Published online Apr 18, 2026. doi: 10.5312/wjo.v17.i4.116916
Revised: December 23, 2025
Accepted: February 9, 2026
Published online: April 18, 2026
Processing time: 137 Days and 3 Hours
Knee osteoarthritis (KOA) is a major cause of disability, especially among older adults and individuals with obesity. Mesenchymal stromal cells (MSCs), particularly those derived from peripheral blood (PB)-MSCs, offer a minimally invasive alternative to bone marrow-derived MSCs for cartilage repair, with comparable chondrogenic potential and reduced procedural risks. However, optimal delivery routes remain underexplored.
To compare clinical and radiological outcomes of intra-articular, subchondral, and combined PB-MSC injections in patients with primary KOA through a double-blinded, unicentric randomized controlled trial.
A total of 150 patients with Kellgren-Lawrence grade 2 or 3 KOA will be randomized into three groups: Intra-articular (group A), subchondral (group B), and combined (group C) PB-MSC injections. PB-MSCs will be harvested post-Filgrastim stimulation via density centrifugation. Functional scores (Visual Analog Scale, Knee Injury and Osteoarthritis Outcome Score, Western Ontario and McMaster Universities Osteoarthritis Index, International Knee Documentation Committee) and imaging (magnetic resonance cartigram, Magnetic Resonance Observation of Cartilage Repair Tissue) will be assessed over 12 months.
Interim results will show that all three PB-MSC delivery routes will improve pain and function in primary KOA. Combined intra-articular plus subchondral injections will demonstrate superior gains in Visual Analog Scale, Knee Injury and Osteoarthritis Outcome Score, Western Ontario and McMaster Universities Osteoarthritis Index, and International Knee Documentation Committee scores, with magnetic resonance imaging expected to confirm better cartilage integrity. Adverse events will be reported, supporting the anticipated safety and regenerative efficacy of PB-MSC therapy.
Identifying the most effective PB-MSC delivery route is central to optimizing regenerative strategies for early-grade knee osteoarthritis. By comparing intra-articular, subchondral, and combined injection techniques, the trial seeks to enhance patient mobility, alleviate pain, and delay disease progression - ultimately informing evidence-based protocols tailored to the Indian clinical landscape.
Core Tip: Optimizing delivery routes for peripheral blood-derived mesenchymal stromal cells is key to enhancing regenerative outcomes in early-grade knee osteoarthritis. This trial compares intra-articular, subchondral, and combined injection techniques using clinical scores and advanced imaging over 12 months. The goal is to establish minimally invasive, evidence-based protocols that improve function, reduce pain, and delay progression - aligned with the Indian healthcare context and regenerative medicine advancements.
- Citation: Ratna HVK, Jeyaraman N, Nallakumarasamy A, Muthu S, Jeyaraman M. Comparative outcomes of intra-articular, subchondral, and combined injections of peripheral blood mesenchymal stromal cells in knee osteoarthritis. World J Orthop 2026; 17(4): 116916
- URL: https://www.wjgnet.com/2218-5836/full/v17/i4/116916.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i4.116916
Osteoarthritis is a prevalent chronic condition that causes significant physical pain and disability, particularly among older adults. In 2020, it was estimated to be the fourth leading cause of disability worldwide[1-3]. Knee osteoarthritis (KOA) is the most prevalent type of osteoarthritis, primarily impacting older adults and people with obesity[4]. Common symptoms include pain, swelling, stiffness, and decreased mobility. Mesenchymal stem cells (MSCs) are multipotent progenitor cells known for their ability to differentiate into multiple cell lineages, self-renew and exhibit low immunogenicity[5,6], and are commonly used as seed cells in cartilage repair and regeneration without raising concerns about an increased risk of cancer[7,8].
A vast range of researches indicate that, peripheral blood (PB) may serve as a viable alternative source of MSCs, demonstrating chondrogenic differentiation potential comparable to that of bone marrow-derived MSCs both in vitro and in vivo studies[9,10]. PB-MSCs can be collected through a minimally invasive procedure that carries fewer risks compared to bone marrow harvesting, which has been linked to complications such as hemorrhage, chronic pain, neurovascular damage, and, in rare cases, death[11,12].
PB-MSCs can also be utilized in autologous transplantation, offering significant benefits for patients in clinical settings[13]. This capability supports the advancement of one-stage surgical procedures and other cell-based therapeutic approaches[14]. Although growing evidence suggests that PB-MSCs are a promising alternative source of chondrogenic progenitor cells for cartilage repair, there is a lack of comprehensive reviews detailing their in vivo application for cartilage repair and regeneration[12,15,16].
There are no published studies on the comparative outcome analysis of intra-articular vs subchondral vs intra-articular plus subchondral injection of PB-MSCs treatment in osteoarthritis knee. But in various centers, studies are going on for individual treatment modalities in KOA. Our study will be the first study to compare the effect of intra-articular vs subchondral vs intra-articular plus subchondral injection of PB-MSCs in primary KOA in terms of pain relief, functional outcome, and cartilage thickness before and after injection.
This study aims to evaluate the effectiveness of intra-articular, subchondral, and combined intra-articular plus subchondral injections of PB-MSCs by analyzing changes in patient-reported outcome measures, including Visual Analog Scale (VAS), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and Knee Injury and Osteoarthritis Outcome Score (KOOS) scores, from baseline through multiple follow-up intervals. Additionally, it seeks to assess the healing of subchondral bone (bone marrow) lesions and cartilage integrity before and after treatment with intra-articular and subchondral PB-MSCs in patients with primary KOA, using magnetic resonance imaging (MRI)-based Magnetic Resonance Observation of Cartilage Repair Tissue scoring at the one-year follow-up point.
This is a single-centre, double-blind, randomized controlled trial aimed at comparing the clinical outcomes of intra-articular, subchondral, and combined intra-articular plus subchondral injections of PB-MSCs in patients with primary KOA. The study will be conducted and reported in accordance with the Consolidated Standards of Reporting Trials guidelines[17]. Participant allocation will be performed using a computer-generated randomization sequence created through a secure web-based platform (randomization.com). To reduce potential bias, blinding will be maintained for participants, outcome assessors, and statisticians.
The trial will be carried out in the Department of Orthopaedics at Sri Lalithambigai Medical College and Hospital. The study centre is well-equipped with advanced facilities for regenerative therapy procedures and imaging technologies essential for comprehensive evaluation of study outcomes.
This study will be conducted in accordance with the Declaration of Helsinki and good clinical practice guidelines. This study was approved by the Ethics Committee of Sri Lalithambigai Medical College and Hospital (Approval No. Dr. MGR-ERI/SLMCH/2025/027). The trial has been prospectively registered with the Clinical Trials Registry of India (No. CTRI/2025/11/097029). All participants will provide written informed consent prior to enrollment. Participant confidentiality will be strictly maintained, with data securely stored in accordance with applicable data protection standards. Any serious adverse events will be reported to the Institutional Review Board within 24 hours, and a Data Safety Monitoring Board will supervise the ongoing safety of the trial.
The sample size was determined using the formula: n = d2Zα/22 × P × (1 - P), where: Zα/2 = 1.96 corresponds to a 95% confidence interval; P = 3.5% (0.035) represents the prevalence of early KOA grade II and III[15]; d = 7% (0.07) is the desired margin of error.
Substituting these values gives: n = [(1.96)2 × 0.035 × (1 - 0.035)]/(0.07)2 ≈ 26.47. The sample size calculation drew on previously published prevalence estimates of early KOA, supplemented by preliminary pilot data from our center. A 95% confidence level and 80% statistical power were applied to detect clinically meaningful differences in pain and functional outcomes. After allowing for an anticipated 15% attrition rate, the total sample size was fixed at 150 participants, with equal allocation across three study arms of 50 patients each.
Participants will be randomly allocated to group using a secure, web-based randomization platform (randomization.com) designed to maintain balance between the groups. The randomization process will be overseen by an independent study coordinator - a physician not involved in data analysis - who will prepare identical syringes for each patient in the procedure room. Investigator will prepare the patient and procedure will be performed by senior orthopaedic surgeon with 10 years of experience. The injections, performed under local anaesthesia, will be administered by a surgeon who follows standardized procedural steps for either intra-articular or subchondral or intra-articular plus subchondral delivery. The surgeon will focus solely on the technical aspects of the procedure and will remain unaware of the treatment allocation and the patient’s clinical details. Although complete masking of procedural variations is not feasible, both patients and outcome assessors will remain blinded. Any potential pain differences are unlikely to reveal treatment allocation due to the use of local anesthesia and standardized post-procedure care (rest, ice, compression, and elevation). In cases of serious adverse events requiring unblinding, affected patients will be withdrawn from the final analysis. Allocation concealment will be maintained through the use of sealed, opaque envelopes. To minimize bias during outcome assessment, blinding will be maintained for participants, outcome assessors, and statisticians.
Inclusion criteria: Patients aged between 30 years and 80 years, irrespective of sex, who have radiologically confirmed primary osteoarthritis of the knee classified as Kellgren-Lawrence grade 2 or 3 based on X-ray findings, experiencing severe pain despite more than three months of anti-inflammatory treatment without improvement, and who have provided informed consent to undergo treatment according to our protocol, were included in the study.
Exclusion criteria: Patients younger than 30 years or older than 80 years of either sex, those with primary osteoarthritis of the knee classified as Kellgren-Lawrence grade 1 or 4 on X-ray or with secondary KOA, individuals with a history of corticosteroid injection at the treatment site within the past three months, patients diagnosed with rheumatoid arthritis, inflammatory arthritis, polyarticular arthritis, or autoimmune diseases, as well as those with haemoglobin levels below 10 g/DL or platelet counts under 105/μL, were excluded. Additional exclusions applied to patients with local infection at the procedure site, human immunodeficiency virus, hepatitis B or hepatitis C, septicaemia, systemic metabolic disorders, bleeding tendencies, coagulation abnormalities, or any other blood dyscrasias.
Following ethics committee approval from the institute and the acquisition of informed written consent from enrolled patients, each participant will undergo a thorough clinical examination to exclude other potential causes of stiff and painful knee syndrome. Baseline investigations will then be performed, including complete hemogram, erythrocyte sedimentation rate, C-reactive protein, renal function tests, random blood glucose, and serological testing for human immunodeficiency virus 1 and human immunodeficiency virus 2 as well as hepatitis B surface antigen. Radiographic evaluation will also be carried out, comprising X-ray imaging and MRI cartigram of the affected knee joint in the standing position, the latter providing assessment of both cartilage integrity and subchondral changes.
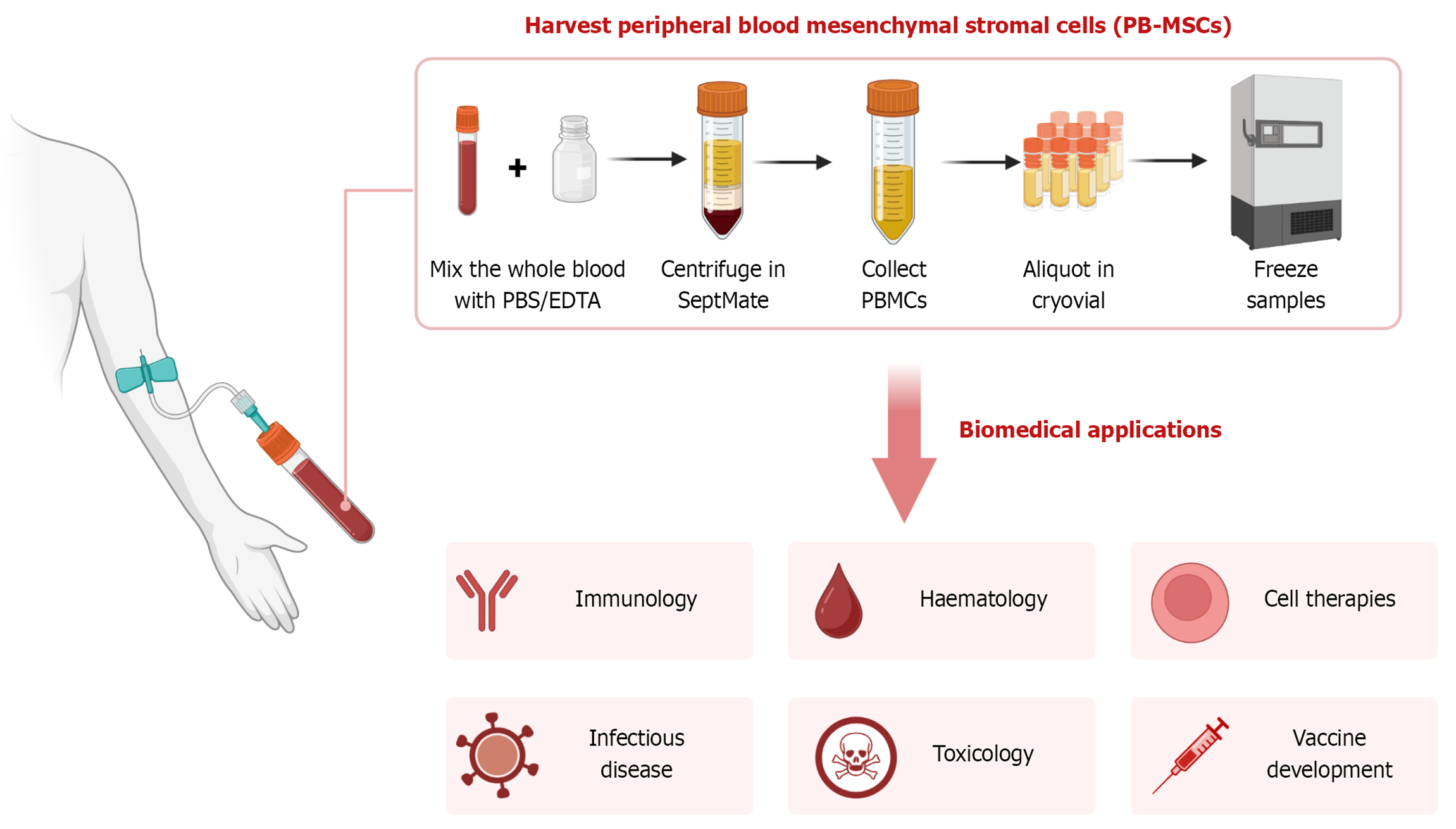
After explaining the procedure to the patients in vernacular language, initially baseline complete blood count of the study participant will be measured. Then, injection filgrastim 300 μg will be given and complete blood count will be repeated on the next day. We will notice three-fold increase in the total count - absolute for MSC/mono nuclear cells. The preparation of PB-MSCs follows double centrifugation technique called differential or density gradient centrifugation.
About 120 mL of blood will be aspirated in a heparinized (1 mL) syringe and transferred into heparinized tubes as shown in Figure 1. Then these tubes will be subjected for first centrifugation at 2400 rpm for 10 minutes. The upper layer with buffy coat, will be subjected for second centrifugation at 1200 rpm for 10 minutes in plain tubes. The final resultant contains 4-5 mL of PB-MSCs.
Intra-articular PB-MSCs injections will be performed using an 18-gauge needle through a superolateral approach, underneath the supralateral surface of the patella, aimed toward the centre of the patella, and then directed slightly posteriorly and inferomedial into the knee joint. Subchondral PB-MSCs injections will be performed by inserting two 4 mm cannulated bone biopsy needles through the superomedial and anteromedial site, under fluoroscopic control, into the bone of both medial femoral condyle and tibial plateau respectively.
Following acquisition of a plain radiograph and MRI cartigram of the involved knee, patients will receive a detailed explanation of the intervention. Written informed consent will then be obtained. Prior to the procedure, patients will be instructed in active quadriceps strengthening exercises.
Participants will be allocated into three groups as follows: Group A (n = 15): A single dose of 4 mL to 5 mL of PB-MSC injectate will be administered intra-articularly into each knee on day 0 under strict aseptic precautions. Group B (n = 15): A single dose of 4 mL to 5 mL of PB-MSC injectate will be delivered via subchondral injection into each knee on day 0 under sterile conditions. Group C (n = 15): A single dose of 4 mL to 5 mL of PB-MSC injectate will be administered using a combined intra-articular and subchondral approach into each knee on day 0 under aseptic conditions.
Upon completion of the injection, a sterile dressing and crepe bandage will be applied. Approximately ten minutes after the procedure, patients will undergo gentle knee mobilization to facilitate uniform dispersion of the injectate within the joint. Instruction will be provided for a home-based program focusing on active quadriceps activation and knee strengthening exercises. Partial weight bearing will be permitted immediately after the intervention. Postprocedural discomfort will be managed with ice pack application, and the use of nonsteroidal anti-inflammatory drugs and other analgesics will be discouraged in all participants.
Once PB-MSCs will be injected into the knee joint and subchondral region for osteoarthritis, the patients will be advised to follow the post-procedural protocol as follows: (1) Give rest, ice, compression, and elevation therapy and supplement collagen; (2) Active range of motion of knee; (3) Active Quadriceps exercises; (4) Partial weight bearing up to 7th post-operative day; (5) Full weight bearing at the end of 1st week; (6) Regular follow-up radiographs at the end of 1 month, 3 months, 6 months, and 12 months; (7) Functional scoring to be done pre-procedurally and post-procedurally at the end of 1 month, 3 months, 6 months, and 12 months with VAS, KOOS, WOMAC, and International Knee Documentation Committee (IKDC) scores[16-19]; and (8) Plan for MRI cartigram of the knee to check the cartilage thickness after 1 year which will be documented with Magnetic Resonance Observation of Cartilage Repair Tissue scores.
Data analysis will be performed using the SPSS version 26 (IBM Corp., Chicago, IL, United States). Both intentions to treat and per protocol analyses will be undertaken to manage missing observations and participant attrition. This dual approach will preserve the benefits of randomization while also evaluating treatment efficacy among participants who adhere fully to the study protocol. Within-group changes in outcome measures will be examined by comparing baseline scores with post-intervention values at 1 month, 2 months, 6 months, and 12 months using paired t tests for the VAS, KOOS, WOMAC, and IKDC scales. Between-group comparisons of continuous variables will be conducted using the Kruskal-Wallis test, while longitudinal intra-group differences will be assessed using the Friedman test. For outcomes demonstrating normal distribution, repeated-measures analysis of variance with Bonferroni-adjusted post hoc testing will be applied. In cases of non-normal data distribution, the Friedman test followed by pairwise Wilcoxon signed-rank comparisons will be used. Categorical variables will be analyzed using Fisher’s exact test. A P-value of less than 0.05 will be considered statistically significant.
Primary outcome: Change in VAS pain scores at 12 months following the injection.
Secondary outcomes: (1) Evaluation of functional improvement using KOOS, WOMAC, and IKDC scores at predetermined follow-up intervals; (2) Assessment of cartilage regeneration through MRI cartilage imaging at 12 months; (3) Monitoring and recording of any treatment-related adverse events; (4) Measurement of patient satisfaction using standardized questionnaires; (5) Evaluation of quality-of-life changes based on EuroQol-5 dimensions scores; and (6) Quantification of patient satisfaction at each follow-up using a 5-point Likert scale (from “very dissatisfied” to “very satisfied”).
Cartilage integrity will be evaluated using T2 mapping, a validated MRI-based technique that quantitatively assesses cartilage health[20]. This method measures the T2 relaxation time within the cartilage, providing information about its biochemical properties, including water content and collagen fiber structure. The use of T2 mapping is justified by its sensitivity in detecting early degenerative changes, making it particularly valuable for assessing cartilage alterations in KOA.
The findings from this study will be published in high-impact, peer-reviewed medical journals and presented at national and international conferences in the fields of orthopedics and regenerative medicine. In addition, comprehensive reports and summaries will be shared with study participants and relevant healthcare stakeholders. Participating institutions will receive detailed reports, and the results will be disseminated to inform and guide future clinical practice.
Articular hyaline cartilage is a specialized connective tissue located in load-bearing diarthrodial joints, providing a smooth, low-friction surface that cushions impact and protects the underlying bone during movement[18]. Due to its avascular and alymphatic nature, this tissue has a limited capacity for self-repair, making regeneration after injury particularly challenging[19,20]. osteoarthritis and traumatic injuries are both common and costly conditions that frequently cause full-thickness cartilage defects incapable of effective healing, often resulting in chronic disability[21]. When conservative management fails, surgical interventions such as arthroscopic microfracture, autologous chondrocyte implantation, high tibial osteotomy, or partial knee replacement are employed; however, these procedures generally achieve only partial restoration of functional cartilage[22,23].
Over the past twenty years, cell-based therapies have evolved toward strategies utilizing MSCs for cartilage regeneration, offering the potential for effective single-stage surgical interventions. These mesoderm-derived progenitor cells represent a valuable source of adult stem cells because of their multipotent differentiation capacity and low immunogenic profile, making them highly suitable for regenerative medicine applications[24,25]. Morphologically, MSCs exhibit a fibroblast-like appearance and are characterized by their ability to self-renew and differentiate into various mesodermal lineages, including osteoblasts (bone cells), adipocytes (fat cells), chondrocytes (cartilage cells), and myocytes (muscle cells)[26,27]. Furthermore, MSCs have demonstrated the potential to differentiate into non-mesodermal cell types, such as neurons and astrocytes[28,29].
Emerging evidence suggests that PB can serve as a practical alternative source of MSCs, exhibiting chondrogenic differentiation potential comparable to that of bone marrow-derived MSCs both in vitro and animal studies[30,31]. Stem cells obtained from PB-MSCs can be collected using a minimally invasive procedure, which carries a lower risk profile than bone marrow extraction - a process associated with complications such as bleeding, prolonged pain, nerve or vascular injury, and, in rare cases, mortality[32]. Additionally, PB-MSCs are suitable for autologous transplantation, providing clear clinical advantages and facilitating the development of single-stage surgical procedures and other cell-based therapies[12].
Human MSCs represent a promising autologous cell source for repairing extensive osteoarthritic cartilage defects and can be guided toward chondrogenic differentiation through targeted biochemical and mechanical cues. Hydrogels are particularly attractive as cell delivery vehicles due to their elasticity and high-water content, which facilitate extracellular matrix formation and efficient nutrient transport while encapsulating cells within their cross-linked network[33-35]. Their biodegradable nature allows for cell migration and the gradual replacement of the scaffold with newly formed tissue. In hydrogels made from natural materials such as collagen and fibrin, degradation occurs through cellular activity, and these materials inherently possess molecular motifs that support cell adhesion and biological recognition[36-38].
Our study design is strengthened by several key features: (1) Randomized controlled design: The use of a doubleblind, randomized allocation minimizes selection bias and confounding variables, thereby strengthening the internal validity of the study; (2) Comprehensive outcome assessment: Primary outcomes, such as pain measured by VAS, are complemented by multiple secondary endpoints - including functional scores, cartilage regeneration assessed via MRI, and the incidence of adverse events - providing a holistic evaluation of both efficacy and safety; (3) Standardized intervention protocols: Uniform, well-defined procedures for administering stromal vascular fraction and nanofat ensure consistency in treatment delivery, enhancing reproducibility and reliability of results; and (4) Ethical rigor: Adherence to ethical principles, including informed consent and strict data confidentiality, safeguards participant rights and reinforces research integrity.
A key strength of this study lies in its double-blind design, which effectively reduces both observer and participant bias in subjective measures such as VAS pain scores. The implementation of a robust randomization strategy - utilizing a permuted block algorithm and neutral syringe labeling - prevents allocation imbalances and eliminates the possibility of predicting group assignments by patients or clinical staff. Through meticulous masking of the injectate and strict allocation concealment, the study achieves a high degree of internal validity.
Despite its strengths, the study has several limitations: (1) Sample size: The inclusion of 150 participants may be insufficient to detect smaller effect sizes and could limit the generalizability of findings across diverse populations. Larger, more extensive studies are required to validate and expand upon these results; (2) Follow-up duration: An 18-month follow-up period may not adequately capture long-term outcomes or delayed adverse events. Future research with extended followup would provide a more comprehensive understanding of treatment durability; and (3) Single-center design: Conducting the trial within a single institution may restrict the applicability of results to other clinical environments with varying patient demographics and treatment practices.
Additionally, a notable limitation involves the difficulty of masking interventions that differ in appearance, texture, or color. Despite the use of opaque syringe covers and standardized injection protocols, subtle visual or tactile cues could inadvertently unblind clinicians or participants. While the randomization strategy helps mitigate selection bias, larger multi-center trials would strengthen external validity and better reflect real-world patient populations.
Based on the results of this study, future research could focus on the following directions: Larger, multi-center trials: Expanding the sample size and involving multiple clinical sites would enhance the generalizability and robustness of findings, providing a clearer understanding of PB-MSCs effectiveness across diverse patient populations. Extended follow-up: Longer monitoring beyond 18 months is needed to assess sustained efficacy and detect delayed adverse events, thereby ensuring long-term safety and therapeutic durability. Mechanistic studies: Investigating the molecular and cellular pathways underlying the regenerative effects of PB-MSCs could elucidate their mechanisms of action, refine treatment protocols, and potentially improve clinical outcomes. Comparative effectiveness research: Direct comparisons of PB-MSCs with other regenerative modalities - such as plateletrich plasma, bone marrow aspirate concentrate, stromal vascular fraction, and nanofat - would help establish their relative clinical value. Incorporating objective biomarkers (e.g., serum or synovial fluid inflammatory markers) could further connect clinical improvements to tissuelevel changes, deepening mechanistic insights. Protocol standardization: Developing universally accepted methods for PB-MSCs isolation and preparation would reduce variability, ensure reproducible therapeutic results, and facilitate broader clinical adoption.
PB-MSCs represent a promising regenerative approach for managing primary KOA, a condition associated with pain, functional decline, and reduced quality of life. This controlled clinical trial bridges a crucial evidence gap by evaluating the safety and efficacy of PB-MSCs in promoting cartilage repair and symptom relief. Through robust methodology, comprehensive outcome assessments, and ethical rigor, the study aims to guide optimal therapeutic strategies and improve patient outcomes. Its findings will advance orthopedic regenerative medicine, yet further research is essential. Standardizing PB-MSC dosage, injection frequency, and preparation protocols will enhance consistency. Comparative studies of autologous vs allogeneic formulations are needed to optimize efficacy, while advanced imaging can deepen understanding of cartilage regeneration. Exploring scalable allogeneic options may improve accessibility and broaden clinical adoption. Together, these efforts will refine evidence-based practices and expand the therapeutic potential of regenerative orthopedics.
| 1. | Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393:1745-1759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3763] [Cited by in RCA: 3189] [Article Influence: 455.6] [Reference Citation Analysis (3)] |
| 2. | Ren JL, Yang J, Hu W. The global burden of osteoarthritis knee: a secondary data analysis of a population-based study. Clin Rheumatol. 2025;44:1769-1810. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 3. | Ma W, Chen H, Yuan Q, Chen X, Li H. Global, regional, and national epidemiology of osteoarthritis in working-age individuals: insights from the global burden of disease study 1990-2021. Sci Rep. 2025;15:7907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 4. | Shumnalieva R, Kotov G, Monov S. Obesity-Related Knee Osteoarthritis-Current Concepts. Life (Basel). 2023;13:1650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 5. | Han X, Liao R, Li X, Zhang C, Huo S, Qin L, Xiong Y, He T, Xiao G, Zhang T. Mesenchymal stem cells in treating human diseases: molecular mechanisms and clinical studies. Signal Transduct Target Ther. 2025;10:262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 100] [Article Influence: 100.0] [Reference Citation Analysis (0)] |
| 6. | Wong RSY, Tan EW, Goh BH. Mesenchymal Stem Cell-Based Therapies: Challenges and Enhancement Strategies. Cell Biochem Biophys. 2025;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 7. | Wu KC, Chang YH, Ding DC, Lin SZ. Mesenchymal Stromal Cells for Aging Cartilage Regeneration: A Review. Int J Mol Sci. 2024;25:12911. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 8. | Lin J, Huang J, Jiao Z, Nian M, Li C, Dai Y, Jia S, Zhang X. Mesenchymal stem cells for osteoarthritis: Recent advances in related cell therapy. Bioeng Transl Med. 2025;10:e10701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 9. | Zhang K, Xu T, Xie H, Li J, Fu W. Donor-Matched Peripheral Blood-Derived Mesenchymal Stem Cells Combined With Platelet-Rich Plasma Synergistically Ameliorate Surgery-Induced Osteoarthritis in Rabbits: An In Vitro and In Vivo Study. Am J Sports Med. 2023;51:3008-3024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 10. | Lin W, Yang Z, Shi L, Wang H, Pan Q, Zhang X, Zhang P, Lin S, Li G. Alleviation of osteoarthritis by intra-articular transplantation of circulating mesenchymal stem cells. Biochem Biophys Res Commun. 2022;636:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 11. | Ouryazdanpanah N, Dabiri S, Derakhshani A, Vahidi R, Farsinejad A. Peripheral Blood-Derived Mesenchymal Stem Cells: Growth Factor-Free Isolation, Molecular Characterization and Differentiation. Iran J Pathol. 2018;13:461-466. [PubMed] |
| 12. | Chen YR, Yan X, Yuan FZ, Ye J, Xu BB, Zhou ZX, Mao ZM, Guan J, Song YF, Sun ZW, Wang XJ, Chen ZY, Wang DY, Fan BS, Yang M, Song ST, Jiang D, Yu JK. The Use of Peripheral Blood-Derived Stem Cells for Cartilage Repair and Regeneration In Vivo: A Review. Front Pharmacol. 2020;11:404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 13. | Saw KY, Anz A, Siew-Yoke Jee C, Merican S, Ching-Soong Ng R, Roohi SA, Ragavanaidu K. Articular cartilage regeneration with autologous peripheral blood stem cells versus hyaluronic acid: a randomized controlled trial. Arthroscopy. 2013;29:684-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 186] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 14. | Saw KY, Anz A, Merican S, Tay YG, Ragavanaidu K, Jee CS, McGuire DA. Articular cartilage regeneration with autologous peripheral blood progenitor cells and hyaluronic acid after arthroscopic subchondral drilling: a report of 5 cases with histology. Arthroscopy. 2011;27:493-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 123] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 15. | Turajane T, Chaveewanakorn U, Fongsarun W, Aojanepong J, Papadopoulos KI. Avoidance of Total Knee Arthroplasty in Early Osteoarthritis of the Knee with Intra-Articular Implantation of Autologous Activated Peripheral Blood Stem Cells versus Hyaluronic Acid: A Randomized Controlled Trial with Differential Effects of Growth Factor Addition. Stem Cells Int. 2017;2017:8925132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 16. | Spaas JH, De Schauwer C, Cornillie P, Meyer E, Van Soom A, Van de Walle GR. Culture and characterisation of equine peripheral blood mesenchymal stromal cells. Vet J. 2013;195:107-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 17. | Cuschieri S. The CONSORT statement. Saudi J Anaesth. 2019;13:S27-S30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 406] [Cited by in RCA: 315] [Article Influence: 45.0] [Reference Citation Analysis (1)] |
| 18. | Krakowski P, Rejniak A, Sobczyk J, Karpiński R. Cartilage Integrity: A Review of Mechanical and Frictional Properties and Repair Approaches in Osteoarthritis. Healthcare (Basel). 2024;12:1648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 100] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 19. | Karuppal R. Current concepts in the articular cartilage repair and regeneration. J Orthop. 2017;14:A1-A3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 92] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 20. | Guo X, Xi L, Yu M, Fan Z, Wang W, Ju A, Liang Z, Zhou G, Ren W. Regeneration of articular cartilage defects: Therapeutic strategies and perspectives. J Tissue Eng. 2023;14:20417314231164765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 65] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 21. | Nazem K, Safdarian A, Fesharaki M, Moulavi F, Motififard M, Zarezadeh A, Shakibaei M, Esfandiari E, Nasr-Esfahani MH. Treatment of full thickness cartilage defects in human knees with Autologous Chondrocyte Transplantation. J Res Med Sci. 2011;16:855-861. [PubMed] |
| 22. | Gracitelli GC, Moraes VY, Franciozi CE, Luzo MV, Belloti JC. Surgical interventions (microfracture, drilling, mosaicplasty, and allograft transplantation) for treating isolated cartilage defects of the knee in adults. Cochrane Database Syst Rev. 2016;9:CD010675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 23. | Jung SH, Jung M, Chung K, Moon HS, Kim TH, Choi CH, Kim SH. Clinical Benefits of Cartilage Repair in High Tibial Osteotomy Can Only Be Expected in Patients with Successfully Regenerated Cartilage. Clin Orthop Surg. 2025;17:607-621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Della Rocca Y, Mazzone A, Marconi GD, Trubiani O, Pizzicannella J, Diomede F. Stem Cells in Regenerative Medicine: A Journey from Adult Stem Cells to Induced Pluripotent Cells. Int J Mol Sci. 2025;26:8255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 25. | Camia B, Monti M. From Biological Waste to Therapeutic Resources: A Comprehensive Review of Stem Cell Sources, Characterization, and Biomedical Potentials. Stem Cell Rev Rep. 2026;22:5-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 26. | Tanaka K, Ogino R, Yamakawa S, Suda S, Hayashida K. Role and Function of Mesenchymal Stem Cells on Fibroblast in Cutaneous Wound Healing. Biomedicines. 2022;10:1391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 27. | Rastegar F, Shenaq D, Huang J, Zhang W, Zhang BQ, He BC, Chen L, Zuo GW, Luo Q, Shi Q, Wagner ER, Huang E, Gao Y, Gao JL, Kim SH, Zhou JZ, Bi Y, Su Y, Zhu G, Luo J, Luo X, Qin J, Reid RR, Luu HH, Haydon RC, Deng ZL, He TC. Mesenchymal stem cells: Molecular characteristics and clinical applications. World J Stem Cells. 2010;2:67-80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 167] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 28. | George S, Hamblin MR, Abrahamse H. Differentiation of Mesenchymal Stem Cells to Neuroglia: in the Context of Cell Signalling. Stem Cell Rev Rep. 2019;15:814-826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 29. | Hernández R, Jiménez-Luna C, Perales-Adán J, Perazzoli G, Melguizo C, Prados J. Differentiation of Human Mesenchymal Stem Cells towards Neuronal Lineage: Clinical Trials in Nervous System Disorders. Biomol Ther (Seoul). 2020;28:34-44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 100] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 30. | Lotfy A, El-Sherbiny YM, Cuthbert R, Jones E, Badawy A. Comparative study of biological characteristics of mesenchymal stem cells isolated from mouse bone marrow and peripheral blood. Biomed Rep. 2019;11:165-170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 31. | Wang P, Zhang S, Meng Q, Zhu P, Yuan W. Treatment and application of stem cells from different sources for cartilage injury: a literature review. Ann Transl Med. 2022;10:610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 32. | Fu WL, Li J, Chen G, Li Q, Tang X, Zhang CH. Mesenchymal Stem Cells Derived from Peripheral Blood Retain Their Pluripotency, but Undergo Senescence During Long-Term Culture. Tissue Eng Part C Methods. 2015;21:1088-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 33. | Xu B, Ye J, Yuan FZ, Zhang JY, Chen YR, Fan BS, Jiang D, Jiang WB, Wang X, Yu JK. Advances of Stem Cell-Laden Hydrogels With Biomimetic Microenvironment for Osteochondral Repair. Front Bioeng Biotechnol. 2020;8:247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 34. | Segneanu AE, Bejenaru LE, Bejenaru C, Blendea A, Mogoşanu GD, Biţă A, Boia ER. Advancements in Hydrogels: A Comprehensive Review of Natural and Synthetic Innovations for Biomedical Applications. Polymers (Basel). 2025;17:2026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 103] [Article Influence: 103.0] [Reference Citation Analysis (0)] |
| 35. | Revete A, Aparicio A, Cisterna BA, Revete J, Luis L, Ibarra E, Segura González EA, Molino J, Reginensi D. Advancements in the Use of Hydrogels for Regenerative Medicine: Properties and Biomedical Applications. Int J Biomater. 2022;2022:3606765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 124] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 36. | Solbu AA, Caballero D, Damigos S, Kundu SC, Reis RL, Halaas Ø, Chahal AS, Strand BL. Assessing cell migration in hydrogels: An overview of relevant materials and methods. Mater Today Bio. 2023;18:100537. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 71] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 37. | Cao H, Duan L, Zhang Y, Cao J, Zhang K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal Transduct Target Ther. 2021;6:426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 989] [Cited by in RCA: 710] [Article Influence: 142.0] [Reference Citation Analysis (1)] |
| 38. | Xu F, Dawson C, Lamb M, Mueller E, Stefanek E, Akbari M, Hoare T. Hydrogels for Tissue Engineering: Addressing Key Design Needs Toward Clinical Translation. Front Bioeng Biotechnol. 2022;10:849831. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 92] [Article Influence: 23.0] [Reference Citation Analysis (0)] |