Published online Apr 18, 2026. doi: 10.5312/wjo.v17.i4.116707
Revised: December 13, 2025
Accepted: January 26, 2026
Published online: April 18, 2026
Processing time: 143 Days and 13.2 Hours
Reconstruction of bone defects remains a major challenge in contemporary orthopedic practice and related reconstructive fields, including dental applications, particularly in cases involving trauma, surgical resections, implants, and prostheses. Regenerative medicine has evolved through the integration of biolo
Core Tip: Regenerative medicine combines biology, engineering, and technology to achieve predictable bone regeneration. While autogenous grafts remain the gold standard for their osteogenic properties, they pose limitations such as donor site morbidity and volume constraints. Alternatives like allogeneic, xenogeneic, and synthetic materials improve biocompatibility and tissue integration. Advanced biomaterials, including bioactive scaffolds, enhance extracellular matrix mimicry, supporting cell adhesion, vascularization, and mineralized tissue formation. Emerging strategies in tissue engineering, cell therapy, and photobiomodulation show promise for osteogenesis and repair. Integrating biological approaches with synthetic solutions offers a path toward individualized, effective, and evidence-based regenerative treatments in orthopedics.
- Citation: Horvath Pereira BO, Horvath-Pereira BO, Reis CHB, da Silva ELR, da Silva-Júnior LN, Buchaim RL, Buchaim DV. Biological approaches and biomaterial-based solutions for bone reconstruction: A comprehensive review. World J Orthop 2026; 17(4): 116707
- URL: https://www.wjgnet.com/2218-5836/full/v17/i4/116707.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i4.116707
Bone tissue is a specialized connective tissue characterized by a highly vascularized, dynamic, and complex structure that undergoes continuous remodeling throughout an organism’s life[1]. Upon injury, bone has a remarkable capacity for regeneration and repair, enabling structural recovery without scar formation, a feature uncommon among other tissue types[2]. However, in certain situations, particularly in large or complex defects, the regenerative process is insufficient to fully restore the original structure. In these cases, additional interventions may be required to stimulate complete bone formation and ensure the functionality of the affected area[3].
In clinical practice, bone loss may result from trauma, degenerative diseases, or other pathological conditions, manifesting as resorption defects with horizontal, vertical, or combined patterns[4]. Consequently, medical (especially orthopedics) and dental research have focused on identifying both natural and synthetic materials capable of effectively replacing lost soft and hard tissues[5]. In orthopedic and reconstructive surgery, bone tissue remains one of the most commonly used materials for interventions, especially in pre-prosthetic procedures, treatment of congenital defects, and correction of dentofacial deformities[6]. Bone grafting is essential in these contexts to promote fracture union where osteotomies have been performed and to prevent collapse of bone segments in iatrogenic defects[7]. This approach is critical for functional restoration and for aesthetic rehabilitation, both vital for patient well-being[8].
To address these challenges, tissue engineering has emerged as a modern, inherently multidisciplinary field that integrates biology and engineering[9]. Its primary goals include creating and improving artificial implants and developing laboratory-synthesized tissues, cells, and molecules to stimulate biological functions or effectively replace damaged body parts, promoting structural and functional restoration[10].
There are currently four main types of bone grafts available for reconstruction: Autogenous grafts harvested from the patient; allogeneic grafts from genetically distinct donors of the same species; xenogeneic grafts derived from other animal species; and synthetic alloplastic biomaterials produced in laboratories[11]. Among these, autogenous grafts are widely recognized as the gold standard for bone regeneration due to their osteoconductive and osteogenic properties, lack of cytotoxicity, and mechanical stability[12]. Nevertheless, their use is often limited by donor site morbidity, defect size, and restricted tissue availability, driving research toward alternative biomaterials that offer effective, less invasive solutions for bone repair[13].
In this context, bone regeneration frequently involves the use of barrier membranes to stabilize blood clots and/or bone substitute materials, while preventing epithelial cell invasion into the regenerating area[14]. These membranes also protect against microbial contamination and promote an environment conducive to angiogenesis and osteogenesis, facilitating bone healing[15]. They act as selective barriers, maintaining space for osteoprogenitor cell migration and proliferation, and enhancing vascularization by transporting oxygen and undifferentiated mesenchymal cells to the defect site, contributing to functional and aesthetic restoration[16].
Membranes are classified as resorbable or non-resorbable. Non-resorbable types include expanded and dense polytetrafluoroethylene, titanium, titanium-reinforced expanded polytetrafluoroethylene, and polypropylene[17]. Resorbable membranes are typically composed of natural polymers such as collagen or synthetic polymers like polylactic acid and polyglycolic acid, which exhibit controlled biodegradability and promote tissue regeneration[15].
Within tissue engineering, alternatives to autogenous bone grafts include xenogeneic biomaterials, such as bovine bone that requires deproteinization, and alloplastic materials including bioactive ceramics, biologically or synthetically derived polymers, and composites, all studied as potential substitutes in bone regeneration and tissue repair[18-20].
Biopolymers find broad applications in medicine, including controlled drug release systems, wound healing, sutures, and scaffolds for tissue engineering[15]. In surgical applications, they play an important role in alveolar bone regeneration, especially through absorbable collagen and gelatin sponges[21]. These biomaterials have gained significant interest in bone tissue engineering due to their ability to provide structural support and modulate cell growth in regenerating regions[22].
Despite many advances, bone regeneration remains a major challenge in contemporary medicine regenerative. Thus, biopolymers are increasingly investigated as promising alternatives for bone repair therapies. Among natural polymers, proteins like collagen, gelatin, and fibroin; polysaccharides such as cellulose, chitin, chitosan, alginate, and hyaluronic acid; and polymers like polynucleotides, polyisoprenes (natural rubber), and polyesters such as poly (β-hydroxybutyrate), poly (β-hydroxyvalerate), and poly (hydroxybutyrate-co-valerate) stand out due to their structural and biocompatible properties[23,24].
In recent years, the use of biotechnology has grown significantly, driven by increased severe injuries from accidents, aging populations, age-associated diseases, and intense research efforts[25]. The global biomaterials market continues to expand annually, reflecting their broad applicability across healthcare. It is estimated that between 200000 devices and 700000 devices, including hip, knee, and shoulder prostheses, implants, and fixation plates, are regularly used, demonstrating their relevance in modern medicine[26].
Recent advances in bone regeneration have expanded the therapeutic arsenal available for orthopedic and reconstructive procedures[27]. Biomaterials have evolved from passive structural fillers to highly engineered scaffolds capable of modulating cellular behavior, controlling degradation rates, and enhancing vascularization. In parallel, biological therapies such as photobiomodulation, growth factors, and cell-based approaches have demonstrated promising results in accelerating bone healing and improving tissue quality. These developments highlight a growing trend toward more biologically responsive and clinically adaptable strategies[28,29].
Despite these advances, current literature remains fragmented, with studies often focusing exclusively on either biomaterial-based solutions or biological stimulation modalities[30]. There is a lack of comprehensive analyses that summarize the main types of biomaterials, their mechanisms of action, and their clinical performance alongside the biological strategies that may complement or enhance regeneration[31]. This fragmentation limits the ability of clinicians to make evidence-based decisions when selecting the most appropriate therapeutic approach[32].
Given the expansion of biomaterial applications, particularly in medical and dental fields, the demand for new materials and technological advances remains constant. The diversity of existing biomaterials, alongside emerging technologies and applications, requires frequent literature reviews to assist professionals in selecting the most appropriate materials according to technique and anatomical region[33].
Considering these advances and the gaps in published studies, a broader and more structured overview of current regenerative strategies is essential. A detailed synthesis of biomaterials, biological therapies, and emerging tissue engineering technologies can better support clinical decision-making and contribute to a more integrated understanding of bone reconstruction modalities.
Therefore, this article aims to provide a comprehensive overview of the main therapeutic strategies used in bone reconstruction within orthopedics and regenerative medicine, including biomaterials, tissue engineering approaches, biostimulatory therapies, and stem cells, summarizing their properties, clinical applications, limitations, and future perspectives.
Bone tissue is one of the most resistant structures in the human body, characterized by its high stiffness combined with a certain degree of elasticity. This property enables bones to support and dissipate mechanical forces, including tension and compression, ensuring its strength and functionality in structural support and protection. Furthermore, as a living tissue, bone comprises various cell types that play essential roles in its formation, remodeling, and repair[34].
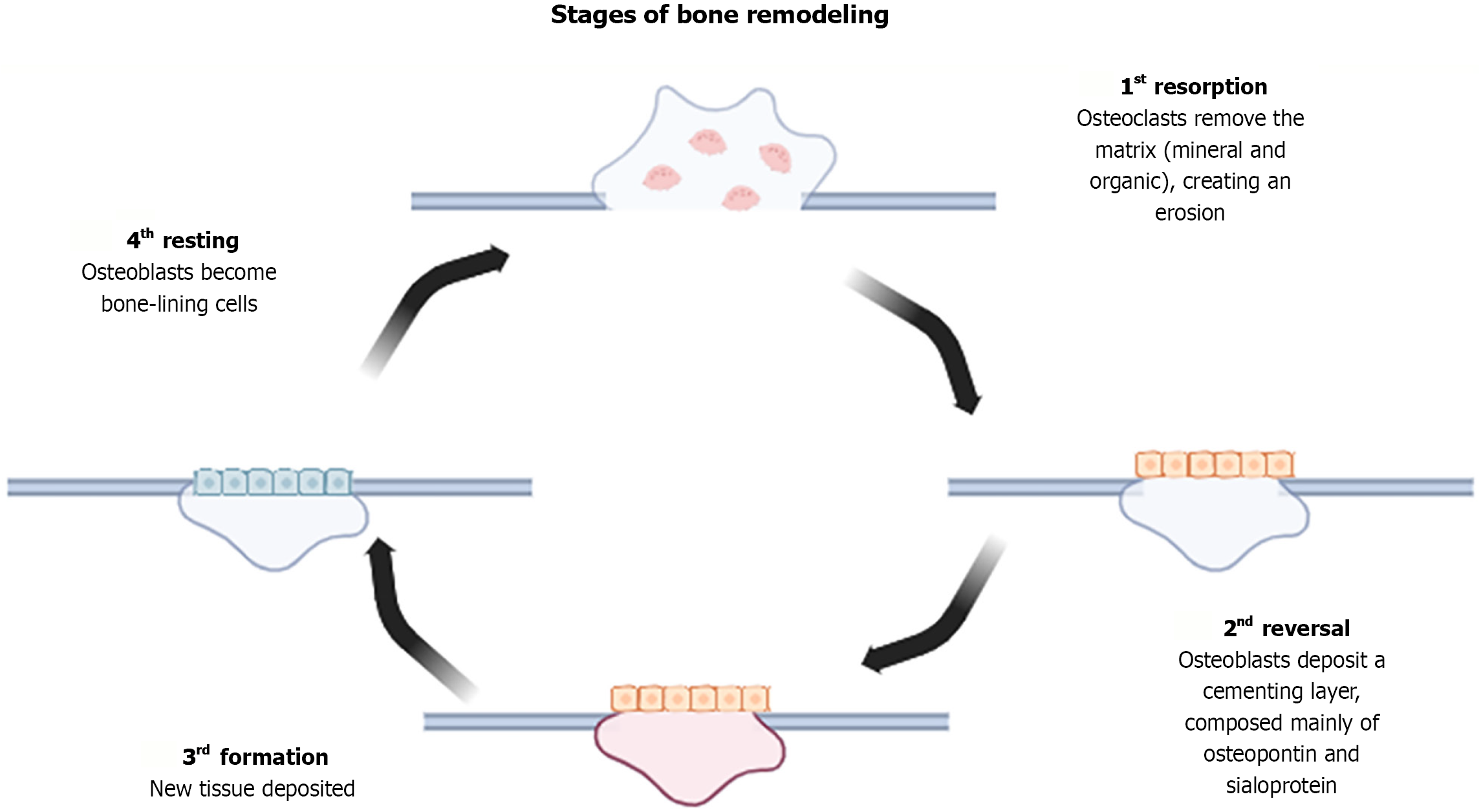
Among these cells, osteoblasts, osteoclasts, osteocytes, and osteoprogenitor cells are particularly noteworthy. Osteoblasts synthesize and deposit the bone matrix, playing a direct role in bone formation and regeneration. In contrast, osteoclasts are multinucleated cells primarily involved in matrix resorption and are crucial for bone remodeling. Osteocytes arise from the differentiation of osteoblasts once they become embedded in the newly formed bone matrix, establishing an interconnected cellular network via cytoplasmic extensions[35]. Osteoprogenitor cells serve as a cellular reservoir and are activated under conditions of tissue injury. When stimulated, they differentiate into osteoblasts and contribute to the deposition of new bone matrix in areas requiring repair[36].
The continuous process of bone tissue formation and resorption is known as bone remodeling. This phenomenon is essential for enabling skeletal growth, microfracture repair, tooth movement, and the adaptation of bone to new physiological demands (Figure 1)[37].
However, despite the significant regenerative capacity of bone tissue, in cases of extensive injuries or critical-size defects, the body is unable to promote spontaneous and effective regeneration. Faced with this limitation, the use of biomaterials becomes essential as temporary bone substitutes, providing structural support and favoring the tissue repair process[38].
Thus, bone neoformation is a biological process resulting from osteoblast activity that depends on several facilitating mechanisms. These mechanisms are directly related to the clinical conditions of the recipient site, type of grafted material, the proper understanding of clinical indications, precision of the surgical technique employed, and the origin of the grafting material[39].
Bone substitutes can be categorized according to their origin, biological properties, and mechanisms of action in the bone regeneration process. These mechanisms include osteoconduction, which provides a three-dimensional matrix for bone growth by supporting the migration of osteoprogenitor cells from the adjacent bone; osteoinduction, which stimulates and recruits undifferentiated mesenchymal cells, inducing their differentiation into osteoblasts and promoting bone tissue formation; and osteogenesis, which refers to the ability of certain biological materials to directly promote bone formation due to the presence of viable osteoblasts in the graft[40,41].
Similarly, biomaterials used in bone regeneration can be classified according to their origin and biological properties, as well as their mechanisms of action in the tissue repair process. Among the main mechanisms are: Osteogenesis, characterized by the ability of certain biological materials to promote direct bone tissue formation through viable osteoblasts present in the graft[42,43]; osteoinduction, which refers to the ability to stimulate the migration and differentiation of undifferentiated mesenchymal cells into osteoblasts or chondroblasts, favoring bone neoformation even in heterotopic sites; and osteoconduction, a property associated with inorganic grafts that provides a three-dimensional matrix for the deposition of new bone tissue, serving as a scaffold for the osteoprogenitor cell proliferation and migration from adjacent bone, promoting bone growth at the recipient site[44,45].
Biomaterials can also be classified based on their biological characteristics as: Biotolerant, which do not promote true osseointegration, resulting in the formation of fibrous connective tissue between the implant and bone tissue; bioinert, which allow bone formation in direct contact with the implant without triggering significant biological reactions between the implanted material and the bone bed; and bioactive, which induce physicochemical interactions between the implant and surrounding bone tissue. Regarding their physical characteristics, biomaterials can be classified as organic, inorganic or mineralized, demineralized, or fresh[46].
Therefore, to understand the use of biomaterials in bone reconstruction, it is essential to understand bone regeneration physiology, which depends on an adequate blood supply from the initial inflammatory response to the remodeling and bone resorption phases[47]. Moreover, biomaterials should promote mechanical stability and be compatible with the dimensions of the bone defect. The biological and physiological processes involved are influenced by both local and systemic factors. Thus, in addition to the aforementioned characteristics, these materials must allow the conduction of osteoblasts or osteoblastic precursor cells to the injured site, presenting osteoconductive and osteoinductive properties, supporting the periosteum, accelerating bone remodeling, and stimulating growth factor release[48].
Bone regeneration follows a well-organized sequence, beginning with an inflammatory phase marked by immune cell recruitment and the release of cytokines that initiate the repair process[49]. This is followed by granulation tissue formation and subsequent osteoid deposition by osteoblasts, which progressively mineralizes. Finally, the remodeling phase restores the structural arrangement of bone through the coordinated actions of osteoclasts and osteoblasts[50]. These sequential mechanisms demonstrate the dynamic and biologically demanding nature of bone repair[51].
Because each stage of bone healing depends on specific cellular and molecular events, the biological requirements of the regenerating tissue directly influence biomaterial design and selection[52]. For example, early angiogenesis requires a biomaterial with an interconnected porous architecture, while osteoblast activity is improved by surfaces with adequate bioactivity and protein adsorption capacity. These biological needs help determine the structural and chemical characteristics desired in bone substitutes[53,54].
Thus, understanding the mechanisms governing bone biology is essential for selecting appropriate bone substitutes[55]. Biomaterials must ensure mechanical stability during the early inflammatory phase, maintain space for tissue ingrowth, support osteoconduction during osteoid deposition, and undergo degradation at a rate compatible with new bone formation[56,57]. Therefore, properties such as porosity, chemical composition, collagen content, crystallinity, and resorption kinetics are designed to mimic or complement physiological processes, ensuring more predictable regenerative outcomes[58,59].
Autogenous grafts (autografts): Autografts are obtained from the patient themselves and are widely used in regenerative therapies due to their essential biological characteristics, particularly the high biocompatibility between donor and recipient sites[60]. The main advantages of autografts include rapid osteoconduction, the ability to transplant viable cells, osteogenic potential, and rapid revascularization, which significantly reduces the risk of transmitting infectious diseases. However, this therapeutic approach presents some limitations, such as postoperative discomfort, a more complex surgical procedure, the risk of aesthetic defects at the donor site, the possibility of paresthesia, and the tendency for partial graft resorption[61].
In reconstructive surgery, donor site selection for autogenous bone grafts depends on defect size and location. In the craniofacial region, intraoral sources are commonly used due to their anatomical accessibility and lower morbidity. The most commonly used intraoral donor sites include the mandibular symphysis, the mandibular ramus, and the maxillary tuberosity, which are frequently employed in clinical cases of reconstructive bone surgery[62]. For extensive bone defect reconstruction, extraoral regions such as the calvarium, iliac crest, rib, and tibia are widely used[63]. According to Reininger et al[64], bone fragments from the mandibular symphysis show superior reconstruction of alveolar defects. However, this area presents higher complication rates, with paresthesia from mental nerve injury being the most frequently reported complication.
Some authors, such as Urban and Monje[65], emphasize that autogenous bone is the only material that has all of the ideal properties for bone regeneration, fulfilling the three main mechanisms of this process: Osteogenesis, osteoconduction, and osteoinduction. The effectiveness of autogenous grafts in interacting with the recipient bed is attributed to the presence of osteoprogenitor cells in the transplanted bone fragment[66]. However, as discussed by Soni et al[67], the need for an additional surgical procedure to obtain the graft and its limited availability have restricted its use, encouraging the search for alternatives such as xenografts, allografts, and alloplasts[65].
Homologous grafts (allografts): Homografts have emerged as a promising alternative to autografts, as they eliminate the need for a donor site and exhibit both osteoconductive and osteoinductive properties[68]. These grafts are often indicated for atrophic maxillae reconstruction and are obtained from individuals of the same species, consisting of cortical bone, cancellous bone, or a combination of both. Additionally, they can be pre-shaped and subjected to various preservation processes such as lyophilization, demineralization, or freezing. The main advantage of this type of graft is the elimination of multiple surgical sites, thereby reducing surgical trauma to the patient. However, the limitations include dependence on tissue banks or donors, as well as the risk of transmission of infectious agents or immunological reactions, especially in fresh grafts[69]. According to Ferraz[11], although the decellularization process effectively reduces the immune response in allografts, it may also impair bone regeneration, making it slower and more challenging.
There is still no consensus in the literature regarding the ideal bone substitute, primarily due to the scarcity of controlled clinical trials[70]. However, both autogenous and homologous grafts show good survival and success rates in osseointegration protocols. In turn, allogeneic grafts have been widely used in combination with bone marrow aspirates through the bone marrow aspirate concentrate method, demonstrating favorable results[69]. A study conducted by Lavareda Corrêa et al[71] compared grafts with and without bone marrow aspirate and demonstrated that bone marrow-associated grafts showed better outcomes, particularly in terms of bone density and bone formation patterns.
Xenogeneic grafts (heterologous): Xenogeneic bone substitutes are derived from individuals of other species and have been widely used due to their high osteoconductive capacity. They are frequently reported in the literature within bone augmentation and reconstructive protocols[72]. The most commonly cited xenograft is deproteinized bovine bone mineral due to its favorable properties, including a composition and porosity similar to human bone, its prolonged residence time at the graft site, and its potential for association with biomaterials such as fibrin. However, a comparative study by Irdem et al[73] concluded that there are no significant advantages in using this material alone or in combination with other bone substitutes.
The bovine bone xenograft offers the advantage of minimizing inflammatory and immunological responses in patients due to deproteinized inorganic matrix. Its main function is to promote host bone cell proliferation, thus favoring new bone formation[74]. This material has been used in tooth cell transplantation, which involves the combination of the xenograft with bone marrow aspirate concentrate, the latter being a potent enhancer of bone regeneration[75].
Thus, heterologous grafts derived from lyophilized bovine bone are among the most commonly used in reconstructive bone procedures. Their production involves a physical-chemical sterilization process aimed at removing proteins in the bone tissue, resulting in the elimination of all organic components[76]. As a residue, hydroxyapatite crystals remain, playing a fundamental role in osteoblast adhesion and subsequent bone matrix deposition. This biomaterial, characterized by its mineralized and porous structure, is available in various formats, including blocks and granules, and may consist of either cortical or cancellous bone[77].
Despite their widespread use, bovine-derived heterologous grafts present some limitations, including the risk of infection and immunological rejection. Moreover, there is the possibility of prion transmission, infectious agents associated with the development of Bovine Spongiform Encephalopathy, commonly known as “mad cow disease”[78]. Another relevant drawback is the potential induction of an immune response with antibody production, which may compromise graft integration with the host tissue[79].
Synthetic grafts (alloplastics): Alloplastic biomaterials, synthetically produced in laboratories from hydroxyapatite and beta-tricalcium phosphate (β-TCP), are widely recognized for their quality and versatility as bone substitutes. Although they exhibit limited or no osteoinductive activity, their low resorption rates provide a stable and long-lasting environment, favoring the time required for new bone formation by the host tissue[80]. These biomaterials are frequently used in combination with other bone grafts due to their unlimited availability and lack of risk of transmitting infectious diseases, characteristics that make them highly advantageous[81,82]. Furthermore, studies by Liu et al[83] highlight that synthetic bone substitutes can satisfactorily repair bone defects, including applications in periodontal infrabony defects.
Hydroxyapatite is a natural bone mineral that can also be synthetically produced in the laboratory. This biomaterial is commercially available in various forms and is notable for its osteoconductive capacity. β-TCP has a phosphate and calcium composition similar to that of human bone tissue; however, it lacks osteogenic properties[84].
Additional studies have shown that the combination of hydroxyapatite and β-TCP in different proportions can influence resorption rate and bone formation, with combinations with higher β-TCP content tending to present greater biomaterial absorption and faster bone formation during healing. These characteristics make hydroxyapatite and β-TCP-based alloplastic biomaterials promising options for various clinical applications in the field of bone regeneration[85].
Synthetic membranes: These biomaterials exhibit significant clinical acceptance and are frequently used in combination with autogenous, xenogeneic, or allogeneic bone grafts. Therefore, resorbable and non-resorbable membranes are widely indicated in guided bone regeneration (GBR) protocols[86]. These membranes act as selective barriers, preventing the migration of epithelial cells and connective tissue into the bone defect, thereby allowing osteogenic cell proliferation and vascularization at the site. This favors mineralized matrix deposition in the area of new bone formation[87].
In other words, the GBR technique optimizes bone graft integration by promoting its compaction and protection within the recipient site. This process creates a favorable environment for localized osteogenic activity, enhancing the formation of new bone tissue[88]. As a result, the membranes most commonly used in GBR protocols are manufactured from materials such as polytetrafluoroethylene, expanded polytetrafluoroethylene, collagen, polylactic acid, polyglactin 910, polyglycolic acid, calcium sulfate, microtitanium mesh, and polyurethane. Among these, expanded polytetrafluoroethylene is widely used due to its high resistance to degradation and low immunogenicity[89].
Resorbable collagen-based membranes have gained popularity among dental professionals because they eliminate the need for a second surgical procedure for removal. Moreover, they present low immunogenicity, adequate cellular adhesion, and effective hemostatic capacity features that support their use as barriers in GBR protocols[90]. These biomaterials significantly contribute to surgical site healing and bone tissue development during the early repair stages, being widely recognized for their proven biocompatibility[91].
The mechanical malleability of these membranes facilitates their adaptation to bone defects and handling during the clinical procedure. Additionally, their properties include semipermeability, allowing the diffusion of essential nutrients for repair, and enzymatic degradation, which enables fibroblast recruitment to the inflammatory site. This process is mediated by collagen fiber cleavage by collagenases, whose degradation products create a microenvironment favorable to the action of nonspecific proteases. These promote fibroblast migration and support tissue regeneration[92].
Platelet-rich fibrin and platelet-rich plasma: Fibrin is a viscoelastic polymer whose mechanical and structural properties influence hemostasis and the development of thrombotic complications[93]. Its polymerization occurs through sequential reactions that form a porous three-dimensional network, whose structure determines clot resistance to blood flow, platelet contraction, and other dynamic forces[94]. This temporary matrix allows for cellular proliferation and organization, promoting tissue regeneration in injured or inflamed areas. Therefore, fibrin is widely applied in surgical procedures, including orthopedics, neurology, plastic surgery, periodontics, implant dentistry, and oral and maxillofacial surgery[93].
Platelet-rich fibrin (PRF) is derived from the patient’s own blood and is classified as an autogenous source, which significantly reduces the risk of immune rejection or allergic reactions and accelerates the healing process[95]. It is widely used in reconstructive surgery and GBR procedures, especially in cases of maxillary atrophy and sinus floor elevation due to its ability to promote soft tissue healing and stimulate the formation of newly formed bone tissue[96]. It is also employed in procedures aimed at post-extraction socket preservation, bone regeneration after cyst enucleation, sinus floor elevation, alveolar ridge maintenance, regenerative therapy in dental implants, and in the repair of intraosseous defects, furcation lesions, and periodontal plastic surgery interventions[97].
The PRF technique induces cell proliferation through growth factors in the plasma, which synergistically interact with the substitute biomaterial used in the procedure[98]. In addition to its favorable biological properties due to its autologous origin, PRF is widely available and low-cost, making it a viable option for patients[99].
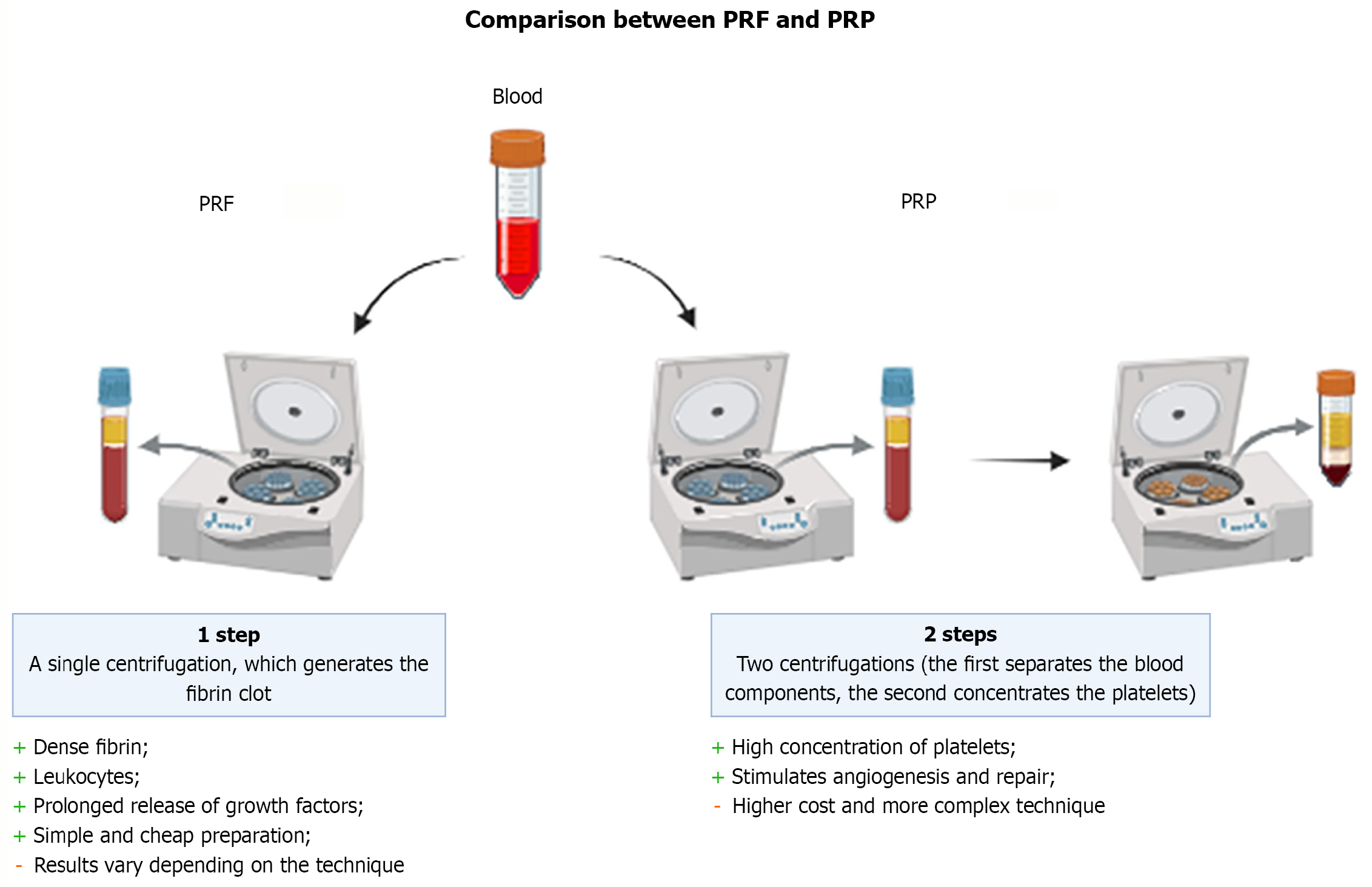
However, despite the benefits of PRF in dentistry, some limitations must be considered. Its quality can vary due to differences in handling methods, preparation techniques, and professional expertise, which may influence its efficacy and clinical outcomes[100]. Moreover, not all dental procedures show the same benefits with PRF use, and in some cases, it must be combined with other biomaterials to enhance therapeutic effects. Finally, although PRF has gained popularity, there is still limited controlled and randomized clinical trials that confirm its effectiveness in specific dental applications (Figure 2)[95].
PRF in tissue regeneration should be highlighted. PRF contains higher concentrations of fibrin and leukocytes than platelet-rich plasma (PRP). Both enhance healing through the release of growth factors; however, PRF sustains this release over a prolonged period, offering long-lasting benefits and promoting faster tissue repair[101].
PRP is characterized by a high concentration of autologous platelets in a reduced volume of the patient’s plasma. It is obtained by collecting the patient’s blood, followed by a first centrifugation to isolate platelets without compromising their integrity[102]. A second centrifugation then separates platelets and white blood cells from the plasma. This two-step process allows for a higher platelet concentration from autologous blood[103].
Platelet granules release various growth factors that stimulate cell proliferation and differentiation, processes essential for osteogenesis. In addition to its pro-coagulant function, PRP plays a role in the initiation and maintenance of tissue repair, accelerating healing and improving its efficiency[104]. Nevertheless, PRP use requires the addition of external chemical agents, significantly increasing costs and complicating the handling process[105].
Given these considerations, PRF has emerged as a desirable option in tissue regeneration. In contrast to PRP, PRF contains higher concentrations of fibrin and leukocytes and has a simplified preparation technique. Although both promote healing through growth factor release, PRF promotes a more prolonged release, delivering long-term benefits and facilitating faster repair[101].
Low-level laser therapy: Laser technology (“Light Amplification by the Stimulated Emission of Radiation”) was introduced into dentistry in the 1960s and has since been widely used across various specialties. Its application encompasses both hard and soft tissues, proving effective in procedures such as caries prevention, treatment of dentin hypersensitivity, healing of herpetic lesions, removal of hyperplastic tissues, hemostasis, and frenectomies[106].
Photobiomodulation therapy, formerly known as low-level laser therapy (LLLT), is a non-thermal process based on the interaction of light with endogenous chromophores. This interaction triggers photophysical and photochemical events at cellular and molecular levels, promoting therapeutic effects such as analgesia, anti-inflammatory action, immune modulation, and stimulation of tissue regeneration[107].
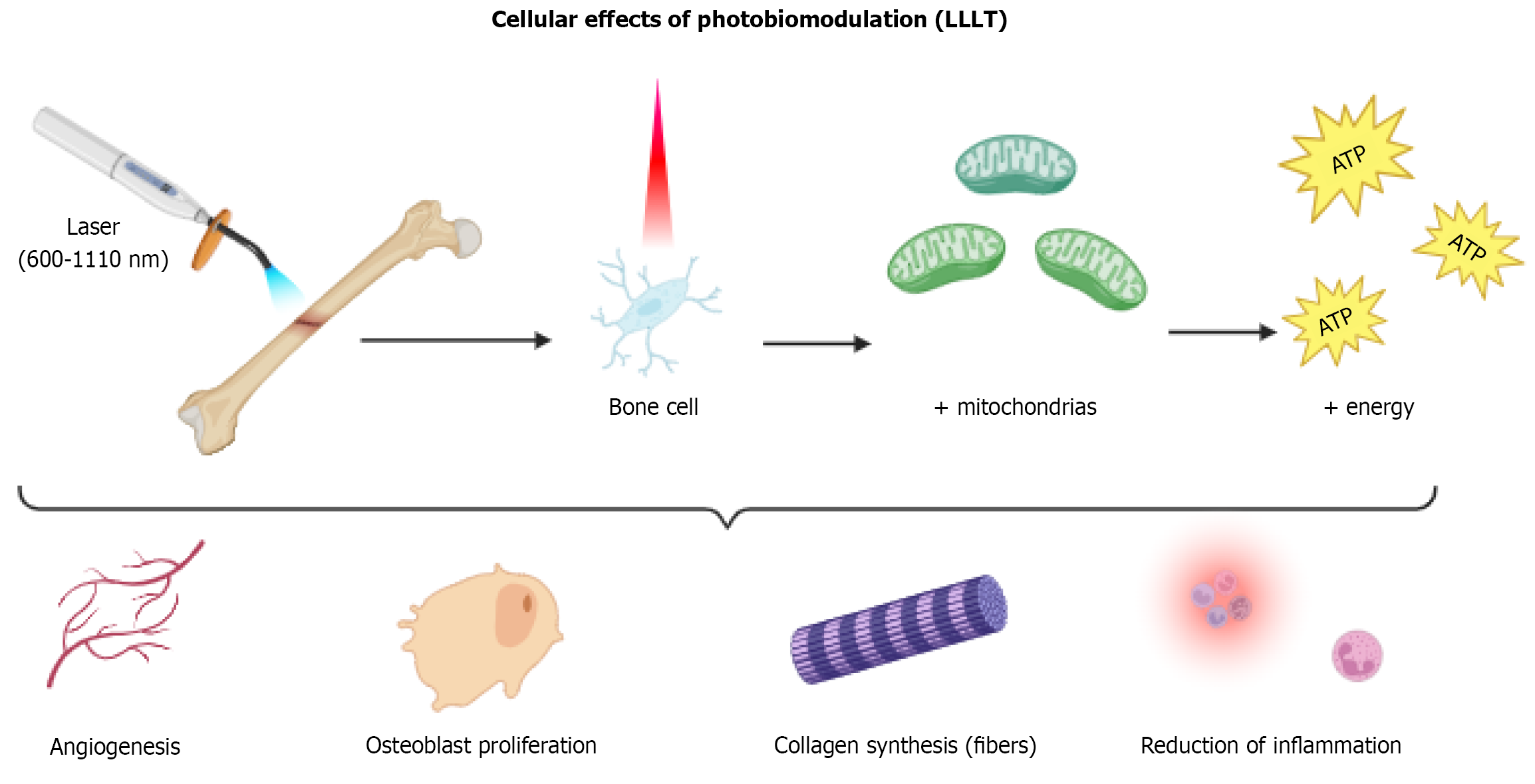
Thus, it is a therapeutic modality that uses non-ionizing light sources within the visible and infrared spectra (600-1110 nm). When absorbed by the irradiated tissue, this light induces biochemical changes that result in growth factor production and increased cellular proliferation. Among the most relevant effects are stimulation of collagen synthesis, increased fibroblast and osteoblast activity, and angiogenesis, all essential for bone regeneration (Figure 3)[108].
These biological effects make photobiomodulation a promising avenue in regenerative medicine. It has been applied as an adjunct therapy in various clinical conditions, including medication-related osteonecrosis of the jaws, antimicrobial therapies, management of acute and chronic pain, and acceleration of orthodontic tooth movement[109]. In areas of fracture, bone defects, or post-extraction sites, laser therapy stimulates osteoblast recruitment and maturation along bone margins, enhances type I collagen expression, the predominant protein in the bone matrix, and promotes neovascularization essential to repair[110].
Recent studies also highlight the potential of photobiomodulation as an adjuvant therapy to biomaterials and scaffolds, promoting greater integration and acceleration of bone regeneration[111].
The effectiveness of photobiomodulation directly depends on variables such as wavelength, energy density, exposure time, and application frequency. Standardization of these parameters is essential to ensure reproducibility and consistent clinical outcomes, representing a current challenge in translating the therapy to broader clinical protocols[112].
Therefore, photobiomodulation with LLLT consolidates itself as a complementary therapeutic strategy with significant potential, substantially contributing to the modulation of the inflammatory response, acceleration of bone repair, and improvement of clinical outcomes in dental procedures[113].
Cell therapy - use of stem cells and advanced scaffolds in bone reconstruction: Tissue engineering represents one of the most innovative and promising approaches in the field of bone regeneration, especially in light of the limitations posed by conventional grafts. This multidisciplinary area aims to achieve the functional reconstruction of tissues through the combination of three essential elements: Regenerative cells, biomaterials (scaffolds), and biochemical or physical stimuli that promote regeneration[114].
Tissue engineering is defined as an interdisciplinary field that integrates biomaterials science, cell biology, and biochemical signaling to create functional tissue substitutes capable of restoring, maintaining, or improving damaged structures[115]. In the context of bone repair, tissue engineering relies on the synergistic interaction among three key components: Stem cells with osteogenic potential, three-dimensional scaffolds that provide structural support and guide tissue formation, and bioactive molecules that regulate cell behavior and promote osteogenesis. This triad forms the foundation of modern regenerative strategies, enabling controlled and predictable bone regeneration in critical-size defects[116,117].
Mesenchymal stem cells (MSCs), obtained from various sources such as bone marrow, adipose tissue, dental pulp, and periosteum, have been extensively studied due to their ability to differentiate into osteoblastic lineages and their role in modulating the inflammatory microenvironment through the release of trophic factors[118]. When associated with biocompatible and bioactive three-dimensional scaffolds, these cells can enhance bone formation in terms of quality and quantity, optimizing the process of neoformation in critical-size defects[119].
Scaffolds used in tissue engineering must exhibit appropriate physicochemical characteristics, such as controlled porosity, mechanical strength compatible with bone tissue, and degradation synchronized with new bone formation. Current advanced biomaterials that mimic the native extracellular matrix have been developed and can be functionalized with signaling molecules, osteoinductive peptides, and even nanoparticles that promote osteogenesis[120]. 3D printing of these scaffolds has enabled the fabrication of customized structures tailored to the morphofunctional needs of each bone defect[121].
Recent advances in tissue engineering have focused on optimizing scaffold–cell interactions and enhancing the biological functionality of implanted constructs. Bioengineered scaffolds incorporating growth factors, nano-structured surfaces, or controlled-release systems have demonstrated improved osteoinduction and vascularization[122]. Three-dimensional bioprinting technologies have also emerged as powerful tools, allowing the fabrication of patient-specific scaffolds with precise architecture and mechanical properties[123]. Additionally, the use of MSC-derived exosomes has gained attention as a cell-free therapeutic strategy, offering comparable osteogenic benefits with lower immunogenic risk. Together, these advances represent significant progress in the development of clinically applicable bone regeneration platforms[124,125].
Despite their therapeutic potential, the clinical use of stem cells and bioengineered scaffolds still faces significant challenges, such as standardization of cell isolation and expansion techniques, cell viability after transplantation, associated costs, and regulatory requirements for human application. Nevertheless, preclinical studies and early clinical trials have shown promising results, indicating the feasibility of incorporating these therapies into future bone regeneration protocols[126].
The clinical application of combined stem-cell and scaffold-based therapies has shown promising results in treating critical bone defects[127]. A recent systematic review (2024) reported that among 138 patients receiving MSC-loaded scaffolds for bone defects, all achieved bone healing with favorable safety and functional outcomes[126]. These findings demonstrate the practical effectiveness of integrating stem cells with osteoconductive scaffolds in promoting predictable bone regeneration in complex defects[128].
Thus, cell therapy and tissue engineering stand out as highly innovative and integrative strategies with the potential to transform the current approach to bone reconstruction in dentistry, offering personalized, less invasive solutions with greater predictability of clinical success[129].
Functionalized acellular periosteum for endogenous stem-cell recruitment: The periosteum plays a central role in natural bone regeneration due to its reservoir of osteoprogenitor cells and its ability to coordinate osteogenic activity[130]. Functionalized acellular periosteum has recently emerged as an innovative biomaterial capable of replicating key aspects of the native periosteal microenvironment[131].
By incorporating bioactive cues, these constructs promote endogenous stem-cell homing to the defect area, stimulate their differentiation into osteoblastic lineages, and enhance the intrinsic regenerative capacity of bone tissue[132]. Experimental models of critical-size defects demonstrate that this strategy supports robust matrix deposition, accelerates osteogenesis, and improves structural repair compared to conventional grafting alone[133,134]. Such findings reinforce the potential of engineered periosteal substitutes to serve as biologically instructive scaffolds that synergize natural healing mechanisms with advanced tissue-engineering approaches[135].
Hyaluronic acid–based multi-responsive hydrogels in infectious bone defects: Recent developments in hydrogel technology have enabled the creation of multifunctional materials designed to simultaneously combat infection and promote bone regeneration[136,137]. Hyaluronic acid-based hydrogel microspheres with multi-responsive properties have shown significant potential in treating Staphylococcus aureus-infected cranial defects[138].
These systems demonstrate the capacity to inhibit bacterial proliferation while supporting osteoblast adhesion, proliferation, and mineralization at the defect site, thereby providing a dual therapeutic effect that integrates antimicrobial action with osteogenic stimulation[139]. Their ability to adapt to local microenvironmental changes and deliver coordinated biological responses highlights their promise as advanced biomaterials for managing infectious bone defects and improving clinical outcomes in regenerative therapies[140-145].
To provide a clear overview of the main categories of bone graft materials, a we developed a comparative table summarizing their fundamental biological properties, advantages, limitations, and typical clinical applications. This comparison facilitates the understanding of how each graft option differs in behavior and clinical performance, supporting more informed decision-making in orthopedic and regenerative procedures[141-151] (Table 1).
| Ref. | Type of graft | Main properties | Advantages | Limitations | Typical clinical applications |
| [142,143] | Autograft | Osteogenic, osteoinductive, osteoconductive; no immune response | Gold standard; high predictability; rapid integration | Donor-site morbidity; limited volume | Large defects, non-unions, orthopedic reconstruction, maxillofacial surgery |
| [144,145] | Allograft | Mainly osteoconductive; may retain osteoinductive potential depending on processing | No second surgical site; wide availability | Possible immune reaction; slower integration; disease transmission risk | Orthopedic revision surgery, spinal fusion, large defects |
| [146,147] | Xenograft | Highly osteoconductive; stable porous structure; slow resorption | Excellent volume maintenance; good scaffold for bone growth | Very slow remodeling; no osteogenic potential | Alveolar ridge preservation, sinus lift, moderate orthopedic defects |
| [148,149] | Synthetic (HA, β-TCP, bioceramics) | Controlled porosity; adjustable resorption; bioinert or bioactive depending on composition | No risk of disease transmission; unlimited availability; tunable properties | Lower mechanical strength; variable integration depending on formulation | Filling small to medium defects, orthopedic revisions, periodontal/trauma defects |
| [150,151] | Composite biomaterials | Combine organic and inorganic components; mechanical reinforcement; enhanced bioactivity | Balanced resorption; improved handling; increased mechanical stability | Higher cost; variable performance depending on formulation | Load-bearing defects, trauma reconstruction, implant dentistry |
The use of biomaterials for bone regeneration has proven essential in the rehabilitation of bone defects across various fields of orthopedic and reconstructive surgery, enabling the restoration of both function and esthetics of affected tissues. These materials exhibit a wide range of compositions, as well as physical, biological, and mechanical properties that directly influence their integration capacity, resorption rate, and clinical efficacy[25,150]. Currently, xenogeneic, alloplastic, and synthetic biomaterials are widely employed, each possessing specific characteristics that make them suitable for distinct clinical contexts and regenerative demands[38,151]. Therefore, the appropriate selection of these materials is crucial for the success of regenerative procedures, considering the complex interaction between the biomaterial, the host biological environment, and the mechanical requirements of the recipient site[152].
All bone substitute biomaterials of both natural and synthetic origin considered in this review are marketed in granular form, which allows optimal handling during surgical procedures and promotes effective osteoconduction by providing space for vascularization and new bone formation. However, beyond descriptive characteristics, their clinical effectiveness varies considerably according to origin, resorption dynamics, and mechanical behavior, requiring a comparative interpretation of evidence.
Naturally derived materials (bovine and porcine xenografts) demonstrated consistently high biocompatibility and predictable osteoconduction, largely due to their trabecular architecture and slow resorption profiles. Products such as Geistlich Bio-Oss®, Cerabone®, and Lumina-Bone Porous® showed superior volumetric stability and long-term clinical performance in alveolar reconstruction and sinus augmentation, as documented in recent clinical and histological studies[144,153].
Collagen-preserving xenografts such as THE Graft®, OsteoBiol Gen-Os®, and Apatos® exhibited integration within 6-12 months and supported cell adhesion due to their organic matrix. In contrast, sintered bovine grafts such as GenOx Inorg® and OrthoGen Granules® demonstrated enhanced mechanical stability and markedly slower resorption (> 24 months), making them suitable for areas subjected to higher functional loading. Composite biomaterials such as SmartBone®, which incorporate collagen and biodegradable polymers into a bovine matrix, showed increased compressive strength and more rapid integration[154].
Synthetic and composite substitutes, including hydroxyapatite/β-TCP materials such as Nanosynt®, GenPhos hydroxyapatite TCP®, and BoneCeramic®, provided tunable porosity and controllable resorption, enabling gradual bone replacement comparable to xenografts[155]. Bioactive materials such as BioGran® (45S5 bioactive glass) promoted rapid angiogenesis and early bone formation, whereas calcium-based materials like Calcigen S® exhibited accelerated resorption suitable for small, contained defects[156,157].
Allogeneic biomaterials such as Grafton DBM® and MinerOss® displayed osteoinductive potential and variable integration (3-12 months), although with mechanical limitations. In contrast, high-purity synthetic materials such as HAP-91® and OsteoGen® exhibited prolonged volumetric maintenance and low immunological risk, being suitable for alveolar and peri-implant reconstructions[158,159].
In contrast, synthetic and hybrid biomaterials offer greater control over physicochemical properties and resorption rates, allowing adaptation to the specific bone defect requirements and reduction of immunological risks. Hydroxyapatite/β-TCP composites exhibit osteoconductive behavior comparable to xenografts, but with more controlled resorption and no risk of biological transmission. Bioactive glasses such as 45S5 have gained prominence for simultaneously stimulating osteogenesis and angiogenesis, thereby accelerating regeneration in critical-size defects[160,161].
Moreover, recent studies indicate that the combination of biomaterials with growth factors and collagen membranes enhances bone regeneration by improving clot stability and promoting osteoblast differentiation[162,163]. This integrative approach reinforces the importance of personalized strategies in which biomaterial selection considers not only the defect size and location but also the patient’s biological profile and restorative treatment plan.
When comparing the materials, the literature indicates that xenogeneic grafts provide the most stable long-term volumetric outcomes due to their slow and predictable resorption, supported by extensive clinical and histological evidence[153,164]. In contrast, HA/β-TCP composites offer more controllable degradation profiles and faster remodeling, making them suitable for medium-sized defects requiring earlier replacement by native tissue[155]. Bioactive glass demonstrates superior early regenerative activity, promoting rapid angiogenesis and accelerated bone deposition, which is advantageous in defects requiring faster biological activation[156,157]. Meanwhile, allogeneic and high-purity synthetic substitutes show variable remodeling rates but low immunogenicity, serving as suitable options when biologic activity or structural maintenance is prioritized[158,159].
Taken together, these findings demonstrate that the choice of biomaterial should be guided by the match between material properties and clinical requirements. Slow-resorbing xenografts are preferable for esthetic areas and sites demanding long-term stability[153,165]; hydroxyapatite/β-TCP composites are appropriate for defects requiring balanced remodeling speed and structural support[155]; bioactive glass is beneficial in cases where rapid angiogenesis and early bone fill are needed[156,157]; and allogeneic or highly pure synthetic materials can be used when immunological safety or reduced donor-site morbidity are priorities[158,159].
Therefore, biomaterial selection should be individualized, taking into account structural characteristics, mechanical properties, and supporting clinical evidence. The future of bone tissue engineering is directed toward the development of hybrid and bioactive biomaterials capable of combining osteoconductivity, osteoinduction, and mechanical stability, thereby maximizing clinical success in bone regeneration and reconstructive orthopedic applications[154].
A number of studies have evaluated the biological and physicochemical characteristics of natural and synthetic bone substitutes, as well as their clinical performance in bone reconstruction[166-179]. Several experimental and clinical studies have investigated the effects of photobiomodulation and other biostimulatory therapies on bone repair[180-197].
Table 2[167-170,172-178,195-197] and 3[171,179-188] summarize the main characteristics of commercially available biomaterials, highlighting their composition, properties, integration mechanisms, and clinical applications, serving as a guide for evidence-based selection. Table 2 synthesizes biomaterials of natural origin, while Table 3 presents synthetic and composite biomaterials.
| Ref. | Commercial name/country/ | Composition | Physical properties | Biological properties | Mechanical properties | Resorption/ | Clinical efficacy | Form/cost |
| [167] | Bonefill | Mineralized bovine spongy xenograft | White granules, irregular, porous | Biocompatible, osteoconductive, high interconnectivity | Lightweight, strong; maintains space and support | Slow and progressive (6-9 months) | Documented use in sinus lift, GBR, and alveolar filling | Granules (0.10-2.5 mm)/affordable |
| [168] | Cerabone®/ | 100% inorganic, calcined bovine bone | Purified ceramic; hard and stable particles | Biocompatible; osteoconductive; bone scaffold | High stability and strength | Very slow; progressive integration | Well-established use in peri-implant grafts, sinus lift, and bone augmentation | Granules and blocks (0.5-4 mm) high |
| [169] | Geistlich | Bovine xenograft | Porous, similar to human bone | Biocompatible, osteoconductive | Preserves volume and structure | Slow, promotes maintenance | Widely used in implant dentistry; proven clinical efficacy | Granules and blocks (0.25-2 mm) high |
| [172] | GenMix®/ | Inorganic bovine xenograft (cortical and cancellous matrix) | Dry granules, lightweight; high volumetric stability; good handling | Biocompatible; osteoconductive; promotes new bone formation; no transmission risk | Good initial strength; intermediate support | Slow and partial (some particles > 12 months); integration 6-9 months | Used in alveolar grafts, sinus lift, ridge augmentation; studies show predictability and maintenance | Granules (0.25 mm; 0.5 mm; 1.0 mm)/ |
| [195] | GenOx | Inorganic bovine xenograft (mineral bone matrix, sintered) | Rigid, stable granules; resistant to rapid resorption; volume-maintaining | Biocompatible; osteoconductive; non-immunogenic; slow bone replacement | High initial strength; prolonged support; indicated for load-bearing areas | Very slow (> 24 months) | Used in sinus lifts, ridge augmentation, alveolar filling, and periodontal applications; proven efficacy | Granules (0.25 mm; 0.5 mm; 1.0 mm; 2.0 mm)/affordable cost |
| [170] | GenOx | Organic and inorganic bovine matrix (type I collagen) | Lightweight, porous, and moldable; trabecular structure | Biocompatible, osteoconductive, potential osteopromotive effect | Lower resistance; suitable for low-load areas | 6 months to 12 months | Effective in GBR, fillings, and esthetic areas; good remodeling | Granules (0.25-2 mm)/affordable |
| [173] | Lumina-Bone | Inorganic lyophilized bovine xenograft | Highly porous, bone-like | Biocompatible, osteoconductive | Preserves volume and structure | Slow (6-12 months or more) | Successful in reconstructions, sinus lift, and preservation | Granules (0.25-1 mm)/affordable |
| [174] | MinerOss | Porcine xenograft (inorganic bone matrix) | Dry, sterile, irregular granules; high volumetric stability | Biocompatible; osteoconductive; good bone integration | Limited structural support; dimensional stability | Slow (6-9 months) | Widely used in GBR, sinus lift, alveolar preservation, and peri-implant defects; good volumetric maintenance and integration | Granules (0.25 mm; 1.0 mm; 2.0 mm)/ |
| [196] | OrthoGen | Bovine xenograft | Rigid granules; good volumetric stability; non-collapsing | Biocompatible; osteoconductive; guides bone growth; no rejection | Adequate structural support; maintains space; compatible with membranes/PRF | Very slow (up to 24 months) | Used in GBR, ridge augmentation, sinus lift, peri-implant, and post-extraction; studies show stability and osseointegration | Granules (0.25 mm; 0.5 mm; 1.0 mm; 2.0 mm)/affordable cost |
| [197] | OsteoBiol | Porcine or equine xenograft | Dry, rigid, trabecular granules | Biocompatible; osteoconductive; matrix for osteoprogenitor cells and new bone formation | High temporary resistance; good support and volume maintenance | Slow (9-12 months) | Effective for sinus lift, alveolar regeneration, and ridge augmentation; extensive clinical and histological evidence | Granules (0.25-2.0 mm)/moderate to high cost |
| [175] | OsteoBiol | Porcine or equine xenograft (cortical and cancellous bone + preserved collagen) | Natural trabecular structure; easy handling | Biocompatible; osteoconductive; native collagen promotes regeneration | Temporary structural support; maintains volume | Slow (6-12 months) | High efficacy; supported by multiple clinical and histological studies | Granules (0.25 mm; 1.0 mm; 2.0 mm)/ |
| [176] | Osteosynt®/ | Bovine xenograft (HA + β-TCP) | Dry, white granules; texture similar to cancellous bone; high volumetric stability | Biocompatible; osteoconductive; no inflammatory response; supports cell colonization and bone formation | Non-deformable; maintains space; limited mechanical load support | Slow (6-18 months) | Used in GBR, sinus lift, post-extraction sockets, and peri-implant defects; maintains volume; can be combined with membranes or PRF | Granules (0.25 mm; 1.0 mm; 2.0 mm), block, wedge, sphere/affordable cost |
| [177] | SmartBone®/ | Bovine xenograft (HA) + biodegradable polymers + type I collagen | Rigid trabecular structure | Biocompatible; osteoconductive; bioactive; rapid tissue integration | High compressive strength; excellent stability; can be fixed with plates or screws | Slow (6-18 months) | Recommended for load-bearing reconstructions in implantology, orthopedics, and oncology; extensive scientific documentation | Granules, blocks, and strips/high cost |
| [178] | THE Graf®/ | Porcine bone mineral matrix, similar to human bone | Interconnected pores, high hydrophilicity | Biocompatible, free of immunogenic agents, osteoconductive | Stable structure for regeneration | From 10 weeks | In vitro, in vivo and clinical studies | Granules/high |
| Ref. | Commercial name/ | Composition | Physical properties | Biological properties | Mechanical properties | Resorption/ | Clinical efficacy | Form/cost |
| [179] | Alobone | Synthetic: Low-crystallinity hydroxyapatite | Porous granules, promote vascularization | Biocompatible, osteoconductive, protein-free | Suitable for filling bone defects | Gradual, replaced by new bone | Positive results in sinus lift, ridge augmentation, and preservation | Granules (0.25-1 mm)/ |
| [180,181] | BioGran®/ | Alloplastic; bioactive glass 45S5 (calcium sodium silicate) | Translucent white; microporous; high surface area | Biocompatible; osteoconductive; potentially osteoinductive; stimulates osteogenesis, angiogenesis, and HA formation | Stable in small to moderate defects; no structural support in load-bearing areas | Rapid (3-6 months); efficient | Good results in alveolar, peri-implant, periodontal, and sinus regeneration | Granules (0.3-0.35 mm)/ |
| [182] | Bonecerami®/ | Alloplastic; 60% HA + 40% β-TCP | 3D structure similar to cancellous bone | Biocompatible; osteoconductive; cell adhesion; no significant inflammatory response | Lightweight and stable; not indicated for high-load areas without protection | 4-6 months; | High predictability in GBR, sinus lift, and alveolar filling; comparable to xenografts | Granules (0.5/1.0-2.0 mm)/ |
| [181] | Calcigen S®/ | Alloplastic; calcium sulfate | Moldable (spheres, blocks, wedges); powder-liquid mix; sets in 4-6 minutes | Biocompatible; osteoconductive; promotes bone growth | Quick application; not indicated for load-bearing support | 8-12 weeks | Used as filler in orthopedics and dentistry; conducive environment for bone regeneration | Powder for mixing/ |
| [171] | GenPhos | Synthetic: 70% HA + 30% β-TCP | Porous granules, similar to cancellous bone | Biocompatible, osteoconductive, protein-free | Good initial strength, lower than ceramics | Slow (7-9 months); gradual integration | Positive use in sockets, ridge augmentation, and peri-implant areas | Granules (0.5-0.75 mm)/ |
| [183] | Grafton | Allogeneic; demineralized human bone + glycerol | Moldable; ready to use; adapts to irregular defects | Biocompatible; osteoconductive and potentially osteoinductive; stimulates osteogenesis and angiogenesis; forms HA in situ | Limited support; not indicated for load-bearing areas | 3 months to 12 months (variable) | Widely used in reconstructions, implants, and orthopedics | Gel, paste, sponge, strip/high cost |
| [184] | HAP-91®/ | Synthetic; pure synthetic HA | White, homogeneous granules; high volumetric stability; non-collapsing | Biocompatible; osteoconductive; non-immunogenic; promotes bone conduction | Good initial strength; adequate support for load and non-load areas; non-moldable | Very slow (18-24 months) | Indicated for alveolar grafts, sinus lift, GBR, peri-implant, and ridge augmentation; volumetric maintenance and osseointegration | Granules (0.15 mm; 0.3 mm; 0.5 mm; 1.0 mm; 2.0 mm)/ |
| [185] | MinerOss®/ | Allogeneic; human cortical and/or cancellous bone | Dry and rehydratable particles; easy adaptation | Biocompatible; osteoconductive; possible residual osteoinduction | Intermediate support | 4 months to 9 months (variable) | Widely used in implant dentistry and GBR | 0.25 mm to 1.0 mm; particulate (cortical, cancellous, or mix)/ |
| [186] | Nanosynt®/ | Synthetic: 60% hydroxyapatite + 40% β-TCP | Porous nanostructure, high capillarity | Biocompatible, osteoconductive, non-immunogenic | Good initial stability; intermediate mechanics | Controlled/ | Good outcomes in preservation, bone augmentation, and peri-implant procedures; systematic reviews | Granules (0.25-2 mm)/ |
| [187] | OsteoGen®/ | Alloplastic; synthetic HA | Dry, lightweight granules; maintains stability | Biocompatible; osteoconductive; non-immunogenic | Fragile; moderate support in cavities | 6-9 months | Used in implants, post-extraction sockets, and periodontics; good but limited literature compared to others | Granules (0.3-0.5 mm), dry/ |
| [188] | Vitoss®/ | Synthetic β-tricalcium phosphate (β-TCP) | Lightweight, porous, hydrophilic texture; easy to handle | Biocompatible; osteoconductive; stimulates vital bone formation | Fragile structure; compressible and adaptable but requires stabilization; not recommended for load-bearing sites | Moderate (4-9 months) | Effective in small to medium defects; indicated for trauma, spine, orthopedics, implantology, and periodontics | Granules and moldable putty/ |
The analysis of the selected literature and commercial data demonstrates that biomaterials play a fundamental role in bone regeneration, particularly in orthopedic and reconstructive rehabilitation. These materials present diverse origins, compositions, and physicochemical characteristics that directly influence their integration capacity, resorption rate, and clinical performance. Among the main categories, xenogeneic, allogeneic, and synthetic biomaterials are the most frequently employed in regenerative bone medicine, each offering specific advantages depending on defect type and clinical demand[25,38,150-152].
Comparative analysis of the evaluated biomaterials demonstrates that their origin and chemical composition directly influence the biological response and rate of tissue replacement. Bovine and porcine xenografts remain the consolidated standard in bone reconstruction due to their high biocompatibility, trabecular structure similar to human bone, and slow resorption, which favors long-term volumetric stability[153,165]. This slow integration is particularly advantageous in esthetic procedures and in regions subjected to moderate loads, ensuring clinical predictability.
In contrast, synthetic and hybrid biomaterials offer greater control over physicochemical properties and resorption rates, allowing adaptation to the specific requirements of the bone defect and reducing immunological risks. Hydroxyapatite/β-TCP composites exhibit osteoconductive behavior comparable to xenografts, but with more controlled resorption and no risk of biological transmission. Bioactive glasses (such as 45S5) have gained prominence for simultaneously stimulating osteogenesis and angiogenesis, thereby accelerating regeneration in critical-size defects[155,160].
Moreover, recent studies indicate that the combination of biomaterials with growth factors and collagen membranes enhances bone regeneration by improving clot stability and promoting osteoblastic differentiation[162,163]. This integrative approach reinforces the importance of personalized strategies in which biomaterial selection considers not only the size and location of the defect but also the patient’s biological profile and the restorative treatment plan[187-191].
Emerging evidence also supports the synergistic combination of biomaterials with adjuvant biological therapies, such as collagen membranes, PRF, and low-level laser photobiomodulation. These associations enhance clot stability, modulate inflammation, and promote osteoblastic differentiation, thereby improving both the speed and quality of bone regeneration in orthopedic and reconstructive contexts[162,163,191]. The incorporation of growth factors or fibrin matrices accelerates vascularization and supports the balance between bone resorption and formation. Photobiomodulation (LLLT) acts through mitochondrial photoreceptors to increases ATP production and stimulates collagen and angiogenic factors, which synergize with scaffold-based regeneration[108,112,113,192-194].
However, despite the substantial technological progress, significant obstacles continue to limit the clinical translation of next-generation biomaterials. Most available products lack standardized manufacturing protocols, leading to variability in porosity, mechanical strength, and biodegradation profiles, which in turn affects the reproducibility of clinical outcomes. Regulatory barriers also remain substantial, particularly for hybrid and bioactive scaffolds that incorporate biologically active components, as their approval requires extensive safety and long-term biocompatibility evidence. Furthermore, cost, manufacturing complexity, and the need for specialized surgical handling continue to restrict the large-scale adoption of advanced biomaterials in routine bone reconstruction[154,195].
In addition, both natural and synthetic biomaterials present inherent limitations that must be acknowledged when interpreting current evidence. Although clinically predictable, xenogeneic grafts exhibit slow remodeling and long-term persistence of residual particles, which may limit their behavior in large defects or in patients requiring accelerated healing[20,38]. Conversely, synthetic ceramics and bioactive glasses offer controlled resorption but may lack the intrinsic biological signaling present in natural matrices, resulting in variable maturation of newly formed bone in some clinical scenarios[160,196]. These limitations illustrate why no single material fulfills all of the requirements for ideal bone regeneration, reinforcing the need for integrative, patient-specific strategies.
Despite these advances, the literature still presents significant gaps that limit the clinical translation of novel biomaterials. Most investigations remain preclinical, with heterogeneous methodologies and limited long-term follow-up. The scarcity of randomized clinical trials and standardized parameters hampers direct comparison among biomaterial types and regenerative strategies[197]. Future research must focus on multicenter clinical studies that assess histological integration, volumetric maintenance and patient-centered outcomes such as function, aesthetics, and long-term stability[25,155].
It is also important to recognize the methodological limitations of the present review. The available literature is highly heterogeneous, with variability in defect models, follow-up periods, biomaterial formulations, and evaluation methods, which restricts direct comparison among studies[154,197]. In addition, most of the evidence originates from preclinical or single-center clinical investigations, limiting the generalizability of outcomes. Because this review synthesizes published data without performing a meta-analysis, the conclusions rely on qualitative integration rather than quantitative effect estimates. These factors should be considered when interpreting the applicability of the findings to broad clinical practice[25].
Despite these limitations, the current body of evidence supports a paradigm shift in regenerative bone medicine from traditional, biologically passive materials to multifunctional, bioactive, and patient-specific scaffolds. The integration of synthetic matrices with osteoinductive molecules, stem cell–based therapies, and photobiomodulatory strategies constitutes a promising frontier for bone regeneration. The future of tissue engineering lies in hybrid systems capable of dynamic interaction with host tissues, ensuring enhanced predictability, safety, and individualized treatment outcomes in orthopedic and reconstructive bone regeneration[52,160].
Future perspectives emphasize the continuous advancement of tissue engineering, focusing on the development of biomimetic three-dimensional scaffolds, 3D printing of customized matrices, bioactive biomaterials, and controlled-release systems for osteoinductive substances. Cell therapy, particularly those using MSCs, emerges as a promising frontier aiming at more functional, predictive, and personalized bone regeneration.
However, despite these advances, several research bottlenecks remain. For 3D-printed scaffolds, the main limitations involve insufficient mechanical resistance of polymer-based constructs, difficulties in reproducing hierarchical microarchitecture, and challenges in ensuring homogeneous vascularization throughout large defects. Overcoming these issues will require the integration of composite materials with tunable stiffness, optimization of pore geometry, and development of bioinks capable of supporting angiogenic signaling.
Regarding bioactive and osteoinductive biomaterials, major obstacles include the unpredictable release kinetics of growth factors, risk of ectopic mineralization, and limited long-term stability of functionalized surfaces. Breakthroughs in this area will depend on engineering stimulus-responsive carriers and refining controlled-release systems to synchronize degradation with new bone formation.
For cell-based therapies, current challenges include variability in MSC quality, high cost of cell expansion, and regulatory barriers that slow clinical translation. Future progress will require standardized manufacturing protocols, scalable bioprocessing methods, and the refinement of allogeneic MSC platforms to reduce costs while maintaining safety and therapeutic potency.
Future research directions include the development of 3D-printed scaffolds with controlled growth factor release, the use of nanobiomaterials with immunomodulatory properties, and the consolidation of clinical protocols involving allogeneic MSCs. These strategies represent feasible pathways for overcoming current technological limitations and strengthening the interface between basic science and clinical practice, ultimately enabling bone regeneration that is more efficient, predictable, and accessible.
Bone regeneration constitutes a fundamental pillar in reconstructive rehabilitation across various medical and surgical specialties, with biomaterials used as grafts being essential for the repair of bone defects of diverse etiologies. This review highlights the diversity of materials currently available in the market, exhibiting distinct physicochemical, biological, and clinical properties, each with specific advantages and limitations.
Autogenous grafts remain the gold standard due to their osteogenic, osteoinductive, and osteoconductive capabilities, although the need for a donor site and the associated morbidity represent significant limitations. Allogeneic and xenogeneic grafts, widely employed in clinical practice, are broadly available, easy to handle, and have demonstrated efficacy through their osteoconductive capacity. However, synthetic materials have gained ground owing to their high biocompatibility, absence of immunological risk, and controllable structural characteristics, although they generally exhibit limited osteoinductive activity. There is no universal biomaterial; optimal outcomes depend on aligning material characteristics with defect morphology and patient-specific biological conditions.
Therefore, the ongoing development and refinement of biomaterials, combined with the integration of innovative biotechnological approaches, represent a promising path toward increasingly safe, effective, and individualized therapies in bone reconstruction applied to orthopedics and regenerative medicine. Despite significant advances, important gaps remain, particularly regarding the standardization of clinical protocols involving synthetic biomaterials and combined therapies. Long-term randomized clinical trials are scarce, hindering validation of new materials and emerging technologies. Furthermore, there is a lack of studies directly comparing the performance of different biomaterials under specific clinical conditions, as well as cost-benefit analyses and assessments of long-term functional impact.
| 1. | Wawrzyniak A, Balawender K. Structural and Metabolic Changes in Bone. Animals (Basel). 2022;12:1946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 75] [Reference Citation Analysis (0)] |
| 2. | Chaudhari S, Khade A, Girase V, Dhatrak P. A Systematic review on Bone grafts and Biomaterials substitutes for Bone Regeneration. J Phys: Conf Ser. 2024;2837:012033. [DOI] [Full Text] |
| 3. | van Dijk LA, Janssen NG, Nurmohamed SJ, Muradin MSM, Longoni A, Bakker RC, de Groot FG, de Bruijn JD, Gawlitta D, Rosenberg AJWP. Osteoinductive calcium phosphate with submicron topography as bone graft substitute for maxillary sinus floor augmentation: A translational study. Clin Oral Implants Res. 2023;34:177-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 4. | de Azambuja Carvalho PH, Dos Santos Trento G, Moura LB, Cunha G, Gabrielli MAC, Pereira-Filho VA. Horizontal ridge augmentation using xenogenous bone graft-systematic review. Oral Maxillofac Surg. 2019;23:271-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 5. | Haidar ZS. Biomimetics-Bridging the Gap (Biomedical Engineering). London: IntechOpen, 2023. [DOI] [Full Text] |
| 6. | Khan FA, Parayaruthottam P, Roshan G, Menon V, Fidha M, Fernandes AK. Platelets and Their Pathways in Dentistry: Systematic Review. J Int Soc Prev Community Dent. 2017;7:S55-S60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Mauffrey C, Barlow BT, Smith W. Management of Segmental Bone Defects. J Am Acad Orthop Sur. 2015;23:143-153. [DOI] [Full Text] |
| 8. | Archual AJ, Bishop AT, Shin AY. Vascularized Bone Grafts in Orthopaedic Surgery: A Review of Options and Indications. J Am Acad Orthop Surg. 2022;30:60-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 9. | Ashammakhi N, GhavamiNejad A, Tutar R, Fricker A, Roy I, Chatzistavrou X, Hoque Apu E, Nguyen KL, Ahsan T, Pountos I, Caterson EJ. Highlights on Advancing Frontiers in Tissue Engineering. Tissue Eng Part B Rev. 2022;28:633-664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 70] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 10. | Cicciù M, Cervino G, Herford AS, Famà F, Bramanti E, Fiorillo L, Lauritano F, Sambataro S, Troiano G, Laino L. Facial Bone Reconstruction Using both Marine or Non-Marine Bone Substitutes: Evaluation of Current Outcomes in a Systematic Literature Review. Mar Drugs. 2018;16:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (13)] |
| 11. | Ferraz MP. Bone Grafts in Dental Medicine: An Overview of Autografts, Allografts and Synthetic Materials. Materials (Basel). 2023;16:4117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 121] [Reference Citation Analysis (0)] |
| 12. | Zhang S, Li X, Qi Y, Ma X, Qiao S, Cai H, Zhao BC, Jiang HB, Lee ES. Comparison of Autogenous Tooth Materials and Other Bone Grafts. Tissue Eng Regen Med. 2021;18:327-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 13. | Sillmann YM, Eber P, Orbeta E, Wilde F, Gross AJ, Guastaldi FPS. Milestones in Mandibular Bone Tissue Engineering: A Systematic Review of Large Animal Models and Critical-Sized Defects. J Clin Med. 2025;14:2717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 14. | Yang Z, Wu C, Shi H, Luo X, Sun H, Wang Q, Zhang D. Advances in Barrier Membranes for Guided Bone Regeneration Techniques. Front Bioeng Biotechnol. 2022;10:921576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 15. | Ren Y, Fan L, Alkildani S, Liu L, Emmert S, Najman S, Rimashevskiy D, Schnettler R, Jung O, Xiong X, Barbeck M. Barrier Membranes for Guided Bone Regeneration (GBR): A Focus on Recent Advances in Collagen Membranes. Int J Mol Sci. 2022;23:14987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 135] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 16. | Alauddin MS, Abdul Hayei NA, Sabarudin MA, Mat Baharin NH. Barrier Membrane in Regenerative Therapy: A Narrative Review. Membranes (Basel). 2022;12:444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 50] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 17. | Mizraji G, Davidzohn A, Gursoy M, Gursoy U, Shapira L, Wilensky A. Membrane barriers for guided bone regeneration: An overview of available biomaterials. Periodontol 2000. 2023;93:56-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 104] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 18. | Zhao R, Yang R, Cooper PR, Khurshid Z, Shavandi A, Ratnayake J. Bone Grafts and Substitutes in Dentistry: A Review of Current Trends and Developments. Molecules. 2021;26:3007. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 348] [Article Influence: 69.6] [Reference Citation Analysis (0)] |
| 19. | Haugen HJ, Lyngstadaas SP, Rossi F, Perale G. Bone grafts: which is the ideal biomaterial? J Clin Periodontol. 2019;46 Suppl 21:92-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 343] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 20. | de Azambuja Carvalho PH, de Oliveira Ciaramicolo N, Ferreira Júnior O, Pereira-filho VA. Clinical and laboratorial outcomes of xenogeneic biomaterials: literature review. Front Oral Maxillofac Med. 2023;5. [DOI] [Full Text] |
| 21. | Catarino M, Castro F, Macedo JP, Lopes O, Pereira J, Lopes P, Fernandes GVO. Mechanisms of Degradation of Collagen or Gelatin Materials (Hemostatic Sponges) in Oral Surgery: A Systematic Review. Surgeries. 2024;5:532-548. [DOI] [Full Text] |
| 22. | Dec P, Modrzejewski A, Pawlik A. Existing and Novel Biomaterials for Bone Tissue Engineering. Int J Mol Sci. 2022;24:529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 85] [Reference Citation Analysis (0)] |
| 23. | Xia D, Chen J, Zhang Z, Dong M. Emerging polymeric biomaterials and manufacturing techniques in regenerative medicine. Aggregate. 2022;3:e176. [RCA] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 24. | Kalirajan C, Dukle A, Nathanael AJ, Oh TH, Manivasagam G. A Critical Review on Polymeric Biomaterials for Biomedical Applications. Polymers (Basel). 2021;13:3015. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 25. | Łuczak JW, Palusińska M, Matak D, Pietrzak D, Nakielski P, Lewicki S, Grodzik M, Szymański Ł. The Future of Bone Repair: Emerging Technologies and Biomaterials in Bone Regeneration. Int J Mol Sci. 2024;25:12766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 51] [Reference Citation Analysis (0)] |
| 26. | Pires ALR, Bierhalz ACK, Moraes ÂM. Biomaterials: Types, Applications, and Markets. Química Nova. 2015;. [DOI] [Full Text] |
| 27. | Wei H, Cui J, Lin K, Xie J, Wang X. Recent advances in smart stimuli-responsive biomaterials for bone therapeutics and regeneration. Bone Res. 2022;10:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 261] [Article Influence: 65.3] [Reference Citation Analysis (0)] |
| 28. | Todd EA, Mirsky NA, Silva BLG, Shinde AR, Arakelians ARL, Nayak VV, Marcantonio RAC, Gupta N, Witek L, Coelho PG. Functional Scaffolds for Bone Tissue Regeneration: A Comprehensive Review of Materials, Methods, and Future Directions. J Funct Biomater. 2024;15:280. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 46] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 29. | Xue G, Wang S, Liu Q, Zhang K, Xin P. Analysis of the effects of concentrated growth factor and low-level laser therapy on the bone healing. Heliyon. 2023;9:e12800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 30. | Marshall KM, Wojciechowski JP, Jayawarna V, Hasan A, Echalier C, Øvrebø Ø, Yang T, Zhou K, Kanczler JM, Mata A, Salmeron-Sanchez M, Stevens MM, Oreffo ROC. Considerations of growth factor and material use in bone tissue engineering using biodegradable scaffolds in vitro and in vivo. Sci Rep. 2024;14:25832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 31. | Avila Narváez JT, Mora Cajilima SM, Carpio Cedillo CP. Injertos, sustitutos óseos, mediadores bioactivos para regeneración ósea: Una revisión actualizada. Res Soc Dev. 2024;13:e14113646042. [DOI] [Full Text] |
| 32. | Laubach M, Whyte S, Chan HF, Frankenbach-Désor T, Mayer-Wagner S, Hildebrand F, Holzapfel BM, Kneser U, Dulleck U, Hutmacher DW. Lost in translation: the lack of agreement between surgeons and scientists regarding biomaterials research and innovation for treating bone defects. BMC Med. 2024;22:517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 33. | Todros S, Todesco M, Bagno A. Biomaterials and Their Biomedical Applications: From Replacement to Regeneration. Processes. 2021;9:1949. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 34. | Dewey MJ, Harley BAC. Biomaterial design strategies to address obstacles in craniomaxillofacial bone repair. RSC Adv. 2021;11:17809-17827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 35. | Győri DS. Research on Bone Cells in Health and Disease. Int J Mol Sci. 2024;25:8758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 36. | Šromová V, Sobola D, Kaspar P. A Brief Review of Bone Cell Function and Importance. Cells. 2023;12:2576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 88] [Reference Citation Analysis (0)] |
| 37. | Singh S, Sarma DK, Verma V, Nagpal R, Kumar M. From Cells to Environment: Exploring the Interplay between Factors Shaping Bone Health and Disease. Medicina (Kaunas). 2023;59:1546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 38. | Santoro A, Voto A, Fortino L, Guida R, Laudisio C, Cillo M, D'Ursi AM. Bone Defect Treatment in Regenerative Medicine: Exploring Natural and Synthetic Bone Substitutes. Int J Mol Sci. 2025;26:3085. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 37] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 39. | Szwed-Georgiou A, Płociński P, Kupikowska-Stobba B, Urbaniak MM, Rusek-Wala P, Szustakiewicz K, Piszko P, Krupa A, Biernat M, Gazińska M, Kasprzak M, Nawrotek K, Mira NP, Rudnicka K. Bioactive Materials for Bone Regeneration: Biomolecules and Delivery Systems. ACS Biomater Sci Eng. 2023;9:5222-5254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 137] [Reference Citation Analysis (0)] |
| 40. | Fernandez de Grado G, Keller L, Idoux-Gillet Y, Wagner Q, Musset AM, Benkirane-Jessel N, Bornert F, Offner D. Bone substitutes: a review of their characteristics, clinical use, and perspectives for large bone defects management. J Tissue Eng. 2018;9:2041731418776819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 449] [Cited by in RCA: 490] [Article Influence: 61.3] [Reference Citation Analysis (1)] |
| 41. | Georgeanu VA, Gingu O, Antoniac IV, Manolea HO. Current Options and Future Perspectives on Bone Graft and Biomaterials Substitutes for Bone Repair, from Clinical Needs to Advanced Biomaterials Research. Appl Sci. 2023;13:8471. [DOI] [Full Text] |
| 42. | Hutchings G, Moncrieff L, Dompe C, Janowicz K, Sibiak R, Bryja A, Jankowski M, Mozdziak P, Bukowska D, Antosik P, Shibli JA, Dyszkiewicz-Konwińska M, Bruska M, Kempisty B, Piotrowska-Kempisty H. Bone Regeneration, Reconstruction and Use of Osteogenic Cells; from Basic Knowledge, Animal Models to Clinical Trials. J Clin Med. 2020;9:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 43. | Cao X, Sun K, Luo J, Chen A, Wan Q, Zhou H, Zhou H, Liu Y, Chen X. Enhancing Osteogenesis and Mechanical Properties through Scaffold Design in 3D Printed Bone Substitutes. ACS Biomater Sci Eng. 2025;11:710-729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 44. | Liu S, Gao M, Liu N, Li R, Zhang Z, Yao Y, Wang W, Tan L, Zhang N. Osteoinduction and Osteoconduction Evaluation of Biodegradable Magnesium Alloy Scaffolds in Repairing Large Segmental Defects in Long Bones of Rabbit Models. ACS Omega. 2024;9:46419-46428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 45. | Wang B, Xie X, Jiang W, Zhan Y, Zhang Y, Guo Y, Wang Z, Guo N, Guo K, Sun J. Osteoinductive micro-nano guided bone regeneration membrane for in situ bone defect repair. Stem Cell Res Ther. 2024;15:135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 46. | Wickramasinghe ML, Dias GJ, Premadasa KMGP. A novel classification of bone graft materials. J Biomed Mater Res B Appl Biomater. 2022;110:1724-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 47. | Duarte ND, Frigério PB, Chica GEA, Okamoto R, Buchaim RL, Buchaim DV, Messora MR, Issa JPM. Biomaterials for Guided Tissue Regeneration and Guided Bone Regeneration: A Review. Dent J (Basel). 2025;13:179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 48. | Kazimierczak P, Przekora A. Osteoconductive and Osteoinductive Surface Modifications of Biomaterials for Bone Regeneration: A Concise Review. Coatings. 2020;10:971. [RCA] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 49. | Zhang H, Qiao W, Liu Y, Yao X, Zhai Y, Du L. Addressing the challenges of infectious bone defects: a review of recent advances in bifunctional biomaterials. J Nanobiotechnology. 2025;23:257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 50. | De Pace R, Iaquinta MR, Benkhalqui A, D'Agostino A, Trevisiol L, Nocini R, Mazziotta C, Rotondo JC, Bononi I, Tognon M, Martini F, Mazzoni E. Revolutionizing bone healing: the role of 3D models. Cell Regen. 2025;14:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 51. | Hadzik J, Jurczyszyn K, Gębarowski T, Trytek A, Gedrange T, Kozakiewicz M, Dominiak M, Kubasiewicz-Ross P, Trzcionka-Szajna A, Szajna E, Simka W. An Experimental Anodized and Low-Pressure Oxygen Plasma-Treated Titanium Dental Implant Surface-Preliminary Report. Int J Mol Sci. 2023;24:3603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 52. | Percival KM, Paul V, Husseini GA. Recent Advancements in Bone Tissue Engineering: Integrating Smart Scaffold Technologies and Bio-Responsive Systems for Enhanced Regeneration. Int J Mol Sci. 2024;25:6012. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 74] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 53. | Ellermann E, Meyer N, Cameron RE, Best SM. In vitro angiogenesis in response to biomaterial properties for bone tissue engineering: a review of the state of the art. Regen Biomater. 2023;10:rbad027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 30] [Reference Citation Analysis (0)] |
| 54. | Zheng W, Bai Z, Huang S, Jiang K, Liu L, Wang X. The Effect of Angiogenesis-Based Scaffold of MesoporousBioactive Glass Nanofiber on Osteogenesis. Int J Mol Sci. 2022;23:12670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Feroz S, Cathro P, Ivanovski S, Muhammad N. Biomimetic bone grafts and substitutes: A review of recent advancements and applications. Biomed Eng Adv. 2023;6:100107. [DOI] [Full Text] |
| 56. | Lv Z, Ji Y, Wen G, Liang X, Zhang K, Zhang W. Structure-optimized and microenvironment-inspired nanocomposite biomaterials in bone tissue engineering. Burns Trauma. 2024;12:tkae036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 57. | Carrascal-Hernández DC, Martínez-Cano JP, Rodríguez Macías JD, Grande-Tovar CD. Evolution in Bone Tissue Regeneration: From Grafts to Innovative Biomaterials. Int J Mol Sci. 2025;26:4242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 58. | Sebastiani S, Buccino F, Qin Z, Vergani LM. Structural influences on bone tissue engineering: A review and perspective. Matter. 2025;8:102252. [DOI] [Full Text] |
| 59. | Pramanik S, Kharche S, More N, Ranglani D, Singh G, Kapusetti G. Natural Biopolymers for Bone Tissue Engineering: A Brief Review. Eng Regen. 2023;4:193-204. [RCA] [DOI] [Full Text] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 60. | Inchingolo AM, Patano A, Di Pede C, Inchingolo AD, Palmieri G, de Ruvo E, Campanelli M, Buongiorno S, Carpentiere V, Piras F, Settanni V, Viapiano F, Hazballa D, Rapone B, Mancini A, Di Venere D, Inchingolo F, Fatone MC, Palermo A, Minetti E, Lorusso F, Scarano A, Sauro S, Tartaglia GM, Bordea IR, Dipalma G, Malcangi G. Autologous Tooth Graft: Innovative Biomaterial for Bone Regeneration. Tooth Transformer(®) and the Role of Microbiota in Regenerative Dentistry. A Systematic Review. J Funct Biomater. 2023;14:132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 44] [Reference Citation Analysis (0)] |
| 61. | Wähnert D, Greiner J, Brianza S, Kaltschmidt C, Vordemvenne T, Kaltschmidt B. Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds. Biomedicines. 2021;9:746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 62. | Cimen T, Saka YY, Ataman-Duruel ET, Duruel O, Güney Yilmaz H, Tözüm TF. Evaluation of symphysis donor site quantity by cone-beam computed tomography and its relation to anterior loop of mental foramen: an observational study. BMC Oral Health. 2025;25:834. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 63. | Kämmerer PW, Al-Nawas B. Bone reconstruction of extensive maxillomandibular defects in adults. Periodontol 2000. 2023;93:340-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 33] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 64. | Reininger D, Cobo-Vázquez C, Monteserín-Matesanz M, López-Quiles J. Complications in the use of the mandibular body, ramus and symphysis as donor sites in bone graft surgery. A systematic review. Med Oral Patol Oral Cir Bucal. 2016;21:e241-e249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 65. | Urban IA, Monje A. Guided Bone Regeneration in Alveolar Bone Reconstruction. Oral Maxillofac Surg Clin North Am. 2019;31:331-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 109] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 66. | Menezes JD, Pereira RDS, Bonardi JP, Griza GL, Okamoto R, Hochuli-Vieira E. Bioactive glass added to autogenous bone graft in maxillary sinus augmentation: a prospective histomorphometric, immunohistochemical, and bone graft resorption assessment. J Appl Oral Sci. 2018;26:e20170296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 67. | Soni R, Priya A, Yadav H, Mishra N, Kumar L. Bone augmentation with sticky bone and platelet-rich fibrin by ridge-split technique and nasal floor engagement for immediate loading of dental implant after extracting impacted canine. Natl J Maxillofac Surg. 2019;10:98-101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 68. | Govoni M, Vivarelli L, Mazzotta A, Stagni C, Maso A, Dallari D. Commercial Bone Grafts Claimed as an Alternative to Autografts: Current Trends for Clinical Applications in Orthopaedics. Materials (Basel). 2021;14:3290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 69. | Ciszyński M, Dominiak S, Dominiak M, Gedrange T, Hadzik J. Allogenic Bone Graft in Dentistry: A Review of Current Trends and Developments. Int J Mol Sci. 2023;24:16598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 70. | Motamedian SR, Khojaste M, Khojasteh A. Success rate of implants placed in autogenous bone blocks versus allogenic bone blocks: A systematic literature review. Ann Maxillofac Surg. 2016;6:78-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 49] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 71. | Lavareda Corrêa SC, Elias de Sousa J, Pasquali PJ, Scavone de Macedo LG, Aloise AC, Teixeira ML, Pelegrine AA. Use of Bone Allograft With or Without Bone Marrow Aspirate Concentrate in Appositional Reconstructions: A Tomographic and Histomorphometric Study. Implant Dent. 2017;26:915-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 72. | Stacchi C, Coyac BR, Helms JA. Biomechanical Basis for Bone Healing and Osseointegration of Implants in Sinus Grafts. Clin Implant Dent Relat Res. 2025;27:e13424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 73. | Irdem HO, Dolanmaz D, Esen A, Ünlükal N, Şimsek S. Evaluation of the Effectiveness of Liquid Platelet-Rich Fibrin and Deproteinized Bovine Bone Mineral Mixture on Newly Formed Bone in Maxillary Sinus Augmentation: A Split-Mouth, Histomorphometric Study. Niger J Clin Pract. 2021;24:1366-1372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 74. | Abdelmoneim D, Coates DE, Schmidlin P, Botter S, Li KC, Porter GC, Seo B, Duncan WJ. In vivo healing of low temperature deproteinized bovine bone xenograft in a rabbit cranial model. J Biomed Mater Res A. 2024;112:1436-1450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 75. | Pasquali PJ, André Dall'Agnol R, Garcia Rezende L, Martinez EF. Clinical, Histological, and Scintigraphic Comparative Study of the Use of Mandibular Bone Marrow and Peripheral Blood in Bone Neoformation. Int J Dent. 2021;2021:4867574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 76. | Sánchez-Pérez A, Rodríguez-Sánchez M, Martínez-Cáceres CM, Jornet-García A, Moya-Villaescusa MJ. The Efficacy of a Deproteinized Bovine Bone Mineral Graft for Alveolar Ridge Preservation: A Histologic Study in Humans. Biomedicines. 2025;13:1358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 77. | Hoveidaei AH, Sadat-Shojai M, Mosalamiaghili S, Salarikia SR, Roghani-Shahraki H, Ghaderpanah R, Ersi MH, Conway JD. Nano-hydroxyapatite structures for bone regenerative medicine: Cell-material interaction. Bone. 2024;179:116956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 78. | Kim Y, Rodriguez AE, Nowzari H. The Risk of Prion Infection through Bovine Grafting Materials. Clin Implant Dent Relat Res. 2016;18:1095-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 79. | Sun X, Liu C, Shi Y, Li C, Sun L, Hou L, Wang X. The assessment of xenogeneic bone immunotoxicity and risk management study. Biomed Eng Online. 2019;18:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 80. | Jeong HJ, Gwak SJ, Seo KD, Lee S, Yun JH, Cho YS, Lee SJ. Fabrication of Three-Dimensional Composite Scaffold for Simultaneous Alveolar Bone Regeneration in Dental Implant Installation. Int J Mol Sci. 2020;21:1863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 81. | Armitage GC. A brief history of periodontics in the United States of America: Pioneers and thought-leaders of the past, and current challenges. Periodontol 2000. 2020;82:12-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 82. | Bozza B, Pesce P, Baldi D, Bagnasco F, Migliorati M, De Angelis N. Synthetic Biomaterials for Alveolar Bone Regeneration: A Systematic Review of Clinical Evidence. Materials (Basel). 2025;18:5328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 83. | Liu CC, Solderer A, Heumann C, Attin T, Schmidlin PR. Tricalcium phosphate (-containing) biomaterials in the treatment of periodontal infra-bony defects: A systematic review and meta-analysis. J Dent. 2021;114:103812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 84. | Jin P, Liu L, Cheng L, Chen X, Xi S, Jiang T. Calcium-to-phosphorus releasing ratio affects osteoinductivity and osteoconductivity of calcium phosphate bioceramics in bone tissue engineering. Biomed Eng Online. 2023;22:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 85. | Tran DL, Ta QTH, Tran MH, Nguyen TMH, Le NTT, Nguyen Hong AP, Park HJ, Park KD, Nguyen DH. Optimized synthesis of biphasic calcium phosphate: enhancing bone regeneration with a tailored β-tricalcium phosphate/hydroxyapatite ratio. Biomater Sci. 2025;13:969-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 86. | Abtahi S, Chen X, Shahabi S, Nasiri N. Resorbable Membranes for Guided Bone Regeneration: Critical Features, Potentials, and Limitations. ACS Mater Au. 2023;3:394-417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 36] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 87. | Chen P, Wu Z, Leung A, Chen X, Landao-Bassonga E, Gao J, Chen L, Zheng M, Yao F, Yang H, Lidgren L, Allan B, Liu Y, Wang T, Zheng M. Fabrication of a silver nanoparticle-coated collagen membrane with anti-bacterial and anti-inflammatory activities for guided bone regeneration. Biomed Mater. 2018;13:065014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 88. | Gan LM, Zhou QR, Zhang Y, Yu YC, Yu ZZ, Sun Y, Li RX, Wu XW, Yang F. Alveolar Bone Morphologic Predictors for Guided Bone Regeneration Outcome in Anterior Maxilla. Int Dent J. 2024;74:102-109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 89. | Alqahtani AM, Moorehead R, Asencio IO. Guided Tissue and Bone Regeneration Membranes: A Review of Biomaterials and Techniques for Periodontal Treatments. Polymers (Basel). 2023;15:3355. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 90. | Nguyen ATM, Landao-Bassonga E, Kontogiorgos ED, Lee C, Cheng T, Ngo HC, Allan B, Goonewardene M, Opperman LA, Zheng M. The efficacy of a novel porcine-derived collagen membrane on guided bone regeneration: a comparative study in canine model. BMC Oral Health. 2025;25:850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 91. | Garcia DC, Mingrone LE, de Sá MJC. Evaluation of Osseointegration and Bone Healing Using Pure-Phase β - TCP Ceramic Implant in Bone Critical Defects. A Systematic Review. Front Vet Sci. 2022;9:859920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 92. | Wang Q, Zhou F, Qiu T, Liu Y, Luo W, Wang Z, Li H, Xiao E, Wei Q, Wu Y. Scalable fabrication of porous membrane incorporating human extracellular matrix-like collagen for guided bone regeneration. J Mater Chem B. 2024;12:11142-11155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 93. | Reis CHB, Buchaim DV, Ortiz AC, Fideles SOM, Dias JA, Miglino MA, Teixeira DB, Pereira ESBM, da Cunha MR, Buchaim RL. Application of Fibrin Associated with Photobiomodulation as a Promising Strategy to Improve Regeneration in Tissue Engineering: A Systematic Review. Polymers (Basel). 2022;14:3150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 94. | Reddy MSB, Ponnamma D, Choudhary R, Sadasivuni KK. A Comparative Review of Natural and Synthetic Biopolymer Composite Scaffolds. Polymers (Basel). 2021;13:1105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 525] [Cited by in RCA: 497] [Article Influence: 99.4] [Reference Citation Analysis (1)] |
| 95. | Egierska D, Perszke M, Mazur M, Duś-Ilnicka I. Platelet-rich plasma and platelet-rich fibrin in oral surgery: A narrative review. Dent Med Probl. 2023;60:177-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 96. | Ohayon L, Del Fabbro M. Sling Suture Technique Used to Stabilize a Collagen Membrane on the Lateral Bone Window During Maxillary Sinus Floor Augmentation with a Lateral Approach: A Retrospective Case Series. Int J Oral Maxillofac Implants. 2024;39:851-856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 97. | La Rosa GRM, Priolo CY, Abiad RS, Romeo VR, Ambu E, Pedullà E. Assessment of bone regeneration after maxillary radicular cyst enucleation with or without bone grafting materials: a retrospective cohort study. Clin Oral Investig. 2024;28:213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 98. | de Lima Barbosa R, Stellet Lourenço E, de Azevedo Dos Santos JV, Rodrigues Santiago Rocha N, Mourão CF, Alves GG. The Effects of Platelet-Rich Fibrin in the Behavior of Mineralizing Cells Related to Bone Tissue Regeneration-A Scoping Review of In Vitro Evidence. J Funct Biomater. 2023;14:503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 99. | Mercader-Ruiz J, Beitia M, Delgado D, Sánchez P, Porras B, Gimeno I, González S, Benito-Lopez F, Basabe-Desmonts L, Sánchez M. Current Challenges in the Development of Platelet-Rich Plasma-Based Therapies. Biomed Res Int. 2024;2024:6444120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 100. | Zwittnig K, Mukaddam K, Vegh D, Herber V, Jakse N, Schlenke P, Zrnc TA, Payer M. Platelet-Rich Fibrin in Oral Surgery and Implantology: A Narrative Review. Transfus Med Hemother. 2023;50:348-359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (5)] |
| 101. | Pietruszka P, Chruścicka I, Duś-Ilnicka I, Paradowska-Stolarz A. PRP and PRF-Subgroups and Divisions When Used in Dentistry. J Pers Med. 2021;11:944. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 102. | Collins T, Alexander D, Barkatali B. Platelet-rich plasma: a narrative review. EFORT Open Rev. 2021;6:225-235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 162] [Cited by in RCA: 132] [Article Influence: 26.4] [Reference Citation Analysis (1)] |
| 103. | Saqlain N, Mazher N, Fateen T, Siddique A. Comparison of single and double centrifugation methods for preparation of Platelet-Rich Plasma (PRP). Pak J Med Sci. 2023;39:634-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 104. | Albanese A, Licata ME, Polizzi B, Campisi G. Platelet-rich plasma (PRP) in dental and oral surgery: from the wound healing to bone regeneration. Immun Ageing. 2013;10:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 208] [Cited by in RCA: 199] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 105. | Pomini KT, Buchaim DV, Andreo JC, Rosso MPO, Della Coletta BB, German ÍJS, Biguetti ACC, Shinohara AL, Rosa Júnior GM, Cosin Shindo JVT, Alcalde MP, Duarte MAH, de Bortoli Teixeira D, Buchaim RL. Fibrin Sealant Derived from Human Plasma as a Scaffold for Bone Grafts Associated with Photobiomodulation Therapy. Int J Mol Sci. 2019;20:1761. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 106. | Luke AM, Mathew S, Altawash MM, Madan BM. Lasers: A Review With Their Applications in Oral Medicine. J Lasers Med Sci. 2019;10:324-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 107. | Anders JJ, Lanzafame RJ, Arany PR. Low-level light/laser therapy versus photobiomodulation therapy. Photomed Laser Surg. 2015;33:183-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 542] [Cited by in RCA: 410] [Article Influence: 37.3] [Reference Citation Analysis (1)] |
| 108. | Della Coletta BB, Jacob TB, Moreira LAC, Pomini KT, Buchaim DV, Eleutério RG, Pereira ESBM, Roque DD, Rosso MPO, Shindo JVTC, Duarte MAH, Alcalde MP, Júnior RSF, Barraviera B, Dias JA, Andreo JC, Buchaim RL. Photobiomodulation Therapy on the Guided Bone Regeneration Process in Defects Filled by Biphasic Calcium Phosphate Associated with Fibrin Biopolymer. Molecules. 2021;26:847. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 109. | Rando RG, Buchaim DV, Cola PC, Buchaim RL. Effects of Photobiomodulation Using Low-Level Laser Therapy on Alveolar Bone Repair. Photonics. 2023;10:734. [RCA] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 110. | Berni M, Brancato AM, Torriani C, Bina V, Annunziata S, Cornella E, Trucchi M, Jannelli E, Mosconi M, Gastaldi G, Caliogna L, Grassi FA, Pasta G. The Role of Low-Level Laser Therapy in Bone Healing: Systematic Review. Int J Mol Sci. 2023;24:7094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 75] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 111. | Moscatel MBM, Pagani BT, Trazzi BFDM, Reis CHB, Ribeiro CA, Buchaim DV, Buchaim RL. Effects of Photobiomodulation in Association with Biomaterials on the Process of Guided Bone Regeneration: An Integrative Review. Ceramics. 2025;8:94. [RCA] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 112. | Fernandes F, Oliveira S, Monteiro F, Gasik M, Silva FS, Sousa N, Carvalho Ó, Catarino SO. Devices used for photobiomodulation of the brain-a comprehensive and systematic review. J Neuroeng Rehabil. 2024;21:53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 113. | Sadeghian A, Rohani B, Salehi-Marzijarani M, Fekrazad R. Radiographical impact of photobiomodulation therapy on bone regeneration in clinical studies: a systematic review. Lasers Med Sci. 2025;40:23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 114. | Bauso LV, La Fauci V, Longo C, Calabrese G. Bone Tissue Engineering and Nanotechnology: A Promising Combination for Bone Regeneration. Biology (Basel). 2024;13:237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 115. | Contessi Negrini N, Angelova Volponi A, Higgins CA, Sharpe PT, Celiz AD. Scaffold-based developmental tissue engineering strategies for ectodermal organ regeneration. Mater Today Bio. 2021;10:100107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 116. | Kwee BJ, Mooney DJ. Biomaterials for skeletal muscle tissue engineering. Curr Opin Biotechnol. 2017;47:16-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 143] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 117. | Abdelaziz AG, Nageh H, Abdo SM, Abdalla MS, Amer AA, Abdal-Hay A, Barhoum A. A Review of 3D Polymeric Scaffolds for Bone Tissue Engineering: Principles, Fabrication Techniques, Immunomodulatory Roles, and Challenges. Bioengineering (Basel). 2023;10:204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 85] [Reference Citation Analysis (0)] |
| 118. | Zhao DZ, Yang RL, Wei HX, Yang K, Yang YB, Wang NX, Zhang Q, Chen F, Zhang T. Advances in the research of immunomodulatory mechanism of mesenchymal stromal/stem cells on periodontal tissue regeneration. Front Immunol. 2024;15:1449411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 119. | Bello SA, Cruz-Lebrón J, Rodríguez-Rivera OA, Nicolau E. Bioactive Scaffolds as a Promising Alternative for Enhancing Critical-Size Bone Defect Regeneration in the Craniomaxillofacial Region. ACS Appl Bio Mater. 2023;6:4465-4503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (2)] |
| 120. | Vahidi M, Rizkalla AS, Mequanint K. Extracellular Matrix-Surrogate Advanced Functional Composite Biomaterials for Tissue Repair and Regeneration. Adv Healthc Mater. 2024;13:e2401218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 121. | Sun J, Chen C, Zhang B, Yao C, Zhang Y. Advances in 3D-printed scaffold technologies for bone defect repair: materials, biomechanics, and clinical prospects. Biomed Eng Online. 2025;24:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 37] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 122. | Ye J, Miao B, Xiong Y, Guan Y, Lu Y, Jia Z, Wu Y, Sun X, Guan C, He R, Xiong X, Jia H, Jiang H, Liu Z, Zhang Y, Wei Y, Lin W, Wang A, Wang Y, Meng H, Xu W, Yuan G, Peng J. 3D printed porous magnesium metal scaffolds with bioactive coating for bone defect repair: enhancing angiogenesis and osteogenesis. J Nanobiotechnology. 2025;23:160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (1)] |
| 123. | Mota C, Puppi D, Chiellini F, Chiellini E. Additive manufacturing techniques for the production of tissue engineering constructs. J Tissue Eng Regen Med. 2015;9:174-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 176] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 124. | Lu Y, Mai Z, Cui L, Zhao X. Engineering exosomes and biomaterial-assisted exosomes as therapeutic carriers for bone regeneration. Stem Cell Res Ther. 2023;14:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 78] [Reference Citation Analysis (0)] |
| 125. | He X, Wang Y, Liu Z, Weng Y, Chen S, Pan Q, Li Y, Wang H, Lin S, Yu H. Osteoporosis treatment using stem cell-derived exosomes: a systematic review and meta-analysis of preclinical studies. Stem Cell Res Ther. 2023;14:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 126. | Theodosaki AM, Tzemi M, Galanis N, Bakopoulou A, Kotsiomiti E, Aggelidou E, Kritis A. Bone Regeneration with Mesenchymal Stem Cells in Scaffolds: Systematic Review of Human Clinical Trials. Stem Cell Rev Rep. 2024;20:938-966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 127. | Cui C, Lin F, Xia L, Zhang X. Mesenchymal stem cells therapy for the treatment of non-union fractures: a systematic review and meta-analysis. BMC Musculoskelet Disord. 2025;26:245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 128. | Qin W, Li L, Niu W, Wang W, Wu D, Song C, Gao C, Mu Z, Tay FR, Jiao K, Niu L. Effects of Electric Field‐Modulated Conductive Hydrogel on Osseoperception and Osseointegration of Dental Implants. Adv Funct Materials. 2024;34:2400256. [DOI] [Full Text] |
| 129. | Huang TH, Chen JY, Suo WH, Shao WR, Huang CY, Li MT, Li YY, Li YH, Liang EL, Chen YH, Lee IT. Unlocking the Future of Periodontal Regeneration: An Interdisciplinary Approach to Tissue Engineering and Advanced Therapeutics. Biomedicines. 2024;12:1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 130. | Cao R, Chen B, Song K, Guo F, Pan H, Cao Y. Characterization and potential of periosteum-derived cells: an overview. Front Med (Lausanne). 2023;10:1235992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 131. | Zhao Y, Xiong Y, Zheng J, Kongling W, Chen J, Li C, Hu P, Yang S, Wang X. A multifaceted biomimetic periosteum with a lamellar architecture and osteogenic/angiogenic dual bioactivities. Biomater Sci. 2023;11:3878-3892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 132. | Wen Z, Li S, Liu Y, Liu X, Qiu H, Che Y, Bian L, Zhou M. An engineered M2 macrophage-derived exosomes-loaded electrospun biomimetic periosteum promotes cell recruitment, immunoregulation, and angiogenesis in bone regeneration. Bioact Mater. 2025;50:95-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 133. | Wang L, Mao J, Cai F, Tang J, Xi K, Feng Y, Xu Y, Liang X, Gu Y, Chen L. A Structured Scaffold Featuring Biomimetic Heterogeneous Architecture for the Regeneration of Critical-Size Bone Defects. Front Bioeng Biotechnol. 2022;10:927050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 134. | Liang L, Wang J, Shang W, Taib H, Liszen T, Ghani ZA. Enhanced bone regeneration in critical-size defects using novel ultralong hydroxyapatite microtubes/chitosan composite scaffolds. Eur J Med Res. 2025;30:1076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 135. | Gupta A, Kim KW, Kurian AG, Jain SK, Bhattacharya S, Singh RK, Kim HW. Advanced therapeutic scaffolds of biomimetic periosteum for functional bone regeneration. J Nanobiotechnology. 2025;23:542. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 136. | Mao Y, Zhang Y, Wang Y, Zhou T, Ma B, Zhou P. A multifunctional nanocomposite hydrogel with controllable release behavior enhances bone regeneration. Regen Biomater. 2023;10:rbad046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 137. | Byun H, Jang GN, Jeong H, Lee J, Huh SJ, Lee S, Kim E, Shin H. Development of a composite hydrogel incorporating anti-inflammatory and osteoinductive nanoparticles for effective bone regeneration. Biomater Res. 2023;27:132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 138. | Lin X, Deng S, Fu T, Lei Y, Wang Y, Yao J, Lu Y, Huang Y, Shang J, Chen J, Zhou X. Hyaluronic acid-based hydrogel microspheres with multi-responsive properties for antibacterial therapy and bone regeneration in Staphylococcus aureus-infected skull defects. Mater Today Bio. 2025;32:101676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 139. | Ullah I, Hussain Z, Ullah S, Zahra QUA, Zhang Y, Mehmood S, Liu X, Kamya E, Waseem Ghani M, Mansoorianfar M, Wang Z, Wang Z, Pei R. An osteogenic, antibacterial, and anti-inflammatory nanocomposite hydrogel platform to accelerate bone reconstruction. J Mater Chem B. 2023;11:5830-5845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 140. | Zhang H, Wang Y, Qiao W, Hu X, Qiang H, Xia K, Du L, Yang L, Bao Y, Gao J, Zhang T, Yu Z. An injectable multifunctional nanocomposite hydrogel promotes vascularized bone regeneration by regulating macrophages. J Nanobiotechnology. 2025;23:283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 141. | Sheikh Z, Sima C, Glogauer M. Bone Replacement Materials and Techniques Used for Achieving Vertical Alveolar Bone Augmentation. Materials. 2015;8:2953-2993. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 142. | Schmidt AH. Autologous bone graft: Is it still the gold standard? Injury. 2021;52 Suppl 2:S18-S22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 359] [Article Influence: 71.8] [Reference Citation Analysis (0)] |
| 143. | Zhu S, Ma R, Qiao R, Zhang J, Hu Y. The results of anterior cruciate ligament reconstruction with allograft versus autograft: a systematic review and meta-analysis. Eur J Orthop Surg Traumatol. 2025;35:337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 144. | Wang K, Zhang J, Ding M, Xie Y, Wang Y, Jin C, Yan M, Liu L, Ding C, Chen X. Comparative evaluation of porcine and bovine bone xenografts in bone grafting: a systematic review and meta-analysis. Int J Implant Dent. 2025;11:45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 145. | Khan RS, Aslam M, Ucer C, Wright S. Success of Xenografts in Alveolar Ridge Preservation Based on Histomorphometric Outcomes. Dent J (Basel). 2023;11:215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 146. | Muñoz E, Loyola AC, Pitol-Palin L, Okamoto R, Shibli J, Messora M, Novaes AB Jr, Scombatti de Souza S. Synthetic Bone Blocks Produced by Additive Manufacturing in the Repair of Critical Bone Defects. Tissue Eng Part C Methods. 2024;30:533-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 147. | Bohner M, Santoni BLG, Döbelin N. β-tricalcium phosphate for bone substitution: Synthesis and properties. Acta Biomater. 2020;113:23-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 666] [Cited by in RCA: 395] [Article Influence: 65.8] [Reference Citation Analysis (0)] |
| 148. | Wang X, Wei W, Guo Z, Liu X, Liu J, Bing T, Yu Y, Yang X, Cai Q. Organic-inorganic composite hydrogels: compositions, properties, and applications in regenerative medicine. Biomater Sci. 2024;12:1079-1114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 149. | Hung GY, Wang CY, Hsiao HY, Tu CS, Mana-Ay H, Chen CT, Lai PL, Feng KC, Chen PY. Composite bone graft of CaO-MgO-SiO(2) glass-ceramics and CaSO(4) ceramics for boosting bone formation rate. J Mater Chem B. 2024;12:6394-6409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 150. | Gandhi Y. Biomaterials for Guided Bone Regeneration: Science and Rationale. J Maxillofac Oral Surg. 2025;24:877-885. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 151. | De Pace R, Molinari S, Mazzoni E, Perale G. Bone Regeneration: A Review of Current Treatment Strategies. J Clin Med. 2025;14:1838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 54] [Reference Citation Analysis (0)] |
| 152. | Luo Y, Kim J. Achieving the ideal balance between biological and mechanical requirements in composite bone scaffolds through a voxel-based approach. Comput Methods Biomech Biomed Engin. 2025;28:923-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 153. | Savadori P, Del Fabbro M, Sora VM, Giardino L, Generali L, Kondic D, Santoro F. Histological and Histomorphometric Evaluation of RegenerOss®: A Porcine-Derived Bone Substitute for Guided Bone Regeneration. J Compos Sci. 2025;9:245. [DOI] [Full Text] |
| 154. | Ivanova N, Ivanov S, Peev S, Dikova T. Types of Bone Substitutes and Their Application in Regenerative Medicine: A Systematic Review. J Funct Biomater. 2025;16:341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 155. | Tang G, Liu Z, Liu Y, Yu J, Wang X, Tan Z, Ye X. Recent Trends in the Development of Bone Regenerative Biomaterials. Front Cell Dev Biol. 2021;9:665813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 119] [Article Influence: 23.8] [Reference Citation Analysis (1)] |
| 156. | Piatti E, Miola M, Verné E. Tailoring of bioactive glass and glass-ceramics properties for in vitro and in vivo response optimization: a review. Biomater Sci. 2024;12:4546-4589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 157. | Wang S, Liu J, Caroprese M, Gianfreda F, Melloni F, DE Santis D. Exploring the potential of calcium-based biomaterials for bone regeneration in dentistry: a systematic review. Minerva Dent Oral Sci. 2024;73:169-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 158. | Li Y, Zhou W, Li P, Luo Q, Li A, Zhang X. Comparison of the osteogenic effectiveness of an autogenous demineralised dentin matrix and Bio-Oss® in bone augmentation: a systematic review and meta-analysis. Br J Oral Maxillofac Surg. 2022;60:868-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 159. | Wüster J, Neckel N, Sterzik F, Xiang-Tischhauser L, Barnewitz D, Genzel A, Koerdt S, Rendenbach C, Müller-Mai C, Heiland M, Nahles S, Knabe C. Effect of a synthetic hydroxyapatite-based bone grafting material compared to established bone substitute materials on regeneration of critical-size bone defects in the ovine scapula. Regen Biomater. 2024;11:rbae041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 160. | Chen S, Michálek M, Galusková D, Michálková M, Švančárek P, Talimian A, Kaňková H, Kraxner J, Zheng K, Liverani L, Galusek D, Boccaccini AR. Multi-targeted B and Co co-doped 45S5 bioactive glasses with angiogenic potential for bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;112:110909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 161. | Han Z, Xu J, Guo X, Liang L, Zhang L, Su Y, Gao W, Tang Q, Chen L, Chen Z, Xiao Y, Chen Z. The Biomaterial and Blood Clot Complex: A Crucial Early Environment for Osteo‐Coagulo‐Immunomodulation and the Advancement of Bone Biomaterials. Small Struct. 2025;6:2400625. [DOI] [Full Text] |
| 162. | Takayama T, Imamura K, Yamano S. Growth Factor Delivery Using a Collagen Membrane for Bone Tissue Regeneration. Biomolecules. 2023;13:809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (2)] |
| 163. | Lima-Sánchez B, Baus-Domínguez M, Serrera-Figallo MA, Torres-Lagares D. Advances in Synthetic Polymer Membranes for Guided Bone Regeneration in Dental Implants: A Scoping Review. J Funct Biomater. 2025;16:149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 164. | Inchingolo AM, Marinelli G, Trilli I, Del Vecchio G, Di Noia A, Inchingolo F, Del Fabbro M, Palermo A, Inchingolo AD, Dipalma G. A Histological and Clinical Evaluation of Long-Term Outcomes of Bovine Bone-Derived Xenografts in Oral Surgery: A Systematic Review. J Funct Biomater. 2025;16:321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 165. | Han HS, Kim H, Lee N, Kim S, Kim S, Cho YD. Comparison of crosslinked and non-crosslinked collagenated xenografts for vertical bone augmentation in rabbit calvaria. Sci Rep. 2025;16:1231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 166. | Stroparo JLDO, Weiss SG, Fonseca SCD, Spisila LJ, Gonzaga CC, Oliveira GCD, Brotto GL, Swiech AM, Vieira ED, Leão Neto RDR, Franco CRC, Leão MP, Deliberador TM, Gabardo MCL, Zielak JC. Xenogenic bone grafting biomaterials do not interfere in the viability and proliferation of stem cells from human exfoliated deciduous teeth - an in vitro pilot study. Res Soc Dev. 2021;10:e34410414249. [DOI] [Full Text] |
| 167. | Macedo LG, Mulinari-Santos G, Siqueira NB, Pitol-Palin L, Silva ACED, Frigério PB, Botacin PR, Lisboa-Filho PN, Okamoto R. Enhancing Bone Repair: Impact of Raloxifene-Functionalized Cerabone(®) on Rat Calvarial Defects. J Funct Biomater. 2025;16:59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 168. | Wang J, Cui W, Zhao Y, Lei L, Li H. Clinical and radiographic evaluation of Bio-Oss granules and Bio-Oss Collagen in the treatment of periodontal intrabony defects: a retrospective cohort study. J Appl Oral Sci. 2024;32:e20230268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 169. | Campos M, Rosa SVD, Nogueira MLG, Consulin MED, Signorelli LG, Marega T, Gonçalves AR. Treatment under general anesthesia after dental trauma in a patient with autism spectrum disorder: a case report. RGO, Rev Gaúch Odontol. 2023;71:e20230005. [DOI] [Full Text] |
| 170. | Egas LS, Matsumoto MA, Hadad H, Tonini KR, de Weert DAB, Perri de Carvalho PS, Ponzoni D. Comparative study of the effect of different temperatures on bovine bone used for bone repair of critical calvaria defects in rats. J Biomed Mater Res B Appl Biomater. 2023;111:1024-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 171. | de Castro HS, Cabral RN, Kehrwald R, Salmeron S, Casaroto AR, Queiroz PM. Apicectomy and simultane-ous obturation of root canals: a clinical case report. Ital J Endod. 2022;6:151-157. [DOI] [Full Text] |
| 172. | da Silva HF, Goulart DR, Sverzut AT, Olate S, de Moraes M. Comparison of two anorganic bovine bone in maxillary sinus lift: a split-mouth study with clinical, radiographical, and histomorphometrical analysis. Int J Implant Dent. 2020;6:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 173. | Kim SR, Song SJ, Urban J, Wu W, Bittner N, Tarnow DP. Comparison of dimensional changes and ridge contour around ovate pontics inserted immediately after extraction with and without buccal bone plate with different grafting procedures. J Prosthodont. 2025;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 174. | Nogueira DMB, Figadoli ALF, Alcantara PL, Pomini KT, Santos German IJ, Reis CHB, Rosa Júnior GM, Rosso MPO, Santos PSDS, Zangrando MSR, Pereira ESBM, Marchi MÂ, Trazzi BFM, Rossi JO, Salmeron S, Pastori CM, Buchaim DV, Buchaim RL. Biological Behavior of Xenogenic Scaffolds in Alcohol-Induced Rats: Histomorphometric and Picrosirius Red Staining Analysis. Polymers (Basel). 2022;14:584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 175. | Menini M, Canullo L, Iacono R, Triestino A, Caponio VCA, Savadori P, Pesce P, Pedetta A, Guerra F. Effect of Different Graft Material Consistencies in the Treatment of Minimal Bone Dehiscence: A Retrospective Pilot Study. Dent J (Basel). 2024;12:198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 176. | Pierfelice TV, Cinquini C, Petrini M, D'Amico E, D'Arcangelo C, Barone A, Iezzi G. Evaluation of Collagenic Porcine Bone Blended with a Collagen Gel for Bone Regeneration: An In Vitro Study. Int J Mol Sci. 2025;26:7621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 177. | Camacho-Alonso F, Tudela-Mulero MR, Navarro JA, Buendía AJ, Mercado-Díaz AM. Use of buccal fat pad-derived stem cells cultured on bioceramics for repair of critical-sized mandibular defects in healthy and osteoporotic rats. Clin Oral Investig. 2022;26:5389-5408. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 178. | Taschieri S, Ofer M, Corbella S, Testori T, Dellavia C, Nemcovsky C, Canciani E, Francetti L, Del Fabbro M, Tartaglia G. The Influence of Residual Alveolar Bone Height on Graft Composition after Maxillary Sinus Augmentation Using Two Different Xenografts: A Histomorphometric Comparative Study. Materials (Basel). 2020;13:5093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 179. | Lee S, Jung J, Lee J, Ko YC, Lee D, Koo KT, Seol YJ, Lee YM. Bone regeneration and biosorption patterns of different bone substitutes: an in vivo study in rabbit skulls. J Periodontal Implant Sci. 2025;55:191-205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 180. | Alves MAL, Both J, Mourão CF, Ghiraldini B, Bezerra F, Granjeiro JM, Sartoretto SC, Calasans-Maia MD. Long-Term Success of Dental Implants in Atrophic Maxillae: A 3-Year Case Series Using Hydroxyapatite and L-PRF. Bioengineering (Basel). 2024;11:1207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 181. | Ashfaq R, Kovács A, Berkó S, Budai-Szűcs M. Developments in Alloplastic Bone Grafts and Barrier Membrane Biomaterials for Periodontal Guided Tissue and Bone Regeneration Therapy. Int J Mol Sci. 2024;25:7746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 33] [Reference Citation Analysis (0)] |
| 182. | Frigério PB, Quirino LC, Gabrielli MAC, Carvalho PHA, Garcia Júnior IR, Pereira-Filho VA. Evaluation of Bone Repair Using a New Biphasic Synthetic Bioceramic (Plenum(®) Oss(hp)) in Critical Calvaria Defect in Rats. Biology (Basel). 2023;12:1417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 183. | Vaziri AS, Tahmasebi MN, Hadi H, Javidmehr S, Keyhani S, Vahdati Z, Nematian H, Farahmand Y. The Impact of Cementing Techniques on Implant Longevity in Relation to Keel Length in Persona and NexGen Knee Arthroplasty: A Comprehensive Study. Arthroplast Today. 2025;31:101608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 184. | Zielak JC, Vendramini I, Corso PFCL, Muller LL, Crivellaro VR, Khajotia SS, Esteban Florez FL, Scariot R, Elsalanty M, Deliberador TM, Storrer CLM. The Role of Marine Organic Extract in Bone Regeneration: A Pilot Study. Biomed Res Int. 2020;2020:2925879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 185. | Chang DG, Park JB, Han Y. Surgical outcomes of two kinds of demineralized bone matrix putties/local autograft composites in instrumented posterolateral lumbar fusion. BMC Musculoskelet Disord. 2021;22:200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 186. | Dorozhkin SV. Calcium Orthophosphate (CaPO4)-Based Bioceramics: Preparation, Properties, and Applications. Coatings. 2022;12:1380. [RCA] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 187. | Nascimento JRB, Sartoretto SC, Alves ATNN, Mourão CFAB, Martinez-Zelaya VR, Uzeda MJ, Granjeiro JM, Montemezzi P, Calasans-Maia MD, Calasans-Maia JA. In Vitro and In Vivo Evaluation of Nanostructured Biphasic Calcium Phosphate in Granules and Putty Configurations. Int J Environ Res Public Health. 2021;18:533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 188. | Mendes LD, Bustamante RP, Vidigal BC, Favato MN, Manzi FR, Cosso MG, Zenóbio EG. Effect of amount of biomaterial used for maxillary sinus lift on volume maintenance of grafts. J Clin Exp Dent. 2020;12:e830-e837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 189. | Apaydin AS, Johnson E, Antwi P, Rocos B, Haglund MM, Shaffrey CI, Passias P, Abd-El-Barr MM, Than K. Synthetic osteobiologics in spine surgery: a review. Turk J Med Sci. 2025;55:43-51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 190. | ElAskary A, Elfana A, Meabed M, Abd-ElWahab Radi I, Akram M, Fawzy El-Sayed K. Immediate implant placement utilizing vestibular socket therapy versus early implant placement with contour augmentation for rehabilitation of compromised extraction sockets in the esthetic zone: A randomized controlled clinical trial. Clin Implant Dent Relat Res. 2022;24:559-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 191. | Gomes YVR, Tavares AA, Barbosa RC, Tomaz AF, Sousa WJB, Oliveira LCC, Silva SML, Fook MVL. Biological responses to biomaterials: a review. Braz J Med Biol Res. 2025;58:e14599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 192. | Bighetti ACC, Cestari TM, Paini S, Pomini KT, Buchaim DV, Ortiz RC, Júnior RSF, Barraviera B, Bullen IRFR, Garlet GP, Buchaim RL, de Assis GF. Efficacy and safety of a new heterologous fibrin biopolymer on socket bone healing after tooth extraction: An experimental pre-clinical study. J Clin Periodontol. 2024;51:1017-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 193. | Hang NLT, Aviña AE, Chang CJ, Yang TS. Photobiomodulation in Promoting Cartilage Regeneration. Int J Mol Sci. 2025;26:5580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 194. | Oliveira ÉR, Nie L, Podstawczyk D, Allahbakhsh A, Ratnayake J, Brasil DL, Shavandi A. Advances in Growth Factor Delivery for Bone Tissue Engineering. Int J Mol Sci. 2021;22:903. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 164] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 195. | Singh AB, Khandelwal C, Dangayach GS. Revolutionizing healthcare materials: Innovations in processing, advancements, and challenges for enhanced medical device integration and performance. J Micromanufacturing. 2024;. [DOI] [Full Text] |
| 196. | Brochu BM, Sturm SR, Kawase De Queiroz Goncalves JA, Mirsky NA, Sandino AI, Panthaki KZ, Panthaki KZ, Nayak VV, Daunert S, Witek L, Coelho PG. Advances in Bioceramics for Bone Regeneration: A Narrative Review. Biomimetics (Basel). 2024;9:690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |