Published online Apr 26, 2026. doi: 10.4330/wjc.v18.i4.116558
Revised: December 4, 2025
Accepted: January 8, 2026
Published online: April 26, 2026
Processing time: 151 Days and 15.5 Hours
We read with great interest the meta-analysis by Parsi et al showing the strong therapeutic effects of sodium-glucose cotransporter-2 inhibitors (SGLT2i) in heart failure. Because the expression of sodium-glucose cotransporter-2 in cardiomyo
Core Tip: Sodium-glucose cotransporter-2 inhibitors (SGLT2i) have revolutionized the treatment of heart failure, but their direct molecular effects in cardiomyocytes, which express little sodium-glucose cotransporter-2, have not been fully described. This letter summarizes current evidence and presents a conceptual framework in which Ca2+/calmodulin-dependent protein kinase II is a central target. We hypothesize that many of the cardioprotective effects of SGLT2i are mediated by the dampening of Ca2+/calmodulin-dependent protein kinase II activity through synergistic modulation of ionic balance, relief of oxidative stress and inflammatory signaling suppression. Together, these processes may provide a unified explanation for the observed improvements in electrical stability, ventricular performance, and cardiac remodeling associated with SGLT2i treatment.
- Citation: Zhang YF, Yang WX, Li XW. Letter to the Editor: Ca2+/calmodulin-dependent protein kinase II in heart failure and the role of sodium-glucose-cotransporter 2 inhibitors. World J Cardiol 2026; 18(4): 116558
- URL: https://www.wjgnet.com/1949-8462/full/v18/i4/116558.htm
- DOI: https://dx.doi.org/10.4330/wjc.v18.i4.116558
We read with great interest the meta-analysis by Parsi et al[1]. This rigorous work offers an excellent and robust synthesis of the randomized evidence, and provides clear evidence that sodium-glucose cotransporter-2 (SGLT2) inhibitors (SGLT2i) significantly reduce the risks of heart failure (HF) hospitalization, cardiovascular death, and overall mortality. Despite these well-established clinical benefits, the molecular mechanisms underlying these changes are not fully defined, particularly as SGLT2 is minimally expressed in cardiomyocytes. For this reason, the extraordinary cardioprotective effects of SGLT2i remain the subject of scientific study. In correspondence, we summarize emerging findings and offer an integrated perspective: Many of the therapeutic effects of SGLT2i in HF may be largely due to indirect suppression of Ca2+/calmodulin-dependent protein kinase II (CaMKII), a central regulator of pathological signaling in the failing heart. This proposed mechanism provides a biologically consistent framework that may help to explain the wide range of improvements seen with SGLT2i therapy.
Traditionally, the cardiovascular benefits of SGLT2i have been attributed primarily to systemic effects such as osmotic diuresis and resulting decreases in cardiac preload and afterload. Increasing evidence, however, indicates that these drugs have a variety of direct effects on the myocardium as well. Reported mechanisms include modulation of cardiomyocyte ionic balance, e.g., inhibition of the sodium-hydrogen exchanger 1 and altered activity of the Na+/Ca2+ exchanger (NCX)[2-4]; changes in cardiac energy metabolism with increased ketone body utilization and improved substrate preference[5,6]; and enhancements in mitochondrial performance, indicated by increased energetic efficiency and reducing oxidative stress[7,8]. While these mechanisms are important, this letter focuses on an additional and increasingly convincing concept: That a large part of the cardioprotective actions of SGLT2i may be mediated by the indirect inhibition of CaMKII, a key regulator of pathological signaling in the failing heart.
HF is a complex clinical syndrome that is linked to a high degree of morbidity, recurrent hospital admissions and high mortality rates[9]. In recent years, SGLT2i have evolved from being exclusively antihyperglycemic drugs to cornerstone medications for HF with benefits that go far beyond glycemic control[10]. Given that little to no SGLT2 protein is ex
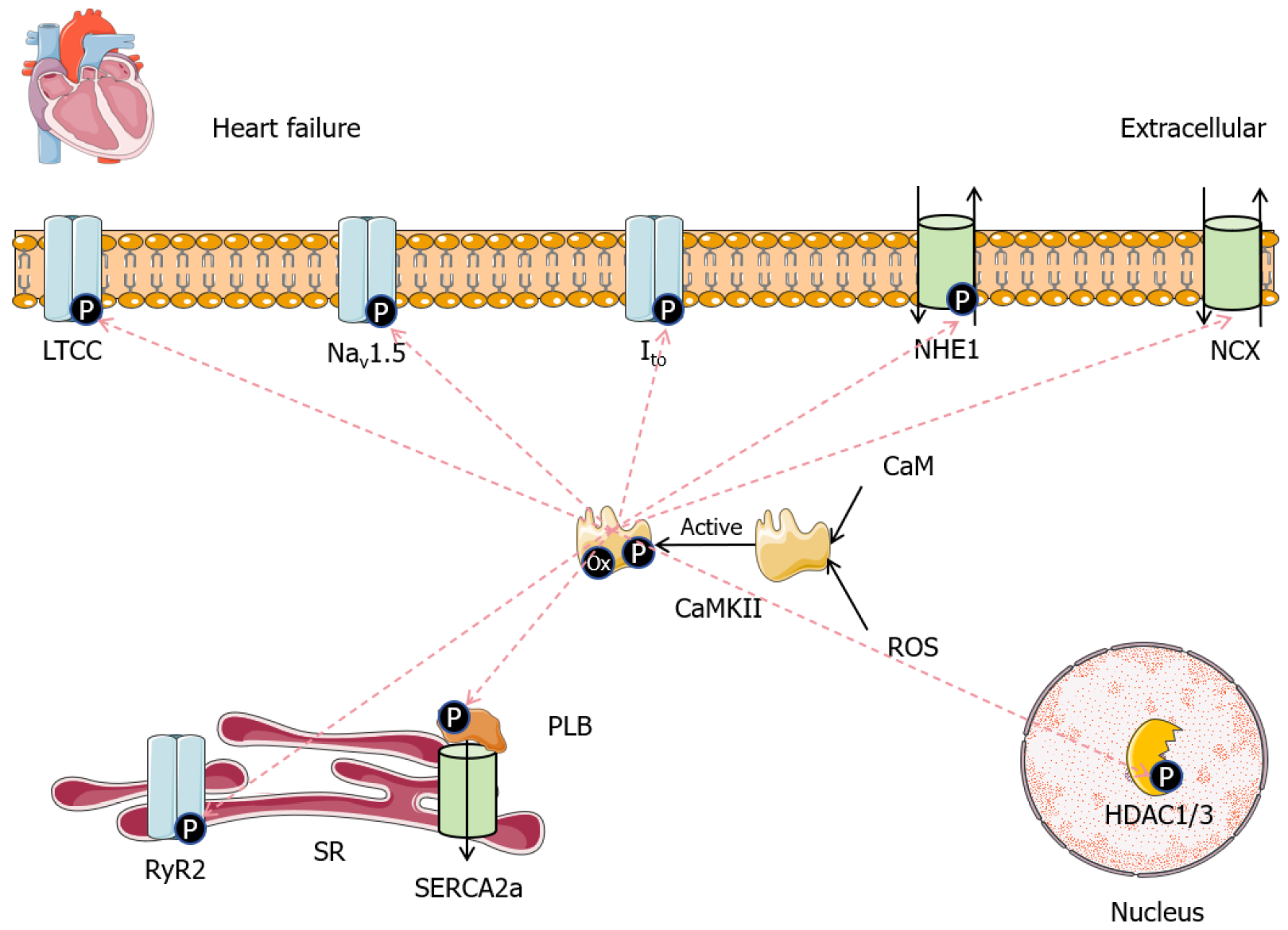
The cardiac isoform of CaMKII predominates, and is CaMKIIδ. Under physiological conditions, CaMKIIδ is activated in response to the binding of Ca2+/calmodulin to the kinase, which leads to autophosphorylation at Thr287, which changes CaMKIIδ into a persistently active form[12-14]. In disease states, however, CaMKII also can be switched on via calcium-independent pathways. It is interesting to note that reactive oxygen species (ROS) can oxidize Met281/282, leading to a constitutively active oxidized version of the enzyme, an important molecular link between oxidative stress and cardiac dysfunction[15].
Being a key mediator of HF progression, pathological hyperactivation of CaMKII is a molecular hub that synthesizes a range of upstream perturbations such as ionic dysplasia and oxidative stress and converts them into widespread maladaptive signaling in cardiomyocytes via the phosphorylation of many key downstream targets[16-18]. These CaMKII-induced effects are pro-arrhythmia susceptibility, myocardial fibrotic remodeling and cardiac function deterioration in HF[19,20]. The dysregulated CaMKII signaling has been attributed to the different pathological pathways including electrical instability, structural remodeling, inflammatory activation and programmed cell death.
CaMKII has extensive impacts on electrophysiology and calcium processing in the heart, and in pathological CaMKII activation a number of these systems are impaired. CaMKII-mediated phosphorylation of ryanodine receptor 2 (RyR2) enhances sarcoplasmic reticulum (SR) calcium leak[21-23], whereas CaMKII-mediated phosphorylation of the L-type calcium channel enhances calcium influx and action potential duration[24]. CaMKII also regulates phospholamban that disrupts the activity of sarcoplasmic/endoplasmic reticulum Ca2+-ATPase 2a and reduces the uptake of SR calcium[25]. These disruptions are also aggravated by CaMKII-mediated up-regulation of NCX[26]. Simultaneously, CaMKII provokes a radical shift in sodium regulation. Its phosphorylation of voltage-gated sodium channel type V 1.5 enhances the late sodium current, which causes intracellular sodium and secondary calcium overload[27,28]. Moreover, CaMKII-modula
| Specific target | CaMKII action | Pathophysiological consequence |
| RyR2 | Phosphorylates RyR2 at Ser2814, increasing its open probability | Increased sarcoplasmic reticulum calcium leak, elevated diastolic calcium spark frequency, leading to abnormal calcium transients and triggered arrhythmias |
| PLB | Phosphorylates PLB at Thr17, relieving its inhibition on SERCA2a | Initially compensatory enhanced calcium reuptake; chronic/over-activation synergizes with SERCA2a downregulation, resulting in reduced SR calcium load and diastolic dysfunction |
| LTCC | Phosphorylates LTCC at Thr1604, prolonging channel opening and slowing inactivation | Increased calcium influx, prolonged action potential duration, establishing the substrate for early afterdepolarizations; exacerbates calcium overload |
| Nav1.5 | Phosphorylates Nav1.5 at Ser516/Thr594, enhancing the late sodium current | Induces intracellular sodium overload, indirectly worsens calcium overload and action potential prolongation, significantly increasing arrhythmia risk |
| NCX | Indirectly promotes NCX protein expression via upstream signals (e.g., oxidative stress) | Increases reverse-mode NCX activity, leading to calcium influx and overload, impairing electrophysiological stability |
| Ito | Reduces Ito current density and function through chronic downregulation and acute phosphorylation-dependent gating modulation | Impairs early repolarization, prolongs action potential duration, and compromises repolarization reserve |
| HDAC | Phosphorylates and activates class I HDACs (e.g., HDAC1, HDAC3), driving pathological gene reprogramming | Promotes myocardial hypertrophy, fibrosis, and pathological remodeling |
Beyond its electrophysiological effects, CaMKII is the driver of a plethora of maladaptive structural, transcriptional and inflammatory responses. By phosphorylating class I histone deacetylases, CaMKII causes pathological transcriptional reprogramming leading to cardiomyocyte hypertrophy and fibrotic remodeling and progressive ventricular structural deterioration[32]. Moreover, oxidized CaMKII is a focal mediator of inflammation and cell injury. It triggers the nuclear factor kappa B pathway and upregulates the NACHT, leucine-rich repeat, and pyrin domain-containing protein 3 (NLRP3) inflammasome, which enhances the pro-inflammatory signaling in the myocardium[33,34]. Programmed cardiomyocyte death is also caused by excessive CaMKII activity. CaMKII overactivation contributes to cellular damage and increased functional impairment of the failing heart through interference with mitochondrial calcium homeostasis, stimulation of mitochondrial permeability transition pore opening, and increased production of ROS[35-37].
In summary, pathological activation of CaMKII orchestrates a range of maladaptive processes, including ionic balance disturbance, mitochondrial dysfunction, programmed cell death, fibrotic remodeling, and arrhythmia susceptibility, which collectively impair electrical stability and lead to decreased overall cardiac performance[18,38].
Accumulating evidence has suggested that SGLT2i can directly inhibit the pathological activation and phosphorylation of CaMKII and its key downstream targets. For example, dapagliflozin has been found to decrease CaMKII Thr287 auto
Importantly, SGLT2i also indirectly suppress CaMKII activity by rectifying the ionic disturbances upstream. Mustroph et al[41] has shown that empagliflozin causes a rapid decrease of intracellular sodium concentration, resulting in long-lasting suppression of CaMKII activity and RyR2 phosphorylation, ultimately leading to improved calcium handling and contractile function. This effect is mediated, to a large extent, by inhibition of overactive sodium-hydrogen exchanger 1[42], by reducing sodium overload at its source. The resulting reduction of reverse-mode NCX activity reduces calcium influx, opposing the resulting accumulation of calcium inside the cell that is responsible for the activation of CaMKII. Notably, CaMKII itself can cause ionic imbalance by phosphorylating voltage-gated sodium channel type V 1.5, increasing the late sodium current, and further accumulating sodium and calcium. Empagliflozin acts on both CaMKII and the late sodium current[43], and acts in a synergistic manner to disrupt this self-perpetuating cycle and stabilize cardiac electrophysiology.
In addition to stabilizing the ionic homeostasis, SGLT2i restrict the excessive activation of CaMKII by modulating the oxidative stress and myocardial metabolism. Empagliflozin, for example, inhibits production of mitochondrial ROS after exposure to doxorubicin, thereby preventing the generation of oxidatively activated CaMKII and the reversal of associated abnormalities of calcium handling and contractile function[30]. Moreover, by improving myocardial metabolic efficiency, empagliflozin reduces ROS production at the source, further blocking CaMKII oxidative activation. Mechanistically, this involves the activation of pathways such as AMP-activated protein kinase, which enhances metabolic flexibility, mitochondrial biogenesis and function and optimizes cellular energy and redox balance, collectively lessening oxidative modification of CaMKII at Met281/282[8,44].
SGLT2i also inhibit CaMKII-induced inflammatory signaling and cell death. Specifically, in HF, pathological CaMKII activation promotes the activation of the NLRP3 inflammasome, thereby triggering cardiomyocyte pyroptosis, exacerbating myocardial inflammation and fibrosis[45,46]. Targeting this inflammatory cascade, empagliflozin has been shown to suppress these pro-inflammatory pathways, reduce NLRP3 inflammasome activation, and improve cardiac function in HF models in a Ca2+-dependent manner[47]. Thus, through re-establishing ionic balance and directly inhibiting CaMKII, SGLT2i achieve a synergistic effect on the upstream triggers of myocardial inflammation and cell death, which improves the inflammatory milieu and limits adverse remodeling. This mechanism provides an important explanation for the broad spectrum of cardioprotective effects observed with SGLT2i therapy[48].
In summary, based on current evidence, it is suggested that SGLT2i achieve their widespread cardioprotective effects through the simultaneous modulation of ionic homeostasis, energy metabolism, redox balance and inflammatory sig
In conclusion, a CaMKII-centered framework offers a coherent explanation for the various cardioprotective effects of SGLT2i in HF. Confirming this model and determining its causal relevance in patients will require rigorous and well-designed clinical investigations.
| 1. | Parsi S, Shirsat PD, Mahali LP, Surani S, Kashyap R. Sodium-glucose cotransporter 2 inhibitor in heart failure patients and their outcomes: A meta-analysis of randomized controlled trials. World J Cardiol. 2025;17:109731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (2)] |
| 2. | Chen S, Wang Q, Bakker D, Hu X, Zhang L, van der Made I, Tebbens AM, Kovácsházi C, Giricz Z, Brenner GB, Ferdinandy P, Schaart G, Gemmink A, Hesselink MKC, Rivaud MR, Pieper MP, Hollmann MW, Weber NC, Balligand JL, Creemers EE, Coronel R, Zuurbier CJ. Empagliflozin prevents heart failure through inhibition of the NHE1-NO pathway, independent of SGLT2. Basic Res Cardiol. 2024;119:751-772. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 37] [Reference Citation Analysis (1)] |
| 3. | Arow M, Waldman M, Yadin D, Nudelman V, Shainberg A, Abraham NG, Freimark D, Kornowski R, Aravot D, Hochhauser E, Arad M. Sodium-glucose cotransporter 2 inhibitor Dapagliflozin attenuates diabetic cardiomyopathy. Cardiovasc Diabetol. 2020;19:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 172] [Article Influence: 28.7] [Reference Citation Analysis (2)] |
| 4. | Wijnker PJM, Dinani R, van der Laan NC, Algül S, Knollmann BC, Verkerk AO, Remme CA, Zuurbier CJ, Kuster DWD, van der Velden J. Hypertrophic cardiomyopathy dysfunction mimicked in human engineered heart tissue and improved by sodium-glucose cotransporter 2 inhibitors. Cardiovasc Res. 2024;120:301-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 5. | Maejima Y. SGLT2 Inhibitors Play a Salutary Role in Heart Failure via Modulation of the Mitochondrial Function. Front Cardiovasc Med. 2019;6:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 102] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 6. | Ekanayake P, Mudaliar S. A novel hypothesis linking low-grade ketonaemia to cardio-renal benefits with sodium-glucose cotransporter-2 inhibitors. Diabetes Obes Metab. 2022;24:3-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 7. | Li X, Flynn ER, do Carmo JM, Wang Z, da Silva AA, Mouton AJ, Omoto ACM, Hall ME, Hall JE. Direct Cardiac Actions of Sodium-Glucose Cotransporter 2 Inhibition Improve Mitochondrial Function and Attenuate Oxidative Stress in Pressure Overload-Induced Heart Failure. Front Cardiovasc Med. 2022;9:859253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 8. | Packer M. SGLT2 inhibitors: role in protective reprogramming of cardiac nutrient transport and metabolism. Nat Rev Cardiol. 2023;20:443-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 123] [Article Influence: 41.0] [Reference Citation Analysis (1)] |
| 9. | Khan MS, Shahid I, Bennis A, Rakisheva A, Metra M, Butler J. Global epidemiology of heart failure. Nat Rev Cardiol. 2024;21:717-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 303] [Article Influence: 151.5] [Reference Citation Analysis (1)] |
| 10. | Fang Y, Chen L, Sun S, Ran X. Sodium-Glucose Transporter 2 Inhibitors in Heart Failure: An Overview of Systematic Reviews. J Cardiovasc Dev Dis. 2024;11:198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 11. | Xie Y, Wei Y, Li D, Pu J, Ding H, Zhang X. Mechanisms of SGLT2 Inhibitors in Heart Failure and Their Clinical Value. J Cardiovasc Pharmacol. 2023;81:4-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 41] [Reference Citation Analysis (1)] |
| 12. | Maier LS, Zhang T, Chen L, DeSantiago J, Brown JH, Bers DM. Transgenic CaMKIIdeltaC overexpression uniquely alters cardiac myocyte Ca2+ handling: reduced SR Ca2+ load and activated SR Ca2+ release. Circ Res. 2003;92:904-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 365] [Article Influence: 15.9] [Reference Citation Analysis (1)] |
| 13. | Nguyen TA, Puhl HL 3rd, Hines K, Liput DJ, Vogel SS. Binary-FRET reveals transient excited-state structure associated with activity-dependent CaMKII - NR2B binding and adaptation. Nat Commun. 2022;13:6335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 14. | Meyer T, Hanson PI, Stryer L, Schulman H. Calmodulin trapping by calcium-calmodulin-dependent protein kinase. Science. 1992;256:1199-1202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 472] [Article Influence: 13.9] [Reference Citation Analysis (4)] |
| 15. | Zhang W, Dong E, Zhang J, Zhang Y. CaMKII, 'jack of all trades' in inflammation during cardiac ischemia/reperfusion injury. J Mol Cell Cardiol. 2023;184:48-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 16. | Reyes Gaido OE, Nkashama LJ, Schole KL, Wang Q, Umapathi P, Mesubi OO, Konstantinidis K, Luczak ED, Anderson ME. CaMKII as a Therapeutic Target in Cardiovascular Disease. Annu Rev Pharmacol Toxicol. 2023;63:249-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 101] [Article Influence: 33.7] [Reference Citation Analysis (1)] |
| 17. | Mustroph J, Baier MJ, Pabel S, Stehle T, Trum M, Provaznik Z, Mohler PJ, Musa H, Hund TJ, Sossalla S, Maier LS, Wagner S. Empagliflozin Inhibits Cardiac Late Sodium Current by Ca/Calmodulin-Dependent Kinase II. Circulation. 2022;146:1259-1261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 18. | Ma K, Ma G, Guo Z, Liu G, Liang W. Regulatory mechanism of calcium/calmodulin-dependent protein kinase II in the occurrence and development of ventricular arrhythmia (Review). Exp Ther Med. 2021;21:656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 19. | Lebek S, Caravia XM, Chemello F, Tan W, McAnally JR, Chen K, Xu L, Liu N, Bassel-Duby R, Olson EN. Elimination of CaMKIIδ Autophosphorylation by CRISPR-Cas9 Base Editing Improves Survival and Cardiac Function in Heart Failure in Mice. Circulation. 2023;148:1490-1504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 20. | Zhu C, Zhao M, Zhao L, Wu M, Xiang YK. Cardiac β2 adrenergic receptor deletion drives calmodulin kinase II upregulation to induce connective tissue growth factor in cardiac fibrosis and diastolic dysfunction. Function (Oxf). 2025;6:zqaf036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 21. | Camors E, Valdivia HH. CaMKII regulation of cardiac ryanodine receptors and inositol triphosphate receptors. Front Pharmacol. 2014;5:101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 22. | Sepúlveda M, Gonano LA, Back TG, Chen SR, Vila Petroff M. Role of CaMKII and ROS in rapid pacing-induced apoptosis. J Mol Cell Cardiol. 2013;63:135-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 23. | Pereira L, Bare DJ, Galice S, Shannon TR, Bers DM. β-Adrenergic induced SR Ca(2+) leak is mediated by an Epac-NOS pathway. J Mol Cell Cardiol. 2017;108:8-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 24. | Li J, Wang S, Zhang J, Liu Y, Zheng X, Ding F, Sun X, Zhao M, Hao L. The CaMKII phosphorylation site Thr1604 in the Ca(V)1.2 channel is involved in pathological myocardial hypertrophy in rats. Channels (Austin). 2020;14:151-162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 25. | Liu Q, Sun J, Zhang L, Xu Y, Wu B, Cao J. The Agonist of Inward Rectifier Potassium Channel (I(K1)) Attenuates Rat Reperfusion Arrhythmias Linked to CaMKII Signaling. Int Heart J. 2021;62:1348-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 26. | Yang Y, He J, Yuan M, Tse G, Zhang K, Ma Z, Li J, Zhang Y, Gao Y, Zhang Y, Wang R, Li G, Liu T. Xanthine oxidase inhibitor allopurinol improves atrial electrical remodeling in diabetic rats by inhibiting CaMKII/NCX signaling. Life Sci. 2020;259:118290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 27. | Ashpole NM, Herren AW, Ginsburg KS, Brogan JD, Johnson DE, Cummins TR, Bers DM, Hudmon A. Ca2+/calmodulin-dependent protein kinase II (CaMKII) regulates cardiac sodium channel NaV1.5 gating by multiple phosphorylation sites. J Biol Chem. 2012;287:19856-19869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 144] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 28. | Wagner S, Dybkova N, Rasenack EC, Jacobshagen C, Fabritz L, Kirchhof P, Maier SK, Zhang T, Hasenfuss G, Brown JH, Bers DM, Maier LS. Ca2+/calmodulin-dependent protein kinase II regulates cardiac Na+ channels. J Clin Invest. 2006;116:3127-3138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 423] [Article Influence: 21.2] [Reference Citation Analysis (1)] |
| 29. | Colinas O, Gallego M, Setién R, López-López JR, Pérez-García MT, Casis O. Differential modulation of Kv4.2 and Kv4.3 channels by calmodulin-dependent protein kinase II in rat cardiac myocytes. Am J Physiol Heart Circ Physiol. 2006;291:H1978-H1987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 48] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 30. | Wagner S, Hacker E, Grandi E, Weber SL, Dybkova N, Sossalla S, Sowa T, Fabritz L, Kirchhof P, Bers DM, Maier LS. Ca/calmodulin kinase II differentially modulates potassium currents. Circ Arrhythm Electrophysiol. 2009;2:285-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 111] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 31. | Hegyi B, Bers DM, Bossuyt J. CaMKII signaling in heart diseases: Emerging role in diabetic cardiomyopathy. J Mol Cell Cardiol. 2019;127:246-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 116] [Article Influence: 16.6] [Reference Citation Analysis (1)] |
| 32. | Zhang M, Yang X, Zimmerman RJ, Wang Q, Ross MA, Granger JM, Luczak ED, Bedja D, Jiang H, Feng N. CaMKII exacerbates heart failure progression by activating class I HDACs. J Mol Cell Cardiol. 2020;149:73-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 33. | Wei Z, Fei Y, Wang Q, Hou J, Cai X, Yang Y, Chen T, Xu Q, Wang Y, Li YG. Loss of Camk2n1 aggravates cardiac remodeling and malignant ventricular arrhythmia after myocardial infarction in mice via NLRP3 inflammasome activation. Free Radic Biol Med. 2021;167:243-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 34. | Yao Y, Li F, Zhang M, Jin L, Xie P, Liu D, Zhang J, Hu X, Lv F, Shang H, Zheng W, Sun X, Duanmu J, Wu F, Lan F, Xiao RP, Zhang Y. Targeting CaMKII-δ9 Ameliorates Cardiac Ischemia/Reperfusion Injury by Inhibiting Myocardial Inflammation. Circ Res. 2022;130:887-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 79] [Article Influence: 19.8] [Reference Citation Analysis (1)] |
| 35. | Zhang T, Zhang Y, Cui M, Jin L, Wang Y, Lv F, Liu Y, Zheng W, Shang H, Zhang J, Zhang M, Wu H, Guo J, Zhang X, Hu X, Cao CM, Xiao RP. CaMKII is a RIP3 substrate mediating ischemia- and oxidative stress-induced myocardial necroptosis. Nat Med. 2016;22:175-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 670] [Article Influence: 67.0] [Reference Citation Analysis (3)] |
| 36. | Joiner ML, Koval OM, Li J, He BJ, Allamargot C, Gao Z, Luczak ED, Hall DD, Fink BD, Chen B, Yang J, Moore SA, Scholz TD, Strack S, Mohler PJ, Sivitz WI, Song LS, Anderson ME. CaMKII determines mitochondrial stress responses in heart. Nature. 2012;491:269-273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 355] [Article Influence: 25.4] [Reference Citation Analysis (1)] |
| 37. | Lin R, Peng X, Li Y, Wang X, Liu X, Jia X, Zhang C, Liu N, Dong J. Empagliflozin attenuates doxorubicin-impaired cardiac contractility by suppressing reactive oxygen species in isolated myocytes. Mol Cell Biochem. 2024;479:2105-2118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 38. | Zhao N, Li Q, Sui H, Zhang H. Role of Oxidation-Dependent CaMKII Activation in the Genesis of Abnormal Action Potentials in Atrial Cardiomyocytes: A Simulation Study. Biomed Res Int. 2020;2020:1597012. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 39. | Liu T, Wu J, Shi S, Cui B, Xiong F, Yang S, Yan M. Dapagliflozin attenuates cardiac remodeling and dysfunction in rats with β-adrenergic receptor overactivation through restoring calcium handling and suppressing cardiomyocyte apoptosis. Diab Vasc Dis Res. 2023;20:14791641231197106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 40. | Kadosaka T, Watanabe M, Natsui H, Koizumi T, Nakao M, Koya T, Hagiwara H, Kamada R, Temma T, Karube F, Fujiyama F, Anzai T. Empagliflozin attenuates arrhythmogenesis in diabetic cardiomyopathy by normalizing intracellular Ca(2+) handling in ventricular cardiomyocytes. Am J Physiol Heart Circ Physiol. 2023;324:H341-H354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 41. | Mustroph J, Wagemann O, Lücht CM, Trum M, Hammer KP, Sag CM, Lebek S, Tarnowski D, Reinders J, Perbellini F, Terracciano C, Schmid C, Schopka S, Hilker M, Zausig Y, Pabel S, Sossalla ST, Schweda F, Maier LS, Wagner S. Empagliflozin reduces Ca/calmodulin-dependent kinase II activity in isolated ventricular cardiomyocytes. ESC Heart Fail. 2018;5:642-648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 184] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 42. | Cappetta D, De Angelis A, Bellocchio G, Telesca M, Cianflone E, Torella D, Rossi F, Urbanek K, Berrino L. Sodium-Glucose Cotransporter 2 Inhibitors and Heart Failure: A Bedside-to-Bench Journey. Front Cardiovasc Med. 2021;8:810791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 43. | Trum M, Riechel J, Schollmeier E, Lebek S, Hegner P, Reuthner K, Heers S, Keller K, Wester M, Klatt S, Hamdani N, Provaznik Z, Schmid C, Maier L, Arzt M, Wagner S. Empagliflozin inhibits increased Na influx in atrial cardiomyocytes of patients with HFpEF. Cardiovasc Res. 2024;120:999-1010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 44. | Xie B, Ramirez W, Mills AM, Huckestein BR, Anderson M, Pangburn MM, Lang EY, Mullet SJ, Chuan BW, Guo L, Sipula I, O'Donnell CP, Wendell SG, Scott I, Jurczak MJ. Empagliflozin restores cardiac metabolic flexibility in diet-induced obese C57BL6/J mice. Curr Res Physiol. 2022;5:232-239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 45. | Wang J, Deng B, Liu Q, Huang Y, Chen W, Li J, Zhou Z, Zhang L, Liang B, He J, Chen Z, Yan C, Yang Z, Xian S, Wang L. Pyroptosis and ferroptosis induced by mixed lineage kinase 3 (MLK3) signaling in cardiomyocytes are essential for myocardial fibrosis in response to pressure overload. Cell Death Dis. 2020;11:574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 174] [Article Influence: 29.0] [Reference Citation Analysis (4)] |
| 46. | Li C, Wang X, Yang R, Liu Y, Wang L, Zhang M, Dong J, Li L, Yu T, Lv L, Li T, Zhang Y, Li B, Liang H, Shan H, Li X. MBNL1 Modulates Nek7 to Facilitate Pathological Cardiac Hypertrophy via NLRP3. Hypertension. 2026;83:103-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 47. | Byrne NJ, Matsumura N, Maayah ZH, Ferdaoussi M, Takahara S, Darwesh AM, Levasseur JL, Jahng JWS, Vos D, Parajuli N, El-Kadi AOS, Braam B, Young ME, Verma S, Light PE, Sweeney G, Seubert JM, Dyck JRB. Empagliflozin Blunts Worsening Cardiac Dysfunction Associated With Reduced NLRP3 (Nucleotide-Binding Domain-Like Receptor Protein 3) Inflammasome Activation in Heart Failure. Circ Heart Fail. 2020;13:e006277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 201] [Article Influence: 33.5] [Reference Citation Analysis (2)] |
| 48. | Andreadou I, Bell RM, Bøtker HE, Zuurbier CJ. SGLT2 inhibitors reduce infarct size in reperfused ischemic heart and improve cardiac function during ischemic episodes in preclinical models. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 79] [Article Influence: 13.2] [Reference Citation Analysis (1)] |
| 49. | Trum M, Riechel J, Wagner S. Cardioprotection by SGLT2 Inhibitors-Does It All Come Down to Na(+)? Int J Mol Sci. 2021;22:7976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (1)] |