-line treatment for insomnia in CKD patients with comorbid depression, followed by zolpidem and zopiclone[63]. In an eight-week trial comparing zolpidem (10 mg nightly) and acupressure in patients undergoing MHD, the zolpidem group showed significant improvements in pruritus, sleep quality, and QoL, with no serious adverse events in either group[64].
Antidepressants and hypnotics benefit patients undergoing MHD with anxiety, depression, poor resilience, and insomnia. However, given the severely impaired renal function in CKD, their use requires careful assessment of risk - benefit ratios. When the clinical benefits of SSRIs or hypnotics outweigh the risks, individualized treatment regimens should be adopted. Drug selection should consider cardiac status, fracture risk, drug interactions, and allergy history of allergies. Treatment should begin with low doses, gradually and slowly increased, and the cardiac QT interval regularly monitored. Tricyclic antidepressants should generally be avoided, as should the concurrent use of multiple antidepressants or hypnotics. Drugs with significant QT prolongation risk should be minimized, and attention paid to bone mineral density and gastrointestinal symptoms. Non-pharmacological interventions should be integrated to enhance efficacy and reduce adverse drug events.
Given the substantial impact of comorbid anxiety, depression, and low resilience in patients with CKD, managing these conditions poses significant challenges. These include diagnostic limitations (nephrologists often lack expertise in mental health assessment), pharmacological risks (renal impairment increases susceptibility to adverse drug reactions), and adherence difficulties (poor patient compliance). These are critical barriers that clinicians must address.
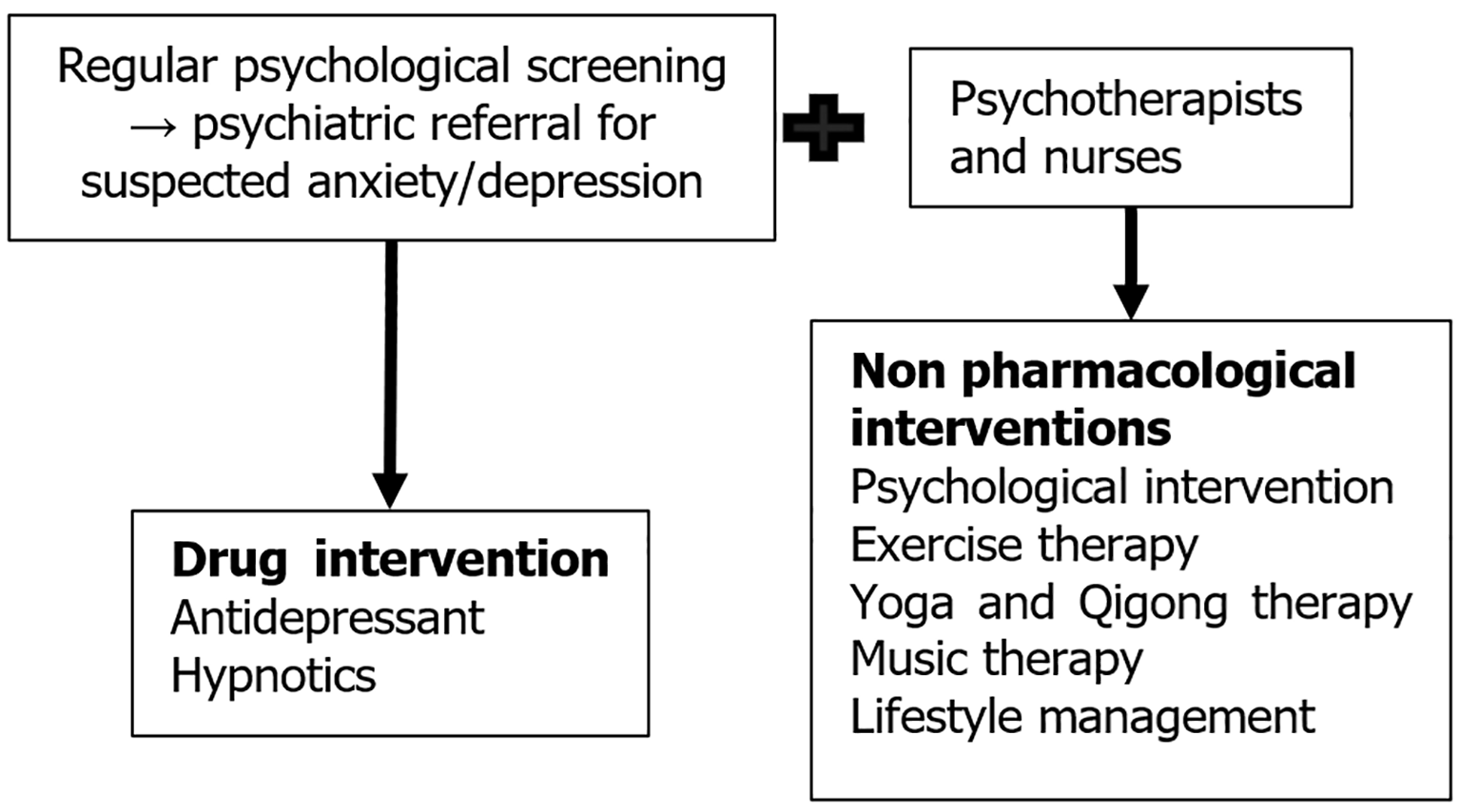
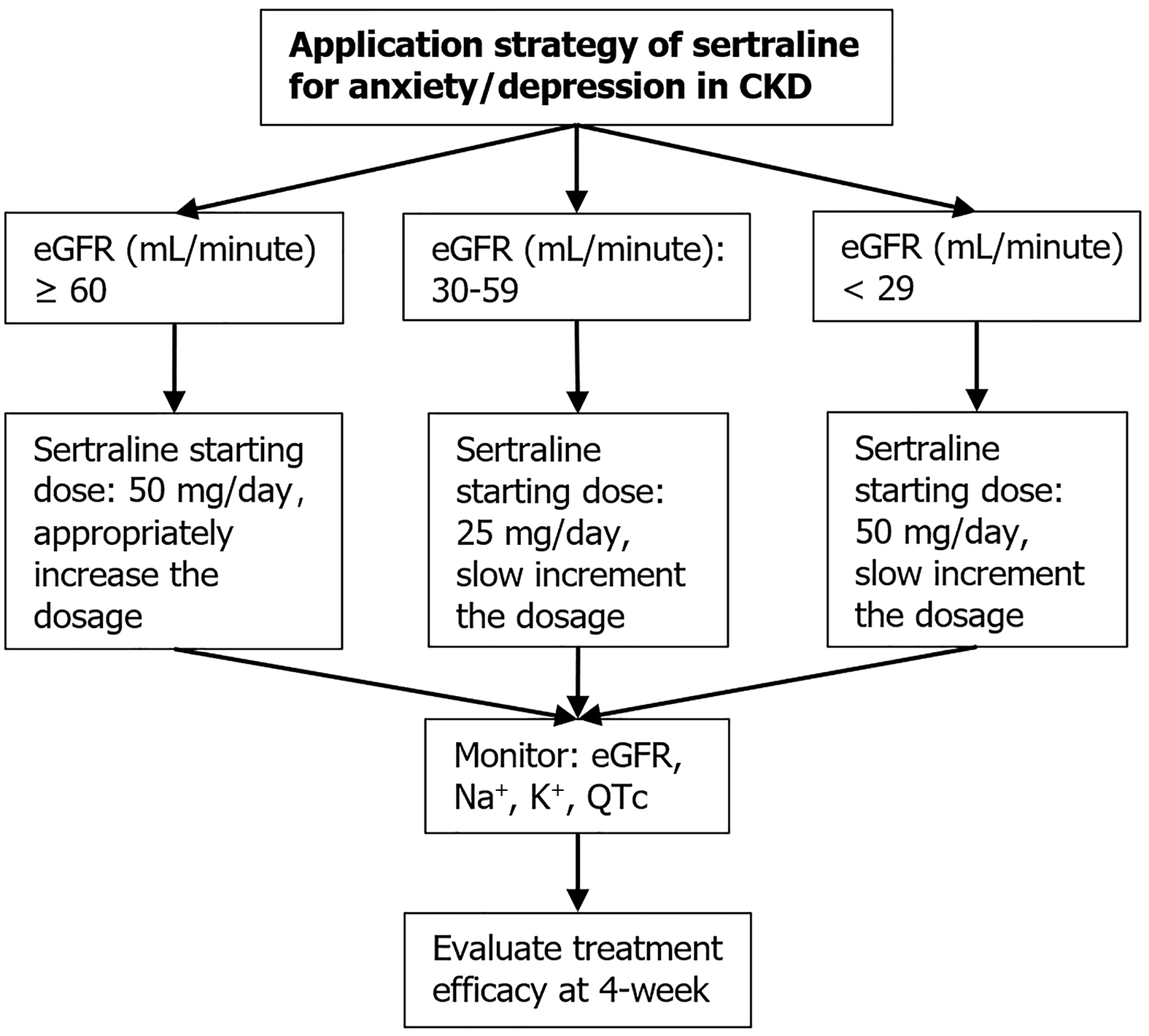
To tackle these challenges, a multidisciplinary approach is essential: (1) Nurses should undergo mental health evaluation training; and (2) They should assist physicians by conducting routine screenings (every 1-2 weeks) using the Patient Health Questionnaire 9 and Generalized Anxiety Disorder 7 scales. Suspected cases require prompt referral to psychiatrists and psychotherapist for diagnosis and treatment. By integrating nephrologists, psychiatrists, psychotherapists, and nurses, personalized care plans can be developed, including pharmacological interventions such as antidepressants or hypnotics. For example, when prescribing sertraline, regular monitoring of estimated glomerular filtration rate, electrolytes (Na+, K+), and electrocardiogram (QTc) intervals is essential (Figure 2).
Nurses can also collaborate with psychotherapists to deliver non-pharmacological interventions, forming a comprehensive, multidimensional care model (Figure 3). Furthermore, digital health technologies may augment traditional approaches, including smart wristbands for continuous monitoring of vital signs (heart rate, blood pressure, oxygen saturation) and virtual reality-assisted relaxation training. Integrating these strategies into CKD management pathways may help disrupt the vicious psychophysiological cycle, thereby achieving synergistic therapeutic outcomes.
Existing studies consistently report a high prevalence of comorbid anxiety and depression in patients with CKD. These negative emotions affect QoL and prognosis through multiple physiological pathways. Their impact may have been previously underestimated, underscoring the need for further investigation and proactive management. Low resilience significantly contributes to the onset and progression of anxiety and depression; however, the psychological and biological mechanisms underlying this relationship remain poorly understood. Most pharmacological and non-pharmacological interventions are based on short-term studies. Such outcomes impose limitations on creating well-rounded interventions to ameliorate QoL and prognosis in CKD populations. Future large-scale, controlled trials are required to assess long-term efficacy and safety. Additionally, research is required to examine cultural and geographical differences in non-pharmacological interventions and to evaluate the cost-effectiveness of comprehensive intervention models. A focus on bidirectional psychosomatic interactions and the development of multidimensional strategies may offer a novel approach to improving QoL and prognosis in patients with CKD.
| 1. | Gu F, Wu Y, Lu J, Zhang P, Qi H. Clinical implications of serum adropin and clusterin in chronic renal failure patients who received hemodialysis. Clin Chim Acta. 2025;573:120287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 2. | Aatif T, Achour I, Arache W, Zajjari Y, Montasser D, Mehssani J, El Kabbaj D. Anxiety and Depression Disorders in Adults on Chronic Hemodialysis. Saudi J Kidney Dis Transpl. 2022;33:566-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | Sharma R, Sharma SC, Chalise P, Regmee J, Sharma S. Anxiety and Depression among Patients with Chronic Kidney Disease Undergoing Haemodialysis in a Tertiary Care Centre: A Descriptive Cross-sectional Study. JNMA J Nepal Med Assoc. 2022;60:634-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 4. | Alencar SBV, de Lima FM, Dias LDA, Dias VDA, Lessa AC, Bezerra JM, Apolinário JF, de Petribu KC. Depression and quality of life in older adults on hemodialysis. Braz J Psychiatry. 2020;42:195-200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 5. | Ye YY, Tao LF, Yang YL, Wang YW, Yang XM, Xu HH. Correlation between anxiety, depression, self-perceived burden, and psychological resilience in patients with chronic renal failure on maintenance hemodialysis. World J Psychiatry. 2025;15:103850. [PubMed] [DOI] [Full Text] |
| 6. | González-Flores CJ, García-García G, Lerma A, Pérez-Grovas H, Meda-Lara RM, Guzmán-Saldaña RME, Lerma C. Resilience: A Protective Factor from Depression and Anxiety in Mexican Dialysis Patients. Int J Environ Res Public Health. 2021;18:11957. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 7. | Liu X, Yang X, Yao L, Zhang Q, Sun D, Zhu X, Xu T, Liu Q, Wang L. Prevalence and related factors of depressive symptoms in hemodialysis patients in northern China. BMC Psychiatry. 2017;17:128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Pretto CR, Winkelmann ER, Hildebrandt LM, Barbosa DA, Colet CF, Stumm EMF. Quality of life of chronic kidney patients on hemodialysis and related factors. Rev Lat Am Enfermagem. 2020;28:e3327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 9. | Freire de Medeiros CM, Arantes EP, Tajra RD, Santiago HR, Carvalho AF, Libório AB. Resilience, religiosity and treatment adherence in hemodialysis patients: a prospective study. Psychol Health Med. 2017;22:570-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 10. | Naeem Alharbi J. Beyond dialysis: Assessing therapeutic regimen compliance and sleep quality in chronic renal failure patients undergoing maintenance hemodialysis in Saudi Arabia. Ther Apher Dial. 2025;29:257-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Poudel B, Timalsina R. Factors associated with resilience among patients with end-stage kidney disease receiving hemodialysis in a teaching hospital: a cross-sectional study. BMC Nephrol. 2025;26:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 12. | Mok MMY, Liu CKM, Lam MF, Kwan LPY, Chan GCW, Ma MKM, Yap DYH, Chiu F, Choy CBY, Tang SCW, Chan TM. A Longitudinal Study on the Prevalence and Risk Factors for Depression and Anxiety, Quality of Life, and Clinical Outcomes in Incident Peritoneal Dialysis Patients. Perit Dial Int. 2019;39:74-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Chilcot J, Guirguis A, Friedli K, Almond M, Day C, Da Silva-Gane M, Davenport A, Fineberg NA, Spencer B, Wellsted D, Farrington K. Depression Symptoms in Haemodialysis Patients Predict All-Cause Mortality but Not Kidney Transplantation: A Cause-Specific Outcome Analysis. Ann Behav Med. 2018;52:1-8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 14. | Zhu N, Virtanen S, Xu H, Carrero JJ, Chang Z. Association between incident depression and clinical outcomes in patients with chronic kidney disease. Clin Kidney J. 2023;16:2243-2253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 15. | Liebman S, Li NC, Lacson E. Change in quality of life and one-year mortality risk in maintenance dialysis patients. Qual Life Res. 2016;25:2295-2306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | El Filali A, Bentata Y, Ada N, Oneib B. Depression and anxiety disorders in chronic hemodialysis patients and their quality of life: A cross-sectional study about 106 cases in the northeast of morocco. Saudi J Kidney Dis Transpl. 2017;28:341-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 17. | Bodessova S, Sultanova B, Bekenova N, Mursalova Z. Annual survival of patients with end-stage chronic kidney disease on supportive hemodialysis and its correlates. Pol Merkur Lekarski. 2024;52:392-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Voorend CGN, van Oevelen M, Nieberg M, Meuleman Y, Franssen CFM, Joosten H, Berkhout-Byrne NC, Abrahams AC, Mooijaart SP, Bos WJW, van Buren M; POLDER investigators. Impact of the COVID-19 pandemic on symptoms of anxiety and depression and health-related quality of life in older patients with chronic kidney disease. BMC Geriatr. 2021;21:650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 19. | Ford EC, Sohansoha GK, Patel NA, Billany RE, Wilkinson TJ, Lightfoot CJ, Smith AC. The association of micro and macro worries with psychological distress in people living with chronic kidney disease during the COVID-19 pandemic. PLoS One. 2024;19:e0309519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Al-Hakeim HK, Twaij BAA, Al-Naqeeb TH, Moustafa SR, Maes M. Neuronal damage and inflammatory biomarkers are associated with the affective and chronic fatigue-like symptoms due to end-stage renal disease. J Affect Disord. 2024;347:220-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 21. | Chagas YW, Vaz de Castro PAS, Simões-E-Silva AC. Neuroinflammation in kidney disease and dialysis. Behav Brain Res. 2025;483:115465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 22. | Jayakumar S, Jennings S, Halvorsrud K, Clesse C, Yaqoob MM, Carvalho LA, Bhui K. A systematic review and meta-analysis of the evidence on inflammation in depressive illness and symptoms in chronic and end-stage kidney disease. Psychol Med. 2023;53:5839-5851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 23. | Brys ADH, Di Stasio E, Lenaert B, Sanguinetti M, Picca A, Calvani R, Marzetti E, Gambaro G, Bossola M. Serum interleukin-6 and endotoxin levels and their relationship with fatigue and depressive symptoms in patients on chronic haemodialysis. Cytokine. 2020;125:154823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 24. | Afsar B, Afsar RE. Brain-derived neurotrophic factor (BDNF): a multifaceted marker in chronic kidney disease. Clin Exp Nephrol. 2022;26:1149-1159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 25. | Zhou H, Qiu Y. Retrospective Study on the Value of Intensive Care Unit Noise Reduction Management in Elderly Patients with Chronic Renal Failure in the Uremic Phase. Noise Health. 2024;26:376-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 26. | Chou YH, Huang WL, Chang CH, Yang CCH, Kuo TBJ, Lin SL, Chiang WC, Chu TS. Heart rate variability as a predictor of rapid renal function deterioration in chronic kidney disease patients. Nephrology (Carlton). 2019;24:806-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 27. | Chang YM, Huang YT, Chen IL, Yang CL, Leu SC, Su HL, Kao JL, Tsai SC, Jhen RN, Shiao CC. Heart rate variability as an independent predictor for 8-year mortality among chronic hemodialysis patients. Sci Rep. 2020;10:881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 28. | da Estrela C, McGrath J, Booij L, Gouin JP. Heart Rate Variability, Sleep Quality, and Depression in the Context of Chronic Stress. Ann Behav Med. 2021;55:155-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 29. | Jankowska-Polańska B, Duczak A, Świątoniowska N, Karniej P, Seń M, Rosińczuk J. The influence of selected psychological variables on quality of life of chronically dialysed patients. Scand J Caring Sci. 2019;33:840-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 30. | Griva K, Chia JMX, Goh ZZS, Wong YP, Loei J, Thach TQ, Chua WB, Khan BA. Effectiveness of a brief positive skills intervention to improve psychological adjustment in patients with end-stage kidney disease newly initiated on haemodialysis: protocol for a randomised controlled trial (HED-Start). BMJ Open. 2021;11:e053588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 31. | Zhang L, Zou L, Zhou L. Effectiveness of psychoeducational interventions on psychological distress and health-related quality of life among patients with maintenance hemodialysis: a systematic review and meta-analysis. Ren Fail. 2024;46:2331613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 32. | Weng Y, Hu L, Zhuang S, Shi Y, Xu C, Liu C. Effects of High-flux Hemodialysis With Narrative Care on Clinical Efficacy and Prognostic Quality of Life of Patients With Chronic Renal Failure. Altern Ther Health Med. 2023;29:164-169. [PubMed] |
| 33. | Gregg LP, Hedayati SS. Pharmacologic and psychological interventions for depression treatment in patients with kidney disease. Curr Opin Nephrol Hypertens. 2020;29:457-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 34. | Chahal JS, Saini S, Bansal P, Arora J, Bansal PD, Saini B. Impact of Psychosocial Interventions on Depression in Chronic Kidney Disease: A Systematic Review and Meta-Analysis. Int J Psychiatry Med. 2025;912174251326009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 35. | González-Flores CJ, Garcia-Garcia G, Lerma C, Guzmán-Saldaña RME, Lerma A. Effect of Cognitive Behavioral Intervention Combined with the Resilience Model to Decrease Depression and Anxiety Symptoms and Increase the Quality of Life in ESRD Patients Treated with Hemodialysis. Int J Environ Res Public Health. 2023;20:5981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 36. | Wu YY, Gao YY, Wang JQ, Zhang C, Xu PJ, Liu J, Yu RZ, Zhang HJ. The influence of mindfulness meditation combined with progressive muscle relaxation training on the clinical efficacy and quality of life of patients with sarcopenia receiving haemodialysis: a randomised controlled trial. BMC Complement Med Ther. 2024;24:194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 37. | Wu Y, Zhang H, Jiang L, Liu Z, Li X, Guo B, Li J, Xu P, Liu J, Yu R. Effects of mindfulness meditation combined with progressive muscle relaxation on sleep disorders, anxiety, and depression in patients with sarcopenia undergoing hemodialysis. Front Psychiatry. 2025;16:1542028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 38. | Sohn BK, Oh YK, Choi JS, Song J, Lim A, Lee JP, An JN, Choi HJ, Hwang JY, Jung HY, Lee JY, Lim CS. Effectiveness of group cognitive behavioral therapy with mindfulness in end-stage renal disease hemodialysis patients. Kidney Res Clin Pract. 2018;37:77-84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 39. | Yang H, Qi L, Pei D. Effect of psychosocial interventions for depression in adults with chronic kidney disease: a systematic review and meta-analysis. BMC Nephrol. 2024;25:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 40. | Hagemann PMS, Martin LC, Neme CMB. The effect of music therapy on hemodialysis patients' quality of life and depression symptoms. J Bras Nefrol. 2019;41:74-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 41. | Debnath S, Rueda R, Bansal S, Kasinath BS, Sharma K, Lorenzo C. Fatigue characteristics on dialysis and non-dialysis days in patients with chronic kidney failure on maintenance hemodialysis. BMC Nephrol. 2021;22:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 42. | Bohm C, Schick-Makaroff K, MacRae JM, Tan M, Thompson S. The role of exercise in improving patient-reported outcomes in individuals on dialysis: A scoping review. Semin Dial. 2019;32:336-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 43. | Hargrove N, El Tobgy N, Zhou O, Pinder M, Plant B, Askin N, Bieber L, Collister D, Whitlock R, Tangri N, Bohm C. Effect of Aerobic Exercise on Dialysis-Related Symptoms in Individuals Undergoing Maintenance Hemodialysis: A Systematic Review and Meta-Analysis of Clinical Trials. Clin J Am Soc Nephrol. 2021;16:560-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 44. | Zelko A, Skoumalova I, Kolarcik P, Rosenberger J, Rabajdova M, Marekova M, Geckova AM, van Dijk JP, Reijneveld SA; NEPHRO-team. The effects of intradialytic resistance training on muscle strength, psychological well-being, clinical outcomes and circulatory micro-ribonucleic acid profiles in haemodialysis patients: Protocol for a quasi-experimental study. Medicine (Baltimore). 2019;98:e15570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 45. | Kanbay M, Copur S, Yildiz AB, Tanriover C, Mallamaci F, Zoccali C. Physical exercise in kidney disease: A commonly undervalued treatment modality. Eur J Clin Invest. 2024;54:e14105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 46. | Shimoda T, Matsuzawa R, Yoneki K, Harada M, Watanabe T, Matsumoto M, Yoshida A, Takeuchi Y, Matsunaga A. Changes in physical activity and risk of all-cause mortality in patients on maintence hemodialysis: a retrospective cohort study. BMC Nephrol. 2017;18:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 47. | Inaguma D, Tanaka A, Shinjo H. Physical function at the time of dialysis initiation is associated with subsequent mortality. Clin Exp Nephrol. 2017;21:425-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 48. | Frih B, Mkacher W, Bouzguenda A, Jaafar H, ALkandari SA, Ben Salah Z, Sas B, Hammami M, Frih A. Effects of listening to Holy Qur'an recitation and physical training on dialysis efficacy, functional capacity, and psychosocial outcomes in elderly patients undergoing haemodialysis. Libyan J Med. 2017;12:1372032. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 49. | Lin CH, Hsu YJ, Hsu PH, Lee YL, Lin CH, Lee MS, Chiang SL. Effects of Intradialytic Exercise on Dialytic Parameters, Health-Related Quality of Life, and Depression Status in Hemodialysis Patients: A Randomized Controlled Trial. Int J Environ Res Public Health. 2021;18:9205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 50. | Piva G, Storari A, Battaglia Y, Manfredini F, Lamberti N. Exercise, Dialysis, and Environment: A Narrative Review in an Ecological Perspective. Kidney Blood Press Res. 2024;49:773-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 51. | Li T, Soh KL, Zakaria NF, Pang Y, Wang P, Hu N. Effects of Laughter Yoga on Patients Receiving Hemodialysis: A Systematic Review. Holist Nurs Pract. 2024;38:202-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 52. | Pardeller S, Kemmler G, Hoertnagl CM, Hofer A. Associations between resilience and quality of life in patients experiencing a depressive episode. Psychiatry Res. 2020;292:113353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 53. | Al-Hakeim HK, Twayej AJ, Al-Dujaili AH. Reduction in serum IL-1β, IL-6, and IL-18 levels and Beck Depression Inventory-II score by combined sertraline and ketoprofen administration in major depressive disorder: A clinical trial. Neurol Psychiatry Brain Res. 2018;30:148-153. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 54. | Sutcigil L, Oktenli C, Musabak U, Bozkurt A, Cansever A, Uzun O, Sanisoglu SY, Yesilova Z, Ozmenler N, Ozsahin A, Sengul A. Pro- and anti-inflammatory cytokine balance in major depression: effect of sertraline therapy. Clin Dev Immunol. 2007;2007:76396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 186] [Cited by in RCA: 244] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 55. | Taraz M, Khatami MR, Dashti-Khavidaki S, Akhonzadeh S, Noorbala AA, Ghaeli P, Taraz S. Sertraline decreases serum level of interleukin-6 (IL-6) in hemodialysis patients with depression: results of a randomized double-blind, placebo-controlled clinical trial. Int Immunopharmacol. 2013;17:917-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 56. | King-Wing Ma T, Kam-Tao Li P. Depression in dialysis patients. Nephrology (Carlton). 2016;21:639-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 112] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 57. | Vangala C, Niu J, Montez-Rath ME, Yan J, Navaneethan SD, Winkelmayer WC. Selective Serotonin Reuptake Inhibitor Use and Hip Fracture Risk Among Patients on Hemodialysis. Am J Kidney Dis. 2020;75:351-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 58. | Assimon MM, Brookhart MA, Flythe JE. Comparative Cardiac Safety of Selective Serotonin Reuptake Inhibitors among Individuals Receiving Maintenance Hemodialysis. J Am Soc Nephrol. 2019;30:611-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 59. | Zhu N, Xu H, Lagerberg T, Johnell K, Carrero JJ, Chang Z. Comparative Safety of Antidepressants in Adults with CKD. Clin J Am Soc Nephrol. 2024;19:178-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 60. | Chen JW, Xie SQ. Agomelatine versus paroxetine in treating depressive and anxiety symptoms in patients with chronic kidney disease. Neuropsychiatr Dis Treat. 2018;14:547-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 61. | Mi WF, Tabarak S, Wang L, Zhang SZ, Lin X, Du LT, Liu Z, Bao YP, Gao XJ, Zhang WH, Wang XQ, Fan TT, Li LZ, Hao XN, Fu Y, Shi Y, Guo LH, Sun HQ, Liu L, Si TM, Zhang HY, Lu L, Li SX. Effects of agomelatine and mirtazapine on sleep disturbances in major depressive disorder: evidence from polysomnographic and resting-state functional connectivity analyses. Sleep. 2020;43:zsaa092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 62. | Asghar MS, Ahsan MN, Jawed R, Rasheed U, Ali Naqvi SA, Hassan M, Yaseen R, Mallick N, Zehra M, Saleem M. A Comparative Study on the Use of Alprazolam and Melatonin for Sleep Disturbances in Hemodialysis Patients. Cureus. 2020;12:e11754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 63. | Nalankilli K, Stallworthy E, Ducharlet K, Hole BD, O'Hara DV, Agarwal N, Snead CM, Caskey FJ, Smyth B. Treatment of insomnia, restless legs, cramps, and pain associated with chronic kidney disease: results from a multinational survey of kidney supportive care practice. BMC Nephrol. 2025;26:204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 64. | Rehman IU, Ahmed R, Rahman AU, Wu DBC, Munib S, Shah Y, Khan NA, Rehman AU, Lee LH, Chan KG, Khan TM. Effectiveness and safety profiling of zolpidem and acupressure in CKD associated pruritus: An interventional study. Medicine (Baltimore). 2021;100:e25995. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/