Published online Mar 20, 2026. doi: 10.5493/wjem.v16.i1.115508
Revised: November 3, 2025
Accepted: December 30, 2025
Published online: March 20, 2026
Processing time: 148 Days and 11.5 Hours
Odontogenic tumors are a heterogeneous group of lesions originating from tissues involved in odontogenesis. Mixed odontogenic tumors comprise a group of le
To determine and compare the expression of caveolin-1, MOC-31, connexin-43 (Cx-43), and Ki-67 in ameloblastic fibromas (AF), ameloblastic fibrodentinomas (AFD), ameloblastic fibro-odontomas (AFO), and odontomas (O).
Immunohistochemical analysis for caveolin-1, MOC-31, Cx-43, and Ki-67 was performed on eight AF, one AFD, five AFO, and six O.
Variable protein expression was identified in the epithelium and mesenchyme of most AF, AFD, and AFO with lower expression observed in O. Immunohistochemical analysis revealed significant differences in protein expression between AF and O (P = 0.0085) and between AFO/AFD and O (P = 0.0250). When eva
This immunohistochemical study suggested that the similarity in protein expression between AFD, AFO, and AF compared with O may be due to the biological proximity among these lesions at the protein expression level. Further studies with larger sample sizes and complementary techniques are required to validate these findings.
Core Tip: This study compared the immunohistochemical expression of caveolin-1, MOC-31, connexin-43, and Ki-67 in ameloblastic fibroma (AF), ameloblastic fibrodentinoma (AFD), ameloblastic fibro-odontoma (AFO), and odontoma (O). AF, AFD, and AFO showed similar epithelial and mesenchymal profiles with lower expression in O. Significant differences in MOC-31 and Cx-43 between AF/AFO and O suggest that AFD and AFO may represent variants of AF rather than de
- Citation: Umpiérrez NG, Sicco E, Silveira F, Beovide AV, Bologna-Molina R. Differences of immunohistochemical profiling of MOC-31, caveolin-1, connexin-43, Ki-67 in ameloblastic fibroma, ameloblastic fibrodentinoma, ameloblastic fibro-odontoma, and odontomas. World J Exp Med 2026; 16(1): 115508
- URL: https://www.wjgnet.com/2220-315x/full/v16/i1/115508.htm
- DOI: https://dx.doi.org/10.5493/wjem.v16.i1.115508
Mixed odontogenic tumors (OT) comprise a group of lesions characterized by the presence of ectodermal and mesenchymal odontogenic tissues with or without the formation of hard dental tissues[1-3]. Ameloblastic fibroma (AF) is a rare, benign, mixed tumor composed of odontogenic mesenchyme resembling dental papilla and odontogenic epithe
Cell signaling molecules are integral components of a complex communication system that regulates basic cellular functions and interactions with the microenvironment. Disruptions such as loss of adhesion, impaired maturation, altered intercellular communication, and increased proliferation may contribute to the development of odontogenic neoplasms[6]. Some of these biomolecules are useful biomarkers for understanding the behavior of some OTs. Our group previously used some biomarkers to provide knowledge to the tumor biology of odontogenic neoplasms[6,7].
MOC-31 is an intercellular adhesion molecule involved in tight junction formation in epithelial cells[8]. Its overexpression has been reported in epithelial-origin neoplasms, including ameloblastomas, and it has also been detected in mixed odontogenic lesions such as primordial OTs[2,6]. Caveolin-1 (Cav-1) is an integral membrane protein involved in various biological processes and expressed in both epithelial and mesenchymal cells with documented expression during different stages of tooth development[9]. Its presence has also been described in odontogenic cystic lesions and neoplastic lesions with locally aggressive clinical behavior, such as ameloblastomas[10].
Connexin-43 (Cx-43) is an integral membrane protein forming channels that facilitate intercellular communication and the exchange of ions and molecules[11]. Its role in osteoblast and osteoclast development has been established[12], and it has been suggested to contribute to ameloblast differentiation. However, its precise role in the development of OT remains poorly understood, with only a few studies investigating its expression in odontogenic lesions[7,13]. Lastly, Ki-67 is a non-histone nuclear protein associated with DNA[14]. Widely recognized as a prototypical biomarker for cellular proliferation, it identifies the total fraction of proliferating cells[15,16].
The biomarkers MOC-31, Cav-1, Ki-67, and Cx-43 are involved in critical cellular functions such as adhesion, proliferation, signaling, and intercellular communication. Although each has a distinct role, they functionally intersect in processes such as tumor progression, tissue remodeling, and cellular homeostasis.
These markers tend to interact in various biological processes. For example, Cx-43 colocalizes with Cav-1 and Cav-2 in keratinocytes and regulates intercellular communication[17]. Cx-43 interacts with specific domains of Cav-1, participating in cellular trafficking and signaling[18], and elevated expression of Cav-1 and Ki-67 has been associated with metastasis and tumor progression[19]. Moreover, Cx-43 and Ki-67 show a positive correlation, suggesting that Cx-43 may play a role in proliferation[20,21]. In summary, Cav-1 modulates the function of Cx-43, and MOC-31 activates proliferative pathways associated with Ki-67. When coexpressed in tumors, these molecules may have combined diagnostic and prognostic value.
In the present study we focused our attention on mixed OTs, a group whose recognition and classification have been the subject of ongoing debate across successive editions of the WHO classification. This diagnostic ambiguity highlights the need for additional tools that may contribute to a better understanding of their histogenesis and biological behavior. In this context we deemed it pertinent to apply this panel of biomarkers previously used by our research group in other OTs[2,6,7]. The selection of this set of biomarkers was based on their indirect involvement in the regulation of cell proliferation, adhesion, and intercellular communication, especially in cancer contexts, and on the possibility of comparing the results obtained with those from previously studied OTs.
The aim of this study was to determine and compare the immunohistochemical expression patterns of MOC-31, Cav-1, Cx-43, and Ki-67 in AF, AFD, AFO, and O to contribute new knowledge that may help to better understand the biological behavior of these OTs.
The cases were randomly selected and consisted of formalin-fixed, paraffin-embedded samples (five AFO, one AFD, eight AF, and six O) obtained from the archives of the Subunit of Pathological Anatomy, Department of Diagnostics in Pa
For the immunohistochemical analyses all samples were subjected to the same standardized mild decalcification protocol implemented in the Department of Pathology at the School of Dentistry. We used Histodecal® Extra (Biopack S.A., Argentina), a chelating decalcifying solution based on ethylenediaminetetraacetic acid, with neutral pH and free from strong or organic acids. This formulation preserves tissue antigenicity and optimizes the detection of selected biomarkers in mineralized tissues.
For immunohistochemical processing 3-μm thick sections mounted on poly-L-lysine-coated slides of each sample were deparaffinized and hydrated. Antigen retrieval was performed at 100 °C for 1 min (Borg Decloaker, RTU; Biocare Medical). Endogenous peroxidases were blocked with 0.9% hydrogen peroxide for 5 min at room temperature (RT). The slides were incubated with the following primary antibodies: Anti-Ki-67 (MIB-1; dilution 1:100, Dako Corporation, United States); anti-Cav-1 (caveolin 1; dilution 1:100, Santa Cruz Biotechnology, Dallas, TX, United States); anti-MOC-31 (Ep-CAM; dilution 1:100, Dako Corporation, United States); or anti-Cx-43 (CX-1 B1; dilution 1:100, Thermo Fisher Scientific, Waltham, MA, United States) for 60 min at RT. Subsequently, the sections were incubated with biotinylated anti-mouse/anti-rabbit antibody and with the streptavidin/peroxidase complex (LSA-B + Labeled streptavidin-biotin, Dako Cor
All histological slides were scanned in digital format using the Motic VM 3.0 Digital Slide Scanning System. For analysis, Motic VM 3.0 - Motic Digital Slide Assistant software (version 1.0.7.46, Copyright Motic China Group Co., Ltd. 2017) was used. The cases were quantified independently, recording protein expression in the epithelial and mesen
Given the rarity and archival nature of these entities, prospective sample-size calculation was not feasible; analyses were treated as exploratory with an emphasis on effect sizes and uncertainty. A sensitivity analysis excluding the single AFD case was conducted. An age-stratified (median split) analysis and an exploratory multivariable logistic mixed model (age, sex, location) assessed robustness of associations.
The results were analyzed using descriptive statistics. To assess the pattern of immunohistochemical expression, a mixed-effects logistic regression model was applied. To compare the expression and establish the degree of similarity, a post-hoc multiple comparison model and the Mann-Whitney test were performed. Additionally, contingency tables with χ2 tests were used to evaluate the relationship between expression and clinical variables. The statistical package Stata was used, and results were considered significant with a P value ≤ 0.05.
In this study OT were in the mandible in 13 (65%) cases and affected 13 (65%) males with a mean age of 23 years (ranging from 4 to 68 years). All AFO were in the mandible with 3 (60%) in males and a mean age of 12 years. AF were in both the maxilla and mandible with 5 (62%) in males and a mean age of 27 years. The AFD was in the mandible in a male aged 24 years. The O were in 3 (50%) cases in the mandible with 4 (67%) in males and a mean age of 23 years. For statistical analysis AFO and AFD were grouped together due to the presence of only one case of AFD and the consideration that they may be similar in biological behavior according to the WHO classification[4,6].
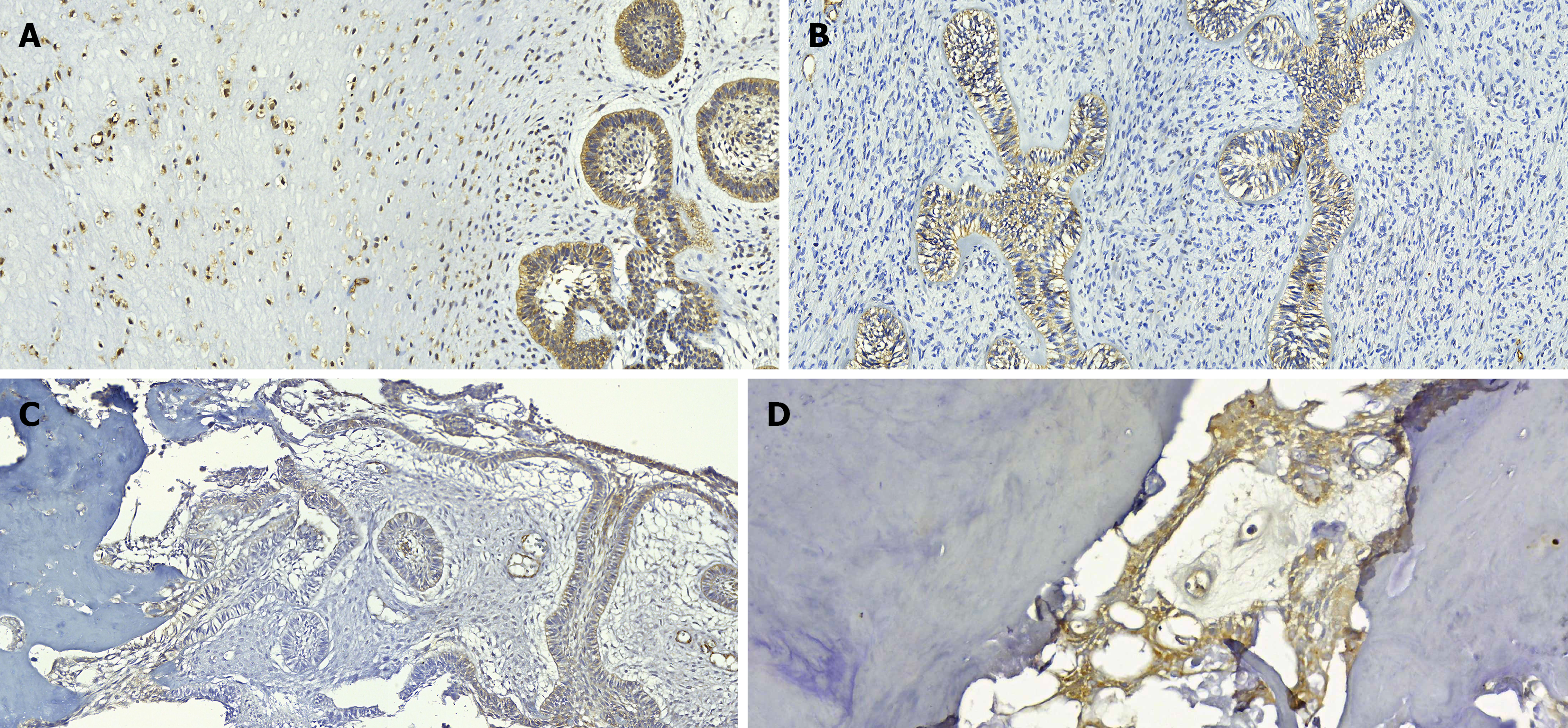
Cav-1 was expressed in 17 (85%) of the 20 OT evaluated with membrane-cytoplasm staining (+) and (++) at the level of ameloblasts, stellate reticulum areas, and odontogenic mesenchyme in all AFO and AFD, 6 AF (75%), and dental papilla areas in 5 O (83.3%). No staining was observed in two of the eight AF (25%), and in the PC, except for one (20%) that showed (+) staining in the reduced enamel epithelium of the enamel organ (Table 1, Figure 1).
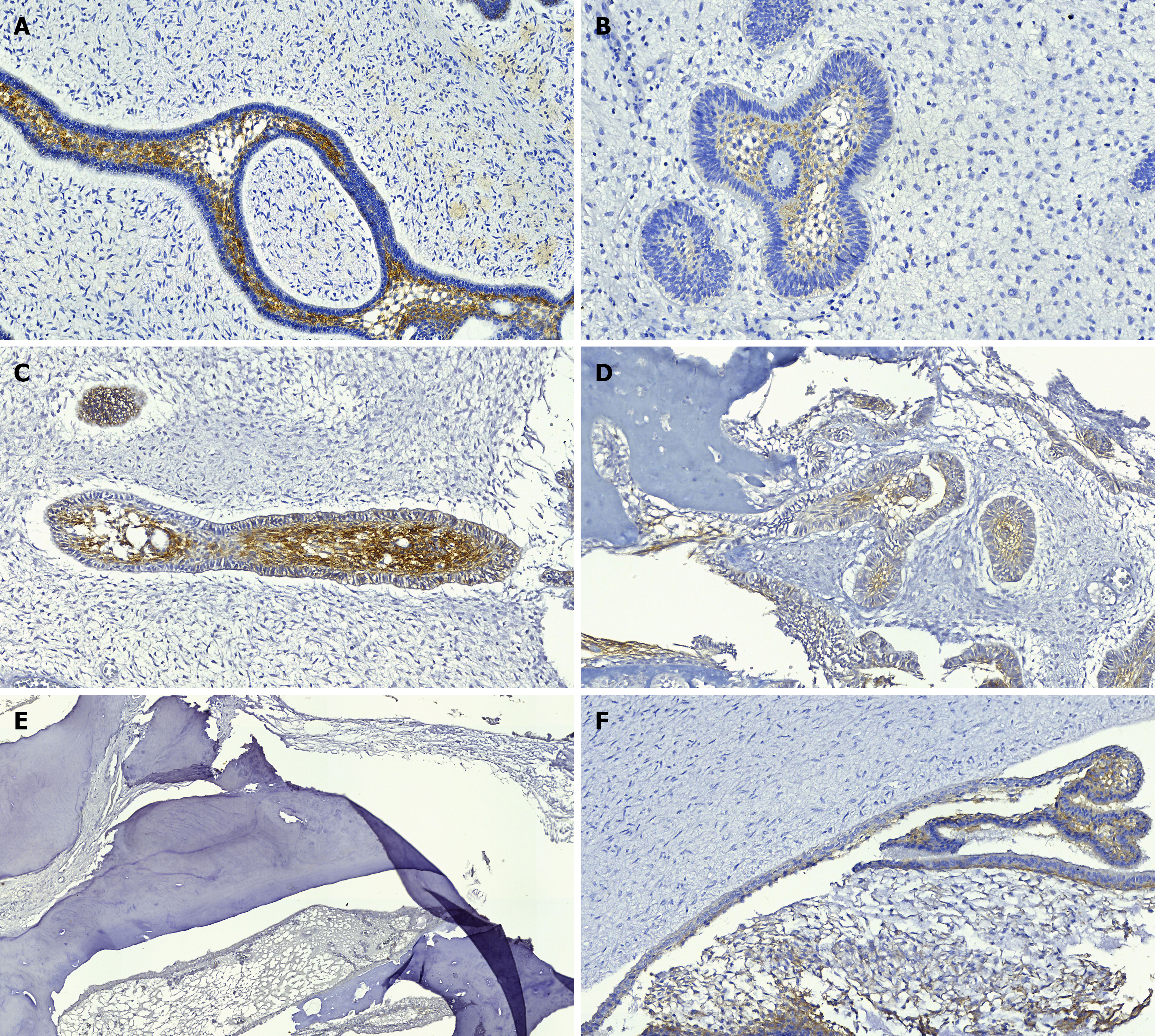
Regarding the tissue expression of MOC-31, membrane-cytoplasm staining (+) and (++) was observed in the ameloblasts and stellate reticulum areas of the single AFD, 2 AFO (40%), 5 AF (63%), and 1 PC (20%). O were all negative for this biomarker (Table 1, Figure 2).
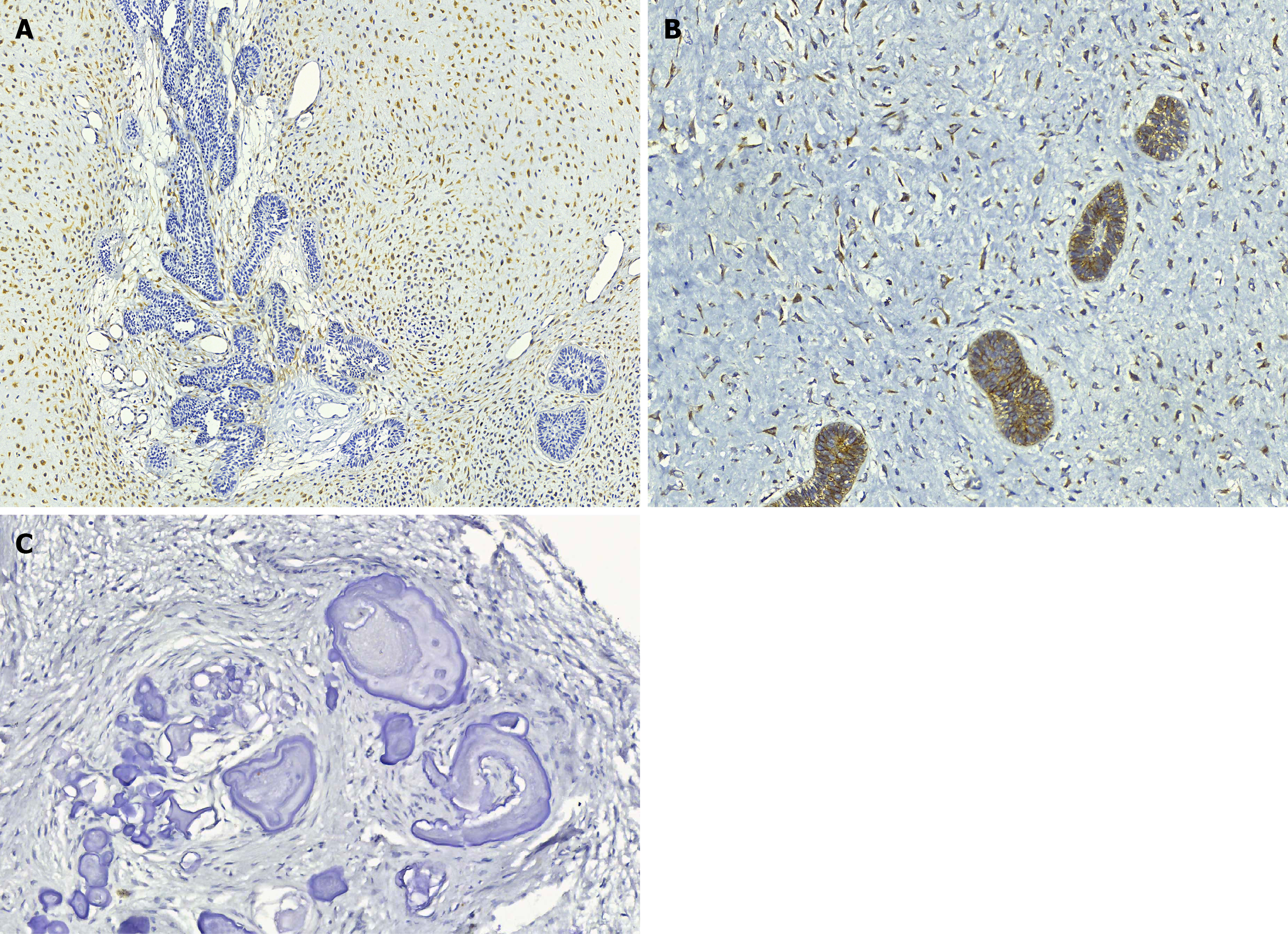
For Cx-43 expression membranous (+) and (++) mesenchymal staining was observed in all AFD and AFO, 5 AF (63%), and 3 PC (60%). In one AFO, one AF, and the AFD, the positivity was (++) and mixed, also involving the ameloblasts. All O were negative for this biomarker (Table 1, Figure 3).
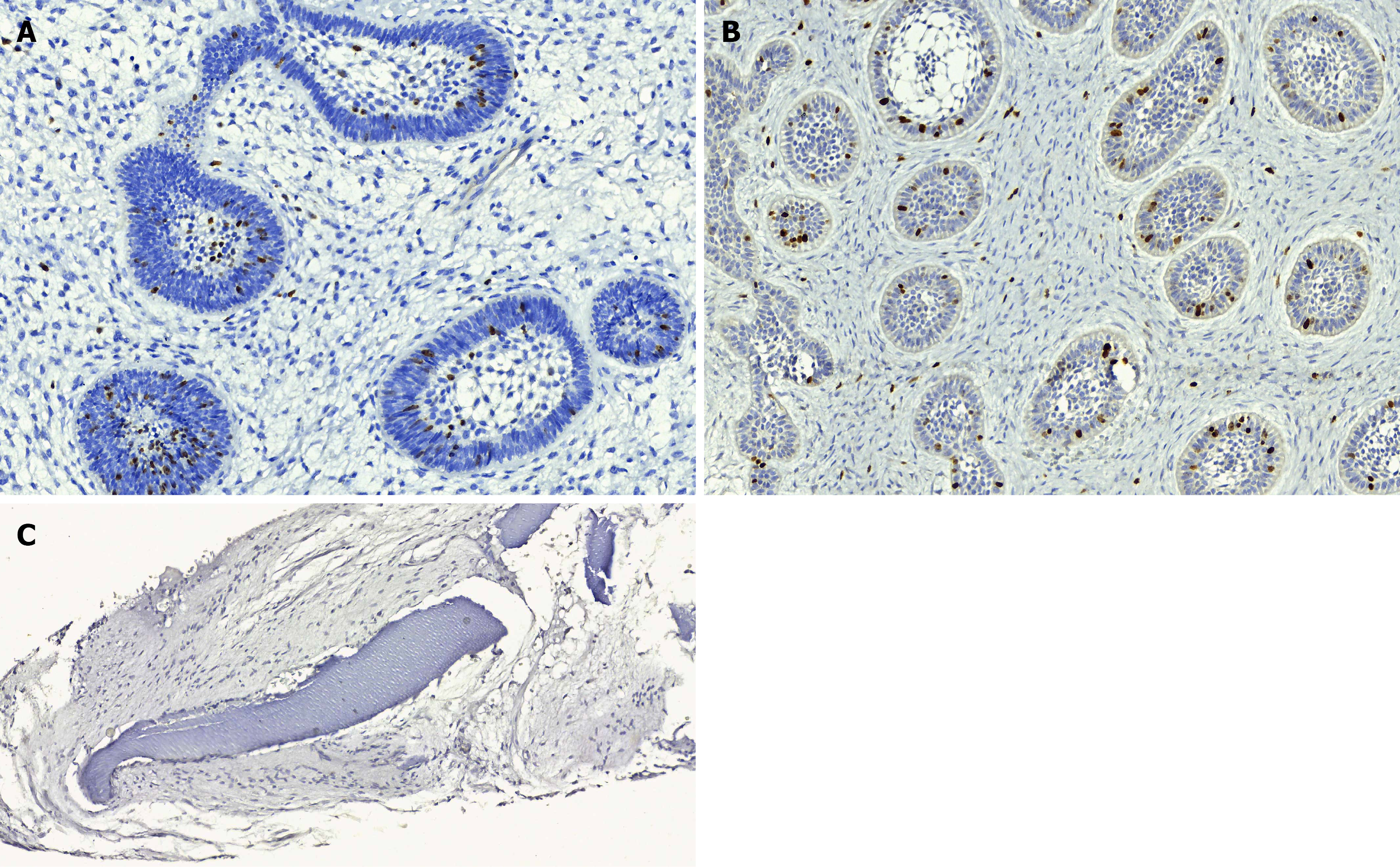
Ki-67 was positive in 3 AFO/AFD (50%) and 4 AF (50%) with a low degree of proliferation (0.5% to 5% of neoplastic cells) while the staining was negative in all O. Positivity was localized in the ameloblastic cells except in one AFO and one AF in which the positivity was mixed, showing both mesenchymal and odontogenic epithelial staining (Table 1, Figure 4).
Observed differences in expression depended on the sample type (P = 0.0020) and the patient’s age (P = 0.0100). Additionally, it was observed that with increasing age, the expression of the biomarkers decreased (odds ratio = 0.93) (Table 2), and a greater tendency to decrease expression with age was observed for MOC-31 and Cav-1 (P = 0.0770). The values in Table 3 correspond to the expression of the biomarkers in all samples (AFD/AFO, AF, O, and PC), considering variables such as sex, age, and location, highlighting differences in expression between AFD/AFO and O (P = 0.0250) and between AF and O (P = 0.0080). The expression of biomarkers in AFD/AFO resembled that in AF, whereas the immunohistochemical expression in O was lower than that in AFD/AFO and AF. Finally, the expression in PC was closer to that observed in O. For Cx-43 a difference in expression was found between O and AFD/AFO (P = 0.0022) and between O and AF (P = 0.0310). Similar trends were also observed for MOC-31 when comparing O with AF (P = 0.0310). In contrast, Cav-1 only showed differences in expression between PC and AFD/AFO (P = 0.0087).
AF, AFD, AFO, and O were classified by the WHO in 2005 as different entities, but starting with the 2017 classification, AFD and AFO began to be considered as entities that represent different stages of a developing odontoma[5,23]. In this study the immunohistochemical assays conducted revealed that the OT under investigation exhibited variable epithelial and mesenchymal expression for Cav-1, Cx-43, and MOC-31 with similar expression patterns among AFD, AFO, and AF as well as low proliferation rates, consistent with lesions of clinically benign behavior. Considering the expression of the biomarkers across different variables through a logistic regression model, a significant effect was found between the immunoexpression of the studied proteins and age with a trend towards lower expression with increasing age. This result was primarily observed for the expression of MOC-31 and Cav-1. These results are consistent with the pediatric nature of these tumors as well as the possible hamartomatous behavior of some cases as proposed by the WHO in 2017 that some of these lesions could exhibit a hamartomatous-like clinical behavior with a decrease in the expression of certain proteins as age progresses[5].
In this study Cav-1 was found to be expressed in most of the tumors, localized in both the epithelial and mesenchymal components with a preference for the stellate reticulum and ameloblast areas. No differences in the expression of this protein were observed in the studied samples; however, differences in expression were determined in the non-neoplastic control tissues (PC) when compared with AFD/AFO (P = 0.0087). This result is similar to that reported by Sánchez-Romero et al[22], who described the expression of Cav-1 in both benign and malignant OTs with lower expression in control gingival tissue that could be related to protumoral events. In some cancers Cav-1 expression is associated with increased cell migration, invasion, and metastasis. It can facilitate the reorganization of the cytoskeleton and enhance the motility of cancer cells[24,25]. Cav-1 can activate prosurvival signaling pathways, such as the phosphoinositide 3-kinase/protein kinase B (known as PI3K/AKT) pathway, which may support tumor growth and survival under stress conditions[26].
Furthermore, in this study a broad, variable expression of Cav-1 was found in the OT while its expression was absent in the non-neoplastic odontogenic control tissues. However, Shi et al[27] demonstrated that Cav-1 is expressed during the embryonic stage of mouse dental germs at both the epithelial and mesenchymal levels during the differentiation and organization stages of the tissues, primarily in the enamel organ.
A strong expression of MOC-31 was found in the odontogenic epithelium (ameloblasts and stellate reticulum areas) of AFD, AFO, and AF with negativity in O and PC. This event could be related to the importance of cellular cohesion during the development and growth of epithelial neoplasms. Ben Rejeb et al[28] observed that MOC-31 was variably expressed in both benign and malignant tumors in noninvasive neoplasms and thyroid carcinomas, supporting the idea that it could be a prognostic molecule. MOC-31 plays a multifaceted role in cancer, influencing cell adhesion, proliferation, invasion, immune evasion, and therapy resistance. Its expression and function make it a valuable biomarker and a potential target for therapeutic interventions in cancer treatment[29,30]. There is little information about the immunoexpression of this protein in OTs.
Cx-43 plays a dual role in cancer, functioning as both a tumor suppressor and promoter, depending on the cellular context, cancer type, and stage. Its role in cell communication, apoptosis, metastasis, and chemoresistance makes it a sig
In this study the expression of Cx-43 was observed in all AFD and AFO, suggesting a role for this protein in the induction of odontogenesis, facilitating the production of dentin matrix in AFD and enamel/dentin in AFO[7]. Based on the differential expression of Cx-43 in the studied OT and the similarity observed between AFD/AFO and AF, it could be suggested that AFD and AFO may be histological variants of AF that in their evolution could eventually initiate inductive processes leading to the formation of mineralized hard tissues, rather than developing as O as established in the last two classifications by the WHO[3,5].
MOC-31, caveolin, Ki-67, and Cx-43 are indirectly involved in the regulation of cell proliferation, adhesion, and intercellular communication, particularly in cancer contexts. Caveolin modulates the function of Cx-43 while Ep-CAM activates proliferative pathways associated with Ki-67. When coexpressed in tumors, these molecules may hold combined diagnostic and prognostic value.
We must remember that the main objective of this study was to determine the expression of several proteins in OT with an epithelial and mesenchymal component. The main limitation of our study was the small sample size. However, it should be noted that these are rare pathologies, and it is difficult to expand the samples. The results obtained could contribute to subsequent discussions that refer to the classification of these pathologies.
Another limitation of this study lies in the absence of complementary imaging data as it is a retrospective analysis based on archived samples from the Department of Pathology. Such information would have been useful for correlating histopathological findings with radiographic characteristics, particularly in OTs in which clinical and radiological fea
Due to the low frequency of OT, the statistical analysis performed was descriptive and based on the formulation of similarities in immunohistochemical expression and post-hoc multiple comparisons. This approach allowed the observation of expression similarities between AFD, AFO, and AF while also highlighting some differences in expression with O and PC (lower tissue immunostaining). These data are more evident when analyzing the expression of MOC-31 and Cx-43. Furthermore, the observed differences in protein expression between O and the other tissue types, coupled with the similarities in MOC-31 and Cx-43 expression among AFD, AFO, and AF, suggest that AFD and AFO share similar biological behavior and are more closely related to AF than to O. Additionally, the absence of MOC-31 and Cx-43 expression in O reinforces the distinction between these tissues and the others examined. On the other hand, in this study when analyzing the relationship between the immunoexpression of various proteins and patient age, a decline in expression with increasing age was observed. This finding underscores the pediatric prevalence of these Ots. This fact is fully justified by the age prevalence of these odontogenic neoplasms although some cases have also been reported in older adult patients[3,5,32].
However, the methodology used only describes the immunolocalization and presence of these proteins in the studied samples. This only allows us to hypothesize possible functions of these proteins within the tumor biology of these odontogenic lesions. Further investigation with more cases is needed to support our results.
AF, AFD, and AFO exhibit similar immunophenotypic profiles for MOC-31 and Cx-43 compared with O, suggesting biological proximity among these lesions at the protein-expression level. Given the small, unbalanced cohort and the absence of molecular assays, these data should be considered exploratory and hypothesis-generating. Confirmation in larger multicenter cohorts integrating molecular profiling and clinicoradiologic correlation is required before any taxonomic change is contemplated.
| 1. | Mosqueda-Taylor A, Ledesma-Montes C, Caballero-Sandoval S, Portilla-Robertson J, Ruíz-Godoy Rivera LM, Meneses-García A. Odontogenic tumors in Mexico: a collaborative retrospective study of 349 cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1997;84:672-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 158] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 2. | Bologna-Molina R, Mikami T, Pereira-Prado V, Pires FR, Carlos-Bregni R, Mosqueda-Taylor A. Primordial odontogenic tumor: An immunohistochemical profile. Med Oral Patol Oral Cir Bucal. 2017;22:e314-e323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 3. | Vered M, Wright JM. Update from the 5th Edition of the World Health Organization Classification of Head and Neck Tumors: Odontogenic and Maxillofacial Bone Tumours. Head Neck Pathol. 2022;16:63-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 289] [Cited by in RCA: 225] [Article Influence: 56.3] [Reference Citation Analysis (1)] |
| 4. | Siwach P, Joy T, Tupkari J, Thakur A. Controversies in Odontogenic Tumours: Review. Sultan Qaboos Univ Med J. 2017;17:e268-e276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 5. | Speight PM, Takata T. New tumour entities in the 4th edition of the World Health Organization Classification of Head and Neck tumours: odontogenic and maxillofacial bone tumours. Virchows Arch. 2018;472:331-339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 267] [Cited by in RCA: 194] [Article Influence: 24.3] [Reference Citation Analysis (1)] |
| 6. | Amm HM, MacDougall M. Molecular Signaling in Benign Odontogenic Neoplasia Pathogenesis. Curr Oral Health Rep. 2016;3:82-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 7. | Silveira FM, Molina-Frechero N, López-Verdín S, Umpiérrez NG, Palacio-Gastellum MG, Almeda-Ojeda O, González-González R, Martins MD, Sicco E, Pereira-Prado V, Bologna-Molina R. Connexin 43 expression in tooth germ and benign odontogenic tumors. Oral Surg Oral Med Oral Pathol Oral Radiol. 2023;135:661-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 8. | Balzar M, Winter MJ, de Boer CJ, Litvinov SV. The biology of the 17-1A antigen (Ep-CAM). J Mol Med (Berl). 1999;77:699-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 429] [Cited by in RCA: 402] [Article Influence: 14.9] [Reference Citation Analysis (1)] |
| 9. | Schwab W, Harada H, Goetz W, Nowicki M, Witt M, Kasper M, Barth K. Immunocytochemical and biochemical detection of EMMPRIN in the rat tooth germ: differentiation-dependent co-expression with MMPs and co-localization with caveolin-1 in membrane rafts of dental epithelial cells. Histochem Cell Biol. 2007;128:195-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 10. | Jaafari-Ashkavandi Z, Mehranmehr F, Roosta E. MCM3 and Ki67 proliferation markers in odontogenic cysts and ameloblastoma. J Oral Biol Craniofac Res. 2019;9:47-50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 11. | Sinyuk M, Mulkearns-Hubert EE, Reizes O, Lathia J. Cancer Connectors: Connexins, Gap Junctions, and Communication. Front Oncol. 2018;8:646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 12. | Lecanda F, Warlow PM, Sheikh S, Furlan F, Steinberg TH, Civitelli R. Connexin43 deficiency causes delayed ossification, craniofacial abnormalities, and osteoblast dysfunction. J Cell Biol. 2000;151:931-944. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 336] [Cited by in RCA: 306] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 13. | Al-Ansari S, Jalali R, Plotkin LI, Bronckers ALJJ, DenBesten P, Zhang Y, Raber-Durlacher JE, de Lange J, Rozema FR. The Importance of Connexin 43 in Enamel Development and Mineralization. Front Physiol. 2018;9:750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 14. | Cuylen S, Blaukopf C, Politi AZ, Müller-Reichert T, Neumann B, Poser I, Ellenberg J, Hyman AA, Gerlich DW. Ki-67 acts as a biological surfactant to disperse mitotic chromosomes. Nature. 2016;535:308-312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 500] [Cited by in RCA: 418] [Article Influence: 41.8] [Reference Citation Analysis (1)] |
| 15. | Gonzalez-Moles MA, Ruiz-Avila I, Gil-Montoya JA, Esteban F, Bravo M. Analysis of Ki-67 expression in oral squamous cell carcinoma: why Ki-67 is not a prognostic indicator. Oral Oncol. 2010;46:525-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 16. | Cortegoso AVB, Laureano NK, Silva ADD, Danilevicz CK, Magnusson AS, Visioli F, Rados PV. Cell proliferation markers at the invasive tumor front of oral squamous cell carcinoma: comparative analysis in relation to clinicopathological parameters of patients. J Appl Oral Sci. 2017;25:318-323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 17. | Langlois S, Cowan KN, Shao Q, Cowan BJ, Laird DW. Caveolin-1 and -2 interact with connexin43 and regulate gap junctional intercellular communication in keratinocytes. Mol Biol Cell. 2008;19:912-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 99] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 18. | Schubert AL, Schubert W, Spray DC, Lisanti MP. Connexin family members target to lipid raft domains and interact with caveolin-1. Biochemistry. 2002;41:5754-5764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 194] [Article Influence: 8.1] [Reference Citation Analysis (1)] |
| 19. | Kim BH, Kim CI, Park CH. Caveolin-1 and Ki-67 Expression as Prognostic Factors in Clear Cell Carcinoma of the Kidney. Korean J Urol. 2008;49:99-106. [DOI] [Full Text] |
| 20. | Krigers A, Moser P, Fritsch H, Demetz M, Kerschbaumer J, Brawanski KR, Thomé C, Freyschlag CF. The relationship between connexin-43 expression and Ki67 in non-glial central nervous system tumors. Int J Biol Markers. 2023;38:46-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 21. | Dias EP, Oliveira NSC, Serra-Campos AO, da Silva AKF, da Silva LE, Cunha KS. A novel evaluation method for Ki-67 immunostaining in paraffin-embedded tissues. Virchows Arch. 2021;479:121-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 22. | Sánchez-Romero C, Pereira-Prado V, Sicco E, Suarez M, Tapia-Repetto G, Carreón-Burciaga R, Gónzalez-Gónzalez R, Villarroel-Dorrego M, Meleti M, Molina-Frechero N, Bologna-Molina R. Expression of caveolin-1 in tooth germ, ameloblastoma and ameloblastic carcinoma. Med Oral Patol Oral Cir Bucal. 2021;26:e238-e245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 23. | Reichart PA, Philipsen HP, Sciubba JJ. The new classification of Head and Neck Tumours (WHO)--any changes? Oral Oncol. 2006;42:757-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 56] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 24. | Arpaia E, Blaser H, Quintela-Fandino M, Duncan G, Leong HS, Ablack A, Nambiar SC, Lind EF, Silvester J, Fleming CK, Rufini A, Tusche MW, Brüstle A, Ohashi PS, Lewis JD, Mak TW. The interaction between caveolin-1 and Rho-GTPases promotes metastasis by controlling the expression of alpha5-integrin and the activation of Src, Ras and Erk. Oncogene. 2012;31:884-896. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 25. | Yang H, Guan L, Li S, Jiang Y, Xiong N, Li L, Wu C, Zeng H, Liu Y. Mechanosensitive caveolin-1 activation-induced PI3K/Akt/mTOR signaling pathway promotes breast cancer motility, invadopodia formation and metastasis in vivo. Oncotarget. 2016;7:16227-16247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 122] [Cited by in RCA: 119] [Article Influence: 11.9] [Reference Citation Analysis (1)] |
| 26. | Matthews LC, Taggart MJ, Westwood M. Modulation of caveolin-1 expression can affect signalling through the phosphatidylinositol 3-kinase/Akt pathway and cellular proliferation in response to insulin-like growth factor I. Endocrinology. 2008;149:5199-5208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 27. | Shi L, Li L, Wang D, Li S, Chen Z, An Z. Spatiotemporal expression of caveolin-1 and EMMPRIN during mouse tooth development. J Mol Histol. 2016;47:337-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 28. | Ben Rejeb S, Beltaifa D, Ghozzi A, Bellil K, Turki S. EpCAM (MOC-31) - immunohistochemical expression in papillary thyroid carcinoma and non invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP). Tunis Med. 2021;99:1066-1071. [PubMed] |
| 29. | Maetzel D, Denzel S, Mack B, Canis M, Went P, Benk M, Kieu C, Papior P, Baeuerle PA, Munz M, Gires O. Nuclear signalling by tumour-associated antigen EpCAM. Nat Cell Biol. 2009;11:162-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 576] [Cited by in RCA: 553] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 30. | Chapel DB, Schulte JJ, Husain AN, Krausz T. Application of immunohistochemistry in diagnosis and management of malignant mesothelioma. Transl Lung Cancer Res. 2020;9:S3-S27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 111] [Article Influence: 18.5] [Reference Citation Analysis (1)] |
| 31. | Bonacquisti EE, Nguyen J. Connexin 43 (Cx43) in cancer: Implications for therapeutic approaches via gap junctions. Cancer Lett. 2019;442:439-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 85] [Article Influence: 12.1] [Reference Citation Analysis (1)] |
| 32. | Escobar E, Gómez-Valenzuela F, Peñafiel C, Ortega-Pinto A. Odontogenic tumours in a Chilean population: a retrospective study of 544 cases based on 2022 WHO classification. Med Oral Patol Oral Cir Bucal. 2023;28:e596-e606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |