Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.115208
Revised: November 4, 2025
Accepted: January 5, 2026
Published online: March 20, 2026
Processing time: 155 Days and 15.7 Hours
In addition to their primary lipid-lowering effects, statins also exhibit pleiotropic properties, including anti-inflammatory, immunomodulatory, antimicrobial, antioxidative, and angiogenic effects, all of which promote wound healing. Ro
To investigate the effects of rosuvastatin and its mechanism of action on burn wound healing process in an experimental study.
Ninety male Wistar albino rats aged 12-16 weeks were randomly assigned to three groups of 30, subjected to burn using specific stainless steel sealer. Burn eschar was removed the following day applying topical rosuvastatin cream (study), Eucerin cream (placebo), normal saline (control), and sterile wound dressing. Each group was divided into three subgroups of ten according to sacrifice day (3rd, 6th, 9th). C-reactive protein (CRP), tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, IL-6, digital assessment of burn healing, and histopathology were performed.
Using the value on the third day in the control group as the baseline, reductions were measured on the sixth and ninth days. Similarly, reduction values were recorded on the third, sixth, and ninth days in the study group. A statistically significant reduction was observed in the study group compared to the control group, with greater reductions corresponding to later sacrifice days (3rd, 6th, and 9th) in CRP (P < 0.01), TNF-α (P < 0.01), IL-1β (P < 0.01), and IL-6 (P < 0.01) levels and burn size (P < 0.01). Histopathology revealed a statistically significant reduction in inflammatory infiltration, coagulative necrosis, and microhemorrhage (P < 0.01). Conversely, statistically significant increases in neovascularization (P = 0.012) and fibroblastic reactions (P = 0.023) were noted. No adverse effects or deaths were observed.
Rosuvastatin reduces inflammation by lowering TNF-α, IL-1β, IL-6, CRP while increasing neo-angiogenesis, fibroblast reactions and microenvironmental protection in burn wounds. These effects promote repair and positively impact burn wound healing.
Core Tip: Overall, the data from this original preclinical study confirm that rosuvastatin does not act unidimensionally but simultaneously influences the inflammatory, angiogenic, and reparative phases of burn wound healing. These findings are consistent with data from other statins and expand our understanding of the mechanisms of action of rosuvastatin, hi
- Citation: Marneri AG, Pavlidis ET, Stavrati KE, Kofinas A, Mouratidou C, Gkoutziotis I, Kotoulas SC, Marouda C, Psalla D, Ballas KD, Pavlidis TE. Experimental beneficial effect of rosuvastatin on burn wound healing in a rat model. World J Exp Med 2026; 16(1): 115208
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/115208.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.115208
In addition to their primary role in lipid reduction, statins have pleiotropic effects, including anti-inflammatory, im
Burn injuries pose a major worldwide public health challenge and can result in serious complications. The link between the delayed healing of burn wounds and higher mortality rates has motivated many researchers to develop new treatments focused on accelerating burn wound recovery[8,10]. The healing process of wounds, particularly burns, is a complex, multistage biological process involving hemostasis, inflammation, cell proliferation, extracellular matrix formation, new blood vessel growth, and tissue remodeling[11-14].
Statins not only reduce cholesterol levels but also promote wound healing[15-19]. Statins studied include lovastatin[20,21], simvastatin[22-26], pravastatin[27,28], fluvastatin[29], and atorvastatin[30-33], and more recently, rosuvastatin, a synthetic hydrophilic statin shown to be effective in wound healing[34-38] and bone healing[39,40]. Specifically, sim
Although rosuvastatin primarily targets the liver, there are concerns about its multiple effects in other tissues where its uptake is low. Nevertheless, various studies have demonstrated the pleiotropic effects of rosuvastatin, such as the in
Recently, stem cells[26,33,41] and platelet-rich plasma[10,35] combined with rosuvastatin have shown promising results in enhancing wound and bone healing[50]. Different statin delivery methods have been utilized to improve absorption and extend release duration[13,15,16,24,34], including the incorporation of rosuvastatin into biological sca
This preclinical study aimed to examine the effects of rosuvastatin and elucidate its underlying mechanism of action on the burn wound healing process using an experimental rat model. Simvastatin and, to a lesser extent, atorvastatin are perhaps the only statins for which the effects on burn wound healing have been investigated. These studies have yielded some positive effects and encouraging results.
To date, the effect of rosuvastatin on partial-thickness burn wounds has not been studied. This research is of great interest because rosuvastatin, as the newest generation of statins, is characterized by fewer side effects and increased efficacy. Therefore, this study is original because it is the first to investigate the effect of rosuvastatin in an experimental model of partial-thickness skin burn wounds in rats.
The animals were handled according to the science-based guidelines for Laboratory Animal Care published by the National Research Council Institute for laboratory animal research in November 2003. The experimental protocol was approved by the Medical School of Aristotle University of Thessaloniki (approval No. 11525-12/05/2023) and the Department of Animal Health, Directorate of Veterinary Policy, Prefecture of Thessaloniki, Greece [approval No. 608344(2695)].
The experimental part of the study was conducted at the Laboratory of Development-Breeding of Animal Models and Biomedical Research, School of Veterinary Medicine, Aristotle University of Thessaloniki (registration number EL-54-BIOexp-10). Ninety male Wistar albino rats, aged 12-16 weeks and weighing approximately 350-400 g, were used in the study. The rats were individually housed in ventilated cages connected to a central ventilation circuit. The cages were maintained at a stable room temperature of 23 °C, with humidity levels between 50% and 60%, noise levels below 50 dB, and an artificial 12-hour light/dark cycle. The rats had ad libitum access to tap water and a standard chow diet. The experimental protocol was designed to minimize pain, discomfort, and distress in the animals. The sample size was calculated using G*Power software on the basis of sample sizes from earlier studies with comparable experimental designs and methods.
The rats were randomly assigned to three main groups (the study group, placebo group and control group) of 30 rats each according to the use or absence of rosuvastatin cream. The animals in all the groups were subjected to burn-causing procedures. These main groups were divided into three subgroups of 10 rats each according to the day of sacrifice (3 days, 6 days, or 9 days after surgery). This study focused on the healing effects of the substance on the 3rd, 6th, and 9th days following the burn. After the burn was induced, the animals were divided on specific days to observe the wound healing process. In the study group, the burn surface was treated with rosuvastatin cream into the Eucerin base, whereas the placebo group received Eucerin cream alone. In the control group, the wounds were washed with normal saline (NaCl 0.9%).
For the study group, the formulation was prepared by incorporating twenty tablets of 20 mg rosuvastatin (Rosuvastatin/Actavis F.C., Reykjavik, Iceland) into 20 g of Eucerin cream base (Beiersdorf AG, Hamburg, Germany), followed by thorough mixing and homogenization to ensure uniform distribution. In contrast, the placebo formulation consisted exclusively of 20 g of the Eucerin cream base without the addition of any active pharmaceutical ingredient.
All the rats were anesthetized with intraperitoneal injections of ketamine hydrochloride (Ketamin-Actavis, Institute of Pharmaceutical Research and Technology, Athens, Greece) and dexmedetomidine (Dexmedetomidine, Ever Valinject, Oberburgau, Austria) at doses of 75 mg/kg and 0.5 mg/kg, respectively. Analgesia was provided with an intramuscular injection of tramadol (Tramal, Vianex, Athens, Greece) at a dosage of 25 mg/kg.
The animals were immobilized and weighed, and their dorsums were shaved on a surface of at least 5 cm2. The skin was then prepared with 10% povidone iodine. To induce the burn, a specialized metal stamp measuring 17 mm in diameter and 2.5 mm in thickness and weighing 200 g was utilized. These dimensions facilitate efficient heat transfer. The stamp is made of stainless steel and features a wooden handle. It was heated to 85 °C and then applied to the skin on the back for 10 seconds to produce a deep partial-thickness burn. The next step involved washing the wound with normal saline, taking photographs of the burn wounds, and documenting the burn surface. Afterward, the wounds were covered with sterile dressings. All surgical procedures were conducted by the same surgeon on a warm, stable dissection table in a sterile environment using sterile surgical instruments. On the first day after the burn, the eschar was removed, and topical treatments were applied. Each day, 200 μL of a mixture of Eucerin cream with rosuvastatin, Eucerin cream, or normal saline was applied to the burn wounds on the basis of the group assignment of each rat. The wounds were then covered with sterile dressings.
Deep tissue excision at the burn site was performed on the scheduled day (3 days, 6 days, or 9 days after surgery) under the same anesthesia as the initial surgery. Blood and burn tissue samples were collected for biochemical and histopathological examinations, and the burn size was photographed for assessment. The rats were then euthanized. A portion of each blood sample was placed in a tube with ethylenediaminetetraacetic acid and centrifuged at 5000 rpm for 15 minutes at +4 °C. The serum samples were then transferred to Eppendorf tubes, immediately frozen in liquid nitrogen, and stored at -80 °C until biochemical analysis.
The serum levels of oxidative stress markers and inflammatory response C-reactive protein (CRP) [rat CRP enzyme-linked immunosorbent assay (ELISA) kit, BD Biosciences, San Diego, CA, United States], tumor necrosis factor-alpha (TNF-α) (rat TNF alpha ELISA kit, Antibodies.com Europe AB, Stockholm, Sweden), interleukin (IL)-1β (rat IL-1β ELISA kit, Antibodies.com Europe AB, Stockholm, Sweden), and IL-6 (rat IL-6 ELISA kit, Antibodies.com Europe AB, Stockholm, Sweden) were assessed using ELISA.
The size of the wound was assessed using digital photography. The burn surface area was measured using ImageJ software, version 1.45 (National Institutes of Health, United States). The burn areas captured in the photographs were utilized to calculate the wound healing rate using the following formula based on the planimetric method: Wound healing rate = (burn surface area on day x/burn surface area on day 0) × 100.
The burn tissue samples were fixed in 10% neutral buffered formalin and embedded in paraffin blocks. Histological sections 2-3 μm in thickness were obtained and then subjected to hematoxylin-eosin staining. The histopathology assessment was performed using a light microscope (Nikon eclipse 50i, Nikon Corporation, Tokio, Japan) and a digital camera (Nikon DS-5 ML1, Nikon Corporation, Tokio, Japan) at 200 × magnification. Inflammatory infiltration, neovascularization and fibroblast activity were assessed using scores ranging from 0 to 3 (0 = absent, 1 = moderate, 2 = severe).
Linear regression analyses were conducted to examine the effects of treatment (rosuvastatin, Eucerin cream or saline) and time (sacrifice days 3, 6, and 9) on the levels of the four inflammation markers (CRP, TNF, IL-1β, and IL-6) as well as on burn size. For each dependent variable, an ordinary least squares linear regression model was employed, using dummy variables to categorize both treatment and sacrifice day. Specifically, the following dummy variables were employed: (1) x1 = 1, if rosuvastatin cream is administered; otherwise, x1 = 0; (2) x2 = 1, if Eucerin cream is administered; otherwise, x2 = 0; (3) The reference group for the treatment was the administration of normal saline. x3 = 1 on sacrifice day 6; otherwise, x3 = 0; and (4) x4 = 1 on sacrifice day 9; otherwise, x4 = 0. The reference group for time is sacrifice day 3. Therefore, the complete reference group in the models consisted of the experimental animals that received normal saline and were sacrificed on the third day (x1 = x2 = x3 = x4 = 0).
In cases where statistically significant interactions between the independent variables were identified, the corresponding interaction terms were included in the final model. Conversely, when the interaction terms were not statistically significant or resulted in a poorer fit on the basis of the Akaike information criterion (AIC) and Bayesian information criterion (BIC) statistical criteria, an additive model without interactions was used.
For each model, the regression equation is presented along with the estimated coefficients, the values of the coefficient of determination (R2 and adjusted R2), the F test statistic for the overall significance of the model, and the corresponding P values. The interpretation of the results is based on calculating the predicted mean responses for each experimental condition relative to the reference group.
For the variables of inflammatory infiltration, neovascularization, and fibroblastic reaction - each recorded on an ordered categorical scale (0 = absent, 1 = moderate, 2 = severe) - multinomial ordinal logistic regression was applied. The group (control as the reference, placebo, study), the day of sacrifice (day 3 as the reference), and the interaction terms between group and day were included as independent variables. All statistical analyses were conducted using RStudio statistical software (Boston, MA, United States). Statistical significance was set at P < 0.05. A biomedical statistician performed the statistical review of the study.
The linear model for CRP does not include interaction terms because these terms were not statistically significant, and the model fit improved without them, as indicated by lower AIC and BIC values. The model equation is CRP = 21.0028 - 6.0037x1 - 2.4567x2 - 4.5233x3 - 9.5600x4. The R2 of the model above is 0.92, indicating that 92% of the variance in the CRP is explained by the independent variables. The adjusted R2 is 0.9163, and the overall model fit is statistically significant [F (4, 85) = 244.5, P < 0.001].
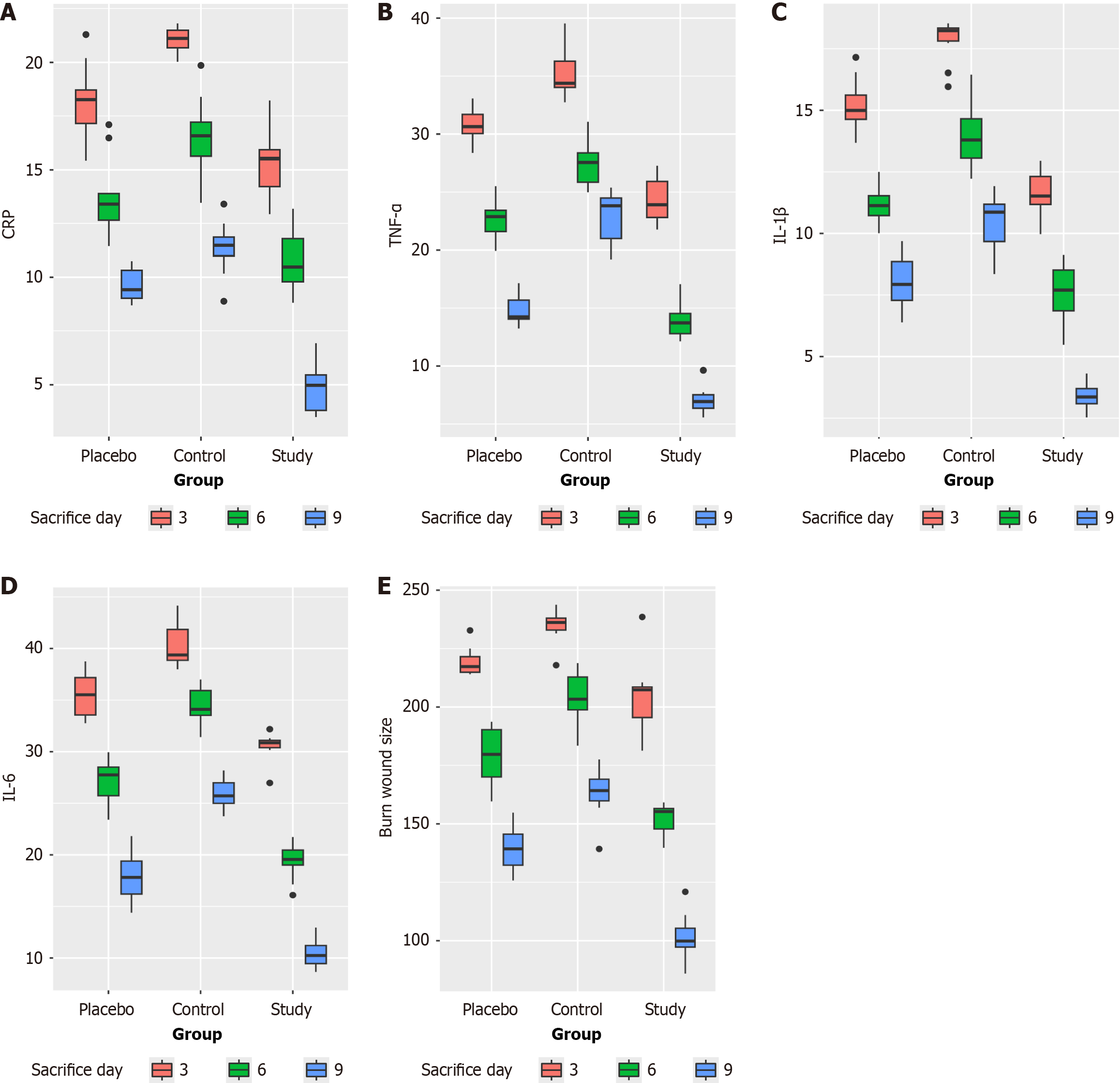
In more detail, the expected CRP concentration for the reference group (normal saline, day 3) was 21.0028 units (P < 0.001). On the same day, the administration of rosuvastatin reduced the CRP concentration by 5.75 units (P < 0.001). Correspondingly, the administration of Eucerin cream reduced the CRP concentration by 2.4567 units (P < 0.001). When normal saline treatment was continued, sacrifice on day 6 was associated with a reduction in CRP levels of 4.8700 units (P < 0.001), whereas sacrifice on day 9 was associated with a reduction of 9.6900 units (P < 0.001). Overall, the results indicate that both rosuvastatin administration and the timing of sacrifice are associated with a statistically significant reduction in CRP levels, with the greatest effect observed when rosuvastatin is administered and when sacrifice occurs later. Changes in CRP levels according to the rat group and day of sacrifice are depicted graphically in Figure 1A, with corresponding descriptive statistics presented in Table 1.
| Group | Day | mean | Range | Median | IQR | SD |
| Group placebo | 3 | 18.22 | 5.85 | 18.26 | 1.56 | 3.85 |
| Group control | 3 | 21.04 | 1.77 | 21.10 | 0.82 | 4.22 |
| Group study | 3 | 15.29 | 5.29 | 15.52 | 1.70 | 4.52 |
| Group placebo | 6 | 13.71 | 5.65 | 13.40 | 1.24 | 3.85 |
| Group control | 6 | 16.53 | 6.40 | 16.57 | 1.59 | 4.22 |
| Group study | 6 | 10.74 | 4.37 | 10.48 | 2.00 | 4.52 |
| Group placebo | 9 | 9.63 | 2.06 | 9.42 | 1.30 | 3.85 |
| Group control | 9 | 11.35 | 4.52 | 11.48 | 0.87 | 4.22 |
| Group study | 9 | 4.89 | 3.43 | 4.98 | 1.66 | 4.52 |
The linear model for TNF-α includes both main effects and interaction terms because statistically significant interactions between the independent variables were identified. The model equation is as follows: TNF-α = 35.288 - 11.020x1 - 4.587x2 - 7.832x3 - 12.513x4 - 2.398x1x3 - 4.679x1x4 - 0.252x2x3 - 3.405x2x4. The R2 of the model is 0.9609, indicating that 96.09% of the variation in TNF-α is explained by the model. The adjusted R2 is 0.957, and the overall fit of the model is statistically significant [F (8, 81) = 248.7, P < 0.001].
In more detail, For the reference group (normal saline, day 3), the expected TNF-α concentration was 35.2880 pg/mL (P < 0.001). On the same day, the administration of rosuvastatin reduced the TNF-α concentration by 11.0200 pg/mL (P < 0.001), whereas the administration of the Eucerin cream resulted in a reduction of 4.5870 pg/mL (P < 0.001). Compared with normal saline treatment, sacrifice on day 6 resulted in a reduction of 7.8320 pg/mL (P < 0.001), whereas sacrifice on day 9 resulted in a reduction of 12.5130 pg/mL (P < 0.001). Owing to interactions, the changes in the combinations of treatment and sacrifice day are more complex. Regarding rosuvastatin on sacrifice day 6, the expected TNF-α concentration decreased by 11.0200 + 7.8320 + 2.3980 = 21.2500 pg/mL (P = 0.036). Regarding rosuvastatin on sacrifice day 9, the total decrease was 11.0200 + 12.5130 + 4.6790 = 28.2120 pg/mL (P < 0.001). Regarding Eucerin cream on sacrifice day 9, the decrease was 4.5870 + 12.5130 + 3.4050 = 20.5050 pg/mL (P = 0.0032). Overall, these results demonstrate that both treatment and time significantly affect TNF-α levels, with rosuvastatin producing the greatest reduction, especially as the day of sacrifice progresses. Changes in TNF-α levels by the rat group and the day of sacrifice are depicted graphically in Figure 1B, with corresponding descriptive statistics presented in Table 2.
| Group | Day | mean | Range | Median | IQR | SD |
| Group placebo | 3 | 30.70 | 4.69 | 30.64 | 1.65 | 6.76 |
| Group control | 3 | 35.29 | 6.80 | 34.39 | 2.26 | 5.65 |
| Group study | 3 | 24.27 | 5.50 | 23.91 | 3.10 | 7.34 |
| Group placebo | 6 | 22.62 | 5.58 | 22.89 | 1.83 | 6.76 |
| Group control | 6 | 27.46 | 6.07 | 27.55 | 2.52 | 5.65 |
| Group study | 6 | 14.04 | 4.93 | 13.72 | 1.74 | 7.34 |
| Group placebo | 9 | 14.78 | 3.91 | 14.24 | 1.63 | 6.76 |
| Group control | 9 | 22.78 | 6.21 | 23.82 | 3.49 | 5.65 |
| Group study | 9 | 7.08 | 4.06 | 6.94 | 1.16 | 7.34 |
The linear model for IL-1β does not include interaction terms because these terms were not statistically significant, and the model demonstrated a better fit without them, as indicated by lower AIC and BIC values. The model equation is as follows: IL-1β = 17.955 - 6.573x1 - 2.623x2 - 3.986x3 - 7.55x4. The R2 of the model is 0.9452, indicating that 94.52% of the variation in IL-1β is explained by the model. The adjusted R2 is 0.9426, and the overall fit is statistically significant [F (4, 85) = 366.7, P < 0.001].
In detail: The expected value of IL-1β for the reference group (normal saline, day 3) was 17.9552 pg/mL (P < 0.001). On the same day, the administration of rosuvastatin reduced the IL-1β concentration by 6.1900 pg/mL (P < 0.001). Correspondingly, the administration of Eucerin cream reduced the IL-1β concentration by 2.6227 pg/mL (P < 0.001). When normal saline treatment was continued, sacrifice on day 6 resulted in a reduction of 3.7200 pg/mL (P < 0.001), whereas sacrifice on day 9 was associated with a reduction of 6.9300 pg/mL (P < 0.001). Overall, the results revealed that the administration of rosuvastatin and an advanced day of sacrifice were associated with a significant reduction in IL-1β levels, with no significant interaction between these factors. Changes in IL-1β levels by the rat group and the day of sacrifice are depicted graphically in Figure 1C.
The linear model for IL-6 includes both main effects and interaction terms, as statistically significant interactions between the independent variables were identified. The model equation is as follows: IL-6 = 40.241 - 9.712x1 - 4.677x2 - 5.755x3 - 14.371x4 - 5.418x1x3 - 5.760x1x4 - 2.728x2x3 - 3.461x2x4. The R2 of the model is 0.9633, indicating that 96.33% of the variation in IL-6 is explained by the model. The adjusted R2 is 0.9597, and the overall fit of the model is statistically significant [F (8, 81) = 266.1, P < 0.001].
In detail: The expected value of IL-6 for the reference group (normal saline, day 3) is 40.2410 pg/mL (P < 0.001). On the same day, the administration of rosuvastatin reduced the IL-6 concentration by 9.7120 pg/mL (P < 0.001), whereas the administration of Eucerin cream reduced the IL-6 concentration by 4.6770 pg/mL (P < 0.001). In the normal saline group, the mean IL-6 concentration decreased by 5.7550 pg/mL on day 6 (P < 0.001) and by 14.3710 units on day 9 (P < 0.001).
When interactions are taken into account: For rosuvastatin on day 6, the total reduction was 9.7120 + 5.7550 + 5.4180 = 20.8850 pg/mL (P < 0.001). For rosuvastatin on day 9, the total reduction was 9.7120 + 14.3710 + 5.7600 = 10.400 pg/mL (P < 0.001). For the Eucerin cream on day 6, the total reduction was 4.6770 + 5.7550 + 2.7280 = 13.1600 pg/mL (P = 0.023). For the Eucerin cream on day 9, the total reduction was 4.6770 + 14.3710 + 3.4610 = 22.5090 pg/mL (P = 0.004). Overall, the results revealed that rosuvastatin administration was associated with a significant reduction in IL-6 levels, which became more pronounced over time, especially when rosuvastatin was combined with sacrifice at later time points. Changes in the level of IL-6 by the rat group and the day of sacrifice are depicted graphically in Figure 1D, with corresponding descriptive statistics presented in Table 3.
| Group | Day | mean | Range | Median | IQR | SD |
| Group placebo | 3 | 35.56 | 6.00 | 35.52 | 3.62 | 7.72 |
| Group control | 3 | 40.24 | 6.17 | 39.37 | 2.99 | 6.25 |
| Group study | 3 | 30.53 | 5.21 | 30.87 | 0.71 | 8.50 |
| Group placebo | 6 | 27.08 | 6.54 | 27.75 | 2.77 | 7.72 |
| Group control | 6 | 34.49 | 5.58 | 34.11 | 2.40 | 6.25 |
| Group study | 6 | 19.36 | 5.63 | 19.56 | 1.45 | 8.50 |
| Group placebo | 9 | 17.73 | 7.43 | 17.82 | 3.19 | 7.72 |
| Group control | 9 | 25.87 | 4.44 | 25.72 | 1.99 | 6.25 |
| Group study | 9 | 10.40 | 4.30 | 10.25 | 1.75 | 8.50 |
The linear model for burn size includes both main effects and interaction terms, as these were found to be statistically significant. The model equation is as follows: Burn size = 235.171 - 30.526x1 - 15.858x2 - 31.476x3 - 71.617x4 - 20.995x1x3 - 31.699x1x4 - 8.548x2x3 - 8.538x2x4. The R2 of the model is 0.9438, indicating that 94.38% of the variance in burn size is explained by the model. The adjusted R2 is 0.9383, and the overall fit is statistically significant [F (8, 81) = 170.1, P < 0.001].
In detail: The expected burn size for the reference group (normal saline, day 3) is 235.171 (P < 0.001). On the same day, the use of rosuvastatin reduced the burn size by 30.526 units (P < 0.001), whereas the use of Eucerin cream reduced it by 15.858 units (P < 0.001). In the normal saline group, sacrifice on day 6 resulted in a reduction of 31.476 units (P < 0.001), whereas sacrifice on day 9 resulted in a reduction of 73.62 units (P < 0.001).
Combined with interaction terms: For rosuvastatin on day 6, the total reduction is 30.526 + 31.476 + 20.995 = 82.997 units (P = 0.0019). For rosuvastatin on day 9, the total reduction is 30.526 + 71.617 + 31.699 = 133.842 units (P < 0.001).
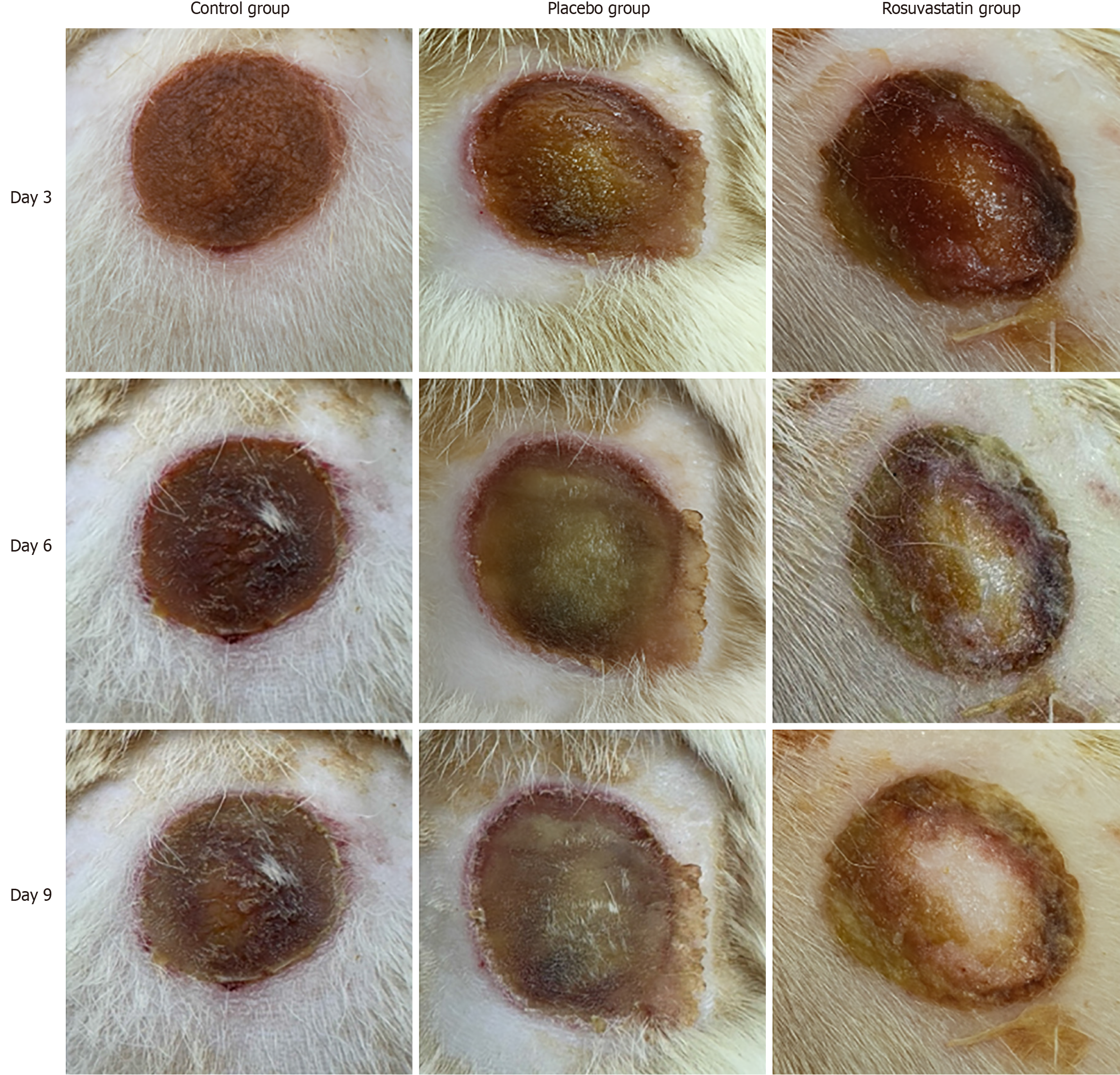
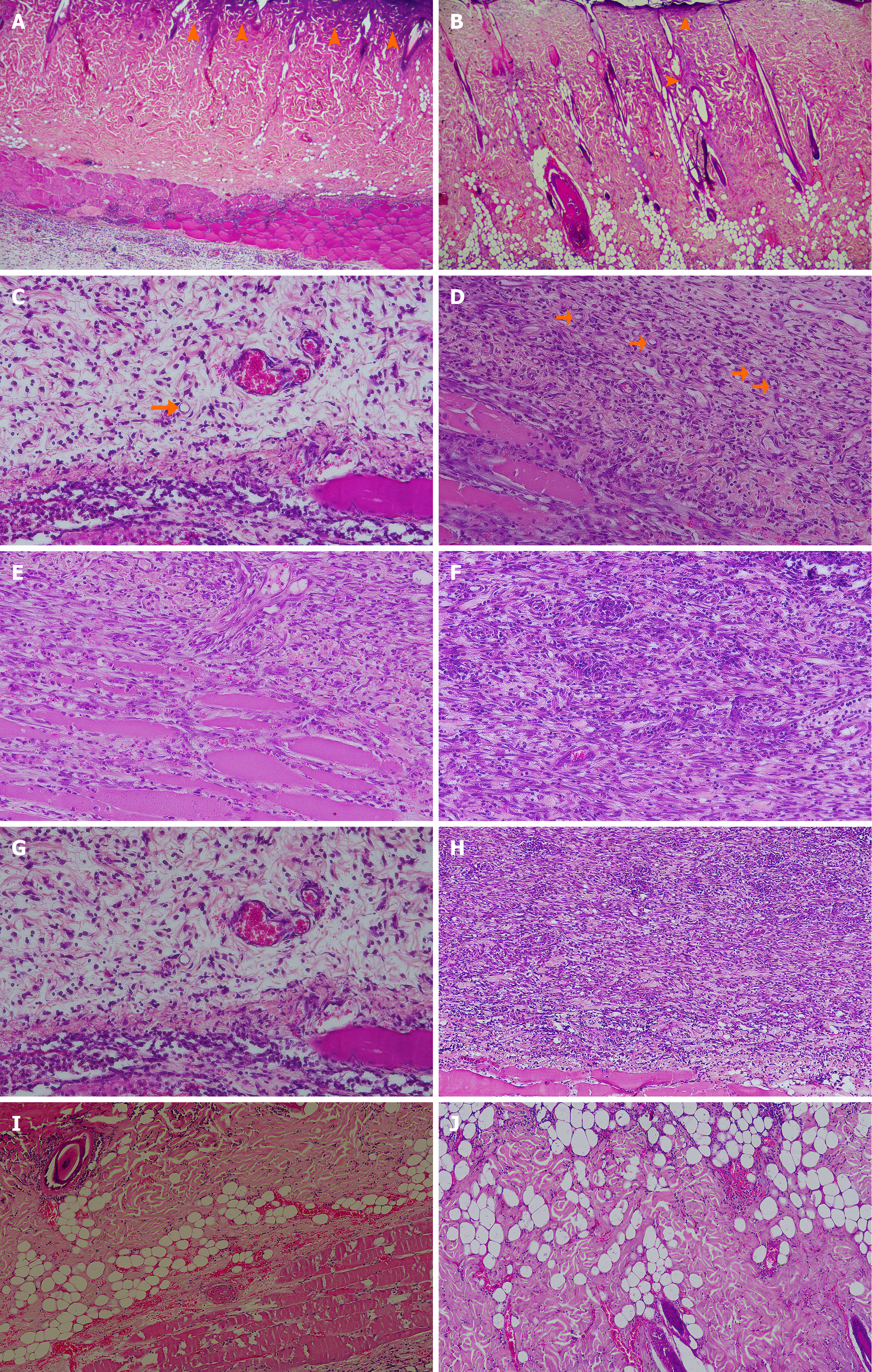
Overall, the results show that rosuvastatin administration and an advanced day of sacrifice are associated with a significant reduction in burn size, with a more pronounced effect noted when the two factors are combined. Changes in burn size by rat group and day of sacrifice are depicted graphically in Figure 1E, with corresponding descriptive statistics presented in Table 4. Representative macroscopic digital images of burn wound healing for the 9 study subgroups are displayed in Figure 2, whereas representative microscopic burn wound healing images are displayed in Figure 3.
| Group | Day | mean | Range | Median | IQR | SD |
| Group placebo | 3 | 219.31 | 18.77 | 217.29 | 6.66 | 34.53 |
| Group control | 3 | 235.17 | 25.88 | 236.17 | 5.08 | 31.40 |
| Group study | 3 | 204.65 | 57.20 | 207.37 | 13.06 | 44.26 |
| Group placebo | 6 | 179.29 | 34.08 | 179.73 | 20.22 | 34.53 |
| Group control | 6 | 203.70 | 35.32 | 203.36 | 14.04 | 31.40 |
| Group study | 6 | 152.17 | 19.42 | 155.25 | 8.73 | 44.26 |
| Group placebo | 9 | 139.16 | 29.02 | 139.29 | 13.26 | 34.53 |
| Group control | 9 | 163.55 | 38.28 | 164.22 | 9.25 | 31.40 |
| Group study | 9 | 101.33 | 34.99 | 99.85 | 8.05 | 44.26 |
For the inflammatory infiltration variable, which was recorded on an ordinal categorical scale (0 = absent, 1 = moderate, 2 = severe), ordinal logistic regression analysis was performed. The independent variables included group (control as the reference, placebo, study), day of sacrifice (day 3 as the reference), and the interaction term group × day. The regression analysis results of the inflammatory infiltration model coefficients are presented in Table 5.
| Predictor | Coefficient (β) | SE | t value | P value |
| Group study | -20.9163 | 0.3231 | -64.73 | < 0.001 |
| Group placebo | -0.4418 | 0.9449 | -0.47 | 0.640 |
| Day 6 | 1.7917 | 1.0206 | 1.76 | 0.079 |
| Day 9 | 1.2528 | 0.9449 | 1.33 | 0.185 |
| Group study × day 6 | -3.5835 | 1.6030 | -2.24 | 0.025 |
| Group study × day 9 | -21.1606 | Approximately 0.0000 | -3.94e + 08 | < 0.001 |
| Group placebo × day 6 | -0.9444 | 1.3849 | -0.68 | 0.495 |
| Group placebo × day 9 | 0.9808 | 1.4121 | 0.69 | 0.487 |
| Cut-point 0|1 | -20.5108 | 0.3224 | -63.62 | < 0.001 |
| Cut-point 1|2 | 0.4055 | 0.6455 | 0.63 | 0.530 |
Model equations: Transition from 0 to 1: Log [P (Y ≤ 0)/P (Y > 0)] = -20.5108 + 20.9163 × group study + 0.4418 × group placebo - 1.7917 × day 6 - 1.2528 × day 9 + 3.5835 × (group study × day 6) + 21.1606 × (group study × day 9) + 0.9444 × (group placebo × day 6) - 0.9808 × (group placebo × day 9). Transition from 1 to 2: Log [P (Y ≤ 1)/P (Y > 1)] = 0.4055 + 20.9163 × group study + 0.4418 × group placebo - 1.7917 × day 6 - 1.2528 × day 9 + 3.5835 × (group study × day 6) + 21.1606 × (group study × day 9) + 0.9444 × (group placebo × day 6) - 0.9808 × (group placebo × day 9).
Commentary on the results: Compared with the control group, the study group demonstrated a highly significant reduction in the likelihood of developing a higher grade of inflammatory infiltrate (β = -20.92; P < 0.001), indicating a strong protective effect of the therapeutic intervention. In contrast, the placebo group did not significantly differ from the control group (β = -0.44, P = 0.64). With respect to the observation period, days 6 and 9 had positive coefficients (β = 1.79 and β = 1.25, respectively), indicating a tendency toward an increase in inflammation; however, the result was not statistically significant (P = 0.079 and P = 0.185, respectively).
The interaction terms highlighted the dynamics of the treatment. Specifically, the interaction between study group and day 6 showed a significant reduction (β = -3.58, P = 0.025), whereas the interaction between study group and day 9 demonstrated a highly significant and strong negative effect (β = -21.16, P < 0.001), indicating almost complete sup
In contrast, the interactions between the placebo group and days 6 and 9 were not statistically significant (P > 0.48), reinforcing the absence of a therapeutic effect. Finally, analysis of the intercepts revealed that the model clearly differentiated between absent and moderate inflammation (0|1, P < 0.001); however, the difference between moderate and severe inflammation (1|2) was not statistically significant (P = 0.53). This finding suggests that while the model effectively captures early differences in inflammatory infiltration, it has limitations in distinguishing between higher levels of intensity.
Neovascularization was assessed as an ordered categorical variable (0 = absent, 1 = moderate, 2 = intense). To investigate the effect of the therapeutic intervention, multinomial logistic regression was applied with the independent variables group (control as reference, placebo, study), observation day (day 3 as reference) and the interaction term group × day. The regression analysis results of the neovascularization model coefficients are presented in Table 6.
| Predictor | Coefficient (β) | SE | t value | P value |
| Group study | 2.3072 | 0.9218 | 2.50 | 0.012 |
| Group placebo | 0.3138 | 0.8292 | 0.38 | 0.705 |
| Day 6 | -0.7534 | 0.9046 | -0.83 | 0.405 |
| Day 9 | -1.2683 | 0.9872 | -1.28 | 0.199 |
| Group study × day 6 | 3.0884 | 1.5367 | 2.01 | 0.044 |
| Group study × day 9 | 2.9249 | 1.4193 | 2.06 | 0.039 |
| Group placebo × day 6 | 1.7872 | 1.2369 | 1.44 | 0.148 |
| Group placebo × day 9 | 1.3427 | 1.3110 | 1.02 | 0.306 |
| Cut-point 0|1 | 0.1451 | 0.5973 | 0.24 | 0.808 |
| Cut-point 1|2 | 2.5504 | 0.7012 | 3.64 | < 0.001 |
Commentary on results: Compared with the control group, the study group had a significantly increased probability of more intense neovascularization (β = 2.31, P = 0.012), demonstrating the strong angiogenic effect of the therapeutic intervention. The interaction terms study group × day 6 (β = 3.09, P = 0.044) and study group × day 9 (β = 2.92, P = 0.039) were statistically significant, confirming that the positive effect of the treatment progressively increased with time and was maintained until day 9. In contrast, the placebo group did not show statistically significant differences in either the main effects or the interactions with days (P > 0.14), which reinforces the indication of the absence of a biological effect. The individual effects of days (day 6, day 9) were not statistically significant (P > 0.19), indicating that the enhancement of neovascularization is attributed mainly to the therapeutic intervention and not to the simple effect of time.
Finally, analysis of the thresholds revealed that the model does not adequately distinguish between absent and moderate neovascularization (0|1, P = 0.808) but clearly distinguishes moderate from intense (1|2, P < 0.001). This finding confirms its ability to capture the highest levels of angiogenesis. Overall, the administration of the substance to the study group was associated with significantly increased neovascularization, especially on days 6 and 9, compared with that in the control and placebo groups. The results clearly show time dependence, as the effect is enhanced over time and remains strong. The modeling proved effective in distinguishing the levels of neovascularization, especially between moderate and intense, enhancing the reliability of the findings and confirming the strong angiogenic effect of the therapeutic intervention.
Fibroblastic response was recorded as an ordinal categorical variable (0 = absent, 1 = moderate, 2 = intense) and analyzed using multinomial logistic regression. The effects of group (control as reference, placebo, study), observation day (day 3 as reference) and the interaction term group × day were examined. The regression analysis results of the neovascularization model coefficients are presented in Table 7.
| Predictor | Coefficient (β) | SE | t value | P value |
| Group study | 2.0683 | 0.9107 | 2.27 | 0.023 |
| Group placebo | 0.2975 | 0.8148 | 0.37 | 0.715 |
| Day 6 | -0.6730 | 0.8562 | -0.79 | 0.432 |
| Day 9 | -1.0816 | 0.8970 | -1.21 | 0.228 |
| Group study × day 6 | 3.0282 | 1.5111 | 2.00 | 0.045 |
| Group study × day 9 | 3.5531 | 1.5307 | 2.32 | 0.020 |
| Group placebo × day 6 | 1.7481 | 1.2163 | 1.44 | 0.151 |
| Group placebo × day 9 | 1.2231 | 1.2378 | 0.99 | 0.323 |
| Cut-point 0|1 | -0.1883 | 0.5850 | -0.32 | 0.748 |
| Cut-point 1|2 | 2.3328 | 0.6766 | 3.45 | < 0.001 |
Commentary on results: Compared with the control group, the study group presented a significantly increased probability of intense fibroblastic response (β = 2.07, P = 0.023). These findings demonstrate that therapeutic intervention strongly stimulates fibroblast activation and collagen production. In contrast, compared with the control group, the placebo group did not significantly differ (P = 0.715), which reinforces the indication of the absence of biological activity.
The individual effects of time (days 6 and 9) were negative but not statistically significant (P > 0.22), suggesting that they do not lead to a reduction in response by themselves unless combined with therapeutic intervention. The interaction terms study group × day 6 (β = 3.03, P = 0.045) and study group × day 9 (β = 3.55, P = 0.020) were statistically significant. This finding indicates that not only did the treatment increase fibroblast activity but also that this phenomenon became more pronounced and more consistent over the following days, confirming the time-dependent enhancement of the healing process. The placebo × day interactions were not significant (P > 0.15), excluding any therapeutic contribution of the placebo.
With respect to the thresholds, the model was unable to satisfactorily distinguish between no response and a moderate response (0|1, P = 0.748), but it accurately distinguished moderate from intense response (1|2, P < 0.001), which reinforces the validity of the conclusions about the higher levels of fibroblast activation.
Overall, therapeutic intervention applied to the study group led to a clear and statistically significant increase in the fibroblast response, especially on days 6 and 9, which is consistent with the activation of tissue repair and remodeling mechanisms. In contrast, the placebo group did not significantly differ. The model reliably distinguished between moderate and intense responses, confirming that the intervention substantially increased the level of fibroblast activation.
In all samples examined in the control group, diffuse coagulative necrosis (corresponding to the area where the special metal sealer was applied) and microhemorrhage were observed. This involved all layers of the epidermis and the middle and deep dermis; however, in some cases, it extended to the muscle layer. In the study group, rosuvastatin induced a significant decrease in both lesions (P < 0.001). The healing process, including hyperemia, microhemorrhages, inflammatory infiltration and the formation of neogranulation tissue and neovascularization, involves the deep dermis, muscularis mucosa and subcutaneous tissue.
No adverse effects or deaths were observed in any experimental group during the study.
Rosuvastatin, a synthetic hydrophilic statin, is distinguished within its class based on its unique pharmacokinetic and pharmacodynamic properties. It has the longest half-life among statins because of its molecular structure, which allows it to bind to multiple sites on 3-hydroxy-3-methylglutaryl-CoA reductase inhibitor that produces cholesterol[7]. This enables effective enzyme inhibition at lower doses of the drug[52]. The primary role of rosuvastatin is associated with two genes: Solute carrier organic anion transporter family member 1B1 and ATP-binding cassette transporter G2[6]. These genes regulate proteins responsible for transporting the drug throughout the body. Data on ATP-binding cassette transporter G2 genotype polymorphisms could be valuable for tailoring rosuvastatin treatment to individual patients because these polymorphisms are associated with the drug’s pharmacokinetics[52,53]. Its liver selectivity leads to reduced bioavailability in other tissues, thereby decreasing the risk of muscle toxicity after administration[7]. Additionally, rosuvastatin is metabolized mainly by the cytochrome P450 2C9 enzyme, with minimal involvement of cytochrome P450 3A4[6], which accounts for its minimal interactions with drugs that inhibit cytochrome P450 enzymes.
The hydrophilic properties of rosuvastatin and its selective uptake by liver cells lead to minimal passive diffusion across cell membranes and almost complete absorption by the liver[7]. As a result, compared with simvastatin and pra
Rosuvastatin improves microvascular function in the skin of individuals with type 2 diabetes mellitus[57] and helps in the healing of diabetic wounds[58]. However, long-term use of statins, particularly atorvastatin followed by rosuvastatin, may increase the risk of developing diabetes mellitus[59].
Compared with oral intake, the topical application of rosuvastatin for wound healing is beneficial. It helps prevent gastrointestinal issues and reduces side effects. Moreover, topical delivery allows the medication to cover a wider area of the skin, promoting faster healing by improving epithelial growth and reducing the presence of inflammatory cells[60]. Additionally, rosuvastatin has antimicrobial effects[4].
Histopathological changes in burn tissue can be classified into three concentric zones: The coagulative necrosis zone (characterized by ischemic necrosis and hypoxia), the stagnation zone (representing borderline viability), and the hyperemia zone. Burns compromise skin integrity and impair both cellular and humoral immunity. Heat-induced vascular damage increases vascular permeability, resulting in the extravasation of plasma and electrolytes into the interstitial space. Dead tissues decrease the number of immune cells in the burned region, allowing pathogens to penetrate deeper into the underlying tissues and facilitating their spread[61].
Considering the aforementioned literature, we chose to evaluate rosuvastatin in partial-thickness burn wounds using a well-established experimental rat model. This model was selected because it accurately replicates the local and systemic pathophysiological processes characteristic of human burns, thereby enabling the extraction of clinically relevant findings. In an experimental study in rats, the optimal topical application dose of rosuvastatin was 20 mg/kg body weight to prevent intra-articular adhesions of the knee[37]. In our study, we used 200 μL of cream once daily, which equates to approximately 7-8 mg of rosuvastatin per rat weighing approximately 350-400 g.
The originality of this study lies in the fact that although previous research has focused on the effects of other statins (e.g., simvastatin, atorvastatin, and pravastatin), with encouraging results in healing various burn wound models, rosuvastatin - as a newer, more potent statin with a better safety profile - has not yet been investigated in this specific context. Therefore, to our knowledge, this is the first study to examine the use of rosuvastatin for burn wound healing in rats.
The study focused on three main areas: (1) The systemic inflammatory response was assessed by quantifying the levels of proinflammatory cytokines (IL-1β, IL-6, and TNF-α) in serum to investigate the extent of inflammatory activation and the potential role of rosuvastatin in modulating this response; (2) Histopathological lesions in the skin were evaluated by examining parameters such as inflammatory infiltration, neovascularization, fibroblastic reactions, coagulative necrosis, and hemorrhages to assess the effects of rosuvastatin at the morphological and cellular levels; and (3) Macroscopic evaluation (size and contraction of the burn wound) was performed.
This integrated approach enabled the simultaneous assessment of both morphological changes in the tissue and the underlying molecular mechanisms, thereby enhancing the validity and interpretative power of the findings. Con
The findings of the present study can be summarized in six key points: (1) The induction of a partial-thickness burn wound is accompanied by an intense inflammatory response, an increase in proinflammatory cytokines, and significant histological alterations in the skin; (2) Rosuvastatin significantly reduced inflammatory infiltration and cytokine levels, indicating an anti-inflammatory effect and a decrease in burn wound size. Using the third-day values from the control group as baseline, the study group demonstrated statistically significant decreases in serum CRP (5.75-10.30-16.15 vs 21.40-4.87-9.69, P < 0.01), TNF-α (11.02-21.25-28.21 vs 35.29-7.83-12.51, P < 0.01), and IL-1β (6.19-10.28-14.41 vs 17.83-3.72-6.93, P < 0.01), and IL-6 levels (9.71-20.88-29.84 vs 40.24-5.75-14.37, P < 0.01) as well as a significant reduction in burn wound size (30.52-83.00-135.84 vs 235.17-31.47-73.62, P < 0.01) across the later sacrifice days. These findings clearly demonstrate a distinct difference in the inflammatory response between the groups during the follow-up period. The study group showed significant and gradual decreases in the levels of the inflammatory markers CRP, TNF-α, IL-1β, and IL-6. This reduction in inflammation correlated with a gradual decrease in burn wound size in the study group, indicating a more effective therapeutic response to rosuvastatin; (3) Rosuvastatin administration was associated with enhanced neovascularization (P = 0.012) and a fibroblastic response (P = 0.023), processes critical for the organization of granulation tissue and the promotion of healing; (4) The presence of coagulative necrosis and hemorrhages was less pronounced in treated animals (P < 0.001), suggesting that rosuvastatin has a protective effect on the wound microenvironment; (5) The combination of the above findings demonstrates that rosuvastatin does not act unidimensionally; rather, it affects both the inflammatory and reparative phases of healing, achieving a better balance between the two; and (6) The administration of rosuvastatin was safe in this experimental model, with no adverse effects or mortality observed.
Overall, our results suggest that rosuvastatin represents a potentially promising therapeutic agent for the treatment of burn wounds, as it combines anti-inflammatory, angiogenic, and reparative effects. This study presents a series of significant findings regarding the effects of rosuvastatin on burn wound healing. These findings not only confirm previous observations about the pleiotropic actions of statins but also provide new data with potential clinical and pathophysiological significance.
The findings of our study support the conclusion that partial-thickness burn induction triggers an intense inflammatory response characterized by elevated levels of proinflammatory cytokines (IL-1β, IL-6, TNF-α) and significant histological alterations, which is consistent with the established pathophysiology of burn wounds. The administration of rosuvastatin resulted in a significant reduction in the levels of these cytokines, demonstrating the anti-inflammatory effects of the substance, which is consistent with the existing international literature. Histopathologically, therapeutic intervention was associated with decreased inflammatory infiltration, reduced coagulative necrosis, and fewer hemorrhages, highlighting a protective effect on the wound microenvironment. Rosuvastatin also significantly enhanced neovascularization and fibroblastic activity - processes critical for tissue remodeling and collagen synthesis - confirming the angiogenic and regenerative effects reported in recent studies. Macroscopically, burn wound contraction was faster and more pro
The authors extend their appreciation to Ioannis Taitzoglou, Professor, School of Veterinary Medicine, Faculty of Health Sciences, Aristotle University of Thessaloniki, for his valuable assistance and guidance in the experimental performance; and Vasilis Chasiotis, Assistant Professor of Official Statistics and Statistical Methodology, Department of Statistics, Athens University of Economics and Business, for performing the statistical analysis and review.
| 1. | Sarhan OM, Gebril MI, Mohamed HH, Gamal GM, Abozaid KA, Sayed EM, Moustafa RH, Hussein YA, Elsayed HH, Hamed MOA. Harnessing nanotechnology for enhanced wound healing: integrating polymeric nanoparticles with rosuvastatin for targeted therapy. J Drug Target. 2025;1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 2. | Kilinc BR, Kostak F, Yilmaz OF, Pehlivanoglu S, Sirin DY. Molecular Docking and Functional Analyses Reveal the Chondroprotective and Anti-Inflammatory Potential of Statins in Osteoarthritis. J Cell Mol Med. 2025;29:e70791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 3. | Sabeel S, Motaung B, Nguyen KA, Ozturk M, Mukasa SL, Wolmarans K, Blom DJ, Sliwa K, Nepolo E, Günther G, Wilkinson RJ, Schacht C, Kengne AP, Thienemann F, Guler R. Impact of statins as immune-modulatory agents on inflammatory markers in adults with chronic diseases: A systematic review and meta-analysis. PLoS One. 2025;20:e0323749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 4. | Al-Kuraishy HM, Al-Gareeb AI, Al-Buhadily AK. Rosuvastatin as forthcoming antibiotic or as adjuvant additive agent: In vitro novel antibacterial study. J Lab Physicians. 2018;10:271-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 5. | Bao JW, Sun B, Ma PP, Gai YS, Sun WZ, Yu HQ, Li J. Rosuvastatin inhibits inflammatory response and resists fibrosis after myocardial infarction. Eur Rev Med Pharmacol Sci. 2018;22:238-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 6. | González-Iglesias E, Novalbos J, Abad-Santos F. The pharmacogenetics of rosuvastatin and implications for treatment: a systematic review. Pharmacogenomics. 2025;26:311-329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 7. | Kanukula R, Salam A, Rodgers A, Kamel B. Pharmacokinetics of Rosuvastatin: A Systematic Review of Randomised Controlled Trials in Healthy Adults. Clin Pharmacokinet. 2021;60:165-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 8. | Marneri AG, Pavlidis ET, Stavrati KE, Mouratidou C, Kotoulas SC, Ballas KD, Pavlidis TE. Effects of rosuvastatin treatment and other statins on burn wound healing. World J Methodol. 2025;In press. |
| 9. | Bedi O, Dhawan V, Sharma PL, Kumar P. Pleiotropic effects of statins: new therapeutic targets in drug design. Naunyn Schmiedebergs Arch Pharmacol. 2016;389:695-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 141] [Article Influence: 14.1] [Reference Citation Analysis (1)] |
| 10. | Santana CDS, Breda MRS, Vicentini YF, Santos SAAD, Justulin LA Jr, Castilho ACS, Nai GA, Santarém CL. Type I and type III collagen immunoexpression in rabbit skin biopsy samples treated with rosuvastatin gel and autologous platelet-rich plasma. Acta Cir Bras. 2025;40:e402725. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 11. | Qin P, Zhou P, Huang Y, Long B, Gao R, Zhang S, Zhu B, Li YQ, Li Q. Upregulation of rate-limiting enzymes in cholesterol metabolism by PKCδ mediates endothelial apoptosis in diabetic wound healing. Cell Death Discov. 2024;10:263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 12. | Zaki RM, Seshadri VD, Mutayran AS, Elsawaf LA, Hamad AM, Almurshedi AS, Yusif RM, Said M. Wound Healing Efficacy of Rosuvastatin Transethosomal Gel, I Optimal Optimization, Histological and In Vivo Evaluation. Pharmaceutics. 2022;14:2521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 13. | Aboelazayem S, Nasra M, Ebada H, Abdallah O. Ethyl-Cellulose Nanosponges for Topical Delivery of Simvastatin with Preferential Skin Retention for Wound Healing in a Full-Thickness Wound Rat Model. AAPS PharmSciTech. 2025;26:126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Gomes Daré R, Beatriz Chieco Costa A, Silva Martins T, Lopes LB. Simvastatin and adenosine-co-loaded nanostructured lipid carriers for wound healing: Development, characterization and cell-based investigation. Eur J Pharm Biopharm. 2024;205:114533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 15. | Abu El Hawa AA, Klein D, Bekeny JC, Severin JH, Zolper EG, Tefera E, Evans KK, Fan KL, Attinger CE. The impact of statins on wound healing: an ally in treating the highly comorbid patient. J Wound Care. 2022;31:S36-S41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 16. | Jull A, Lu H, Jiang Y. Statins and venous leg ulcer healing: Secondary analysis of data from a cohort of three randomised controlled trials. Wound Repair Regen. 2022;30:186-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 17. | Heydari MB, Ghanbari-Movahed Z, Heydari M, Farzaei MH. In vitro study of the mesenchymal stem cells-conditional media role in skin wound healing process: A systematic review. Int Wound J. 2022;19:2210-2223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (2)] |
| 18. | Chello C, Nenna A, Chello M, Satriano UM, Cardetta F, Lusini M, Nappi F, Dianzani C. Statin treatment and hypertrophic scarring after cardiac surgery. Wound Repair Regen. 2021;29:129-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 19. | O'Dell B, Rothenberg G, Holmes C, Priesand S, Mizokami-Stout K, Brandt EJ, Schmidt BM. The impact of statin therapy on the healing of diabetic foot ulcers: a case-control series. Clin Diabetes Endocrinol. 2024;10:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 20. | AlJunaydil NA, Lambarte RNA, Sumague TS, Alghamdi OG, Niazy AA. Lovastatin and Resveratrol Synergistically Improve Wound Healing and Inhibit Bacterial Growth. Int J Mol Sci. 2025;26:851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 21. | Chen Z, Xiao L, Hu C, Shen Z, Zhou E, Zhang S, Wang Y. Aligned lovastatin-loaded electrospun nanofibers regulate collagen organization and reduce scar formation. Acta Biomater. 2023;164:240-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 22. | Janipour Z, Najafi H, Abolmaali SS, Heidari R, Azarpira N, Özyılmaz ED, Tamaddon AM. Simvastatin-Releasing Nanofibrous Peptide Hydrogels for Accelerated Healing of Diabetic Wounds. ACS Appl Bio Mater. 2023;6:4620-4628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 23. | Ahmed LM, Hassanein KMA, Mohamed FA, Elfaham TH. Formulation and evaluation of simvastatin cubosomal nanoparticles for assessing its wound healing effect. Sci Rep. 2023;13:17941. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 24. | Patel VN, Patel HV, Agrawal K, Soni I, Shah P, Mangrulkar SV, Umekar MJ, Lalan MS. Comprehensive developmental investigation on simvastatin enriched bioactive film forming spray using the quality by design paradigm: a prospective strategy for improved wound healing. J Drug Target. 2024;32:1139-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 25. | Rahamathulla M, Pokale R, Al-Ebini Y, Osmani RAM, Thajudeen KY, Gundawar R, Ahmed MM, Farhana SA, Shivanandappa TB. Simvastatin-Encapsulated Topical Liposomal Gel for Augmented Wound Healing: Optimization Using the Box-Behnken Model, Evaluations, and In Vivo Studies. Pharmaceuticals (Basel). 2024;17:697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 26. | Rezvanian M, Ng SF, Alavi T, Ahmad W. In-vivo evaluation of Alginate-Pectin hydrogel film loaded with Simvastatin for diabetic wound healing in Streptozotocin-induced diabetic rats. Int J Biol Macromol. 2021;171:308-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 27. | Laing T, Hanson R, Chan F, Bouchier-Hayes D. Effect of pravastatin on experimental diabetic wound healing. J Surg Res. 2010;161:336-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 28. | Holler V, Buard V, Gaugler MH, Guipaud O, Baudelin C, Sache A, Perez Mdel R, Squiban C, Tamarat R, Milliat F, Benderitter M. Pravastatin limits radiation-induced vascular dysfunction in the skin. J Invest Dermatol. 2009;129:1280-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 93] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 29. | Yasunami N, Ayukawa Y, Furuhashi A, Atsuta I, Rakhmatia YD, Moriyama Y, Masuzaki T, Koyano K. Acceleration of hard and soft tissue healing in the oral cavity by a single transmucosal injection of fluvastatin-impregnated poly (lactic-co-glycolic acid) microspheres. An in vitro and rodent in vivo study. Biomed Mater. 2015;11:015001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 30. | Yoon JP, Park SJ, Kim DH, Choi YS, Lee HJ, Park EJJ, Cho CH, Chung SW. Ezetimibe/Atorvastatin, a Treatment for Hyperlipidemia, Inhibits Supraspinatus Fatty Infiltration and Improves Bone-Tendon Interface Healing in a Rotator Cuff Tear Rat Model. Am J Sports Med. 2025;53:80-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 31. | Miranda-Filho FV, Barbosa S, Panigali OA, Silva MC, Costa MGD, Flores FDS, Ervolino E, Theodoro LH, Magro-Filho O, Faverani LP. Effect of local and systemic administration of atorvastatin for improving bone healing on critical defects. Braz Dent J. 2024;35:e246114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 32. | Xiang J, Zhou L, Xie Y, Zhu Y, Xiao L, Chen Y, Zhou W, Chen D, Wang M, Cai L, Guo L. Mesh-like electrospun membrane loaded with atorvastatin facilitates cutaneous wound healing by promoting the paracrine function of mesenchymal stem cells. Stem Cell Res Ther. 2022;13:190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 33. | Yu M, Liu W, Li J, Lu J, Lu H, Jia W, Liu F. Exosomes derived from atorvastatin-pretreated MSC accelerate diabetic wound repair by enhancing angiogenesis via AKT/eNOS pathway. Stem Cell Res Ther. 2020;11:350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 331] [Cited by in RCA: 314] [Article Influence: 52.3] [Reference Citation Analysis (1)] |
| 34. | Salem HF, Nafady MM, Ewees MGE, Hassan H, Khallaf RA. Rosuvastatin calcium-based novel nanocubic vesicles capped with silver nanoparticles-loaded hydrogel for wound healing management: optimization employing Box-Behnken design: in vitro and in vivo assessment. J Liposome Res. 2022;32:45-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 35. | Tetila AF, Breda MRS, Nogueira RMB, Nai GA, Laposy CB. The Use of Platelet-Rich Plasma and Rosuvastatin in Wound Healing in Rabbits: A Longitudinal Study. Adv Skin Wound Care. 2019;32:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 36. | Maged A, Abdelkhalek AA, Mahmoud AA, Salah S, Ammar MM, Ghorab MM. Mesenchymal stem cells associated with chitosan scaffolds loaded with rosuvastatin to improve wound healing. Eur J Pharm Sci. 2019;127:185-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 37. | Wu H, Germanov AV, Goryaeva GL, Yachmenev AN, Gordienko DI, Kuzin VV, Skoroglyadov AV. The Topical Application of Rosuvastatin in Preventing Knee Intra-Articular Adhesion in Rats. Med Sci Monit. 2016;22:1403-1409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 38. | Gürer B, Kahveci R, Gökçe EC, Ozevren H, Turkoglu E, Gökçe A. Evaluation of topical application and systemic administration of rosuvastatin in preventing epidural fibrosis in rats. Spine J. 2015;15:522-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 39. | Türer A, Coşkun Türer Ç, Durmuşlar MC, Balli U, Önger ME. The influence of oral administration of rosuvastatin on calvarial bone healing in rats. J Craniomaxillofac Surg. 2016;44:1327-1332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 40. | Ibrahim HK, Fahmy RH. Localized rosuvastatin via implantable bioerodible sponge and its potential role in augmenting bone healing and regeneration. Drug Deliv. 2016;23:3181-3192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 41. | Mohajer Ansari J, Ramhormozi P, Shabani R, Pazoki-Toroudi H, Yari A, Barati M, Dahmardehei M, Babakhani A, Nobakht M. Simvastatin combined with bone marrow mesenchymal stromal cells (BMSCs) improve burn wound healing by ameliorating angiogenesis through SDF-1α/CXCR4 pathway. Iran J Basic Med Sci. 2020;23:751-759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 42. | Ramhormozi P, Ansari JM, Simorgh S, Asgari HR, Najafi M, Barati M, Babakhani A, Nobakht M. Simvastatin accelerates the healing process of burn wound in Wistar rats through Akt/mTOR signaling pathway. Ann Anat. 2021;236:151652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 43. | Hosny KM, Alhakamy NA, Sindi AM, Khallaf RA. Coconut Oil Nanoemulsion Loaded with a Statin Hypolipidemic Drug for Management of Burns: Formulation and In Vivo Evaluation. Pharmaceutics. 2020;12:1061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 44. | Zhao G, Yu YM, Kaneki M, Tompkins RG, Fischman AJ. Simvastatin protects hepatocytes from apoptosis by suppressing the TNF-α/caspase-3 signaling pathway in mice with burn injury. Ann Surg. 2013;257:1129-1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 45. | Beffa DC, Fischman AJ, Fagan SP, Hamrahi VF, Paul KW, Kaneki M, Yu YM, Tompkins RG, Carter EA. Simvastatin treatment improves survival in a murine model of burn sepsis: Role of interleukin 6. Burns. 2011;37:222-226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 46. | Akershoek JJ, Brouwer KM, Vlig M, Boekema BKHL, Beelen RHJ, Middelkoop E, Ulrich MMW. Differential effects of Losartan and Atorvastatin in partial and full thickness burn wounds. PLoS One. 2017;12:e0179350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 47. | Boyko T, Marin C, Furnari G, Flynn W, Lukan JK. Safety profile of atorvastatin in the role of burn wound injury conversion. Am J Surg. 2020;220:1323-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 48. | Tiwari VK. Burn wound: How it differs from other wounds? Indian J Plast Surg. 2012;45:364-373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 235] [Cited by in RCA: 182] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 49. | Tan JQ, Zhang HH, Lei ZJ, Ren P, Deng C, Li XY, Chen SZ. The roles of autophagy and apoptosis in burn wound progression in rats. Burns. 2013;39:1551-1556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 50. | Pradeep AR, Karvekar S, Nagpal K, Patnaik K, Raju A, Singh P. Rosuvastatin 1.2 mg In Situ Gel Combined With 1:1 Mixture of Autologous Platelet-Rich Fibrin and Porous Hydroxyapatite Bone Graft in Surgical Treatment of Mandibular Class II Furcation Defects: A Randomized Clinical Control Trial. J Periodontol. 2016;87:5-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 51. | Balakumar K, Raghavan CV, selvan NT, prasad RH, Abdu S. Self nanoemulsifying drug delivery system (SNEDDS) of rosuvastatin calcium: design, formulation, bioavailability and pharmacokinetic evaluation. Colloids Surf B Biointerfaces. 2013;112:337-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 188] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 52. | Kasten A, Cascorbi I. Understanding the impact of ABCG2 polymorphisms on drug pharmacokinetics: focus on rosuvastatin and allopurinol. Expert Opin Drug Metab Toxicol. 2024;20:519-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 53. | Song Y, Lim HH, Yee J, Yoon HY, Gwak HS. The Association between ABCG2 421C>A (rs2231142) Polymorphism and Rosuvastatin Pharmacokinetics: A Systematic Review and Meta-Analysis. Pharmaceutics. 2022;14:501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 54. | Zhang C, Wang Q, Li K, Fu M, Gao K, Lv C. Rosuvastatin: A Potential Therapeutic Agent for Inhibition of Mechanical Pressure-Induced Intervertebral Disc Degeneration. J Inflamm Res. 2024;17:3825-3838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 55. | Yan W, Alagarsamy KN, Srivastava A, Rafieerad A, Arora RC, Dhingra S. Rosuvastatin-loaded injectable immunomodulatory hydrogel mitigates local immune response against transplanted stem cells and promotes heart repair in vivo. Biomater Adv. 2026;178:214481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 56. | Momen YS, Kandeil MA, Mahmoud MO. A novel cardioprotective mechanism of rosuvastatin: restoring PINK1/parkin-mediated mitophagy via SIRT1/FOXO1 activation in doxorubicin-induced cardiotoxicity. Cancer Chemother Pharmacol. 2025;95:84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 57. | Parson HK, Bundy MA, Dublin CB, Boyd AL, Paulson JF, Vinik AI. Pleiotropic effects of rosuvastatin on microvascular function in type 2 diabetes. Diabetes Metab Syndr Obes. 2010;3:19-26. [PubMed] [DOI] [Full Text] |
| 58. | Hao H, Hu J, Kuai Z, Hao F, Jiang W, Ran N, He Y, Zhang Y, Huang Y, Qi Y, Luo Q. Enzyme-mediated multifunctional self-healing lysozyme hydrogel for synergistic treatment of chronic diabetic wounds. Int J Biol Macromol. 2024;282:136719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 59. | Zheng D, Ren J, Lv D, Zhao Q, Hong D. Ranking the Diabetes-related Safety Profile of Different Statin Drugs. Curr Pharm Des. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 60. | Farghaly Aly U, Abou-Taleb HA, Abdellatif AA, Sameh Tolba N. Formulation and evaluation of simvastatin polymeric nanoparticles loaded in hydrogel for optimum wound healing purpose. Drug Des Devel Ther. 2019;13:1567-1580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 61. | Shariati A, Moradabadi A, Azimi T, Ghaznavi-Rad E. Wound healing properties and antimicrobial activity of platelet-derived biomaterials. Sci Rep. 2020;10:1032. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |