Published online Apr 18, 2026. doi: 10.5312/wjo.v17.i4.116046
Revised: November 17, 2025
Accepted: January 9, 2026
Published online: April 18, 2026
Processing time: 160 Days and 18.7 Hours
Osteoporosis is a prevalent metabolic bone disorder that poses significant health burdens. The Xianling Gubao capsule (XLGB), a traditional Chinese medicine, has demonstrated clinical effectiveness in combating osteoporosis; however, its underlying mechanism, particularly concerning the gut-bone axis, remains ina
To investigate the anti-osteoporotic mechanism of XLGB via the gut-bone axis and Wnt/β-catenin signaling.
An ovariectomized rat model of postmenopausal osteoporosis was established. The rats received low, medium, or high doses of XLGB for eight weeks. Bone microstructure was assessed using micro-computed tomography, serum bone metabolism markers were quantified by enzyme-linked immunosorbent assay, gut microbiota composition was characterized through 16S rRNA gene seque
The medium dose of XLGB produced the most significant therapeutic effects. The medium dose of XLGB sig
XLGB alleviates osteoporosis by modulating the gut-bone axis, correcting metabolic disturbances, and rectifying the osteogenic-adipogenic imbalance through the Wnt/β-catenin and peroxisome proliferator-activated receptor gamma pathways.
Core Tip: This study demonstrates that the Xianling Gubao capsule (XLGB) alleviates osteoporosis by modulating the gut-bone axis. Utilizing a multi-omics approach, we reveal that XLGB not only improves bone microstructure and serum bone markers but also rectifies ovariectomy-induced gut microbiota dysbiosis and metabolic disturbances. More importantly, we identify that its core mechanism involves the activation of the osteogenic Wnt/β-catenin pathway while simultaneously suppressing the adipogenic peroxisome proliferator-activated receptor gamma pathway, thereby rebalancing osteogenic-adipogenic differentiation in the bone marrow. This provides a novel mechanistic basis for XLGB’s anti-osteoporotic effects and underscores the gut-bone axis as a promising therapeutic target.
- Citation: Yu AX, Liang WH, Zhou YS, Li P, Ji JM, Zhang FW, Yu P, Liu J. Xianling Gubao capsule alleviates osteoporosis: Modulation of gut-bone axis and Wnt signaling. World J Orthop 2026; 17(4): 116046
- URL: https://www.wjgnet.com/2218-5836/full/v17/i4/116046.htm
- DOI: https://dx.doi.org/10.5312/wjo.v17.i4.116046
Osteoporosis is a prevalent metabolic bone disorder that poses significant economic burdens and public health challenges[1]. This systemic skeletal disorder is characterized by reduced bone mineral density (BMD) and deteriorated bone microarchitecture, which together increase bone fragility and the risk of fractures. The pathogenesis of osteoporosis is multifactorial, involving aging processes, endocrine changes, lifestyle factors, and genetic influences[2]. Bone loss in postmenopausal women with osteoporosis is strongly linked to estrogen deficiency[3]. Although current pharmacological treatments - such as bisphosphonates, selective estrogen receptor modulators, and estrogen replacement therapy - are available, they often cause significant adverse effects, highlighting the urgent need to develop safer and more cost-effective therapeutic options for osteoporosis management[4].
The gut microbiota, recognized as the largest microbial ecosystem within the human body, plays a crucial role in maintaining host health. Dysbiosis of this microbial community has been linked to the pathogenesis of numerous diseases. Emerging evidence suggests that the gut microbiota contributes to the development of osteoporosis by modulating bone metabolism[5,6]. Through the production of metabolites, the gut microbiota can influence osteoclast and osteoblast activity by regulating host bone metabolism, immune responses, and hormone secretion, thereby offering potential avenues for the prevention or treatment of osteoporosis[7]. However, the specific microbial taxa fundamentally involved in the pathogenesis of osteoporosis have yet to be comprehensively identified.
Xianling Gubao capsule (XLGB), a well-established formulation in traditional Chinese medicine, has been demon
XLGBs were produced by Guizhou Tongjitang Pharmaceutical Co., Ltd. (Guizhou Province, China), while estradiol (E2) valerate tablets were manufactured by Bayer Pharmaceutical Healthcare Co., Ltd (Beijing, China).
In accordance with the intragastric dosing regimen for the various groups of rats in the XLGB administration group, the appropriate powder content from the XLGBs was dissolved in normal saline to prepare different doses of XLGB suspension. The E2 valerate tablets were ground and subsequently dissolved in normal saline. The intragastric administration of these drugs was performed daily.
A total of sixty specific-pathogen-free female Sprague-Dawley (SD) rats, aged 6-8 weeks and weighing 200 ± 10 g, were purchased from Liaoning Changsheng Biotechnology Co., Ltd. [License No. SCXK (Liaoning) 2020-0001; China]. All animals were maintained under standardized laboratory conditions at the Experimental Animal Center of Changchun University of Chinese Medicine. These conditions included a 12-hour light/dark cycle, an ambient temperature regulated between 22 °C and 25 °C, and relative humidity maintained at 55% ± 5%. The rats had unrestricted access to standard laboratory chow and water throughout the study. All experimental procedures were approved by the Institutional Animal Care and Use Committee of Changchun University of Chinese Medicine (Approval No. 2023559). The rat model of postmenopausal osteoporosis was established through bilateral ovariectomy (OVX). SD rats were initially anesthetized via intraperitoneal injection of tribromoethanol at a dose of 150 mg/kg, followed by maintenance anesthesia using inhalation of 1.4% isoflurane at a ventilation flow rate of 2 L/minutes. After induction of anesthesia, the dorsal fur was shaved, and the surgical site was disinfected with iodophor. Bilateral incisions, approximately 1.5 cm in length, were made flanking the midline, 1 cm below the costal margin and 2 cm from the spine. The ovaries were identified and excised. In the sham-operated (Sham) group, only the periovarian adipose tissue was removed, leaving the ovaries intact. After surgery, the muscle and skin layers were sutured sequentially, and the incision was disinfected again. To mitigate the risk of postoperative infection, penicillin sodium was administered intraperitoneally at a dose of 80000 units daily for three consecutive days.
Sixty SD rats were randomly assigned to six groups (n = 10 per group): Sham, OVX, E2, XLGB-low (XLGB-L), XLGB-middle (XLGB-M), and XLGB-high (XLGB-H). Treatment began two months after surgery and was administered daily via oral gavage. The E2 group received 0.1 mg/kg/day of E2, while the XLGB-L, XLGB-M, and XLGB-H groups were given 157.5 mg/kg/day, 315 mg/kg/day, and mg/kg/day, respectively. The Sham and OVX control groups were administered an equivalent volume of distilled water. After two months of treatment, all rats were euthanized. Serum, bilateral femurs, and fresh fecal samples were collected for analysis, and the uteri were harvested and weighed to calculate the uterine index.
The micro-architecture of the distal femur was evaluated using a micro-computed tomography (micro-computed tomography) scanner (PerkinElmer, MA, United States). The scanned images were reconstructed with SkyScan software (Bruker, Belgium). Bone morphometric parameters, including BMD, bone volume (BV)/tissue volume (TV), trabecular number, trabecular thickness, and trabecular separation, were quantified from the reconstructed images.
Femurs from rats were preserved in a 10% neutral buffered formalin solution and later encased in paraffin for slicing. The slices obtained were stained using hematoxylin and eosin to examine the structural alterations in the bone tissue.
Blood specimens were obtained from the abdominal aorta of rats in each experimental group and were allowed to coagulate at ambient temperature for half an hour. Afterward, serum was extracted through centrifugation at 3000 rpm for 10 minutes at a temperature of 4 °C. The levels of procollagen I N-terminal propeptide (PINP), collagen type I alpha 1, osteocalcin (OCN), runt-related transcription factor 2 (RUNX2), and E2 in the serum were measured using enzyme-linked immunosorbent assay kits from (MEIMIAN, Jiangsu Province, China). Furthermore, the serum levels of calcium, pho
The tissue sections were dewaxed, rehydrated, and incubated overnight at 4 ℃ with a primary antibody at a dilution of 1:600. For detection, the sections were incubated with a horseradish peroxidase-labeled secondary antibody for 60 minutes, followed by the application of a liquid 3,3’-diaminobenzidine substrate (Thermo Fisher Scientific, MA, United States). Subsequently, the sections were counterstained with hematoxylin, dehydrated, and mounted using a quick-drying medium (Joint Biosciences, Australia). Scanning was conducted using an Aperio Scanscope (Leica Biosystems, IL, United States), and bone histomorphometry was analyzed with BIOQUANT OSTEO software (Bioquant Image Analysis Corporation, TN, United States).
Rat femurs were frozen in liquid nitrogen and subsequently ground into a fine powder using a mortar maintained at low temperatures with liquid nitrogen. Total RNA was extracted from the powdered tissue employing TRIzol reagent (TIANGEN, Beijing, China) and quantified utilizing a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, MA, United States). Reverse transcription was conducted using the FastKing gDNA Dispelling RT SuperMix (TIANGEN, Beijing, China). Quantitative real-time polymerase chain reaction (PCR) was executed using FastReal gPCR PreMix (SYBR Green; Thermo Fisher Scientific, MA, United States) on a QuantStudio 3 system (Thermo Fisher Scientific, MA, United States) to analyze the expression levels of wingless-type MMTV integration site family, member 3A (Wnt3a), β-catenin, RUNX2, glycogen synthase kinase-3 beta (GSK3β), receptor activator of nuclear factor-kappaB (RANK), RANK ligand (RANKL) and osteoprotegerin (OPG) mRNA. Gene expression levels were calculated using the 2-ΔΔCt method, with glyceraldehyde 3-phosphate dehydrogenase or β-actin serving as reference genes. The primer sequences are detailed in Table 1.
| Target gene | Primer | |
| GAPDH | Forward | AAGATGGTGAAGGTCGGTGT |
| Reverse | GCTTCCCATTCTCAGCCTTG | |
| Wnt3a | Forward | CTCCTCTCGGATACCTCTTAGTG |
| Reverse | GCATGATCTCCACGTAGTTCCTG | |
| β-catenin | Forward | CTTCCAGACACGCCATCATG |
| Reverse | AAGGTTGTGTAGGGTCCCAG | |
| RUNX2 | Forward | CGCCACCACTCACTACCACAC |
| Reverse | ATAGGACGCTGACGAAGTACCA | |
| GSK3β | Forward | GAGCCACCGATTACACGTC |
| Reverse | TATTTCCACCAACTGATCCACAC | |
| β-actin | Forward | CTGAACGTGAAATTGTCCGAGA |
| Reverse | TTGCCAATGGTGATGACCTG | |
| RANK | Forward | GCCAGCAAGAAGTGTGTGAA |
| Reverse | TCGCTGACTAAGGTGAACGT | |
| RANKL | Forward | CACACGAGGGTCCGCTGCATC |
| Reverse | TGCAGACCACCTGACCCAGTCC | |
| OPG | Forward | ACATCATTGAATGGACAACCCAG |
| Reverse | TGCCAGGAGCACATTTGTCA | |
Left tibial samples (0.1 g each) were pulverized using a cryogenic grinder in liquid nitrogen. Total protein was extracted from the powdered tissue with tissue lysis buffer (Biyuntian, Shanghai, China). The protein concentration was de
The extraction of microbial DNA and the sequencing of the 16S rRNA gene were performed according to the guidelines set forth by Shanghai Applied Protein Technology (China). For an in-depth description of the procedures and parameters (Supplementary material).
Non-target metabolomics metabolite analysis was executed using liquid chromatography-mass spectrometry in ac
The data measurements are presented as mean ± SD. A one-way ANOVA was performed utilizing GraphPad Prism version 8.0.1. A P-value of less than 0.05 was deemed statistically significant.
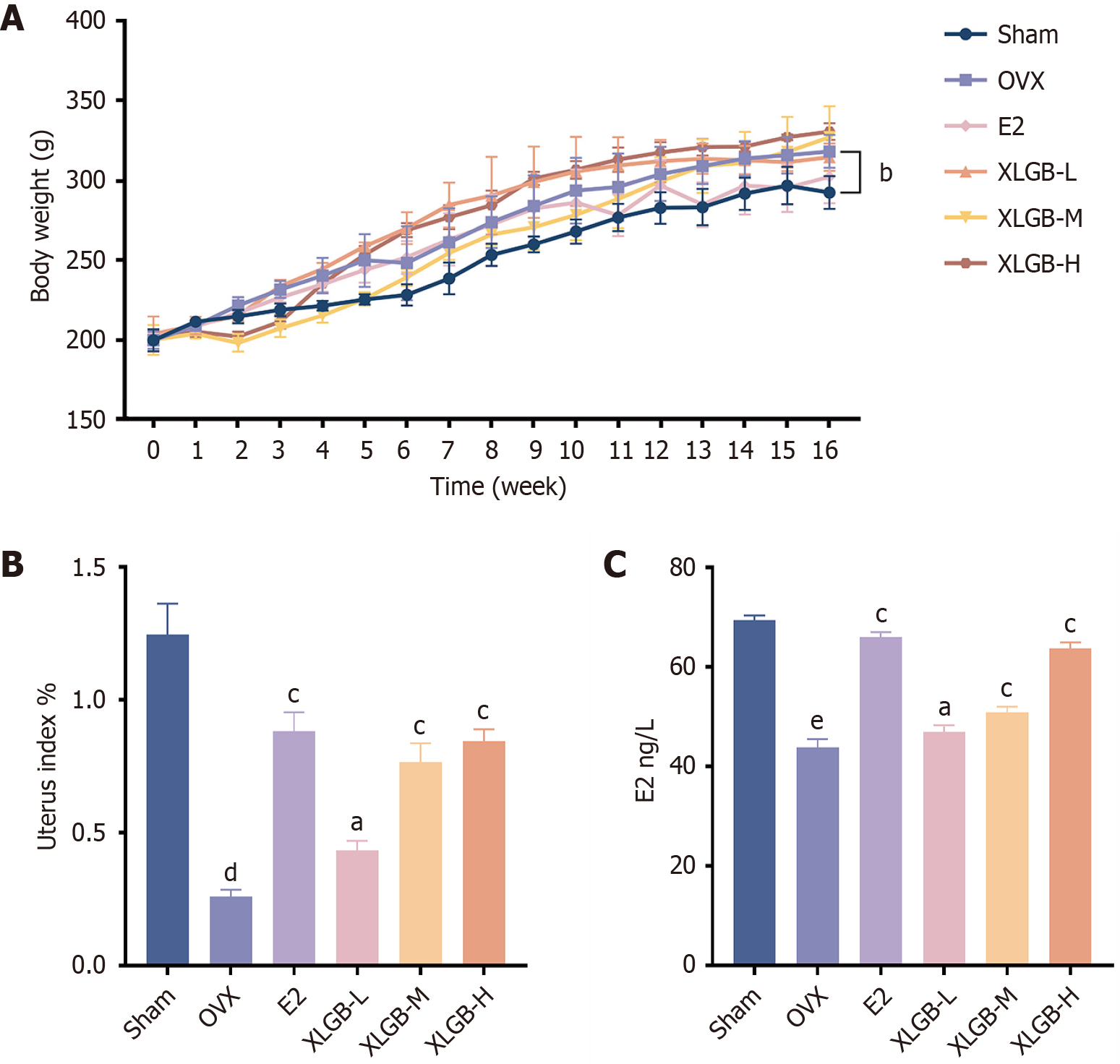
OVX rats typically exhibit increased body weight and uterine atrophy post-surgery, which are hallmarks of estrogen deficiency[3]. As illustrated in Figure 1A, the body weight in the OVX group was significantly higher than that in the Sham group. In contrast, none of the drug treatments resulted in a significant alteration of body weight compared to the OVX group. The uterine index was significantly reduced in the OVX group compared to the Sham group (Figure 1B). Treatment with E2 or various doses of XLGB significantly increased the uterine index in OVX rats. Serum E2 concentrations were significantly elevated in the E2-treated group compared to the OVX group. Similarly, XLGB treatment effectively increased serum E2 levels in a dose-dependent manner (Figure 1C).
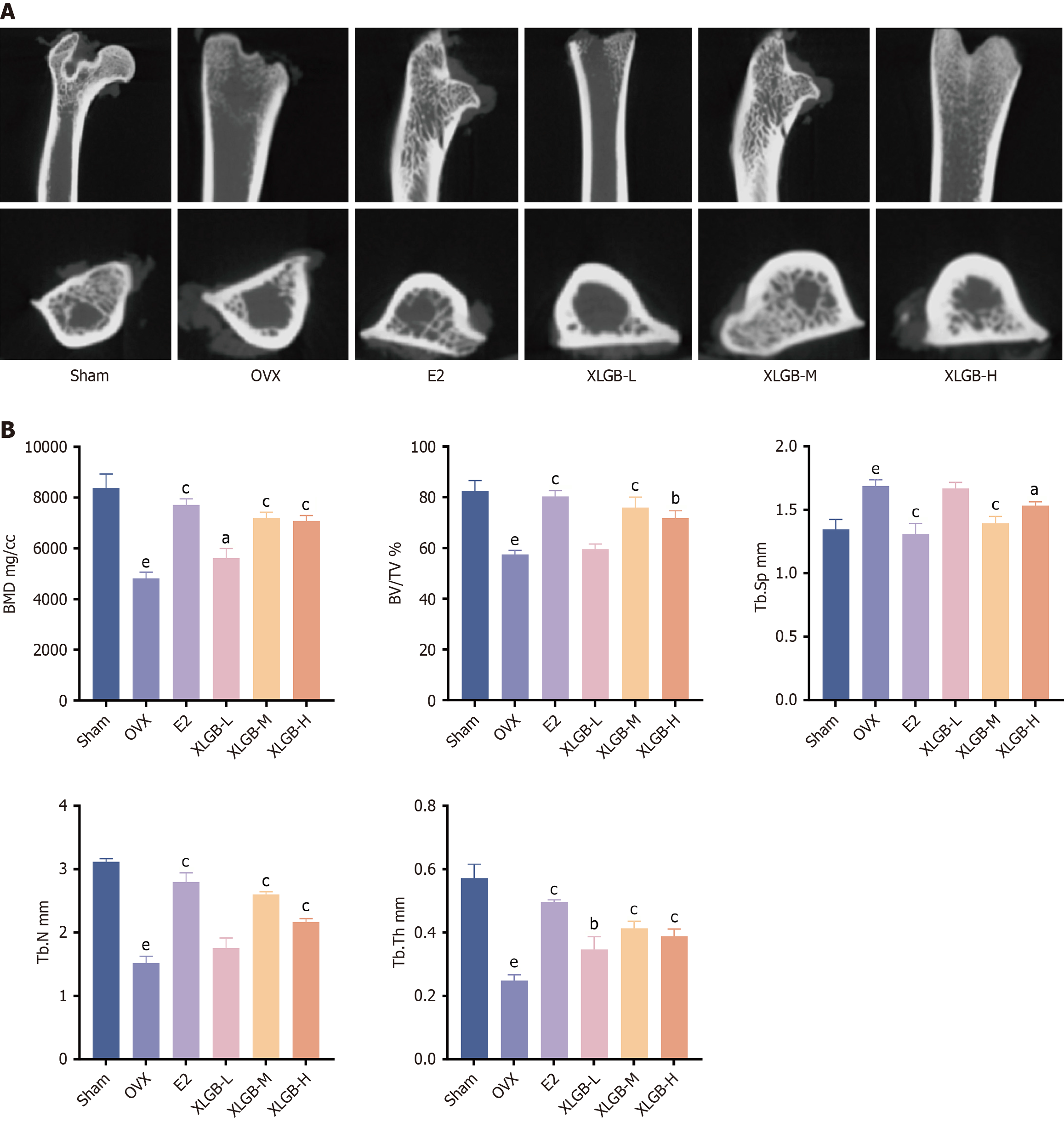
To evaluate the effects of XLGB on bone microstructure in OVX rats, micro-computed tomography analysis was conducted. Representative images showed that, in comparison to the Sham group, the OVX group exhibited a marked reduction in trabecular number and density, with trabeculae appearing more sparsely distributed (Figure 2A). Histomo
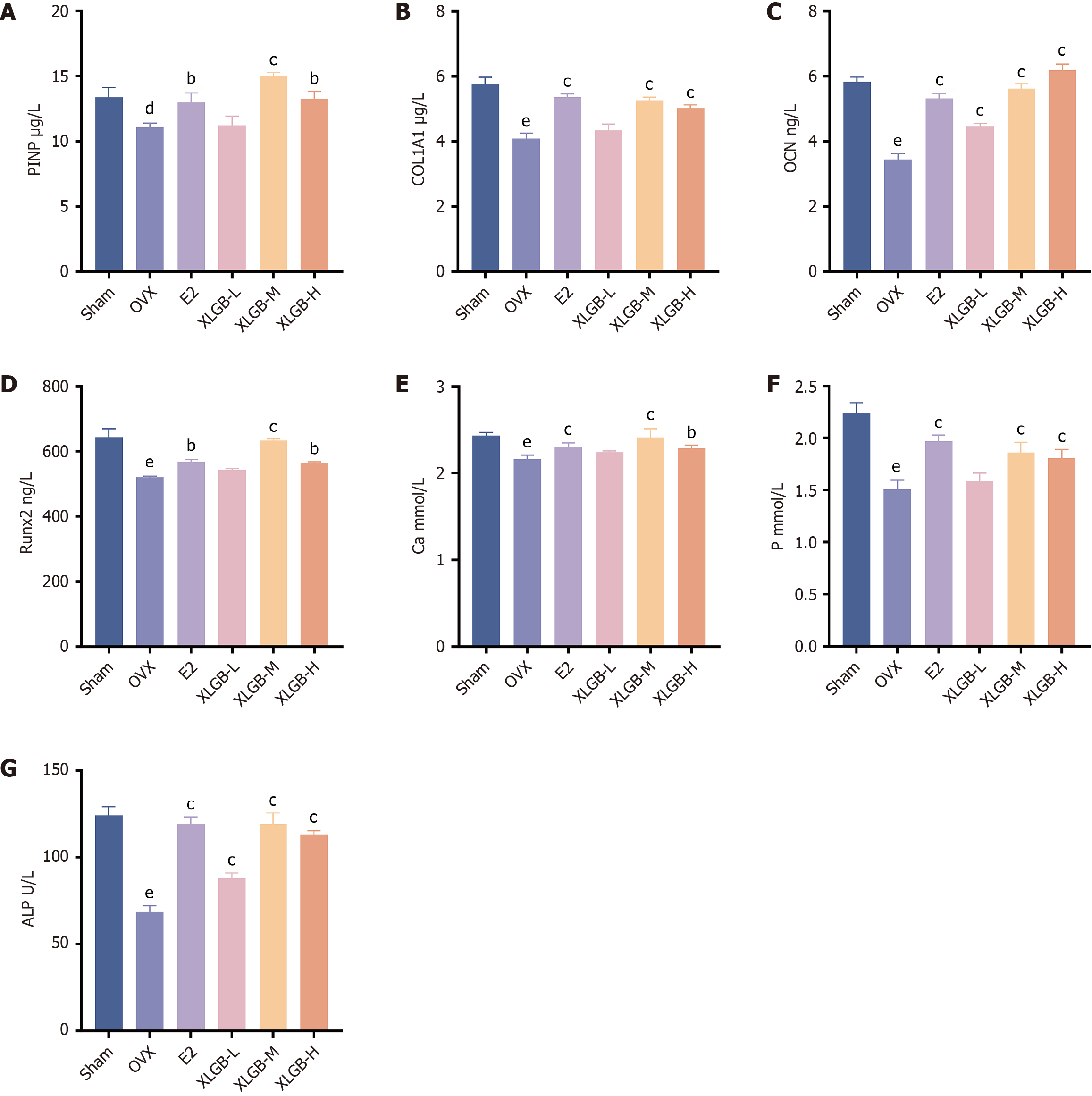
The enzyme-linked immunosorbent assay results for serum bone metabolism markers are presented in Figure 3A-D. Compared to the Sham group, the expression levels of PINP, collagen type I alpha 1, OCN, and RUNX2 were significantly decreased in the OVX group. Treatment with XLGB at various concentrations increased the levels of these markers, with the most pronounced improvement observed in the XLGB-M group. Similarly, serum biochemical analysis indicated that the levels of calcium, phosphorus, and ALP were significantly lower in the OVX group compared to the Sham group (Figure 3E-G). E2, along with both XLGB-M and XLGB-H, significantly increased serum calcium and phosphorus levels. Additionally, E2 and all doses of XLGB significantly elevated ALP levels. Notably, the upregulation of serum calcium by XLGB-M was significantly greater than that observed with other XLGB doses. In summary, XLGB enhances osteoblast function, improves bone matrix synthesis, and promotes bone mineralization, with XLGB-M demonstrating the optimal therapeutic effect.
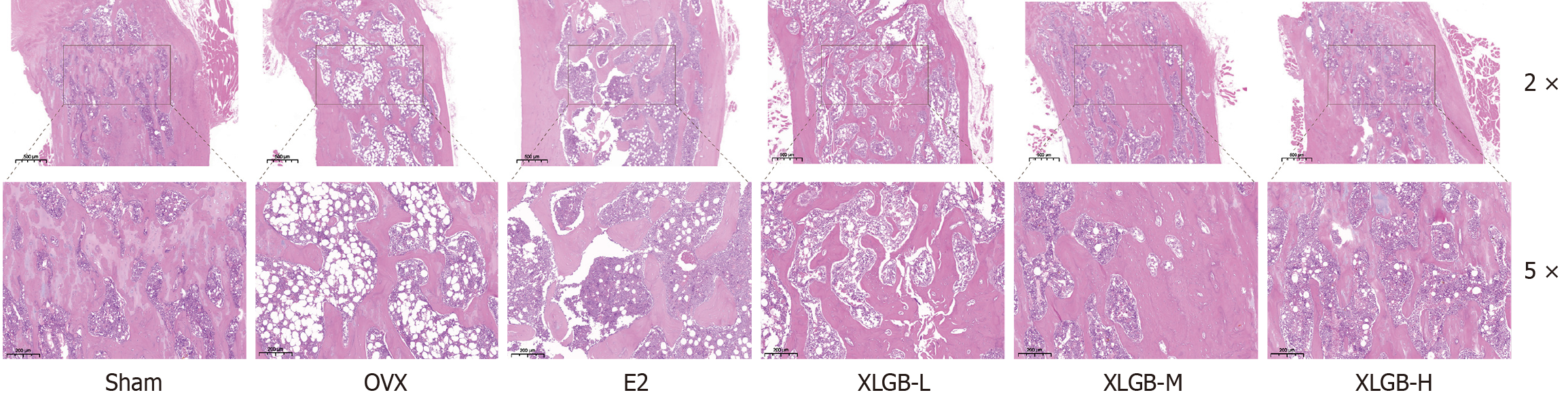
Histopathological examination of bone tissue was conducted using hematoxylin and eosin staining (Figure 4). The Sham group exhibited a closely arranged and intact trabecular structure with minimal lipid accumulation. In contrast, the OVX group displayed sparse, loose, and discontinuous trabeculae, accompanied by an increase in lipid droplet accumulation. Compared to the OVX group, both the E2 and XLGB treatment groups demonstrated a widened trabecular bone area, a denser arrangement, continuous structural improvement, and a significantly reduced number of lipid vacuoles. Notably, among the XLGB doses, XLGB-M exhibited the most pronounced effect in ameliorating bone tissue damage.
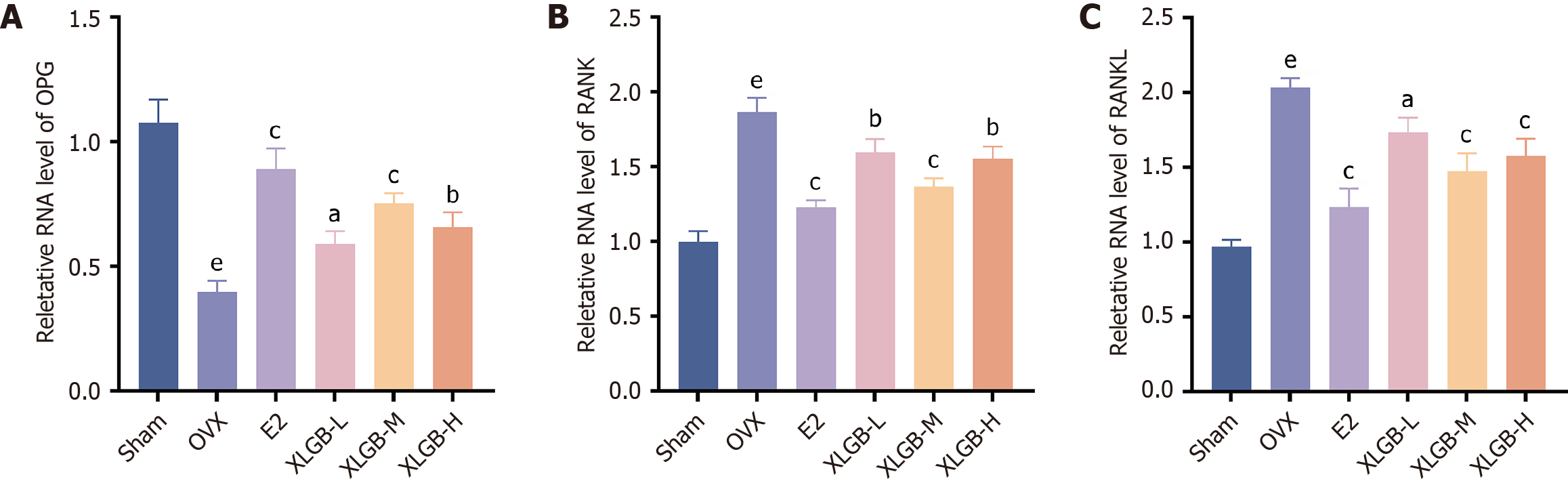
The RANK, RANKL, and OPG signaling pathway plays a crucial role in the pathogenesis of osteoporosis[15]. OPG functions as a decoy receptor that inhibits osteoclastogenesis and osteoclastic activity by disrupting the RANK-RANKL interaction, making it a key therapeutic target for osteoporosis[16]. To determine whether the effects of XLGB are mediated through this pathway, we measured the mRNA expression levels of RANK, RANKL, and OPG in bone tissue. Compared to the Sham group, OVX rats showed significantly increased mRNA expression of RANK and RANKL, alongside a significant reduction in OPG expression. Treatment with E2 or XLGB effectively reversed these changes, significantly downregulating RANK and RANKL expression while upregulating OPG expression relative to the OVX group (Figure 5). Among the XLGB treatment groups, XLGB-M produced the most pronounced regulatory effects, comparable to those observed with E2 treatment. In conclusion, these pharmacodynamic findings indicate that XLGB-M offers optimal therapeutic efficacy against osteoporosis, providing a mechanistic rationale for its clinical application.
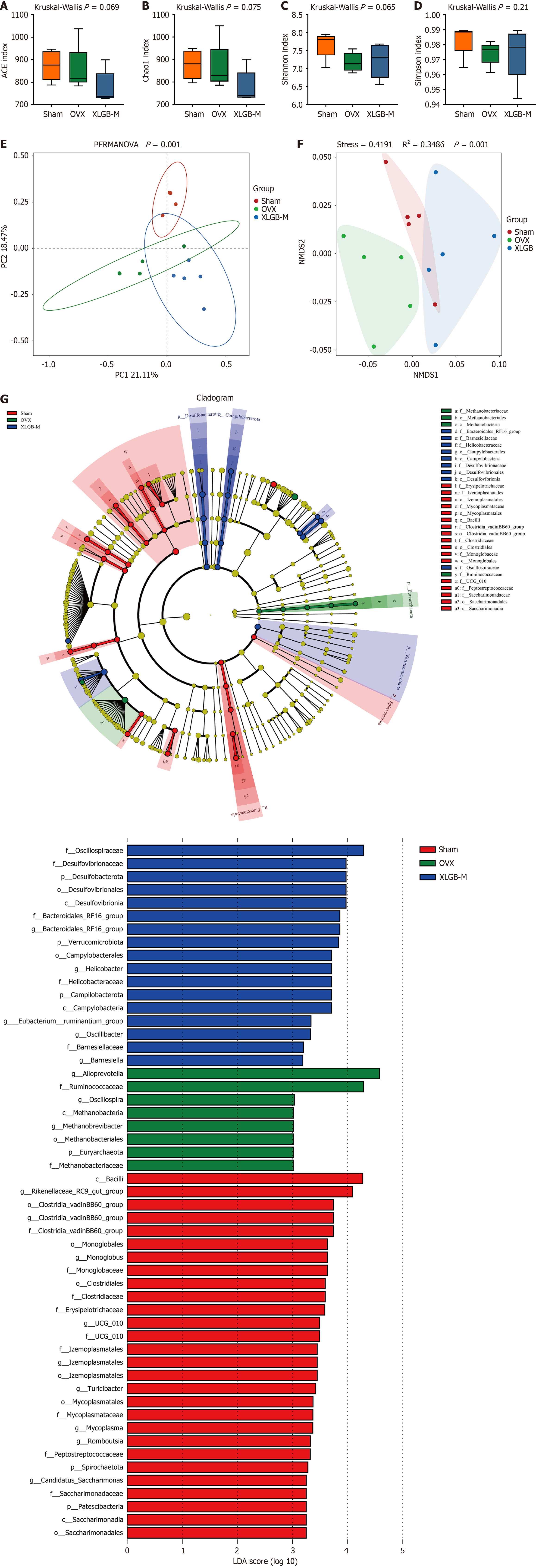
Studies have demonstrated a close association between gut microbiota and osteoporosis[17]. Pharmacodynamic results indicated that XLGB-M exhibited the most potent therapeutic effect against osteoporosis. Accordingly, fecal samples were collected from the Sham, OVX, and XLGB-M groups for 16S rRNA gene sequencing to elucidate the influence of XLGB on gut microbial communities in an OVX-induced rat model of osteoporosis.
We first assessed the within-sample (alpha) diversity. The species richness, as reflected by the abundance-based coverage estimator and Chao1 indices, exhibited a decreasing trend in the XLGB-M group compared to the OVX group; however, this difference was not statistically significant (P = 0.069 and P = 0.075, respectively; Figure 6A and B). Conversely, measures that incorporate both richness and evenness, specifically the Shannon and Simpson indices, indicated a tendency toward increased diversity in the XLGB-M group compared to the OVX group, although these findings were also not statistically significant (P = 0.065 and P = 0.21, respectively; Figure 6C and D). Collectively, these results suggest that XLGB-M treatment did not significantly alter the overall within-sample diversity under the experimental conditions.
In marked contrast, the between-sample (beta) diversity analysis revealed profound structural alterations in the gut microbial community. Both principal coordinates analysis and non-metric multidimensional scaling demonstrated distinct and significant separation among the Sham, OVX, and XLGB-M groups (Figure 6E and F). The distinct clustering indicates that while the local diversity within each sample remained relatively stable, the overall composition of the microbial ecosystem was significantly reshaped by OVX and subsequently modulated by XLGB-M intervention.
To identify specific bacterial taxa contributing to these compositional changes, linear discriminant analysis effect size was performed. This analysis revealed characteristic microbial biomarkers unique to each group (Figure 6G). The Sham group was predominantly enriched with taxa from the families Erysipelotrichaceae, Mycoplasmataceae, Clostridiaceae, Monoglobaceae, and Saccharimonadaceae. In contrast, the OVX group was characterized by a significant increase in methanogenic archaea, such as family (f)_Methanobacteriaceae and order (o)_Methanobacteriales, alongside f_Ruminococcaceae. Notably, the XLGB-M group exhibited a unique microbial signature, enriched with f_Bacteroidales_RFl6_ group, f_Barnesiellaceae, f_Helicobacteraceae, o_Campylobacterales, f_Desulfovibrionaceae, and f_Oscillospiraceae.
In summary, the stability in α-diversity, coupled with significant β-diversity shifts and distinct taxonomic alterations, indicates that XLGB-M induces a qualitative restructuring of the gut microbiota in OVX rats. This suggests a mechanism of action that specifically modulates microbial community composition rather than globally enhancing species richness.
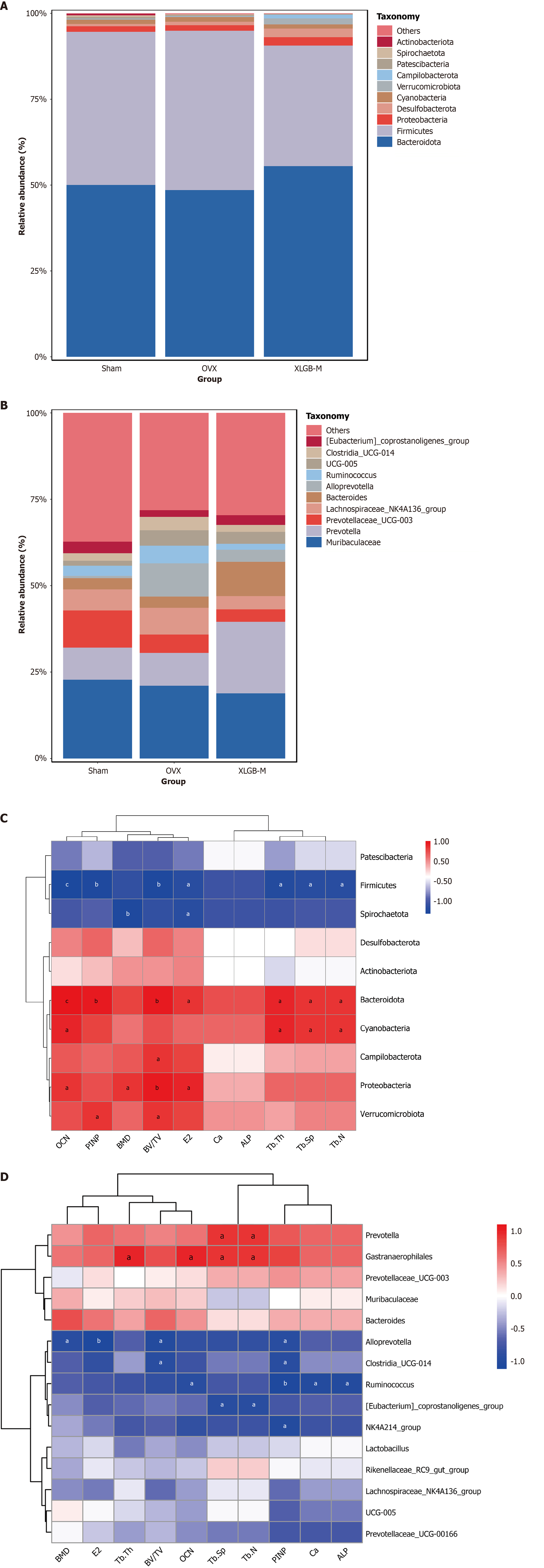
At the phylum level, Bacteroidota and Firmicutes emerged as the dominant phyla in the intestine, collectively accounting for over 90% of the total microbiota across all groups. Compared the Sham group, the OVX group exhibited a decreased relative abundance of Bacteroidota and Proteobacteria, alongside an increased abundance of Firmicutes, Cyanobacteria, and Desulfobacterota. Notably, XLGB-M treatment significantly reversed these OVX-induced alterations by reducing the proportions of Firmicutes and Cyanobacteria while increasing the proportions of Bacteroidota, Proteobacteria, and Desulfobacterota (Figure 7A). The Firmicutes/Bacteroidota ratio serves as a critical indicator of gut microbiota composition and is closely linked to bone metabolism. The Firmicutes/Bacteroidota ratios were 0.89 for the Sham group, 0.96 for the OVX group, and 0.63 for the XLGB-M group, indicating that XLGB-M significantly modulated the gut microbiota structure at the phylum level. At the genus level, OVX rats demonstrated an increased relative abundance of Muribaculaceae, Prevotella, Lachnospiraceae_NK4A136_group, Alloprevotella, Ruminococcus, UCG-005, and Clostridia_ UCG-014. Conversely, XLGB-M treatment reduced in the abundance of these genera while enhancing the relative abundance of Prevotella, Bacteroides, and Eubacterium_coprostanoligenes_group compared to the OVX group (Figure 7B). Spearman correlation analysis was conducted to evaluate the relationships between gut microbiota composition, bone microstructural parameters, and bone formation markers. At the phylum level, the abundance of Firmicutes was negatively correlated with OCN, PINP, BV/TV, trabecular thickness, and trabecular number. In contrast, Spirochaetota exhibited a negative correlation with BMD. Conversely, Bacteroidota, Cyanobacteria, Campilobacterota, Proteobacteria, and Verrucomicrobiota demonstrated strong positive correlations with bone formation markers (OCN and PINP) and parameters (BMD, BV/TV, trabecular thickness, and trabecular number; Figure 7C). At the genus level, Prevotella and Gastranaerophilales displayed positive correlations with several bone parameters. In contrast, Alloprevotella was negatively correlated with BMD, BV/TV, and PINP; Ruminococcus was negatively correlated with calcium, ALP, OCN, and PINP; Clostridia_UCG-014 was negatively correlated with BV/TV and PINP; Eubacterium_coprostanoligenes_group was negatively correlated with trabecular separation and trabecular number; and NK4A214_group was negatively correlated with PINP (Figure 7D). In summary, our data suggest that XLGB-M treatment ameliorates OVX-induced gut microbiota dysbiosis in osteoporotic rats.
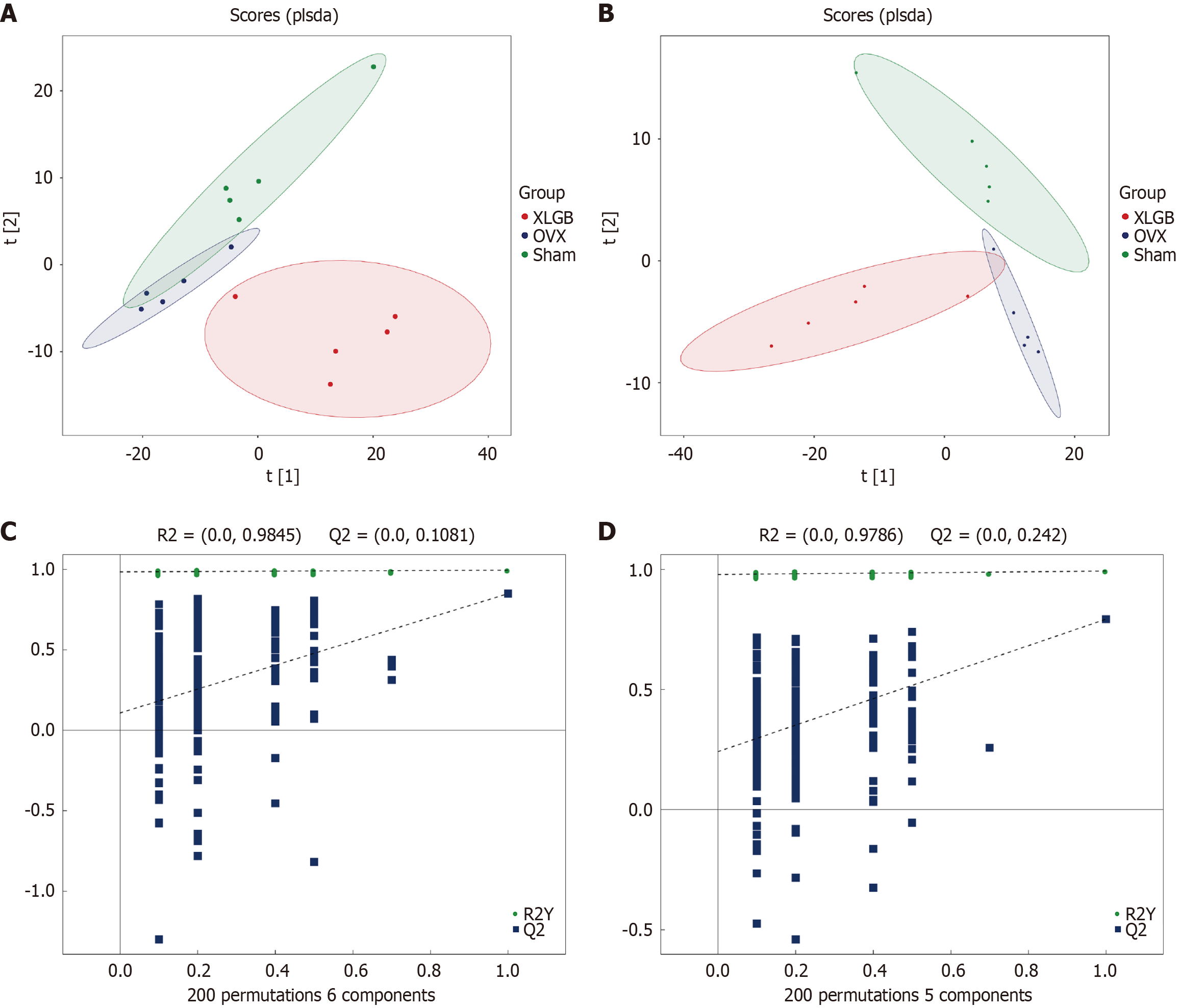
Alterations in intestinal microbial composition can significantly influence the profile of intestinal metabolites. We further investigated the effects of XLGB-M on the metabolome through a non-targeted metabolomic analysis of fecal samples using liquid chromatography-mass spectrometry. In both positive and negative ion modes, the orthogonal projections to latent structures discriminant analysis (OPLS-DA) score plots demonstrated clear separation between the experimental groups, while samples within the same group exhibited tight clustering (Figure 8A and B). This pattern indicates high intra-group consistency in metabolite composition and concentration, with minimal biological variation among indivi
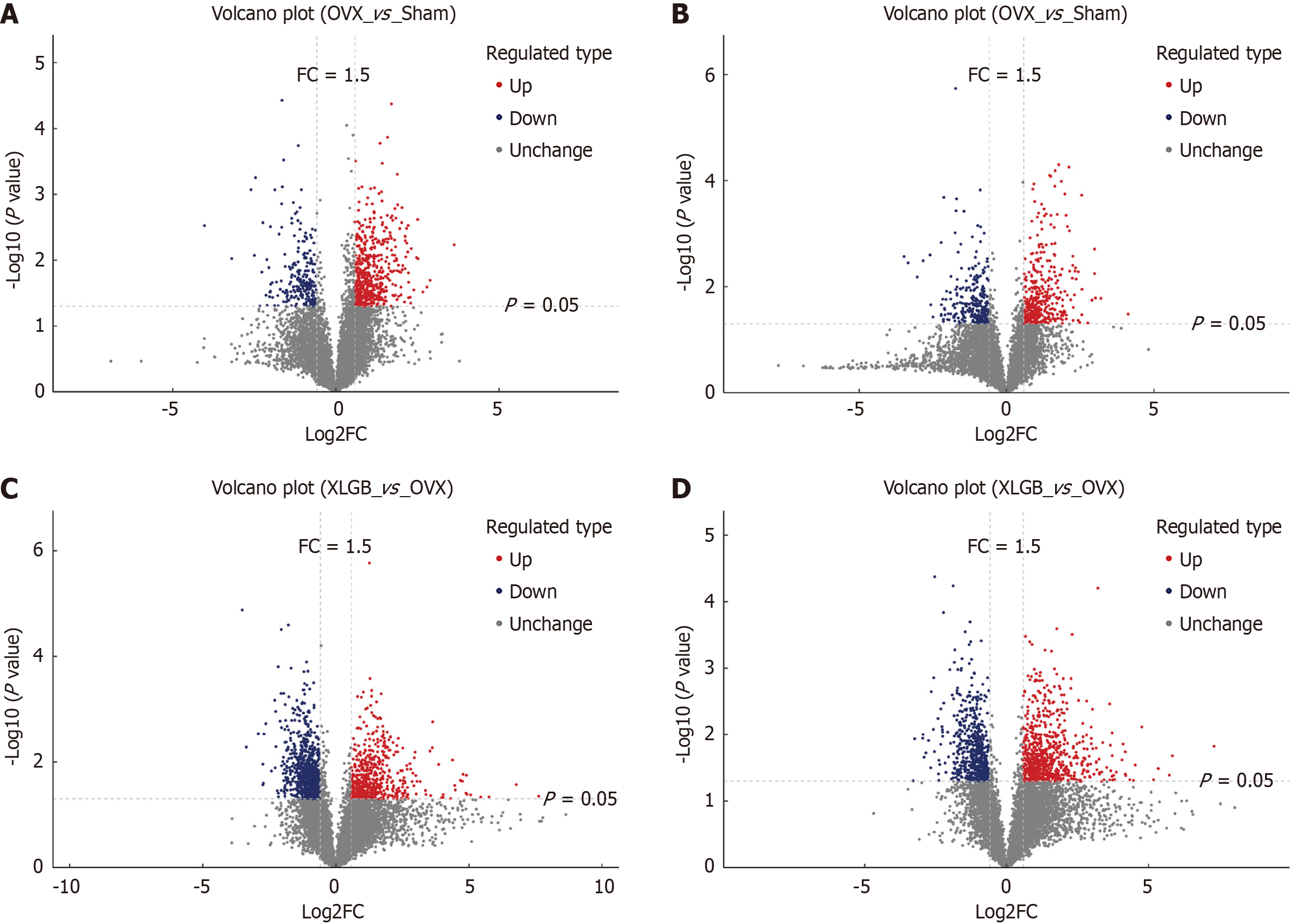
A total of 2086 compounds were detected by cross-referencing the HMDB and METLIN databases across both positive and negative ionization modes. Specifically, 1314 compounds were detected in positive ion mode, while 772 compounds were identified in negative ion mode. Differential metabolites were screened using thresholds of a fold change > 1.5 or fold change < 0.67 and P < 0.05 (Student’s t-test), which are visualized in the volcano plots presented in Figure 9. In the negative ion mode, the OVX group exhibited 22 significantly up-regulated and 12 down-regulated metabolites compared to the Sham group. Conversely, the XLGB-M group demonstrated 34 up-regulated and 27 down-regulated metabolites relative to the OVX group. In the positive ion mode, 33 metabolites were up-regulated and 18 were down-regulated in the OVX group vs the Sham group, while the XLGB-M group displayed 34 up-regulated and 72 down-regulated metabolites compared to the OVX group.
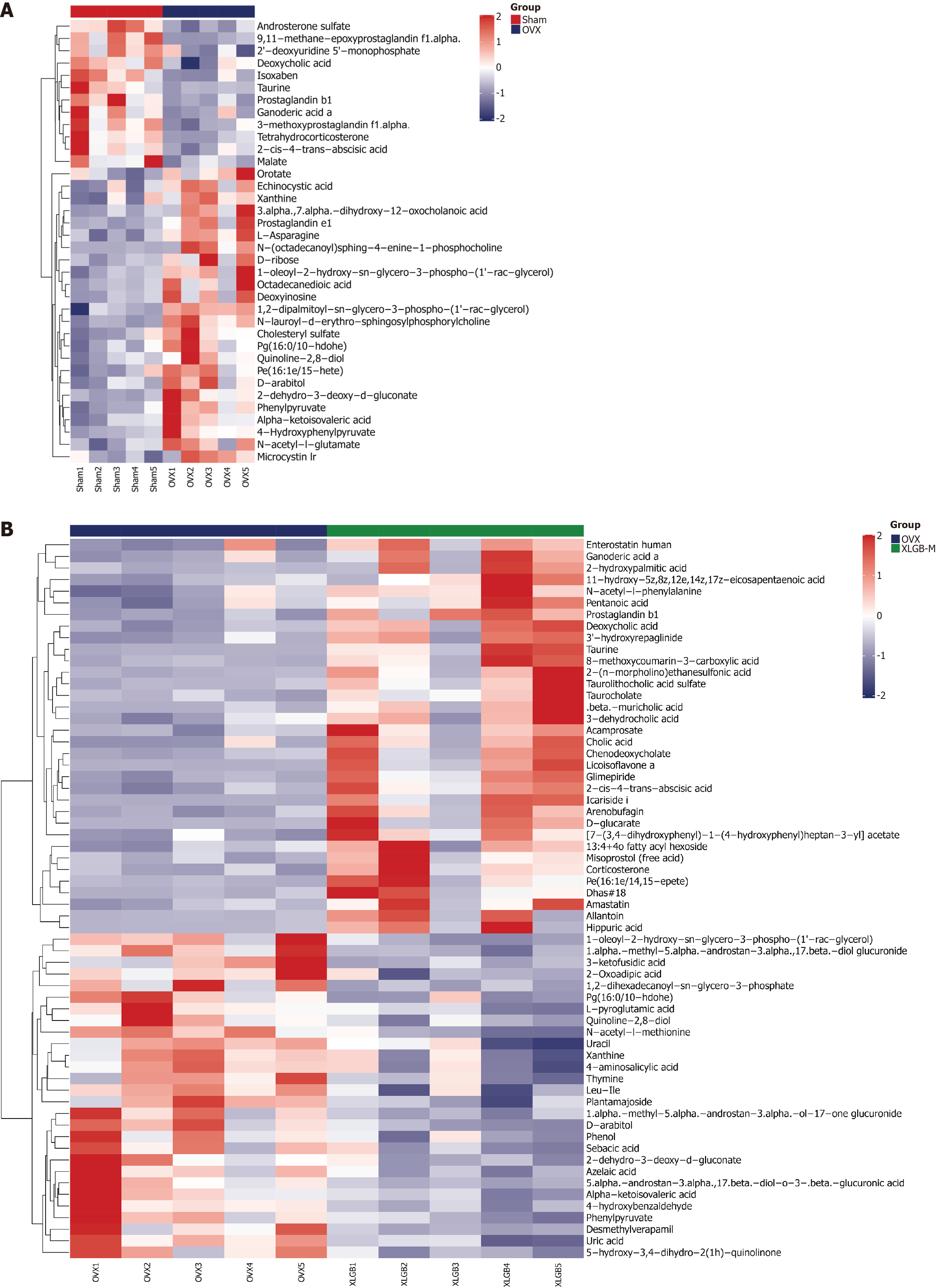
A clustered heatmap was generated to illustrate the average levels of fecal differential metabolites across the Sham, OVX, and XLGB-M groups, thereby visually representing the relative abundance of biomarkers among these groups (Figure 10). The heatmap displays hierarchical clustering of both metabolites and samples, with red and blue colors indicating high and low relative abundance, respectively.
In metabolomics, the variable importance in projection (VIP) score derived from the OPLS-DA model quantifies a variable’s contribution to intergroup separation and reflects its correlation with the model’s predictive capacity. Consequently, the VIP score serves as a crucial criterion for identifying potential biomarkers, significantly contributing to the elucidation of underlying metabolic mechanisms. By applying a threshold of VIP > 1 and P < 0.05 (Student’s t-test), we identified 35 significant differential metabolites that were consistent across the three groups (Supplementary Table 1).
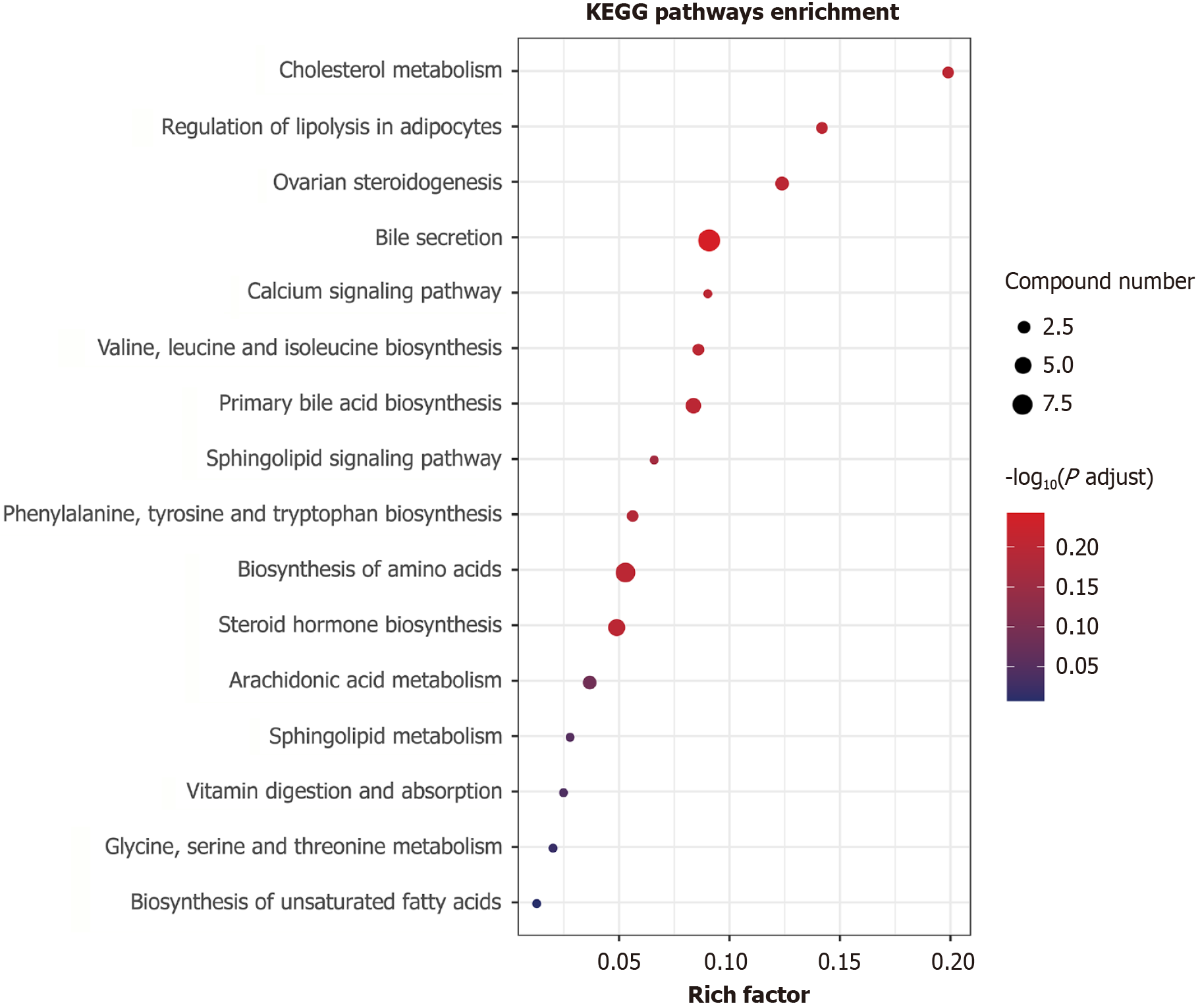
To further elucidate the anti-osteoporotic metabolic mechanisms of XLGB, the identified differential metabolites were subjected to pathway enrichment analysis using the MetaboAnalyst 5.0 database (https://www.metaboanalyst.ca/). This analysis revealed significant involvement of pathways including ovarian steroidogenesis, regulation of adipocyte lipolysis, and primary bile acid biosynthesis (Figure 11). Previous studies have demonstrated a complex interaction between PPAR-γ and Wnt/β-catenin signaling in maintaining the balance between osteogenesis and adipogenesis[18]. In the bone marrow microenvironment, bone marrow mesenchymal stem cells (BMSCs) can differentiate into either os
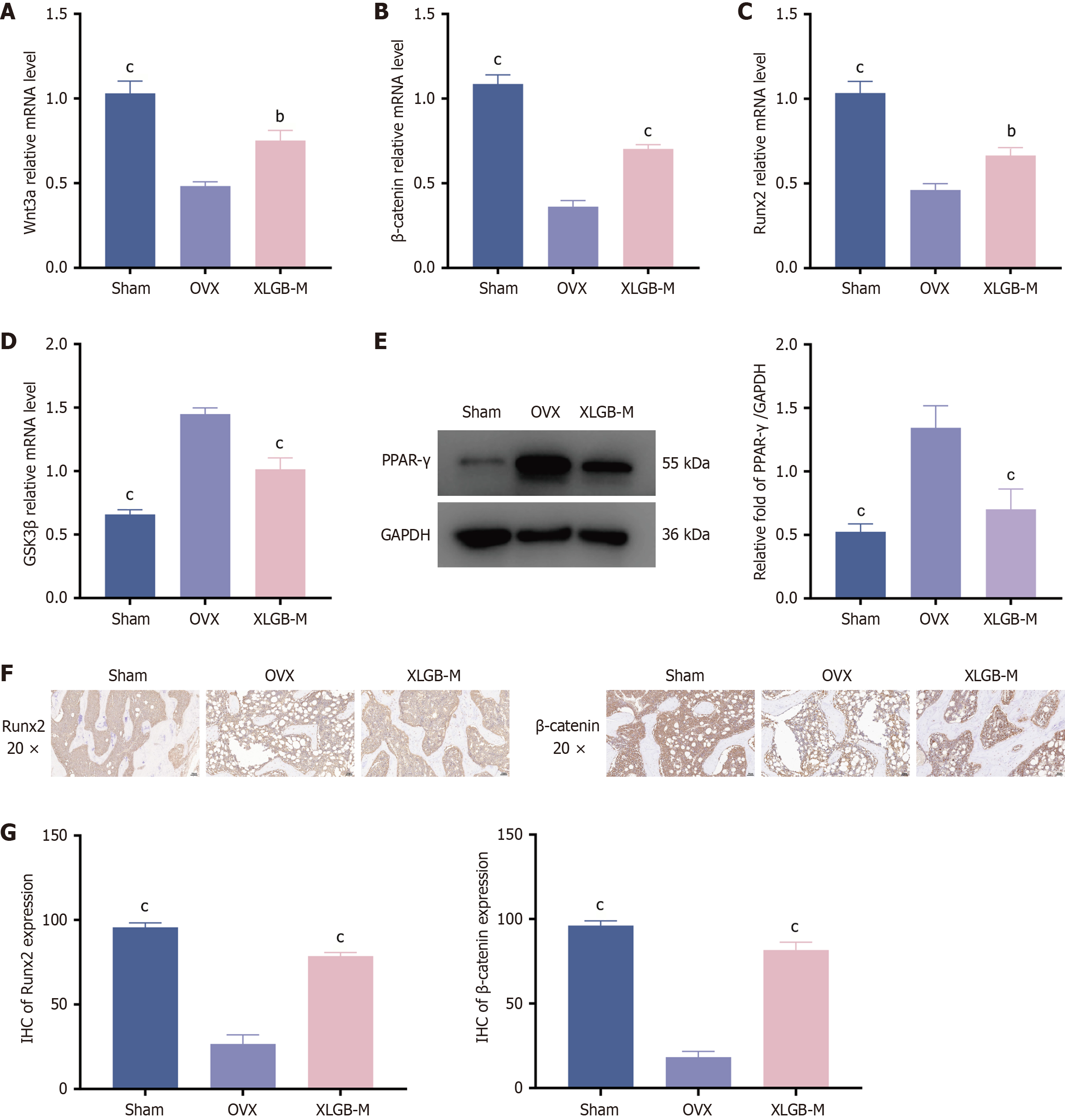
Guided by the Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis, we investigated the roles of key Wnt/β-catenin pathway genes and the master adipogenic regulator PPAR-γ in osteogenesis and lipid metabolism within bone tissue. quantitative PCR analysis revealed that the mRNA expression levels of Wnt3a, β-catenin, and RUNX2 were significantly decreased in the OVX group compared to the Sham group, while GSK3β expression was markedly increased. XLGB-M treatment effectively reversed these alterations in gene expression induced by OVX (Figure 12A-D). Conversely, Western blot analysis demonstrated a significant upregulation of PPAR-γ protein in the OVX group, which was notably attenuated by XLGB-M intervention (Figure 12E). Consistent with the quantitative PCR findings, immunohistochemistry confirmed that the protein levels of RUNX2 and β-catenin were significantly lower in OVX rats and were robustly increased following XLGB-M treatment (Figure 12F and G).
This study employed a multi-omics approach to elucidate the anti-osteoporotic mechanisms of XLGB in an OVX rat model. The results demonstrate that the medium dose of XLGB provides therapeutic benefits not only by directly enhancing bone microstructure and modulating the OPG/RANK/RANKL axis but also, and perhaps more importantly, by orchestrating restorative changes along the gut-bone axis. This mechanism involves significant remodeling of gut microbiota composition, correction of associated metabolic imbalances, and critical reprogramming of BMSC differentiation. The coordinated regulation of the Wnt/β-catenin and PPAR-γ pathways may underlie this shift towards osteo
In alignment with the traditional Chinese medicine principle that posits the kidney as governing bone health, as well as the established clinical efficacy of XLGB[20-22], our study substantiates the direct osteoprotective effects of XLGB in an estrogen-deficient state. Administration of XLGB-M effectively mitigated OVX-induced deterioration of bone microarchitecture, as evidenced by increased BMD, improved trabecular parameters (BV/TV, trabecular number, and trabecular thickness), and reduced trabecular separation. Concurrently, XLGB-M elevated serum levels of bone formation markers, including PINP, OCN, and RUNX2, while modulating the critical OPG/RANK/RANKL signaling pathway, thereby favoring bone formation and attenuating excessive bone resorption[23,24]. Collectively, these findings provide a robust mechanistic basis for the bone-protective properties of XLGB, with XLGB-M identified as the most effective dosage.
Beyond its direct skeletal effects, our 16S rRNA gene sequencing analyses revealed that the therapeutic efficacy of XLGB is closely associated with a significant restructuring of the gut microbial community. Although alpha diversity metrics remained relatively unchanged, beta diversity analysis demonstrated distinct clustering among the Sham, OVX, and XLGB-M groups, indicating a qualitative shift in community structure[25,26]. Notably, XLGB-M normalized the OVX-induced elevation of the Firmicutes/Bacteroidota ratio, a parameter often linked to a pro-inflammatory states and disturbances detrimental to bone metabolism[27,28]. The reduction of this ratio, primarily driven by an increase in Bacteroidota abundance, suggests a potential shift towards a less inflammatory gut environment, possibly mediated by bacterial metabolites such as short-chain fatty acids, which have been implicated in promoting bone health[29,30].
At the genus level, XLGB-M treatment increased the relative of Prevotella, a genus previously reported to mitigate bone loss by reinforcing gut barrier function and suppressing systemic inflammation[31]. Concurrently, XLGB-M reduced the abundance of Ruminococcus, a genus frequently enriched in intestinal inflammatory disorders[32-35]. It is essential to note that our Spearman correlation analysis identified a negative correlation between Ruminococcus and bone formation markers (e.g., OCN, PINP), suggesting a potential link but not establishing causality. The complex, context-dependent nature of host-microbiota interactions implies that the influence of any single taxon is not absolute[36,37]. For instance, the observed decrease in Alloprevotella, which was positively correlated with BV, further highlights this complexity[38,39]. Therefore, the therapeutic benefits of XLGB likely arises from a comprehensive restoration of microbial ecosystem integrity homeostasis rather than from the modulation of individual bacterial species.
The restructuring of the gut microbiota was accompanied by significant alterations in the host metabolome[40,41]. Pathway enrichment analysis identified several metabolic processes in the therapeutic effects of XLGB, including ovarian steroidogenesis, primary bile acid biosynthesis, and the regulation of adipocyte lipolysis. The XLGB-M intervention effectively reversed the OVX-induced depletion of key bile acids, such as deoxycholic acid. Bile acids act as signaling molecules that modulate bone metabolism through receptors such as farnesoid X receptor and G-protein coupled bile acid receptor 1, and dysregulation of these receptors is associated with bone loss[42,43]. Furthermore, XLGB-M restored levels of lipid species, including sphingomyelins and phospholipids, indicating a systemic improvement in lipid me
Integrating the findings of this study, we propose a comprehensive mechanistic model in which XLGB likely influences cell fate decisions within the bone marrow microenvironment by modulating the gut microbiota-metabolite axis. BMSCs have the capacity to differentiate into osteoblasts or adipocytes, with an imbalance favoring adipogenesis over osteo
Although the sample sizes varied between the multi-omics and molecular biology analyses, the principal findings related to gut microbiota composition, metabolomic profiles, and Wnt/β-catenin and PPAR-γ signaling pathways were highly consistent and mutually corroborative. This convergence of evidence enhances the overall validity of our con
Future studies, such as fecal microbiota transplantation or the utilization of knockout models, will be valuable for further elucidating the causal relationships within this pathway.
In summary, our multi-omics study indicates that the XLGB may alleviate estrogen deficiency-induced osteoporosis by modulating the gut-bone axis. The XLGB-M was found to improved bone microstructure and serum biomarkers of bone formation, which correlated with a restructuring of the gut microbiota and correction of related metabolic disturbances. Further mechanistic investigations suggest that these therapeutic effects are potentially mediated, at least in part, through the coordinated activation of the osteogenic Wnt/β-catenin pathway signaling pathway alongside the inhibition of the adipogenic PPAR-γ pathway within the bone marrow. These findings provide experimental evidence supporting the potential of XLGB as a multi-targeted therapy for osteoporosis.
| 1. | Adejuyigbe B, Kallini J, Chiou D, Kallini JR. Osteoporosis: Molecular Pathology, Diagnostics, and Therapeutics. Int J Mol Sci. 2023;24:14583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 91] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 2. | Foessl I, Dimai HP, Obermayer-Pietsch B. Long-term and sequential treatment for osteoporosis. Nat Rev Endocrinol. 2023;19:520-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 167] [Article Influence: 55.7] [Reference Citation Analysis (1)] |
| 3. | Cheng CH, Chen LR, Chen KH. Osteoporosis Due to Hormone Imbalance: An Overview of the Effects of Estrogen Deficiency and Glucocorticoid Overuse on Bone Turnover. Int J Mol Sci. 2022;23:1376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 267] [Cited by in RCA: 368] [Article Influence: 92.0] [Reference Citation Analysis (1)] |
| 4. | Muñoz M, Robinson K, Shibli-Rahhal A. Bone Health and Osteoporosis Prevention and Treatment. Clin Obstet Gynecol. 2020;63:770-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 5. | Seely KD, Kotelko CA, Douglas H, Bealer B, Brooks AE. The Human Gut Microbiota: A Key Mediator of Osteoporosis and Osteogenesis. Int J Mol Sci. 2021;22:9452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 134] [Article Influence: 26.8] [Reference Citation Analysis (1)] |
| 6. | Guan Z, Xuanqi Z, Zhu J, Yuan W, Jia J, Zhang C, Sun T, Leng H, Jiang C, Xu Y, Song C. Estrogen deficiency induces bone loss through the gut microbiota. Pharmacol Res. 2023;196:106930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 66] [Article Influence: 22.0] [Reference Citation Analysis (2)] |
| 7. | Li L, Rao S, Cheng Y, Zhuo X, Deng C, Xu N, Zhang H, Yang L. Microbial osteoporosis: The interplay between the gut microbiota and bones via host metabolism and immunity. Microbiologyopen. 2019;8:e00810. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 78] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 8. | Luo MH, Zhao JL, Xu NJ, Xiao X, Feng WX, Li ZP, Zeng LF. Comparative Efficacy of Xianling Gubao Capsules in Improving Bone Mineral Density in Postmenopausal Osteoporosis: A Network Meta-Analysis. Front Endocrinol (Lausanne). 2022;13:839885. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 9. | Tang XY, Gao MX, Xiao HH, Yun WJ, Dai Y, Yao ZH, Wong MS, Yao XS. Simultaneous Quantitative Analysis of Multiple Biotransformation Products of Xian-Ling-Gu-Bao, a Traditional Chinese Medicine Prescription, with Rat Intestinal Microflora by Ultra-Performance Liquid Chromatography Tandem Triple Quadrupole Mass Spectrometry. J Chromatogr Sci. 2020;58:494-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 10. | Lei Y, Bu XZ, Zhang XB, Wang K, Meng DF, Li HY. [Research Progress on Clinical Application and Pharmacological Mechanism of Xianling Gubao Capsule]. Liaoning Zhongyiyao Daxue Xuebao. 2025;27:119-125. [DOI] [Full Text] |
| 11. | Zheng XX, Wei LW, Xie Y, Zhang H, Qin N. [Gusong Yigu Formula for Treating Postmenopausal Osteoporosis Based on Gut Microbiota and Metabolites]. Zhongyi Xuebao. 2024;39:1388-1393. [DOI] [Full Text] |
| 12. | Chen C, Lei H, Zhao Y, Hou Y, Zheng H, Zhang C, Cao Z, Wu F, Chen G, Song Y, Zhang C, Zhou J, Lu Y, Xie D, Zhang L. A novel small molecule effectively ameliorates estrogen deficiency-induced osteoporosis by targeting the gut-bone signaling axis. Eur J Pharmacol. 2023;954:175868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 13. | Wang S, Wang S, Wang X, Xu Y, Zhang X, Han Y, Yan H, Liu L, Wang L, Ye H, Li X. Effects of Icariin on Modulating Gut Microbiota and Regulating Metabolite Alterations to Prevent Bone Loss in Ovariectomized Rat Model. Front Endocrinol (Lausanne). 2022;13:874849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 14. | Lv H, Jia H, Cai W, Cao R, Xue C, Dong N. Rehmannia glutinosa polysaccharides attenuates colitis via reshaping gut microbiota and short-chain fatty acid production. J Sci Food Agric. 2023;103:3926-3938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (1)] |
| 15. | Yang Y, Cheng R, Liu J, Fang J, Wang X, Cui Y, Zhang P, Du B. Linarin Protects against Cadmium-Induced Osteoporosis Via Reducing Oxidative Stress and Inflammation and Altering RANK/RANKL/OPG Pathway. Biol Trace Elem Res. 2022;200:3688-3700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 16. | Tobeiha M, Moghadasian MH, Amin N, Jafarnejad S. RANKL/RANK/OPG Pathway: A Mechanism Involved in Exercise-Induced Bone Remodeling. Biomed Res Int. 2020;2020:6910312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 216] [Article Influence: 36.0] [Reference Citation Analysis (1)] |
| 17. | Ding K, Hua F, Ding W. Gut Microbiome and Osteoporosis. Aging Dis. 2020;11:438-447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 96] [Article Influence: 16.0] [Reference Citation Analysis (5)] |
| 18. | Yuan Z, Li Q, Luo S, Liu Z, Luo D, Zhang B, Zhang D, Rao P, Xiao J. PPARγ and Wnt Signaling in Adipogenic and Osteogenic Differentiation of Mesenchymal Stem Cells. Curr Stem Cell Res Ther. 2016;11:216-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 173] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 19. | Vallée A, Lecarpentier Y. Crosstalk Between Peroxisome Proliferator-Activated Receptor Gamma and the Canonical WNT/β-Catenin Pathway in Chronic Inflammation and Oxidative Stress During Carcinogenesis. Front Immunol. 2018;9:745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 326] [Article Influence: 40.8] [Reference Citation Analysis (5)] |
| 20. | Yang Y, Nian H, Tang X, Wang X, Liu R. Effects of the combined Herba Epimedii and Fructus Ligustri Lucidi on bone turnover and TGF-β1/Smads pathway in GIOP rats. J Ethnopharmacol. 2017;201:91-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 21. | Cheng BR, Wu RY, Gao QY, Jiang KX, Li SS, Qi SH, Yuan MY, Liu JP. Chinese Proprietary Medicine Xianling Gubao Capsule for Osteoporosis: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Front Endocrinol (Lausanne). 2022;13:870277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 22. | Jia Y, Sun J, Zhao Y, Tang K, Zhu R, Zhao W, Wang R, Zhang Y, Lin N, Chen W. Chinese patent medicine for osteoporosis: a systematic review and meta-analysis. Bioengineered. 2022;13:5581-5597. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 23. | Liao M, He Q, Yang J, Gou L, Ma K, Wang H. Study on the Mechanism of Xianling Gubao Capsule Regulating Runt-Related Transcription Factor 2 (RUNX2) and Promoting Osteoblast Differentiation by N6-Methyladenosine (m6A) Methyltransferase-Like 3 (METTL3). Altern Ther Health Med. 2024;AT10075. [PubMed] |
| 24. | Chen X, Li Y, Zhang Z, Chen L, Liu Y, Huang S, Zhang X. Xianling Gubao attenuates high glucose-induced bone metabolism disorder in MG63 osteoblast-like cells. PLoS One. 2022;17:e0276328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 25. | Tang XY, Gao MX, Xiao HH, Dai ZQ, Yao ZH, Dai Y, Yao XS. Effects of Xian-Ling-Gu-Bao capsule on the gut microbiota in ovariectomized rats: Metabolism and modulation. J Chromatogr B Analyt Technol Biomed Life Sci. 2021;1176:122771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 26. | Zhou J, Wang R, Zhao R, Guo X, Gou P, Bai H, Lei P, Xue Y. Intermittent Parathyroid Hormone Alters Gut Microbiota in Ovariectomized Osteoporotic Rats. Orthop Surg. 2022;14:2330-2338. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 27. | D'Amelio P, Sassi F. Gut Microbiota, Immune System, and Bone. Calcif Tissue Int. 2018;102:415-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 221] [Article Influence: 27.6] [Reference Citation Analysis (4)] |
| 28. | Guo M, Liu H, Yu Y, Zhu X, Xie H, Wei C, Mei C, Shi Y, Zhou N, Qin K, Li W. Lactobacillus rhamnosus GG ameliorates osteoporosis in ovariectomized rats by regulating the Th17/Treg balance and gut microbiota structure. Gut Microbes. 2023;15:2190304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 202] [Reference Citation Analysis (1)] |
| 29. | Feng R, Wang Q, Yu T, Hu H, Wu G, Duan X, Jiang R, Xu Y, Huang Y. Quercetin ameliorates bone loss in OVX rats by modulating the intestinal flora-SCFAs-inflammatory signaling axis. Int Immunopharmacol. 2024;136:112341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (1)] |
| 30. | Weaver CM. Diet, gut microbiome, and bone health. Curr Osteoporos Rep. 2015;13:125-130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 207] [Cited by in RCA: 182] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 31. | Zhang YW, Cao MM, Li YJ, Sheng RW, Zhang RL, Wu MT, Chi JY, Zhou RX, Rui YF. The Preventive Effects of Probiotic Prevotella histicola on the Bone Loss of Mice with Ovariectomy-Mediated Osteoporosis. Microorganisms. 2023;11:950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 32. | Li N, Wang H, Pei H, Wu Y, Li L, Ren Y, Wang S, Ma Y, Luo M, Yuan J, Li L, Qin D. Genus_Ruminococcus and order_Burkholderiales affect osteoporosis by regulating the microbiota-gut-bone axis. Front Microbiol. 2024;15:1373013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 33. | Nishino K, Nishida A, Inoue R, Kawada Y, Ohno M, Sakai S, Inatomi O, Bamba S, Sugimoto M, Kawahara M, Naito Y, Andoh A. Analysis of endoscopic brush samples identified mucosa-associated dysbiosis in inflammatory bowel disease. J Gastroenterol. 2018;53:95-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 335] [Article Influence: 41.9] [Reference Citation Analysis (1)] |
| 34. | Čipčić Paljetak H, Barešić A, Panek M, Perić M, Matijašić M, Lojkić I, Barišić A, Vranešić Bender D, Ljubas Kelečić D, Brinar M, Kalauz M, Miličević M, Grgić D, Turk N, Karas I, Čuković-Čavka S, Krznarić Ž, Verbanac D. Gut microbiota in mucosa and feces of newly diagnosed, treatment-naïve adult inflammatory bowel disease and irritable bowel syndrome patients. Gut Microbes. 2022;14:2083419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 35. | Buisson A, Sokol H, Hammoudi N, Nancey S, Treton X, Nachury M, Fumery M, Hébuterne X, Rodrigues M, Hugot JP, Boschetti G, Stefanescu C, Wils P, Seksik P, Le Bourhis L, Bezault M, Sauvanet P, Pereira B, Allez M, Barnich N; Remind study group. Role of adherent and invasive Escherichia coli in Crohn's disease: lessons from the postoperative recurrence model. Gut. 2023;72:39-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 36. | Zhang YW, Song PR, Wang SC, Liu H, Shi ZM, Su JC. Diets intervene osteoporosis via gut-bone axis. Gut Microbes. 2024;16:2295432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 113] [Article Influence: 56.5] [Reference Citation Analysis (1)] |
| 37. | Knudsen JK, Leutscher P, Sørensen S. Gut Microbiota in Bone Health and Diabetes. Curr Osteoporos Rep. 2021;19:462-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 38. | Yang J, Chen C, Zhang H, Chen B, Xiao K, Tang Y, Meng K, Qin L, Chen P. Serum metabolomics and 16S rRNA amplicon sequencing reveal the role of puerarin in alleviating bone loss aggravated by antidiabetic agent pioglitazone in type 2 diabetic mice. J Ethnopharmacol. 2025;340:119128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 39. | Wang H, Huang J, Ding Y, Zhou J, Gao G, Han H, Zhou J, Ke L, Rao P, Chen T, Zhang L. Nanoparticles Isolated From Porcine Bone Soup Ameliorated Dextran Sulfate Sodium-Induced Colitis and Regulated Gut Microbiota in Mice. Front Nutr. 2022;9:821404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 40. | Lin X, Xiao HM, Liu HM, Lv WQ, Greenbaum J, Gong R, Zhang Q, Chen YC, Peng C, Xu XJ, Pan DY, Chen Z, Li ZF, Zhou R, Wang XF, Lu JM, Ao ZX, Song YQ, Zhang YH, Su KJ, Meng XH, Ge CL, Lv FY, Luo Z, Shi XM, Zhao Q, Guo BY, Yi NJ, Shen H, Papasian CJ, Shen J, Deng HW. Gut microbiota impacts bone via Bacteroides vulgatus-valeric acid-related pathways. Nat Commun. 2023;14:6853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 62] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 41. | Zhang YW, Cao MM, Li YJ, Lu PP, Dai GC, Zhang M, Wang H, Rui YF. Fecal microbiota transplantation ameliorates bone loss in mice with ovariectomy-induced osteoporosis via modulating gut microbiota and metabolic function. J Orthop Translat. 2022;37:46-60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 114] [Article Influence: 28.5] [Reference Citation Analysis (1)] |
| 42. | Yu C, Sun R, Yang W, Gu T, Ying X, Ye L, Zheng Y, Fan S, Zeng X, Yao S. Exercise ameliorates osteopenia in mice via intestinal microbial-mediated bile acid metabolism pathway. Theranostics. 2025;15:1741-1759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 27] [Article Influence: 27.0] [Reference Citation Analysis (1)] |
| 43. | Liu J, Chen Y, Luo Q. The Association of Serum Total Bile Acids With Bone Mineral Density in Chinese Adults Aged 20-59: A Retrospective Cross-Sectional Study. Front Endocrinol (Lausanne). 2022;13:817437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 44. | Li B, Wang Y, Gong S, Yao W, Gao H, Liu M, Wei M. Puerarin improves OVX-induced osteoporosis by regulating phospholipid metabolism and biosynthesis of unsaturated fatty acids based on serum metabolomics. Phytomedicine. 2022;102:154198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 45. | Lin WY, Radu F, Schuler C, Leggett RE, Mannikarottu A, Levin RM. The effect of ovariectomy and oestrogen therapy on the free fatty acid content, endogenous lipase activity, and phospholipid content of the rabbit urinary bladder. BJU Int. 2008;102:885-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 46. | Wei X, Zhang Y, Sun C, Qi B, Huang X, Chen M, Liu N, Sun K, Chen X, Qin X, Xie Y, Zhu L. Elucidating the relationship between dyslipidemia and osteoporosis: A multicenter, prospective cohort study protocol. Front Cardiovasc Med. 2022;9:901786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 47. | Hu L, Yin C, Zhao F, Ali A, Ma J, Qian A. Mesenchymal Stem Cells: Cell Fate Decision to Osteoblast or Adipocyte and Application in Osteoporosis Treatment. Int J Mol Sci. 2018;19:360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 365] [Cited by in RCA: 329] [Article Influence: 41.1] [Reference Citation Analysis (1)] |
| 48. | Hu L, Chen W, Qian A, Li YP. Wnt/β-catenin signaling components and mechanisms in bone formation, homeostasis, and disease. Bone Res. 2024;12:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 214] [Reference Citation Analysis (5)] |
| 49. | Lefterova MI, Haakonsson AK, Lazar MA, Mandrup S. PPARγ and the global map of adipogenesis and beyond. Trends Endocrinol Metab. 2014;25:293-302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 596] [Cited by in RCA: 517] [Article Influence: 43.1] [Reference Citation Analysis (4)] |
| 50. | Xu C, Wang J, Zhu T, Shen Y, Tang X, Fang L, Xu Y. Cross-Talking Between PPAR and WNT Signaling and its Regulation in Mesenchymal Stem Cell Differentiation. Curr Stem Cell Res Ther. 2016;11:247-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 79] [Article Influence: 7.9] [Reference Citation Analysis (1)] |