Published online Feb 28, 2026. doi: 10.4329/wjr.v18.i2.116610
Revised: December 28, 2025
Accepted: January 22, 2026
Published online: February 28, 2026
Processing time: 102 Days and 2.1 Hours
Surgical strategies for cochlear implantation in patients with chronic otitis media (COM) are diverse and largely depend on the extent of the underlying pathology.
To develop a high-resolution computed tomography (HRCT)-based algorithm for guiding surgical strategy using correlations between imaging and operative findings.
We retrospectively analyzed the preoperative HRCT scans of 12 consecutive adult patients (n = 12) with COM who underwent cochlear implantation. Specific radio
Preoperative HRCT accurately predicted the necessary surgical approach in all cases in our cohort. Disease limited to the epitympanum with an intact posterior canal wall required a canal wall-up surgical approach (n = 7), whereas extensive soft tissue opacity involving the mastoid cavity necessitated a canal wall-down/subtotal petrosectomy approach (n = 5). HRCT achieved 100% sensitivity for detecting the single case of significant cochlear ossification in this preliminary series, allowing for appropriate preoperative planning. Postoperative computed tomography confirmed successful electrode placement in all cases. Clinical outcomes, including a low complication rate (one minor infection) and no disease recurrence, confirmed the accuracy of the imaging-based strategy.
Preoperative HRCT reliably predicts the required surgical approach in COM. The proposed imaging-based algorithm may help standardize planning for successful cochlear implantation.
Core Tip: This study demonstrates that preoperative high-resolution computed tomography (HRCT) is a decisive tool for planning cochlear implantation in patients with chronic otitis media. Complete mastoid opacification on HRCT reliably predicts the need for radical surgery (canal wall-down/subtotal petrosectomy), whereas a clear mastoid permits a conservative (canal wall-up) approach. HRCT also accurately detects cochlear ossification, enabling crucial preoperative pre
- Citation: Zhao LJ, Fu Y, Zhang ZL. High-resolution computed tomography predicts optimal cochlear implantation strategy in patients with chronic otitis media. World J Radiol 2026; 18(2): 116610
- URL: https://www.wjgnet.com/1949-8470/full/v18/i2/116610.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i2.116610
Cochlear implantation has become a standard and effective treatment for severe to profound sensorineural hearing loss[1-3]. However, its application in patients with chronic otitis media (COM) or middle ear cholesteatoma presents considerable challenges, including the need for thorough pathological removal to prevent recurrence, management of distorted middle ear and mastoid anatomy, and mitigation of long-term risks, such as infection, device migration, or extrusion[4]. Therefore, careful preoperative planning is essential to ensure both oncological safety and successful auditory rehabilitation in these patients.
The selection of surgical strategy is crucial and continues to be debated[5-7]. Surgical options span a spectrum, from the conservative canal wall-up (CWU) mastoidectomy - which preserves the native ear canal anatomy but risks leaving residual disease - to more radical procedures. The latter include the canal wall-down (CWD) mastoidectomy - where the posterior ear canal wall is removed to create an open cavity - and the definitive subtotal petrosectomy (STP) - which entails complete removal of the middle ear and mastoid contents, followed by closure of the external auditory canal and cavity obliteration to establish a sterile, isolated environment suitable for cochlear implantation[3]. These extensive techniques prioritize thorough disease eradication and the creation of a secure implant site over anatomical preservation. Given the inherent complexity of and variability among COM cases, objective and standardized preoperative criteria are essential to guide decision-making, although clinical judgment and intraoperative findings remain paramount. In this context, high-resolution computed tomography (HRCT) of the temporal bone serves as the cornerstone of preoperative assessment, offering unparalleled visualization of bony anatomy and disease extent.
Despite the widespread use of HRCT, few studies have systematically correlated specific imaging findings with surgical requirements. This underrepresentation in the research may stem from a lack of consensus on which radiological features are most predictive, limiting the development of a standardized, imaging-based decision-making framework for clinical practice. While many studies have focused on reporting outcomes of different surgical techniques[8-13], few have concentrated on using preoperative imaging alone to reliably guide the choice of surgical approach. Consequently, current practice often relies on surgeon experience rather than on objective, reproducible imaging criteria, highlighting a critical gap that this study aimed to address.
This study, thus, retrospectively analyzed a cohort of complex cochlear implantation candidates. We had two primary objectives: (1) To identify key HRCT markers that reliably predict the required surgical approach; and (2) To develop a clear, imaging-based algorithm that standardizes surgical planning and improves outcomes in this challenging population. Importantly, our algorithm builds upon the concept of anatomical preservation in selected cases (e.g., Jeong et al[14]) but also advances the field by providing a systematic, radiology-driven framework specifically tailored for cochlear implantation planning, integrating both mastoid status and cochlear ossification (CO) assessment. By esta
This single-center, observational study included 12 consecutive adult patients (> 18 years) with documented COM or middle ear cholesteatoma who underwent cochlear implantation at our tertiary referral institution between June 2022 and October 2025. Patient demographics and clinical characteristics are detailed in Table 1. The patient population exhibited a wide spectrum of middle ear and mastoid pathologies, including active cholesteatoma, pars flaccida retraction pockets, middle ear granuloma, and a history of prior CWU or CWD mastoidectomy. Key exclusion criteria were: (1) Pediatric pa
| Demographic characteristics | CWU group (n = 7) | CWD/STP group (n = 5) | P value |
| Sex, female | 3 (42.9) | 2 (40.0) | 1.000 |
| Age in years | 66.3 ± 5.5 | 65.0 ± 9.9 | 0.778 |
| Duration of hearing loss in years | 27.6 ± 20.3 | 14.4 ± 8.2 | 0.201 |
| Etiology | |||
| Pars flaccida retraction pocket | 4 | 0 | |
| MEC/history of MEC | 2 | 3 | |
| MEC relapse | 0 | 2 | |
| Middle ear CG | 1 | 0 | |
| Follow-up duration in months | 26.9 ± 3.0 | 30.2 ± 14.2 | 0.558 |
All patients underwent a standardized preoperative workup, including a full otologic history, microscopic otoscopic examination, comprehensive audiological testing (e.g., pure-tone audiometry, speech discrimination scores), and the radiological imaging protocol detailed below. All surgeries were performed by the same senior otologic surgeon to minimize technical variability. This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (Approval No. 2025B IIT Ethics Approval No. 1088). The requirement for written informed consent was formally waived by the ethics committee due to the retrospective nature of the study.
All patients underwent preoperative temporal bone HRCT using the GE Revolution EVO scanner (General Electric, Boston, MA, United States). The scan parameters were as follows: Tube voltage 120 kV, tube current modulated automatically (range 100-300 mA), slice thickness 0.625 mm with 0.3 mm reconstruction interval, and bone reconstruction algorithm (kernel). Axial and coronal planes were reconstructed for comprehensive analysis.
Qualitative evaluation was preferred over semi-quantitative scoring systems (e.g., Lund-Mackay) as it better reflects the holistic, pattern-based interpretation commonly used in real-world clinical decision-making for surgical planning in this context. Assessment relied solely on the qualitative evaluation of morphological changes, without a formal quantitative scoring system. Due to the limitations of HRCT in detecting minute dehiscence’s (e.g., facial nerve canal), MRI was performed using the GE MR 750 scanner (General Electric, Boston, MA, United States) to complement HRCT findings and differentiate cholesteatoma from inflammatory tissue. MRI was performed using the GE MR 750 scanner (General Electric, Boston, MA, United States) with a dedicated head coil. The protocol included: Axial and coronal T2-weighted fast spin-echo sequences (TR/TE: 3000/100 milliseconds), axial T1-weighted fast spin-echo sequences (TR/TE: 600/10 milliseconds), axial diffusion-weighted imaging (DWI) with single-shot spin-echo echo-planar imaging (b-values: 0 second/mm2 and 1000 second/mm2; spatial resolution: 1.2 mm × 1.2 mm × 3.0 mm). DWI and contrast-enhanced se
To ensure a standardized and reproducible evaluation, preoperative HRCT scans were assessed using explicit, hierar
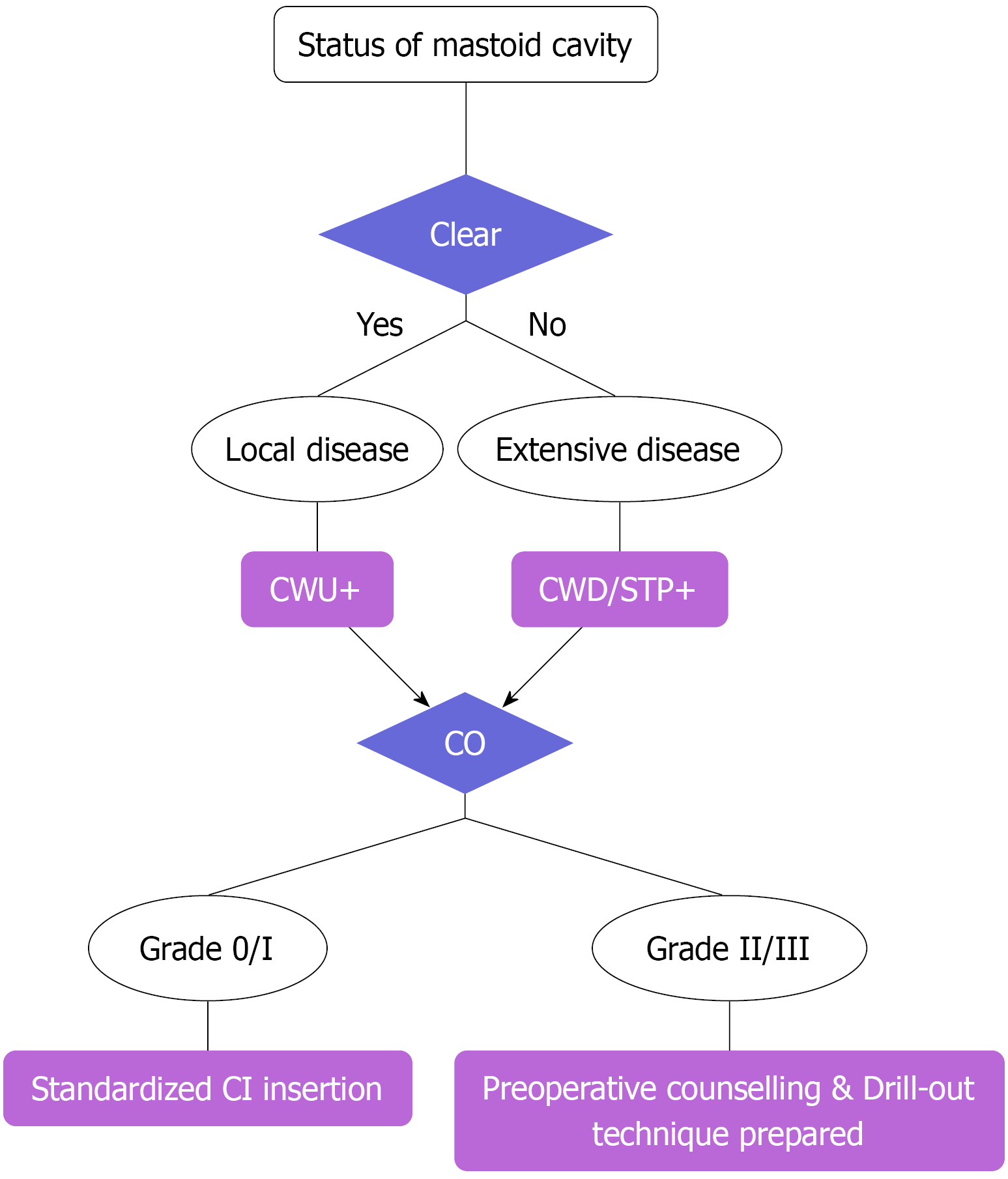
The status of the mastoid cavity served as the primary determinant: A clear or well-aerated mastoid - defined as the absence of soft-tissue opacification within the mastoid air cell system, or the presence of disease confined to the middle ear spaces (epitympanum/mesotympanum) with an intact posterior canal wall - was planned for a CWU procedure. Conversely, complete opacification of the mastoid cavity - defined as diffuse soft-tissue density occupying the entire mastoid air cell system, irrespective of middle ear findings - indicated the need for a more extensive procedure, namely CWD mastoidectomy or STP, to ensure complete disease eradication.
Secondly, CO was assessed for technical planning. The severity of CO was graded according to established radiological criteria, namely: Grade 0 (none); grade I (ossification confined to the round window niche, defined as obliteration of the round window membrane with bony tissue thickness > 4 mm); grade II (ossification extending into the basal turn of the cochlea but limited to within 180°); and grade III (ossification extending beyond the basal turn, involving > 180° of the cochlear lumen). For grade II and grade III, the linear extent of ossification along the basal turn was also measured from the round window niche; an extent of ≥ 8 mm was considered indicative of more severe obstruction within those grades[16]. It is important to note that the presence of severe CO did not alter the primary choice between a CWU or CWD/STP approach, but it triggered specific preoperative preparations. These included counseling the patient regarding the potential for only partial electrode insertion and preparing surgically for specialized techniques, such as drill-out procedures.
This stepwise evaluation framework directly informed the decision algorithm depicted in Figure 1. The final imaging-based prediction (CWU or CWD/STP, plus note on severe CO) was recorded prior to surgery for later validation against the actual procedure performed (the gold standard).
A comprehensive correlation analysis was conducted to quantify the relationship between radiological parameters and surgical outcomes. Key imaging features - including mastoid opacification, scutum erosion, tegmen tympani thickness, facial nerve canal integrity, and CO - were qualitatively assessed by a senior neuroradiologist (> 10 years of experience) and the lead otologic surgeon using a structured reporting template (Supplementary material). Inter-rater agreement for these categorical assessments was excellent (Cohen’s κ = 0.85).
All statistical analyses were conducted with SPSS Statistics version 29.0 (IBM Corp., Armonk, NY, United States). The predictive accuracy of the HRCT-based criteria was evaluated by calculating sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV), using intraoperative findings as the gold standard. Given the small sample size, confidence intervals (CIs) were calculated for sensitivity, specificity, PPV, and NPV via the exact binomial method (Clopper-Pearson) in order to provide a measure of statistical uncertainty. Of note, due to the exploratory nature of this study (with a limited cohort), the statistical results presented herein represent preliminary evidence rather than definitive performance metrics. The association between specific radiological findings (e.g., complete mastoid opaci
Applying the standardized imaging criteria to the cohort yielded a preoperative prediction for all 12 cases. In this cohort, inter-rater agreement for the qualitative assessment of key HRCT features was excellent (κ = 0.85). The HRCT-based predictions showed full concordance with the actual surgical approach performed in all cases. The scans correctly predicted the need for CWU or CWD/STP, demonstrating 100% sensitivity (95%CI: 63.1%-100%), specificity (95%CI: 59.0%-100%), PPV (95%CI: 63.1%-100%), and NPV (95%CI: 59.0%-100%) in this series. The width of these CIs underscores the statistical imprecision inherent in small-sample studies and reinforces the need for cautious interpretation within this exploratory framework. The association between complete mastoid opacification on HRCT and the necessity for a CWD/STP procedure was statistically significant (P < 0.001).
The analysis revealed a direct and consistent relationship between preoperative HRCT features and the surgical approach. In all 12 cases, the procedure performed matched the preoperative plan derived from imaging, resulting in 100% concordance (see complete case-by-case data in Supplementary Table 1). The status of the mastoid cavity served as the primary and decisive factor in every case: A clear or well-aerated mastoid was uniformly associated with successful management via a CWU mastoidectomy (n = 7). Conversely, complete opacification of the mastoid cavity was an unequivocal indicator for a more extensive CWD mastoidectomy or STP procedure (n = 5). Within the CWD/STP group, the presence of CO (grade I/II/III) on HRCT accurately identified cases requiring specialized preoperative counseling and preparation for a drill-out technique. This stepwise correlation confirms that systematic HRCT evaluation can effectively stratify surgical strategy based on disease extent.
The predictive power of HRCT is best illustrated by comparing two distinct clinical scenarios.
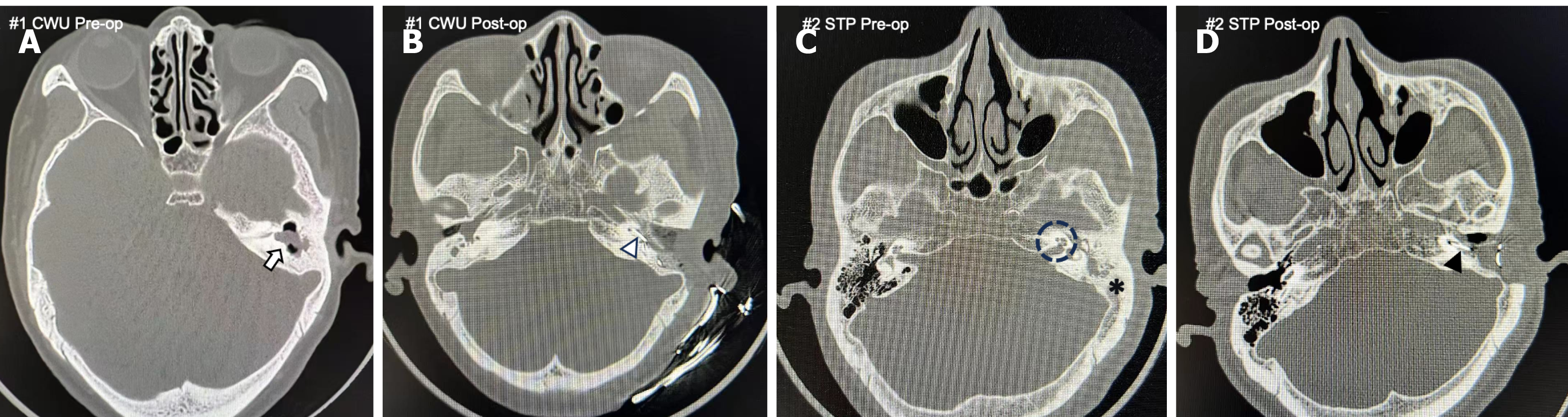
Case 1: A 71-year-old male with cholesteatoma presented with classic, localized disease on preoperative HRCT (Figure 2A and B). The scan showed soft tissue pathology confined to the epitympanic space, with a well-aerated mastoid and intact posterior canal wall. The clear radiological evidence allowed for a conservative CWU mastoidectomy to safely remove the disease while preserving the natural ear canal anatomy. Surgery proceeded as planned, confirming localized cholesteatoma and allowing successful simultaneous cochlear implantation.
Case 2: By contrast, a 60-year-old female presented with a more complex radiological profile (Figure 2C and D). HRCT revealed widespread opacification throughout the middle ear and entire mastoid cavity. Critically, the scan revealed severe (grade III) CO, posing a significant challenge for electrode insertion and requiring specialized techniques. Imaging unequivocally indicated that a limited, conservative approach would be insufficient and unsafe. Consequently, a radical STP was scheduled to ensure complete disease removal. Preoperative awareness of severe CO allowed for patient counseling regarding the high probability of only partial electrode insertion. The extent of the disease and CO was confirmed during surgery, validating the radical surgical approach taken.
These cases illustrate our central finding: Complete opacification of the mastoid cavity was the most reliable predictor for the necessity of a radical procedure (CWD/STP), which was present in all 5 cases where such a procedure was needed. Conversely, when the disease was confined to the middle ear spaces with a clear mastoid, the CWU approach was successful in all 7 applicable cases. Postoperative computed tomography confirmed satisfactory electrode placement in all 12 patients. The baseline demographic characteristics were comparable between the CWU and CWD/STP groups (all P > 0.05; Table 1). Of note, Table 1 includes key preoperative and postoperative auditory outcomes (speech discrimination scores) for both groups where available, enhancing the clinical relevance of our study’s findings.
This study demonstrates that systematic analysis of preoperative HRCT scans can reliably predict the optimal surgical strategy for cochlear implantation in patients with COM. Our findings reposition HRCT from a descriptive tool to a powerful predictive instrument for surgical planning. The 100% concordance between an imaging-based plan and executed surgery in this preliminary cohort underscores its potential value, though the associated 95%CIs for all predictive metrics (sensitivity, specificity, PPV, and NPV) were wide, with lower bounds ranging from 59.0% to 63.1%. This reflects the substantial statistical uncertainty inherent in our small cohort and indicates that the perfect point estimates should be interpreted as promising preliminary findings. Furthermore, the small sample size increases the risk of overfitting the decision algorithm to our specific cohort, potentially limiting its generalizability. Future validation studies should employ crossvalidation techniques to better assess model performance. Therefore, while these results underscore the predictive potential of HRCT, they also emphasize the need for validation in larger, prospective studies before this approach can be generalized into routine practice.
The primary contribution of this study is confirmation that the extent of mastoid opacification on HRCT is the single most critical predictor for surgical planning. The consistent finding that well-aerated mastoids could be managed with the CWU procedure, whereas extensive mastoid disease required a radical approach, offers a clear and practical guideline. This aligns with the concept proposed by Jeong et al[14], who advocated a CWU approach for cholesteatoma confined to the epitympanum. Our research expands on this by specifically demonstrating the applicability of this technique for cochlear implantation candidates, highlighting the advantages of preserving the canal wall for improved device stability and reduced risk of infection, while also systematically integrating the assessment of CO - a cochlear implantation-specific challenge - into the decision framework. For extensive disease, our findings support the necessity of radical procedures like STP, which have proven to be safe and effective in cochlear implantation across multiple large-scale studies[3,5,6,10,17].
Based on our findings, we propose a simple, stepwise, imaging-based algorithm to guide surgical planning (Figure 1). Its primary utility lies in standardizing the initial assessment and providing a clear, visual framework for surgeons, especially trainees, when approaching complex cases. This structured approach can reduce cognitive load and decision fatigue by providing a clear starting point, thereby potentially reducing inter-surgeon variability. It is expected to also serve as an excellent educational tool for systematically evaluating key imaging features. This may help reduce inter-surgeon variability and serves as an excellent educational tool. We emphasize that this algorithm represents a pre
Preoperative HRCT plays a critical role in detecting CO, a common complication of longstanding COM[18,19]. As illustrated in case 2, HRCT reliably identifies severe ossification of the basal turn - a finding that directly influences surgical planning. Critically, the imaging-based grading of CO (grades I-III) directly informs the selection of specialized electrode insertion techniques[20]. For grade I ossification, surgery typically involves removing the fibroosseous tissue and proceeds with a standard cochleostomy anterior-inferior to the round window. In grade II cases, a 1-mm diamond burr is often used to drill through the ossified segment, remove fibrous tissue, and allow for full electrode insertion; if the round window is obstructed, an alternative scala vestibuli approach may be employed. For advanced grade III ossification, a cochlearostomy at the second turn or a more extensive drill-out of the basal and middle turns may be required to create a patent channel for electrode placement.
While HRCT excels in visualizing dense, established ossification, it may be less sensitive to subtle or early fibrotic changes. In such scenarios, MRI provides valuable supplementary information. The MRI protocol used in this study, including DWI and post-contrast T1-weighted sequences, serves a complementary diagnostic role: DWI is highly sensitive for detecting cholesteatoma (manifested as hyperintensity), aiding in its differentiation from other soft-tissue pathologies, such as cholesterol granuloma or granulation tissue. Future refinements of our imaging-based algorithm could incorporate specific MRI parameters to further improve soft-tissue characterization and diagnostic confidence. Although HRCT remains the primary modality for delineating bony anatomy and guiding surgical navigation, the combined use of HRCT and MRI enhances the overall preoperative assessment. Our findings underscore that a syste
This study has several limitations. Its retrospective design and small sample size (n = 12) may introduce selection bias and limit generalizability. Although no formal sample size calculation was performed - consistent with exploratory pilot studies - the limited cohort increases overfitting risk; thus, the reported perfect predictive metrics should be interpreted cautiously and validated in larger prospective cohorts. Second, despite excellent inter-rater agreement (κ = 0.85), the small sample precluded precise estimation of diagnostic performance, as reflected by wide CIs. Third, while HRCT provides essential anatomical guidance, clinical decision-making in COM also incorporates patient comorbidities, eustachian tube function, and surgical judgment - factors beyond the scope of this imaging-focused analysis. Fourth, although all patients underwent MRI (with known value for differentiating cholesteatoma), its features were not systematically integrated into the predictive model, which relied primarily on HRCT. Future studies combining HRCT and MRI parameters could improve preoperative stratification. Finally, the mean follow-up of 26.9-30.2 months allowed assessment of early complications and electrode stability but is insufficient to evaluate long-term outcomes, such as late infection, device survival, or cholesteatoma recurrence. Moreover, this study focused on establishing imaging criteria for surgical approach selection - a critical prerequisite for safe implantation. Prospective evaluation of auditory outcomes (e.g., speech discrimination scores) in future studies will further clarify the clinical benefits of this standardized planning strategy.
Preoperative HRCT provides robust, predictive information that appears essential for managing cochlear implantation in patients with COM. Our findings in this preliminary cohort highlight that the radiological extent of mastoid opacification is the most critical determinant for selecting conservative (CWU) or radical (CWD/STP) mastoidectomy. By synthesizing these findings into a practical decision-making framework, we can standardize surgical planning and better predict potential intraoperative challenges like CO. Prospective, multicenter studies are essential to validate and refine this pre
| 1. | Vincenti V, Pasanisi E, Bacciu A, Bacciu S, Zini C. Cochlear implantation in chronic otitis media and previous middle ear surgery: 20 years of experience. Acta Otorhinolaryngol Ital. 2014;34:272-277. [PubMed] |
| 2. | Vashishth A, Fulcheri A, Prasad SC, Dandinarasaiah M, Caruso A, Sanna M. Cochlear Implantation in Chronic Otitis Media With Cholesteatoma and Open Cavities: Long-term Surgical Outcomes. Otol Neurotol. 2018;39:45-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 3. | Morelli L, Damam SK, Yilala M H, Fancello G, Ferraro M, Caruso A, Sanna M. Subtotal petrosectomy in cochlear implantation: Gruppo Otologico experience after 348 cases. Eur Arch Otorhinolaryngol. 2025;282:5091-5099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 4. | Hunter JB, O'Connell BP, Wanna GB. Systematic Review and Meta-analysis of Surgical Complications following Cochlear Implantation in Canal Wall Down Mastoid Cavities. Otolaryngol Head Neck Surg. 2016;155:555-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 5. | Szymański M, Ataide A, Linder T. The use of subtotal petrosectomy in cochlear implant candidates with chronic otitis media. Eur Arch Otorhinolaryngol. 2016;273:363-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Canzi P, Berrettini S, Albera A, Barbara M, Bruschini L, Canale A, Carlotto E, Covelli E, Cuda D, Dispenza F, Falcioni M, Forli F, Franchella S, Gaini L, Gallina S, Laborai A, Lapenna R, Lazzerini F, Malpede S, Mandalà M, Minervini D, Pasanisi E, Ricci G, Viberti F, Zanetti D, Zanoletti E, Benazzo M. Current trends on subtotal petrosectomy with cochlear implantation in recalcitrant chronic middle ear disorders. Acta Otorhinolaryngol Ital. 2023;43:S67-S75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 7. | Yoon YH, Lee JB, Chung JH, Park KW, Kim BJ, Choi JW. Cochlear Implantation in Patients with Chronic Suppurative Otitis Media: Surgical Outcomes and a Management Algorithm. Audiol Neurootol. 2020;25:151-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | Yang H, Cao KL, Chen XW, Wang Y, Wei CG, Jin X, Wei BJ. [Cochlear implantation in patients with otitis media-related diseases]. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2006;41:908-912. [PubMed] |
| 9. | Jang JH, Park MH, Song JJ, Lee JH, Oh SH, Kim CS, Chang SO. Long-term outcome of cochlear implant in patients with chronic otitis media: one-stage surgery is equivalent to two-stage surgery. J Korean Med Sci. 2015;30:82-87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Polo R, Del Mar Medina M, Arístegui M, Lassaletta L, Gutierrez A, Aránguez G, Prasad SC, Alonso A, Gavilán J, Sanna M. Subtotal Petrosectomy for Cochlear Implantation: Lessons Learned After 110 Cases. Ann Otol Rhinol Laryngol. 2016;125:485-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 11. | Wong MC, Shipp DB, Nedzelski JM, Chen JM, Lin VY. Cochlear implantation in patients with chronic suppurative otitis media. Otol Neurotol. 2014;35:810-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Gao S, Jiang Y, Wang GJ, Li BC, Yuan YY, Gao B, Zhang D, Li J, Yu Q, Dai P. Cochlear implantation in patients with canal wall down mastoidectomy cavities. Acta Otolaryngol. 2018;138:993-997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 13. | Velasco G, Alshaikh H, Lee CM, Jeon MC, Han JS, Seo JH, Park SN. Surgical outcomes of simultaneous subtotal petrosectomy and cochlear implantation: a single institutional study. Eur Arch Otorhinolaryngol. 2025;282:1791-1799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Jeong Y, Hur YK, Choi JY, Kim SH, Moon IS, Jung J, Song CI. One-stage cochlear implantation in patients with chronic otitis media using canal wall up mastoidectomy. Acta Otolaryngol. 2021;141:354-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | De Foer B, Vercruysse JP, Pilet B, Michiels J, Vertriest R, Pouillon M, Somers T, Casselman JW, Offeciers E. Single-shot, turbo spin-echo, diffusion-weighted imaging versus spin-echo-planar, diffusion-weighted imaging in the detection of acquired middle ear cholesteatoma. AJNR Am J Neuroradiol. 2006;27:1480-1482. [PubMed] |
| 16. | Yan T, Zong F, Ma X, Xu X, Chen W, Song Z, Han X, Wang X, Zhang H. Cochlear implantation in patients with ossified cochleas. Am J Otolaryngol. 2019;40:183-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Prasad SC, Roustan V, Piras G, Caruso A, Lauda L, Sanna M. Subtotal petrosectomy: Surgical technique, indications, outcomes, and comprehensive review of literature. Laryngoscope. 2017;127:2833-2842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 18. | Kim CS, Chang SO, Lee HJ, Shim WS, Oh SH, Kim YH. Cochlear implantation in patients with a history of chronic otitis media. Acta Otolaryngol. 2004;124:1033-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Xenellis J, Nikolopoulos TP, Marangoudakis P, Vlastarakos PV, Tsangaroulakis A, Ferekidis E. Cochlear implantation in atelectasis and chronic otitis media: long-term follow-up. Otol Neurotol. 2008;29:499-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 20. | Berrettini S, Forli F, Neri E, Segnini G, Franceschini SS. Scala vestibuli cochlear implantation in patients with partially ossified cochleas. J Laryngol Otol. 2002;116:946-950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Isaacson B, Roland PS, Wright CG. Anatomy of the middle-turn cochleostomy. Laryngoscope. 2008;118:2200-2204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/