Published online Feb 28, 2026. doi: 10.4329/wjr.v18.i2.116799
Revised: December 25, 2025
Accepted: January 9, 2026
Published online: February 28, 2026
Processing time: 96 Days and 3.1 Hours
Cognitive impairment is a common functional impairment after stroke that severely affects the quality of life of patients. The underlying neurobiological mechanisms of poststroke cognitive impairment (PSCI) remain unclear.
To investigate the changes in functional connectivity (FC) in the brains of patients with PSCI.
A total of 21 patients with PSCI and 12 healthy controls were selected as study subjects, and resting-state functional magnetic resonance imaging was performed. The brain region [Cerebellum_6_R (aal)] with significant differences identified by regional homogeneity analysis and the left thalamus, right thalamus, left basal ganglia, and right basal ganglia in the Brainnetome Atlas were selected as the seeds (regions of interest), and the FC between the seeds and whole-brain voxels was analyzed. Moreover, the 116 brain regions defined in the AAL116 atlas were selected as seeds (regions of interest), and the FC between the whole-brain seeds was calculated.
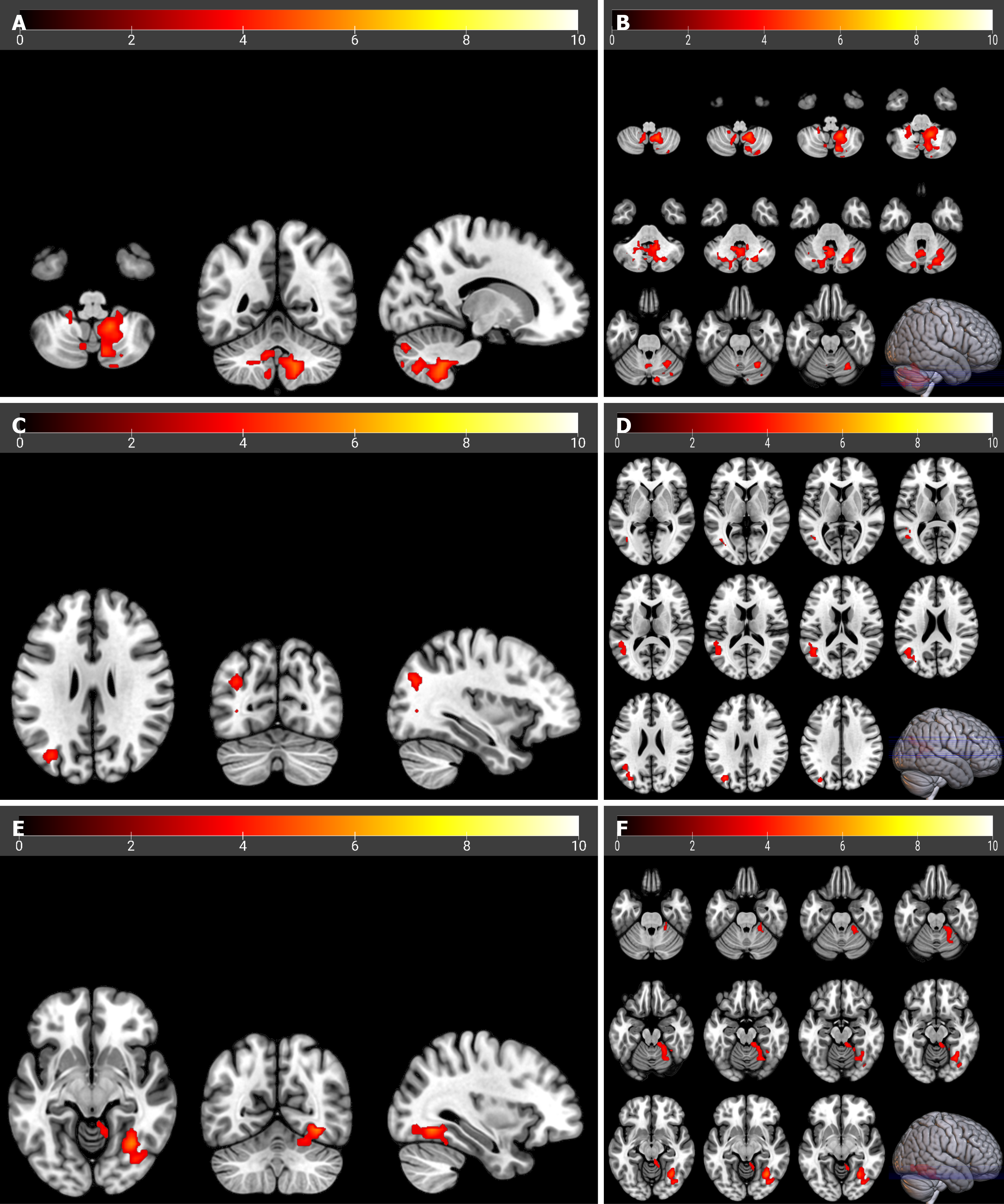
The results of the seed-based FC analysis revealed that the FC of the Cerebelum_9_R, Occipital_Mid_L, and Fusiform_R in the PSCI group was significantly greater than that in the control group. FC analysis of whole-brain seeds revealed that the FC of 20 pairs (Cerebelum_4_5_R and Cerebelum_6_R, etc.) in the PSCI group was significantly greater than that in the healthy control group.
Patients with PSCI exhibit changes in the FC of specific brain regions in the resting state, which may help re
Core Tip: Cognitive impairment is a common functional impairment after stroke that seriously affects the quality of life of patients. The underlying neurobiological mechanisms of poststroke cognitive impairment (PSCI) remain unclear. This study aimed to investigate the changes in brain functional connectivity of patients with PSCI, which may help explore the neurobiological mechanisms of PSCI from a new perspective.
- Citation: Tao YY, Wang R, Zhang P, Huang XH, Yang L. Functional connectivity alterations in patients with poststroke cognitive impairment: A resting-state functional magnetic resonance imaging study. World J Radiol 2026; 18(2): 116799
- URL: https://www.wjgnet.com/1949-8470/full/v18/i2/116799.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i2.116799
Cognitive impairment (CI) is a common form of functional impairment after stroke[1]. If not effectively managed, poststroke CI (PSCI) can result in abnormal mental behaviors and may even progress to rapidly advancing dementia[2-4]. However, the specific neurobiological mechanisms that cause PSCI remain unclear[5,6]. Images of brain activation obtained via blood-oxygenation-level-dependent functional magnetic resonance imaging (fMRI) based on the hemodynamic changes in different functionally active areas of the brain[7] have been widely used to study brain function in patients with neurological and psychiatric diseases[8]. Resting-state fMRI (rs-fMRI) measures the time correlation of changes in blood-oxygenation-level-dependent signals in various brain regions in the resting state[9]. Spontaneous neuronal activity in brain regions at baseline is detected through magnetic resonance imaging (MRI) scans, and the network connectivity of each relevant brain region is determined, reflecting spontaneous functional activity in the basal state[10]. Functional connectivity (FC), a commonly used method to assess brain function, is a term that refers to the level of functional connections between areas and is calculated by analyzing the correlation between one region and the rest of the brain[11]. FC can be used to quantify the temporal correlation of neurophysiological events in different brain regions and reveal the functional interactions between damaged brain regions and other brain regions, thus playing a critical role in the elucidation of the neurobiological mechanisms of PSCI[12,13]. Research on changes in FC in patients with PSCI may help researchers explore the underlying mechanisms of PSCI from a new perspective[6]. However, few studies have focused on changes in brain function in these patients. Thus, this study investigated changes in FC in patients with PSCI.
T1-weighted structural imaging and rs-fMRI scans were performed on 21 patients with PSCI before treatment and 12 age- and sex-matched healthy controls (HCs) with a 32-channel head-neck coil (MR750 3.0 T, GE). T1W images were parameterized as follows: Repetition time = 8.2 milliseconds, echo time = 3.1 milliseconds, flip angle = 7°, matrix = 256 × 256, field of view = 256 × 256 (mm2), and slice thickness = 1 mm. The parameters of the rs-fMRI protocol were set as follows: Re
| Clinical characteristics | PSCI | HC | χ2/t/Z | P value |
| Sex (males/females) | 12/9 | 4/8 | 1.733 | 0.188 |
| Age (years) | 60.2 ± 9.2 | 56.8 ± 5.0 | 1.393 | 0.174 |
| Education level (years) | 6.0 (6.0, 9.0) | 6.0 (6.0, 9.0) | -0.514 | 0.645 |
| Lesion laterality (left/right) | 9/12 | - | - | - |
| Onset time (days) | 3.0 (2.3, 4.0) | - | - | - |
| Lesion volume (mm3) | 1528.1 (630.5, 3055.3) | - | - | - |
| Hypertension (yes/no) | 13/8 | - | - | - |
| Diabetes (yes/no) | 4/17 | - | - | - |
| Smoking history (yes/no) | 9/12 | - | - | - |
| Alcoholism history (yes/no) | 4/17 | - | - | - |
MRI data were preprocessed using the SPM12 and DPARSF5.4 toolkits in MATLAB 2022b[14,15]. The main preprocessing pipelines were as follows: (1) The first 10 time points were removed to ensure signal stability; (2) Time correction and head-motion correction were performed by excluding subjects whose head rotated more than 3° or moved more than 3 mm along the x, y, and z axes; (3) Spatial normalization of the MRI images of the subjects to the standard Montreal Neurological Institute space with a 3 mm × 3 mm × 3 mm resample was performed; (4) Spatial smoothing using a Gaussian kernel of a 6 mm × 6 mm × 6 mm full width at half maximum; (5) Linear drift was removed to eliminate the baseline drift caused by the machine; (6) Covariates were removed to eliminate the influences of head motion, white matter and cerebrospinal fluid signals; and (7) Low-frequency filtering was performed on all images by processing them with a horizontal bandwidth of 0.01-0.08 Hz to remove the high-frequency signals.
Within the MATLAB 2022b platform, the SPM12 and DPARSF5.4 toolkits were used for data preprocessing[14,15]. The FC analysis was conducted as previously described[11]. The significant brain regions [Cerebellum_6_R (aal)] identified in the regional homogeneity analysis and the left thalamus[14], right thalamus, left basal ganglia, and right basal ganglia in the Brainnetome Atlas were used as the seeds (regions of interest); the FC between the seeds and whole-brain voxels was analyzed. In addition, the 116 brain regions defined in the AAL116 atlas were selected as seeds (regions of interests), and the FC between the whole-brain seeds was calculated.
SPM12 was used for the analysis of FC between seeds and whole-brain voxels, and the GRETNA toolkit was used for the analysis of FC between whole-brain seeds. Paired t tests were performed on data from the PSCI group, with no covariates. Two-sample t tests were performed to compare the results between the PSCI group and the control group, and sex and age were used as covariates. For the analysis of FC between seeds and whole-brain
An analysis of FC between seeds and whole-brain voxels showed that the FC of the Cerebelum_9_R, Occipital_Mid_L, and Fusiform_R in the PSCI group was significantly greater than that in the control group (Figure 1, Table 2).
| Seeds | Direction | Brain region | Coordinates of MNI peak | t value | Cluster size | ||
| X | Y | Z | |||||
| Brain region identified in the regional homogeneity analysis | PSCI group > control group | Cerebelum_9_R (aal) | 15 | -51 | -45 | 5.4862 | 644 |
| Left basal ganglia | PSCI group > control group | Occipital_Mid_L (aal) | -33 | -72 | 27 | 4.4355 | 154 |
| Left thalamus | PSCI group > control group | Fusiform_R (aal) | 33 | -60 | -9 | 5.7931 | 221 |
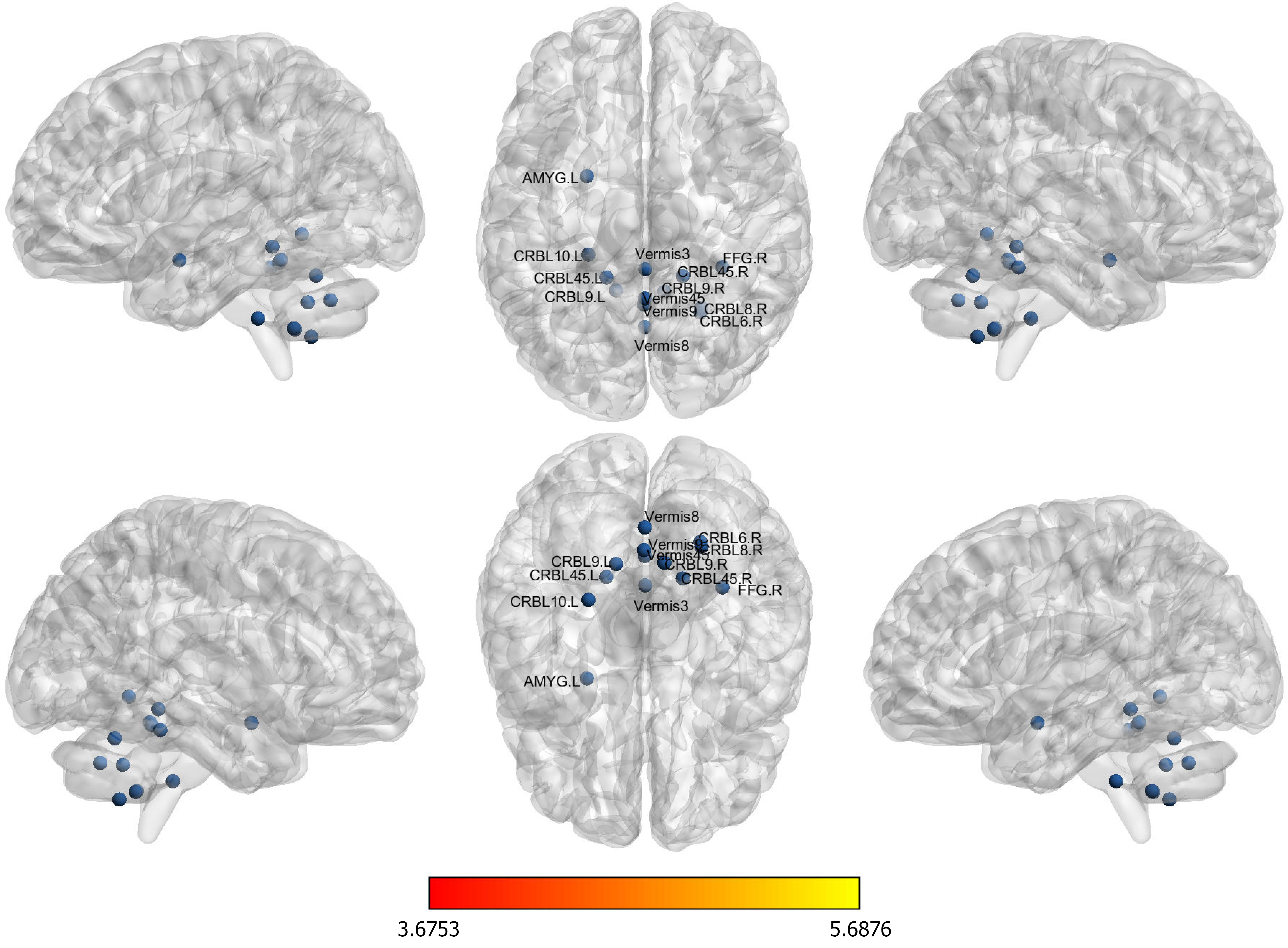
An analysis of the FC of whole-brain seeds revealed that the FC of 20 pairs in the PSCI group was significantly greater than that in the HC group. No decrease in FC was observed between the PSCI group and the control group (Figure 2, Table 3).
| Inter-regional FC | PSCI group | Control group | t value | P value |
| Cerebelum_4_5_R and Cerebelum_6_R | 1.06 ± 0.26 | 0.58 ± 0.22 | 4.5326 | 0.0001 |
| Cerebelum_4_5_R and Cerebelum_8_R | 0.68 ± 0.25 | 0.29 ± 0.24 | 4.0434 | 0.0004 |
| Amygdala_L and Cerebelum_9_L | 0.37 ± 0.21 | 0.06 ± 0.26 | 3.8097 | 0.0007 |
| Cerebelum_4_5_L and Cerebelum_9_L | 0.65 ± 0.20 | 0.24 ± 0.25 | 4.1890 | 0.0002 |
| Cerebelum_4_5_R and Cerebelum_9_L | 0.65 ± 0.17 | 0.19 ± 0.26 | 4.7038 | 0.0001 |
| Cerebelum_6_R and Cerebelum_9_L | 0.73 ± 0.32 | 0.33 ± 0.23 | 3.6753 | 0.0010 |
| Cerebelum_4_5_L and Cerebelum_9_R | 0.61 ± 0.25 | 0.20 ± 0.28 | 3.8625 | 0.0006 |
| Cerebelum_4_5_R and Cerebelum_9_R | 0.68 ± 0.21 | 0.20 ± 0.30 | 4.0530 | 0.0003 |
| Fusiform_R and Vermis_3 | 0.44 ± 0.26 | 0.17 ± 0.15 | 3.8251 | 0.0006 |
| Cerebelum_4_5_R and Vermis_3 | 0.71 ± 0.23 | 0.35 ± 0.25 | 3.8691 | 0.0006 |
| Cerebelum_6_R and Vermis_3 | 0.62 ± 0.28 | 0.24 ± 0.17 | 3.9236 | 0.0005 |
| Cerebelum_8_R and Vermis_3 | 0.56 ± 0.17 | 0.08 ± 0.17 | 4.4017 | 0.0001 |
| Cerebelum_9_L and Vermis_3 | 0.52 ± 0.17 | 0.08 ± 0.29 | 3.7043 | 0.0009 |
| Cerebelum_9_R and Vermis_3 | 0.53 ± 0.16 | 0.06 ± 0.30 | 3.9206 | 0.0005 |
| Cerebelum_10_L and Vermis_3 | 0.44 ± 0.26 | 0.01 ± 0.17 | 4.4302 | 0.0001 |
| Cerebelum_4_5_R and Vermis_4_5 | 0.85 ± 0.22 | 0.54 ± 0.14 | 5.6876 | 0.0000 |
| Cerebelum_4_5_R and Vermis_8 | 0.72 ± 0.33 | 0.16 ± 0.35 | 4.2980 | 0.0002 |
| Cerebelum_4_5_L and Vermis_9 | 0.61 ± 0.29 | 0.17 ± 0.30 | 3.9113 | 0.0005 |
| Cerebelum_4_5_R and Vermis_9 | 0.61 ± 0.33 | 0.13 ± 0.26 | 4.3557 | 0.0002 |
| Cerebelum_6_R and Vermis_9 | 0.75 ± 0.36 | 0.36 ± 0.23 | 4.2176 | 0.0002 |
In the PSCI group, the partial correlation analysis (controlling for age) revealed that among the significant brain regions, the FC values of the Fusiform_R were significantly correlated with the MMSE scores (r = 0.464; P = 0.039; without correction for multiple comparisons), but no other significant correlations were identified.
Previous studies have shown that PSCI is closely related to infarcted brain areas[21]. The basal ganglia[22], internal capsule, thalamus, corpus callosum[23], angular gyrus[24], cortex cingulate[25] and subfrontal cortical areas[26] are key brain regions involved in global PSCI. Patients with PSCI have disrupted low-degree rich-club organization, relatively preserved functional core networks, and decreased feeder and local connectivity in cognition-related networks[27]. In the present study, as the ischemic stroke lesions of patients were located mainly in the bilateral thalamus or basal ganglia, in addition to the significant brain regions identified in the regional homogeneity analysis, the bilateral thalamus and basal ganglia were also used as the seeds. The results revealed that the FC of the PSCI group in Cerebelum_9_R, Occipi
In recent years, researchers have studied patterns of altered FC in patients with PSCI from different perspectives[6,35-37]. Yue et al[6] performed fMRI examinations on 17 patients with PSCI and 24 HCs and used independent component analysis combined with sliding-window and K-means clustering approaches to examine the FC of 11 resting-state networks. Their results revealed that in terms of static functional network connectivity, the PSCI group presented decreased dorsal default mode network-ventral mode network, ventral mode network-salience network, dorsal default mode network-higher visual network, auditory network-right executive control network, and auditory network-visuospatial network interactions. Li et al[36] used rs-fMRI to study the effects of repetitive transcranial magnetic stimulation on brain function in patients with PSCI and reported that, compared with the control group, the repetitive transcranial magnetic stimulation group presented increased FC between the left dorsolateral prefrontal cortex and the precuneus, the inferior temporal gyrus, the middle and inferior frontal gyri and the marginal gyrus and decreased FC between the left dorsolateral prefrontal cortex and the middle temporal gyrus and thalamus. Lu et al[37] investigated functional alterations in the nucleus basalis of Meynert (NBM) and its projections in patients with mild CI and assessed the effects of computerized cognitive training (CCT). They analyzed FC between the NBM and three correlated brain regions. Their findings demonstrated that CCT intervention elevated the FC between the NBM and the right putamen, which lends support to the neuroplastic potential of impaired brains and highlights the clinical value of CCT in patients with MC.
As a core node in brain networks underlying social cognition, the cerebellum collaborates with multiple other cerebral regions to exert critical effects on cognitive processing. Nevertheless, the specific mechanism through which this cerebellumcentered network coordinates to ameliorate CI remains largely unclear. A growing body of prior research has also emphasized a robust link between cerebellar function and the pathogenesis of CI[32,38-42]. Zhang et al[43] used rs-fMRI to examine the difference in the FC of the limbic system and the cerebellum between patients with and without PSCI and identified the intracerebellar brain regions that exhibited functional changes during the onset of CI; their results revealed that the functional connections between the Cerebelum_Crus2_R and Frontal_Mid_Orb_L brain regions decreased. The functional connections between the Cerebelum_Crus2_R and Hippocampus_L brain regions decreased. Tang et al[44] collected rs-fMRI data from three different groups (28 patients with Alzheimer’s disease, 26 patients with mild CI and 30 HCs) and defined the cerebellar lobes (Crus II and IX) as seed regions to assess differences in cortical-cerebellar connectivity within groups, and reported decreased FC between the cerebellum and the medial frontal gyrus in patients with Alzheimer’s disease. However, the FC between the right-sided lobule IX and the medial frontal gyrus was significantly increased in patients with mild CI, which may compensate for the impaired memory that is often observed in these patients. Hong et al[42] collected rs-fMRI data from thirty-six participants with subcortical chronic stroke and thirty-eight HCs and performed FC analysis with the bilateral cerebellar anterior lobe and cerebellar posterior lobe as seeds for each participant. They reported that the cerebellar anterior lobe showed increased FC with the prefrontal cortex, superior/inferior temporal gyrus, and lingual gyrus, while the cerebellar posterior lobe showed increased FC with the inferior parietal lobule, precuneus, and cingulum gyrus in stroke participants compared with HCs. Jung et al[32] explored changes in hippocampal FC following ischemic stroke using rs-fMRI. Thirty-three patients with CI after ischemic stroke and sixteen HCs were recruited. Their results showed that across all the hippocampal subfields, FC with the inferior parietal lobule was reduced in participants with stroke compared with HCs, and the FC of the hippocampal subfields with the cerebellum was increased. In addition, a few studies have reported that cerebellar–hippocampal interactions are associated with various cognitive functions and have indicated the importance of the cerebellum and cerebellar-hippocampal connections for cognitive tasks. The results of this study differ from those reported by Zhang et al[43] and are consistent with those reported by Hong et al[42].
The results of this study revealed a positive correlation between the FC values of the Fusiform_R and MMSE scores after controlling for age, suggesting that it may be specifically associated with PSCI disease status; other regions with increased FC did not significantly correlate with MMSE scores, which may be related to the complexity of compensation and other factors[45].
This study had several limitations: (1) The sample size was relatively small, which significantly reduced the statistical power and significantly increased the risk of both type I (false positive) and type II (false negative) errors; (2) The patients with PSCI had mild CI according to the Petersen criteria[46], and the differences in brain FC between patients with different degrees of CI were not analyzed; and (3) The control for multiple comparisons was inadequate. The whole-brain seed analysis used the AAL116 atlas, and the significance threshold was set to edge_P < 0.001 and component_P < 0.05 (NBS-corrected). While the NBS is appropriate, the initial uncorrected edge threshold of P < 0.001 for such a massive number of tests may still be too lenient. In future studies, the sample size should be increased[47,48], subgroups with different degrees of CI should be established for further validation, and the impact of the test used to correct for multiple comparisons on the results should be further investigated.
In summary, this study preliminarily revealed that in the resting state, patients with PSCI exhibited changes in FC in specific brain regions, which may be important mechanisms of PSCI and may be potential imaging biomarkers for the evaluation of CI in PSCI patients.
| 1. | Qu Y, Zhuo L, Li N, Hu Y, Chen W, Zhou Y, Wang J, Tao Q, Hu J, Nie X, Zhan S. Prevalence of post-stroke cognitive impairment in china: a community-based, cross-sectional study. PLoS One. 2015;10:e0122864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 124] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 2. | Mijajlović MD, Pavlović A, Brainin M, Heiss WD, Quinn TJ, Ihle-Hansen HB, Hermann DM, Assayag EB, Richard E, Thiel A, Kliper E, Shin YI, Kim YH, Choi S, Jung S, Lee YB, Sinanović O, Levine DA, Schlesinger I, Mead G, Milošević V, Leys D, Hagberg G, Ursin MH, Teuschl Y, Prokopenko S, Mozheyko E, Bezdenezhnykh A, Matz K, Aleksić V, Muresanu D, Korczyn AD, Bornstein NM. Post-stroke dementia - a comprehensive review. BMC Med. 2017;15:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 493] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 3. | Rost NS, Brodtmann A, Pase MP, van Veluw SJ, Biffi A, Duering M, Hinman JD, Dichgans M. Post-Stroke Cognitive Impairment and Dementia. Circ Res. 2022;130:1252-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 543] [Article Influence: 135.8] [Reference Citation Analysis (0)] |
| 4. | Gallucci L, Sperber C, Guggisberg AG, Kaller CP, Heldner MR, Monsch AU, Hakim A, Silimon N, Fischer U, Arnold M, Umarova RM. Post-stroke cognitive impairment remains highly prevalent and disabling despite state-of-the-art stroke treatment. Int J Stroke. 2024;19:888-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 41] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 5. | Min Y, Liu C, Zuo L, Wang Y, Li Z. The relationship between altered degree centrality and cognitive function in mild subcortical stroke: A resting-state fMRI study. Brain Res. 2023;1798:148125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 6. | Yue X, Li Z, Li Y, Gao J, Han H, Zhang G, Li X, Shen Y, Wei W, Bai Y, Xie J, Luo Z, Zhang X, Wang M. Altered static and dynamic functional network connectivity in post-stroke cognitive impairment. Neurosci Lett. 2023;799:137097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 7. | Conklin CJ, Faro SH, Mohamed FB. Technical considerations for functional magnetic resonance imaging analysis. Neuroimaging Clin N Am. 2014;24:695-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Sun YW, Qin LD, Zhou Y, Xu Q, Qian LJ, Tao J, Xu JR. Abnormal functional connectivity in patients with vascular cognitive impairment, no dementia: a resting-state functional magnetic resonance imaging study. Behav Brain Res. 2011;223:388-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 87] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 9. | Azeez AK, Biswal BB. A Review of Resting-State Analysis Methods. Neuroimaging Clin N Am. 2017;27:581-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 10. | Raichle ME, Snyder AZ. A default mode of brain function: a brief history of an evolving idea. Neuroimage. 2007;37:1083-90; discussion 1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1462] [Cited by in RCA: 1482] [Article Influence: 78.0] [Reference Citation Analysis (0)] |
| 11. | van den Heuvel MP, Hulshoff Pol HE. Exploring the brain network: a review on resting-state fMRI functional connectivity. Eur Neuropsychopharmacol. 2010;20:519-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2902] [Cited by in RCA: 2251] [Article Influence: 140.7] [Reference Citation Analysis (3)] |
| 12. | Auer DP. Spontaneous low-frequency blood oxygenation level-dependent fluctuations and functional connectivity analysis of the 'resting' brain. Magn Reson Imaging. 2008;26:1055-1064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 165] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 13. | Han K, Dong L, Liao X, Long J, Chen J, Lu H, Zhang H. Alterations in brain function in patients with post-stroke cognitive impairment: a resting-state functional magnetic resonance imaging study. Front Aging Neurosci. 2025;17:1501082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Zhang P, Wang R, Shi Y, Huang X, Yang L. Regional Coherence Alterations in Patients with Poststroke Cognitive Impairment after Acupuncture Therapy: A Resting-State fMRI Study. Health. 2024;16:1176-1186. [DOI] [Full Text] |
| 15. | Yan CG, Wang XD, Zuo XN, Zang YF. DPABI: Data Processing & Analysis for (Resting-State) Brain Imaging. Neuroinformatics. 2016;14:339-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3467] [Cited by in RCA: 3050] [Article Influence: 305.0] [Reference Citation Analysis (1)] |
| 16. | Zalesky A, Fornito A, Bullmore ET. Network-based statistic: identifying differences in brain networks. Neuroimage. 2010;53:1197-1207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1577] [Cited by in RCA: 2132] [Article Influence: 133.3] [Reference Citation Analysis (1)] |
| 17. | Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10:186-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9905] [Cited by in RCA: 7307] [Article Influence: 429.8] [Reference Citation Analysis (0)] |
| 18. | Nichols T, Hayasaka S. Controlling the familywise error rate in functional neuroimaging: a comparative review. Stat Methods Med Res. 2003;12:419-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 830] [Cited by in RCA: 840] [Article Influence: 38.2] [Reference Citation Analysis (0)] |
| 19. | Qian L, Li Y, Wang Y, Wang Y, Cheng X, Li C, Cui X, Jiao G, Ke X. Shared and Distinct Topologically Structural Connectivity Patterns in Autism Spectrum Disorder and Attention-Deficit/Hyperactivity Disorder. Front Neurosci. 2021;15:664363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 20. | Amorim L, Magalhães R, Coelho A, Moreira PS, Portugal-Nunes C, Castanho TC, Marques P, Sousa N, Santos NC. Poor Sleep Quality Associates With Decreased Functional and Structural Brain Connectivity in Normative Aging: A MRI Multimodal Approach. Front Aging Neurosci. 2018;10:375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 21. | Zhao L, Biesbroek JM, Shi L, Liu W, Kuijf HJ, Chu WW, Abrigo JM, Lee RK, Leung TW, Lau AY, Biessels GJ, Mok V, Wong A. Strategic infarct location for post-stroke cognitive impairment: A multivariate lesion-symptom mapping study. J Cereb Blood Flow Metab. 2018;38:1299-1311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 140] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 22. | Narasimhalu K, Wiryasaputra L, Sitoh YY, Kandiah N. Post-stroke subjective cognitive impairment is associated with acute lacunar infarcts in the basal ganglia. Eur J Neurol. 2013;20:547-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 28] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 23. | Biesbroek JM, Weaver NA, Biessels GJ. Lesion location and cognitive impact of cerebral small vessel disease. Clin Sci (Lond). 2017;131:715-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 144] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 24. | Benson DF, Cummings JL. Angular gyrus syndrome simulating Alzheimer's disease. Arch Neurol. 1982;39:616-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 56] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 25. | Munsch F, Sagnier S, Asselineau J, Bigourdan A, Guttmann CR, Debruxelles S, Poli M, Renou P, Perez P, Dousset V, Sibon I, Tourdias T. Stroke Location Is an Independent Predictor of Cognitive Outcome. Stroke. 2016;47:66-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 110] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 26. | Kandiah N, Wiryasaputra L, Narasimhalu K, Karandikar A, Marmin M, Chua EV, Sitoh YY. Frontal subcortical ischemia is crucial for post stroke cognitive impairment. J Neurol Sci. 2011;309:92-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 27. | Miao G, Rao B, Wang S, Fang P, Chen Z, Chen L, Zhang X, Zheng J, Xu H, Liao W. Decreased Functional Connectivities of Low-Degree Level Rich Club Organization and Caudate in Post-stroke Cognitive Impairment Based on Resting-State fMRI and Radiomics Features. Front Neurosci. 2021;15:796530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 28. | Dipasquale O, Cercignani M. Network functional connectivity and whole-brain functional connectomics to investigate cognitive decline in neurodegenerative conditions. Funct Neurol. 2016;31:191-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 29. | Cole DM, Smith SM, Beckmann CF. Advances and pitfalls in the analysis and interpretation of resting-state FMRI data. Front Syst Neurosci. 2010;4:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 247] [Cited by in RCA: 521] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 30. | Liu C, Zuo L, Li Z, Jing J, Wang Y, Liu T. Brain structural-functional coupling mechanism in mild subcortical stroke and its relationship with cognition. Brain Res. 2024;1845:149167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 31. | Sagues E, Alfaro F, Ramos-Rodríguez R, García-Casares N. Resting-state functional connectivity alterations in post-stroke cognitive impairment: a systematic review. Brain Imaging Behav. 2025;19:1117-1131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 32. | Jung J, Laverick R, Nader K, Brown T, Morris H, Wilson M, Auer DP, Rotshtein P, Hosseini AA. Altered hippocampal functional connectivity patterns in patients with cognitive impairments following ischaemic stroke: A resting-state fMRI study. Neuroimage Clin. 2021;32:102742. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 33. | Cramer SC. Treatments to Promote Neural Repair after Stroke. J Stroke. 2018;20:57-70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 34. | Schmahmann JD. The cerebellum and cognition. Neurosci Lett. 2019;688:62-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 398] [Cited by in RCA: 829] [Article Influence: 118.4] [Reference Citation Analysis (0)] |
| 35. | Tuladhar AM, Snaphaan L, Shumskaya E, Rijpkema M, Fernandez G, Norris DG, de Leeuw FE. Default Mode Network Connectivity in Stroke Patients. PLoS One. 2013;8:e66556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 102] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 36. | Li Y, Luo H, Yu Q, Yin L, Li K, Li Y, Fu J. Cerebral Functional Manipulation of Repetitive Transcranial Magnetic Stimulation in Cognitive Impairment Patients After Stroke: An fMRI Study. Front Neurol. 2020;11:977. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 37. | Lu Q, Wang Y, Qu B, Wang C, Su X, Wang S, Xing Y, Qin W, Tang Y, Zhang N. Structural and Functional Projections of the Nucleus Basalis of Meynert and Their Changes After Cognitive Training in Individuals With Mild Cognitive Impairment. CNS Neurosci Ther. 2024;30:e70194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 38. | Jacobs HIL, Hopkins DA, Mayrhofer HC, Bruner E, van Leeuwen FW, Raaijmakers W, Schmahmann JD. The cerebellum in Alzheimer's disease: evaluating its role in cognitive decline. Brain. 2018;141:37-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 263] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 39. | Rudolph S, Badura A, Lutzu S, Pathak SS, Thieme A, Verpeut JL, Wagner MJ, Yang YM, Fioravante D. Cognitive-Affective Functions of the Cerebellum. J Neurosci. 2023;43:7554-7564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 110] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 40. | Yin K, Zhou C, Yin L, Zhu Y, Yin W, Lu Y, Liu B, Ren H, Xu Z, Yang X. Resting-state functional magnetic resonance imaging of the cerebellar vermis in patients with Parkinson's disease and visuospatial disorder. Neurosci Lett. 2021;760:136082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 41. | Wang Y, Liu W, Yang W, Chai X, Yu H, Ma H, Liu L, Rao J, Xu G, Hu Z. Differential Abnormality in Regional Brain Spontaneous Activity and Functional Connectivity in Patients of Non-Acute Subcortical Stroke With Versus Without Global Cognitive Functional Impairment. Brain Behav. 2025;15:e70356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 42. | Hong W, Du Y, Xu R, Zhang X, Liu Z, Li M, Yu Z, Wang Y, Wang M, Yang B, Sun F, Xu G. Altered cerebellar functional connectivity in chronic subcortical stroke patients. Front Hum Neurosci. 2022;16:1046378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 43. | Zhang H, Lu J, Zhang L, Hu J, Yue J, Ma Y, Yao Q, Jie P, Fan M, Fang J, Zhao J. Abnormal cerebellar activity and connectivity alterations of the cerebellar-limbic system in post-stroke cognitive impairment: a study based on resting state functional magnetic resonance imaging. Front Neurosci. 2025;19:1543760. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 44. | Tang F, Zhu D, Ma W, Yao Q, Li Q, Shi J. Differences Changes in Cerebellar Functional Connectivity Between Mild Cognitive Impairment and Alzheimer's Disease: A Seed-Based Approach. Front Neurol. 2021;12:645171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 45. | Hillary FG, Grafman JH. Injured Brains and Adaptive Networks: The Benefits and Costs of Hyperconnectivity. Trends Cogn Sci. 2017;21:385-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 251] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 46. | Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol. 1999;56:303-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7065] [Cited by in RCA: 6235] [Article Influence: 230.9] [Reference Citation Analysis (0)] |
| 47. | Marek S, Tervo-Clemmens B, Calabro FJ, Montez DF, Kay BP, Hatoum AS, Donohue MR, Foran W, Miller RL, Hendrickson TJ, Malone SM, Kandala S, Feczko E, Miranda-Dominguez O, Graham AM, Earl EA, Perrone AJ, Cordova M, Doyle O, Moore LA, Conan GM, Uriarte J, Snider K, Lynch BJ, Wilgenbusch JC, Pengo T, Tam A, Chen J, Newbold DJ, Zheng A, Seider NA, Van AN, Metoki A, Chauvin RJ, Laumann TO, Greene DJ, Petersen SE, Garavan H, Thompson WK, Nichols TE, Yeo BTT, Barch DM, Luna B, Fair DA, Dosenbach NUF. Reproducible brain-wide association studies require thousands of individuals. Nature. 2022;603:654-660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1792] [Cited by in RCA: 1393] [Article Influence: 348.3] [Reference Citation Analysis (0)] |
| 48. | DeYoung CG, Hilger K, Hanson JL, Abend R, Allen TA, Beaty RE, Blain SD, Chavez RS, Engel SA, Feilong M, Fornito A, Genç E, Goghari V, Grazioplene RG, Homan P, Joyner K, Kaczkurkin AN, Latzman RD, Martin EA, Nikolaidis A, Pickering AD, Safron A, Sassenberg TA, Servaas MN, Smillie LD, Spreng RN, Viding E, Wacker J. Beyond Increasing Sample Sizes: Optimizing Effect Sizes in Neuroimaging Research on Individual Differences. J Cogn Neurosci. 2025;37:1023-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/