Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.118003
Revised: January 16, 2026
Accepted: February 24, 2026
Published online: April 27, 2026
Processing time: 124 Days and 21.1 Hours
Anastomotic leakage (AL) is a serious complication following McKeown eso
To evaluate the association between preoperative CTA-measured RGEA length and AL after MKE.
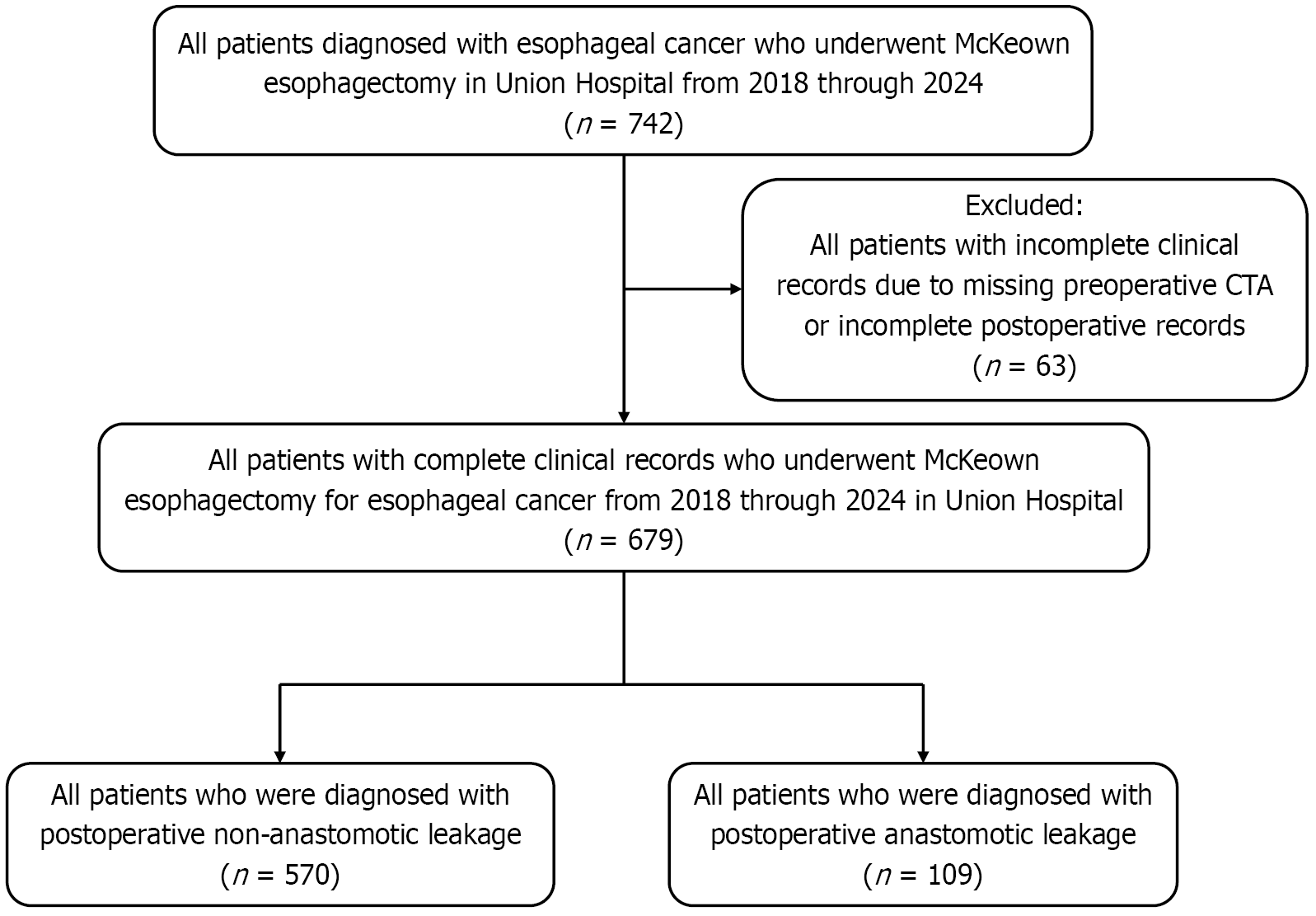
A total of 679 patients who underwent MKE for esophageal cancer were included and stratified into two groups based on the presence or absence of AL (AL and Non-AL). The primary endpoint was to assess the association between AL incidence and the length of the RGEA as measured by CTA. Statistical analyses were performed using EmpowerStats and R software version 4.5.1.
Among the 679 patients, 109 (16.10%) developed AL. Patients in the AL group had significantly shorter RGEA lengths compared with the non-AL group (P < 0.001). Segmented regression analysis identified a cohort-specific threshold of 27.8 cm, below which the risk of AL increased. These findings suggest that shorter RGEA length is associated with elevated AL risk.
Shorter preoperative CTA-measured RGEA length is associated with a higher risk of AL. This cohort-specific threshold may serve as a risk-enrichment marker to inform perioperative planning and patient counseling.
Core Tip: Anastomotic leakage is a serious complication following McKeown esophagectomy. In this single-center re
- Citation: Sowe B, Lei P, Tang GX, Yang FJ, Wang SH, Song T. Computed tomographic angiography measured right gastroepiploic artery length and anastomotic leakage after McKeown esophagectomy: A retrospective cohort study. World J Gastrointest Surg 2026; 18(4): 118003
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/118003.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.118003
Esophageal cancer remains a significant global public health burden, with high morbidity and mortality despite advances in diagnostic modalities and multimodal treatment strategies[1]. Surgical resection continues to represent a cornerstone of curative-intent therapy, and McKeown esophagectomy (MKE) is widely performed for tumors of the thoracic esophagus. However, this procedure is associated with a substantial risk of postoperative complications[2,3], among which anasto
AL following esophagectomy is associated with prolonged hospitalization, increased healthcare costs, infectious complications, and impaired short- and long-term survival[4,6,7]. Reported AL rates range from 10% to 20%, with mortality rates of approximately 3%-4%[5,6,8-10]. Although multiple patient-related, tumor-related, and perioperative risk factors for AL have been identified, impaired conduit perfusion and excessive anastomotic tension remain central mechanisms in its pathogenesis[4,6,11-13].
In the context of MKE, the right gastroepiploic artery (RGEA) constitutes the dominant arterial supply to the gastric conduit. Inadequate arterial length may increase conduit tension and compromise perfusion at the anastomotic site, thereby increasing the risk of leakage[11]. Conversely, sufficient arterial flow facilitates the construction of a tension-free and well-perfused anastomosis. Recent studies have demonstrated an association between RGEA anatomy and anasto
Computed tomographic angiography (CTA) enables non-invasive, preoperative visualization of vascular anatomy with spatial resolution, allowing objective measurement of arterial length and course[14]. From a surgical planning perspec
Despite growing interest in vascular predictors of AL, data specifically evaluating the relationship between preopera
Accordingly, this retrospective cohort study aimed to evaluate the association between preoperative CTA-measured RGEA length and the incidence of AL following MKE for esophageal cancer. Additionally, we explored whether RGEA length thresholds within our cohort could serve as risk-enrichment markers to support preoperative planning. We hypothesized that shorter RGEA length is associated with higher risk of AL and that objective preoperative vascular assessment may provide incremental value when integrated into comprehensive perioperative decision-making.
This retrospective cohort study included patients diagnosed with esophageal cancer who underwent MKE at the Department of Thoracic Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, between January 2018 and December 2024. All surgeries were performed by experienced thoracic surgeons following standardized institutional protocols.
Medical records of patients with histologically confirmed esophageal cancer were reviewed. Postoperative tumor staging was determined according to the 8th edition of the American Joint Committee on Cancer Tumor, Node, Metastasis classification system[15]. The majority of the patients presented with stage II-III disease, while a smaller proportion had stage I disease. Tumor stage was not included in subsequent univariate or multivariate analyses, as it was not considered a primary determinant of AL risk in the context of this study.
Inclusion criteria: (1) Availability of preoperative CTA with clear visualization of the RGEA; (2) Completion of curative-intent MKE with gastric conduit reconstruction; and (3) Availability of complete clinical data for postoperative complica
Exclusion criteria: (1) Prior gastric or esophageal surgery that affected vascular anatomy; (2) Poor-quality CTA images precluding accurate measurement of RGEA length; (3) Patients who underwent other esophagectomy techniques (Ivor-Lewis or Transhiatal approaches); and (4) Incomplete clinical data.
A total of 679 patients met the inclusion criteria and were enrolled, as illustrated in Figure 1 (flowchart of patient inclusion, exclusion, and analysis).
The study protocol was reviewed and approved by the Institutional Review Board of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, approval No. 2025 (0895). Given the retrospective nature of the research, the requirement for informed consent was waived.
All enrolled patients underwent preoperative CTA using a Siemens scanner with standardized arterial-phase acquisition parameters. CTA datasets were independently reviewed by two board-certified radiologists, each with more than five years of experience in vascular imaging, who were blinded to postoperative outcomes.
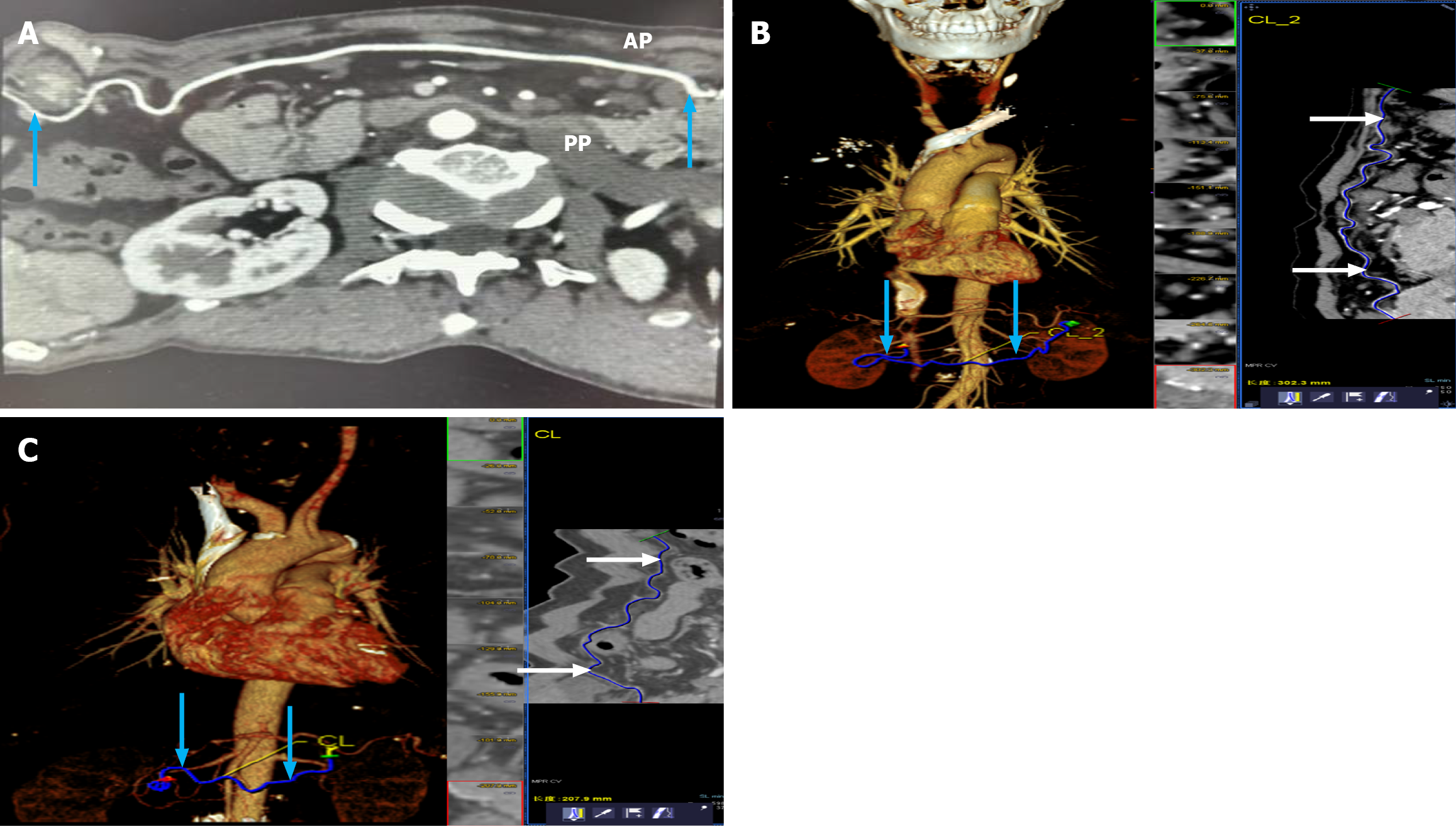
The length of the RGEA was defined as the distance from its origin at the gastroduodenal artery to its terminal branch along the greater curvature of the stomach. In cases where the gastroepiploic arcade demonstrated a relatively uniform caliber and the transition between the right and the left gastroepiploic arteries was not clearly distinguishable, the terminal point of the RGEA was defined as the point where the vessel crossed the midline of the greater curvature and began to course toward the splenic hilum, consistent with anatomical descriptions in prior CTA-based studies.
Measurements were performed along the arterial centerline using multiplanar reconstruction to accurately account for vessel curvature. Representative examples of measurement techniques for shorter and longer RGEAs, including two-dimensional CTA images, three-dimensional reconstructions, and integrated multiplanar views, are shown in Figure 2. Any interobserver measurement discrepancy greater than 2 mm was resolved through consensus.
All patients underwent MKE with cervical esophagogastric anastomosis at our institution. The procedures were performed by experienced thoracic surgeons following a standardized departmental protocol.
The vast majority of patients underwent minimally invasive MKE. Only two patients in the entire cohort underwent an open procedure, one in the AL group and one in the no-AL group. Given the minimal number of open cases, no subgroup or comparative analysis based on surgical approach was performed.
The procedure was initiated with the patient in a standard left lateral decubitus position. Mobilization of the thoracic esophagus and mediastinal lymphadenectomy were performed, adhering to the principle of en bloc resection of the esophagus with its adjacent lymphatic tissue.
During the abdominal phase, the stomach was fully mobilized with meticulous preservation of the RGEA, which served as the primary arterial supply to the gastric conduit. The left gastroepiploic artery, short gastric vessels, and left gastric artery were routinely divided. Care was taken to minimize traction, spasm, and thermal injury to the RGEA along the greater curvature.
A tubular gastric conduit was constructed along the greater curvature using linear staplers. The conduit width was standardized to approximately 3-4 cm across all cases to balance adequate perfusion with sufficient length for cervical anastomosis. Resection of the lesser curvature was performed consistently, extending from the gastric angle toward the cardia, thereby ensuring uniform conduit geometry. Although multiple surgeons participated in the procedures, conduit construction followed a uniform institutional protocol to reduce technical variability. Meticulous care was taken during transposition of the gastric conduit through the posterior mediastinum to ensure optimal orientation and to avoid undue tension or rotational torsion.
A left cervical incision was used to mobilize the cervical esophagus. The gastric conduit was delivered to the neck in a tension-free manner. A cervical esophagogastric anastomosis was performed using either a hand-sewn or stapled technique, according to the surgeon's preference. Adequate conduit reach, orientation, and perfusion were confirmed before completion of the anastomosis.
During the study period, intraoperative perfusion assessment techniques, such as indocyanine green fluorescence angiography, were not routinely employed. Evaluation of conduit viability relied on visual assessment, bleeding from the conduit edge, and the surgeon's experience.
Postoperative management followed the institutional enhanced recovery after surgery protocols. Routine contrast swallow or computed tomographic esophagography was performed on postoperative day 7, or earlier when clinical suspicion of leakage arose.
AL was defined according to the Esophagectomy Complications Consensus Group criteria as a full-thickness defect involving the esophagus, anastomosis, staple line, or conduit, with communication between intraluminal and extralumi
Clinical data, including demographic characteristics, comorbidities, tumor features, neoadjuvant therapy status, RGEA length, intraoperative details, and postoperative outcomes, were collected from electronic medical records. The occurrence, severity, and management of AL were documented according to Esophagectomy Complications Consensus Group standards.
Patients were divided into AL and non-AL groups based on postoperative outcomes. Continuous variables were expressed as mean ± SD, while categorical variables were expressed as frequencies and n (%).
Between-groups differences were analyzed using the t-test for continuous variables and the χ2 test for categorical variables. Univariate analyses were performed to identify potential risk factors.
Statistical analyses were conducted using EmpowerStats (http://www.empowerstats.com; X&Y Solutions, Inc., Boston, MA, United States) and R software version 4.5.1 (http://www.R-project.org; The R Foundation for Statistical Computing, Vienna, Austria). A 95% confidence interval (CI) was applied, and a two-sided P value < 0.050 was consi
A total of 679 patients who underwent MKE for esophageal cancer between January 2018 and December 2024 were included in the study. The cohort comprised 555 males (81.7%) and 124 females (18.3%), with a mean age of 62.08 ± 7.45 years and a mean body mass index of 22.50 ± 2.78 kg/m2.
Baseline demographic, clinical, and perioperative characteristics are summarized in Table 1. The AL and non-AL groups were well balanced with respect to age, sex, body mass index, smoking history, comorbidities, neoadjuvant therapy, and surgical approach, with no statistically significant differences observed.
| Parameters | Non-AL | AL | SD | P value | P value1 |
| Number of patients | 570 | 109 | - | - | - |
| Age (years), mean ± SD | 61.66 ± 7.67 | 62.50 ± 6.55 | 0.12 (-0.09 to 0.32) | 0.286 | 0.435 |
| BMI (kg/m2), mean ± SD | 22.63 ± 2.79 | 22.37 ± 2.76 | 0.10 (-0.11 to 0.30) | 0.365 | 0.369 |
| Length of RGEA measured with CTA (cm), mean ± SD | 28.36 ± 3.69 | 23.53 ± 2.46 | 1.54 (1.32, 1.76) | < 0.001 | < 0.001 |
| Sex (male/female) | 468/102 | 82/22 | 0.06 (-0.15 to 0.26) | 0.571 | - |
| Smoking history | 0.14 (-0.07 to 0.34) | 0.396 | - | ||
| Never smoking | 332 (58.25) | 61 (55.96) | - | ||
| Previously smoking | 120 (21.05) | 29 (26.61) | - | ||
| Presently smoking | 118 (20.70) | 19 (17.43) | - | ||
| Neoadjuvant therapy | 0.23 (0.04, 0.43) | 0.115 | - | ||
| No | 342 (61.62) | 75 (60.48) | - | ||
| Chemotherapy | 141 (25.41) | 24 (19.35) | - | ||
| Chemotherapy with immunotherapy | 49 (8.83) | 19 (15.32) | - | ||
| Others | 23 (4.14) | 6 (4.84) | - | ||
| Hypertension (yes/no) | 59/511 | 15/94 | 0.10 (-0.10 to 0.31) | 0.295 | - |
| Diabetes mellitus (yes/no) | 19/551 | 15/94 | 0.06 (-0.14 to 0.27) | 0.516 | - |
| Family history of cancer (yes/no) | 12/558 | 3/106 | 0.04 (-0.16 to 0.25) | 0.674 | - |
| Surgical method (MI/open) | 569/1 | 108/1 | 0.10 (-0.10 to 0.31) | 0.190 | 0.295 |
In contrast, preoperative CTA-measured RGEA length differed markedly between groups. Patients who developed AL had a significantly shorter RGEA compared with those without AL (23.53 ± 2.46 cm vs 28.36 ± 3.69 cm; P < 0.001), indicating that RGEA length may serve as an anatomical risk marker for AL.
Among the 679 patients, 109 (16.10%) developed AL. The remaining 570 patients (83.95%) did not experience AL, although some patients may have experienced other postoperative complications, as shown in Table 1.
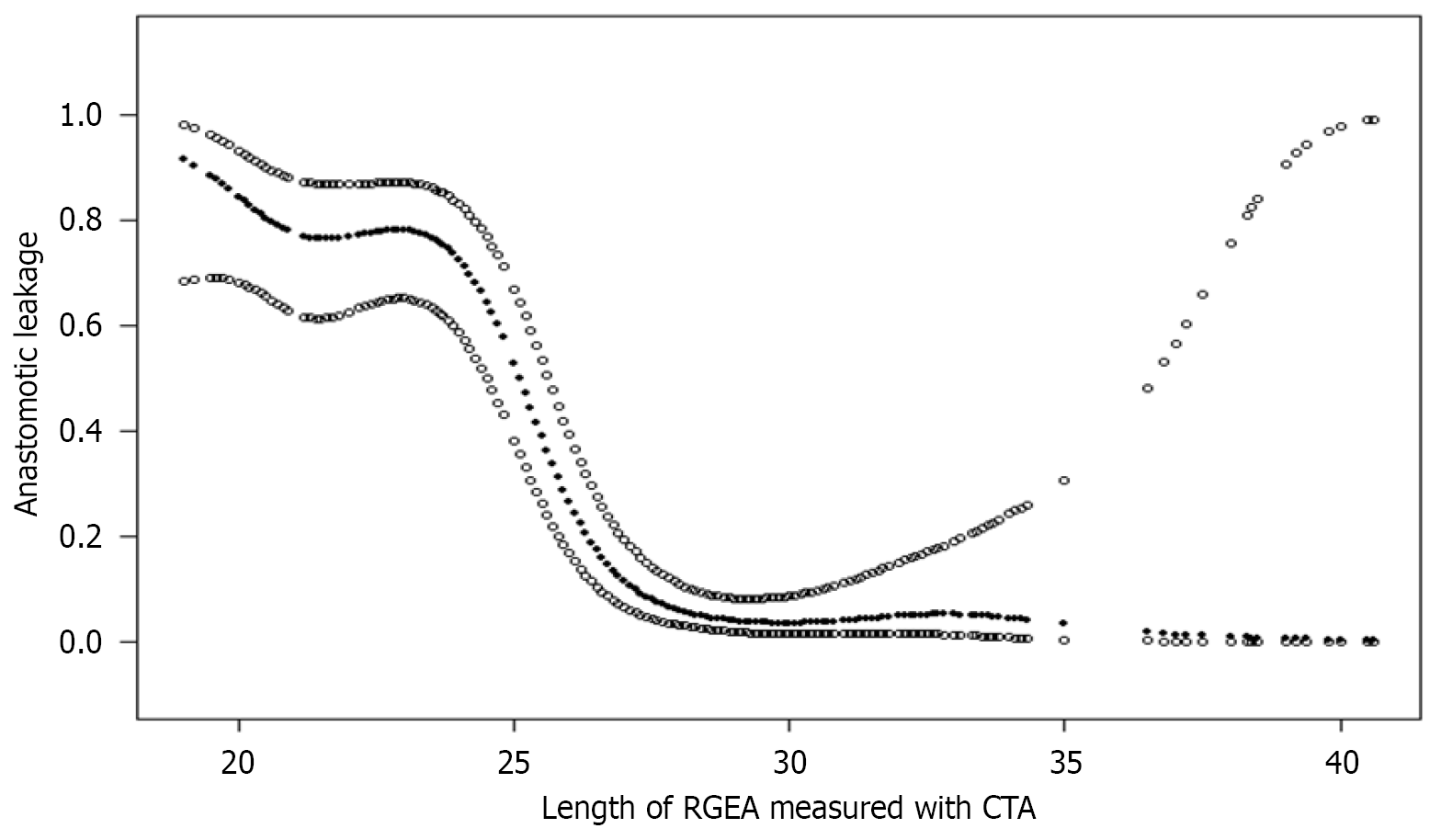
Statistical analysis revealed that patients with shorter RGEA length exhibited a markedly higher incidence of AL. To further delineate this association, a threshold (segmented regression) analysis was performed, demonstrating a clear inflection point. As shown in Figure 3, the probability of AL increased sharply when RGEA length fell below 27.80 cm, which was identified as the critical cut-off (K) value. This relationship was further quantified in Table 2, where patients with an RGEA shorter than 27.80 cm exhibited a significantly elevated risk of AL (P < 0.001).
| Outcome | Estimated (95%CI) | P value |
| Model I: Linear effect | 0.5 (0.4, 0.6) | < 0.001 |
| Model II: Threshold (K) | 27.80 cm | - |
| Segmental effect 1 (< K) | 0.4 (0.3, 0.5) | < 0.001 |
| Segmental effect 2 (> K) | 0.9 (0.7, 1.2) | 0.638 |
| Difference between 2 and 1 | 2.7 (1.7, 4.2) | < 0.001 |
| Predicted log relative risk at the cutoff point | -4.0 (-4.7 to -3.2) | - |
| Likelihood ratio test | - | < 0.001 |
To explore this relationship further, segmented regression analysis was performed. An inflection point was observed at an RGEA length of 27.8 cm (Table 2, Figure 3). Below this threshold, patients exhibited a significantly higher risk of AL [segmental effect: 0.4 (95%CI: 0.3-0.5), P < 0.001], whereas no significant association was observed above the threshold [0.9 (95%CI: 0.7-1.2), P = 0.638]. The difference between segments was 2.7 (95%CI: 1.7-4.2, P < 0.001), and the predicted log relative risk at the cutoff point was -4.0 (95%CI: -4.7 to -3.2). The likelihood ratio test confirmed the improved fit of the segmented model (P < 0.001).
These findings are consistent with the unadjusted analysis and indicate that shorter RGEA length is associated with increased risk of AL, particularly below the cohort-specific threshold of 27.8 cm. This threshold is interpreted as a risk-enrichment marker rather than a prescriptive clinical cut-off.
In this retrospective cohort study, we investigated the association between preoperative CTA-measured RGEA length and the risk of AL following MKE for esophageal cancer. Our findings demonstrated that shorter RGEA length was significantly associated with a higher incidence of AL. Segmental regression analysis identified a cohort-specific threshold of 27.8 cm, below which the probability of leakage increased markedly. These results indicate that preoperative quantitative assessment of RGEA length may serve as a risk-enrichment marker, providing objective anatomical information to support perioperative planning.
AL remains one of the most serious complications after esophagectomy, with reported incidences ranging from 10% to 20%, consistent with our observed rate of 16.10%[4-6,8,17]. The etiology of AL is multifactorial, including patient-related, tumor-related, and surgery-related factors[5,6,8,10].
Our analysis emphasizes the importance of adequate vascular supply to the gastric conduit, particularly via the RGEA. Patients with shorter RGEA length exhibited higher rates of AL, supporting the hypothesis that limited arterial length may increase conduit tension and compromise microvascular perfusion at the anastomotic site[11,18]. The identified 27.8 cm threshold represents a cohort-specific risk-enrichment marker rather than a universal clinical cut-off.
However, due to the retrospective nature of this study and the variability in CTA image acquisition protocols over the study period, reliable and standardized assessment of the extra-organ vascular arcade between the right and left gastroepiploic arteries was not feasible for all patients. In particular, distinguishing between a single dominant arch and dual or weak interconnections was inconsistent across cases, precluding meaningful quantitative analysis.
Therefore, RGEA length was investigated as a measurable, reproducible, and preoperatively accessible surrogate vascular parameter, rather than as a comprehensive representation of gastric conduit perfusion. We acknowledge that patients with a short RGEA but a robust inter-gastroepiploic connection may indeed have a lower leakage risk than those with a long RGEA and poor collateralization.
These observations align with previous anatomical and physiological studies suggesting that tension, ischemia, and impaired perfusion are central contributors to anastomotic dehiscence[19-26]. Preoperative quantification of RGEA length provides objective data that can complement intraoperative judgment and conventional risk assessment.
Several prior studies have explored AL risk factors - including nutritional status, neoadjuvant therapy, tumor characteristics, and surgical technique - but few have systematically examined vascular anatomy as a measurable predictor[4,6,8,10,27-29]. While anatomical variations of the RGEA and its importance for gastric conduit perfusion have been described, most studies relied on intraoperative or postoperative observations rather than standardized preoperative imaging[11,30-32].
Li et al[11] reported an association between RGEA length and AL, but an important limitation of this study was the intraoperative determination of arterial length and the lack of threshold quantification. Our findings extend this knowledge by providing a non-invasive, reproducible, preoperative measurement of RGEA length and by identifying a cohort-specific threshold that may aid in risk stratification.
While the relationship between shorter RGEA and AL is largely confirmatory of known surgical principles, quantifying arterial length preoperatively may support anticipatory planning. For patients with relatively short RGEA, surgeons may consider strategies such as: (1) Enhanced intraoperative vigilance for anastomotic tension; (2) Use of perfusion assessment techniques (e.g., indocyanine green fluorescence); and (3) Consideration of reinforcement or supercharging techniques.
It is important to emphasize that the identified threshold is exploratory, cohort-specific, and intended as a risk-enrichment tool rather than a prescriptive or universal criterion.
Future studies may aim to validate these findings prospectively in multicenter cohorts to assess reproducibility across different populations and surgical practices. Integration of preoperative anatomical measurements with other risk factors may improve predictive models for AL. The potential role of automated imaging analysis or three-dimensional vascular modeling could be explored in the future, but such applications remain experimental and were beyond the scope of the present study.
Several limitations should be acknowledged: (1) This is a retrospective, single-center study, and selection or information bias cannot be excluded; (2) Although CTA provides high-resolution vascular imaging, interobserver variability may affect measurement accuracy; (3) While baseline patient characteristics were comparable and surgical technique was standardized, unmeasured intraoperative factors (e.g., conduit length, surgeon experience, perfusion assessment) may contribute to residual confounding; (4) The 27.8 cm threshold is derived from this specific cohort and may not be generalizable to other populations or institutions. External and prospective validations are therefore required; and (5) CTA is not universally performed preoperatively; its utility should be considered complementary rather than mandatory.
In conclusion, shorter preoperative CTA-measured RGEA length is associated with an increased risk of AL following MKE. Segmental analysis identified a cohort-specific threshold of 27.8 cm, below which patients were at higher risk. The cohort-specific threshold highlights the potential value of preoperative vascular assessment as a risk-enrichment marker to support perioperative planning and patient counseling.
The authors express their sincere gratitude to all the doctors and nurses in the Department of Thoracic Surgery, Union Hospital, Tongji Medical College, for their invaluable support and collaboration throughout this study.
| 1. | Lander S, Lander E, Gibson MK. Esophageal Cancer: Overview, Risk Factors, and Reasons for the Rise. Curr Gastroenterol Rep. 2023;25:275-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 85] [Article Influence: 28.3] [Reference Citation Analysis (3)] |
| 2. | Zheng XD, Li SC, Lu C, Zhang WM, Hou JB, Shi KF, Zhang P. Safety and efficacy of minimally invasive McKeown esophagectomy in 1023 consecutive esophageal cancer patients: a single-center experience. J Cardiothorac Surg. 2022;17:36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 3. | Zhai C, Liu Y, Li W, Xu T, Yang G, Lu H, Hu D. A comparison of short-term outcomes between Ivor-Lewis and McKeown minimally invasive esophagectomy. J Thorac Dis. 2015;7:2352-2358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 31] [Reference Citation Analysis (0)] |
| 4. | Hagens ERC, Reijntjes MA, Anderegg MCJ, Eshuis WJ, van Berge Henegouwen MI, Gisbertz SS. Risk Factors and Consequences of Anastomotic Leakage After Esophagectomy for Cancer. Ann Thorac Surg. 2021;112:255-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 65] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 5. | Fabbi M, Hagens ERC, van Berge Henegouwen MI, Gisbertz SS. Anastomotic leakage after esophagectomy for esophageal cancer: definitions, diagnostics, and treatment. Dis Esophagus. 2021;34:doaa039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (7)] |
| 6. | Moon SW, Kim JJ, Cho DG, Park JK. Early detection of complications: anastomotic leakage. J Thorac Dis. 2019;11:S805-S811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 7. | Tiainen E, Wirta EV, Tyrväinen T, Kuorilehto T, Toikkanen V, Laukkarinen J, Ukkonen M. Calcifications in the descending thoracic aorta predict postoperative anastomotic leakages after esophagectomy for cancer. World J Surg. 2024;48:1209-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Weber MC, Jorek N, Neumann PA, Bachmann J, Dimpel R, Martignoni M, Feith M, Friess H, Novotny A, Berlet M, Reim D. Incidence and treatment of anastomotic leakage after esophagectomy in German acute care hospitals: a retrospective cohort study. Int J Surg. 2025;111:2953-2961. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 9. | Pace M, Minervini A, Goglia M, Cinquepalmi M, Moschetta G, Antolino L, D'Angelo F, Valabrega S, Petrucciani N, Berardi G, Aurello P. Overall Survival Following Anastomotic Leakage After Surgery for Carcinoma of the Esophagus and Gastroesophageal Junction: A Systematic Review. In Vivo. 2023;37:1423-1431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Scognamiglio P, Reeh M, Melling N, Kantowski M, Eichelmann AK, Chon SH, El-Sourani N, Schön G, Höller A, Izbicki JR, Tachezy M. Management of intra-thoracic anastomotic leakages after esophagectomy: updated systematic review and meta-analysis of endoscopic vacuum therapy versus stenting. BMC Surg. 2022;22:309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 11. | Li KK, Wang YJ, Zhang TM, Zhang L, Zhao SL, Chen L, Bao T, Zhao XL, Xie XF, Guo W. Right gastroepiploic artery length determined anastomotic leakage after minimally invasive esophagectomy for esophageal cancer: a prospective cohort study. Int J Surg. 2024;110:2757-2764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 12. | Luo RJ, Zhu ZY, He ZF, Xu Y, Wang YZ, Chen P. Efficacy of Indocyanine Green Fluorescence Angiography in Preventing Anastomotic Leakage After McKeown Minimally Invasive Esophagectomy. Front Oncol. 2020;10:619822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Hoek VT, Edomskis PP, Menon AG, Kleinrensink GJ, Lagarde SM, Lange JF, Wijnhoven BPL. Arterial calcification is a risk factor for anastomotic leakage after esophagectomy: A systematic review and meta-analysis. Eur J Surg Oncol. 2020;46:1975-1988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 14. | Lim B, Cevik J, Seth I, Sofiadellis F, Ross RJ, Rozen WM, Cuomo R. Evaluating Artificial Intelligence's Role in Teaching the Reporting and Interpretation of Computed Tomographic Angiography for Preoperative Planning of the Deep Inferior Epigastric Artery Perforator Flap. JPRAS Open. 2024;40:273-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 15. | Rice TW, Ishwaran H, Hofstetter WL, Kelsen DP, Apperson-Hansen C, Blackstone EH; Worldwide Esophageal Cancer Collaboration Investigators. Recommendations for pathologic staging (pTNM) of cancer of the esophagus and esophagogastric junction for the 8th edition AJCC/UICC staging manuals. Dis Esophagus. 2016;29:897-905. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 151] [Article Influence: 15.1] [Reference Citation Analysis (4)] |
| 16. | Low DE, Alderson D, Cecconello I, Chang AC, Darling GE, DʼJourno XB, Griffin SM, Hölscher AH, Hofstetter WL, Jobe BA, Kitagawa Y, Kucharczuk JC, Law SY, Lerut TE, Maynard N, Pera M, Peters JH, Pramesh CS, Reynolds JV, Smithers BM, van Lanschot JJ. International Consensus on Standardization of Data Collection for Complications Associated With Esophagectomy: Esophagectomy Complications Consensus Group (ECCG). Ann Surg. 2015;262:286-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 975] [Cited by in RCA: 917] [Article Influence: 83.4] [Reference Citation Analysis (4)] |
| 17. | Low DE, Kuppusamy MK, Alderson D, Cecconello I, Chang AC, Darling G, Davies A, D'Journo XB, Gisbertz SS, Griffin SM, Hardwick R, Hoelscher A, Hofstetter W, Jobe B, Kitagawa Y, Law S, Mariette C, Maynard N, Morse CR, Nafteux P, Pera M, Pramesh CS, Puig S, Reynolds JV, Schroeder W, Smithers M, Wijnhoven BPL. Benchmarking Complications Associated with Esophagectomy. Ann Surg. 2019;269:291-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 699] [Cited by in RCA: 612] [Article Influence: 87.4] [Reference Citation Analysis (3)] |
| 18. | Prasetya H, Jansen SM, Marquering HA, van Leeuwen TG, Gisbertz SS, de Bruin DM, van Bavel E. Estimation of microvascular perfusion after esophagectomy: a quantitative model of dynamic fluorescence imaging. Med Biol Eng Comput. 2019;57:1889-1900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 19. | Kiełbowski K, Wojtyś M, Kostopanagiotou K, Janowski H, Wójcik J. Association of major postoperative wound and anastomotic complications in thoracic surgery with COVID-19 infection. Surg Open Sci. 2022;10:208-215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 20. | Duan X, Bai W, Ma Z, Yue J, Shang X, Jiang H, Yu Z. Management and outcomes of anastomotic leakage after McKeown esophagectomy: A retrospective analysis of 749 consecutive patients with esophageal cancer. Surg Oncol. 2020;34:304-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 21. | Farran L, Llop J, Sans M, Kreisler E, Miró M, Galan M, Rafecas A. Efficacy of enteral decontamination in the prevention of anastomotic dehiscence and pulmonary infection in esophagogastric surgery. Dis Esophagus. 2008;21:159-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Junemann-Ramirez M, Awan MY, Khan ZM, Rahamim JS. Anastomotic leakage post-esophagogastrectomy for esophageal carcinoma: retrospective analysis of predictive factors, management and influence on longterm survival in a high volume centre. Eur J Cardiothorac Surg. 2005;27:3-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 154] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 23. | Lu M, Luketich JD, Levy RM, Awais O, Sarkaria I, Visintainer P, Nason KS. Anastomotic complications after esophagectomy: Influence of omentoplasty in propensity-weighted cohorts. J Thorac Cardiovasc Surg. 2020;159:2096-2105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Bakken JC, Law R, Wigle D, Baron TH. Anastomotic dehiscence after esophagogastrectomy treated with stent and tissue matrix graft. Surg Endosc. 2013;27:4383-4384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 25. | Siska D, Janik M, Laucek P, Lucenic M, Tarabova K, Juhos P, Balaz R, Turcinova M, Gallikova Z, Benej R. Management of anastomotic leaks after oesophagectomy and gastric pull-up. Bratisl Lek Listy. 2023;124:508-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 26. | Murphy T, Jobe BA. Endoluminal management of anastomotic dehiscence after esophagectomy: an increasing quiver of options reflects the difficulty in realizing a definitive therapy. Gastrointest Endosc. 2010;71:387-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 27. | Sun ZW, Du H, Li JR, Qin HY. Constructing a risk prediction model for anastomotic leakage after esophageal cancer resection. J Int Med Res. 2020;48:300060519896726. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 28. | Klontzas ME, Ri M, Koltsakis E, Stenqvist E, Kalarakis G, Boström E, Kechagias A, Schizas D, Rouvelas I, Tzortzakakis A. Prediction of Anastomotic Leakage in Esophageal Cancer Surgery: A Multimodal Machine Learning Model Integrating Imaging and Clinical Data. Acad Radiol. 2024;31:4878-4885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 29. | Nie X, Nie X, Xie Q, Li C, He W, Ni K, Lu S, Wang C, Miao Y, Jiang L, Wang K, Liu G, Fang Q, Peng L, Shi Q, Kanamori J, Aiolfi A, Deana C, Han Y, Leng X. Patient-reported outcomes of the prognostic nutritional index for resectable esophageal squamous cell carcinoma. J Thorac Dis. 2025;17:5133-5145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 30. | Nickel F, Studier-Fischer A, Özdemir B, Odenthal J, Müller LR, Knoedler S, Kowalewski KF, Camplisson I, Allers MM, Dietrich M, Schmidt K, Salg GA, Kenngott HG, Billeter AT, Gockel I, Sagiv C, Hadar OE, Gildenblat J, Ayala L, Seidlitz S, Maier-Hein L, Müller-Stich BP. Optimization of anastomotic technique and gastric conduit perfusion with hyperspectral imaging and machine learning in an experimental model for minimally invasive esophagectomy. Eur J Surg Oncol. 2025;51:106908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 31. | Hayashi E, Yuasa N, Sasaki E, Kamiya J, Nagino M, Nimura Y, Hirooka Y. Right gastroepiploic artery occlusion test for resection of recurrent lesion after esophageal reconstruction using a gastric tube. Am J Surg. 2004;187:446-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 32. | Hijab A, Tova Y, Alani S. The Right Gastroepiploic Artery as a Potential Organ at Risk in Neoadjuvant Chemoradiation for Esophageal and Gastroesophageal Cancers. Cureus. 2024;16:e61342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |