Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.116033
Revised: December 29, 2025
Accepted: February 27, 2026
Published online: April 26, 2026
Processing time: 165 Days and 0.2 Hours
Wire-based pressure pullback gradient (PPG) is the reference method for differentiating focal from diffuse coronary artery disease (CAD). However, it requires invasive instrumentation and hyperaemia. The quantitative flow ratio (QFR)-derived PPG [QFR virtual pullback (QVP) index] is a non-invasive alternative.
To evaluate the correlation between QVP index and PPG, and to explore the diagnostic performance of QVP index for identifying focal CAD.
We retrospectively studied 74 patients (86 vessels) who underwent coronary angiography, fractional flow reserve (FFR), wire-based PPG, angio-based QFR and QVP index between December 2021 and October 2023. The primary analysis focused on FFR-significant lesions (FFR ≤ 0.75, n = 31), as these are clinically relevant for guiding percutaneous coronary intervention. QVP index was cal
QFR was strongly correlated with FFR (r = 0.84, P < 0.001). In FFR significant vessels (FFR ≤ 0.75, n = 31), QVP index showed a moderate correlation with PPG (r = 0.45, P = 0.01). QVP index demonstrated excellent intra-observer and inter-observer variability with intraclass correlation coefficients of 0.918 (P < 0.001) and 0.932 (P < 0.001), respectively. QVP index predicted focal disease (defined as PPG > 0.73) with area under the curve of 0.73 (P = 0.02). A retrospectively derived threshold of QVP index > 0.53 yielded 90% sensitivity and 53% specificity (P = 0.04), though this cut-off was derived from the same dataset and should be regarded as hypothesis-generating.
QVP index correlates with PPG in FFR-significant lesions and may help to identify focal CAD patterns. However, these findings are hypothesis-generating and derived from a small, retrospective, single-centre cohort without external validation. Prospective multicentre studies are needed to validate cut-offs and determine whether QVP index provides incremental clinical value beyond existing physiological and imaging tools.
Core Tip: Quantitative flow ratio virtual pullback index is a non-invasive, high-sensitivity tool that accurately predicts focal coronary artery disease, offering an alternative to pressure pullback gradient for percutaneous coronary intervention decision-making without the need for hyperemia or further invasive procedures.
- Citation: Teoh ZH, Wardill T, Zhang M, O’Brien J, Chew DP, Ko B, Wong DTL. Quantitative flow ratio virtual pullback index predicts focal coronary artery disease identified on wire-based pullback pressure gradient. World J Cardiol 2026; 18(4): 116033
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/116033.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.116033
The outcome of percutaneous coronary intervention (PCI) guided by functional assessment using fractional flow reserve (FFR) is superior to angiography guided PCI[1-3]. The outcome from PCI differs depending on the pattern of coronary artery disease (CAD). The pattern of CAD can be evaluated invasively by measuring invasive FFR pullback pressure gradients during continuous hyperemia (pressure pullback gradient (PPG))[4-6].
Recent studies have demonstrated that PCI on diffuse patterns of CAD is associated with suboptimal post-procedural results, as compared to focal disease[7,8]. This disparity has been linked to poorer long-term outcomes and recurrent anginal symptoms, underscoring the importance of accurately assessing disease patterns prior to undertaking PCI[9].
However, routine assessment of FFR in all coronary lesions does not result in significant improvement in quality of life, nor is this cost effective[10]. Hyperaemia can also be contraindicated in certain patient groups and invasive wiring, albeit rare, can also result in complications[11-14].
Given these limitations, there is a growing interest in non-invasive techniques that circumvent the need for inducing hyperemia. Angiogram-based quantitative flow ratio (QFR) has emerged as a promising alternative, demonstrating performance comparable to its invasive counterpart, FFR[15,16]. Moreover, QFR has the added advantage of being non-invasive and not requiring hyperemic agents, thus potentially mitigating patient discomfort and procedural risks.
Using a similar concept to that of invasively derived PPG, a PPG can be derived from the QFR i.e. the QFR-derived PPG [QFR virtual pullback (QVP) index]. This would serve as an ideal alternative to PPG without the need for further invasive wiring and does not require hyperaemia. This technology is relatively new and not as established as PPG, but several studies have shown that, when utilized appropriately, it can also help predict favourable post-PCI outcomes in a similar manner[17-19].
Severe knowledge gaps remain. Although both PPG and QVP index are used to delineate patterns of CAD, there is no universally agreed cut-off point for significance in either metric, and no study has directly compared the performance of PPG with that of QVP index. Clinically, there is an unmet need for a non-invasive, hyperaemia-free method to characterize focal vs diffuse disease patterns in lesions that are already functionally significant, in order to support PCI planning without additional wire-based instrumentation. Accordingly, our study aimed primarily to evaluate the correlation between QVP index and PPG and to assess the diagnostic performance of QVP index for identifying focal CAD, with an exploratory secondary objective of examining its association with post-PCI FFR.
Our study seeks to address these gaps by directly comparing the diagnostic accuracy of QVP index to PPG. Ad
A retrospective single-centre study was performed on patients who underwent FFR and PPG assessments at a high volume, metropolitan Australian network. The study protocol was approved by the Local Human Research Ethics Com
Invasive coronary angiography was performed as per standard practice via either femoral or radial approach at the discretion of the operator. For FFR, the pressure wire (PressureWire X; Abbott Vascular) was calibrated and electronically equalised with the aortic pressure before being placed distal to the stenosis in the distal third of the coronary artery being interrogated. Intracoronary glyceryl trinitrate (100 μg) was injected to minimise vasospasm. Intravenous adenosine was administered (180 μg/kg/minute) through an intravenous line in the antecubital fossa. At steady-state hyperaemia, FFR was assessed using the Coroflow v3.5.1 software (Coroventis Research AB) calculated by dividing the mean coronary pressure measured with the pressure sensor placed distal to the stenosis by the mean aortic pressure measured through the guide catheter. FFR of ≤ 0.75 was taken to define ischemia in the interrogated artery and its supplied territory[2,20-23].
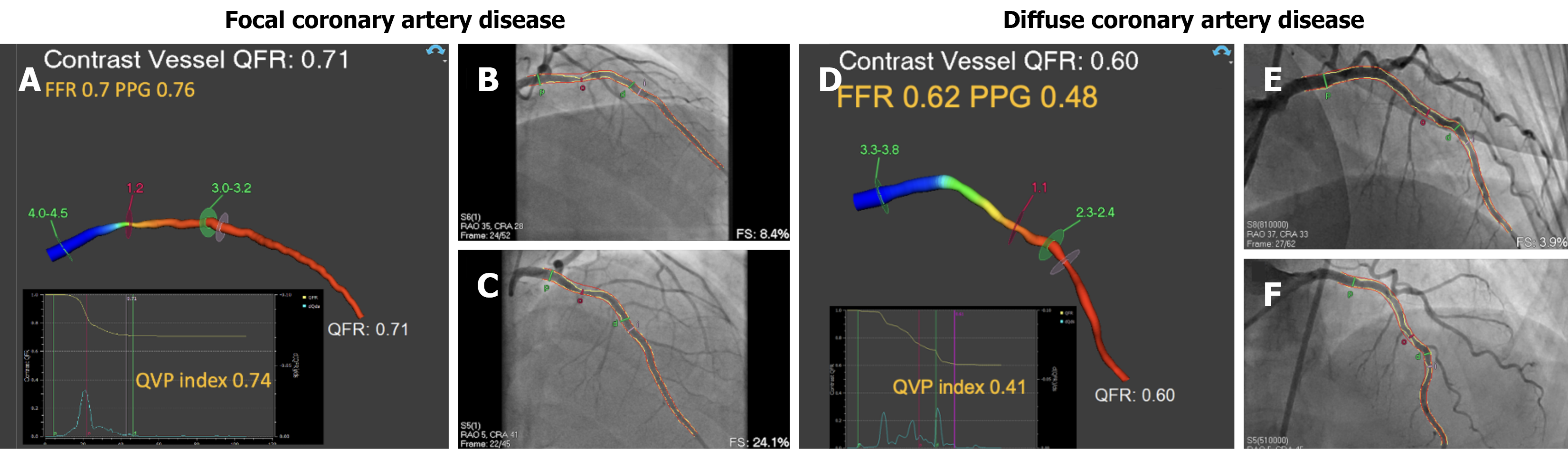
The calculation of the PPG is derived from the FFR curve by combining two parameters: (1) Maximal PPG over 20 mm, depicting the magnitude FFR drop; and (2) The length of epicardial coronary segments with FFR deterioration.
PPG = [Max PPG20mm/∆FFRvessel + (1 - length with functional disease/total vessel length)]/2. PPG is a continuous metric with values approaching 1.0 representing focal CAD whereas values close to 0 representing diffuse CAD. For our study, focal pattern of CAD was defined as PPG > 0.73[7].
QFR analysis was undertaken using Quantitative Flow Ratio Research Edition v2.2 (Medis Medical Imaging System, Leiden, Netherlands) by an independent operator, blinded to clinical information. Analysis was performed on two angiographic acquisitions that were separated by ≥ 25°, ensuring that the angiographic projections had minimal foreshortening of the stenosis, and minimal overlap of the main vessel and the side branches. Vessel QFR was recorded. Two-dimensional quantitative coronary angiography was performed, and percentage diameter stenosis, lesion length and minimum lumen diameter recorded.
The QVP index combined the maximal drop of the QFR value over 20 mm (Max QFR20mm) and the length of epicardial coronary segments with QFR deterioration. Specifically, the QVP index was calculated as follows: QVP = [Max QFR20mm/∆QFRvessel + (1 - length with functional disease/total vessel length)]/2. QVP index is a continuous metric with values approaching 1 representing focal CAD whereas values close to 0 representing diffuse CAD.
Continuous variables are expressed as mean ± SD or median (quartiles) as appropriate, whereas categorical variables are expressed as percentage. Continuous and categorical variables were compared using t-test, Mann-Whitney or χ² test as appropriate. Correlation was assessed using a Pearson’s or Spearman’s correlation coefficient as appropriate. Receiver operating characteristic analysis was performed to evaluate the discriminatory ability of QVP index for focal PPG defined as PPG > 0.73. Statistical analysis was performed using SPSS version 30 and a P value of < 0.05 was considered statistically significant (IBM Corporation, Armonk, NY, United States).
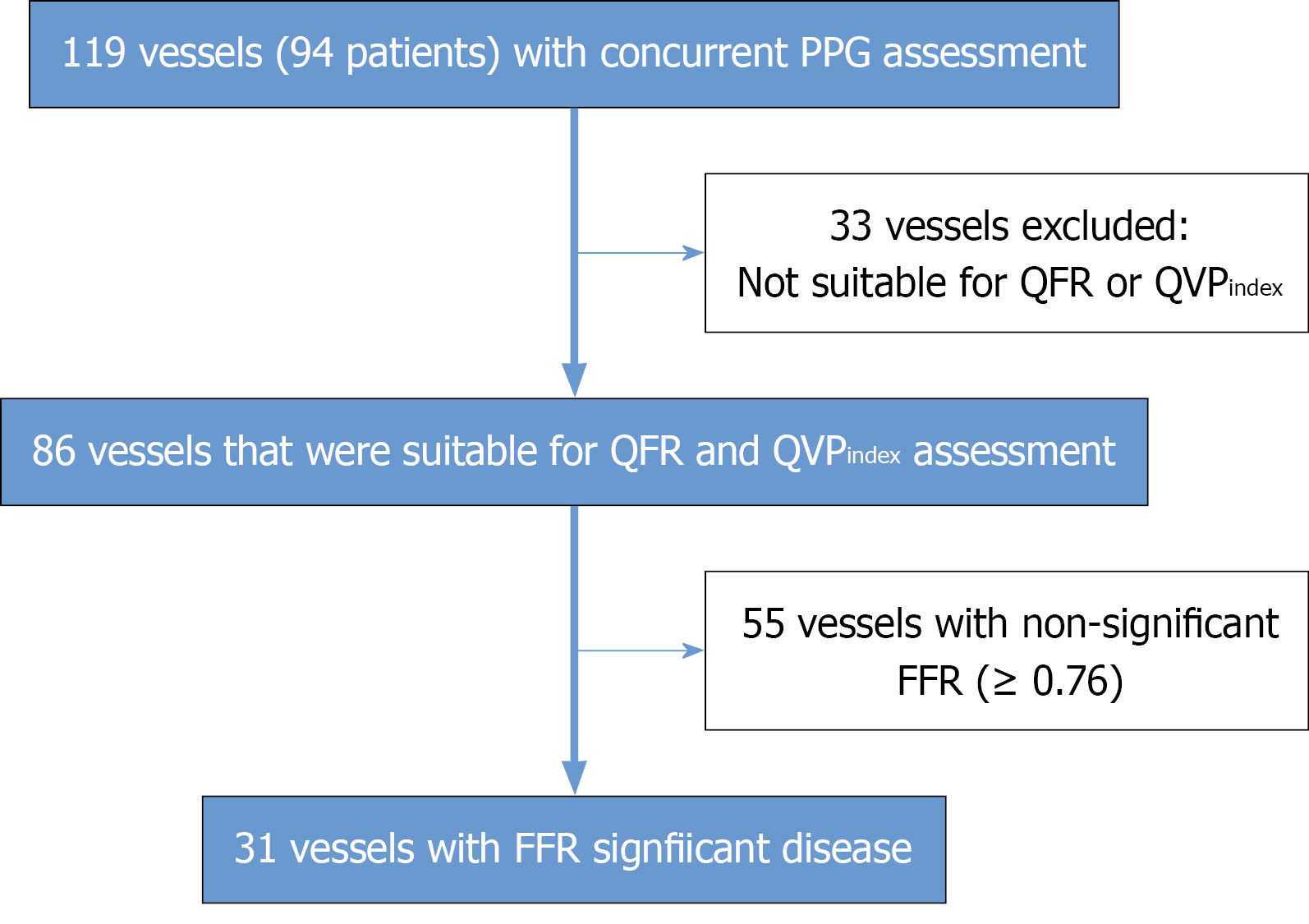
From December 2021 to October 2023, 119 vessels (94 patients) had FFR and concurrent wire-based PPG assessment but QVP index was measurable in only 75% of the vessels i.e. 86 vessels (74 patients). The study flowchart is shown in Figure 1. Reasons for exclusion include inadequate orthogonal views by ≥ 25°, significant overlap of the main vessel and side branches, ostial lesions, inadequate lesion visualization and vessels providing collateral circulation to chronic total occlusion territory.
The mean age was 65 ± 10.5 years with majority of patients being male (75.7%). The median FFR was 0.78 (interquartile range = 0.12) while the median QFR was 0.79 (interquartile range = 0.16).
A total of 31 vessels (36%) were found to be FFR significant (defined as FFR ≤ 0.75). Clinical characteristics stratified according to disease pattern i.e. focal and diffuse are displayed in Table 1.
| Patient characteristics | Overall (n = 74) | Focal (n = 10) | Diffuse (n = 21) | P value |
| Age (years) | 65 ± 10.5 | 61 ± 11.3 | 62 ± 12.2 | 0.42 |
| Male | 56 (75.7) | 9 (90) | 18 (86) | 0.74 |
| Body mass index (kg/m2) | 28.8 ± 4.6 | 28.7 ± 4.8 | 28.5 ± 3.4 | 0.97 |
| Caucasian | 51 (68.9) | 6 (60) | 16 (76) | 0.35 |
| Atherosclerotic cardiovascular risk factors | ||||
| Hypertension | 53 (71.6) | 6 (60) | 15 (71) | 0.53 |
| Dyslipidaemia | 49 (66.2) | 7 (70) | 12 (57) | 0.49 |
| Diabetes mellitus | 25 (33.8) | 2 (20) | 7 (33) | 0.45 |
| Cigarette smoking | 9 (12.2) | 2 (20) | 0 | 0.34 |
| Previous myocardial infarction | 10 (13.5) | 1 (10) | 2 (9.5) | 0.97 |
| Previous percutaneous coronary intervention | 14 (18.9) | 1 (10) | 3 (14.3) | 0.74 |
| Vessels | n = 86 | 0.99 | ||
| Left anterior descending artery | 62 (72.1) | 7 (70) | 15 (71.4) | |
| Left circumflex | 8 (9.3) | 1 (10) | 2 (9.5) | |
| Right coronary artery | 16 (18.6) | 2 (20) | 4 (19.0) |
QFR demonstrated a strong correlation with FFR across all analysable vessels (n = 86), with a correlation coefficient of r = 0.84 (P < 0.001). QFR also effectively identified FFR-significant lesions (defined as FFR ≤ 0.75, n = 31), with an area under the curve (AUC) of 0.94 (P < 0.001).
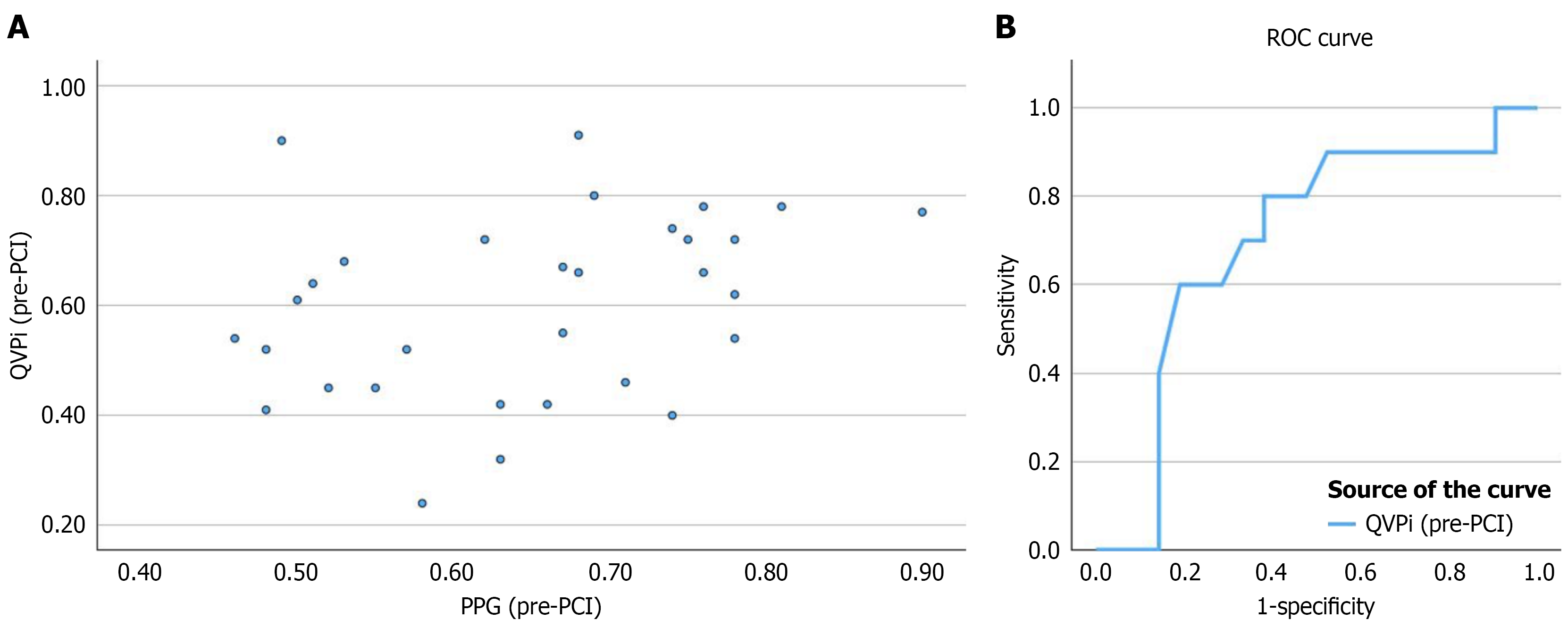
When assessed across the entire cohort (all vessels with QVP index measurements, n = 86), QVP index showed only a weak correlation with PPG (r = 0.22, P = 0.04). In the clinically relevant subgroup of FFR-significant lesions (n = 31), QVP index displayed a stronger, moderate correlation with PPG (r = 0.45, P = 0.01; Figures 2 and 3).
QVP index predicted focal disease (defined as PPG > 0.73) with AUC of 0.73 (P = 0.02). A retrospectively derived QVP index threshold of > 0.53 to define focal disease achieved a sensitivity of 90% and specificity of 48% (P = 0.041). This cut-off was derived from the same dataset and should be regarded as hypothesis-generating rather than clinically actionable.
When focal disease was alternatively defined by the median PPG value within our cohort (PPG > 0.67), QVP index demonstrated an improved discriminatory ability (AUC = 0.81, P < 0.001). In this analysis, QVP index > 0.53 identified focal disease with 86% sensitivity and 53% specificity (P = 0.025). Again, these findings are exploratory and require external validation.
QVP index demonstrated excellent reproducibility, with intra-observer and inter-observer intraclass correlation coefficients of 0.918 (P < 0.001) and 0.932 (P < 0.001), respectively.
In the subset of 21 patients with available post-PCI FFR measurements, we performed exploratory receiver operating characteristic analyses to assess whether any pre-procedural or post-procedural physiological indices could identify vessels achieving a post-PCI FFR > 0.90. Among the indices assessed, the pre-PCI QVP index demonstrated the highest numerical AUC = 0.71, P = 0.07, although this did not reach statistical significance. Pre-PCI PPG showed a lower AUC of 0.66 (P = 0.20), while pre-PCI FFR and pre-PCI QFR yielded AUCs of 0.42 (P = 0.59) and 0.49 (P = 0.84), respectively. Post-PCI QFR and post-PCI QVP index had AUCs of 0.51 (P = 0.37) and 0.64 (P = 0.37).
Given the small sample size, particularly the limited number of vessels with post-PCI FFR ≤ 0.90, these findings should be regarded as purely hypothesis-generating, and no comparative testing between AUC values was undertaken.
This study provides insights into the diagnostic performance of the QVP index compared with the PPG in identifying focal patterns of CAD. Several key findings emerged. First, QFR correlated strongly with FFR across the cohort and reliably identified FFR-significant lesions. Second, QVP index demonstrated a moderate correlation with PPG specifically within the FFR-significant subgroup, which represents the clinically relevant population for PCI planning. Finally, in an exploratory analysis of the small subset with post-PCI FFR measurements, the pre-PCI QVP index showed the highest numerical AUC for predicting post-PCI FFR > 0.90, including when compared with pre-PCI FFR and other physiological indices. However, none of these results reached statistical significance, and this observation should therefore be regarded solely as hypothesis-generating rather than indicative of any true predictive advantage.
When considering potential clinical implementation, it is important to place QVP index in the context of existing tools used to integrate anatomical and functional information, such as expert visual-functional assessment, intravascular imaging (intravascular ultrasound-optical coherence tomography), and QFR alone. In our study, QVP index showed only a moderate correlation with PPG in FFR-significant lesions, and its diagnostic performance for focal disease must be interpreted cautiously in light of the sample size and retrospective design. At present, there are insufficient data to conclude that QVP index provides superior or incremental value over these simpler or more established approaches. Rather, our findings support its role as a candidate non-invasive metric that warrants further prospective validation alongside, and not in place of, current physiological and imaging strategies.
PPG is an innovative metric that enables clinicians to differentiate between focal CAD and diffuse lesions, which are associated with poorer outcomes post-PCI[4-6]. However, this technique necessitates invasive wiring and the induction of hyperemia, both of which carry inherent risks[8-11]. In contrast, QVP index utilizes non-invasive, non-hyperemia technology, presenting a potential solution to these limitations. Our findings show that QVP index moderately correlates with PPG, particularly in FFR-significant lesions, highlighting its potential to serve as a surrogate tool for assessing CAD pattern.
The choice of an FFR cutoff of 0.75 is based on several considerations. First, PCI performed on lesions above this threshold has not been shown to improve mortality or myocardial infarction rates[22]. Furthermore, research by Petraco et al[23] indicates that at a cutoff of 0.80, the inherent biological variability of FFR can lead to significant changes in the probability of a lesion remaining FFR-significant if measured again after 10 minutes, approaching a 50% chance. Con
The use of cut-offs in continuous metrics such as FFR, QFR, PPG, and QVP index underscores their arbitrary nature; however, these thresholds are essential for aiding and standardizing clinical decision-making. Currently, there is no internationally accepted cut-off for identifying focal disease using PPG. In the largest PPG trial to date, the PPG Global Registry study, a median PPG value of 0.62 was employed to define focal disease. Nonetheless, subsequent sensitivity analyses revealed that a PPG cut-off of 0.73 better predicted post-PCI outcomes. The authors concluded that the es
In our study, we implemented two different cut-offs for defining focal disease. The first was a PPG > 0.73, based on evidence indicating superior outcomes following PCI in this cohort[7,8]. Alternatively, we defined focal disease as PPG > 0.67, aligning with the median PPG observed in our cohort, similar to the approach taken by Collet et al[4] and Collet et al[8] in their landmark PPG paper. Regardless of the cut-off chosen, QVP index accurately predicted focal disease, demonstrating AUC of 0.73 and 0.81, respectively.
Previous QVP index studies have similarly used a cut-off of 0.78, based on the median value within their cohorts, to delineate focal disease[25]. In our study, we propose a cut-off of 0.53, which yields high sensitivity in predicting the presence of focal disease.
To implement this in daily clinical practice, we recommend performing QFR and assessing the QVP index following angiography. If the values exceed 0.53, this indicates a focal pattern of disease as suggested by both the PPG and QVP indices, thereby supporting the decision for PCI[7,9].
However, it is important to note that a QVP index below 0.53 does not necessarily indicate a diffuse pattern of disease, given its lower specificity. In such cases, further confirmation with PPG may still be warranted to ensure accurate diagnosis and treatment planning.
In regard to defining optimal post-PCI FFR, there is no universally accepted cut-off, with thresholds reported in the literature ranging from approximately 0.80 to 0.92[8,26-28]. This variability reflects differences in study design, vessel territory, and clinical endpoints and underscores the lack of consensus on the physiological target most strongly associated with favourable outcomes. In our study, we selected a post-PCI FFR threshold of > 0.90 based on large meta-analyses, most notably Rimac et al[29], demonstrating that patients who achieve values above this level tend to experience lower rates of adverse events and repeat revascularisation.
In this context, our exploratory analysis provides additional insight into the behaviour of angio-derived and pressure-derived indices after PCI. Although the pre-PCI QVP index showed the highest numerical AUC for predicting post-PCI FFR > 0.90, including when compared with pre-PCI PPG, pre-PCI FFR, and pre-PCI QFR, none of the assessed phy
This study has several limitations. Firstly, the sample size was small, particularly in the FFR-significant cohort (n = 31) and in the subset with post-PCI FFR measurements (n = 21), and analyses were performed retrospectively at a single centre. As a result, the study lacks external validation and is underpowered for definitive clinical inference and should be regarded as hypothesis-generating rather than practice-changing.
Furthermore, PPG itself lacks a universally accepted cut-off for defining focal CAD, adding complexity to the interpretation of results. However, in order to address this issue, we used several different cut-offs based on existing literature[3-5], all of which suggest that QVP index is able to predict focal disease, as defined by PPG.
Finally, we used manual rather than motorized FFR pullback for practical reasons. However, this is unlikely to significantly affect our results, as studies have shown that PPG derived from manual pullbacks exhibits excellent reproducibility compared to motorized pullbacks[30].
In FFR-significant lesions, QVP index correlated with PPG and showed the ability to identify focal CAD patterns. The diagnostic thresholds proposed here were derived retrospectively in a single-centre cohort and require validation in larger, multicentre studies with external datasets before any clinical adoption can be considered. QVP index should be viewed as an investigational, non-invasive tool for evaluating disease distribution, and its precise clinical role relative to existing physiological and imaging strategies remains to be defined.
| 1. | van Nunen LX, Zimmermann FM, Tonino PA, Barbato E, Baumbach A, Engstrøm T, Klauss V, MacCarthy PA, Manoharan G, Oldroyd KG, Ver Lee PN, Van't Veer M, Fearon WF, De Bruyne B, Pijls NH; FAME Study Investigators. Fractional flow reserve versus angiography for guidance of PCI in patients with multivessel coronary artery disease (FAME): 5-year follow-up of a randomised controlled trial. Lancet. 2015;386:1853-1860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 379] [Cited by in RCA: 470] [Article Influence: 42.7] [Reference Citation Analysis (0)] |
| 2. | Tonino PA, De Bruyne B, Pijls NH, Siebert U, Ikeno F, van' t Veer M, Klauss V, Manoharan G, Engstrøm T, Oldroyd KG, Ver Lee PN, MacCarthy PA, Fearon WF; FAME Study Investigators. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360:213-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3610] [Cited by in RCA: 3054] [Article Influence: 179.6] [Reference Citation Analysis (2)] |
| 3. | Mangiacapra F, Paolucci L, De Bruyne B, Rioufol G, Hahn JY, Chen SL, Koo BK, Tonino PAL, van 't Veer M, Motreff P, Angoulvant D, Lee JM, Hwang D, Yang S, Pijls NHJ, Barbato E; Physiology and Revascularization for Myocardial Endpoints (PRIME) Collaboration. Fractional flow reserve vs angiography to guide percutaneous coronary intervention: an individual patient data meta-analysis. Eur Heart J. 2025;46:3851-3859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 4. | Collet C, Sonck J, Vandeloo B, Mizukami T, Roosens B, Lochy S, Argacha JF, Schoors D, Colaiori I, Di Gioia G, Kodeboina M, Suzuki H, Van 't Veer M, Bartunek J, Barbato E, Cosyns B, De Bruyne B. Measurement of Hyperemic Pullback Pressure Gradients to Characterize Patterns of Coronary Atherosclerosis. J Am Coll Cardiol. 2019;74:1772-1784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 212] [Article Influence: 30.3] [Reference Citation Analysis (1)] |
| 5. | Omari M, Ibrahem A, Bawamia B, Cartlidge T, Bagnall A, Purcell I, Egred M, Zaman A, Farag M, Alkhalil M. Pullback Pressure Gradient-An Emerging Concept in Patients with Coronary Artery Disease. Rev Cardiovasc Med. 2024;25:278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 6. | Carvalho PEP, Collet C, De Bruyne B, Munhoz D, Sonck J, Sara JDS, Strepkos D, Mutlu D, Alexandrou M, Ser OS, Brilakis ES, Sandoval Y. The Pullback Pressure Gradient: A Physiologic Index to Differentiate Focal From Diffuse Coronary Artery Disease. JACC Adv. 2025;4:101679. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 7. | Mizukami T, Sonck J, Sakai K, Ko B, Maeng M, Otake H, Koo BK, Nagumo S, Nørgaard BL, Leipsic J, Shinke T, Munhoz D, Mileva N, Belmonte M, Ohashi H, Barbato E, Johnson NP, De Bruyne B, Collet C. Procedural Outcomes After Percutaneous Coronary Interventions in Focal and Diffuse Coronary Artery Disease. J Am Heart Assoc. 2022;11:e026960. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 8. | Collet C, Munhoz D, Mizukami T, Sonck J, Matsuo H, Shinke T, Ando H, Ko B, Biscaglia S, Rivero F, Engstrøm T, Arslani K, Leone AM, van Nunen LX, Fearon WF, Christiansen EH, Fournier S, Desta L, Yong A, Adjedj J, Escaned J, Nakayama M, Eftekhari A, Zimmermann FM, Sakai K, Storozhenko T, da Costa BR, Campo G, West NEJ, De Potter T, Heggermont W, Buytaert D, Bartunek J, Berry C, Collison D, Johnson T, Amano T, Perera D, Jeremias A, Ali Z, Pijls NHJ, De Bruyne B, Johnson NP. Influence of Pathophysiologic Patterns of Coronary Artery Disease on Immediate Percutaneous Coronary Intervention Outcomes. Circulation. 2024;150:586-597. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 59] [Article Influence: 29.5] [Reference Citation Analysis (1)] |
| 9. | Collet C, Collison D, Mizukami T, McCartney P, Sonck J, Ford T, Munhoz D, Berry C, De Bruyne B, Oldroyd K. Differential Improvement in Angina and Health-Related Quality of Life After PCI in Focal and Diffuse Coronary Artery Disease. JACC Cardiovasc Interv. 2022;15:2506-2518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 10. | Stables RH, Mullen LJ, Elguindy M, Nicholas Z, Aboul-Enien YH, Kemp I, O'Kane P, Hobson A, Johnson TW, Khan SQ, Wheatcroft SB, Garg S, Zaman AG, Mamas MA, Nolan J, Jadhav S, Berry C, Watkins S, Hildick-Smith D, Gunn J, Conway D, Hoye A, Fazal IA, Hanratty CG, De Bruyne B, Curzen N. Routine Pressure Wire Assessment Versus Conventional Angiography in the Management of Patients With Coronary Artery Disease: The RIPCORD 2 Trial. Circulation. 2022;146:687-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 11. | Altstidl JM, Gaede L, Troebs M, Marwan M, Achenbach S. Side effects and major adverse cardiac events caused by fractional flow reserve measurement: a systematic review and meta-analysis of 12,215 patients. Eur Heart J. 2022;43:ehac544.2022. [DOI] [Full Text] |
| 12. | Sen S, Escaned J, Malik IS, Mikhail GW, Foale RA, Mila R, Tarkin J, Petraco R, Broyd C, Jabbour R, Sethi A, Baker CS, Bellamy M, Al-Bustami M, Hackett D, Khan M, Lefroy D, Parker KH, Hughes AD, Francis DP, Di Mario C, Mayet J, Davies JE. Development and validation of a new adenosine-independent index of stenosis severity from coronary wave-intensity analysis: results of the ADVISE (ADenosine Vasodilator Independent Stenosis Evaluation) study. J Am Coll Cardiol. 2012;59:1392-1402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 599] [Cited by in RCA: 521] [Article Influence: 37.2] [Reference Citation Analysis (1)] |
| 13. | Al-Lamee R, Thompson D, Dehbi HM, Sen S, Tang K, Davies J, Keeble T, Mielewczik M, Kaprielian R, Malik IS, Nijjer SS, Petraco R, Cook C, Ahmad Y, Howard J, Baker C, Sharp A, Gerber R, Talwar S, Assomull R, Mayet J, Wensel R, Collier D, Shun-Shin M, Thom SA, Davies JE, Francis DP; ORBITA investigators. Percutaneous coronary intervention in stable angina (ORBITA): a double-blind, randomised controlled trial. Lancet. 2018;391:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 830] [Cited by in RCA: 758] [Article Influence: 94.8] [Reference Citation Analysis (1)] |
| 14. | Van Nunen LX. Physiology-based treatment in stable angina and acute myocardial infarction. PhD Thesis. Technische Universiteit Eindhoven. |
| 15. | Tanigaki T, Emori H, Kawase Y, Kubo T, Omori H, Shiono Y, Sobue Y, Shimamura K, Hirata T, Matsuo Y, Ota H, Kitabata H, Okubo M, Ino Y, Matsuo H, Akasaka T. QFR Versus FFR Derived From Computed Tomography for Functional Assessment of Coronary Artery Stenosis. JACC Cardiovasc Interv. 2019;12:2050-2059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 16. | Xu B, Tu S, Song L, Jin Z, Yu B, Fu G, Zhou Y, Wang J, Chen Y, Pu J, Chen L, Qu X, Yang J, Liu X, Guo L, Shen C, Zhang Y, Zhang Q, Pan H, Fu X, Liu J, Zhao Y, Escaned J, Wang Y, Fearon WF, Dou K, Kirtane AJ, Wu Y, Serruys PW, Yang W, Wijns W, Guan C, Leon MB, Qiao S, Stone GW; FAVOR III China study group. Angiographic quantitative flow ratio-guided coronary intervention (FAVOR III China): a multicentre, randomised, sham-controlled trial. Lancet. 2021;398:2149-2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 314] [Article Influence: 62.8] [Reference Citation Analysis (1)] |
| 17. | Dai N, Zhang R, Hu N, Guan C, Zou T, Qiao Z, Zhang M, Duan S, Xie L, Dou K, Zhang Y, Xu B, Ge J. Integrated coronary disease burden and patterns to discriminate vessels benefiting from percutaneous coronary intervention. Catheter Cardiovasc Interv. 2022;99:E12-E21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 18. | Dai N, Zhang R, Yuan S, Hu N, Guan C, Zou T, Qiao Z, He J, Duan S, Xie L, Dou K, Zhang Y, Xu B, Ge J. Prognostic Implications of Quantitative Flow Ratio-Derived Physiological 2-Dimensional Residual Disease Patterns After Stenting. JACC Cardiovasc Interv. 2022;15:1624-1634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 19. | Biscaglia S, Uretsky BF, Tebaldi M, Erriquez A, Brugaletta S, Cerrato E, Quadri G, Spitaleri G, Colaiori I, Di Girolamo D, Scoccia A, Zucchetti O, D'Aniello E, Manfrini M, Pavasini R, Barbato E, Campo G. Angio-Based Fractional Flow Reserve, Functional Pattern of Coronary Artery Disease, and Prediction of Percutaneous Coronary Intervention Result: a Proof-of-Concept Study. Cardiovasc Drugs Ther. 2022;36:645-653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 20. | Kern MJ, Samady H. Current concepts of integrated coronary physiology in the catheterization laboratory. J Am Coll Cardiol. 2010;55:173-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 200] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 21. | Pijls NH, van Schaardenburgh P, Manoharan G, Boersma E, Bech JW, van't Veer M, Bär F, Hoorntje J, Koolen J, Wijns W, de Bruyne B. Percutaneous coronary intervention of functionally nonsignificant stenosis: 5-year follow-up of the DEFER Study. J Am Coll Cardiol. 2007;49:2105-2111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1301] [Cited by in RCA: 1131] [Article Influence: 59.5] [Reference Citation Analysis (1)] |
| 22. | Mohdnazri SR, Keeble TR, Sharp AS. Fractional Flow Reserve: Does a Cut-off Value add Value? Interv Cardiol. 2016;11:17-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 23. | Petraco R, Sen S, Nijjer S, Echavarria-Pinto M, Escaned J, Francis DP, Davies JE. Fractional flow reserve-guided revascularization: practical implications of a diagnostic gray zone and measurement variability on clinical decisions. JACC Cardiovasc Interv. 2013;6:222-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 134] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 24. | Munhoz D, Collet C, Mizukami T, Yong A, Leone AM, Eftekhari A, Ko B, da Costa BR, Berry C, Collison D, Perera D, Christiansen EH, Rivero F, Zimmermann FM, Ando H, Matsuo H, Nakayama M, Escaned J, Sonck J, Sakai K, Adjedj J, Desta L, van Nunen LX, West NEJ, Fournier S, Storozhenko T, Amano T, Engstrøm T, Johnson T, Shinke T, Biscaglia S, Fearon WF, Ali Z, De Bruyne B, Johnson NP. Rationale and design of the pullback pressure gradient (PPG) global registry. Am Heart J. 2023;265:170-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 25. | Shin D, Dai N, Lee SH, Choi KH, Lefieux A, Molony D, Hwang D, Kim HK, Jeon KH, Lee HJ, Jang HJ, Ha SJ, Park TK, Yang JH, Song YB, Hahn JY, Choi SH, Doh JH, Shin ES, Nam CW, Koo BK, Gwon HC, Ge J, Lee JM. Physiological Distribution and Local Severity of Coronary Artery Disease and Outcomes After Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2021;14:1771-1785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 26. | Piroth Z, Otsuki H, Zimmermann FM, Ferenci T, Keulards DCJ, Yeung AC, Pijls NHJ, De Bruyne B, Fearon WF. Prognostic Value of Measuring Fractional Flow Reserve After Percutaneous Coronary Intervention in Patients With Complex Coronary Artery Disease: Insights From the FAME 3 Trial. Circ Cardiovasc Interv. 2022;15:884-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 27. | Piroth Z, Toth GG, Tonino PAL, Barbato E, Aghlmandi S, Curzen N, Rioufol G, Pijls NHJ, Fearon WF, Jüni P, De Bruyne B. Prognostic Value of Fractional Flow Reserve Measured Immediately After Drug-Eluting Stent Implantation. Circ Cardiovasc Interv. 2017;10:e005233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 114] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 28. | Hwang D, Koo BK, Zhang J, Park J, Yang S, Kim M, Yun JP, Lee JM, Nam CW, Shin ES, Doh JH, Chen SL, Kakuta T, Toth GG, Piroth Z, Johnson NP, Pijls NHJ, Hakeem A, Uretsky BF, Hokama Y, Tanaka N, Lim HS, Ito T, Matsuo A, Azzalini L, Leesar MA, Neleman T, van Mieghem NM, Diletti R, Daemen J, Collison D, Collet C, De Bruyne B. Prognostic Implications of Fractional Flow Reserve After Coronary Stenting: A Systematic Review and Meta-analysis. JAMA Netw Open. 2022;5:e2232842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (1)] |
| 29. | Rimac G, Fearon WF, De Bruyne B, Ikeno F, Matsuo H, Piroth Z, Costerousse O, Bertrand OF. Clinical value of post-percutaneous coronary intervention fractional flow reserve value: A systematic review and meta-analysis. Am Heart J. 2017;183:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 112] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 30. | Sonck J, Mizukami T, Johnson NP, Nagumo S, Gallinoro E, Candreva A, Mileva N, Munhoz D, Shinke T, Svanerud J, Barbato E, De Bruyne B, Collet C. Development, validation, and reproducibility of the pullback pressure gradient (PPG) derived from manual fractional flow reserve pullbacks. Catheter Cardiovasc Interv. 2022;99:1518-1525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |