Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.113326
Revised: September 25, 2025
Accepted: January 23, 2026
Published online: March 24, 2026
Processing time: 213 Days and 16.3 Hours
The components of the tumor microenvironment are crucial in tumor growth, metastasis, immune evasion and therapeutic resistance. To adapt to the low-oxygen and nutrient-deficient conditions, cancer cells generate new blood vessels to promote tumor expansion and metastatic spread via tumor angiogenesis. Recent research has revealed that tumor endothelial cells reprogram their me
Core Tip: In recent years, the tumor microenvironment (TME) has emerged as a critical target in cancer research due to its role in regulating tumor growth, metastasis and treatment response. Immune cells are core components of the TME, and the effects of tumor cell metabolic reprogramming on immune cells are well understood. However, research on the impact of metabolic reprogramming of tumor endothelial cells on the tumor immune microenvironment is limited. This review summarizes metabolic changes in endothelial cells within the TME and their effects on the tumor immune microenvironment. It also suggests combining anti-angiogenic therapy that targets endothelial metabolism with immunotherapy as a future treatment option.
- Citation: Chen M, Chen ZG. Endothelial metabolic reprogramming influences tumor immune microenvironment. World J Clin Oncol 2026; 17(3): 113326
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/113326.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.113326
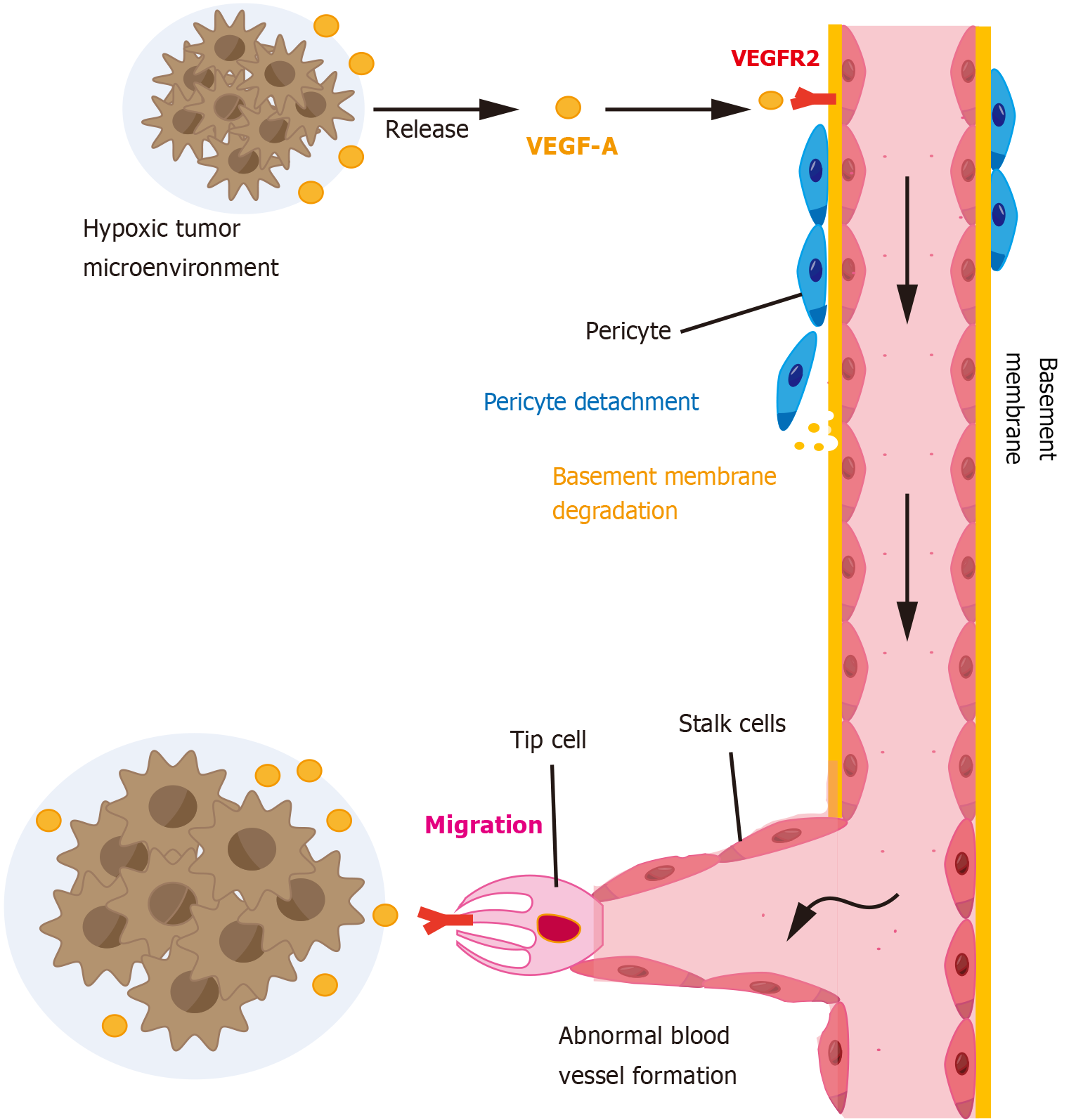
The constant and dynamic interactions between cancer cells and the tumor microenvironment (TME) are essential for tumor progression, metastasis, compromised immune surveillance and therapeutic response[1]. Within the TME, the main components are non-tumor cells, including fibroblasts, endothelial cells, pericytes, adipocytes, and immune cells, as well as non-cellular parts like the extracellular matrix and soluble molecules such as cytokines and growth factors[2]. The highly specific and complex TME can severely hinder the study of mechanisms and treatments for certain cancers. For instance, the efficacy of immunotherapy and anti-angiogenic therapy (AAT) in pancreatic neuroendocrine neoplasms remains limited due to their unique characteristics[3]. Therefore, analyzing the TME may provide crucial insights for identifying new targets in refractory tumors. Tumor endothelial cells (TECs) play a significant role in the TME and execute distinct functions that are stimulated by various inflammatory factors or angiogenesis-inducing cytokines, facilitating tumor growth and promoting distant metastasis[4]. Thus, the deadly tumor progression is heavily dependent on the tumor-vessel interactions. Under hypoxic conditions in the TME, quiescent endothelial cells become active, vascular endothelial growth factor (VEGF) expression is upregulated, and the angiogenic switch occurs when VEGF binds to the receptors on endothelial cells[5,6] (Figure 1). In addition, pericytes detachment and basement membrane degradation promote the migration of tip cells towards the tumor[7]. However, the new vessels from tumor angiogenesis are structurally and functionally abnormal. Twisted vascular structures and aberrant metabolic reprogramming of TECs could influence the infiltration and function of immune cells. For instance, TECs induce tumor-infiltrating T cells ex
A defining feature of cancer is the alteration of metabolism[10]. In recent times, it is becoming clear that the components of the TME regulates its own metabolism, including TECs, to meet the high energy needs and fast growth of tumors. TECs survive and form new blood vessels depending on various metabolic pathways, including glycolysis, oxidation of fatty acids (FAs) and glutamine metabolism[11,12]. Despite extensive research into the metabolic alterations of cancer cells and individual cell types within the TME, the interactions between TECs and other TME components (such as immune cells) via metabolites are still unclear. Recently, research has reported that hypoxia induces endothelial metabolic reprogramming and DGKG-high expressing TECs promote angiogenesis and immune evasion by recruiting ubiquitin-specific peptidase 16 to facilitate zinc finger E-box binding homeobox 2 deubiquitination and then initiating the transforming growth factor-β1 positive feedback loop[13].
Immunotherapy has ushered in a new era of cancer therapy. It has offered long-lasting clinical responses for certain groups of cancer patients. It has been demonstrated that the TME significantly influences tumor growth and immunotherapy resistance[14-16]. Immunotherapy in combination with AAT is one of the classic cancer treatment strategies[17-19]. The metabolism of TECs is a key regulator of angiogenesis, and tumor vessel normalization has emerged as a novel therapeutic strategy[20,21]. Therefore, combining new agents that target the endothelial metabolism with immune modulators may be a promising therapeutic approach. This review highlights the metabolic reprogramming of TECs, the impact on the tumor immune microenvironment, and discusses a potential therapeutic possibility.
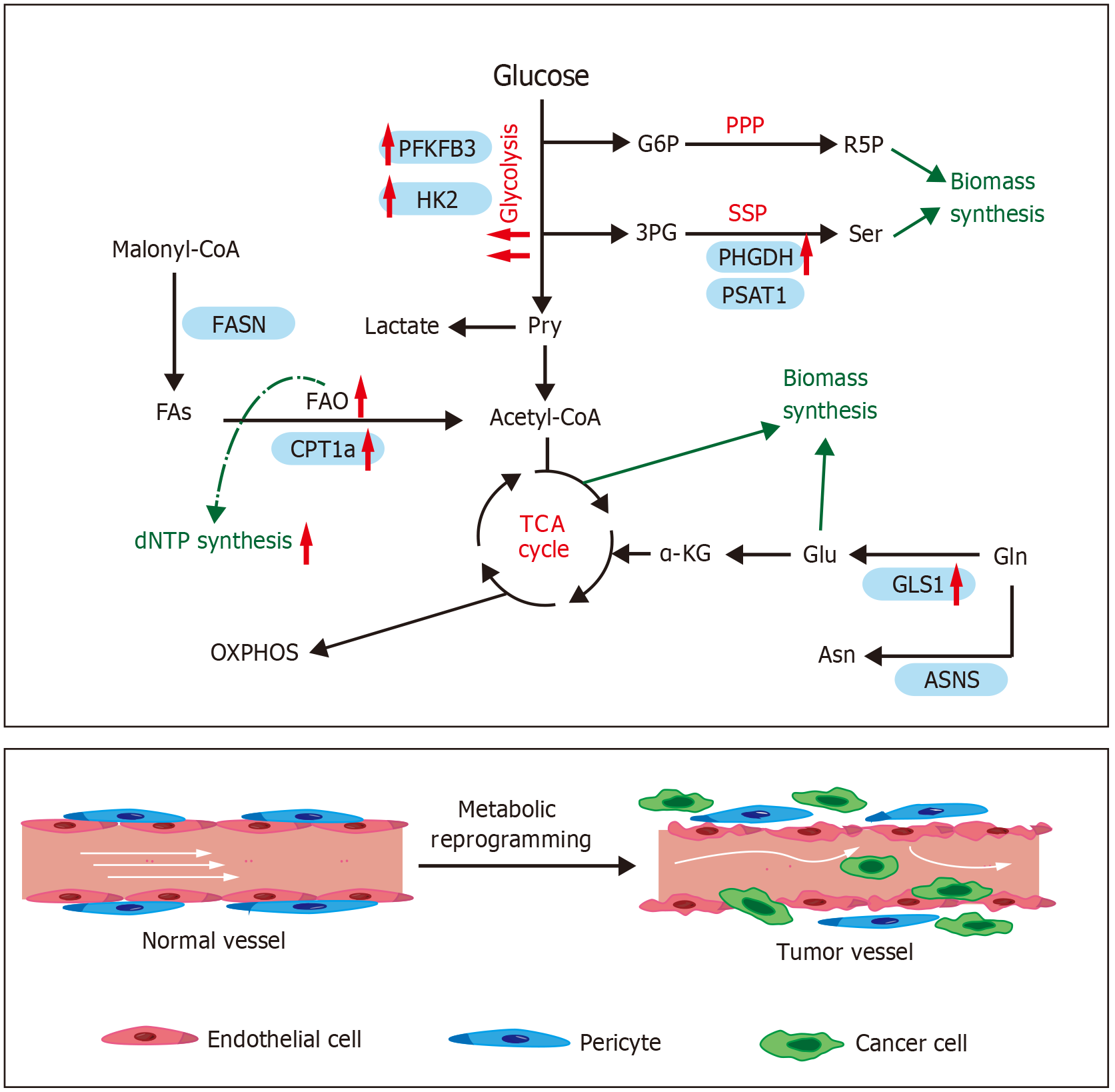
Despite being in an oxygen-rich condition, energy production in resting endothelial cells is primarily dependent on anaerobic glycolysis. In addition, FA oxidation (FAO) in endothelial cells provides energy and a substrate for nucleotide synthesis[12,22]. In contrast to the normal state, the structural malformation and dysfunction of blood vessels in tumors, coupled with the vigorous proliferation of the cancer cells, lead to poor perfusion, hypoxic states and metabolic disorders in the TME[10]. TECs act as a metabolic interface between the blood flow and the tumor, and alterations of their metabolism will affect cancer cells and other components within the TME (Figure 2).
Compared to normal endothelial cells, TECs have higher levels of glycolysis. Some studies have found that TECs have higher expression levels of glycolytic genes, including 6-phosphofructo-2-kinase/fructose-2,6-bisphosph-atase 3 (PFKFB3), the glucose transporter glucose transporter 2 and rate-limiting enzymes of the glycolytic bypass pathway[23,24]. The glycolytic pathway is regulated by three enzymes: Phosphofructokinase-1 (PFK1), hexokinase 2 (HK2) and pyruvate kinase[25]. PFKFB3, a glycolytic activator, activates PFK1 and is overexpressed on TECs. Researchers have demonstrated that reducing or inhibiting PFKFB3 does not impact tumor growth, but instead hinders tumor angiogenesis, which in turn decreases cancer cell invasion, intravasation, and metastasis[24,26-28]. Therefore, targeting the PFKFB3-mediated glycolytic process of TECs could be an effective anti-angiogenic treatment approach. HK2 is another rate-limiting enzyme for glycolysis. Research into lung cancer has revealed the key role of the Foxp1-hypoxia-inducible factor 1-alpha-Hk2 pathway in regulating glycolysis in TECs, which in turn affects tumor angiogenesis[29]. Knockdown of Foxp1 results in attenuated inhibition of hypoxia-inducible factor 1-alpha-Hk2 pathway, which promotes glycolysis of TECs and neo-angiogenesis in lung tumor.
Research indicates that highly differentiated tumors exhibit lower vascularization compared to poorly differentiated tumors, and vascularization levels correlate with tumor prognosis and survival rates[3]. In highly vascularized tumors such as neuroendocrine tumors, VEGF levels in the microenvironment can reach up to 80%, thereby driving angiogenesis and tumorigenesis. The upregulation and sustained effects of angiogenesis inducers are crucial for boosting glycolysis in TECs. VEGF-A is a well-known molecule that encourages angiogenesis[10], and it increases the PFKFB3-driven glycolysis[30]. Fibroblast growth factor (FGF) is another element that can influence HK2 expression by controlling c-MYC expression[31]. The transcription factors forkhead box O (FOXO) are essential for cellular metabolism and tissue homeostasis[32]. FOXO1 maintains endothelial cells quiescence by inhibiting c-MYC signal[33,34]. Inhibition of FOXO1 expression in TECs is a driver of glycolysis and angiogenesis[35,36].
Another study in colorectal cancer (CRC) found that high glyceraldehyde-3-phosphate dehydrogenase expression in CRC patients was associated with enhanced glycolysis in TECs and the application of low-dose glyceraldehyde-3-phosphate dehydrogenase inhibitors normalized tumor vasculature and increased the infiltration of immune effector cells[37]. In addition to the high glycolytic metabolism, TECs have elevated transcript levels of the pentose phosphate pathway and the serine biosynthesis pathway related enzymes, representing an upregulation of the nucleotide synthesis pathway in TECs[12,24].
Fructose is a common food additive in today’s society. Studies have shown that excessive fructose intake is strongly associated with obesity, metabolic syndromes and cancer[38]. Some other studies have found that reprogramming of fructose metabolism in the TME promotes the progression and metastasis of various cancers[39-41]. As components of the TME and important participants in tumor angiogenesis, TECs are also affected by fructose metabolism. Fang et al[42] and Cui et al[43] discovered that vascular endothelial cells can metabolize fructose, thereby activating the protein kinase B, Scr and AMP-activated protein kinase signaling pathways, which enhances their proliferation and migration. Additionally, fructose indirectly promotes angiogenesis by increasing VEGF expression in cancer cells.
In addition to glycolysis, activated endothelial cells increase their levels of oxidative phosphorylation (OXPHOS) to meet their energy needs. Hyperproliferating endothelial cells consume a lot of oxygen, and mitochondrial overuse makes them sensitive to uncoupling agents. When endothelial cells are treated with embelin, neoangiogenesis is effectively inhibited, which inhibits tumor growth[44]. Another genetic evidence similarly demonstrates the critical role of OXPHOS in the vascular endothelium for tumor angiogenesis and cancer progression. The researchers conducted experiments using mice with endothelium-specific deletion of cox10 and found that wound healing was slowed and tumor growth was impaired in mice under conditions of high energy consumption[45]. Elevated fructose metabolism in the TME can promote mitochondrial respiration and enhance endothelial cell migration by activating the AMP-activated protein kinase signaling pathway[42].
The above studies suggest that blocking the OXPHOS process in endothelial cells is a promising approach for treating cancer progression. However, not all mitochondrial respiration inhibitors target endothelial cells specifically. The β1 receptor inhibitor nebivolol, for example, can interfere with the OXPHOS process in cancer cells by inhibiting the activity of the OXPHOS complexes, yet it does not affect the mitochondrial respiratory function of TECs[46]. However, nebivolol interferes with endothelial cell proliferation because it blocks extracellular signal-regulated kinase activation and inhibits the glycolytic process of TECs[46].
Endothelial cells metabolize FA to acetyl-CoA for maintenance of the tricarboxylic acid (TCA) cycle for energy. More importantly, FAO facilitates deoxy nucleotide triphosphate (dNTP) synthesis in proliferating endothelial cells[34,47,48]. Reduced FAO in endothelial cells doesn’t lead to energy depletion, but it impacts DNA synthesis. Studies of isotopically labelled endothelial cells have shown that the carbons in FA enter the Krebs cycle and become part of aspartate, uridine monophosphate and DNA[49]. Carnitine palmitoyltransferase 1A (CPT1A) is the enzyme that limits the rate of FAO and is responsible for transporting FAs to mitochondria for β-oxidation. It plays an important role in regulating the proliferation of endothelial cells. Pharmacological inhibition of CPT1 by using etomoxir as well as selective knockdown of the CPT1a gene in endothelial cells resulted in reduced endothelial cells proliferation, decreased dNTPs synthesis and impaired neo-angiogenesis[34].
FA synthase (FASN) is an important enzyme for synthesizing lipids endogenously. It catalyzes the production of palmitate, a long-chain FA, from acetyl-CoA and malonyl-CoA in the presence of NADPH[50,51]. Numerous past studies have shown that FASN is highly expressed in various tumors and is linked to a poor prognosis, including cervical[52], gastric[53], breast[54] and gallbladder cancer[55]. However, there are fewer studies of FASN in TECs. FASN has a promoting effect on tumor vessel sprouting[56]. Knocking down FASN in endothelial cells led to higher malonyl-CoA levels, resulting in mammalian target of rapamycin (mTOR) malonylation, impaired the activity of mTOR1 kinase and ultimately inhibited endothelial cells proliferation[56]. In addition, an interesting study in lymphatic endothelial cells (LECs) showed that FASN plays a role in supporting human LECs viability, proliferation and migration[57].
In the last ten years, numerous studies have emphasized the significance of glutamine metabolic reprogramming in cancer[58,59], establishing it as a potential target for treatment[60,61]. Nonetheless, the impact of endothelial glutamine metabolism reprogramming on tumor cells and TME have not been thoroughly investigated. Glutamine, which is the most prevalent non-essential amino acid in the somatic circulation, is absorbed by cells and catabolized by glutaminase (GLS) into glutamate and ammonia, with glutamate entering the TCA cycle as α-ketoglutarate[5]. One of the few studies focusing on endothelial cell metabolism found that endothelial cells have a high rate of glutamine metabolism[62]. Glutamine supplies carbon to the TCA cycle and nitrogen for the synthesis of pyrimidines and purines, and is meta
Endothelial cells can also use glutaminolysis-derived glutamate to synthesize aspartate with the help of aspartate aminotransferase[68]. Through activation of the mTOR complex 1 pathway, aspartate regulates VEGF receptor 2 and FGF receptor 1 expression in endothelial cells and pyrimidine synthesis, thereby promoting tumor angiogenesis[68]. In addition to the above amino acids, proliferating endothelial cells also use serine to support intracellular biosynthesis. Serine can be taken up directly from extracellular sources or synthesized from 3-phosphoglycerate[5]. The phosphoglycerate dehydrogenase (PHGDH) is a key enzyme of the serine synthesis pathway and is essential for endothelial cell survival and proliferation. Knockdown of PHGDH in endothelial cells severely impairs endothelial cell survival and proliferation. This is due to mitochondrial dysfunction caused by insufficient heme synthesis[69]. Together, these findings emphasize the importance of endothelial metabolic reprogramming in solid tumor progression.
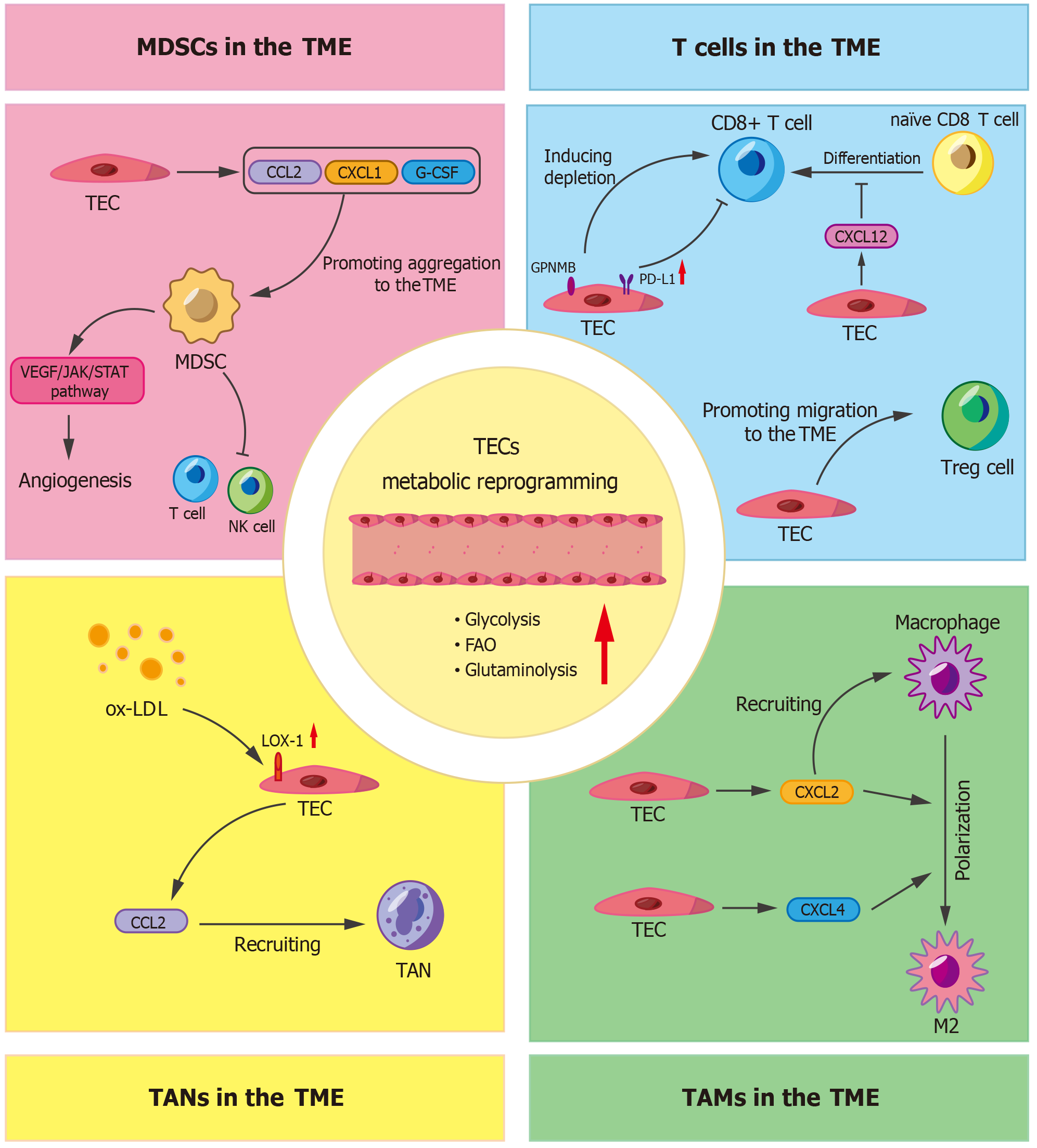
In tumors, endothelial cells show increased glycolysis, FA metabolism and glutamine metabolism to sustain their survival and proliferation. In addition to tumor cells, metabolic reprogramming of endothelial cells also help to create a metabolic environment conducive to immune evasion and tumor progression[70]. A study focusing on endothelial cell phenotypes in lung TME found that nucleotide metabolic pathways were significantly enhanced in TECs, and other pathways affected were OXPHOS and glycolysis[71]. However, genes related to immune activation and immune cell homing were expressed at lower levels in TECs[71]. DGKG is an enzyme associated with lipid metabolism, and hypoxia induces endothelial cell-specific high expression of DGKG, which would promote angiogenesis and immune evasion in hepatocellular carcinoma[13]. More importantly, tumor progression can be synergistically inhibited when targeting enzymes involved in glucolysis or lipid metabolism in TECs is combined with anti-programmed cell death-1 blockade[13,37]. Thus, TECs metabolism may differently affect anti-tumor immunity and contribute to immune evasion (Figure 3). The effects of TECs metabolic reprogramming on various immune cells will be discussed in detail below.
T cells not only interact with adjacent antigen-presenting cells but also with endothelial cells, which are regarded as the metabolic bridge between the blood and various tissues and organs in the body[4]. Researchers have identified an immunoregulatory endothelial cell subtype in triple-negative breast cancer (TNBC) that express high levels of tumor necrosis factor receptor 2 in chemoresistant TNBC patients[72]. Tumor necrosis factor receptor 2 activates the nuclear factor kappa B pathway to inhibit glycolysis and increases programmed death-ligand 1 (PD-L1) expression in endothelial cells, thereby inducing CD8+ T cells suppression[72]. In hepatocellular carcinoma, TECs induced CD8+ T cell depletion by upregulating the expression of the membrane glycoprotein nonmetastatic melanoma protein B[8]. This new target offers another potential treatment option for hepatocellular carcinoma. Lu et al[73] identified a novel TEC subpopulations that secretes C-X-C motif ligand 12 (CXCL12) and inhibits the differentiation of naïve CD8 T cells into cytotoxic CD8 T cells. Therefore, these TECs play an important role in shaping the immunosuppressive microenvironment.
It has been found that human umbilical vein endothelial cells (HUVECs) regulate the proliferation and activation of CD4+ T cells and regulatory T (Treg) cells[74]. Treg cells within the TME promote tumor development by inhibiting anti-tumor responses and interacting with tumor stromal cells such as fibroblasts and endothelial cells[75]. A high pro-angiogenic state is associated with the accumulation of tumor-infiltrating Treg cells in renal cell carcinoma, and the angiogenic network and immune tolerance cooperate with each other to ultimately promote tumor growth and progression[76]. In addition, in LECs, metabolic reprogramming affects immune cell stability and infiltration. Abnormal metabolism of LECs induced by a high cholesterol environment limits their ability to stabilize Treg cells[77]. High levels of cholesterol metabolites induced hyper-activation of the interleukin-18/nuclear factor kappa B/vascular cell adhesion molecule 1 axis in LECs, which inhibit Treg cells migration[77].
TAMs are a primary type of immune cell found within tumors. They are usually polarized into two functionally contradictory subtypes, pro-inflammatory or immuno-suppressive[78,79]. To date, numerous studies have demonstrated that TAMs can promote tumor angiogenesis by secreting pro-angiogenic factors[78]. In pancreatic neuroendocrine neoplasms, the cellular density of TAMs is significantly correlated with invasive and malignant features, and these cells show markedly increased infiltration in liver metastases. This may be due to TAMs promoting the angiogenic switch[80,81]. Accordingly, angiogenic factors can shape the immuno-suppressive microenvironment by directly suppressing antigen-presenting cells and immune effector cells or by enhancing the effect of Treg cells, myeloid-derived suppressor cells (MDSCs) and TAMs[82]. Interactions between angiogenesis and TAMs polarization drive tumor initiation and progression. Some researchers have also found a close relationship between TECs and TAMs in tumorigenesis and progression. A senescence-associated secretory phenotype can be produced by senescent cells in the TME, and this can promote tumorigenesis. Researchers found that the most common types of senescent cells in KRAS-driven mouse lung cancer were macrophages and endothelial cells[83]. They also found that removing senescent macrophages reduced tumor load and improved survival. Although the potential mechanism of interaction between senescent endothelial cells and macrophages was not fully explored, this study provides ideas and rationale for future research. Further evidence of a strong link between TECs and TAMs in the TME is that reprogramming the tuberous sclerosis complex-mTOR complex 1 signaling pathway of TAM enhances its competitive advantage over endothelial progenitor cells, thereby inhibiting tumor neoangiogenesis[84].
A wide variety of metabolites in the TME are involved in shaping the TAMs phenotype[85,86], and the role of me
TANs are also one of the immune cells that play a unique role in the TME. TANs not only stimulate tumor angiogenesis and epithelial-to-mesenchymal transition, but also specifically suppress the immune function of T cells[90]. Additionally, TANs interact with tumor cells. Under the stimulation of certain cytokines, such as transforming growth factor-β, they may transition from an antitumor (N1) phenotype to a pro-tumor (N2) phenotype[91,92]. The recruitment of these neutrophils requires close interaction with endothelial cells. Therefore, metabolic reprogramming of endothelial cells in the TME may affect the function of neutrophils in close contact with them. A multi-omics study focusing on the TME of intrahepatic cholangiocarcinoma found that intrahepatic cholangiocarcinoma patients with a high tumor-promoting propensity of TECs had an increased infiltration of neutrophils and a decreased number of natural killer cells and effector memory T cells in the TME[93]. More interestingly, TECs highly express lectin-like oxidized low-density lipoprotein receptor-1, which is recognized as a receptor for oxidized low-density lipoprotein, a key molecule in cardiovascular disease. The lectin-like oxidized low-density lipoprotein receptor-1/oxidized low-density lipoprotein axis induces TECs to secrete C-C motif ligand 2 to recruit neutrophils, thereby creating a microenvironment conducive to tumor metastasis[94].
MDSCs play a complicated role in regulating the tumor immune microenvironment. On the one hand, they can inhibit the killing effects of T lymphocytes and natural killer cells, and recruit Treg cells to further enhance the immunosuppressive effects; on the other hand, MDSCs can also induce tumor angiogenesis through the VEGF/Janus kinase/signal transducer and activator of the transcription signaling pathway[95]. Although a large number of previous studies have thoroughly explored the vital role of MDSCs in immunosuppression and angiogenesis, research on how metabolic alterations in TECs impact MDSCs status remains limited. A study by He et al[96] found that knocking out the Shb gene in mouse endothelial cells promoted the recruitment of MDSCs in breast cancer. The exact mechanism of this phenomenon is unclear, but elevated expression of C-C motif ligand 2, CXCL1, and granulocyte colony-stimulating factor was found in Shb-KO endothelial cell-derived CD45+ cells, which creates an excellent microenvironment for the aggregation of MDSCs.
In conclusion, these discoveries reveal a strong connection between TECs and tumor-infiltrating immune cells. TECs’ metabolic changes influence both the recruitment and the function of tumor-infiltrating lymphocytes and immune-regulatory cells, while the secretion of angiogenic factors by immuno-regulatory cells promotes abnormal neoangiogenesis of tumors, further aggravating the hypoxic state of TME.
By uncovering the features and processes of tumor angiogenesis, numerous AAT have been created, focusing on blocking blood vessel growth to deprive cancer cells of nutrients. Tumor angiogenesis is mainly induced by pro-angiogenic factors in the TME, and common classical pro-angiogenic factors include VEGF, FGF-2 and platelet derived growth factor[97]. Common drugs that selectively target VEGF are bevacizumab, aflibercept and ranibizumab[97]. However, some cancers that are initially effective often develop resistance during treatment, resulting in poor prognosis. Numerous studies have identified and summarized the mechanisms behind AAT resistance: Increased recruitment of bone marrow-derived cells[98], cancer cells achieve vasculogenic mimicry by upregulating Foxc2[99], microRNAs regulate VEGF expression[100], and long noncoding RNAs promote vessel co-option by inhibiting the process of autophagy degradation of alanine-serine cysteine-preferring transporter 2[101,102]. Additionally, tumor cells are also able to develop AAT resistance through metabolic symbiosis[103,104].
The emergence of these drug-resistant responses suggests that the paradigm of blocking angiogenesis in solid tumors must change. Therefore, in the following years, the concept of “tumor vascular normalization” was proposed. Rather than inhibiting neoangiogenesis, the concept was to promote the transformation of the tumor vasculature to a more mature phenotype, which would facilitate the delivery of therapeutic agents and the infiltration of immune effector cells[105-109]. Abnormal TECs metabolism drives angiogenesis and worsens hypoxia in the TME. Targeting TECs metabolism to interfere with pathological angiogenesis and thereby normalize the tumor blood vessels has become a new method[110-112] (Table 1).
| Metabolic type | Target | Agent | Conditions | Mechanism | Ref. |
| Glycolysis | PFKFB3 | PFK15 | Hepatocellular carcinoma | Suppressing glycolysis in TECs and tumor cells and inducing apoptosis. Inducing tumor vessel normalization | [28] |
| Apatinib | Hepatocellular carcinoma | Suppressed glycolysis in ECs via blocking PI3K/AKT/PFKFB3 pathway | [113] | ||
| 3PO | Pathological neovascularization | Inhibiting glycolysis in ECs partially and reversibly. Enhancing the antiangiogenic efficacy of VEGF blockade | [114] | ||
| PKM2 | SAA | Melanoma and lung cancer | Reducing glycolysis in ECs and activating the PKM2-dependent β-catenin/claudin-5 signaling axis improves endothelial integrity | [115] | |
| Fatty acid metabolism | FASN | Orlistat | Retinopathy of prematurity | Inhibiting pathologic ocular neovascularization by decreasing mTOR activity | [56] |
| Orlistat | Melanoma | Reducing HDLEC cells proliferation and migration. Inhibiting the secretion of VEGF-C and promoted the secretion of VEGF-D by melanoma cells | [57] | ||
| CPT1 | Etomoxir | Lymphangiogenesis | Inhibiting injury-induced lymphangiogenesis | [119] | |
| Glutamine metabolism | GLS | miR-367b-3p | Choroidal neovascularization | Inhibiting CEBPB leads to the inactivation of GLS1 transcription. Inhibiting ECs proliferation, migration and tubular structure formation while promoting apoptosis | [122] |
| GS | MSO | Lung cancer | Inducing a shift from the M2 phenotype to the M1 phenotype in macrophages. Promoting tumor vessel normalization and inhibiting the metastasis of cancer cells | [124] | |
| OXPHOS | Mitochondria | Embelin | Pathological neoangiogenesis in tumor and injury | Depleting the low respiratory reserve of proliferating ECs without deleterious effects on resting ECs | [44] |
Compared to normal endothelial cells, TECs upregulated the expression of the glycolytic activator PFKFB3 and exhibited stronger glycolysis. In recent years, it has been discovered that inhibiting PFKFB3-driven glycolysis, directly or indirectly, induces tumor vascular normalization and inhibits tumor progression. For example, PFK15 specifically inhibit PFKFB3 in TECs to normalize the tumor vasculature[28]. Apatinib, the drug of choice for the treatment of hepatocellular carcinoma, reduces glycolysis in TECs by blocking the phosphoinositide 3-kinases/protein kinase B/PFKFB3 pathway[113]. Schoors et al[114] found that 3-(3-pyridinyl)-1-(4-pyridinyl)-2-propen-1-one, a small molecule inhibitor of PFKFB3, transiently reduced glycolysis in vivo and inhibited endothelial cell proliferation and migration, but this was sufficient to reduce pathological neovascularization. Meanwhile, blocking PFKFB3 with 3-(3-pyridinyl)-1-(4-pyridinyl)-2-propen-1-one did not impact tumor growth, but it did decrease cancer cell invasion, intravasation, and metastasis by promoting vascular maturation and improving blood perfusion[24].
PFKFB3 is not the only target that regulates the metabolism of TECs. Under hypoxic conditions, salvianic acid A binds to the glycolytic enzyme pyruvate kinase M2 in HUVECs and significantly reduces glycolysis[115]. In a study of TNBC, electroacupuncture was discovered to inhibit glyoxalase in TECs to attenuate endothelial cell glycolysis, inhibit cell proliferation, and promote vascular normalization[116]. It has been found that transient receptor potential melastatin 7 channels might be a regulator of glycolytic reprogramming in endothelial cells, and inhibiting transient receptor potential melastatin 7-dependent glycolysis could offer a new therapeutic approach for treating cancer[117].
In previous discussions, we have known that FAO can provide carbon for dNTP synthesis, which is essential for endothelial cell proliferation[48,49]. Targeting enzymes involved in FA metabolism in endothelial cells, such as FASN and CPT1, appears to be an ideal therapeutic approach. FASN inhibition with cerulenin and orlistat reduced LECs viability, proliferation and migration[57]. And in vivo experiments showed that FASN inhibitors reduced lymphatic metastasis of melanoma[57]. In a study of metastasis and angiogenesis in B16-F10 melanoma, FASN inhibitor treatment was found to reduce endothelial cell (rat aortic endothelial cell and HUVEC) viability, proliferation and formation of abnormal vascular structures[118]. Some researchers, in order to test the therapeutic potential of FASN blockade in pathologic angiogenesis, used a mouse model of retinopathy of prematurity and found that low-dose orlistat treatment inhibited ocular pathologic angiogenesis in mice by decreasing mTOR activity[56]. In addition, blocking CPT1 with etomoxir inhibits injury-induced lymphangiogenesis[119]. Thus, FASN and CPT1 may be potential targets for tumor vascular normalization. Targeting these two enzymes may be a novel way to stop tumor progression and metastasis.
The enzyme GLS1 converts glutamine into glutamate and ammonia. In endothelial cells, this GLS1-driven metabolism encourages their growth, movement, and longevity[120]. When HUVECs were treated with 6-diazo-5-oxo-L-norleucine, a specific inhibitor of GLS, the proliferative capacity of the cells was significantly reduced and shifted toward a senescent phenotype[121]. In addition, another GLS1 inhibitor, bis-2(5-phenylacetamido-1,2,4-thiadiazol-2-yl)ethylsulfide, also normalizes HUVECs metabolism by modulating glutamine metabolism and reduces proliferation and migration[66]. MicroRNAs have also been found to block pathologic angiogenesis by inhibiting glutamine metabolism in studies of choroidal neovascularization-related therapeutic targets[122]. Asparagine synthetase (ASNS) synthesizes asparagine using aspartic acid and ammonia from glutamine[123]. Since ASNS silencing impairs endothelial cell proliferation, not only GLS1 but also ASNS may be a potential target for regulating endothelial metabolism[123]. Interestingly, pharmacological or genetic targeting of glutamine metabolism may indirectly interfere with tumor angiogenesis and inhibit cancer metastasis. Glutamine-synthetase (GS) is the enzyme responsible for catalyzing the conversion of glutamate into glutamine. Pharmacological inhibition of GS induces macrophage polarization from the M2 phenotype to the M1 type, which promotes endothelial cell branching and deletion of the macrophage GS gene promotes normalization of the tumor vasculature[124].
Numerous previous studies have confirmed the effectiveness of combining conventional anti-angiogenic agents with immune checkpoint blockades for cancer treatment[17-19,125,126] (Table 2). In addition to targeting angiogenic factors, targeting endothelial metabolism may be another useful perspective in the study of tumor vasculature normalization in combination with immunotherapy. For example, it has been found that aerobic glycolysis mediates the upregulation of cellular PD-L1 expression[127], whereas TECs express PD-L1[72,128], and targeting glycolysis may be able to reduce PD-L1 expression on both tumor cells and TECs, which would be beneficial for enhancing the anti-tumor effects of immune checkpoint blockade. Additionally, enhanced endothelial glycolysis mediated by PHGDH leads to excessive proliferation of TECs, which is one of the factors that drive glioblastoma resistance to chimaeric antigen receptor T therapy[129]. Targeting endothelial PHGDH inhibits abnormal vascular proliferation, improves the hypoxic microenvironment, and promotes immune cell infiltration into tumors. This discovery reveals the potential of targeting endothelial metabolic reprogramming in combination with chimaeric antigen receptor T therapy. Although AAT has become the standard treatment, overcoming resistance to it remains a challenge. It is believed that metabolic reprogramming of TECs plays a significant role in treatment resistance[130]. A study on metastatic CRC revealed that, under anti-VEGFA treatment, the TEC-specific F3 protein inhibits the degradation of the CPT1A protein via the mitogen-activated protein kinase/c-Jun NH2-terminal kinase-mitogen-activated protein kinase/extracellular signal-regulated kinase-TP53/p53 signaling axis, thereby enhancing the FAO of TECs[131]. Further studies confirm that targeting FAO effectively overcomes resistance to anti-VEGFA therapy. Therefore, in addition to immunotherapy, combining anti-angiogenic drugs with endothelial metabolism targeting can further enhance tumor treatment efficacy.
| Inhibitor | Clinical stage | Study status | Conditions | Intervention | Ref. |
| Anlotinib | II | Recruiting | NSCLC | In combination with penpulimab | NCT06341530 |
| II | Recruiting | Breast cancer | In combination with sintilimab and chemotherapy | NCT04877821 | |
| Apatinib | II | Ongoing | Solid tumor | In combination with adebrelimab and microwave ablation | NCT06537505 |
| Axitinib | II | Completed | Advanced melanoma | In combination with nivolumab | NCT04493203 |
| Bevacizumab | I/II | Completed | Malignant solid tumor | In combination with NK immunotherapy | NCT02857920 |
| II | Completed | Colorectal cancer | In combination with atezolizumab and chemotherapy | NCT02873195 | |
| I/II | Recruiting | Ovarian cancer | In combination with oregovomab and chemotherapy | NCT04938583 | |
| II/III | Recruiting | Colorectal cancer liver metastases | In combination with serplulimab and chemotherapy | NCT06280495 | |
| II | Recruiting | Gastric cancer | In combination with neoadjuvant chemotherapy and | NCT06371586 | |
| II | Recruiting | Recurrent TNBC | In combination with pembrolizumab and chemotherapy | NCT06976944 | |
| IV | Ongoing | Advanced melanoma | In combination with iparomlimab and tuvonralimab | NCT07004335 | |
| Ramucirumab | II | Recruiting | Gastric cancer | In combination with pembrolizumab and chemotherapy | NCT04069273 |
| Sorafenib | II | Completed | Hepatocellular carcinoma | In combination with PD-1 mAb | NCT04518852 |
Research exploring targets for endothelial metabolic reprogramming is gaining momentum. An increasing number of studies have demonstrated that targeting endothelial metabolism can promote tumor vasculature normalization, thereby enhancing the efficacy of both immunotherapy and conventional anti-angiogenic treatments. However, further clinical validation is required to determine its translational value in the field of cancer therapy.
Currently, studies targeting the non-cancerous cellular components of the TME have become increasingly intensive. In this context, some progress has been made in the development of tumor vascular normalization therapy focusing on TECs. However, conventional antiangiogenic therapy has drawbacks, and there is now a need to develop a new approach that is different from previous antiangiogenic therapies. With the increasing research on endothelial cell metabolism and its effects on normalizing tumor vasculature, the application of modulating TECs metabolism in cancer therapy is promising. Furthermore, metabolic reprogramming of TECs plays an important role in chemotherapeutic drug delivery and immune evasion. In the future, in addition to the use of monotherapies targeting the metabolism of TECs, combinations of chemotherapy and immunotherapy should be explored to achieve greater therapeutic potential. Previous studies have demonstrated that risk models based on genes associated with lipid metabolism may predict the prognosis of CRC and response to immunotherapy[132]. This also provides us with a new perspective: Evaluating differences in TECs metabolic characteristics between tumor samples obtained before and after treatment may offer novel insights for AAT or immunotherapy. In addition, correlating these metabolic characteristics with clinical endpoints, such as response rates and progression-free survival, could indicate that TECs metabolic reprogramming serves as a new prognostic indicator.
The authors thank Chen C and Liao WX for critically reviewing the manuscript.
| 1. | Xiao Y, Yu D. Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther. 2021;221:107753. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1650] [Cited by in RCA: 1461] [Article Influence: 292.2] [Reference Citation Analysis (5)] |
| 2. | Fane M, Weeraratna AT. How the ageing microenvironment influences tumour progression. Nat Rev Cancer. 2020;20:89-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 640] [Article Influence: 106.7] [Reference Citation Analysis (0)] |
| 3. | Neves FR, Martins AL, Oliveira RC, Martins R. Characterization of the Pancreatic Neuroendocrine Neoplasm Immune Microenvironment. Cancer Med. 2025;14:e70798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 4. | Augustin HG, Koh GY. A systems view of the vascular endothelium in health and disease. Cell. 2024;187:4833-4858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 206] [Article Influence: 103.0] [Reference Citation Analysis (0)] |
| 5. | Kane K, Edwards D, Chen J. The influence of endothelial metabolic reprogramming on the tumor microenvironment. Oncogene. 2025;44:51-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 36] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 6. | Lugano R, Ramachandran M, Dimberg A. Tumor angiogenesis: causes, consequences, challenges and opportunities. Cell Mol Life Sci. 2020;77:1745-1770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 422] [Cited by in RCA: 1403] [Article Influence: 200.4] [Reference Citation Analysis (4)] |
| 7. | Jiang Z, Zhou J, Li L, Liao S, He J, Zhou S, Zhou Y. Pericytes in the tumor microenvironment. Cancer Lett. 2023;556:216074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 107] [Reference Citation Analysis (0)] |
| 8. | Sakano Y, Noda T, Kobayashi S, Sasaki K, Iwagami Y, Yamada D, Tomimaru Y, Akita H, Gotoh K, Takahashi H, Asaoka T, Tanemura M, Wada H, Doki Y, Eguchi H. Tumor endothelial cell-induced CD8(+) T-cell exhaustion via GPNMB in hepatocellular carcinoma. Cancer Sci. 2022;113:1625-1638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 9. | McCoy MG, Nascimento DW, Veleeparambil M, Murtazina R, Gao D, Tkachenko S, Podrez E, Byzova TV. Endothelial TLR2 promotes proangiogenic immune cell recruitment and tumor angiogenesis. Sci Signal. 2021;14:eabc5371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 10. | Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55210] [Cited by in RCA: 48658] [Article Influence: 3243.9] [Reference Citation Analysis (11)] |
| 11. | Li X, Sun X, Carmeliet P. Hallmarks of Endothelial Cell Metabolism in Health and Disease. Cell Metab. 2019;30:414-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 360] [Article Influence: 51.4] [Reference Citation Analysis (0)] |
| 12. | Rohlenova K, Veys K, Miranda-Santos I, De Bock K, Carmeliet P. Endothelial Cell Metabolism in Health and Disease. Trends Cell Biol. 2018;28:224-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 249] [Article Influence: 31.1] [Reference Citation Analysis (3)] |
| 13. | Zhang L, Xu J, Zhou S, Yao F, Zhang R, You W, Dai J, Yu K, Zhang Y, Baheti T, Pu L, Xu J, Qian X, Zhang C, Xia Y, Dai X, Li Q, Wang X. Endothelial DGKG promotes tumor angiogenesis and immune evasion in hepatocellular carcinoma. J Hepatol. 2024;80:82-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 116] [Article Influence: 58.0] [Reference Citation Analysis (2)] |
| 14. | Cha JH, Chan LC, Song MS, Hung MC. New Approaches on Cancer Immunotherapy. Cold Spring Harb Perspect Med. 2020;10:a036863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 96] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 15. | Yang Y. Cancer immunotherapy: harnessing the immune system to battle cancer. J Clin Invest. 2015;125:3335-3337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1113] [Cited by in RCA: 1006] [Article Influence: 91.5] [Reference Citation Analysis (4)] |
| 16. | Zhang Y, Zhang Z. The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol. 2020;17:807-821. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2469] [Cited by in RCA: 2129] [Article Influence: 354.8] [Reference Citation Analysis (5)] |
| 17. | Furukawa K, Nagano T, Tachihara M, Yamamoto M, Nishimura Y. Interaction between Immunotherapy and Antiangiogenic Therapy for Cancer. Molecules. 2020;25:3900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 18. | Kang Y, Kang Y, Zhang D, Yao J. Antiangiogenic therapy exerts antitumor effects by altering the tumor microenvironment: bibliometric analysis. Front Immunol. 2024;15:1460533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 19. | Padda SK, Reckamp KL. Combination of Immunotherapy and Antiangiogenic Therapy in Cancer-a Rational Approach. J Thorac Oncol. 2021;16:178-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Liu Z, Zhao Q, Zheng Z, Liu S, Meng L, Dong L, Jiang X. Vascular normalization in immunotherapy: A promising mechanisms combined with radiotherapy. Biomed Pharmacother. 2021;139:111607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (5)] |
| 21. | Yang T, Xiao H, Liu X, Wang Z, Zhang Q, Wei N, Guo X. Vascular Normalization: A New Window Opened for Cancer Therapies. Front Oncol. 2021;11:719836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 22. | García-Caballero M, Sokol L, Cuypers A, Carmeliet P. Metabolic Reprogramming in Tumor Endothelial Cells. Int J Mol Sci. 2022;23:11052. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 53] [Reference Citation Analysis (0)] |
| 23. | Rohlenova K, Goveia J, García-Caballero M, Subramanian A, Kalucka J, Treps L, Falkenberg KD, de Rooij LPMH, Zheng Y, Lin L, Sokol L, Teuwen LA, Geldhof V, Taverna F, Pircher A, Conradi LC, Khan S, Stegen S, Panovska D, De Smet F, Staal FJT, Mclaughlin RJ, Vinckier S, Van Bergen T, Ectors N, De Haes P, Wang J, Bolund L, Schoonjans L, Karakach TK, Yang H, Carmeliet G, Liu Y, Thienpont B, Dewerchin M, Eelen G, Li X, Luo Y, Carmeliet P. Single-Cell RNA Sequencing Maps Endothelial Metabolic Plasticity in Pathological Angiogenesis. Cell Metab. 2020;31:862-877.e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 232] [Article Influence: 38.7] [Reference Citation Analysis (3)] |
| 24. | Cantelmo AR, Conradi LC, Brajic A, Goveia J, Kalucka J, Pircher A, Chaturvedi P, Hol J, Thienpont B, Teuwen LA, Schoors S, Boeckx B, Vriens J, Kuchnio A, Veys K, Cruys B, Finotto L, Treps L, Stav-Noraas TE, Bifari F, Stapor P, Decimo I, Kampen K, De Bock K, Haraldsen G, Schoonjans L, Rabelink T, Eelen G, Ghesquière B, Rehman J, Lambrechts D, Malik AB, Dewerchin M, Carmeliet P. Inhibition of the Glycolytic Activator PFKFB3 in Endothelium Induces Tumor Vessel Normalization, Impairs Metastasis, and Improves Chemotherapy. Cancer Cell. 2016;30:968-985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 527] [Article Influence: 52.7] [Reference Citation Analysis (4)] |
| 25. | Xu S, Liao J, Liu B, Zhang C, Xu X. Aerobic glycolysis of vascular endothelial cells: a novel perspective in cancer therapy. Mol Biol Rep. 2024;51:717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 26. | Horváthová J, Moravčík R, Matúšková M, Šišovský V, Boháč A, Zeman M. Inhibition of Glycolysis Suppresses Cell Proliferation and Tumor Progression In Vivo: Perspectives for Chronotherapy. Int J Mol Sci. 2021;22:4390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Liu XT, Huang Y, Liu D, Jiang YC, Zhao M, Chung LH, Han XD, Zhao Y, Chen J, Coleman P, Ting KK, Tran C, Su Y, Dennis CV, Bhatnagar A, Liu K, Don AS, Vadas MA, Gorrell MD, Zhang S, Murray M, Kavurma MM, McCaughan GW, Gamble JR, Qi Y. Targeting the SphK1/S1P/PFKFB3 axis suppresses hepatocellular carcinoma progression by disrupting glycolytic energy supply that drives tumor angiogenesis. J Transl Med. 2024;22:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 28. | Matsumoto K, Noda T, Kobayashi S, Sakano Y, Yokota Y, Iwagami Y, Yamada D, Tomimaru Y, Akita H, Gotoh K, Takeda Y, Tanemura M, Umeshita K, Doki Y, Eguchi H. Inhibition of glycolytic activator PFKFB3 suppresses tumor growth and induces tumor vessel normalization in hepatocellular carcinoma. Cancer Lett. 2021;500:29-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (3)] |
| 29. | Pi J, Liu J, Chang H, Chen X, Pan W, Zhang Q, Zhuang T, Liu J, Wang H, Tomlinson B, Chan P, Cheng Y, Yu Z, Zhang L, Zhao Z, Liu Z, Liu J, Zhang Y. Therapeutic efficacy of ECs Foxp1 targeting Hif1α-Hk2 glycolysis signal to restrict angiogenesis. Redox Biol. 2024;75:103281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 30. | De Bock K, Georgiadou M, Carmeliet P. Role of endothelial cell metabolism in vessel sprouting. Cell Metab. 2013;18:634-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 322] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 31. | Yu P, Wilhelm K, Dubrac A, Tung JK, Alves TC, Fang JS, Xie Y, Zhu J, Chen Z, De Smet F, Zhang J, Jin SW, Sun L, Sun H, Kibbey RG, Hirschi KK, Hay N, Carmeliet P, Chittenden TW, Eichmann A, Potente M, Simons M. FGF-dependent metabolic control of vascular development. Nature. 2017;545:224-228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 258] [Cited by in RCA: 302] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 32. | Eijkelenboom A, Burgering BM. FOXOs: signalling integrators for homeostasis maintenance. Nat Rev Mol Cell Biol. 2013;14:83-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 968] [Cited by in RCA: 890] [Article Influence: 68.5] [Reference Citation Analysis (3)] |
| 33. | Miyamura Y, Kamei S, Matsuo M, Yamazaki M, Usuki S, Yasunaga K, Uemura A, Satou Y, Ohguchi H, Minami T. FOXO1 stimulates tip cell-enriched gene expression in endothelial cells. iScience. 2024;27:109161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 34. | Teuwen LA, Draoui N, Dubois C, Carmeliet P. Endothelial cell metabolism: an update anno 2017. Curr Opin Hematol. 2017;24:240-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 35. | Sun D, Chen S, Li S, Wang N, Zhang S, Xu L, Zhu S, Li H, Gu Q, Xu X, Wei F. Enhancement of glycolysis-dependent DNA repair regulated by FOXO1 knockdown via PFKFB3 attenuates hyperglycemia-induced endothelial oxidative stress injury. Redox Biol. 2023;59:102589. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 49] [Reference Citation Analysis (0)] |
| 36. | Liu Z, Du D, Zhang S. Tumor-derived exosomal miR-1247-3p promotes angiogenesis in bladder cancer by targeting FOXO1. Cancer Biol Ther. 2024;25:2290033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 37. | Shan Y, Ni Q, Zhang Q, Zhang M, Wei B, Cheng L, Zhong C, Wang X, Wang Q, Liu J, Zhang J, Wu J, Wang G, Zhou F. Targeting tumor endothelial hyperglycolysis enhances immunotherapy through remodeling tumor microenvironment. Acta Pharm Sin B. 2022;12:1825-1839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (4)] |
| 38. | Li Z, Fan X, Gao F, Pan S, Ma X, Cheng H, Nakatsukasa H, Zhang W, Zhang D. Fructose metabolism and its roles in metabolic diseases, inflammatory diseases, and cancer. Mol Biomed. 2025;6:43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 39. | Cui Y, Tian J, Wang Z, Guo H, Zhang H, Wang Z, Liu H, Song W, Liu L, Tian R, Zuo X, Ren S, Niu R, Zhang F. Fructose-Induced mTORC1 Activation Promotes Pancreatic Cancer Progression through Inhibition of Autophagy. Cancer Res. 2023;83:4063-4079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 29] [Reference Citation Analysis (0)] |
| 40. | Bu P, Chen KY, Xiang K, Johnson C, Crown SB, Rakhilin N, Ai Y, Wang L, Xi R, Astapova I, Han Y, Li J, Barth BB, Lu M, Gao Z, Mines R, Zhang L, Herman M, Hsu D, Zhang GF, Shen X. Aldolase B-Mediated Fructose Metabolism Drives Metabolic Reprogramming of Colon Cancer Liver Metastasis. Cell Metab. 2018;27:1249-1262.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 241] [Article Influence: 30.1] [Reference Citation Analysis (4)] |
| 41. | Yan H, Wang Z, Teng D, Chen X, Zhu Z, Chen H, Wang W, Wei Z, Wu Z, Chai Q, Zhang F, Wang Y, Shu K, Li S, Shi G, Zhu M, Piao HL, Shen X, Bu P. Hexokinase 2 senses fructose in tumor-associated macrophages to promote colorectal cancer growth. Cell Metab. 2024;36:2449-2467.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 37] [Reference Citation Analysis (0)] |
| 42. | Fang JH, Chen JY, Zheng JL, Zeng HX, Chen JG, Wu CH, Cai JL, Wang ZY, Zhuang SM. Fructose Metabolism in Tumor Endothelial Cells Promotes Angiogenesis by Activating AMPK Signaling and Mitochondrial Respiration. Cancer Res. 2023;83:1249-1263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 40] [Reference Citation Analysis (3)] |
| 43. | Cui Y, Liu H, Wang Z, Zhang H, Tian J, Wang Z, Song W, Guo H, Liu L, Tian R, Zuo X, Ren S, Zhang F, Niu R. Fructose promotes angiogenesis by improving vascular endothelial cell function and upregulating VEGF expression in cancer cells. J Exp Clin Cancer Res. 2023;42:184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 44. | Coutelle O, Hornig-Do HT, Witt A, Andree M, Schiffmann LM, Piekarek M, Brinkmann K, Seeger JM, Liwschitz M, Miwa S, Hallek M, Krönke M, Trifunovic A, Eming SA, Wiesner RJ, Hacker UT, Kashkar H. Embelin inhibits endothelial mitochondrial respiration and impairs neoangiogenesis during tumor growth and wound healing. EMBO Mol Med. 2014;6:624-639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 45. | Schiffmann LM, Werthenbach JP, Heintges-Kleinhofer F, Seeger JM, Fritsch M, Günther SD, Willenborg S, Brodesser S, Lucas C, Jüngst C, Albert MC, Schorn F, Witt A, Moraes CT, Bruns CJ, Pasparakis M, Krönke M, Eming SA, Coutelle O, Kashkar H. Mitochondrial respiration controls neoangiogenesis during wound healing and tumour growth. Nat Commun. 2020;11:3653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 46. | Nuevo-Tapioles C, Santacatterina F, Stamatakis K, Núñez de Arenas C, Gómez de Cedrón M, Formentini L, Cuezva JM. Coordinate β-adrenergic inhibition of mitochondrial activity and angiogenesis arrest tumor growth. Nat Commun. 2020;11:3606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 47. | Li X, Kumar A, Carmeliet P. Metabolic Pathways Fueling the Endothelial Cell Drive. Annu Rev Physiol. 2019;81:483-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 128] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 48. | Harjes U, Kalucka J, Carmeliet P. Targeting fatty acid metabolism in cancer and endothelial cells. Crit Rev Oncol Hematol. 2016;97:15-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 49. | Schoors S, Bruning U, Missiaen R, Queiroz KC, Borgers G, Elia I, Zecchin A, Cantelmo AR, Christen S, Goveia J, Heggermont W, Goddé L, Vinckier S, Van Veldhoven PP, Eelen G, Schoonjans L, Gerhardt H, Dewerchin M, Baes M, De Bock K, Ghesquière B, Lunt SY, Fendt SM, Carmeliet P. Fatty acid carbon is essential for dNTP synthesis in endothelial cells. Nature. 2015;520:192-197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 554] [Article Influence: 50.4] [Reference Citation Analysis (3)] |
| 50. | Fhu CW, Ali A. Fatty Acid Synthase: An Emerging Target in Cancer. Molecules. 2020;25:3935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 300] [Article Influence: 50.0] [Reference Citation Analysis (1)] |
| 51. | Menendez JA, Lupu R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat Rev Cancer. 2007;7:763-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2468] [Cited by in RCA: 2289] [Article Influence: 120.5] [Reference Citation Analysis (3)] |
| 52. | Du Q, Liu P, Zhang C, Liu T, Wang W, Shang C, Wu J, Liao Y, Chen Y, Huang J, Tan H, Zhao Y, Xia M, Liu J, Yao S. FASN promotes lymph node metastasis in cervical cancer via cholesterol reprogramming and lymphangiogenesis. Cell Death Dis. 2022;13:488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 96] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 53. | Li C, Zhang L, Qiu Z, Deng W, Wang W. Key Molecules of Fatty Acid Metabolism in Gastric Cancer. Biomolecules. 2022;12:706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 77] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 54. | Menendez JA, Lupu R. Fatty acid synthase (FASN) as a therapeutic target in breast cancer. Expert Opin Ther Targets. 2017;21:1001-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 219] [Article Influence: 24.3] [Reference Citation Analysis (2)] |
| 55. | Cheng H, Sun Y, Yu X, Zhou D, Ding J, Wang S, Ma F. FASN promotes gallbladder cancer progression and reduces cancer cell sensitivity to gemcitabine through PI3K/AKT signaling. Drug Discov Ther. 2023;17:328-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (2)] |
| 56. | Bruning U, Morales-Rodriguez F, Kalucka J, Goveia J, Taverna F, Queiroz KCS, Dubois C, Cantelmo AR, Chen R, Loroch S, Timmerman E, Caixeta V, Bloch K, Conradi LC, Treps L, Staes A, Gevaert K, Tee A, Dewerchin M, Semenkovich CF, Impens F, Schilling B, Verdin E, Swinnen JV, Meier JL, Kulkarni RA, Sickmann A, Ghesquière B, Schoonjans L, Li X, Mazzone M, Carmeliet P. Impairment of Angiogenesis by Fatty Acid Synthase Inhibition Involves mTOR Malonylation. Cell Metab. 2018;28:866-880.e15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 217] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 57. | Bastos DC, Paupert J, Maillard C, Seguin F, Carvalho MA, Agostini M, Coletta RD, Noël A, Graner E. Effects of fatty acid synthase inhibitors on lymphatic vessels: an in vitro and in vivo study in a melanoma model. Lab Invest. 2017;97:194-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 58. | Altman BJ, Stine ZE, Dang CV. From Krebs to clinic: glutamine metabolism to cancer therapy. Nat Rev Cancer. 2016;16:619-634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1619] [Cited by in RCA: 1443] [Article Influence: 144.3] [Reference Citation Analysis (5)] |
| 59. | Cluntun AA, Lukey MJ, Cerione RA, Locasale JW. Glutamine Metabolism in Cancer: Understanding the Heterogeneity. Trends Cancer. 2017;3:169-180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 703] [Cited by in RCA: 627] [Article Influence: 69.7] [Reference Citation Analysis (0)] |
| 60. | Yang WH, Qiu Y, Stamatatos O, Janowitz T, Lukey MJ. Enhancing the Efficacy of Glutamine Metabolism Inhibitors in Cancer Therapy. Trends Cancer. 2021;7:790-804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 221] [Article Influence: 44.2] [Reference Citation Analysis (4)] |
| 61. | Jin J, Byun JK, Choi YK, Park KG. Targeting glutamine metabolism as a therapeutic strategy for cancer. Exp Mol Med. 2023;55:706-715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 397] [Reference Citation Analysis (0)] |
| 62. | Leighton B, Curi R, Hussein A, Newsholme EA. Maximum activities of some key enzymes of glycolysis, glutaminolysis, Krebs cycle and fatty acid utilization in bovine pulmonary endothelial cells. FEBS Lett. 1987;225:93-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 63. | Eelen G, de Zeeuw P, Simons M, Carmeliet P. Endothelial cell metabolism in normal and diseased vasculature. Circ Res. 2015;116:1231-1244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 515] [Article Influence: 46.8] [Reference Citation Analysis (4)] |
| 64. | Polet F, Feron O. Endothelial cell metabolism and tumour angiogenesis: glucose and glutamine as essential fuels and lactate as the driving force. J Intern Med. 2013;273:156-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 192] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 65. | Drogat B, Bouchecareilh M, North S, Petibois C, Déléris G, Chevet E, Bikfalvi A, Moenner M. Acute L-glutamine deprivation compromises VEGF-a upregulation in A549/8 human carcinoma cells. J Cell Physiol. 2007;212:463-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 66. | Schoonjans CA, Mathieu B, Joudiou N, Zampieri LX, Brusa D, Sonveaux P, Feron O, Gallez B. Targeting Endothelial Cell Metabolism by Inhibition of Pyruvate Dehydrogenase Kinase and Glutaminase-1. J Clin Med. 2020;9:3308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 67. | Ngwa VM, Edwards DN, Hwang Y, Karno B, Wang X, Yan C, Richmond A, Brantley-Sieders DM, Chen J. Loss of vascular endothelial glutaminase inhibits tumor growth and metastasis, and increases sensitivity to chemotherapy. Cancer Res Commun. 2022;2:694-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 68. | Oberkersch RE, Pontarin G, Astone M, Spizzotin M, Arslanbaeva L, Tosi G, Panieri E, Ricciardi S, Allega MF, Brossa A, Grumati P, Bussolati B, Biffo S, Tardito S, Santoro MM. Aspartate metabolism in endothelial cells activates the mTORC1 pathway to initiate translation during angiogenesis. Dev Cell. 2022;57:1241-1256.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 69. | Vandekeere S, Dubois C, Kalucka J, Sullivan MR, García-Caballero M, Goveia J, Chen R, Diehl FF, Bar-Lev L, Souffreau J, Pircher A, Kumar S, Vinckier S, Hirabayashi Y, Furuya S, Schoonjans L, Eelen G, Ghesquière B, Keshet E, Li X, Vander Heiden MG, Dewerchin M, Carmeliet P. Serine Synthesis via PHGDH Is Essential for Heme Production in Endothelial Cells. Cell Metab. 2018;28:573-587.e13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 157] [Article Influence: 19.6] [Reference Citation Analysis (3)] |
| 70. | Ocaña MC, Martínez-Poveda B, Quesada AR, Medina MÁ. Metabolism within the tumor microenvironment and its implication on cancer progression: An ongoing therapeutic target. Med Res Rev. 2019;39:70-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 68] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 71. | Lambrechts D, Wauters E, Boeckx B, Aibar S, Nittner D, Burton O, Bassez A, Decaluwé H, Pircher A, Van den Eynde K, Weynand B, Verbeken E, De Leyn P, Liston A, Vansteenkiste J, Carmeliet P, Aerts S, Thienpont B. Phenotype molding of stromal cells in the lung tumor microenvironment. Nat Med. 2018;24:1277-1289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 725] [Cited by in RCA: 1227] [Article Influence: 153.4] [Reference Citation Analysis (3)] |
| 72. | Hu Y, Lou X, Zhang K, Pan L, Bai Y, Wang L, Wang M, Yan Y, Wan J, Yao X, Duan X, Ni C, Qin Z. Tumor necrosis factor receptor 2 promotes endothelial cell-mediated suppression of CD8+ T cells through tuning glycolysis in chemoresistance of breast cancer. J Transl Med. 2024;22:672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 73. | Lu Y, Liu Y, Zuo X, Li G, Wang J, Liu J, Wang X, Wang S, Zhang W, Zhang K, Lei X, Hao Q, Li W, Liu L, Li M, Zhang C, Zhang HM, Zhang Y, Gao Y. CXCL12(+) tumor-associated endothelial cells promote immune resistance in hepatocellular carcinoma. J Hepatol. 2025;82:634-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 74. | Lim WC, Olding M, Healy E, Millar TM. Human Endothelial Cells Modulate CD4(+) T Cell Populations and Enhance Regulatory T Cell Suppressive Capacity. Front Immunol. 2018;9:565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 75. | Glasner A, Plitas G. Tumor resident regulatory T cells. Semin Immunol. 2021;52:101476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 76. | Ning H, Shao QQ, Ding KJ, Gao DX, Lu QL, Cao QW, Niu ZH, Fu Q, Zhang CH, Qu X, Lü JJ. Tumor-infiltrating regulatory T cells are positively correlated with angiogenic status in renal cell carcinoma. Chin Med J (Engl). 2012;125:2120-2125. [PubMed] |
| 77. | Miyazaki T, Taketomi Y, Higashi T, Ohtaki H, Takaki T, Ohnishi K, Hosonuma M, Kono N, Akasu R, Haraguchi S, Kim-Kaneyama JR, Otsu K, Arai H, Murakami M, Miyazaki A. Hypercholesterolemic Dysregulation of Calpain in Lymphatic Endothelial Cells Interferes With Regulatory T-Cell Stability and Trafficking. Arterioscler Thromb Vasc Biol. 2023;43:e66-e82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 78. | Baradaran A, Asadzadeh Z, Hemmat N, Baghbanzadeh A, Shadbad MA, Khosravi N, Derakhshani A, Alemohammad H, Afrashteh Nour M, Safarpour H, Silvestris N, Brunetti O, Baradaran B. The cross-talk between tumor-associated macrophages and tumor endothelium: Recent advances in macrophage-based cancer immunotherapy. Biomed Pharmacother. 2022;146:112588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 79. | Zhang W, Wang M, Ji C, Liu X, Gu B, Dong T. Macrophage polarization in the tumor microenvironment: Emerging roles and therapeutic potentials. Biomed Pharmacother. 2024;177:116930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 114] [Reference Citation Analysis (0)] |
| 80. | Pyonteck SM, Gadea BB, Wang HW, Gocheva V, Hunter KE, Tang LH, Joyce JA. Deficiency of the macrophage growth factor CSF-1 disrupts pancreatic neuroendocrine tumor development. Oncogene. 2012;31:1459-1467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 101] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 81. | Krug S, Abbassi R, Griesmann H, Sipos B, Wiese D, Rexin P, Blank A, Perren A, Haybaeck J, Hüttelmaier S, Rinke A, Gress TM, Michl P. Therapeutic targeting of tumor-associated macrophages in pancreatic neuroendocrine tumors. Int J Cancer. 2018;143:1806-1816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 82. | Rahma OE, Hodi FS. The Intersection between Tumor Angiogenesis and Immune Suppression. Clin Cancer Res. 2019;25:5449-5457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 424] [Cited by in RCA: 376] [Article Influence: 53.7] [Reference Citation Analysis (3)] |
| 83. | Haston S, Gonzalez-Gualda E, Morsli S, Ge J, Reen V, Calderwood A, Moutsopoulos I, Panousopoulos L, Deletic P, Carreno G, Guiho R, Manshaei S, Gonzalez-Meljem JM, Lim HY, Simpson DJ, Birch J, Pallikonda HA, Chandra T, Macias D, Doherty GJ, Rassl DM, Rintoul RC, Signore M, Mohorianu I, Akbar AN, Gil J, Muñoz-Espín D, Martinez-Barbera JP. Clearance of senescent macrophages ameliorates tumorigenesis in KRAS-driven lung cancer. Cancer Cell. 2023;41:1242-1260.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 175] [Article Influence: 58.3] [Reference Citation Analysis (0)] |
| 84. | Do MH, Shi W, Ji L, Ladewig E, Zhang X, Srivastava RM, Capistrano KJ, Edwards C, Malik I, Nixon BG, Stamatiades EG, Liu M, Li S, Li P, Chou C, Xu K, Hsu TW, Wang X, Chan TA, Leslie CS, Li MO. Reprogramming tumor-associated macrophages to outcompete endovascular endothelial progenitor cells and suppress tumor neoangiogenesis. Immunity. 2023;56:2555-2569.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 85. | Kim SW, Kim CW, Moon YA, Kim HS. Reprogramming of tumor-associated macrophages by metabolites generated from tumor microenvironment. Anim Cells Syst (Seoul). 2024;28:123-136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 86. | Ghesquière B, Wong BW, Kuchnio A, Carmeliet P. Metabolism of stromal and immune cells in health and disease. Nature. 2014;511:167-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 373] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 87. | Alsina-Sanchis E, Mülfarth R, Moll I, Böhn S, Wiedmann L, Jordana-Urriza L, Ziegelbauer T, Zimmer E, Taylor J, De Angelis Rigotti F, Stögbauer A, Giaimo BD, Cerwenka A, Borggrefe T, Fischer A, Rodriguez-Vita J. Endothelial RBPJ Is Essential for the Education of Tumor-Associated Macrophages. Cancer Res. 2022;82:4414-4428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 88. | Yu G, Corn PG, Mak CSL, Liang X, Zhang M, Troncoso P, Song JH, Lin SC, Song X, Liu J, Zhang J, Logothetis CJ, Melancon MP, Panaretakis T, Wang G, Lin SH. Prostate cancer-induced endothelial-cell-to-osteoblast transition drives immunosuppression in the bone-tumor microenvironment through Wnt pathway-induced M2 macrophage polarization. Proc Natl Acad Sci U S A. 2024;121:e2402903121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 89. | Kuo FC, Tsai HY, Cheng BL, Tsai KJ, Chen PC, Huang YB, Liu CJ, Wu DC, Wu MC, Huang B, Lin MW. Endothelial Mitochondria Transfer to Melanoma Induces M2-Type Macrophage Polarization and Promotes Tumor Growth by the Nrf2/HO-1-Mediated Pathway. Int J Mol Sci. 2024;25:1857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 90. | Segal BH, Giridharan T, Suzuki S, Khan ANH, Zsiros E, Emmons TR, Yaffe MB, Gankema AAF, Hoogeboom M, Goetschalckx I, Matlung HL, Kuijpers TW. Neutrophil interactions with T cells, platelets, endothelial cells, and of course tumor cells. Immunol Rev. 2023;314:13-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 52] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 91. | Fridlender ZG, Sun J, Kim S, Kapoor V, Cheng G, Ling L, Worthen GS, Albelda SM. Polarization of tumor-associated neutrophil phenotype by TGF-beta: "N1" versus "N2" TAN. Cancer Cell. 2009;16:183-194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2930] [Cited by in RCA: 2715] [Article Influence: 159.7] [Reference Citation Analysis (5)] |
| 92. | Antuamwine BB, Bosnjakovic R, Hofmann-Vega F, Wang X, Theodosiou T, Iliopoulos I, Brandau S. N1 versus N2 and PMN-MDSC: A critical appraisal of current concepts on tumor-associated neutrophils and new directions for human oncology. Immunol Rev. 2023;314:250-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 108] [Reference Citation Analysis (3)] |
| 93. | Jiang H, Gao B, Meng Z, Wang Y, Jiao T, Li J, Li X, Cao Y, Zhang X, Li C, Lu S. Integrative multi-omics analysis reveals the role of tumor-associated endothelial cells and their signature in prognosis of intrahepatic cholangiocarcinoma. J Transl Med. 2024;22:948. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 94. | Tsumita T, Maishi N, Annan DA, Towfik MA, Matsuda A, Onodera Y, Nam JM, Hida Y, Hida K. The oxidized-LDL/LOX-1 axis in tumor endothelial cells enhances metastasis by recruiting neutrophils and cancer cells. Int J Cancer. 2022;151:944-956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 95. | Vetsika EK, Koukos A, Kotsakis A. Myeloid-Derived Suppressor Cells: Major Figures that Shape the Immunosuppressive and Angiogenic Network in Cancer. Cells. 2019;8:1647. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 98] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 96. | He Q, Jamalpour M, Bergquist E, Anderson RL, Gustafsson K, Welsh M. Mouse Breast Carcinoma Monocytic/Macrophagic Myeloid-Derived Suppressor Cell Infiltration as a Consequence of Endothelial Dysfunction in Shb-Deficient Endothelial Cells Increases Tumor Lung Metastasis. Int J Mol Sci. 2021;22:11478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 97. | Marech I, Leporini C, Ammendola M, Porcelli M, Gadaleta CD, Russo E, De Sarro G, Ranieri G. Classical and non-classical proangiogenic factors as a target of antiangiogenic therapy in tumor microenvironment. Cancer Lett. 2016;380:216-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 98. | Huang M, Lin Y, Wang C, Deng L, Chen M, Assaraf YG, Chen ZS, Ye W, Zhang D. New insights into antiangiogenic therapy resistance in cancer: Mechanisms and therapeutic aspects. Drug Resist Updat. 2022;64:100849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 126] [Reference Citation Analysis (3)] |
| 99. | Cannell IG, Sawicka K, Pearsall I, Wild SA, Deighton L, Pearsall SM, Lerda G, Joud F, Khan S, Bruna A, Simpson KL, Mulvey CM, Nugent F, Qosaj F, Bressan D; CRUK IMAXT Grand Challenge Team, Dive C, Caldas C, Hannon GJ. FOXC2 promotes vasculogenic mimicry and resistance to anti-angiogenic therapy. Cell Rep. 2023;42:112791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 100. | Hu C, Hui K, Jiang X. Effects of microRNA regulation on antiangiogenic therapy resistance in non-small cell lung cancer. Biomed Pharmacother. 2020;131:110557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 101. | Wen Q, Huang M, Xie J, Liu R, Miao Q, Huang J, Zhang J, Lyu W, Qi M, Wu C, Qi Q, Zhang Z, Deng R, Wang C, Chen ZS, Zhang D, Ye W, Chen M. lncRNA SYTL5-OT4 promotes vessel co-option by inhibiting the autophagic degradation of ASCT2. Drug Resist Updat. 2023;69:100975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 102. | Liu J, Zhang Q, Yang D, Xie F, Wang Z. The role of long non-coding RNAs in angiogenesis and anti-angiogenic therapy resistance in cancer. Mol Ther Nucleic Acids. 2022;28:397-407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 103. | Jiménez-Valerio G, Casanovas O. Antiangiogenic resistance via metabolic symbiosis. Mol Cell Oncol. 2016;3:e1211979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 104. | Jiménez-Valerio G, Casanovas O. Angiogenesis and Metabolism: Entwined for Therapy Resistance. Trends Cancer. 2017;3:10-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 105. | Goel S, Duda DG, Xu L, Munn LL, Boucher Y, Fukumura D, Jain RK. Normalization of the vasculature for treatment of cancer and other diseases. Physiol Rev. 2011;91:1071-1121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1342] [Cited by in RCA: 1248] [Article Influence: 83.2] [Reference Citation Analysis (4)] |
| 106. | Martin JD, Seano G, Jain RK. Normalizing Function of Tumor Vessels: Progress, Opportunities, and Challenges. Annu Rev Physiol. 2019;81:505-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 403] [Article Influence: 67.2] [Reference Citation Analysis (3)] |
| 107. | Zheng R, Li F, Li F, Gong A. Targeting tumor vascularization: promising strategies for vascular normalization. J Cancer Res Clin Oncol. 2021;147:2489-2505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 108. | Viallard C, Larrivée B. Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis. 2017;20:409-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1201] [Cited by in RCA: 1087] [Article Influence: 120.8] [Reference Citation Analysis (4)] |
| 109. | Jászai J, Schmidt MHH. Trends and Challenges in Tumor Anti-Angiogenic Therapies. Cells. 2019;8:1102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 158] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 110. | Cantelmo AR, Pircher A, Kalucka J, Carmeliet P. Vessel pruning or healing: endothelial metabolism as a novel target? Expert Opin Ther Targets. 2017;21:239-247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 111. | Stapor P, Wang X, Goveia J, Moens S, Carmeliet P. Angiogenesis revisited - role and therapeutic potential of targeting endothelial metabolism. J Cell Sci. 2014;127:4331-4341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 112. | Mohammadi P, Yarani R, Rahimpour A, Ranjbarnejad F, Mendes Lopes de Melo J, Mansouri K. Targeting endothelial cell metabolism in cancerous microenvironment: a new approach for anti-angiogenic therapy. Drug Metab Rev. 2022;54:386-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 113. | Wu Y, Xie BB, Zhang BL, Zhuang QX, Liu SW, Pan HM. Apatinib regulates the glycolysis of vascular endothelial cells through PI3K/AKT/PFKFB3 pathway in hepatocellular carcinoma. World J Gastroenterol. 2025;31:102848. [PubMed] [DOI] [Full Text] |
| 114. | Schoors S, De Bock K, Cantelmo AR, Georgiadou M, Ghesquière B, Cauwenberghs S, Kuchnio A, Wong BW, Quaegebeur A, Goveia J, Bifari F, Wang X, Blanco R, Tembuyser B, Cornelissen I, Bouché A, Vinckier S, Diaz-Moralli S, Gerhardt H, Telang S, Cascante M, Chesney J, Dewerchin M, Carmeliet P. Partial and transient reduction of glycolysis by PFKFB3 blockade reduces pathological angiogenesis. Cell Metab. 2014;19:37-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 434] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 115. | Qian C, Zhou Y, Zhang T, Dong G, Song M, Tang Y, Wei Z, Yu S, Shen Q, Chen W, Choi JP, Yan J, Zhong C, Wan L, Li J, Wang A, Lu Y, Zhao Y. Targeting PKM2 signaling cascade with salvianic acid A normalizes tumor blood vessels to facilitate chemotherapeutic drug delivery. Acta Pharm Sin B. 2024;14:2077-2096. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 35] [Reference Citation Analysis (0)] |
| 116. | Wan YX, Qi XW, Lian YY, Liu ZY, Wang H, Qiu YQ, Zhang CG, Li WN, Jiang HL, Yang DH, Zhao W, Chen ZS, Huang JC. Electroacupuncture facilitates vascular normalization by inhibiting Glyoxalase1 in endothelial cells to attenuate glycolysis and angiogenesis in triple-negative breast cancer. Cancer Lett. 2024;598:217094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 117. | Wu W, Wang X, Liao L, Chen J, Wang Y, Yao M, Zhu L, Li J, Wang X, Chen AF, Zhang G, Zhang Z, Bai Y. The TRPM7 channel reprograms cellular glycolysis to drive tumorigenesis and angiogenesis. Cell Death Dis. 2023;14:183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (1)] |
| 118. | Seguin F, Carvalho MA, Bastos DC, Agostini M, Zecchin KG, Alvarez-Flores MP, Chudzinski-Tavassi AM, Coletta RD, Graner E. The fatty acid synthase inhibitor orlistat reduces experimental metastases and angiogenesis in B16-F10 melanomas. Br J Cancer. 2012;107:977-987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 124] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 119. | Wong BW, Wang X, Zecchin A, Thienpont B, Cornelissen I, Kalucka J, García-Caballero M, Missiaen R, Huang H, Brüning U, Blacher S, Vinckier S, Goveia J, Knobloch M, Zhao H, Dierkes C, Shi C, Hägerling R, Moral-Dardé V, Wyns S, Lippens M, Jessberger S, Fendt SM, Luttun A, Noel A, Kiefer F, Ghesquière B, Moons L, Schoonjans L, Dewerchin M, Eelen G, Lambrechts D, Carmeliet P. The role of fatty acid β-oxidation in lymphangiogenesis. Nature. 2017;542:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 278] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 120. | Peyton KJ, Liu XM, Yu Y, Yates B, Behnammanesh G, Durante W. Glutaminase-1 stimulates the proliferation, migration, and survival of human endothelial cells. Biochem Pharmacol. 2018;156:204-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 121. | Unterluggauer H, Mazurek S, Lener B, Hütter E, Eigenbrodt E, Zwerschke W, Jansen-Dürr P. Premature senescence of human endothelial cells induced by inhibition of glutaminase. Biogerontology. 2008;9:247-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 96] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 122. | Feng Y, Wang L, Dong C, Yang X, Wang J, Zhang X, Yuan Y, Dai J, Huang J, Yuan F. MicroRNA-376b-3p Suppresses Choroidal Neovascularization by Regulating Glutaminolysis in Endothelial Cells. Invest Ophthalmol Vis Sci. 2023;64:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 123. | Huang H, Vandekeere S, Kalucka J, Bierhansl L, Zecchin A, Brüning U, Visnagri A, Yuldasheva N, Goveia J, Cruys B, Brepoels K, Wyns S, Rayport S, Ghesquière B, Vinckier S, Schoonjans L, Cubbon R, Dewerchin M, Eelen G, Carmeliet P. Role of glutamine and interlinked asparagine metabolism in vessel formation. EMBO J. 2017;36:2334-2352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 263] [Article Influence: 29.2] [Reference Citation Analysis (3)] |
| 124. | Palmieri EM, Menga A, Martín-Pérez R, Quinto A, Riera-Domingo C, De Tullio G, Hooper DC, Lamers WH, Ghesquière B, McVicar DW, Guarini A, Mazzone M, Castegna A. Pharmacologic or Genetic Targeting of Glutamine Synthetase Skews Macrophages toward an M1-like Phenotype and Inhibits Tumor Metastasis. Cell Rep. 2017;20:1654-1666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 327] [Article Influence: 40.9] [Reference Citation Analysis (10)] |
| 125. | Melo V, Bremer E, Martin JD. Towards Immunotherapy-Induced Normalization of the Tumor Microenvironment. Front Cell Dev Biol. 2022;10:908389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 126. | Li S, Zhang Q, Hong Y. Tumor Vessel Normalization: A Window to Enhancing Cancer Immunotherapy. Technol Cancer Res Treat. 2020;19:1533033820980116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 127. | Guo D, Tong Y, Jiang X, Meng Y, Jiang H, Du L, Wu Q, Li S, Luo S, Li M, Xiao L, He H, He X, Yu Q, Fang J, Lu Z. Aerobic glycolysis promotes tumor immune evasion by hexokinase2-mediated phosphorylation of IκBα. Cell Metab. 2022;34:1312-1324.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 308] [Article Influence: 77.0] [Reference Citation Analysis (0)] |
| 128. | Taguchi K, Onoe T, Yoshida T, Yamashita Y, Tanaka Y, Ohdan H. Tumor Endothelial Cell-Mediated Antigen-Specific T-cell Suppression via the PD-1/PD-L1 Pathway. Mol Cancer Res. 2020;18:1427-1440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 129. | Zhang D, Li AM, Hu G, Huang M, Yang F, Zhang L, Wellen KE, Xu X, Conn CS, Zou W, Kahn M, Rhoades SD, Weljie AM, Fuchs SY, Amankulor N, Yoshor D, Ye J, Koumenis C, Gong Y, Fan Y. PHGDH-mediated endothelial metabolism drives glioblastoma resistance to chimeric antigen receptor T cell immunotherapy. Cell Metab. 2023;35:517-534.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 104] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 130. | Missiaen R, Morales-Rodriguez F, Eelen G, Carmeliet P. Targeting endothelial metabolism for anti-angiogenesis therapy: A pharmacological perspective. Vascul Pharmacol. 2017;90:8-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 131. | Huang N, Ren J, Deng X, Bao Q, Huang G, Zhi S, Li Y, Li J, Hu B, Zeng D, Sun H, Zeng W, Shi M, Liao W, Wu J, Huang N. Endothelial F3-mediated autolysosome and lipid metabolism promote resistance to anti-VEGFA therapy in metastatic colorectal cancer. Autophagy. 2025;21:3004-3023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 132. | Liu L, Mo M, Chen X, Chao D, Zhang Y, Chen X, Wang Y, Zhang N, He N, Yuan X, Chen H, Yang J. Targeting inhibition of prognosis-related lipid metabolism genes including CYP19A1 enhances immunotherapeutic response in colon cancer. J Exp Clin Cancer Res. 2023;42:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 67] [Reference Citation Analysis (0)] |