Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116655
Revised: January 15, 2026
Accepted: February 3, 2026
Published online: April 27, 2026
Processing time: 140 Days and 2.1 Hours
Laparoscopic anterior resection for colorectal cancer often necessitates a tem
To investigate the effect of mDCQD on stoma reversal time and incidence of po
A retrospective analysis was conducted on 283 patients who underwent laparoscopic anterior resection for colorectal cancer with prophylactic stoma at our hospital from January 2022 to August 2024. According to whether mDCQD was used postoperatively, patients were divided into observation group (132 cases) and control group (151 cases). The control group received conventional treatment postoperatively, while the observation group received oral mDCQD (raw rhubarb 12 g, mirabilite 10 g, immature bitter orange 15 g, magnolia bark 15 g, radish seed 15 g, aucklandia root 10 g, areca seed 10 g, cannabis seed 15 g) starting from postoperative day 2-3 (the day of gastric tube removal) in addition to conventional treatment, twice daily, 150 mL each time, continued until anal gas passage and defecation. Observation indicators included stoma reversal time, incidence of postoperative ileus, time to first anal gas passage, time to first defecation, time to bowel sound recovery, TCM symptom scores, quality of life scores European Organisation for Research and Treatment of Cancer Quality of Life Core Questionnaire (EORTC QLQ-C30), gastrointestinal hormone levels [motilin (MTL), gastrin (GAS), vasoactive intestinal peptide (VIP)], and inflammatory and nutritional indicators [C-reactive protein (CRP), procalcitonin (PCT), albumin (ALB), prealbumin (PA)]. Kaplan-Meier method and Cox proportional hazards regression model were used to analyze factors affecting stoma reversal time.
The stoma reversal time in the observation group was 118.5 ± 23.7 days, shorter than 142.8 ± 28.4 days in the control group (t = 7.623, P < 0.001). The incidence of postoperative ileus in the observation group was 6.1% (8/132), lower than 18.5% (28/151) in the control group (χ2 = 10.245, P = 0.001). The time to first anal gas passage (52.3 ± 10.8 hours vs 68.7 ± 14.2 hours), time to first defecation (78.6 ± 15.3 hours vs 96.4 ± 18.7 hours), and time to bowel sound recovery (38.2 ± 8.5 hours vs 51.6 ± 11.3 hours) in the observation group were all shorter than those in the control group (P < 0.001). On postoperative days 5 and 7, TCM symptom scores in the observation group were lower than those in the control group (P < 0.001). At 3 months after stoma surgery, the EORTC QLQ-C30 overall health status score in the observation group was 68.5 ± 11.7 points, higher than 62.3 ± 13.2 points in the control group (t = 4.19, P < 0.001). On postoperative days 5 and 7, MTL and GAS levels in the observation group were higher than those in the control group, while VIP levels were lower (P < 0.001); CRP and PCT levels in the observation group were lower than those in the control group, while ALB and PA levels were higher (P < 0.05). Multivariate Cox regression analysis showed that the use of mDCQD was an independent factor for shortening stoma reversal time (hazard ratio = 2.147, 95% confidence interval: 1.658-2.781, P < 0.001).
mDCQD can shorten stoma reversal time after laparoscopic anterior resection for colorectal cancer, reduce the incidence of postoperative ileus, promote intestinal function recovery, and improve patients’ quality of life. It is a safe and effective integrated traditional Chinese and Western medicine treatment method.
Core Tip: This study demonstrates that modified Da-Cheng-Qi decoction significantly shortens stoma reversal time and reduces postoperative ileus after laparoscopic anterior resection for colorectal cancer. By promoting gastrointestinal motility, regulating gastrointestinal hormones, and attenuating postoperative inflammation, the modified formula accelerates bowel recovery and improves patients’ nutritional status and quality of life. The study highlights the value of integrating traditional Chinese medicine with enhanced recovery pathways and identifies the decoction as an independent factor associated with earlier stoma closure.
- Citation: Cheng XY, Chen T. Effect of modified Da-Cheng-Qi decoction on stoma reversal and postoperative ileus after laparoscopic anterior resection. World J Gastrointest Surg 2026; 18(4): 116655
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116655.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116655
Colorectal cancer is one of the most common malignant tumors of the digestive system worldwide, with both incidence and mortality rates ranking among the top malignant tumors[1]. With continuous advances in surgical techniques, laparoscopic anterior resection (Dixon surgery) for colorectal cancer has become the standard surgical procedure for treating middle and low rectal cancer, with advantages of minimal trauma and rapid recovery[2]. However, due to factors such as low tumor location, high anastomotic tension, and complex pelvic anatomy, the incidence of postoperative anastomotic leakage can reach 3%-15%, seriously affecting patient prognosis[3]. Therefore, prophylactic stoma surgery is routinely performed clinically to reduce the risk of anastomotic leakage, but the presence of a stoma not only affects patients' quality of life but also increases medical burden. How to shorten stoma reversal time has become a key clinical concern.
Postoperative ileus is one of the common complications after colorectal cancer surgery, manifested by weakened or absent intestinal peristalsis, abdominal distension, nausea and vomiting. It not only delays postoperative recovery but may also lead to serious consequences such as intestinal obstruction and poor anastomotic healing, thereby prolonging stoma reversal time[4]. Currently, western medicine mainly adopts measures such as early mobilization and prokinetic drugs to promote intestinal function recovery, but with limited effectiveness. In recent years, the application of traditional Chinese medicine (TCM) in enhanced recovery after surgery has received increasing attention, and multiple studies have shown that Chinese herbal medicine can effectively improve postoperative gastrointestinal function and shorten hospital stay[5].
Da-Cheng-Qi decoction originated from Zhang Zhongjing’s “Treatise on Febrile Diseases” in the Han Dynasty, composed of rhubarb, mirabilite, immature bitter orange, and magnolia bark. It has the efficacy of drastically purging heat accumulation and cleansing the stomach and intestines, and is a classic prescription for treating Yangming bowel excess syndrome[6]. Modern pharmacological studies have shown that Da-Cheng-Qi decoction can promote gastroin
A retrospective analysis was conducted on clinical data of patients who underwent laparoscopic anterior resection for colorectal cancer with prophylactic stoma at our hospital from January 2022 to August 2024. A total of 283 patients meeting the criteria were finally included. According to whether mDCQD was used postoperatively, patients were divided into observation group (132 cases, receiving oral mDCQD in addition to conventional treatment postoperatively) and control group (151 cases, receiving conventional treatment only postoperatively). This study was approved by the Ethics Committee of our hospital and complied with the principles of the Declaration of Helsinki.
Inclusion criteria: (1) Age 18-75 years; (2) Pathologically confirmed colorectal cancer; (3) Underwent laparoscopic anterior resection (Dixon surgery) with prophylactic stoma, with tumor location in upper rectum, middle rectum, lower rectum, or lower sigmoid colon (≤ 15 cm from anal verge); (4) Preoperative American Society of Anesthesiologists (ASA) grade I-III; (5) Postoperative pathological stage I-III; and (6) Complete clinical data.
Exclusion criteria: (1) Emergency surgery or conversion to open surgery; (2) Combined with other organ malignancies; (3) Received neoadjuvant radiotherapy or chemotherapy preoperatively; (4) Distant metastasis found intraoperatively; (5) Combined with severe heart, liver, or kidney dysfunction; (6) Postoperative occurrence of serious complications such as anastomotic leakage or abdominal infection; (7) TCM syndrome differentiation performed preoperatively by two TCM physicians with associate senior professional titles or above at our hospital, with syndrome differentiation not belonging to heat-bound bowel excess syndrome (diagnostic criteria referenced from the 10th edition of “Traditional Chinese Internal Medicine” and “Criteria of Diagnosis and Therapeutic Effect of Diseases and Syndromes in TCM”); (8) Allergy or intolerance to mDCQD component drugs; and (9) Poor compliance, failure to take medication as prescribed, or incom
All patients underwent laparoscopic anterior resection for colorectal cancer performed by the same surgical team. The surgery followed the principles of total mesorectal excision. According to tumor location, colectomy or proctectomy was performed, intestinal reconstruction was completed using double stapling technique or manual anastomosis, and prophylactic ileostomy or colostomy was routinely performed.
Conventional postoperative treatment (implemented in both groups): (1) Fasting until intestinal function recovery (anal gas passage and defecation); (2) Continuous gastrointestinal decompression until gastric fluid < 200 mL/day, generally removing gastric tube 2-3 days postoperatively; (3) Fluid replacement, maintaining water-electrolyte and acid-base balance; (4) Prophylactic antibiotic use for 3-5 days; (5) Starting bedside activity on postoperative day 1, gradually in
On the basis of the above conventional treatment, the observation group received oral or nasointestinal tube (if retained) administration of mDCQD starting from the day of gastric tube removal (generally postoperative day 2-3), twice daily, 150 mL each time, continued until anal gas passage and defecation (generally used for 3-7 days). mDCQD formula: Raw rhubarb 12 g (decocted later), mirabilite 10 g (dissolved in decoction), immature bitter orange 15 g, magnolia bark 15 g, radish seed 15 g, aucklandia root 10 g, areca seed 10 g, cannabis seed 15 g. The above dosages were standard adult doses (applicable to patients weighing 50-70 kg), reduced by 20% for patients weighing < 50 kg, and increased by 20% for patients weighing > 70 kg. The above formula was uniformly decocted by the hospital’s TCM pharmacy, with each dose decocted to 300 mL, divided into 2 administrations. Drug sources complied with the 2020 edition of the “Pharmacopoeia of the People’s Republic of China”.
Stoma reversal criteria: (1) 3-6 months after stoma surgery, with specific reversal time comprehensively evaluated and determined by multidisciplinary team according to patient-specific conditions; (2) Good healing of intestinal anastomosis (confirmed by colonoscopy or contrast examination); (3) No anastomotic stenosis; (4) No pelvic abscess or infection; (5) No need for repeated chemotherapy or completed adjuvant chemotherapy; and (6) Patient in good general condition, with cardiopulmonary function tolerating surgery. All patients underwent initial assessment 3 months after stoma surgery. Those meeting criteria were scheduled for reversal surgery as soon as possible, while those not meeting criteria were reviewed and assessed monthly until meeting criteria or reaching 6 months postoperatively.
Baseline data of both groups were collected and compared, including: (1) Age, gender, body mass index (BMI); (2) Tumor location (upper rectum, middle rectum, lower rectum, lower sigmoid colon); (3) Tumor tumor-nodes-metastasis (TNM) staging (according to American Joint Committee on Cancer AJCC 8th edition standards); (4) ASA grade; (5) Comorbidities (hypertension, diabetes, coronary heart disease, etc.); and (6) Stoma type (ileostomy, colostomy).
Stoma reversal time: Defined as the time interval between prophylactic stoma surgery date and stoma reversal surgery date, calculated in days.
Incidence of postoperative ileus: Ileus was defined as the occurrence of any of the following conditions postoperatively: (1) No anal gas passage within 5 days postoperatively; (2) After gas passage, recurrence of persistent abdominal disten
Postoperative recovery indicators: (1) Gastric tube retention time: Time from postoperative gastric tube placement to removal (days); (2) Time to first oral liquid intake: Time from end of surgery to first oral liquid diet intake (days); and (3) Incidence of postoperative complications: Recording various complications occurring within 30 days postoperatively, including wound infection, pulmonary infection, urinary tract infection, anastomotic bleeding, intestinal obstruction, etc.
Using the TCM spleen-stomach disease symptom quantification scale, symptoms of abdominal distension, abdominal pain, nausea and vomiting, and poor appetite were assessed on postoperative days 3, 5, and 7. The scale included 4 dimensions: (1) Abdominal distension: 0 points = no distension, 1 point = mild distension not affecting activity, 2 points = moderate distension affecting activity, 3 points = severe distension with dyspnea; (2) Abdominal pain: 0 points = no pain, 1 point = mild tolerable pain, 2 points = moderate pain requiring analgesics, 3 points = severe pain with poor analgesic effect; (3) Nausea and vomiting: 0 points = no nausea or vomiting, 1 point = mild nausea without vomiting, 2 points = nausea with small amount of vomiting (1-2 times/day), 3 points = frequent vomiting (≥ 3 times/day); and (4) Poor appetite: 0 points = normal appetite, 1 point = slightly reduced appetite, 2 points = significantly reduced appetite, 3 points = complete inability to eat. Total score was the sum of scores from each dimension, ranging 0-12 points, with higher scores indicating more severe gastrointestinal dysfunction symptoms.
The European Organisation for Research and Treatment of Cancer Quality of Life Core Questionnaire (EORTC QLQ-C30, Chinese version) was used to assess patients’ quality of life at 3 months after stoma surgery. This scale contains 30 items, divided into 5 functional domains, 3 symptom domains, and 1 global health status domain: (1) Functional domains: Including physical functioning (5 items), role functioning (2 items), emotional functioning (4 items), cognitive functioning (2 items), social functioning (2 items); (2) Symptom domains: Including fatigue (3 items), nausea and vomiting (2 items), pain (2 items), and other single-item symptoms (dyspnea, insomnia, appetite loss, constipation, diarrhea, financial difficulties, 1 item each); and (3) Global health status/quality of life (2 items). Scoring principles: Items 1-28 used 4-point Likert scale (1 = not at all, 2 = a little, 3 = quite a bit, 4 = very much), items 29-30 used 7-point Likert scale (1 = very poor, 7 = excellent). According to the EORTC QLQ-C30 scoring manual, raw scores from each domain were linearly transformed to 0-100 points, with higher scores in functional domains and global health status indicating better functional status and higher quality of life, while higher scores in symptom domains indicating more severe symptoms and poorer quality of life.
(1) Incidence of stoma complications: Recording parastomal hernia, stoma prolapse, stoma retraction, peristomal dermatitis, and other complications occurring during the stoma period; (2) Stoma reversal operation time: Operation time for stoma reversal surgery (minutes); and (3) Postoperative complications after stoma reversal: Recording wound infection, intestinal obstruction, anastomotic leakage, and other complications occurring within 30 days after stoma reversal.
Fasting venous blood was collected 1 day preoperatively and on postoperative days 3, 5, and 7 to measure: (1) Inflammatory indicators: White blood cell count, C-reactive protein (CRP), procalcitonin (PCT); (2) Nutritional indicators: Serum albumin (ALB), prealbumin (PA), hemoglobin; and (3) Gastrointestinal hormones: Motilin (MTL), gastrin (GAS), vasoactive intestinal peptide (VIP), measured by enzyme-linked immunosorbent assay, with kits purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd., strictly following kit instructions. Normal reference ranges: MTL 200-400 pg/mL, GAS 40-150 pg/mL, VIP 20-80 pg/mL.
SPSS 26.0 statistical software was used for data analysis. Continuous variables were first tested for normality (Shapiro-Wilk test) and homogeneity of variance (Levene test). Normally distributed continuous variables were expressed as mean ± SD, with independent samples t-test for between-group comparisons. Non-normally distributed continuous variables were expressed as median (interquartile range) [M (Q1, Q3)], with Mann-Whitney U test for between-group comparisons. Categorical variables were expressed as n (%), with χ2 test or Fisher’s exact test (when theoretical frequency < 5) for between-group comparisons.
For repeated measurement data (such as TCM symptom scores, laboratory indicators), independent samples t-test or Mann-Whitney U test was used for between-group comparisons at each time point, with Bonferroni correction for multiple comparisons.
Kaplan-Meier method was used to plot cumulative curves of stoma reversal time, with log-rank test for between-group comparisons. Cox proportional hazards regression model was used for multivariate analysis, including variables with P < 0.10 in univariate analysis, to evaluate independent risk factors affecting stoma reversal time. Results were expressed as hazard ratio (HR) and 95% confidence interval (95%CI).
The significance level was α = 0.05, with P < 0.05 considered statistically significant. All statistical tests were two-sided. Missing data were handled as follows: The overall missing data rate in this study was < 5%. For continuous variables, multiple imputation method was employed using predictive mean matching with five imputations, incorporating relevant covariates (age, gender, tumor stage, and surgical approach) in the imputation model. For categorical variables, complete case analysis was performed. Sensitivity analyses were conducted comparing results from complete case analysis with those from multiple imputation to assess the robustness of findings, which demonstrated consistent results across both approaches (data not shown).
This study included a total of 283 patients, with 132 cases in the observation group and 151 cases in the control group. There were no statistically significant differences between the two groups in baseline data including age, gender, BMI, tumor location, TNM stage, ASA grade, comorbidities, and stoma type (P > 0.05), indicating comparability (Table 1).
| Item | Observation group (n = 132) | Control group (n = 151) | Statistic | P value |
| Age (years) | 58.3 ± 10.6 | 59.1 ± 11.2 | t = 0.596 | 0.552 |
| Gender | χ2 = 0.428 | 0.513 | ||
| Male | 78 (59.1) | 94 (62.3) | ||
| Female | 54 (40.9) | 57 (37.7) | ||
| BMI (kg/m2) | 23.4 ± 2.8 | 23.7 ± 3.1 | t = 0.827 | 0.409 |
| Tumor location | χ2 = 2.156 | 0.54 | ||
| Upper rectum | 35 (26.5) | 38 (25.2) | ||
| Middle rectum | 42 (31.8) | 55 (36.4) | ||
| Lower rectum | 38 (28.8) | 38 (25.2) | ||
| Lower sigmoid colon | 17 (12.9) | 20 (13.2) | ||
| TNM stage | χ2 = 0.894 | 0.64 | ||
| Stage I | 28 (21.2) | 35 (23.2) | ||
| Stage II | 52 (39.4) | 63 (41.7) | ||
| Stage III | 52 (39.4) | 53 (35.1) | ||
| ASA grade | χ2 = 1.267 | 0.531 | ||
| Grade I | 38 (28.8) | 48 (31.8) | ||
| Grade II | 72 (54.5) | 79 (52.3) | ||
| Grade III | 22 (16.7) | 24 (15.9) | ||
| Comorbidities | ||||
| Hypertension | 42 (31.8) | 51 (33.8) | χ2 = 0.122 | 0.727 |
| Diabetes | 23 (17.4) | 28 (18.5) | χ2 = 0.057 | 0.811 |
| Coronary heart disease | 15 (11.4) | 19 (12.6) | χ2 = 0.106 | 0.745 |
| Stoma type | χ2 = 0.015 | 0.904 | ||
| Ileostomy | 95 (72.0) | 108 (71.5) | ||
| Colostomy | 37 (28.0) | 43 (28.5) |
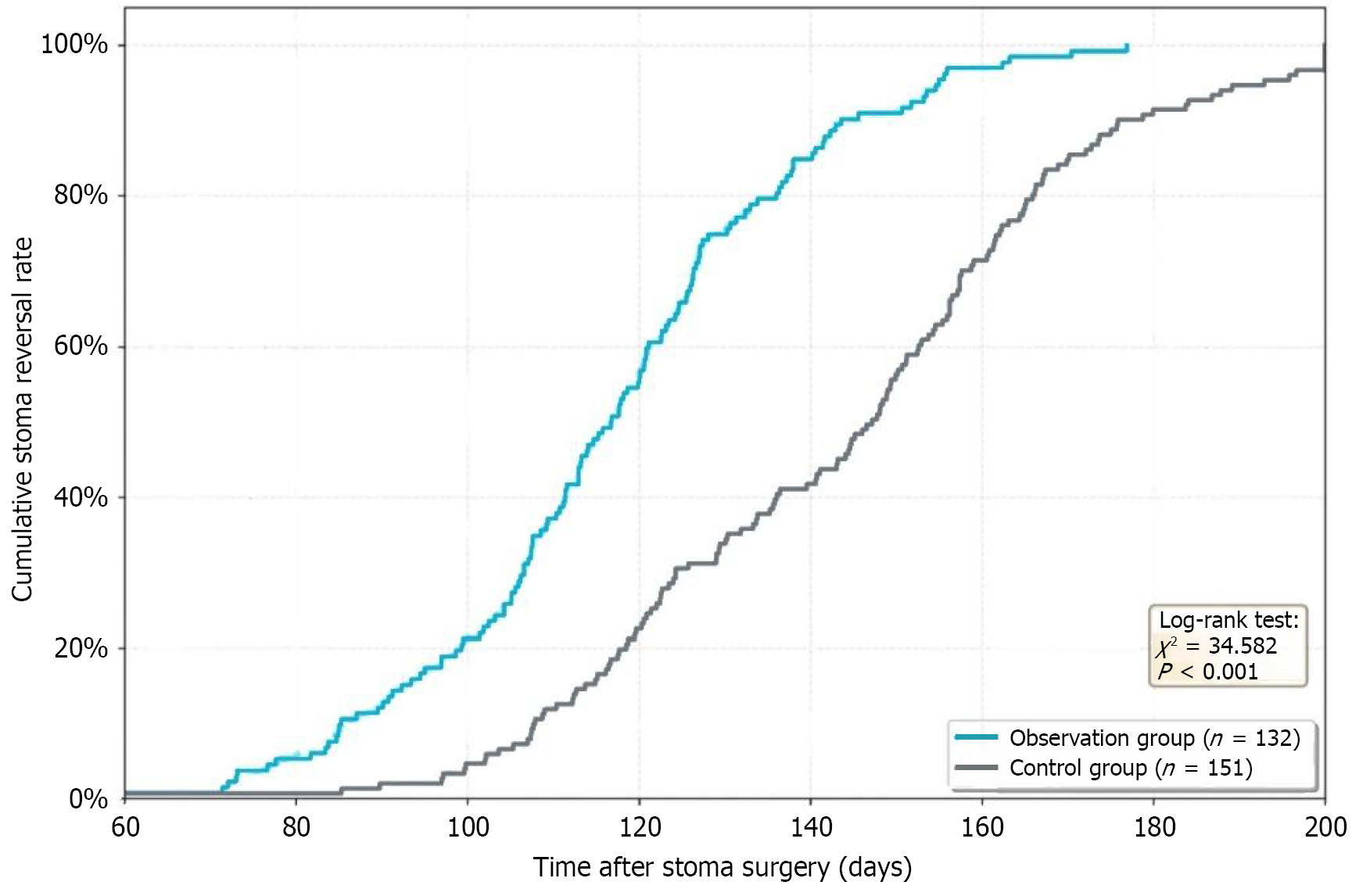
The stoma reversal time in the observation group was 118.5 ± 23.7 days, and in the control group was 142.8 ± 28.4 days. The stoma reversal time in the observation group was shorter than that in the control group (t = 7.623, P < 0.001). Kaplan-Meier survival analysis showed that the cumulative stoma reversal rate in the observation group was higher than that in the control group (Log-rank χ2 = 34.582, P < 0.001) (Figure 1).
Univariate Cox proportional hazards regression analysis showed that mDCQD treatment (P < 0.001), TNM stage (P = 0.042), ASA grade (P = 0.068), and age (P = 0.082) were associated with stoma reversal time. Variables with P < 0.10 were included in multivariate analysis (Table 2). Multivariate Cox proportional hazards regression analysis showed that mDCQD treatment (HR = 2.147, 95%CI: 1.658-2.781, P < 0.001) and TNM stage I (HR = 1.523, 95%CI: 1.082-2.145, P = 0.016) were independent factors for shortened stoma reversal time (Table 3).
| Variable | HR | 95%CI | P value |
| Age (≥ 60 years vs < 60 years) | 0.823 | 0.662-1.024 | 0.082 |
| Gender (female vs male) | 1.085 | 0.872-1.350 | 0.467 |
| BMI (≥ 24 kg/m2 vs < 24 kg/m2) | 0.946 | 0.761-1.176 | 0.618 |
| TNM stage | - | - | 0.042 |
| Stage I vs stage III | 1.428 | 1.045-1.951 | 0.025 |
| Stage II vs stage III | 1.186 | 0.914-1.539 | 0.198 |
| ASA grade (III vs I-II) | 0.758 | 0.562-1.023 | 0.068 |
| Tumor location | - | - | 0.526 |
| Hypertension (yes vs no) | 0.892 | 0.703-1.131 | 0.345 |
| Diabetes (yes vs no) | 0.856 | 0.634-1.156 | 0.312 |
| Stoma type (colostomy vs ileostomy) | 1.124 | 0.876-1.442 | 0.358 |
| Modified Da-Cheng-Qi Decoction (yes vs no) | 2.235 | 1.738-2.875 | < 0.001 |
| Variable | HR | 95%CI | P value |
| Modified Da-Cheng-Qi Decoction (yes vs no) | 2.147 | 1.658-2.781 | < 0.001 |
| TNM stage I (vs stage III) | 1.523 | 1.082-2.145 | 0.016 |
| TNM stage II (vs stage III) | 1.248 | 0.956-1.629 | 0.104 |
| Age (≥ 60 years vs < 60 years) | 0.865 | 0.693-1.079 | 0.196 |
| ASA grade (III vs I-II) | 0.812 | 0.599-1.100 | 0.178 |
The incidence of postoperative ileus in the observation group was 6.1%, lower than 18.5% in the control group (χ2 = 10.245, P = 0.001). The time to first anal gas passage, time to first defecation, and time to bowel sound recovery in the observation group were all shorter than those in the control group (P < 0.001) (Table 4).
| Indicator | Observation group (n = 132) | Control group (n = 151) | Statistic | P value |
| Postoperative ileus | 8 (6.1) | 28 (18.5) | χ2 = 10.245 | 0.001 |
| Time to first anal gas passage (hours) | 52.3 ± 10.8 | 68.7 ± 14.2 | t = 11.01 | < 0.001 |
| Time to first defecation (hours) | 78.6 ± 15.3 | 96.4 ± 18.7 | t = 8.454 | < 0.001 |
| Time to bowel sound recovery (hours) | 38.2 ± 8.5 | 51.6 ± 11.3 | t = 10.963 | < 0.001 |
| Gastric tube retention time (days) | 2.5 ± 0.6 | 3.0 ± 0.8 | t = 5.741 | < 0.001 |
| Time to first oral liquid intake (days) | 3.4 ± 0.7 | 4.3 ± 0.9 | t = 9.120 | < 0.001 |
| Postoperative complications | 12 (9.1) | 18 (11.9) | χ2 = 0.668 | 0.414 |
Gastric tube retention time and time to first oral liquid intake in the observation group were both shorter than those in the control group (P < 0.001). There was no statistically significant difference in the incidence of postoperative complications between the two groups (P = 0.414). Types of postoperative complications are shown in Table 5.
| Complication type | Observation group (n = 132) | Control group (n = 151) | χ2 | P value |
| Wound infection | 4 (3.0) | 6 (4.0) | 0.214 | 0.644 |
| Pulmonary infection | 3 (2.3) | 5 (3.3) | 0.285 | 0.594 |
| Urinary tract infection | 2 (1.5) | 3 (2.0) | - | 1.0001 |
| Anastomotic bleeding | 2 (1.5) | 2 (1.3) | - | 1.0001 |
| Intestinal obstruction | 1 (0.8) | 2 (1.3) | - | 1.0001 |
| Total | 12 (9.1) | 18 (11.9) | 0.668 | 0.414 |
On postoperative day 3, the difference in total TCM symptom scores between the two groups was statistically significant (P = 0.043), but after Bonferroni correction for multiple comparisons (α’ = 0.05/3 ≈ 0.017), the difference was not statistically significant (corrected P > 0.017). On postoperative days 5 and 7, scores for abdominal distension, abdominal pain, nausea and vomiting, poor appetite, and total scores in the observation group were all lower than those in the control group (P < 0.001) (Table 6). Using Bonferroni correction for multiple comparisons at 3 postoperative time points, differences in all TCM symptom scores and total scores between groups on postoperative days 5 and 7 remained statistically significant (corrected P < 0.017).
| Time point | Group | Abdominal distension | Abdominal pain | Nausea/vomiting | Poor appetite | Total score |
| Postoperative day 3 | Observation | 2.1 ± 0.6 | 1.8 ± 0.5 | 1.6 ± 0.5 | 1.9 ± 0.5 | 7.4 ± 1.5 |
| Control | 2.2 ± 0.6 | 1.9 ± 0.6 | 1.7 ± 0.6 | 2.0 ± 0.6 | 7.8 ± 1.7 | |
| t value | 1.386 | 1.491 | 1.496 | 1.5 | 2.033 | |
| P value | 0.167 | 0.137 | 0.136 | 0.135 | 0.043 | |
| Corrected P value | > 0.017 | > 0.017 | > 0.017 | > 0.017 | > 0.017 | |
| Postoperative day 5 | Observation | 1.2 ± 0.4 | 0.9 ± 0.4 | 0.7 ± 0.3 | 1.1 ± 0.4 | 3.9 ± 1.0 |
| Control | 1.8 ± 0.5 | 1.4 ± 0.5 | 1.3 ± 0.5 | 1.6 ± 0.5 | 6.1 ± 1.4 | |
| t value | 10.713 | 9.109 | 11.572 | 9.086 | 14.679 | |
| P value | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | |
| Corrected P value | < 0.017 | < 0.017 | < 0.017 | < 0.017 | < 0.017 | |
| Postoperative day 7 | Observation | 0.6 ± 0.3 | 0.4 ± 0.3 | 0.3 ± 0.2 | 0.5 ± 0.3 | 1.8 ± 0.7 |
| Control | 1.3 ± 0.5 | 0.9 ± 0.4 | 0.8 ± 0.4 | 1.0 ± 0.4 | 4.0 ± 1.1 | |
| t value | 13.929 | 11.729 | 12.818 | 11.729 | 19.39 | |
| P value | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | |
| Corrected P value | < 0.017 | < 0.017 | < 0.017 | < 0.017 | < 0.017 |
At 3 months after stoma surgery, the EORTC QLQ-C30 global health status score in the observation group was 68.5 ± 11.7 points, higher than 62.3 ± 13.2 points in the control group (t = 4.19, P < 0.001) (Table 7).
| Group | n | Global health status | t | P value |
| Observation group | 132 | 68.5 ± 11.7 | 4.19 | < 0.001 |
| Control group | 151 | 62.3 ± 13.2 |
There was no statistically significant difference in the incidence of stoma complications between the two groups (P = 0.685). The stoma reversal operation time in the observation group was shorter than that in the control group (P = 0.002). There was no statistically significant difference in the incidence of postoperative complications after stoma reversal between the two groups (P = 0.440) (Table 8).
| Indicator | Observation group (n = 132) | Control group (n = 151) | Statistic | P value |
| Stoma complications | 15 (11.4) | 20 (13.2) | χ2 = 0.165 | 0.685 |
| Parastomal hernia | 5 (3.8) | 7 (4.6) | - | - |
| Stoma prolapse | 4 (3.0) | 6 (4.0) | - | - |
| Stoma retraction | 3 (2.3) | 4 (2.6) | - | - |
| Peristomal dermatitis | 3 (2.3) | 3 (2.0) | - | - |
| Stoma reversal operation time (minute) | 125.3 ± 28.6 | 136.8 ± 32.4 | t = 3.134 | 0.002 |
| Postoperative complications after stoma reversal | 6 (4.5) | 10 (6.6) | χ2 = 0.596 | 0.44 |
| Wound infection | 3 (2.3) | 5 (3.3) | - | - |
| Intestinal obstruction | 2 (1.5) | 3 (2.0) | - | - |
| Anastomotic leakage | 1 (0.8) | 2 (1.3) | - | - |
On preoperative day 1, there were no statistically significant differences in all laboratory indicators between the two groups (P > 0.05). On postoperative day 3, there were no statistically significant differences in inflammatory and nutri
| Indicator | Time point | Observation group (n = 132) | Control group (n = 151) | t value | P value | Corrected P value |
| WBC (× 109/L) | Preoperative day 1 | 6.8 ± 1.5 | 6.9 ± 1.6 | 0.529 | 0.597 | - |
| Postoperative day 3 | 10.2 ± 2.3 | 10.6 ± 2.5 | 1.385 | 0.167 | > 0.017 | |
| Postoperative day 5 | 8.5 ± 1.8 | 8.9 ± 2.0 | 1.737 | 0.084 | > 0.017 | |
| Postoperative day 7 | 7.3 ± 1.6 | 7.6 ± 1.7 | 1.519 | 0.13 | > 0.017 | |
| CRP (mg/L) | Preoperative day 1 | 5.2 ± 2.1 | 5.4 ± 2.3 | 0.759 | 0.449 | - |
| Postoperative day 3 | 78.6 ± 18.4 | 82.3 ± 19.7 | 1.609 | 0.109 | > 0.017 | |
| Postoperative day 5 | 42.3 ± 12.5 | 48.7 ± 14.3 | 3.916 | < 0.001 | < 0.017 | |
| Postoperative day 7 | 18.5 ± 6.8 | 22.4 ± 8.2 | 4.282 | < 0.001 | < 0.017 | |
| PCT (ng/mL) | Preoperative day 1 | 0.08 ± 0.03 | 0.08 ± 0.03 | 0 | 1 | - |
| Postoperative day 3 | 0.35 ± 0.12 | 0.37 ± 0.13 | 1.326 | 0.186 | > 0.017 | |
| Postoperative day 5 | 0.18 ± 0.08 | 0.21 ± 0.09 | 2.914 | 0.004 | < 0.017 | |
| Postoperative day 7 | 0.11 ± 0.05 | 0.13 ± 0.06 | 3.007 | 0.003 | < 0.017 | |
| ALB (g/L) | Preoperative day 1 | 41.2 ± 3.8 | 40.8 ± 4.1 | 0.847 | 0.398 | - |
| Postoperative day 3 | 35.6 ± 3.2 | 35.2 ± 3.5 | 0.992 | 0.322 | > 0.017 | |
| Postoperative day 5 | 37.8 ± 3.1 | 36.5 ± 3.4 | 3.343 | 0.001 | < 0.017 | |
| Postoperative day 7 | 39.5 ± 3.0 | 38.2 ± 3.3 | 3.447 | 0.001 | < 0.017 | |
| PA (mg/L) | Preoperative day 1 | 268.5 ± 42.3 | 265.2 ± 45.7 | 0.63 | 0.529 | - |
| Postoperative day 3 | 185.3 ± 35.6 | 182.7 ± 38.2 | 0.595 | 0.552 | > 0.017 | |
| Postoperative day 5 | 218.6 ± 32.4 | 205.3 ± 36.8 | 3.179 | 0.002 | < 0.017 | |
| Postoperative day 7 | 245.8 ± 31.5 | 228.4 ± 38.6 | 4.089 | < 0.001 | < 0.017 | |
| Hb (g/L) | Preoperative day 1 | 128.5 ± 15.6 | 127.3 ± 16.8 | 0.617 | 0.538 | - |
| Postoperative day 3 | 112.3 ± 12.4 | 111.5 ± 13.2 | 0.52 | 0.603 | > 0.017 | |
| Postoperative day 5 | 115.7 ± 11.8 | 113.8 ± 12.5 | 1.309 | 0.192 | > 0.017 | |
| Postoperative day 7 | 120.4 ± 11.3 | 118.6 ± 12.1 | 1.286 | 0.2 | > 0.017 |
On postoperative day 3, there were no statistically significant differences in gastrointestinal hormone levels between the two groups (P > 0.05). On postoperative days 5 and 7, MTL and GAS levels in the observation group were higher than those in the control group, while VIP levels were lower (P < 0.001) (Table 10).
| Indicator | Time point | Observation group (n = 132) | Control group (n = 151) | t value | P value | Corrected P value |
| MTL | Preoperative day 1 | 285.6 ± 42.3 | 282.4 ± 45.8 | 0.608 | 0.544 | - |
| Postoperative day 3 | 198.5 ± 35.7 | 195.2 ± 38.4 | 0.746 | 0.456 | > 0.017 | |
| Postoperative day 5 | 245.8 ± 38.6 | 218.3 ± 42.5 | 5.641 | < 0.001 | < 0.017 | |
| Postoperative day 7 | 265.4 ± 39.8 | 238.7 ± 44.2 | 5.328 | < 0.001 | < 0.017 | |
| GAS | Preoperative day 1 | 92.5 ± 18.6 | 90.8 ± 19.7 | 0.743 | 0.458 | - |
| Postoperative day 3 | 68.3 ± 15.2 | 64.5 ± 16.8 | 1.985 | 0.048 | > 0.017 | |
| Postoperative day 5 | 82.6 ± 16.4 | 72.8 ± 17.9 | 4.799 | < 0.001 | < 0.017 | |
| Postoperative day 7 | 87.5 ± 16.9 | 76.2 ± 18.3 | 5.421 | < 0.001 | < 0.017 | |
| VIP | Preoperative day 1 | 48.3 ± 10.5 | 49.1 ± 11.2 | 0.622 | 0.534 | - |
| Postoperative day 3 | 72.5 ± 14.8 | 74.2 ± 15.6 | 0.94 | 0.348 | > 0.017 | |
| Postoperative day 5 | 58.6 ± 12.3 | 66.8 ± 14.5 | 5.022 | < 0.001 | < 0.017 | |
| Postoperative day 7 | 52.8 ± 11.4 | 60.5 ± 13.8 | 5.035 | < 0.001 | < 0.017 |
Using Bonferroni correction for multiple comparisons at 3 postoperative time points (α’ = 0.05/3 ≈ 0.017), differences in CRP, PCT, ALB, and PA levels between the observation group and control group on postoperative days 5 and 7 remained statistically significant (corrected P < 0.017); differences in MTL, GAS, and VIP levels between the observation group and control group on postoperative days 5 and 7 remained statistically significant (corrected P < 0.017).
Stoma reversal time is influenced by multiple factors, including anastomotic healing, patient general condition, postoperative complications, and adjuvant therapy progress[12]. The primary finding of this study-a significant reduction in stoma reversal time from 142.8 ± 28.4 days in the control group to 118.5 ± 23.7 days in the mDCQD group (P < 0.001)-carries substantial clinical implications. This 24-day reduction represents approximately a 17% decrease in time to stoma closure, which can meaningfully improve patient quality of life by earlier restoration of normal bowel continuity, reduction in stoma-related complications, and decreased psychological burden associated with living with a stoma. Moreover, the economic impact of shortened hospitalization and reduced stoma care needs should not be underestimated. This study’s multivariate Cox regression analysis showed that mDCQD was an independent factor for shortening stoma reversal time (HR = 2.147, 95%CI: 1.658-2.781), suggesting that mDCQD administration actively modifies the clinical course rather than merely correlating with improved outcomes. A HR greater than 2 indicates that patients receiving mDCQD were more than twice as likely to achieve stoma reversal at any given time point compared to controls, which is consistent with recent research on the application of TCM in enhanced postoperative recovery[13].
From a TCM theoretical perspective, patients after colorectal cancer surgery often suffer from qi stagnation and bowel obstruction due to factors such as surgical trauma, anesthesia, and intraoperative organ traction, belonging to the category of heat-bound bowel excess syndrome[14]. Da-Cheng-Qi decoction, as a classic prescription for drastically purging heat accumulation, consists of rhubarb (bitter and cold, purgative and heat-clearing), mirabilite (softening hardness, moistening and heat-purging), immature bitter orange (breaking qi and eliminating stagnation), and magnolia bark (moving qi and eliminating distension). The four herbs work together to achieve the effects of opening bowels and purging heat, cleansing the stomach and intestines[15]. This study added radish seed (moving qi downward and eliminating distension), aucklandia root (moving qi and relieving pain), areca seed (moving qi downward and eliminating accumulation), and cannabis seed (moistening intestines and facilitating defecation) to the classical formula, enhancing the effects of moving qi, guiding stagnation, moistening intestines, and facilitating defecation, which better fits the pathological mechanism of postoperative patients with qi stagnation and bowel excess combined with qi-blood deficiency[16].
Modern pharmacological studies have revealed multiple mechanisms by which mDCQD promotes stoma reversal. The mechanisms appear to operate through complementary pathways involving gastrointestinal motility enhancement, hormonal regulation, and anti-inflammatory effects. First, anthraquinone compounds in rhubarb can stimulate intestinal smooth muscle contraction, increase intestinal propulsive movement, and promote intestinal content discharge[17]. Studies have shown that emodin enhances intestinal motility by activating 5-hydroxytryptamine receptors and cholinergic receptors in the enteric nervous plexus[18]. Second, herbs such as immature bitter orange and magnolia bark, rich in flavonoids and volatile oils, can regulate gastrointestinal hormone secretion. The gastrointestinal hormone data from our study provides direct evidence for this mechanism: This study’s results showed that on postoperative days 5 and 7, MTL and GAS levels in the observation group were significantly higher than those in the control group, while VIP levels were significantly lower, consistent with previous studies[19]. MTL and GAS are important hormones promoting gastrointestinal motility, while VIP has an inhibitory effect on gastrointestinal motility. The improvement in these three hormone levels suggests that mDCQD can promote intestinal function recovery by regulating gastrointestinal hormone secretion[20]. Third, the anti-inflammatory effects documented through reduced CRP and PCT levels on postoperative days 5 and 7 in the mDCQD group suggest that the intervention addresses the underlying inflammatory cascade that typically impairs anastomotic healing. Studies have shown that rhubarb and immature bitter orange can inhibit inflammatory mediator release and reduce intestinal inflammatory response[21]. The concurrent improvement in nutritional indicators (higher ALB and PA levels in the observation group) further supports optimal conditions for tissue healing. Collectively, these mechanisms-enhanced motility through hormonal modulation, reduced inflammation, and improved nutritional status-create a favorable microenvironment for anastomotic healing, thereby enabling earlier safe stoma reversal[22].
Postoperative ileus is one of the common complications affecting prognosis after colorectal surgery, related to multiple factors including surgical trauma, anesthetic drugs, electrolyte disturbances, and inflammatory response[23]. This study showed that the incidence of postoperative ileus in the observation group was 6.1%, significantly lower than 18.5% in the control group, with time to first anal gas passage shortened by approximately 16 hours, time to first defecation shortened by nearly 18 hours, and time to bowel sound recovery shortened by approximately 13 hours. These results were superior to previous reports of Western medicine treatment alone[24].
The mechanisms by which mDCQD reduces the incidence of ileus are multifaceted. From an intestinal motility perspective, the formula’s herbs promote intestinal peristalsis through multiple pathways: Anthraquinone compounds in rhubarb directly stimulate intestinal wall smooth muscle; the hypertonic effect of mirabilite increases intestinal lumen water content, softens stool, and stimulates reflex intestinal peristalsis; the qi-moving effects of immature bitter orange and magnolia bark relieve gastrointestinal qi stagnation; the qi-descending and stagnation-guiding effects of radish seed and areca seed accelerate intestinal content discharge[25]. This multi-target, multi-pathway action mode may be more effective than single prokinetic drugs.
From a neuro-endocrine regulation perspective, the occurrence of postoperative ileus is closely related to enteric nervous system dysfunction and hormonal regulation imbalance[26]. The improvement in gastrointestinal hormone levels in the observation group in this study indicates that mDCQD can restore normal intestinal physiological function by regulating the neuro-endocrine system. Studies have shown that alkaloid components in aucklandia root and areca seed can regulate intestinal neurotransmitter release and improve enteric nervous system function[27].
From an inflammatory response perspective, systemic inflammatory response and local intestinal inflammation caused by surgical trauma are important causes of ileus[28]. The significant improvement in inflammatory indicators in the observation group suggests that mDCQD can reduce postoperative inflammatory response. Emodin in rhubarb has the effect of inhibiting NF-κB signaling pathway, reducing the release of pro-inflammatory factors such as tumor necrosis factor-α and interleukin-6[29]. Immature bitter orange, magnolia bark, and other herbs have also been proven to have antioxidant and anti-inflammatory effects. These synergistic effects reduce postoperative inflammatory response, creating favorable conditions for intestinal function recovery[30].
This study showed that the EORTC QLQ-C30 global health status score at 3 months after stoma in the observation group was significantly higher than that in the control group, which has important clinical significance. The improvement in quality of life may be related to the following factors: First, rapid recovery of intestinal function reduced patients' discomfort symptoms such as abdominal distension and nausea, which is fully demonstrated by the improvement in TCM symptom scores in this study[31]. Second, shortened stoma reversal time means patients are freed from the physical and psychological burden of stoma earlier, which is crucial for improving quality of life. Studies have shown that the presence of a stoma significantly affects patients' social activities, sexual life, and mental health. Early stoma reversal can significantly improve patients' quality of life and psychological state[32].
Additionally, the improvement in nutritional indicators (ALB, PA) in the observation group in this study may also have a positive impact on quality of life. Nutritional status is not only related to patients’ physical recovery but also affects immune function and tolerance to anti-tumor treatment. Cannabis seed and other herbs have intestinal-moistening and nourishing effects, helping to improve patients' postoperative nutritional absorption[33]. Good nutritional status provides a foundation for patients to receive subsequent anti-tumor treatment and creates conditions for improving quality of life.
The results of this study fully demonstrate the unique advantages of integrated traditional Chinese and Western medicine in postoperative recovery after colorectal cancer surgery. The concept of fast-track surgery in western medicine emphasizes early mobilization, early feeding, and application of prokinetic drugs, which to some extent promote postoperative recovery[34]. However, Western medicine treatment alone still has limitations in improving intestinal function. The intervention of TCM provides new approaches for postoperative recovery. mDCQD, through multi-target and multi-pathway mechanisms, not only promotes intestinal motility recovery but also regulates gastrointestinal hormone secretion, reduces inflammatory response, and improves intestinal microecology. This holistic regulatory effect is difficult to achieve with single Western drugs.
It is worth noting that in this study, the timing of mDCQD use was chosen to begin on the day of gastric tube removal (postoperative day 2-3). This timing ensures adequate gastrointestinal decompression while allowing early intervention for intestinal function recovery, embodying the TCM treatment philosophy of “preventing disease before it occurs and preventing progression of existing disease”. At the same time, adjusting drug dosage according to patient body weight reflects the TCM principle of individualized treatment based on syndrome differentiation. This integrated traditional Chinese and western medicine treatment model with complementary advantages provides new ideas for enhanced postoperative recovery[35].
The innovations of this study include: First, applying mDCQD to colorectal cancer patients with postoperative stoma and exploring its effect on stoma reversal time fills a gap in this research area. Previous studies have mostly focused on the effect of Chinese herbs on early postoperative intestinal function recovery, while this study is the first to systematically evaluate its effect on the long-term indicator of stoma reversal time. Second, this study has a large sample size and comprehensive observation indicators, including not only clinical indicators such as stoma reversal time and ileus incidence but also multi-dimensional indicators such as TCM symptom scores, quality of life assessment, and gastrointestinal hormone levels, providing objective evidence for comprehensively evaluating the efficacy of mDCQD.
The clinical significance of this study lies in: Providing a safe and effective integrated traditional Chinese and Western medicine treatment plan for colorectal cancer patients with postoperative stoma, which can significantly shorten stoma reversal time, reduce the incidence of ileus, and improve quality of life, with good clinical application prospects. The shortening of stoma reversal time can not only reduce patients’ physical and psychological burden but also reduce medical costs, having important social and economic value.
This study has the following limitations: First, as a retrospective study, there may be selection bias and confounding factors. Although baseline data were balanced between the two groups, prospective randomized controlled trials are still needed for further verification. Second, patients included in this study all belonged to heat-bound bowel excess syndrome according to TCM syndrome differentiation. The efficacy for patients with other syndrome types remains to be explored. Third, this study did not conduct long-term follow-up, and the effect of mDCQD on patients' long-term prognosis is still unclear. Fourth, this study did not deeply explore the molecular mechanisms of mDCQD action, such as specific effects on intestinal flora and intestinal mucosal barrier function, which require further clarification through basic research.
Future research should conduct multi-center, large-sample, prospective randomized controlled trials to further verify the efficacy and safety of mDCQD. Specifically, prospective trials should include: (1) Broader patient inclusion criteria encompassing different tumor locations, TNM stages, and surgical approaches to enhance generalizability; (2) Longer-term follow-up periods (≥ 12 months) to assess the intervention’s impact on cancer recurrence, quality of life sustainability, and cost-effectiveness; (3) Standardized outcome measures including patient-reported outcomes and healthcare resource utilization; and (4) Pre-specified subgroup analyses to identify patient populations most likely to benefit from mDCQD therapy. At the same time, its mechanisms of action should be deeply studied, including effects on intestinal flora structure, intestinal epithelial cell function, immune regulation, and other aspects, to provide theoretical basis for optimizing treatment plans. Advanced techniques such as metagenomics, metabolomics, and transcriptomics could elucidate the complex interactions between mDCQD components and host physiology. Such mechanistic insights would not only validate the traditional use of Da-Cheng-Qi decoction but also facilitate the development of optimized formulations and potentially identify biomarkers predictive of treatment response. Additionally, the optimal treatment plans for patients with different TCM syndrome types should be explored to achieve truly individualized precision treatment.
In summary, this study shows that mDCQD can significantly shorten stoma reversal time after laparoscopic anterior resection for colorectal cancer, reduce the incidence of postoperative ileus, promote intestinal function recovery, and improve patients' quality of life. It is a safe and effective integrated traditional Chinese and western medicine treatment method worthy of clinical promotion and application.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15061] [Article Influence: 7530.5] [Reference Citation Analysis (23)] |
| 2. | Jiang W, Xu J, Cui M, Qiu H, Wang Z, Kang L, Deng H, Chen W, Zhang Q, Du X, Yang C, Guo Y, Zhong M, Ye K, You J, Xu D, Li X, Xiong Z, Tao K, Ding K, Zang W, Feng Y, Pan Z, Wu A, Huang F, Huang Y, Wei Y, Su X, Chi P; LASRE trial investigators. Laparoscopy-assisted versus open surgery for low rectal cancer (LASRE): 3-year survival outcomes of a multicentre, randomised, controlled, non-inferiority trial. Lancet Gastroenterol Hepatol. 2025;10:34-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 3. | Xu F, Li H, Guo C, Yang Z, Gao J, Zhang X, Wei Q, Meng C, Sun L, Wu G, Yao H, Zhang Z. Incidence and Risk Factors of Surgical Complications and Anastomotic Leakage After Transanal Total Mesorectal Excision for Middle and Low Rectal Cancer. J Gastrointest Surg. 2023;27:373-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 4. | Feng JY, Wang SF, Yan J. The Application of Enhanced Recovery After Surgery for Gastrectomy and Colorectal Resection: A Systematic Review and Meta-Analysis. J Laparoendosc Adv Surg Tech A. 2023;33:586-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 5. | Xue J, Xu Z, Wang Q, Hou H, Wei L, Zhang J, Zhao X, Chen L, Ding F, Ma L, Zhao Y, Wang Y, Ma D, Wang T, Liu R, Gan TJ, Robinson N, Frank Y, Su F, Chi Y, Yang D, Liu S, Cui S, Wei Y, Chen Z, Qin Y, Cao L, Chen G, Shu K, Xiao Z, Zhang H, Yu J, Hu Z, Cheng H, Ma W, Liu G, Wang X, Cao X, Gao J, Kong G, Tao Q, Wang B, Wang J, Li H, Lyu C, Zhang Z, Li T, Yang K. Clinical practice guidelines for prevention and treatment of postoperative gastrointestinal disorder with Integrated Traditional Chinese and Western Medicine (2023). J Evid Based Med. 2024;17:207-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 6. | Duan WQ, Cai MC, Ma QQ, Huang P, Zhang JH, Wei TF, Shang D, Leng AJ, Qu JL. Exploring the chemical components of Kuanchang-Shu granule and its protective effects of postoperative ileus in rats by regulating AKT/HSP90AA1/eNOS pathway. Chin Med. 2024;19:29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 7. | Guo W, Zhang X, Zhou X, Lan W, Liu J, Liu Y, Li L, Li Z. Dachengqi decoction for the treatment of acute pancreatitis: a comprehensive analysis based on metabolites, pharmacokinetics, and metabolites efficacy mechanisms. Front Pharmacol. 2025;16:1549909. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 8. | Ye Z, Wei X, Feng S, Gu Q, Li J, Kuai L, Luo Y, Xi Z, Wang K, Zhou J. Effectiveness and safety of acupuncture for postoperative ileus following gastrointestinal surgery: A systematic review and meta-analysis. PLoS One. 2022;17:e0271580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (3)] |
| 9. | Hu Q, Chen XP, Tang ZJ, Zhu XY, Liu C. Therapeutic effects of Buzhong Yiqi decoction in patients with spleen and stomach qi deficiency after routine surgery and chemotherapy for colorectal cancer. World J Gastrointest Surg. 2024;16:2183-2193. [PubMed] [DOI] [Full Text] |
| 10. | Hou YT, Pan YY, Wan L, Zhao WS, Luo Y, Yan Q, Zhang Y, Zhang WX, Mo YC, Huang LP, Dai QX, Jia DY, Yang AM, An HY, Wu AS, Tian M, Fang JQ, Wang JL, Feng Y. Transcutaneous electrical acupoint stimulation in adult patients receiving gastrectomy/colorectal resection: A randomized controlled trial. World J Gastrointest Surg. 2023;15:1474-1484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Yin FT, Zhou XH, Kang SY, Li XH, Li J, Ullah I, Zhang AH, Sun H, Wang XJ. Prediction of the mechanism of Dachengqi Decoction treating colorectal cancer based on the analysis method of " into serum components -action target-key pathway". J Ethnopharmacol. 2022;293:115286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (6)] |
| 12. | MacDonald S, Gallagher A, McNicholl L, McElroy L, Hughes R, Quasim T, Moug S. Stoma reversal after emergency stoma formation-the importance of timing: a multi-centre retrospective cohort study. World J Emerg Surg. 2025;20:26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 13. | Jin O, Xu T, Lai J, He J, Wu Y, Yang X. Impact of enhanced recovery after surgery concept process optimization on the perioperative period of gynecologic laparoscopic surgery. BMC Womens Health. 2025;25:120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 14. | Abernethy EK, Aly EH. Postoperative Ileus after Minimally Invasive Colorectal Surgery: A Summary of Current Strategies for Prevention and Management. Dig Surg. 2024;41:79-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 15. | Liu Y, Lei Y, Shang L, Yu Z, Wang L, Zhou J, Pan G. Protective effects of Dachengqi Decoction in sepsis-associated liver injury based on network pharmacology analysis and experimental validation. J Ethnopharmacol. 2025;353:120450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Luo Y, Feng X, Wu D, Wang J, Lyv Z, Zheng J, Lim KT, Li Y. A randomized controlled trial of Chinese traditional medicine Dachengqi Decoction in the treatment of postoperative intestinal function recovery. Transl Cancer Res. 2020;9:4498-4506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Ma Q, Wang CZ, Sawadogo WR, Bian ZX, Yuan CS. Herbal Medicines for Constipation and Phytochemical Comparison of Active Components. Am J Chin Med. 2022;50:723-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 18. | Guzel T, Mirowska-Guzel D. The Role of Serotonin Neurotransmission in Gastrointestinal Tract and Pharmacotherapy. Molecules. 2022;27:1680. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 109] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 19. | Liang C, Qiu FX, Zhang XC, Hu QL. Effects of gastrointestinal motility therapy combined with acupuncture on gastrointestinal function in patients after laparoscopic radical surgery. World J Gastrointest Surg. 2025;17:104325. [PubMed] [DOI] [Full Text] |
| 20. | Wang J, Lu FF, Ge MM, Wang LW, Wang G, Gong GW, Liu XX, Zhang WZ, Ning FL, Chen BH, Liu Y, Quan HG, Jiang ZW. Transcutaneous Electrical Acupoint Stimulation Improves Postoperative Sleep Quality in Patients Undergoing Laparoscopic Gastrointestinal Tumor Surgery: A Prospective, Randomized Controlled Trial. Pain Ther. 2023;12:707-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 21. | Zhou Y, Gao C, Vong CT, Tao H, Li H, Wang S, Wang Y. Rhein regulates redox-mediated activation of NLRP3 inflammasomes in intestinal inflammation through macrophage-activated crosstalk. Br J Pharmacol. 2022;179:1978-1997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 22. | Boyer ME, Dhimal T, Hilty Chu BK, Loria A, Cai X, Gao S, Jacobs ML, Li Y, Cupertino P, Fleming FJ. Assessing the pathway and timing for ileostomy reversal in rectal cancer: A retrospective cohort study. Surgery. 2025;185:109507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 23. | Quiroga-Centeno AC, Jerez-Torra KA, Martin-Mojica PA, Castañeda-Alfonso SA, Castillo-Sánchez ME, Calvo-Corredor OF, Gómez-Ochoa SA. Risk Factors for Prolonged Postoperative Ileus in Colorectal Surgery: A Systematic Review and Meta-analysis. World J Surg. 2020;44:1612-1626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 24. | Wolthuis AM, Bislenghi G, Lambrecht M, Fieuws S, de Buck van Overstraeten A, Boeckxstaens G, D'Hoore A. Preoperative risk factors for prolonged postoperative ileus after colorectal resection. Int J Colorectal Dis. 2017;32:883-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Montgomery LE, Tansey EA, Johnson CD, Roe SM, Quinn JG. Autonomic modification of intestinal smooth muscle contractility. Adv Physiol Educ. 2016;40:104-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 26. | Wang Z, Stakenborg N, Boeckxstaens G. Postoperative ileus-Immune mechanisms and potential therapeutic interventions. Neurogastroenterol Motil. 2025;37:e14951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 27. | Zhang YM, Zhu L, Zhao XL, Chen H, Kang HX, Zhao JL, Wan MH, Li J, Zhu L, Tang WF. Optimal timing for the oral administration of Da-Cheng-Qi decoction based on the pharmacokinetic and pharmacodynamic targeting of the pancreas in rats with acute pancreatitis. World J Gastroenterol. 2017;23:7098-7109. [PubMed] [DOI] [Full Text] |
| 28. | Schneider R, Leven P, Mallesh S, Breßer M, Schneider L, Mazzotta E, Fadda P, Glowka T, Vilz TO, Lingohr P, Kalff JC, Christofi FL, Wehner S. IL-1-dependent enteric gliosis guides intestinal inflammation and dysmotility and modulates macrophage function. Commun Biol. 2022;5:811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (4)] |
| 29. | HaoShang, Jia X, Liu H, Zhang X, Shao Y. A comprehensive review of emodin in fibrosis treatment. Fitoterapia. 2023;165:105358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 30. | Shang H, Guo J, Wang P, Li L, Tian Y, Li X, Zou Z. Design, synthesis and anti-inflammatory evaluation of aloe-emodin derivatives as potential modulators of Akt, NF-κB and JNK signaling pathways. Eur J Med Chem. 2022;238:114511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 31. | Milne T, Liu C, O'Grady G, Woodfield J, Bissett I. Effect of prucalopride to improve time to gut function recovery following elective colorectal surgery: randomized clinical trial. Br J Surg. 2022;109:704-710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 32. | Xi Z, Rong CM, Ling LJ, Hua ZP, Rui G, Fang HG, Long W, Zhen ZH, Hong L. The influence of stigma and disability acceptance on psychosocial adaptation in patients with stoma: A multicenter cross-sectional study. Front Psychol. 2022;13:937374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 33. | McKechnie T, Kazi T, Jessani G, Shi V, Sne N, Doumouras A, Hong D, Eskicioglu C. The use of preoperative enteral immunonutrition in patients undergoing elective colorectal cancer surgery: A systematic review and meta-analysis. Colorectal Dis. 2025;27:e70061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 34. | Kannan V, Ullah N, Geddada S, Ibrahiam A, Munaf Shakir Al-Qassab Z, Ahmed O, Malasevskaia I. Impact of "Enhanced Recovery After Surgery" (ERAS) protocols vs. traditional perioperative care on patient outcomes after colorectal surgery: a systematic review. Patient Saf Surg. 2025;19:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 31] [Reference Citation Analysis (1)] |
| 35. | Cao LX, Chen ZQ, Jiang Z, Chen QC, Fan XH, Xia SJ, Lin JX, Gan HC, Wang T, Huang YX. Rapid rehabilitation technique with integrated traditional Chinese and Western medicine promotes postoperative gastrointestinal function recovery. World J Gastroenterol. 2020;26:3271-3282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |