Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116480
Revised: January 15, 2026
Accepted: February 26, 2026
Published online: April 27, 2026
Processing time: 133 Days and 1.9 Hours
Laparoscopic sleeve gastrectomy (LSG) is an effective bariatric procedure; how
To evaluate short-term weight loss outcomes and comorbidity improvements in patients with obesity within 12 months after LSG.
A single-center retrospective analysis was conducted on 122 patients with obesity who underwent LSG between June 2019 and June 2024. Weight, body mass index, excess weight loss percentage, total weight loss percentage, and metabolic pa
Significant reductions in weight (115.36 ± 18.72 kg to 90.65 ± 18.59 kg) and body mass index (38.73 ± 5.17 kg/m2 to 30.32 ± 5.01 kg/m2) were observed at 12 months (P < 0.05). Excess weight loss percentage reached 71.84% and total weight loss percentage showed a consistent upward trend. Marked improvements were also observed in type 2 diabetes, hypertension, dyslipidemia, hyperuricemia, and fatty liver disease (all P < 0.05).
LSG is effective for achieving significant short-term weight loss and ameliorating obesity-related metabolic comorbidities, highlighting its value as a therapeutic intervention for severe obesity.
Core Tip: In this single-center retrospective study, 122 Chinese adults with obesity underwent laparoscopic sleeve gastrectomy and were followed up for 12 months. Mean body mass index fell from 38.7 kg/m2 to 30.3 kg/m2, excess weight loss reached 71.8%, and 72% of patients achieved effective weight loss. Among 28 patients with type 2 diabetes, 54% achieved complete remission; significant reductions were also observed in hypertension, hyperuricemia, and fatty liver. No major surgical complications were observed. These results confirm that laparoscopic sleeve gastrectomy produces rapid, sustained weight loss and broad metabolic benefits within the first postoperative year.
- Citation: Fu Q, Xiang HF, Zhang SY, Liu XL, Yu JH, Zhang ZQ. Short-term weight control and complications in patients with obesity following laparoscopic sleeve gastrectomy. World J Gastrointest Surg 2026; 18(4): 116480
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116480.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116480
Obesity is an increasingly serious public health issue worldwide, with its annual incidence rate rising, posing a serious threat to human health and quality of life[1,2]. According to an epidemiological survey, the prevalence rates of obesity and overweight in China are 14.1% and 34.8%, respectively, and there is a significant trend toward younger age[3]. Obesity is an independent risk factor for metabolic diseases such as type 2 diabetes mellitus (T2DM) and hypertension, and is closely associated with a significant increase in cardiovascular disease, malignant tumors, and all-cause mortality[4,5]. The traditional treatments for obesity include diet control, exercise, and medication[6]. However, these methods often have limitations in terms of long-term efficacy and patient compliance[7]. Traditional nonsurgical treatment methods often fail to achieve ideal weight control and comorbidity relief[8]. Therefore, surgery has gradually become an important treatment method for patients with moderate to severe obesity.
Laparoscopic sleeve gastrectomy (LSG) is an emerging bariatric surgical procedure that has been widely used in clinical practice in recent years[9]. LSG involves removing approximately 70%-80% of the gastric body to form a narrow tubular gastric cavity. This procedure significantly reduces gastric capacity and ghrelin secretion while preserving pyloric function, enabling rapid weight loss and reducing the risk of long-term nutritional complications[10]. Compared with traditional gastric bypass surgery, LSG has the advantages of a simple operation, a low complication rate, and preservation of gastrointestinal tract continuity[11]. However, most current research on LSG has focused on long-term weight changes and improvements in comorbidities, and relatively little research has been conducted on short-term weight control outcomes and early relief of comorbidities after surgery.
In bariatric surgery research, “short-term” follow-up is commonly defined as 12-24 months postoperatively, distinguishing it from “medium-term” and “long-term” follow-ups[12,13]. The 12-month timeframe is critical as it reflects the peak early weight loss, stabilizes metabolic adaptations, and serves as a strong predictor of long-term surgical success[14,15]. Short-term weight control and early relief from complications are crucial for boosting patients' confidence in treatment, supporting postoperative recovery, and predicting long-term outcomes. These short-term weight benefits can also enhance adherence to healthy lifestyle changes after surgery. The early identification and treatment of possible weight rebound or unresolved comorbidities after surgery can facilitate timely adjustment of the treatment plan and improve the long-term prognosis of patients. This study aims to evaluate the short-term therapeutic effects of LSG by retrospectively analyzing short-term weight control and relief of comorbidities in patients with obesity after LSG and to explore the factors affecting short-term weight control and relief of comorbidities. These findings are expected to provide scientific evidence for clinical practice, further optimize surgical treatment strategies for patients with obesity, and improve their quality of life and long-term prognoses. Additionally, this study will serve as a reference for future research, contributing to the ongoing advancement of bariatric surgery.
This single-center, retrospective study involved patients with obesity who underwent LSG at the Third People’s Hospital of Chengdu between June 2019 and June 2024. All clinical patients’ data were obtained from the hospital's electronic medical record system and surgical database. After screening, 122 patients were included in the study. This study was reviewed and approved by the Institutional Review Board of the Third People’s Hospital of Chengdu, approval No. 2024-S-252.
Inclusion criteria: (1) Age between 18 years and 65 years; (2) Patients who met the diagnostic criteria for simple obesity in the 2019 edition of the “Chinese Guidelines for Surgical Treatment of Obesity and Type 2 Diabetes”[16]: Body mass index (BMI) ≥ 32.5 kg/m2, or BMI of 27.5 kg/m2 ≤ 32.5 kg/m2 and two components of metabolic syndrome or accompanied by TDM; (3) Patients who met the surgical indications for LSG and had undergone surgical treatment after comprehensive consideration; and (4) patients with complete baseline data and follow-up visit data.
Exclusion criteria: (1) Patients who had undergone other bariatric or upper gastrointestinal surgeries in the past; (2) Patients with severe functional disorders of vital organs such as the heart, liver, and kidneys, or with malignant tumors; (3) Patients who were pregnant or planning pregnancy within 1 year; and (4) Patients participating in other clinical trials related to weight loss.
All patients were administered general anesthesia. Surgical procedures were performed by surgeons with more than 5 years of experience in bariatric surgery, and the standardized four-port laparoscopic technique was uniformly adopted. A 10-mm trocar was inserted through an infraumbilical incision to serve as the camera port, maintaining a pneumoperitoneum of 12-14 mmHg. Three additional trocars - 5 mm, 5 mm, and 12 mm - were placed under the left costal margin, right costal margin, and left lower quadrant, respectively, as working ports. After diagnostic laparoscopy, the gastrocolic ligament along the greater curvature was opened, and the greater curvature of the stomach was fully mobilized toward the fundus, preserving the right gastric and right gastroepiploic vessels while carefully avoiding injury to the spleen and pancreas. A 36-Fr or 40-Fr gastric calibration tube was inserted as a guide. Starting 2-3 cm proximal to the pylorus and continuing along the lesser curvature to the gastroesophageal junction, a linear stapler loaded with a blue cartridge was used sequentially to resect approximately 70%-80% of the gastric greater curvature, creating a tubular gastric sleeve approximately 10-15 cm in length and 3-4 cm in diameter. The patients received a liquid diet for 1 day preoperatively. Postoperatively, oral intake was withheld for 6 hours. From postoperative day 1, a graded dietary progression from clear to full liquids, semi-solids, and soft solids was instituted. Prophylactic antibiotics (second-generation cephalosporins) were administered for 24-48 hours, and patient-controlled analgesia was continued for 48 hours.
Baseline data and postoperative follow-up data of patients were extracted through the hospital's electronic medical record system, specifically including: (1) Baseline data: Sex, age, preoperative weight, BMI, preoperative laboratory tests [fasting plasma glucose (FPG), glycated hemoglobin (HbA1c), total cholesterol (TC), triglycerides (TG), and uric acid levels, and systolic and diastolic blood pressure], and preoperative medication history (hypoglycemic drugs, anti-hypertensive drugs, lipid-lowering drugs, etc.); (2) Surgery-related data: Operation duration, gastric tube model, weight of resected gastric tissue, postoperative hospital stay, and occurrence of postoperative complications (such as bleeding, gastric fistula, and intestinal obstruction); and (3) Follow-up data: Weight, BMI, laboratory examination indicators, adjustment of medication for comorbidities, and occurrence of adverse reactions at 1 month, 3 months, 6 months, and 12 months postoperatively.
Weight and BMI: The patient’s weight was measured before surgery and at 1 month, 3 months, 6 months, and 12 months postoperatively, and BMI was calculated [BMI = weight (kg)/height2 (m2)].
Excess weight loss percentage: It was used to assess the degree of weight loss in patients and was calculated as excess weight loss percentage (EWL%) = (preoperative weight - follow-up weight)/(preoperative weight - ideal weight) × 100%. The ideal weight was calculated based on an ideal BMI of 25 kg/m2 using the following formula: Ideal weight = 25 × height2 (m2).
Overall weight reduction percentage: It was used to assess the change in a patient’s overall weight. It was calculated as weight reduction percentage (TWL%) = (preoperative weight - follow-up weight)/preoperative weight × 100%.
Therapeutic effect: This was calculated based on three criteria: (1) Weight loss efficacy, classified as invalid when EWL% < 25%; effective when 25% ≤ EWL% < 50%; significantly effective when 50% ≤ EWL% < 75%; (2) Diabetes remission, defined as complete remission when FPG level < 5.6 mmol/L and HbA1c level < 5.7%; partial remission when 5.6 ≤ FPG level < 6.9 mmol/L or 5.7% ≤ HbA1c level < 6.5%, with a reduction in hypoglycemic drug dosage of ≥ 50%; improvement when the HbA1c level decreased by ≥ 1% but did not meet the partial remission criteria; and, invalid when none of these criteria were met; (3) Hypertension remission was defined as systolic blood pressure < 130 mmHg and diastolic blood pressure < 80 mmHg on two consecutive visits at least 1 month apart, with complete withdrawal of anti-hypertensive drugs (complete remission) or ≥ 50% reduction in drug number/dosage (partial remission); and (4) Hyperuricemia remission was defined as serum uric acid < 420 μmol/L in men and < 360 μmol/L in women, accompanied by the discontinuation of urate-lowering therapy or maintenance on ≤ 50% of the pre-operative dose.
A combination of outpatient and telephone follow-ups was adopted. The follow-up periods were 1 month, 3 months, 6 months, and 12 months postoperatively. Patients were advised to visit the hospital’s weight loss clinic at these time points for re-examination. Anthropometric indicators such as height and weight were measured, venous blood was collected to evaluate the biochemical indicators, and related examinations for comorbidities (such as blood pressure and blood sugar) were completed. Data on weight changes, medication adjustments, and symptom changes were collected via phone from patients unable to attend follow-up visits on schedule, and they were reminded to complete the necessary examinations.
Data analysis was conducted using SPSS 26.0 statistical software, and GraphPad Prism 9.0 was used to draw statistical graphs. Measurement data that conformed to a normal distribution were expressed as mean ± SD, and independent sample t-tests or paired t-tests were used for comparisons between groups. Count data were expressed as n (%), and comparisons between groups were conducted using the χ2 test or Fisher’s exact probability method. Weight loss effect was taken as the dependent variable [0: Effective group (EWL% ≥ 50%); 1: General group (EWL% < 50%)], and indepen
A total of 122 patients who successfully underwent LSG were included in this study. The average age of all patients was 33.11 ± 7.69 years, and the age range was 18-52 years. The operation time averaged 119.33 minutes, ranging from 57.76 minutes to 203.97 minutes. The preoperative average weight and BMI were 115.36 ± 18.72 kg and 38.73 ± 5.17 kg/m2, respectively. The average postoperative hospital stay was 3.84 ± 1.31 days.
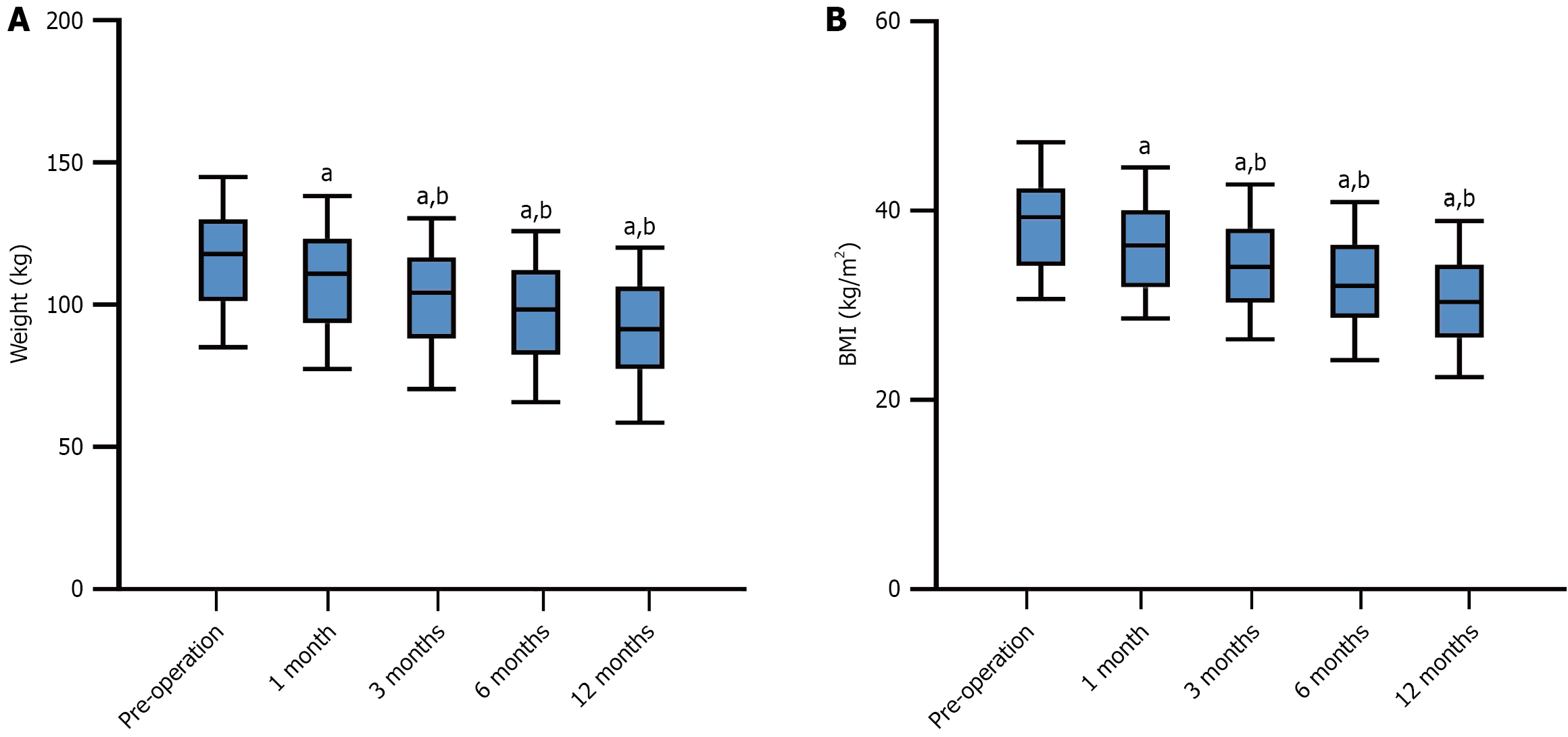
The 1-year weight and BMI change trends of patients who underwent LSG are shown in Figure 1. After 12 months of LSG, the average weight and average BMI of the patients decreased from 115.36 ± 18.72 kg and 38.73 ± 5.17 kg/m2 before surgery to 90.65 ± 18.59 kg and 30.32 ± 5.01 kg/m2, respectively. The patients’ weight and BMI at each follow-up time point (1 month, 3 months, 6 months, and 12 months) decreased significantly compared with those before the surgery (P < 0.05). Moreover, significant reductions in weight and BMI were observed at each follow-up point compared with the previous visit (P < 0.05).
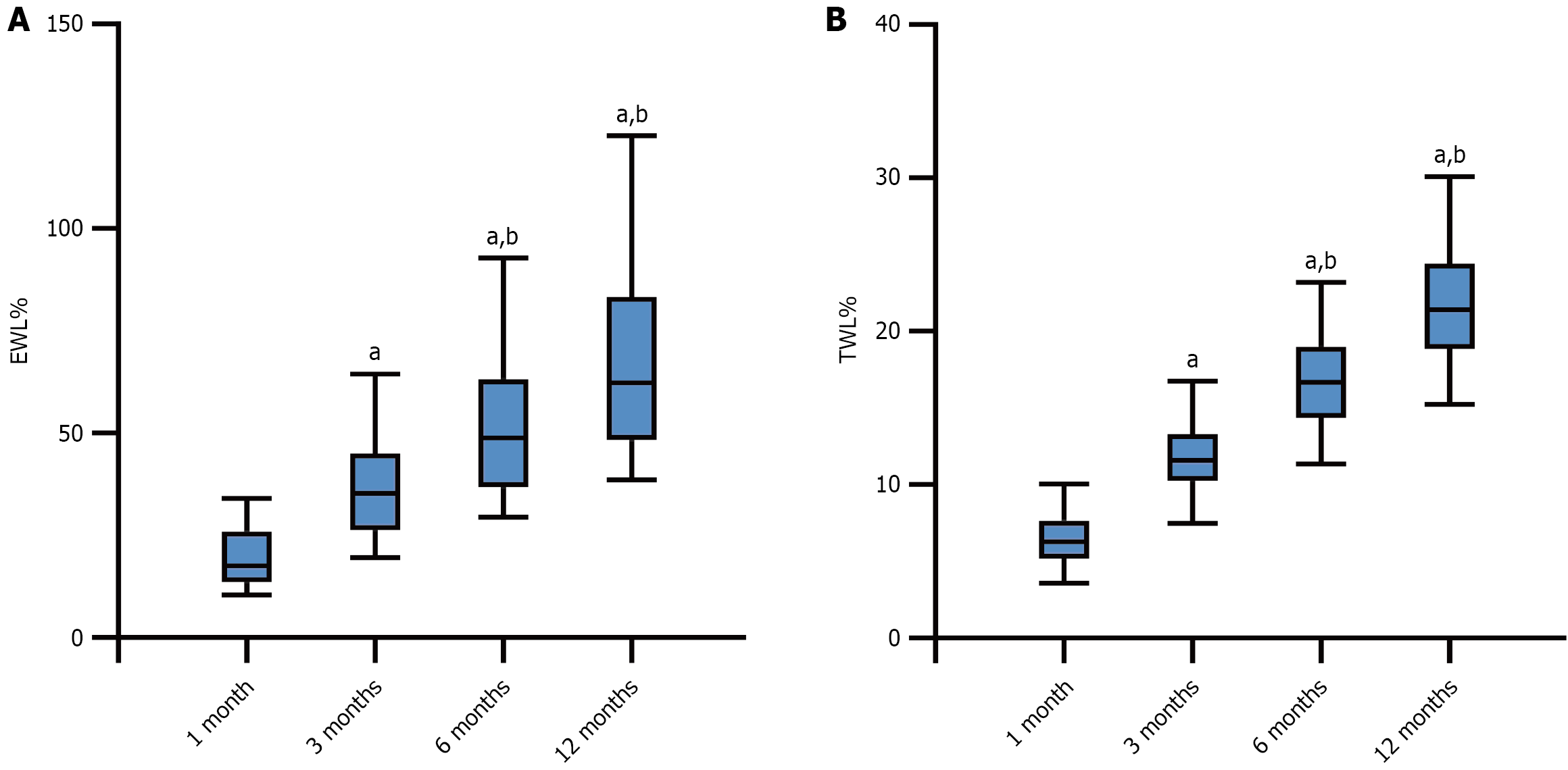
The EWL% and TWL% of the patients decreased continuously (Figure 2) The EWL% at 1 month, 3 months, 6 months, and 12 months was 20.80%, 38.63%, 54.65%, and 71.84%, respectively. The EWL% significantly increased compared with the baseline and the time point of the last follow-up (P < 0.05). Additionally, at 6 months postoperatively, 39 (31.97%), 21 (17.21%), and 60 (49.18%) patients achieved a good, excellent, and effective EWL%, respectively. At 12 months, 50 patients (40.98%) achieved good, 38 (31.15%) achieved excellent, and 88 (72.13%) achieved effective EWL%. During the 12-month postoperative period, the TWL% of patients also showed a significant upward trend (Figure 2). TWL% also showed a significant increase at each follow-up time point (P < 0.05).
In this study, 28 patients were diagnosed with T2DM. All patients took hypoglycemic drugs before surgery; however, the effect on blood sugar control was poor. The average preoperative FPG level was 9.13 ± 2.84 mmol/L, and the average HbA1c level was 7.72 ± 1.21 (Table 1). Starting from the third month postoperative follow-up, the FPG and HbA1c levels of the patients decreased significantly compared with the preoperative period (P < 0.05). At 12 months follow-up, the FPG and HbA1c levels decreased to 5.95 ± 1.49 mmol/L and 6.03 ± 0.39%, respectively.
The preoperative levels of TC, TG, and uric acid were 5.86 ± 0.20 mmol/L, 2.71 ± 0.71 mmol/L and 354.68 ± 55.28 μmol/L, respectively (Table 2). Compared with the preoperative levels, the levels of TC, TG, and uric acid at each follow-up time point decreased significantly (P < 0.05). At the 12-month follow-up, the levels of TC, TG and uric acid of the patients were 4.75 ± 0.62 mmol/L, 1.55 ± 0.38 mmol/L and 353.68 μmol/L, respectively.
| Time | Number | TC level (mmol/L) | TG level (mmol/L) | Uric acid level (μmol/L) |
| Pre-operation | 122 | 5.86 ± 0.20 | 2.71 ± 0.71 | 441.60 ± 62.27 |
| 1 month | 122 | 5.62 ± 0.45a | 2.46 ± 0.78a | 409.83 ± 55.27a |
| 3 months | 122 | 5.37 ± 0.58a,b | 2.04 ± 0.56a,b | 388.25 ± 48.58a,b |
| 6 months | 122 | 5.09 ± 0.67a,b | 1.72 ± 0.41a,b | 366.51 ± 46.30a,b |
| 12 months | 122 | 4.75 ± 0.62a,b | 1.55 ± 0.38a,b | 354.68 ± 55.28a |
The average systolic and diastolic blood pressures of the patients were 139.66 ± 17.54 mmHg and 85.09 ± 13.14 mmHg, respectively, at baseline (Table 3). Compared with the preoperative period, the patients’ systolic blood pressure showed a significant decrease starting from the third month (P < 0.05). Diastolic blood pressure began to decrease significantly from the sixth month. By the end of the follow-up (12 months), the average systolic and diastolic blood pressures of the patients were 128.81 ± 14.08 mmHg and 79.24 ± 9.81 mmHg, respectively.
Preoperatively, the prevalence of fatty liver, hypertension, hyperuricemia, and diabetes were 75 (61.48%), 38 (31.15%), 35 (28.69%), and 28 (22.96%) cases, respectively (Table 4). At the 12-month follow-up, fatty liver (P < 0.001), hypertension (P < 0.001), hyperuricemia (P = 0.014), and diabetes (P = 0.029) were significantly alleviated compared with those before the operation.
| Time | Fatty liver | Hypertension | Hyperuricemia | T2DM |
| Pre-operation | 75 (61.48) | 38 (31.15) | 35 (28.69) | 28 (22.95) |
| 12 months | 36 (29.51) | 13 (10.66) | 19 (15.57) | 15 (12.30) |
| χ2 value | 25.14 | 15.49 | 6.09 | 4.77 |
| P value | < 0.001 | < 0.001 | 0.014 | 0.029 |
Taking EWL% as the dependent variable [0: Effective group (EWL% ≥ 50%); 1: General group (EWL% < 50%)], the influencing factors of weight loss effect after LSD in patients were explored. The results showed that preoperative weight (odds ratio = 1.13, 95% confidence interval: 1.06-1.21, P < 0.001) and BMI (odds ratio = 2.50, 95% confidence interval: 1.58-3.97, P < 0.001) were independent risk factors affecting the weight loss effect, suggesting that patients with more severe obesity require closer postoperative monitoring (Table 5).
| Variables | Univariate logistic | Multivariate logistic | ||||
| β | P value | OR (95%CI) | β | P value | OR (95%CI) | |
| Age | 0.01 | 0.686 | 1.01 (0.96-1.06) | - | - | - |
| Operation time | 0.00 | 0.765 | 1.00 (0.99-1.01) | - | - | - |
| Hospital stay | 0.18 | 0.246 | 1.20 (0.88-1.62) | - | - | - |
| Preoperative weight | 0.09 | < 0.001 | 1.09 (1.05-1.13) | 0.12 | < 0.001 | 1.13 (1.06-1.21) |
| Preoperative BMI | 0.64 | < 0.001 | 1.89 (1.48-2.43) | 0.92 | < 0.001 | 2.50 (1.58-3.97) |
| Preoperative TC | -1.07 | 0.281 | 0.34 (0.05-2.41) | - | - | - |
| Preoperative TG | -0.34 | 0.240 | 0.71 (0.40-1.26) | - | - | - |
| Preoperative uric acid | 0.00 | 0.551 | 1.00 (1.00-1.01) | - | - | - |
| Preoperative systolic blood pressure | 0.01 | 0.657 | 1.01 (0.98-1.03) | - | - | - |
| Preoperative diastolic blood pressure | -0.02 | 0.146 | 0.98 (0.95-1.01) | - | - | - |
Obesity and associated metabolic complications have become significant public health challenges for adults in China[2]. This study systematically evaluated the short-term weight loss outcomes and resolution of comorbidities following LSG by retrospectively analyzing 1-year postoperative clinical data from 122 patients with obesity who underwent the procedure. Patients exhibited sustained significant reductions in weight and BMI within 12 months post-surgery, achie
The results showed a mean reduction in body weight and BMI of 24.71 kg/m2 and 8.41 kg/m2, respectively, at 12 months postoperatively. Furthermore, body weight and BMI were significantly lower at each follow-up time point (1 month, 3 months, 6 months, and 12 months) than those at the preceding interval. Goldenshluger et al[17] conducted a 10-year follow-up study and observed that 10 years after LSG surgery, the BMI of patients decreased by 33.24% and the long-term TWL% was 32.31%. Sivakumar et al[18] also reported that the average BMI of patients who underwent LSG surgery could decrease by up to 11.12 kg/m2 in 12 months postoperatively. These findings are consistent with the results of the present study. The core mechanism underlying this sustained weight-loss trend can be explained in two ways. Additionally, LSG directly reduces gastric capacity by excising 70%-80% of the gastric body to form a tubular stomach, thereby significantly restricting food intake[19]. Simultaneously, the removal of the gastric fundus diminishes ghrelin secretion[20]. As a key hormone regulating appetite, reduced ghrelin levels effectively suppress hunger sensations, decreasing both the frequency and total volume of food consumption[21]. Beyond these two core mechanisms, accumulating evidence suggests that LSG-induced metabolic improvements are also closely linked to altered incretin hormone secretion, particularly glucagon-like peptide-1 (GLP-1)[22,23]. GLP-1, secreted by intestinal L-cells, enhances insulin secretion, suppresses glucagon release, and delays gastric emptying - all contributing to glycemic control and weight regulation[24]. Although this study did not directly measure GLP-1 levels, previous clinical studies have reported significant elevations in circulating GLP-1 post-LSG, which synergize with reduced ghrelin to amplify the metabolic benefits[25]. This incretin-mediated pathway may partially explain the rapid remission of T2DM observed in our study, even before substantial weight loss. Furthermore, strict management of the postoperative diet structure (gradually transitioning from liquid to soft food) and the enhanced compliance of patients with a healthy lifestyle have further strengthened the weight loss effect[26].
In terms of the remission of comorbidities, the results of this study further reveal the multidimensional improvement effect of LSG on metabolic abnormalities. For patients with T2DM, the FPG level decreased from 9.13 mmol/L to 5.95 mmol/L at 12 months postoperatively, whereas HbA1c levels decreased from 7.72% to 6.03%. The reduction in blood glucose levels was statistically significant from 3 months postoperatively. In a previous study, Murshid et al[27] reported through a retrospective study involving 320 patients that, within 1 year after the operation, the HbA1c of the patients decreased from 8.38% to 6.43%, and the HbA1c of 75% of the patients dropped below 6.5%. First, this improvement can be attributed primarily to weight reduction that directly improves insulin sensitivity, particularly as the reduction in visceral fat decreases the release of inflammatory cytokines (such as tumor necrosis factor-α and interleukin-6) from adipose tissue[28,29]. Second, LSG may directly regulate glucose metabolism through non-weight-dependent mechanisms, such as altering gastrointestinal hormone secretion (elevated GLP-1 and decreased GIP levels) and improving the structure of the intestinal flora[30,31]. Moreover, a reduction in hepatic fat content markedly improved hepatic insulin resistance, thereby restoring normal hepatic glucose output[32]. Improvement in dyslipidemia is closely associated with a reduced influx of free fatty acids into the liver and the inhibition of very low-density lipoprotein synthesis[33]. This reduction in blood pressure may stem from decreased sympathetic nervous system activity, diminished renal sodium reabsorption, and reduced adipose tissue-derived angiotensinogen secretion[34]. Collectively, these multiple mechanisms enable LSG to ameliorate metabolic disorders and restore metabolic homeostasis.
The results of our study have clinical and practical significance. First, they provide reliable localized evidence of the short-term efficacy of LSG in the obese population in China, which is conducive to promoting the standardized development of bariatric and metabolic surgeries in our country. The weight loss effect at 12 months post-surgery is regarded as a key indicator for predicting long-term prognosis. In the present study, 72.13% of the patients achieved an effective weight loss standard, indicating a favorable long-term outcome. This requires clinicians to attach great importance to the “golden window period” in the first year post-surgery and maximize the surgical benefits by strengthening nutritional guidance, exercise advice and psychological support. At the postoperative follow-up management level, the study found that there were time differences in the improvement of indicators such as blood pressure and uric acid levels, suggesting that personalized follow-up plans should be formulated for different comor
However, the study has some limitations that should be addressed in future studies. First, single-center retrospective designs have inherent selection (only including patients with complete data, possibly excluding those lost to follow-up and those with severe complications) and information biases (some follow-up data rely on telephone recall, which may lead to weight reporting errors). In the future, multicenter prospective cohort studies should be conducted to reduce bias through random grouping and blinded assessments. Second, telephone-based weight recall was permitted when patients could not attend the clinic; this may introduce recall or social-desirability bias. To mitigate this, interviewers asked patients to report the most recent weight documented on home scales and pharmacy or primary-care records and to state the date and setting of that measurement; 78% of phone respondents supplied a photo or written record, which was cross-checked against clinic files when available. Nevertheless, lack of standardized, calibrated scales for every time point remains a limitation that may have modestly inflated the apparent weight-loss success. Moreover, the proportion of telephone vs in-person weight assessments (28% vs 72%) was not equally distributed across follow-up intervals; future studies should pre-specify a validation sub-cohort with mandatory on-site weighing to quantify the exact magnitude of telephone-report bias. Third, the follow-up period was only 12 months, which made it impossible to assess the medium- and long-term efficacy of LSG (such as the weight rebound rate within 2-5 years and the recurrence rate of complications). Subsequently, follow-up needs to be extended to 3-5 years to further verify the sustainability of efficacy. Furthermore, the study did not analyze the individual differences in factors influencing the therapeutic effect (such as the stratified impact of sex, age, and preoperative BMI on the weight loss effect), nor did it explore the occurrence of postoperative nutritional complications (such as iron deficiency anemia and vitamin B12 deficiency). In the future, multivariate logistic regression analysis will be needed to screen for predictive factors of therapeutic effects, strengthen postoperative nutritional monitoring, and improve the risk–benefit assessment of LSG. Finally, quantitative data on lifestyle interventions (such as dietary structure and exercise frequency) were not included in this study, making it impossible to clearly define their contribution to short-term efficacy. Subsequent research should collect objective data through tools such as dietary diaries and exercise bracelets to analyze the interaction between lifestyle and surgical efficacy.
This study demonstrated that LSG yielded favorable short-term weight loss outcomes and metabolic improvements in patients with obesity. At the 12-month follow-up, patients achieved an average EWL% of 71.84%, with significant reductions in BMI and marked amelioration of multiple comorbidities, including T2DM, hypertension, and dyslipidemia. These findings confirmed that LSG leads to effective weight loss and metabolic improvement through the dual mechanisms of mechanical restriction and neuroendocrine regulation. This study provides clinical evidence supporting the application of LSG in the obese Chinese population and recommends it as an effective treatment option for patients with obesity meeting the surgical criteria, particularly those with concomitant metabolic disorders. Future multicenter studies with longer follow-up periods are required to further evaluate the long-term efficacy and safety of LSG.
| 1. | Perdomo CM, Cohen RV, Sumithran P, Clément K, Frühbeck G. Contemporary medical, device, and surgical therapies for obesity in adults. Lancet. 2023;401:1116-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 490] [Article Influence: 163.3] [Reference Citation Analysis (3)] |
| 2. | Wang Y, Zhao L, Gao L, Pan A, Xue H. Health policy and public health implications of obesity in China. Lancet Diabetes Endocrinol. 2021;9:446-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 359] [Article Influence: 71.8] [Reference Citation Analysis (0)] |
| 3. | Chen K, Shen Z, Gu W, Lyu Z, Qi X, Mu Y, Ning Y; Meinian Investigator Group. Prevalence of obesity and associated complications in China: A cross-sectional, real-world study in 15.8 million adults. Diabetes Obes Metab. 2023;25:3390-3399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 255] [Article Influence: 85.0] [Reference Citation Analysis (0)] |
| 4. | Tsur AM, Twig G. The actual burden of obesity-accounting for multimorbidity. Lancet Diabetes Endocrinol. 2022;10:233-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 5. | Welsh A, Hammad M, Piña IL, Kulinski J. Obesity and cardiovascular health. Eur J Prev Cardiol. 2024;31:1026-1035. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 127] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 6. | Yanovski SZ, Yanovski JA. Approach to Obesity Treatment in Primary Care: A Review. JAMA Intern Med. 2024;184:818-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 54] [Reference Citation Analysis (0)] |
| 7. | Hood MM, Corsica J, Bradley L, Wilson R, Chirinos DA, Vivo A. Managing severe obesity: understanding and improving treatment adherence in bariatric surgery. J Behav Med. 2016;39:1092-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 8. | Abhishek F, Ogunkoya GD, Gugnani JS, Kaur H, Muskawad S, Singh M, Singh G, Soni U, Julka D, Udoyen AO. Comparative Analysis of Bariatric Surgery and Non-surgical Therapies: Impact on Obesity-Related Comorbidities. Cureus. 2024;16:e69653. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 9. | Vitiello A, Abu-Abeid A, Dayan D, Berardi G, Musella M. Long-Term Results of Laparoscopic Sleeve Gastrectomy: a Review of Studies Reporting 10+ Years Outcomes. Obes Surg. 2023;33:3565-3570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 10. | Al-Mohaidly MT, Al-Asmari AK, Khan HA, Alshngeetee AS, Khan I, Al-Asmari YA, Al-Hussain GO, Alsalem SS, Khan A, Babtain AM, Aljorfi EA, Alshumiesy HA, Aluraifej MA. Laparoscopic sleeve gastrectomy for obesity treatment in adolescents and young adults: a systematic review and meta-analysis. Langenbecks Arch Surg. 2023;408:158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 11. | Hany M, Zidan A, Aboelsoud MR, Torensma B. Laparoscopic sleeve gastrectomy vs one-anastomosis gastric bypass 5-year follow-up: a single-blinded randomized controlled trial. J Gastrointest Surg. 2024;28:621-633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Ismael NH, Shabila NP. Short-Term Weight Loss Outcomes of 104 Mini-Gastric Bypass or One-Anastomosis Gastric Bypass Operations: Retrospective study. Sultan Qaboos Univ Med J. 2024;24:515-522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Ram Sohan P, Mahakalkar C, Kshirsagar S, Bikkumalla S, Reddy S, Hatewar A, Dixit S. Long-Term Effectiveness and Outcomes of Bariatric Surgery: A Comprehensive Review of Current Evidence and Emerging Trends. Cureus. 2024;16:e66500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 14. | Pawłuszewicz P, Wojciak PA, Łukaszewicz A, Chilmonczyk J, Ładny JR, Nadolny K, Razak Hady H. Assessment of Lipid Balance Parameters after Laparoscopic Sleeve Gastrectomy in 1-Year Observation. J Clin Med. 2023;12:4079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 15. | Xu T, Wang C, Zhang H, Han X, Liu W, Han J, Yu H, Chen J, Zhang P, Di J. Timing of Maximal Weight Reduction Following Bariatric Surgery: A Study in Chinese Patients. Front Endocrinol (Lausanne). 2020;11:615. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | The Thyroid and Metabolic Surgery Group of the Chinese Society of Surgery; the Obesity and Diabetes Surgery Committee of the Chinese Medical Association's Surgical Physicians' Branch. [China surgical treatment for obesity and type 2 diabetes guide (2019 edition)]. Zhongguo Shiyong Waike Zazhi. 2019;39:301-306. [DOI] [Full Text] |
| 17. | Goldenshluger M, Iluz R, Beck T, Adileh M, Segev L, Froilych D, Goldenshluger A, Geron N, Goitein D, Hazzan D. Laparoscopic Sleeve Gastrectomy in Adolescents: Ten-Years Follow-up. Obes Surg. 2023;33:32-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Sivakumar J, Chen Q, Chong L, Read M, Ward S, Winter N, Sutherland TR, Hii MW. Effect of laparoscopic sleeve gastrectomy versus laparoscopic Roux-en-Y gastric bypass on body composition. ANZ J Surg. 2024;94:1317-1323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 19. | Chemaly R, Ibrahim Z, Lainas P, Ghaida MA, Kassir NE, Al-Hajj G, Tayar C, Safadi B. Laparoscopic Sleeve Gastrectomy as a First Step Procedure for Oncologic Purposes: An Indication Beyond the Updated Guidelines. Obes Surg. 2024;34:2026-2032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 20. | Dobrescu A, Copaescu C, Zmeu B, Duta C, Bedreag OH, Stoica L, Tarta C, Rogobete AF, Lazar F. Ghrelin Levels and Hunger Sensation after Laparoscopic Sleeve Gastrectomy Compared with Laparoscopic Greater Curvature Plication in Obese Patients. Clin Lab. 2020;66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 21. | van Loenen MR, Geenen B, Arnoldussen IAC, Kiliaan AJ. Ghrelin as a prominent endocrine factor in stress-induced obesity. Nutr Neurosci. 2022;25:1413-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 22. | Ji Y, Lee H, Kaura S, Yip J, Sun H, Guan L, Han W, Ding Y. Effect of Bariatric Surgery on Metabolic Diseases and Underlying Mechanisms. Biomolecules. 2021;11:1582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 23. | Çalık Başaran N, Dotan I, Dicker D. Post metabolic bariatric surgery weight regain: the importance of GLP-1 levels. Int J Obes (Lond). 2025;49:412-417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 48] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 24. | Alonso SP, Valdes S, Doulatram-Gamgaram VK. GLP-1 agonists in glycemic and weight control of type 2 diabetes. New perspectives. Med Clin (Barc). 2025;165:107042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Gala K, Ghusn W, Abu Dayyeh BK. Gut motility and hormone changes after bariatric procedures. Curr Opin Endocrinol Diabetes Obes. 2024;31:131-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 26. | Lin S, Guan W, Hans P, Liang H. Status of Laparoscopic Sleeve Gastrectomy in China: A National Survey. Obes Surg. 2017;27:2968-2973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Murshid KR, Alsisi GH, Almansouri FA, Zahid MM, Boghdadi AA, Mahmoud EH. Laparoscopic sleeve gastrectomy for weight loss and treatment of type 2 diabetes mellitus. J Taibah Univ Med Sci. 2021;16:387-394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 28. | Tong Y, Xu S, Huang L, Chen C. Obesity and insulin resistance: Pathophysiology and treatment. Drug Discov Today. 2022;27:822-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 139] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 29. | Merovci A, Finley B, Hansis-Diarte A, Neppala S, Abdul-Ghani MA, Cersosimo E, Triplitt C, DeFronzo RA. Effect of weight-maintaining ketogenic diet on glycemic control and insulin sensitivity in obese T2D subjects. BMJ Open Diabetes Res Care. 2024;12:e004199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 30. | Kural A, Khan I, Seyit H, Caglar TR, Toklu P, Vural M. Changes in the gut microbiota of morbidly obese patients after laparoscopic sleeve gastrectomy. Future Microbiol. 2022;17:5-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 31. | Cibičková Ľ, Grega M, Dohnal R, Schovánek J. Effect of Laparoscopic Sleeve Gastrectomy on Serum Adipokine Levels. Physiol Res. 2023;72:S165-S172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 32. | Kerr AG, Andersson DP, Dahlman I, Rydén M, Arner P. Adipose Insulin Resistance Associates With Dyslipidemia Independent of Liver Resistance and Involves Early Hormone Signaling. Arterioscler Thromb Vasc Biol. 2023;43:1054-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 33. | Yin J, Du L, Sheng C, You H, Wang X, Qu S. Vitamin C status and its change in relation to glucose-lipid metabolism in overweight and obesity patients following laparoscopic sleeve gastrectomy. Eur J Clin Nutr. 2022;76:1387-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 34. | Elkan H, Baş MM, Kaya B. Impact of Laparoscopic Sleeve Gastrectomy on Thrombomodulin Concentration and Early Markers of Atherosclerosis. J Interv Cardiol. 2022;2022:6152571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |