Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.114756
Revised: December 3, 2025
Accepted: February 2, 2026
Published online: March 24, 2026
Processing time: 172 Days and 10.5 Hours
Microsatellite instability (MSI) of colorectal cancer (CRC) is an important bio
To identify CT-based imaging features without radiomics to differentiate between MSI status in CRC.
Imaging features of 109 subjects (58 male, 51 female) from 2011 to 2018 were stratified based on MSI status using CT scan images and pathological records. These variables included primary tumor location, primary tumor size, initial cancer stage, metastatic locations, primary attenuation compared to the liver, primary tumor growth pattern, primary tumor content, tumor margin, tumor density, and the presence or absence of mesenteric infiltration for primary tumor. Statistical analysis was performed using χ2, t tests, and Mann-Whitney U tests.
Four clinical characteristics were statistically significant for MSI-high (MSI-H). Although the MSI-H mass is larger (P < 0.001), the MSI-H tumors have a lower cancer stage at initial diagnosis (P = 0.012) as the MSI-H CRC tends to have regional metastasis than distant metastasis (P < 0.001). MSI-H CRC tends to be right-sided while MSI-low CRC tends to be left-sided (P < 0.001). The Hounsfield unit SD was lower in the MSI-H than the MSI-low CRC, P = 0.002. The remaining primary imaging characteristics (tumor attenuation relative to the liver, tumor content, tumor growth pattern, and tumor margin) were not statistically different. Both classes of tumors were more likely to be well-defined with mucosal growth and hypoattenuating relative to the liver on portovenous phase imaging.
Four clinical and one imaging characteristics were statistically significant for MSI-H CRC tumors. Larger and pro
Core Tip: Microsatellite instability in colorectal tumors tends to have right-sided and larger tumor and is less likely to have distant metastasis at the time of initial diagnosis. Therefore, if a left-sided cancer with distant metastasis is seen at initial diagnosis, it has a higher likelihood of being a microsatellite instability low tumor.
- Citation: Stearns J, McLaughlin S, Kuckelman T, Hutzley S, Chiu A, Kloft L, Bay C, Kundranda M, Chang J. Colorectal cancer computed tomography imaging features involving microsatellite instability. World J Clin Oncol 2026; 17(3): 114756
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/114756.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.114756
Colorectal cancer (CRC) is the third most frequently diagnosed cancer and the third most common cause of cancer-related death in the United States. In 2023, approximately 153000 Americans would be diagnosed with CRC while 52500 would die[1]. Treatment for this disease has evolved over time from cytotoxic chemotherapy to targeted molecular therapy. Most recently, immunotherapy targeting immune inhibition in tumors has yielded outstanding response and survival data, although this is limited to tumors with high tumor mutational burden typically seen in microsatellite unstable/de
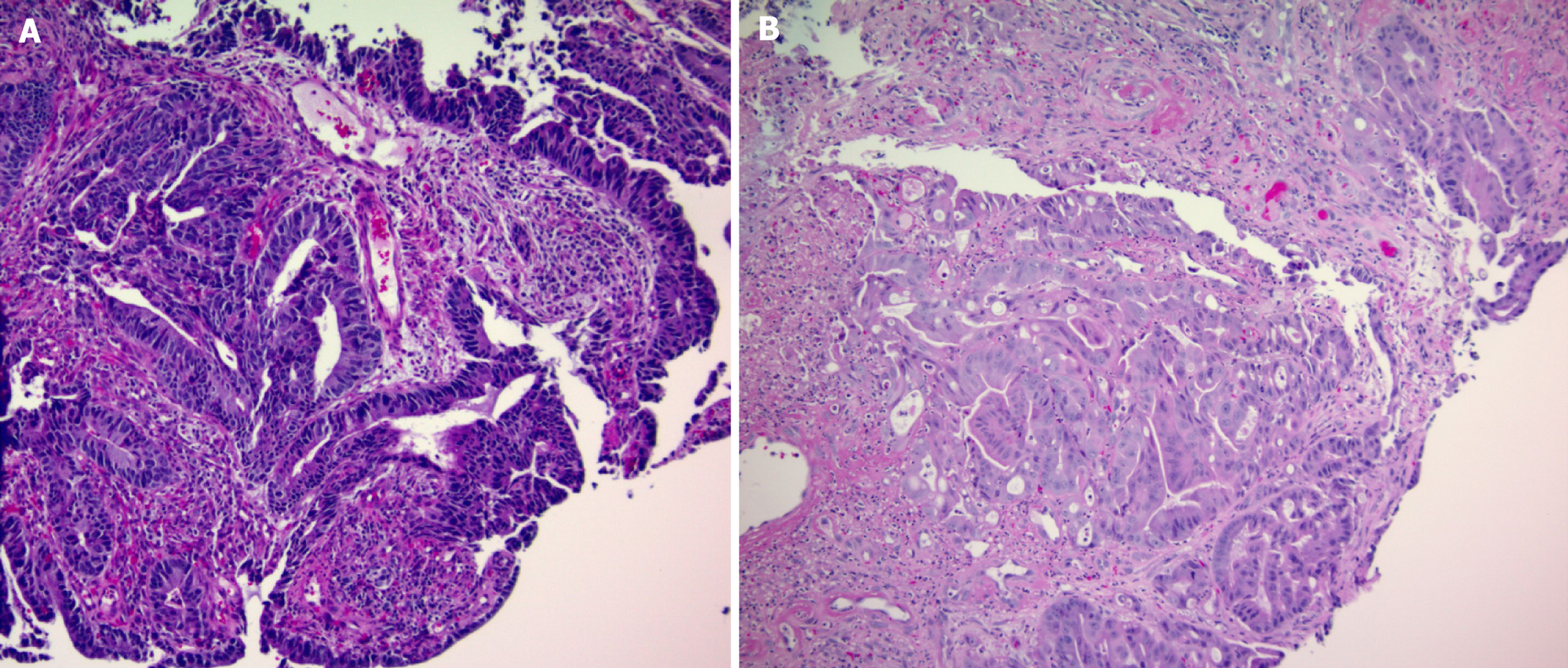
Microsatellites are short, repeating DNA sequences found throughout the human genome and have recently become important for treatment decisions[4,5]. Due to their repetitive structure, microsatellites have an increased propensity for replication errors, which under normal circumstance are corrected by the cellular DNA MMR system. Abnormalities in the MMR system prevent the cell from correcting these errors resulting in high mutational burden that can lead to cancer[6]. Microsatellite instability (MSI) refers to genetic hypermutability that results from defects in the MMR system, which is classified into three different categories: Microsatellite stability (MSS), MSI-low (MSI-L), or MSI-high (MSI-H). Histologically, these difference manifest as greater regions of mucin and infiltrating lymphocytes in the tumor[7] (Figure 1). For treatment purposes, MSI-L and MSS are grouped together due to similarity of their clinical response to treatment. MSI-H is defined by mutations in > 30% of microsatellite and is linked to several diseases such as Lynch syndrome[4,8]. MSI status is an important consideration when determining treatment modality as MSS and MSI-L tumors do not respond to programmed death-ligand 1 or programmed death-ligand 1-cytotoxic T-lymphocyte-associated protein 4 combination inhibitors[2,3,9]. MSI-H tumors also have a more favorable stage-adjusted prognosis when compared to those with MSI-L or MSS[9]. Earlier detection and categorization of MSI status via computed tomography (CT) imaging could improve out
Research on predicting MSI status using both radiomics and patient clinical factors have shown predictive accuracy ranging between 50%-90% depending on the classification model as well as training and validation data sets[10-12]. Patient age, tumor location, carcinoembryonic antigen levels, and smoking history are a few examples of the many characteristics considered to be valuable predictors of MSI status[10-12]. These factors, combined with radiomic features, could be used to predict MSI status of colorectal tumors non-invasively[6,10]. Study by Cao et al[10] found that a MSI predictive system utilizing a combination of clinical factors and CT-based radiomic features successfully predicts MSI status. The group developed a nomogram combining 32 radiomic features (out of 1032) into a numerical value in the final model with 3 additional clinical factors to achieve an area under the curve of 0.96. Likewise, the study from Pei et al[11] also demonstrated predictive radiomic features with 16 features (out of 352) combined with 4 clinical factors achieved area under the curve of 0.74 using a large training and validation dataset of 762 subjects. These studies were included as part of a systematic review of radiomic-based predictive models for MSI in CRC[13]. However, despite these promising findings, the validity of radiomic CT analysis for MSI status remains debated. Many studies have shown the potential of radiomic features in categorizing MSI; however, more research is needed to further validate the predictive quality of these features and its impact on patient outcomes.
The past research has focused on radiomic findings which require additional image processing and image trans
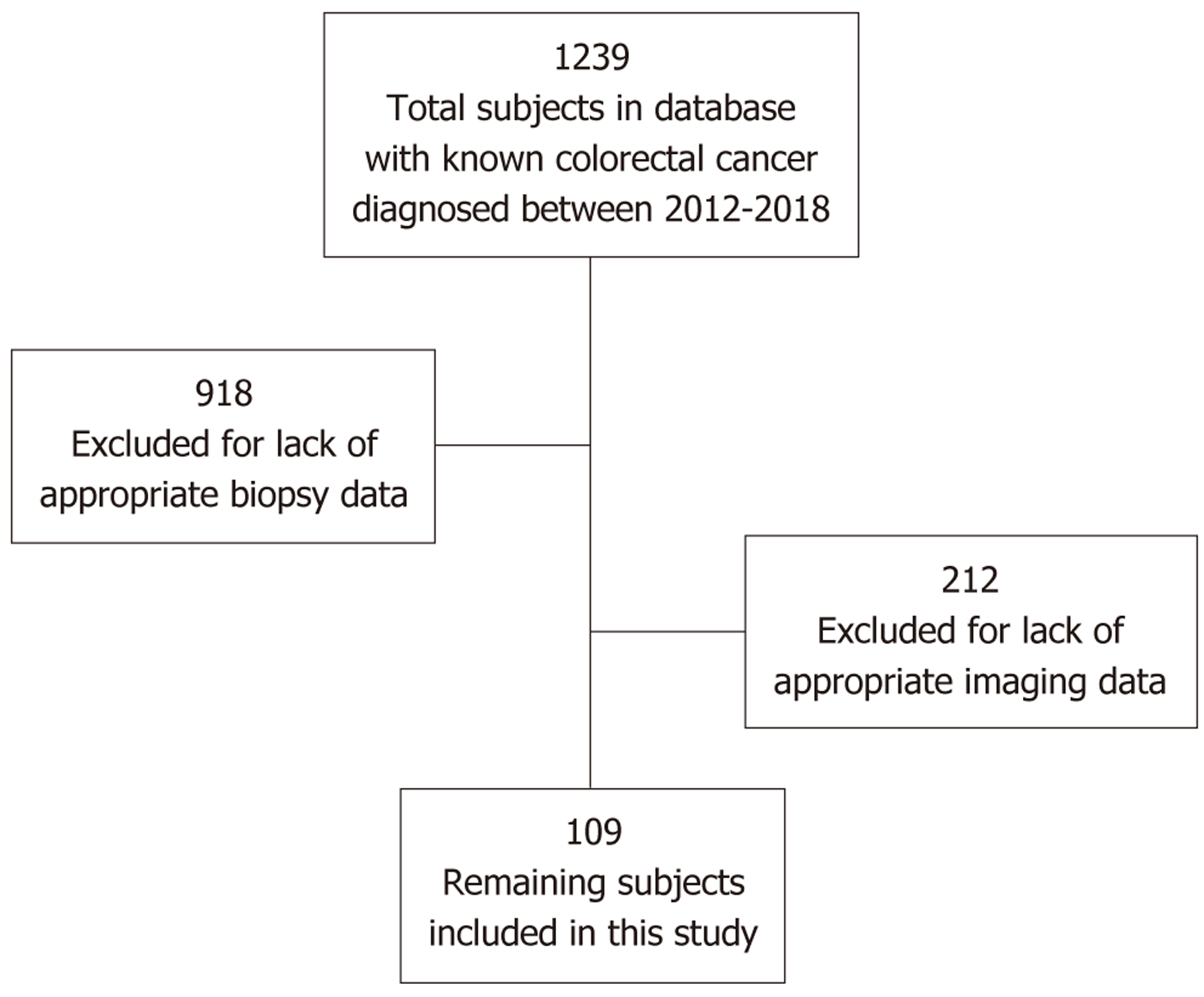
The retrospective study protocol was approved by our local institutional review board. The list of subjects were obtained from our institutional tumor registry. Subjects were selected from a database of 1239 patients with known CRC diagnosed from 2012 to 2018 at our local, tertiary cancer center. For each subject, the electronic medical record was searched for information regarding MSI status. This was done using the Cerner electronic medical record search function for key words such as MSI, pathology, staging, and genetics. Clinical notes were also manually searched to find pathology results that may have been obtained at outside institution on initial investigation. If there were genetic results for MSI status available, the patient was included for further evaluation. Additional exclusion criteria included previous treatment of the tumor with chemotherapy or radiation, history of colon resection, or colon involvement by metastasis of a prior malignancy. This resulted in 54 subjects with MSI-H pathology and 79 subjects with MSI-L pathology. Then, each patient chart was reviewed again to determine if there was available CT imaging at the time of initial diagnosis with an identifiable colorectal tumor. This excluded an additional 24 subjects. The final list of eligible subjects had 31 MSI-H and 78 MSI-L tumors. These eligible subjects did not have to receive treatment at our cancer center. Figure 2 shows the inclusion and exclusion criteria.
The retrospectively collected data included clinical data and imaging features. Clinical data included subject age, gender, primary tumor site location, clinical tumor-nodes-metastasis (TNM), clinical stage, pathologic TNM, pathologic stage, Kirsten rat sarcoma viral oncogene homolog mutation status, B-Raf proto-oncogene, serine/threonine kinase mutation status, and MSI status. Various imaging features of the primary mass were obtained. These features included the location of primary tumor (determined by the location of the primary mass which is then converted into right or left sided defined by the mid-transverse colon), size of primary tumor mass defined in the transverse plane, location of the metastasis, primary mass attenuation compared to the liver, primary mass content (heterogeneity), primary mass density, growth pattern, tumor margin, the presence or absence of mesenteric infiltration, and normal liver enhancement density. Imaging features were reviewed by a board certified body imaging radiologist after initial identification and classification of the tumor by participating medical students. The medical students were instructed to identify the colon based on anatomy and identify lesions that were either mass-like or wall-thickening. These lesions were then reviewed with Chang J (with 13 years of independent body imaging practice).
Data for tumor stage were gathered through the patient’s clinical chart, imaging report, and pathology report. This combination of information provides the involvement of colonic wall layers, regional lymph nodes, and the distant organs. These data provide the initial TNM staging which were then converted to the standard anatomical staging from 0-4 with 0 being in situ disease and 4 being metastatic disease. In terms of metastatic classification, if the disease involved only the colon without nodal or distant metastasis, then it was classified as local disease (N and M = 0). For any invol
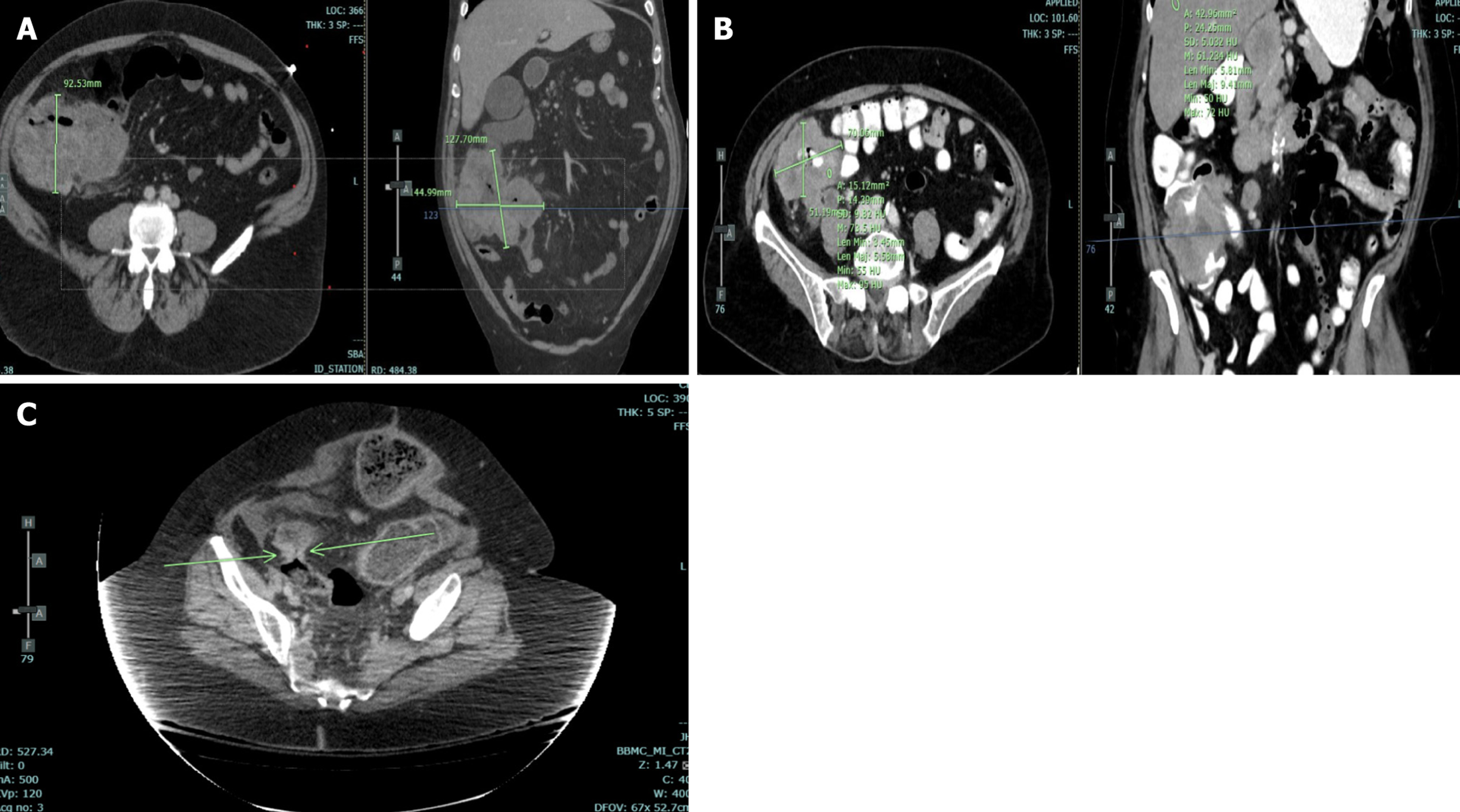
Imaging data are obtained according to the following description. Size of the primary tumor is obtained in the axial plane on the image with the largest cross-section area. The size is then measured along the longest axis with the short-axis measurement obtained perpendicular to the long axis to obtain the largest cross-section area of the tumor. Lymph nodes are considered abnormal if the short axis is ≥ 1 cm or if the nodes are rounded in appearance. Metastasis is determined by pathology report or clinical notes. Tumor heterogeneity/content classification was determined if the primary mass has at least 30% of the area being different in density than the dominant, enhancing area. The type of growth pattern is classified as either along the wall or mass-like. Growth along the wall is when the mass extends mostly along the wall of the colon while mass-like growth tends to be rounded or polypoid in morphology. Tumor margin is classified as either well-defined or ill-defined. Well-defined tumors have sharply marginated borders whereas ill-defined tumors have borders that could not clearly be identified. Mesenteric infiltration is defined as whether or not the images show pericolonic stranding. This definition may be confounded by the edema that is present rather than true tumor infiltration. Unfortunately, with CT, this difference could not be clearly differentiated. Primary tumor enhancement is determined relative to the normal liver density. It is considered hypoattenuating if the density is less than normal liver while hyperattenuating is density greater than normal liver. Isoattenuating is when the mass density is within ± 10% of the liver attenuation. The density is measured in the enhancing regions of the primary mass by using a rounded region of interest measurement tool with at least 0.5 cm diameter. An example of the features described above are shown in Figure 3.
Summary descriptive statistics, including mean ± SD, median (interquartile ranges) and n (%) are provided, as appr
Of the 109 eligible subjects, 58 were male and 51 were female, 31 of the subjects had MSI-H tumors, and 78 had MSI-L tumors. The average age of the subjects in years was 61.4 ± 14.8. Female subjects averaged 63.0 ± 14.7 years old while the male subjects averaged 60.1 ± 14.9. MSI-H subjects averaged 62.4 ± 14.2 while the MSI-L subjects averaged 61 ± 15.1 years in age. Table 1 lists the demographics of the subjects and their disease locations and tumor stage.
| Demographic | Number of subjects | Percentage of total | |
| Sex | Male | 58 | 53.2% |
| Female | 51 | 46.8% | |
| Age distribution | 20-29 | 1 | 0.9% |
| 30-39 | 10 | 9.2% | |
| 40-49 | 15 | 13.8% | |
| 50-59 | 18 | 16.5% | |
| 60-69 | 25 | 22.9% | |
| 70-79 | 29 | 26.6% | |
| 80-89 | 10 | 9.2% | |
| 90-99 | 1 | 0.9% | |
| MSI status | MSI-high | 31 | 28.4% |
| MSI-low | 78 | 71.6% | |
| Primary tumor clinical stage | 0 | 2 | 1.8% |
| I | 7 | 6.4% | |
| II | 29 | 26.6% | |
| III | 41 | 37.6% | |
| IV | 30 | 27.5% | |
| Primary tumor location | Right colon | 55 | 50.5% |
| Left colon | 54 | 49.5% |
Tumor staging: Of the 78 MSI-L lesions, 2.6% (2) were clinical stage 0, 5.1% (4) were clinical stage 1, 17.9% (14) were clinical stage 2, 42.3% (33) were clinical stage 3, and 32.1% (25) were clinical stage 4. Of the 31 MSI-H lesions, 9.7% (3) were clinical stage 1, 48.4% (15) were clinical stage 2, 25.8% (8) were clinical stage 3, and 16.1% (5) were clinical stage 4. The distributions across MSI categories were significantly different, P = 0.012. Most notably, when analyzing frequencies of clinical stages 2 and 3 between MSI categories, MSI-H lesions were more likely to be clinical stage 2, and MSI-L lesions were more likely to be clinical stage 3.
Tumor size: MSI-H had a higher primary mass total area (average area of 31.7 cm2) than MSI-L tumors (16.2 cm2), P = 0.001. The distribution of primary mass area was larger in the MSI-H than MSI-L group; for the MSI-L group: Median = 13.98 cm2, [interquartile range (IQR): 7.89 to 20.18] vs MSI-H group: Median = 21.0 cm2 (IQR: 12.04 to 38.94).
Tumor location: In the MSI-L group, 64.1% (50) of tumors were found in the left colon, while 35.9% (28) were found in the right colon; in the MSI-H group, 12.9% (4) were found in the left colon, while 87.1% (27) were found in the right colon, P < 0.001.
Metastasis: Among MSI-L, 52.6% (41) showed no metastasis, 16.7% (13) showed regional metastasis, and 30.8% (24) showed distant metastasis. Among MSI-H, it was found that 51.6% (16) showed no metastasis, 38.7% (12) showed regi
Mesenteric infiltration: In the MSI-L group, 66.7% (52) of tumors were positive for mesenteric infiltration, and 33.3% (26) were negative; in the MSI-H group, 51.6% (16) were positive for mesenteric infiltration, and 48.4% (15) were negative, P = 0.189.
Tumor margin: Both MSI-L (69.2%, 54) and MSI-H (67.7%, 21) tumors were more likely to be well-defined. In the MSI-L group, 69.2% (54) were well-defined and 30.8% (24) were ill-defined. In the MSI-H group, 67.7% (21) were well-defined and 32.3% (10) were ill-defined. MSI-H or MSI-L did not show a difference in this regard, P = 1.0.
Content: Both MSI groups had 1 patient who was indeterminant when evaluating their content and were excluded from statistical analysis. The MSI-L group had 68.8% (53) in the homogeneous group and 31.2% (24) in the heterogeneous group. The MSI-H group had 63.3% (19) in the homogenous group and 36.7% (11) in the heterogeneous group. These distributions did not differ significantly, P = 0.498.
Growth pattern: The MSI-L group had 17.9% (14) tumors exhibiting mass-like growth and 82.1% (64) with mucosal growth; the MSI-H group had 32.3% (10) patients who exhibited mass-like growth and 67.7% (21) patients who had mucosal growth, P = 0.127.
Tumor enhancement: Among the MSI-L group, 6 patients could not be evaluated and 2 patients were excluded for not receiving contrast on imaging. In the MSI-L group, 81.4% (57) were hypoattenuating, 15.7% (11) were isoattenuating, and 2.9% (2) were hyperattenuating. Among the MSI-H group, 6 patients could not be evaluated and 4 patients did not receive contrast on imaging. In the MSI-H group, 95.2% (20) were hypoattenuating, 4.8% (1) were isoattenuating, and 0% were hyperattenuating. The distributions of tumor enhancement did not differ across MSI categories, P = 0.376.
Primary mass density measurement: The distribution of mean primary tumor Hounsfield unit: Hyperdense region was the same across both categories of MSI, P = 0.162. For the MSI-L group: Median = 70.0 (IQR: 54.35 to 83.50) vs MSI-H group: Median = 63.70 (IQR: 46.85 to 77.95). However, the SD of the primary mass enhancement; the primary tumor Hounsfield unit standard deviation was found to differ, P = 0.002. The MSI-H samples had a lower SD than MSI-L samples. For the MSI-L group: Median = 16.400 (IQR: 12.400 to 21.000) vs MSI-H group: Median = 12.550 (IQR: 10.000 to 15.175).
Mean normal liver Hounsfield unit: The distribution of mean liver Hounsfield unit did not differ across categories of MSI, P = 0.105. For the MSI-L group: Median = 100.00 (IQR: 86.90 to 114.60) vs MSI-H group: Median = 92.10 (IQR: 74.73 to 105.68).
CRC remains a significant public health challenge with substantial morbidity and mortality rates, underscoring the importance of improved diagnostic and prognostic methods to optimize patient outcomes. The classification of tumors based on molecular characteristics, such as MSI status, has become integral to decision-making regarding treatment. Tumors with MSI-H status have been shown to have distinct responses to certain treatments[6], making accurate and accessible methods for predicting MSI status vital to improving patient outcomes. In this study, we aimed to investigate the potential of CT imaging features that may predict MSI status. Although our current subject number is insufficient, we aim to eventually acquire sufficient cases to develop an algorithm based on CT imaging features to predict the likelihood of MSI-H status.
One finding of statistical significance was that MSI-H tumors were less likely to show distant metastasis but more likely to be confined to regional lymph nodes. When analyzing the presence of metastasis, these results showed an increased likelihood for CRC tumors with distant metastasis to be MSI-L, and in contrast, showed an increased likelihood for CRC tumors with local metastasis to be MSI-H. These findings are concordant with pathological findings in stage IV CRC[14,15]. The study from Fujiyoshi et al[14] and Kang et al[15] showed that MSI-H is most frequently seen in stage II CRC, while stage IV CRC is associated with MSI-L tumors. These findings are similar to our findings from clinical and CT imaging staging. Of note, the study by Kang et al[15] showed that MSI-H tumors tend to have lower lymph node ratio while MSI-L tumors overall had fewer number of lymph nodes. This is thought to be related to the immunogenicity of MSI-H tumors from neopeptides[16]. This concordance shows that there is potential for CT imaging features to identify underlying molecular features of CRC and has clinical relevance for rapid prediction of MSI statue of CRC.
There are additional findings from the current study that correlated with prior research on the relationship between imaging features and MSI status prediction: Size of the primary mass, location of the tumor, and the standard deviation of the primary tumor density. Our findings align with those of previous studies that showed MSI-H tumors are more likely to occur on the right side of the colon and are more likely to be larger in size[10,11]. These findings are also corroborated by clinical research findings[15]. The MSI-L tumors are also more likely to have distant metastasis both on imaging and clinical studies[11,14,15]. In addition, the lower SD for the primary mass with MSI-H status is consistent with radi
In the published literature, radiomic models have contributed to the prediction of MSI status, although there is significant parameter variability which may limit the generalizability of these models. In this research, the number of radiomic parameters included in the prediction models varied significantly. Although these variables are based on similar radiomic techniques, each provided a different flavor to the technique with varying data transformation and parameter selection. Either all or part of the data was first transformed using different techniques including Laplacian of Gaussian and wavelet filter[10,17], Gabor transformation[18], and Haralick transformation; others used untransformed radiomics data[11,12,19]. The transformations were suggested to have denoising function[10]. The final number of radiomic features selected for analysis also varied from study to study ranging between 6 to 51 variables[10-12,17-20]. These do not all correlate as some had plain radiomic while others had radiomic variables in transformed spaces. The selected clinical variables also varied between studies with commonly included variables of age, tumor location, and carcinoembryonic antigen while other less commonly included variables include tumor grade, smoking history, diabetes history, hypertension history, and family history[10-12,17-20]. Some of these clinical variables are directly related to tumor biological behavior (age and tumor location) while others appear less so (carcinoembryonic antigen, smoking, hypertension, diabetes, and family history). The variability of these parameters makes clinical application more difficult. In addition, there are also intrinsic data acquisition variability that limit general application of this technique, which will require significant research and technique standardization prior to generalized clinical application[21,22]. This suggests that detailed imaging analysis may have easier standardization than second order or higher radiomic techniques.
The remaining analyses showed statistically insignificant relationships between mesenteric infiltration, tumor margin, tumor content, growth pattern, tumor enhancement, mean primary Hounsfield unit, and mean liver Hounsfield units. Even though prior studies did reveal MSI-H tumors to be more poorly differentiated, this feature is not identifiable on the CT imaging features that we evaluated including tumor content, tumor margin, growth pattern, and mesenteric infiltration. These imaging features may not be capable of differentiating the underlying molecular pathological differences as there is significant interplay between the mutations and chromosomal abnormalities with the tumor microenvironment[14,23]. There are other mutations which may confer different physiological differences that may manifest similarly on CT imaging features; these mutations include B-Raf proto-oncogene, serine/threonine kinase, Kirsten rat sarcoma viral oncogene homolog, neuroblastoma Ras viral oncogene homolog, and chromosomal instability[14]. Perhaps the differentiation may require a larger sample size or better definition of imaging features (as our definition of tumor homogeneity included tumor with up to 30% heterogeneous appearance). Alternatively, CT imaging features are a macroscopic eva
There are several limitations to our studies. First, the study is limited in size with 109 subjects (78 MSI-L, 31 MSI-H) and is a single center study which does limit generalizability. However, this is the only study that evaluates CT imaging features without radiomics. Also, we evaluated a broader range of CT imaging features than others which have limited the variables to the location of the tumor plus other clinical factors. Second, a total of 31 MSI-H subjects limits our ability to derive a nomogram to predict MSI pathology, as others have done. This will require additional subjects to achieve sufficient statistical power to predict a probability of MSI status. In addition, there are additional plausible variables that could be tested, one of which includes the number of abnormal regional lymph nodes, which may be a physiologically relevant characteristic. This will be reanalyzed and included in future studies.
Our data shows the potential of using strictly clinical and imaging features for identifying MSI status of CRC at initial diagnosis. However, larger, blinded, prospective trial will be necessary to assess the true clinical validity of imaging approach to determine MSI status. Moreover, artificial intelligence model may improve adoption of detailed image analysis by providing an automated approach to determine MSI status.
The authors would like to thank Bryce Dursteler, MD, for his assistance in obtaining the histological images in Figure 1.
| 1. | Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A. Colorectal cancer statistics, 2023. CA Cancer J Clin. 2023;73:233-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2171] [Cited by in RCA: 1972] [Article Influence: 657.3] [Reference Citation Analysis (12)] |
| 2. | André T, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, Morse MA, Van Cutsem E, McDermott R, Hill A, Sawyer MB, Hendlisz A, Neyns B, Abdullaev S, Memaj A, Lei M, Dixon M, Kopetz S, Overman MJ. Nivolumab plus low-dose ipilimumab in previously treated patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: 4-year follow-up from CheckMate 142. Ann Oncol. 2022;33:1052-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 192] [Article Influence: 48.0] [Reference Citation Analysis (5)] |
| 3. | André T, Shiu KK, Kim TW, Jensen BV, Jensen LH, Punt C, Smith D, Garcia-Carbonero R, Benavides M, Gibbs P, de la Fouchardiere C, Rivera F, Elez E, Bendell J, Le DT, Yoshino T, Van Cutsem E, Yang P, Farooqui MZH, Marinello P, Diaz LA Jr; KEYNOTE-177 Investigators. Pembrolizumab in Microsatellite-Instability-High Advanced Colorectal Cancer. N Engl J Med. 2020;383:2207-2218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2522] [Cited by in RCA: 2185] [Article Influence: 364.2] [Reference Citation Analysis (6)] |
| 4. | Battaglin F, Naseem M, Lenz HJ, Salem ME. Microsatellite instability in colorectal cancer: overview of its clinical significance and novel perspectives. Clin Adv Hematol Oncol. 2018;16:735-745. [PubMed] |
| 5. | Shaikh R, Bhattacharya S, Prajapati BG. Microsatellite instability: A potential game-changer in colorectal cancer diagnosis and treatment. Result Chem. 2024;7:101461. [DOI] [Full Text] |
| 6. | De' Angelis GL, Bottarelli L, Azzoni C, De' Angelis N, Leandro G, Di Mario F, Gaiani F, Negri F. Microsatellite instability in colorectal cancer. Acta Biomed. 2018;89:97-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 119] [Reference Citation Analysis (0)] |
| 7. | Greenson JK, Huang SC, Herron C, Moreno V, Bonner JD, Tomsho LP, Ben-Izhak O, Cohen HI, Trougouboff P, Bejhar J, Sova Y, Pinchev M, Rennert G, Gruber SB. Pathologic predictors of microsatellite instability in colorectal cancer. Am J Surg Pathol. 2009;33:126-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 207] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 8. | Yang G, Zheng RY, Jin ZS. Correlations between microsatellite instability and the biological behaviour of tumours. J Cancer Res Clin Oncol. 2019;145:2891-2899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 9. | Samowitz WS, Curtin K, Ma KN, Schaffer D, Coleman LW, Leppert M, Slattery ML. Microsatellite instability in sporadic colon cancer is associated with an improved prognosis at the population level. Cancer Epidemiol Biomarkers Prev. 2001;10:917-923. [PubMed] |
| 10. | Cao Y, Zhang G, Zhang J, Yang Y, Ren J, Yan X, Wang Z, Zhao Z, Huang X, Bao H, Zhou J. Predicting Microsatellite Instability Status in Colorectal Cancer Based on Triphasic Enhanced Computed Tomography Radiomics Signatures: A Multicenter Study. Front Oncol. 2021;11:687771. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 11. | Pei Q, Yi X, Chen C, Pang P, Fu Y, Lei G, Chen C, Tan F, Gong G, Li Q, Zai H, Chen BT. Pre-treatment CT-based radiomics nomogram for predicting microsatellite instability status in colorectal cancer. Eur Radiol. 2022;32:714-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 12. | Golia Pernicka JS, Gagniere J, Chakraborty J, Yamashita R, Nardo L, Creasy JM, Petkovska I, Do RRK, Bates DDB, Paroder V, Gonen M, Weiser MR, Simpson AL, Gollub MJ. Radiomics-based prediction of microsatellite instability in colorectal cancer at initial computed tomography evaluation. Abdom Radiol (NY). 2019;44:3755-3763. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 13. | Wang Q, Xu J, Wang A, Chen Y, Wang T, Chen D, Zhang J, Brismar TB. Systematic review of machine learning-based radiomics approach for predicting microsatellite instability status in colorectal cancer. Radiol Med. 2023;128:136-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 14. | Fujiyoshi K, Yamamoto G, Takenoya T, Takahashi A, Arai Y, Yamada M, Kakuta M, Yamaguchi K, Akagi Y, Nishimura Y, Sakamoto H, Akagi K. Metastatic Pattern of Stage IV Colorectal Cancer with High-Frequency Microsatellite Instability as a Prognostic Factor. Anticancer Res. 2017;37:239-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 79] [Article Influence: 8.8] [Reference Citation Analysis (3)] |
| 15. | Kang S, Na Y, Joung SY, Lee SI, Oh SC, Min BW. The significance of microsatellite instability in colorectal cancer after controlling for clinicopathological factors. Medicine (Baltimore). 2018;97:e0019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 98] [Article Influence: 12.3] [Reference Citation Analysis (3)] |
| 16. | Kim WK, Park M, Park M, Kim YJ, Shin N, Kim HK, You KT, Kim H. Identification and selective degradation of neopeptide-containing truncated mutant proteins in the tumors with high microsatellite instability. Clin Cancer Res. 2013;19:3369-3382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Ying M, Pan J, Lu G, Zhou S, Fu J, Wang Q, Wang L, Hu B, Wei Y, Shen J. Development and validation of a radiomics-based nomogram for the preoperative prediction of microsatellite instability in colorectal cancer. BMC Cancer. 2022;22:524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 18. | Fan S, Li X, Cui X, Zheng L, Ren X, Ma W, Ye Z. Computed Tomography-Based Radiomic Features Could Potentially Predict Microsatellite Instability Status in Stage II Colorectal Cancer: A Preliminary Study. Acad Radiol. 2019;26:1633-1640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (3)] |
| 19. | Chen X, He L, Li Q, Liu L, Li S, Zhang Y, Liu Z, Huang Y, Mao Y, Chen X. Non-invasive prediction of microsatellite instability in colorectal cancer by a genetic algorithm-enhanced artificial neural network-based CT radiomics signature. Eur Radiol. 2023;33:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 20. | Yuan H, Peng Y, Xu X, Tu S, Wei Y, Ma Y. A Tumoral and Peritumoral CT-Based Radiomics and Machine Learning Approach to Predict the Microsatellite Instability of Rectal Carcinoma. Cancer Manag Res. 2022;14:2409-2418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 21. | Jahanshahi A, Soleymani Y, Fazel Ghaziani M, Khezerloo D. Radiomics reproducibility challenge in computed tomography imaging as a nuisance to clinical generalization: a mini-review. Egypt J Radiol Nucl Med. 2023;54:83. [DOI] [Full Text] |
| 22. | Ge G, Zhang J. Feature selection methods and predictive models in CT lung cancer radiomics. J Appl Clin Med Phys. 2023;24:e13869. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 23. | Nguyen LH, Goel A, Chung DC. Pathways of Colorectal Carcinogenesis. Gastroenterology. 2020;158:291-302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 483] [Cited by in RCA: 412] [Article Influence: 68.7] [Reference Citation Analysis (6)] |
| 24. | Kim JC, Bodmer WF. Genomic landscape of colorectal carcinogenesis. J Cancer Res Clin Oncol. 2022;148:533-545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |