Published online Apr 18, 2026. doi: 10.5312/wjo.v17.i4.117533
Revised: January 25, 2026
Accepted: February 28, 2026
Published online: April 18, 2026
Processing time: 122 Days and 2.6 Hours
Myxofibrosarcoma (MFS) is a rare soft-tissue tumor composed mainly of fib
Here, we report a case involving a giant neoplasm at the thoracoabdominal jun
3D-printed titanium prostheses combined with bilateral femoral anterolateral thi
Core Tip: This case report details the management of a massive thoracoabdominal wall soft tissue sarcoma using a multidisciplinary, collaborative approach combined with medicine-engineering integration. Following meticulous perioperative optimization, three-dimensional printed titanium alloy prostheses and free bilateral anterolateral thigh myocutaneous flaps were used to reconstruct the extensive bony and soft tissue defects of the chest wall, respectively, with highly satisfactory short-term postoperative outcomes. Notably, the combined thoracoabdominal wall defect at the surgical site measured 750 cm2, which, to the best of our knowledge, represents the largest reported repair of a thoracoabdominal wall defect secondary to soft tissue sarcoma to date.
- Citation: Li XA, Chen JY, Bu J, Wu HW. Customized three-dimensional printed titanium ribs combined with bilateral flaps for reconstruction of chest wall defect: A case report. World J Orthop 2026; 17(4): 117533
- URL: https://www.wjgnet.com/2218-5836/full/v17/i4/117533.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i4.117533
Extensive chest wall defects are usually caused by tumors, infections, or trauma. Myxofibrosarcoma (MFS) is characterized by a high incidence of local recurrence and distant metastasis. It most commonly occurs in the extremities of older patients and rarely in the chest wall[1,2]. Radical resection with wide tumor-free margins remains the cornerstone of treatment for MFS[3-6]. Given the high mortality and morbidity associated with MFS, achieving optimal resection and reconstruction poses a major challenge when the tumor involves the chest wall. For soft tissue reconstruction, large chest wall defects caused by malignant tumors can usually be repaired using a free anterolateral thigh flap or a free deep inferior epigastric perforator flap when local flaps are insufficient to meet reconstructive requirements[7].
However, controversy remains regarding the most advantageous approach to osseous chest wall reconstruction, and the choice of prosthetic material largely remains at the surgeon’s discretion. With the development of new materials and reconstructive technologies, particularly custom-made prostheses produced using three-dimensional (3D) printing, postoperative outcomes have improved substantially. A variety of materials, including silicone-based implants, polydioxanone mesh, titanium alloy plates, polypropylene mesh, and fascia lata, have recently been used for chest wall recon
The principle of biomimesis involves respecting anatomy, preserving function, selecting appropriate reconstructive materials, and integrating multidisciplinary efforts for complex reconstructions[12]. Biomimesis has become a promising reconstructive strategy after chest wall resection for primary or secondary tumors. The reconstruction technique should fulfill two opposing functional requirements: Adequate rigidity and flexibility[9,13]. Balancing these parameters remains a significant challenge. Here, we report a case in which 3D custom-made titanium ribs and bilateral anterolateral thigh musculocutaneous flaps were combined to reconstruct a chest wall defect. To our knowledge, this is the first case report describing the combination of these two biotechnologies to mimic both the osseous and soft tissue components of the chest wall. This case may contribute to the growing body of knowledge regarding the clinical presentation, diagnostic workup, and therapeutic strategies for this disease.
Right chest wall mass, 12 years post-operation, with the last recurrence 6 months ago.
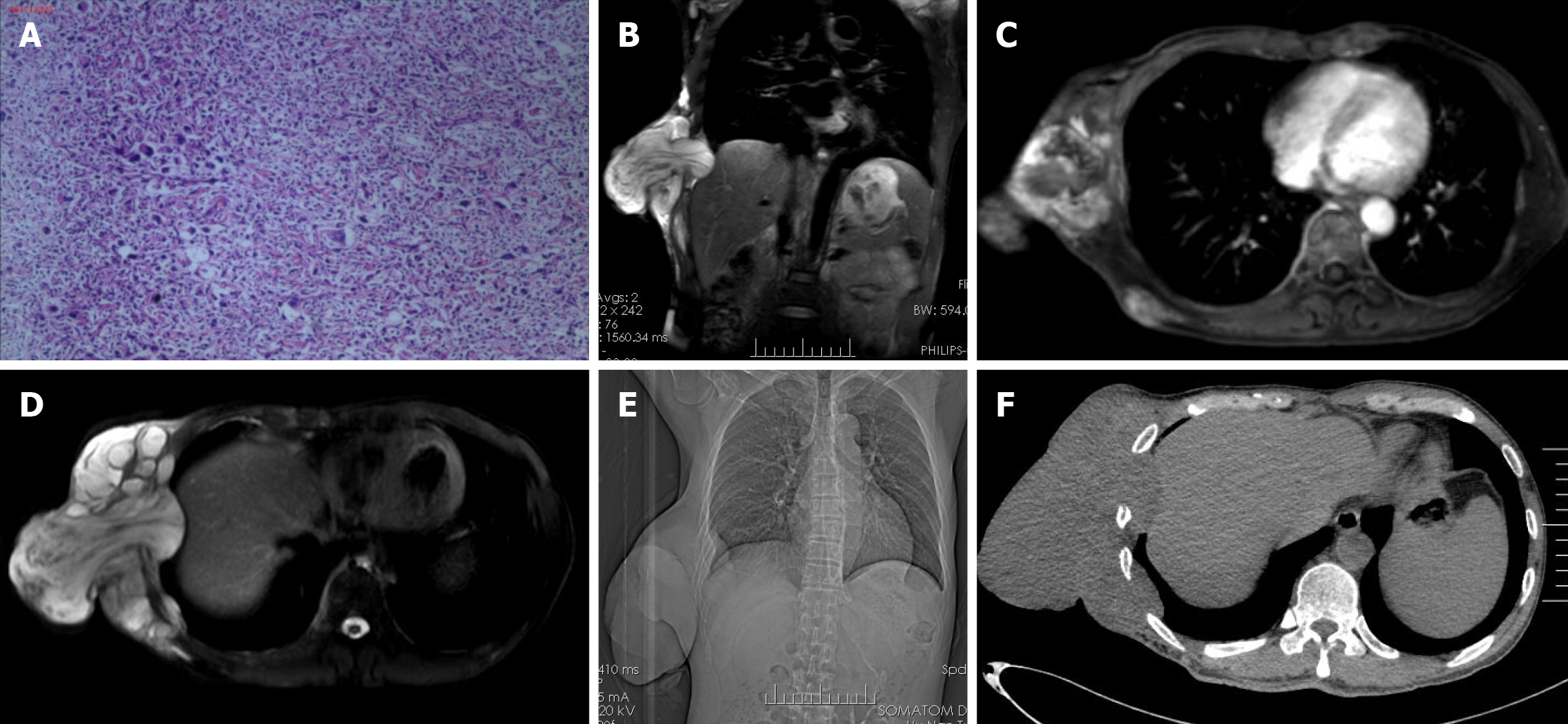
A 56-year-old male patient presented with a bean-sized mass on his right lower chest wall. The mass was primarily observed in 2006 and resected in a local hospital. The resected mass was pathologically confirmed as low-grade MFS at Hunan Cancer Hospital. The patient did not receive subsequent treatment. In 2008, the mass reappeared and was resected again in the same hospital. The second pathology diagnosis was comparable to the initial diagnosis (Figure 1A). After that, the patient experienced multiple surgeries to treat the mass as before. During multiple treatments, the lesion's border gradually extended, and its size increased rapidly. Upon this hospitalization, the large mass protruded out of the chest wall, and ulceration formed with heavy exudation.
No special history of past illness.
No special family history.
The patient developed a symptom complex of severe hypoproteinemia, anemia, and malnutrition due to massive and long-term tumor ulcer exudation.
Routine blood and liver function tests showed lower hemoglobin content (66 g/L), higher white blood cell content (15.19 × 109/L), lower albumin content (24.9 g/L), and higher high-sensitivity C-reactive protein content (69 mg/L). Although vital signs were normal, these data indicate the patient developed severe malnutrition and an infection.
The results of magnetic resonance imaging (MRI) and computed tomography (CT) suggested that the lesions had invaded the chest wall and ribs. Multiple nodules, suspected as metastatic lesions, were found in both lungs (Figure 1B-F). Furthermore, two enlarged lymph nodes were detected in the right auxiliary fossa by ultrasonography, and malignant cells were identified by needle biopsy. Although an obvious abnormality was not observed in the abdominal organs by ultrasonography, radioactive accumulation in the right 6th-8th ribs was detected by bone scanning and was suspected to be a metastatic lesion.
A comprehensive discussion of this case was conducted by a multidisciplinary team, including experts in Orthopaedic and Soft Tissue Oncology, Radiation Oncology, Medical Oncology, Anesthesiology, Radiology, Transfusion Medicine, Microsurgical Reconstructive Surgery, Pathology, and Critical Care Medicine. The team recommended thorough communication with the patient and his family to explain the disease status, clarify the necessity of surgery and its potential risks, and outline the expected surgical benefit and long-term prognosis. The primary objectives were complete resection of the lesion, reduction of tumor burden, improvement of quality of life, and support of the patient’s confidence during rehabilitation.
First, meticulous preoperative preparation was imperative, including nutritional support to correct a negative nitrogen balance, maximal efforts to mitigate the risk of infection at the tumor site, and fractional transfusions of whole blood and plasma to correct anemia and hypoalbuminemia. Second, intraoperatively, adequate units of red blood cells and plasma were prepared in advance. The surgical procedure included extensive tumor resection, reconstruction of the bony thoracic cage using 3D-printed titanium alloy rib prostheses, and repair of the soft tissue defect with free bilateral anterolateral thigh myocutaneous flaps. Postoperatively, a series of targeted interventions were implemented to promote early wound healing and rehabilitation, including adequate wound drainage, correction of water and electrolyte disturbances, management of anemia and hypoalbuminemia, prophylactic anti-infective therapy for the surgical site, and intensive nutritional support.
The patient was diagnosed with MFS.
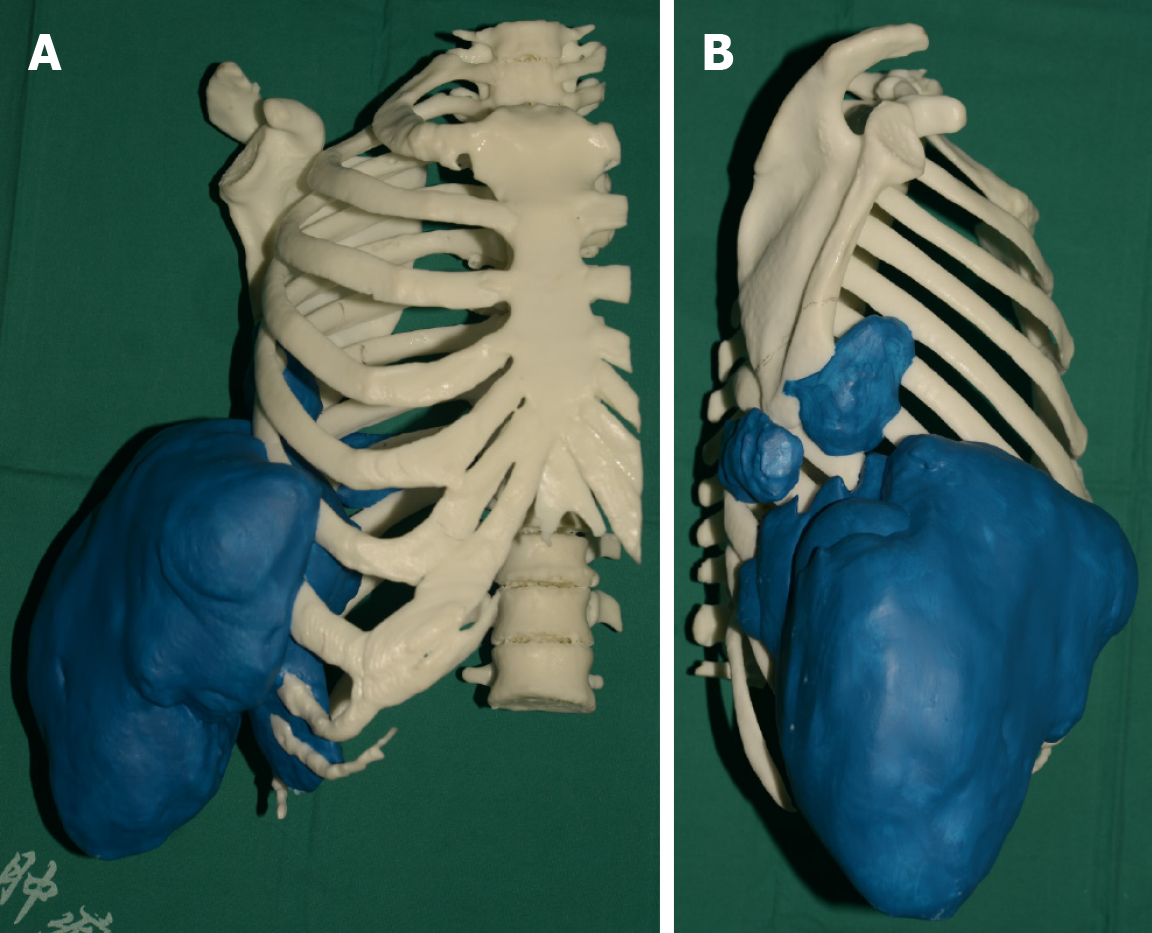
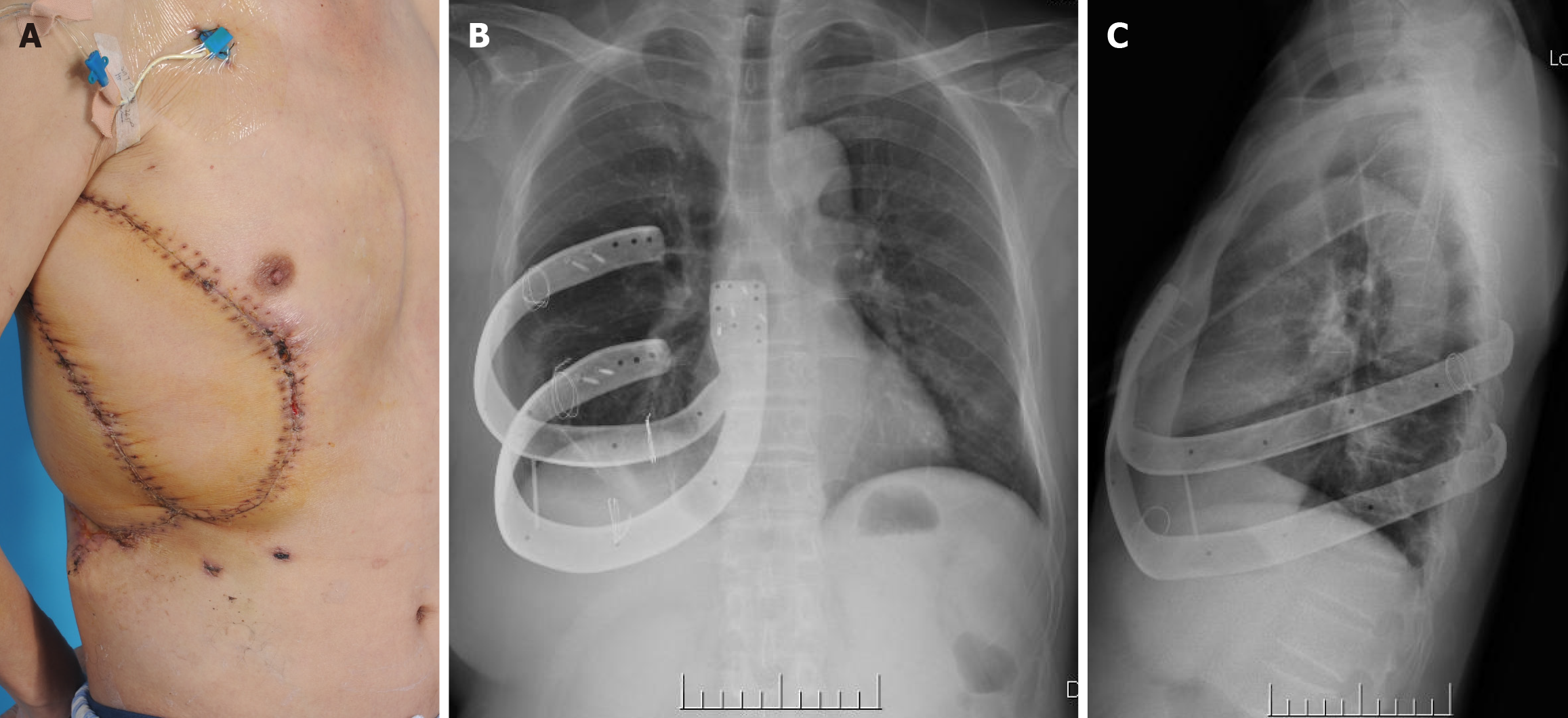
To overcome the challenge of having a massive defect of the chest-abdomen wall and exposure of internal organs after tumor resection, including thoracic defects and massive skin and soft tissue elimination, orthopedic, head, and neck microsurgery, and thoracic surgeons developed a combined solution. This was accomplished by reconstructing the stable and integral thoracic cavity with a 3D metal printing thoracic consisting of two ribs and a superior segment of sternum (Figure 2), as well as reconstructing the skin and soft tissue with two anterolateral thigh musculocutaneous flaps from the bilateral thigh.
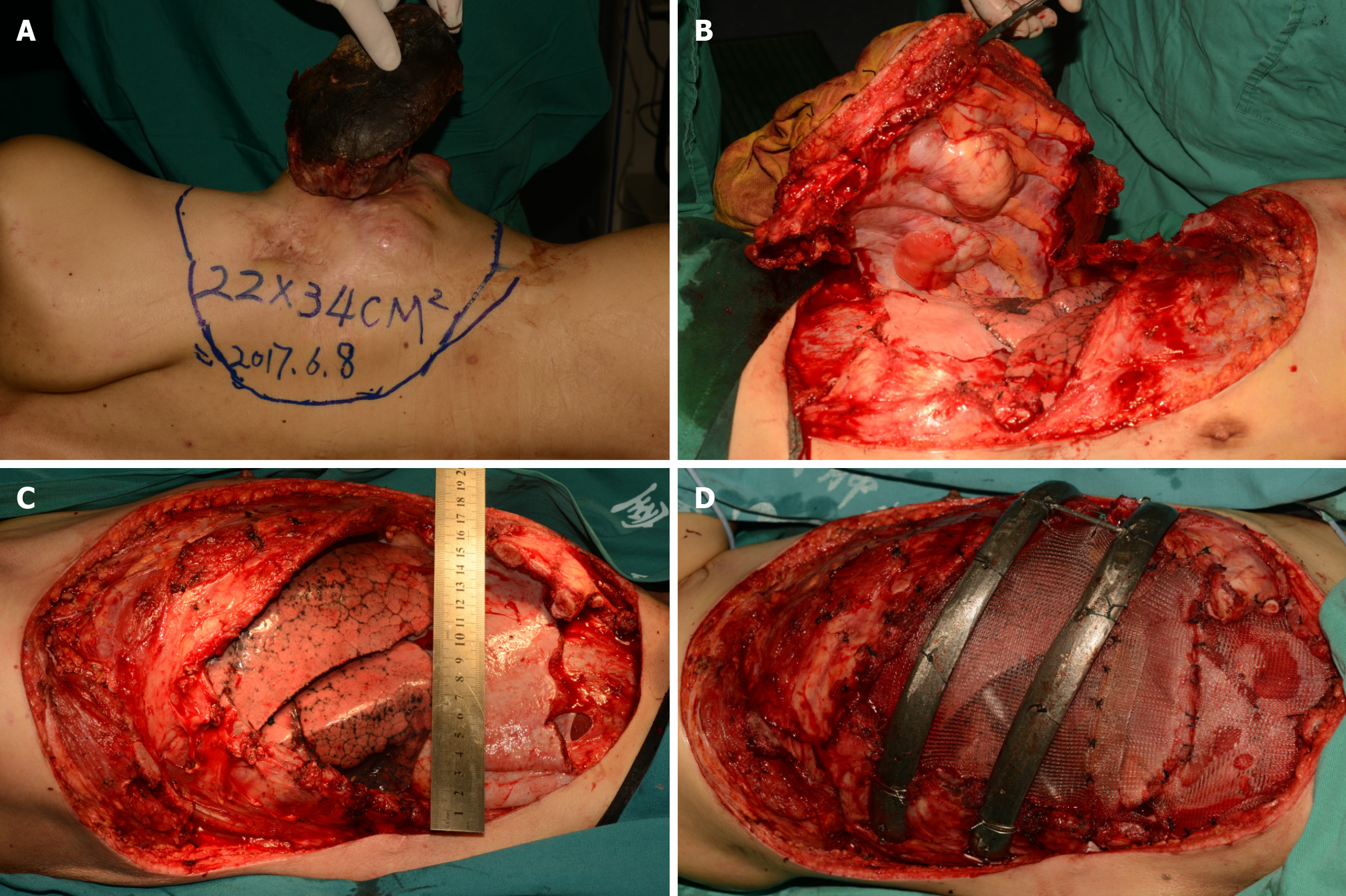
To maintain a relatively aseptic environment, the ulcer wound dressings were changed every day before surgery. The patient’s albumin level and anti-infection capacity were supported by discontinuous transfusion of packed red blood cells and plasma. Prior to surgery, hemoglobin should be above 90 g/L, and albumin should be above 30 g/L. During general anesthesia, the patient was placed in the left lateral position. An extended round-shaped incision was carried out 3 cm away from the tumor edge via a combined thoracic and abdominal approach (Figure 3A). Lesions distributed in the skin, chest wall muscle, inferior angle of the scapula, parts of the 5th-12th ribs, and part of the diaphragm and liver capsular were all completely removed (Figure 3B). Fortunately, though the capsular could not be distinguished from the images (Figure 3B), it appeared intact and without obvious invasion. The resected lesion was 33 cm × 28 cm × 15 cm in size (Figure 3C). The small lung lesion was also resected during the routine thoracic exploration. Axillary lymph nodes were cleaned up by lengthening the incision up to the axillary fossa. Then, the deficient pleura and the liver capsular were repaired by sewing a nylon mesh along the edge of the left capsular. The 3D printed metal rib prosthesis was implanted and fixed with matched screws and metal wire to the other end of the left normal thoracic (Figure 3D). The defect area was approximately 22 cm × 34 cm.
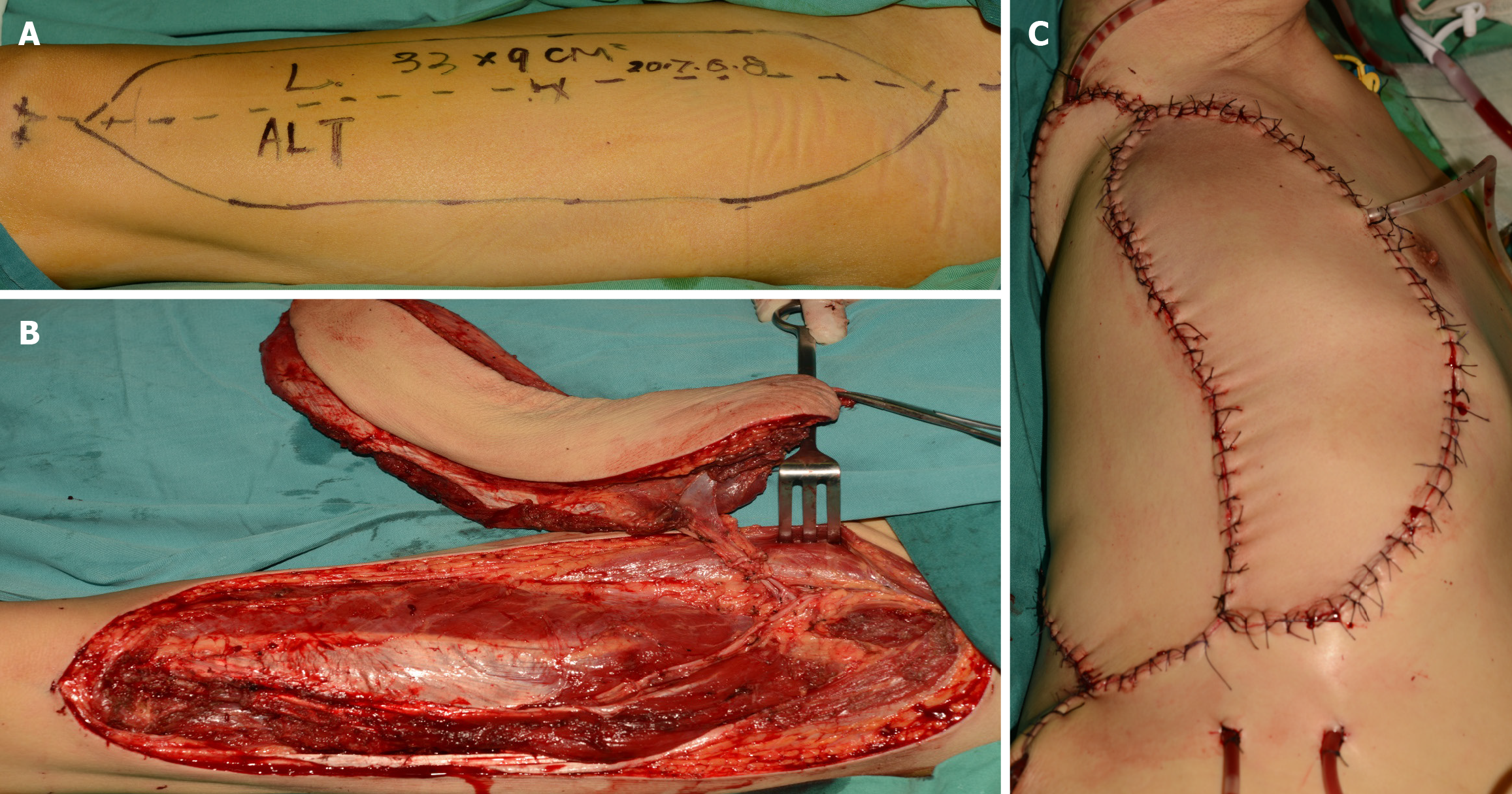
Next, the subscapular artery, vein, and subscapular vein were dissected for vascular anastomosis. An anterolateral thigh musculocutaneous flap with a size of 33 cm × 9 cm and lateral femoral vessels was dissected from the right thigh and then preserved in the heparin-saline (Figure 4A and B). After that, the patient was placed in a supine position, and the other anterolateral thigh musculocutaneous flap of the same size was prepared in the same way. Both flaps were transplanted onto the chest wall wound and anastomosed via the vascular pedicle in an end-to-end manner. One artery and two veins were anastomosed for each flap. After confirming patency of the blood circulation, the two flaps were sutured to the defect site (Figure 4C). Five vacuum drainages were placed beneath the flaps until exudation decreased to minimal levels.
Pathological results confirmed the lesion (18 cm × 14 cm × 9 cm) as recurrent MFS (Figure 1A). Microscopic images depicted common characteristics of MFS, including dominant tissue structural distortion in fibrous and histiocyte cells, emergence of heteromultinuclear giant cells, high nucleus/cytoplasm ratio, and sporadic inflammation cells. Though adjacent to the lesion, no infiltration was detected in the ribs. In addition to the inflammatory changes caused by fibrosis calcification in the right lung nodule, no metastatic lesion was detected in the auxiliary fossa lymph node. This indicates that no residual tumor was detected at the skin’s cutting edge or at the bottom, and was therefore a good prognostic sign.
The patient received antibiotics, fluid, and blood transfusions after the operation until a healthy internal environment and healthy flaps were confirmed. No postoperative complications such as bleeding, pneumothorax, abdominal infection, or vessel crisis or flap necrosis were displayed. The patient was in good condition and discharged 14 days after surgery, when the wound was confirmed to be well healed (Figure 5).
During the first 3 years after surgery, the patient remained asymptomatic, and no evidence of tumor recurrence was detected. However, he died of lung metastases 3 years later without further treatment.
This report describes a case of recurrent MFS presenting as a large mass on the right chest wall. MFS is a subtype of undifferentiated pleomorphic sarcoma. The most common site is the lower limb, followed by the upper limb and the retroperitoneum, and it is rarely encountered in the chest wall[14]. Owing to its rarity, MFS presents challenges in establishing standardized treatment protocols. This report shares our experience in the treatment and management of this disease, emphasizing meticulous surgical planning, multidisciplinary collaboration, and innovative reconstruction techniques as key factors in achieving optimal outcomes in patients with large chest wall MFS.
First, achieving a safe R0 surgical margin is essential to prevent recurrence of malignant tumors[15]. In this case, we used a 3D-printed poly-lactic acid tumor model to assist with preoperative planning of safe surgical margins. Second, the preoperative examinations showed limited invasion into adjacent tissues and structures; therefore, we recommended extensive resection.
Because of the large defect created by tumor resection, postoperative reconstruction of the chest and abdominal walls was vital to the success of the procedure. The objectives of chest wall reconstruction include restoring chest wall rigidity and protecting underlying organs, preventing pulmonary or cardiac herniation, preserving respiratory function, avoiding scapular trapping, and achieving an acceptable cosmetic outcome[16]. Strategies for reconstructing the osseous chest wall include prosthetic mesh and cement or metal prostheses. Mesh is advantageous because it is easy to manipulate, has good biocompatibility, and is relatively flexible, whereas bone cement is often preferred for skeletal thoracic reconstruction because of its mechanical strength. Metal implants are conventional materials for bone fixation. However, because of the irregular shape of the thorax, particularly the long rib segments, few metal prostheses are used for thoracic reconstruction[17,18]. In the early 21st century, 3D printing technology emerged and was widely adopted in oral and maxillofacial surgery, neurosurgery, and orthopedics[19]. This technology enables prostheses to be custom-made to match the geometry of the resected thoracic skeleton. 3D custom-made titanium ribs can provide a stable osseous thorax, effective protection of underlying organs, and smooth respiratory motion while maintaining an acceptable cosmetic appearance. In this case, we successfully reconstructed a complex chest wall defect by combining 3D custom-made titanium ribs with bilateral anterolateral thigh musculocutaneous flaps.
In recent years, the anterolateral thigh musculocutaneous flap has been regarded as one of the most versatile free flaps in clinical practice and is widely used for wound repair[20,21]. Owing to its many advantages, including a large skin paddle, a long vascular pedicle with a favorable caliber, relatively straightforward dissection, minimal donor-site mor
During 3 years of follow-up, no prosthesis displacement, infection, or respiratory complications were observed, in
Overall, this case illustrates the management of a complex soft tissue sarcoma through a multidisciplinary oncology team approach combined with medicine–engineering integration. It highlights the importance of prioritizing individual patient needs, meticulously evaluating each component of care, and formulating tailored therapeutic strategies.
A well-defined, precisely executed oncologic surgical margin is indispensable for preventing tumor recurrence and therefore requires preoperative, collaborative planning by a multidisciplinary team. Furthermore, 3D-printed prostheses enabled by medicine–engineering integration, in conjunction with microsurgical free flap transplantation, are pivotal for successful reconstruction of the thoracic cage and preservation of respiratory function. Accordingly, when managing similar cases, clinicians should actively leverage the synergistic strengths of multidisciplinary collaboration and cross-disciplinary medicine–engineering technologies to address the therapeutic challenges posed by large-volume tumors and thereby improve patients’ survival prospects.
In this case, a 3D-printed titanium prosthesis was successfully integrated with bilateral anterolateral thigh myocutaneous flaps to reconstruct a large thoracoabdominal wall defect caused by recurrent malignant tumors. Short-term follow-up demonstrated satisfactory functional outcomes without local recurrence, suggesting that this approach is a feasible option in clinical practice.
The authors acknowledgement the clinical support from Dr. Xue-Zheng Xu and Dr. Jian-Fan Liu.
| 1. | Pagnoni C, Improta L, Alloni R, Mallozzi Santa Maria F, Aprile I, Brunetti B, Greco C, Vincenzi B, Gronchi A, Valeri S. Recurrent neck myxofibrosarcoma: a case report. J Med Case Rep. 2021;15:484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 2. | Yoshida N, Miyanari N, Yamamoto Y, Egami H. Successful treatment of malignant fibrous histiocytoma originating in the chest wall: report of a case. Surg Today. 2006;36:714-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Zhong L, Zhang Y, Kang H, Wang Z. Giant myxofibrosarcoma of neck: A case report and review of the literature. Medicine (Baltimore). 2025;104:e42150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 4. | Teurneau H, Engellau J, Ghanei I, Vult von Steyern F, Styring E. High Recurrence Rate of Myxofibrosarcoma: The Effect of Radiotherapy Is Not Clear. Sarcoma. 2019;2019:8517371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 5. | Bagheri R, Haghi SZ, Kalantari MR, Sharifian Attar A, Salehi M, Tabari A, Soudaneh M. Primary malignant chest wall tumors: analysis of 40 patients. J Cardiothorac Surg. 2014;9:106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 6. | Fujiwara T, Stevenson J, Parry M, Tsuda Y, Tsoi K, Jeys L. What is an adequate margin for infiltrative soft-tissue sarcomas? Eur J Surg Oncol. 2020;46:277-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 7. | Hameed A, Akhtar S, Naqvi A, Pervaiz Z. Reconstruction of complex chest wall defects by using polypropylene mesh and a pedicled latissimus dorsi flap: a 6-year experience. J Plast Reconstr Aesthet Surg. 2008;61:628-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 46] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 8. | Wu Y, Zhang G, Zhu Z, Chai Y. Chest wall reconstruction after resection using hernia repair piece. J Thorac Dis. 2016;8:1353-1355. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Puviani L, Fazio N, Boriani L, Ruggieri P, Fornasari PM, Briccoli A. Reconstruction with fascia lata after extensive chest wall resection: results. Eur J Cardiothorac Surg. 2013;44:125-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Robinson LA, Grubbs DM. Using "Rebar" to Stabilize Rigid Chest Wall Reconstruction. Thorac Cardiovasc Surg. 2016;64:266-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 11. | Gunasekaran V, Chaturvedi G, Thanneeru SK, Gupta A, Ahmad R. Innovative Approach to Chest Wall Reconstruction Using Autologous Tissue - The Fascia Lata in Pediatric Patients. J Indian Assoc Pediatr Surg. 2024;29:381-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Rocco G. Chest wall resection and reconstruction according to the principles of biomimesis. Semin Thorac Cardiovasc Surg. 2011;23:307-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 13. | Huffman GR, Piper C, Gupta R, Hast MW. Similar Biomechanics Between the Double-Cortical Button and Docking Techniques for Ulnar Collateral Ligament Reconstruction: A Cadaveric Evaluation. Orthop J Sports Med. 2023;11:23259671221123342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 14. | Kocaman G, Yenigün MB, Kaya B, Sefa Özden N, Özdemir A, Koçak ME, Kıvrak H, Kayı Cangır A. A rare giant sarcoma of the chest wall: Undifferentiated pleomorphic sarcoma. Turk Gogus Kalp Damar Cerrahisi Derg. 2021;29:552-555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Wang L, Yan X, Zhao J, Chen C, Chen C, Chen J, Chen KN, Cao T, Chen MW, Duan H, Fan J, Fu J, Gao S, Guo H, Guo S, Guo W, Han Y, Jiang GN, Jiang H, Jiao WJ, Kang M, Leng X, Li HC, Li J, Li J, Li SM, Li S, Li Z, Li Z, Liang C, Mao NQ, Mei H, Sun D, Wang D, Wang L, Wang Q, Wang S, Wang T, Liu L, Xiao G, Xu S, Yang J, Ye T, Zhang G, Zhang L, Zhao G, Zhao J, Zhong WZ, Zhu Y, Hulsewé KWE, Vissers YLJ, de Loos ER, Jeong JY, Marulli G, Sandri A, Sziklavari Z, Vannucci J, Ampollini L, Ueda Y, Liu C, Bille A, Hamaji M, Aramini B, Inci I, Pompili C, Van Veer H, Fiorelli A, Sara R, Sarkaria IS, Davoli F, Kuroda H, Bölükbas S, Li XF, Huang L, Jiang T. Expert consensus on resection of chest wall tumors and chest wall reconstruction. Transl Lung Cancer Res. 2021;10:4057-4083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 16. | Sandler G, Hayes-Jordan A. Chest wall reconstruction after tumor resection. Semin Pediatr Surg. 2018;27:200-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 17. | Berthet JP, Wihlm JM, Canaud L, Joyeux F, Cosma C, Hireche K, Alric P, Marty-Ané CH. The combination of polytetrafluoroethylene mesh and titanium rib implants: an innovative process for reconstructing large full thickness chest wall defects. Eur J Cardiothorac Surg. 2012;42:444-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 18. | Billè A, Okiror L, Karenovics W, Routledge T. Experience with titanium devices for rib fixation and coverage of chest wall defects. Interact Cardiovasc Thorac Surg. 2012;15:588-595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 19. | Cheng GZ, San Jose Estepar R, Folch E, Onieva J, Gangadharan S, Majid A. Three-dimensional Printing and 3D Slicer: Powerful Tools in Understanding and Treating Structural Lung Disease. Chest. 2016;149:1136-1142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 77] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 20. | Hage JJ, Woerdeman LA. Lower limb necrosis after use of the anterolateral thigh free flap: is preoperative angiography indicated? Ann Plast Surg. 2004;52:315-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Pu JJ, Atia A, Yu P, Su YX. The Anterolateral Thigh Flap in Head and Neck Reconstruction. Oral Maxillofac Surg Clin North Am. 2024;36:451-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Graboyes EM, Hornig JD. Evolution of the anterolateral thigh free flap. Curr Opin Otolaryngol Head Neck Surg. 2017;25:416-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 23. | Sananpanich K, Tu YK, Kraisarin J, Chalidapong P. Flow-through anterolateral thigh flap for simultaneous soft tissue and long vascular gap reconstruction in extremity injuries: anatomical study and case report. Injury. 2008;39 Suppl 4:47-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Hung NT, Doan LV, Trung VH, Cuong NV. A vascular anatomical study of the anterolateral thigh flap in the Vietnamese's adult cadavers. Ann Med Surg (Lond). 2022;76:103416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |