Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116768
Revised: February 5, 2026
Accepted: March 6, 2026
Published online: April 27, 2026
Processing time: 101 Days and 1.4 Hours
Perioperative hypothermia is a common complication of gastrointestinal surgery and is associated with an increased risk of surgical site infections and other com
To investigate the impact and underlying pathophysiological mechanisms of pe
A retrospective analysis was conducted on 45 patients who underwent elective gastrointestinal surgery at our institution between 2020 and 2025: Nine patients with postoperative infections within 30 days and 36 uninfected controls. A tem
The case group had significantly lower intraoperative and immediate post
Perioperative hypothermia and inadequate nursing quality for temperature management are independent risk factors for postoperative infections following gastrointestinal surgery. Enhanced temperature monitoring and active warming via standardized individualized temperature management protocols may reduce the risk of post
Core Tip: This 1:4 retrospective study of 45 patients who underwent elective gastrointestinal surgery identified the lowest intraoperative body temperature and temperature management compliance scores as independent risk factors for postoperative infectious complications. This association was significantly modified by the surgical approach (open vs laparoscopic) and the diabetes status. The underlying mechanisms involve impaired microcirculation (increased core-to-skin temperature gradient and decreased peripheral perfusion index), metabolic dysregulation (reduced lactate clearance), and increased systemic inflammation (elevated interleukin-6). Standardized individualized temperature management protocols are crucial, especially in open surgery and patients with diabetes.
- Citation: Jiang HY. Impact of perioperative temperature management nursing quality on postoperative infectious complications in patients undergoing gastrointestinal surgery. World J Gastrointest Surg 2026; 18(4): 116768
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116768.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116768
Gastrointestinal surgery is one of the most common surgical procedures and includes a wide spectrum of interventions, ranging from minimally invasive appendectomies to complex resections for gastric and colorectal malignancies. Despite continued advances in surgical techniques, anesthesia, and perioperative care, postoperative infectious complications remain a major clinical challenge, leading to prolonged hospitalization, increased medical costs, delayed recovery, and increased mortality[1]. The incidence of postoperative infections after gastrointestinal surgery ranges from 5% to 15%, highlighting the need for effective preventive strategies[2].
Perioperative hypothermia, defined as a core body temperature less than 36.0 °C, is a frequent yet preventable adverse event during gastrointestinal surgery. Accumulating evidence has identified perioperative hypothermia as an inde
In recent years, perioperative temperature management has been recognized as a core component of enhanced recovery after surgery (ERAS) programs, emphasizing continuous temperature monitoring, timely warming interven
Therefore, this study aimed to evaluate the association between perioperative temperature management nursing qua
Adult patients who underwent elective gastrointestinal surgery in the Department of General Surgery of our hospital between January 2020 and May 2025 were retrospectively enrolled. The inclusion criteria included: (1) Age ≥ 18 years; (2) Elective open or laparoscopic gastrointestinal surgery (e.g., laparoscopic appendectomy, radical resection for rectal or gastric cancer) under general anesthesia, with an operation duration ≥ 60 minutes; and (3) Complete medical and nursing records. The exclusion criteria included: (1) Emergency surgery; (2) Pre-existing infection or fever (body temperature ≥
All data were independently extracted from the hospital’s electronic medical record system by two trained researchers and crosschecked to ensure accuracy.
Demographic and basic clinical data: The demographic characteristics included age and sex. The clinical characteristics included body mass index, American Society of Anesthesiologists physical status classification, presence of type 2 diabetes mellitus, and preoperative serum albumin level (g/L). Surgical variables included the surgical approach (open vs laparoscopic surgery), total operation duration (minutes), total intraoperative fluid infusion volume (mL), and total intraoperative blood loss (mL).
Perioperative temperature management-related indicators: In the preoperative phase, nursing records were reviewed to verify whether patients received active warming (e.g., warming blanket and forced-air warmer) 30-60 minutes before entering the operating room, recorded as “yes/no”. In the intraoperative phase, the following were reviewed: (1) Temperature data: Core body temperature (nasopharyngeal or bladder) recorded every 15 minutes was extracted from the anesthesia records. Two metrics were extracted, namely intraoperative lowest body temperature (°C) and duration of hypothermia (cumulative minutes with temperature < 36.0 °C); and (2) Warming intervention: Surgical nursing records were reviewed to confirm whether active warming devices (forced-air warming blanket and/or fluid/blood warmer for infusions/irrigation fluids) were used, recorded as “yes/no”. Finally, in the postoperative phase, the core body temperature immediately upon admission to the post-anesthesia care unit was extracted and recorded as the Immediate postoperative body temperature (°C). In terms of the temperature management nursing quality composite score, to quantify the nursing team’s adherence to perioperative temperature management protocols, a 5-dimension scale was constructed (each dimension scored 0-2: 0 = not performed, 1 = partially performed, 2 = fully performed) with a total score ranging from 0 to 10. The dimensions included: (1) Preoperative warming; (2) Intraoperative use of active warming devices; (3) Intraoperative temperature monitoring frequency (≥ every 15-30 minutes); (4) Completeness of temperature and intervention documentation; and (5) Timely management of intraoperative hypothermia. The scores were inde
Potential pathophysiological mechanism indicators between perioperative hypothermia and postoperative infection: To elucidate the potential pathophysiological links between perioperative hypothermia and postoperative infection and enhance the precision of temperature management quality assessment, data on the following indicators across four dimensions were collected and analyzed: Microcirculatory perfusion, inflammatory response, metabolic status, and nursing response efficiency. To this end, several measurements were taken: (1) Intraoperative core temperature vari
Data were analyzed using SPSS software (version 26.0). Measurement data are presented as the mean ± SD, and group comparisons were made using paired t-tests. Count data are presented as n (%), and comparisons were made using the χ2 test or Fisher’s exact test. Conditional logistic regression analysis was used to identify factors influencing postoperative infection, calculating the odds ratios (ORs) and their 95% confidence intervals (CIs). All tests were two-sided, and P < 0.05 was considered statistically significant.
No statistically significant differences were observed between the case and control groups in the baseline demographic and clinical characteristics, including age, sex, body mass index, American Society of Anesthesiologists classification, diabetes status, preoperative albumin level, surgical approach, operative duration, intraoperative fluid volume, and blood loss. Sex distribution was compared based on the proportion of men, with women constituting the remaining pro
| Variable | Case group (n = 9) | Control group (n = 36) | t/χ2 value | P value |
| Age (years) | 62.32 ± 8.71 | 61.85 ± 9.12 | 0.139 | 0.890 |
| Sex, male | 6 (66.67) | 21 (58.33) | 0.721 | |
| BMI (kg/m2) | 23.53 ± 3.21 | 24.16 ± 2.91 | 0.570 | 0.572 |
| ASA ≥ III | 4 (44.44) | 13 (36.11) | 0.711 | |
| Complicated with diabetes | 3 (33.33) | 10 (27.78) | 0.704 | |
| Preoperative albumin (g/L) | 36.22 ± 5.11 | 37.85 ± 4.72 | 0.912 | 0.367 |
| Open surgery | 5 (55.56) | 17 (47.22) | 0.722 | |
| Operation time (minutes) | 185.65 ± 42.36 | 178.92 ± 38.71 | 0.458 | 0.649 |
| Intraoperative fluid volume (mL) | 1520.09 ± 320.23 | 1480.45 ± 290.67 | 0.359 | 0.721 |
| Intraoperative blood loss (mL) | 210.62 ± 85.78 | 195.09 ± 78.80 | 0.520 | 0.606 |
Compared with the control group, the case group showed a significantly lower intraoperative body temperature (35.22 ± 0.61 °C vs 36.15 ± 0.42 °C), longer duration of hypothermia (82.35 ± 25.61 minutes vs 28.75 ± 9.26 minutes), lower immediate postoperative body temperature (35.54 ± 0.72 °C vs 36.34 ± 0.52 °C), and a lower temperature management compliance score (5.13 ± 1.31 vs 8.47 ± 1.11; all P < 0.001). The proportion of patients receiving intraoperative active warming was also significantly lower in the case group (22.22% vs 75.00%, P = 0.006). No significant difference was observed in preoperative warming between the two groups (33.33% vs 61.11%, P > 0.05; Table 2).
| Indicator | Case group (n = 9) | Control group (n = 36) | t/χ2 value | P value |
| Preoperative warming | 3 (33.33) | 22 (61.11) | 0.157 | |
| Lowest intraoperative body temperature (°C) | 35.22 ± 0.61 | 36.15 ± 0.42 | 5.409 | < 0.001 |
| Duration of hypothermia (minutes) | 82.35 ± 25.61 | 28.75 ± 9.26 | 10.385 | < 0.001 |
| Intraoperative active warming | 2 (22.22) | 27 (75.00) | 0.006 | |
| Immediate postoperative temperature (°C) | 35.54 ± 0.72 | 36.34 ± 0.52 | 3.815 | < 0.001 |
| Temperature management compliance score | 5.13 ± 1.31 | 8.47 ± 1.11 | 7.794 | < 0.001 |
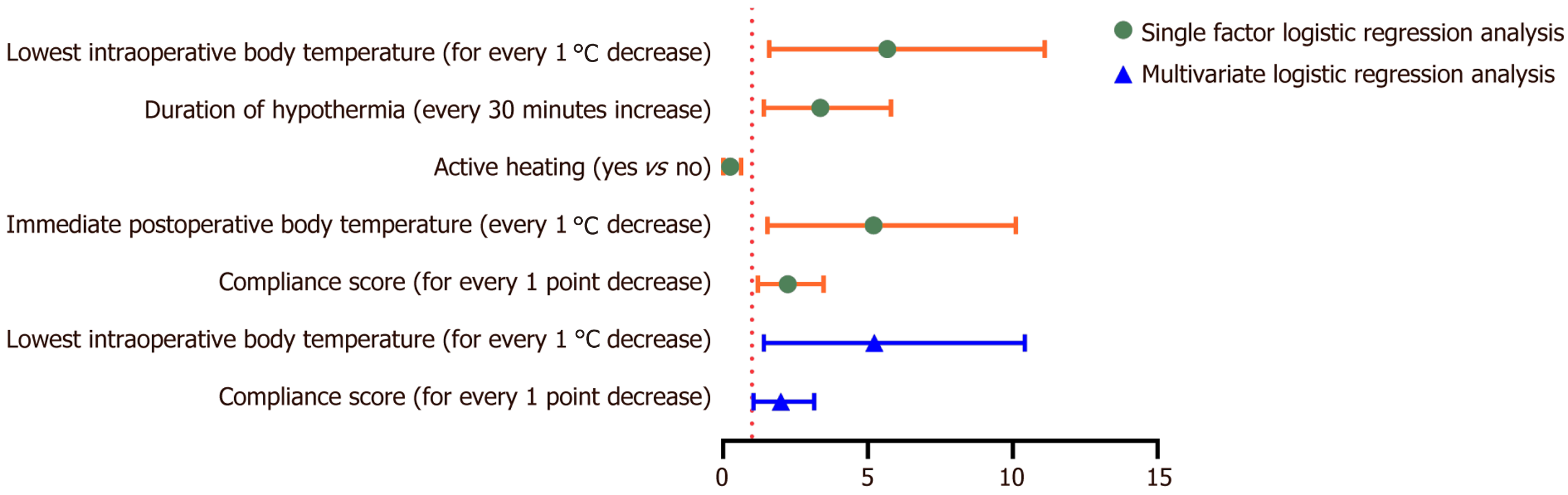
Univariate conditional logistic regression analysis showed that the lowest intraoperative body temperature, duration of hypothermia, use of active warming, immediate postoperative body temperature, and compliance score were signi
| Variable | Univariate analysis | Multiplicity | ||||
| OR | 95%CI | P value | OR | 95%CI | P value | |
| Minimum intraoperative body temperature (per 1 °C decrease) | 4.320 | 1.608-11.121 | 0.002 | 3.850 | 1.420-10.430 | 0.008 |
| Duration of hypothermia (per 30-minute increment) | 2.870 | 1.420 to -5.811 | 0.003 | |||
| Active heating (yes/no) | 0.110 | 0.020-0.630 | 0.013 | |||
| Intraoperative body temperature (per 1 °C decrease) | 3.950 | 1.540-10.122 | 0.004 | |||
| Compliance score (each point down) | 2.050 | 1.210-3.480 | 0.008 | 1.820 | 1.050-3.160 | 0.032 |
In the open surgery subgroup, each 1 °C decrease in the lowest intraoperative body temperature and each 1-point decrease in the temperature management compliance score significantly increased the risk of postoperative infection (P < 0.05). These associations were not statistically significant in the laparoscopic surgery subgroup (P > 0.05). The surgical approach demonstrated a significant modifying effect on the association between low intraoperative body temperature and postoperative infection (P for interaction = 0.046). In the subgroup with diabetes, each 1 °C decrease in the intraoperative lowest body temperature was associated with a greater increase in infection risk than in the non-diabetic sub
| Stratification factor | Variable | Subgroup | Case/control | OR (95%CI) | P value | P for interaction |
| Surgical approach | Lowest intraoperative temperature (per 1 °C decrease) | Open surgery | 5/17 | 5.231 (2.905-9.437) | 0.002 | 0.046 |
| Laparoscopic surgery | 4/19 | 2.152 (1.441-3.222) | 0.138 | |||
| Compliance score (per 1-point decrease) | Open surgery | 5/17 | 2.311 (1.564-3.421) | 0.023 | 0.118 | |
| Laparoscopic surgery | 4/19 | 1.572 (0.951-2.602) | 0.121 | |||
| Diabetes status | Lowest intraoperative temperature (per 1 °C decrease) | With diabetes | 3/10 | 6.125 (3.214-11.680) | 0.001 | 0.049 |
| Without diabetes | 6/26 | 3.012 (1.822-4.983) | 0.045 | |||
| Compliance score (per 1-point decrease) | With diabetes | 3/10 | 1.927 (0.994-3.751) | 0.047 | 0.812 | |
| Without diabetes | 6/26 | 1.785 (0.911-3.501) | 0.058 |
The case group had a significantly higher intraoperative core temperature variability, intraoperative core-to-skin tem
| Indicator | Case group (n = 9) | Control group (n = 36) | t value | P value |
| Intraoperative core temperature variability (°C) | 0.48 ± 0.12 | 0.21 ± 0.07 | 8.873 | < 0.001 |
| Intraoperative core-to-skin temperature gradient (°C) | 4.35 ± 1.02 | 2.67 ± 0.84 | 5.144 | < 0.001 |
| Intraoperative lactate clearance rate (%/hour) | 18.32 ± 3.51 | 32.73 ± 4.11 | 9.654 | < 0.001 |
| Serum PCT (ng/mL) 2 hours postoperative | 2.65 ± 0.76 | 0.82 ± 0.21 | 12.969 | < 0.001 |
| Immediate postoperative peripheral perfusion index | 0.82 ± 0.31 | 1.45 ± 0.47 | 3.802 | < 0.001 |
| Temperature management response delay time (minutes) | 24.65 ± 3.81 | 17.33 ± 2.24 | 7.541 | < 0.001 |
| Intraoperative FiO2 exposure index (%·minutes) | 4820.20 ± 1050.13 | 4100.04 ± 890.23 | 2.096 | 0.042 |
Utilizing a real-world clinical database and a 1:4 matched case-control design, this study systematically evaluated the impact of perioperative temperature management nursing quality on infectious complications after gastrointestinal surgery. The results demonstrated that each 1 °C decrease in lowest intraoperative body temperature and each 1-point decrease in the temperature management compliance score were independent risk factors for postoperative infection. This association showed significant effect modification in patients undergoing open surgery and those with diabetes. Furthermore, by integrating mechanism indicators, such as core temperature variability, core-to-skin temperature gradient, lactate clearance rate, PCT potency, peripheral perfusion index, and temperature intervention response delay time, a pathophysiological pathway model of perioperative hypothermia linking perioperative hypothermia with microcirculatory dysfunction, metabolic disturbance, immunosuppression, and inflammatory imbalance was constructed. This study provides mechanistic evidence clarifying the biological basis of temperature management for perioperative infection prevention and control.
Perioperative hypothermia (< 36 °C) has been identified as an intervenable risk factor for postoperative complications[6-8]. The results of this study showed that each 1 °C decreases in lowest intraoperative body temperature increased the risk of postoperative infection by 2.85 times (OR = 3.85; 95%CI: 1.420-10.430). Mechanistic analysis suggested that hypothermia directly suppresses innate immunity, significantly impairing neutrophil chemotaxis, migration, and oxidative burst activity, thereby reducing pathogen recognition and clearance efficiency[9,10]. Moreover, hypothermia induces peripheral vasoconstriction, reduces tissue perfusion, and decreases subcutaneous tissue oxygen tension (PtO2), which is the rate-limiting factor in collagen synthesis and wound healing[11]. Additionally, the hypoperfusion state prolongs lactate clearance time, further exacerbating local acidosis and hypoxia. In this study, the intraoperative lactate clearance rate was significantly lower in the case group compared to the control group (18.32 ± 3.51 %/hour vs 32.73 ± 4.11 %/hour), and the immediate postoperative peripheral perfusion index was also concurrently reduced (0.82 ± 0.31 vs 1.45 ± 0.47). This cycle of “hypoxia-metabolic disorder-delayed repair” creates a favorable microenvironment for bacterial colonization and invasion, ultimately manifesting as an exponential increase in the infection risk[12-14].
The results of this study showed that the temperature management response delay time was significantly longer in the case group compared to the control group (24.65 ± 3.81 minutes vs 17.33 ± 2.24 minutes). This delay increases the “time window” of hypothermia exposure for patients and results in the lack of intervention during the critical early window for microcirculatory dysfunction[15]. From a systems perspective, response delay essentially represents a breakdown in the “recognition, decision, and action” chain, revealing the structural defect of “disruption between monitoring and action” in clinical practice[16]. Further analysis of the temperature management compliance score revealed it was significantly lower in the case group (5.13 ± 1.31 vs 8.47 ± 1.11), suggesting that inadequate temperature management is not merely a technical error but a reflection of nursing system quality. Logistic regression showed that each 1-point decrease in the compliance score increased the infection risk by 1.82 times (OR = 1.82, 95%CI: 1.05-3.16), strengthening from an evidence-based perspective the core value of structured, standardized temperature management processes in infection prevention and control.
Further analysis of the pathophysiological mechanisms by which hypothermia promotes infection revealed that the intraoperative core temperature fluctuation amplitude was significantly larger in the case group (0.48 ± 0.12 °C vs 0.21 ± 0.07 °C), indicating thermoregulatory imbalance[17,18]. Concurrently, the core-to-skin temperature gradient increased from 2.67 ± 0.84 °C to 4.35 ± 1.02 °C, reflecting intense peripheral vasoconstriction and an increased core-to-peripheral heat gradient, which is a typical sign of inadequate tissue perfusion[19,20]. In this context, the body increases oxygen consumption to maintain core functions. If hypothermia persists, anaerobic metabolism increases, leading to lactate accumulation. The decreased lactate clearance rate further supports mitochondrial dysfunction and energy metabolism disturbance[21,22]. Crucially, postoperative PCT levels in the case group were significantly elevated at 2 hours (2.65 ± 0.76 ng/mL vs 0.82 ± 0.21 ng/mL), indicating that systemic inflammatory responses to bacterial infection were markedly activated under hypothermic conditions. As a highly specific biomarker for bacterial infection, elevated PCT indicates an increased pathogen load and signals an overactivated immune system. Persistently high PCT expression is frequently associated with endothelial injury, coagulation dysfunction, and increased risk of multiple organ dysfunction, resulting in a cycle of “infection-inflammation-coagulation-microcirculatory impairment”. This cycle ultimately compromises the tissue barrier integrity and accelerates the spread of infection[23].
The subgroup analysis indicated that clinical heterogeneity had a significant modifying effect on the association between hypothermia and infection. In the open surgery subgroup, each 1 °C decrease in intraoperative lowest body temperature increased the postoperative infection risk by 5.23 times, far exceeding the corresponding increase in the laparoscopic surgery subgroup, and a significant interaction existed between surgical approach and body temperature. This difference may be attributed to the larger exposed body cavity area, longer duration of heat loss, and stronger traumatic stress in open surgery, which more easily disrupts the thermoregulatory center and amplifies the immunosuppressive effects of hypothermia. Similarly, patients with diabetes exhibited a hypersensitivity to hypothermia: Each 1 °C decrease in the lowest body temperature drastically increased their infection risk by 6.13 times. Patients with diabetes are in a state of chronic low-grade inflammation, impaired neutrophil chemotaxis, phagocytosis, and microangiopathy. Hypothermia can further aggravate vascular endothelial injury and tissue hypoxia, creating a metabolic-immune-microcirculatory triple hit that synergistically amplifies infection susceptibility[24]. These results suggest that high-risk populations urgently require individualized temperature management to reduce infection incidence, including inten
This study has several limitations. First, this was a single-center retrospective study with a relatively small sample size, which may have limited the statistical power and generalizability of the findings. However, a 1:4 retrospective study design was employed to control major confounding factors, and conditional logistic regression was applied to enhance analytical robustness. Given the relatively low incidence of postoperative infections in elective gastrointestinal surgery, this study should be regarded as exploratory and hypothesis generating. Larger multicenter prospective studies are warranted to validate these findings.
From a clinical perspective, these findings highlight the importance of incorporating perioperative temperature management into routine quality assessment indicators within ERAS pathways. Establishing standardized protocol-driven temperature monitoring and intervention workflows supported by electronic medical record-based early warning systems may reduce response delays and improve patient outcomes. Future studies should focus on multicenter vali
In summary, this study verified the protective role of perioperative temperature management in reducing the incidence of postoperative infections and systematically constructed a complete pathological pathway linking hypothermia with microcirculatory dysfunction, metabolic disturbance, immunosuppression, inflammatory imbalance, and subsequent infection occurrence through mechanistic indicators, thereby clarifying the key regulatory role of body temperature in the occurrence of perioperative infections. These findings highlight the importance of incorporating temperature mana
| 1. | Wang HY, Zou Y, Shi LY, Qin X, Hong LJ. Effect of perioperative disinfection and isolation measures in infection control after gastrointestinal surgery: A retrospective analysis. World J Gastrointest Surg. 2025;17:102799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 2. | Chen R, Du Y, Chen L, Bai Y, Zhang Y, Yu T, Li H, Wang G. The impact of perioperative hypothermia on surgical site infection risk: a meta-analysis. BMC Anesthesiol. 2025;25:443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 3. | Tan R, Chen Y, Yang D, Long X, Ma H, Yang C. Risk factors for postoperative hypothermia in non-cardiac surgery patients: a systematic review and meta-analysis. BMC Anesthesiol. 2025;25:223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 4. | Cho SA, Lee SJ, Yoon S, Sung TY. Risk Factors for Postoperative Hypothermia in Patients Undergoing Robot-Assisted Gynecological Surgery: A Retrospective Cohort Study. Int J Med Sci. 2022;19:1147-1154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 5. | Werner LM, Kevorkian RT, Getnet D, Rios KE, Hull DM, Robben PM, Cybulski RJ, Bobrov AG. Hypothermia: Pathophysiology and the propensity for infection. Am J Emerg Med. 2025;88:64-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 6. | Whitelaw A, Thoresen M. Therapeutic Hypothermia for Hypoxic-Ischemic Brain Injury Is More Effective in Newborn Infants than in Older Patients: Review and Hypotheses. Ther Hypothermia Temp Manag. 2023;13:170-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | McGinigle KL, Spangler EL, Ayyash K, Arya S, Settembrini AM, Thomas MM, Dell KE, Swiderski IJ, Davies MG, Setacci C, Urman RD, Howell SJ, Garg J, Ljungvist O, de Boer HD. A framework for perioperative care for lower extremity vascular bypasses: A Consensus Statement by the Enhanced Recovery after Surgery (ERAS®) Society and Society for Vascular Surgery. J Vasc Surg. 2023;77:1295-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 8. | Goswami K, Stevenson KL, Parvizi J. Intraoperative and Postoperative Infection Prevention. J Arthroplasty. 2020;35:S2-S8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 9. | Janko J, Bečka E, Kmeťová K, Hudecová L, Konečná B, Celec P, Bajaj-Elliott M, Pastorek M. Neutrophil extracellular traps formation and clearance is enhanced in fever and attenuated in hypothermia. Front Immunol. 2023;14:1257422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 10. | Masso-Silva JA, Bowlin GL. Editorial: Neutrophil heterogeneity in healing and tissue damage. Front Immunol. 2024;15:1537469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Güven B, İbrahimoğlu Ö, Kuş İ. Inadvertent Perioperative Hypothermia in Ambulatory Surgery Patients: Incidence, Risk Factors, and Prevention Initiatives. J Perianesth Nurs. 2023;38:792-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 12. | Jyoti D, Kumar A, Halim T, Hai AA. The Association Between Serum Lactate Concentration, Base Deficit, and Mortality in Polytrauma Patients as a Prognostic Factor: An Observational Study. Cureus. 2022;14:e28200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 13. | Çeleğen M, Çeleğen K. Lactate Clearance as an Early Prognostic Marker of Mortality for Pediatric Trauma. Klin Padiatr. 2023;235:270-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 14. | Lee C, Lee J, Cho H, Song J, Jung H, Ma X, Hwang J. The Association of Perioperative Serum Lactate Levels with Postoperative Delirium in Elderly Trauma Patients. Biomed Res Int. 2019;2019:3963780. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Xia F, Li Q, Cheng Z, Yan Q, Hu J, Liu J, Li G, Chen R, Zhang J, Li J. Development, validation and visualization of risk prediction model for postoperative shivering in patients undergoing video-assisted thoracoscopic lobectomy: a real-world retrospective study. Sci Rep. 2025;15:35849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 16. | Ji N, Wang J, Li X, Shang Y. Strategies for perioperative hypothermia management: advances in warming techniques and clinical implications: a narrative review. BMC Surg. 2024;24:425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 17. | Wang G, He S, Yu M, Zhang Y, Mu D, Wang D. Intraoperative body temperature and emergence delirium in elderly patients after non-cardiac surgery: A secondary analysis of a prospective observational study. Chin Med J (Engl). 2023;136:2330-2339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 18. | Ding N, Yang J, Wu C. Comparison of prewarming plus intraoperative warming with intraoperative warming alone in patients undergoing minimally invasive thoracic or abdominal surgery: A systematic review and meta-analysis. PLoS One. 2024;19:e0310096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 19. | Guo Q, Liu D, Wang X; Chinese Critical Ultrasound Study Group (CCUSG). Early peripheral perfusion monitoring in septic shock. Eur J Med Res. 2024;29:477. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 20. | Amson H, Vacheron CH, Thiolliere F, Piriou V, Magnin M, Allaouchiche B. Core-to-skin temperature gradient measured by thermography predicts day-8 mortality in septic shock: A prospective observational study. J Crit Care. 2020;60:294-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 21. | Ma F, Yu W. The Roles of Lactate and Lactylation in Diseases Related to Mitochondrial Dysfunction. Int J Mol Sci. 2025;26:7149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 22. | Cai M, Li S, Cai K, Du X, Han J, Hu J. Empowering mitochondrial metabolism: Exploring L-lactate supplementation as a promising therapeutic approach for metabolic syndrome. Metabolism. 2024;152:155787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 23. | Xu HG, Tian M, Pan SY. Clinical utility of procalcitonin and its association with pathogenic microorganisms. Crit Rev Clin Lab Sci. 2022;59:93-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 24. | Maruhashi T, Higashi Y. Pathophysiological Association between Diabetes Mellitus and Endothelial Dysfunction. Antioxidants (Basel). 2021;10:1306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 113] [Article Influence: 22.6] [Reference Citation Analysis (0)] |