Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116546
Revised: January 15, 2026
Accepted: February 12, 2026
Published online: April 27, 2026
Processing time: 133 Days and 1.9 Hours
Colorectal cancer remains a leading cause of cancer-related morbidity and mor
To investigate multiple clinical parameters in patients undergoing laparoscopic radical colorectal cancer surgery, specifically examining pain control effectiveness, markers of early postoperative recovery, physiological stress indicators, and tre
Clinical records of 117 laparoscopic radical colorectal cancer surgery patients treated at our institution between December 2022 and December 2024 underwent retrospective examination. Patient allocation into study arms was determined by their postoperative pain management protocol: 59 patients constituted the ob
Pain intensity measurements using VAS methodology demonstrated significantly lower values in the observation cohort compared to controls at all assessment points (6 hours, 12 hours, 24 hours, 48 hours, and 72 hours postoperatively) during both rest and activity (P < 0.001). Within the initial 72-hour postoperative period, the observation group demonstrated reduced PCIA pump activation frequency, diminished total sufentanil consumption, lower rescue analgesia requirement rate (10.2% vs 31.0%), and superior patient satisfaction ratings regarding pain control (P < 0.05). Recovery milestones were achieved earlier in the observation cohort, with shorter intervals to first ambulation, initial flatus passage, and first oral intake compared to the control cohort (P < 0.001). Inflammatory and stress biomarker analysis revealed lower serum concentrations of IL-6, CRP, and Cor in the observation group on both postoperative day 1 and day 3 (P < 0.001). Recovery quality assessment using QoR-15 scoring demon
Ultrasound-guided TAP block combined with multimodal analgesia applied after laparoscopic radical colorectal cancer surgery can provide excellent analgesic effects, reduce opioid consumption, promote early ambulation and gastrointestinal function recovery, reduce postoperative stress response, improve recovery quality, reduce opioid-related adverse reactions, and has good safety, which is helpful in achieving ERAS goals.
Core Tip: This retrospective study evaluates whether integrating an ultrasound-guided transversus abdominis plane (TAP) block into multimodal analgesia can further enhance postoperative recovery after laparoscopic radical colorectal cancer surgery. The findings show that TAP block significantly improves pain control, reduces opioid consumption, accelerates gastrointestinal and functional recovery, attenuates stress responses, and enhances early quality-of-recovery outcomes without increasing complications. These results provide important clinical evidence supporting optimized analgesic strategies and enhanced recovery after surgery implementation for colorectal cancer patients.
- Citation: Zhang SX, Liao XB, Lin XL, Yang J. Role of transversus abdominis plane block combined with multimodal analgesia in early recovery after radical colorectal cancer surgery. World J Gastrointest Surg 2026; 18(4): 116546
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116546.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116546
Among malignant neoplasms worldwide, colorectal cancer represents a highly prevalent condition, with its incidence and mortality figures ranking prominently in cancer statistics[1]. Radical surgical excision remains the definitive therapeutic intervention for colorectal malignancies. Laparoscopic techniques have evolved to become the preferred surgical methodology for radical colorectal cancer resection, offering benefits including reduced surgical trauma and expedited convalescence[2]. Despite these advances, moderate-to-severe postoperative pain afflicts 30%-40% of patients, creating discomfort while potentially triggering amplified stress responses, compromised immune function, delayed recovery trajectories, and elevated complication risks[3].
The enhanced recovery after surgery (ERAS) framework prioritizes comprehensive perioperative care optimization, positioning effective postoperative analgesia as a fundamental component[4]. Through synergistic application of analgesic agents and techniques operating via distinct mechanisms, multimodal analgesia achieves enhanced pain relief while simultaneously reducing opioid requirements, establishing itself as the reference standard for postoperative pain control strategies[5]. As a regional anesthetic technique, transversus abdominis plane (TAP) block delivers prolonged abdominal wall analgesia by infiltrating local anesthetic agents into the TAP, effectively interrupting signal transmission through the anterior branches of spinal nerves T7-L1[6]. Contemporary research has demonstrated TAP block's analgesic value in abdominal surgical procedures; however, insufficient investigation exists regarding its potential to further refine multimodal analgesic protocols and facilitate early recovery following radical colorectal cancer operations[7]. Furthermore, TAP block’s influence on postoperative stress physiology and recovery quality metrics requires additional clinical substantiation[8].
Consequently, this investigation employed retrospective methodology to evaluate pain control effectiveness, early recovery parameters, stress response characteristics, and safety considerations associated with ultrasound-guided TAP block integration into multimodal analgesia protocols following laparoscopic radical colorectal cancer surgery. The objective was generating clinical evidence to support perioperative pain management optimization and enhanced recovery facilitation in the colorectal cancer patient population.
Therefore, this study aims to evaluate the analgesic efficacy, early recovery indicators, stress response, and safety of ultrasound-guided TAP block combined with multimodal analgesia after laparoscopic radical colorectal cancer surgery through retrospective analysis, providing clinical evidence for optimizing perioperative pain management and promoting rapid recovery in colorectal cancer patients.
This study retrospectively analyzed clinical data from 132 patients who underwent radical colorectal cancer surgery at our hospital from December 2022 to December 2024. This study was approved by the hospital ethics committee. As the study was a retrospective analysis, the ethics committee approved exemption from patient informed consent.
Inclusion criteria: (1) Age 18-75 years; (2) Pathologically confirmed colorectal cancer meeting indications for radical surgery; (3) American Society of Anesthesiologists (ASA) physical status classification grade I-III; (4) Body mass index (BMI) 18-30 kg/m2; (5) Underwent laparoscopic radical colorectal cancer surgery; and (6) Complete clinical data.
Exclusion criteria: (1) Severe cardiac, pulmonary, hepatic, or renal dysfunction; (2) Coagulation disorders or long-term use of anticoagulant drugs; (3) Infection or deformity at the puncture site; (4) Allergy to local anesthetics or study-related drugs; (5) Long-term use of opioids or history of drug abuse; (6) Mental illness or cognitive impairment, unable to cooperate with pain assessment; (7) Intraoperative changes in surgical approach or conversion to open surgery; (8) Severe postoperative complications requiring reoperation, or withdrawal from ERAS management protocol for various reasons postoperatively; and (9) Missing key data such as pain assessment and laboratory examinations.
According to the above criteria, 117 patients were finally included for analysis. Based on different postoperative analgesia protocols, the 117 patients were divided into two groups: (1) Observation group (n = 59): TAP block combined with multimodal analgesia; and (2) Control group (n = 58): Conventional multimodal analgesia.
A single surgical team performed all laparoscopic radical colorectal cancer operations, adhering to established onco
Postoperative management incorporated all patients into ERAS protocols, featuring early mobilization, early oral feeding, and avoidance of routine nasogastric decompression and abdominal drainage.
Group assignment was determined through review of anesthesia documentation and postoperative analgesia orders. Observation group patients received intraoperative ultrasound-guided bilateral TAP block combined with multimodal analgesia; control group patients received exclusively conventional multimodal analgesia.
General anesthesia was administered to all patients following preoperative fasting periods of 8-12 hours for solids and 2-4 hours for liquids. Upon operating room arrival, peripheral venous access was secured and standard monitoring established [electrocardiogram; blood pressure; heart rate; saturation of peripheral oxygen (SpO2)]. Anesthetic induction consisted of midazolam 0.05 mg/kg, sufentanil 0.3-0.5 μg/kg, etomidate 0.2-0.3 mg/kg, and cisatracurium 0.15-0.2
Control cohort protocol: Both groups received intravenous flurbiprofen axetil 50 mg administered thirty minutes before surgical completion. Postoperative analgesia employed patient-controlled intravenous analgesia (PCIA): Sufentanil 100 μg + ondansetron 12 mg diluted with normal saline to 100 mL total volume, programmed with 2 mL/hour conti
Observation cohort protocol: In addition to all interventions in the control protocol, ultrasound-guided bilateral TAP block was incorporated. Per anesthesia documentation, while patients remained under general anesthesia prior to surgical conclusion, a high-frequency linear transducer (6-13 MHz) was positioned at the mid-axillary line/iliac crest intersection to visualize the three muscular layers: External oblique, internal oblique, and transversus abdominis muscles. Under continuous ultrasound visualization utilizing in-plane technique, a 22G needle was advanced into the TAP between internal oblique and transversus abdominis muscles. Following negative aspiration confirmation for blood and air, 0.375% ropivacaine 20 mL was deposited on each side (total volume 40 mL, total ropivacaine dose 150 mg; all patients weighed ≥ 50 kg, ensuring ropivacaine dosing ≤ 3 mg/kg), with observation of homogeneous medication spread within the TAP. Postoperative analgesia protocols matched the control cohort.
If patients had Visual Analog Scale (VAS) scores > 4 postoperatively, rescue analgesia was administered: Intramuscular injection of tramadol 100 mg or intravenous injection of flurbiprofen axetil 50 mg, which could be repeated if necessary, with an interval of ≥ 6 hours between administrations.
General data: Baseline data of patients in both groups were recorded through the electronic medical record system, including: Gender, age, BMI, ASA physical status classification, tumor location (colon/rectum), tumor, node, and metastasis (TNM) staging, operative time, intraoperative blood loss, and surgical approach.
Primary observation indicators: VAS scores at rest and during movement were collected by reviewing nursing records and pain assessment forms: A 0-10 points scale was used to assess pain severity at rest and during movement (coughing) at 6 hours, 12 hours, 24 hours, 48 hours, and 72 hours postoperatively, with 0 being no pain and 10 being severe pain. VAS scores were assessed by trained nurses who were blinded to patient grouping.
Secondary observation indicators: Analgesia-related indicators: The number of effective PCIA pump presses within 72 hours postoperatively and cumulative sufentanil consumption (μg) at 72 hours postoperatively were recorded. A 5-point scale was used to assess analgesia satisfaction at 48 hours postoperatively through patient satisfaction questionnaires, with 1 being very dissatisfied and 5 being very satisfied. The incidence of rescue analgesia was recorded, defined as the proportion of patients requiring additional analgesic medication due to VAS scores > 4 within 72 hours postoperatively.
Early recovery indicators: Time to first ambulation (time from end of surgery to patient’s first standing or walking from bed), time to first flatus (time from end of surgery to patient’s first spontaneous flatus), and time to first oral intake (time from end of surgery to patient's first liquid diet intake) were recorded, all in hours.
Stress response indicators: Serum interleukin-6 (IL-6), C-reactive protein (CRP), and cortisol (Cor) levels were collected by reviewing laboratory report systems for peripheral venous blood samples collected fasting in the morning on preoperative day 1 and postoperative days 1 and 3. The above indicators were all tested by enzyme-linked immuno
Postoperative adverse reactions: The occurrence and severity of adverse reactions including nausea and vomiting, urinary retention, skin itching, respiratory depression (respiratory rate < 8 breaths/min or SpO2 < 90%), and dizziness and drowsiness within 72 hours postoperatively were recorded. Nausea and vomiting grading: Grade 0 for no nausea or vomiting, grade I for mild nausea, grade II for nausea with 1-2 episodes of vomiting, grade III for frequent vomiting ≥ 3 episodes.
Postoperative recovery quality: The Quality of Recovery-15 (QoR-15) scale was used to assess patient recovery quality at 24 hours and 48 hours postoperatively. This scale contains 15 items with a total score of 0-150 points, with higher scores indicating better recovery quality. This scale data was collected by trained researchers through review of nursing records or telephone follow-up.
Postoperative complications: The occurrence of complications including wound infection, pulmonary infection, anastomotic leakage, and intestinal obstruction within 30 days postoperatively was recorded, and complication severity was assessed using the Clavien-Dindo classification system.
SPSS 26.0 statistical software was used for data analysis. Continuous variables were first tested for normality (Shapiro-Wilk test) and homogeneity of variance (Levene’s test). Continuous variables conforming to normal distribution were expressed as mean ± SD, and independent samples t-test was used for comparison between groups; continuous variables not conforming to normal distribution were expressed as median interquartile range (IQR) [M (Q1, Q3)], and Mann-Whitney U test was used for comparison between groups. For variables, normality testing confirmed that “effective PCIA pump presses” showed non-normal distribution (P = 0.038 in observation group); therefore, this variable was re-analyzed using Mann-Whitney U test and presented as median (IQR). Other continuous variables passed normality testing (P > 0.05) and were appropriately analyzed using parametric tests. Categorical variables were expressed as n (%), and χ2 test or Fisher’s exact test (when theoretical frequency < 5) was used for comparison between groups. Mann-Whitney U test was used for ordinal data. Bonferroni method was used for multiple comparison correction of data measured at multiple time points. All tests were two-sided, with P < 0.05 considered statistically significant.
There were no statistically significant differences between the two groups in gender, age, BMI, ASA physical status classification, tumor location, tumor TNM staging, operative time, intraoperative blood loss, and surgical approach (P > 0.05), indicating comparability (Table 1).
| Item | Observation group (n = 59) | Control group (n = 58) | Statistic | P value |
| Gender | χ2 = 0.145 | 0.703 | ||
| Male | 35 (59.3) | 33 (56.9) | ||
| Female | 24 (40.7) | 25 (43.1) | ||
| Age (years, mean ± SD) | 58.6 ± 10.2 | 59.3 ± 9.8 | t = 0.389 | 0.698 |
| BMI (kg/m2, mean ± SD) | 23.4 ± 2.8 | 23.7 ± 2.6 | t = 0.618 | 0.538 |
| ASA classification | Z = -0.326 | 0.744 | ||
| Grade I | 18 (30.5) | 16 (27.6) | ||
| Grade II | 32 (54.2) | 34 (58.6) | ||
| Grade III | 9 (15.3) | 8 (13.8) | ||
| Tumor location | χ2 = 0.069 | 0.793 | ||
| Colon | 37 (62.7) | 35 (60.3) | ||
| Rectum | 22 (37.3) | 23 (39.7) | ||
| TNM staging | Z = -0.158 | 0.875 | ||
| Stage I | 12 (20.3) | 11 (19.0) | ||
| Stage II | 26 (44.1) | 27 (46.6) | ||
| Stage III | 21 (35.6) | 20 (34.4) | ||
| Surgical approach | χ2 = 0.892 | 0.926 | ||
| Right hemicolectomy | 19 (32.2) | 17 (29.3) | ||
| Left hemicolectomy | 11 (18.6) | 13 (22.4) | ||
| Sigmoid colectomy | 7 (11.9) | 5 (8.6) | ||
| Anterior rectal resection | 18 (30.5) | 19 (32.8) | ||
| Abdominoperineal resection | 4 (6.8) | 4 (6.9) | ||
| Operative time (minute, mean ± SD) | 186.4 ± 35.7 | 189.2 ± 38.1 | t = 0.415 | 0.679 |
| Intraoperative blood loss (mL, mean ± SD) | 82.3 ± 28.6 | 85.7 ± 31.2 | t = 0.622 | 0.535 |
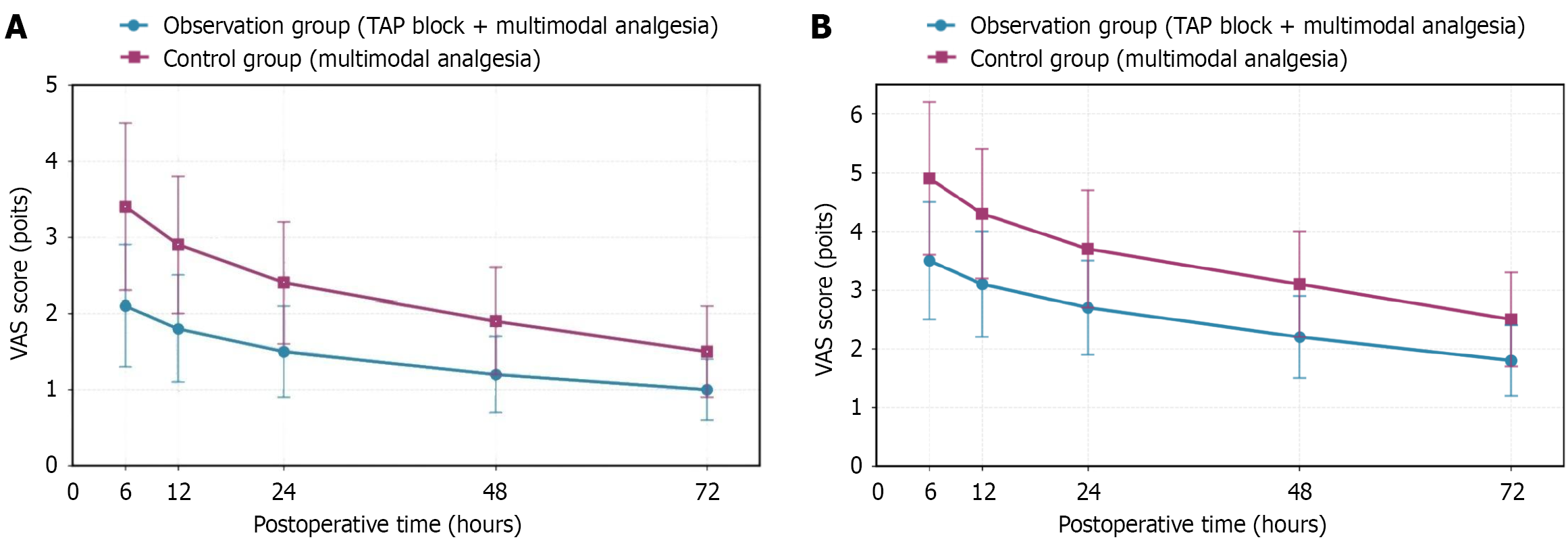
VAS scores at rest and during movement at all postoperative time points were lower in the observation group than in the control group. Bonferroni method was applied for multiple comparison correction of data from 5 time points (corrected significance level α = 0.01), and P values at all time points were < 0.001, with differences remaining statistically significant after correction (Table 2 and Figure 1).
| Time point | Resting VAS score | Movement VAS score | ||||
| Observation group | Control group | P value | Observation group | Control group | P value | |
| Postop 6 hours | 2.1 ± 0.8 | 3.4 ± 1.1 | < 0.001 | 3.5 ± 1.0 | 4.9 ± 1.3 | < 0.001 |
| Postop 12 hours | 1.8 ± 0.7 | 2.9 ± 0.9 | < 0.001 | 3.1 ± 0.9 | 4.3 ± 1.1 | < 0.001 |
| Postop 24 hours | 1.5 ± 0.6 | 2.4 ± 0.8 | < 0.001 | 2.7 ± 0.8 | 3.7 ± 1.0 | < 0.001 |
| Postop 48 hours | 1.2 ± 0.5 | 1.9 ± 0.7 | < 0.001 | 2.2 ± 0.7 | 3.1 ± 0.9 | < 0.001 |
| Postop 72 hours | 1.0 ± 0.4 | 1.5 ± 0.6 | < 0.001 | 1.8 ± 0.6 | 2.5 ± 0.8 | < 0.001 |
The observation group had fewer effective PCIA pump presses within 72 hours postoperatively, lower cumulative sufentanil consumption at 72 hours postoperatively, and lower rescue analgesia incidence rate compared with the control group (P < 0.05), and higher analgesia satisfaction scores at 48 hours postoperatively than the control group (P < 0.05) (Table 3). Cumulative sufentanil consumption at 72 hours postoperatively included the sum of PCIA pump background infusion and patient-controlled bolus doses. PCIA pumps were replaced with new pumps after drug depletion at 48-50 hours postoperatively to ensure continuous analgesia. For patients requiring rescue analgesia, this mainly occurred when VAS scores during movement were > 4 within 6-24 hours postoperatively.
| Indicator | Observation group (n = 59) | Control group (n = 58) | Statistic | P value |
| Effective PCIA pump presses [times, M (Q1, Q3)] | 8.0 (6.0, 10.0) | 14.0 (11.0, 18.0) | Z = -6.825 | < 0.001 |
| Cumulative sufentanil at 72 hours postop (μg, mean ± SD) | 68.5 ± 15.3 | 89.7 ± 18.6 | t = 6.842 | < 0.001 |
| Analgesia satisfaction score (points, mean ± SD) | 4.3 ± 0.6 | 3.7 ± 0.7 | t = 5.025 | < 0.001 |
| Rescue analgesia incidence rate, n (%) | 6 (10.2) | 18 (31.0) | χ2 = 7.783 | 0.005 |
Time to first ambulation (defined as the time for patients to stand from bed and walk ≥ 3 meters postoperatively), time to first flatus, and time to first oral intake (defined as the time for patients to consume ≥ 100 mL liquid diet postoperatively) were shorter in the observation group than in the control group (P < 0.05) (Table 4).
| Indicator | Observation group (n = 59) | Control group (n = 58) | t value | P value |
| Time to first ambulation | 18.6 ± 4.3 | 24.8 ± 5.7 | 6.731 | < 0.001 |
| Time to first flatus | 42.3 ± 10.8 | 56.7 ± 13.4 | 6.475 | < 0.001 |
| Time to first oral Intake | 16.2 ± 3.8 | 21.5 ± 5.2 | 6.332 | < 0.001 |
There were no statistically significant differences in stress response indicators on preoperative day 1 between the two groups (P > 0.05). Serum IL-6, CRP, and Cor levels on postoperative days 1 and 3 were elevated compared with preoperative levels in both groups. Bonferroni method was applied for comparison correction of 3 time points for each stress indicator (corrected significance level α = 0.017), and all indicator levels on postoperative days 1 and 3 were lower in the observation group than in the control group, with differences remaining statistically significant after correction (Table 5).
| Indicator | Time point | Observation group | Control group | t value | P value | After correction |
| IL-6 (pg/mL) | Preop day 1 | 12.8 ± 3.6 | 13.1 ± 3.8 | 0.447 | 0.656 | NS |
| Postop day 1 | 86.5 ± 18.4 | 112.3 ± 22.6 | 6.878 | < 0.001 | P < 0.001 | |
| Postop day 3 | 45.2 ± 12.7 | 63.8 ± 15.3 | 7.256 | < 0.001 | P < 0.001 | |
| CRP (mg/L) | Preop day 1 | 5.2 ± 1.8 | 5.4 ± 1.9 | 0.588 | 0.558 | NS |
| Postop day 1 | 68.4 ± 15.2 | 89.6 ± 18.9 | 6.898 | < 0.001 | P < 0.001 | |
| Postop day 3 | 38.7 ± 10.5 | 54.3 ± 13.8 | 6.95 | < 0.001 | P < 0.001 | |
| Cor (nmol/L) | Preop day 1 | 285.3 ± 45.6 | 289.7 ± 48.2 | 0.517 | 0.606 | NS |
| Postop day 1 | 486.8 ± 68.4 | 562.5 ± 82.3 | 5.538 | < 0.001 | P < 0.001 | |
| Postop day 3 | 368.2 ± 52.7 | 428.6 ± 64.5 | 5.685 | < 0.001 | P < 0.001 |
The severity of nausea and vomiting (assessed using 4-grade classification) and the incidence of dizziness and drowsiness were lower in the observation group than in the control group (P < 0.05), while there were no statistically significant differences in the incidence of urinary retention and skin itching between the two groups (P > 0.05). No respiratory depression occurred in either group (Table 6).
| Adverse reaction | Observation group (n = 59) | Control group (n = 58) | Statistic | P value |
| Nausea and vomiting | Z = -2.534 | 0.011 | ||
| Grade 0 | 45 (76.3) | 32 (55.2) | ||
| Grade I | 11 (18.6) | 17 (29.3) | ||
| Grade II | 3 (5.1) | 7 (12.1) | ||
| Grade III | 0 (0) | 2 (3.4) | ||
| Urinary retention | 3 (5.1) | 6 (10.3) | Fisher’s exact test | 0.312 |
| Skin itching | 2 (3.4) | 4 (6.9) | Fisher’s exact test | 0.431 |
| Respiratory depression | 0 (0) | 0 (0) | - | - |
| Dizziness and drowsiness | 8 (13.6) | 18 (31.0) | χ2 = 5.123 | 0.024 |
QoR-15 scores at 24 hours and 48 hours postoperatively were higher in the observation group than in the control group. Bonferroni method was applied for multiple comparison correction of data from 2 time points (corrected significance level α = 0.025), with differences remaining statistically significant after correction (Table 7).
| Time point | Observation group (n = 59) | Control group (n = 58) | t value | P value | After correction |
| Postop 24 hours | 108.6 ± 12.4 | 95.3 ± 14.7 | 5.436 | < 0.001 | P < 0.001 |
| Postop 48 hours | 122.5 ± 11.8 | 110.2 ± 13.5 | 5.389 | < 0.001 | P < 0.001 |
There were no statistically significant differences in the incidence of complications including wound infection, pulmonary infection, anastomotic leakage, and intestinal obstruction within 30 days postoperatively between the two groups (P > 0.05). There was no statistically significant difference in Clavien-Dindo classification of complications between the two groups (P > 0.05) (Table 8).
| Complication | Observation group (n = 59) | Control group (n = 58) | Statistic | P value |
| Wound infection | 2 (3.4) | 3 (5.2) | Fisher’s exact test | 0.677 |
| Pulmonary infection | 3 (5.1) | 5 (8.6) | Fisher’s exact test | 0.492 |
| Anastomotic leakage | 1 (1.7) | 2 (3.4) | Fisher’s exact test | 0.617 |
| Intestinal obstruction | 2 (3.4) | 3 (5.2) | Fisher’s exact test | 0.677 |
| Total incidence | 8 (13.6) | 13 (22.4) | χ2 = 1.541 | 0.214 |
| Clavien-Dindo classification | Z = -0.985 | 0.325 | ||
| Grade I | 4 (6.8) | 5 (8.6) | ||
| Grade II | 3 (5.1) | 6 (10.3) | ||
| Grade IIIa | 1 (1.7) | 2 (3.4) | ||
| Grade IIIb and above | 0 (0) | 0 (0) |
This study demonstrates an integrated mechanistic pathway by which TAP block enhances ERAS outcomes in colorectal cancer surgery: Superior pain control enables earlier mobilization and feeding, which in turn reduces opioid requirements and attenuates the surgical stress response, ultimately improving recovery quality. Our findings align with and extend previous literature. Comparable analgesic efficacy between TAP block combined with multimodal analgesia vs epidural analgesia in laparoscopic colon cancer surgery, supporting TAP block as a viable regional technique. However, our study uniquely demonstrates TAP block’s impact on stress biomarkers (IL-6, CRP, Cor) and QoR-15 scores, outcomes not systematically evaluated and confirmed TAP block’s opioid-sparing benefits in bariatric surgery, consistent with our 23.6% reduction in sufentanil consumption. Building on this foundation, we demonstrate that this opioid-sparing effect translates to accelerated gastrointestinal recovery (14-hour earlier flatus) and reduced stress response-mechanistic links not established in the bariatric population. Most notably, recently examined multimodal analgesia in elderly colorectal cancer patients but did not specifically evaluate TAP block’s incremental contribution. Our controlled comparison isolates TAP block’s specific effects within a multimodal framework, demonstrating clear additional benefits beyond systemic analgesia alone.
TAP block provides effective sensory blockade for the anterolateral abdominal wall by injecting local anesthetics in the fascial plane between the internal oblique muscle and transversus abdominis muscle, blocking the anterior branches of T7-L1 spinal nerves[9]. This study used bilateral TAP block with 20 mL of 0.375% ropivacaine injected on each side, totaling 150 mg, which ensured both the range and duration of blockade while remaining within safe dosage limits[10]. Results showed that VAS scores at all postoperative time points were significantly lower in the observation group than in the control group, especially the analgesic effect during movement was more prominent, which is closely related to TAP block’s effective blockade of somatic nerve conduction in the abdominal wall, reducing pain caused by incision and abdominal wall traction[11].
It is worth noting that the analgesic effect of TAP block in this study lasted until 72 hours postoperatively, which may be related to the pharmacokinetic characteristics of ropivacaine and the anatomical features of the TAP. Studies have shown that ropivacaine is absorbed slowly in the TAP and can maintain local anesthetic effects for an extended period[12]. In addition, TAP block mainly blocks somatic pain conduction and has limited effects on visceral pain, so it needs to be combined with systemic analgesic drugs to achieve optimal analgesic effects[13].
Multimodal analgesia is a core element of ERAS. Through the combined application of analgesic techniques and drugs with different mechanisms of action, it can block pain conduction pathways at different levels, producing synergistic and additive effects[14]. The multimodal analgesia protocol in this study included TAP block, PCIA (sufentanil), and non-steroidal anti-inflammatory drugs (celecoxib, flurbiprofen axetil), acting at different levels including peripheral, spinal, and central, forming a complete analgesic system[15].
Study results showed that cumulative sufentanil consumption at 72 hours postoperatively was reduced by 23.6% in the observation group compared with the control group, effective PCIA pump presses were reduced by 43.2%, and rescue analgesia incidence rate was reduced by 67.1%. This indicates that the addition of TAP block significantly reduced opioid demand and reduced the risk of opioid-related adverse reactions[16]. In this study, both the severity of nausea and vomiting and the incidence of dizziness and drowsiness were lower in the observation group than in the control group, further confirming the clinical value of reducing opioid consumption. This opioid-sparing effect not only improves patient comfort but also helps reduce opioid-induced intestinal dysfunction and promotes gastrointestinal function recovery[17].
Effective postoperative analgesia is a prerequisite for achieving rapid recovery. Pain not only affects patient comfort but may also lead patients to reduce activity and delay feeding due to pain, thereby affecting the recovery process[18]. This study showed that time to first ambulation, time to first flatus, and time to first oral intake were all significantly earlier in the observation group than in the control group, which is closely related to the excellent analgesic effect provided by TAP block.
Early ambulation can reduce the risk of postoperative deep vein thrombosis and pulmonary complications and promote gastrointestinal function recovery[19]. In this study, patients in the observation group were able to ambulate an average of 18.6 hours postoperatively, approximately 6 hours earlier than the control group, a difference that is clinically significant. Early mobilization requires patients to tolerate pain during movement, and study results showed that movement VAS scores were significantly lower in the observation group than in the control group, providing guarantee for early mobilization[20].
Time to first flatus is an important indicator reflecting gastrointestinal function recovery. Studies showed that time to first flatus was approximately 14 hours earlier in the observation group than in the control group. This may be related to the following mechanisms: First, TAP block reduced opioid consumption, reducing the inhibitory effect of opioids on the intestine[21]; second, good analgesic effects promoted early mobilization and feeding, promoting intestinal motility through mechanical stimulation; in addition, reducing pain stress response also helps reduce sympathetic nervous tension and improve intestinal blood supply and function[22].
Surgical trauma can cause the body to produce stress responses, manifested as proinflammatory cytokine release, acute phase reactant protein elevation, and hypothalamic-pituitary-adrenal axis activation[23]. Excessive stress response may lead to immune function suppression and enhanced catabolism, which is unfavorable for postoperative recovery. This study measured three stress response indicators: IL-6, CRP, and Cor, and results showed that all indicator levels on postoperative days 1 and 3 were significantly lower in the observation group than in the control group.
IL-6 is an important proinflammatory cytokine, and its level is related to the degree of surgical trauma and risk of postoperative complications[24]. CRP is an acute phase reactant protein synthesized by the liver, regulated by cytokines such as IL-6, and is a sensitive indicator reflecting systemic inflammatory response[25]. Cor is a key hormone in stress response, and its elevated level reflects the degree of hypothalamic-pituitary-adrenal axis activation[26]. TAP block combined with multimodal analgesia in this study was able to reduce these stress response indicators, which may be related to the following mechanisms: First, effective analgesia reduced neuroendocrine responses caused by nociceptive stimulation; second, reducing opioid consumption may have reduced the immunosuppressive effects of the drugs themselves; in addition, early mobilization and feeding also help regulate stress response and promote immune function recovery[27,28].
The QoR-15 scale is an effective tool for assessing postoperative recovery quality, covering five dimensions: Pain, physical comfort, psychological support, physical independence, and emotional state[29]. This study showed that QoR-15 scores at 24 hours and 48 hours postoperatively were significantly higher in the observation group than in the control group, indicating that TAP block combined with multimodal analgesia can comprehensively improve patients’ postope
In this study, there was no significant difference in postoperative complication incidence rates between the two groups, indicating that TAP block is safe when applied after radical colorectal cancer surgery. No serious TAP block-related complications such as local anesthetic toxicity or intra-abdominal organ injury occurred in the observation group, which benefited from the application of ultrasound-guided technology and strict operating protocols[31]. It is worth noting that the incidence of opioid-related adverse reactions such as nausea, vomiting, dizziness, and drowsiness was lower in the observation group than in the control group, further confirming the opioid-sparing effect of TAP block. No respiratory depression occurred in either group, indicating that the analgesia protocols in this study were within safe limits.
This study has the following limitations: First, as a retrospective study, there may be selection bias and information bias, and some confounding factors are difficult to completely control; second, this study was a single-center study with a relatively limited sample size, and the generalizability of results requires further verification; third, this study did not explore optimization of optimal local anesthetic concentration, volume, and timing for TAP block; fourth, this study had a short follow-up period and did not evaluate the impact of TAP block on long-term prognosis and chronic pain incidence; finally, this study did not conduct cost-benefit analysis, and the health economic value of TAP block requires further research[32].
Study results indicate that ultrasound-guided TAP block combined with multimodal analgesia after laparoscopic radical colorectal cancer surgery can provide excellent analgesic effects, reduce opioid consumption, promote early recovery, reduce stress response, improve recovery quality, and has good safety. This provides clinical evidence for optimizing perioperative pain management in colorectal cancer and improving ERAS implementation protocols. Future research could consider conducting multi-center, large-sample randomized controlled trials to further verify the efficacy of TAP block; exploring optimal implementation protocols for TAP block, including local anesthetic selection, timing of administration, and comparison of single vs continuous administration; evaluating the impact of TAP block on long-term prognosis, including chronic pain, tumor recurrence, and long-term survival; and conducting health economic evaluations to provide more comprehensive evidence support for clinical decision-making.
| 1. | Morgan E, Arnold M, Gini A, Lorenzoni V, Cabasag CJ, Laversanne M, Vignat J, Ferlay J, Murphy N, Bray F. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023;72:338-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1885] [Cited by in RCA: 1536] [Article Influence: 512.0] [Reference Citation Analysis (8)] |
| 2. | Kajiwara Y, Ueno H. Essential updates 2022-2023: Surgical and adjuvant therapies for locally advanced colorectal cancer. Ann Gastroenterol Surg. 2024;8:977-986. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 3. | Albers-Warlé KI, Helder LS, Groh LA, Polat F, Panhuizen IF, Snoeck MMJ, Kox M, van Eijk L, Joosten LAB, Netea MG, Negishi Y, Mhlanga M, Keijzer C, Scheffer GJ, Warlé MC. Postoperative Innate Immune Dysregulation, Proteomic, and Monocyte Epigenomic Changes After Colorectal Surgery: A Substudy of a Randomized Controlled Trial. Anesth Analg. 2025;140:185-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 4. | Kannan V, Ullah N, Geddada S, Ibrahiam A, Munaf Shakir Al-Qassab Z, Ahmed O, Malasevskaia I. Impact of "Enhanced Recovery After Surgery" (ERAS) protocols vs. traditional perioperative care on patient outcomes after colorectal surgery: a systematic review. Patient Saf Surg. 2025;19:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 31] [Reference Citation Analysis (1)] |
| 5. | Negm EM, Younus MA, Morsy AA, El Gammal SMS, El-Harrisi MA, Sameaa FAA, Rashad RAM, Elserafy TS, Gouda AM. Opioid-sparing multimodal analgesia for post-craniotomy pain: a randomized, double-blind, placebo-controlled trial. BMC Anesthesiol. 2025;25:437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 6. | Zhang L, Jia Z, Gao T, Wang Y, Zhao Y, Li J, Yu Y, Li Q, Wang G. A randomized controlled trial evaluating the effects of transversus abdominis plane block with compound lidocaine hydrochloride injection on postoperative pain and opioid consumption and gastrointestinal motility in patients undergoing gynecological laparotomy. Front Mol Neurosci. 2023;16:967917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 7. | Kitagawa H, Manabe T, Yamada Y, Sato H, Takesue S, Hiraki M, Kawaguchi A, Sakaguchi Y, Noshiro H. A prospective randomized study of multimodal analgesia combined with single injection transversus abdominis plane block versus epidural analgesia against postoperative pain after laparoscopic colon cancer surgery. Int J Colorectal Dis. 2023;39:12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 8. | Geng ZY, Zhang Y, Bi H, Zhang D, Li Z, Jiang L, Song LL, Li XY. Addition of preoperative transversus abdominis plane block to multimodal analgesia in open gynecological surgery: a randomized controlled trial. BMC Anesthesiol. 2023;23:21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 9. | Ma N, Duncan JK, Scarfe AJ, Schuhmann S, Cameron AL. Clinical safety and effectiveness of transversus abdominis plane (TAP) block in post-operative analgesia: a systematic review and meta-analysis. J Anesth. 2017;31:432-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 85] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 10. | Zhou M, Yu F, Xu Y, Wu J, Luowu L, Tang Q, Hao X, Shao K, Ye M, Bo L, Zhou L, Jiang C. Combining ropivacaine transversus abdominis plane block with intravenous lidocaine infusion in adults undergoing colorectal cancer surgery: an open-label, dose-escalation exploratory trial. BMC Anesthesiol. 2025;25:357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Erol MK, Şengel A, Tammo Ö, Seçilmiş S, Kaya F. The effect of TAP block use in postoperative analgesic in cesarean section. Eur Rev Med Pharmacol Sci. 2023;27:2786-2793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 12. | Matsota P, Karalis V, Saranteas T, Kiospe F, Markantonis SL. Ropivacaine pharmacokinetics in the arterial and venous pools after ultrasound-guided continuous thoracic paravertebral nerve block. J Anaesthesiol Clin Pharmacol. 2024;40:283-292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 13. | Földi M, Soós A, Hegyi P, Kiss S, Szakács Z, Solymár M, Pétervári E, Balaskó M, Kusza K, Molnár Z. Transversus Abdominis Plane Block Appears to Be Effective and Safe as a Part of Multimodal Analgesia in Bariatric Surgery: a Meta-analysis and Systematic Review of Randomized Controlled Trials. Obes Surg. 2021;31:531-543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 14. | Gelman D, Gelmanas A, Urbanaitė D, Tamošiūnas R, Sadauskas S, Bilskienė D, Naudžiūnas A, Širvinskas E, Benetis R, Macas A. Role of Multimodal Analgesia in the Evolving Enhanced Recovery after Surgery Pathways. Medicina (Kaunas). 2018;54:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 15. | He J, Qin S, Wang Y, Ye Q, Wang P, Zhang Y, Wu Y. Rescue analgesia with a transversus abdominis plane block alleviates moderate-to-severe pain and improves oxygenation after abdominal surgery: a randomized controlled trial. Front Pain Res (Lausanne). 2024;5:1454665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Darras M, Schneider C, Marguerite S, Saadé S, Maechel AL, Oulehri W, Collange O, Mazzucotelli JP, Mertes PM, Kindo M. Multimodal analgesia with parasternal plane block protocol within an enhanced recovery after cardiac surgery program decreases opioid use. JTCVS Open. 2024;22:25-35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 17. | Lohsiriwat V. Opioid-sparing effect of selective cyclooxygenase-2 inhibitors on surgical outcomes after open colorectal surgery within an enhanced recovery after surgery protocol. World J Gastrointest Oncol. 2016;8:543-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 17] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 18. | Doronio GM, Lee ASD. The Effect of Implementing a Standardized Enhanced Recovery After Surgery Pain Management Pathway at an Urban Medical Center in Hawaii. AORN J. 2023;118:391-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Liu F, Tan J, Pan Y. Prevention of deep vein thrombosis in postoperative orthopedic patients: a hybrid meta-analysis and clinical case study. Front Med (Lausanne). 2025;12:1603191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Zhang L, Zhang F, Xiao CJ, Shu YF, Li Z, Wang J, Tang WJ. Impact of a Quantitative Early Activity Program on Gastrointestinal Function Following Laparoscopic Pancreaticoduodenectomy: A Single-Center Retrospective Analysis. Surg Laparosc Endosc Percutan Tech. 2025;35:e1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 21. | Squeo F, Celiberto F, Ierardi E, Russo F, Riezzo G, D'Attoma B, Di Leo A, Losurdo G. Opioid-induced Constipation: Old and New Concepts in Diagnosis and Treatment. J Neurogastroenterol Motil. 2024;30:131-142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 22. | Schneider KM, Blank N, Alvarez Y, Thum K, Lundgren P, Litichevskiy L, Sleeman M, Bahnsen K, Kim J, Kardo S, Patel S, Dohnalová L, Uhr GT, Descamps HC, Kircher S, McSween AM, Ardabili AR, Nemec KM, Jimenez MT, Glotfelty LG, Eisenberg JD, Furth EE, Henao-Mejia J, Bennett FC, Pierik MJ, Romberg-Camps M, Mujagic Z, Prinz M, Schneider CV, Wherry EJ, Bewtra M, Heuckeroth RO, Levy M, Thaiss CA. The enteric nervous system relays psychological stress to intestinal inflammation. Cell. 2023;186:2823-2838.e20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 278] [Cited by in RCA: 247] [Article Influence: 82.3] [Reference Citation Analysis (5)] |
| 23. | Li R, Ye JJ, Gan L, Zhang M, Sun D, Li Y, Wang T, Chang P. Traumatic inflammatory response: pathophysiological role and clinical value of cytokines. Eur J Trauma Emerg Surg. 2024;50:1313-1330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 46] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 24. | Procházka V, Lacina L, Smetana K Jr, Svoboda M, Skřivanová K, Beňovská M, Jarkovský J, Křen L, Kala Z. Serum concentrations of proinflammatory biomarker interleukin-6 (IL-6) as a predictor of postoperative complications after elective colorectal surgery. World J Surg Oncol. 2023;21:384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 25. | Ali SB, Cecchin A, Lucchesi C, Putty T, Edwards S, Petrou T, Coates P, Ferrante A, Pucar PA, King J, Banovic T. Can C-reactive protein be used as a surrogate marker of IL-6 in a broad array of clinical entities? Biomark Med. 2023;17:1001-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (13)] |
| 26. | Feng Y, Chang P, Liu J, Zhang WS. Effects and mechanisms of perioperative medications on the hypothalamic pituitary adrenal response to surgical injury: A narrative review. J Clin Anesth. 2024;94:111367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 27. | Ma M, Zheng Z, Zeng Z, Li J, Ye X, Kang W. Perioperative Enteral Immunonutrition Support for the Immune Function and Intestinal Mucosal Barrier in Gastric Cancer Patients Undergoing Gastrectomy: A Prospective Randomized Controlled Study. Nutrients. 2023;15:4566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 28. | Fariha Kaunain S, Kumar Pandurangan A. Immunotherapy for Colorectal Cancer: Recent Advancements. Innov Discov. 2025;2:12. [DOI] [Full Text] |
| 29. | Campfort M, Cayla C, Lasocki S, Rineau E, Léger M. Early quality of recovery according to QoR-15 score is associated with one-month postoperative complications after elective surgery. J Clin Anesth. 2022;78:110638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 30. | Huang L, Zhang T, Wang K, Chang B, Fu D, Chen X. Postoperative Multimodal Analgesia Strategy for Enhanced Recovery After Surgery in Elderly Colorectal Cancer Patients. Pain Ther. 2024;13:745-766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 31. | Algyar MF, Abdelsamee KS. Laparoscopic assisted versus ultrasound guided transversus abdominis plane block in laparoscopic bariatric surgery: a randomized controlled trial. BMC Anesthesiol. 2024;24:133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 32. | Nair P, Behera CR, Patra RK, Shekar N, Rao LS, Pujari P, Panda B, Mishra A. Efficacy and Cost-Effectiveness of Laparoscopic Transversus Abdominis Plane (TAP) Block in Laparoscopic Cholecystectomy: A Comparison With the Non-TAP Group. Cureus. 2022;14:e32038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |