INTRODUCTION
Despite being the cornerstone of gastrointestinal surgery, postoperative complications, such as anastomotic leakage (AL), delayed healing, and infections, continue to be significant causes of patient morbidity and death. Among the many influencing factors, perioperative nutrition is one of the most important modifiable factors affecting anastomotic integrity and surgical outcomes. The risk of postoperative complications, such as AL, extended hospitalization, and death, is significantly increased by malnutrition[1]. In colorectal surgeries, hypoalbuminemia was identified as an independent risk factor for AL[2]. Delays in wound healing and higher infection rates have also been connected to nutritional status[3]. These results highlight how crucial and pivotal it is to perform preoperative nutritional evaluation.
In malnourished patients or in those with extensive gastrointestinal resections, preoperative nutritional supplementation has also been shown to be useful for anastomosis healing[4]. Gündoğdu et al[5] found that preoperative nutritional supplementation enhanced colonic anastomotic healing in malnourished rats. Clinical evidence also suggests that perioperative nutritional support can decrease complications following intestinal resection and anastomosis[6].
Early enteral feeding [early enteral nutrition (EEN)] introduced within the first 24 postoperative hours, is also more advantageous. Multiple studies have shown that EEN reduces the incidence of postoperative complications and hospital stay in lower gastrointestinal surgery[7] by shortening the time of flatus and stool passage[8,9]. Animal and clinical research also suggests the importance of nutritional interventions beyond mere caloric and protein replacement. Glutamine supplementation and hydrogen-rich saline improved anastomotic strength and reduced inflammation in pediatric rat models[10]. Castilho et al[11] demonstrated that probiotic supplementation altered the gut microbiota to support anastomosis healing.
Enteral immunonutrition has also been reported to enhance mucosal immunity and reduce infectious complications during the postoperative period[12]. Early immunonutrition in the postoperative period for patients with gastric cancer improved both clinical and immunologic outcomes in a randomized trial[13]. Recent evidence suggests that the gut microbiome is a significant determinant of anastomotic healing. Microbial flora disruption by antibiotics or malnutrition can jeopardize the mucosal lining, thereby affecting anastomotic healing[14]. This study attempts to synthesize current evidence regarding the effects of nutrition, including preoperative status, EEN, immunonutrition, and microbiome-targeted therapies, on the healing of intestinal anastomosis and the subsequent complications.
NARRATIVE REVIEW STRATEGY
Our search included recent articles from the following databases: PubMed, Google Scholar, Scopus, ScienceDirect, and Cochrane. The main study types included randomized controlled trials, cohort studies, case-control studies, and systematic reviews written in English. The inclusion criteria were as follows: Adult population (> 18 years of age) after all types of gastrointestinal anastomotic surgeries, individuals who underwent gastrointestinal surgery with a known perioperative nutritional status, and animal studies were included if they were found to have an impact on this subject.
The following objectives were used as primary outcomes for our study: AL, complications, recovery, length of hospital stay, and survival rates. There were several exclusion criteria, as follows: Studies that did not correlate perioperative nutritional status and its effect on gastrointestinal anastomosis; case reports; opinion studies; and studies with insufficient data on nutritional status and its impact on anastomosis.
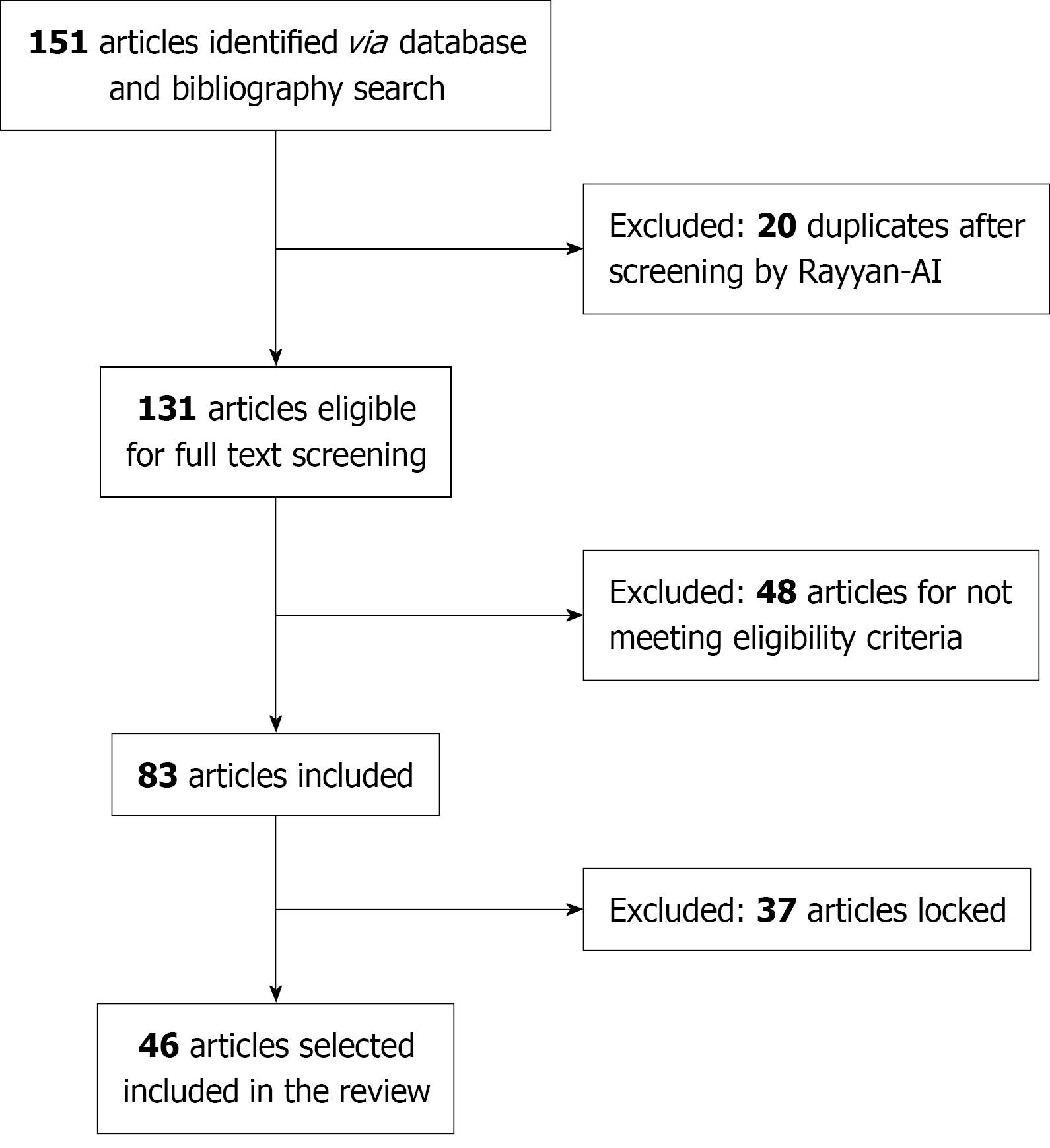
Our search yielded 151 articles that met our criteria across all the databases; these papers were input into the Rayyan artificial intelligence-powered systematic review management platform (https://www.rayyan.ai/) for title and abstract screening. After careful review, 20 studies were found to be duplicates, and the remaining 131 were independently reviewed by two authors. A third author was responsible for conflict resolution, and 83 papers were ultimately included. Among these 83 articles, 37 studies were locked behind paywalls and were not accessible through institutional subscriptions, and a decision to exclude them was made. In the final stage, 46 studies met the inclusion criteria and were readily accessible (Figure 1).
Figure 1 Flow chart.
AI: Artificial intelligence.
FINDINGS
Parameters used to assess nutritional status
Malnutrition is a significant predictor of poor surgical outcomes, especially because it impairs anastomotic safety and slows wound healing by affecting the immune system, collagen synthesis, and general tissue repair processes[7]. Research from surgical wards indicates that patients who arrive in the operating room undernourished have significantly greater risks of AL, surgical site infection, and a longer recovery time following surgery[15-17].
A broad spectrum of nutritional assessment tools has been investigated. Screening instruments such as the NRS-2002 are commonly used among surgical patients. Higher values (≥ 3) on malnutrition screening tools strongly correlate with increased rates of postoperative complications and anastomotic leaks following gastrointestinal surgery[7,16]. Similar findings emerged from the Malnutrition Universal Screening Tool and Subjective Global Assessment (SGA) tools, both of which reliably identified malnourished patients prior to surgery. The subset of patients with positive screening results presented significantly higher incidences of surgical site infections, as well as prolonged wound healing times[16].
Among the anthropometric measures, body mass index (BMI) yielded various outcomes. Kwag et al[18] affirmed that both low and excessive BMIs carry heightened risks in surgical settings. Conversely, gains in risk are evident when weight decreases involuntarily by more than 10% within 6 months; this change correlates robustly with complications, including anastomotic leak[19].
Biochemical markers, such as serum albumin (ALB), remain the most widely reported. A concentration less than 3.5 g/dL serves as a leading marker for anastomotic failure in colorectal resections, underscoring the predictive value identified by El Nakeeb et al[20]. Moreover, the prognostic nutritional index, which integrates ALB and lymphocyte counts, further refines risk stratification and continues to be validated across multiple studies. A low prognostic nutritional index (< 45) is a significant predictor of increased rates of leaks and infectious complications[13]. Other indicators, such as the C-reactive protein/ALB ratio, are similarly correlated with postoperative results in various surgical groups[3]. However, it is important to recognize that ALB is a negative acute phase reactant and reflects systemic inflammation, in addition to fluid status and nutritional reserves. Hypoalbuminemia, therefore, should be interpreted as an integrated indicator of physiologic stress and inflammatory burden.
In addition to laboratory values, preoperative computed tomography scans facilitate the assessment of the skeletal muscle index and psoas muscle area. Sarcopenia, characterized by a decreased skeletal muscle index, is regularly associated with increased leakage and infection rates following colorectal and esophageal anastomoses[2]. Frasson et al[21] also associated myosteatosis and visceral fat accumulation with compromised anastomotic healing; however, the results were less consistent. Functional measures, including handgrip strength and phase angle derived from bioelectrical impedance, have surfaced as new indicators. In surgical groups, lower grip strength was linked to hindered recovery and a greater risk of leakage[5].
Finally, the concept of immunonutrition (nutrition adapted to the immune response) has gained popularity, especially with the targeted administration of omega-3 lipids and enteral feeds enhanced with arginine. Although the impact on the incidence of AL shows differing results, Barlow et al[15] reported a decrease in surgical-site infections. In line with this, Sapidis et al[22] reported that co-supplementing glutamine with symbiotic agents enhanced the operator’s ability to perform colonic anastomoses. However, there is still a dearth of research on individual micronutrients, particularly zinc and vitamin D, and the results are not always consistent. Overall, the evidence to date indicates that the best predictive ability for identifying patients at risk for less-than-ideal intestinal anastomosis healing is a thorough, multimodal evaluation that incorporates biochemical markers, anthropometric monitoring, and a thorough body composition analysis.
Anastomosis site
Following a literature review, we identified several gastrointestinal anastomosis sites depending on the underlying pathology and surgical approach, including inflammatory conditions, such as Crohn’s disease and ulcerative colitis, and if surgical intervention was required, including ileal pouch-anal anastomosis (IPAA)[23], total proctocolectomy with IPAA[24], and ileocecal resection[19]. Other surgical approaches included resections of the transverse colon, left hemicolectomy, rectosigmoid resection, low anterior resection, abdominoperineal resection[19], and colon-colonic anastomoses[25] on the basis of disease location and severity, with the goal of preserving bowel length and function whenever possible.
In contrast, surgical treatment for gastrointestinal malignancies varies depending on the tumor location. For cancers in the upper gastrointestinal tract, procedures include Ivor-Lewis esophagectomy (with esophagogastric anastomosis)[12,15,16], total gastrectomy (with esophagojejunostomy and jejunojejunostomy)[13], distal gastrectomy (with gastro-jejunal anastomosis)[1,26], and bilio-enteric anastomosis[27]. On the other hand, lower gastrointestinal malignancies require more extensive resections, such as right colectomy[20,21,28] with ileocolic anastomosis[29,30], left colectomy with colon-colonic anastomosis[29,31], low anterior resection[32,33], or sigmoidectomy[29]. Our pooled data included over 42000 patients with upper gastrointestinal cancers and more than 72000 individuals with lower gastrointestinal malignancies.
Complication types and rates
Various complications, such as anastomotic leaks, infections, and nutritional imbalances, are associated with gastrointestinal surgeries and can strongly affect patient outcome and satisfaction[34]. The rates of AL vary depending on the type of surgery. For example, 2%-20% of colorectal procedures (higher for low rectal anastomoses) and 9.6%-14% of esophageal surgeries resulted in AL, with severe cases requiring reoperation in 76.9% of instances[14,28,30,35-37]. Malnourished patients, particularly those with hypoalbuminemia (< 3.0 g/dL), are more likely to experience infectious complications after surgery, especially surgical site infections (4.5%-28.1%) and pneumonia (3.3%-21.1%)[2,15,18]. Malnutrition itself makes things worse. Additionally, malnutrition itself increases risk; 50% of defunctionalized-stage patients have protein-caloric deficits, and 60% of IPAA patients have severe electrolyte imbalances (such as zinc deficiency), which makes recovery even harder[16,23]. Systemic complications, such as wound dehiscence (11%) and postoperative ileus (35%-45%), are also common. Although several studies have suggested EEN as safe and beneficial for lower rate of complications and shorter hospital stay[12,15,38,39], the data regarding its effect on AL remain inconsistent. The meta-analyses have demonstrated no statistically significant reduction in leak rates, and due to limited information size and heterogeneity the available evidence is considered weak. Moreover, most trials have included heterogeneous surgical populations, and few have been adequately powered with AL as a primary endpoint.
Men, smokers, people with low anastomotic heights, and people who have undergone emergency surgery are at greater risk for these problems, with mortality due to AL reaching 27% in severe cases[14,28,30]. Improving nutrition, surgical techniques (such as side-to-side anastomoses), and microbiome modulation may help reduce these risks, but more research is needed because of the heterogeneity of AL definitions and study designs[28,40,41].
Correlation between nutritional status and complications
The articles included in our narrative review explored how a patient’s nutritional status may play a role in a multitude of complications that can arise postoperatively, with a particular focus on patients undergoing surgical procedures with anastomosis of any type. Importantly, the studies are heterogeneous in the qualifications and methods used to assess a patient’s nutritional status and the types of complications quantified. Thus, reviews exploring this correlation across the literature are reviewed below and subcategorized in accordance with overlapping themes of nutritional status assessment tools or interventions.
Patient nutritional status scoring: Across this review, different studies adopted distinct methods to assess and quantify patients’ nutritional status, including scoring surveys and systems such as the Mini Nutritional Assessment score, arm and calf circumference, NRS-2002 score, PG-SGA score, and ALB levels. Xu and Kong[42] collected data from 382 patients with rectal cancer who underwent surgery and analyzed several nutritional indicators to correlate them with postoperative complications. Single-factor analysis revealed that patients with high NRS-2002 scores, a tool used to identify the risk of malnutrition in patients (P < 0.001), high PG-SGA scores, a tool used to identify malnutrition risk in cancer patients specifically (P < 0.001), and low postoperative ALB levels (mean, 29.907 ± 5.478 g/L, P = 0.046) were more likely to have AL. Furthermore, multivariate analysis revealed that low postoperative ALB levels (P = 0.044) and high PG-SGA scores (P < 0.001) were “independent risk factors for AL after rectal cancer surgery”[42]. This demonstrates that the risk of leakage complications among patients with poor nutritional status remains high, and the replenishment of perioperative nutrition and ALB is important. Similarly, Garth et al[16] carried out a prospective study to assess whether certain perioperative nutritional practices and protocols improved outcomes in patients undergoing upper gastrointestinal and colorectal cancer surgery. Nutritional status was also assessed by SGA. The investigators reported that malnourished patients of all subclasses within the SGA tool were more than twice as likely (41.7%) to develop one or more complications (of which AL was included) than preoperatively well-nourished patients (15.4%). However, this difference did not reach statistical significance (P = 0.20), which may be attributed to the small sample size used for SGA analysis. Another prospective study by Kwag et al[18] assessed a similar outcome, using the NRS-2002 score instead of the screening tool. They reported that malnutrition was a statistically significant independent risk factor for anastomosis leakage (P = 0.027) and wound infection (P = 0.01). In a literature review of preoperative, intraoperative, and postoperative factors that increase the risk of AL, Sripathi et al[43] reported that multiple studies suggest that malnutrition is highly correlated with AL, as are low preoperative ALB levels, ALB ≤ 32 g/L; or ALB < 3.5 mg/dL, odds ratio of 2.8 (P = 0.03)]. Additionally, a retrospective analysis in a single center conducted by Galata et al[19] employed multivariate analysis and found that that low levels of preoperative ALB at a cutoff of 32.5 g/L are independent risk factors for major postoperative complications in patients surgically treated for Crohn’s disease (P = 0.0005) and that this was the most statistically significant factor.
EEN
A study by Chatterjee et al[27] explored the potential benefits of EEN following enteric anastomosis compared with conventional feeding (within 24 hours of anastomosis vs after 48-72 hours or the return of peristaltic sounds) and reported that there was no statistically significant difference in clinical leakage rate (P = 0.206), rate of re-exploration (P = 0.36), or mortality due to AL (P = 0.61) between the two groups. The authors also claimed that their results were in accordance with previously published literature that demonstrated enhanced anastomotic healing and strength with early feeding without any increase in AL or dehiscence. Similarly, Ilyas and Raza[44] investigated the same early feeding intervention and reported 0% leakage in both groups, as well as reduced surgical stress and wound infection. In a systematic review, Herbert et al[7] screened 17 randomized controlled trials that studied the effects of early feeding following elective lower gastrointestinal surgery, 13 of which reported AL and dehiscence. In the early feeding group, the range of dehiscence among patients was 0%-8.3%, whereas it was 0%-13.3% in the control group. Furthermore, their statistical analysis of the combined studies revealed a combined relative risk of 0.78 (95% confidence interval: 0.38-1.61, P = 0.51) and an absolute risk reduction of 0.01. However, it is important to note that the Trial Sequential Analyses showed that the “information size was not large enough to rule out that early feeding vs control reduces the risk ratio of AL/dehiscence events by 30% or more. AL/dehiscence was also judged as having low-quality evidence.”[7]. Similarly, El Nakeeb et al[20] and Wang et al[33] did not identify a statistically significant difference in AL or postoperative complications between early-feeding and control groups; however, the former study revealed that the incidence of AL in the early-feeding group was lower than that in the traditional feeding group.
Probiotic supplements and the microbiome
The microbiota plays an important role in anastomosis wound healing and recovery, where pathogenic bacteria bred by gut dysbiosis caused by surgery can interrupt the wound healing cascade. This process degrades the injured tissue, primarily through the release of collagenases and host metalloproteinases[43], leading to dehiscence and subsequent leakage. Nonetheless, the literature still lacks tangible significant evidence on the influence of the microbiota on postoperative anastomosis complications, particularly outside of animal models. Castilho et al[11] studied the microbiota metagenomics in rats supplemented with probiotics following intestinal anastomosis surgery, specifically by measuring the alpha diversity index (mathematical measure of species variation in a community) and the beta diversity index (“variation in composition between different communities of bacterial species” reflecting relative abundance in bacterial genera in postop vs preop groups), as well as anastomosis success by tensiometry. They verified that probiotic use was associated with increased anastomosis tensile strength, where the maximum traction forces of the control and probiotic groups were 1.1 ± 0.2 N and 1.4 ± 0.3 N, respectively (P = 0.0116, 95% confidence interval: 95.27%). For the diversity indices, there was no significant change for the alpha index; however, the beta index significantly differed between the two groups, where the probiotic intervention group preserved and increased the amount of beneficial bacteria and decreased the abundance of pathogenic bacteria, creating a favorable environment for anastomosis healing. It is important to note, though, that the evidence supporting probiotic supplementation remains limited. Most available data have been derived from small randomized trials or animal models, and substantial heterogeneity exists regarding probiotic strains, dosing regimens, timing of administration, and surgical subtypes. Therefore, while mechanistic data are promising, the current clinical evidence is insufficient to support routine probiotic use specifically for the prevention of AL.
DISCUSSION
Malnutrition adversely affects various physiologic processes, notably anastomotic healing, in the context of gastrointestinal surgery[7,15]. To assess the nutritional status of patients, several parameters have been utilized and reliably indicate relatively high complication rates in high-scoring individuals[7,15,16]. Among these factors, BMI is correlated with increased risk in extremis individuals[18,19]. A more widely employed tool would be ALB levels, which is an improved risk assessment when combined with other tools, such as the lymphocytic count or C-reactive protein[3,13,20]. Other evaluation modalities include imaging to assess muscle mass, functional (bioelectrical) measures to evaluate handgrip strength, and immunonutrition, which is still in its early phases[2,5,15,21,22,45]. All the modalities combined would yield the best approach for assessing nutritional status instead of focusing on only one tool.
For a better understanding of how malnutrition affects an anastomosis, we examined the different locations along the gastrointestinal tract where one was created. The location of the anastomosis primarily depended on the underlying pathology. The different anastomoses included in this paper are listed below: IPAA[23,24], colon-colonic[25,29,31], esophagogastric[12,15,16], esophagojejunostomy[13], jejunojejunostomy[13], gastro-jejunal[1,26], bilio-enteric[27], and ileocolic[29,30]. Among these anastomoses, colorectal, mainly low rectal, and esophageal operations had the highest rates of leakage[14,28,30,35-37]. Malnourishment leads to an increase in complications, which are most commonly infectious in nature, in individuals whose ALB levels are decreased[2,18,19]. Moreover, electrolyte disturbances, macronutrient deficits, abnormal wound healing, and ileus are common findings among this population[16,23]. These problems were found to be mitigated by starting EEN[12,15,38]. Additionally, several risk factors have been shown to increase the probability of complications and can be classified as modifiable, such as smoking and nutritional status, and nonmodifiable, such as male sex and surgery in an emergent setting[14,28,30].
Multiple authors have adopted a collection of scoring systems to stratify the risk of complications related to malnutrition. Some of these assessments include the Mini Nutritional Assessment score, NRS-2002, and PG-SGA[42]. Although some papers did not identify a statistically significant correlation between malnutrition and perioperative complications[16], the majority of the findings supported a statistically significant association, which was most evident in patients with inflammatory diseases[18,19,43]. While some papers have established that starting enteral feeding early can reduce perioperative complications[12,15,38], others have not identified a statistical correlation with leak rates, re-exploration rate, or mortality due to leakage if enteral nutrition is started at 48-72 hours postoperative or if enteral peristalsis is returned while improving anastomotic healing and strength in both groups[27]. Other studies reported no significant difference in the likelihood of starting early feeding compared with traditional food introduction; nonetheless, owing to the low information size, the authors could not rule out a reduction in the AL risk ratio and reported that AL incidence was lower when feeding was started earlier[7,20,33,44]. Surgery induces dysregulation of the enteral microbiome, leading to a disruption in wound healing via the disruption of collagen, which is essential in the injured tissue repair process and, in turn, facilitates AL[43]. One study concluded that mechanical loading on anastomotic fibroblasts led to an increase in types I and III collagen expression, enhancing anastomotic healing[46]. A new area of study targets probiotics to offset that dysregulation; although the data are still scarce, they reported an increase in tensile strength as well as beneficial bacteria in the group with probiotic supplementation[11]. Importantly, gastrointestinal anastomoses do not represent a homogeneous entity. Leak rates, healing biology, and perioperative management differ significantly between colorectal, esophageal, gastric, and small bowel surgeries. Moreover, elective oncologic resections differ substantially from emergency procedures in the setting of inflammation or sepsis. These variations limit the generalizability of pooled conclusions and necessitate caution in interpretation when extrapolating findings across surgical subtypes.