Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.117397
Revised: December 25, 2025
Accepted: February 5, 2026
Published online: April 27, 2026
Processing time: 139 Days and 19.7 Hours
In addition to gastric cancer, emerging evidence indicate that individuals treated for Helicobacter pylori (H. pylori) infection experience a significant reduction in both colorectal cancer (CRC) proportion and mortality, suggesting a potential causative role of this infection in CRC. However, in the Chinese population, it remains unclear whether H. pylori infection is involved in the early stages of colorectal carcinogenesis or is associated with the risk of colorectal polyps (CPs). Therefore, we conducted a retrospective study based on 5986 gastroscopy and colonoscopy patients without a treatment history of H. pylori in Zhejiang Province (China).
To investigate H. pylori’s association with CRC and the different types of CPs, and its potential to alter their risks.
A retrospective cross-sectional study was performed with 5986 patients who had undergone gastroscopy and colonoscopy. Pearson χ2 test was used for analyzing how H. pylori are correlated to various CPs and CRC. H. pylori as a risk factor for various CPs and CRC was determined by multivariable logistic regression.
H. pylori-positive patients demonstrated a significantly higher proportion of advanced adenomas (AAs) than negative patients (7.51% vs 5.61%; P = 0.006), with a non-significant increase in CRC proportion (1.45% vs 1.15%). No significant differences were observed for non-adenomatous polyps or low-risk adenomas, and infection levels showed no association with CRC or CPs proportion. H. pylori infection was linked to a more distal CRC location (P < 0.05) but not to AAs characteristics. Furthermore, the analysis showed an association between H. pylori infection and increased odds of AAs (odds ratio = 1.53, 95% confidence interval: 1.20-1.93) and RC (odds ratio = 1.70, 95% confidence interval: 1.01-2.80).
In a similar clinical population of Chinese, H. pylori infection may be a risk factor for both AAs and CRC but not non-adenomatous polyps or low-risk adenomas. This suggests that H. pylori-positive patients should prioritize colonoscopy.
Core Tip: In the Chinese population, it remains unclear whether Helicobacter pylori (H. pylori) infection is involved in the early stages of colorectal carcinogenesis or is associated with the risk of colorectal polyps. We conducted a retrospective study based on 5986 gastroscopy and colonoscopy patients without a treatment history of H. pylori in Zhejiang Province (China). We found that H. pylori infection was a risk factor for both advanced adenomas and colorectal cancer in a similar clinical population of Chinese, but not non-adenomatous polyps or low-risk adenomas. Therefore, H. pylori-positive patients should undergo colonoscopy.
- Citation: Shan N, Hou JJ, Jin CQ, Jin Q, Qin YH, Li WW. Helicobacter pylori positively associated with colorectal cancer and advanced, but not low-risk, adenomas: A retrospective study in China. World J Gastrointest Surg 2026; 18(4): 117397
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/117397.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.117397
As the third most diagnosed cancer and the second foremost cause of cancer mortality, colorectal cancer (CRC) represents a paramount global health burden[1]. China is experiencing rising CRC rates driven by dietary and lifestyle factors[2]. In 2022, China was estimated to have 517100 CRC cases and 240000 deaths[3]. Most CRCs evolved from colorectal polyps (CPs)[4]. Therefore, it is imperative to investigate the drivers of malignant transformation in CPs and reduce modifiable risk factors.
CPs include non-adenomatous polyps (NAPs) and adenoma polyps. Furthermore, adenoma polyps can be divided into low-risk adenomas (LRAs) and advanced adenomas (AAs), with the latter more likely to become CRC. The proportion and mortality of CRC can be effectively reduced through the screening, detection, and endoscopic resection of these precancerous lesions[5,6].
The infection of Helicobacter pylori (H. pylori) is globally evolved as a health concern related to the public, besides its recognition as the major pathogenic factor of gastric cancer[7,8]. In addition to gastric cancer, emerging evidences indicate that individuals treated for H. pylori infection experience a significant reduction in both CRC proportion and mortality, suggesting a potential causative role of this infection in CRC[9,10]. Besides modulating the host intestinal immune response, H. pylori is thought to increase CRC risk by inducing gut microbiota alterations[11]. The widespread use of the carbon-13 urea breath test has made detecting H. pylori infection straightforward and accessible. A clearer understanding of its associations with CRC and CPs will yield critical evidence necessary for devising effective preventive and therapeutic approaches against CRC. However, in the Chinese population, it remains unclear whether H. pylori infection is involved in the early stages of colorectal carcinogenesis or is associated with the risk of CPs.
Therefore, we conducted a retrospective study based on 5986 gastroscopy and colonoscopy patients without a treatment history of H. pylori in Zhejiang Province (China). Our study aimed to investigate the associations of H. pylori infection with CRC and the different types of CPs and evaluate whether H. pylori infection may alter their risks.
Owing to the retrospective-observational nature of our study, the Medical Ethics Committee of Shaoxing People’s Hospital (Approval No. IEC-K-AF-016-1.3) discussed and approved the research plan. The enrolled patients were who had gastroscopies and colonoscopies on the same day in the Endoscopy Center of the Shaoxing People’s Hospital between August 1, 2019 and May 31, 2020. The patients were included based on the following: Complete clinical data, well-prepared bowel, and gastroenterology examination. Meanwhile, the exclusion criteria were patients with inflammatory bowel disease, hereditary polyposis (e.g., familial adenomatous polyposis and Peutz-Jeghers syndrome), partial or total gastrointestinal resection (e.g., CRC and gastric cancer), other intestinal diseases, H. pylori treatment history, and no endoscopic pathology.
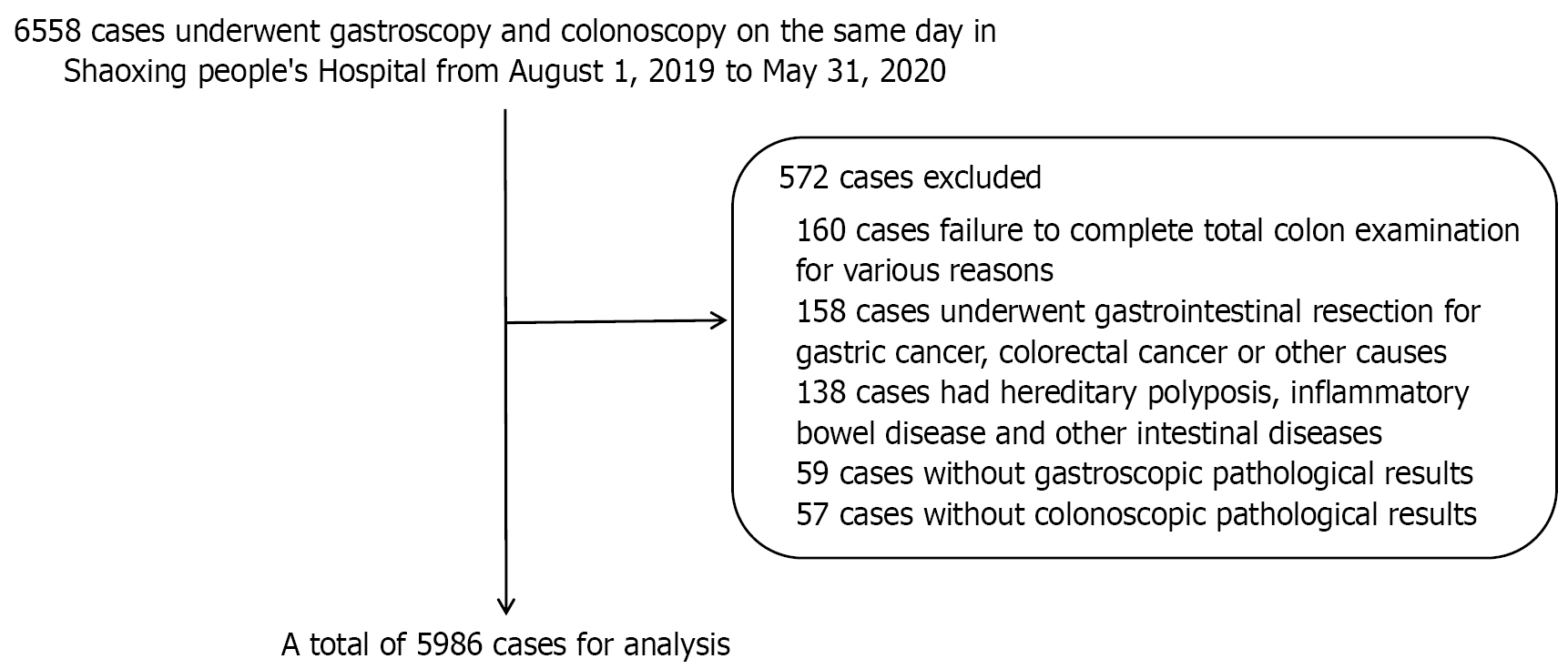
By accessing the clinical information system, image archival, and communication system, the gastrointestinal endoscopy and pathology analysis data of 6558 outpatients, inpatients, and physical examinees in the Shaoxing People’s Hospital between August 1, 2019 and May 31, 2020 were collected. According to the exclusion criteria, 5986 cases were analyzed (Figure 1). The various CPs and CRC forms were diagnosed by gastroscopy and pathological examination. In the Department of Pathology, pathological specimens were investigated and then followed by fixation with a 10% formaldehyde solution, embedding, sectioning, and staining with H. pylori staining solution for histological classification and assessment.
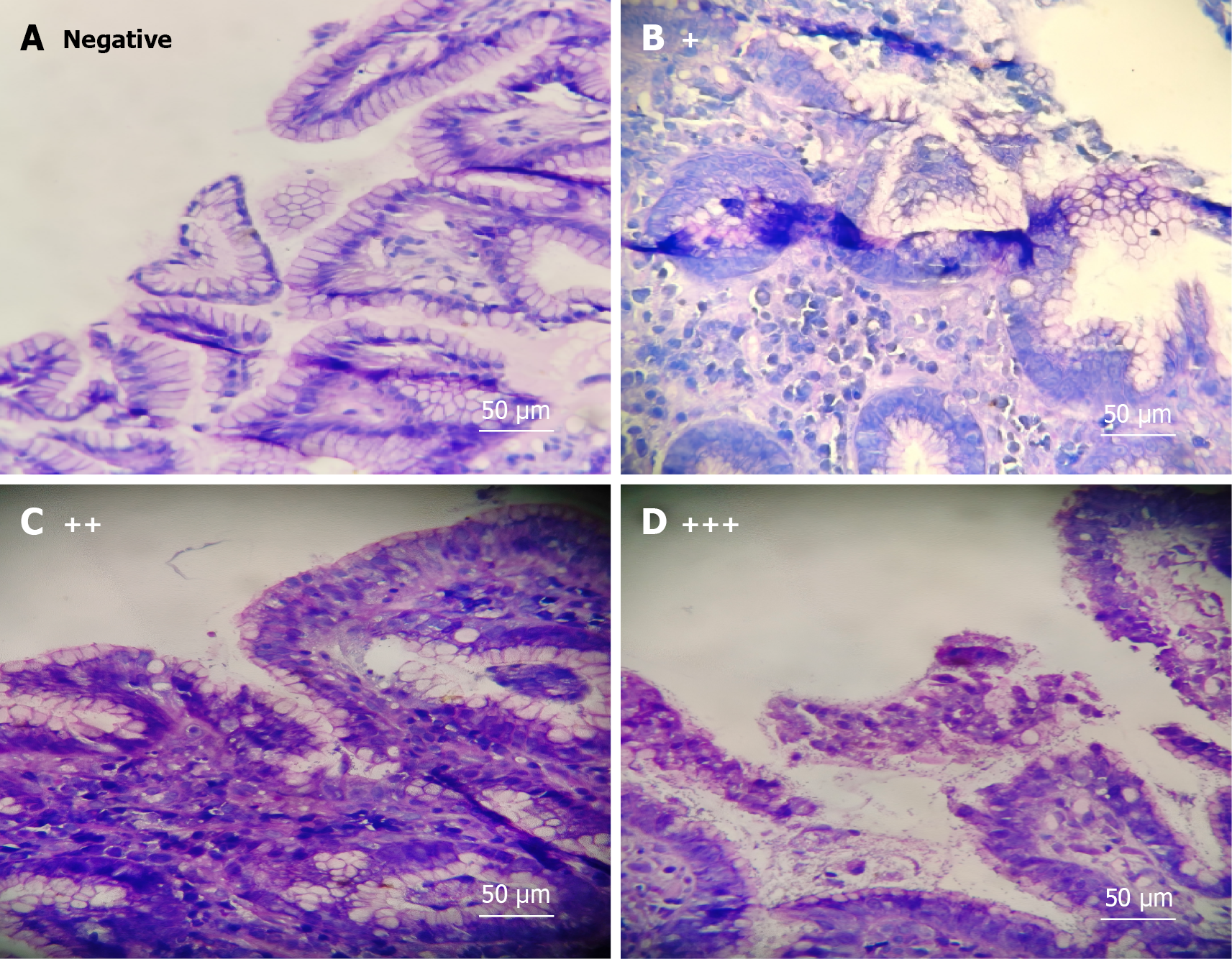
The colonoscopy results were categorized into five groups: Normal, NAPs (including proliferative and inflammatory polyps), LRAs (one or two small (< 10 mm) tubular adenomas and serrated adenoma with no high-grade dysplasia), AAs (numerous (≥ 3), large (≥ 10 mm), and/or of high-risk histology (tubulovillous or high-grade dysplastic changes), and CRC. Following Giemsa staining of gastric mucosa for H. pylori assessment, samples were categorized as either positive (including H. pylori+, H. pylori++, and H. pylori+++) or negative based on the number of bacteria per high-power microscope field. The histological grading criteria and representative high-power field (× 400 magnification) images were shown in Figure 2. To assess the accuracy and consistency of H. pylori status classification, two pathologists performed the histopathological evaluation independently. In cases of discrepant results, the final decision was made by the more senior pathologist. The specific category referred to the Consensus Opinions on Chronic Gastritis in China (2012, Shanghai, China). The general information on AAs and CRC, including the number (single and multiple) and site (distal, proximal, and multiple), underwent a retrospective analysis. Regarding the site, the distal end refers to the left colon and rectum, the proximal end refers to the right colon and multiple polyps refer to both proximal and distal.
The R language (version 4.0.4) was utilized to analyze and visualize the data. The dplyr and compareGroups packages were conducted to process the data and statistics. By using the ggplot2 package, the data was visualized, while using the sjlabelled, ggpmisc, and forest packages, both the R2 adjustment and regression equation were calculated, and forest figures were drawn. The pROC package was utilized to plot the receiver operating characteristic curve and calculate the area under the curve (AUC). Representing the categorical variables were statistically reported as proportions and compared by Fisher’s exact probability method or χ2 test, while reported the normally distributed continuous variables as means ± SD with the t-test. The expression of the enumeration data was reported as n (%), and by using the χ2 statistical test, the two groups were compared. Multivariable logistic regression was utilized for the determination of whether H. pylori could be a risk factor for various CPs and CRC types, reporting the findings as adjusted odds ratio (OR) with a corresponding 95% confidence interval (CI). By setting P < 0.05, a statistic significant difference was indicated. The statistical methods of this study were reviewed by Li WW from Shaoxing People’s Hospital, The First Affiliated Hospital of Shaoxing University.
Among the 5986 cases, 4334 cases gave H. pylori-negative, while 1652 cases gave positive. The rate of H. pylori infection was 27.6%. The proportion in males was 29.07%, and in females was 26.34%. The results revealed a more elevated H. pylori infection rate in males than females (P < 0.05). The average age of H. pylori-negative cases (52.3 ± 10.8) was older than H. pylori-positive cases (51.1 ± 10.5; P < 0.05). Group-based analysis by age revealed a higher H. pylori infection rate in patients under 40 years old (29.48%) compared to those aged 40-60 years (28.75%) and those over 60 years old (23.15%), with the difference being statistically significant (P < 0.05). Hence, H. pylori infection might be associated with the selection and region of cases (Table 1).
| H. pylori negative | H. pylori positive | H. pylori positive rate (%) | Test statistic | P value | |
| Total | 4334 | 1652 | 27.60 | ||
| Sex | 5.601 | 0.020 | |||
| Male | 1957 (45.15) | 802 (48.55) | 29.07 | ||
| Female | 2377 (54.85) | 850 (51.45) | 26.34 | ||
| Average age | 52.3 ± 10.8 | 51.1 ± 10.5 | 3.920 | < 0.001 | |
| Different ages | 17.220 | < 0.001 | |||
| < 40 | 579 (13.36) | 242 (14.65) | 29.48 | ||
| 40-60 | 2726 (62.90) | 1100 (66.59) | 28.75 | ||
| > 60 | 1029 (23.74) | 310 (18.76) | 23.15 |
Amongst the 5986 cases, 3707 cases were normal, 689 cases were NAPs, 1149 cases were LRAs, 367 cases were AAs, and 74 cases were CRC. The proportion of normal females (59.24%) was higher than normal males (40.76%). On the contrary, in NAPs (52.54%; 47.46%), LRAs (53.61%; 46.39%), AAs (62.67%; 37.33%), and CRC (54.05%; 45.95%), the proportion of males was more elevated than females (P < 0.05). Regarding the average age, normal subjects were the youngest (49.9 ± 10.9), while the CRC (62.3 ± 10.4) was older than NAPs (53.3 ± 9.5), LRAs (55.2 ± 9.14), and AAs (57.9 ± 9.23; P < 0.05). Different types of CPs and CRC were also distributed differently at different ages. The proportion of normal (18.6%) under the age of 40 was the highest, while the proportion of AAs (40.6%) and CRC (63.5%) in patients significantly increased over the age of 60 (P < 0.05; Table 2).
| Normal | NAPs | LRAs | AAs | CRC | P value | |
| Total | 3707 | 689 | 1149 | 367 | 74 | |
| Sex | < 0.05 | |||||
| Male | 1511 (40.76) | 362 (52.54) | 616 (53.61) | 230 (62.67) | 40 (54.05) | |
| Female | 2196 (59.24) | 327 (47.46) | 533 (46.39) | 137 (37.33) | 34 (45.95) | |
| Average age | 49.9 ± 10.9 | 53.3 ± 9.50 | 55.2 ± 9.14 | 57.9 ± 9.23 | 62.3 ± 10.4 | < 0.001 |
| Different ages | < 0.001 | |||||
| < 40 | 689 (18.6) | 57 (8.27) | 56 (4.87) | 17 (4.63) | 2 (2.70) | |
| 40-60 | 2378 (64.1) | 472 (68.5) | 750 (65.3) | 201 (54.8) | 25 (33.8) | |
| > 60 | 640 (17.3) | 160 (23.2) | 343 (29.9) | 149 (40.6) | 4 7(63.5) |
The proportion of various CPs and CRC differed between the two H. pylori infection groups. The proportion of NAPs was slightly higher in the H. pylori-negative group (11.61%) than in the H. pylori-positive group (11.26%). Conversely, the proportion of LRAs was somewhat lower in the H. pylori-negative group (18.90%) compared to the H. pylori-positive group (19.98%). However, none of these differences reached statistical significance (P > 0.05). The proportion of AAs in the H. pylori-positive group (7.51%) was significantly higher than that in the H. pylori-negative group (5.61%; P = 0.006). A similar trend was observed for CRC proportion, which was 1.45% in the positive group compared to 1.15% in the negative group. No indicated statistical difference (P > 0.05) between the groups, which might be due to the small number of CRC cases (Table 3).
| H. pylori negative | H. pylori positive | P value | |
| Total | 4334 | 1652 | |
| Normal | 2719 (62.73) | 988 (59.81) | 0.037 |
| NAPs | 503 (11.61) | 186 (11.26) | 0.707 |
| LRAs | 819 (18.90) | 330 (19.98) | 0.344 |
| AAs | 243 (5.61) | 124 (7.51) | 0.006 |
| CRC | 50 (1.15) | 24 (1.45) | 0.349 |
Utilizing the multivariable logistic regression for analyzing whether H. pylori is a risk factor for different types of CPs and CRC: Sex, age, and H. pylori were included for multivariable logistic regression analysis. As compared with the normal group, the OR value of H. pylori with NAPs and LRAs were 1.03 (95%CI: 0.86-1.24) and 1.15 (95%CI: 0.99-1.34), indicating that H. pylori infection was not correlated to NAPs and LRAs occurrence (P > 0.05). The OR value of H. pylori with AAs and CRC were 1.53 (95%CI: 1.20-1.93; P < 0.001) and 1.65 (95%CI: 0.98-2.71), revealing a statistically significant association of H. pylori infection with the occurrence of AAs and CRC. The P value was 0.051 in CRC, which could be due to the small number (Table 4).
| H. pylori negative | H. pylori positive | Odds ratio | 95%CI | P value | |
| Normal | 2719 (73.35) | 988 (26.65) | 1 | ||
| NAPs | 503 (73.00) | 186 (27.00) | 1.03 | 0.86-1.24 | 0.72 |
| LRAs | 819 (71.28) | 330 (28.72) | 1.15 | 0.99-1.34 | 0.06 |
| AAs | 243 (66.21) | 124 (33.79) | 1.53 | 1.20-1.93 | < 0.001 |
| CRC | 50 (67.57) | 24 (32.43) | 1.65 | 0.98-2.71 | 0.051 |
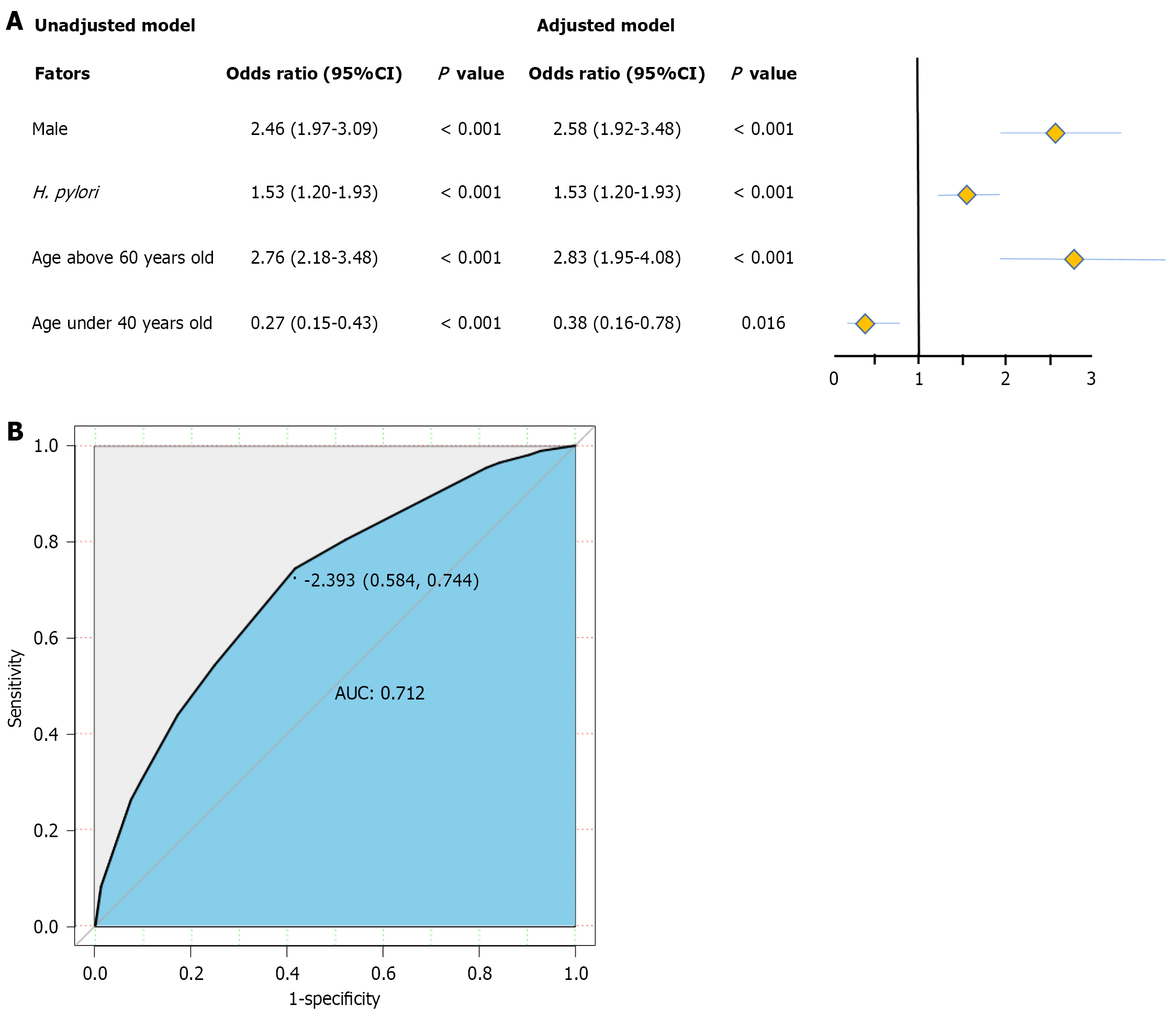
Multivariable logistic regression for AAs: Multivariable logistic regression of the unadjusted and adjusted (age and sex) models yielded the same results. Males (adjusted model: OR = 2.58, 95%CI: 1.92-3.48, P < 0.001), H. pylori infection (adjusted model: OR = 1.53, 95%CI: 1.20-1.93, P < 0.001), and old age (adjusted model: OR = 2.83, 95%CI: 1.95-4.08, P < 0.001) were AAs risk factors (Figure 3A). The AUC was 0.712 (95%CI: 0.687-0.741; Figure 3B).
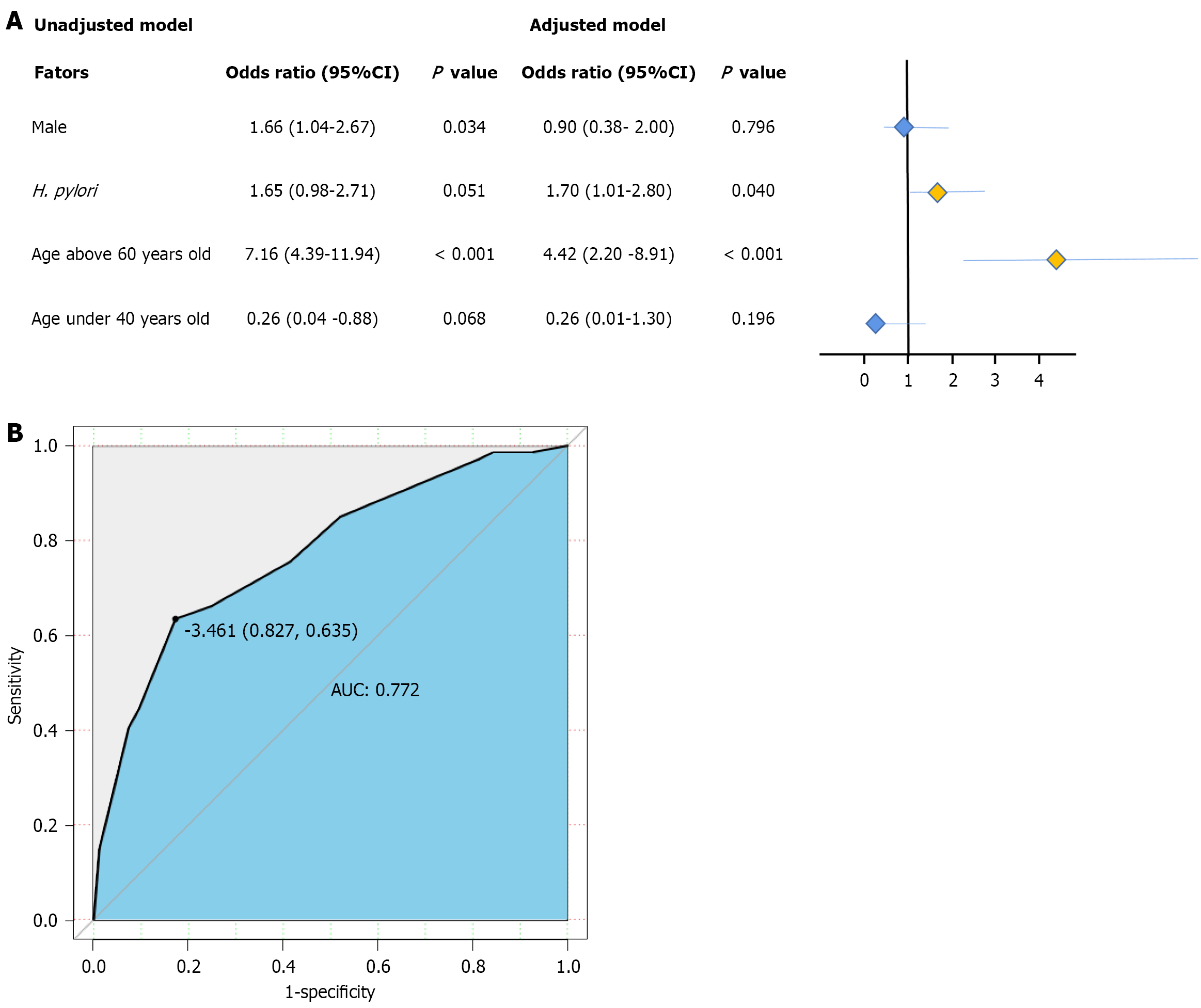
Multivariable logistic regression for CRC: The P value was 0.051 for H. pylori infection in the unadjusted model. In the adjusted model, H. pylori infection (adjusted model: OR = 1.70, 95%CI: 1.01-2.80, P < 0.05) and old age (adjusted model: OR = 4.42, 95%CI: 2.20-8.91, P < 0.001) were CRC risk factors (Figure 4A). The AUC was 0.772 (95%CI: 0.725-0.836; Figure 4B).
According to the H. pylori infection levels, 1652 H. pylori-positive cases were categorized into H. pylori+ (1034 cases), H. pylori++ (557 cases), and H. pylori+++ (61 cases) groups. The proportion of AAs (9.84%) and CRC (3.28%) in the H. pylori+++ group showed more elevation than in the H. pylori++ (7.00%, 1.62%) and H. pylori+ (7.64%, 1.26%) groups, but no significant difference was found (P > 0.05). The results were similar for NAPs and LRAs (P > 0.05; Table 5).
| H. pylori + | H. pylori ++ | H. pylori +++ | P value | |
| Total | 1034 | 557 | 61 | |
| Colonoscopy results | ||||
| Normal | 612 (59.19) | 342 (61.40) | 34 (55.74) | 0.556 |
| NAPs | 121 (11.70) | 60 (10.77) | 5 (8.20) | 0.635 |
| LRAs | 209 (20.21) | 107 (19.21) | 14 (22.95) | 0.749 |
| AAs | 79 (7.64) | 39 (7.00) | 6 (9.84) | 0.702 |
| CRC | 13 (1.26) | 9 (1.62) | 2 (3.28) | 0.406 |
The association between H. pylori and the site and the number of AAs: H. pylori-positive patients exhibited a more distal distribution (43.55%) and multiple in number (64.52%) when compared with H. pylori-negative patients (41.98%; 58.44%), but there were no significant differences (P > 0.05; Table 6).
| H. pylori negative | H. pylori positive | P value | |
| Total | 243 | 124 | |
| Site | 0.811 | ||
| Distal | 102 (41.98) | 54 (43.55) | |
| Proximal | 50 (20.58) | 22 (17.74) | |
| Multiple | 91 (37.45) | 48 (38.71) | |
| Number | 0.260 | ||
| Single | 101 (41.56) | 44 (35.48) | |
| Multiple | 142 (58.44) | 80 (64.52) |
The association between H. pylori and the site of CRC: H. pylori-positive patients exhibited a significantly more distal distribution (91.67%) than H. pylori-negative patients (68%; P < 0.05; Table 7).
| H. pylori negative | H. pylori positive | P value | |
| Total | 50 | 24 | |
| Site | 0.026 | ||
| Distal | 34 (68.00) | 22 (91.67) | |
| Proximal | 16 (32.00) | 2 (8.3) |
Based on the retrospective cross-sectional study, we found that the rate of H. pylori infection was 27.6%, lower than reported in the Chinese literature[12], possibly due to the good economic conditions and high awareness of health in the study population. H. pylori-positive patients had a higher proportion of AAs (7.51% vs 5.61%; P = 0.006) and CRC (1.45% vs 1.15%; P > 0.05) than H. pylori-negative patients. The proportions of NAPs and LRAs in the two groups were similar (P > 0.05). No significant association was found between H. pylori infection levels and the proportion of various CPs and CRC (P > 0.05). Specifically, H. pylori infection was not correlated with the site (43.55% vs 41.98%) or number (64.52% vs 58.44%) of AAs (P > 0.05). However, in CRC, the lesions were distributed more distally in H. pylori-positive patients than in negative patients (91.67% vs 68%, P < 0.05). Furthermore, multivariable logistic regression revealed that H. pylori may be an AAs (OR = 1.53, 95%CI: 1.20-1.93) and CRC (OR = 1.70, 95%CI: 1.01-2.80) risk factor.
Prevalence and death of CRC are rising both in China and worldwide[13], with the majority evolving from adenomatous CPs. Most previous studies have focused on the effects of H. pylori infection on CRC risk, but lack analysis on precancerous lesions[11,14,15]. AAs, the most important precancerous lesions of CRC, have not been significantly reported based in China. Emerging evidence indicates that H. pylori infection may contribute to hypergastrinemia and colorectal mucosal proliferation through mechanisms such as direct mucosal stimulation, gut microbiome dysbiosis, and alterations in microbial composition, potentially facilitating the development of CPs[16]. However, there is still no consensus on whether H. pylori infection is independently associated with CPs or whether this association varies by the histological type of the polyp. Therefore, a retrospective study founded on 5986 cases in China was performed for determining if H. pylori could be a risk factor for different types of CPs and CRC and the need for colonoscopy in H. pylori-positive patients. Given that most CRCs develop through the multi-stage adenoma-carcinoma sequence, we conducted a detailed analysis of all types of CPs and rule out the effect of H. pylori infection intensity. In our results, H. pylori infection was not related to NAPs and LRAs but associated with AAs and CRC, and H. pylori infection may function as an early “promoter” in the carcinogenesis process of AAs.
H. pylori have been recognized as a class I carcinogen for gastric cancer, besides its recognition as the major pathogenic factor of atrophic gastritis, and peptic ulcer[7,8,17]. Moreover, H. pylori take part in the carcinogenesis of extragastric target organs, including hepatocellular carcinoma, lung cancer, and neurological diseases[18]. The mechanism underlying the promotion of AAs development and carcinogenesis by H. pylori warrants further investigation. Previous studies described the possible mechanisms as follows. H. pylori is a major immunostimulant. Ralser et al[14] discovered that in the Apc mutant mouse model H. pylori infection recruited CD3+ T cells within the colon tissue epithelium, upregulated CD8+ T cells, and caused Treg cell loss and differentiation into potentially pathogenic forkhead box P3+ Interleukin-17A+ T cells, resulting in a specific pro-inflammatory immune response[19]. Similar immune and epithelial alterations were found in human colon biopsies from H. pylori-infected patients[14]. These pro-inflammatory factors may create a pro-carcinogenic microenvironment. Secondly, gastric atrophy produced by H. pylori-related gastritis results in hypergastrinemia following hypochlorhydria[20], and it has been discovered that gastrin receptors are overexpressed in CPs and CRC[21]. Gastrin has the capability to both stimulate the colonic mucosal cell to proliferate, promoting CRC development[22], and also acts on the gastrointestinal epithelial cells and promote cyclooxygenase-2 formation, affecting the colorectal neoplasia ability to occur, develop, invade, and proliferate[23].
These findings propose a mechanism whereby H. pylori contribute to the development of CPs and CRC through the alteration of gastrin levels. In the same way, the alteration of the intestinal microbiome has been demonstrated with H. pylori infection, including clostridia and anaerobes[24].
Our strength in this study was that histopathological findings were used to determine H. pylori infection status, which displayed high specificity and sensitivity. Based on both endoscopic and pathological results, classifying CPs was performed in detail following the progression risk to CRC, which was significantly beneficial in clinical practice. But there were some constraints in our study. First, this study adopted a single-center retrospective cross-sectional design, which inherently limited its ability to establish causal relationships or temporality and control for confounding factors. Second, the population who underwent both gastroscopy and colonoscopy on the same day was likely to have underlying upper gastrointestinal symptoms, health check-up indications, or other relevant medical histories, which may result in a higher prevalence of H. pylori infection and a greater detection rate of colorectal lesions compared to the general population. Third, the probable effects of the H. pylori infection duration on CPs and CRC were not considered due to the difficulty to be tracking. Prospective multicenter cohort studies comprising larger sample sizes should be performed with an additional assessment to determine the ability of H. pylori eradication to reduce CPs and CRC occurrence for a better understanding of the molecular mechanisms. Fourth, the multivariable logistic regression model was not adjusted for potential confounding factors such as body mass index, smoking, alcohol intake, family history, metabolic factors, and non-steroidal anti-inflammatory drugs use. In addition, the prediction model was not subjected to calibration assessment, which may have influenced the results.
Overall, the study provides valuable insights into the association between H. pylori infection and the risk of CPs and CRC, highlighting the importance of early colonoscopy in H. pylori-positive patients to prevent CRC occurrence. However, further studies are needed to confirm the results with a larger sample size of CRC and to evaluate the mechanisms underlying the association between H. pylori infection and AAs.
In a similar clinical population of Chinese, H. pylori infection may be a risk factor for both AAs and CRC but not NAPs or LRAs. This suggests that H. pylori-positive patients should prioritize colonoscopy.
We would particularly like to acknowledge my team members, for their wonderful collaboration and patient support.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14537] [Article Influence: 7268.5] [Reference Citation Analysis (17)] |
| 2. | Matsuda T, Fujimoto A, Igarashi Y. Colorectal Cancer: Epidemiology, Risk Factors, and Public Health Strategies. Digestion. 2025;106:91-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 85] [Article Influence: 85.0] [Reference Citation Analysis (0)] |
| 3. | Han B, Zheng R, Zeng H, Wang S, Sun K, Chen R, Li L, Wei W, He J. Cancer incidence and mortality in China, 2022. J Natl Cancer Cent. 2024;4:47-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1395] [Cited by in RCA: 1400] [Article Influence: 700.0] [Reference Citation Analysis (1)] |
| 4. | Sun M, Sun M, Zhang L, Shi S. Colorectal polyp risk is linked to an elevated level of homocysteine. Biosci Rep. 2018;38:BSR20171699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 5. | Forbes N. Outcomes Associated With Colorectal Cancer After Population-Based Colonoscopy Screening: Results From a European Pragmatic Randomized Trial. Gastroenterology. 2023;164:493-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Mehta SJ, Morris AM, Kupfer SS. Colorectal Cancer Screening Starting at Age 45 Years-Ensuring Benefits Are Realized by All. JAMA Netw Open. 2021;4:e2112593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 7. | Peleteiro B, Bastos A, Ferro A, Lunet N. Prevalence of Helicobacter pylori infection worldwide: a systematic review of studies with national coverage. Dig Dis Sci. 2014;59:1698-1709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 228] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 8. | Nolen LD, Vindigni SM, Parsonnet J; Symposium leaders. Combating Gastric Cancer in Alaska Native People: An Expert and Community Symposium. Gastroenterology. 2020;158:1197-1201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 9. | Frías-Ordoñez JS, Riquelme A, Marulanda-Fernandez H, Otero-Parra L, Urrego JA, Otero-Ramos E, Portillo-Miño JD, Regino WO. Helicobacter pylori and Colorectal Cancer: Meeting Sir Austin Bradford Hill's Causality Criteria. Helicobacter. 2025;30:e70024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | Kuo YC, Ko HJ, Yu LY, Shih SC, Wang HY, Lin YC, Hu KC. Kill Two Birds with One Stone? The Effect of Helicobacter pylori Eradication in Decreased Prevalence of Gastric Cancer and Colorectal Cancer. Cancers (Basel). 2024;16:3881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 11. | Engelsberger V, Gerhard M, Mejías-Luque R. Effects of Helicobacter pylori infection on intestinal microbiota, immunity and colorectal cancer risk. Front Cell Infect Microbiol. 2024;14:1339750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 55] [Reference Citation Analysis (0)] |
| 12. | Hooi JKY, Lai WY, Ng WK, Suen MMY, Underwood FE, Tanyingoh D, Malfertheiner P, Graham DY, Wong VWS, Wu JCY, Chan FKL, Sung JJY, Kaplan GG, Ng SC. Global Prevalence of Helicobacter pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology. 2017;153:420-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2676] [Cited by in RCA: 2250] [Article Influence: 250.0] [Reference Citation Analysis (6)] |
| 13. | Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71:7-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12117] [Cited by in RCA: 12179] [Article Influence: 2435.8] [Reference Citation Analysis (4)] |
| 14. | Ralser A, Dietl A, Jarosch S, Engelsberger V, Wanisch A, Janssen KP, Middelhoff M, Vieth M, Quante M, Haller D, Busch DH, Deng L, Mejías-Luque R, Gerhard M. Helicobacter pylori promotes colorectal carcinogenesis by deregulating intestinal immunity and inducing a mucus-degrading microbiota signature. Gut. 2023;72:1258-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 97] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 15. | Shah SC, Camargo MC, Lamm M, Bustamante R, Roumie CL, Wilson O, Halvorson AE, Greevy R, Liu L, Gupta S, Demb J. Impact of Helicobacter pylori Infection and Treatment on Colorectal Cancer in a Large, Nationwide Cohort. J Clin Oncol. 2024;42:1881-1889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 16. | Lu D, Wang M, Ke X, Wang Q, Wang J, Li D, Wang M, Wang Q. Association Between H. pylori Infection and Colorectal Polyps: A Meta-Analysis of Observational Studies. Front Med (Lausanne). 2021;8:706036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 17. | Ansari S, Yamaoka Y. Current understanding and management of Helicobacter pylori infection: an updated appraisal. F1000Res. 2018;7:F1000 Faculty Rev-F1000 Faculty 721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 18. | Franceschi F, Covino M, Roubaud Baudron C. Review: Helicobacter pylori and extragastric diseases. Helicobacter. 2019;24 Suppl 1:e12636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 19. | Yang S, Wang B, Guan C, Wu B, Cai C, Wang M, Zhang B, Liu T, Yang P. Foxp3+IL-17+ T cells promote development of cancer-initiating cells in colorectal cancer. J Leukoc Biol. 2011;89:85-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 116] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 20. | Duan S, Rico K, Merchant JL. Gastrin: From Physiology to Gastrointestinal Malignancies. Function (Oxf). 2022;3:zqab062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 21. | Smith AM, Watson SA. Gastrin and gastrin receptor activation: an early event in the adenoma-carcinoma sequence. Gut. 2000;47:820-824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 3.3] [Reference Citation Analysis (6)] |
| 22. | Thorburn CM, Friedman GD, Dickinson CJ, Vogelman JH, Orentreich N, Parsonnet J. Gastrin and colorectal cancer: a prospective study. Gastroenterology. 1998;115:275-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 204] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 23. | Chao C, Hellmich MR. Gastrin, inflammation, and carcinogenesis. Curr Opin Endocrinol Diabetes Obes. 2010;17:33-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (6)] |
| 24. | Myllyluoma E, Ahlroos T, Veijola L, Rautelin H, Tynkkynen S, Korpela R. Effects of anti-Helicobacter pylori treatment and probiotic supplementation on intestinal microbiota. Int J Antimicrob Agents. 2007;29:66-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 64] [Article Influence: 3.2] [Reference Citation Analysis (1)] |