Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116578
Revised: December 30, 2025
Accepted: February 9, 2026
Published online: April 27, 2026
Processing time: 147 Days and 1.6 Hours
Hirschsprung’s disease (HD) is a congenital intestinal malformation defined by intestinal aganglionosis. Persistent spasm of the aganglionic segment is commonly followed by fecal retention and impaction, with consequent proximal colonic dilation and wall thickening. The only established curative treatment is surgery; nevertheless, postoperative Hirschsprung-associated enterocolitis (HAEC) occurs in approximately 30% of affected children, compromising physiological recovery and causing potentially life-threatening morbidity. Postoperative HAEC has been linked to impaired intestinal barrier function, gut microbial dysbiosis and im
To characterize the gut microbiota and serum levels of intestinal fatty acid-binding protein (I-FABP)/peroxiredoxin 1 (PRDX1) in children with HAEC fol
This retrospective study included 334 children with HD who underwent surgery at our hospital between March 2022 and March 2025. Patients were categorized into two groups based on the occurrence of HAEC within 3 months postoperatively: The HAEC group (n = 109) and the non-HAEC group (n = 225). The two groups were compared in terms of general characteristics, preoperative gut microbiota α-diversity indices, phylum and genus abundances, and serum levels of C-reactive protein (CRP), tumor necrosis factor-alpha (TNF-α), interleukin-6
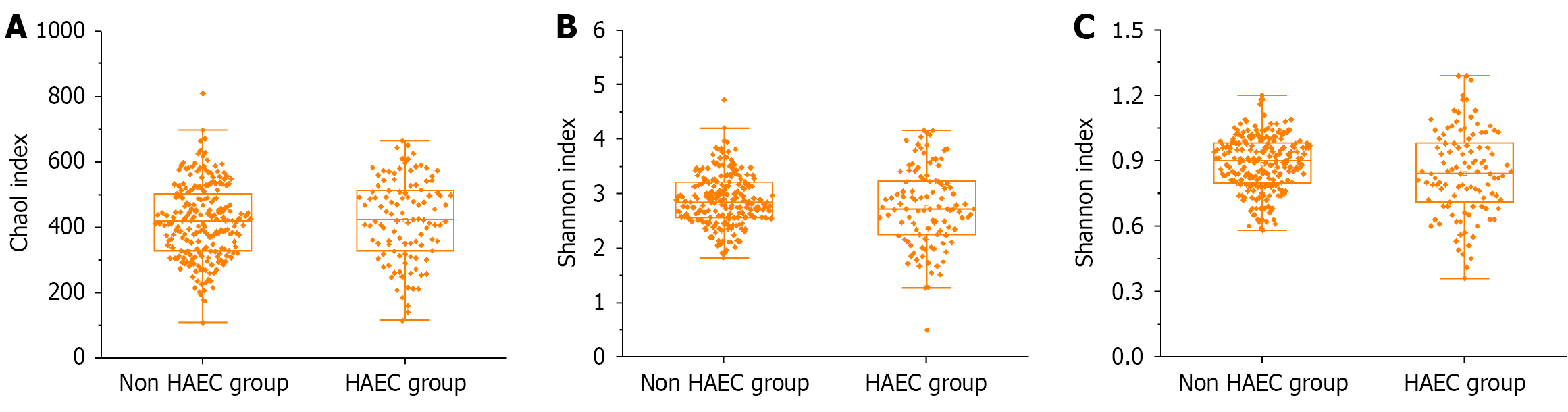
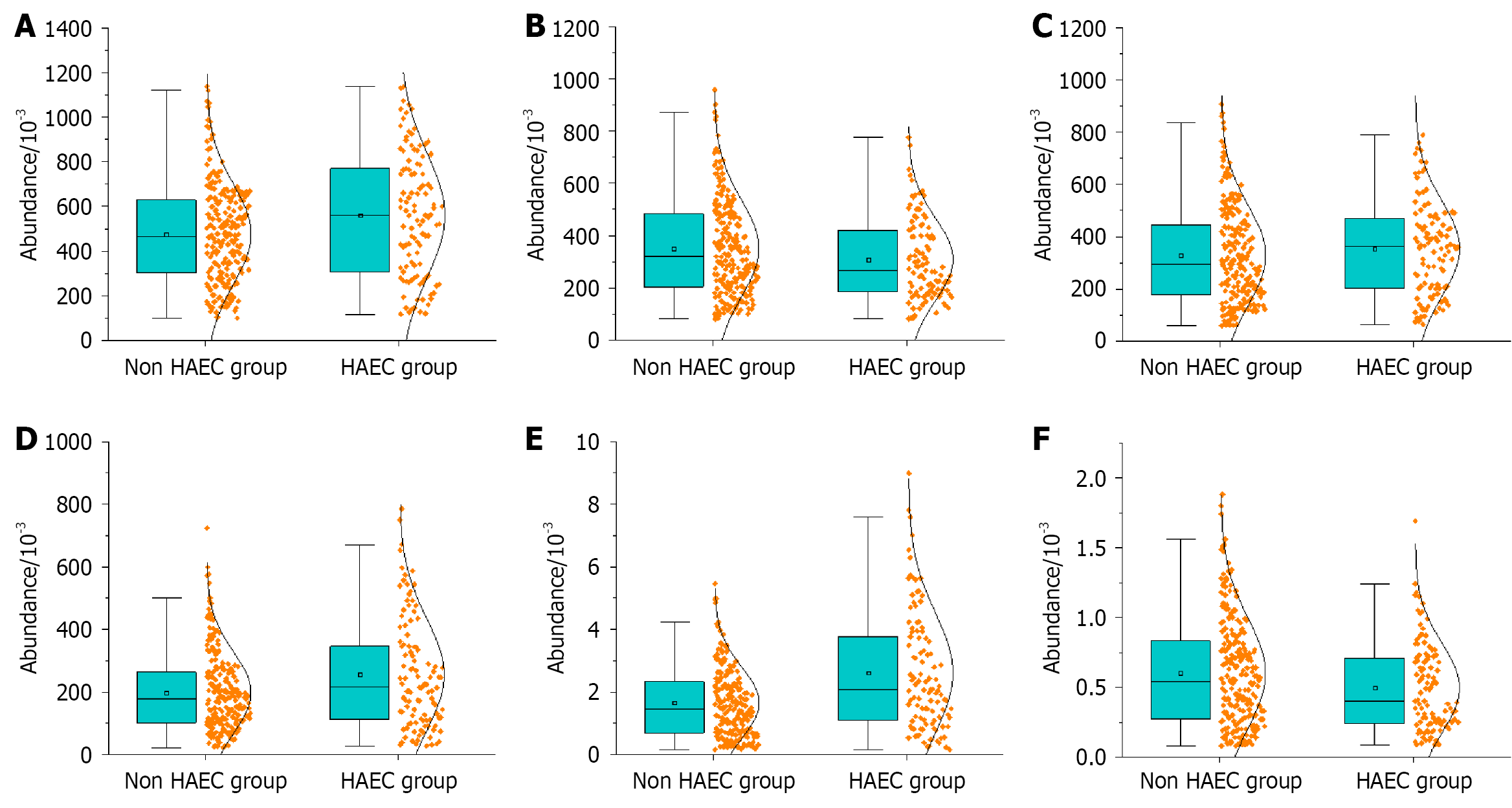
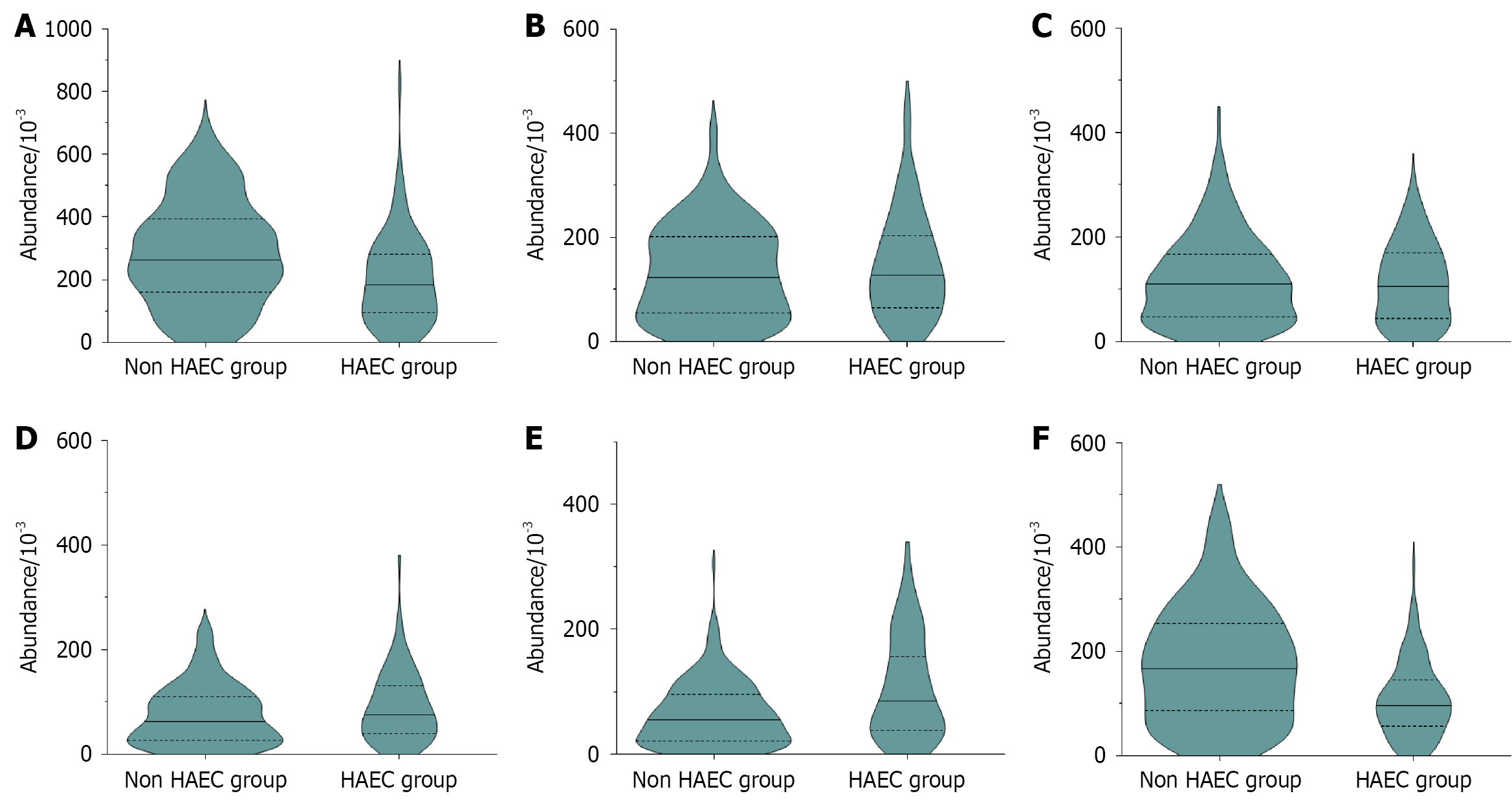
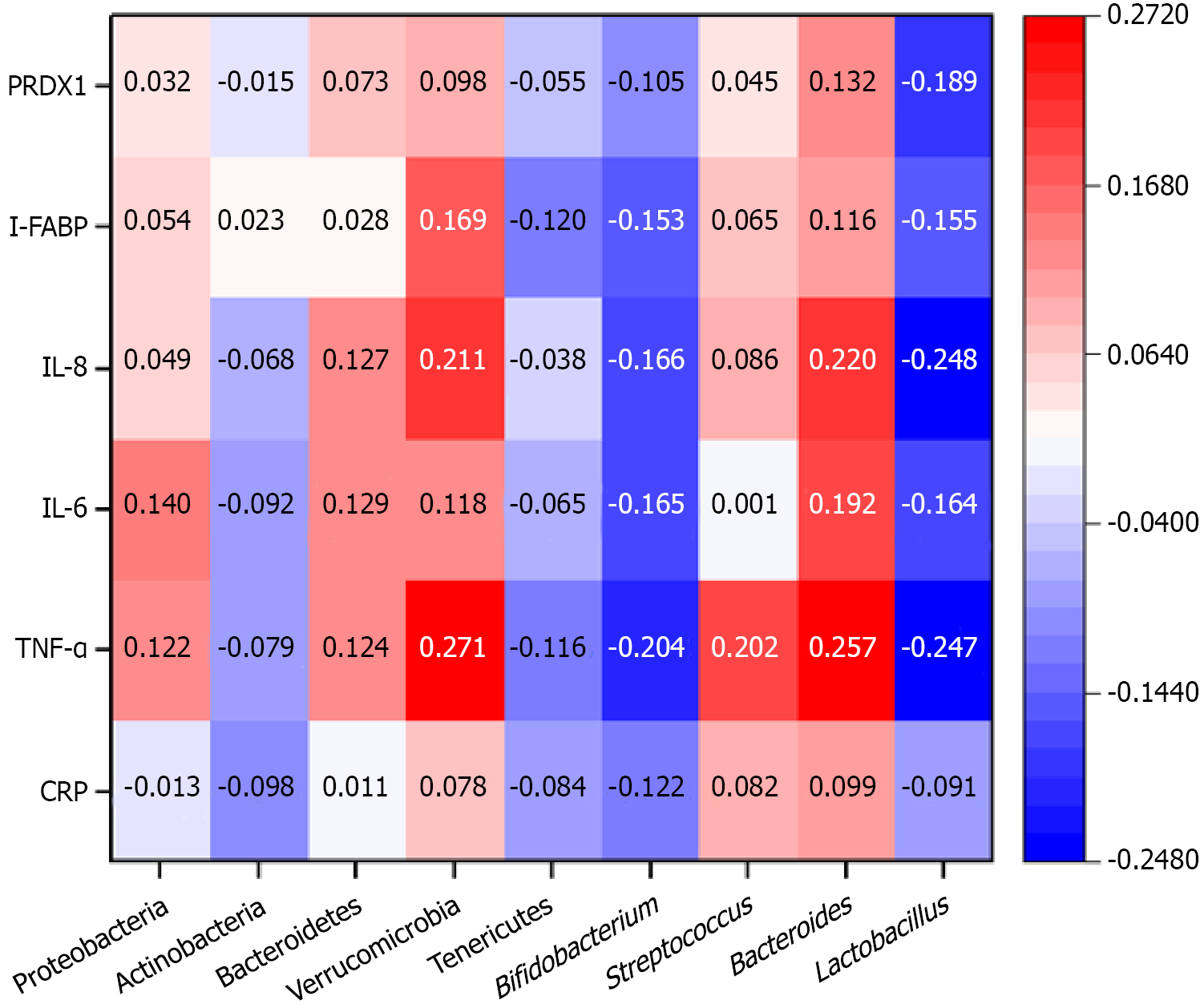
The HAEC group was characterized by a longer aganglionic segment and a higher incidence of preoperative enteritis and postoperative intestinal obstruction than the non-HAEC group (P < 0.05). No significant between-group differences were observed in gut microbiota α-diversity, as assessed by the Chao1, Shannon and Simpson indices (P > 0.05). By contrast, relative abundances of Proteobacteria, Bacteroidetes, Verrucomicrobia, Streptococcus and Bacteroides were increased, whereas those of Actinobacteria, Tenericutes, Bifidobacterium and Lactobacillus were decreased, in the HAEC group compared with the non-HAEC group (P < 0.05). Preoperative serum concentrations of CRP, TNF-α, IL-6, IL-8, I-FABP and PRDX1 were also significantly elevated in the HAEC group (P < 0.05). Correlation analysis demonstrated negative associations between these serum markers and the abundance of beneficial bacteria, alongside positive associations with pathogenic bacterial abundance. Multivariable regression analysis identified serum CRP, I-FABP and PRDX1 concentrations as independent risk factors for postoperative HAEC, whereas Bifidobacterium abundance emerged as an independent protective factor. On this basis, a nomogram prediction model was constructed incorporating preoperative abundances of Verrucomicrobia, Bacteroides and Bifidobacterium, together with serum CRP, I-FABP and PRDX1 levels. The model showed excellent discriminative performance, with an area under the receiver operating characteristic curve of 0.951 (95%CI: 0.927-0.974) and good calibration (Hosmer-Lemeshow χ2 = 3.573, P = 0.893).
Children who develop HAEC following HD surgery exhibit a characteristic preoperative profile of reduced beneficial bacteria, increased pathogenic bacteria, and elevated levels of CRP, TNF-α, IL-6, IL-8, I-FABP, and PRDX1. Preoperative levels of these serum markers were significantly correlated with gut microbiota abundance. Furthermore, the risk of postoperative HAEC was strongly associated with preoperative gut microbiota structure as well as serum I-FABP and PRDX1 levels.
Core Tip: This retrospective study examined the characteristics of, and correlations between, preoperative gut microbiota and serum intestinal fatty acid-binding protein (I-FABP) and peroxiredoxin 1 (PRDX1) levels in children who developed Hirschsprung-associated enterocolitis (HAEC) after Hirschsprung’s disease (HD) surgery, with the aim of informing risk assessment and preventive intervention strategies. Preliminary analyses indicated that children with postoperative HAEC exhibited decreased abundance of beneficial bacteria, increased abundances of pathogenic bacteria and elevated preoperative levels of C-reactive protein (CRP), tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), interleukin-8 (IL-8), I-FABP and PRDX1. Preoperative levels of CRP, TNF-α, IL-6, IL-8, I-FABP and PRDX1 were significantly correlated with gut microbiota abundance, and the risk of HAEC after HD was strongly associated with preoperative gut microbiota structure and with I-FABP and PRDX1 levels.
- Citation: Sun HL, Cui YT, Huo BL, Zheng XC, Kang WL. Analysis of gut microbiota and serum I-FABP and PRDX1 levels in children with enterocolitis after congenital megacolon surgery. World J Gastrointest Surg 2026; 18(4): 116578
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116578.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116578
Hirschsprung’s disease (HD) is a congenital intestinal disorder characterized by the absence of ganglion cells in the intestinal tract. This defect often results in persistent intestinal spasms, fecal retention, and subsequent hypertrophy and dilation of the proximal colon[1]. Surgical intervention remains the only definitive treatment for HD; however, approximately 30% of children experience postoperative Hirschsprung-associated enterocolitis (HAEC), a life-threatening complication that can impair functional recovery and even be life-threatening[2,3]. Existing research indicates that the occurrence of HAEC after HD surgery is associated with multiple factors, including impaired intestinal barrier function, gut microbiota dysbiosis, and dysregulated immune responses, although the precise pathogenesis remains unclear[4,5]. While some studies have reported that prophylactic probiotics can decrease the incidence and severity of postoperative HAEC[6,7], others have found no significant preventive or therapeutic benefits[8,9]. Consequently, the relationship between postoperative HAEC and gut microbiota structure, as well as the potential benefits of prophylactic probiotic administration, requires further investigation. Intestinal fatty acid-binding protein (I-FABP) is a low-molecular-weight protein that is predominantly located in the epithelial cells of the small intestine. It is rapidly released into the bloodstream following epithelial cell damage or necrosis caused by ischemia, infection, or inflammation and serves as a specific serum biomarker of intestinal mucosal injury[10]. Peroxiredoxin 1 (PRDX1) is an antioxidant protein widely expressed in human cells, protects cells from oxidative stress, and plays a key role in regulating cell proliferation, differentiation, and apoptosis[11]. Previous studies have shown significantly elevated serum levels of I-FABP and PRDX1 in children who develop HAEC following HD surgery[12]. Nevertheless, the relationships between the gut microbiota, I-FABP, and PRDX1, and their collective association with postoperative HAEC remain poorly understood. Therefore, this retrospective study aimed to characterize the preoperative gut microbiota and serum I-FABP/PRDX1 levels in children with postoperative HAEC, analyze their correlations, and offer evidence to support risk assessment and preventive strategies.
This retrospective study utilized logistic regression analysis to evaluate the association between gut microbiota, serum I-FABP, and PRDX1 levels, and the risk of postoperative HAEC in children with HD. The sample size was determined according to the events-per-variable (EPV) criterion for logistic regression using the formula: n = (EPV × m)/P, where n represents the required total sample size, EPV denotes the number of events required per independent variable, m is the number of independent variables, and P is the expected overall event rate. With reference to the relevant literature, the EPV was set at 10, and the postoperative HAEC incidence was assumed to be 30%[2-5,13]. Considering nine independent variables for inclusion in the logistic regression analysis, the minimum required sample size was 300. After accounting for an estimated 10% attrition rate, a total of 334 participants were targeted for enrollment.
We retrospectively enrolled 334 pediatric patients who underwent surgical treatment for HD at our hospital between March 2022 and March 2025. Based on the occurrence of HAEC within 3 months postoperatively, the patients were categorized into HAEC and non-HAEC groups. This study was approved by the Ethics Committee of Hengshui People's Hospital (Approval No. AF/SC-08/02.0). The inclusion criteria were as follows: (1) Aged 1-3 years; (2) Confirmed diagnosis of HD; (3) Undergoing primary surgical treatment; (4) Absence of HAEC within 6 months before surgery; (5) No use of medications affecting gut microbiota within 1 month before surgery; (6) Underwent laparoscopic modified Soave pull-through procedure; and (7) Underwent preoperative 16S rRNA gene high-throughput sequencing of gut microbiota. The exclusion criteria were as follows: (1) Presence of major organ dysfunction; (2) Hematological disorders; (3) History of major previous surgery; (4) Presence of other congenital disorders; (5) Postoperative pathological confir
Surgical procedure for laparoscopy-assisted modified Soave pull-through: Pneumoperitoneum was established. The Veress needle was then withdrawn and replaced with a trocar to create an observation port for the laparoscope. Two additional operating ports were placed on the left and right sides of the umbilical ring to accommodate the ultrasonic scalpel and grasping forceps. The aganglionic segments of the bowel were identified laparoscopically, and the mesenteric vessels of the sigmoid colon and upper rectum were dissected and divided. The affected bowel segment was mobilized along the rectal mesentery to a level below the peritoneal reflection of the pelvic floor, extending to the rectoanal junction. An anal retractor was used to dilate the anus, and a circumferential incision was made in the rectal mucosa, approximately 5-10 mm above the dentate line. Dissection proceeded cephalad in the plane between the rectal mucosa and muscular sheath until the abdominal and perineal dissection planes met. The mobilized aganglionic colon was then delivered telescopically through the muscular sheath and exteriorized through the anus. Finally, the dilated and hypertrophied aganglionic colon was resected. The well-perfused proximal segment of the normal colon was anasto
All enrolled children were followed up for 3 months postoperatively. HAEC was diagnosed based on the diagnostic and grading criteria issued by the American Pediatric Surgical Association[14]. This scoring system integrates clinical manifestations (such as abdominal distension, vomiting, fever, diarrhea, foul-smelling watery stools, or bloody stools), physical signs (e.g., explosive release of gas or loose stools during digital rectal examination), imaging findings (abdominal plain films showing intestinal dilatation and air-fluid levels indicative of distal intestinal obstruction), and laboratory results (e.g., elevated white blood cell count). A total score of 10 or higher was required to confirm the diagnosis of HAEC.
General patient data, including age, sex, body mass index (BMI), aganglionic segment length, pathological type, history of enteritis, postoperative intestinal obstruction, and postoperative wound infections, were collected.
Gut microbiota profiling was performed using high-throughput 16S rRNA gene sequencing of fecal samples to analyze the microbial community structure, α-diversity, and relative abundance of the major bacterial phyla and genera. The specific procedure is as follows: (1) Sample collection and DNA extraction: Fresh fecal samples were collected preoperatively from the children. Total microbial DNA was extracted, and its integrity and concentration were assessed for quality control using agarose gel electrophoresis and a Nanodrop spectrophotometer; (2) PCR amplification and library preparation: The V3-V4 hypervariable regions of the 16S rRNA gene were amplified by PCR using specific barcoded primers. The reaction mixture contained high-fidelity DNA polymerase, dNTPs, and template DNA. The amplification protocol consisted of initial denaturation at 95 °C for 5 minutes; 25-30 cycles of denaturation at 95 °C (30 seconds), annealing at 55 °C (30 seconds), and extension at 72 °C (45 seconds); followed by a final extension at 72 °C for 10 minutes. The amplified products were purified and recovered, and sequencing libraries were constructed using the Illumina MiSeq platform; (3) High-throughput sequencing: The prepared libraries were subjected to paired-end sequencing (2 × 250 bp) using the Illumina platform. A minimum of 10000 high-quality sequences per sample was used for the downstream analysis; and (4) Bioinformatic analysis: Raw sequencing data were filtered to eliminate low-quality reads and adapter contaminants. The UPARSE algorithm was employed to cluster sequences with ≥ 97% similarity into operational taxonomic units (OTUs). To ensure comparability across samples, sequencing depth was normalized (rarefied) to 30000 reads per sample, corresponding to the minimum effective read count across all samples. Representative sequences from each OTU were taxonomically annotated against the SILVA 16S rRNA reference database, enabling classification at the phylum, class, order, family, and genus levels. Statistical analysis focused on relative abundances at the phylum and genus levels. α-diversity indices (including the Shannon, Simpson, and Chao1 indices) were calculated using the QIIME 2 software package to assess microbial diversity within individual samples.
Serum C-reactive protein (CRP) levels were measured using immunoturbidimetry (Roche Diagnostics, Basel, Switzer
Statistical analyses were performed using SPSS software (version 26.0). The normality of continuous data was assessed using the Kolmogorov-Smirnov test. Normally distributed data with homogeneity of variance are presented as mean ± SD and were compared between groups using the independent samples t-test. Non-normally distributed data are expressed as median (first quartile, third quartile), and were compared using the Mann-Whitney U test. Categorical data are presented as n (%) and were analyzed using the χ2 test. Fisher's exact test was applied when the expected cell count was < 5. To investigate associations between the gut microbiota and biomarkers, compositional data on relative microbial abundance were first subjected to centered log-ratio transformation to mitigate constant-sum constraints. Subsequently, partial correlation analysis was performed to assess correlations between the transformed gut microbial diversity indices and phylum- and genus-level relative abundances and I-FABP and PRDX1 levels. Variables that demonstrated statistically significant differences in the univariate analysis between the HAEC and non-HAEC groups were tested for multicollinearity. After excluding severe multicollinearity [variance inflation factor, (VIF) < 5], binary logistic regression analysis was conducted, adjusting for potential confounders, such as age and sex, to identify factors associated with the development of postoperative HAEC in children with HD. A nomogram prediction model was constructed based on logistic regression results. The performance of the model was internally validated using bootstrap resampling with 1000 repetitions. The Hosmer-Lemeshow test and decision curve analysis (DCA) were used to evaluate the model's calibration and clinical utility, respectively, in predicting the risk of HAEC. Statistical significance was defined as a two-sided P value < 0.05.
Among the 334 children included, 109 (32.63%) developed HAEC within the 3-month postoperative follow-up period. No statistically significant between-group differences were observed for sex, age, BMI, pathological type or postoperative wound infection (P > 0.05). By contrast, the HAEC group had a significantly longer aganglionic segment and a higher prevalence of preoperative enteritis and postoperative intestinal obstruction than the non-HAEC group (P < 0.05; Table 1).
| Non HAEC group (n = 225) | HAEC group (n = 109) | t/χ2 | P value | |
| Gender | 0.490 | 0.484 | ||
| Male | 137 (60.89) | 62 (56.88) | ||
| Female | 88 (39.1) | 47 (43.12) | ||
| Age (years) | 2.04 ± 0.58 | 1.96 ± 0.57 | 1.127 | 0.261 |
| BMI (kg/m2) | 16.62 ± 1.67 | 16.38 ± 1.81 | 1.812 | 0.085 |
| Spasmodic segment lesion length (cm) | 17.69 ± 4.77 | 25.82 ± 8.74 | -10.982 | < 0.001 |
| Pathological classification | 1.495 | 0.683 | ||
| Short segment type | 91 (40.44) | 42 (38.53) | ||
| Common types | 95 (42.22) | 45 (41.28) | ||
| Long segment type | 23 (10.22) | 10 (9.17) | ||
| Whole segment type | 16 (7.11) | 12 (11.02) | ||
| History of enteritis | 7.263 | 0.007 | ||
| Yes | 75 (33.33) | 53 (48.62) | ||
| No | 150 (66.67) | 56 (51.38) | ||
| Postoperative ileus (cases) | - | 0.0121 | ||
| Yes | 4 (1.78) | 9 (8.26) | ||
| No | 221 (98.22) | 100 (91.74) | ||
| Postoperative wound infection (cases) | - | 0.0631 | ||
| Yes | 3 (1.33) | 6 (5.5) | ||
| No | 222 (98.67) | 103 (94.50) |
No significant differences in gut microbiota α-diversity indices, including the Chao1, Shannon and Simpson indices, were observed between the HAEC and non-HAEC groups (P > 0.05; Figure 1).
No significant difference in the abundance of Firmicutes was observed between the HAEC and non-HAEC groups (P > 0.05). By contrast, the HAEC group showed significantly higher abundances of Proteobacteria, Bacteroidetes and Verrucomicrobia and lower abundances of Actinobacteria and Tenericutes than the non-HAEC group (P < 0.05; Figure 2).
No significant differences in the abundances of Escherichia and Shigella were observed between the HAEC and non-HAEC groups (P > 0.05). By contrast, the HAEC group showed significantly lower abundances of Bifidobacterium and Lactobacillus and higher abundances of Streptococcus and Bacteroides than the non-HAEC group (P < 0.05; Figure 3).
The HAEC group exhibited significantly higher preoperative serum levels of CRP, TNF-α, IL-6, IL-8, I-FABP and PRDX1 than the non-HAEC group (P < 0.05; Table 2).
| Items | Non HAEC group (n = 225) | HAEC group (n = 109) | t value | P value |
| CRP (mg/L) | 3.58 ± 0.88 | 4.63 ± 1.29 | -8.742 | < 0.001 |
| TNF-α (pg/mL) | 32.76 ± 5.49 | 48.50 ± 5.76 | -24.183 | < 0.001 |
| IL-6 (pg/mL) | 26.00 ± 3.54 | 33.26 ± 5.24 | -14.910 | < 0.001 |
| IL-8 (pg/mL) | 16.33 ± 3.63 | 24.98 ± 5.04 | -17.875 | < 0.001 |
| I-FABP (pg/mL) | 84.35 ± 30.14 | 113.16 ± 35.10 | -7.756 | < 0.001 |
| PRDX1 (μg/mL) | 22.91 ± 6.40 | 32.05 ± 5.94 | -12.523 | < 0.001 |
After adjustment for age, sex and other confounding variables, correlation analysis showed that PRDX1 levels were positively correlated with the abundance of the Bacteroides genus and negatively correlated with the abundance of the Lactobacillus genus. I-FABP levels were positively correlated with the abundance of the Verrucomicrobia phylum and the Bacteroides genus and negatively correlated with the abundance of the Firmicutes phylum and the Bifidobacterium and Lactobacillus genera. IL-8 levels were positively correlated with the abundance of the Bacteroidetes and Verrucomicrobia phyla and the Bacteroides genus and negatively correlated with the abundance of the Firmicutes phylum and the Bifidobacterium and Lactobacillus genera. IL-6 levels were positively correlated with the abundance of the Proteobacteria, Bacteroidetes and Verrucomicrobia phyla and the Bacteroides genus and negatively correlated with the abundance of the Firmicutes phylum and the Bifidobacterium and Lactobacillus genera. TNF-α levels were positively correlated with the abundance of the Proteobacteria, Bacteroidetes and Verrucomicrobia phyla and the Streptococcus and Bacteroides genera and negatively correlated with the abundance of the Firmicutes phylum and the Bifidobacterium and Lactobacillus genera. CRP levels were negatively correlated with the abundance of the Bifidobacterium genus (P < 0.05; Figure 4).
Variables showing significant differences in the univariate analysis were subjected to a multicollinearity assessment. The initial assessment identified moderate multicollinearity (VIF ≥ 5.0) among length of the aganglionic segment, history of enteritis, abundances of Bacteroidetes, Bifidobacterium, Lactobacillus, Proteobacteria, Actinobacteria, Bacteroides, and serum concentrations of CRP, TNF-α, IL-6, IL-8, I-FABP, and PRDX1. Sequential exclusion, guided by inter-variable correlation analysis, removed length of the aganglionic segment, history of enteritis, abundances of Bacteroidetes, Lactobacillus, Proteobacteria, Actinobacteria, and serum concentrations of TNF-α, IL-6, and IL-8. The residual variables—including postoperative intestinal obstruction, abundances of Tenericutes, Verrucomicrobia, Bifidobacterium, Streptococcus, Bacte
| β | SE | Wals | P value | OR (95%CI) | |
| Postoperative ileus | 0.038 | 0.364 | 0.011 | 0.916 | 1.039 (0.509-2.120) |
| Tenericutes abundance | -0.672 | 0.567 | 1.405 | 0.236 | 0.511 (0.168-1.552) |
| Verrucomicrobia abundance | 0.492 | 0.157 | 9.820 | 0.002 | 1.636 (1.204-2.224) |
| Bifidobacterium abundance | -0.003 | 0.001 | 4.379 | 0.036 | 0.997 (0.995-1.000) |
| Streptococcus abundance | 0.004 | 0.003 | 1.778 | 0.250 | 1.004 (0.997-1.011) |
| Bacteroides abundance | 0.010 | 0.003 | 11.111 | 0.003 | 1.010 (1.003-1.016) |
| CRP | 0.961 | 0.235 | 16.723 | < 0.001 | 2.614 (1.648-4.146) |
| I-FABP | 0.031 | 0.007 | 19.621 | < 0.001 | 1.032 (1.018-1.045) |
| PRDX1 | 0.279 | 0.041 | 46.306 | < 0.001 | 1.322 (1.219-1.433) |
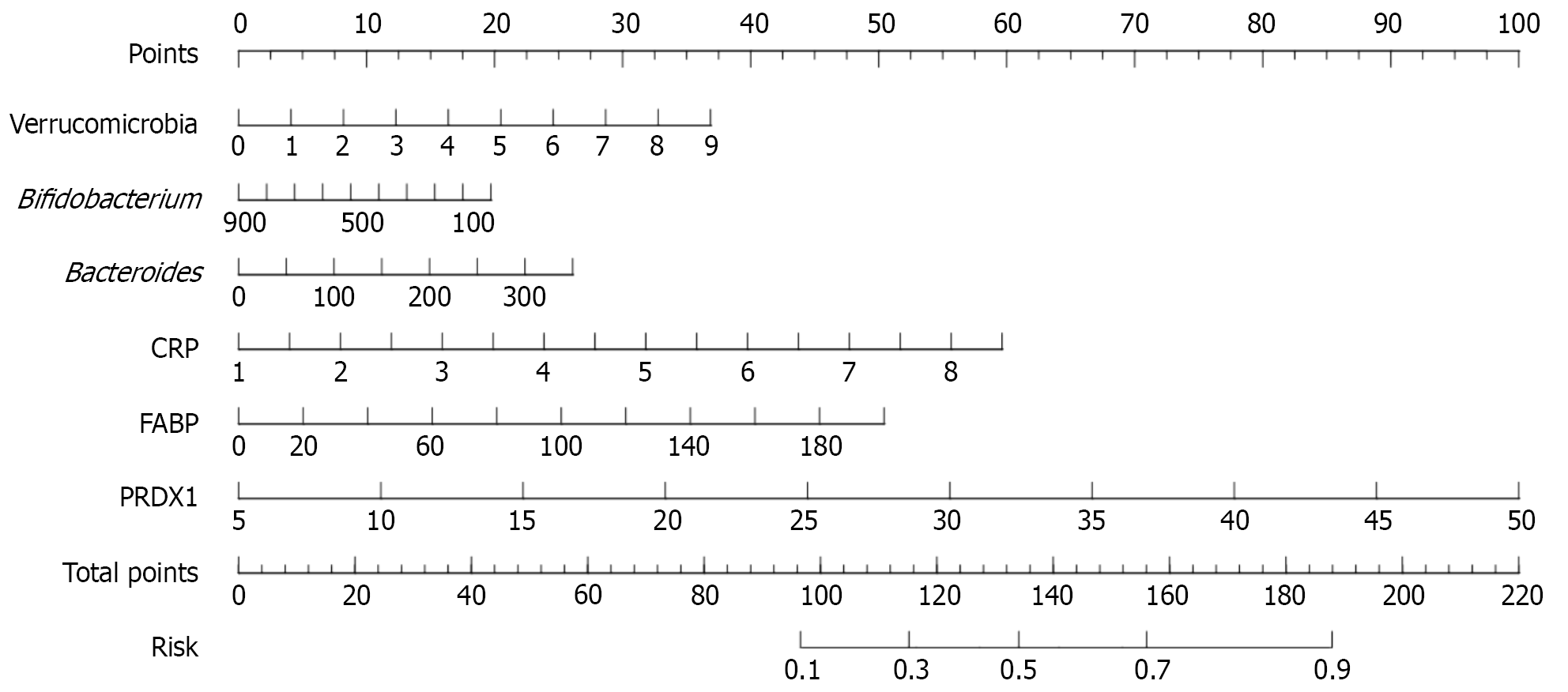
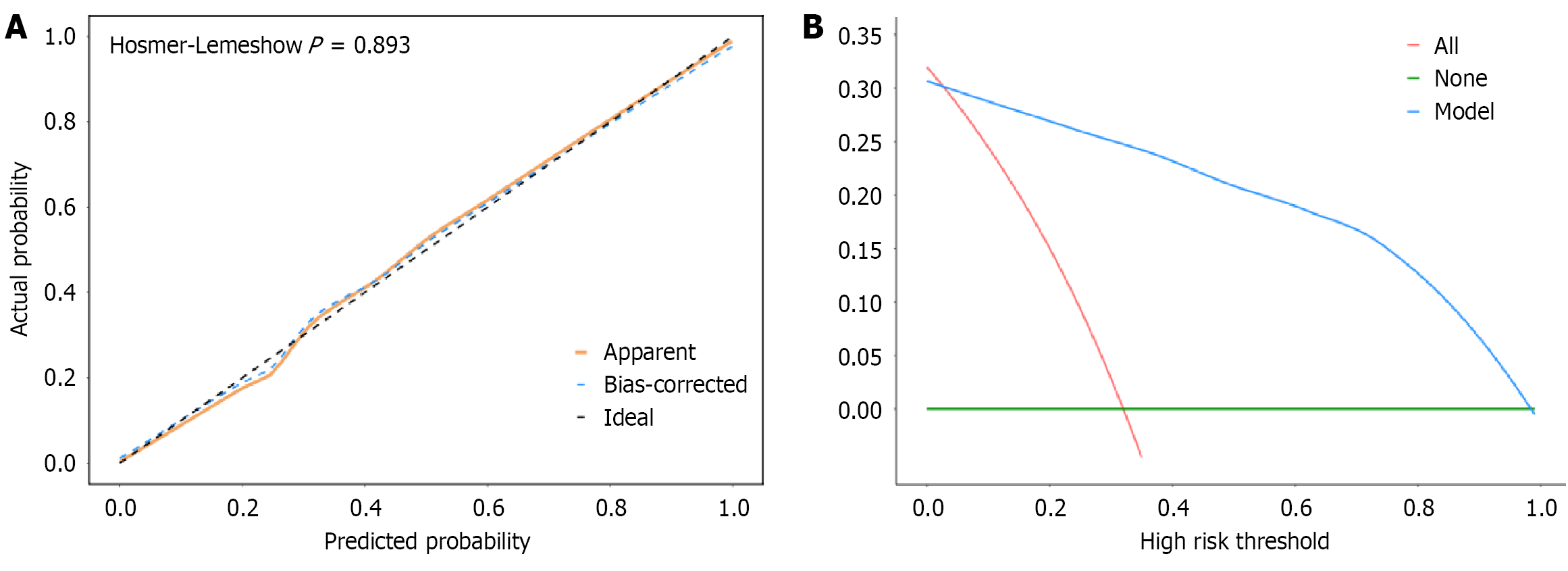
Based on logistic regression analysis, a nomogram prediction model was constructed to estimate the risk of postoperative HAEC in children with HD, as shown in Figure 5. Internal validation using bootstrap resampling (1000 iterations) demonstrated excellent predictive performance. The area under the receiver operating characteristic curve (AUC) for postoperative HAEC prediction was 0.951 (95%CI: 0.927-0.974). The Hosmer-Lemeshow test yielded a χ2 value of 3.573 (P = 0.893), indicating good calibration and close agreement between predicted and observed probabilities (Figure 6A). DCA further showed that application of the model conferred clinical net benefit across a threshold probability range of 0.05-1.00 (Figure 6B).
Children undergoing surgical treatment for HD have a substantial risk of developing HAEC, with previous studies reporting an incidence of approximately 30%[2,3]. The findings of this study align closely with the existing literature, revealing an HAEC incidence of 32.63% within 3 months following the modified Soave pull-through procedure. This underscores the critical need for increased awareness and management of postoperative HAEC risk. Srinivas et al[15] identified longer aganglionic segments, a history of enteritis, and postoperative intestinal obstruction as factors that increase HAEC risk, and our results corroborate these findings. The underlying rationale may be that an extended aganglionic segment results in a more widespread area of functional obstruction owing to the absence of normal ganglion cells. This leads to more severe compensatory colonic dilation and hypertrophy. This, in turn, causes prolonged and aggravated fecal stasis, fostering an environment conducive to pathogenic growth. Furthermore, the chronically distended bowel wall, under sustained pressure, exacerbates the impairment of intestinal barrier function[16]. Patients with a history of preoperative enteritis present with a pre-sensitized intestinal mucosal immune system and compro
Several studies have established that gut microbiota dysbiosis can directly lead to impaired intestinal barrier function and immune-inflammatory imbalance. Parker et al[19] reported that children who developed HAEC after HD surgery exhibited significantly lower gut microbiota diversity than those who did not. However, because microbial diversity in that study was assessed postoperatively, the observed differences may have reflected a consequence rather than a cause of HAEC. By contrast, no statistically significant differences in preoperative α-diversity, as assessed by the Chao1, Shannon and Simpson indices, were observed between the HAEC and non-HAEC groups in the present study. This finding suggests that preoperative gut microbiota α-diversity may be only weakly associated with subsequent deve
Further genus-level analysis showed that the HAEC group exhibited higher abundances of Streptococcus and Bacteroides and lower abundances of Bifidobacterium and Lactobacillus than the non-HAEC group. These findings suggest that preoperative depletion of protective bacterial genera, together with expansion of potentially harmful genera, may contribute to the development of postoperative HAEC in children with HD. The underlying rationale is that Bifidobacterium and Lactobacillus are key beneficial genera in the gut, supporting intestinal homeostasis through mechanisms including short-chain fatty acid production, regulation of T cell differentiation and reinforcement of epithelial tight junctions. The depletion of these beneficial bacteria indicates a substantial weakening of the host's intestinal protective functions, thereby creating a permissive environment for pathogen colonization and inflammatory outbreaks[23]. Conversely, Streptococcus includes several pro-inflammatory species, the expansion of which may amplify local immune and inflammatory responses[24]. Although Bacteroides constitutes a major component of the normal gut microbiota, specific species can act as opportunistic pathogens under dysbiotic conditions. However, this proposed functional shift requires further validation through metagenomic analyses[25].
Due to factors such as functional obstruction and impaired intestinal barrier function, the gut environment of children with HD is characterized by a state of chronic, low-grade inflammation. Consistent with the findings of Elkrewi et al[26], who reported significantly higher preoperative inflammatory markers in children who later developed HAEC, our study similarly demonstrated elevated preoperative serum levels of CRP, TNF-α, IL-6, IL-8, I-FABP, and PRDX1 in the HAEC group than in the non-HAEC group. These results indicate that postoperative HAEC development is associated with severe preoperative intestinal barrier damage and a heightened inflammatory response. Elevated preoperative levels of pro-inflammatory cytokines (TNF-α, IL-6, and IL-8) and CRP indicate a heightened systemic inflammation state before surgery, likely rooted in gut dysbiosis and barrier disruption[27]. Persistent stimulation by opportunistic pathogens and other pathogen-associated molecular patterns triggers overproduction of proinflammatory factors by the intestinal mucosal immune system. This process drives a systemic inflammatory response, exacerbates tissue injury, and stimulates hepatic CRP synthesis, further amplifying the inflammatory cascade. I-FABP is specific to the cytoplasm of intestinal epithelial cells and is rapidly released into the bloodstream following epithelial cell necrosis or apoptosis. Thus, elevated I-FABP levels serve as direct evidence of a significant compromise in intestinal barrier integrity[28,29]. Similarly, PRDX1 is a crucial antioxidant protein that is upregulated during severe oxidative stress to protect cells. A significant increase in PRDX1 levels indicated that the intestinal epithelial cells in these children were substantially oxidatively damaged, rendering them more vulnerable to bacterial and toxin invasion[30].
Correlation analysis identified a distinct “harmful axis”, comprising Proteobacteria, Bacteroidetes/Bacteroides, Verrucomicrobia and Streptococcus, which was positively correlated with pro-inflammatory markers and indices of intestinal injury. Conversely, a “protective axis”, comprising Actinobacteria (particularly Bifidobacterium), Tenericutes, and Lactobacillus, was negatively correlated with these markers, indicating an association with anti-inflammatory effects and intestinal integrity. These findings suggest that gut microbiota dysbiosis, characterized by depletion of beneficial taxa and expansion of potentially pathogenic taxa, may contribute to inflammation and intestinal injury rather than representing a passive consequence. Accordingly, severe dysbiosis could constitute a predisposing factor for postoperative HAEC in children undergoing HD; however, this putative causal relationship requires validation through detailed mechanistic studies.
In this study, logistic regression analysis identified the abundances of Verrucomicrobia and Bacteroides, together with preoperative serum levels of CRP, I-FABP and PRDX1, as independent risk factors for postoperative HAEC in children with HD, whereas Bifidobacterium abundance emerged as an independent protective factor. A nomogram prediction model constructed using these variables demonstrated excellent predictive performance during internal validation with bootstrap resampling (1000 iterations), achieving an AUC of 0.951 and showing strong agreement between predicted and observed risks. These findings suggest that assessment of these indicators could enable effective stratification of postoperative HAEC risk, and that preoperative interventions targeting these potentially modifiable factors may reduce HAEC incidence. Notably, several clinically relevant variables, including aganglionic segment length, history of enteritis, Bacteroidetes abundance and preoperative TNF-α levels, were excluded from the final model because of significant multicollinearity. This exclusion does not indicate a lack of association with HAEC risk; rather, their predictive effects are likely captured by the more robust variables retained in the model, namely Verrucomicrobia and Bacteroides abundance and preoperative CRP, I-FABP and PRDX1 Levels.
Although this study included a relatively large overall cohort, all participants were recruited from a single center, and the number of children with HAEC was limited, which may have introduced selection bias and reduced statistical power. Future multicenter studies with larger cohorts are therefore required to further elucidate the relationships between postoperative HAEC, gut microbiota composition, inflammatory markers, and I-FABP and PRDX1 levels. In addition, prospective clinical trials are needed to evaluate whether preventive strategies targeting these modifiable factors can reduce the incidence of HAEC after surgery for HD.
Children who develop HAEC following surgery for HD demonstrate a characteristic profile of reduced beneficial gut bacteria, increased pathogenic bacteria, a heightened immune-inflammatory response, and elevated levels of I-FABP and PRDX1. The serum levels of I-FABP and PRDX1 were significantly correlated with gut microbiota abundance. Preope
| 1. | Klein M, Varga I. Hirschsprung's Disease-Recent Understanding of Embryonic Aspects, Etiopathogenesis and Future Treatment Avenues. Medicina (Kaunas). 2020;56:611. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 2. | Chantakhow S, Tepmalai K, Singhavejsakul J, Tantraworasin A, Khorana J. Prognostic factors of postoperative Hirschsprung-associated enterocolitis: a cohort study. Pediatr Surg Int. 2023;39:77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 3. | Ziogas IA, Kuruvilla KP, Fu M, Gosain A. Hirschsprung-associated enterocolitis: a comprehensive review. World J Pediatr Surg. 2024;7:e000878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Zhang Y, Xiang X, Li X, Feng W, Guo Z. Early intervention in Hirschsprung's disease: effects on enterocolitis and surgical outcomes. BMC Pediatr. 2024;24:476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 5. | Roorda D, Oosterlaan J, van Heurn E, Derikx JPM. Risk factors for enterocolitis in patients with Hirschsprung disease: A retrospective observational study. J Pediatr Surg. 2021;56:1791-1798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Svetanoff WJ, Lim-Beutal IIP, Wood RJ, Levitt MA, Rentea RM. The utilization of botulinum toxin for Hirschsprung disease. Semin Pediatr Surg. 2022;31:151161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 7. | Cheng T, Yao Y, Zhang S, Hou C. Effect of live combined Bifidobacterium and Lactobacillus tablets combined with Soave radical operation on postoperative intestinal flora and immune function in children with Hirschsprung disease. Am J Transl Res. 2023;15:3572-3578. [PubMed] |
| 8. | Mei F, Wu M, Zhao L, Hu K, Gao Q, Chen F, Kwong JS, Wang Y, Shen XP, Ma B. Probiotics for the prevention of Hirschsprung-associated enterocolitis. Cochrane Database Syst Rev. 2022;4:CD013714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (2)] |
| 9. | Chantakhow S, Khorana J, Tepmalai K, Boonchooduang N, Chattipakorn N, Chattipakorn SC. Alterations of Gut Bacteria in Hirschsprung Disease and Hirschsprung-Associated Enterocolitis. Microorganisms. 2021;9:2241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 10. | Rehman N, Pandey A. Insight of Intestinal Fatty Acid Binding Protein as a Potential Biomarker in the Biology of Epithelial Damage of Gastrointestinal Membrane. Curr Protein Pept Sci. 2025;26:321-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Ledgerwood EC, Marshall JW, Weijman JF. The role of peroxiredoxin 1 in redox sensing and transducing. Arch Biochem Biophys. 2017;617:60-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 12. | Sbragia L, Gualberto IJN, Xia J, Gadde R, Saulsbery A, Hameedi S, Mársico Dalto ALF, Olutoye OO. Intestinal Fatty Acid-binding Protein as a Marker of Necrotizing Enterocolitis Incidence and Severity: a Scoping Review. J Surg Res. 2024;303:613-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 13. | Pastor AC, Osman F, Teitelbaum DH, Caty MG, Langer JC. Development of a standardized definition for Hirschsprung's-associated enterocolitis: a Delphi analysis. J Pediatr Surg. 2009;44:251-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 136] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 14. | Gosain A, Frykman PK, Cowles RA, Horton J, Levitt M, Rothstein DH, Langer JC, Goldstein AM; American Pediatric Surgical Association Hirschsprung Disease Interest Group. Guidelines for the diagnosis and management of Hirschsprung-associated enterocolitis. Pediatr Surg Int. 2017;33:517-521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 163] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 15. | Srinivas S, Henderson K, Griffin KL, Thomas E, Wood RJ, Langer JC, Halaweish I. Rates of Hirschsprung-Associated Enterocolitis Decrease With Increasing Child Opportunity Index. J Pediatr Surg. 2024;59:1240-1244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 16. | Bokova E, Elhalaby I, Dow B, Reeves PT, Rentea RM. Hirschsprung-associated Enterocolitis Action Plan: A Pictographic Tool for Caregivers of Children With Hirschsprung Disease. J Pediatr Surg. 2025;60:162269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Svetanoff WJ, Dekonenko C, Osuchukwu O, Oyetunji TA, Aguayo P, Fraser JD, Juang D, Snyder CL, Hendrickson R, Peter SS, Rentea RM. Inpatient management of Hirschsprung's associated enterocolitis treatment: the benefits of standardized care. Pediatr Surg Int. 2020;36:1413-1421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Benibol Y, Önenerk Men AM, Hakalmaz AE, Çomunoğlu N, Topuzlu Tekant G, Özcan R. The Effect of the Ganglionic Segment Inflammatory Response to Postoperative Enterocolitis in Hirschsprung Disease. Fetal Pediatr Pathol. 2024;43:140-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Parker KD, Mueller JL, Westfal M, Goldstein AM, Ward NL. A pilot study characterizing longitudinal changes in fecal microbiota of patients with Hirschsprung-associated enterocolitis. Pediatr Surg Int. 2022;38:1541-1553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Hidalgo-Cantabrana C, Delgado S, Ruiz L, Ruas-Madiedo P, Sánchez B, Margolles A. Bifidobacteria and Their Health-Promoting Effects. Microbiol Spectr. 2017;5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 314] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 21. | Furuichi M, Kawaguchi T, Pust MM, Yasuma-Mitobe K, Plichta DR, Hasegawa N, Ohya T, Bhattarai SK, Sasajima S, Aoto Y, Tuganbaev T, Yaginuma M, Ueda M, Okahashi N, Amafuji K, Kiridoshi Y, Sugita K, Stražar M, Avila-Pacheco J, Pierce K, Clish CB, Skelly AN, Hattori M, Nakamoto N, Caballero S, Norman JM, Olle B, Tanoue T, Suda W, Arita M, Bucci V, Atarashi K, Xavier RJ, Honda K. Commensal consortia decolonize Enterobacteriaceae via ecological control. Nature. 2024;633:878-886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 71] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 22. | Rodrigues VF, Elias-Oliveira J, Pereira ÍS, Pereira JA, Barbosa SC, Machado MSG, Carlos D. Akkermansia muciniphila and Gut Immune System: A Good Friendship That Attenuates Inflammatory Bowel Disease, Obesity, and Diabetes. Front Immunol. 2022;13:934695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 330] [Cited by in RCA: 282] [Article Influence: 70.5] [Reference Citation Analysis (0)] |
| 23. | Gavzy SJ, Kensiski A, Lee ZL, Mongodin EF, Ma B, Bromberg JS. Bifidobacterium mechanisms of immune modulation and tolerance. Gut Microbes. 2023;15:2291164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 204] [Article Influence: 68.0] [Reference Citation Analysis (0)] |
| 24. | Laterza L, Cremon C, Coppola G, Settanni CR, Maresca R, Strazzeri M, Durini E, Petito V, Scaldaferri F, Gargari G, Mora D, Vojoudi Yazdi E, Marangelo C, Ianiro G, Putignani L, Barbaro MR, Marasco G, Barbara G, Gasbarrini A. Multistrain Probiotics Plus Vitamin D Improve Gut Barrier Function and Gut Microbiota Composition in Irritable Bowel Syndrome Without Constipation: Results from a Double-Blind, Randomized, Placebo-Controlled Trial. Nutrients. 2025;17:1708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 25. | Zhang S, Nie Q, Sun Y, Zuo S, Chen C, Li S, Yang J, Hu J, Zhou X, Yu Y, Huang P, Lian L, Xie M, Nie S. Bacteroides uniformis degrades β-glucan to promote Lactobacillus johnsonii improving indole-3-lactic acid levels in alleviating colitis. Microbiome. 2024;12:177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 88] [Reference Citation Analysis (0)] |
| 26. | Elkrewi EZ, Al Abdulqader AA, Khasanov R, Maas-Omlor S, Boettcher M, Wessel LM, Schäfer KH, Tapia-Laliena MÁ. Role of Inflammation and the NF-κB Signaling Pathway in Hirschsprung's Disease. Biomolecules. 2024;14:992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Sutthatarn P, Lapidus-Krol E, Smith C, Halaweish I, Rialon K, Ralls MW, Rentea RM, Madonna MB, Haddock C, Rocca AM, Gosain A, Frischer J, Piper H, Goldstein AM, Saadai P, Durham MM, Dickie B, Jafri M, Langer JC. Hirschsprung-associated inflammatory bowel disease: A multicenter study from the APSA Hirschsprung disease interest group. J Pediatr Surg. 2023;58:856-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 28. | Pimenta S, Pissarra S, Soares P, Azevedo I, Pereira-Nunes J. Serum biomarkers in the early detection of necrotizing enterocolitis: a systematic review. J Perinat Med. 2025;53:966-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 29. | Seethaler B, Basrai M, Neyrinck AM, Nazare JA, Walter J, Delzenne NM, Bischoff SC. Biomarkers for assessment of intestinal permeability in clinical practice. Am J Physiol Gastrointest Liver Physiol. 2021;321:G11-G17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 174] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 30. | Li S, Zhang Y, Lu R, Lv X, Lei Q, Tang D, Dai Q, Deng Z, Liao X, Tu S, Yang H, Xie Y, Meng J, Yuan Q, Qin J, Pu J, Peng Z, Tao L. Peroxiredoxin 1 aggravates acute kidney injury by promoting inflammation through Mincle/Syk/NF-κB signaling. Kidney Int. 2023;104:305-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 61] [Reference Citation Analysis (0)] |