Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.114774
Revised: November 30, 2025
Accepted: February 2, 2026
Published online: March 24, 2026
Processing time: 176 Days and 18.8 Hours
Obesity is a significant global public health challenge and a key risk factor for several types of cancer. Breast cancer, the most common malignant tumor among women, is strongly associated with obesity. Currently, bariatric surgery is the most effective approach for long-term weight management and improving metabolic health. Although research indicates that bariatric surgery decreases the likelihood of developing certain cancers, its impact on breast cancer incidence is unclear.
To investigate the relationship between bariatric surgery and breast cancer risk in women.
We conducted a comprehensive search of multiple electronic databases for original studies comparing the incidence of breast cancer in women who underwent bariatric surgery to that in a control group. For the meta-analysis, we used a random-effects model and performed stratified analyses based on variables such as menopausal status, tumor stage, and hormone receptor status to examine effect modification. We evaluated the rigor of the study methods using the Risk of Bias in Non-randomized Studies of Interventions tool. We quantified heterogeneity using the I2 statistic and assessed the quality of the evidence using the Grading of Recommendations, Assessment, Development and Evaluation approach. Additionally, we created a funnel plot and performed a Begg’s test to evaluate publication bias.
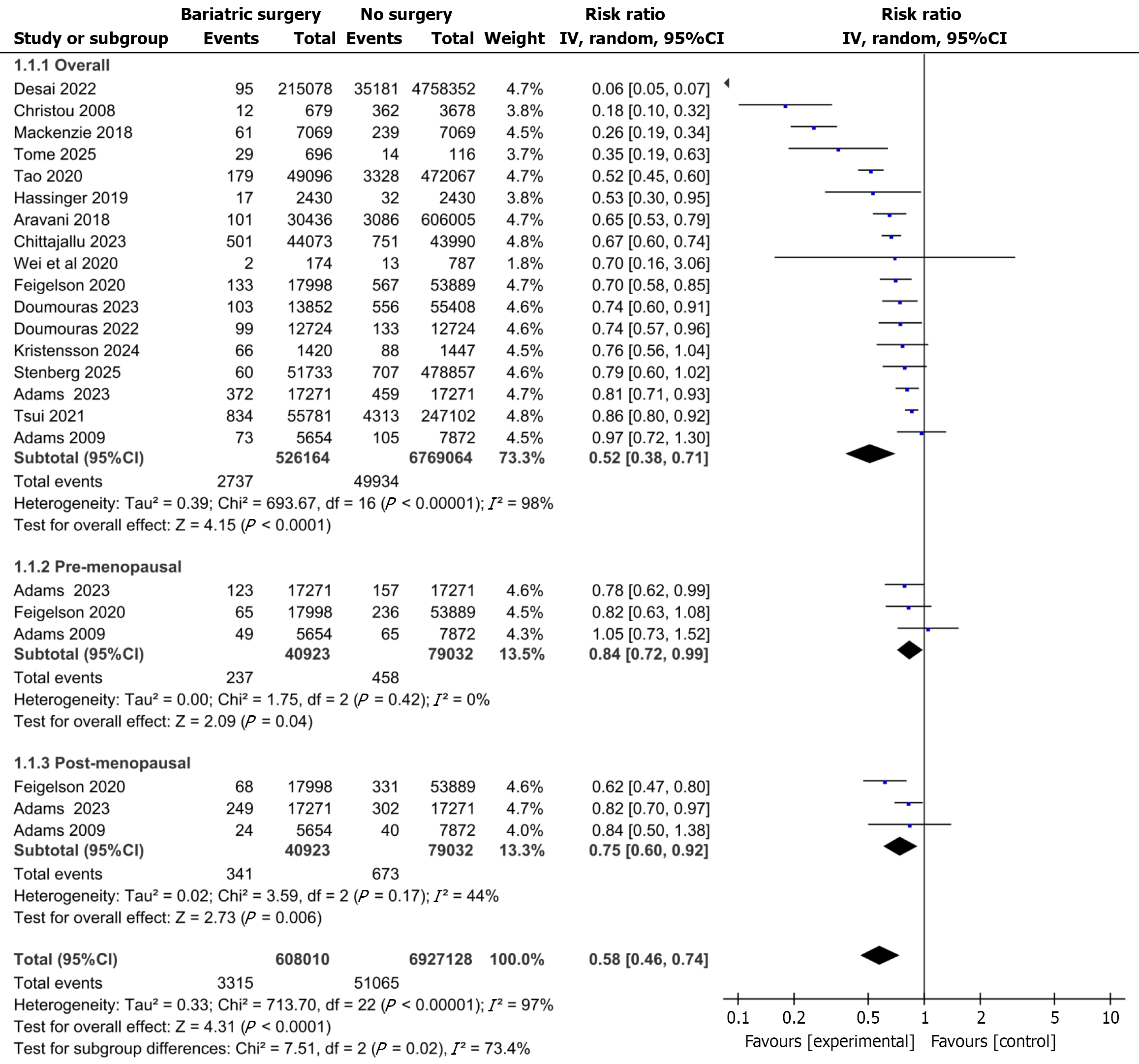
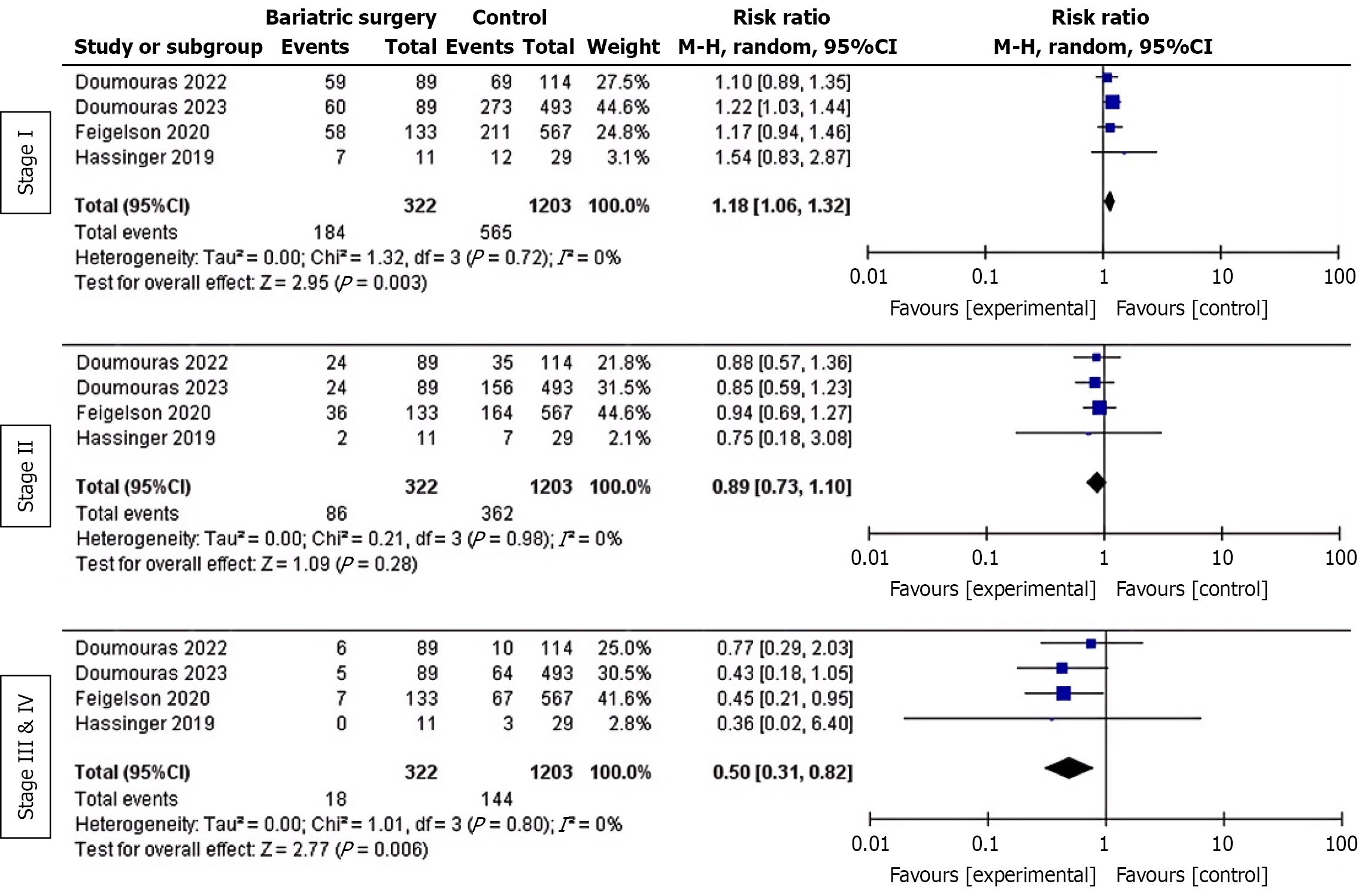
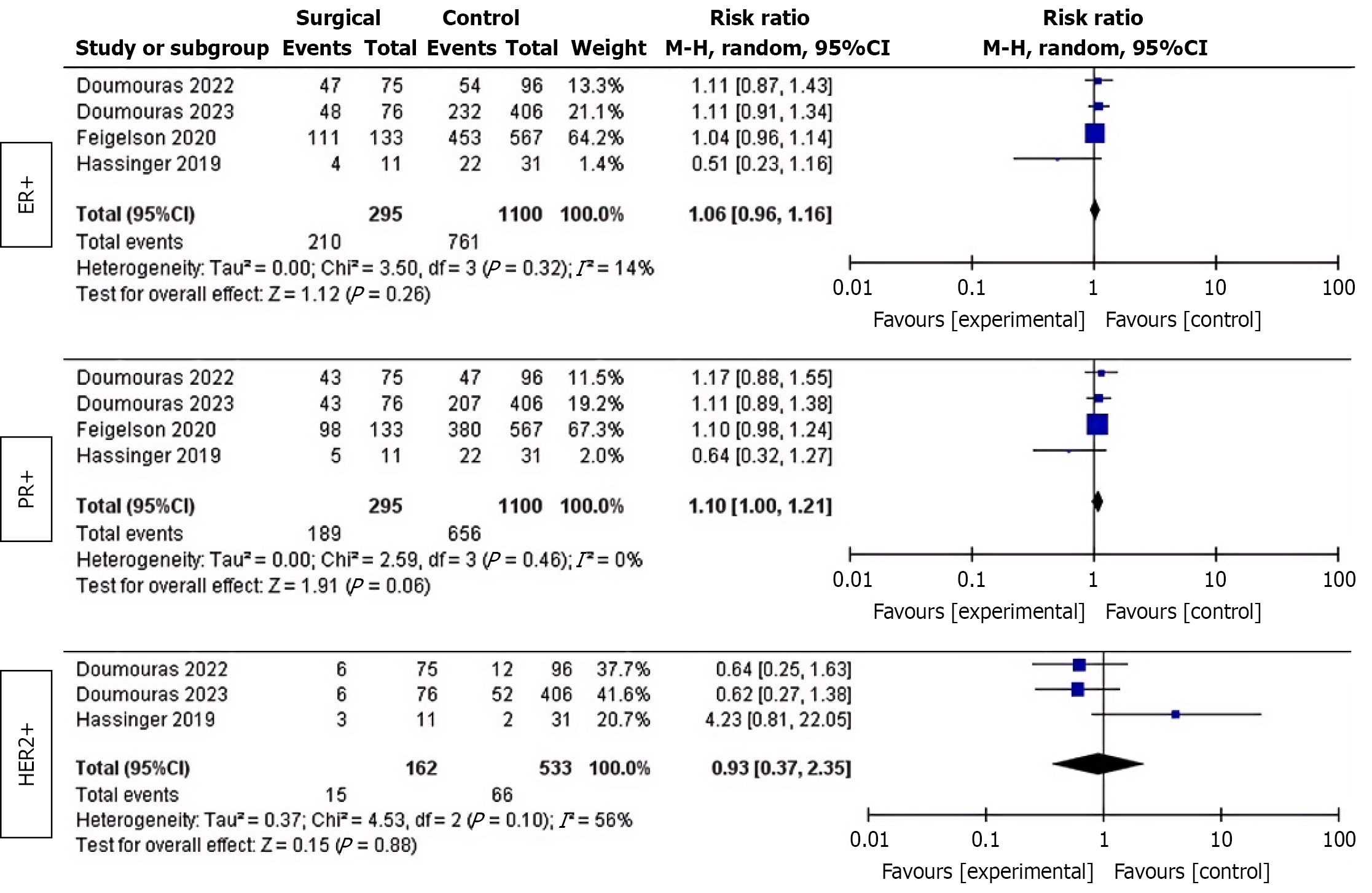
A total of 17 observational studies, including 7129194 female patients, were included in the analysis. Of those patients, 513601 underwent bariatric surgery, and 6615593 were in the control group. A meta-analysis revealed that bariatric surgery is significantly associated with a reduced overall risk of breast cancer [relative risk (RR) = 0.52; 95% confidence interval (CI): 0.38-0.71; P < 0.00001]. However, substantial heterogeneity was observed (I2 = 98%). Subgroup analysis showed a consistent reduction in risk among premenopausal (RR = 0.84; 95%CI: 0.72-0.99; P = 0.04) and postmenopausal (RR = 0.75; 95%CI: 0.60-0.92; P = 0.006) women. Analysis by disease stage revealed an 18% increase in the RR of stage I cancer in the surgery group (RR = 1.18; 95%CI: 1.06-1.32; P = 0.003). Conversely, the incidence of stage III-IV cancer decreased significantly (RR = 0.50; 95%CI: 0.31-0.82; P = 0.006). The incidence of stage II cancer did not change significantly (RR = 0.89; 95%CI: 0.73-1.10; P = 0.28). No significant association was observed for hormone receptor-positive (estrogen receptor-positive, progesterone receptor-positive, and human epidermal growth factor receptor 2+) subtypes. The overall risk of bias was moderate to high. Based on the Grading of Recommendations, Assessment, Development and Evaluation criteria and other considerations, the quality of the evidence was ultimately rated as “moderate”.
According to observational data, bariatric surgery appears to reduce the overall risk of breast cancer, especially among postmenopausal women. However, the observed association with an increased risk of stage I cancer should be interpreted with caution. Further prospective studies are needed to establish causality.
Core Tip: Obesity is a well-established risk factor for breast cancer. Although bariatric surgery is an effective intervention for long-term weight management and metabolic improvement, its impact on breast cancer incidence remains unclear. Our systematic review and meta-analysis of 17 observational studies indicates that bariatric surgery is associated with an overall reduced risk of breast cancer in obese women. This protective effect was observed in both premenopausal and postmenopausal women, though it was more pronounced in the latter group. However, the observational design precludes establishing causality. An observed increased risk of stage I cancer requires cautious interpretation. These findings offer a new per
- Citation: He YF, Wang HZ, Bian N, Hu XD, He QT, Liu JQ, Li HM, Lu SF, Wu N. Correlation between bariatric surgery and breast cancer risk in women: A systematic review and meta-analysis. World J Clin Oncol 2026; 17(3): 114774
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/114774.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.114774
Obesity has become a global public health crisis. Currently, approximately 15% of the population is obese[1], and projections indicate that this figure will rise to 20% by 2030[2]. Obesity is closely associated with cardiovascular disease and type 2 diabetes. It is also a confirmed risk factor for several cancers, including breast, colorectal, and endometrial cancers[3,4]. Breast cancer is the most common malignant tumor in women and the fourth leading cause of cancer-related deaths[5,6].
The relationship between obesity and breast cancer is complex and multifaceted. Notably, this relationship varies based on menopausal status[7]. Studies indicate that obesity is a clear risk factor for postmenopausal women[8,9]. However, its impact on premenopausal women is controversial[10]. Some studies suggest a protective effect[11], while others find no significant association[12,13]. These discrepancies highlight the complexity of obesity’s influence on breast cancer risk.
Bariatric surgery is currently the most effective treatment for severe obesity. It achieves long-term weight loss and significantly improves metabolic syndrome[14,15]. Recent studies suggest that bariatric surgery may reduce the incidence and mortality of certain cancers, especially hormone-related cancers, by altering metabolic and hormonal pathways[16,17]. However, studies examining the impact of bariatric surgery on breast cancer risk in women are limited, and existing conclusions are inconsistent. Some studies support its role in reducing breast cancer risk[18,19], while others suggest that it only affects specific subtypes[20,21]. Furthermore, other research has failed to observe a significant protective effect[22]. Due to the persistently rising global prevalence of obesity and the growing popularity of bariatric surgery, it is crucial to compile and evaluate existing evidence on this topic systematically. This systematic review and meta-analysis aims to quantitatively synthesize existing studies to clarify the strength of the association between bariatric surgery and breast cancer incidence in women. The review will also explore potential sources of heterogeneity and address important questions. Ultimately, the review will provide updated, evidence-based support for optimizing primary prevention strategies and clinical decision-making regarding breast cancer in obese women.
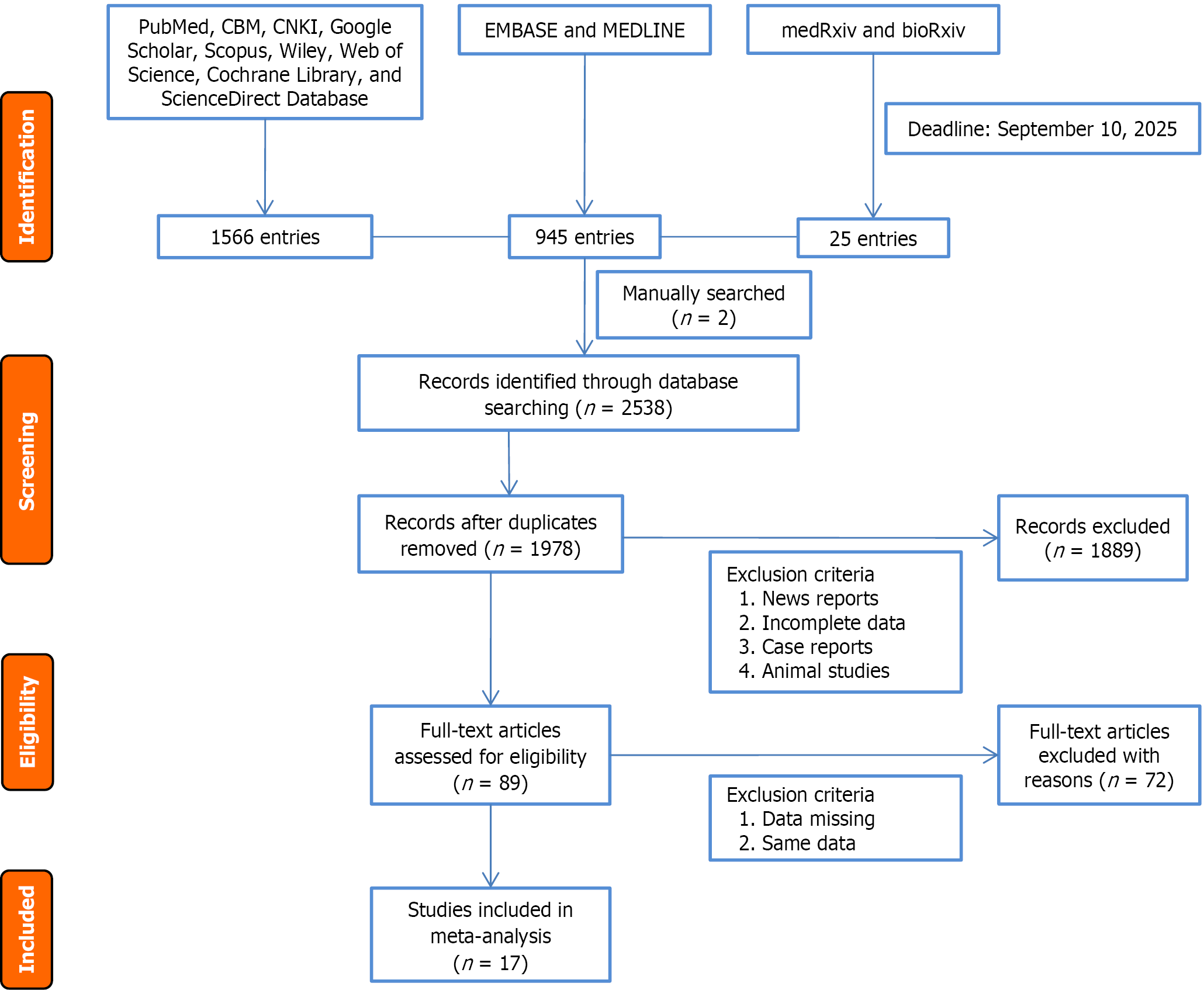
This study was conducted as a systematic review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The study protocol was registered on the PROSPERO platform with the registration number 1234543.
Does bariatric surgery affect the incidence of breast cancer in women?
Information librarians conducted a systematic search of professional databases, including PubMed, EMBASE, MEDLINE, CBM, CNKI, Google Scholar, Scopus, Wiley, Web of Science, Cochrane, and ScienceDirect. They also searched the preprint platforms medRxiv and bioRxiv for gray literature and manually traced the reference lists of the included studies to ensure comprehensive retrieval. The search employed Boolean operators and keyword combinations such as “bariatric surgery”, “Roux-en-Y gastric bypass (RYGB)”, “metabolic surgery”, and “breast cancer”, as well as their relevant synonyms. The final search was conducted on September 10, 2025. There were no restrictions based on publication language or date. All original studies published online that reported on the incidence of breast cancer in women after bariatric surgery were included. News reports, case reports, animal studies, and studies with incomplete data were excluded. Studies were included if their abstracts provided sufficient data for extraction and analysis.
Two investigators conducted the research screening independently and resolved any discrepancies through negotiation or third-party arbitration. The inclusion and exclusion criteria were predefined. The study population consisted of adult women undergoing bariatric surgery. The primary outcome was the incidence of breast cancer. Secondary outcomes included menopausal status, hormone receptor status, and tumor staging. In cases of duplicate publications[23-26], the study with the longest follow-up duration or the most complete data was included[24,26].
The initial search yielded 2538 relevant publications. Of those, 2536 were found through database searches, and two were added manually. After removing duplicates and conducting a preliminary screening based on titles and abstracts, 89 publications remained for full-text reading and evaluation. Ultimately, this systematic review and meta-analysis included 17 studies[18,19,21,22,24,26-37]. Figure 1 shows the PRISMA flow diagram illustrating the literature screening and inclusion process. Two researchers independently extracted data from the included studies. The extracted information included the first author, study design, type of surgery, and the patients’ baseline demographic and clinical characteristics. It also included the main conclusions of each study. All extracted information was cross-checked, systematically categorized, and summarized in Table 1.
| Ref. | Country of origin | Study design | Time period | Follow-up period | Sample size | Female (%) | Mean age (years), mean ± SD | BMI (kg/m2) | Breast cancer risk, n (%) | Type of bariatric surgery (%) | Main findings | HR (95%CI) |
| Surgery | Surgery | Surgery | Surgery | Surgery | Surgery | |||||||
| Control | Control | Control | Control | Control | Control | |||||||
| Wei et al[22], 2021 | Hong Kong SAR, China | Retrospective cohort study | 2006-2017 | 37 months | 345 | 174 (50.4) | 51.0 ± 11.0 | 37.5 ± 4.8 | 2 (0.011)1 | SG: 83.2, RYGB: 14.2, AGB: 2.6 | Bariatric surgery does not significantly impact the incidence of breast cancer | 0.394 (0.048-1.424) |
| 1599 | 787 (49.2) | 51.7 ± 12.8 | 36.9 ± 6.3 | 13 (0.017)1 | 0.521 (0.277-0.891) | |||||||
| Chittajallu et al[27], 2023 | United States | Retrospective cohort study | 2002-2022 | 10 years | 557892 | 44073 (79.0) | 43.1 ± 14.1 | > 35 | 501 (0.9) | NA | The incidence rate of new breast cancer cases was significantly lower among obese adults who had undergone bariatric surgery compared to those who had not | 0.753 (0.678-0.836) |
| 557892 | 43990 (78.9) | 44.6 ± 15.3 | 751 (13.5) | |||||||||
| Doumouras et al[28], 2022 | Canada | Retrospective population-based, matched cohort study | From January 2010 to December 2016 | 9 years | 12724 | 12724 (100.0) | 45.09 ± 0.98 | > 35 | 99 (0.79)3 | NA | Bariatric surgery is associated with a reduced incidence of breast cancer and lower tumor grade in obese women | 0.81 (0.69-0.95) |
| 12724 | 12724 (100.0) | 45.02 ± 1.08 | 133 (1.09)3 | |||||||||
| Feigelson et al[24], 2020 | United States | Retrospective cohort study | From January 1, 2005 to December 31, 2012 | 47.5 ± 23.2 months | 17998 | 17998 (100.0) | 44.6 ± 11.1 | 44.6 ± 6.6 | Premenopausal 65 | SG: 27.0, RYGB: 61.3, AGB: 5.6 | Bariatric surgery is associated with a reduced risk of breast cancer in both premenopausal and postmenopausal women | Premenopausal 072 (0.54-0.94) |
| Postmenopausal 68 | Postmenopausal 055 (0.42-0.72) | |||||||||||
| 40.8 ± 24.7 months | 53889 | 53889 (100.0) | 44.7 ± 11.0 | 44.1 ± 6.2 | Premenopausal 236 | Overall 063 (0.52-0.76) | ||||||
| postmenopausal 331 | ||||||||||||
| Christou et al[29], 2008 | Canada | Observational cohort study | From January 1986 to June 2002 | 5 years | 1035 | 679 (65.6) | 45.1 ± 11.6 | NA | 12 (1.16) | RYGB: 81.3, VGB: 18.7 | Patients in the bariatric surgery group had a lower incidence of breast cancer than those in the control group | 0.17 (0.098-0.311) |
| 5746 | 3678 (64.0) | 46.7 ± 13.1 | NA | 362 (6.31) | ||||||||
| Tome et al[21], 2025 | Israel | Retrospective cohort study | 2012-2020 | NA | 696 | 696 (100.0) | 60.7 ± 10.0 | 32.4 ± 6.5 | 29 (4.17) | NA | Bariatric surgery is associated with a reduced overall incidence of breast cancer | NA |
| 1164 | 116 (100.0) | 61.0 ± 10.1 | 38.6 ± 4.1 | 14 (12.10) | ||||||||
| Kristensson et al[26], 2024 | Sweden | Prospective, matched, intervention study | From September 1, 1987, to January 31, 2001 | 23.9 years (20.1-27.1) | 1420 | 1420 (100.0) | 47.2 ± 6.0 | 42.8 ± 4.3 | 66 (4.65) | AGB: 18.3, RYGB: 68.3, VGB: 13.4 | The bariatric surgery group showed a significantly reduced risk of breast cancer compared with the conventional treatment group | 0.68 (0.49-0.94) |
| 1447 | 1447 (100.0) | 48.8 ± 6.3 | 40.7 ± 4.6 | 88 (3.32) | ||||||||
| Adams et al[30], 2009 | United States | Retrospective cohort study | 1984-2002 | 12.3 ± 5.7 years | 6596 | 5654 (85.7) | 38.9 ± 10.3 | 44.9 ± 7.6 | Premenopausal 49 | RYGB: 100.0 | Gastric bypass surgery may reduce the overall incidence of cancer, but it does not affect breast cancer cases in premenopausal or postmenopausal women | Premenopausal 093 (0.63-1.37) |
| Postmenopausal 24 | Postmenopausal 096 (0.57-1.63) | |||||||||||
| 11.8 ± 5.6 years | 9442 | 7872 (83.4) | 39.1 ± 10.7 | 47.4 ± 6.5 | Premenopausal 65 | Overall 091 (0.67-1.24) | ||||||
| Postmenopausal 40 | ||||||||||||
| Tao et al[31], 2020 | Denmark, Finland, Iceland, Norway, and Sweden | Retrospective cohort study | From January 1, 1980 to December 31, 2012 | 33 years5 | 49096 | 36533 (74.5) | ≥ 18 | NA | 179 (0.36)6 | RYGB: 72.4, restrictive: 220, other: 5.6 | Women experience a reduced risk of breast cancer after bariatric surgery | 0.81 (0.69-0.95) |
| 472067 | 318,596 (67.5) | NA | 3328 (0.70)7 | |||||||||
| Desai et al[32], 2022 | United States | Retrospective cohort study | 1999-2014 | NA | 279171 | 215078 (77.04) | 43.9 ± 25.2 | NA | 95 (0.04) | NA | Patients with morbid obesity who undergo bariatric surgery have a significantly lower risk of developing breast cancer | 0.07 (0.06-0.09) |
| NA | 7393337 | 4758352 (64.36) | 54.9 ± 36.4 | NA | 35181 (0.74) | |||||||
| Adams et al[18], 2023 | United States | Retrospective cohort study | 1982-2019 | 38 years5 | 21837 | 17271 (100.0) | 42.2 ± 11.7 | 46.0 ± 8.3 | Premenopausal 123 | AGB: 11.8, RYGB: 71.1, duodenal switch: 3.9, SG: 13.1 | Women who underwent bariatric surgery were significantly less likely to develop breast cancer than those who did not, including both pre- and postmenopausal cases | Premenopausal 072 (0.54-0.95) |
| Postmenopausal 249 | Postmenopausal 0.79 (0.64-0.97) | |||||||||||
| 21837 | 17271 (100.0) | 42.3 ± 11.9 | 46.2 ± 6.8 | Premenopausal 157 | NA | |||||||
| Postmenopausal 302 | ||||||||||||
| Mackenzie et al[33], 2018 | United Kingdom | Retrospective population-based, matched cohort study | 1997-2012 | 55 (30-94) months7 | 8794 | 7069 (80.4) | 42.0 (35.0-50.0)7 | NA | 61 (0.86) | RYGB: 56.6, VGB: 33.6, SG: 9.8 | Patients who underwent bariatric surgery had a reduced risk of developing breast cancer compared to those who did not undergo the surgery | 0.25 (0.19-0.33) |
| 8794 | 7069 (80.4) | 42.0 (35.0-50.0)7 | NA | 239 (3.38) | ||||||||
| Aravani et al[34], 2018 | United Kingdom | Retrospective population-based, matched cohort study | 1997-2013 | 3.0 (range 1-16) years | 39474 | 30436 (76.6) | 44.8 | NA | 101 (0.33) | Restrictive: 520, other: 48.0 | Bariatric surgery and subsequent weight loss are associated with a reduced risk of developing breast cancer | SIR, 0.76 (0.62-0.92) |
| 2.5 (range 1-16) years | 962860 | 606005 (62.9) | 53.1 | NA | 3086 (0.51) | SIR 1.08 (1.04-1.11) | ||||||
| Hassinger et al[35], 2019 | United States | Retrospective propensity-matched cohort study | 1985-2015 | 6.0 (2.6-10.8)7 | 24302 | 2430b (100.0) | 42.0 (35.0-51.0)7 | 48.0 (43.0-53.0)7 | 17 (0.70) | GB: 11.9, RYGB: 79.4, SG: 7.5, other 1.2 | The rate of breast cancer of any type among female patients who underwent bariatric surgery was lower than that observed among propensity-matched controls | NA |
| 5.0 (3.6-10.3)7 | 24302 | 2430b (100.0) | 42.0 (31.0-53.0)7 | 47.1 (40.6-55.5)7 | 32 (1.31) | |||||||
| Doumouras et al[36], 2023 | Canada | Retrospective matched cohort study | From January 1, 2010 to December 31, 2016 | 5 years | 13852 | 13852 (100.0) | 45.1 ± 10.8 | NA | 103 (0.74) | NA | Bariatric surgery is associated with a reduced risk of breast cancer in obese women, bringing their risk level comparable to that of women with a BMI below 25 | 1.40 (1.18-1.57)3 |
| 5 years | 55408 | 55408 (100.0) | 45.1 ± 10.9 | NA | 556 (1.00) | |||||||
| Stenberg et al[19], 2025 | Sweden | Retrospective matched cohort study | From January 1, 2007 to December 31, 2020 | 7.7 ± 3.39 years | 68424 | 51733 (75.7) | 40.5 ± 11.13 | 41.8 ± 5.62 | 60 (0.12) | RYGB: 80.90, SG: 19.10 | The overall cancer incidence risk after bariatric surgery has approached that of the general population, and the risk of breast cancer has decreased | IRR 0.78 (0.71-0.86) |
| 7.8 ± 3.39 years | 640944 | 478857 (74.7) | 40.1 ± 11.07 | NA | 707 (0.15) | |||||||
| Tsui et al[37], 2021 | United States | Retrospective matched cohort study | 2006-2012 | 10 years | 55781 | 55781 (100.0) | NA | NA | 834 (1.50) | GB: 26.79, RYGB: 43.98, SG: 11.33, other 18.06 | Patients who underwent bariatric surgery had a lower incidence of breast cancer compared with obese patients who did not undergo the procedure | NA |
| 247102 | 247102 (100.0) | NA | NA | 4313 (1.75) |
Two independent reviewers assessed the risk of bias in each observational study using the Risk of Bias in Non-Randomized Studies of Interventions tool. Any disagreements were resolved through discussion. They evaluated the quality of the evidence using the Grading of Recommendations, Assessment, Development and Evaluation approach. For observational studies, the initial quality was set as “low” and adjusted based on various factors. Additionally, we constructed funnel plots and performed a Begg’s test to assess publication bias.
Data analysis was performed using Microsoft Excel and RevMan (version 5.3) software. For the meta-analysis of dichotomous variables, we used the DerSimonian-Laird random-effects model to calculate the relative risk (RR) and its 95% confidence interval (CI). A minimum of three studies was required for pooling. Statistical significance was set at P < 0.05. Heterogeneity among the studies was quantified using the I2 statistic, where 0% indicates no observed heterogeneity and higher values represent greater heterogeneity. An I2 value greater than 50% was considered indicative of substantial heterogeneity. Pre-specified subgroup analyses were conducted based on menopausal status, tumor stage, and hormone receptor status.
A total of 17 observational studies were included. Sixteen of these were retrospective cohort studies, and one was a prospective, non-randomized, matched cohort study derived from the Swedish obese subjects study[26]. These studies were conducted in North America, Europe, and parts of Asia, including the United States, the United Kingdom, Canada, Sweden, other Nordic countries, Israel, and Hong Kong, China. The total sample size was 10580993, 67.4% of whom were women. Of the female patients, 513601 underwent bariatric surgery, and 6615593 were in the control group. The largest study used the United States National Inpatient Sample database, enrolling 7672508 morbidly obese patients[32]. The earliest study inception date was 1980[31], and the longest follow-up period was 38 years[18].
Thirteen studies reported on specific surgical procedures, primarily RYGB, sleeve gastrectomy, and gastric banding, including vertical banded gastroplasty and adjustable gastric banding. Other or unclassified procedures were also included. Three of these studies conducted additional stratified analyses based on surgical type[19,33,37]. Three studies conducted subgroup analyses based on menopausal status, but their conclusions were inconsistent. Two of the studies suggested that bariatric surgery could lower the risk of breast cancer in both premenopausal and postmenopausal women[18,24]. However, the third study found no significant effect[30].
A total of four studies examined staging information at the time of breast cancer diagnosis in 47004 patients who underwent bariatric surgery and 171455 control subjects (Table 2). Two studies used the 0-IV staging system[24,35] and two used the I-IV system[28,36]. All four studies showed no statistically significant difference in breast cancer stage distribution between the surgical and control groups. These four studies provided hormone receptor status data but reached different conclusions. Two of the studies suggested that the incidence of hormone receptor-positive breast cancer was significantly lower in the bariatric surgery group than in the nonsurgical group[28,35]. The other two studies observed no differences between the groups[24,36]. Additionally, one study analyzed patients who had undergone breast cancer surgery[21], and another conducted a stratified analysis based on baseline body mass index (BMI)[36]. Of the 17 included studies, 15 concluded that bariatric surgery reduces the risk of developing breast cancer, while two found no significant association[22,30].
| Ref. | Arm | Incident breast cancer | Stage at diagnosis | ER+ breast cancer | PR+ breast cancer | HER2+ breast cancer |
| Doumouras et al[28], 2022 | Surgery | 99 (0.79)1 | Stage 1: 59 (66.3) | ER+: 47 (64.8) | PR+: 43 (59.1) | HER2+: 6 (8.0) |
| Stage 2: 24 (27) | ER-: 6 (9.1) | PR-: 10 (14.8) | HER2-: 46 (63.6) | |||
| Stage 3 and 4: 6 (6.7) | Unknown: 22 (26.1) | Unknown: 22 (26.1) | Unknown: 23 (28.4) | |||
| Control | 133 (1.09)1 | Stage 1: 69 (60.5) | ER+: 54 (61.8) | PR+: 47 (54.5) | HER2+: 12 (12.2) | |
| Stage 2: 35 (30.7) | ER-: 7 (8.1) | PR-: 14 (15.4) | HER2-: 44 (51.2) | |||
| Stage 3 and 4: 10 (8.8) | Unknown: 35 (30.1) | Unknown: 35 (30.1) | Unknown: 40 (36.6) | |||
| P value | 0.423 | 0.045 | 0.041 | 0.012 | ||
| Feigelson et al[24], 2020 | Surgery | 133 (0.7) | Stage 0: 32 (24.1) | ER+: 111 (83.5) | PR+: 98 (73.7) | |
| Stage 1: 58 (43.6) | ER-: 19 (14.3) | PR-: 19 (14.3) | ||||
| Stage 2: 36 (27.1) | Unknown: 3 (2.2) | Unknown: 16 (12.0) | ||||
| Stage 3 and 4: 7 (5.3) | ||||||
| Control | 567 (1.1) | Stage 0: 120 (21.2) | ER+: 453 (79.9) | PR+: 380 (67.0) | ||
| Stage 1: 211 (37.2) | ER-: 91 (16.0) | PR-: 89 (15.7) | ||||
| Stage 2: 164 (28.9) | Unknown: 23 (4.1) | Unknown: 98 (17.3) | ||||
| Stage 3 and 4: 67 (11.8) | ||||||
| P value | 0.20 | 0.60 | 0.60 | |||
| Hassinger et al[35], 2019 | Surgery | 17 (0.7) | Stage 0: 2/11 (18.2) | ER+: 4/11 (36.4) | PR+: 5/11 (45.5) | HER2+: 3/11 (27.3) |
| Stage 1: 7/11 (63.6) | ER-: 7/11 (63.6) | PR-: 6/11 (54.5) | HER2-: 8/11 (72.7) | |||
| Stage 2: 2/11 (18.2) | ||||||
| Stage 3 and 4: 0 (0) | ||||||
| Control | 32 (1.3) | Stage 0: 7/29 (24.1) | ER+: 22/31 (71.0) | PR+: 22/31 (71.0) | HER2+: 2/31 (6.5) | |
| Stage 1: 12/29 (41.4) | ER-: 9/31 (29.0) | PR-: 9/31 (29.0) | HER2-: 29/31 (93.5) | |||
| Stage 2: 7/29 (24.1) | ||||||
| Stage 3 and 4: 3/29 (10.4) | ||||||
| P value | 0.53 | 0.04 | 0.13 | 0.07 | ||
| Doumouras et al[36], 2023 | Surgery | 103 (0.74) | Stage 1: 273 (55.4) | ER+: 48/76 (63.2) | PR+: 43/76 (56.6) | HER2+: 6/76 (7.9) |
| Stage 2: 156 (31.6) | ER-: 5/76 (6.5) | PR-: 10/76 (13.2) | HER2-: 46/76 (60.5) | |||
| Stage 3 and 4: 64 (13.0) | Unknown: 23/76 (30.3) | Unknown: 23 (30.3) | Unknown: 24/76 (31.6) | |||
| Control | 556 (1.00) | Stage 1: 60 (67.4) | ER+: 232/406 (57.1) | PR+: 207/406 (51.0) | HER2+: 52/406 (12.8) | |
| Stage 2: 24 (27.0) | ER-: 48/406 (11.8) | PR-: 73/406 (18.0) | HER2-: 221/406 (54.4) | |||
| Stage 3 and 4: 5 (5.6) | Unknown: 126/406 (31.0) | Unknown: 126/406 (31.0) | Unknown: 133/406 (32.8) | |||
| P value | 0.29 | 0.94 | 0.9 | 0.65 |
Impact on overall breast cancer incidence and menopausal status: A meta-analysis using a random-effects model revealed a significant correlation between undergoing bariatric surgery and a reduced risk of developing breast cancer (RR = 0.52; 95%CI: 0.38-0.71; P < 0.00001). However, substantial heterogeneity was observed among the studies (I2 = 98%). The subgroup analysis was based on data from three studies examining the incidence of premenopausal and postmenopausal breast cancer[18,24,30]. These studies included a total of 119955 patients who underwent bariatric surgery and 79032 control subjects. Significant reductions in breast cancer risk were observed among premenopausal (RR: 0.84; 95%CI: 0.72-0.99; P = 0.04) and postmenopausal (RR: 0.75; 95%CI: 0.60-0.92; P = 0.006) women. The reduction was greater for postmenopausal women (25% vs 16%), and the heterogeneity was low (I2 = 0% and I2 = 44%, respectively). Figure 2 shows the overall impact of bariatric surgery on breast cancer incidence, with subgroup analyses conducted according to menopausal status.
Impact on the risk of breast cancer at different stages: A stratified analysis of data from four studies (comprising 47004 bariatric surgery patients and 171455 control participants) concerning breast cancer staging at diagnosis revealed an increased risk of stage I breast cancer (RR: 1.18; 95%CI: 1.06-1.32; P = 0.003) and a decreased risk of stages III-IV (RR: 0.50; 95%CI: 0.31-0.82; P = 0.006) following bariatric surgery. However, there was no significant effect on the risk of stage II breast cancer (RR: 0.89; 95%CI: 0.73-1.10; P = 0.28). No heterogeneity was detected in any staging analysis (I2 = 0%). Due to the limited availability of stage 0 breast cancer data (only two studies provided such information), a combined analysis for this stage could not be performed. Figure 3 illustrates the association between bariatric surgery and the risk of developing breast cancer at different stages.
Impact on hormone receptor status: Four studies examined the impact of bariatric surgery on estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 expression in individuals newly diagnosed with breast cancer. A stratified analysis of patients with available receptor status data (295 in the surgical group and 1100 in the control group) revealed that bariatric surgery did not significantly impact the likelihood of developing estrogen receptor-positive, progesterone receptor-positive, or human epidermal growth factor receptor 2-positive (HER2+) breast cancer. The respective RR were 1.06 (95%CI: 0.96-1.16, P = 0.26), 1.10 (95%CI: 1.00-1.21, P = 0.06), and 0.93 (95%CI: 0.37-2.35, P = 0.88), with respective heterogeneity indices of I2 = 14%, 0%, and 56%, respectively. Figure 4 shows the effects of bariatric surgery on hormone receptor status using a random-effects meta-analysis and a forest plot. Due to the small sample size and high level of heterogeneity among the studies, a subgroup analysis of the type of bariatric surgery was not conducted (only three studies addressed this topic[19,33,37]).
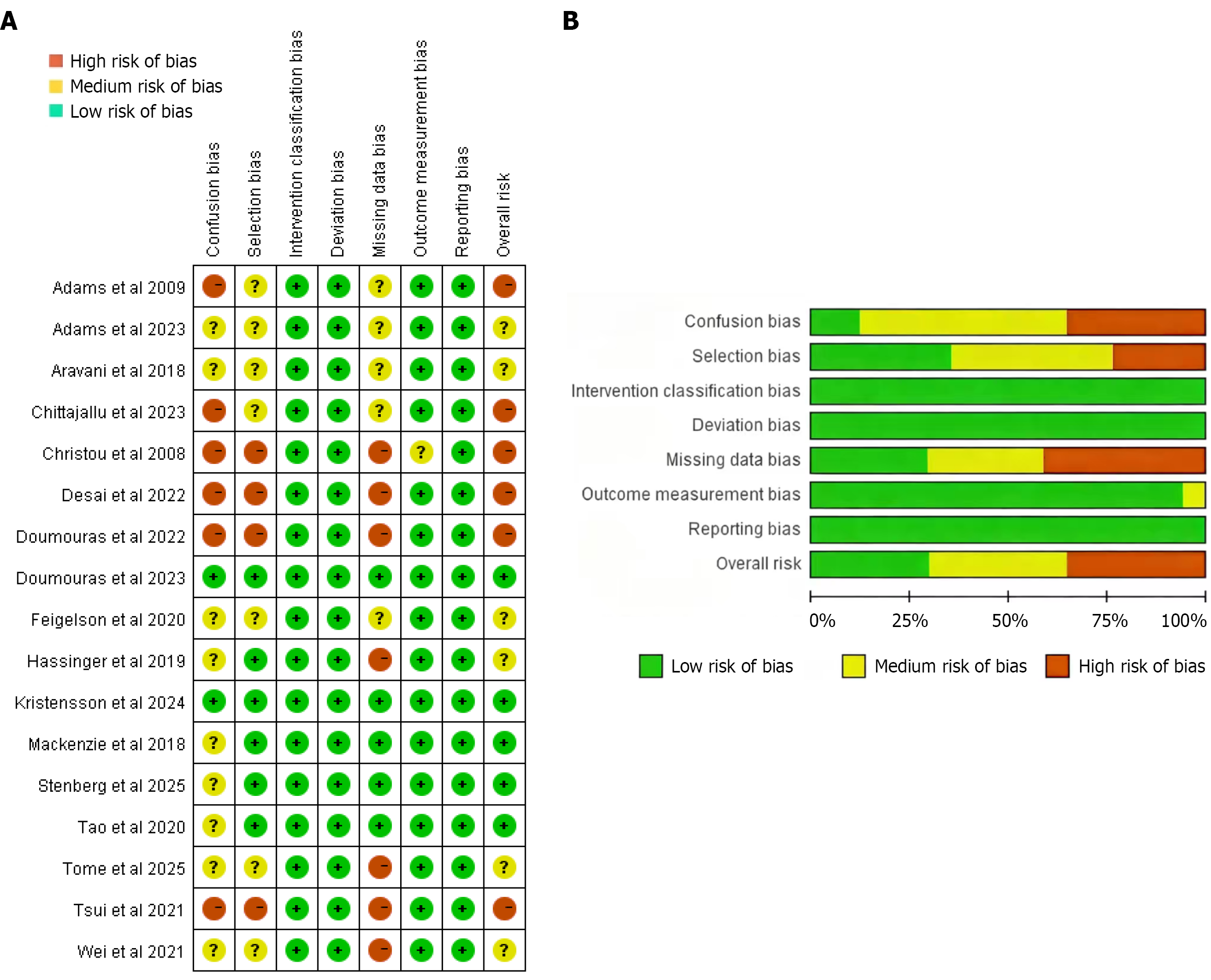
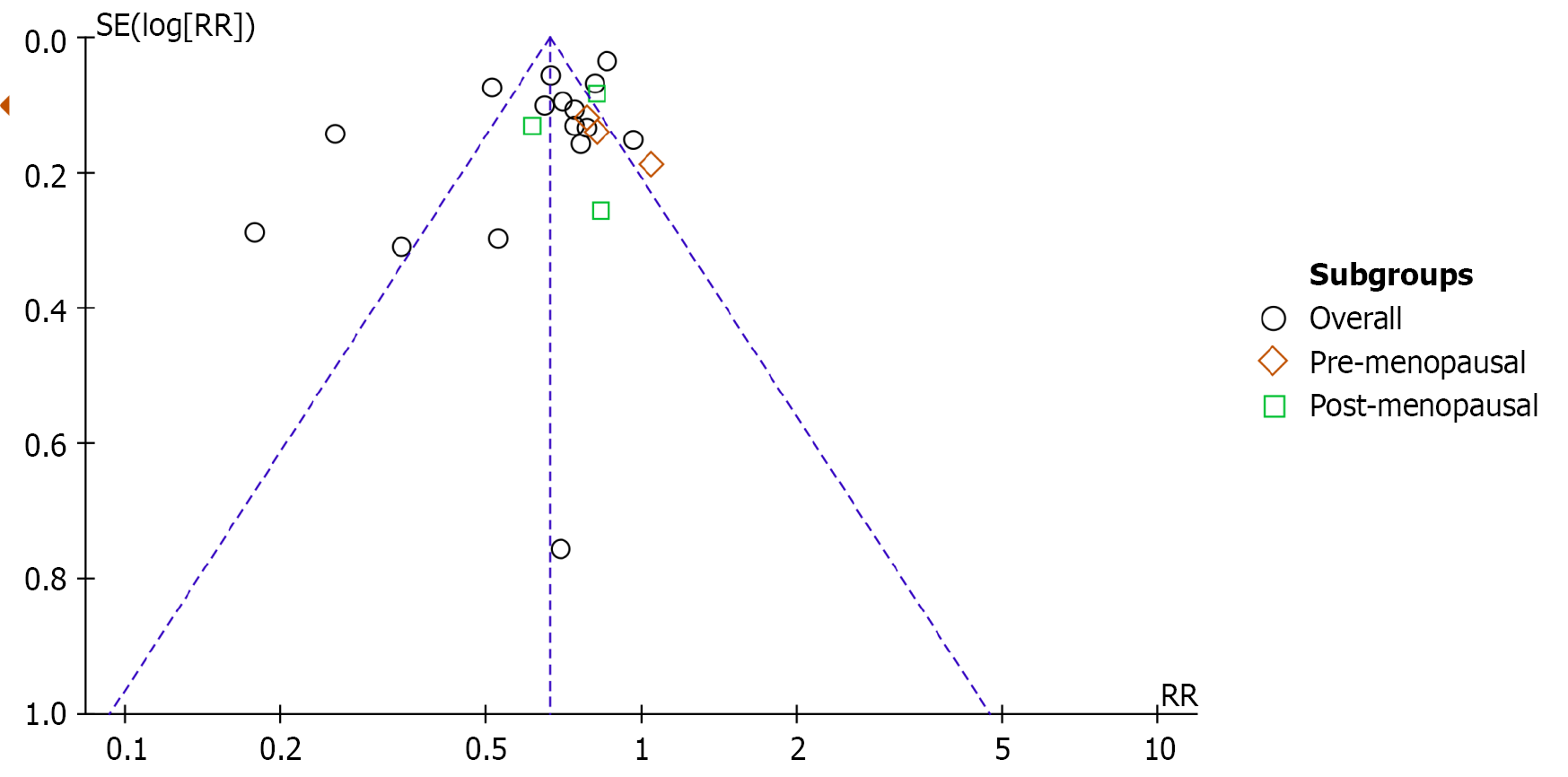
The included studies exhibited a moderate to high risk of bias in the confounding and selection domains due to their retrospective designs. These designs prevented researchers from fully controlling for confounding factors. Figure 5A provides a detailed Risk of Bias in Non-Randomized Studies of Interventions risk-of-bias assessment for each study, and Figure 5B illustrates these assessments. The funnel plot (Figure 6) revealed asymmetry (Begg’s test, P < 0.05), which suggests the presence of publication bias. Since all of the included studies were observational, the initial quality was rated as “low”. However, due to consistent effect sizes and large sample sizes, the overall quality of the evidence was upgraded to “moderate”.
This systematic review and meta-analysis of 17 observational studies examined the relationship between undergoing bariatric surgery and the likelihood of developing breast cancer. The analysis indicates that bariatric surgery is associated with a significant reduction in overall breast cancer risk, with a potentially stronger protective effect observed in postmenopausal women. However, the studies also revealed a paradoxical finding: Despite the overall reduction in risk, the incidence of stage I breast cancer increased in the surgical group.
Previous studies have confirmed that bariatric surgery significantly reduces the overall incidence of obesity-related cancers[17,36,38]. For example, Doumouras et al[36] demonstrated that this surgery lowers the risk of breast cancer in obese women to levels comparable with those of normal-weight individuals (BMI < 25). Although some studies showed heterogeneity, the majority of the studies included in this review, as well as the review’s overall findings, support the protective effect of bariatric surgery against breast cancer. However, there is some divergence in conclusions across different studies. For example, Wei et al’s study[22], based on data from Hong Kong, China, revealed that surgery did not significantly affect the incidence of breast cancer. The researchers attributed this discrepancy to racial or regional factors[22]. Similarly, Adams et al[30] observed no significant effect of gastric bypass surgery on breast cancer incidence, attributing this to factors such as limited sample size, healthy user bias, and short follow-up duration. These discrepancies suggest that the association between bariatric surgery and breast cancer risk may be modulated by multiple factors, including population characteristics, surgical techniques, follow-up duration, and methodological differences.
It should be noted that the current evidence is limited by the observational design of the included studies, substantial heterogeneity between the studies, and a moderate to high risk of bias. Therefore, more rigorous research designs and stronger bias control are needed to validate this association in future studies.
Bariatric surgery primarily reduces the risk of breast cancer by interacting with multiple physiological pathways. The procedure focuses on weight reduction, metabolic improvement, and hormone regulation. It decreases the volume of adipose tissue, the primary site of estrogen synthesis. This lowers estrogen levels and improves insulin resistance[39,40]. Hyperinsulinemia is considered one of the key mechanisms of this improvement[26]. Additionally, the procedure may influence gut microbiota composition by altering gastrointestinal architecture, thereby modulating systemic inflammation and estrogen metabolism[41]. These changes create a metabolic and immune microenvironment that is unfavorable for tumorigenesis, thereby exerting a preventive effect against hormone-sensitive breast cancers. However, the specific molecular mechanisms and variations among different surgical approaches require further investigation.
Although evidence suggests that bariatric surgery may reduce the risk of breast cancer in obese women, the clinical applicability of this treatment and which populations would benefit from it are still controversial topics. This presents multiple challenges. Addressing these issues is essential to defining the role of bariatric surgery in primary cancer prevention and developing personalized intervention strategies.
Which group benefits more from bariatric surgery: Women who undergo the procedure before or after menopause? Menopausal status is a key factor in the preventive effects of bariatric surgery and exhibits physiological complexity and apparent paradoxes. This meta-analysis shows that bariatric surgery significantly reduces the risk of breast cancer in both premenopausal (RR: 0.84; 95%CI: 0.72-0.99; P = 0.04) and postmenopausal (RR: 0.75; 95%CI: 0.60-0.92; P = 0.006) women. A greater reduction was observed in the postmenopausal group (25% vs 16%). These results suggest that the benefits may be more pronounced in postmenopausal women. Mechanistically, obesity in premenopausal women is often accompanied by estrogen feedback inhibition, which provides a certain “protective effect”. In contrast, postmenopausal women rely on adipose tissue for estrogen synthesis. Bariatric surgery reduces fat mass, lowers aromatase activity, and improves insulin resistance, thereby exerting a stronger preventive effect on hormone receptor-positive breast cancer[42]. Although data suggest greater benefits from surgery after menopause, surgery before menopause may provide long-term advantages by promoting early metabolic improvements. Future research should explore how surgical intervention interacts with endocrine and tumor characteristics.
Is there an association between bariatric surgery and the biological characteristics of breast cancer? This study found that bariatric surgery has different effects on the various stages of breast cancer. Specifically, it has been associated with an increased risk of stage I breast cancer (RR: 1.18; 95%CI: 1.06-1.32; P = 0.003) and a decreased risk of stages III-IV (RR: 0.50; 95%CI: 0.31-0.82; P = 0.006). However, it has had no significant impact on stage II breast cancer risk (RR: 0.89; 95%CI: 0.73-1.10; P = 0.28). Bariatric surgery is associated with an increased risk of stage I breast cancer (RR: 1.18). Previous studies have confirmed this association between bariatric surgery and stage I breast cancer[28,43]. However, this finding may reflect detection bias resulting from more intensive postoperative monitoring rather than the “carcinogenic effect” of bariatric surgery[44]. Specifically, earlier-stage cases were more frequently detected in the surgical group due to regular checkups. Meanwhile, the number of advanced-stage cases decreased thanks to early intervention. Regarding molecular subtypes, Heshmati et al[20] matched 42 patients with surgically confirmed breast cancer to 84 nonsurgical patients and found significantly lower HER2+ rates in the surgical group. These results suggest that bariatric surgery may modulate tumor molecular phenotypes. Of the four studies included in this review that addressed hormone receptor status, the results were inconsistent. Hassinger et al[35] found a higher proportion of estrogen receptor-positive tumors in the nonsurgical group. These results suggest that surgery may offer specific protection for the estrogen receptor-positive subtype. Doumouras et al’s findings[36] were similar to Heshmati et al[20] but different from Hassinger et al[35] (P < 0.05). This meta-analysis found no significant association between bariatric surgery and hormone receptor status (estrogen receptor-positive, progesterone receptor-positive, or HER2+). However, given the small sample sizes and high heterogeneity of the included studies, the current conclusions should be interpreted with caution. Future research with larger sample sizes is needed to clarify the impact of surgery on tumor biological characteristics.
Cancer patients and bariatric surgery: Traditional contraindication or new therapeutic pathway? How should clinicians make decisions for obese patients with a history of or current cancer? Studies indicate that patients with a history of malignancy achieve similar weight loss results as those with a new cancer diagnosis after bariatric surgery[45]. The procedure does not negatively impact tumor treatment and may facilitate early diagnosis through routine screening[46]. Parmar et al’s research[47] suggests that preoperative weight loss is an effective treatment strategy for low-grade, less invasive tumors. A retrospective study by Tome et al[21] showed that the incidence of postoperative breast cancer was significantly lower in the bariatric surgery group (4%, or 29 out of 696 patients) than in the obese control group (12%, or 14 out of 116 patients). These findings suggest that bariatric surgery is not an absolute contraindication for obese patients with a history of cancer and may offer additional benefits. However, enhanced perioperative screening and individualized assessment are essential.
Which type of surgery offers a greater risk-benefit ratio for breast cancer? Existing studies demonstrate inconsistencies in the selection of surgical procedures. Of the 17 studies included in the analysis, only three conducted stratified analyses based on surgical technique. Mackenzie et al[33] found no significant differences among different surgical types. However, Stenberg et al[19] and Tsui et al[37] observed that RYGB surgery was more effective than sleeve gastrectomy at reducing the risk of breast cancer. The advantages of RYGB may stem from its stronger metabolic improvement effects. RYGB typically results in more significant weight loss, and sustained weight reduction is associated with decreased circulating hormone levels, such as estrogen. On the other hand, RYGB achieves higher remission rates for type 2 diabetes and may offer further protection by enhancing insulin sensitivity. Hyperinsulinemic states are known to correlate positively with the incidence of postmenopausal breast cancer[26]. However, this study did not perform subgroup analyses due to substantial heterogeneity among the studies and incomplete information on key covariates such as age, BMI, and tumor grade/type. Future research requires higher-quality, prospective data to explore stratified analyses that incorporate surgical type, patient baseline characteristics, and tumor molecular subtypes.
This study is the largest and most comprehensive systematic review and meta-analysis examining the association between bariatric surgery and breast cancer risk. It provides a clear, representative perspective for obese women with various characteristics. As one of the few studies deeply exploring changes in breast cancer incidence following bariatric surgery, this study has several advantages. First, it is the first study to conduct pre-specified subgroup analyses across key dimensions such as menopausal status, hormone receptor expression, and breast cancer stage. The study revealed the protective effect of bariatric surgery on breast cancer risk, providing crucial theoretical foundations for guiding clinical practice in this field. Secondly, this study had a large sample size and a long follow-up period (with the longest lasting 38 years). Furthermore, some studies employed multidimensional matching and propensity score methods to statistically adjust for various confounding factors, including comorbidities, socioeconomic status, history of cancer screening, and mental health conditions. These methods significantly enhanced the accuracy of effect estimates and the strength of causal inferences.
However, this study has several limitations. First, it was not possible to conduct subgroup analyses based on surgical type, age, baseline BMI, and other factors due to high heterogeneity and incomplete reporting of covariates in the original studies. In fact, only seven studies reported baseline BMI, and only six mentioned intergroup differences. Second, all of the included studies employed observational designs. Despite the use of multivariate adjustments, residual confounding factors such as healthy user bias and monitoring bias could not be entirely ruled out. This could lead to an overestimation of the protective effect. Additionally, some studies had small sample sizes and short follow-up periods (most of which were less than five years), resulting in limited statistical power that may underestimate long-term risks. Other limitations include missing reports on key confounding variables (e.g., lifestyle and family history), heterogeneity arising from evolving surgical techniques, a lack of postoperative weight dynamics data, and insufficient tumor molecular subtyping information. These factors hindered the exploration of the dose-response relationship between weight loss and risk, as well as the effects of biological subtypes. Finally, the results of the statistical analysis indicate that the study may be subject to potential bias. Therefore, current conclusions should be interpreted with caution.
The findings of this study have significant clinical and public health value. Obesity is a well-established risk factor for breast cancer. Bariatric surgery is an effective means of achieving long-term weight control, and numerous studies have demonstrated its ability to lower breast cancer risk. Our research indicates that bariatric surgery can significantly reduce breast cancer incidence in obese women. Although bariatric surgery is not currently recommended for cancer prevention, our results suggest its significant potential as a primary prevention strategy for managing breast cancer risk in obese women. Future research should focus on the dose-response relationship between weight loss and risk reduction. Additionally, researchers should develop precision intervention strategies based on patients’ metabolic characteristics, tumor subtypes, and genetic backgrounds. These strategies would optimize individualized prevention and treatment decisions.
This systematic review and meta-analysis indicates that bariatric surgery may reduce the overall risk of breast cancer in obese women. However, as the studies are based on observational data, a causal relationship cannot be confirmed. Furthermore, the finding of an increased risk of stage I disease should be interpreted with caution. Further prospective studies are necessary to distinguish between different breast cancer subtypes and investigate the relationship between weight loss and changes in risk.
| 1. | World Health Organization. Obesity and overweight. [cited 3 August 2025]. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. |
| 2. | World Obesity. World Obesity Atlas 2025. [cited 3 August 2025]. Available from: https://www.worldobesity.org/resources/resource-library/world-obesity-atlas-2025. |
| 3. | Wang J, Yang DL, Chen ZZ, Gou BF. Associations of body mass index with cancer incidence among populations, genders, and menopausal status: A systematic review and meta-analysis. Cancer Epidemiol. 2016;42:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 102] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 4. | Paragomi P, Zhang Z, Abe SK, Islam MR, Rahman MS, Saito E, Shu XO, Dabo B, Pham YT, Chen Y, Gao YT, Koh WP, Sawada N, Malekzadeh R, Sakata R, Hozawa A, Kim J, Kanemura S, Nagata C, You SL, Ito H, Park SK, Yuan JM, Pan WH, Wen W, Wang R, Cai H, Tsugane S, Pourshams A, Sugawara Y, Wada K, Chen CJ, Oze I, Shin A, Ahsan H, Boffetta P, Chia KS, Matsuo K, Qiao YL, Rothman N, Zheng W, Inoue M, Kang D, Luu HN. Body Mass Index and Risk of Colorectal Cancer Incidence and Mortality in Asia. JAMA Netw Open. 2024;7:e2429494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 5. | Newman L. Oncologic anthropology: Global variations in breast cancer risk, biology, and outcome. J Surg Oncol. 2023;128:959-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 6. | Wagle NS, Nogueira L, Devasia TP, Mariotto AB, Yabroff KR, Islami F, Jemal A, Alteri R, Ganz PA, Siegel RL. Cancer treatment and survivorship statistics, 2025. CA Cancer J Clin. 2025;75:308-340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 125] [Article Influence: 125.0] [Reference Citation Analysis (0)] |
| 7. | Park JW, Han K, Shin DW, Yeo Y, Chang JW, Yoo JE, Jeong SM, Lee SK, Ryu JM, Park YM. Obesity and breast cancer risk for pre- and postmenopausal women among over 6 million Korean women. Breast Cancer Res Treat. 2021;185:495-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 8. | Yoon KH, Yoon Y, Jeong S, Kang J, Oh JH, Koh HW, Shin HC, Kim EK. Impact of obesity on breast cancer recurrence by menopausal status and subtype: a retrospective cohort study. Breast Cancer Res Treat. 2025;214:387-395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 9. | García-Estévez L, Cortés J, Pérez S, Calvo I, Gallegos I, Moreno-Bueno G. Obesity and Breast Cancer: A Paradoxical and Controversial Relationship Influenced by Menopausal Status. Front Oncol. 2021;11:705911. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 99] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 10. | Cheraghi Z, Poorolajal J, Hashem T, Esmailnasab N, Doosti Irani A. Effect of body mass index on breast cancer during premenopausal and postmenopausal periods: a meta-analysis. PLoS One. 2012;7:e51446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 183] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 11. | Smith SG, Sestak I, Morris MA, Harvie M, Howell A, Forbes J, Cuzick J. The impact of body mass index on breast cancer incidence among women at increased risk: an observational study from the International Breast Intervention Studies. Breast Cancer Res Treat. 2021;188:215-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 12. | Ayoub NM, Yaghan RJ, Abdo NM, Matalka II, Akhu-Zaheya LM, Al-Mohtaseb AH. Impact of Obesity on Clinicopathologic Characteristics and Disease Prognosis in Pre- and Postmenopausal Breast Cancer Patients: A Retrospective Institutional Study. J Obes. 2019;2019:3820759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 13. | Moore J, Pal T, Beeghly-Fadiel A, Fadden MK, Munro HM, Dujon SA, Reid S, Tezak A, Blasingame M, Ware J, Blot WJ, Shu XO, Zheng W, Sanderson M, Lipworth L. A pooled case-only analysis of obesity and breast cancer subtype among Black women in the southeastern United States. Cancer Causes Control. 2022;33:515-524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Carlsson LMS, Sjöholm K, Jacobson P, Andersson-Assarsson JC, Svensson PA, Taube M, Carlsson B, Peltonen M. Life Expectancy after Bariatric Surgery in the Swedish Obese Subjects Study. N Engl J Med. 2020;383:1535-1543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 369] [Article Influence: 61.5] [Reference Citation Analysis (3)] |
| 15. | Gloy VL, Briel M, Bhatt DL, Kashyap SR, Schauer PR, Mingrone G, Bucher HC, Nordmann AJ. Bariatric surgery versus non-surgical treatment for obesity: a systematic review and meta-analysis of randomised controlled trials. BMJ. 2013;347:f5934. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1087] [Cited by in RCA: 989] [Article Influence: 76.1] [Reference Citation Analysis (0)] |
| 16. | Sjöström L, Gummesson A, Sjöström CD, Narbro K, Peltonen M, Wedel H, Bengtsson C, Bouchard C, Carlsson B, Dahlgren S, Jacobson P, Karason K, Karlsson J, Larsson B, Lindroos AK, Lönroth H, Näslund I, Olbers T, Stenlöf K, Torgerson J, Carlsson LM; Swedish Obese Subjects Study. Effects of bariatric surgery on cancer incidence in obese patients in Sweden (Swedish Obese Subjects Study): a prospective, controlled intervention trial. Lancet Oncol. 2009;10:653-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 633] [Cited by in RCA: 528] [Article Influence: 31.1] [Reference Citation Analysis (3)] |
| 17. | Aminian A, Wilson R, Al-Kurd A, Tu C, Milinovich A, Kroh M, Rosenthal RJ, Brethauer SA, Schauer PR, Kattan MW, Brown JC, Berger NA, Abraham J, Nissen SE. Association of Bariatric Surgery With Cancer Risk and Mortality in Adults With Obesity. JAMA. 2022;327:2423-2433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 267] [Article Influence: 66.8] [Reference Citation Analysis (4)] |
| 18. | Adams TD, Meeks H, Fraser A, Davidson LE, Holmen J, Newman M, Ibele AR, Playdon M, Hardikar S, Richards N, Hunt SC, Kim J. Long-term cancer outcomes after bariatric surgery. Obesity (Silver Spring). 2023;31:2386-2397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 19. | Stenberg E, Ottosson J, Cao Y, Lagstam I, Näslund E. Risk for cancer after bariatric surgery compared with the general population: a nationwide matched cohort study. Obesity (Silver Spring). 2025;33:1725-1733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 20. | Heshmati K, Harris DA, Rosner B, Pranckevicius E, Ardestani A, Cho N, Golshan M, Ng WT, Ligibel J, Tavakkoli A, Sheu E. Association of Bariatric Surgery Status with Reduced HER2+ Breast Cancers: a Retrospective Cohort Study. Obes Surg. 2019;29:1092-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 21. | Tome J, Khatib M, Nizri E, Grigg LM, Orbach L, Lahat G, Eldar SM, Abu-Abeid A. Breast Cancer Characteristics after Metabolic and Bariatric Surgery: A Matched Comparison to Patients with Severe Obesity. Obes Surg. 2025;35:2847-2853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 22. | Wei Y, Wong SKH, Wu T, Law BTT, Ng EKW, Lam CLK, Wong CKH. Association between bariatric surgery and risks of cancer among Chinese patients with type 2 diabetes mellitus: A retrospective cohort study. J Diabetes. 2021;13:868-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 23. | Schauer DP, Feigelson HS, Koebnick C, Caan B, Weinmann S, Leonard AC, Powers JD, Yenumula PR, Arterburn DE. Bariatric Surgery and the Risk of Cancer in a Large Multisite Cohort. Ann Surg. 2019;269:95-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 283] [Article Influence: 40.4] [Reference Citation Analysis (2)] |
| 24. | Feigelson HS, Caan B, Weinmann S, Leonard AC, Powers JD, Yenumula PR, Arterburn DE, Koebnick C, Altaye M, Schauer DP. Bariatric Surgery is Associated With Reduced Risk of Breast Cancer in Both Premenopausal and Postmenopausal Women. Ann Surg. 2020;272:1053-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 68] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 25. | Anveden Å, Taube M, Peltonen M, Jacobson P, Andersson-Assarsson JC, Sjöholm K, Svensson PA, Carlsson LMS. Long-term incidence of female-specific cancer after bariatric surgery or usual care in the Swedish Obese Subjects Study. Gynecol Oncol. 2017;145:224-229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 103] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 26. | Kristensson FM, Andersson-Assarsson JC, Peltonen M, Jacobson P, Ahlin S, Svensson PA, Sjöholm K, Carlsson LMS, Taube M. Breast Cancer Risk After Bariatric Surgery and Influence of Insulin Levels: A Nonrandomized Controlled Trial. JAMA Surg. 2024;159:856-863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 27. | Chittajallu V, Mansoor E, Perez J, Omar YA, Firkins SA, Yoo H, Baggott B, Simons-Linares R. Association of Bariatric Surgery with Risk of Incident Obesity-Associated Malignancies: a Multi-center Population-Based Study. Obes Surg. 2023;33:4065-4069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 28. | Doumouras AG, Lovrics O, Paterson JM, Sutradhar R, Paszat L, Sivapathasundaram B, Tarride JE, Anvari M. Bariatric Surgery and Breast Cancer Incidence: a Population-Based, Matched Cohort Study. Obes Surg. 2022;32:1261-1269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 29. | Christou NV, Lieberman M, Sampalis F, Sampalis JS. Bariatric surgery reduces cancer risk in morbidly obese patients. Surg Obes Relat Dis. 2008;4:691-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 186] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 30. | Adams TD, Stroup AM, Gress RE, Adams KF, Calle EE, Smith SC, Halverson RC, Simper SC, Hopkins PN, Hunt SC. Cancer incidence and mortality after gastric bypass surgery. Obesity (Silver Spring). 2009;17:796-802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 333] [Cited by in RCA: 269] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 31. | Tao W, Santoni G, von Euler-Chelpin M, Ljung R, Lynge E, Pukkala E, Ness-Jensen E, Romundstad P, Tryggvadottir L, Lagergren J. Cancer Risk After Bariatric Surgery in a Cohort Study from the Five Nordic Countries. Obes Surg. 2020;30:3761-3767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 32. | Desai D, Singhal S, Koka J. Evaluating the Correlation of Bariatric Surgery and the Prevalence of Cancers in Obese Patients: A Study of the National Inpatient Sample (NIS) Database. Cureus. 2022;14:e23976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 33. | Mackenzie H, Markar SR, Askari A, Faiz O, Hull M, Purkayastha S, Møller H, Lagergren J. Obesity surgery and risk of cancer. Br J Surg. 2018;105:1650-1657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 34. | Aravani A, Downing A, Thomas JD, Lagergren J, Morris EJA, Hull MA. Obesity surgery and risk of colorectal and other obesity-related cancers: An English population-based cohort study. Cancer Epidemiol. 2018;53:99-104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 35. | Hassinger TE, Mehaffey JH, Hawkins RB, Schirmer BD, Hallowell PT, Schroen AT, Showalter SL. Overall and Estrogen Receptor-Positive Breast Cancer Incidences Are Decreased Following Bariatric Surgery. Obes Surg. 2019;29:776-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 36. | Doumouras AG, Lovrics O, Paterson JM, Sutradhar R, Paszat L, Sivapathasundaram B, Tarride JE, Anvari M. Residual Risk of Breast Cancer After Bariatric Surgery. JAMA Surg. 2023;158:634-641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 37. | Tsui ST, Yang J, Zhang X, Spaniolas K, Kim S, Griffin T, Burke WM, Pryor AD. The risk of female-specific cancer after bariatric surgery in the state of New York. Surg Endosc. 2021;35:4267-4274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Sjöholm K, Carlsson LMS, Svensson PA, Andersson-Assarsson JC, Kristensson F, Jacobson P, Peltonen M, Taube M. Association of Bariatric Surgery With Cancer Incidence in Patients With Obesity and Diabetes: Long-term Results From the Swedish Obese Subjects Study. Diabetes Care. 2022;45:444-450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 39. | Gérard C, Brown KA. Obesity and breast cancer - Role of estrogens and the molecular underpinnings of aromatase regulation in breast adipose tissue. Mol Cell Endocrinol. 2018;466:15-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 40. | Pollak M. Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer. 2008;8:915-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1697] [Cited by in RCA: 1581] [Article Influence: 87.8] [Reference Citation Analysis (0)] |
| 41. | Wong CC, Yu J. Gut microbiota in colorectal cancer development and therapy. Nat Rev Clin Oncol. 2023;20:429-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 536] [Cited by in RCA: 466] [Article Influence: 155.3] [Reference Citation Analysis (4)] |
| 42. | Ashrafian H, Ahmed K, Rowland SP, Patel VM, Gooderham NJ, Holmes E, Darzi A, Athanasiou T. Metabolic surgery and cancer: protective effects of bariatric procedures. Cancer. 2011;117:1788-1799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 112] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 43. | Lovrics O, Butt J, Lee Y, Lovrics P, Boudreau V, Anvari M, Hong D, Doumouras AG. The effect of bariatric surgery on breast cancer incidence and characteristics: A meta-analysis and systematic review. Am J Surg. 2021;222:715-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 44. | Alexander A, Brown N, Horns JJ, Hardikar S, Playdon M, Das R, Driggs N, Paudel N, Matsen C, Ibele A. Screening Mammography Adherence Improves After Bariatric Surgery. J Surg Res. 2024;303:476-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 45. | Tranchart H, Gaillard M, Kassir R, Gronnier C, Moszkowicz D, Ben Amor I, Lainas P, Savala N, Meunier H, Le Roux Y, Degrandi O, Baratte C, Vychnevskaia K, Gugenheim J, Dagher I. Malignancy in bariatric surgery patients: a French multisite cohort. Surg Endosc. 2021;35:6021-6030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 46. | Gagné DJ, Papasavas PK, Maalouf M, Urbandt JE, Caushaj PF. Obesity surgery and malignancy: our experience after 1500 cases. Surg Obes Relat Dis. 2009;5:160-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 47. | Parmar C, Abi Mosleh K, Aeschbacher P, Halfdanarson TR, McKenzie TJ, Rosenthal RJ, Ghanem OM; BRIDGE Study Group. The feasibility and outcomes of metabolic and bariatric surgery prior to neoplastic therapy. Surg Obes Relat Dis. 2024;20:717-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |