Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.115712
Revised: December 2, 2025
Accepted: February 5, 2026
Published online: April 26, 2026
Processing time: 173 Days and 13.6 Hours
Metformin, a first-line therapy for type 2 diabetes mellitus (T2DM), has demon
To investigate the effectiveness of metformin in preventing T2DM among patients with CVD who do not have diabetes.
We searched PubMed, the Cochrane Central Register of Controlled Trials, and Scopus (from January 1, 2000 to July 31, 2024) for eligible randomized controlled trials (RCTs). A meta-analysis was conducted to evaluate the effect of metformin on the prevention of T2DM in patients with CVD and or coronary artery disease (CAD) without diabetes.
A total of 933 patients with CVD, all of whom had CAD, were included (470 in the metformin group and 463 in the control group). Fifty-six participants (11.9%) in the intervention groups and fifty-seven (12.3%) in the control groups developed T2DM. Patients with CAD receiving metformin showed no statistically significant difference in the development of T2DM compared with those not receiving metformin (odds ratio: 0.97; 95% confidence interval: 0.65-1.45; P = 0.89). Heterogeneity was rather low (Q = 2.38, P = 0.50; I2 = 0%, 95% confidence interval: 0%-84%), showing satisfactory results across studies; however, the overall quality of evidence was very low. The results remained non-significant in subgroup analyses restricted to: (1) Studies conducted in similar countries; (2) Studies with or without post-intervention follow-up; (3) Studies with a predominance of male or female participants; (4) Studies with a mean participant age above or below 60 years; (5) Studies including central adiposity as an ad
The administration of metformin does not appear to be effective in preventing the development of T2DM in non-diabetic patients with CAD. However, this finding is based on a limited number of small RCTs. Therefore, results should be interpreted cautiously, and further high-quality studies are needed before definitive clinical recommendations can be made.
Core Tip: This meta-analysis evaluated the role of metformin in preventing type 2 diabetes among non-diabetic patients with coronary artery disease. Across 933 participants from randomized controlled trials, metformin did not significantly reduce the incidence of diabetes compared to control. The findings were consistent across multiple subgroup analyses, including variations in age, gender, comorbidities, dosage, and follow-up duration. Despite low heterogeneity, the overall quality of evidence was very low, limiting the strength of conclusions. These results suggest that metformin should not be routinely used for diabetes prevention in non-diabetic coronary artery disease patients until more robust evidence from larger trials becomes available.
- Citation: Tsironikos GΙ, Zakynthinos GΕ, Kyprianidou D, Rammou V, Antonogiannis T, Bargiota A, Zakynthinos E, Tsolaki V. Metformin fails to prevent diabetes in non-diabetic cardiovascular patients: Systematic review and meta-analysis. World J Cardiol 2026; 18(4): 115712
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/115712.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.115712
Hyperglycemia may produce microvascular and macrovascular chronic disorders[1]. Particularly, cardiovascular disease (CVD) is the leading morbidity, and mortality cause in type 2 diabetes mellitus (T2DM)[2]. The global prevalence of T2DM continues to rise[3,4], with projections estimating 642 million people will be affected by T2DM by 2040[3], and T2DM-related deaths expected to reach 592 million by 2035[5].
Diabetes and CVD frequently coexist, a combination that significantly increases the risk of major adverse car
Prediabetes, defined as impaired fasting glucose (IFG), impaired glucose tolerance (IGT), or elevated hemoglobin A1c (HbA1c) (5.7%-6.4%), is a strong independent predictor of progression to T2DM[1]. Additional risk factors include a history of gestational diabetes mellitus (GDM), family history of diabetes in first-degree relatives, and age over 45 years[1]. Lifestyle and metabolic contributors-such as poor diet[4], physical inactivity, overweight/obesity, and central adiposity[9]-further elevate risk. Other associated clinical conditions include hypertension (HY) or use of antihypertensive therapy and hypertriglyceridemia (> 250 mg/dL)[1]. Importantly, CVD is recognized as an independent risk factor for incident T2DM[1], suggesting that CVD itself predisposes individuals to diabetes and thus complete a vicious, bidirectional, and potentially self-perpetuating relationship between the two conditions.
Preventing T2DM in high-risk populations is a pressing public health objective, particularly in light of the increasing global burden of diabetes and its complications. Metformin alone or combined with incretin-based therapies may induce secretion of glucagon-like-peptide-1 (GLP-1), reduce blood glucose and associated inflammation and oxidative stress, ameliorate body weight and improve coagulation mechanism enhancing cardioprotective benefit in patients with diabetes[10].
Metformin has undergone extensive research regarding its efficacy in delaying or preventing the progression from prediabetes to overt T2DM, especially among individuals with IFG, IGT, or metabolic syndrome. The Diabetes Prevention Program (DPP) and its long-term follow-up (DPPOS) showed that metformin significantly slowed the progression from prediabetes to T2DM, especially in younger, overweight people and women with a history of GDM[11,12]. This supports its use in high-risk groups. Moreover, several meta-analyses (MAs) have substantiated that metformin, whether ad
Given that CVD is recognized as an independent risk factor for incident T2DM, delaying the onset of diabetes in individuals with CVD who are not yet diabetic represents a clinically meaningful intervention. Τo our knowledge, no studies have systematically evaluated the efficacy of metformin in preventing T2DM specifically in patients with pre-existing CVD. In this study, we aimed to investigate the effect of metformin on the prevention of T2DM in non-diabetic individuals with CVD.
This study was pre-registered in the Open Science Framework (OSF) [Registration DOI 10.17605/OSF.IO/8XH4G (OSF, available online: https://archive.org/details/osf-registrations-8xh4g-v1)]. The systematic review was performed according to PRISMA extension guideline for complex interventions[16].
For eligible trials, we searched PubMed, Cochrane Library Central Register of Controlled Trials and Scopus (from January 1, 2000 to July 31, 2024). In our pre-registered protocol, we describe the performance of a broad search for interventions aiming at preventing T2DM in high-risk individuals (OSF). Available online: https://archive.org/details/osf-registrations-8xh4g-v1. The keyword that were used for the initial search were related to metformin, GLP-1 receptor agonists (GLP-1 RAs), sodium-glucose cotransporter 2 (SGLT2) inhibitors, diet or nutrition, exercise or physical activity, lifestyle and diabetes mellitus (Supplementary Table 1). The Cochrane collaboration search algorithm for randomized controlled trials (RCTs) was applied for the search in PubMed (Supplementary Table 1). The same keywords were used for the search in Cochrane Library Central Register of Controlled Trials and Scopus (Supplementary Table 1). Duplicates were removed by EndNote 21.1 software. Four investigators (Tsironikos GI, Zakynthinos GE, Kyprianidou D, Rammou V) performed the screening and detected potentially eligible trials after retrieving items full-text based on title, and/or abstract. A fifth researcher (Tsolaki V) contributed to the conclusion for studies that the four researchers (Tsironikos GI, Zakynthinos GE, Kyprianidou D, Rammou V) could not decide. Discrepancies were solved through consensus.
RCTs were accepted based on population, intervention, control, outcome approach. Trials were considered as eligible if they included non-diabetic patients with CVD receiving metformin vs not administration of metformin and reporting the diagnosis of T2DM by any method. Studies in non-English language, with pilot or feasibility design, from conference proceedings, considering antidiabetic regiments except for metformin and without T2DM events were excluded.
Four researchers (Tsironikos GI, Zakynthinos GE, Kyprianidou D, Rammou V) extracted the data. A fifth attributor (Tsolaki V) participated if required. The extracted items included the first author’s name, year of publication, country, type of RCT and numbers of centers or clusters if multicenter or cluster RCT, study duration, follow-up duration and drop-out rate. They also included the total sample size, specific characteristics of participants with CVD-coronary artery disease (CAD) such as gender, age and T2DM risk factors and characteristics of interventions such as dosage and duration, as well as the comparator arm. Finally, the number of patients that were analysed for new cases of T2DM by any diagnostic modality and any adverse event were recorded.
Four investigators (Tsironikos GI, Zakynthinos GE, Kyprianidou D, Rammou V) extracted items relevant to quality of eligible RCTs. A fifth investigator (Tsolaki V) contributed to the final decision, if necessary. To evaluate the quality of eligible trials, we used the revised Collaboration risk of bias tool proposed by Cochrane[17]. To rate the overall evidence, we used the Grading of Recommendations, Assessment, Development, and Evaluation tool (GRADEpro, version 3.6.1 McMaster University, Hamilton, Ontario, Canada, 2011).
The software of the Review Manager version 5.4.1 (Cochrane Collaboration, London, United Kingdom) and the Statistical Package for the Social Sciences version 26.0 (SPSS, Inc., Chicago, IL, United States) were used to perform our analyses. All available data were included in the main analyses. The overall level of statistical significance was set at P < 0.05 and for Cochran’s Q statistic at P < 0.1[18]. MA was performed to combine the events of T2DM. We assessed and measured heterogeneity between studies with the Cochran’s Q statistic (statistically significant for P < 0.1)[18] and the I2 index (< 25%, low; 25%-49%, moderate; 50%-75%, large; > 75%, very large), respectively[19]. Both fixed effects and random effects model of MA was performed. For large heterogeneity, the synthesis was performed by random effects model[19].
Subgroup analyses were performed based on studies’ characteristics (similar countries, follow-up duration), po
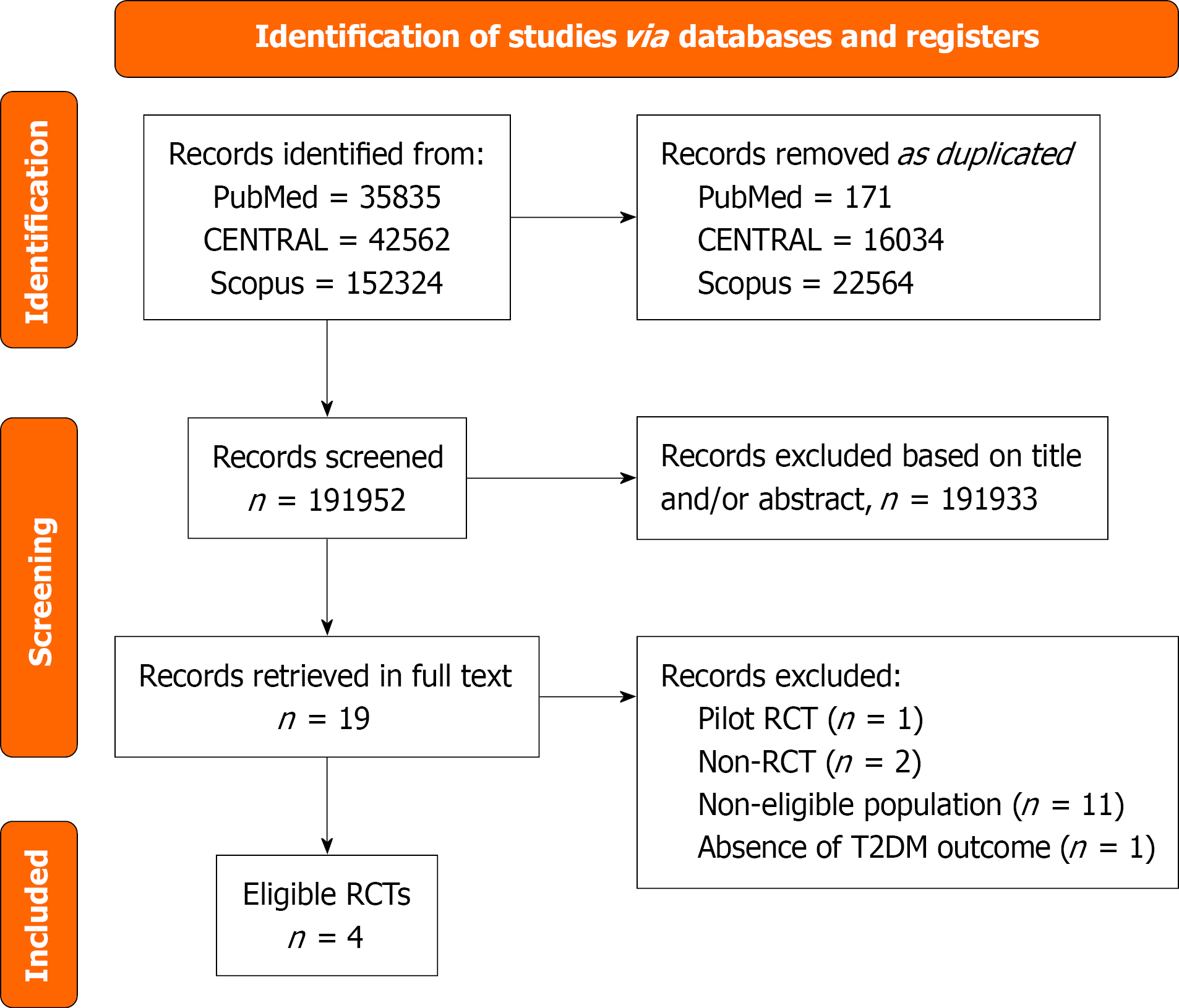
The initial broad search yielded 230721 items. Of 191952 items were assessed for potential eligibility, after removing 38769 duplicates. Of 191933 papers were excluded based on title and/or abstract. Then, we retrieved the remaining 19 papers in full text. Of 15 studies were excluded: Two of them had non-RCT design, one was pilot RCT, 11 did not include eligible population and one study did not report the outcome of T2DM. Finally, we accepted four RCTs (Figure 1).
The eligible RCTs were published from 2014 to 2019[22-25]. They all had a parallel design and were single-center performed[22-25]. All studies were performed in Europe[22-25]; two in Scotland, United Kingdom[22,25] and two in the Netherlands[23,24]. Studies’ duration varied between four and 28 months[22-25]. One study reported follow-up post-intervention duration of 24 months[24]. Two RCTs had no drop-out[23,24] and the other two had drop-out below 10%; 7.5%[22] and 7.4%[25], respectively (Table 1).
The total sample size was consisted of 966 patients[22-25]. Men participants were more than women in two studies[22,24] and two studies had more women participants than men[23,25]. Patients were all Caucasians in two RCTs[22-25]. Also, they comprised the majority of participants in the other two studies[23,24]. Both of them included minorities Asians and African ethnicities included living in Europe[23,24] (Table 2).
| Ref. | Sample size | Gender %, male/female | Mean age in years | Ethnicities (%) | Mean baseline BMI (I/ C) | Risk factors for T2DM | |
| Overall | Potential coexistence | ||||||
| Preiss et al[22], 2014 | 173 | 54.5/45.5 | 63.5 | Caucasian (100) | 30.2/30.5 | CHD, central adiposity, obesity | N/A |
| Lexis et al[23], 2015 | 346 | 13.0/87.0 | 58.1 | Caucasian (97), Asian (2.0), African (1.0) | 32.2/31.9 | CAD (STEMI), obesity | N/A |
| Hartman et al[24], 2017 | 379 | 75.0/25.0 | 58.1 | Caucasian (97.0), Asian (2.0), African (1.0) | 26.5/26.9 | CAD (STEMI), overweight | N/A |
| Mohan et al[25], 2019 | 68 | 47.0/53.0 | 64.5 | Caucasians (100) | 26.5/26.9 | CAD, overweight | Prediabetes, HY |
All participants had CAD[22-25]. Moreover, one study reported central adiposity as an additional risk factor[22]. The participants were obese in two trials[22,23] and overweight in the other two[24,25]. One RCT reported that prediabetes and/or HY may influence outcomes in some patients[24] (Table 2).
All studies reported metformin use in intervention arms[22-25]. No lifestyle modification was reported[22-25]. Metformin was compared to matching placebo in two RCTs[22,25]. Furthermore, metformin was applied to standard care compared to standard care in the other two studies[23,24]. The dosage of metformin varied between 1000 mg and 2000 mg daily[22-25]. Particularly, 1000 mg of metformin’s daily consumption were assessed in two studies[23,24], 1700 mg in one study[22] and 2000 mg in another study[25]. Two studies reported titration of drug[22,25]. Two studies had an intervention duration of four months[23,24] and the other two 12 months[22] and 18 months[25], respectively. In one of the studies with a four-month intervention period, an extended follow-up of 24 months was included[24]. Pill accountability was used to evaluate patients’ adherence in the intervention in one study[22]. Data regarding adherence were not reported in three RCTs[23-25] (Table 3).
| Ref. | Characteristics of compared arms | |||
| Component of compared arms | Dosage, description | Duration (month) | Assessment of adherence | |
| Preiss et al[22], 2014 | Metformin | 850 mg twice daily; initial dose 850 mg once daily for one week | 18.0 | Tablet counts of numbered bottles |
| Placebo | Matching placebo | |||
| Lexis et al[23], 2015 | Metformin plus standard care | Metformin 500 mg twice daily | 4.0 | NR |
| Standard care | ||||
| Standard care | Standard care | N/A | ||
| Hartman et al[24], 2017 | Metformin plus standard care | Metformin 500 mg twice daily | 4.0 | NR |
| Standard care | ||||
| Standard care | Standard care | N/A | ||
| Mohan et al[25], 2019 | Metformin | Metformin 1000 mg twice daily; initial dosage 500 mg twice daily for two weeks | 12.0 | NR |
| Placebo | Matching placebo | |||
T2DM was secondary outcome in all eligible RCTs[22-25]. Their primary outcomes were the progression of mean distal carotid intima-media thickness in patients with CAD[22], the cardiovascular risk (CV) profile[23] and the ventricular ejection fraction in patients with CAD[24] and the left ventricular hypertrophy in patients with CAD and prediabetes[25] (Table 4). T2DM diagnosis was set by either HbA1c alone[22,25], or combined with either fasting plasma glucose[23,24], or two hours 75 g oral glucose tolerance test[23] (Table 4).
| Overall effectiveness and safety of metformin | |||||
| Ref. | Outcome of T2DM assessed as primary or secondary | Participants at risk for T2DM (I/C) | Events of T2DM n (%) (I/C) | Diagnostic modalities | Total adverse events, side effects, n (%) (I/C) |
| Preiss et al[22], 2014 | Secondary | 86/87 | 2 (2)/6 (7) | HbA1c | Gastrointestinal symptoms 28 (32.6)/5 (5.7), CVD events 7 (8.1)/16 (18.4), newly diagnosed neoplasm 1 (1.0)/4 (5.0), deaths 1 (1.0)/0 (0.0) |
| Lexis et al[23], 2015 | Secondary | 162/156 | 20 (12.3)/18 (11.5) | FPG, 2 hours 75 g OGTT, HbA1c | NR |
| Hartman et al[24], 2017 | Secondary | 191/188 | 34 (17.8)/32 (17.0) | HbA1c | NR |
| Mohan et al[25], 2019 | Secondary | 31/32 | 0 (0)/1 (3.1) | FPG, HbA1c | Mild-to-serious gastrointestinal symptoms 24 (70.6)/19 (55.9), stroke 1 (2.9)/0 (0.0) |
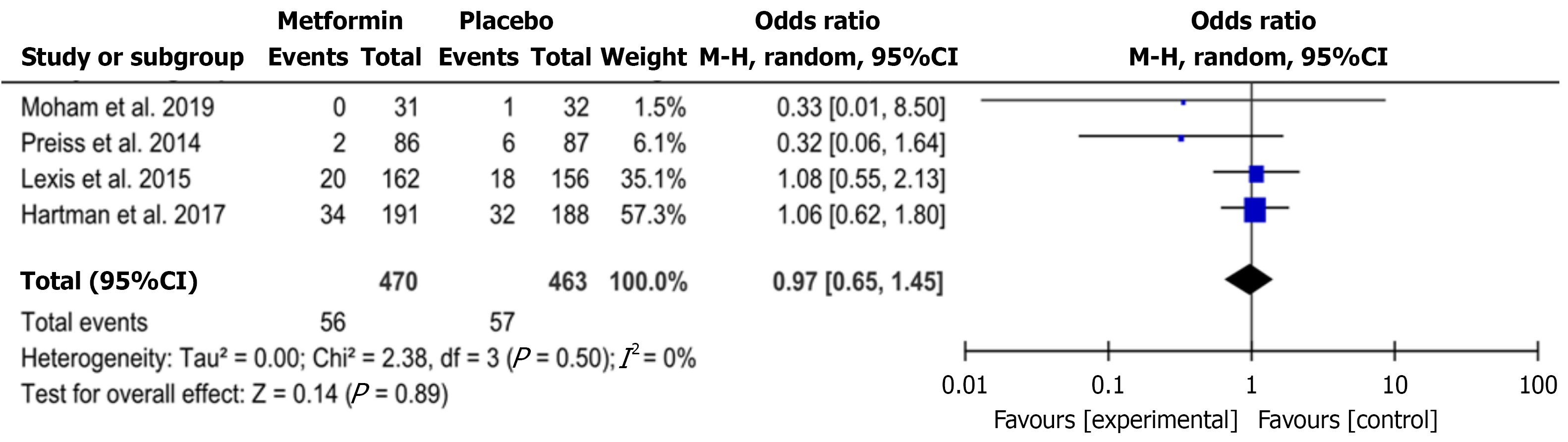
Effectiveness of metformin: A total of 933 patients with CAD (470 in metformin groups and 463 in control groups) were analysed for the outcome of diabetes[22-25] (Table 4, Figure 2). Fifty-six patients (11.9%) were diagnosed with T2DM in intervention and 57 (12.3%) in control group, respectively[22-25] (Table 4, Figure 2). The result of MA was non-significant (odds ratio: 0.97, 95% confidence interval: 0.65-1.45; P = 0.89) (Figure 2). Although Q statistic denotes absence of heterogeneity (Q = 2.38, P = 0.50), a large variability cannot be excluded as the upper limit of I2 is more than 50% (I2 = 0%, 95% confidence interval: 0%-84%) (Figure 2). Thus, the synthesis was performed with the random effects model (Figure 2).
Subgroup, sensitivity analyses and meta-regressions for the effect of metformin: Subgroup analyses were performed to assess heterogeneity across studies. They were based on similar countries of studies’ performance, adoption of follow-up post-intervention duration or not, genders’ propositions, mean age of participants, central adiposity as additional diabetes’ risk factor or not, baseline overweight or obesity, prediabetes and/or HY as potential coexisting risk factor or not, metformin’s daily dosage and intervention’s duration. However, there was not found significant results in any separate analysis (Supplementary Table 2).
Evaluating the effect of study with the largest sample size and with follow-up post-intervention[24] and of studies without drop-out[23,24] in sensitivity analyses, the result remained non-significant (Supplementary Table 2). Finally, meta-regressions including studies’ duration and baseline risk as covariates did not influence the summary odds ratio (Supplementary Table 3).
Safety: Adverse events, when reported, were mainly mild and related to gastrointestinal manifestations[22,25]. Other adverse events, probably not associated with metformin, included neoplasms, CVD events and deaths[22,25]. Two studies did not report adverse events[23,24] (Table 4).
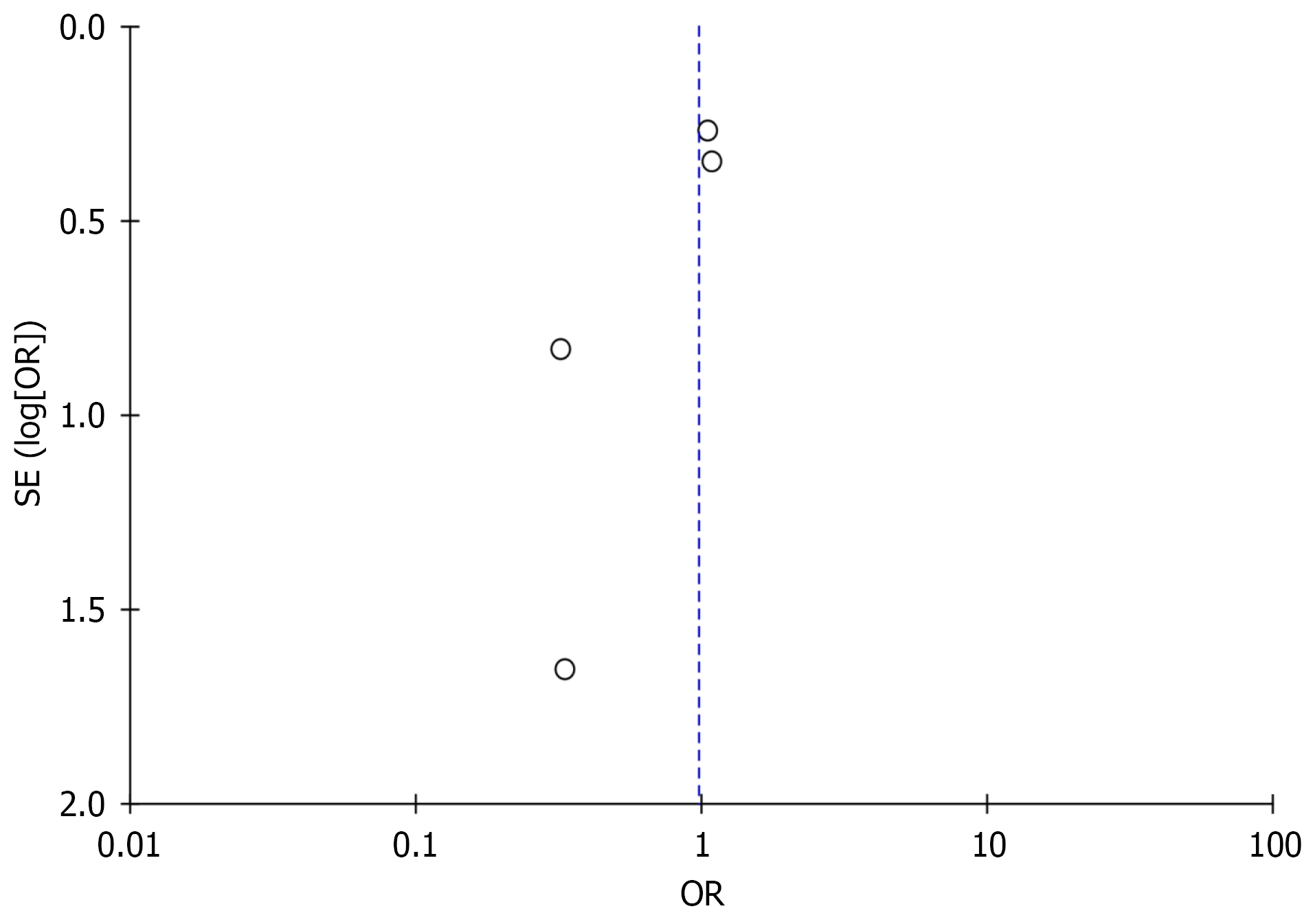
The risk of selection, performance, detection, attrition, reporting and other bias was assessed as low in all included studies[22-25] (Table 5). Performing Eggers’ test, we found an absence of potential publication bias (P = 0.127). However, publication bias cannot be excluded due to the absence of symmetry in funnel plot (Figure 3). The overall quality of evidence was very low (Table 6).
| Ref. | Random sequence generation (selection bias) | Allocation concealment (selection bias) | Blinding of participants and personnel (performance bias) | Blinding of outcome assessment (detection bias) | Incomplete outcome data (attrition bias) | Selective reporting (reporting bias) | Other bias |
| Preiss et al[22], 2014 | L | L | L | L | L | L | L |
| Lexis et al[23], 2015 | L | L | L | L | L | L | L |
| Hartman et al[24], 2017 | L | L | L | L | L | L | L |
| Mohan et al[25], 2019 | L | L | L | L | L | L | L |
| Metformin compared to placebo for T2DM prevention | ||||||
| Patient or population: Patients with CAD without T2DM; settings: Randomized controlled trials; intervention: Metformin; comparison: Placebo | ||||||
| Outcomes | Illustrative comparative risks1 (95%CI) | Relative effect (95%CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Placebo | Metformin | |||||
| T2DM | Study population | OR: 0.97 (0.65-1.45) | 933 (4 studies) | Very low | ||
| 123 per 1000 | 120 per 1000 (84-169) | |||||
To the best of our knowledge, this is the first MA to systematically evaluate the efficacy of metformin in preventing T2DM specifically in patients with pre-existing CVD and/or CAD; CVD is widely recognized as a significant risk factor for the development of T2DM[1].
Given that T2DM is considered the most significant risk factor for the development of CVD, particularly CAD, it is important to consider how detrimental it would be for a patient with established CAD to subsequently develop T2DM. Equally important is the question of how beneficial it could be to prevent or delay the onset of T2DM in these patients. Therefore, preventing T2DM, especially in individuals who already have CAD, is of critical importance, as the development of diabetes would introduce an additional major CV risk factor, further compounding their disease burden. However, the results were less favorable than desired, as this study found no statistically significant benefit of metformin for the prevention of T2DM in patients with CAD.
In T2DM, chronically elevated blood glucose, insulin resistance, dyslipidemia, HY, and low-grade inflammation act synergistically to damage both small and large blood vessels, resulting in a wide spectrum of vascular complications. From a CV perspective, patients with T2DM have a two- to four-fold higher risk of developing CAD, myocardial infraction (MI), stroke, and peripheral arterial disease[26]. In addition, heart failure (HF) is among the most common and severe CV complications of T2DM[25]. Overall, CVD accounts for a substantial proportion of mortality in this population[26]. These individuals experience higher rates of morbidity and premature mortality, with over one-third of deaths occurring before the age of 60 years[3]. Hyperglycemia and insulin resistance contribute to endothelial dysfunction, oxidative stress, and accelerated atherosclerosis. Additionally, diabetic cardiomyopathy-independent of underlying CAD-plays a key role in the pathogenesis of HF in patients with T2DM[27].
During the past few years, a growing body of evidence from large cohort studies, mechanistic investigations, and long-term observational analyses has highlighted the pleiotropic effects of metformin beyond glycemic control. Metformin is increasingly recognized as a multifaceted agent that acts through metabolic, inflammatory, and vascular pathways. In patients with prediabetes, it has been shown to improve markers of endothelial dysfunction, including soluble intercellular adhesion molecule-1, soluble vascular cell adhesion molecule-1, and von Willebrand factor[28]. Its anti-inflammatory effects are largely mediated through activation of adenosine monophosphate-activated protein kinase (AMPK), which suppresses pro-inflammatory signaling pathways[29,30]. Through combined mitochondrial actions and AMPK activation, metformin also enhances antioxidant defenses and reduces the production of reactive oxygen species[29,31]. Additionally, metformin has been shown to reduce triglyceride levels, low-density lipoprotein (LDL) cholesterol, and total cholesterol, likely through AMPK-mediated inhibition of hepatic lipogenic enzymes[31,32].
Metformin also exerts modest weight-reducing effects, in part by increasing circulating levels of growth differentiation factor-15, a hormone that suppresses appetite and reduces food intake. These effects appear to be reinforced by gut-brain and intestinal-brown adipose tissue signaling pathways[33].
The CV benefits of metformin in T2DM have been extensively documented over the past three decades, beginning with the landmark United Kingdom Prospective Diabetes Study. In this pivotal trial, metformin use over a median follow-up of 10.7 years was associated with a 32% reduction in any diabetes-related endpoint (P = 0.002), a 42% reduction in diabetes-related mortality (P = 0.017), and a 36% reduction in all-cause mortality (P = 0.011) among overweight patients with T2DM[34]. These findings have since been corroborated by multiple subsequent studies, which consistently report reductions in diabetes-related complications and mortality among metformin-treated patients[35,36]. More recently, a nationwide retrospective cohort study demonstrated a significantly lower incidence of acute MI among patients with T2DM receiving metformin in real-world clinical settings[37]. Supporting observational evidence has further revealed sustained reductions in MACEs, CV mortality, and all-cause mortality with metformin therapy over follow-up periods extending up to 10 years[38].
While the CV protective effects of metformin are well-established in patients with T2DM, emerging evidence suggests that similar benefits may extend to non-diabetic individuals[39]. However, findings from RCTs in this population have been mixed. Early studies reported no significant effect of metformin on surrogate markers of atherosclerosis, such as carotid intima-media thickness, or on MACEs in patients with established CAD who were already receiving statin therapy[22]. Similarly, trials involving non-diabetic individuals-including those with a history of myocardial infarction but without overt diabetes-did not demonstrate a clear CV benefit, potentially due to limitations such as small sample sizes and short follow-up durations[40,41].
Among the studies included in our MA involving patients with CAD but without diabetes-designed to evaluate whether metformin could improve cardiovascular outcomes and secondary reduce the incidence of diabetes-a few key findings merit attention. Preiss et al[22] and Hartman et al[24] both reported that metformin conferred no significant CV benefit in non-diabetic individuals with established CAD. Preiss et al[22] found no improvement in CV outcomes among patients with CAD and central adiposity[22], while Hartman et al[24] observed no significant reduction in cardiovascular events in patients with ST-elevation MI (STEMI). Moreover, Hartman et al[24] noted a non-significant reduction in carotid plaque progression in the metformin-treated group. Lexis et al[23] assessed the effects of a four-month course of metformin in patients post-STEMI and found a modest improvement in CV risk markers, including reductions in total and LDL cholesterol, despite concurrent optimal statin therapy.
Further supporting potential CV benefit, Mohan et al[25] demonstrated that 12 months of metformin administered at a daily dose of 2000 mg significantly reduced left ventricular hypertrophy and left ventricular mass in patients with CAD and insulin resistance and/or prediabetes. Metformin was also associated with reductions in blood pressure, body weight, and oxidative stress. However, no significant differences were observed between the metformin and placebo groups in terms of HbA1c or fasting insulin resistance index at the end of the study.
Independent of diabetic status, Dludla et al[42] demonstrated in their systematic review that patients with HF receiving metformin treatment may experience improved myocardial perfusion, characterized by reduced oxygen consumption, lower levels of LDL cholesterol, and decreased concentrations of N-terminal pro-brain natriuretic peptide. Furthermore, in patients with HF and insulin resistance without overt diabetes, metformin has been associated with enhanced myocardial efficiency, primarily through reductions in myocardial oxygen demand[43].
The 2025 American Diabetes Association Standards of Care continue to recommend metformin as first-line therapy for patients without renal impairment, though SGLT-2 inhibitors and GLP-1 RAs are prioritized for those with high CV or renal risk[44]. Similarly, the 2023 European Society of Cardiology guidelines recognize metformin’s metabolic and weight-related benefits but favor newer agents in patients with established CVD[45].
However, emerging evidence suggests that metformin may offer CV comparable to SGLT-2 inhibitors and GLP-1 RAs[46]. This is supported by real-world data, including a nationwide cohort study that demonstrated lower rates of acute MI among metformin users[37], as well as an intensive care unit study that reported better outcomes after cardiac surgery[47]. Moreover, in a comparative analysis, Wong et al[46] found that both metformin and SGLT-2 inhibitors reduced all-cause and CV mortality in T2DM patients with CVD, but metformin was linked to a slightly greater estimated survival gain (23.26 months vs 23.04 months) and a lower hazard ratio (HR) for all-cause mortality (HR: 1.308, 95% confidence interval: 1.103-1.550). In the same context, in prediabetic patients with obesity, the GLP-1 RA exenatide was not superior to metformin in improving endothelial function[48].
Halting progression to T2DM is vital given its global burden and complications. Metformin has been shown to delay-or even prevent-the transition from prediabetes to overt T2DM, particularly in individuals with IFG, IGT, or metabolic syndrome[49]. A landmark multicenter randomized trial involving 3234 participants with IFG and IGT demonstrated that, over 2.8 years, intensive lifestyle modification reduced diabetes incidence by 58%, while metformin reduced it by 31% (both P < 0.001)[49]. Metformin’s preventive effect was most pronounced in participants under 60 years of age, those with a body mass index ≥ 35 kg/m2, and women with a history of GDM, supporting its targeted use in these subgroups. Long-term results from the DPPOS showed sustained benefit. After 15 years, diabetes incidence was reduced by 27% in the lifestyle group and 18% in the metformin group, compared to placebo[50]. These benefits persisted through 21 years of follow-up[12].
Multiple MAs reinforce metformin’s preventive efficacy. An earlier analysis confirmed metformin’s ability to reduce T2DM risk in high-risk individuals[13]. More recently, a MA of 2720 participants found that adding metformin to lifestyle intervention significantly reduced T2DM incidence and improved HbA1c and fasting glucose at 12 months, though no significant differences were observed in secondary outcomes such as blood pressure, lipid levels, or body weight[14].
A 2023 systematic review highlighted metformin’s greatest preventive effect in younger individuals, those with elevated body mass index, and women with a history of GDM, consistent with DPP findings[15]. Additionally, a recent MA confirmed that metformin reduced T2DM incidence by 23% in high-risk adults and 25% in those with prediabetes[8]. The most substantial benefits were observed with treatment durations ≥ 18 months and doses around 1700 mg/day. Combination therapy with lifestyle modification further amplified the effect, lowering diabetes incidence by up to 52% compared with standard care-even at lower metformin doses[8].
In contrast to what might have been anticipated based on prior data, this MA found no significant effect of metformin in preventing the onset of T2DM in patients with CAD but without established diabetes. However, several critical limitations of the included studies may have contributed to the neutral result in our analysis and must be taken into account.
First, the total number of eligible RCTs was small (n = 4), and their cumulative sample size (n = 933) was limited. The small number of included patients significantly restricts the statistical power of the analysis, increasing the risk of type II error and the potential to overlook a modest yet clinically meaningful effect.
Second, the duration of intervention was also very short in two studies-only four months in the trials by Lexis et al[23] and Hartman et al[24], and just 12 months in Mohan et al[25]. It is unlikely that such brief interventions would be sufficient to detect a meaningful reduction in diabetes incidence. In contrast, trials that demonstrated preventive benefits of metformin in individuals with IFG or IGT-such as the DPP and its DPPOS-included significantly larger populations and longer treatment periods, often exceeding 2-3 years[49,50].
Third, the design of the included trials and the low quality of evidence may have limited the ability to detect preventive effects. In all the studies included in our MA, diabetes prevention or delay was only a secondary outcome, not the primary endpoint, with primary outcomes typically focused on cardiovascular or surrogate imaging endpoints. No RCT has yet assessed the prevention of T2DM as a primary outcome specifically in patients with CAD but without diabetes receiving metformin, limiting the rigor of event ascertainment and follow-up for this endpoint. This is in contrast to two recent trials involving newer oral antidiabetic agents, in which the prevention of T2DM was designated as a primary endpoint[51,52]. Both studies included large populations and had extended treatment durations, contributing to the strength of their findings. In the SELECT trial by Kahn et al[51], semaglutide was evaluated in 17604 individuals with established CVD and overweight/obesity but without diabetes. After approximately three years of follow-up, sema
Additional factors that may have influenced the neutral findings of our analysis include heterogeneity in metformin dosing (ranging from 1000 mg/day[23,24] to 2000 mg/day[25]). Notably, a recent MA by Tsironikos et al[8] showed that metformin reduced T2DM incidence by 25% in individuals with prediabetes. The strongest effects were observed with treatment durations of at least 18 months and daily doses of 1700 mg, particularly when combined with lifestyle in
As suggested by the newer antidiabetic agents discussed above, the reduction in diabetes incidence appears to be closely linked to improvements in cardiometabolic outcomes-particularly weight loss[4,52]. Metformin also demonstrated favorable effects on cardiometabolic profiles in the studies included in our MA. Specifically, reductions in body weight, body fat, waist circumference, and subcutaneous adipose tissue were observed in trials where these parameters were measured[22,23,25]. Given the observed weight loss, one would expect a corresponding reduction in diabetes risk. However, none of the included studies showed a significant difference in the incidence of new-onset T2DM, despite measurable reductions in weight. It is plausible that metformin might have demonstrated a significant effect on diabetes prevention had these studies included larger sample sizes, longer intervention periods and been specifically designed with diabetes incidence as the primary endpoint. Alternatively, it may suggest that in the presence of high background therapy (e.g., statins), the incremental benefit of metformin for diabetes prevention is modest in this population.
Ιn the study by Hartman et al[24], patients with STEMI received metformin treatment for only four months, with follow-up extending to two years. The study reported no long-term benefit of metformin on the incidence of MACEs or new-onset T2DM[24]. The study design may have been suboptimal, as any potential protective effect of metformin on diabetes development would likely manifest during the active treatment phase or shortly thereafter[24]. Recent evidence also suggests that any protective effect of metformin on diabetes prevention in non-diabetic, high-risk populations may diminish after treatment discontinuation[8].
In addition to the limitations noted above-namely small sample sizes, short intervention durations, heterogeneous metformin dosing, and secondary endpoint designation- significant heterogeneity among the included studies cannot be ruled out, which could not be fully explained by subgroup or meta-regression analyses. In fact, we conducted extensive subgroup, sensitivity, and meta-regression analyses, none of which revealed a significant association between metformin and diabetes prevention in any stratified subgroup. This further supports the consistency of the null finding across various patient and intervention characteristics. However, we acknowledge that the analyses are limited by the small number of included studies and events.
While all included RCTs were high-quality[22-25], the overall quality of evidence was rated as very low using the Grading of Recommendations, Assessment, Development, and Evaluation framework, primarily due to imprecision, indirectness (secondary endpoint designation), and strongly suspected publication bias. We also note the absence of any RCTs specifically powered to assess T2DM prevention in non-diabetic patients with CVD/CAD. In contrast, as already noted, recent large-scale trials of GLP-1 RAs and SGLT2 inhibitors have assessed diabetes prevention as a primary outcome, and, importantly, have included substantially longer treatment durations, ultimately demonstrating preventive benefit in selected high-risk populations[51,52]. These trials serve as important models for future research design[51,52].
In conclusion, metformin was not associated with a significant reduction in the incidence of T2DM among patients with CAD. Although the preventive effect of metformin is well established in prediabetic and other high-risk individuals, our analysis highlights the lack of definitive evidence supporting its use for diabetes prevention in patients with established CAD but without diabetes. However, these findings should be interpreted with caution. The included studies were few in number, limited in sample size, and not designed with diabetes prevention as the primary outcome. Additionally, the intervention durations were short, and treatment adherence were variably reported. Given these limitations and the very low overall quality of evidence, we do not conclude that metformin is ineffective in this population-but rather that current evidence is insufficient to support its preventive use in all non-diabetic patients with CAD. Results should not preclude individualized use of metformin based on clinical judgment.
To address this gap, as evidence remains limited and preliminary, more robust data are needed from large-scale, long-term RCTs specifically designed to evaluate diabetes prevention as a primary outcome. Such trials should enroll larger populations of high-risk individuals-particularly those with CVD or CAD without diabetes-who may stand to benefit most from early intervention. Additionally, further research is warranted to determine the optimal dose and duration of metformin therapy, as well as its potential synergistic effects when combined with newer glucose-lowering agents.
| 1. | ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA; on behalf of the American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46:S19-S40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2057] [Cited by in RCA: 1712] [Article Influence: 570.7] [Reference Citation Analysis (5)] |
| 2. | Joseph JJ, Deedwania P, Acharya T, Aguilar D, Bhatt DL, Chyun DA, Di Palo KE, Golden SH, Sperling LS; American Heart Association Diabetes Committee of the Council on Lifestyle and Cardiometabolic Health; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Clinical Cardiology; and Council on Hypertension. Comprehensive Management of Cardiovascular Risk Factors for Adults With Type 2 Diabetes: A Scientific Statement From the American Heart Association. Circulation. 2022;145:e722-e759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 528] [Cited by in RCA: 447] [Article Influence: 111.8] [Reference Citation Analysis (2)] |
| 3. | Petrie JR, Guzik TJ, Touyz RM. Diabetes, Hypertension, and Cardiovascular Disease: Clinical Insights and Vascular Mechanisms. Can J Cardiol. 2018;34:575-584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1249] [Cited by in RCA: 1136] [Article Influence: 142.0] [Reference Citation Analysis (1)] |
| 4. | Khan MAB, Hashim MJ, King JK, Govender RD, Mustafa H, Al Kaabi J. Epidemiology of Type 2 Diabetes - Global Burden of Disease and Forecasted Trends. J Epidemiol Glob Health. 2020;10:107-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2498] [Cited by in RCA: 1929] [Article Influence: 321.5] [Reference Citation Analysis (7)] |
| 5. | Tao Z, Shi A, Zhao J. Epidemiological Perspectives of Diabetes. Cell Biochem Biophys. 2015;73:181-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 159] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 6. | Cichosz SL, Johansen MD, Hejlesen O. Toward Big Data Analytics: Review of Predictive Models in Management of Diabetes and Its Complications. J Diabetes Sci Technol. 2015;10:27-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 7. | Chen L, Islam RM, Wang J, Hird TR, Pavkov ME, Gregg EW, Salim A, Tabesh M, Koye DN, Harding JL, Sacre JW, Barr ELM, Magliano DJ, Shaw JE. A systematic review of trends in all-cause mortality among people with diabetes. Diabetologia. 2020;63:1718-1735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 8. | Tsironikos GI, Tsolaki V, Zakynthinos GE, Rammou V, Kyprianidou D, Antonogiannis T, Zakynthinos E, Bargiota A. Metformin's Overall Effectiveness and Combined Action with Lifestyle Interventions in Preventing Type-2 Diabetes Mellitus in High-Risk Metformin-Naïve Patients: An Updated Systematic Review and Meta-Analysis of Published RCTs. J Clin Med. 2025;14:4947. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 9. | Moore SM, Hardie EA, Hackworth NJ, Critchley CR, Kyrios M, Buzwell SA, Crafti NA. Can the onset of type 2 diabetes be delayed by a group-based lifestyle intervention? A randomised control trial. Psychol Health. 2011;26:485-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Zilov AV, Abdelaziz SI, AlShammary A, Al Zahrani A, Amir A, Assaad Khalil SH, Brand K, Elkafrawy N, Hassoun AAK, Jahed A, Jarrah N, Mrabeti S, Paruk I. Mechanisms of action of metformin with special reference to cardiovascular protection. Diabetes Metab Res Rev. 2019;35:e3173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (1)] |
| 11. | Diabetes Prevention Program Research Group. Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care. 2012;35:731-737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 395] [Cited by in RCA: 352] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 12. | Goldberg RB, Orchard TJ, Crandall JP, Boyko EJ, Budoff M, Dabelea D, Gadde KM, Knowler WC, Lee CG, Nathan DM, Watson K, Temprosa M; Diabetes Prevention Program Research Group*. Effects of Long-term Metformin and Lifestyle Interventions on Cardiovascular Events in the Diabetes Prevention Program and Its Outcome Study. Circulation. 2022;145:1632-1641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 117] [Article Influence: 29.3] [Reference Citation Analysis (1)] |
| 13. | Salpeter SR, Buckley NS, Kahn JA, Salpeter EE. Meta-analysis: metformin treatment in persons at risk for diabetes mellitus. Am J Med. 2008;121:149-157.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 160] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 14. | Amer BE, Abdelgalil MS, Hamad AA, Abdelsayed K, Elaraby A, Abozaid AM, Abd-ElGawad M. Metformin plus lifestyle interventions versus lifestyle interventions alone for the delay or prevention of type 2 diabetes in individuals with prediabetes: a meta-analysis of randomized controlled trials. Diabetol Metab Syndr. 2024;16:273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 15. | Patel D, Ayesha IE, Monson NR, Klair N, Patel U, Saxena A, Hamid P. The Effectiveness of Metformin in Diabetes Prevention: A Systematic Review and Meta-Analysis. Cureus. 2023;15:e46108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 16. | Guise JM, Butler ME, Chang C, Viswanathan M, Pigott T, Tugwell P; Complex Interventions Workgroup. AHRQ series on complex intervention systematic reviews-paper 6: PRISMA-CI extension statement and checklist. J Clin Epidemiol. 2017;90:43-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 77] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 17. | Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA; Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27927] [Cited by in RCA: 26515] [Article Influence: 1767.7] [Reference Citation Analysis (7)] |
| 18. | DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32714] [Cited by in RCA: 30831] [Article Influence: 770.8] [Reference Citation Analysis (5)] |
| 19. | Engels EA, Schmid CH, Terrin N, Olkin I, Lau J. Heterogeneity and statistical significance in meta-analysis: an empirical study of 125 meta-analyses. Stat Med. 2000;19:1707-1728. [PubMed] [DOI] [Full Text] |
| 20. | Higgins JP, Thompson SG. Controlling the risk of spurious findings from meta-regression. Stat Med. 2004;23:1663-1682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 899] [Article Influence: 40.9] [Reference Citation Analysis (4)] |
| 21. | Lin L, Chu H. Quantifying publication bias in meta-analysis. Biometrics. 2018;74:785-794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1366] [Cited by in RCA: 1112] [Article Influence: 139.0] [Reference Citation Analysis (4)] |
| 22. | Preiss D, Lloyd SM, Ford I, McMurray JJ, Holman RR, Welsh P, Fisher M, Packard CJ, Sattar N. Metformin for non-diabetic patients with coronary heart disease (the CAMERA study): a randomised controlled trial. Lancet Diabetes Endocrinol. 2014;2:116-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 156] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 23. | Lexis CP, van der Horst-Schrivers AN, Lipsic E, Valente MA, Muller Kobold AC, de Boer RA, van Veldhuisen DJ, van der Harst P, van der Horst IC. The effect of metformin on cardiovascular risk profile in patients without diabetes presenting with acute myocardial infarction: data from the Glycometabolic Intervention as adjunct to Primary Coronary Intervention in ST Elevation Myocardial Infarction (GIPS-III) trial. BMJ Open Diabetes Res Care. 2015;3:e000090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 24. | Hartman MHT, Prins JKB, Schurer RAJ, Lipsic E, Lexis CPH, van der Horst-Schrivers ANA, van Veldhuisen DJ, van der Horst ICC, van der Harst P. Two-year follow-up of 4 months metformin treatment vs. placebo in ST-elevation myocardial infarction: data from the GIPS-III RCT. Clin Res Cardiol. 2017;106:939-946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 25. | Mohan M, Al-Talabany S, McKinnie A, Mordi IR, Singh JSS, Gandy SJ, Baig F, Hussain MS, Bhalraam U, Khan F, Choy AM, Matthew S, Houston JG, Struthers AD, George J, Lang CC. A randomized controlled trial of metformin on left ventricular hypertrophy in patients with coronary artery disease without diabetes: the MET-REMODEL trial. Eur Heart J. 2019;40:3409-3417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 126] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 26. | Siam NH, Snigdha NN, Tabasumma N, Parvin I. Diabetes Mellitus and Cardiovascular Disease: Exploring Epidemiology, Pathophysiology, and Treatment Strategies. Rev Cardiovasc Med. 2024;25:436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 27. | Yang DR, Wang MY, Zhang CL, Wang Y. Endothelial dysfunction in vascular complications of diabetes: a comprehensive review of mechanisms and implications. Front Endocrinol (Lausanne). 2024;15:1359255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 145] [Article Influence: 72.5] [Reference Citation Analysis (1)] |
| 28. | Caballero AE, Delgado A, Aguilar-Salinas CA, Herrera AN, Castillo JL, Cabrera T, Gomez-Perez FJ, Rull JA. The differential effects of metformin on markers of endothelial activation and inflammation in subjects with impaired glucose tolerance: a placebo-controlled, randomized clinical trial. J Clin Endocrinol Metab. 2004;89:3943-3948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 102] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 29. | Jeon SM. Regulation and function of AMPK in physiology and diseases. Exp Mol Med. 2016;48:e245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 526] [Cited by in RCA: 886] [Article Influence: 88.6] [Reference Citation Analysis (2)] |
| 30. | Shivaprakash P, Beeraka NM, Madhunapantula SRV, Nikolenko VN, Basalingappa KM. Metformin Effects on SHIP2, AMPKs and Gut Microbiota: Recent Updates on Pharmacology. Curr Med Chem. 2025;32:1732-1754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 31. | Markowicz-Piasecka M, Huttunen KM, Mateusiak L, Mikiciuk-Olasik E, Sikora J. Is Metformin a Perfect Drug? Updates in Pharmacokinetics and Pharmacodynamics. Curr Pharm Des. 2017;23:2532-2550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 63] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 32. | Dunn CJ, Peters DH. Metformin. A review of its pharmacological properties and therapeutic use in non-insulin-dependent diabetes mellitus. Drugs. 1995;49:721-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 233] [Article Influence: 7.5] [Reference Citation Analysis (4)] |
| 33. | Zhang E, Jin L, Wang Y, Tu J, Zheng R, Ding L, Fang Z, Fan M, Al-Abdullah I, Natarajan R, Ma K, Wang Z, Riggs AD, Shuck SC, Yang L, Huang W. Intestinal AMPK modulation of microbiota mediates crosstalk with brown fat to control thermogenesis. Nat Commun. 2022;13:1135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (2)] |
| 34. | Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;352:854-865. [PubMed] |
| 35. | Zhang K, Yang W, Dai H, Deng Z. Cardiovascular risk following metformin treatment in patients with type 2 diabetes mellitus: Results from meta-analysis. Diabetes Res Clin Pract. 2020;160:108001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 36. | Campbell JM, Bellman SM, Stephenson MD, Lisy K. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: A systematic review and meta-analysis. Ageing Res Rev. 2017;40:31-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 340] [Article Influence: 37.8] [Reference Citation Analysis (1)] |
| 37. | Chang CC, Chou YC, Yang T, Chang JY, Sun CA. Effects of metformin treatment on the risk of acute myocardial infarction. Sci Rep. 2025;15:27707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 38. | Bahardoust M, Hadaegh F, Mehrabi Y, Delpisheh A, Khalili D. Medication time of metformin and sulfonylureas and incidence of cardiovascular diseases and mortality in type 2 diabetes: a pooled cohort analysis. Sci Rep. 2025;15:8401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 39. | Larsen AH, Wiggers H, Dollerup OL, Jespersen NR, Hansson NH, Frøkiær J, Brøsen K, Nørrelund H, Bøtker HE, Møller N, Jessen N. Metformin Lowers Body Weight But Fails to Increase Insulin Sensitivity in Chronic Heart Failure Patients without Diabetes: a Randomized, Double-Blind, Placebo-Controlled Study. Cardiovasc Drugs Ther. 2021;35:491-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 40. | Griffin SJ, Bethel MA, Holman RR, Khunti K, Wareham N, Brierley G, Davies M, Dymond A, Eichenberger R, Evans P, Gray A, Greaves C, Harrington K, Hitman G, Irving G, Lessels S, Millward A, Petrie JR, Rutter M, Sampson M, Sattar N, Sharp S. Metformin in non-diabetic hyperglycaemia: the GLINT feasibility RCT. Health Technol Assess. 2018;22:1-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 41. | Han Y, Xie H, Liu Y, Gao P, Yang X, Shen Z. Effect of metformin on all-cause and cardiovascular mortality in patients with coronary artery diseases: a systematic review and an updated meta-analysis. Cardiovasc Diabetol. 2019;18:96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 250] [Article Influence: 35.7] [Reference Citation Analysis (5)] |
| 42. | Dludla PV, Nyambuya TM, Johnson R, Silvestri S, Orlando P, Mazibuko-Mbeje SE, Gabuza KB, Mxinwa V, Mokgalaboni K, Tiano L, Muller CJF, Louw J, Nkambule BB. Metformin and heart failure-related outcomes in patients with or without diabetes: a systematic review of randomized controlled trials. Heart Fail Rev. 2021;26:1437-1445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (3)] |
| 43. | Larsen AH, Jessen N, Nørrelund H, Tolbod LP, Harms HJ, Feddersen S, Nielsen F, Brøsen K, Hansson NH, Frøkiaer J, Poulsen SH, Sörensen J, Wiggers H. A randomised, double-blind, placebo-controlled trial of metformin on myocardial efficiency in insulin-resistant chronic heart failure patients without diabetes. Eur J Heart Fail. 2020;22:1628-1637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 44. | American Diabetes Association Professional Practice Committee. 10. Cardiovascular Disease and Risk Management: Standards of Care in Diabetes-2025. Diabetes Care. 2025;48:S207-S238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 148] [Article Influence: 148.0] [Reference Citation Analysis (1)] |
| 45. | Marx N, Federici M, Schütt K, Müller-Wieland D, Ajjan RA, Antunes MJ, Christodorescu RM, Crawford C, Di Angelantonio E, Eliasson B, Espinola-Klein C, Fauchier L, Halle M, Herrington WG, Kautzky-Willer A, Lambrinou E, Lesiak M, Lettino M, McGuire DK, Mullens W, Rocca B, Sattar N; ESC Scientific Document Group. 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes. Eur Heart J. 2023;44:4043-4140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1212] [Cited by in RCA: 989] [Article Influence: 329.7] [Reference Citation Analysis (8)] |
| 46. | Wong HJ, Lin NH, Teo YN, Syn NL, Teo YH, Sia CH. Evaluation of the Lifetime Benefits of Metformin and SGLT2 Inhibitors in Type 2 Diabetes Mellitus Patients with Cardiovascular Disease: A Systematic Review and Two-Stage Meta-Analysis. Am J Cardiovasc Drugs. 2024;24:371-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 47. | Chen B, Tai Y, Kong Y, Wang C. Association between metformin use and reduced short- and long-term all-cause mortality in patients undergoing cardiac surgery: a retrospective cohort study. Eur J Med Res. 2025;30:606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 48. | Kelly AS, Bergenstal RM, Gonzalez-Campoy JM, Katz H, Bank AJ. Effects of exenatide vs. metformin on endothelial function in obese patients with pre-diabetes: a randomized trial. Cardiovasc Diabetol. 2012;11:64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 49. | Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14659] [Cited by in RCA: 12460] [Article Influence: 519.2] [Reference Citation Analysis (5)] |
| 50. | Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the Diabetes Prevention Program Outcomes Study. Lancet Diabetes Endocrinol. 2015;3:866-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 869] [Cited by in RCA: 755] [Article Influence: 68.6] [Reference Citation Analysis (4)] |
| 51. | Kahn SE, Deanfield JE, Jeppesen OK, Emerson SS, Boesgaard TW, Colhoun HM, Kushner RF, Lingvay I, Burguera B, Gajos G, Horn DB, Hramiak IM, Jastreboff AM, Kokkinos A, Maeng M, Matos ALSA, Tinahones FJ, Lincoff AM, Ryan DH; SELECT Trial Investigators. Effect of Semaglutide on Regression and Progression of Glycemia in People With Overweight or Obesity but Without Diabetes in the SELECT Trial. Diabetes Care. 2024;47:1350-1359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 62] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 52. | James S, Erlinge D, Storey RF, McGuire DK, de Belder M, Eriksson N, Andersen K, Austin D, Arefalk G, Carrick D, Hofmann R, Hoole SP, Jones DA, Lee K, Tygesen H, Johansson PA, Langkilde AM, Ridderstråle W, Parvaresh Rizi E, Deanfield J, Oldgren J. Dapagliflozin in Myocardial Infarction without Diabetes or Heart Failure. NEJM Evid. 2024;3:EVIDoa2300286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 215] [Article Influence: 107.5] [Reference Citation Analysis (0)] |