Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116412
Revised: January 14, 2026
Accepted: February 12, 2026
Published online: April 27, 2026
Processing time: 150 Days and 1.5 Hours
Primary hepatocellular carcinoma (HCC) is a common malignancy worldwide, with surgical resection being the most effective treatment for long-term survival. For complex HCC (large diameter, central location, multiple lesions, or proximity to major vessels), traditional surgery relying on surgeon experience and palpation has limitations including difficulty identifying deep lesions and high positive ma
To evaluate the clinical application value of IOUS combined with ICG fluores
Clinical data of 200 patients with complex HCC who underwent radical he
Baseline characteristics were balanced between the two groups. The combined navigation group had shorter tumor localization time (P < 0.001), less intraoperative blood loss (P = 0.004), lower intraoperative transfusion rate (P = 0.021), higher detection rate of occult lesions (23.3% vs 6.2%, P < 0.001), and higher anatomical resection rate (P = 0.040). The combined navigation group had lower positive margin rate (2.9% vs 13.4%, P = 0.006), higher R0 resection rate (97.1% vs 86.6%, P = 0.006), and greater margin distance (P < 0.001). The combined navigation group had lower overall postoperative complication rate (18.4% vs 28.9%, P = 0.042) and severe complication rate (5.8% vs 12.4%, P = 0.042), and shorter postoperative hospital stay (P = 0.003). With a median follow-up of 28.6 months, the combined navigation group had higher 2-year overall survival rate (76.8% vs 65.2%, P = 0.033) and 2-year disease-free survival rate (58.4% vs 45.7%, P = 0.022), and lower postoperative recurrence rate (36.9% vs 50.5%, P = 0.048). Multivariate analysis showed that application of combined navigation technology was an independent protective factor for postoperative complications (odds ratio = 0.498, P = 0.027), and was also an independent protective factor for overall survival (hazard ratio = 0.584, P = 0.028) and disease-free survival (hazard ratio = 0.631, P = 0.025).
IOUS combined with ICG fluorescence imaging technology can improve tumor localization accuracy in complex HCC surgery, improve margin control, reduce intraoperative blood loss, decrease the incidence of postoperative complications, and improve long-term survival prognosis in patients, demonstrating important clinical application value.
Core Tip: This retrospective study evaluated 200 patients with complex hepatocellular carcinoma undergoing radical hepatectomy. Intraoperative ultrasound combined with indocyanine green fluorescence imaging significantly improved tumor localization, increased R0 resection and anatomical hepatectomy rates, reduced intraoperative blood loss and postoperative complications, and enhanced long-term survival compared with conventional surgery. The combined navigation approach provides real-time visualization and precise resection guidance, offering a safe, effective, and clinically valuable strategy for precision hepatectomy in complex hepatocellular carcinoma.
- Citation: Zhang S, Li SY, Zhou B. Navigation value of intraoperative ultrasound combined with indocyanine green fluorescence in complex hepatocellular carcinoma resection. World J Gastrointest Surg 2026; 18(4): 116412
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116412.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116412
Primary hepatocellular carcinoma (HCC) is one of the most common malignant tumors worldwide, and surgical resection remains the most effective treatment for achieving long-term survival[1]. However, for complex HCC, including cases with large tumor diameter, central location, multiple lesions, or close proximity to important vascular structures, surgical difficulty is significantly increased, and accurate intraoperative localization of tumor boundaries, determination of resection range, and protection of important vascular structures become key to surgical success[2]. Traditional surgery mainly relies on the surgeon’s experience, intraoperative palpation, and hepatic surface anatomical landmarks for tumor localization, but there are obvious limitations in judging deep lesions, small lesions not shown on preoperative imaging, and the precise spatial relationship between tumors and vessels, which may lead to increased positive margin rates, missed occult lesions, and elevated postoperative recurrence rates[3]. Intraoperative ultrasound (IOUS) technology can display liver internal structures and vessel courses in real-time, significantly improving tumor localization accuracy, but its ability to identify isoechoic or small lesions is limited[4]. Indocyanine green (ICG) fluorescence imaging technology utilizes the selective uptake of ICG by hepatocytes and its retention characteristics in tumor tissue, and through near-infrared fluorescence imaging systems can display fluorescence signal differences between tumors and normal liver tissue in real-time, helping to identify tumor boundaries and occult lesions intraoperatively, and can mark the anatomical resection plane[5,6]. Recent studies suggest that IOUS and ICG fluorescence imaging technology have complementary advantages, with the former providing deep anatomical information and vascular relationships, and the latter achieving real-time fluorescence visualization of tumors, and combined application may further improve the precision and safety of complex HCC surgery[7]. However, there are still few systematic evaluation studies on the combined application of the two technologies in complex HCC resection, and their effects on surgical outcomes, oncological outcomes, and long-term prognosis need further verification[8]. Therefore, this study retrospectively analyzed clinical data of 200 patients with complex HCC to compare the therapeutic differences between IOUS combined with ICG fluorescence imaging technology and conventional surgical methods, aiming to evaluate the clinical application value of combined navigation technology in radical resection of complex HCC.
Clinical data of patients who underwent radical hepatectomy for HCC at our hospital from January 2019 to August 2024 were retrospectively analyzed. Patient allocation was based on patient/surgeon preference and equipment availability rather than temporal criteria. The conventional surgery group had cases distributed across the entire period (mean: 17.3 cases/year), and the combined navigation group also had cases throughout the period (mean: 18.4 cases/year), with a temporal overlap coefficient of 0.87, indicating substantial concurrency. All surgeries were performed by the same surgical team (three senior surgeons with > 15 years of hepatobiliary experience), and standardized perioperative care protocols were maintained throughout the study period to minimize temporal confounding. All patients underwent surgery by the same surgical team, with identical surgical indications and technical standards.
Inclusion criteria: (1) Preoperative imaging examination [enhanced computed tomography (CT) or magnetic resonance imaging (MRI)] confirmed diagnosis of primary HCC; (2) Met the definition of complex HCC: Tumor diameter ≥ 5 cm, or central location (according to Makuuchi classification, tumor located in Couinaud segments I, IV, V, VIII, or involving the first or second hepatic hilum region), or multiple lesions (≥ 3), or close to important vascular structures (main hepatic vein, main portal vein or first-order branches, inferior vena cava); (3) Liver function Child-Pugh grade A or B (7-9 points); (4) No surgical contraindications, with indications for hepatectomy; (5) For Child-Pugh A patients, preoperative ICG 15-minute retention rate (ICG-R15) < 30%; for Child-Pugh B patients, ICG-R15 < 20%; (6) Complete clinical data; and (7) Complete follow-up data, follow-up time ≥ 6 months or death during observation period (based on International Consensus Guidelines for HCC and Japanese Clinical Practice Guidelines).
Exclusion criteria: (1) Combined with extrahepatic metastasis; (2) History of previous liver surgery; (3) Received anti-tumor treatment such as transarterial chemoembolization, ablation therapy, or radiotherapy within 6 months before surgery; (4) Severe coagulation dysfunction [prothrombin time-international normalized ratio (PT-INR) > 1.5 or platelets < 50 × 109/L] or dysfunction of other important organs (cardiac function New York Heart Association class ≥ III, renal function serum creatinine > 177 μmol/L, severe pulmonary dysfunction); (5) Allergy to ICG or iodinated contrast agents; (6) Pregnant or lactating women; (7) Combined with other malignant tumors; and (8) Intraoperative exploration found unresectable.
Equipment and standardization: All IOUS examinations used the Aloka ProSound Alpha 7 system (7.5 MHz probe), and ICG fluorescence imaging used the Stryker SPY Elite system. Equipment remained unchanged from January 2019 to August 2024, ensuring standardization. Combined navigation group protocol: All cases received comprehensive IOUS scanning of all 8 liver segments with tumor-vessel relationship mapping and occult lesion search (mean duration: 8.7 ± 2.3 minutes), plus ICG fluorescence imaging with preoperative ICG injection (0.5 mg/kg, 12-24 hours before surgery) and real-time fluorescence visualization during hepatic transection. Conventional surgery group protocol: This group relied on manual palpation, preoperative CT/MRI imaging, and visual inspection. IOUS was used selectively in only 23 of 97 cases (23.7%) for problem-solving scans (mean duration: 3.2 ± 1.1 minutes) when tumors were non-palpable, deep-seated, or location was uncertain. No ICG fluorescence imaging was used.
Detailed surgical protocols preoperative evaluation: Enhanced CT/MRI with three-dimensional reconstruction (Iqqa-Liver or MI-3DVS) to plan resection; liver function assessment including ICG-R15 and CT volumetry ensuring remnant liver volume > 40% (> 50% for cirrhosis). Combined navigation group received peripheral intravenous ICG (0.5 mg/kg, maximum 25 mg) 24 hours preoperatively. Surgical procedure: All surgeries performed by the same senior team (> 10 years’ experience). After laparotomy and exploration, the conventional group used palpation and preoperative imaging with selective IOUS assistance, determining resection range through anatomical landmarks and ischemic boundaries. The combined navigation group systematically: (1) Performed IOUS scanning of entire liver (right/Left lobes, hepatic hilum, IVC and hepatic veins) to confirm tumor characteristics and vessel relationships; (2) Used near-infrared fluorescence imaging (excitation 760 nm, emission 800-830 nm) to observe tumor development, record fluorescence differences (target-to-normal ratio), identify anatomical resection planes after vascular occlusion, and search for occult lesions (< 1 cm not on preoperative imaging); (3) Precisely planned resection with ≥ 1 cm margins, performed parenchymal dissection under fluorescence guidance with real-time adjustments, rechecking resection plane every 2-3 cm; and (4) Examined resection surface post-dissection to confirm no residual tumor, with frozen section for suspicious margins. Both groups used individualized Pringle maneuver (15-minute block, 5-minute release, cumulative < 120 minutes), ultrasonic scalpel/cavitron ultrasonic surgical aspirator for dissection, regional lymphadenectomy, and drainage placement.
Baseline characteristics: Age, gender, body mass index (BMI); hepatitis B virus/hepatitis C virus infection, cirrhosis grade; Child-Pugh class, total bilirubin (TBIL), albumin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), PT-INR, ICG-R15; tumor diameter/number/Location/vascular involvement, alpha-fetoprotein (AFP) level; CT/MRI volumetry; Eastern Cooperative Oncology Group score; comorbidities.
Intraoperative indicators: Operation time, blood loss, transfusion volume, hepatic hilar blocking time, tumor localization time; occult lesion detection (confirmed by pathology); surgical procedure adjustments; actual procedures performed.
Oncological indicators: Margin status (positive rate, distance: < 0.5 cm/0.5-1.0 cm/> 1.0 cm); R0 resection rate; anato
Postoperative recovery: Complications within 90 days (Clavien-Dindo classification): Bile leakage (International Study Group of Liver Surgery criteria), abdominal infection/hemorrhage, liver failure (“50-50” criteria), pleural effusion, pulmonary/wound infection, renal insufficiency, other events; liver function (TBIL, ALT, AST, albumin, PT-INR on day 1, day 3, day 5, and day 7); reoperation rate; 30-day/90-day mortality; hospital stay; time to ambulation/diet/drain removal.
Follow-up: Reexaminations every 2 months (year 1), 3 months (year 2), 6 months (thereafter) including liver function, AFP, enhanced CT/MRI. Follow-up until September 30, 2025 or death.
Recorded: Overall survival (OS), disease-free survival (DFS), recurrence (intrahepatic/extrahepatic sites and timing), 1-year and 2-year OS/DFS rates. Recurrence defined as new lesions with characteristic enhancement patterns confirmed by multi-disciplinary team/pathology.
Subgroup analyses: Subgroup comparison of conventional cases with IOUS (n = 23) vs without (n = 74); temporal analysis comparing early (2019-2021) vs late (2022-2024) periods.
SPSS version 26.0 and R version 4.2.0 used for analysis. Normality tested by Shapiro-Wilk, variance by Levene test. Continuous data: mean ± SD or median (interquartile range); t-test or Mann-Whitney U test for comparison. Categorical data: n (%); χ2 or Fisher’s exact test for comparison. Multivariate logistic regression for complication risk factors (back
This study enrolled a total of 200 patients with complex HCC, including 103 cases in the combined navigation group and 97 cases in the conventional surgery group. There were no statistically significant differences between the two groups in age, gender, BMI, hepatitis B virus infection rate, hepatitis C virus infection rate, degree of cirrhosis, Child-Pugh grade, preoperative liver function indicators, tumor characteristics (tumor size, number, location, vascular involvement, AFP level), imaging evaluation indicators, Eastern Cooperative Oncology Group score, and comorbidities (P > 0.05; Table 1).
| Indicator | Combined navigation group | Conventional surgery group | Statistic | P value |
| General demographic characteristics | ||||
| Age (years) | 54.3 ± 10.7 | 55.8 ± 11.2 | t = 0.978 | 0.329 |
| Gender (male) | 86 (83.5) | 79 (81.4) | χ2 = 0.168 | 0.682 |
| BMI (kg/m2) | 23.8 ± 3.2 | 24.1 ± 3.4 | t = 0.654 | 0.514 |
| Underlying liver disease | ||||
| HBV infection | 89 (86.4) | 82 (84.5) | χ2 = 0.156 | 0.693 |
| HCV infection | 8 (7.8) | 9 (9.3) | χ2 = 0.156 | 0.693 |
| Cirrhosis | χ2 = 0.824 | 0.662 | ||
| No cirrhosis | 28 (27.2) | 24 (24.7) | ||
| Mild cirrhosis | 51 (49.5) | 51 (52.6) | ||
| Moderate to severe cirrhosis | 24 (23.3) | 22 (22.7) | ||
| Preoperative liver function indicators | ||||
| Child-Pugh A | 91 (88.3) | 87 (89.7) | χ2 = 0.092 | 0.762 |
| TBIL (μmol/L) | 16.8 ± 6.3 | 17.2 ± 6.8 | t = 0.431 | 0.667 |
| ALB (g/L) | 41.2 ± 4.6 | 40.8 ± 4.9 | t = 0.601 | 0.548 |
| ALT (U/L) | 42 (28-68) | 45 (30-71) | Z = 0.721 | 0.471 |
| AST (U/L) | 38 (26-59) | 40 (28-62) | Z = 0.683 | 0.495 |
| PT-INR | 1.08 ± 0.12 | 1.09 ± 0.13 | t = 0.567 | 0.571 |
| ICG-R15 (%) | 12.4 ± 5.8 | 12.9 ± 6.1 | t = 0.603 | 0.547 |
| Tumor characteristics | ||||
| Maximum tumor diameter (cm) | 7.8 ± 3.2 | 7.6 ± 3.4 | t = 0.431 | 0.667 |
| Tumor number | χ2 = 0.412 | 0.814 | ||
| Single | 67 (65.0) | 66 (68.0) | ||
| 2 lesions | 24 (23.3) | 21 (21.6) | ||
| ≥ 3 lesions | 12 (11.7) | 10 (10.3) | ||
| Central tumor | 58 (56.3) | 52 (53.6) | χ2 = 0.147 | 0.701 |
| Large vessel involvement | 47 (45.6) | 42 (43.3) | χ2 = 0.109 | 0.741 |
| AFP > 400 ng/mL | 52 (50.5) | 46 (47.4) | χ2 = 0.191 | 0.662 |
| Imaging evaluation | ||||
| Tumor volume (cm3) | 286 (145-512) | 273 (138-498) | Z = 0.512 | 0.609 |
| Estimated resection liver volume (cm3) | 618 (425-856) | 602 (410-841) | Z = 0.621 | 0.535 |
| Residual liver volume percentage (%) | 58.3 ± 12.4 | 57.6 ± 13.1 | t = 0.394 | 0.694 |
| ECOG score | χ2 = 0.286 | 0.867 | ||
| 0 points | 64 (62.1) | 62 (63.9) | ||
| 1 point | 32 (31.1) | 28 (28.9) | ||
| 2 points | 7 (6.8) | 7 (7.2) | ||
| Comorbidities | ||||
| Hypertension | 31 (30.1) | 27 (27.8) | χ2 = 0.127 | 0.722 |
| Diabetes | 18 (17.5) | 15 (15.5) | χ2 = 0.147 | 0.701 |
| Cardiovascular and cerebrovascular disease | 12 (11.7) | 10 (10.3) | χ2 = 0.093 | 0.761 |
The combined navigation group had significantly shorter tumor localization time (P < 0.001) but longer operation time (P = 0.012) than the conventional surgery group. The navigation group demonstrated less intraoperative blood loss (P = 0.004) and lower transfusion rate (P = 0.021). Hepatic hilar blocking time showed no significant difference between groups (P = 0.156). The navigation group achieved higher rates of occult lesion detection (P < 0.001, referring to additional lesions detected immediately intraoperatively and confirmed by pathology, excluding lesions < 5 mm found only on postoperative pathology), surgical procedure adjustment (P = 0.007), and anatomical resection (P = 0.040; Table 2).
| Indicator | Combined navigation group | Conventional surgery group | Statistic | P value |
| Operation time (minutes) | 298.5 ± 68.3 | 276.2 ± 71.6 | t = 2.255 | 0.012 |
| Intraoperative blood loss (mL) | 420 (280-680) | 550 (350-850) | Z = 2.891 | 0.004 |
| Intraoperative transfusion | 28 (27.2) | 41 (42.3) | χ2 = 5.343 | 0.021 |
| Red blood cell units (U)1 | 4 (2-6) | 4 (2-8) | Z = 1.234 | 0.217 |
| Plasma volume (mL)1 | 200 (100-400) | 300 (150-450) | Z = 1.156 | 0.248 |
| Hepatic hilar blocking time (minutes) | 48 (28-72) | 54 (32-78) | Z = 1.421 | 0.156 |
| Tumor localization time (minutes) | 12.6 ± 4.3 | 18.9 ± 6.7 | t = 7.831 | < 0.001 |
| Intraoperative detection of occult lesions2 | 24 (23.3) | 6 (6.2) | χ2 = 12.358 | < 0.001 |
| Number of lesions detected3 | 1 (1-2) | 1 (1-1.5) | Z = 1.156 | 0.248 |
| Surgical procedure adjustment | 31 (30.1) | 14 (14.4) | χ2 = 7.296 | 0.007 |
| Surgical procedure | χ2 = 7.891/Fisher | 0.048 | ||
| Hemi-hepatectomy | 42 (40.8) | 35 (36.1) | ||
| Extended hemi-hepatectomy | 18 (17.5) | 22 (22.7) | ||
| Segmentectomy | 38 (36.9) | 28 (28.9) | ||
| Local resection | 5 (4.9) | 12 (12.4) | ||
| Anatomical resection | 98 (95.1) | 85 (87.6) | χ2 = 4.218 | 0.04 |
The increased operative time (median 15 minutes) is attributed to comprehensive IOUS scanning (approximately 8-10 minutes) and ICG fluorescence imaging (approximately 5-7 minutes). This represents a justified trade-off given significant benefit: 3.6-fold higher occult lesion detection, 10.5% increase in R0 resection rate, and 10.5% reduction in complications. Learning curve analysis showed operative time decreased from 245 ± 48 minutes to 225 ± 42 minutes (P = 0.021) between early (2019-2021) and late (2022-2024) phases while maintaining outcomes, indicating the time overhead diminishes with experience.
The combined navigation group demonstrated lower positive margin rate (P = 0.006), higher R0 resection rate (P = 0.006), and greater margin distance (P < 0.001) compared to the conventional surgery group. No significant differences were observed in postoperative pathological features including maximum tumor diameter, final tumor number, pathological type, differentiation degree, MVI grade, liver capsule invasion, or satellite nodules (all P > 0.05).
Postoperative final tumor number was defined as: Preoperative imaging lesions + intraoperative occult lesions (≥ 5 mm) + small lesions found only on pathology (< 5 mm). In the navigation group, 24 cases (23.3%) had occult lesions detected intraoperatively with 0 additional small lesions on pathology. In the conventional group, 6 cases (6.2%) had intraoperative occult lesion detection while 12 cases had small lesions found only on pathology, totaling 18 cases (18.6%) exceeding preoperative imaging. Although the navigation group had higher intraoperative occult lesion detection (23.3% vs 6.2%, P < 0.001), the proportion with final tumor numbers exceeding preoperative imaging showed no significant difference (P = 0.365), indicating the main advantage lies in immediate intraoperative detection of larger occult lesions (≥ 5 mm; Table 3).
| Indicator | Combined navigation group (n = 103) | Conventional surgery group | Statistic | P value |
| Positive margin | 3 (2.9) | 13 (13.4) | χ2 = 7.621 | 0.006 |
| Margin distance (cm) | 1.2 (0.8-1.8) | 0.8 (0.5-1.3) | Z = 3.912 | < 0.001 |
| Margin distance grouping1 | χ2 = 14.328 | 0.001 | ||
| < 0.5 cm | 8 (7.8) | 22 (22.7) | ||
| 0.5-1.0 cm | 32 (31.1) | 35 (36.1) | ||
| > 1.0 cm | 63 (61.2) | 40 (41.2) | ||
| R0 resection | 100 (97.1) | 84 (86.6) | χ2 = 7.621 | 0.006 |
| Pathological features | ||||
| Maximum tumor diameter (cm) | 7.9 ± 3.3 | 7.7 ± 3.5 | t = 0.421 | 0.674 |
| Postoperative final confirmed total tumor number2 | χ2 = 0.821 | 0.365 | ||
| Consistent with preoperative imaging | 79 (76.7) | 79 (81.4) | ||
| More than preoperative imaging | 24 (23.3) | 18 (18.6) | ||
| Among which: Detected immediately intraoperatively (≥ 5 mm) | 24 (23.3) | 6 (6.2) | ||
| Found on postoperative pathology (< 5 mm) | 0 (0) | 12 (12.4) | ||
| Pathological type | Fisher | 0.782 | ||
| Hepatocellular carcinoma | 98 (95.1) | 93 (95.9) | ||
| Cholangiocarcinoma | 3 (2.9) | 2 (2.1) | ||
| Mixed type | 2 (1.9) | 2 (2.1) | ||
| Differentiation degree | χ2 = 1.234 | 0.745 | ||
| Well-differentiated | 21 (20.4) | 18 (18.6) | ||
| Moderately differentiated | 56 (54.4) | 54 (55.7) | ||
| Poorly differentiated | 23 (22.3) | 22 (22.7) | ||
| Undifferentiated | 3 (2.9) | 3 (3.1) | ||
| MVI grade | χ2 = 0.621 | 0.733 | ||
| M0 | 48 (46.6) | 42 (43.3) | ||
| M1 | 38 (36.9) | 38 (39.2) | ||
| M2 | 17 (16.5) | 17 (17.5) | ||
| Liver capsule invasion | 42 (40.8) | 37 (38.1) | χ2 = 0.149 | 0.7 |
| Satellite nodules | 28 (27.2) | 24 (24.7) | χ2 = 0.156 | 0.693 |
| Intact tumor capsule | 67 (65.0) | 61 (62.9) | χ2 = 0.101 | 0.751 |
The combined navigation group had lower overall complication rate than the conventional surgery group (18.4% vs 28.9%, P = 0.042). The incidence of Clavien-Dindo grade III-V complications was also lower (P = 0.042), though overall grade distribution showed no significant difference (P = 0.078). Multiple complications occurred in some patients: 19 patients with 26 episodes in the navigation group vs 28 patients with 46 episodes in the conventional group.
Bile leakage incidence was significantly lower in the navigation group (P = 0.041), while other specific complications showed no differences (P > 0.05). Postoperative liver function recovered faster in the navigation group, with lower TBIL, ALT, and AST levels on day 3 and day 5 (P < 0.05). The navigation group had shorter hospital stays (P = 0.003). Reoperation rates (1.9% vs 6.2%, P = 0.104) and 30-day/90-day mortality rates showed no significant differences between groups (P > 0.05; Tables 4 and 5).
| Indicator | Combined navigation group (n = 103) | Conventional surgery group | Statistic | P value |
| Overall complications | 19 (18.4) | 28 (28.9) | χ2 = 4.123 | 0.042 |
| Clavien-Dindo grade1 | χ2 = 6.821 | 0.078 | ||
| Grade I-II | 13 (12.6) | 16 (16.5) | ||
| Grade III | 4 (3.9) | 9 (9.3) | ||
| Grade IV | 2 (1.9) | 2 (2.1) | ||
| Grade V | 0 (0) | 1 (1.0) | ||
| Grade III-V complications | 6 (5.8) | 12 (12.4) | χ2 = 4.128 | 0.042 |
| Total complication episodes | 26 | 46 | ||
| Specific complication types (counted by episodes) | ||||
| Bile leakage | 5 (4.9) | 10 (10.3) | χ2 = 4.178 | 0.041 |
| Grade A | 3 (2.9) | 5 (5.2) | ||
| Grade B | 1 (1.0) | 4 (4.1) | ||
| Grade C | 1 (1.0) | 1 (1.0) | ||
| Abdominal infection | 4 (3.9) | 6 (6.2) | χ2 = 0.628 | 0.428 |
| Abdominal hemorrhage | 2 (1.9) | 5 (5.2) | Fisher | 0.133 |
| Liver failure | 1 (1.0) | 3 (3.1) | Fisher | 0.354 |
| Pleural effusion | 6 (5.8) | 8 (8.2) | χ2 = 0.521 | 0.47 |
| Pulmonary infection | 4 (3.9) | 6 (6.2) | χ2 = 0.628 | 0.428 |
| Wound infection | 2 (1.9) | 4 (4.1) | Fisher | 0.428 |
| Renal insufficiency | 1 (1.0) | 2 (2.1) | Fisher | 0.607 |
| Other complications | 1 (1.0) | 2 (2.1) | Fisher | 0.607 |
| Postoperative reoperation | 2 (1.9) | 6 (6.2) | Fisher | 0.104 |
| 30-day postoperative mortality | 0 (0) | 1 (1.0) | Fisher | 0.485 |
| 90-day postoperative mortality | 1 (1.0) | 3 (3.1) | Fisher | 0.354 |
| Indicator | Combined navigation group | Conventional surgery group | Statistic | P value |
| Postoperative day 1 | ||||
| TBIL (μmol/L) | 28.6 ± 12.4 | 31.2 ± 14.8 | t = 1.342 | 0.181 |
| ALT (U/L) | 286 (198-412) | 312 (218-445) | Z = 1.523 | 0.128 |
| AST (U/L) | 268 (182-385) | 295 (201-421) | Z = 1.612 | 0.107 |
| ALB (g/L) | 35.8 ± 4.2 | 35.2 ± 4.6 | t = 0.962 | 0.337 |
| PT-INR | 1.28 ± 0.18 | 1.32 ± 0.21 | t = 1.456 | 0.147 |
| Postoperative day 3 | ||||
| TBIL (μmol/L) | 32.4 ± 14.6 | 38.7 ± 17.2 | t = 2.781 | 0.006 |
| ALT (U/L) | 156 (98-238) | 198 (128-285) | Z = 2.634 | 0.008 |
| AST (U/L) | 142 (88-215) | 178 (112-256) | Z = 2.512 | 0.012 |
| ALB (g/L) | 34.6 ± 4.1 | 33.8 ± 4.5 | t = 1.321 | 0.188 |
| PT-INR | 1.24 ± 0.16 | 1.29 ± 0.19 | t = 2.012 | 0.045 |
| Postoperative day 5 | ||||
| TBIL (μmol/L) | 28.3 ± 12.8 | 34.6 ± 15.4 | t = 3.112 | 0.002 |
| ALT (U/L) | 98 (62-148) | 128 (82-186) | Z = 2.891 | 0.004 |
| AST (U/L) | 86 (54-132) | 112 (72-165) | Z = 2.734 | 0.006 |
| ALB (g/L) | 35.2 ± 4.3 | 34.6 ± 4.6 | t = 0.962 | 0.337 |
| PT-INR | 1.18 ± 0.14 | 1.23 ± 0.17 | t = 2.234 | 0.027 |
| Postoperative day 7 | ||||
| TBIL (μmol/L) | 22.4 ± 9.6 | 26.8 ± 12.3 | t = 2.812 | 0.005 |
| ALT (U/L) | 68 (45-102) | 82 (56-125) | Z = 2.123 | 0.034 |
| AST (U/L) | 58 (38-88) | 72 (48-108) | Z = 2.234 | 0.026 |
| ALB (g/L) | 36.4 ± 4.2 | 35.8 ± 4.5 | t = 0.981 | 0.328 |
| PT-INR | 1.12 ± 0.12 | 1.16 ± 0.14 | t = 2.156 | 0.032 |
| Postoperative recovery indicators | ||||
| Postoperative hospital stays (days) | 12 (9-16) | 15 (11-20) | Z = 3.012 | 0.003 |
| Time to first ambulation (days) | 2 (1-3) | 2 (2-3) | Z = 1.823 | 0.068 |
| Time to first liquid diet (days) | 2 (2-3) | 3 (2-4) | Z = 2.234 | 0.026 |
| Time to drainage tube removal (days) | 5 (4-7) | 6 (5-9) | Z = 2.567 | 0.01 |
To identify independent factors influencing postoperative complications, univariate analysis was first performed, and variables with P < 0.10 were included in multivariate Logistic regression model. Variables included were age, BMI, Child-Pugh grade, degree of cirrhosis, tumor diameter, tumor number, central tumor, intraoperative blood loss, operation time, application of combined navigation technology, etc. Backward method was used for variable screening, with P < 0.05 as the variable retention standard. Multivariate Logistic regression analysis showed that intraoperative blood loss ≥ 600 mL [odds ratio (OR) = 2.856, 95% confidence interval (CI): 1.423-5.732, P = 0.003], operation time ≥ 300 minutes (OR = 2.134, 95%CI: 1.087-4.189, P = 0.028), and moderate to severe cirrhosis (OR = 2.687, 95%CI: 1.298-5.563, P = 0.008) were independent risk factors for postoperative complications, and application of combined navigation technology (OR = 0.498, 95%CI: 0.268-0.925, P = 0.027) was an independent protective factor for postoperative complications (Table 6).
| Factor | β | SE | Wald χ2 | OR | 95%CI | P value |
| Combined navigation technology | -0.697 | 0.318 | 4.812 | 0.498 | 0.268-0.925 | 0.027 |
| Intraoperative blood loss ≥ 600 mL | 1.049 | 0.356 | 8.689 | 2.856 | 1.423-5.732 | 0.003 |
| Operation time ≥ 300 minutes | 0.758 | 0.345 | 4.826 | 2.134 | 1.087-4.189 | 0.028 |
| Moderate to severe cirrhosis | 0.988 | 0.371 | 7.098 | 2.687 | 1.298-5.563 | 0.008 |
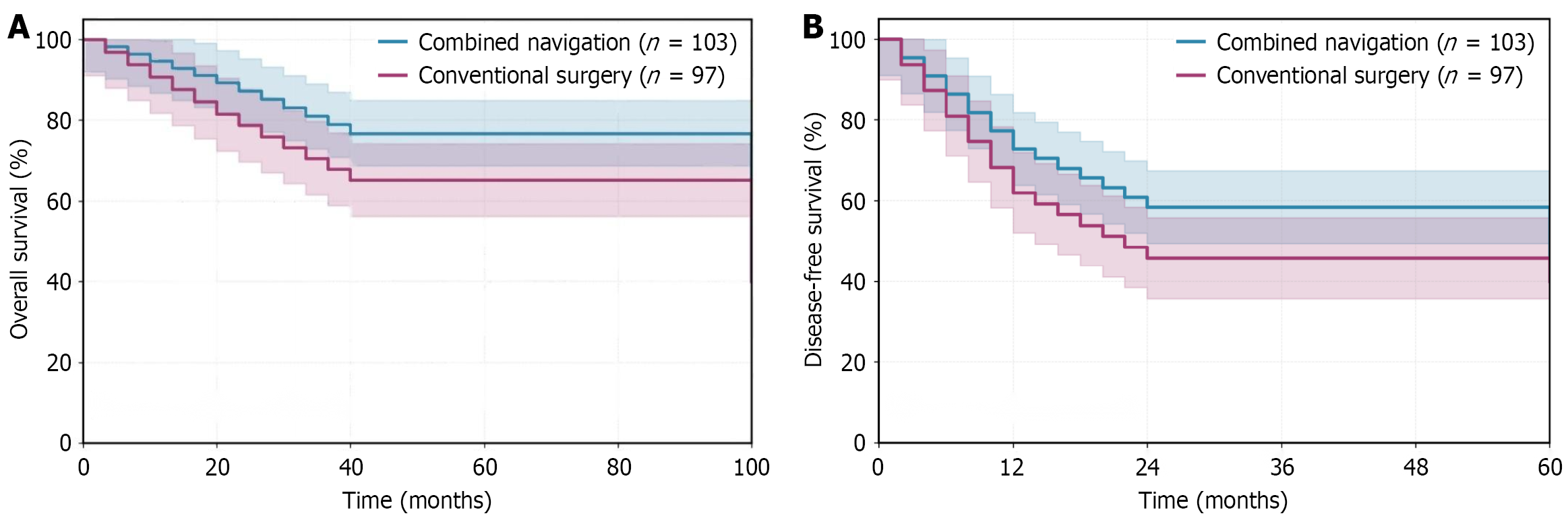
Median follow-up time was 28.6 months (range 6-68 months). Kaplan-Meier survival analysis included all 200 patients, with log-rank test comparing entire survival curves between groups. For OS, the combined navigation group achieved 1-year and 2-year rates of 89.3% and 76.8% respectively, compared to 81.4% and 65.2% in the conventional surgery group, with significantly different survival curves (log-rank χ2 = 4.521, P = 0.033). For DFS, the navigation group achieved 1-year and 2-year rates of 72.8% and 58.4% vs 61.9% and 45.7% in the conventional group, also showing significantly different curves (log-rank χ2 = 5.234, P = 0.022). The navigation group demonstrated lower postoperative recurrence rate (P = 0.048) and marginally lower intrahepatic recurrence rate (P = 0.050), while extrahepatic metastasis rates showed no significant difference (P = 0.431; Table 7).
| Indicator | Combined navigation group (n = 103) | Conventional surgery group | Statistic | P value |
| Median follow-up time (months) | 29.5 (18-42) | 27.8 (16-40) | Z = 0.823 | 0.411 |
| 1-year overall survival rate (%) | 89.3 | 81.4 | ||
| 2-year overall survival rate (%) | 76.8 | 65.2 | ||
| Overall survival curve comparison1 | Log-rank χ2 = 4.521 | 0.033 | ||
| 1-year disease-free survival rate (%) | 72.8 | 61.9 | ||
| 2-year disease-free survival rate (%) | 58.4 | 45.7 | ||
| Disease-free survival curve comparison1 | Log-rank χ2 = 5.234 | 0.022 | ||
| Postoperative recurrence | 38 (36.9) | 49 (50.5) | χ2 = 3.921 | 0.048 |
| Recurrence time (months) | 16.8 (10.2-24.5) | 13.5 (8.6-20.3) | Z = 2.012 | 0.044 |
| Recurrence site classification | ||||
| Intrahepatic recurrence only | 26 (25.2) | 34 (35.1) | χ2 = 2.512 | 0.113 |
| Single | 18 (17.5) | 19 (19.6) | ||
| Multiple | 8 (7.8) | 15 (15.5) | ||
| Extrahepatic metastasis only | 6 (5.8) | 6 (6.2) | χ2 = 0.012 | 0.913 |
| Lung metastasis | 4 (3.9) | 4 (4.1) | ||
| Bone metastasis | 1 (1.0) | 1 (1.0) | ||
| Peritoneal metastasis | 1 (1.0) | 1 (1.0) | ||
| Intrahepatic + extrahepatic metastasis | 6 (5.8) | 9 (9.3) | χ2 = 0.921 | 0.337 |
| Total intrahepatic recurrence2 | 32 (31.1) | 43 (44.3) | χ2 = 3.847 | 0.05 |
| Total extrahepatic metastasis2 | 12 (11.7) | 15 (15.5) | χ2 = 0.621 | 0.431 |
Univariate and multivariate Cox regression analyses identified independent prognostic factors for OS and DFS. Variables with P < 0.10 in univariate analysis were included in multivariate models using backward elimination (P < 0.05 retention).
For OS, independent protective factors were combined navigation technology [hazard ratio (HR) = 0.584, 95%CI: 0.362-0.942, P = 0.028] and R0 resection (HR = 0.412, 95%CI: 0.248-0.684, P < 0.001); independent risk factors were tumor diameter ≥ 8 cm (HR = 1.876, 95%CI: 1.165-3.022, P = 0.010), MVI M2 grade (HR = 2.234, 95%CI: 1.324-3.769, P = 0.003), and AFP > 400 ng/mL (HR = 1.698, 95%CI: 1.056-2.731, P = 0.029; Figure 1A; Table 8).
| Factor | β | SE | Wald χ2 | HR | 95%CI | P value |
| Combined navigation technology | -0.538 | 0.245 | 4.821 | 0.584 | 0.362-0.942 | 0.028 |
| R0 resection | -0.886 | 0.261 | 11.523 | 0.412 | 0.248-0.684 | < 0.001 |
| Tumor diameter ≥ 8 cm | 0.629 | 0.243 | 6.698 | 1.876 | 1.165-3.022 | 0.01 |
| MVI M2 grade | 0.804 | 0.268 | 8.989 | 2.234 | 1.324-3.769 | 0.003 |
| AFP > 400 ng/mL | 0.529 | 0.242 | 4.781 | 1.698 | 1.056-2.731 | 0.029 |
For DFS, independent protective factors were combined navigation technology (HR = 0.631, 95%CI: 0.421-0.945, P = 0.025), R0 resection (HR = 0.468, 95%CI: 0.306-0.715, P < 0.001), and margin distance > 1.0 cm (HR = 0.598, 95%CI: 0.389-0.919, P = 0.019); independent risk factors were tumor number ≥ 3 (HR = 2.156, 95%CI: 1.234-3.767, P = 0.007), MVI M2 grade (HR = 2.487, 95%CI: 1.568-3.945, P < 0.001), and central tumor (HR = 1.623, 95%CI: 1.078-2.443, P = 0.020; Figure 1B; Table 9).
| Factor | β | SE | Wald χ2 | HR | 95%CI | P value |
| Combined navigation technology | -0.461 | 0.207 | 4.961 | 0.631 | 0.421-0.945 | 0.025 |
| R0 resection | -0.759 | 0.218 | 12.123 | 0.468 | 0.306-0.715 | < 0.001 |
| Margin distance > 1.0 cm | -0.514 | 0.22 | 5.461 | 0.598 | 0.389-0.919 | 0.019 |
| Tumor number ≥ 3 | 0.768 | 0.285 | 7.267 | 2.156 | 1.234-3.767 | 0.007 |
| MVI M2 grade | 0.911 | 0.235 | 15.021 | 2.487 | 1.568-3.945 | < 0.001 |
| Central tumor | 0.485 | 0.208 | 5.436 | 1.623 | 1.078-2.443 | 0.02 |
This study systematically evaluated the navigation value of IOUS combined with ICG fluorescence imaging technology in complex HCC resection through retrospective analysis of clinical data from 200 patients with complex HCC. The results showed that combined navigation technology demonstrated significant advantages in tumor localization, margin control, intraoperative blood loss, postoperative complications, and long-term prognosis, providing important technical support for precision surgical treatment of complex HCC.
This study found that the tumor localization time in the combined navigation group was significantly shorter than the conventional surgery group (12.6 minutes vs 18.9 minutes, P < 0.001), which is consistent with previous research results[9]. IOUS can display liver deep structures and vessel courses in real-time, while ICG fluorescence imaging can intuitively display tumor boundaries through fluorescence signal differences, and the combined application of the two technologies achieves complementary advantages, significantly improving the efficiency and accuracy of tumor localization[10]. However, the operation time in the combined navigation group was slightly longer than the conventional surgery group (298.5 minutes vs 276.2 minutes, P = 0.012), which may be related to the need for IOUS scanning and fluorescence imaging examination, but the increased time is within clinically acceptable range, and this time investment resulted in more precise tumor resection[11].
The intraoperative detection of occult lesions is one of the important findings of this study. The rate of immediate intraoperative detection of occult lesions in the combined navigation group was significantly higher than the conventional surgery group (23.3% vs 6.2%, P < 0.001). Studies have shown that approximately 15%-25% of HCC patients have small lesions not shown on preoperative imaging, and these occult lesions are an important cause of early postoperative recurrence[12]. ICG fluorescence imaging technology utilizes the retention characteristics of tumor tissue for ICG and can identify lesions with diameter greater than 5 mm, significantly improving intraoperative detection rate[13]. It is worth noting that in this study, 12 cases of small lesions (diameter < 5 mm) were found on postoperative pathological examination in the conventional surgery group, while no such lesions were found in the combined navigation group, which may be related to the combined navigation group expanding resection range due to finding occult lesions intraoperatively, but it also suggests that current technology still has limitations in detecting extremely small lesions[14].
In terms of oncological outcomes, the positive margin rate in the combined navigation group was significantly lower than the conventional surgery group (2.9% vs 13.4%, P = 0.006), R0 resection rate was higher (97.1% vs 86.6%, P = 0.006), and margin distance was greater (median 1.2 cm vs 0.8 cm, P < 0.001). Adequate margin distance is a key factor affecting postoperative recurrence and long-term survival of HCC, and multiple studies have shown that margin distance > 1.0 cm can significantly reduce recurrence risk[15,16]. In this study, 61.2% of patients in the combined navigation group had margin distance > 1.0 cm, while only 41.2% in the conventional surgery group, and this difference partly stems from the ability of combined navigation technology to display tumor boundaries in real-time, guiding surgeons to expand margin distance as much as possible while ensuring complete tumor resection[17]. In addition, the anatomical resection rate in the combined navigation group was higher than the conventional surgery group (95.1% vs 87.6%, P = 0.040), which is closely related to the characteristic of ICG fluorescence imaging being able to precisely mark the anatomical resection plane by observing fluorescence disappearance interface after blocking target hepatic segment blood flow[18].
Control of intraoperative blood loss is an important challenge in complex HCC surgery. This study showed that intraoperative blood loss in the combined navigation group was significantly less than the conventional surgery group (median 420 mL vs 550 mL, P = 0.004), and transfusion rate was also significantly lower (27.2% vs 42.3%, P = 0.021). This result can be attributed to multiple factors: First, IOUS can clearly display vessel courses, avoiding injury to important vessels; second, ICG fluorescence imaging can monitor resection plane in real-time, reducing bleeding caused by blind dissection; third, precise anatomical resection reduces the risk of unplanned vascular injury[19,20]. Reducing intraoperative blood loss not only decreases transfusion requirements but may also improve patient prognosis by reducing immunosuppression and inflammatory response[21].
In terms of postoperative complications, the overall complication rate in the combined navigation group (18.4% vs 28.9%, P = 0.042) and severe complication rate (Clavien-Dindo grade III-V: 5.8% vs 12.4%, P = 0.042) were significantly lower than the conventional surgery group. Multivariate logistic regression analysis confirmed that application of combined navigation technology is an independent protective factor for postoperative complications (OR = 0.498, P = 0.027). Specifically, the incidence of bile leakage in the combined navigation group was lower (4.9% vs 10.3%, P = 0.041), which may be related to precise anatomical resection reducing bile duct injury[22]. In addition, postoperative liver function indicators in the combined navigation group recovered faster, with TBIL, ALT, and AST levels on postoperative day 3, day 5, and day 7 significantly lower than the conventional surgery group, suggesting that combined navigation technology promoted early recovery of liver function by reducing unnecessary liver tissue damage[23]. Postoperative hospital stay in the combined navigation group was also significantly shortened (median 12 days vs 15 days, P = 0.003), which not only reflects improvement in patient recovery quality but also has important health economics significance[24].
Long-term prognosis analysis showed that the combined navigation group was superior to the conventional surgery group in both OS and DFS. The 2-year OS rate in the combined navigation group was 76.8%, significantly higher than 65.2% in the conventional surgery group (log-rank P = 0.033); the 2-year DFS rate was 58.4%, also higher than 45.7% in the conventional surgery group (log-rank P = 0.022). Cox multivariate regression analysis confirmed that application of combined navigation technology is an independent protective factor for OS (HR = 0.584, P = 0.028) and DFS (HR = 0.631, P = 0.025). This result is consistent with domestic and international multicenter research reports[25,26]. The mechanism by which combined navigation technology improves prognosis may be multifaceted: First, higher R0 resection rate and greater margin distance directly reduce local recurrence risk; second, finding and treating occult lesions intraoperatively reduces the possibility of residual tumor; third, reducing intraoperative blood loss and postoperative complications may indirectly improve prognosis by reducing immunosuppression and inflammatory response[27]. This study also found that the intrahepatic recurrence rate in the combined navigation group showed a marginally significant trend lower than the conventional surgery group (31.1% vs 44.3%, P = 0.050), while there was no significant difference in extrahepatic metastasis rate between the two groups, suggesting that combined navigation technology mainly reduces recurrence risk by improving local control.
This study has certain clinical significance. For patients with complex HCC, especially cases with tumors located in central regions, multiple lesions, or close to important vascular structures, IOUS combined with ICG fluorescence imaging technology provides a safe and effective surgical navigation method that can significantly improve tumor localization accuracy, improve margin quality, reduce intraoperative blood loss and postoperative complication incidence, and ultimately improve patients’ long-term prognosis. The combined application of these technologies embodies the concept of precision hepatobiliary surgery and is worthy of promotion in clinical practice.
This study has several important limitations that warrant critical consideration. First, the retrospective design introduces potential selection bias, as technology adoption was gradual and early cases in the navigation group may have been selected for greater complexity. However, our comprehensive baseline matching and multivariate adjustments help mitigate these concerns. Second, as a single-center study from a specialized hepatobiliary center, our results may not be directly generalizable to community hospitals with different resources and experience levels; external validation through multicenter prospective trials is needed. Third, regarding economic considerations, while navigation equipment requires substantial initial investment and per-care costs, potential reductions in complications, reoperations, and recurrence may justify these expenses, though formal cost-effectiveness analysis is needed, particularly for resource-limited settings[28].
In conclusion, IOUS combined with ICG fluorescence imaging technology has important navigation value in complex HCC resection, can improve tumor localization accuracy, improve margin control, reduce intraoperative blood loss, decrease postoperative complication incidence, and significantly improve patients’ long-term survival. This combined navigation strategy provides effective technical support for precision surgical treatment of complex HCC and has good clinical application prospects. In the future, multicenter prospective randomized controlled trials need to be conducted to further verify the clinical value of combined navigation technology and explore its optimal application strategies in different HCC subtypes.
| 1. | Kim NR, Choi GH, Han DH, Kim KS, Choi JS. Significant Improvement in Long-Term Survival after Liver Resection for Hepatocellular Carcinoma: Evolving Outcomes over 20 Years. Yonsei Med J. 2025;66:637-646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 2. | Takamoto T, Mihara Y, Nishioka Y, Ichida A, Kawaguchi Y, Akamatsu N, Hasegawa K. Surgical treatment for hepatocellular carcinoma in era of multidisciplinary strategies. Int J Clin Oncol. 2025;30:417-426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (4)] |
| 3. | Herrero A, Toubert C, Bedoya JU, Assenat E, Guiu B, Panaro F, Bardol T, Cassese G. Management of hepatocellular carcinoma recurrence after liver surgery and thermal ablations: state of the art and future perspectives. Hepatobiliary Surg Nutr. 2024;13:71-88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 4. | Chang GY, Fetzer DT, Porembka MR. Contrast-Enhanced Intraoperative Ultrasound of the Liver. Surg Oncol Clin N Am. 2022;31:707-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Wakabayashi T, Cacciaguerra AB, Abe Y, Bona ED, Nicolini D, Mocchegiani F, Kabeshima Y, Vivarelli M, Wakabayashi G, Kitagawa Y. Indocyanine Green Fluorescence Navigation in Liver Surgery: A Systematic Review on Dose and Timing of Administration. Ann Surg. 2022;275:1025-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 125] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 6. | Cai X, Hong H, Pan W, Chen J, Jiang L, Du Q, Li G, Lin S, Chen Y. Does Using Indocyanine Green Fluorescence Imaging for Tumors Help in Determining the Safe Surgical Margin in Real-Time Navigation of Laparoscopic Hepatectomy? A Retrospective Study. Ann Surg Oncol. 2023;30:1981-1987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 7. | Ingallinella S, Aldrighetti L, Marino R, Ratti F. Indocyanine green (ICG)-guided robotic resection for liver adenoma: combined technologies for precision surgery. Updates Surg. 2024;76:1105-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 8. | Zhou K, Zhou S, Du L, Liu E, Dong H, Ma F, Sun Y, Li Y. Safety and effectiveness of indocyanine green fluorescence imaging-guided laparoscopic hepatectomy for hepatic tumor: a systematic review and meta-analysis. Front Oncol. 2023;13:1309593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 9. | Avella P, Spiezia S, Rotondo M, Cappuccio M, Scacchi A, Inglese G, Guerra G, Brunese MC, Bianco P, Tedesco GA, Ceccarelli G, Rocca A. Real-Time Navigation in Liver Surgery Through Indocyanine Green Fluorescence: An Updated Analysis of Worldwide Protocols and Applications. Cancers (Basel). 2025;17:872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 10. | Potharazu AV, Gangemi A. Indocyanine green (ICG) fluorescence in robotic hepatobiliary surgery: A systematic review. Int J Med Robot. 2023;19:e2485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 11. | Boogerd LS, Handgraaf HJ, Lam HD, Huurman VA, Farina-Sarasqueta A, Frangioni JV, van de Velde CJ, Braat AE, Vahrmeijer AL. Laparoscopic detection and resection of occult liver tumors of multiple cancer types using real-time near-infrared fluorescence guidance. Surg Endosc. 2017;31:952-961. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 73] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 12. | Liu S, Feng J, Ren Q, Qin H, Yang W, Cheng H, Yao X, Xu J, Han J, Chang S, Yang S, Mou J, Lin Y, He L, Wang H. Evaluating the clinical efficacy and limitations of indocyanine green fluorescence-guided surgery in childhood hepatoblastoma: A retrospective study. Photodiagnosis Photodyn Ther. 2023;44:103790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 13. | Zhou J, Tan Z, Sun B, Leng Y, Liu S. Application of indocyanine green fluorescence imaging in hepatobiliary surgery. Int J Surg. 2024;110:7948-7961. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 14. | Liu Y, Wu D, Zhang K, Ren R, Liu Y, Zhang S, Zhang X, Cheng J, Chen L, Huang J. Detection technology and clinical applications of serum viral products of hepatitis B virus infection. Front Cell Infect Microbiol. 2024;14:1402001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 15. | Liu J, Zhuang G, Bai S, Hu Z, Xia Y, Lu C, Wang J, Wang C, Liu L, Li F, Wu Y, Shen F, Wang K. The Comparison of Surgical Margins and Type of Hepatic Resection for Hepatocellular Carcinoma With Microvascular Invasion. Oncologist. 2023;28:e1043-e1051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 16. | Wang Y, Qu Y, Yang C, Wu Y, Wei H, Qin Y, Yang J, Zheng T, Chen J, Cannella R, Vernuccio F, Ronot M, Chen W, Song B, Jiang H. MRI-based prediction of the need for wide resection margins in patients with single hepatocellular carcinoma. Eur Radiol. 2025;35:1772-1784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Zhu W, Zeng X, Hu H, Xiang N, Zeng N, Wen S, Tian J, Yang J, Fang C. Perioperative and Disease-Free Survival Outcomes after Hepatectomy for Centrally Located Hepatocellular Carcinoma Guided by Augmented Reality and Indocyanine Green Fluorescence Imaging: A Single-Center Experience. J Am Coll Surg. 2023;236:328-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 18. | Huang Z, Zeng X, Zeng N. Laparoscopic anatomic segment Ⅷ resection for hepatocellular carcinoma using the augmented reality-assisted navigation and indocyanine green fluorescence imaging. J Gastrointest Surg. 2024;28:195-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Zeindler J, Hess GF, von Heesen M, Aegerter N, Reber C, Schmitt AM, Muenst S, Bolli M, Soysal SD, Kollmar O. Anatomic versus non-anatomic liver resection for hepatocellular carcinoma-A European multicentre cohort study in cirrhotic and non-cirrhotic patients. Cancer Med. 2024;13:e6981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 20. | De Gasperi A, Petrò L, Amici O, Scaffidi I, Molinari P, Barbaglio C, Cibelli E, Penzo B, Roselli E, Brunetti A, Neganov M, Giacomoni A, Aseni P, Guffanti E. Major liver resections, perioperative issues and posthepatectomy liver failure: A comprehensive update for the anesthesiologist. World J Crit Care Med. 2024;13:92751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (7)] |
| 21. | Hu L, Li Z, Qiao Y, Wang A. Does perioperative allogeneic blood transfusion worsen the prognosis of patients with hepatocellular carcinoma? A meta-analysis of propensity score-matched studies. Front Oncol. 2023;13:1230882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 22. | Ueno M, Hayami S, Miyamoto A, Okada KI, Kitahata Y, Shimizu A, Motobayashi H, Matsumoto K, Kawai M. Relationship between postoperative biliary complications and biliary anatomical aspects in performing right anterior- or central bisectionectomy: Single-center retrospective observational study. Ann Gastroenterol Surg. 2024;8:1076-1083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 23. | Sijberden JP, Kuemmerli C, Ratti F, D'Hondt M, Sutcliffe RP, Troisi RI, Efanov M, Fichtinger RS, Díaz-Nieto R, Ettorre GM, Sheen AJ, Menon KV, Besselink MG, Soonawalla Z, Aroori S, Marino R, De Meyere C, Marudanayagam R, Zimmitti G, Olij B, Eminton Z, Brandts L, Ferrari C, M van Dam R, Aldrighetti LA, Pugh S, Primrose JN, Abu Hilal M. Laparoscopic versus open parenchymal preserving liver resections in the posterosuperior segments (ORANGE Segments): a multicentre, single-blind, randomised controlled trial. Lancet Reg Health Eur. 2025;51:101228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 24. | Koh YX, Zhao Y, Tan IE, Tan HL, Chua DW, Loh WL, Tan EK, Teo JY, Au MKH, Goh BKP. Comparative cost-effectiveness of open, laparoscopic, and robotic liver resection: A systematic review and network meta-analysis. Surgery. 2024;176:11-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 25. | Martinet-Kosinski F, Le Roy B, Lopez YE, Bartoli A, Buc E. Improved tumour localisation during minimally invasive liver surgery using augmented reality: a retrospective study with propensity score analysis. Surg Endosc. 2025;39:5027-5034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 26. | Xiao T, Chen D, Peng L, Li Z, Pan W, Dong Y, Zhang J, Li M. Fluorescence-guided Surgery for Hepatocellular Carcinoma: From Clinical Practice to Laboratories. J Clin Transl Hepatol. 2025;13:216-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Endo Y, Tsilimigras DI, Munir MM, Woldesenbet S, Yang J, Guglielmi A, Ratti F, Marques HP, Cauchy F, Lam V, Poultsides GA, Kitago M, Popescu I, Alexandrescu S, Martel G, Gleisner A, Hugh T, Aldrighetti L, Shen F, Endo I, Pawlik TM. Prognostic significance of postoperative complications for patients with hepatocellular carcinoma relative to alpha-feto protein and tumor burden score. HPB (Oxford). 2024;26:998-1006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 28. | Fichtinger RS, Aldrighetti LA, Abu Hilal M, Troisi RI, Sutcliffe RP, Besselink MG, Aroori S, Menon KV, Edwin B, D'Hondt M, Lucidi V, Ulmer TF, Díaz-Nieto R, Soonawalla Z, White S, Sergeant G, Olij B, Ratti F, Kuemmerli C, Scuderi V, Berrevoet F, Vanlander A, Marudanayagam R, Tanis P, Dewulf MJL, Dejong CHC, Eminton Z, Kimman ML, Brandts L, Neumann UP, Fretland ÅA, Pugh SA, van Breukelen GJP, Primrose JN, van Dam RM; ORANGE II PLUS Collaborative. Laparoscopic Versus Open Hemihepatectomy: The ORANGE II PLUS Multicenter Randomized Controlled Trial. J Clin Oncol. 2024;42:1799-1809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 56] [Article Influence: 28.0] [Reference Citation Analysis (4)] |