Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.115942
Revised: December 22, 2025
Accepted: February 6, 2026
Published online: April 26, 2026
Processing time: 167 Days and 10.7 Hours
Sudden cardiac death in adolescent athletes, although rare, carries substantial public health impact. Screening methods remain debated internationally. Cost effectiveness, diagnostic yield and logistical ability remain as points of contention.
To evaluate the diagnostic yield, accuracy, and cost-effectiveness of electrocardiogram (ECG)-based screening programs for adolescent athletes. The secondary aim is to discuss ethical and psychological issues that may arise from universal sc
A systematic review of literature from 1998-2025 was conducted, assessing ECG-inclusive screening, transthoracic echocardiography follow-up, and comparative international models. An additional segment was created to analyze more quali
ECG screening demonstrated superior sensitivity compared to history and physical exam alone. International programs varied in diagnostic yield, false-positive rates, and feasibility. A majority of physicians support ECG screening for athletes if proper training and financial feasibility were possible.
ECG-inclusive screening improves detection of high-risk cardiac conditions but requires careful consideration of cost, resource availability, and implementation strategies. A targeted approach to specific high-risk populations would benefit most from targeted screening and may serve as a pilot framework for future universal screening.
Core Tip: This review evaluates the role of electrocardiogram screening in preventing sudden cardiac death among adolescent athletes, emphasizing diagnostic accuracy, cost-effectiveness, and international policy differences. By comparing American and European screening models, we highlight the balance between sensitivity, feasibility, and resource utilization. Advances such as standardized electrocardiogram interpretation criteria and artificial intelligence-assisted analysis may improve detection accuracy while reducing false positives and financial burden. These insights underscore the potential for evidence-based, equitable, and efficient cardiovascular screening strategies in young athletes.
- Citation: Celebi TB, Landman SM, Hayes JP, Makaryus AN. Electrocardiogram screening in adolescent athletes - evaluating clinical protocols, outcomes, and cost-effectiveness a systemic literature review. World J Cardiol 2026; 18(4): 115942
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/115942.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.115942
Sudden cardiac death (SCD) in adolescent athletes is defined as an unexpected death attributable to cardiac causes that occurs during or immediately following exercise, often in individuals without prior symptoms or diagnosis[1]. Although rare, with an estimated incidence of 1 in 50000 to 1 in 80000 young athletes per year, SCD remains the leading cause of mortality in this population. Regardless of its rarity, the incidence means very little to families and friends who lose loved ones to these pathologies. The most frequent underlying causes include hypertrophic cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy, anomalous coronary artery anatomy, and primary arrhythmogenic disorders[1,2]. Although these deaths have had a decreased incidence with policies increasing the use of on-site automated external defibrillator (AED) devices, these situations can be significantly minimized with more detailed screening methods in youth athletes. Furthermore, the increased availability and use of AEDs can only serve as a treatment for SCD after the athlete has collapsed, rather than in the prevention of the event. The sports with the highest risk of SCD in the United States are basketball, American football, and soccer[3]. The risk is highest in male athletes, especially black male basketball players, who make up a majority of these SCDs[1].
The public health significance of SCD extends beyond its numerical rarity, as each event represents a catastrophic loss in otherwise healthy adolescents. The collapse and possible death of a young athlete can profoundly impact families, peers, and communities, often draw widespread media attention and raise concerns about the adequacy of current prevention strategies[4,5]. These events can erode community trust in organized sports, place psychological strain on teammates and coaches, and prompt calls for policy change within athletic associations[6]. As physicians who complete pre-participation physicals that require documentation of sports eligibility, it is our responsibility to consider ways to reduce SCD in these populations.
Pre-participation cardiovascular screening is designed to identify athletes at risk before participation. In the United States, current guidelines from the American Heart Association (AHA) and the preparticipation physical evaluation monograph recommend a 14-element history and physical examination, without routine electrocardiogram (ECG)[7]. This level of history taking often requires significant time, something little afforded to primary care providers for their patients. In contrast, European societies, particularly in Italy, mandate universal ECG screening as part of pre-participation clearance, citing evidence of reduced SCD incidence following implementation[8]. This divergence has fueled significant controversy and adds to the discussion of insurance types and costs in the American healthcare system compared to the rest of the world.
Proponents of ECG argue that history and physical examination alone are insensitive, with detection rates as low as 20% for high-risk conditions[9,10]. Conversely, critics emphasize the cost, logistical burden, potential false positives, and limited availability of pediatric cardiologists to interpret abnormal ECG results and the associated tests, such as pediatric echocardiograms or Holter monitors[11,12]. Likewise, no two patient populations are the same, and pre-participation screening may outweigh its benefits in certain cultures and genetic predispositions.
Given the ongoing debate, this review seeks to synthesize current evidence regarding ECG screening in adolescent athletes. Specifically, we will compare the sensitivity and specificity of history/physical vs ECG, explore models of care in the United States and United Kingdom, assess follow-up strategies such as transthoracic echocardiography (TTE) interpreted by pediatric cardiologists, and examine cost-effectiveness analyses, including estimates of cost per life saved. These quantitative measures will be discussed and analyzed, giving explanations to incorporate or omit ECG screening. Likewise, qualitative measures such as psychological impacts on patients and the community will be explored. This creates a more holistic approach to analyzation without focusing on a specific data type, allowing incorporation of difficult to measure variables. The purpose of this review is to provide clinicians, policymakers, and athletic organizations with a balanced understanding of the evidence to guide future screening protocols.
A comprehensive literature search was conducted to identify studies evaluating the role of ECG screening in the prevention of SCD among adolescent and young adult athletes. The databases PubMed/MEDLINE, EMBASE, Scopus, and the Cochrane Library were systematically searched from January 1998 through October 2025. This end date reflects the most recent month for which complete database updates were available prior to manuscript preparation and submission. With increased literature and diagnostic understanding, it was decided that more recent literature should be utilized to formulate arguments due to more relevant data, while older landmark papers were still discussed as points of evolution in screening processes.
The search strategy incorporated both Medical Subject Headings and free-text terms. The following keywords and Boolean combinations were used: “sudden cardiac death” OR “SCD” AND “adolescent athlete” OR “young athlete” AND “pre-participation screening” AND (“electrocardiogram” OR “ECG” OR “EKG”) AND (“transthoracic echocardiography” OR “TTE” OR “echocardiography”) AND (“cost-effectiveness” OR “economic evaluation” OR “screening protocol”).
These search terms were utilized as they would receive the broadest returns without jeopardizing the focused nature of the search results and topic in discussion. Filters were applied to limit results to English-language (primary language of the authors), human studies, and subjects aged 10-25 years.
Studies were eligible for inclusion if they met the following criteria: (1) Examined pre-participation cardiovascular screening programs in adolescent or young adult athletes; (2) Compared ECG-inclusive screening with traditional history and physical examination; (3) Reported measurable outcomes such as diagnostic yield, SCD incidence, cost-effectiveness, test accuracy, or downstream resource utilization; and (4) Represented original research, systematic reviews, or meta-analyses. Studies were excluded if they: (1) Involved non-athlete populations or ages outside 10-25 years; (2) Were editorials, opinion pieces, or single case reports; and (3) Did not report quantitative or clinically interpretable outcomes related to SCD prevention.
Increased emphasis was placed on studies published between 2015 and 2025, reflecting modern interpretation frameworks such as the Seattle criteria and international recommendations for athlete ECG evaluation. These refinements have substantially reduced false-positive rates and reshaped cost-effectiveness modeling. Classic landmark studies predating 2010 were retained due to their foundational role in establishing screening efficacy, shaping international guidelines, and providing the longest duration of outcome data.
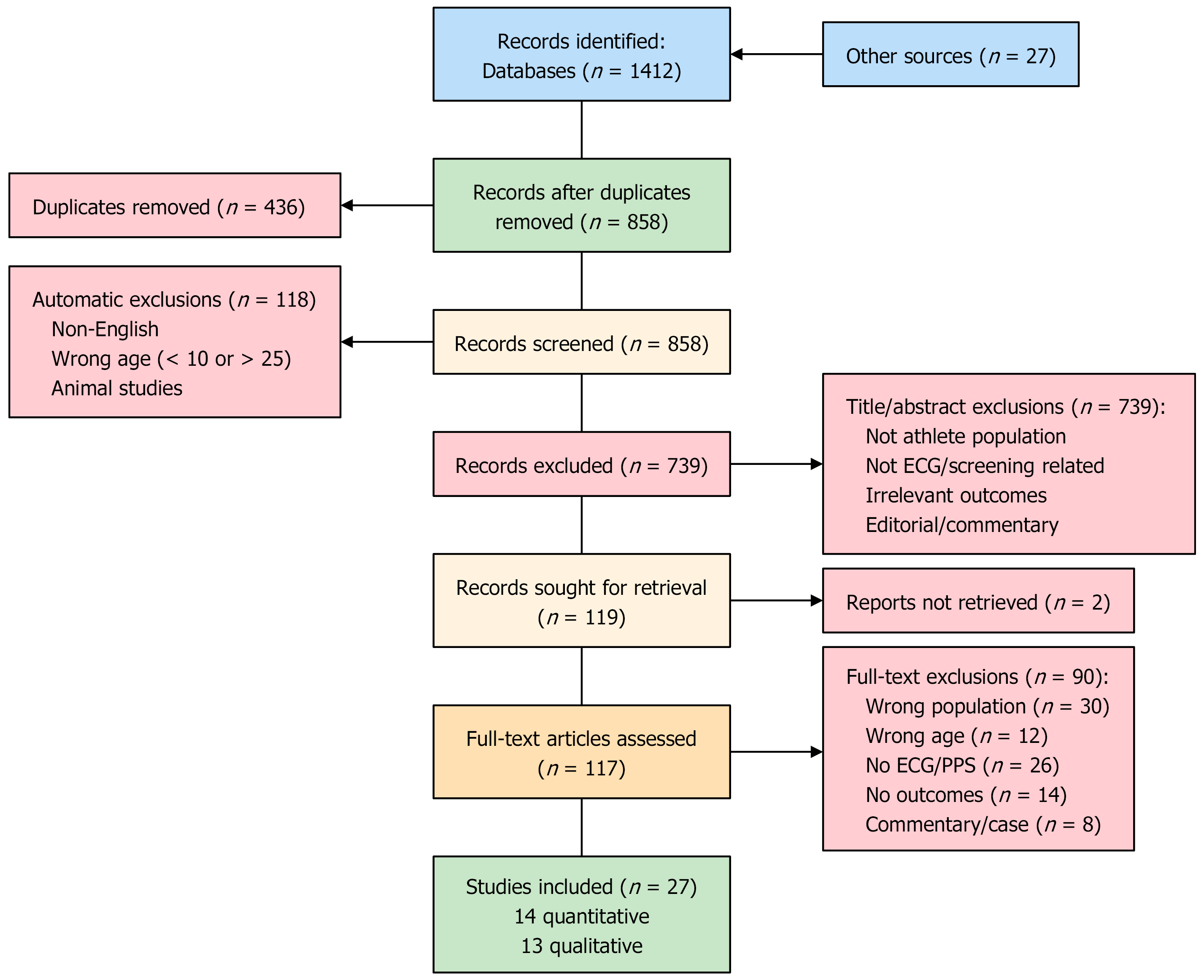
A Preferred Reporting Items for Systematic reviews and Meta-Analyses-compliant flow diagram (Figure 1) outlines the number of records identified, screened, assessed for eligibility, and included in the final synthesis, with specific exclusion reasons noted. The articles excluded from the original analysis secondary to final exclusion criteria were then re-evaluated. This was deemed necessary as a significant portion of data and information that cannot be measured using qualitative means was discussed and found to be pertinent for the discussion of ECG screening in young athletes.
The final number of articles reviewed was 27, with 14 of those being quantitative in nature. These quantitative articles, along with their corresponding key points, primary outcomes, sample size, study design, and screening modality are illustrated in Table 1. The originally excluded 13 articles that were qualitative in nature were then analyzed. Key ethical, philosophical, and logistic concerns were found in each manuscript. The findings, design, and focus area of each study can be found in Table 2.
| Ref. | Country | Population/region | Design | Screening modality | Sample size | Primary outcome | Key findings/quantitative result |
| Corrado et al[26], 2006 | Italy | Competitive athletes, Veneto region | Prospective cohort | H&P + ECG (mandatory) | 42386 | SCD incidence | 89% SCD reduction (3.6/100000 to 0.4/100000 person-years) |
| Papadakis et al[25], 2011 | United Kingdom | Voluntary youth program | Prospective registry | H&P + ECG ± TTE | 32561 | Diagnostic yield | 0.4% serious cardiac findings; 80% detected by ECG |
| Harmon et al[1], 2016 | United States | HS and college athletes | Observational registry | H&P only | 2046000 | SCD incidence | 1.21/100000 person-years; low sensitivity (< 20 %) |
| Drezner et al[24], 2013 | United States | Collegiate athletes | Comparative | H&P vs ECG ± TTE | 11168 | Sensitivity/specificity | ECG 94%/93%; false positives < 5 % (Seattle criteria) |
| Leslie et al[30], 2012 | United States | Modeled athlete cohort | Economic model | ECG ± TTE vs H&P | Cost-effectiveness | 42000 dollars per life-year saved (ICER) | |
| Malhotra and Sharma[5], 2018 | United Kingdom | Elite adolescent athletes | Prospective 20-year follow-up | H&P + ECG | 11168 | Long-term SCD prevention | 0.38/100000 person-years; 80% of SCD etiologies identified |
| Wheeler et al[11], 2010 | United States | Modeled United State athletes | Economic simulation | H&P vs ECG ± ECHO | Cost per life saved | > 10 thousand dollars/life year saved (early); improved < 7 thousand dollars with newer criteria | |
| Steinvil et al[22], 2011 | Israel | National cohort | Retrospective trend | Mandatory ECG | National registry data over 12 years | SCD trend | No statistically significant SCD reduction |
| Baggish et al[15], 2017 | United States | Collegiate athletes | Mixed observational | H&P ± ECG | 510 | Diagnostic yield | 6 pathologies found only by ECG; cost per case approximately 11000 dollars |
| Moulson et al[33], 2021 | Canada | Multicenter athletes | Prospective pilot | H&P + ECG | 2120 | Feasibility/FP rate | FP 3.2%; supports international criteria use |
| Petek and Baggish[23], 2020 | United States | Sports-medicine clinicians | Survey-based quantitative | H&P ± ECG | 125 | Implementation feasibility | 68% cited training barriers; 72% favored ECG inclusion |
| Hernelahti et al[36], 2008 | Nordic | National registries | Policy analysis/quantitative comparison | H&P vs proposed ECG | Policy outcome | Cost-burden model > 150 thousand dollars/life year saved; ECG not adopted | |
| Dhutia et al[12], 2021 | United Kingdom | Health-policy review | Quantitative + ethical | H&P + ECG ± genetic | Cost/risk model | Supported selective screening in high-risk sports | |
| Asif et al[38], 2014 | United States | HS athletes ± parents | Mixed-methods (survey + rates) | H&P + ECG | 410 | Psychological/compliance data | 92% reported reassurance post-ECG; FP 4% |
| Ref. | Country | Population/region | Design/approach | Focus area | Key themes/findings |
| Asif et al[38], 2014 | United States | High-school athletes ± parents | Survey + focus groups | Psychologic impact of ECG screening | (1) False-positive ECGs did not raise anxiety; and (2) Most athletes felt safer and more confident resuming play |
| Dhutia et al[12], 2021 | United Kingdom | Policy experts/clinicians | Narrative review + ethical analysis | Ethical proportionality of mass screening | Advocates context-specific, risk-proportionate screening and shared decision-making |
| Petek and Baggish[23], 2020 | United States | Sports medicine physicians/ATs | Semi-structured survey | Implementation feasibility | Training deficits, variable institutional support, and limited follow-up access identified as key barriers |
| Hernelahti et al[36], 2008 | Nordic countries | Public-health authorities | Policy comparison/qualitative | National screening policy | Nordic taskforce declined national ECG program citing cost, limited yield, and equity concerns |
| Baggish et al[15], 2017 | United States | Collegiate athletes | Mixed-methods observational | Clinical utility + attitudes | (1) ECG added clinical reassurance; and (2) Athletes and clinicians viewed program as acceptable and feasible |
| Moulson et al[33], 2021 | Canada | Multicenter pilot participants | Prospective + feasibility interviews | Workflow + perception | (1) Positive reception; (2) 3.2 % false positives; and (3) Supports standardized interpretation criteria |
| Steinvil et al[22], 2011 | Israel | Policymakers/registry review | Mixed quantitative-qualitative | Policy evaluation | (1) National mandate showed no clear SCD decline; and (2) Prompted debate about cost-benefit and implementation |
| Harmon et al[35], 2015 | United States | Collegiate sports programs | Qualitative analysis of registries | Health-system disparities | Highlighted racial/socioeconomic inequities in SCD events and screening access |
| Drezner et al[34], 2017 | United States | Athletic trainers + team physicians | Survey study | Knowledge and training attitudes | 78% desired ECG training; 64% supported integration if cardiology follow-up available |
| Malhotra and Sharma[5], 2018 | United Kingdom | Elite adolescent athletes | Thematic follow-up interviews | Emotional impact of disqualification | (1) Temporary disqualification caused distress; and (2) Long-term acceptance after counseling is possible unforeseen expense |
| Sharma et al[32], 2017 | International | Consensus experts/societies | Delphi consensus process | Guideline development | Produced International ECG interpretation criteria, standardized approach reducing false positives |
| Roberts et al[37], 2015 | United States | Sports medicine professionals | Narrative review/ethical discourse | Screening philosophy | (1) Compared ECG screening with other public-health screenings (Pap, PSA); and (2) Discussed utilitarian ethics |
| Corrado et al[39], 1998 | Italy | Young athletes/families | Retrospective interviews | Psychosocial effects of disqualification | (1) Emotional burden significant; and (2) Counseling mitigates risk of depression/suicide in disqualified youth |
Data from international ECG screening programs show variable diagnostic yield and differing impacts on SCD incidence. Italy’s Veneto region demonstrates an 89% reduction in SCD following mandatory ECG screening, while other European regions show more modest effects. As summarized in Table 1, these findings illustrate that the highest documented population-level reduction in SCD associated with ECG screening was in areas notable for electroconductive diseases. In contrast, data from the United Kingdom’s Cardiac Risk in the Young (CRY) program show a diagnostic yield of 0.3%-0.4% with high community uptake but do not replicate the magnitude of Italy’s reduction. This can be attributed to a likely genetic variation to the sample cohorts.
Qualitative considerations such as socioeconomic inequities, patient education, and psychological stress were noted in these manuscripts, which correspond to themes presented in Table 2 when more qualitative studies were completed.
SCD in adolescent athletes usually results from a heterogeneous group of cardiovascular disorders that predispose young athletes to fatal arrhythmias, often during or immediately after exertion. Pathophysiology typically involves either structural cardiac abnormalities or primary electrical disorders, both of which can be asymptomatic until a catastrophic event occurs on the athletic field.
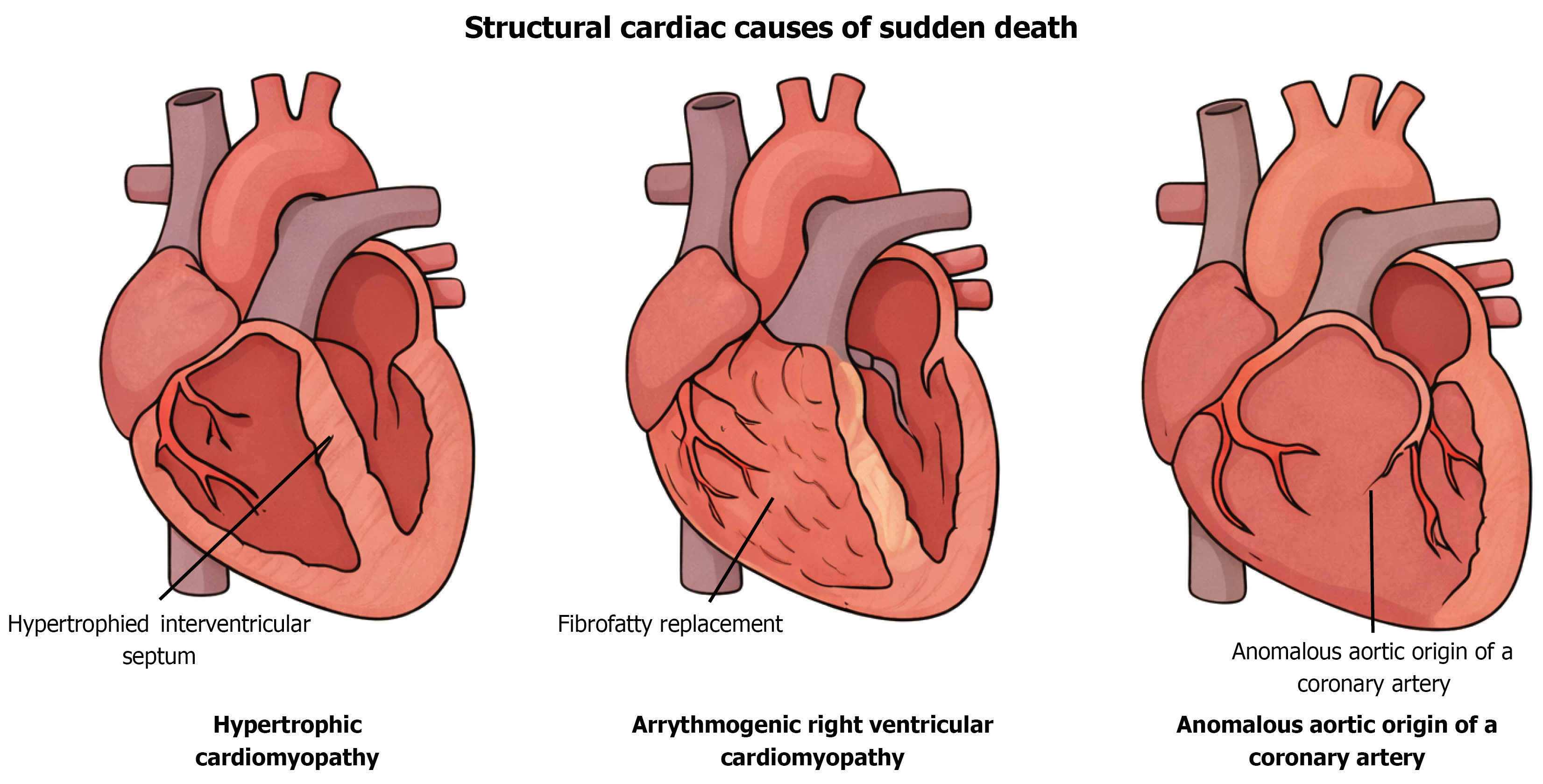
Structural cardiac abnormalities are among the most common reasons for SCD. Hypertrophic cardiomyopathy remains the leading cause in the United States, accounting for up to one-third of athletic SCD cases[6]. Hypertrophic cardiomyopathy has a global incidence of 1 in 500, with an equal distribution between both men and women[13]. This incidence is likely underreported, given marked disparities in access to care and the financial burden of the diagnosis[13]. The asymmetric hypertrophy of the interventricular septum can promote ventricular arrhythmias, particularly during high-stress states such as athletic competition (Figure 2).
Arrhythmogenic right ventricular cardiomyopathy represents another significant contributor, particularly in European populations, and is characterized by fibro-fatty replacement of the right ventricular myocardium, leading to ventricular tachyarrhythmias[8]. The general prevalence of the pathology is difficult to estimate, largely because of the variable expressivity of the disease. However, an overall estimate of the prevalence of arrhythmogenic right ventricular cardiomyopathy is between 1 in 2000 and 1 in 5000 in the general population[2].
Anomalous coronary artery anatomy, such as anomalous origin of the left coronary artery from the right sinus of Valsalva, can result in ischemia during exercise due to dynamic compression of the aberrant vessel and has been implicated in collapse in otherwise healthy athletes[6]. The incidence of this pathology is roughly 1% in the general population; however, based on the patient population and area being tested, the incidence can get as high as 2.6%[14].
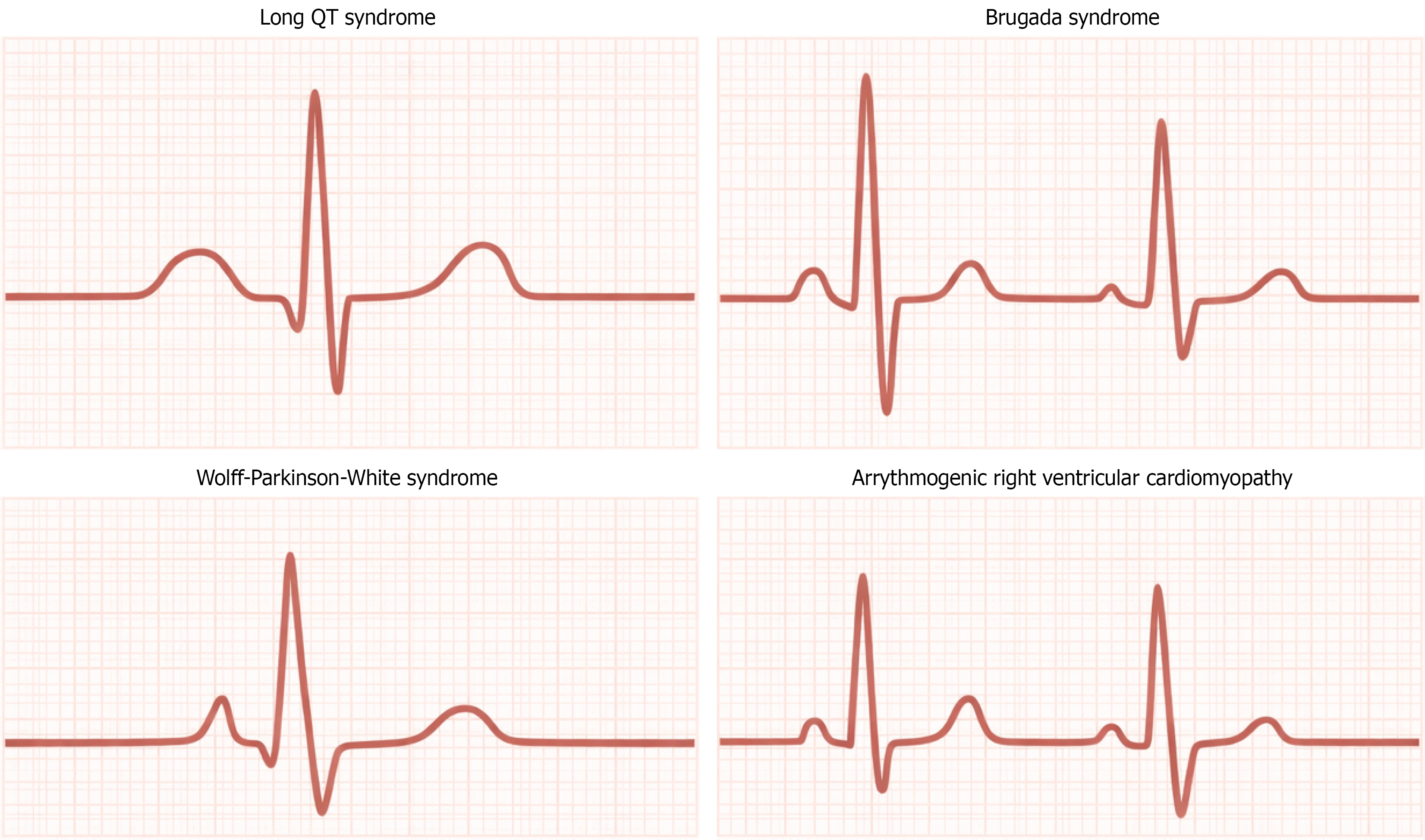
Primary electrical disorders represent another important group of etiologies. Long QT syndrome, caused by genetic mutations in cardiac ion channels, predisposes affected individuals to torsade de pointes and ventricular fibrillation, particularly due to adrenergic stimulation during exertion[15]. Incidence ranges in the general population, but it can be estimated to be 1 in 2000 to 1 in 2500 based on a prospective neonatal EKG screening[16].
Brugada syndrome, an inherited sodium channelopathy, can precipitate polymorphic ventricular tachycardia and SCD, often during rest but occasionally unmasked by exertion[2]. Wolff-Parkinson-White syndrome, characterized by an accessory conduction pathway, predisposes those affected to rapid atrioventricular reentrant tachycardia and atrial fibrillation with rapid ventricular conduction, which can degenerate into ventricular fibrillation during intense physical activity[17]. Brugada syndrome has an estimated prevalence of 1-5 per 10000; however, in certain populations, such as Southwest Asian populations, there is a marked increase[18]. Likewise, the incidence of Wolff-Parkinson-White is 1-3 per 1000, with regional variation and a notably increased incidence in China and Taiwan[19,20].
The distribution of causes varies between geographic regions, reflecting both genetic prevalence and differences in screening strategies. In the United States, hypertrophic cardiomyopathy and congenital coronary anomalies are con
Since 1996, the AHA has recommended its 14-element history and physical as the standard for pre-participation screening of pediatric athletes[17]. The guidelines and checklist focus on personal history (e.g., syncope, chest pain with exertion), family history (e.g., sudden unexplained death, cardiomyopathy), and require a targeted physical examination (e.g., heart murmurs, femoral pulses). While widely implemented due to its low cost and logistical simplicity, evidence from both domestic and international studies has demonstrated limited diagnostic sensitivity. A systematic review found that history and physical examination alone identify fewer than 20% of athletes with conditions associated with SCD, and specificity is further compromised by subjective symptom reporting and examiner variability[21]. Table 1 reinforces the improved diagnostic abilities of an ECG through multiple studies. Additionally, the time required to conduct such a significant and thorough exam for each patient who presents for a full “sports physical” places a large burden on providers. If a sudden cardiac arrest or death occurs following one of these sports physicals, it also creates an air of blame and frustration with the physicians who completed the physical. Consequently, this mainstay approach may under-detect asymptomatic structural or electrical heart diseases, such as hypertrophic cardiomyopathy or long QT syndrome. Some studies have even illustrated no significant change in SCD when ECG screening is performed[22]. Continued research must be completed to illustrate how new guidelines could prevent SCD. Surveys to discuss this among providers have shown that there is a split between the desire to incorporate ECG screening, with many citing financial barriers as a major limitation[23], which was illustrated throughout multiple manuscripts in Table 2.
The addition of a 12-lead ECG to pre-participation evaluation substantially increases diagnostic yield by identifying electrical abnormalities or indirect markers of structural disease. Therefore, this type of screening could theoretically decrease the cost of a physical exam and reduce second-guessing if a murmur or irregular heartbeat is heard. Comparative analyses indicate that ECG screening has a sensitivity exceeding 80% for detecting high-risk cardiovascular conditions[24,25]. Italy’s Veneto region, where mandatory ECG screening has been in place since the 1980s, reports an 89% reduction in SCD incidence among young athletes, largely attributed to the identification and disqualification of athletes with cardiomyopathies[26]. It should be stated that this region also has an increased risk of arrhythmic cardiac conditions[26].
In contrast, the United Kingdom has pursued a voluntary model initiative, the CRY program, which offers ECG and follow-up echocardiography on a smaller scale to patients who may be at higher risk or who are concerned about their general health competing at higher levels. While not population-wide, CRY has screened over 200000 young individuals since 1995 and demonstrated high feasibility and strong community uptake[27]. The heterogeneity across countries reflects differences in healthcare systems, resource allocation, and societal tolerance for false positives.
Abnormal or indeterminate ECG findings typically prompt secondary evaluation with TTE, which serves as the gold standard for non-invasive assessment of structural heart disease. Indications include suspected cardiomyopathy, valvular pathology, or anomalous coronary artery origins. The use of TTE enhances diagnostic specificity, filtering false-positive ECG results and ensuring that only athletes with clinically significant abnormalities are disqualified or treated[28]. The reliance on a TTE also leads to increased financial burden and healthcare stress, as this exam must be reviewed and acted on by a pediatric cardiologist. Major financial and logistical barriers frequently arise not from the initial ECG itself, but from the need for escalated care and further diagnostic testing when abnormalities are detected. Due to the limited number of pediatric cardiologists able to read a TTE, this can be an extra barrier to an already stressed healthcare system. This seems to be the major survey point in a majority of studies found in Table 2. Emphasizing standardized ECG interpretation guidelines provides a straightforward strategy to reduce false positives and alleviate the burden on the healthcare system.
The cost-effectiveness of ECG screening is directly tied to the yield of secondary imaging. Studies from both Europe and North America suggest that approximately 2-5% of athletes undergoing ECG screening require TTE follow-up, with only a small fraction ultimately diagnosed with conditions linked to SCD risk[11,29]. Cost models have estimated the incremental cost-effectiveness ratio (ICER) of ECG plus TTE strategies to range from 40000 dollars to 100000 dollars per life-year saved, depending on assumptions about incidence rates, test performance, and downstream healthcare costs[30,31]. This falls near the accepted thresholds for public health interventions in some systems; the variability underscores the need for context-specific economic analysis. For example, routine public health interventions such as mammography fall within a similar range, suggesting ECG screening may be acceptable from an economic perspective, depending on assumptions. Likewise, certain emotional and social costs cannot be calculated accurately. Lastly, advances in standardized ECG interpretation criteria, such as the Seattle criteria[24] and international criteria[32], have reduced false-positive rates from > 15% to < 5%, thereby decreasing unnecessary secondary imaging and improving overall cost-effectiveness. This was reiterated again in pilot studies by Moulson et al[33]. It has even promoted discussion about increased desire for cardiology follow-up for athletes[34]. This does require physicians to also feel comfortable with reading pediatric ECGs and applying these criteria to assist with diagnosis. Comfort level in ECG interpretation was a notable survey point found in Table 2.
Taken together, these approaches underscore the ongoing tension between sensitivity, specificity, and feasibility in pre-participation screening. The United States strategy, which emphasizes history and physical examination, prioritizes broad applicability and low cost but risks under-detection of silent pathologies. However, with the room for error and the speed at which appointments must be completed due to the financial pressures of the healthcare system, there are areas of concern during completion of the visit. Likewise, with increased reliance on imaging and technology when it comes to the diagnosis of pathology, physical exams may be lacking.
In contrast, as seen in Table 1, European models incorporating ECG screening demonstrate higher sensitivity and have documented population-level reductions in SCD but carry greater financial and logistical demands, as well as challenges related to false positives and secondary imaging. Burdens like this can be exacerbated by the number of individuals being tested, the cost of testing, and the ease of access to these resources. Smaller countries may be able to do this more effectively than larger, more diverse countries.
Emerging evidence suggests that refining ECG interpretation criteria and applying targeted echocardiographic follow-up may represent a balanced pathway forward, potentially reconciling diagnostic accuracy with cost-effectiveness. Likewise, because areas of Europe do not encompass the size and complexity of the United States’ diverse patient population, different approaches may be more applicable in certain regions.
These comparisons highlight that optimal screening approaches are highly dependent on healthcare infrastructure, resource availability, and societal willingness to invest in preventive measures, which ultimately summarizes all the information illustrated across both Tables 1 and 2.
Diverse screening methods exist worldwide, and each protocol has been studied to varying degrees. These different protocols and their effectiveness can be illustrated through Table 1. Italy represents the most extensively studied model of population-wide ECG screening in young adult athletes. The foundational study in the Veneto region, a mandatory ECG program initiated in 1982, required all competitive athletes to undergo a 12-lead ECG in addition to history and physical examination. This program was a result of an initiative to decrease SCD in young athletes. Encompassing two decades, Corrado et al[26] documented an 89% reduction in SCD among screened athletes, from 3.6 per 100000 person-years to 0.4 per 100000 person-years. However, critics have noted limitations to the study, including the potential underreporting of SCD in non-athletes used as a comparison group, which could have skewed the reduction percentage[35]. Likewise, generalizability concerns for non-European populations have often been raised as a major concern when planning to initiate this program in much more diverse patient populations[35]. Despite these criticisms, the Italian experience remains the strongest population-level evidence supporting ECG screening as a preventive measure.
Data from the United Kingdom’s CRY initiative indicates a prevalence of potentially serious cardiac conditions of about 0.3%-0.4% of screened individuals, with detection rates far surpassing those achievable through history/physical examination alone[27]. However, critics suggest that population trust and education about SCD can limit the utilization of these ECG screenings. Interestingly, Table 2 illustrates results of mixed feelings about the incorporation of ECGs by the public, and the potential unforeseen aftermath of false positives on athletes. Therefore, the populations most at risk for SCD may not obtain their screenings, and ultimately, the programs may just burden the healthcare system without any benefit.
Elsewhere in Europe, approaches vary. Denmark specifically has documented formal consideration and then rejection of national ECG screenings. Ultimately, citing the cost of screening as a major deterrent, Denmark has shifted towards voluntary screening for its elite athletes[36]. This again aligns with the survey results found in Table 2 regarding cost and financial feasibility.
Overall, European data suggest meaningful diagnostic yield, as seen in Table 1, but have not consistently replicated the dramatic reductions in SCD observed in Italy, which appears to be an outlier. Hypotheses include differences in patient population, logistical implementation, and cost efficiency as major factors affecting the yield and effectiveness.
In contrast, the AHA and American College of Cardiology continue to recommend pre-participation screening limited to history and physical examination[17]. United States guidelines emphasize avoiding harm from unnecessary disqualification and downstream testing, prioritizing feasibility and resource stewardship over maximal sensitivity. Pilot programs at select institutions (e.g., Stanford University, University of Washington) have explored ECG-inclusive screening. They often demonstrate increased diagnostic yield but face significant implementation challenges. Barriers discussed in these pilot programs consistently cited high rates of false positives under earlier interpretation standards. Moreover, the logistical difficulty of scaling cardiology follow-up across a fragmented healthcare system, and cost-effectiveness concerns in a payer-driven environment, remain obstacles that would need to be addressed[17,28].
Multiple studies demonstrate that ECG screening detects over 80% of conditions associated with SCD, such as hypertrophic cardiomyopathy and channelopathies, compared to < 20% with history/physical examination alone[24,35]. As shown in Table 1, quantitative comparisons from multiple studies illustrate this marked difference in diagnostic performance. This diagnostic performance then leads to better outcomes for patients by reducing SCD. These statistical analyses have historically been limited, with false-positive rates ranging from 80%-90% when applied to large populations. Positive predictive value varies widely, influenced by disease prevalence and population characteristics.
False positives, and the need for follow-up diagnostics, have been a central criticism of ECG screening. Early studies reported abnormal findings in up to 15%-20% of athletes, many of whom were ultimately healthy[29]. These cases not only generate unnecessary follow-up testing and costs but can also impose psychological stress and risk of premature athletic disqualification. This emotional burden and distress were mostly noted in Table 2 through survey analysis. Athletes with abnormal ECGs will need to avoid sports until further workup is completed, which, given the current medical infrastructure, may take months. Conversely, false negatives, though less common, remain a concern, particularly for pathologies with minimal ECG manifestations like anomalous coronary arteries. Increasing screening techniques that ultimately fail to catch disease cause strife between patients and providers. The balance of harms and benefits depends heavily on the interpretation criteria and the robustness of follow-up pathways. The ineffectiveness of this aspect of a screening program is likely to be exacerbated in suburban and rural areas, particularly in low socio-economic regions.
Substantial progress has been made in refining ECG interpretation to reduce unnecessary testing by increasing sensitivity and specificity. The Seattle criteria[24] and subsequent international recommendations[32] standardized definitions of normal, borderline, and abnormal findings in athletes, accounting for physiological adaptations to exercise. These criteria reduced false-positive rates from > 15% to < 5% without compromising sensitivity[32]. Such advances have reshaped the debate, with increasing recognition that previous criticisms of ECG screening may be less valid under modern interpretive frameworks. However, as these more refined ECG criteria are unlikely to be known to frontline providers, this leaves a knowledge gap in interpretation.
Financial burden is another major criticism. Cost-effectiveness models have yielded mixed results, which are seen across both Tables 1 and 2. Early United States-based analyses concluded that universal ECG screening was not cost-effective, with incremental costs exceeding 100000 dollars per life-year saved[11]. In contrast, more recent studies incorporating improved ECG specificity suggest more favorable ratios, often between 40000 dollars and 70000 dollars per life-year saved[30]. With the enhanced spotlight on SCD in young, seemingly healthy athletes from outlets like social media, the inability of current practices to reduce these traumatic events may add enough pressure to allow for a greater leeway when it comes to the cost of this public intervention.
Corrado et al[26] estimated that 1 SCD was prevented for every 25000 athletes screened in Italy, yielding a favorable cost-benefit profile in a universal healthcare context. United States analyses, however, have generally reported higher costs per life saved due to the fragmented healthcare system, greater expense of follow-up imaging, and lack of centralized screening infrastructure. This also raises questions about the need for a more targeted approach, selecting opt-in screening programs rather than universal screening as a more financially savvy decision.
Healthcare financing and delivery models strongly influence feasibility. In single-payer systems such as Italy’s or the United Kingdom’s National Health Service, costs are spread across populations, and benefits accrue to the same system that funds the intervention. In the United States, private insurers face weaker incentives to support upfront screening, given high short-term costs and uncertain long-term benefits. Moreover, regional disparities in access to pediatric cardiologists and echocardiography capacity further challenge equitable implementation. These systemic differences largely explain divergent policy adoption despite similar clinical evidence seen in Table 1.
The central ethical dilemma in ECG-based screening of adolescent athletes lies in reconciling population-level prevention of rare but catastrophic events with the burdens placed on individuals. While universal screening may avert SCD in a small subset of athletes, the majority will never benefit directly yet may face additional costs, anxiety, or restrictions from sport. This tension raises questions about proportionality: Is it ethically justifiable to impose widespread testing and potential disqualification on thousands to prevent a handful of deaths? Advocates argue that even a small number of lives saved justifies the intervention, drawing parallels to neonatal metabolic screening and cancer screening programs. Critics, however, caution against medicalizing otherwise healthy populations when the risk of harm is exceedingly low, particularly in resource-limited healthcare systems.
Screening outcomes can profoundly affect the psychological well-being of adolescents and their families. False-positive findings may generate unnecessary anxiety, stigmatization, and concerns about long-term health despite eventual clearance. Athletes may experience identity disruption if disqualified from sport, with potential implications for self-esteem, social relationships, and scholarship opportunities. Furthermore, athletes who continue to compete in sports, even after testing with parental permission, bear a risk of death. A discussion and decision on allowing a child to compete, regardless of diagnosis, would raise future debates. Conversely, identification of true pathology can provide reassurance and enable risk-reducing interventions, such as implantable cardioverter-defibrillators or safe participation guidelines. Ethical screening, therefore, requires robust counseling frameworks and shared decision-making processes to mitigate adverse psychosocial effects.
The implementation of ECG screening also raises equity concerns. In the United States, screening initiatives are often supported by philanthropic organizations or private institutions, creating uneven access across socioeconomic and racial/ethnic groups. Evidence suggests that minority and underserved populations already face disparities in cardiovascular outcomes; the inequitable deployment of screening programs risks exacerbating these divides. In contrast, countries with centralized healthcare systems may achieve more equitable access, though geographic disparities in cardiology services still exist. Policymakers must therefore consider not only the clinical validity of screening but also distributive justice in its provision.
Medicolegal considerations further complicate screening policy. Institutions and athletic organizations face potential liability if an athlete suffers from SCD after being cleared without an ECG, creating pressure to adopt more comprehensive protocols even in the absence of national mandates. Conversely, widespread ECG screening could expose institutions to litigation over disqualification decisions, false positives, or missed diagnoses despite testing. From a legal standpoint, standards of care are often shaped by professional society guidelines. In the United States, the AHA/American College of Cardiology’s continued endorsement of history/physical examination as the standard means institutions adopting broader ECG screening operate outside consensus guidelines, potentially heightening liability risk. Balancing risk management, institutional responsibility, and adherence to evolving evidence thus remains a major policy challenge.
To fully evaluate the implementation of a pre-participation screening for SCD-causing disease in young athletes, further consideration is required. Screening tests, like hemoglobin A1c for type II diabetes risk, Pap smears for cervical cancer, and prostate-specific antigen levels for prostate cancer, all share a commonality. They screen for asymptomatic disease or for conditions for which early detection reduces disease burden[37]. Furthermore, these screening tests have a significant impact on public health. While the event of SCD of a young athlete often occurs in a widely public setting and can be a highly emotional event, cardiovascular death occurs much more commonly in those over the age of 50 who experience atherosclerosis of the coronary arteries or heart failure[12]. In trying to fulfill the physician’s promise for benevolence, screening for such rare conditions may be an inappropriate use of funding.
For a screening program that mandates that all young athletes receive an ECG for cardiac clearance, psychosocial factors must be considered for the athlete and their family. Even a screening program modified with narrower ECG guidelines will still yield many false positives when applied to a large population. There is notable concern for the mental health and well-being of these athletes. For this concern, there is nuance in the body of evidence. Studies investigating athletes’ relative anxiety have shown that a false positive did not significantly increase their anxiety, and many reported they felt safer playing sports after the screening[38]. Conversely, for many young athletes, disqualification from sports comes with detrimental psychological effects that may lead to an increase in suicide[39].
In a financial light, “over screening” young patients may impact their ability to obtain affordable health insurance by generating unnecessary medical costs, false positives, and downstream testing that inflate both individual and system-wide expenditures. While the intent of widespread ECG screening in adolescent athletes is to prevent rare but catastrophic cardiac events, the economic consequences of non-guideline-based testing can be significant. Insurers may classify such screenings as non-preventive, leaving families responsible for out-of-pocket expenses, and the accumulation of incidental findings can label otherwise healthy adolescents as higher-risk, potentially influencing future premium structures[40,41].
With these debates occurring regularly, there are future variables that have yet to leave an impact. Emerging technologies and evolving consensus offer promising avenues to refine pre-participation cardiovascular screening in adolescent athletes and could optimize and streamline these screening methods. One of the most significant developments lies in the application of artificial intelligence (AI) to ECG interpretation. AI-assisted algorithms have demonstrated potential to enhance diagnostic accuracy, reduce false positives, and improve efficiency by standardizing interpretation across clinicians with varying levels of expertise[42]. The pattern recognition capabilities of AI have already been incorporated in fields such as radiology and dermatology to assist with screening for significant pathology. Incorporating this technology into the pre-participation screening of young athletes can assist similarly.
Such tools may help address the shortage of pediatric cardiologists available to review abnormal tracings and lower the downstream burden of unnecessary follow-up testing. Especially with the incorporation of electronic medical record utilization and ECG documentation, a large database can be easily used to train such an intervention.
In parallel, portable ECG devices and wearable cardiac monitoring technologies are becoming increasingly easy to use and wear and could potentially be incorporated into screening. Lightweight, user-friendly platforms capable of detecting arrhythmias in real time may allow for longitudinal monitoring during training and competition, providing dynamic insights into athletes’ cardiac health that static, one-time screenings cannot capture[5]. These technologies could expand access to underserved regions where traditional equipment and expertise are limited, offering more equitable opportunities for early detection, without needing direct physician contact with the specialist.
Secondary prevention has been another potential intervention for SCD in young athletes. With many of these events occurring at public sporting events or at school, investing in cardiopulmonary resuscitation and AED training for school/team staff, as well as for laypeople, should be further explored. Programs like the medical emergency response plan for schools have estimated that such programs would be considerably more affordable than incorporating ECG into screening[43,44].
Integration of genetic testing into select high-risk populations can also be a pragmatic way to screen if methods become more streamlined. Genetic screening may facilitate identification of inherited cardiomyopathies and channelopathies such as hypertrophic cardiomyopathy, long QT syndrome, or Brugada syndrome, conditions often implicated in athletic SCD[16,18] (Figure 3). While ethical and cost considerations remain, the capacity to detect silent carriers before clinical manifestations represents a potential paradigm shift in athlete safety, with a more preventative than post-occurrence mentality.
The detection and prevention of SCD in young athletes remain top priorities in pediatric cardiology and sports medicine. With the increased spotlight on these traumatic events, questions about the current United States protocol have led to vigorous debate. There is little doubt that an ECG screening program for young athletes before entering the sports world has greater specificity and sensitivity than the AHA’s 14-point protocol. What does remain unclear is the practicality of such a program. It is challenging to accurately compare cost-effectiveness between the United States and European countries, such as the United Kingdom, which differ significantly in their medical payment structures and population demographics. The few studies that have investigated cost-effectiveness in the United States have yielded wide variation in conclusions, warranting further investigation. Another difficulty in implementing an ECG screening program is the dramatic increase in diagnostic yield. Because ECG testing is more sensitive and specific, more patients at risk for SCD will be identified, but more patients with cardiac conditions unrelated to SCD, as well as false positives, will be flagged for further testing. Given the already stressed pediatric cardiologists’ infrastructure, preparations for this increase in necessary secondary and follow-up testing must be made. Point-of-care echocardiography and AI interpretation of pediatric athlete ECGs are proposed solutions to this problem that should be explored. Ultimately, we recommend a targeted, risk-stratified ECG screening strategy. This represents the most evidence-supported pathway forward, particularly for athletes participating in higher-risk sports such as basketball and American football. Those with genetic descent from high-risk populations such as northern Italians, Southeast Asians, and black individuals would benefit most from these screenings and assist with the disproportionately higher mortality rates in cardiac conduction disorders. Regional pilot ECG screening programs within integrated healthcare networks may help assess feasibility, cost-effectiveness, and follow-up prior to broader adoption. With cost analysis studies illustrating similar costs to already established screening methods for other pathologies, such as breast cancer screening, it would be financially acceptable comparatively. Advances in AI-supported ECG interpretation offer further potential to reduce false positives and streamline triage processes. These interventions collectively may contribute to reducing the risk of SCD in adolescent athletes.
| 1. | Harmon KG, Asif IM, Maleszewski JJ, Owens DS, Prutkin JM, Salerno JC, Zigman ML, Ellenbogen R, Rao AL, Ackerman MJ, Drezner JA. Incidence and Etiology of Sudden Cardiac Arrest and Death in High School Athletes in the United States. Mayo Clin Proc. 2016;91:1493-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 94] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 2. | Corrado D, Link MS, Calkins H. Arrhythmogenic Right Ventricular Cardiomyopathy. N Engl J Med. 2017;376:61-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 607] [Cited by in RCA: 508] [Article Influence: 56.4] [Reference Citation Analysis (0)] |
| 3. | Peterson DF, Kucera K, Thomas LC, Maleszewski J, Siebert D, Lopez-Anderson M, Zigman M, Schattenkerk J, Harmon KG, Drezner JA. Aetiology and incidence of sudden cardiac arrest and death in young competitive athletes in the USA: a 4-year prospective study. Br J Sports Med. 2021;55:1196-1203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 119] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 4. | Lear A, Patel N, Mullen C, Simonson M, Leone V, Koshiaris C, Nunan D. Screening Electrocardiogram in Young Athletes and Military Members: A Systematic Review and Meta-Analysis. J Athl Train. 2022;57:444-451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Malhotra A, Sharma S. Outcomes of Cardiac Screening in Adolescent Soccer Players. N Engl J Med. 2018;379:2084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 6. | Maron BJ, Haas TS, Doerer JJ, Thompson PD, Hodges JS. Comparison of U.S. and Italian experiences with sudden cardiac deaths in young competitive athletes and implications for preparticipation screening strategies. Am J Cardiol. 2009;104:276-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 149] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 7. | Friedman KG, Alexander ME. Chest pain and syncope in children: a practical approach to the diagnosis of cardiac disease. J Pediatr. 2013;163:896-901.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | Corrado D, Pelliccia A, Heidbuchel H, Sharma S, Link M, Basso C, Biffi A, Buja G, Delise P, Gussac I, Anastasakis A, Borjesson M, Bjørnstad HH, Carrè F, Deligiannis A, Dugmore D, Fagard R, Hoogsteen J, Mellwig KP, Panhuyzen-Goedkoop N, Solberg E, Vanhees L, Drezner J, Estes NA 3rd, Iliceto S, Maron BJ, Peidro R, Schwartz PJ, Stein R, Thiene G, Zeppilli P, McKenna WJ; Section of Sports Cardiology, European Association of Cardiovascular Prevention and Rehabilitation. Recommendations for interpretation of 12-lead electrocardiogram in the athlete. Eur Heart J. 2010;31:243-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 677] [Cited by in RCA: 522] [Article Influence: 32.6] [Reference Citation Analysis (1)] |
| 9. | Basso C, Perazzolo Marra M, Rizzo S, De Lazzari M, Giorgi B, Cipriani A, Frigo AC, Rigato I, Migliore F, Pilichou K, Bertaglia E, Cacciavillani L, Bauce B, Corrado D, Thiene G, Iliceto S. Arrhythmic Mitral Valve Prolapse and Sudden Cardiac Death. Circulation. 2015;132:556-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 464] [Article Influence: 42.2] [Reference Citation Analysis (0)] |
| 10. | Sarto P, Zorzi A, Merlo L, Vessella T, Pegoraro C, Giorgiano F, Graziano F, Basso C, Drezner JA, Corrado D. Value of screening for the risk of sudden cardiac death in young competitive athletes. Eur Heart J. 2023;44:1084-1092. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 70] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 11. | Wheeler MT, Heidenreich PA, Froelicher VF, Hlatky MA, Ashley EA. Cost-effectiveness of preparticipation screening for prevention of sudden cardiac death in young athletes. Ann Intern Med. 2010;152:276-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 160] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 12. | Dhutia H, Malhotra A, Finocchiaro G, Parpia S, Bhatia R, D'Silva A, Gati S, Mellor G, Narain R, Chandra N, Behr E, Tome M, Papadakis M, Sharma S. Diagnostic yield and financial implications of a nationwide electrocardiographic screening programme to detect cardiac disease in the young. Europace. 2021;23:1295-1301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 13. | Braunwald E. Hypertrophic Cardiomyopathy. N Engl J Med. 2025;393:1004-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 14. | Saade C, Fakhredin RB, El Achkar B, Ghieh D, Mayat A, Abchee A, Refaat M, Ismail H, El-Rayess H, Karout L, El Merhi F. Coronary Artery Anomalies and Associated Radiologic Findings. J Comput Assist Tomogr. 2019;43:572-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Baggish AL, Ackerman MJ, Lampert R. Competitive Sport Participation Among Athletes With Heart Disease: A Call for a Paradigm Shift in Decision Making. Circulation. 2017;136:1569-1571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 59] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 16. | Schwartz PJ, Stramba-Badiale M, Crotti L, Pedrazzini M, Besana A, Bosi G, Gabbarini F, Goulene K, Insolia R, Mannarino S, Mosca F, Nespoli L, Rimini A, Rosati E, Salice P, Spazzolini C. Prevalence of the congenital long-QT syndrome. Circulation. 2009;120:1761-1767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 892] [Cited by in RCA: 751] [Article Influence: 44.2] [Reference Citation Analysis (0)] |
| 17. | Maron BJ, Friedman RA, Kligfield P, Levine BD, Viskin S, Chaitman BR, Okin PM, Saul JP, Salberg L, Van Hare GF, Soliman EZ, Chen J, Matherne GP, Bolling SF, Mitten MJ, Caplan A, Balady GJ, Thompson PD; American Heart Association Council on Clinical Cardiology, Advocacy Coordinating Committee, Council on Cardiovascular Disease in the Young, Council on Cardiovascular Surgery and Anesthesia, Council on Epidemiology and Prevention, Council on Functional Genomics and Translational Biology, Council on Quality of Care and Outcomes Research, and American College of Cardiology. Assessment of the 12-lead ECG as a screening test for detection of cardiovascular disease in healthy general populations of young people (12-25 Years of Age): a scientific statement from the American Heart Association and the American College of Cardiology. Circulation. 2014;130:1303-1334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 167] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 18. | Steinfurt J, Biermann J, Bode C, Odening KE. The Diagnosis, Risk Stratification, and Treatment of Brugada Syndrome. Dtsch Arztebl Int. 2015;112:394-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Gollob MH, Green MS, Tang AS, Gollob T, Karibe A, Ali Hassan AS, Ahmad F, Lozado R, Shah G, Fananapazir L, Bachinski LL, Roberts R. Identification of a gene responsible for familial Wolff-Parkinson-White syndrome. N Engl J Med. 2001;344:1823-1831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 550] [Cited by in RCA: 413] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 20. | Lu CW, Wu MH, Chen HC, Kao FY, Huang SK. Epidemiological profile of Wolff-Parkinson-White syndrome in a general population younger than 50 years of age in an era of radiofrequency catheter ablation. Int J Cardiol. 2014;174:530-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 21. | Wilson MG, Basavarajaiah S, Whyte GP, Cox S, Loosemore M, Sharma S. Efficacy of personal symptom and family history questionnaires when screening for inherited cardiac pathologies: the role of electrocardiography. Br J Sports Med. 2008;42:207-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 88] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 22. | Steinvil A, Chundadze T, Zeltser D, Rogowski O, Halkin A, Galily Y, Perluk H, Viskin S. Mandatory electrocardiographic screening of athletes to reduce their risk for sudden death proven fact or wishful thinking? J Am Coll Cardiol. 2011;57:1291-1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 191] [Article Influence: 12.7] [Reference Citation Analysis (2)] |
| 23. | Petek BJ, Baggish AL. Current controversies in pre-participation cardiovascular screening for young competitive athletes. Expert Rev Cardiovasc Ther. 2020;18:435-442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 24. | Drezner JA, Ackerman MJ, Anderson J, Ashley E, Asplund CA, Baggish AL, Börjesson M, Cannon BC, Corrado D, DiFiori JP, Fischbach P, Froelicher V, Harmon KG, Heidbuchel H, Marek J, Owens DS, Paul S, Pelliccia A, Prutkin JM, Salerno JC, Schmied CM, Sharma S, Stein R, Vetter VL, Wilson MG. Electrocardiographic interpretation in athletes: the 'Seattle criteria'. Br J Sports Med. 2013;47:122-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 454] [Cited by in RCA: 358] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 25. | Papadakis M, Carre F, Kervio G, Rawlins J, Panoulas VF, Chandra N, Basavarajaiah S, Carby L, Fonseca T, Sharma S. The prevalence, distribution, and clinical outcomes of electrocardiographic repolarization patterns in male athletes of African/Afro-Caribbean origin. Eur Heart J. 2011;32:2304-2313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 233] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 26. | Corrado D, Basso C, Pavei A, Michieli P, Schiavon M, Thiene G. Trends in sudden cardiovascular death in young competitive athletes after implementation of a preparticipation screening program. JAMA. 2006;296:1593-1601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1238] [Cited by in RCA: 926] [Article Influence: 46.3] [Reference Citation Analysis (0)] |
| 27. | Papadakis M, Sharma S, Cox S, Sheppard MN, Panoulas VF, Behr ER. The magnitude of sudden cardiac death in the young: a death certificate-based review in England and Wales. Europace. 2009;11:1353-1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 113] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 28. | Maron BJ, Zipes DP. Introduction: eligibility recommendations for competitive athletes with cardiovascular abnormalities-general considerations. J Am Coll Cardiol. 2005;45:1318-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 362] [Article Influence: 17.2] [Reference Citation Analysis (1)] |
| 29. | Fuller CM. Cost effectiveness analysis of screening of high school athletes for risk of sudden cardiac death. Med Sci Sports Exerc. 2000;32:887-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 101] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 30. | Leslie LK, Cohen JT, Newburger JW, Alexander ME, Wong JB, Sherwin ED, Rodday AM, Parsons SK, Triedman JK. Costs and benefits of targeted screening for causes of sudden cardiac death in children and adolescents. Circulation. 2012;125:2621-2629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 31. | Lampert R, Olshansky B, Heidbuchel H, Lawless C, Saarel E, Ackerman M, Calkins H, Estes NAM, Link MS, Maron BJ, Marcus F, Scheinman M, Wilkoff BL, Zipes DP, Berul CI, Cheng A, Jordaens L, Law I, Loomis M, Willems R, Barth C, Broos K, Brandt C, Dziura J, Li F, Simone L, Vandenberghe K, Cannom D. Safety of Sports for Athletes With Implantable Cardioverter-Defibrillators: Long-Term Results of a Prospective Multinational Registry. Circulation. 2017;135:2310-2312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 111] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 32. | Sharma S, Drezner JA, Baggish A, Papadakis M, Wilson MG, Prutkin JM, La Gerche A, Ackerman MJ, Borjesson M, Salerno JC, Asif IM, Owens DS, Chung EH, Emery MS, Froelicher VF, Heidbuchel H, Adamuz C, Asplund CA, Cohen G, Harmon KG, Marek JC, Molossi S, Niebauer J, Pelto HF, Perez MV, Riding NR, Saarel T, Schmied CM, Shipon DM, Stein R, Vetter VL, Pelliccia A, Corrado D. International Recommendations for Electrocardiographic Interpretation in Athletes. J Am Coll Cardiol. 2017;69:1057-1075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 261] [Article Influence: 29.0] [Reference Citation Analysis (1)] |
| 33. | Moulson N, Petek BJ, Drezner JA, Harmon KG, Kliethermes SA, Patel MR, Baggish AL; Outcomes Registry for Cardiac Conditions in Athletes Investigators. SARS-CoV-2 Cardiac Involvement in Young Competitive Athletes. Circulation. 2021;144:256-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 198] [Article Influence: 39.6] [Reference Citation Analysis (0)] |
| 34. | Drezner JA, Sharma S, Baggish A, Papadakis M, Wilson MG, Prutkin JM, Gerche A, Ackerman MJ, Borjesson M, Salerno JC, Asif IM, Owens DS, Chung EH, Emery MS, Froelicher VF, Heidbuchel H, Adamuz C, Asplund CA, Cohen G, Harmon KG, Marek JC, Molossi S, Niebauer J, Pelto HF, Perez MV, Riding NR, Saarel T, Schmied CM, Shipon DM, Stein R, Vetter VL, Pelliccia A, Corrado D. International criteria for electrocardiographic interpretation in athletes: Consensus statement. Br J Sports Med. 2017;51:704-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 325] [Article Influence: 36.1] [Reference Citation Analysis (1)] |
| 35. | Harmon KG, Asif IM, Maleszewski JJ, Owens DS, Prutkin JM, Salerno JC, Zigman ML, Ellenbogen R, Rao AL, Ackerman MJ, Drezner JA. Incidence, Cause, and Comparative Frequency of Sudden Cardiac Death in National Collegiate Athletic Association Athletes: A Decade in Review. Circulation. 2015;132:10-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 500] [Cited by in RCA: 404] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 36. | Hernelahti M, Heinonen OJ, Karjalainen J, Nylander E, Börjesson M. Sudden cardiac death in young athletes: time for a Nordic approach in screening? Scand J Med Sci Sports. 2008;18:132-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 37. | Roberts WO, Asplund CA, O'Connor FG, Stovitz SD. Cardiac preparticipation screening for the young athlete: why the routine use of ECG is not necessary. J Electrocardiol. 2015;48:311-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 38. | Asif IM, Johnson S, Schmieg J, Smith T, Rao AL, Harmon KG, Salerno JC, Drezner JA. The psychological impact of cardiovascular screening: the athlete's perspective. Br J Sports Med. 2014;48:1162-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 39. | Corrado D, Basso C, Schiavon M, Thiene G. Screening for hypertrophic cardiomyopathy in young athletes. N Engl J Med. 1998;339:364-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 745] [Cited by in RCA: 544] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 40. | Kusma JD, Raphael JL, Perrin JM, Hudak ML; Committee on Child Health Financing. Medicaid and the Children's Health Insurance Program: Optimization to Promote Equity in Child and Young Adult Health. Pediatrics. 2023;152:e2023064088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 41. | Hoagland A, Yu O, Horný M. Inequities in Unexpected Cost-Sharing for Preventive Care in the United States. Am J Prev Med. 2025;68:5-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 42. | Armoundas AA, Narayan SM, Arnett DK, Spector-Bagdady K, Bennett DA, Celi LA, Friedman PA, Gollob MH, Hall JL, Kwitek AE, Lett E, Menon BK, Sheehan KA, Al-Zaiti SS; American Heart Association Institute for Precision Cardiovascular Medicine; Council on Cardiovascular and Stroke Nursing; Council on Lifelong Congenital Heart Disease and Heart Health in the Young; Council on Cardiovascular Radiology and Intervention; Council on Hypertension; Council on the Kidney in Cardiovascular Disease; and Stroke Council. Use of Artificial Intelligence in Improving Outcomes in Heart Disease: A Scientific Statement From the American Heart Association. Circulation. 2024;149:e1028-e1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 169] [Article Influence: 84.5] [Reference Citation Analysis (0)] |
| 43. | Halkin A, Steinvil A, Rosso R, Adler A, Rozovski U, Viskin S. Preventing sudden death of athletes with electrocardiographic screening: what is the absolute benefit and how much will it cost? J Am Coll Cardiol. 2012;60:2271-2276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 44. | Hazinski MF, Markenson D, Neish S, Gerardi M, Hootman J, Nichol G, Taras H, Hickey R, OConnor R, Potts J, van der Jagt E, Berger S, Schexnayder S, Garson A Jr, Doherty A, Smith S; American Heart Association; American Academy of Pediatrics; American College of Emergency Physicians; American National Red Cross; National Association of School Nurses; National Association of State EMS Directors; National Association of EMS Physicians; National Association of Emergency Medical Technicians; Program for School Preparedness and Planning; National Center for Disaster Preparedness; Columbia University Mailman School of Public Health. Response to cardiac arrest and selected life-threatening medical emergencies: the medical emergency response plan for schools: A statement for healthcare providers, policymakers, school administrators, and community leaders. Circulation. 2004;109:278-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 102] [Article Influence: 4.6] [Reference Citation Analysis (0)] |