Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.117318
Revised: January 20, 2026
Accepted: February 11, 2026
Published online: April 27, 2026
Processing time: 136 Days and 9 Hours
Prolonged postoperative ileus (PPOI) is a common post-surgery iatrogenic complication, with an incidence rate of 10%-28%. Severe PPOI delays postoperative recovery, prolongs hospital stay, and further increases healthcare costs. The pathogenesis of PPOI is extremely complex. The persistent and excessive immune-inflammatory response is currently considered to be the main pathological mechanism of PPOI. Gut vascular barrier (GVB) damage and the resulting vascular inflammatory response are two key factors responsible for sustaining persistent intestinal inflammation in PPOI. This article comprehensively reviews the effects of intestinal microvascular endothelial cell (IMVEC) dysfunction, increased GVB permeability, pathological angiogenesis, and intestinal microvascular hemodynamic alterations on PPOI. Our review indicates that these GVB-related mechanisms collectively exacerbate intestinal inflammation and dysmotility, contributing to the persistence of PPOI. It also discusses the potential of improving IMVEC function, reducing GVB permeability, inhibiting pathological angiogenesis, and ameliorating intestinal microcirculation for preventing and treating PPOI. In conclusion, targeting GVB dysfunction and associated vascular pathology represents a promising therapeutic strategy. Thus, we propose that restoring the impaired GVB may serve as a potential target for therapeutic intervention in PPOI.
Core Tip: Prolonged postoperative ileus (PPOI) is a common iatrogenic complication following surgery, with an incidence rate ranging from 10% to 28%. Damage to the gut vascular barrier (GVB) and the subsequent vascular inflammatory response are key factors that contribute to the persistence of intestinal inflammation in PPOI. This review comprehensively examines the impact of intestinal microvascular endothelial cell dysfunction, increased GVB permeability, pathological angiogenesis, and alterations in intestinal microvascular hemodynamics on PPOI. It suggests that restoring the impaired GVB may present a promising therapeutic target for managing PPOI.
- Citation: Shen DL, Wan L, Zhang XC, Fang YD, Jiang HC, Wei YC, Chen LF, Wu ZM, Ye CC, Pei C, Zhou H, Qian L. Repairing the impaired gut vascular barrier as a novel therapeutic target for prolonged postoperative ileus: A scoping review. World J Gastrointest Surg 2026; 18(4): 117318
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/117318.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.117318
Prolonged postoperative ileus (PPOI) is a temporary gastrointestinal motility disorder caused by non-mechanical factors after surgery. The incidence rate of PPOI is as high as 10%-28%[1]. PPOI not only prolongs the postoperative hospital stay but also increases the postoperative mortality rate and total medical costs[2]. In 2011, the total annual expenditure on PPOI in the United States was up to 12.3 billion USD[3]. Despite this, the pathophysiological mechanisms of PPOI have not yet been fully elucidated. Current studies suggest that the immune-inflammatory response following intestinal barrier damage is a key factor in the pathogenesis of PPOI[4,5]. Therefore, improving intestinal barrier permeability has been proposed as a novel therapeutic target for intervention in PPOI[5].
The gut vascular barrier (GVB) was first reported by Spadoni et al[6] in the intestinal subepithelial layer of mice infected with Salmonella typhimurium in 2015. It has been identified as a novel barrier, distinct from the conventional microbial, chemical, physical, and immune barriers[6,7]. The GVB exhibits selective permeability: Under normal physio
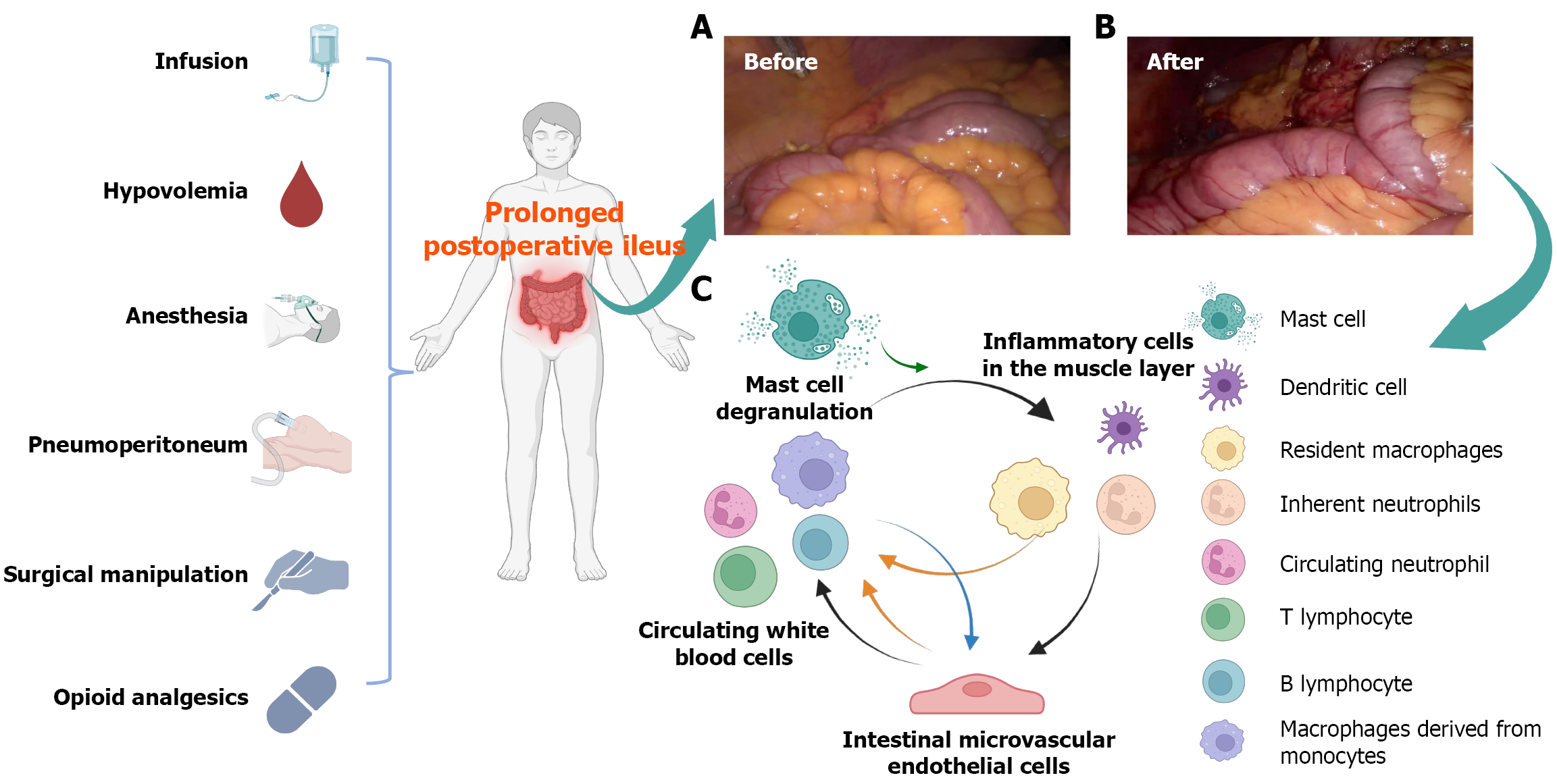
Anesthesia, pneumoperitoneum, surgical manipulation, massive hemorrhage, large-volume fluid administration, and use of opioid analgesics are the main inducing factors of PPOI (Figure 1). The fundamental pathological mechanism of PPOI involves abnormal neuronal signaling caused by inflammatory cell infiltration into the muscular layer following intestinal barrier injury[14]. Prolonged intravenous anesthesia and excessive use of opioids lead to the systemic nonselective activation of mu-opioid receptors in the central nervous system, which results in the inhibition of intestinal peristalsis[15,16]. Carbon dioxide (CO2) is the most commonly employed gas for establishing pneumoperitoneum. Research indicates that CO2 suppresses the infiltration of anti-inflammatory macrophages into the intestinal muscular layer by modulating their intracellular pH via carbonic anhydrases[17]. Additionally, CO2 potentiates intestinal inflammation by promoting the release of immunoglobulin A from immunoinflammatory cells[18]. Perioperative fluid overload activates the renin-angiotensin-aldosterone system, resulting in intestinal tissue edema. This edema elevates intra-abdominal pressure, reduces mesenteric blood flow, and induces tissue ischemia and hypoxia, thereby exacerbating intestinal inflammation[19]. Massive hemorrhage induces circulatory hypovolemia and the consequent ischemic hypoxia, thereby intensifying existing intestinal inflammation. Surgical manipulation and injury release an enormous amount of inflammatory factors, chemokines, and damage-associated molecular patterns[20,21]. These cytokines promote mast cell degranulation by inhibiting the transient receptor potential ankyrin 1-mediated cyclic AMP response element-binding protein/WNT1-inducible signaling protein 1 signaling pathway[4]. Substances [including chymase, histamine, and interleukin (IL)] released during mast cell degranulation compromise the intestinal epithelial barrier and simultaneously activate inflammatory cells[20,22]. Dendritic cells exert their antigen-presenting function following activation by CCR7[21]. Orchestrated by the gut microbiota, they activate muscularis resident macrophages and neutrophils[23]. Dendritic cells additionally induce the migration of T helper type 1 memory cells to intestinal regions remote from the surgical site, where they trigger gut hypomotility[24]. The binding of damage-associated molecular patterns to the purinergic 2X7 receptor activates macrophages[25]. The activated macrophages trigger inflammatory cascades through various signaling pathways, including toll like receptors (TLR), nuclear factor kappa-B (NF-κB)/mitogen-activated protein kinases (MAPK), phosphatidylinositol 3 kinase/protein kinase B (Akt)/NF-κB, and Janus kinase 2/signal transducer and activator of transcription 3[26-29]. IL-17A released by macrophages induces monocytes and neutrophils to express nitric oxide (NO), resulting in the relaxation of the intestinal circular muscle[30]. C-X-C motif chemokine ligand 1 secreted by macrophages can directly inhibit the intestinal contractile activity[31]. Activated neutrophils infiltrate the muscular layer and exacerbate intestinal inflammation by releasing reactive oxygen species (ROS) and neutrophil extracellular traps[32,33]. Granzyme B, interferon-γ, and tumor necrosis factor-α (TNF-α) produced by T lymphocytes activate the inflammatory phenotype in enteric glial cells[34,35]. These inflammatory signals are transmitted via visceral afferent nerves to the spinal cord, activating the sympathetic nervous system, and are subsequently relayed through efferent nerves to the intestinal muscular layer[36]. Additionally, severe inflammatory stimuli can ascend via spinal nerves to the brainstem, activating the hypothalamic and pontine nuclei, which suppress vagal nerve activity and further delay intestinal peristalsis[36].
Recent studies have established that intestinal microvascular endothelial cells (IMVECs), a critical component of the intestinal barrier, actively participate in the occurrence and development of PPOI by mediating vascular inflammatory responses (Figure 1). Ischemia-reperfusion injury signaling activates the focal adhesion kinase pathway, promoting the generation of oxygen-free radicals in endothelial cells and reducing the expression of vascular endothelial-cadherin (VE-cadherin) and β-catenin between endothelial cells[37]. Damaged endothelial cells highly express intercellular adhesion molecule-1 (ICAM-1), transforming growth factor-β, insulin-like growth factor-1, and insulin-like growth factor-1 receptor and promote the infiltration of inflammatory cells into the muscular layer[38]. Under hemodynamic dis
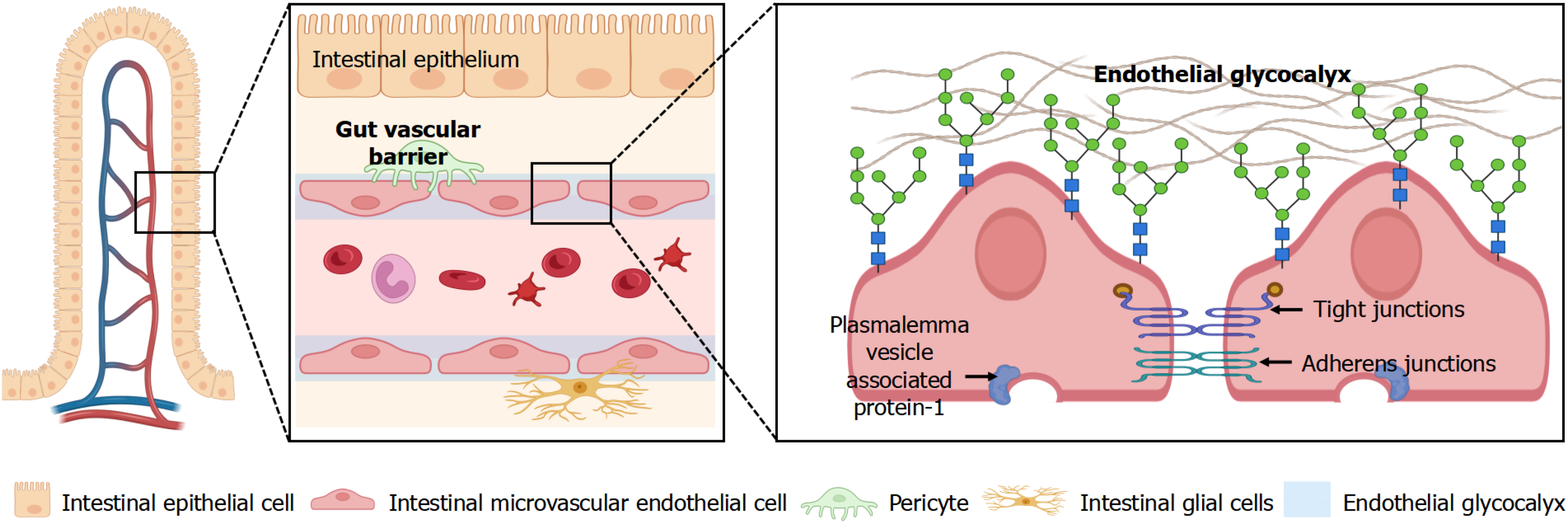
The GVB is present underneath the intestinal epithelium. It is primarily composed of a monolayer of IMVEC and is supported by enteric glial cells and pericytes[6] (Figure 2). IMVECs are connected through TJ and adherens junction (AJ). The TJ is composed of occludin, zonula occludens-1 (ZO-1), cingulin, and junctional adhesion molecule-A. The AJ comprises VE-cadherin and β-catenin[6]. CD31 and vWF are characteristic phenotypic markers of IMVECs. PV-1 is a distinctive protein of IMVECs[6,47]. Mature IMVECs have abundant vesicles, microfilament bundles, and Weibel-Palade bodies in the cytoplasm and exhibit tube-forming ability[48]. Under physiological conditions, 4-kD fluorescein isothiocyanate (FITC) dextran can freely pass through the GVB, whereas ≥ 70-kD FITC-dextran, bacteria, and cells cannot[6]. This size-selective property is critical because it prevents translocation of bacteria and large macromolecules from the gut lumen into the circulation, thereby protecting the host from systemic endotoxemia and excessive immune activation.
The canonical Wnt/β-catenin signaling pathway plays a crucial role in maintaining the structural and functional maturity of the endothelial barrier[49]. Following the activation of the Wnt/β-catenin signaling pathway, β-catenin activity and Axin2 expression are upregulated[50]. This effect increases the expression of TJ and AJ while simultaneously reducing that of PV-1, thereby maintaining the structural and functional integrity of the GVB[50]. The VEGF/NOTCH pathway regulates the normal structure and function of the intestinal vasculature, though additional studies are required[51]. S100B, a cytosolic calcium-binding protein secreted by enteric glial cells, regulates the expression of PV-1, β-catenin, and occludin in IMVECs through the S100B/ADAM10 and S100B/caspase-8/β-catenin signaling pathways[52,53]. Pericytes play a crucial role in maintaining the endothelial barrier function. Angiopoietin-1 secreted by pericytes can reduce the permeability of the intestinal vasculature and promote the proliferation of endothelial cells and angiogenesis[54]. The gut microbiota can facilitate the maturation and functional development of the GVB[55]. In neonates, the gut microbiota is not fully developed, and the Wnt pathway shows increased activity, leading to the incomplete establishment of the barrier function of the GVB[55]. Post-colonization, microbial signals downregulate PV-1 and enhance the production of TJ/AJ proteins, thereby accelerating GVB maturation. Additionally, bacterial ligands enhance the proliferation and tube-formation capability of IMVECs, thereby promoting angiogenesis and vessel sprouting[55,56]. The maturation of GVB relies on the integration of multiple signaling pathways (such as Wnt/β-catenin, VEGF/Notch, and gut microbiota signals). These mechanisms are inhibited or disrupted to varying degrees following surgery, which may result in PPOI.
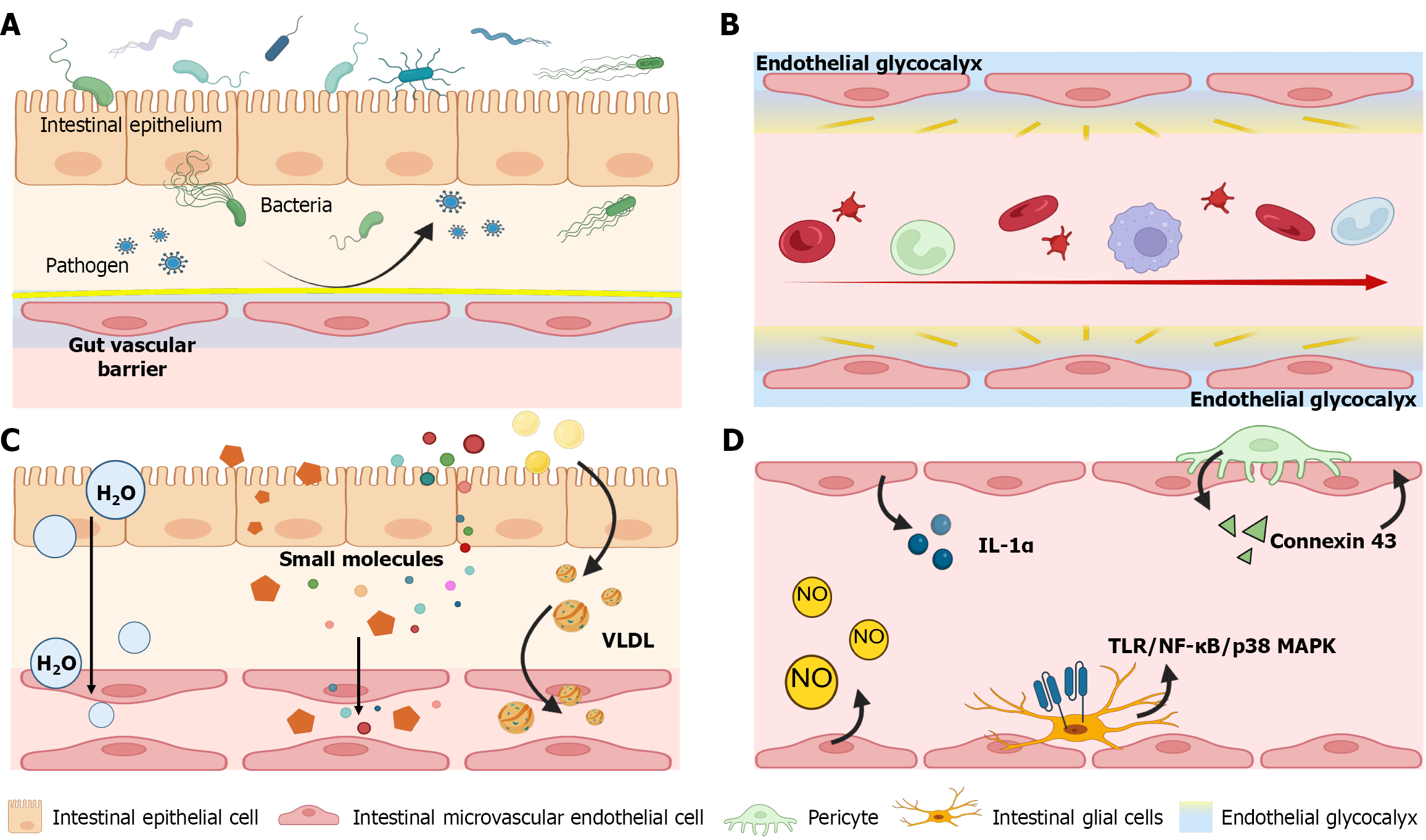
Barrier function of GVB: The GVB constitutes the final barrier preventing translocation of bacteria, macromolecules, cells, and other luminal constituents into the bloodstream[57]. The endothelial glycocalyx, intercellular junctions, and cellular components collectively constitute the barrier function of the GVB[58] (Figure 3A). The endothelial glycocalyx coating the surface of IMVECs prevents these cells from direct exposure to blood flow, blocking fluid shear stress-mediated activation of the NF-κB signaling pathway and phosphorylation of Akt, which maintains the physical barrier function of the GVB[59,60]. The intercellular junctional complexes form a “fence” between adjacent IMVECs, preventing bacteria and cells from entering the circulatory system[61]. PV-1 and VE-cadherin constitute the essential components of fenestrae and caveolae in IMVECs. In gut-derived sepsis, IMVECs exhibit upregulated PV-1 expression and reduce the level of VE-cadherin, compromising the GVB[62]. The increased permeability of the GVB allows macromolecules and bacteria from the gut to enter the systemic circulation and invade distant organs[62].
Beyond its physical barrier role, the GVB also functions as an anti-adhesive interface (Figure 3B). Under normal pH, the endothelial glycocalyx is a negatively charged gel layer that repels the adhesion of similarly negatively charged leuko
Absorptive function of GVB: Nutrient absorption refers to a direct transfer of substances or particles from the intestinal lumen into the bloodstream without prior digestion[69] (Figure 3C). The permeability-surface area product of the GVB and the capillary blood flow in the intestinal villi directly influence the rate and efficiency of absorption[70]. Small-molecule lipids in the chyme are first processed into chylomicrons and very-low-density lipoproteins with a diameter of less than 75 nm in the smooth endoplasmic reticulum and Golgi apparatus of intestinal epithelial cells and are then released into the basolateral plasma membrane[71]. IMVECs transport these chylomicrons and very-low-density li
Immune function of GVB: The GVB also functions as an immunoprotective barrier (Figure 3D). The proteoglycans in the glycocalyx (e.g., syndecan-1, syndecan-3, and biglycan) can scavenge chemokines, inhibit pro-inflammatory factor expression, block leukocyte migration, and induce autophagy of proinflammatory M1 macrophages[74]. High-molecular-weight hyaluronic acid exhibits anti-inflammatory and immunosuppressive properties. In addition, it inhibits M1 macrophage polarization and downregulates inflammatory factor expression by suppressing the activation of the glucoseregulated protein 78 kD/NF-κB, MAPK, and NF-κB signaling pathways and by binding to CD44 variants[75-77]. In contrast, the accumulation of low-molecular-weight hyaluronic acid promotes immune-inflammatory responses[78]. It downregulates VE-cadherin, increases endothelial permeability[79], and binds erythrocyte CD44 to induce red blood cell aggregation[80]. Endothelial-derived IL-1α initially promotes neutrophil adhesion in the early phase of PPOI and enhances the bactericidal activity of transendothelial neutrophils in the later phase by triggering oxidative phosph
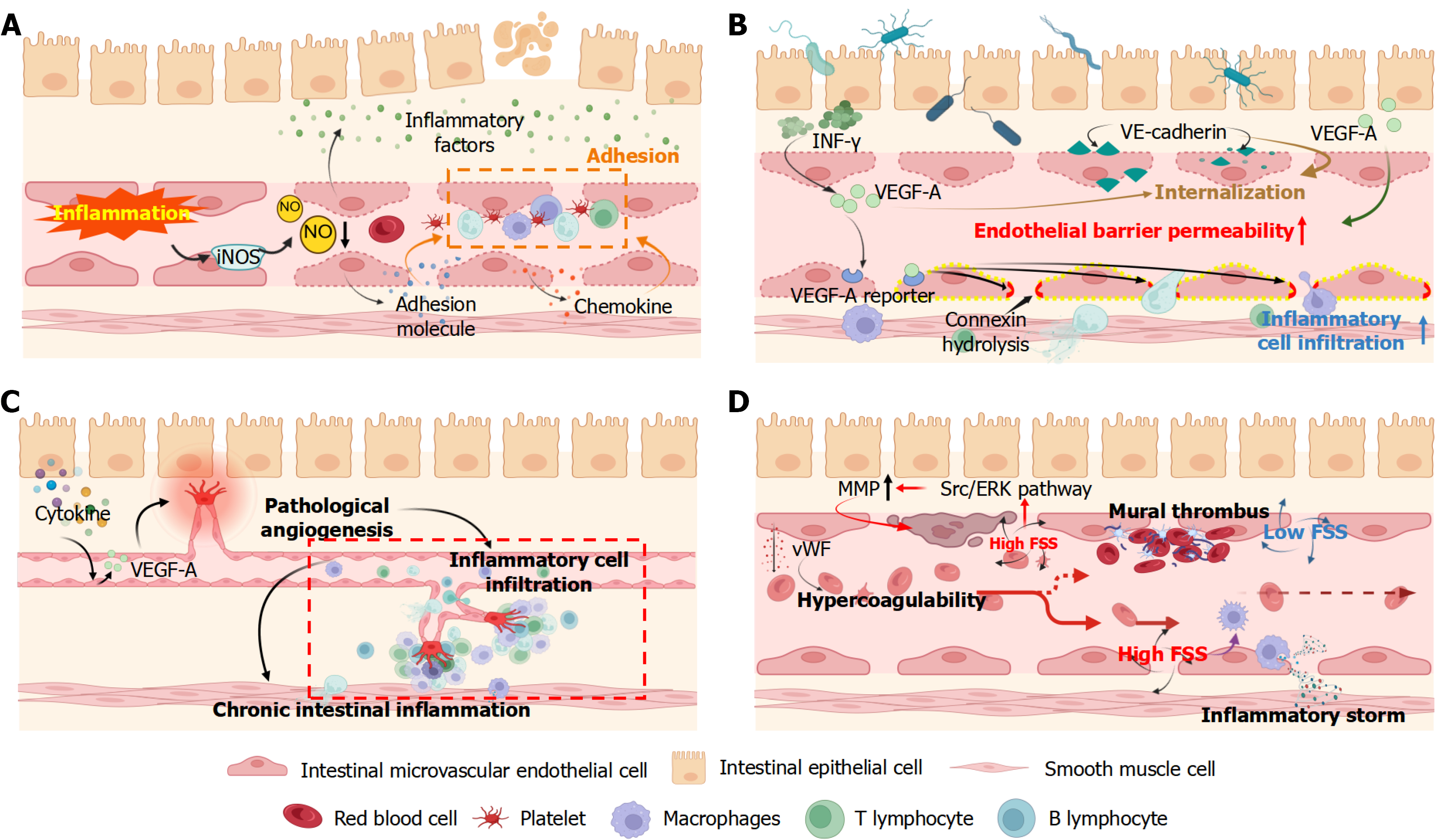
Disruption of the GVB and subsequent transendothelial migration of circulating leukocytes into the muscularis are central pathological events in PPOI (Figure 4). Loss of GVB integrity allows immune cells to infiltrate the intestinal wall, where they release pro-inflammatory cytokines, proteases, and reactive species. These mediators further degrade endothelial junctional proteins (e.g., VE-cadherin and ZO-1), increase vascular permeability, and upregulate adhesion and procoagulant molecules on IMVECs, such as ICAM-1, VCAM-1, and tissue factor. Together, these processes can form a feed-forward loop in which inflammation perpetuates GVB impairment, leading to tissue edema, microcirculatory dysfunction, altered neuronal signaling, and impaired gastrointestinal motility[57]. Thus, in PPOI, the GVB serves not merely as a regulatory factor for intestinal inflammation but also as a target of potential attack.
IMVEC dysfunction refers to a spectrum of maladaptive alterations in their functional phenotype, including reduced NO bioavailability, activation of pro-inflammatory phenotypes, and enhanced membrane permeability, among others[85]. These changes can initiate local tissue inflammation, edema, and hypoxic injury, ultimately contributing to functional disturbance. Key regulators implicated in endothelial dysfunction include the angiopoietin-Tie2 axis, adrenomedullin, and VE-cadherin[58]. Under inflammatory conditions, IMVECs exhibit reduced iNOS expression and diminished NO production, resulting in impaired endothelial anti-adhesive function[86]. Dysfunctional IMVECs secrete inflammatory factors (IL-6, IL-8, and TNF-α), chemokines [integrin α(v)β(3)], adhesion molecules (E-selectin, thromboxane B2, and ICAM-1), pro-angiogenic factors (VEGF-A), and apoptosis-related proteins (caspase-3)[8,9]. Inflammatory cytokines IL-6, IL-10, and TNF-α are considered independent risk factors for inducing PPOI[87]. After the NLRP3 inflammasome in endothelial cells is activated, the released IL-1β and caspase-1 exacerbate intestinal ischemia/reperfusion injury[88]. ICAM-1, integrin α(v)β(3), and fractalkine promote platelet activation, rolling, and adhesion to endothelial cells, thereby inducing intestinal inflammation[9]. IMVECs overexpressing VEGF-A and matrix metallopeptidase 14 promote microvascular angiogenesis in inflammatory bowel diseases[10,11]. Pathological angiogenesis is recognized as an active driver of chronic intestinal inflammation. Under TNF-α induction, the highly expressed apoptotic proteins in IMVECs—caspase-1 and caspase-3—can disrupt the intestinal mucosal barrier[89].
Surgical and traumatic stress activates the hypothalamic-pituitary-adrenal axis and increases the total number of circulating glucocorticoids, which can downregulate TJ proteins in both epithelial and endothelial cells. This phenomenon leads to impaired barrier function[90-92]. Sorribas et al[93] reported that chronic intraperitoneal administration of isoproterenol in mice upregulates PV-1 expression in IMVECs, increases GVB permeability, and permits trans-barrier passage of 70-kDa FITC dextran into systemic circulation. Increased GVB permeability promotes transendothelial migration of circulating macrophages, monocytes, and lymphocytes[94]. Infiltration of the muscularis propria by myeloid leukocytes induces enteric neuron paralysis, precipitating PPOI[29]. Inflammatory triggers cause IMVECs to synthesize and secrete a large amount of VEGF-A[95] also known as vascular permeability factor. The activation of the VEGF-A/phospholipase Cβ2, hypoxia-inducible factor/VEGF-A, and VEGF/Akt/MAPK signaling pathways directly induces an increase in GVB permeability[95-97]. Increased GVB permeability is primarily manifested as the degradation of the endothelial glycocalyx, hydrolysis of junction proteins, and proliferation of fenestrae/caveolae[59,98,99]. Elevated levels of TNF-α promote the degradation of the endothelial glycocalyx[100], which further activates plasma cells and neuroinflammatory responses, thereby triggering mucosal immunity[101]. Infection of IMVECs by interferon-γ disrupts the VE-cadherin membrane localization and induces the internalization of VE-cadherin through the VEGF-A pathway, ultimately leading to increased GVB permeability and intestinal inflammation[102]. Intestinal inflammation further impairs the GVB through a positive feedback loop, leading to reduced endothelial TJ and AJ proteins, increased PV-1 expression, and elevated fenestrae density in endothelial cells[103]. To circulate repeatedly, the inflammatory storm continuously up
Pathological angiogenesis is closely associated with chronic intestinal inflammation, postoperative intra-abdominal adhesions, and postoperative ileus[104,105]. In the inflamed intestinal tissue, a cascade of endothelial and platelet-derived signals drives the formation of new microvessels that are structurally and functionally abnormal. These nascent vessels frequently lack proper maturation and barrier function, which in turn amplifies tissue inflammation and compromises organ function. For example, activated STAT1 upregulates transglutaminase-2 in IMVECs; transglutaminase-2 can interact with VEGF receptor 2 and enhance its phosphorylation (reported at residues such as Tyr1059 and Tyr1214), potentiating downstream VEGF-dependent angiogenic signaling[105]. Platelets, displaced toward the vessel wall under disturbed flow conditions, adhere to activated IMVECs and release pro-angiogenic mediators, including VEGF-A, further stimulating endothelial proliferation, migration, and tubulogenesis[9]. Inflammatory mediators, tissue factors, chemo
Damaged endothelial cells release large amounts of vWF, rendering the blood hypercoagulable[43]. The vWF exposes binding sites and interacts with the glycoprotein Ib-IX-V complex on platelets and integrin αIIbβ3 to form aggregates[117]. These aggregates adhere to the damaged and activated IMVECs and serve as binding sites to further recruit leukocytes for adhesion[117]. The accumulation of platelet-leukocyte aggregates along with elevated inflammatory mediators increases blood viscosity and predisposes the blood to flow stasis[118]. Endothelial injury, a hypercoagulable milieu, and stasis act synergistically to promote microvascular thrombosis; when microthrombi organize into stable mural thrombi, local luminal stenosis can develop[118]. Blood flow through stenotic segments becomes turbulent, producing vortices and marked hemodynamic disturbance that can exacerbate barrier injury, local inflammation, and ileus[119-121]. Under mechanical stress, elevated expression of macrophage chemoattractant protein 1 induces ma
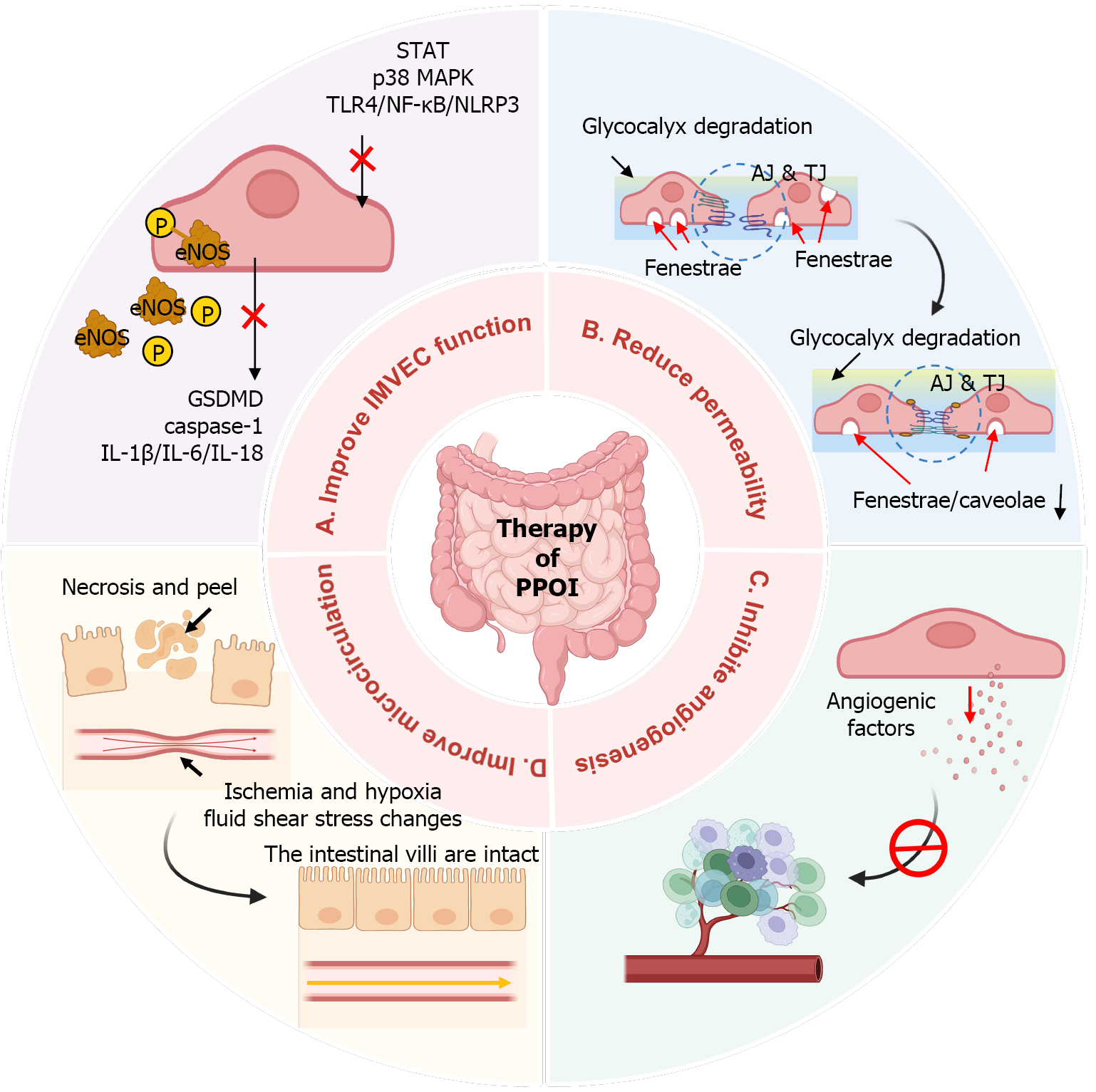
Dysfunction of IMVECs and the damage to the GVB are considered important pathological changes in PPOI. Therefore, improving the function of IMVECs and repairing the damaged GVB may be potential therapeutic targets for PPOI (Figure 5). Table 1 reviews drugs that can restore the GVB to improve PPOI.
| Drug | Mechanism | Biological activity | Animals/cells | Ref. |
| Kaempferol | Inhibiting the activation of inflammatory phenotype in IMVECs and strengthening the intestinal epithelial–endothelial barrier | Attenuating the overexpression of pro-inflammatory cytokines and adhesion molecules in IMVECs through inhibiting TLR4 overexpression and NF-κB, p38 MAPK, and STAT phosphorylation | RIMVECs | Bian et al[128], 2019; and Bian et al[133], 2020 |
| Prostaglandin E2 | Promoting the activity of IMVECs | Through the PGE2-EP4-eNOS signaling axis, prostaglandin E2 promotes IMVEC proliferation and NO production, which result in improved intestinal microcirculation | Mice/MIMVECs | Mo et al[129], 2023 |
| Naringin | Inhibiting apoptosis, promoting cell migration, and upregulating the expression of endothelial cell junction proteins | Preventing TNF-α-induced apoptosis and migration suppression in IMVECs; Upregulating the expression of endothelial TJ, such as ZO-1, occludin, claudin-1, and claudin-2 | RIMVECs | Liu et al[130], 2020 |
| Quercetin | Protecting IMVECs from pyroptotic inflammation | Inhibiting the release of NLRP3 inflammasomes and the expression of pyroptotic proteins via the TLR4/NF-κB/NLRP3 signaling pathway to alleviate intestinal inflammation | Rat/RIMVECs | Zhang et al[131], 2022 |
| Berberine | Reducing the permeability of the GVB | Increasing the expression of β-catenin, claudin-12, and VE-cadherin via modulating the Wnt/β-catenin pathway | Rat/RIMVECs | He et al[13], 2018 |
| Dexmedetomidine | Reducing the permeability of the GVB | Increasing the expression of β-catenin, VE-cadherin, ZO-1, and occludin | Mice/MIMVECs | Zhang et al[61], 2022 |
| Dihydroartemisinin | Repairing the intestinal epithelial–endothelial barrier | Increasing the expression of TJ and AJ and decreasing that of PV-1 | Mice | Qiu et al[103], 2024 |
| G6-31 | Inhibiting angiogenesis; improving microcirculatory hemodynamics | Specifically binding to VEGF-A, inhibiting angiogenesis and improving microcirculatory hemodynamics | Mice | Ardelean et al[115], 2014 |
| AP-Cav | Inhibiting angiogenesis | Inhibiting the proliferation of VECs and inducing VEC apoptosis through the suppression of the MAP signaling pathway and induction of JNK-dependent apoptosis | ECs | Fang and Kevil[135], 2020 |
| N-palmitoyl-d-glucosamine | Inhibiting angiogenesis | Inhibiting the expression of inflammatory and pro-angiogenic factors by blocking the pAkt/mTOR/hypoxia-inducible factor1α pathway | Mice | Palenca et al[136], 2024 |
| Heparin-binding epidermal growth factor-like growth factor | Improving intestinal microcirculation | Alleviating intestinal ischemia/reperfusion injury by reducing JNK phosphorylation and inhibiting the activation of the p38/MAPK signaling pathway Increasing the expression of the endothelin B receptor and triggering intracellular calcium mobilization | Rat/HIMVECs | Ming et al[138], 2019 and Zhou et al[139], 2009 |
| Melatonin | Improving intestinal microcirculation | Attenuating pathological intestinal microvascular congestion and suppressing the infiltration of mast cells and granulocytes into intestinal tissues | Rat | Lansink et al[141], 2017 |
IMVECs are the primary components of GVB and serve as trigger cells for inflammation. Suppression of the inflammatory phenotype activation in IMVECs and improving IMVEC function may effectively mitigate intestinal inflammation. Through the suppression of TLR4 overexpression and inhibition of NF-κB, p38 MAPK, and STAT phosphory
Increased GVB permeability facilitates the circulating immune cells to respond to local inflammatory signals and migrate across the endothelium[132]. Restoring the barrier function of the GVB may mitigate immune-inflammatory cell infiltration into the intestinal muscular layer in PPOI[50]. Reportedly, kaempferol enhances the GVB integrity by elevating transepithelial electrical resistance and upregulating TJ and AJ protein expression, thereby counteracting lipopolysaccharide-induced hyperpermeability[133]. In an inflammatory factor-induced GVB injury model, naringin has been shown to enhance the barrier function of GVB by increasing the expression and distribution of ZO-1, occludin, claudin-1, and claudin-2 proteins in IMVECs[130]. During sepsis, berberine protects the GVB and reduces microvascular leakage by regulating the Wnt/β-catenin signaling pathway and increasing the expression of β-catenin, claudin-12, and VE-cadherin[13]. In colitis models, dihydroartemisinin repairs the intestinal epithelial-endothelial barrier by increasing the expression of TJ and AJ and decreasing that of PV-1, ultimately ameliorating colonic inflammation[103]. Dexmedetomidine protects against GVB damage caused by intestinal ischemia-reperfusion by increasing the expression of β-catenin, VE-cadherin, ZO-1, and occludin[61].
Pathological angiogenesis is a hallmark of chronic intestinal inflammation. Therefore, inhibiting the aberrant proliferation of IMVECs and inflammatory angiogenesis may control the extent of infiltration by circulating leukocytes into the muscularis layer[134]. G6-31 is a monoclonal antibody with high affinity and specificity for VEGF-A. When colitis-afflicted mice were treated with G6-31, a significant reduction in intestinal microvascular density was observed (430 ± 24 mm-2 vs 250 ± 26 mm-2, P < 0.001)[115]. This reduction in microvessel density in colitis mice is accompanied by improved microvascular hemodynamics, decreased inflammatory cell infiltration into the muscular layer, and alleviated intestinal inflammation[115]. The anti-angiogenic effect of the antennapedia-conjugated caveolin-1 scaffolding domain is achieved through the suppression of the mitogen-activated protein signaling pathway and induction of c-Jun N-terminal kinase-dependent apoptosis, which inhibit the proliferation and promote the apoptosis of endothelial cells[135]. Micronized N-palmitoyl-d-glucosamine inhibits the expression of inflammatory and pro-angiogenic factors by blocking the pAkt/mammalian target of rapamycin/hypoxia-inducible factor 1α pathway, countering inflammatory angiogenesis in colorectal carcinoma[136].
Surgical manipulation decreases the bowel wall perfusion by 29% and impairs intestinal motility[137]. Thus, we propose that improving intestinal microcirculation can improve intestinal motility. Heparin-binding epidermal growth factor-like growth factor can alleviate intestinal ischemia/reperfusion injury by reducing c-Jun N-terminal kinase phosphorylation and inhibiting the activation of the p38/MAPK signaling pathway[138]. Additionally, heparin-binding epidermal growth factor-like growth factor dilates the intestinal microvasculature and ameliorates intestinal microcirculation by increasing the expression of endothelin B receptor and provoking intracellular calcium mobilization[139]. Inflammation leads to increased intestinal microvascular hemodynamics and induces vascular inflammation. Anti-VEGF therapy can decrease intestinal microvascular hemodynamics and vascular inflammation, resulting in reduced intestinal inflammation[115]. Necrotizing enterocolitis leads to impaired intestinal microcirculatory perfusion. Promotion of endogenous NO and production of hydrogen sulfide through remote ischemic conditioning have been proven to maintain intestinal microcirculatory perfusion and reduce the ischemic necrosis of intestinal villi[140]. Intestinal congestion, edema, and hemorrhage are adaptive changes in response to inflammation. Using murine models of systemic inflammation, it has been shown that intravenous melatonin attenuates pathological intestinal microvascular congestion and suppresses the infiltration of mast cells and granulocytes into intestinal tissues[141].
PPOI is one of the most common postoperative complications, especially after abdominal surgery. In PPOI, both local inflammation and vascular inflammation mutually reinforce each other, triggering a cascade of events that disrupts the intestinal barrier, impairs intestinal neuronal conduction, and inhibits intestinal contractile activity. IMVEC dysfunction, increased GVB permeability, pathological angiogenesis, and intestinal microcirculatory hemodynamic alterations are the fundamental pathological alterations in PPOI. Thus, the GVB may present a potential therapeutic target for PPOI. Improving IMVEC function, reducing GVB permeability, inhibiting pathological angiogenesis, and improving intestinal microcirculation are highly likely to shorten or even block the progression of PPOI. However, further in vivo and in vitro studies are required to substantiate this hypothesis.
| 1. | Liu GXH, Milne T, Xu W, Varghese C, Keane C, O'Grady G, Bissett IP, Wells CI. Risk prediction algorithms for prolonged postoperative ileus: A systematic review. Colorectal Dis. 2024;26:1101-1113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 2. | Traeger L, Koullouros M, Bedrikovetski S, Kroon HM, Moore JW, Sammour T. Global cost of postoperative ileus following abdominal surgery: meta-analysis. BJS Open. 2023;7:zrad054. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 3. | Solanki S, Chakinala RC, Haq KF, Singh J, Khan MA, Solanki D, Vyas MJ, Kichloo A, Mansuri U, Shah H, Patel A, Haq KS, Iqbal U, Nabors C, Khan HMA, Aronow WS. Paralytic ileus in the United States: A cross-sectional study from the national inpatient sample. SAGE Open Med. 2020;8:2050312120962636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 4. | Yi K, An L, Qi Y, Yang T, Duan Y, Zhao X, Zhang P, Huang X, Su X, Tang Z, Sun D. Docosahexaenoic acid (DHA) promotes recovery from postoperative ileus and the repair of the injured intestinal barrier through mast cell-nerve crosstalk. Int Immunopharmacol. 2024;136:112316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | Kim MJ, Lee YJ, Hussain Z, Park H. Effect of Probiotics on Improving Intestinal Mucosal Permeability and Inflammation after Surgery. Gut Liver. 2025;19:207-218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 6. | Spadoni I, Zagato E, Bertocchi A, Paolinelli R, Hot E, Di Sabatino A, Caprioli F, Bottiglieri L, Oldani A, Viale G, Penna G, Dejana E, Rescigno M. A gut-vascular barrier controls the systemic dissemination of bacteria. Science. 2015;350:830-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 607] [Cited by in RCA: 555] [Article Influence: 50.5] [Reference Citation Analysis (1)] |
| 7. | Di Tommaso N, Santopaolo F, Gasbarrini A, Ponziani FR. The Gut-Vascular Barrier as a New Protagonist in Intestinal and Extraintestinal Diseases. Int J Mol Sci. 2023;24:1470. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 8. | Hu Y, Chen X, Duan H, Hu Y, Mu X. Chinese herbal medicinal ingredients inhibit secretion of IL-6, IL-8, E-selectin and TXB2 in LPS-induced rat intestinal microvascular endothelial cells. Immunopharmacol Immunotoxicol. 2009;31:550-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Rutella S, Vetrano S, Correale C, Graziani C, Sturm A, Spinelli A, De Cristofaro R, Repici A, Malesci A, Danese S. Enhanced platelet adhesion induces angiogenesis in intestinal inflammation and inflammatory bowel disease microvasculature. J Cell Mol Med. 2011;15:625-634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 10. | Scaldaferri F, Vetrano S, Sans M, Arena V, Straface G, Stigliano E, Repici A, Sturm A, Malesci A, Panes J, Yla-Herttuala S, Fiocchi C, Danese S. VEGF-A links angiogenesis and inflammation in inflammatory bowel disease pathogenesis. Gastroenterology. 2009;136:585-95.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 263] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 11. | Esteban S, Clemente C, Koziol A, Gonzalo P, Rius C, Martínez F, Linares PM, Chaparro M, Urzainqui A, Andrés V, Seiki M, Gisbert JP, Arroyo AG. Endothelial MT1-MMP targeting limits intussusceptive angiogenesis and colitis via TSP1/nitric oxide axis. EMBO Mol Med. 2020;12:e10862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 12. | Chu T, Yu R, Gu Y, Wang Y, Chang H, Li Y, Li J, Bian Y. Kaempferol protects gut-vascular barrier from high glucose-induced disorder via NF-κB pathway. J Nutr Biochem. 2024;123:109496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 13. | He Y, Yuan X, Zuo H, Sun Y, Feng A. Berberine Exerts a Protective Effect on Gut-Vascular Barrier via the Modulation of the Wnt/Beta-Catenin Signaling Pathway During Sepsis. Cell Physiol Biochem. 2018;49:1342-1351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 14. | Kalff JC, Carlos TM, Schraut WH, Billiar TR, Simmons RL, Bauer AJ. Surgically induced leukocytic infiltrates within the rat intestinal muscularis mediate postoperative ileus. Gastroenterology. 1999;117:378-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 238] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 15. | Sherrod BA, Kim R, Hunsaker J, Rada C, Christensen C, Stoddard GJ, Brodke D, Mahan MA, Mazur MD, Bisson EF, Dailey AT. Postoperative ileus risk after posterior thoracolumbar fusion performed with total intravenous anesthesia versus inhaled anesthesia. J Neurosurg Spine. 2023;38:307-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 16. | Ye Q, Hu Y, Xing Q, Wu Y, Zhang Y. The Effects of Opioid-Free Anesthesia with Dexmedetomidine and Esketamine on Postoperative Anesthetic-Related Complications for Hip Surgery in the Elderly. Int J Gen Med. 2024;17:6291-6302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 17. | Kusano T, Etoh T, Inomata M, Shiraishi N, Kitano S. CO(2) pneumoperitoneum increases secretory IgA levels in the gut compared with laparotomy in an experimental animal model. Surg Endosc. 2014;28:1879-1885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Strowitzki MJ, Nelson R, Garcia MP, Tuffs C, Bleul MB, Fitzsimons S, Navas J, Uzieliene I, Ritter AS, Phelan D, Kierans SJ, Blanco A, Bernotiene E, Belton O, Schneider M, Cummins EP, Taylor CT. Carbon Dioxide Sensing by Immune Cells Occurs through Carbonic Anhydrase 2-Dependent Changes in Intracellular pH. J Immunol. 2022;208:2363-2375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 19. | Abd El Aziz MA, Grass F, Calini G, Lovely JK, Jacob AK, Behm KT, D'Angelo AD, Shawki SF, Mathis KL, Larson DW. Intraoperative Fluid Management a Modifiable Risk Factor for Surgical Quality - Improving Standardized Practice. Ann Surg. 2022;275:891-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 20. | Snoek SA, Dhawan S, van Bree SH, Cailotto C, van Diest SA, Duarte JM, Stanisor OI, Hilbers FW, Nijhuis L, Koeman A, van den Wijngaard RM, Zuurbier CJ, Boeckxstaens GE, de Jonge WJ. Mast cells trigger epithelial barrier dysfunction, bacterial translocation and postoperative ileus in a mouse model. Neurogastroenterol Motil. 2012;24:172-184, e91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 21. | Koscielny A, Engel D, Maurer J, Hirner A, Kurts C, Kalff JC. Impact of CCR7 on the gastrointestinal field effect. Am J Physiol Gastrointest Liver Physiol. 2011;300:G665-G675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | The FO, Bennink RJ, Ankum WM, Buist MR, Busch OR, Gouma DJ, van der Heide S, van den Wijngaard RM, de Jonge WJ, Boeckxstaens GE. Intestinal handling-induced mast cell activation and inflammation in human postoperative ileus. Gut. 2008;57:33-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 116] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 23. | Pohl JM, Gutweiler S, Thiebes S, Volke JK, Klein-Hitpass L, Zwanziger D, Gunzer M, Jung S, Agace WW, Kurts C, Engel DR. Irf4-dependent CD103(+)CD11b(+) dendritic cells and the intestinal microbiome regulate monocyte and macrophage activation and intestinal peristalsis in postoperative ileus. Gut. 2017;66:2110-2120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 24. | Engel DR, Koscielny A, Wehner S, Maurer J, Schiwon M, Franken L, Schumak B, Limmer A, Sparwasser T, Hirner A, Knolle PA, Kalff JC, Kurts C. T helper type 1 memory cells disseminate postoperative ileus over the entire intestinal tract. Nat Med. 2010;16:1407-1413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 89] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 25. | Kimura H, Yamazaki T, Mihara T, Kaji N, Kishi K, Hori M. Purinergic P2X7 receptor antagonist ameliorates intestinal inflammation in postoperative ileus. J Vet Med Sci. 2022;84:610-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | Lin SS, Zhang RQ, Shen L, Xu XJ, Li K, Bazhin AV, Fichna J, Li YY. Alterations in the gut barrier and involvement of Toll-like receptor 4 in murine postoperative ileus. Neurogastroenterol Motil. 2018;30:e13286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 27. | Delfini M, Stakenborg N, Viola MF, Boeckxstaens G. Macrophages in the gut: Masters in multitasking. Immunity. 2022;55:1530-1548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 130] [Reference Citation Analysis (1)] |
| 28. | Liu T, Xu M, Shi Z, Li M, Wang R, Shi Y, Xu X, Shao T, Sun Q. Shenhuang plaster ameliorates the Inflammation of postoperative ileus through inhibiting PI3K/Akt/NF-κB pathway. Biomed Pharmacother. 2022;156:113922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 29. | Yang NN, Yang JW, Ye Y, Huang J, Wang L, Wang Y, Su XT, Lin Y, Yu FT, Ma SM, Qi LY, Lin LL, Wang LQ, Shi GX, Li HP, Liu CZ. Electroacupuncture ameliorates intestinal inflammation by activating α7nAChR-mediated JAK2/STAT3 signaling pathway in postoperative ileus. Theranostics. 2021;11:4078-4089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 162] [Article Influence: 32.4] [Reference Citation Analysis (0)] |
| 30. | Yip JLK, Balasuriya GK, Spencer SJ, Hill-Yardin EL. The Role of Intestinal Macrophages in Gastrointestinal Homeostasis: Heterogeneity and Implications in Disease. Cell Mol Gastroenterol Hepatol. 2021;12:1701-1718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 31. | Docsa T, Bhattarai D, Sipos A, Wade CE, Cox CS Jr, Uray K. CXCL1 is upregulated during the development of ileus resulting in decreased intestinal contractile activity. Neurogastroenterol Motil. 2020;32:e13757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 32. | Tsuchida Y, Hatao F, Fujisawa M, Murata T, Kaminishi M, Seto Y, Hori M, Ozaki H. Neuronal stimulation with 5-hydroxytryptamine 4 receptor induces anti-inflammatory actions via α7nACh receptors on muscularis macrophages associated with postoperative ileus. Gut. 2011;60:638-647. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 114] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 33. | Sui C, Tao L, Bai C, Shao L, Miao J, Chen K, Wang M, Hu Q, Wang F. Molecular and cellular mechanisms underlying postoperative paralytic ileus by various immune cell types. Front Pharmacol. 2022;13:929901. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 34. | Bu F, Huang S, Yang X, Wei L, Zhang D, Zhang Z, Tian D. Damage-induced NAD release activates intestinal CD4+ and CD8+ T cell via P2X7R signaling. Cell Immunol. 2023;385:104677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 35. | Schneider R, Leven P, Glowka T, Kuzmanov I, Lysson M, Schneiker B, Miesen A, Baqi Y, Spanier C, Grants I, Mazzotta E, Villalobos-Hernandez E, Kalff JC, Müller CE, Christofi FL, Wehner S. A novel P2X2-dependent purinergic mechanism of enteric gliosis in intestinal inflammation. EMBO Mol Med. 2021;13:e12724. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 36. | Chapman SJ, Pericleous A, Downey C, Jayne DG. Postoperative ileus following major colorectal surgery. Br J Surg. 2018;105:797-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 119] [Article Influence: 14.9] [Reference Citation Analysis (1)] |
| 37. | Patrick R, Pando BD, Yang C, Aponte A, Wang F, Ewing T, Ma Y, Yuan SY, Wu MH. Focal adhesion kinase mediates microvascular leakage and endothelial barrier dysfunction in ischemia-reperfusion injury. Microvasc Res. 2025;159:104791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Morini S, Elias G, Brown M, Subbotin V, Rastellini C, Cicalese L. Chronic morpho-functional damage as a consequence of transient ischemia/reperfusion injury of the small bowel. Histol Histopathol. 2010;25:277-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 39. | Krüger-Genge A, Blocki A, Franke RP, Jung F. Vascular Endothelial Cell Biology: An Update. Int J Mol Sci. 2019;20:4411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 949] [Cited by in RCA: 833] [Article Influence: 119.0] [Reference Citation Analysis (0)] |
| 40. | Fleming I, Fisslthaler B, Dixit M, Busse R. Role of PECAM-1 in the shear-stress-induced activation of Akt and the endothelial nitric oxide synthase (eNOS) in endothelial cells. J Cell Sci. 2005;118:4103-4111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 242] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 41. | Andrabi SM, Sharma NS, Karan A, Shahriar SMS, Cordon B, Ma B, Xie J. Nitric Oxide: Physiological Functions, Delivery, and Biomedical Applications. Adv Sci (Weinh). 2023;10:e2303259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 483] [Cited by in RCA: 348] [Article Influence: 116.0] [Reference Citation Analysis (0)] |
| 42. | Soufli I, Toumi R, Rafa H, Touil-Boukoffa C. Overview of cytokines and nitric oxide involvement in immuno-pathogenesis of inflammatory bowel diseases. World J Gastrointest Pharmacol Ther. 2016;7:353-360. [PubMed] [DOI] [Full Text] |
| 43. | Shihata WA, Michell DL, Andrews KL, Chin-Dusting JP. Caveolae: A Role in Endothelial Inflammation and Mechanotransduction? Front Physiol. 2016;7:628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 44. | Zhuang CL, Chen FF, Lu JX, Zheng BS, Liu S, Zhou CJ, Huang DD, Shen X, Yu Z. Impact of different surgical traumas on postoperative ileus in rats and the mechanisms involved. Int J Clin Exp Med. 2015;8:16778-16786. [PubMed] |
| 45. | Suzuki S, Suzuki H, Horiguchi K, Tsugawa H, Matsuzaki J, Takagi T, Shimojima N, Hibi T. Delayed gastric emptying and disruption of the interstitial cells of Cajal network after gastric ischaemia and reperfusion. Neurogastroenterol Motil. 2010;22:585-593, e126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 46. | Silva MA, de Meirelles LR, Bustorff-Silva JM. Changes in intestinal motility and in the myenteric plexus in a rat model of intestinal ischemia-reperfusion. J Pediatr Surg. 2007;42:1062-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 47. | Liu P, Bian Y, Zhong J, Yang Y, Mu X, Liu Z. Establishment and characterization of a rat intestinal microvascular endothelial cell line. Tissue Cell. 2021;72:101573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 48. | Haraldsen G, Rugtveit J, Kvale D, Scholz T, Muller WA, Hovig T, Brandtzaeg P. Isolation and longterm culture of human intestinal microvascular endothelial cells. Gut. 1995;37:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 44] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 49. | Nayak G, Odaka Y, Prasad V, Solano AF, Yeo EJ, Vemaraju S, Molkentin JD, Trumpp A, Williams B, Rao S, Lang RA. Developmental vascular regression is regulated by a Wnt/β-catenin, MYC and CDKN1A pathway that controls cell proliferation and cell death. Development. 2018;145:dev154898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 50. | Ke Z, Huang Y, Xu J, Liu Y, Zhang Y, Wang Y, Zhang Y, Liu Y. Escherichia coli NF73-1 disrupts the gut-vascular barrier and aggravates high-fat diet-induced fatty liver disease via inhibiting Wnt/β-catenin signalling pathway. Liver Int. 2024;44:776-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 51. | Wang WG, Jiang XF, Zhang C, Zhan XP, Cheng JG, Tao LM, Xu WP, Li Z, Zhang Y. Avermectin induced vascular damage in zebrafish larvae: association with mitochondria-mediated apoptosis and VEGF/Notch signaling pathway. J Hazard Mater. 2024;477:135376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 52. | Feng A, Li C, Su S, Liu Y. 1,25(OH)2D3 supplementation alleviates gut-vascular barrier disruption via inhibition of S100B/ADAM10 pathway. Tissue Barriers. 2024;12:2327776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 53. | Feng A, Su S, Li C, Kang Y, Qiu J, Zhou J. Berberine decreases S100B generation to regulate gut vascular barrier permeability in mice with burn injury. Pharm Biol. 2024;62:53-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 54. | Zhang ZS, Yang A, Luo X, Zhou HN, Liu YY, Bao DQ, Zhang J, Zang JT, Li QH, Li T, Liu LM. Pericyte-derived extracellular vesicles improve vascular barrier function in sepsis via the Angpt1/PI3K/AKT pathway and pericyte recruitment: an in vivo and in vitro study. Stem Cell Res Ther. 2025;16:70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 55. | Travier L, Alonso M, Andronico A, Hafner L, Disson O, Lledo PM, Cauchemez S, Lecuit M. Neonatal susceptibility to meningitis results from the immaturity of epithelial barriers and gut microbiota. Cell Rep. 2021;35:109319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 56. | Schirbel A, Kessler S, Rieder F, West G, Rebert N, Asosingh K, McDonald C, Fiocchi C. Pro-angiogenic activity of TLRs and NLRs: a novel link between gut microbiota and intestinal angiogenesis. Gastroenterology. 2013;144:613-623.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 106] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 57. | Britzen-Laurent N, Weidinger C, Stürzl M. Contribution of Blood Vessel Activation, Remodeling and Barrier Function to Inflammatory Bowel Diseases. Int J Mol Sci. 2023;24:5517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 58. | Hellenthal KEM, Brabenec L, Wagner NM. Regulation and Dysregulation of Endothelial Permeability during Systemic Inflammation. Cells. 2022;11:1935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 125] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 59. | Ying J, Zhang C, Wang Y, Liu T, Yu Z, Wang K, Chen W, Zhou Y, Lu G. Sulodexide improves vascular permeability via glycocalyx remodelling in endothelial cells during sepsis. Front Immunol. 2023;14:1172892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 60. | Sugita S, Naito Y, Zhou L, He H, Hao Q, Sakamoto A, Lee JW. Hyaluronic acid restored protein permeability across injured human lung microvascular endothelial cells. FASEB Bioadv. 2022;4:619-631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 61. | Zhang YN, Chang ZN, Liu ZM, Wen SH, Zhan YQ, Lai HJ, Zhang HF, Guo Y, Zhang XY. Dexmedetomidine Alleviates Gut-Vascular Barrier Damage and Distant Hepatic Injury Following Intestinal Ischemia/Reperfusion Injury in Mice. Anesth Analg. 2022;134:419-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 62. | Chang Z, Zhang Y, Lin M, Wen S, Lai H, Zhan Y, Zhu X, Huang Z, Zhang X, Liu Z. Improvement of gut-vascular barrier by terlipressin reduces bacterial translocation and remote organ injuries in gut-derived sepsis. Front Pharmacol. 2022;13:1019109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 63. | Jedlicka J, Becker BF, Chappell D. Endothelial Glycocalyx. Crit Care Clin. 2020;36:217-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 91] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 64. | Dragovich MA, Genemaras K, Dailey HL, Jedlicka S, Frank Zhang X. Dual Regulation of L-Selectin-Mediated Leukocyte Adhesion by Endothelial Surface Glycocalyx. Cell Mol Bioeng. 2017;10:102-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 65. | Aguilar G, Córdova F, Koning T, Sarmiento J, Boric MP, Birukov K, Cancino J, Varas-Godoy M, Soza A, Alves NG, Mujica PE, Durán WN, Ehrenfeld P, Sánchez FA. TNF-α-activated eNOS signaling increases leukocyte adhesion through the S-nitrosylation pathway. Am J Physiol Heart Circ Physiol. 2021;321:H1083-H1095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 66. | Aguilar G, Koning T, Ehrenfeld P, Sánchez FA. Role of NO and S-nitrosylation in the Expression of Endothelial Adhesion Proteins That Regulate Leukocyte and Tumor Cell Adhesion. Front Physiol. 2020;11:595526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 67. | Parsanathan R, Jain SK. Glucose-6-phosphate dehydrogenase deficiency increases cell adhesion molecules and activates human monocyte-endothelial cell adhesion: Protective role of l-cysteine. Arch Biochem Biophys. 2019;663:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 68. | Gao F, Lucke-Wold BP, Li X, Logsdon AF, Xu LC, Xu S, LaPenna KB, Wang H, Talukder MAH, Siedlecki CA, Huber JD, Rosen CL, He P. Reduction of Endothelial Nitric Oxide Increases the Adhesiveness of Constitutive Endothelial Membrane ICAM-1 through Src-Mediated Phosphorylation. Front Physiol. 2017;8:1124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 69. | Takahara E, Mantani Y, Udayanga KG, Qi WM, Tanida T, Takeuchi T, Yokoyama T, Hoshi N, Kitagawa H. Ultrastructural demonstration of the absorption and transportation of minute chylomicrons by subepithelial blood capillaries in rat jejunal villi. J Vet Med Sci. 2013;75:1563-1569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 70. | Pappenheimer JR, Michel CC. Role of villus microcirculation in intestinal absorption of glucose: coupling of epithelial with endothelial transport. J Physiol. 2003;553:561-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 71. | Sesorova IS, Dimov ID, Kashin AD, Sesorov VV, Karelina NR, Zdorikova MA, Beznoussenko GV, Mirоnоv AA. Cellular and sub-cellular mechanisms of lipid transport from gut to lymph. Tissue Cell. 2021;72:101529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 72. | Klip A, De Bock K, Bilan PJ, Richter EA. Transcellular Barriers to Glucose Delivery in the Body. Annu Rev Physiol. 2024;86:149-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 73. | Cheng Y, Liu Y, Chen D, Zhou Y, Yu S, Lin H, Liao CK, Lin H, Xu P, Huang M. Dual effects of quercetin on protein digestion and absorption in the digestive tract. Food Chem. 2021;358:129891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 74. | Kunnathattil M, Rahul P, Skaria T. Soluble vascular endothelial glycocalyx proteoglycans as potential therapeutic targets in inflammatory diseases. Immunol Cell Biol. 2024;102:97-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 75. | Lee CH, Chiang CF, Kuo FC, Su SC, Huang CL, Liu JS, Lu CH, Hsieh CH, Wang CC, Lee CH, Shen PH. High-Molecular-Weight Hyaluronic Acid Inhibits IL-1β-Induced Synovial Inflammation and Macrophage Polarization through the GRP78-NF-κB Signaling Pathway. Int J Mol Sci. 2021;22:11917. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 76. | Chen M, Li L, Wang Z, Li P, Feng F, Zheng X. High molecular weight hyaluronic acid regulates P. gingivalis-induced inflammation and migration in human gingival fibroblasts via MAPK and NF-κB signaling pathway. Arch Oral Biol. 2019;98:75-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 77. | Hu L, Nomura S, Sato Y, Takagi K, Ishii T, Honma Y, Watanabe K, Mizukami Y, Muto J. Anti-inflammatory effects of differential molecular weight Hyaluronic acids on UVB-induced calprotectin-mediated keratinocyte inflammation. J Dermatol Sci. 2022;107:24-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 42] [Reference Citation Analysis (0)] |
| 78. | Imani J, Liu K, Cui Y, Assaker JP, Han J, Ghosh AJ, Ng J, Shrestha S, Lamattina AM, Louis PH, Hentschel A, Esposito AJ, Rosas IO, Liu X, Perrella MA, Azzi J, Visner G, El-Chemaly S. Blocking hyaluronan synthesis alleviates acute lung allograft rejection. JCI Insight. 2021;6:e142217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 79. | Queisser KA, Mellema RA, Middleton EA, Portier I, Manne BK, Denorme F, Beswick EJ, Rondina MT, Campbell RA, Petrey AC. COVID-19 generates hyaluronan fragments that directly induce endothelial barrier dysfunction. JCI Insight. 2021;6:e147472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 80. | Ma X, Wang X, Jia X, Hui JH, Shofaro JH, Tao R, Hui MM. Size-dependent aggregation of erythrocytes by low molecular weight hyaluronic acids of different sizes: bioactivity and quality control potential. Front Physiol. 2025;16:1527354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 81. | Liu X, Zhang H, He S, Mu X, Hu G, Dong H. Endothelial-Derived Interleukin-1α Activates Innate Immunity by Promoting the Bactericidal Activity of Transendothelial Neutrophils. Front Cell Dev Biol. 2020;8:590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 82. | Brown RM, Le HH, Babcock IW, Harris TH, Gaultier A. Functional analysis of antigen presentation by enteric glial cells during intestinal inflammation. Glia. 2025;73:291-308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 83. | Antunes J, Sobral P, Martins M, Branco V. Nanoplastics activate a TLR4/p38-mediated pro-inflammatory response in human intestinal and mouse microglia cells. Environ Toxicol Pharmacol. 2023;104:104298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 84. | Carvalho TP, Toledo FAO, Bautista DFA, Silva MF, Oliveira JBS, Lima PA, Costa FB, Ribeiro NQ, Lee J-Y, Birbrair A, Paixão TA, Tsolis RM, Santos RL. Pericytes modulate endothelial inflammatory response during bacterial infection. mBio. 2024;15:e0325223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 85. | Gimbrone MA Jr, García-Cardeña G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ Res. 2016;118:620-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2941] [Cited by in RCA: 2651] [Article Influence: 265.1] [Reference Citation Analysis (0)] |
| 86. | Binion DG, Rafiee P, Ramanujam KS, Fu S, Fisher PJ, Rivera MT, Johnson CP, Otterson MF, Telford GL, Wilson KT. Deficient iNOS in inflammatory bowel disease intestinal microvascular endothelial cells results in increased leukocyte adhesion. Free Radic Biol Med. 2000;29:881-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 45] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 87. | Sui C, Wang B, Zhao Y, Guo Y, Zhu J, Yu F, Zhou X, Bu X, Zhang J. Establishment of an inflammatory cytokine-based predictive model for the onset of prolonged postoperative ileus after radical gastrectomy: a prospective cohort study. Front Immunol. 2025;16:1552944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 88. | Ito H, Kimura H, Karasawa T, Hisata S, Sadatomo A, Inoue Y, Yamada N, Aizawa E, Hishida E, Kamata R, Komada T, Watanabe S, Kasahara T, Suzuki T, Horie H, Kitayama J, Sata N, Yamaji-Kegan K, Takahashi M. NLRP3 Inflammasome Activation in Lung Vascular Endothelial Cells Contributes to Intestinal Ischemia/Reperfusion-Induced Acute Lung Injury. J Immunol. 2020;205:1393-1405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 89. | Yu S, Qian H, Zhang D, Jiang Z. Ferulic acid relieved ulcerative colitis by inhibiting the TXNIP/NLRP3 pathway in rats. Cell Biol Int. 2023;47:417-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 90. | Hussain Z, Park H. Inflammation and Impaired Gut Physiology in Post-operative Ileus: Mechanisms and the Treatment Options. J Neurogastroenterol Motil. 2022;28:517-530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 25] [Reference Citation Analysis (1)] |
| 91. | Varanoske AN, McClung HL, Sepowitz JJ, Halagarda CJ, Farina EK, Berryman CE, Lieberman HR, McClung JP, Pasiakos SM, Philip Karl J. Stress and the gut-brain axis: Cognitive performance, mood state, and biomarkers of blood-brain barrier and intestinal permeability following severe physical and psychological stress. Brain Behav Immun. 2022;101:383-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 92. | Boschetti E, Accarino A, Malagelada C, Malagelada JR, Cogliandro RF, Gori A, Tugnoli V, Giancola F, Bianco F, Bonora E, Clavenzani P, Volta U, Caio G, Sternini C, Stanghellini V, Azpiroz F, De Giorgio R. Gut epithelial and vascular barrier abnormalities in patients with chronic intestinal pseudo-obstruction. Neurogastroenterol Motil. 2019;31:e13652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 93. | Sorribas M, de Gottardi A, Moghadamrad S, Hassan M, Spadoni I, Rescigno M, Wiest R. Isoproterenol Disrupts Intestinal Barriers Activating Gut-Liver-Axis: Effects on Intestinal Mucus and Vascular Barrier as Entry Sites. Digestion. 2020;101:717-729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 94. | Keuschnigg J, Henttinen T, Auvinen K, Karikoski M, Salmi M, Jalkanen S. The prototype endothelial marker PAL-E is a leukocyte trafficking molecule. Blood. 2009;114:478-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 95. | Bischoff J. Novel Target for Limiting VEGF-A (Vascular Endothelial Growth Factor A)-Induced Vascular Permeability. Arterioscler Thromb Vasc Biol. 2022;42:1242-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 96. | Huang J, Kelly CP, Bakirtzi K, Villafuerte Gálvez JA, Lyras D, Mileto SJ, Larcombe S, Xu H, Yang X, Shields KS, Zhu W, Zhang Y, Goldsmith JD, Patel IJ, Hansen J, Huang M, Yla-Herttuala S, Moss AC, Paredes-Sabja D, Pothoulakis C, Shah YM, Wang J, Chen X. Clostridium difficile toxins induce VEGF-A and vascular permeability to promote disease pathogenesis. Nat Microbiol. 2019;4:269-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 97. | Yu R, Zhong J, Zhou Q, Ren W, Liu Z, Bian Y. Kaempferol prevents angiogenesis of rat intestinal microvascular endothelial cells induced by LPS and TNF-α via inhibiting VEGF/Akt/p38 signaling pathways and maintaining gut-vascular barrier integrity. Chem Biol Interact. 2022;366:110135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 98. | Barbaro MR, Cremon C, Marasco G, Savarino E, Guglielmetti S, Bonomini F, Palombo M, Fuschi D, Rotondo L, Mantegazza G, Duncan R, di Sabatino A, Valente S, Pasquinelli G, Vergnolle N, Stanghellini V, Collins SM, Barbara G. Molecular Mechanisms Underlying Loss of Vascular and Epithelial Integrity in Irritable Bowel Syndrome. Gastroenterology. 2024;167:1152-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 99. | Bodor C, Nagy JP, Végh B, Németh A, Jenei A, MirzaHosseini S, Sebe A, Rosivall L. Angiotensin II increases the permeability and PV-1 expression of endothelial cells. Am J Physiol Cell Physiol. 2012;302:C267-C276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 100. | Gao SL, Zhang Y, Zhang SY, Liang ZY, Yu WQ, Liang TB. The hydrocortisone protection of glycocalyx on the intestinal capillary endothelium during severe acute pancreatitis. Shock. 2015;43:512-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 101. | Pardo-Camacho C, Ganda Mall JP, Martínez C, Pigrau M, Expósito E, Albert-Bayo M, Melón-Ardanaz E, Nieto A, Rodiño-Janeiro B, Fortea M, Guagnozzi D, Rodriguez-Urrutia A, Torres I, Santos-Briones I, Azpiroz F, Lobo B, Alonso-Cotoner C, Santos J, González-Castro AM, Vicario M. Mucosal Plasma Cell Activation and Proximity to Nerve Fibres Are Associated with Glycocalyx Reduction in Diarrhoea-Predominant Irritable Bowel Syndrome: Jejunal Barrier Alterations Underlying Clinical Manifestations. Cells. 2022;11:2046. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 102. | Langer V, Vivi E, Regensburger D, Winkler TH, Waldner MJ, Rath T, Schmid B, Skottke L, Lee S, Jeon NL, Wohlfahrt T, Kramer V, Tripal P, Schumann M, Kersting S, Handtrack C, Geppert CI, Suchowski K, Adams RH, Becker C, Ramming A, Naschberger E, Britzen-Laurent N, Stürzl M. IFN-γ drives inflammatory bowel disease pathogenesis through VE-cadherin-directed vascular barrier disruption. J Clin Invest. 2019;129:4691-4707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 206] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 103. | Qiu P, Chang Y, Chen X, Wang S, Nie H, Hong Y, Zhang M, Wang H, Xiao C, Chen Y, Liu L, Zhao Q. Dihydroartemisinin Modulates Enteric Glial Cell Heterogeneity to Alleviate Colitis. Adv Sci (Weinh). 2024;11:e2403461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 104. | Greene AK, Alwayn IP, Nose V, Flynn E, Sampson D, Zurakowski D, Folkman J, Puder M. Prevention of intra-abdominal adhesions using the antiangiogenic COX-2 inhibitor celecoxib. Ann Surg. 2005;242:140-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 105. | Zhou G, Zhang M, Zheng S, Yang G, Li L, Huang S, Zeng Z, Chen R, Zhang S, Chen M. Transglutaminase 2 modulates inflammatory angiogenesis via vascular endothelial growth factor receptor 2 pathway in inflammatory bowel disease. J Adv Res. 2025;S2090-1232(25)00500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 106. | Chidlow JH Jr, Shukla D, Grisham MB, Kevil CG. Pathogenic angiogenesis in IBD and experimental colitis: new ideas and therapeutic avenues. Am J Physiol Gastrointest Liver Physiol. 2007;293:G5-G18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 125] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 107. | Zhang G, Qin Q, Zhang C, Sun X, Kazama K, Yi B, Cheng F, Guo ZF, Sun J. NDRG1 Signaling Is Essential for Endothelial Inflammation and Vascular Remodeling. Circ Res. 2023;132:306-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 57] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 108. | Zhang M, Yang H, Wan L, Wang Z, Wang H, Ge C, Liu Y, Hao Y, Zhang D, Shi G, Gong Y, Ni Y, Wang C, Zhang Y, Xi J, Wang S, Shi L, Zhang L, Yue W, Pei X, Liu B, Yan X. Single-cell transcriptomic architecture and intercellular crosstalk of human intrahepatic cholangiocarcinoma. J Hepatol. 2020;73:1118-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 505] [Cited by in RCA: 435] [Article Influence: 72.5] [Reference Citation Analysis (0)] |
| 109. | Wang T, Lian P, Zhan J, Li Y, Liu B, Zhao X, Wu Q, Li H, Lu L, Chen S. The landscape of angiogenesis and inflammatory factors in eyes with myopic choroidal neovascularization before and after anti-VEGF injection. Cytokine. 2024;179:156640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 110. | Zhou S, He Y, Lin J, Yang F, Zhou W, Cai J, Liao Y, Lu F. Brown Adipose Tissue Improves Angiogenesis and M2 Macrophage Polarization in Burn Wounds by Activating Interleukin-17 Signaling. Plast Reconstr Surg. 2025;155:649-658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 111. | Zou J, Fei Q, Xiao H, Wang H, Liu K, Liu M, Zhang H, Xiao X, Wang K, Wang N. VEGF-A promotes angiogenesis after acute myocardial infarction through increasing ROS production and enhancing ER stress-mediated autophagy. J Cell Physiol. 2019;234:17690-17703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 140] [Article Influence: 20.0] [Reference Citation Analysis (2)] |
| 112. | Surman M, Wilczak M, Bzowska M, Tylko G, Przybyło M. The Proangiogenic Effects of Melanoma-Derived Ectosomes Are Mediated by αvβ5 Integrin Rather than αvβ3 Integrin. Cells. 2024;13:1336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 113. | Yuzhuo W, Li L, Shu B, Jing Y, Yongqi W, Zhiwei M, Yi X. Sanguisorba officinalis L. and Sophora japonica L. Inhibit Angiogenesis in Ulcerative Colitis. J Gastroenterol Hepatol. 2025;40:1991-2006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 114. | Zhang Y, Davis C, Shah S, Hughes D, Ryan JC, Altomare D, Peña MM. IL-33 promotes growth and liver metastasis of colorectal cancer in mice by remodeling the tumor microenvironment and inducing angiogenesis. Mol Carcinog. 2017;56:272-287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 126] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 115. | Ardelean DS, Yin M, Jerkic M, Peter M, Ngan B, Kerbel RS, Foster FS, Letarte M. Anti-VEGF therapy reduces intestinal inflammation in Endoglin heterozygous mice subjected to experimental colitis. Angiogenesis. 2014;17:641-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 116. | Jerkic M, Peter M, Ardelean D, Fine M, Konerding MA, Letarte M. Dextran sulfate sodium leads to chronic colitis and pathological angiogenesis in Endoglin heterozygous mice. Inflamm Bowel Dis. 2010;16:1859-1870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 117. | Okhota S, Melnikov I, Avtaeva Y, Kozlov S, Gabbasov Z. Shear Stress-Induced Activation of von Willebrand Factor and Cardiovascular Pathology. Int J Mol Sci. 2020;21:7804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 118. | Sastry S, Cuomo F, Muthusamy J. COVID-19 and thrombosis: The role of hemodynamics. Thromb Res. 2022;212:51-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 119. | Jia Y, Cui R, Wang C, Feng Y, Li Z, Tong Y, Qu K, Liu C, Zhang J. Metformin protects against intestinal ischemia-reperfusion injury and cell pyroptosis via TXNIP-NLRP3-GSDMD pathway. Redox Biol. 2020;32:101534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 273] [Article Influence: 45.5] [Reference Citation Analysis (0)] |
| 120. | Kitai T, Nemet I, Engelman T, Morales R, Chaikijurajai T, Morales K, Hazen SL, Tang WHW. Intestinal barrier dysfunction is associated with elevated right atrial pressure in patients with advanced decompensated heart failure. Am Heart J. 2022;245:78-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 121. | Kalia N, Pockley AG, Wood RF, Brown NJ. Effects of FK409 on intestinal ischemia-reperfusion injury and ischemia-induced changes in the rat mucosal villus microcirculation. Transplantation. 2001;72:1875-1880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 122. | Frösen J, Cebral J, Robertson AM, Aoki T. Flow-induced, inflammation-mediated arterial wall remodeling in the formation and progression of intracranial aneurysms. Neurosurg Focus. 2019;47:E21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 245] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 123. | Koseki H, Miyata H, Shimo S, Ohno N, Mifune K, Shimano K, Yamamoto K, Nozaki K, Kasuya H, Narumiya S, Aoki T. Two Diverse Hemodynamic Forces, a Mechanical Stretch and a High Wall Shear Stress, Determine Intracranial Aneurysm Formation. Transl Stroke Res. 2020;11:80-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 124. | He P, Zhang H, Zhu L, Jiang Y, Zhou X. Leukocyte-platelet aggregate adhesion and vascular permeability in intact microvessels: role of activated endothelial cells. Am J Physiol Heart Circ Physiol. 2006;291:H591-H599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 125. | Staarmann B, Smith M, Prestigiacomo CJ. Shear stress and aneurysms: a review. Neurosurg Focus. 2019;47:E2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 67] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 126. | Wei H, Wang G, Tian Q, Liu C, Han W, Wang J, He P, Li M. Low shear stress induces macrophage infiltration and aggravates aneurysm wall inflammation via CCL7/CCR1/TAK1/ NF-κB axis. Cell Signal. 2024;117:111122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 127. | Zaidi D, Churchill L, Huynh HQ, Carroll MW, Persad R, Wine E. Capillary Flow Rates in the Duodenum of Pediatric Ulcerative Colitis Patients Are Increased and Unrelated to Inflammation. J Pediatr Gastroenterol Nutr. 2017;65:306-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 128. | Bian Y, Liu P, Zhong J, Hu Y, Fan Y, Zhuang S, Liu Z. Kaempferol inhibits multiple pathways involved in the secretion of inflammatory mediators from LPSinduced rat intestinal microvascular endothelial cells. Mol Med Rep. 2019;19:1958-1964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 129. | Mo D, Deng C, Chen B, Ding X, Deng Q, Guo H, Chen G, Ye C, Guo C. The severity of NEC is ameliorated by prostaglandin E2 through regulating intestinal microcirculation. Sci Rep. 2023;13:13395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 130. | Liu P, Bian Y, Fan Y, Zhong J, Liu Z. Protective Effect of Naringin on In Vitro Gut-Vascular Barrier Disruption of Intestinal Microvascular Endothelial Cells Induced by TNF-α. J Agric Food Chem. 2020;68:168-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 131. | Zhang HX, Li YY, Liu ZJ, Wang JF. Quercetin effectively improves LPS-induced intestinal inflammation, pyroptosis, and disruption of the barrier function through the TLR4/NF-κB/NLRP3 signaling pathway in vivo and in vitro. Food Nutr Res. 2022;66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 45] [Reference Citation Analysis (0)] |
| 132. | Carloni S, Bertocchi A, Mancinelli S, Bellini M, Erreni M, Borreca A, Braga D, Giugliano S, Mozzarelli AM, Manganaro D, Fernandez Perez D, Colombo F, Di Sabatino A, Pasini D, Penna G, Matteoli M, Lodato S, Rescigno M. Identification of a choroid plexus vascular barrier closing during intestinal inflammation. Science. 2021;374:439-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 215] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 133. | Bian Y, Dong Y, Sun J, Sun M, Hou Q, Lai Y, Zhang B. Protective Effect of Kaempferol on LPS-Induced Inflammation and Barrier Dysfunction in a Coculture Model of Intestinal Epithelial Cells and Intestinal Microvascular Endothelial Cells. J Agric Food Chem. 2020;68:160-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 99] [Article Influence: 16.5] [Reference Citation Analysis (3)] |
| 134. | Haep L, Britzen-Laurent N, Weber TG, Naschberger E, Schaefer A, Kremmer E, Foersch S, Vieth M, Scheuer W, Wirtz S, Waldner M, Stürzl M. Interferon Gamma Counteracts the Angiogenic Switch and Induces Vascular Permeability in Dextran Sulfate Sodium Colitis in Mice. Inflamm Bowel Dis. 2015;21:2360-2371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 135. | Fang K, Kevil CG. Caveolin-1 Scaffolding Domain Peptide Regulates Colon Endothelial Cell Survival through JNK Pathway. Int J Inflam. 2020;2020:6150942. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 136. | Palenca I, Basili Franzin S, Zilli A, Seguella L, Troiani A, Pepi F, Vincenzi M, Giugliano G, Catapano V, Di Filippo I, Sarnelli G, Esposito G. N-palmitoyl-d-glucosamine limits mucosal damage and VEGF-mediated angiogenesis by PPARα-dependent suppression of pAkt/mTOR/HIF1α pathway and increase in PEA levels in AOM/DSS colorectal carcinoma in mice. Phytother Res. 2024;38:5350-5362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 137. | Behrendt FF, Tolba RH, Overhaus M, Hirner A, Minor T, Kalff JC. Indocyanine green fluorescence measurement of intestinal transit and gut perfusion after intestinal manipulation. Eur Surg Res. 2004;36:210-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 138. | Ming YC, Chao HC, Chu SM, Luo CC. Heparin-binding epidermal growth factor-like growth factor (HB-EGF) protected intestinal ischemia-reperfusion injury through JNK and p38/MAPK-dependent pathway for anti-apoptosis. Pediatr Neonatol. 2019;60:332-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 139. | Zhou Y, Brigstock D, Besner GE. Heparin-binding EGF-like growth factor is a potent dilator of terminal mesenteric arterioles. Microvasc Res. 2009;78:78-85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 140. | Koike Y, Li B, Ganji N, Zhu H, Miyake H, Chen Y, Lee C, Janssen Lok M, Zozaya C, Lau E, Lee D, Chusilp S, Zhang Z, Yamoto M, Wu RY, Inoue M, Uchida K, Kusunoki M, Delgado-Olguin P, Mertens L, Daneman A, Eaton S, Sherman PM, Pierro A. Remote ischemic conditioning counteracts the intestinal damage of necrotizing enterocolitis by improving intestinal microcirculation. Nat Commun. 2020;11:4950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 141. | Lansink MO, Patyk V, de Groot H, Effenberger-Neidnicht K. Melatonin reduces changes to small intestinal microvasculature during systemic inflammation. J Surg Res. 2017;211:114-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |