Copyright: ©Author(s) 2026.
World J Exp Med. Mar 20, 2026; 16(1): 115478
Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.115478
Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.115478
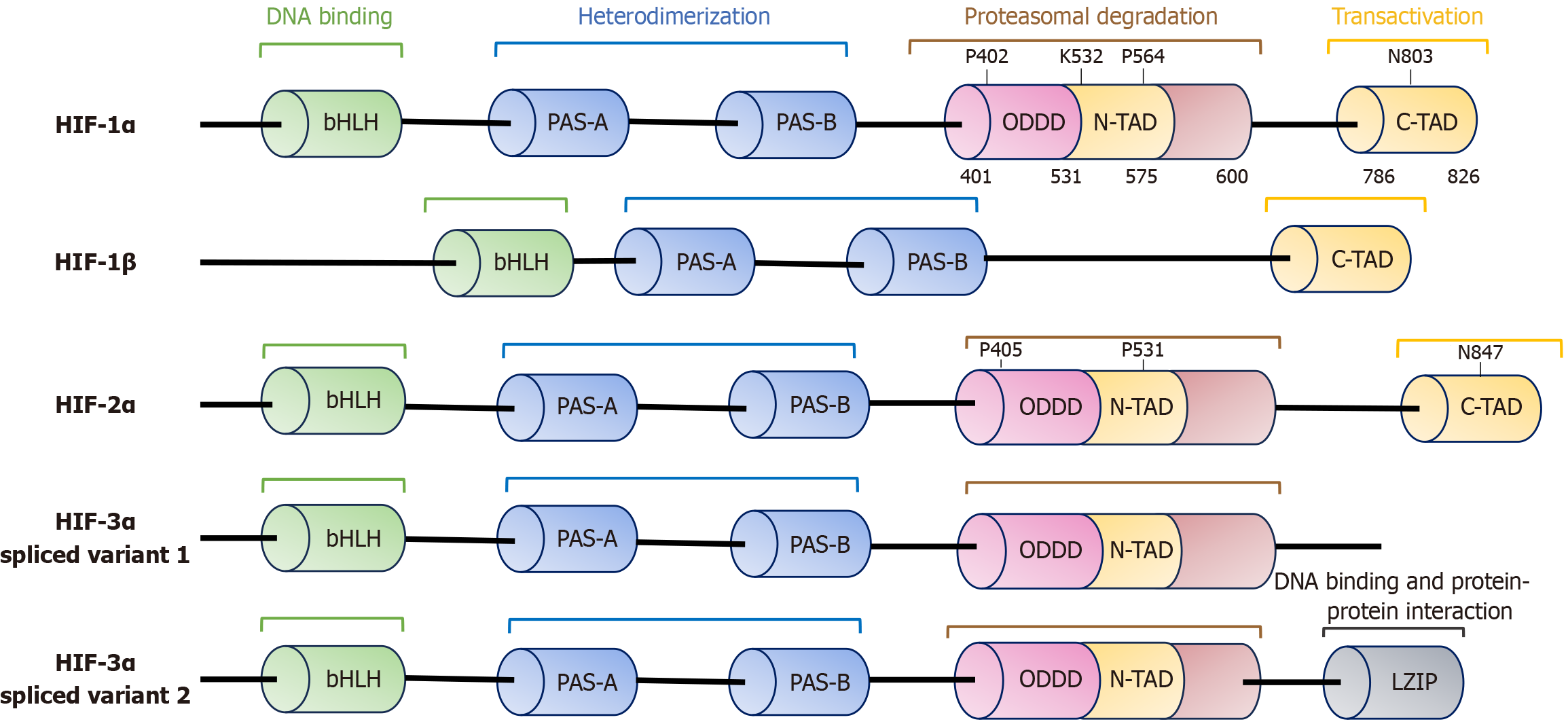
Figure 1 Schematic representation of the functional domains and potential roles of hypoxia inducible factor isoforms: Each column represents a distinct domain, with hydroxylation sites indicated above.
All hypoxia inducible factor (HIF) isoforms belong to the bHLH-PAS family containing a bHLH and two PAS domains (PAS-A and PAS-B) essential for HIF-1α and HIF-1β heterodimerization. Unlike HIF-1β, HIF-1α subunits possess an oxygen-dependent degradation domain (ODDD) that mediates proline hydroxylation and lysine acetylation, leading to proteasomal degradation. The ODDD contains an N-terminal activation domain and a C-terminal transactivation domain that regulate transcriptional activity. Conserved proline residues are present in HIF-1α and HIF-2α. Several HIF-3α splice variants exist, including variant 1, which lacks the C-terminal transcriptional activation domain, and variant 2, which contains a leucine zipper domain involved in DNA binding and protein interactions. HIF: Hypoxia inducible factor; N-TAD: N-terminal activation domain; C-TAD: C-terminal transcriptional activation domain; ODDD: Oxygen-dependent degradation domain; LZIP: Leucine zipper.
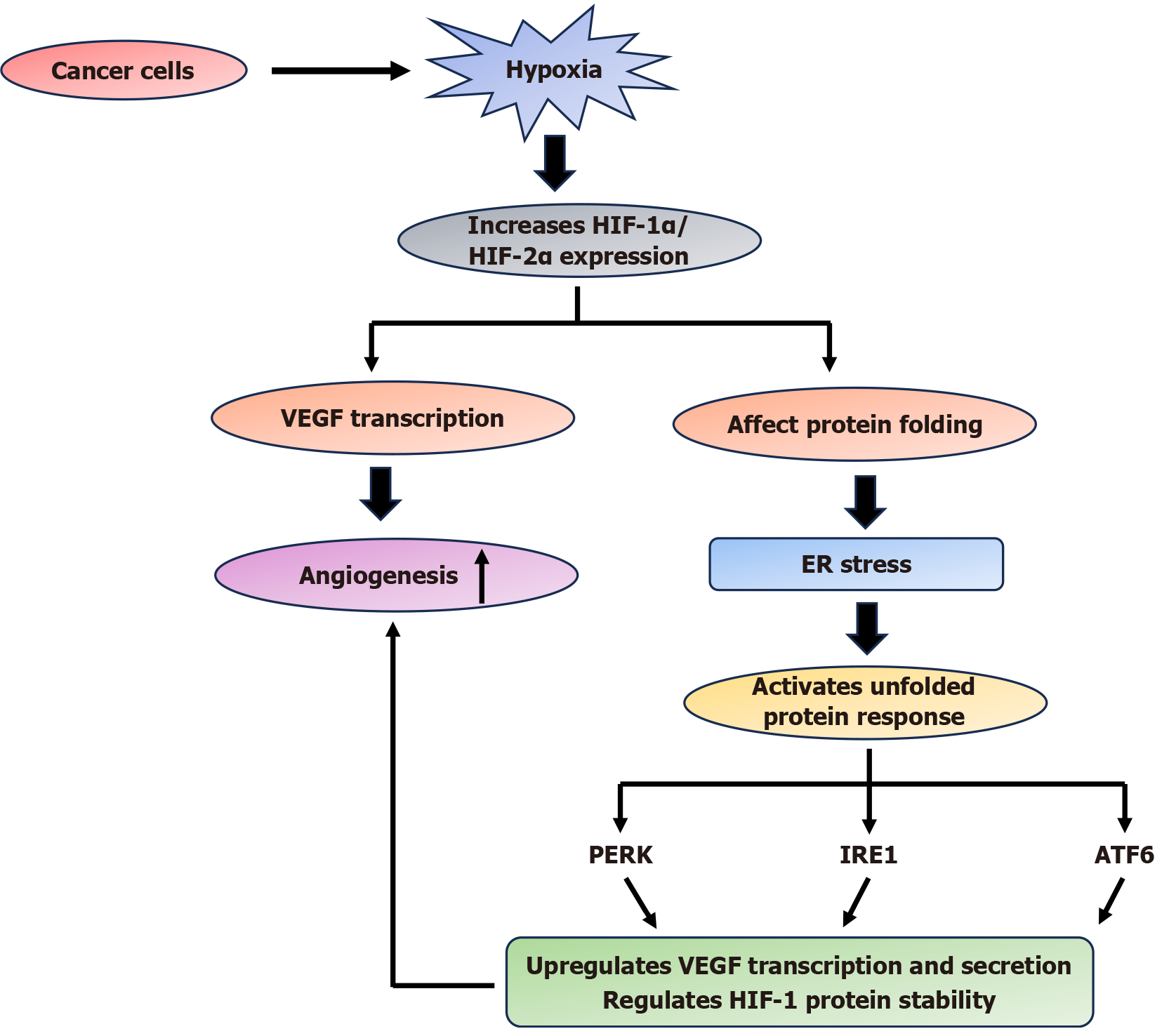
Figure 2 Diagram showing the interplay between hypoxia, endoplasmic reticulum stress, and angiogenesis in cancer progression: Under hypoxic conditions, cancer cells upregulate the expression of hypoxia inducible factor-1α and hypoxia inducible factor-2α, which in turn enhances vascular endothelial growth factor transcription, promoting angiogenesis.
Simultaneously, hypoxia disrupts protein folding within the endoplasmic reticulum (ER), leading to the ER stress and activation of the unfolded protein response through its three primary signaling branches: PERK, IRE1, and ATF6 The PERK and IRE1 branches further contribute to vascular endothelial growth factor transcription and secretion, helping to regulate HIF-1 protein stability and thereby creating a feedback loop that supports tumor vascularization and survival under low-oxygen conditions. HIF: Hypoxia inducible factor; VEGF: Vascular endothelial growth factor; ER: Endoplasmic reticulum.
- Citation: Bhardwaj S, Pandey S, Ghosh DK, Sharma T, Jain BP. Deciphering the interplay between hypoxia, angiogenesis, and endoplasmic reticulum stress in carcinogenesis: A narrative review. World J Exp Med 2026; 16(1): 115478
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/115478.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.115478