Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.119293
Revised: February 12, 2026
Accepted: March 10, 2026
Published online: April 27, 2026
Processing time: 88 Days and 4.6 Hours
Postoperative delirium (POD) is common in older surgical patients and increases morbidity and mortality. Oxiracetam, a nootropic agent, has not been evaluated for POD prevention.
To assess the efficacy of perioperative oxiracetam in reducing POD incidence.
In this multicenter, randomized, double-blind, placebo-controlled trial, a total of 173 elderly patients (age ≥ 65 years) scheduled for elective laparoscopic gastro
POD incidence was lower in the OG [13.4% (11/82)] than in the placebo group [28.6% (26/91)] (P < 0.01). Oxiracetam remained an independent protective factor after multivariate adjustment (adjusted odds ratio = 0.12, 95% confidence interval: 0.04-0.36, P < 0.01). The OG also showed better preservation of cognitive function (Montreal Cognitive Assessment score). Inflammatory profiling revealed a distinct immunomodulatory pattern: Oxiracetam attenuated postoperative rises in interleukin (IL)-6, IL-8, and IL-1β while enhancing levels of IL-10 and IL-2.
Perioperative oxiracetam significantly reduces POD risk and preserves cognitive function in elderly surgical patients, potentially through a multi-target immunomodulatory mechanism rather than simple anti-inflammation.
Core Tip: Perioperative intravenous oxiracetam safely and effectively reduces the risk of postoperative delirium in elderly patients undergoing laparoscopic gastrointestinal surgery, and maintains postoperative cognitive functionality. Its protective effect likely involves multi-target mechanisms, including stabilization complex modulation of the systemic immune response, rather than dependence on simple suppression of a single inflammatory pathway. This study provides a foundation for the clinical application of oxiracetam as a perioperative brain protection strategy for elderly surgical patients.
- Citation: Lv BS, Wang W, Jin M, Ma Y, Wang JL, Gao H. Impact of oxiracetam on postoperative delirium in elderly patients undergoing laparoscopic gastrointestinal surgery. World J Gastrointest Surg 2026; 18(4): 119293
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/119293.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.119293
Delirium is an acute disturbance in attention and cognition that is not explained by a preexisting neurocognitive disorder or severe reduction in arousability[1,2]. Some of the common characteristics include disturbance in attention, impaired cognition and memory, disorientation, or perceptual disturbances[1]. The subtypes include hypoactive delirium, characterized by reduced activity, and alertness; hyperactive delirium, characterized by agitation, irritability, and hallucinations; and mixed delirium[3]. Delirium is common in the postoperative settings, especially in the older population, ranging from 10%-26% depending on the complexity and type of surgery[4]. Postoperative delirium (POD) is associated with increased complications and mortality, unplanned intensive care unit admissions, prolonged length of hospital stay, and higher non-home discharges[5].
Several mechanisms are observed in the development of POD, including neurotransmitter imbalance, impaired neural connectivity, and neuroinflammation. Systemic inflammatory response triggered by surgical trauma activates the innate immune system and initiates the inflammatory process, ultimately leading to POD[6]. Therefore, modulating the perioperative inflammatory state may prevent POD, ultimately preventing the undesired impact of POD on surgical outcomes.
Oxiracetam is a nootropic drug used to improvememory and cognitive function[7]. It passes through the blood-brain barrier and is distributed throughout the cortex and hippocampus[8,9]. Rodent studies indicate its neuroprotective effects through enhanced acetylcholine (ACh) release and choline acetyltransferase activity in the hippocampus[10]. Studies have assessed the efficacy of oxiracetam in various neurological disorders, such as dementia[11] and traumatic brain injury (TBI)[12]. However, it’s effect on the incidence of POD is yet to be studied.
Therefore, this multicenter randomized controlled trial (RCT) was conducted to test whether the perioperative administration of Oxiracetam can reduce the incidence of POD in elderly patients undergoing laparoscopic gastro
This was a prospective, randomized, double-blind, placebo-controlled, multicenter clinical trial. The included patients were aged ≥ 65 years, with American Society of Anesthesiologists (ASA) physical status I-III, who were scheduled for elective laparoscopic gastrointestinal surgery. Patient recruitment took place between March 2021 and November 2025.The study protocol was approved by the ethics committees of Hebei Yanda Hospital (2021-3-008) and registered in the Chinese Clinical Trial Registry (Registration Number: ChiCTR2300076112). Written informed consent was obtained from all patients or their legal guardians. The patients with a baseline Montreal Cognitive Assessment (MoCA) score < 23, indicating pre-existing cognitive impairment were excluded. Those with a history of psychiatric disorders, dementia, or severe cerebrovascular disease were also excluded. Other exclusion criteria included patients with severe hepatic or renal insufficiency, long-term use of antipsychotics or sedative-hypnotics, and a known allergy to oxiracetam.
The sample size was calculated based on the primary outcome of POD incidence. According to previous literature and pilot data, we assumed a POD incidence of 55% in the control group. To ensure clinical significance, we hypothesized that oxiracetam would reduce the incidence to 30% (an absolute reduction of 25%). Using a two-sided test with a significance level (α) of 0.05, a power (1 - β) of 0.80, and an allocation ratio of 1:1, the formula for comparing two independent proportions was applied.
With p1 = 0.55, p2 = 0.30, Z1 - α/2 = 1.96, and Z1 - β = 0.84, the calculation yielded a minimum required sample size of approximately 61 patients per group (total n ≈ 122). Accounting for an estimated 10% dropout rate, the final target sample size was 68-69 patients per group (total n ≈ 136). This study ultimately enrolled 173 patients [91 in the control group and 82 in the oxiracetam group (OG)], which meets and exceeds the calculated requirement, ensuring adequate statistical power to detect the hypothesized effect size.
Computer-generated block randomization sequences were used to assign patients in a 1:1 ratio to the OG or the normal saline control group (SG).
Study medications (oxiracetam or identical-appearing normal saline) were prepared by pharmacists not involved in patient management or outcome assessment, according to the randomization codes. Patients, investigators, and outcome assessors were blinded to group assignment.
OG: Intravenous infusion of oxiracetam injection (4.0 g dissolved in 250 mL normal saline) was initiated 30 minutes before anesthesia induction and repeated daily at the same time point from postoperative day 1 to day 6.
SG: Received an equal volume of normal saline infusion at the same time points.
Both groups received standardized anesthesia and perioperative management protocols.
The primary outcome assessment was the incidence of POD within 7 postoperative days.
POD is diagnosed using Jacobson and Truax’s reliable change index: Z-score = [(X1-X2)-△xc]/standard deviation of change. POD was diagnosed if the Z-score of two tests was greater than 1.64. As many patients developed POD in the ward, confusion assessment method scoring was not routinely conducted in the ward, and thus this data was not routinely collected.
Additional or secondary measures included: (1) Cognitive function assessed using the MoCA one day before surgery and on postoperative day 7; and (2) Inflammatory markers: Venous blood samples were collected preoperatively (T0), and on postoperative day 1 (T1), day 2 (T2), and day 7 (T3), and serum levels of interleukin (IL)-1β, IL-2, IL-6, IL-8, IL-10, and tumor necrosis factor-α (TNF-α) were measured using enzyme-linked immunosorbent assay. The MoCA scale test, Stroop word color interference test and cross-connection test were conducted respectively before the operation and 7 days after the operation.
Data were analyzed using SPSS software (version 24.0). Continuous data are presented as mean ± SD or median (interquartile range) and were compared between groups using the t-test or Mann-Whitney U test. Categorical data are presented as n (%) and were compared using the χ2 test. The independent effect of Oxiracetam on POD was analyzed using multivariate logistic regression models, with stepwise adjustment for demographic, clinical, and preoperative inflammatory factors. All tests were two-sided, and a P value < 0.05 was considered statistically significant.
To ensure the robustness of the regression model, we calculated the variance inflation factor (VIF) to detect collinearity between variables. All VIF values were less than 5, indicating that the collinearity problem was not significant. In addition, the influence points in the data were evaluated using the Cook’s distance, and no influence points were found. All Cook’s distances were less than 1.
When dealing with missing data, we adopted Multiple imputation to handle the missing postoperative inflammatory factor data. Multiple sets of missing data were generated through interpolation method, and sensitivity analysis was conducted to ensure the rationality of the interpolation results and the consistency of the data. In addition, for variables with less missing data, the complete case analysis method was used to further ensure the robustness of the results.
This study included a total of 173 elderly patients undergoing laparoscopic gastrointestinal surgery, with 91 in the control group and 82 in the OG. Comparison of baseline data between the two groups showed no statistically significant differences in demographic characteristics [age, gender, body mass index body mass index (BMI)], clinical risk classification (ASA grade), disease diagnosis type, surgery-related indicators (operation time, intraoperative blood loss, intraoperative blood transfusion rate), or hospital stay (all P > 0.05). The baseline characteristics of the two groups were well comparable. The baseline data of the study subjects are shown in Table 1.
| Characteristic | Control group | Oxiracetam group | Statistic | P value |
| n | 91 | 82 | ||
| Age (years old) | 71 ± 9 | 71 ± 9 | t = 0.09 | 0.9261 |
| Gender | χ2 = 0.31 | 0.5762 | ||
| Female | 38 (41.8) | 33 (40.2) | ||
| Male | 53 (58.2) | 49 (59.8) | ||
| BMI, mean ± SD | 23.1 ± 4.1 | 23.3 ± 3.9 | t = -0.46 | 0.6451 |
| ASA grade | χ2 = 1.66 | 0.1982 | ||
| II | 72 (79.1) | 65 (79.3) | ||
| III | 19 (20.9) | 17 (20.7) | ||
| Diagnosis | χ2 = 3.68 | 0.1652 | ||
| Colorectal cancer | 63 (69.2) | 57(69.5) | ||
| Others | 28 (30.8) | 25 (30.5) | ||
| Operation time (minute) | 246 ± 132 | 246 ± 127 | t = -0.05 | 0.9621 |
| Intraoperative blood loss (mL) | 103 ± 134 | 113 ± 242 | t = -0.34 | 0.7361 |
| Intraoperative blood transfusion | 11 (12.1) | 9 (11.0) | χ2 = 0.20 | 0.6542 |
| Hospital stay (days) | 22.5 ± 5.0 | 20.7 ± 7.9 | t = 1.83 | 0.0701 |
The MoCA was used to evaluate perioperative cognitive function changes. Results showed comparable baseline cognitive function levels between the two groups preoperatively. The control group showed a mild decline in postoperative cognitive function compared to baseline, while the OG maintained cognitive function essentially at preoperative levels. These results suggest that perioperative intravenous infusion of Oxiracetam can, to some extent, mitigate cognitive fluctuations in elderly patients after laparoscopic gastrointestinal surgery, exerting a protective effect on postoperative cognitive function. Changes in MoCA before and after intervention are shown in Table 2.
| Treatment | Pre-operative | Post-operative | Change from baseline | |
| mean (SD) | LS mean (95%CI) | |||
| Control group | 24.7 (1.25) | 24.3 (1.21) | -0.3 (0.81) | -0.36 (-0.61, -0.11) |
| Oxiracetam group | 25.2 (1.70) | 25.1 (2.21) | -0.2 (1.48) | -0.14 (-0.39, 0.11) |
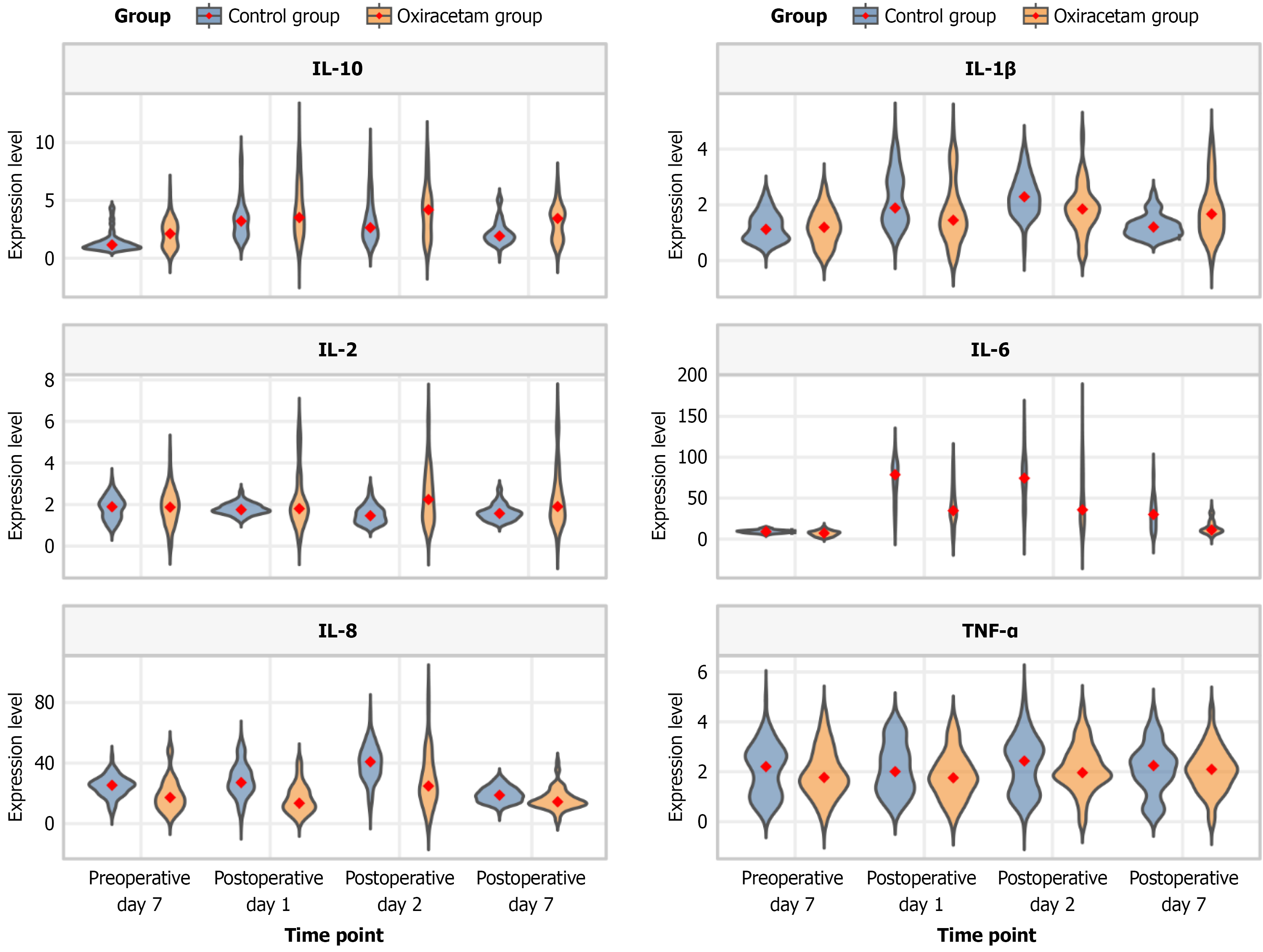
Preoperatively, there were no significant differences in the levels of inflammatory factors between the OG and the control group (all P > 0.05). On postoperative day 1, IL-6, IL-8, and IL-1β levels in the OG were significantly lower than in the control group (all P < 0.05); there were no between-group differences in IL-2, IL-10, and TNF-α. On postoperative day 2, IL-6, IL-8, and IL-1β levels in the OG remained significantly lower than in the control group (all P < 0.05), while IL-2 and IL-10 were significantly higher than in the control group (both P < 0.01); there was no between-group difference in TNF-α. On postoperative day 7, IL-6 levels in the OG were significantly lower than in the control group (P < 0.001), while IL-10 and IL-2 were significantly higher and IL-1β were lower than in the control group (all P < 0.05); there were no between-group differences in IL-8 and TNF-α. The relationship between Oxiracetam and inflammatory factor levels is shown in Table 3 and Figure 1.
| Time | Characteristic | Control group | Oxiracetam group | P value |
| n | 91 | 82 | ||
| Pre | IL-1β | 1.25 (0.80, 1.71) | 1.27 (0.87, 1.89) | 0.792 |
| IL-2 | 1.95 (1.45, 2.34) | 2.12 (1.54, 2.81) | 0.371 | |
| IL-6 | 9.5 (7.9, 10.7) | 9.4 (7.8, 10.6) | 0.831 | |
| IL-8 | 26 (21, 30) | 25 (20, 31) | 0.582 | |
| IL-10 | 1.24 (0.89, 2.34) | 1.30 (0.90, 2.30) | 0.711 | |
| TNF-α | 2.28 (1.07, 2.84) | 1.78 (1.25, 2.56) | 0.408 | |
| Day 1 | IL-1β | 1.95 (1.39, 2.98) | 1.63 (1.08, 3.10) | 0.012 |
| IL-2 | 1.79 (1.65, 2.07) | 1.92 (1.42, 3.50) | 0.222 | |
| IL-6 | 79 (58, 89) | 36 (27, 55) | < 0.001 | |
| IL-8 | 28 (22, 36) | 15 (11, 24) | < 0.001 | |
| IL-10 | 3.4 (2.2, 4.9) | 3.8 (2.4, 6.4) | 0.314 | |
| TNF-α | 2.01 (1.31, 2.86) | 1.78 (1.24, 2.53) | 0.218 | |
| Day 2 | IL-1β | 2.33 (1.79, 2.89) | 1.92 (1.45, 2.77) | 0.011 |
| IL-2 | 1.56 (1.18, 1.85) | 2.34 (1.34, 3.47) | < 0.001 | |
| IL-6 | 76 (60, 94) | 39 (22, 77) | < 0.001 | |
| IL-8 | 41 (32, 49) | 27 (17, 46) | < 0.001 | |
| IL-10 | 2.9 (2.0, 5.1) | 4.5 (2.5, 6.0) | 0.008 | |
| TNF-α | 2.45 (1.18, 2.99) | 2.01 (1.70, 2.92) | 0.892 | |
| Day 7 | IL-1β | 1.76 (1.09, 2.58) | 1.25 (0.97, 1.58) | 0.005 |
| IL-2 | 1.61 (1.39, 2.01) | 2.14 (1.34, 3.48) | 0.013 | |
| IL-6 | 31 (16, 44) | 12 (9, 22) | < 0.001 | |
| IL-8 | 19 (15, 24) | 16 (12, 26) | 0.093 | |
| IL-10 | 2.15 (1.65, 3.27) | 3.57 (1.78, 4.27) | 0.012 | |
| TNF-α | 2.25 (1.65, 2.78) | 2.12 (1.54, 2.76) | 0.705 | |
Among the 173 patients included in this study, 37 were in the delirium group and 136 in the non-delirium group. Preoperatively, there were no differences in the levels of inflammatory factors between two groups. On postoperative day 1, median IL-6 and IL-8 levels in the delirium group were significantly higher than in the non-delirium group (both P < 0.001), while IL-2 and IL-10 levels were significantly lower (both P < 0.001); there were no statistical differences in IL-1β and TNF-α levels. On postoperative day 2, IL-8 levels were higher in the delirium group (P = 0.019), while IL-2, IL-10, and TNF-α levels were lower (P < 0.001 to P = 0.021); there were no between-group differences in IL-1β and IL-6 levels. By postoperative day 7, IL-2 and IL-10 levels in the delirium group remained significantly lower than in the non-delirium group (P < 0.001 to P = 0.003), IL-1β, TNF-α and IL-6 levels were higher ((P < 0.001 to P = 0.038), and there was no significant difference in IL-8 levels. The relationship between delirium and inflammatory factor levels is shown in Table 4.
| Time | Characteristic | Delirium | Non-delirium | P value |
| n | 37 | 136 | ||
| Pre | IL-1β | 1.11 (0.75, 1.54) | 1.15 (0.78, 1.56) | < 0.753 |
| IL-2 | 1.91 (1.35, 2.21) | 1.95 (1.38, 2.25) | < 0.657 | |
| IL-6 | 8.8 (7.7, 10.1) | 8.6 (7.6, 10.3) | 0.843 | |
| IL-8 | 24 (19, 28) | 20 (12, 48) | 0.579 | |
| IL-10 | 1.11 (0.88, 1.44) | 1.18 (0.90, 1.46) | 0.765 | |
| TNF-α | 1.69 (1.03, 2.54) | 1.72 (1.05, 2.50) | 0.821 | |
| Day 1 | IL-1β | 1.84 (1.44, 2.89) | 1.68 (0.99, 3.88) | 0.259 |
| IL-2 | 1.76 (1.57, 1.95) | 2.39 (1.69, 4.07) | < 0.001 | |
| IL-6 | 64 (42, 85) | 38 (22, 83) | < 0.001 | |
| IL-8 | 26 (20, 35) | 15 (9, 26) | < 0.001 | |
| IL-10 | 3.2 (2.1, 3.8) | 6.0 (3.1, 8.8) | < 0.001 | |
| TNF-α | 1.79 (1.22, 2.67) | 1.94 (1.39, 2.72) | 0.461 | |
| Day 2 | IL-1β | 2.21 (1.78, 2.71) | 2.06 (1.44, 3.89) | 0.789 |
| IL-2 | 1.36 (1.11, 1.84) | 2.48 (1.63, 3.83) | < 0.001 | |
| IL-6 | 66 (37, 87) | 63 (28, 107) | 0.770 | |
| IL-8 | 40 (31, 46) | 28 (16, 54) | 0.019 | |
| IL-10 | 2.4 (1.7, 3.7) | 5.8 (4.2, 9.3) | < 0.001 | |
| TNF-α | 2.00 (1.21, 2.84) | 2.42 (1.76, 3.44) | 0.021 | |
| Day 7 | IL-1β | 1.93 (1.13, 3.58) | 1.24 (0.93, 1.55) | < 0.001 |
| IL-2 | 1.55 (1.30, 1.80) | 2.34 (1.67, 4.22) | < 0.001 | |
| IL-6 | 22 (11, 39) | 17 (9, 32) | 0.038 | |
| IL-8 | 18 (14, 22) | 22 (11, 62) | 0.165 | |
| IL-10 | 1.89 (1.45, 2.50) | 4.18 (3.17, 6.58) | < 0.001 | |
| TNF-α | 2.30 (1.76, 3.21) | 1.99 (1.35, 2.53) | 0.003 | |
The incidence of delirium was 13.4% (11/82) in the OG and 28.6% (26/91) in the control group. The impact of the OG compared to the control group on the outcome was assessed using multivariate logistic regression analysis. In Model 1 without adjusting for any covariates, the risk of the outcome in the OG was 0.27 times that of the control group [odds ratio (OR) = 0.27, 95% confidence interval (CI): 0.15-0.51, P < 0.001]. In Model 2, adjusted for gender, age, BMI, diagnosis, and ASA grade, the association remained significant, and the effect size decreased further (OR = 0.20, 95%CI: 0.09-0.42, P < 0.001). In Model 3, which further adjusted for preoperative MoCA scale score and multiple preoperative inflammatory factors (IL-1β, IL-2, IL-6, IL-8, IL-10, TNF-α), the protective effect of the OG was most significant (OR = 0.12, 95%CI: 0.04-0.36, P < 0.001). These results indicate that Oxiracetam is an independent protective factor for the outcome, and after correcting for a series of demographic, clinical, and inflammatory indicators, its protective effect remains significant and the effect strength gradually increases. The relationship between Oxiracetam and delirium incidence is shown in Table 5.
| Characteristic | Model 1 | Model 2 | Model 3 | ||||||
| OR | 95%CI | P value | OR | 95%CI | P value | OR | 95%CI | P value | |
| Intervention | |||||||||
| Control group | - | - | - | - | - | - | |||
| Oxiracetam group | 0.27 | 0.15, 0.51 | < 0.001 | 0.20 | 0.09, 0.42 | < 0.001 | 0.12 | 0.04, 0.36 | < 0.001 |
This multicenter RCT confirms that perioperative intravenous Oxiracetam significantly reduces the incidence of POD in elderly patients undergoing laparoscopic gastrointestinal surgery. This protective effect remained independent and was even strengthened after rigorous adjustment for multiple confounders, including preoperative inflammatory status.
The pathophysiology of delirium’s may be explained by cholinergic deficiency and neuroinflammation[13]. Anticholinergic drugs precipitate delirium, and that many delirium patients have impaired ACh signaling[13]. Low ACh levels can also disinhibit inflammation, for example, cholinergic blockade enhances IL-1β, IL-6 and TNF-α release[14]. Therefore, prophylactic ACh inhibitors which raise ACh have been shown to significantly reduced POD[15]. The neuroprotextive effect of Oxiracetam may be attributed to various effects, including its indirect impact on cholinergic neurotransmission possibly by increasing the availability of choline chloride for ACh synthesis[16]. In rodent models of cholinergic hypofunction, Oxiracetam restored ACh release and uptake. In rats with reduced striatal ACh, acute Oxiracetam increased ACh levels to control levels, whereas in normal rats Oxiracetam increased ACh utilization in cortex and hippocampus. Another rodent study showed that Oxiracetam also increased glutamate and ACh release in the hippocampus[17]. Additionally, Oxiracetam has been shown to promote the biosynthesis of brain phospholipids like phosphatidyl choline and phosphatidyl ethanolamine, stabilizing the structure and function of neural cell membranes[18].
Neuroinflammation also plays a vital role in the pathophysiology of delirium, with mitochondrial and synaptic dysfunction, and neural death resulting from neuroinflammation being important mechanisms underlying delirium[13]. Our results showed that oxiracetam it reduced levels of key pro-inflammatory factors such as IL-6 and IL-8, while promoting the expression of anti-inflammatory factors such as IL-10. This pattern suggests that oxiracetam’s perioperative action may be closer to immunomodulation that just anti-inflammation. Similar findings have been shown in Alzheimer’s disease models where oxiracetam may exert a protective role by attenuating the damage caused by inflammation and oxidative stress[19], and in rodent TBI models where oxiracetam down regulated neuroinflammatory markers: COX-2, NLRP3 inflammasome, caspase-1, IL-6, TNF-α, and IL-1β expression, and upregulated antioxidant enzymes SOD1/2[12]. Another crucial finding is that the protective effect of Oxiracetam was significantly enhanced after adjusting for preoperative inflammatory factors, and the preoperative inflammatory profile in patients who developed POD (like lower levels of IL-10, IL-2) suggests an inherent vulnerability in immune regulation among high-risk patients.
As a known nootropic, Oxiracetam has cognitive enhancing effects[7]. In our study, the OG maintained cognitive function at preoperative levels, whereas the control group showed a mild decline. These results suggest that perioperative intravenous infusion of Oxiracetam can, to some extent, mitigate cognitive fluctuations in elderly patients after laparoscopic gastrointestinal surgery, exerting a protective effect on postoperative cognitive function.
This study was a multicenter RCT with a rigorous multivariate adjustment model, however, it is important to acknowledge the limitations. Firstly, POD diagnosis relied primarily on clinical scales, not objective neuropsychological assessments. Second, inflammatory markers were measured in peripheral blood and may not fully reflect central neuroinflammatory changes. Finally, the analysis of inflammatory factors was based on discrete time-point comparisons; future research with dense longitudinal sampling could delineate a more complete dynamic trajectory. Additionally, the 55% POD incidence assumed for sample size calculation was a conservative estimate. The observed 28.6% incidence in our control group aligns with reported rates in comparable high-risk surgical cohorts, supporting the generalizability of our findings and confirming adequate statistical power.
Perioperative intravenous oxiracetam safely and effectively reduces the risk of POD in elderly patients undergoing laparoscopic gastrointestinal surgery, and maintains postoperative cognitive functionality. Its protective effect likely involves multi-target mechanisms, including stabilization complex modulation of the systemic immune response, rather than dependence on simple suppression of a single inflammatory pathway. This study provides a foundation for the clinical application of oxiracetam as a perioperative brain protection strategy for elderly surgical patients. We further plan to extend our research to assess long-term follow-up data and functional prognosis.
| 1. | Rengel KF, Pandharipande PP, Hughes CG. Postoperative delirium. Presse Med. 2018;47:e53-e64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 85] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 2. | Oh ST, Park JY. Postoperative delirium. Korean J Anesthesiol. 2019;72:4-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 111] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 3. | Meagher D. Motor subtypes of delirium: past, present and future. Int Rev Psychiatry. 2009;21:59-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 4. | Igwe EO, Nealon J, O'Shaughnessy P, Bowden A, Chang HR, Ho MH, Montayre J, Montgomery A, Rolls K, Chou KR, Chen KH, Traynor V, Smerdely P. Incidence of postoperative delirium in older adults undergoing surgical procedures: A systematic literature review and meta-analysis. Worldviews Evid Based Nurs. 2023;20:220-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 50] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 5. | Yan E, Veitch M, Saripella A, Alhamdah Y, Butris N, Tang-Wai DF, Tartaglia MC, Nagappa M, Englesakis M, He D, Chung F. Association between postoperative delirium and adverse outcomes in older surgical patients: A systematic review and meta-analysis. J Clin Anesth. 2023;90:111221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 107] [Article Influence: 35.7] [Reference Citation Analysis (0)] |
| 6. | Xiao MZ, Liu CX, Zhou LG, Yang Y, Wang Y. Postoperative delirium, neuroinflammation, and influencing factors of postoperative delirium: A review. Medicine (Baltimore). 2023;102:e32991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 75] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 7. | Malík M, Tlustoš P. Nootropics as Cognitive Enhancers: Types, Dosage and Side Effects of Smart Drugs. Nutrients. 2022;14:3367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 8. | Li W, Liu H, Jiang H, Wang C, Guo Y, Sun Y, Zhao X, Xiong X, Zhang X, Zhang K, Nie Z, Pu X. (S)-Oxiracetam is the Active Ingredient in Oxiracetam that Alleviates the Cognitive Impairment Induced by Chronic Cerebral Hypoperfusion in Rats. Sci Rep. 2017;7:10052. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 9. | Ponzio F, Pozzi O, Banfi S, Dorigotti L. Brain entry and direct central pharmacological effects of the nootropic drug oxiracetam. Oxiracetam: brain entry and pharmacological effects. Pharmacopsychiatry. 1989;22 Suppl 2:111-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Mochizuki D, Sugiyama S, Shinoda Y. [Biochemical studies of oxiracetam (CT-848) on cholinergic neurons]. Nihon Yakurigaku Zasshi. 1992;99:27-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 11. | Bottini G, Vallar G, Cappa S, Monza GC, Scarpini E, Baron P, Cheldi A, Scarlato G. Oxiracetam in dementia: a double-blind, placebo-controlled study. Acta Neurol Scand. 1992;86:237-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Youn DH, Han SW, Kim JT, Choi H, Lee A, Kim N, Jung H, Hong EP, Park CH, Lee Y, Cho SM, Jeon JP. Oxiracetam alleviates anti-inflammatory activity and ameliorates cognitive impairment in the early phase of traumatic brain injury. Acta Neurochir (Wien). 2023;165:2201-2210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Fan YY, Luo RY, Wang MT, Yuan CY, Sun YY, Jing JY. Mechanisms underlying delirium in patients with critical illness. Front Aging Neurosci. 2024;16:1446523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 34] [Reference Citation Analysis (0)] |
| 14. | Saeed RW, Varma S, Peng-Nemeroff T, Sherry B, Balakhaneh D, Huston J, Tracey KJ, Al-Abed Y, Metz CN. Cholinergic stimulation blocks endothelial cell activation and leukocyte recruitment during inflammation. J Exp Med. 2005;201:1113-1123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 417] [Cited by in RCA: 387] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 15. | Pipek LZ, Pascual GS, Nascimento RFV, Silva GD, Castro LH. Acetylcholinesterase Inhibitors for Delirium Prevention: A Systematic Review and Meta-Analysis. Crit Care Med. 2025;53:e2054-e2061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 16. | Consolo S, Salmoiraghi P, Amoroso D, Kolasa K. Treatment with oxiracetam or choline restores cholinergic biochemical and pharmacological activities in striata of decorticated rats. J Neurochem. 1990;54:571-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 17. | Raiteri M, Costa R, Marchi M. Effects of oxiracetam on neurotransmitter release from rat hippocampus slices and synaptosomes. Neurosci Lett. 1992;145:109-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Lim JS, Lee J, Kang Y, Park HT, Kim DE, Cha JK, Park TH, Heo JH, Lee KB, Park JM, Oh MS, Kim EG, Chang DI, Heo SH, Park MS, Park H, Yi S, Lee YB, Park KY, Lee SJ, Kim JG, Lee J, Cho KH, Rha JH, Kim YI, Lee JH, Choi JC, Oh KM, Kwon JH, Kim C, Park JH, Jung KH, Sung SM, Chung JW, Lee YS, Kim HY, Cho HJ, Park JW, Moon WJ, Bae HJ. Efficacy and safety of oxiracetam in patients with vascular cognitive impairment: A multicenter, randomized, double-blinded, placebo-controlled, phase IV clinical trial. Contemp Clin Trials. 2023;126:107108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 19. | Zhang H, Jia L, Jia J. Oxiracetam Offers Neuroprotection by Reducing Amyloid β-Induced Microglial Activation and Inflammation in Alzheimer's Disease. Front Neurol. 2020;11:623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |