Copyright: ©Author(s) 2026.
World J Clin Oncol. Mar 24, 2026; 17(3): 113326
Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.113326
Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.113326
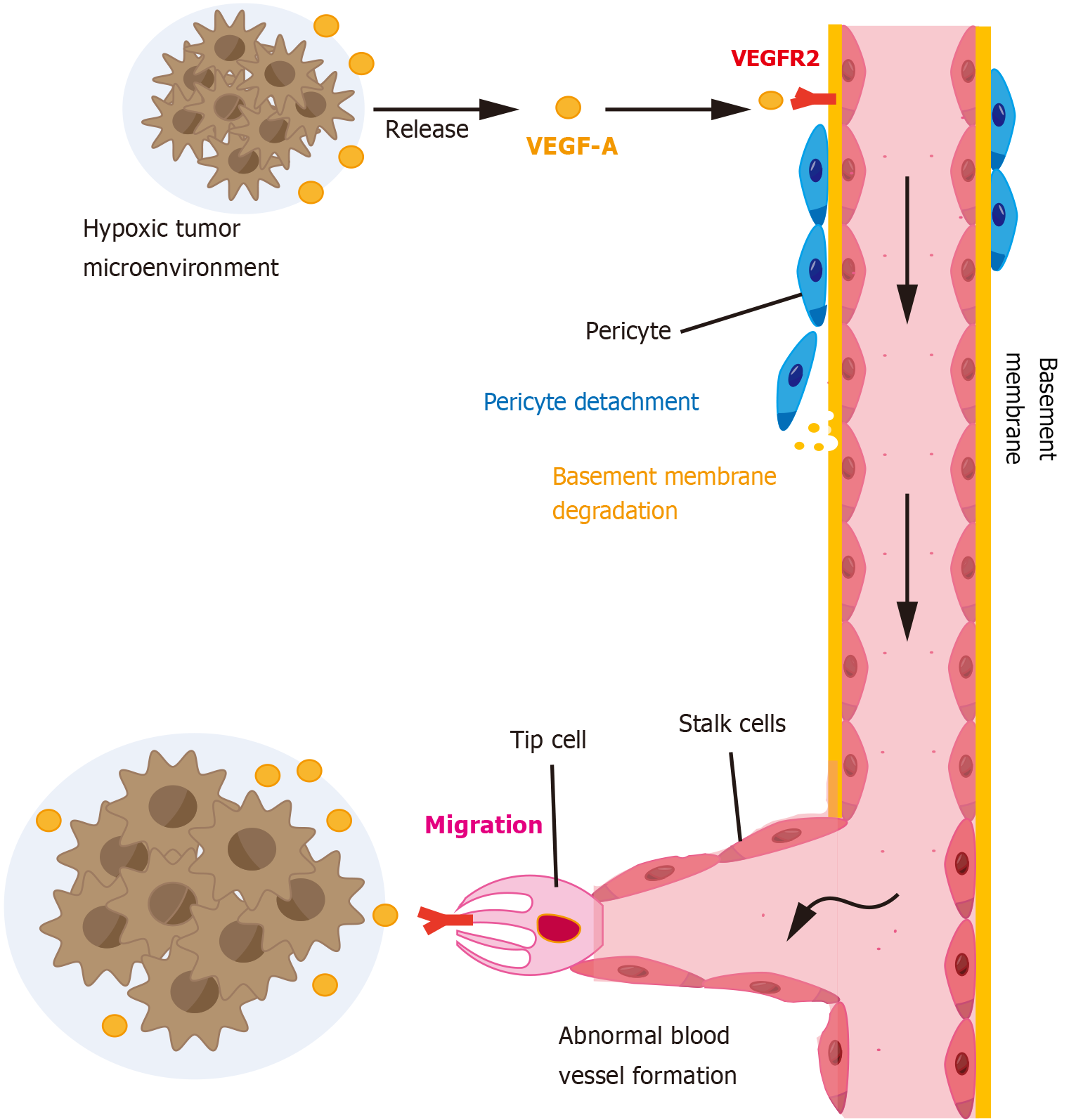
Figure 1 Angiogenic processes in the tumor microenvironment.
The hypoxic tumor microenvironment releases vascular endothelial growth factor (VEGF). When VEGF binds to VEGF receptors on quiescent endothelial cells, it activates the endothelial cells and initiates the angiogenic program. At the same time, pericytes detach and the basement membrane degrades. During angiogenesis, tip cell migrates toward the tumor and stalk cells proliferate to lengthen the vessels. Finally, structurally and functionally abnormal tumor vasculature is formed. VEGF-A: Vascular endothelial growth factor A; VEGFR2: Vascular endothelial growth factor receptor 2.
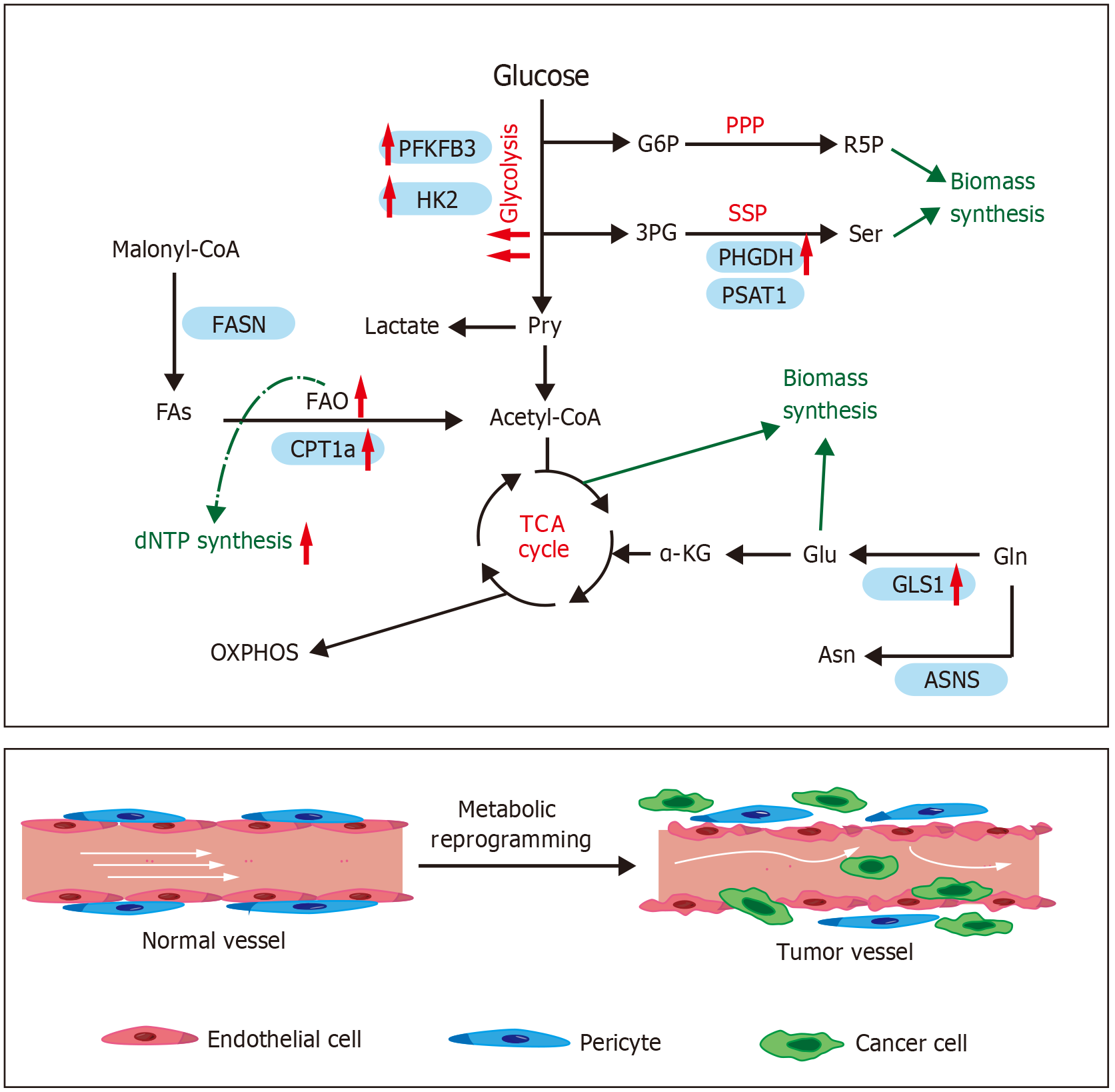
Figure 2 Metabolism of tumor endothelial cells.
Tumor endothelial cell (TEC) exhibits higher levels of glycolysis than normal endothelial cell. 6-phosphofructo-2-kinase/fructose-2,6-bisphosph-atase 3, an activator of glycolysis, is overexpressed in TEC. Endothelial cells transport fatty acids into mitochondria via carnitine palmitoyltransferase 1A. In proliferating endothelial cells, carnitine palmitoyltransferase 1A is the rate-limiting enzyme for fatty acid oxidation and fatty acid oxidation provides a significant amount of substrate for deoxy nucleotide triphosphate synthesis. Enhanced glutamine metabolism in TEC provides amino acid substrates for nucleotide and biomass synthesis. Additionally, serine can be synthesized directly from 3-phosphoglycerate, which is produced by glycolysis, via serine synthesis pathway. Compared to normal blood vessels, tumor blood vessels have a disrupted barrier and distorted structure. They are more likely to develop hypoxia and metabolic abnormalities, which promote the dissemination of tumor cells. TEC: Tumor endothelial cell; PFKFB3: 6-phosphofructo-2-kinase/fructose-2,6-bisphosph-atase 3; HK2: Hexokinase 2; PPP: Pentose phosphate pathway; SSP: Serine synthesis pathway; PHGDH: Phosphoglycerate dehydrogenase; PSAT1: Phosphoserine aminotransferase 1; FASN: Fatty acid synthase; FAs: Fatty acids; FAO: Fatty acid oxidation; CPT1a: Carnitine palmitoyltransferase 1a; dNTP: Deoxy nucleotide triphosphate; TCA: Tricarboxylic acid; α-KG: Α-ketoglutarate; Glu: Glutamate; Gln: Glutamine; GLS1: Glutaminase 1; OXPHOS: Oxidative phosphorylation; ASNS: Asparagine synthetase.
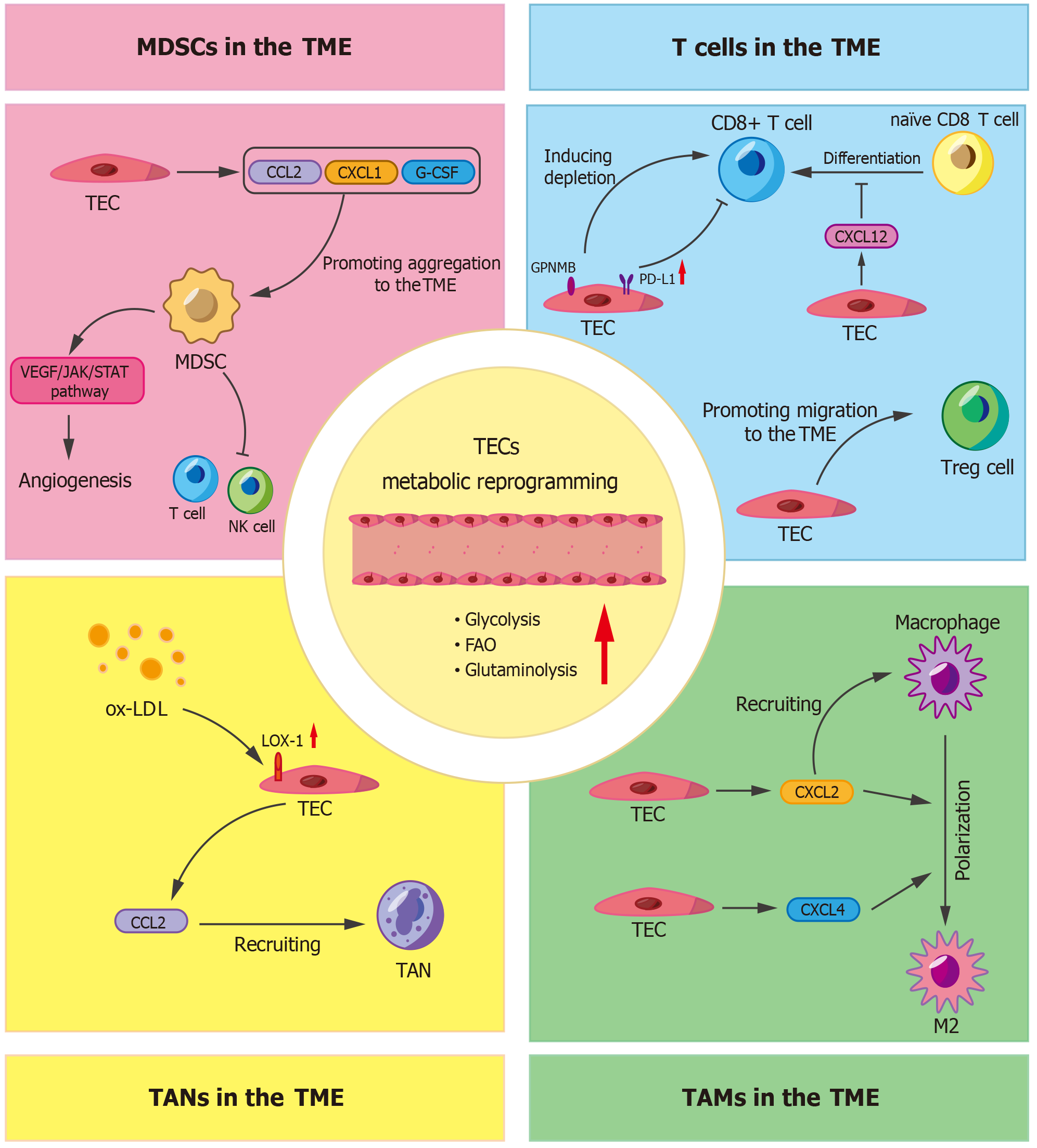
Figure 3 Endothelial metabolic reprogramming alters the tumor immune microenvironment.
In the tumor microenvironment (TME), endothelial cells undergo metabolic reprogramming, including enhanced glycolysis, fatty acid oxidation and glutaminolysis. These alterations will not only affect the proliferation and migration of endothelial cells, but also have an impact on immune cells in the TME. The expression of programmed death-ligand 1 is increased on tumor endothelial cell (TEC), which in turn induces CD8+ T cell suppression. TECs induce the exhaustion of CD8+ T cells via glycoprotein nonmetastatic melanoma protein B. Moreover, they secrete C-X-C motif ligand 12 (CXCL12) to impede the differentiation of naïve CD8 T cells into cytotoxic T cells. TECs can also recruit regulatory T cells, tumor-associated macrophages, tumor-associated neutrophils and myeloid-derived suppressor cells to the TME by secreting factors including C-C motif ligand 2, CXCL1, CXCL2 and granulocyte colony-stimulating factor. This process contributes to the establishment of an immunosuppressive state. Furthermore, TECs secrete CXCL2 and CXCL4, which drive the polarization of macrophages towards the M2 phenotype. The accumulation of immunosuppressive cells within the TME leads to the further secretion of pro-angiogenic factors, thereby inducing abnormal angiogenesis. The structurally and functionally dysregulated blood vessels exacerbate the hypoxic condition of the TME, which in turn further promotes the metabolic reprogramming of TECs. MDSCs: Myeloid-derived suppressor cells; TME: Tumor microenvironment; TEC: Tumor endothelial cell; CCL2: C-C motif ligand 2; CXCL1: C-X-C motif ligand 1; G-CSF: Granulocyte colony-stimulating factor; VEGF: Vascular endothelial growth factor; JAK: Janus kinase; STAT: Signal transducer and activator of the transcription; NK: Natural killer; GPNMB: Glycoprotein non-metastatic melanoma protein B; PD-L1: Programmed death-ligand 1; Treg: Regulatory T; FAO: Fatty acid oxidation; ox-LDL: Oxidized low-density lipoprotein; LOX-1: Lectin-like oxidized low-density lipoprotein receptor-1; TAN: Tumor-associated neutrophil.
- Citation: Chen M, Chen ZG. Endothelial metabolic reprogramming influences tumor immune microenvironment. World J Clin Oncol 2026; 17(3): 113326
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/113326.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.113326