Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.117929
Revised: January 12, 2026
Accepted: March 12, 2026
Published online: April 26, 2026
Processing time: 116 Days and 17.2 Hours
Diabetes mellitus (DM) poses a high-risk factor for cardiovascular disease (CVD), not only increasing the susceptibility to CVD but also worsening the prognosis and increasing mortality. Daming capsule (DMC), a hypolipidemic drug inde
To reveal the effects and the protective mechanism of DMC combined with SGLT2i on DM with I/R-induced ferroptosis.
We administered continuous drugs interventions to diabetic mice, then established I/R mice model. The cardiac function of DM with I/R mice was significantly impaired, with notable downregulation of the ejection fraction (EF), fractional shortening (FS), and early to late diastolic transmitral flow velocity.
Our data identified that ferroptosis contributed to cardiac damage during DM with I/R. The systolic and diastolic functions of the DM + I/R group mice were significantly improved after DMC and SGLT2i intervention. Fur
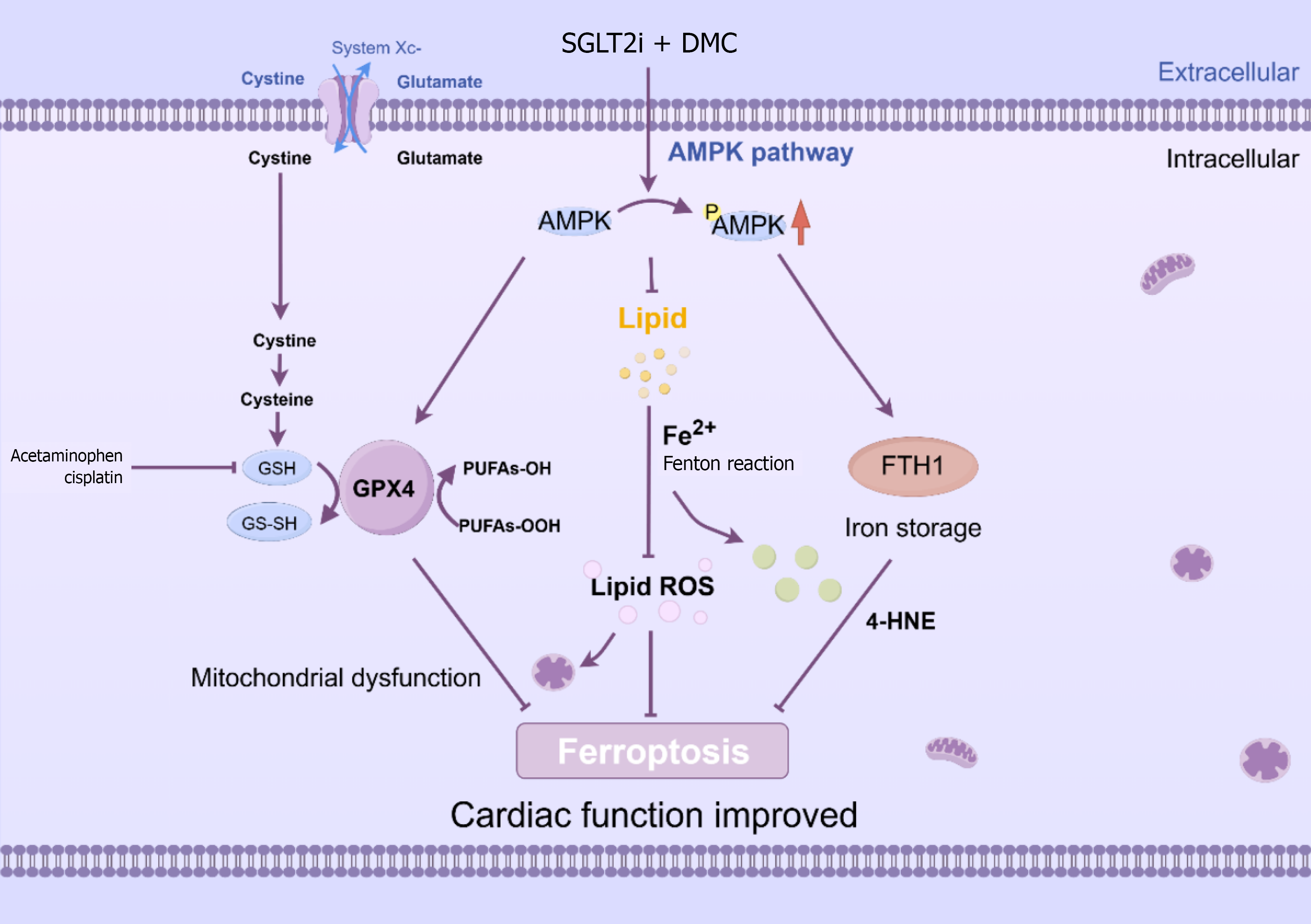
Mechanistically, it’s hypothesized that DMC and SGLT2i may suppress ferroptosis and attenuate cardiac dys
Core Tip: This study demonstrated the important role of ferroptosis in diabetes mellitus (DM) with myocardial ischemia-reperfusion (I/R) injury. For the first time, we advocated for an integrated Chinese and Western medicine approach to treating DM with I/R. Daming capsule and sodium-dependent glucose transporters 2 inhibitor reduced lipid peroxidation and iron deposition synergistically via adenosine monophosphate-activated protein kinase, thereby inhibiting ferroptosis and improving cardiac function.
- Citation: Yang X, Zhao YT, Liu H, Wang RX, Wu LY, Ye HW, Wen Y, Wang JX, Yu MX, Ma CX, Zhang XF, Wang LH. Daming capsule combined with SGLT2i confers protection against diabetes with myocardial ischemia/reperfusion injury induced ferroptosis via AMPK. World J Cardiol 2026; 18(4): 117929
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/117929.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.117929
According to the latest Global Diabetes Map, as of 2024, the cumulative number of adult Diabetes mellitus (DM) patients reached 589 million, and it was expected to increase to 853 million by 2050[1]. DM poses a high-risk factor for car
Previous foundational work has indicated that ferroptosis may play a pivotal role in the progression of DM with I/R. The dysregulation of lipid metabolism in the context of DM leads to the accumulation of polyunsaturated fatty acids. Moreover, I/R injury further promoted the generation of a large amount of reactive oxygen species (ROS) in car
Traditional Chinese medicine (TCM) is a rich resource treasure. As early as in the “Huangdi Neijing”, it’s mentioned that the superior doctor prevents illness before it occurs. TCM, due to its multi-target therapy approach and fewer side effects, has unique advantages in disease prevention and management, as well as in the regulation of chronic conditions. Many active ingredients of TCM have been proven to play an important role in preventing and treating CVD, such as berberine, matrine, sophoridine, resveratrol, and flavonoids from mistletoe. These medicines have shown efficacy in preventing myocardial damage[10]. Daming capsule (DMC), a hypolipidemic drug independently developed by our team, was composed of six Chinese herbal medicines: Rheum palmatum, Cassia obtusifolia L, Salvia miltiorrhiza, Panax ginseng C.A., Citri reticulatae pericarpium, and Poria cocos. Clinical trials found that DMC had cardioprotective effects in the treatment of hyperlipidemia. Our previous study demonstrated that DMC’s cardioprotective effects in diabetic rats were due to its ability to restore prolonged QT and PR intervals by increasing Kv4.2 expression and decreasing α1c subunit expression. Furthermore, DMC reduced serum lactate dehydrogenase (LDH) and creatine kinase (CK) activity, decreased infarction size in rats with myocardial infarction, and improved cardiac function by promoting mitophagy through the silent information regulator 1 (SIRT1)/adenosine monophosphate-activated protein kinase (AMPK) signaling pathway[11-13]. On the other hand, current evidence suggested that sodium-dependent glucose transporters 2 inhibitor (SGLT2i), a novel hypoglycemic drug, improved cardiac function in a short time.
DM is a chronic disease characterized primarily by glucose and lipid metabolism disorders. Addressing the problem of treating acute exacerbations of chronic diseases, we put forward the integration of Chinese and western medical treatments. It’s hypothesized that DMC combined with SGLT2i can reduce the accumulation of iron ions in cardio
C57BL/6J adult male (8 weeks old) mice were obtained from Beijing Viton Lever Laboratory Animal Technology Co Ltd (Charles River Laboratories, Beijing, China). Food and water were provided ad libitum under standard animal room conditions (temperature 23 ± 3 °C, humidity 30%-70%). All experimental procedures followed the Guidelines for the Care and Use of Laboratory Animals of Jinan University and were approved by the Ethics Committees of Jinan University (No. IACUC-20220512-06).
Following one week of acclimatization, DM model was established by using high-fat diet (60% fat, 20% carbohydrate, and 20% protein) for 8 weeks combined with streptozotocin (STZ, 50 mg/kg/day, Sigma-Aldrich, St Louis, MO, United States) injections for 5 consecutive days. After two weeks, mice that exhibited fasting blood glucose ≥ 11.1 mmol/L on two consecutive days were considered to have established diabetes and were included in the study. Subsequently, diabetic mice were divided into five groups: DM, DM + I/R, DM + I/R + SGLT2i, DM + I/R + DMC, DM + I/R + SGLT2i + DMC. Based on our previous research, we determined that 200 mg/kg/d was the optimal protective dose for DMC and collected extensive literatures to decide the dosage of empagliflozin. For the next three months, we continuously gavaged the mice with empagliflozin (10 mg/kg/day), DMC (200 mg/kg/day) and empagliflozin combined with DMC (empagliflozin 10 mg/kg/day, DMC 200 mg/kg/day) for 12 weeks[11,12,14,15]. Mice of the same age and sex main
After anesthesia, the chest was bluntly dissected at the fourth intercostal space and a surgical knot was tied on the left anterior descending coronary artery for 45 minutes of myocardial ischemia, then releasing the knot to allow for cardiac reperfusion. The mice were then returned to their cages for 24 hours before echocardiography and harvest.
Rat cardiomyocyte cell line (H9C2 cells) were cultured with complete medium which supplemented with 10% fetal bovine serum (FBS, Vivacell VC, Biological Industries, Israel) and 1% penicillin and streptomycin (Gibco, Carlsbad, CA, United States) in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Carlsbad, CA, United States). The cardiomyocytes were subjected to various treatments based on their group assignments [low glucose (5.5 mmol/L) or high glucose (50 mmol/L) and palmitic acid (0.2 mmol/L)] medium containing 10% medicated serum (blank group serum or DMC group serum) for 48 hours in the absence of FBS, and empagliflozin (5 μM) was added to the treatment group containing SGLT2i) when they reached 70% confluence. After 12 hours of treatment, cells were subjected to hypoxic condition (5% CO2 and 95% N2) for 12 hours, followed by reoxygenation for 24 hours to establish an in vitro I/R model hypoxic/reoxygenation (H/R)[16].
C57BL/6 mice were randomly classified into blank serum group and DMC serum group, which were treated with an equivalent volume of solvent or 200 mg/kg DMC twice a day for a total of seven times. Blood was obtained from the abdominal aorta 1 hours after the final administration and centrifuged at 2000 g for 15 minutes to separate serum from whole blood after standing for 2 hours. Serum was inactivated at 56 °C for 30 minutes, filtered through 0.22 μM microporous membrane, and stored at -20 °C[11,12,15,17].
Cardiac function was evaluated before and after I/R modeling using echocardiography (Vevo 3100 imaging system). Mice were anesthetized with isoflurane by inhalation (heart rate 400-500/minutes). After chest hair was removed, the animals were secured on a thermostatic heating pad. The chest was fully exposed and coupling agent was evenly applied, ultrasound images of the left ventricle and four-chambered heart of the mice were captured with a mouse-specific high-frequency ultrasound probe. Ejection fraction (EF%) and fractional shortening (FS%) were measured to assess myocardial systolic function, and early to late diastolic transmitral flow velocity was calculated to evaluate myocardial diastolic function.
The heart samples (1 mm × 1 mm × 1 mm) were immersed in the electron microscope fixative. They were post-fixed, embedded, cut and mounted at the electron microscope core facility (Servicebio, Wuhan, Hubei Province, China), then ultrathin sections were imaged employing Hitachi H-7800 transmission electron microscope (Hitachi, Tokyo, Japan).
Standard hematoxylin and eosin (HE) staining was performed using a hematoxylin-eosin staining kit (G1005, Servicebio, Wuhan, China). Specifically, cardiac paraffin sections were deparaffinized to water, stained sequentially with HE, and finally the sections were dehydrated and sealed. Imaging was conducted applying a TissueGnostics Strata FAXS P-S (TissueGnostics, Austria).
Standard Masson staining was carried out utilizing the Masson trichrome staining kit (G1006, Servicebio, Wuhan, China). Detailly, cardiac paraffin sections were deparaffinized to water, and placed in potassium dichromate overnight, then stained in order with hematoxylin, lachrymose red acid magenta, phosphomolybdic acid, and aniline blue, and finally dehydrated and sealed. Imaging was performed employing TissueGnostics Strata FAXS P-S.
Tissue sections were treated sequentially in xylene and ethanol and then rinsed with water. They were stained with a mixture of potassium ferrous hydride and hydrochloric acid, followed by diaminobenzidine solution, observing color development under the microscope. After rinsing, the sections were counterstained with hematoxylin, which restored the blue color after differentiation. After dehydration and removal in xylene, the sections were mounted. Imaging was performed employing TissueGnostics Strata FAXS P-S.
BODIPY 493/503 (life technologies) was diluted to a concentration of 10 mmol/L using dimethyl sulfoxide. Frozen cardiac tissue or H9C2 cells fixed with 4% paraformaldehyde for 20 minutes and then immersed in BODIPY 493/503 solution (tissue 1 mg/mL, cells 4 μM) for 30 minutes at 37 °C[18]. After washing 3 times with PBS, the stained droplets were observed using laser scanning confocal microscope (LSCM).
H9C2 cells were adhered to slides and treated differently, then fixed with 4% paraformaldehyde. After rinsing with PBS, cardiomyocytes were treated with 0.1% Triton X-100 for 8 minutes, followed by blocking with 10% normal goat serum for 1 hour at room temperature. Subsequently, the slides were covered with glutathione peroxidase 4 (GPX4, Proteintech Group, Inc, 67763-1-Ig, Wuhan, China), 4-hydroxynonenal (4-HNE) (Invitrogen, MA5-27570, Carlsbad, CA, United States) and incubated at 4 °C overnight. After rinsing with PBS, fluorescein 488 antibody (1:500) was added and incubated for 1 hour at room temperature. After rinsing with PBS, nuclei were stained with 4,6-diamidino-2-phenylindole (DAPI, Sigma-Aldrich, D9542, St Louis, MO, United States). Immunofluorescence was examined using LSCM (Leica, STELLARIS 8, Weizler, Germany).
JC-1 dye (Thermo Fisher Scientific Inc, T3168, Waltham, MA, United States) was applied to detect mitochondrial membrane potential (MMP) in H9C2 cells. JC-1 accumulates in mitochondria in a potential-dependent manner, leading to a shift in fluorescence emission from green (529 nm) to red (590 nm). H9C2 cells were stained with JC-1 dye (2 μM) and incubated for 30 minutes at 37 °C, 5% CO2. Observation was conducted using LSCM and fluorescence intensity was measured by ImageJ. Mitochondrial depolarization was presented by a decrease in the red/green fluorescence intensity ratio.
Lipid ROS levels in H9C2 cells were detected by lipid peroxidation sensor BODIPY™ 581/591 C11 (Thermo Fisher Scientific Inc, D3861, Waltham, MA, United States), excitation/emission maxima of 581/591 nm in reduced state, which shift to 488/510 nm upon oxidation. H9C2 cells were incubated with BODIPY™ 581/591 C11 (5 μM) for 30 minutes at 37 °C, 5% CO2. Changes in the green to red fluorescence ratio, reflecting lipid peroxidation. Ultimately, we employed LSCM observing, and applied ImageJ to calculate fluorescence intensity.
FerroOrange (Dojindo, F374, Kyushu Island, Japan), excitation/emission wavelengths of 532/580 nm, was applied to measure intracellular Fe2+ content according to the instructions. H9C2 cells were incubated with FerroOrange (1 μM) for 30 minutes at 37 °C, 5% CO2. Ultimately, we employed LSCM observing, and applied ImageJ to calculate fluorescence intensity.
LDH assay kit (Nanjing Jiancheng Bioengineering Research Institute, A020-2-2, Nanjing, China) were employed to detect serum LDH levels, following the manufacturer's instructions. And serum total cholesterol (TC), triglycerides, low-density lipoprotein (LDL) and high-density lipoprotein (HDL) were measured with respective assay kits (Nanjing Jiancheng Bioengineering Research Institute, A110-1-1, A111-1-1, A113-1-1, A112-1-1).
The cell viability was detected by Cell counting kit-8 (CCK8, Li Ji Bio, AC11 L054, Shanghai, China). H9C2 cells were inoculated at 1 × 104 cells/well in 96-well plate. CCK-8 reagent was added according to the manufacturer’s instructions, incubated at 37 °C for proper time (0.5-2 hours). The absorbance value at 450 nm was measured by an enzyme labeler, and the relative cell activity was calculated from the absorbance value.
Total protein samples from H9C2 cells and tissues were extracted using radio immunoprecipitation assay (RIPA) lysis buffer (Beyotime, P0013B, Beijing, China), supplemented with phenylmethylsulfonyl fluoride (PMSF) (RIPA: PMSF = 100:1, Beyotime, ST506, Beijing, China). Subsequently, Proteins were separated using sodium dodecyl-sulfate polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. The membranes were blocked with Tris-buffered saline with 5% milk and incubated overnight at 4 °C with primary antibodies, using β-actin for normalization. After washing with Tris-buffered saline containing Tween, the membranes were incubated with the secondary antibody for 1 hour at room temperature. Finally, the membranes were scanned and analyzed using a compatible developer.
The following antibodies were employed for Western blotting analysis: Β-actin (Boster, BM5422, Wuhan, China), ferritin heavy chain 1 (FTH1, Absci, AB32180, Oregon, United States), GPX4 (Proteintech Group, Inc, 67763-1-Ig, Wuhan, China), 4-HNE (Invitrogen, MA5-27570, Carlsbad, CA, United States), AMPK (Abcam, ab207442, Cambridge, United Kingdom), p-AMPK (Cell Signaling Technology, Inc, 2535 s, Boston, MA, United States).
All results in this experiment are presented as the mean ± SD. The comparison between two or more groups was conducted by t-test or one-way analysis of variance, and P-value of less than 0.05 was considered statistically significant. Data were analyzed using GraphPad Prism 9.4.1.
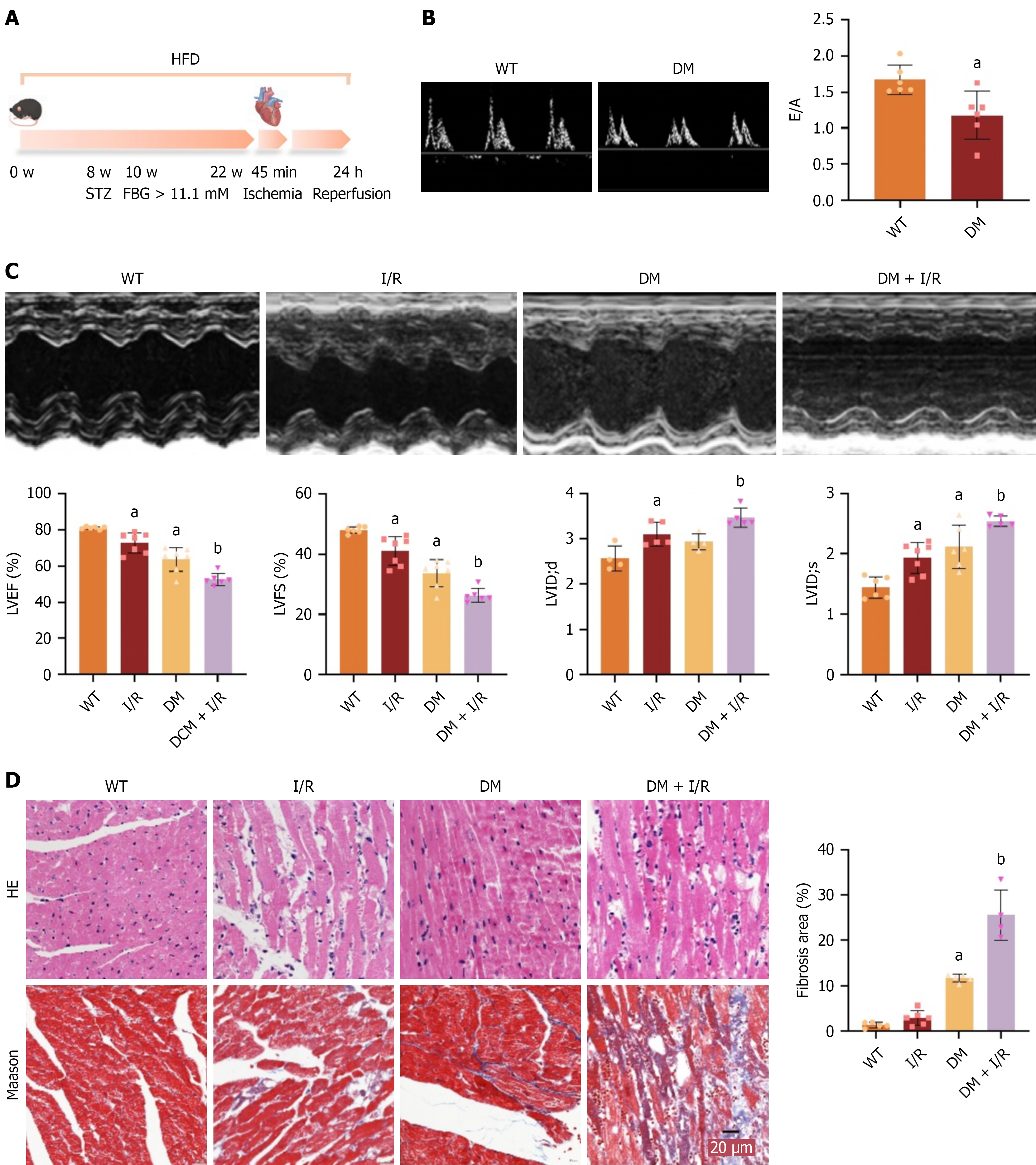
Three months after successfully establishing the DM model, we constructed the I/R model (Figure 1A). To validate the successful establishment of the model, echocardiography was utilized to assess cardiac function (Figure 1B). The mice in the DM + I/R group exhibited significantly impaired cardiac systolic and diastolic functions, characterized by a notable decrease in EF% and FS%, as well as a significant increase in LVID in diastole and LVID in systole compared to the DM group (Figure 1C). Consistently, immunohistochemistry of myocardial tissue revealed severe disarray of cardiomyocytes, inflammatory infiltration, and pronounced fibrosis in the DM + I/R group compared with the DM group (Figure 1D). These findings indicate that DM combined with I/R produces the most severe myocardial dysfunction.
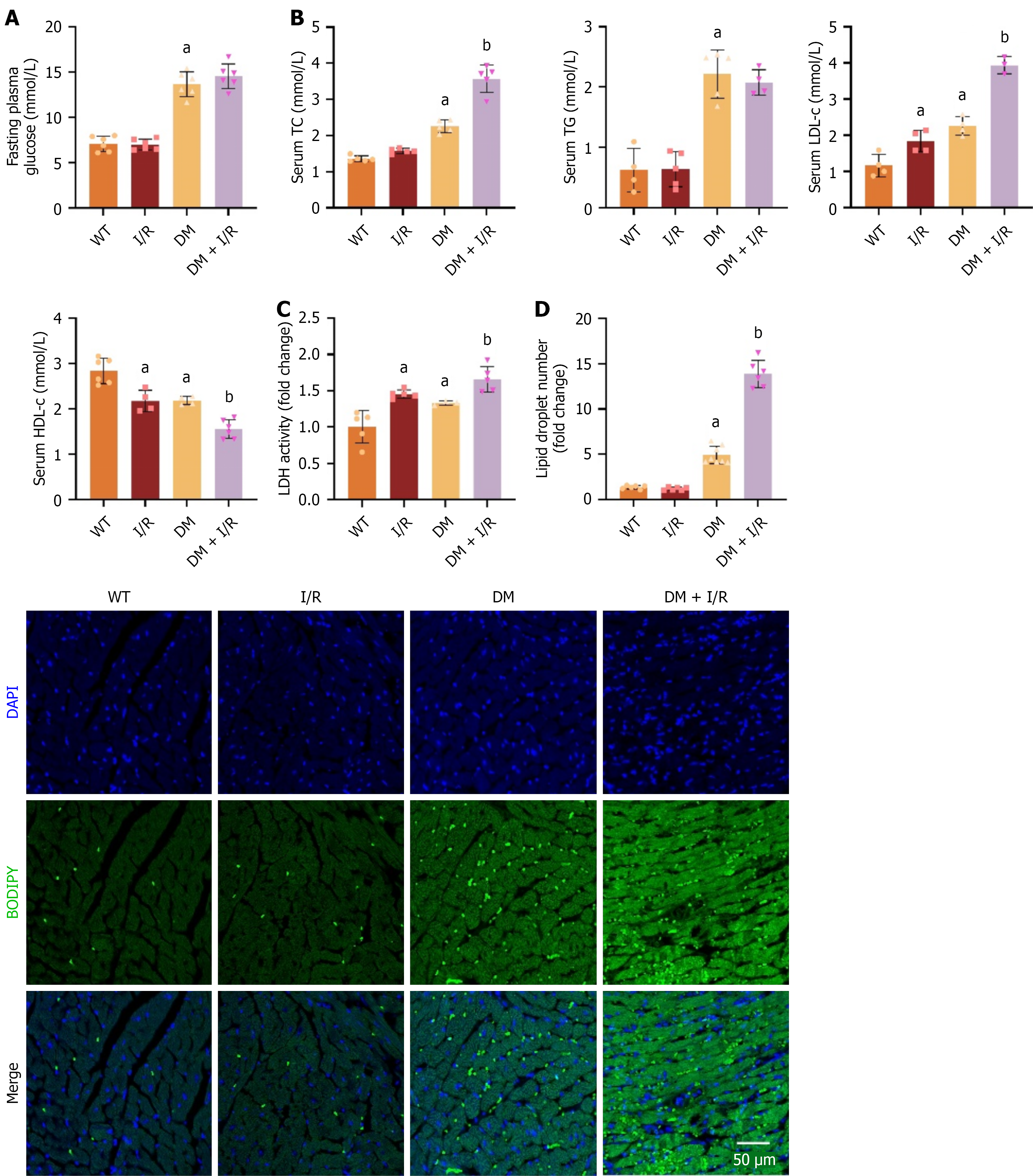
DM is a chronic condition characterized by disorders in glucose and lipid metabolism. The DM with I/R group exhibited severe metabolic disorders. The fasting blood glucose of the DM + I/R group was significantly higher than that of the wild type group, although there was no statistical difference compared with the DM group, there was a trend of increasing (Figure 2A). Similarly, blood lipids examination in the DM + I/R group revealed that TC, LDL increased and HDL decreased (Figure 2B). Upon myocardial injury, LDH was released into the circulatory system, with the most pronounced elevation in serum LDH activity observed in the DM + I/R group (Figure 2C). Additionally, lipid droplet storage within the DM + I/R cardiomyocytes was the highest, as demonstrated by BODIPY 493/503 staining (Figure 2D). On this basis, we conclude that DM with I/R aggravated metabolic abnormalities.
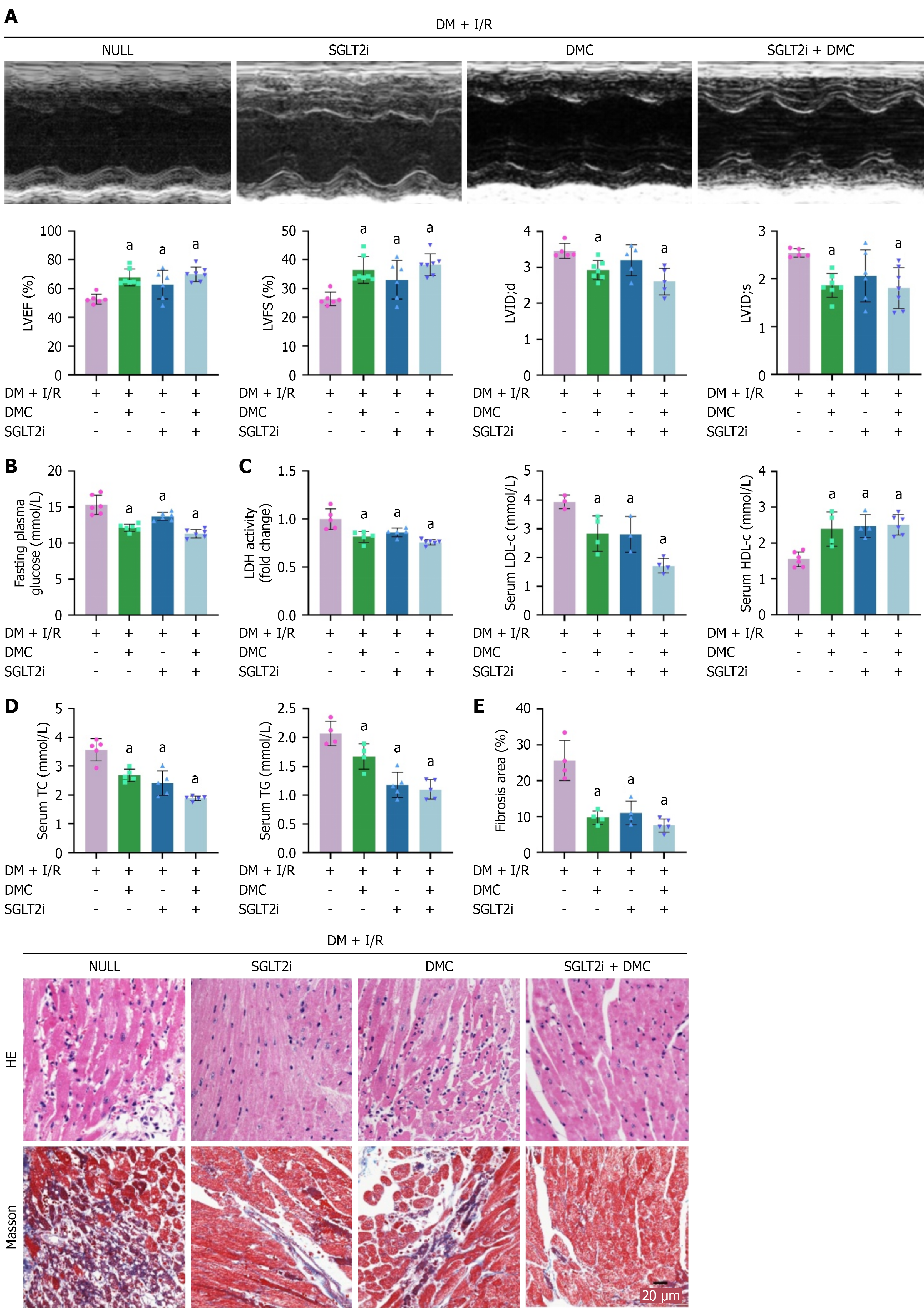
To investigate the protective effects of DMC combined with SGLT2i on DM with I/R, we initiated continuous gastric gavage for 3 months after establishing DM model, followed by the construction of the I/R model[16]. The echocardiographic results showed significant improvements in cardiac function in the treatment groups, as evidenced by increased EF% and FS%, along with decreased LVID at end-diastole and LVID at end-systole (Figure 3A). Of paramount im
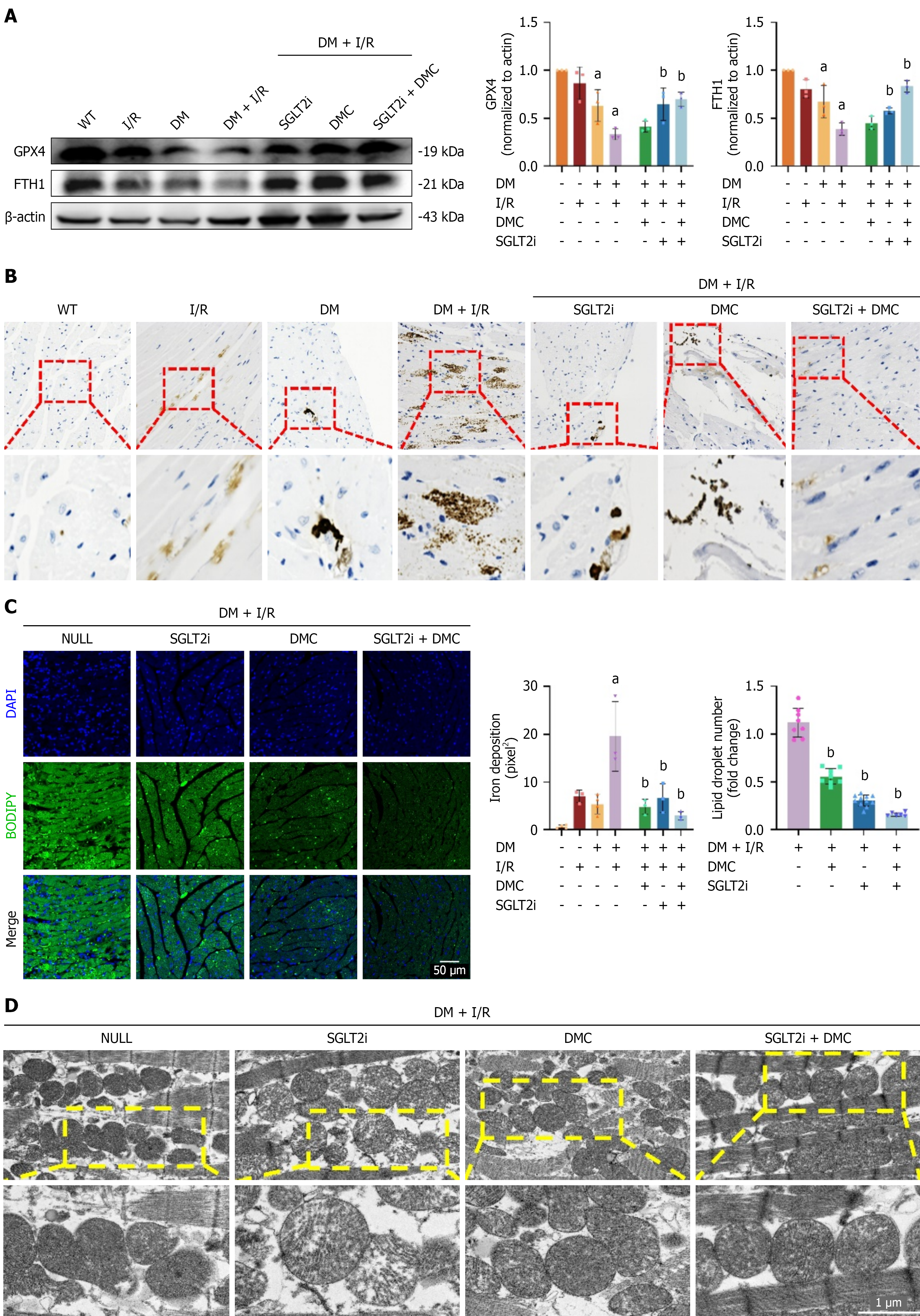
Based on emerging evidence, ferroptosis may play a pivotal role in the progression of DM and I/R, and it was considered as a principal mode of myocardial cell death[19-21]. In this study, we investigated whether DMC combined with SGLT2i inhibited the process of ferroptosis and ameliorated DM with I/R mice. GPX4, a key resistance factor against ferroptosis, employed reduced glutathion in a selenium-dependent manner to catalyze the reduction reaction of lipid peroxides[22]. FTH1, an iron storage protein, modulated intracellular Fe2+ concentration by reversibly sequestering free Fe2+, thereby mitigating the toxicity of Fe2+ caused by the production of ROS[23]. Initially, we observed the expression of GPX4 and FTH1 was significantly downregulated in the I/R group, DM group, and DM + I/R group, with the most pronounced decrease occurring in the DM + I/R group. After drug intervention, GPX4 and FTH1 expression was up-regulated, with the most pronounced increase observed in the combination-therapy group (Figure 4A). Next, we employed prussian blue staining to assess tissue iron deposition, and BODIPY 493/503 staining to detect the accumulation of lipid droplets within myocardial cells. The conclusion is consistent with the preceding results (Figure 4B and C). Additionally, transmission electron microscopy revealed that mitochondria appeared shrunken, their membranes were ruptured, matrix density was increased, and cristae were reduced or even absent. Following pharmacological intervention, there was a discernible enhancement in mitochondrial integrity, with most optimal mitochondrial morphology observed in the combination therapy group, as presented in Figure 4D. Overall, these results elucidated that SGLT2i combined with DMC impeded the onset of ferroptosis in diabetic mice experiencing myocardial I/R injury.
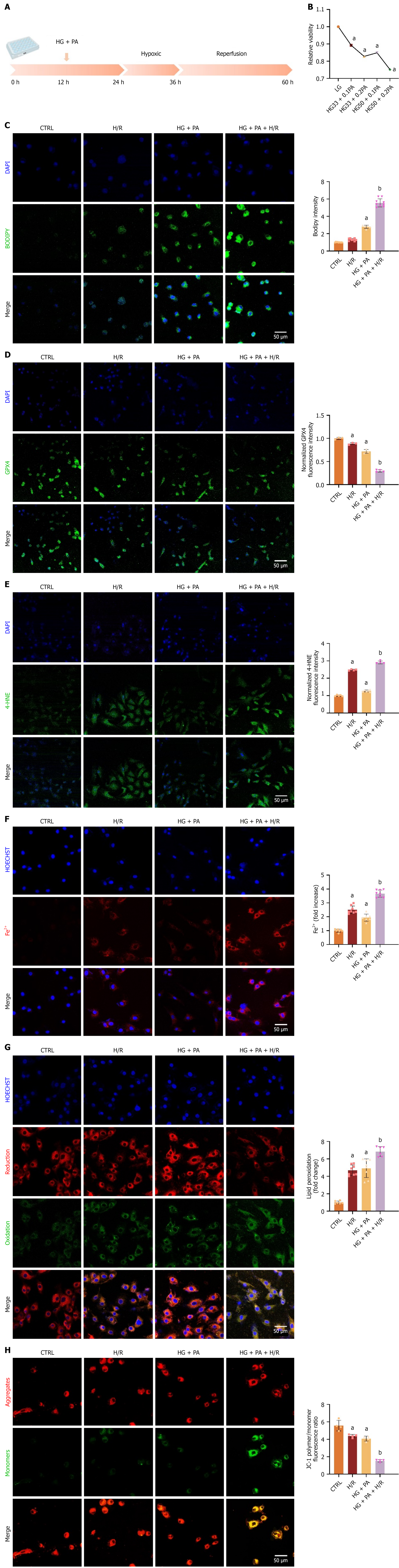
With substantial in-vivo evidence in hand, we next sought to corroborate the role of ferroptosis in vitro (Figure 5). First, using the CCK-8 assay, we selected 50 mmol/L glucose and 0.2 mmol/L palmitate as the high-glucose and high-lipid concentrations for subsequent experiments (Figure 5B). Following this, we simulated in vitro DM + I/R according to Figure 5A. BODIPY 493/503 staining detected the highest storage of H9C2 in DM + I/R group (Figure 5C). Immunofluorescence showed that GPX4 decreased greatly in the DM + I/R group (Figure 5D). 4-HNE, a prominent marker of lipid peroxidation during ferroptosis, accumulated substantially (Figure 5E)[24]. The accumulation of iron and lipid pero
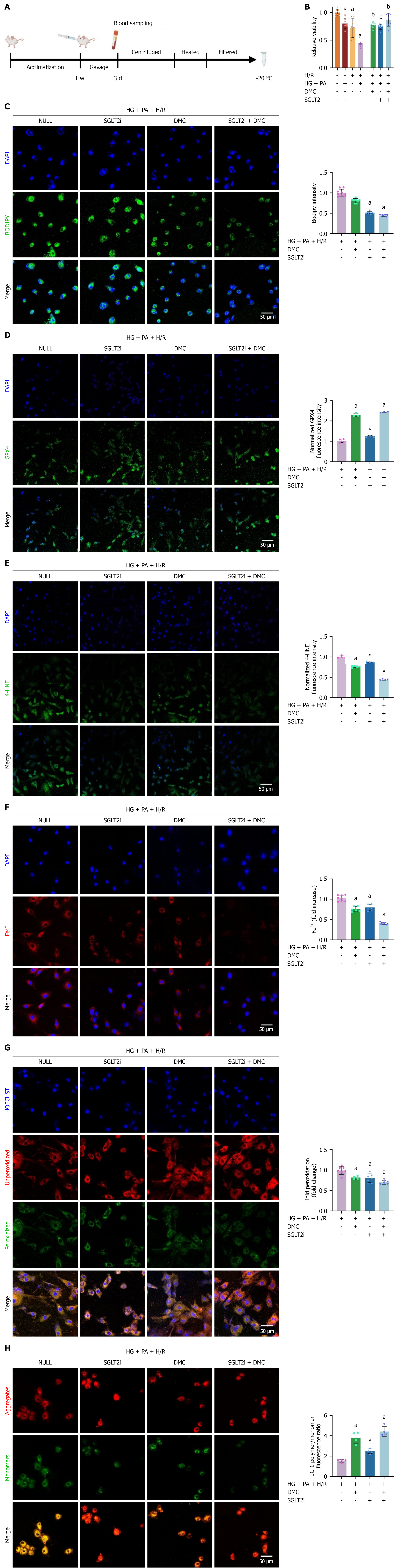
Initially, we extracted medicated serum and prepared culture medium for the further validation (Figure 6A). CCK-8 assay was then used to assess cell viability and evaluate the protective effect of the drug combination in vitro (Figure 6B). To further substantiate the ferroptosis inhibition of DMC and SGLT2i. We continued to utilize BODIPY 493/503 to examine the accumulation of lipid droplets. Immunofluorescence was used to detect GPX4, 4-HNE within H9C2 cells after drug intervention. The findings demonstrated that the combination therapy group markedly ameliorated DM with I/R in vitro (Figure 6C-E). Likewise, there was a significant reduction in iron accumulation and lipid peroxides (Figure 6F and G), and MMP increased subsequent to drug intervention (Figure 6H). In summation, the above results confirmed that DMC and SGLT2i effectively suppressed ferroptosis in vitro and mitigated DM with I/R. Notably, the combination drug group demonstrated the most optimal improvement effect.
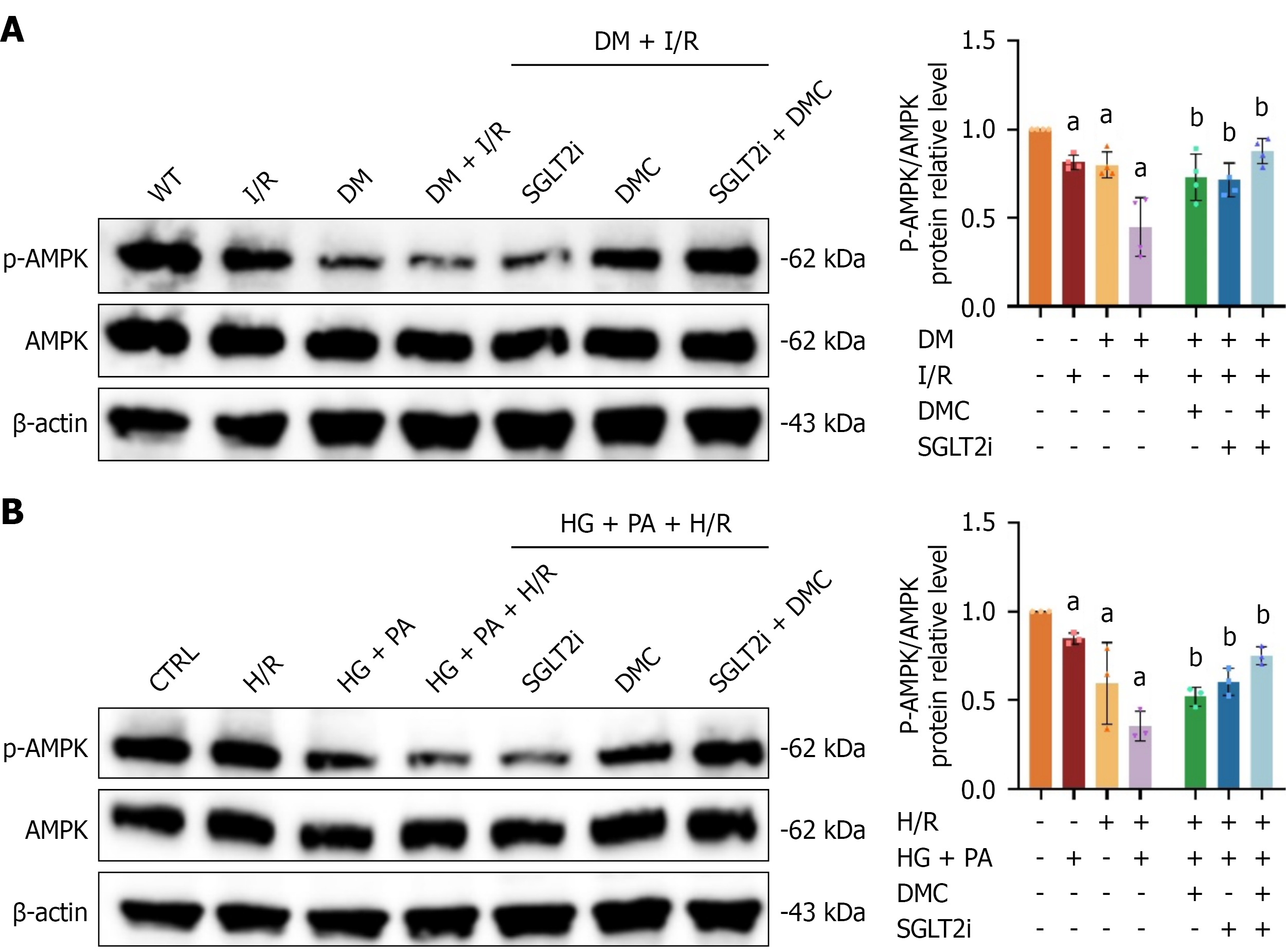
The core molecular mechanism of ferroptosis involves regulating the balance between oxidative damage and antioxidant defense[26]. Our team previously discovered that DMC reduced lipid levels through the AMPK pathway[11,12], de
DM is a systemic metabolic disease mainly characterized by glucose and lipid metabolism disorders. This condition can progressively lead to microvascular injury and macrovascular events, such as atherosclerosis and ischemia[33,34]. Currently, hypoglycemic drugs are inadequate for the prevention and treatment of CVD, with common side effects including weight gain, hypoglycemia, gastrointestinal reactions and urinary tract infections[35,36]. Hence, there is an urgent need to develop new, safe, and effective treatments for DM complicated with CVD. In our study, we embraced a holistic approach that integrated traditional Chinese and Western medicine. Our findings further solidified the important role of ferroptosis in the mechanism of DM with IR. It has been demonstrated that DMC combined with SGLT2i synergistically reduces lipid peroxidation and iron deposition via the AMPK signaling pathway, alleviates oxidative stress, and thereby inhibits ferroptosis and alleviates myocardial injury in DM with I/R.
A substantial body of studies illustrated that ferroptosis was closely related to the onset and progression of DM, myocardial infarction, I/R and heart failure. Recent studies indicated that ferroptosis was the predominant mode of cardiomyocyte death in the advanced stages of the disease[7,8,19]. In our study, we observed that DM with I/R exa
SGLT2i was recognized for providing short-term myocardial protection and was frequently used in combination with other drugs. At present, the clinical efficacy of SGLT2i in managing DM was well-established[27,39], and their role in preventing and treating I/R has garnered widespread attention[40,41]. Studies showed that Dapagliflozin can prevent I/R by upregulating SLC7A11/GPX4 and FTH1 via the MAPK signaling pathway, thereby inhibiting ACSL4 and ferroptosis[42]. Additionally, Canagliflozin was found to modulate cardiac ferroptosis in HFpEF rats through the AMPK/PGC-1α/Nrf2 pathway[43]. However, it remained unclear whether SGLT2i could enhance DM with I/R by suppressing ferroptosis. Our work demonstrated it.
TCM placed a priority on promptly treating symptoms and addressing the underlying causes when there was less urgency. DMC is renowned for its lipid-lowering effects, while SGLT2i are established anti-hyperglycemic drugs that offer immediate cardiac protection. The integration of Chinese and Western medical treatments focused on simultaneously addressing both symptoms and root causes, emphasizing both prevention and treatment. Our research demon
To sum up, this paper shed new light on the treatment of acute exacerbations of chronic diseases. We demonstrated that the combination therapy reduced lipid peroxidation and iron deposition synergistically through the AMPK signaling pathway, mitigated oxidative stress and thereby effectively inhibited ferroptosis induced by DM with I/R. This research boasted two strengths. On one hand, we advocated for an integrated Chinese and Western medicine approach to treating acute exacerbations of chronic disease-DM with I/R. This holistic treatment approach significantly enhanced both preventive and therapeutic efficacy. On the other hand, we elucidated the underlying mechanism of the combination therapy. There are some limitations in our study, which need to be further explored and confirmed. Moving forward, we will advance our research in two ways to improve it: By collecting clinical samples to further substantiate our experimental findings, or by conducting a comprehensive mechanistic investigation to identify the key target and further confirm them with relevant inhibitors, which will ultimately enhance our clinical treatment strategies.
Summarizing the key findings, our research revealed that DMC combined with empagliflozin reduced lipid peroxidation and iron deposition synergistically through the AMPK signaling pathway, thereby inhibiting ferroptosis and alleviating diabetes with I/R. The scholarly contributions of this research broaden the clinical application of novel drugs and provide a clear path toward the treatment of acute exacerbations of chronic diseases. In the future, we contend that a combination of traditional Chinese and Western medicine will be utilized to address conditions such as asthma, COPD, eczema, diabetic ketoacidosis and others.
We thank Guangzhou Key Laboratory of Basic and Translational Research on Chronic Diseases for providing the platform.
| 1. | Duncan BB, Magliano DJ, Boyko EJ. IDF Diabetes Atlas 11th edition 2025: global prevalence and projections for 2050. Nephrol Dial Transplant. 2025;41:7-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 145] [Article Influence: 145.0] [Reference Citation Analysis (2)] |
| 2. | Lejay A, Fang F, John R, Van JA, Barr M, Thaveau F, Chakfe N, Geny B, Scholey JW. Ischemia reperfusion injury, ischemic conditioning and diabetes mellitus. J Mol Cell Cardiol. 2016;91:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 190] [Article Influence: 19.0] [Reference Citation Analysis (5)] |
| 3. | Pluijmert NJ, Atsma DE, Quax PHA. Post-ischemic Myocardial Inflammatory Response: A Complex and Dynamic Process Susceptible to Immunomodulatory Therapies. Front Cardiovasc Med. 2021;8:647785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 4. | Das A, Salloum FN, Filippone SM, Durrant DE, Rokosh G, Bolli R, Kukreja RC. Inhibition of mammalian target of rapamycin protects against reperfusion injury in diabetic heart through STAT3 signaling. Basic Res Cardiol. 2015;110:31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 5. | Gao Y, Xu HY, Guo YK, Wen XL, Shi R, Li Y, Yang ZG. Impact of myocardial scars on left ventricular deformation in type 2 diabetes mellitus after myocardial infarction by contrast-enhanced cardiac magnetic resonance. Cardiovasc Diabetol. 2021;20:215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 6. | Heusch G, Rassaf T. Left Ventricular Unloading in Myocardial Infarction: Gentle Reperfusion Through the Backdoor? J Am Coll Cardiol. 2020;76:700-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 7. | Li W, Li W, Leng Y, Xiong Y, Xia Z. Ferroptosis Is Involved in Diabetes Myocardial Ischemia/Reperfusion Injury Through Endoplasmic Reticulum Stress. DNA Cell Biol. 2020;39:210-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 320] [Article Influence: 53.3] [Reference Citation Analysis (6)] |
| 8. | Huang Q, Tian H, Tian L, Zhao X, Li L, Zhang Y, Qiu Z, Lei S, Xia Z. Inhibiting Rev-erbα-mediated ferroptosis alleviates susceptibility to myocardial ischemia-reperfusion injury in type 2 diabetes. Free Radic Biol Med. 2023;209:135-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 9. | Fang X, Ardehali H, Min J, Wang F. The molecular and metabolic landscape of iron and ferroptosis in cardiovascular disease. Nat Rev Cardiol. 2023;20:7-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 804] [Article Influence: 268.0] [Reference Citation Analysis (8)] |
| 10. | Hao P, Jiang F, Cheng J, Ma L, Zhang Y, Zhao Y. Traditional Chinese Medicine for Cardiovascular Disease: Evidence and Potential Mechanisms. J Am Coll Cardiol. 2017;69:2952-2966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 455] [Article Influence: 50.6] [Reference Citation Analysis (0)] |
| 11. | Sun X, Han Y, Dong C, Qu H, Yu Y, Ju J, Bai Y, Yang B. Daming capsule protects against myocardial infarction by promoting mitophagy via the SIRT1/AMPK signaling pathway. Biomed Pharmacother. 2022;151:113162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 12. | Bian Y, Li X, Li X, Ju J, Liang H, Hu X, Dong L, Wang N, Li J, Zhang Y, Yang B. Daming capsule, a hypolipidaemic drug, lowers blood lipids by activating the AMPK signalling pathway. Biomed Pharmacother. 2019;117:109176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 13. | Ai J, Wang LH, Zhang R, Qiao GF, Wang N, Sun LH, Lu GY, Sun C, Yang BF. Protective effect of the daming capsule on impaired baroreflexes in STZ-induced diabetic rats with hyperlipoidemia. BMC Complement Altern Med. 2010;10:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 14. | Liu Y, Wu M, Xu B, Kang L. Empagliflozin Alleviates Atherosclerosis Progression by Inhibiting Inflammation and Sympathetic Activity in a Normoglycemic Mouse Model. J Inflamm Res. 2021;14:2277-2287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | Zhang Y, Li X, Li J, Zhang Q, Chen X, Liu X, Zhang Y, Zhang H, Yang H, Hu Y, Wu X, Li X, Ju J, Yang B. The anti-hyperglycemic efficacy of a lipid-lowering drug Daming capsule and the underlying signaling mechanisms in a rat model of diabetes mellitus. Sci Rep. 2016;6:34284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 16. | Zhang Y, Zhang X, Cai B, Li Y, Jiang Y, Fu X, Zhao Y, Gao H, Yang Y, Yang J, Li S, Wu H, Jin X, Xue G, Yang J, Ma W, Han Q, Tian T, Li Y, Yang B, Lu Y, Pan Z. The long noncoding RNA lncCIRBIL disrupts the nuclear translocation of Bclaf1 alleviating cardiac ischemia-reperfusion injury. Nat Commun. 2021;12:522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 17. | An R, Li Y, Li M, Bai Y, Lu Y, Du Z. Plasma pharmacochemistry combined with pharmacokinetics and pattern recognition analysis to screen potentially bioactive components from Daming capsule using ultra high performance liquid chromatography with electrospray ionization quadrupole time-of-flight mass spectrometry. J Sep Sci. 2015;38:1507-1514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Yu M, Du H, Wang B, Chen J, Lu F, Peng S, Sun Y, Liu N, Sun X, Shiyun D, Zhao Y, Wang Y, Zhao D, Lu F, Zhang W. Exogenous H(2)S Induces Hrd1 S-sulfhydration and Prevents CD36 Translocation via VAMP3 Ubiquitylation in Diabetic Hearts. Aging Dis. 2020;11:286-300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 19. | Cai W, Liu L, Shi X, Liu Y, Wang J, Fang X, Chen Z, Ai D, Zhu Y, Zhang X. Alox15/15-HpETE Aggravates Myocardial Ischemia-Reperfusion Injury by Promoting Cardiomyocyte Ferroptosis. Circulation. 2023;147:1444-1460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 338] [Article Influence: 112.7] [Reference Citation Analysis (4)] |
| 20. | Yang XD, Yang YY. Ferroptosis as a Novel Therapeutic Target for Diabetes and Its Complications. Front Endocrinol (Lausanne). 2022;13:853822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (1)] |
| 21. | Sha W, Hu F, Xi Y, Chu Y, Bu S. Mechanism of Ferroptosis and Its Role in Type 2 Diabetes Mellitus. J Diabetes Res. 2021;2021:9999612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 216] [Article Influence: 43.2] [Reference Citation Analysis (8)] |
| 22. | Xue Q, Yan D, Chen X, Li X, Kang R, Klionsky DJ, Kroemer G, Chen X, Tang D, Liu J. Copper-dependent autophagic degradation of GPX4 drives ferroptosis. Autophagy. 2023;19:1982-1996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 465] [Reference Citation Analysis (4)] |
| 23. | Kong N, Chen X, Feng J, Duan T, Liu S, Sun X, Chen P, Pan T, Yan L, Jin T, Xiang Y, Gao Q, Wen C, Ma W, Liu W, Zhang M, Yang Z, Wang W, Zhang R, Chen B, Xie T, Sui X, Tao W. Baicalin induces ferroptosis in bladder cancer cells by downregulating FTH1. Acta Pharm Sin B. 2021;11:4045-4054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 232] [Article Influence: 46.4] [Reference Citation Analysis (5)] |
| 24. | Liu L, Pang J, Qin D, Li R, Zou D, Chi K, Wu W, Rui H, Yu H, Zhu W, Liu K, Wu X, Wang J, Xu P, Song X, Cao Y, Wang J, Xu F, Xue L, Chen Y. Deubiquitinase OTUD5 as a Novel Protector against 4-HNE-Triggered Ferroptosis in Myocardial Ischemia/Reperfusion Injury. Adv Sci (Weinh). 2023;10:e2301852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 76] [Article Influence: 25.3] [Reference Citation Analysis (1)] |
| 25. | Tang D, Chen X, Kang R, Kroemer G. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31:107-125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3500] [Cited by in RCA: 3222] [Article Influence: 644.4] [Reference Citation Analysis (4)] |
| 26. | Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6378] [Cited by in RCA: 5754] [Article Influence: 1150.8] [Reference Citation Analysis (8)] |
| 27. | Zhang W, Lu J, Wang Y, Sun P, Gao T, Xu N, Zhang Y, Xie W. Canagliflozin Attenuates Lipotoxicity in Cardiomyocytes by Inhibiting Inflammation and Ferroptosis through Activating AMPK Pathway. Int J Mol Sci. 2023;24:858. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 69] [Reference Citation Analysis (5)] |
| 28. | Hu X, Li D, Chen W, Kuang H, Yang D, Gong Z, Long Y, Liu G, Wang K, Xia M, Xu Y, Yin Y. Sodium Glucose Transporter 2 Inhibitor Protects Against Heart Failure With Preserved Ejection Fraction: Preclinical "2-Hit" Model Reveals Autophagy Enhancement Via AMP-Activated Protein Kinase/Mammalian Target of Rapamycin Complex 1 Pathway. J Am Heart Assoc. 2025;14:e040093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 29. | Tadokoro T, Ikeda M, Ide T, Deguchi H, Ikeda S, Okabe K, Ishikita A, Matsushima S, Koumura T, Yamada KI, Imai H, Tsutsui H. Mitochondria-dependent ferroptosis plays a pivotal role in doxorubicin cardiotoxicity. JCI Insight. 2020;5:e132747. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 556] [Cited by in RCA: 510] [Article Influence: 85.0] [Reference Citation Analysis (5)] |
| 30. | Astudillo AM, Balboa MA, Balsinde J. Compartmentalized regulation of lipid signaling in oxidative stress and inflammation: Plasmalogens, oxidized lipids and ferroptosis as new paradigms of bioactive lipid research. Prog Lipid Res. 2023;89:101207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 66] [Reference Citation Analysis (1)] |
| 31. | Vamecq J, Dessein AF, Fontaine M, Briand G, Porchet N, Latruffe N, Andreolotti P, Cherkaoui-Malki M. Mitochondrial dysfunction and lipid homeostasis. Curr Drug Metab. 2012;13:1388-1400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 32. | Wang S, Han Y, Liu R, Hou M, Neumann D, Zhang J, Wang F, Li Y, Zhao X, Schianchi F, Dai C, Liu L, Nabben M, Glatz JFC, Wu X, Lu X, Li X, Luiken JJFP. Glycolysis-Mediated Activation of v-ATPase by Nicotinamide Mononucleotide Ameliorates Lipid-Induced Cardiomyopathy by Repressing the CD36-TLR4 Axis. Circ Res. 2024;134:505-525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 33. | Nauck MA, Wefers J, Meier JJ. Treatment of type 2 diabetes: challenges, hopes, and anticipated successes. Lancet Diabetes Endocrinol. 2021;9:525-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 205] [Article Influence: 41.0] [Reference Citation Analysis (4)] |
| 34. | Zhao M, Song L, Sun L, Wang M, Wang C, Yao S, Li Y, Yun C, Zhang S, Sun Y, Hou Z, Wu S, Xue H. Associations of Type 2 Diabetes Onset Age With Cardiovascular Disease and Mortality: The Kailuan Study. Diabetes Care. 2021;44:1426-1432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (5)] |
| 35. | McGovern A, Tippu Z, Hinton W, Munro N, Whyte M, de Lusignan S. Comparison of medication adherence and persistence in type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes Metab. 2018;20:1040-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 123] [Article Influence: 15.4] [Reference Citation Analysis (4)] |
| 36. | Tawulie D, Jin L, Shang X, Li Y, Sun L, Xie H, Zhao J, Liao J, Zhu Z, Cui H, Wen W. Jiang-Tang-San-Huang pill alleviates type 2 diabetes mellitus through modulating the gut microbiota and bile acids metabolism. Phytomedicine. 2023;113:154733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 55] [Article Influence: 18.3] [Reference Citation Analysis (1)] |
| 37. | Ai J, Yan X, Zhao L, Lu Y, Liang F, Cai B, Li G, Lu Y, Yang B. The protective effect of Daming capsule on heart function in streptozocin-induced diabetic rats with hyperlipidemia. Biol Pharm Bull. 2009;32:1354-1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 38. | He Y, Xi J, Fang J, Zhang B, Cai W. Aloe-emodin alleviates doxorubicin-induced cardiotoxicity via inhibition of ferroptosis. Free Radic Biol Med. 2023;206:13-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 49] [Reference Citation Analysis (1)] |
| 39. | Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Furtado RHM, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Sabatine MS. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2383] [Cited by in RCA: 2076] [Article Influence: 296.6] [Reference Citation Analysis (4)] |
| 40. | Chen R, Zhang Y, Zhang H, Zhou H, Tong W, Wu Y, Ma M, Chen Y. SGLT2 inhibitor dapagliflozin alleviates intramyocardial hemorrhage and adverse ventricular remodeling via suppressing hepcidin in myocardial ischemia-reperfusion injury. Eur J Pharmacol. 2023;950:175729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 41. | Nikolaou PE, Mylonas N, Makridakis M, Makrecka-Kuka M, Iliou A, Zerikiotis S, Efentakis P, Kampoukos S, Kostomitsopoulos N, Vilskersts R, Ikonomidis I, Lambadiari V, Zuurbier CJ, Latosinska A, Vlahou A, Dimitriadis G, Iliodromitis EK, Andreadou I. Cardioprotection by selective SGLT-2 inhibitors in a non-diabetic mouse model of myocardial ischemia/reperfusion injury: a class or a drug effect? Basic Res Cardiol. 2022;117:27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 42. | Chen W, Zhang Y, Wang Z, Tan M, Lin J, Qian X, Li H, Jiang T. Dapagliflozin alleviates myocardial ischemia/reperfusion injury by reducing ferroptosis via MAPK signaling inhibition. Front Pharmacol. 2023;14:1078205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 91] [Reference Citation Analysis (0)] |
| 43. | Ma S, He LL, Zhang GR, Zuo QJ, Wang ZL, Zhai JL, Zhang TT, Wang Y, Ma HJ, Guo YF. Canagliflozin mitigates ferroptosis and ameliorates heart failure in rats with preserved ejection fraction. Naunyn Schmiedebergs Arch Pharmacol. 2022;395:945-962. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 59] [Article Influence: 14.8] [Reference Citation Analysis (1)] |