Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.117422
Revised: January 5, 2026
Accepted: February 10, 2026
Published online: April 27, 2026
Processing time: 139 Days and 0.8 Hours
Pancreaticoduodenectomy has achieved significant clinical advancements, offe
To observe the characteristics of intestinal mucosal cell migration and the changes of inflammatory factors during the pancreaticojejunal anastomosis.
Thirty-six beagle dogs were randomly divided into two groups, namely, the PM-PJ group (n = 18) and the control group (n = 18), which underwent classic end-to-side invagination PJ. Six beagles were sacrificed on postoperative days 7, days 14, and days 21 to obtain PJ tissue. The primary outcomes were postoperative morta
All surgeries were successful. In the study group, the incidence rates of anastomotic leakage and pancreatitis were 0% and 11.1% (2/18), respectively, whereas in the control group, these rates were 5.6% (1/18) and 16.7% (3/18), respectively. Serum levels of transforming growth factor β1, interleukin (IL)-10, tumor necrosis factor-α, and IL-6 were elevated at all postoperative time points in both groups, with more significant increases observed in the study group at certain time points. Pathological examination revealed superior anastomotic healing in the study group at all time points. The expression levels of collagen I/III and alpha smooth muscle actin proteins in the anastomoses of both groups showed temporal changes, with more pronounced variations in the study group at specific time points.
PM-PJ surgery proved safe and reliable, with lower anastomotic leakage vs controls; complications were com
Core Tip: This study investigates the migration of intestinal mucosal cells and the role of inflammatory mediators in pancreaticojejunal anastomosis using a beagle model. The novel pancreaticojejunostomy with mucosal-priority healing technique demonstrates a lower incidence of anastomotic leakage and reduced postoperative complications. Elevated levels of transforming growth factor-β1 and interleukin-6 are observed, indicating their critical role in early healing through collagen synthesis. These findings highlight the benefits of prioritizing mucosal healing and offer insights into optimizing surgical techniques for improved clinical outcomes.
- Citation: Wu SX, Abulimiti P, Zhang BL, Abuduwaili A, Wang C, Li JG, Ba Y, Tuniyazi D, Geng C, Xu XJ. Migration and repair of intestinal mucosal cells in pancreas-intestine anastomosis via inflammatory mediators: A beagle study. World J Gastrointest Surg 2026; 18(4): 117422
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/117422.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.117422
In the past few decades, substantial clinical progress has been made in pancreatoduodenectomy (PD), providing patients with the best long-term survival opportunities. However, even in high-volume centres, 40%-60% of patients experience postoperative complications and 2%-5% of patients are at risk of in-hospital death[1]. Pancreaticojejunostomy (PJ), a key step in PD, is technically challenging because of the soft texture and rich blood supply of the pancreas. Severe postoperative complications, such as pancreatic fistula, bleeding, and death, may occur[2]; thus, its safety of PJ remains a major challenge and is directly related to the success of the operation as well as the patient’s postoperative recovery. A key research area in PJ concerns the necessity of duct-to-mucosa anastomosis[3], which ensures direct drainage of the main pancreatic duct into the intestine. This technique maintains the continuity between the pancreatic duct and jejunal mucosa, facilitates tissue healing, and is consistent with normal physiological processes.
In clinical practice, through analysis of pathological sections obtained during repancreaticojejunostomy in patients with pancreatic duct stenosis after PD, our team observed that the healing process of the PJ anastomosis can be divided into two components: A mucosal connection (inner layer) and a mechanical connection (outer layer). The inner layer involves the healing of jejunal mucosal epithelial cells and pancreatic duct epithelial cells, whereas the outer layer involves the healing of fibrous tissue. Regardless of the PJ method used, the final anastomosis is established between the intestinal mucosa and the epithelial cells of the pancreatic duct. Different anastomotic methods result in different crawling distances of the intestinal mucosa. Based on the crawling distance of the intestinal mucosa, we propose the theory of “mucosal-priority healing during the PJ anastomosis healing process”. On the basis of this theory, our research team has performed a series of PJ surgeries[4]. This study aimed to observe the migration of intestinal mucosal cells at the anastomosis site and to assess how inflammatory factors influence PJ healing. To do so, we compared histological findings from PJ in beagles. The pancreatic anatomy of beagle dogs is similar to that of humans, and their pancreatic duct diameter (1-3 mm) effectively simulates the surgical challenges associated with all small pancreatic ducts in clinical practice.
Healthy 12-month-old female Beagle dogs with a body weight of 9 ± 1 kg were used (Zhenjiang Wanwei Experimental Animal Breeding Co., Ltd., Zhenjiang, Jiangsu Province, China). Before surgery, the dogs were fasted for 6 hours, deprived of water for 12 hours, and intravenously injected with 250 mg of cefotaxime sodium as an antibiotic prophy
A midline laparotomy incision was made in the upper abdomen. The distal jejunum was transected 15-20 cm distal to the ligament of Treitz and sutured intermittently with absorbable sutures. The proximal jejunum was anastomosed end-to-side with the distal end of the transected jejunum to ensure the patency of the gastrointestinal tract. The main pancreatic duct was carefully dissected and transected at the junction of the descending part of the duodenum and the pancreatic duct. The proximal end was ligated, and the other end was dissected and wrapped with the pancreatic duct tissue for the PJ anastomosis. A pancreatic duct stent was placed in the intestine. After PJ, the surgical area was examined for bleeding, and then the abdominal incision was sutured.
This study was approved by the Experimental Animal Ethics Committee of Xinjiang Medical University, approval No. IACUC-20210303-55. All experimental procedures were carried out in accordance with the relevant guidelines and regulations as per approved animal protocols. We confirm that all methods are reported in accordance with the ARRIVE guidelines[5]. Analgesia was administered before and after surgery, the animal’s pain score was monitored daily after surgery, and in accordance with ethical guidelines, euthanasia was performed immediately if obvious signs of distress were observed.
For the animal experiments, 36 dogs were randomly divided into two groups: The study group (n = 18), which under
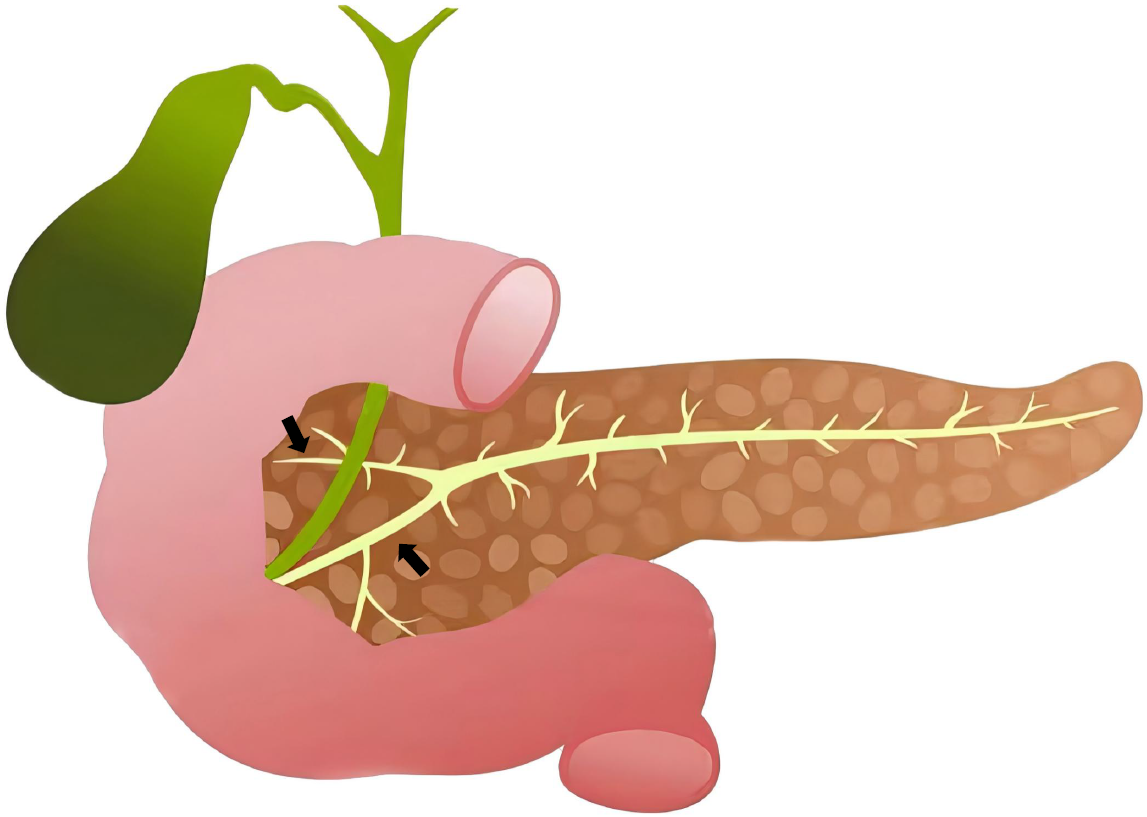
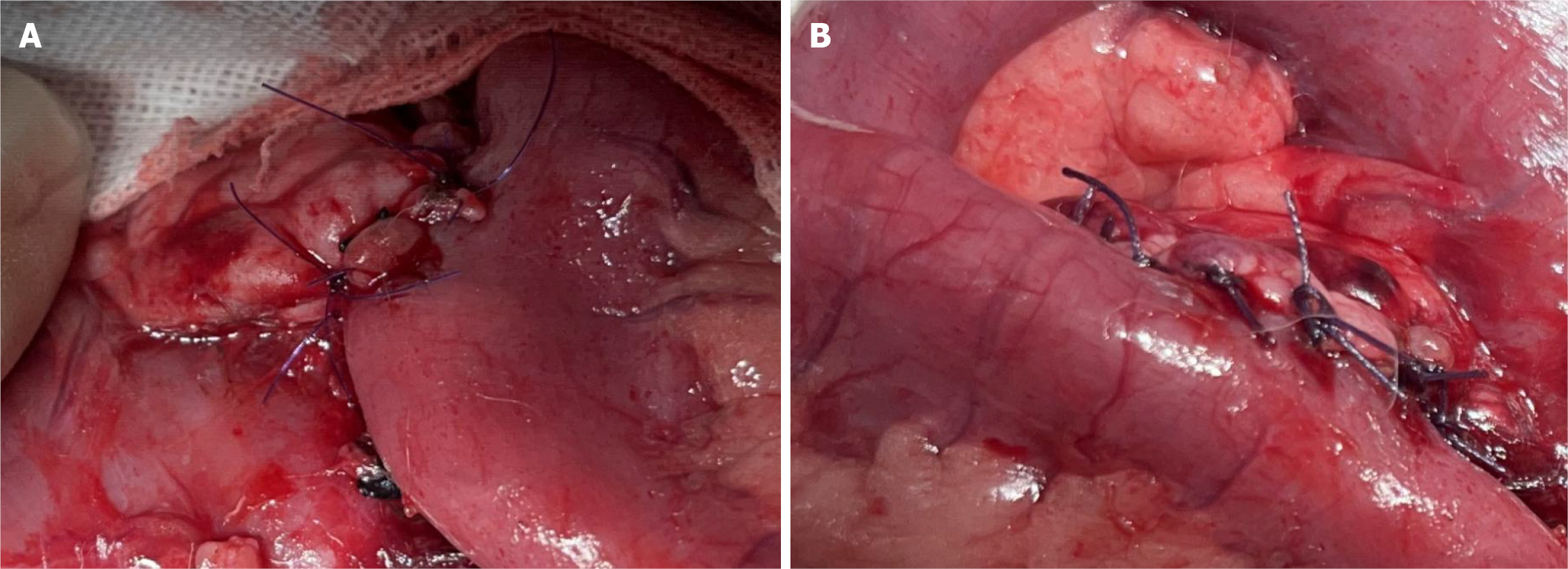
In dogs, the pancreas is a V-shaped organ (Figure 1) consisting of the left lobe, right lobe, and pancreatic body. The diameter of the pancreatic duct is usually small (approximately 1-3 mm). PJ in 9-kg beagles is technically difficult because the diameter of the transected pancreatic duct is approximately 2 mm. In the study group, the jejunum was intermittently sutured to 2 stitches at the upper and lower edges of the pancreatic stump to ensure that the jejunum and the pancreatic stump were in close proximity. Afterwards, an opening similar to that in the pancreatic duct was made in the jejunum, and the seromuscular layer of the jejunal opening was sutured to the pancreatic stump centred on the pancreatic duct. The anterior and posterior walls of the anastomosis were sutured with 5/0 silk threads or 4/0 silk threads, respectively, using 3-5 stitches. A stent was then placed in the pancreatic duct. It was not necessary to achieve perfect mucosa-to-mucosa anastomosis between the pancreatic duct and the jejunum. For small pancreatic ducts, to avoid tearing, it was necessary only to facilitate close migration and connection between the intestinal mucosa and the pancreatic duct epithelium (Figure 2). In the control group, when the PJ was performed, an appropriate position was selected to incise the full thickness of the jejunum. The size of the incision was the same as or slightly smaller than the diameter of the pancreatic stump. The pancreatic stump that had been inserted into the jejunum was sutured intermittently in a single layer. The posterior wall of the pancreatic capsule and the seromuscular layer of the jejunum were sutured intermittently with silk threads. The pancreatic stump was subsequently anastomosed with the full thickness of the jejunum in a circle. Finally, the anterior wall of the pancreatic capsule and the seromuscular layer of the jejunum were sutured intermittently. At a distance of 0.5-1 cm from the anastomosis, the seromuscular layer of the jejunum and the pancreatic parenchyma were continuously buried and sutured. On postoperative days 7, days 14, and days 21, 6 dogs in each group were euthanized by excessive anaesthesia injection (first given tranquilizers and then intravenously injected with excess barbiturates when the beagles entered a quiet state) to obtain PJ anastomosis tissues for corresponding laboratory tests.
The primary outcome indicators were postoperative anastomotic healing and intestinal mucosal migration. The secondary outcome measures were postoperative mortality and morbidity.
Peripheral venous blood was drawn before surgery and before sacrifice to measure fasting blood glucose and serum amylase levels. During the surgical procedure, the texture, shape, and size of the pancreas, diameter of the pancreatic duct, and anastomosis time were recorded. The general conditions of the experimental animals after surgery were recorded, and the healing of the anastomosis at each postoperative stage was observed through gross pathology. The anastomosis tissues at each postoperative stage were fixed in 4% neutral paraformaldehyde for 24 hours and embedded in paraffin. The paraffin-embedded anastomosis tissue samples were cut into 4-mm sections for hematoxylin and eosin (HE) staining, immunohistochemistry, and immunofluorescence staining.
The serum concentrations of transforming growth factor β1 (TGF-β1), interleukin (IL)-10, tumor necrosis factor-α (TNF-α), and IL-6 were detected via ELISA. Serum samples obtained from the dogs were stored at room temperature for 2 hours or at 4 °C overnight. To measure the contents of TGF-β1, IL-10, TNF-α, and IL-6 in the serum, 100 μL of serum or standards of different concentrations were added to each well of the microplate. After dilution, the microplate was covered with a sealing film and incubated for 2 hours. After the cells were washed, enzyme-linked polyclonal antibodies specific for TGF-β1, IL-10, TNF-α, and IL-6 were added to the wells of the microplate, which was subsequently incubated at room temperature for 2 hours. After the samples were washed again with washing buffer, the colour intensity was measured at 450 nm by using a microplate reader. The detection range and sensitivity of the ELISA kits (Wuhan Huamei Biological, CSB-E04639m) were 15.6-1000 pg/mL (sensitivity 4.5 pg/mL) for IL-6, TNF-α, and TGF-β1 and 7.8-500 pg/mL (sensitivity 3.5 pg/mL) for IL-10. The intrabatch coefficient of variation of the kit is < 8%, and the interbatch coefficient of variation is < 12%.
The measurement data of the observed outcome indicators are presented as the means ± SDs. Count data are presented as n (%). Continuous variables following a normal distribution are expressed as the mean ± SD, with intergroup compa
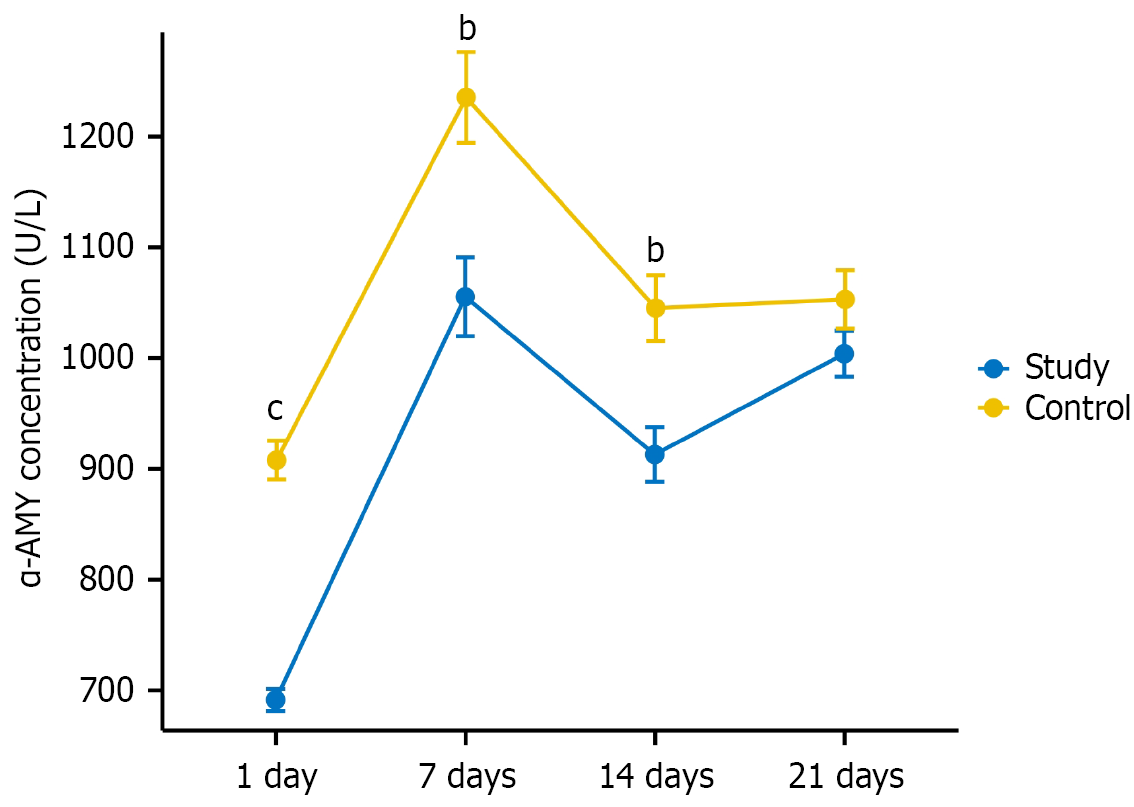
No significant differences were observed in the average body weight, fasting blood glucose level, or serum amylase level between the dogs in the two groups before surgery. No significant differences were observed in the average pancreatic duct diameter, pancreatic texture, or PJ anastomosis time between the two groups (Table 1). The serum amylase levels in the study and control groups at different postoperative time points were as follows: 1 day (691.42 ± 24.34 vs 907.87 ± 42.86); 7 days (1055.42 ± 87.29 vs 1235.41 ± 100.30); 14 days (912.95 ± 60.80 vs 1045.14 ± 72.74); and 21 days (1003.92 ± 51.06 vs 1052.97 ± 64.45). In both groups, the serum amylase levels of the dogs increased within 7 days after surgery. From 7 days to 14 days after surgery, the serum amylase levels in both groups tended to decrease. From 14 days to 21 days after surgery, the levels in the study group tended to increase slowly, whereas the trend in the control group was flat, with no significant change. The serum amylase levels of the two groups were the same but were higher than the preoperative values (Figure 3).
| Characteristics | Study group (n = 18) | Control group (n = 18) | P value | 95%CI |
| Weight (kg) | 9.08 ± 0.56 | 9.03 ± 0.36 | 0.787 | -0.258 to 0.358 |
| FBG (mmol/L) | 4.86 ± 0.24 | 5.06 ± 0.26 | 0.066 | -0.363 to 0.037 |
| Α-AMY (U/L) | 648.3 ± 11.90 | 654.7 ± 13.16 | 0.266 | -14.6 to 1.8 |
| Duct size (mm) | 2.24 ± 0.16 | 2.16 ± 0.08 | 0.164 | -0.002 to 0.162 |
| Soft gland texture, n (%) | 18 (100) | 18 (100) | - | - |
| Operative time (minute), median (IQR) | 31.5 (28.75-33) | 29 (28-31.25) | 0.138 | 0.7-4.3 |
The postoperative mortality, morbidity, and causes of death of the dogs in this study were investigated. The surgeries in both the study and control groups were successfully performed. Dead dogs were included in the intention-to-treat analysis, with their final pathological data carried forward based on the last observed value. Before the specified sacrifice time, two dogs (2/18) in the study group died: One from pulmonary infection and the other from gastric perforation. Three dogs (3/18) in the control group died before the specified sacrifice time. The causes of death were anastomotic leakage in 1 case, postoperative bleeding in another case, and pulmonary infection in the third case. The complication rates of the study and control groups were 5/18 and 7/18, respectively (P > 0.05, χ2 test) (Table 2).
| Characteristics | Study group (n = 18) | Control group (n = 18) | P value | 95%CI |
| Postoperative complications | 5 (27.78) | 7 (38.89) | 0.724 | -0.417 to 0.195 |
| Anastomotic leakage | 0 (0.00) | 1 (5.56) | > 0.999 | -0.161 to 0.050 |
| Pancreatitis | 2 (11.11) | 3 (16.67) | > 0.999 | -0.281 to 0.170 |
| Pulmonary infection | 1 (5.56) | 1 (5.56) | > 0.999 | -0.150 to 0.150 |
| Intestinal infection | 1 (5.56) | 1 (5.56) | > 0.999 | -0.150 to 0.150 |
| Gastric perforation | 1 (5.56) | 0 (0.00) | > 0.999 | -0.050 to 0.161 |
| Hemorrhage | 0 (0.00) | 1 (5.56) | > 0.999 | -0.161 to 0.050 |
| Postoperative death | 2 (11.11) | 3 (16.67) | > 0.999 | -0.281 to 0.170 |
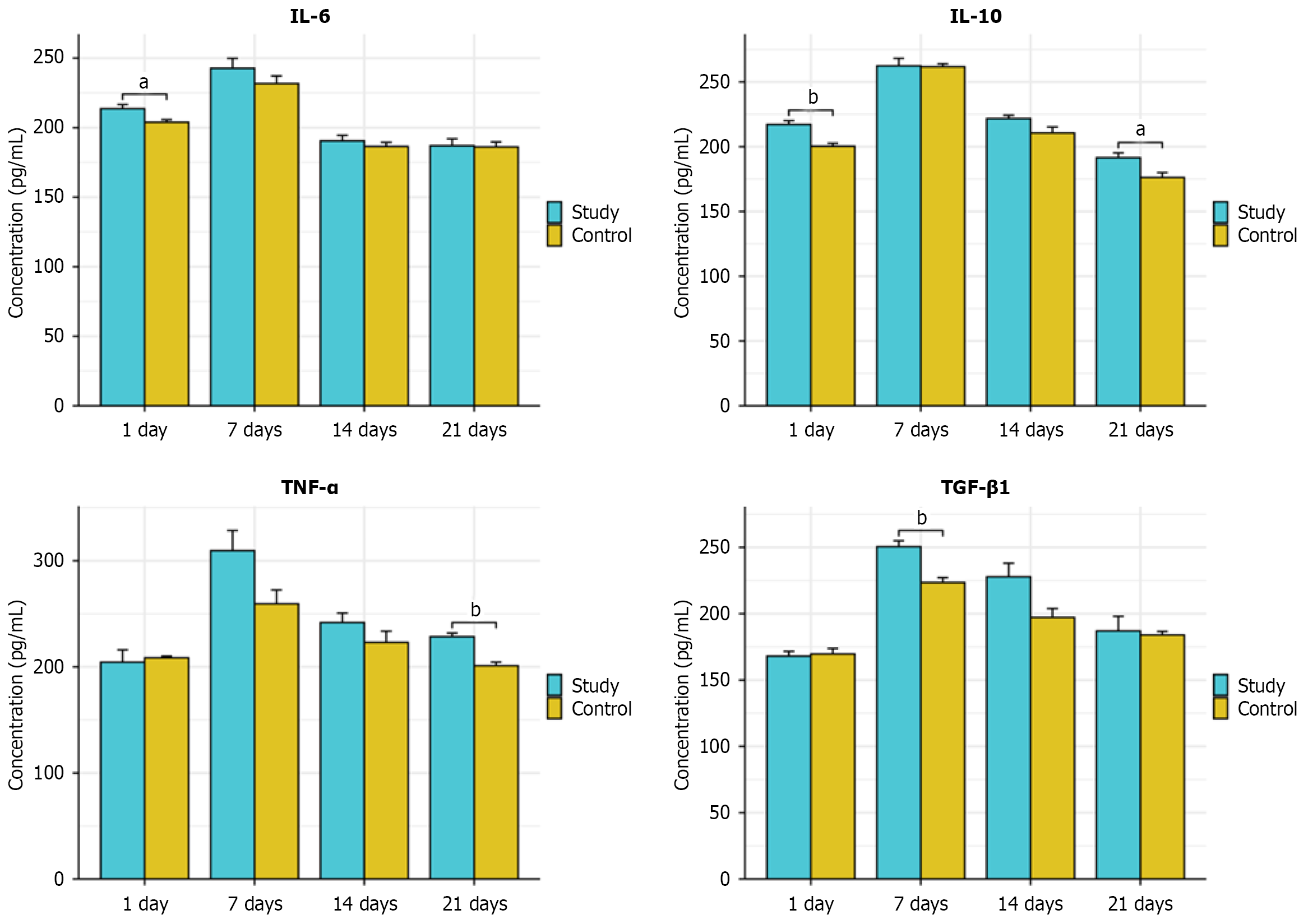
The serum levels of TGF-β1, IL-10, TNF-α, and IL-6 in dogs were detected using ELISA kits. The levels of TGF-β1, IL-10, TNF-α, and IL-6 in both groups increased at all time points. Compared with those in the control group, the increases at some time points in the study group were significant (aP < 0.05; bP < 0.01) (Figure 4).
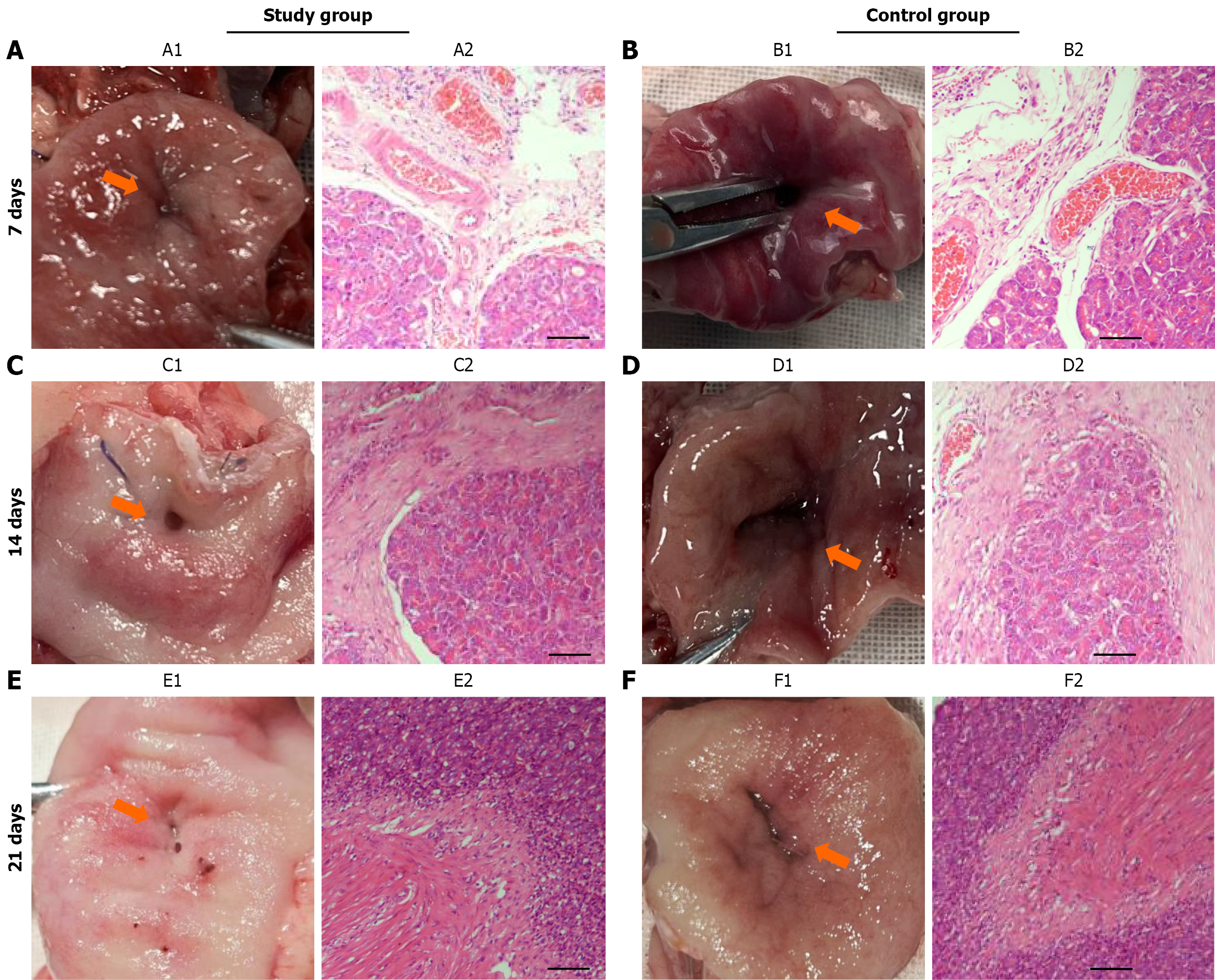
In the study group, the pancreatic tissue in front of the anastomosis was closely adhered to the jejunal surface at the suture site on the 7th day after surgery. Obvious tissue inflammatory reactions and oedema were also observed. The anastomosis was not completely healed, and a small volume of exudate and a large amount of pale-yellow necrotic substance could be seen (Figure 5A). In the control group, the suture site of the anastomosis was slightly everted on the 7th day after surgery. A small amount of necrotic tissue was observed on the pancreatic section side, and oedema was obvious (Figure 5B). The pancreatic ducts in both groups were patent and slightly dilated, and both contained clear pancreatic juice. By the 14th day after surgery, the oedema in both groups had significantly subsided. The suture site of the posterior wall of the anastomosis in the study group became thicker, the necrotic tissue at the anastomosis was sloughed off (Figure 5C), and the anastomosis healed well. HE staining and microscopic examination revealed that the intestinal mucosa and pancreatic duct mucosa at the suture site of the anastomosis were closely connected. A small amount of scar connective tissue was observed under the mucosa (Figure 5C). Moreover, the tissue at the suture site in the control group was thickened (Figure 5D). Microscopy analysis of HE-stained sections revealed that the intestinal mucosal epithelial tissue covered the proliferative connective tissue (Figure 5D). by the 21st day after surgery, the pancreatic cross-section of the anastomosis in the study group was completely covered by the intestinal mucosa. The anterior wall of the PJ anastomosis was everted, and the posterior wall protruded in a semicircular shape (Figure 5E). The anastomosis was obviously scarred, and the pancreatic ducts were dilated to different degrees. The intestinal mucosal cells migrated from the periphery to the centre over an increasingly wide range, where they proliferated and differentiated into the lumen. The degree of cell differentiation was high near the anastomosis and approached that of normal intestinal mucosa. On the 21st day after surgery, in the control group, the pancreatic duct was obviously dilated (Figure 5F). Scar hyperplasia at the anastomosis site was more obvious in the control group than in the study group. The degree of pancreatic inflammation was severe in 2 dogs. Partial mucosal necrosis and a high level of inflammatory cell infiltration were observed via microscopy.
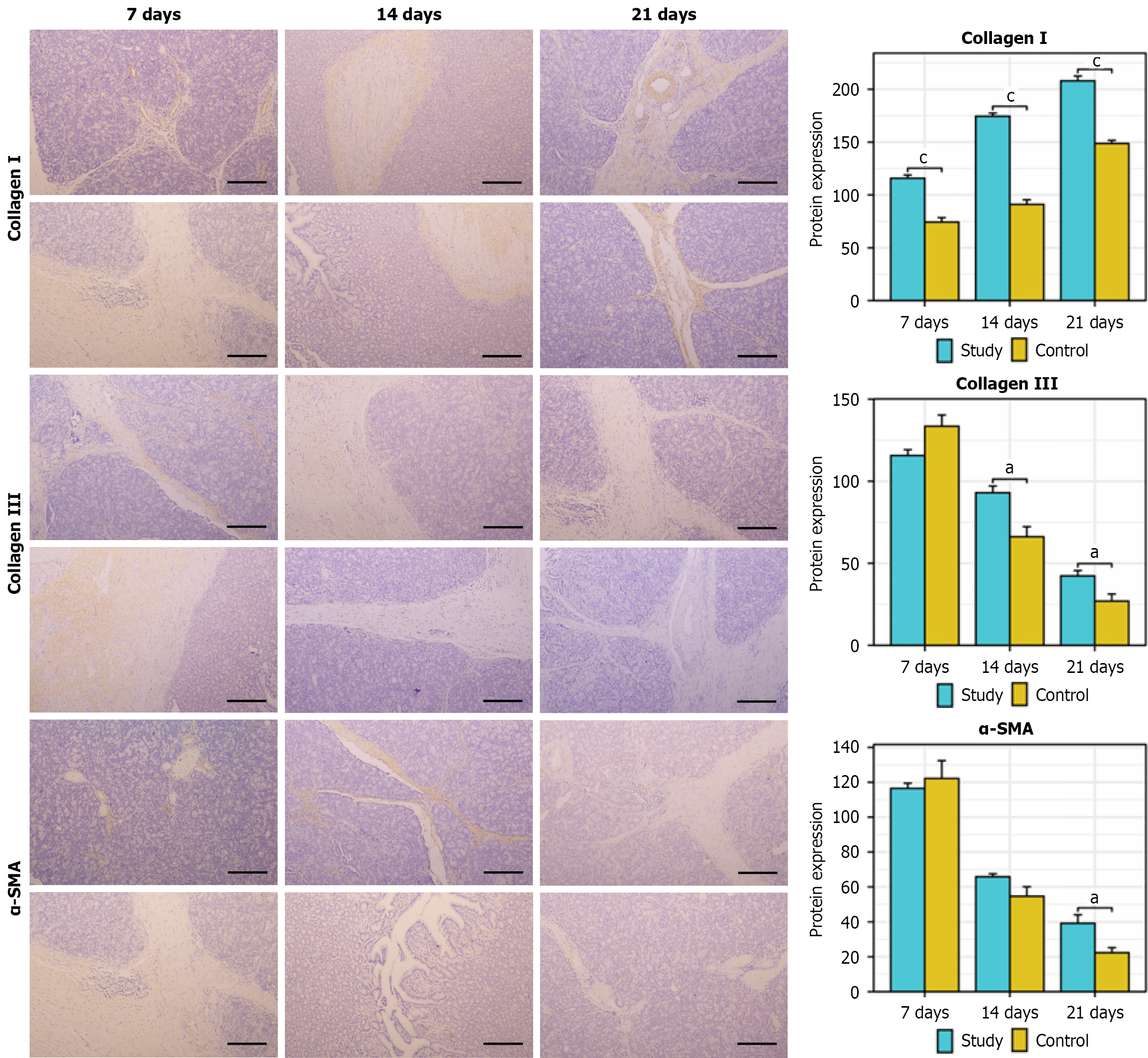
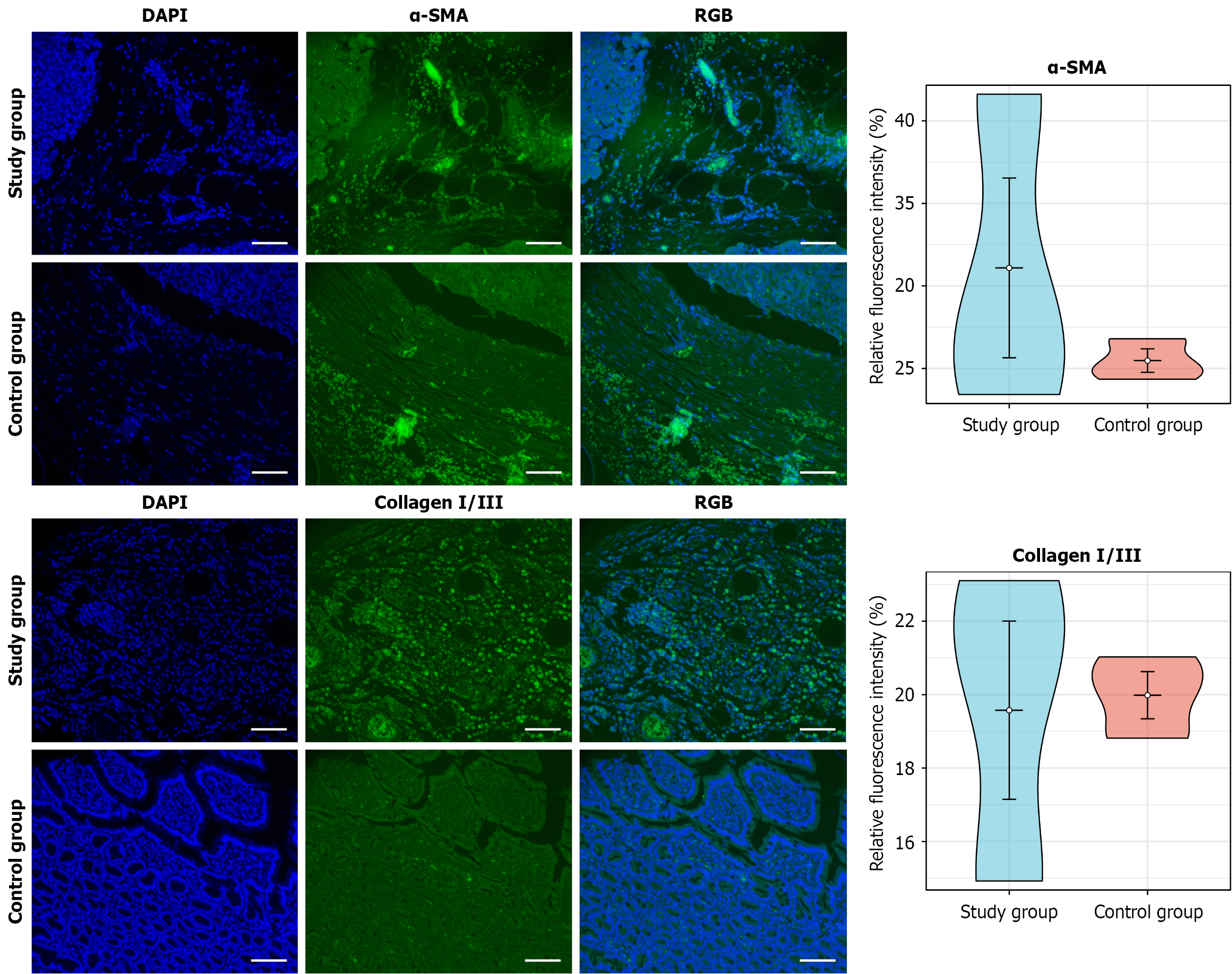
The immunohistochemical results revealed that, at different time points, the protein expression levels of collagen I/III and alpha smooth muscle actin (α-SMA) in the anastomosis were increased in both groups. The collagen I protein level gradually increased over time, whereas the collagen III and α-SMA protein levels gradually decreased over time. Compared with those in the control group, the changes in the protein expression levels of collagen I/III and α-SMA in the study group were significant at some time points (aP < 0.05; cP < 0.001) (Figure 6). Immunofluorescence staining of the PJ anastomosis on the 21st day after surgery revealed no significant difference in the fluorescence intensity of the collagen I/III and α-SMA proteins between the study group and the control group (P > 0.05) (Figure 7). ImageJ software was used for semiquantitative analysis of collagen I/III and α-SMA staining (integral absorbance values), which were indepen
Experimental results confirm that inflammatory factors play a positive role in the early healing of pancreaticojejunal anastomoses by stimulating collagen secretion and synthesis. Moreover, for postpancreatic resection reconstruction, the PM-PJ offers several advantages. First, fewer sutures are required for pancreatic tissue, minimizing trauma to the ana
Animal experiments revealed that in the PM-PJ, the posterior wall of the anastomosis essentially healed within 7 days. While one dog in the control group died of anastomotic leakage, none of the dogs in the study group experienced this adverse event. This may be related to the relatively long free pancreas, which affects the blood supply of the stump and leads to poor local blood supply at the anastomosis site[9]. On the 21st day after surgery, the section of the pancreas at the anastomosis in the dogs in the study group was covered by proliferative mucosal epithelium, and the unsutured anterior wall of the pancreatic duct had healed in a slightly everted state. This typical healing is only possible when healing of the pancreatic section of the anastomosis is delayed. In the early stage of tissue healing, the wound defect is covered by granulation tissue. In the middle stage, this defect contracts under the action of myofibroblasts. Afterwards, epithelial cells grow towards the centre and finally cover the defect. After epithelialization, the growth of granulation tissue is inhibited, and scar hyperplasia stops[10]. In our study, the early stage of anastomosis healing was the inflammatory reaction stage, during which coagulation, haemostasis, inflammatory exudation and neutrophil infiltration occur and inflammatory cells release various inflammatory and tissue growth factors. This process subsequently leads to the tissue-healing stage, in which fibroblasts proliferate, collagen is secreted, capillaries regenerate, and the microcirculation is established. The maturity stage (remodelling) is the stage at which granulation tissue is remodelled. Due to the unique anatomical structure of the pancreas and intestine and the presence of pancreatic juice, the repair and healing processes after PJ occur according to their own principles and characteristics. When the defect is covered by proliferative mucosal epithelial tissue, the entire anastomosis is epithelialized, thereby preventing further scar hyperplasia. Moreover, the proliferative mucosal epithelium protects the anastomosis tissue from the corrosive effect of pancreatic juice and prevents bleeding caused by tissue corrosion.
During the 21 days of the animal experiment, the migration and differentiation of intestinal mucosal cells were observed in the anastomosis, and the serum levels of inflammatory factors, collagen and α-SMA in the anastomosis increased to different degrees. No difference in immunofluorescence was detected between the two groups at 21 days after surgery, which may be related to the stabilization of collagen remodeling in the late healing stage, whereas the difference in immunohistochemistry at the early stage (7-14 days) reflected the different repair kinetics in the inflammatory stage. This study revealed that prioritized mucosal healing during PM-PJ surgery can shorten the migration distance of epithelial cells, which is consistent with the intestinal stem cell migration model proposed by Azkanaz et al[11]. Moreover, the peaks of TGF-β1 and IL-6 precede the collagen deposition peak, suggesting that they may activate fibroblasts through the Janus kinases/signal transducer and activator of transcription pathway[12]. In the early stage, platelet degranulation provides certain signals that initiate various aspects of the repair process, including the recruit
In this study, although we explored the mechanism by which inflammatory factors affect the migration and repair of intestinal mucosal cells at the site of the PJ anastomosis, the use of animals in research inevitably has several limitations. First, because of the small diameter of the pancreatic duct in Beagle dogs, pancreatic duct jejunostomy is very difficult to perform. Second, a small sample size may lead to type II errors, such as a failure to achieve statistical significance in terms of the difference in complication rates (power less than 45%). Finally, in this study, only the effects of inflammatory factors on anastomosis healing were observed within 21 days after surgery, which may not be sufficient to fully capture the entire process of anastomosis healing and its long-term effects. Future studies should conduct multicentre large-sample research and utilize gene knockout animal models to further explore the role of the Janus kinases/signal transducer and activator of transcription and TGF-β signaling pathways in mucosal repair.
This study revealed that inflammatory factors can stimulate collagen secretion and synthesis during the early healing stage of pancreaticojejunal anastomosis. The advantages of the PM-PJ procedure in animal models include a reduced incidence of anastomotic leakage, postoperative bleeding, and mortality. However, further validation of its long-term safety is needed before its clinical application.
| 1. | Smits FJ, Verweij ME, Daamen LA, van Werkhoven CH, Goense L, Besselink MG, Bonsing BA, Busch OR, van Dam RM, van Eijck CHJ, Festen S, Koerkamp BG, van der Harst E, de Hingh IH, Kazemier G, Klaase JM, van der Kolk M, Liem M, Luyer MDP, Meerdink M, Mieog JSD, Nieuwenhuijs VB, Roos D, Schreinemakers JM, Stommel MW, Wit F, Zonderhuis BM, de Meijer VE, van Santvoort HC, Molenaar IQ; Dutch Pancreatic Cancer Group. Impact of Complications After Pancreatoduodenectomy on Mortality, Organ Failure, Hospital Stay, and Readmission: Analysis of a Nationwide Audit. Ann Surg. 2022;275:e222-e228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 2. | Martin AN, Narayanan S, Turrentine FE, Bauer TW, Adams RB, Zaydfudim VM. Pancreatic duct size and gland texture are associated with pancreatic fistula after pancreaticoduodenectomy but not after distal pancreatectomy. PLoS One. 2018;13:e0203841. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (5)] |
| 3. | Zhang S, Lan Z, Zhang J, Chen Y, Xu Q, Jiang Q, Zhao Y, Wang C, Bi X, Huang X. Duct-to-mucosa versus invagination pancreaticojejunostomy after pancreaticoduodenectomy: a meta-analysis. Oncotarget. 2017;8:46449-46460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 4. | Wu S, Li J, Zhang B, Wang C, Abuduwaili A, Zhang J, Zhang S, Geng C, Xu X. Comparison of pancreaticojejunostomy under the theory of mucosal priority healing with duct-to-mucosa anastomosis and invagination pancreaticojejunostomy after pancreaticoduodenectomy: A single-centre case-control study. Sci Prog. 2025;108:368504251345016. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 5. | Percie du Sert N, Ahluwalia A, Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl U, Emerson M, Garner P, Holgate ST, Howells DW, Hurst V, Karp NA, Lazic SE, Lidster K, MacCallum CJ, Macleod M, Pearl EJ, Petersen OH, Rawle F, Reynolds P, Rooney K, Sena ES, Silberberg SD, Steckler T, Würbel H. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol. 2020;18:e3000411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 835] [Cited by in RCA: 1629] [Article Influence: 271.5] [Reference Citation Analysis (0)] |
| 6. | Casciani F, Bassi C, Vollmer CM Jr. Decision points in pancreatoduodenectomy: Insights from the contemporary experts on prevention, mitigation, and management of postoperative pancreatic fistula. Surgery. 2021;170:889-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 7. | Bai X, Zhang Q, Gao S, Lou J, Li G, Zhang Y, Ma T, Zhang Y, Xu Y, Liang T. Duct-to-Mucosa vs Invagination for Pancreaticojejunostomy after Pancreaticoduodenectomy: A Prospective, Randomized Controlled Trial from a Single Surgeon. J Am Coll Surg. 2016;222:10-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 8. | Li J, Xiang J, Zhu J, Wang M, Lin M, Wang H, Li H. A new method of pancreaticojejunostomy in laparoscopic pancreaticoduodenectomy: A retrospective analysis of 93 cases. J Minim Access Surg. 2025;21:34-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Daamen LA, Smits FJ, Besselink MG, Busch OR, Borel Rinkes IH, van Santvoort HC, Molenaar IQ; Dutch Pancreatic Cancer Group. A web-based overview, systematic review and meta-analysis of pancreatic anastomosis techniques following pancreatoduodenectomy. HPB (Oxford). 2018;20:777-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 10. | Martin P, Pardo-Pastor C, Jenkins RG, Rosenblatt J. Imperfect wound healing sets the stage for chronic diseases. Science. 2024;386:eadp2974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 119] [Article Influence: 59.5] [Reference Citation Analysis (0)] |
| 11. | Azkanaz M, Corominas-Murtra B, Ellenbroek SIJ, Bruens L, Webb AT, Laskaris D, Oost KC, Lafirenze SJA, Annusver K, Messal HA, Iqbal S, Flanagan DJ, Huels DJ, Rojas-Rodríguez F, Vizoso M, Kasper M, Sansom OJ, Snippert HJ, Liberali P, Simons BD, Katajisto P, Hannezo E, van Rheenen J. Retrograde movements determine effective stem cell numbers in the intestine. Nature. 2022;607:548-554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 50] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 12. | Makabe K, Okada H, Tachibana N, Ishikura H, Ito N, Tanaka M, Chijimatsu R, Terashima A, Yano F, Asaka M, Yanagihara D, Taketomi S, Matsumoto T, Tanaka S, Omata Y, Saito T. Baricitinib ameliorates inflammatory and neuropathic pain in collagen antibody-induced arthritis mice by modulating the IL-6/JAK/STAT3 pathway and CSF-1 expression in dorsal root ganglion neurons. Arthritis Res Ther. 2024;26:121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 37] [Reference Citation Analysis (0)] |
| 13. | Wang M, Li X, Wang Q, Zhang M, He J, Ming S, Wang Z, Cao C, Zhang S, Geng L, Gong S, Huang X, Chen K, Wu Y. TLT-1 Promotes Platelet-Monocyte Aggregate Formation to Induce IL-10-Producing B Cells in Tuberculosis. J Immunol. 2022;208:1642-1651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Metelli A, Salem M, Wallace CH, Wu BX, Li A, Li X, Li Z. Immunoregulatory functions and the therapeutic implications of GARP-TGF-β in inflammation and cancer. J Hematol Oncol. 2018;11:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 92] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 15. | Scopelliti F, Cattani C, Dimartino V, Mirisola C, Cavani A. Platelet Derivatives and the Immunomodulation of Wound Healing. Int J Mol Sci. 2022;23:8370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 16. | Zeng YY, Hu WP, Zuo YH, Wang XR, Zhang J. Altered serum levels of type I collagen turnover indicators accompanied by IL-6 and IL-8 release in stable COPD. Int J Chron Obstruct Pulmon Dis. 2019;14:163-168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 17. | Martin P, Leibovich SJ. Inflammatory cells during wound repair: the good, the bad and the ugly. Trends Cell Biol. 2005;15:599-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 883] [Cited by in RCA: 876] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 18. | Mori R, Shaw TJ, Martin P. Molecular mechanisms linking wound inflammation and fibrosis: knockdown of osteopontin leads to rapid repair and reduced scarring. J Exp Med. 2008;205:43-51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 234] [Article Influence: 13.0] [Reference Citation Analysis (3)] |
| 19. | Shook BA, Wasko RR, Rivera-Gonzalez GC, Salazar-Gatzimas E, López-Giráldez F, Dash BC, Muñoz-Rojas AR, Aultman KD, Zwick RK, Lei V, Arbiser JL, Miller-Jensen K, Clark DA, Hsia HC, Horsley V. Myofibroblast proliferation and heterogeneity are supported by macrophages during skin repair. Science. 2018;362:eaar2971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 421] [Cited by in RCA: 412] [Article Influence: 51.5] [Reference Citation Analysis (4)] |