Published online Apr 27, 2026. doi: 10.4240/wjgs.v18.i4.116371
Revised: December 21, 2025
Accepted: February 11, 2026
Published online: April 27, 2026
Processing time: 165 Days and 20.4 Hours
Gastric tumors originating from the muscularis propria layer, particularly those with extraluminal growth, pose therapeutic challenges because treatment must balance complete resection with preservation of gastric integrity. Although both digestive endoscopic super-minimally invasive surgery (DESMIS) and laparoscopic wedge resection (lap MWR) are increasingly used, objective preoperative criteria to guide selection between these approaches remain unclear.
To target observable preoperative indicators and compare the efficacy of DESMIS and Lap MWR for treating EGT-MP, aiming to guide optimal treatment plan selection for such tumors at different locations.
Patients who underwent DESMIS or Lap MWR for EGT-MP at the First Medical Center of Chinese People’s Liberation Army General Hospital between January 2019 and December 2023 were retrospectively selected. Basic patient information, surgical, health economics, and efficacy evaluation indicators; pathological types; postoperative medical management; and laboratory tests were recorded. They were categorized into the endoscopic full-thickness resection, submucosal tun
Overall, 542 patients were included in the study, with 98, 99, and 345 in the en
DESMIS provides several advantages for EGT-MP, achieving surgical outcomes comparable to those of Lap MWR while yielding better health economics benefits. Our findings suggest that Lap MWR and DESMIS are more advantageous for EGT-MP in the antrum and fundus of the stomach, respectively.
Core Tip: This study compared digestive endoscopic super minimally invasive surgery and laparoscopic wedge resection (Lap MWR) for treating extraluminal gastric tumors originating from the muscularis propria layer (EGT-MP). Analysis of 542 patients revealed that the Lap MWR group had larger tumors, longer operative times, greater blood loss, longer hospital stays, and higher costs, but a lower incidence of adverse postoperative reactions and a simpler medication regimen. Outcomes were location-dependent. For antral EGT-MP, Lap MWR did not prolong surgery or hospitalization and simplified postoperative management. For fundic EGT-MP, digestive endoscopic super minimally invasive surgery provided shorter operative times, less blood loss, and lower costs.
- Citation: Wang LH, Wu X, Sun YT, Liu PF, Wang JF, Yuan YQ, Li HK, Linghu EQ, Chen QQ. Endoscopic super-minimally invasive vs laparoscopic wedge resection for muscularis-propria-originating extraluminal gastric tumors: Efficacy and selection. World J Gastrointest Surg 2026; 18(4): 116371
- URL: https://www.wjgnet.com/1948-9366/full/v18/i4/116371.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i4.116371
Gastric tumors originating from the muscularis propria layer represent a type of gastric submucosal tumor with potential for malignancy, primarily including gastrointestinal stromal tumors (GISTs), leiomyomas, and schwannomas. These tumors predominantly originate from the gastric muscularis propria, grow intermuscularly, and can protrude toward the submucosal layer or approach the thin serosal layer. Additionally, the tumor may be predominantly external in some cases, with only a small portion of tissue or a thin pedicle connected to the gastric wall[1]. Surgical resection is the traditional method for treating gastric tumors originating from the muscularis propria layer. While effective, this approach compromises the gastrointestinal tract’s integrity. The use of digestive endoscopic super minimally invasive surgery (DESMIS) provides a more advantageous therapeutic approach for localized and low-malignancy lesions[2]. With advances in endoscopic resection and suturing techniques, the tumor location, size, and pattern of protrusion are no longer relative contraindications for endoscopic resection therapy[3-5]. The application of endoscopic full-thickness resection (EFR) and submucosal tunneling endoscopic resection (STER) has further expanded the indications for the sole endoscopic treatment of extraluminal-growing gastric tumors originating from the muscularis propria layer (EGT-MP)[5-7]. Recently, various organ-preserving surgeries have been used for treating EGT-MP, primarily including DESMIS and laparoscopic minimally invasive wedge resection (Lap MWR)[8]. However, the efficacy evaluations of these procedures vary, and preoperatively selecting the optimal surgical method is challenging due to the absence of objective indicators. Therefore, this study aimed to target observable preoperative indicators and compare the therapeutic outcomes of DESMIS and Lap MWR for treating EGT-MP, aiming to guide the optimal treatment plan selection for such tumors at different locations.
Research methods: Patients with EGT-MP who underwent DESMIS and Lap MWR at the First Medical Center of Chinese PLA General Hospital between January 2019 and December 2023 were retrospectively analyzed. The inclusion criteria were as follows: (1) Preoperative endoscopic ultrasound gastroscopy identified the lesion as originating from the muscularis propria with extraluminal growth; (2) Patients who received DESMIS and Lap MWR; (3) Single lesion; and (4) No lymph node metastasis confirmed by computed tomography. In contrast, the exclusion criteria were: (1) Evidence of lymph node metastasis or invasion; (2) Treatment with open surgery or subtotal gastrectomy; and (3) Missing infor
Surgical method: In this retrospective cohort, treatment was allocated based on the referring specialty clinic. Patients from General Surgery underwent Lap MWR, whereas those from gastroenterology underwent endoscopic resection, primarily STER for cardiac lesions. Final technical decisions were at the surgeon’s discretion.
DESMIS - EFR: This technique involves endoscopically determining the lesion boundary, marking it, performing submucosal injection to sufficiently elevate the lesion, resecting the entire local layer using an electric knife, retrieving the lesion, and managing the wound surface. Patients who underwent this procedure are categorized as the EFR group.
DESMIS - STER: This involves endoscopically establishing a submucosal tunnel and resecting the entire tumor in the muscularis propria layer within the tunnel. STER is a sub-technique of digestive endoscopic tunnel technology applied to muscularis propria lesions. The operational steps include establishing a tunnel opening, creating a submucosal tunnel, resecting the tumor, treating the wound surface, and sealing the tunnel opening. Patients who underwent this type of surgery are designated as the STER group.
Three primary endoscopic suturing techniques were utilized for defect closure following resection: (1) Pre-cutting and simultaneous closure: This technique employs a dual-knife pre-cutting method to define the resection margin, followed by immediate closure using a through-the-scope clip or similar device during the same procedural sequence; (2) Endoscopic metal clip closure: Post-resection defects were approximated using through-the-scope or over-the-scope metal clips. The choice between through-the-scope systems and over-the-scope clip systems was based on the defect size and location, aiming for secure, watertight apposition of the mucosal and submucosal layers; and (3) Endoscopic purse-string suture: For larger or irregular defects, a purse-string suture technique was applied. This involved placing a nylon loop around the periphery of the defect using multiple anchoring clips. Subsequent tightening of the loop achieved concentric closure of the tissue edges. All endoscopic clips and suture materials used in this study were commercially available products manufactured by Nanjing Micro-Tech Medical Technology Co., Ltd. (Nanjing, China).
Lap MWR: This procedure involves laparoscopic local tumor resection of solitary gastric masses, with or without robotic assistance, excluding proximal or distal subtotal gastrectomy.
Routine fasting, fluid restriction, fluid resuscitation, and intravenous infusion or bolus administration of proton pump inhibitors (PPIs) were performed postoperatively. Gastrointestinal decompression and anti-infective treatments are also administered depending on the condition. Patients who underwent this type of surgery are designated as the laparoscopic minimally invasive surgery (LMIS) group.
Fever: Temperature of > 38°C.
Abdominal pain: Abdominal pain conditions that require intervention.
Tumor size: Tumor size was determined by the longest diameter of the excised specimen.
The following data were collected from patients treated with DESMIS or Lap MWR for EGT-MP: (1) Basic information: Age; sex; smoking history; alcohol consumption; history of cardiovascular disease, diabetes, and fatty liver disease; and use of anticoagulant or antiplatelet medications; (2) Tumor characteristics (EGT-MP): Tumor location, long-axis diameter, and pathological type. The malignant potential of all GISTs was assessed postoperatively according to the modified National Institutes of Health risk stratification criteria, which classify tumors as very low, low, intermediate, or high risk based on tumor size, mitotic index, and primary site]; (3) Surgical indicators: Surgical approach, operative time, intraoperative blood loss, and intraoperative complications, including tumor or tissue clip detachment into the abdominal cavity and perforation; (4) Health economics indicators: Surgical costs and length of hospital stay. For international comparison, all costs, originally recorded in Chinese Yuan, were converted to United States dollars using the mean monthly exchange rate between January 2019 and December 2023 (1 United States Dollars = 6.88 Chinese Yuan), based on official data from the State Administration of Foreign Exchange; (5) Therapeutic outcome indicators: Complete resection and postoperative adverse events, including abdominal pain, infection, and bleeding; (6) Postoperative medical management indicators: Duration of gastric tube decompression, mode (intravenous or pump infusion) and duration of PPI therapy, type of anti-infective treatment (first- or second-line monotherapy, combination therapy with metronidazole for anaerobic coverage, or escalation to higher-level antimicrobials such as meropenem, imipenem-cilastatin, or vancomycin), duration of anti-infective therapy, and time to resumption of oral intake; and (7) Postoperative laboratory indicators: Hemoglobin (Hb) levels and changes from preoperative values, infection markers, including white blood cell (WBC) count and C-reactive protein (CRP)], alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels, along with changes relative to preoperative values.
Data analysis was conducted using IBM SPSS Statistics for Windows, version 26.0 (IBM Corp., Armonk, NY, United States). Continuous data were first tested for normality using the Shapiro-Wilk test (n < 100) or the Kolmogorov-Smirnov test (n > 100). In the Kolmogorov-Smirnov test, the statistic D measures the maximum vertical distance between the empirical and theoretical normal cumulative distribution functions. For the Shapiro-Wilk test, the statistic W assesses goodness-of-fit to a normal distribution. In both tests, the corresponding P value is derived from the calculated statistic, and P > 0.05 indicates no significant deviation from normality. Normally distributed continuous variables are expressed as mean ± SD, and differences were assessed using the independent samples t-test. Non-normally distributed continuous variables are expressed as M (Q1, Q3), and the Mann-Whitney U or Kruskal-Wallis H test was used for group comparisons (The statistic is denoted as U or H). The Bonferroni post hoc test was applied for pairwise comparisons to control for type I error inflation due to multiple testing. Categorical variables are expressed as n (%), and differences were analyzed using the χ2 or Fisher’s exact test. To identify the independent factors associated with the primary continuous outcome, multiple linear regression analysis was performed. All variables were entered into the model simultaneously based on clinical relevance. The results are reported as unstandardized regression coefficients (B) with their 95% confidence intervals. The coefficient of determination (R2) was calculated to assess the model’s goodness-of-fit. The assumptions of linearity, independence, homoscedasticity, and normality of residuals were examined graphically. P < 0.05 was considered statistically significant.
Basic patient characteristics: A total of 542 patients were included in the study, with 98 (42 males and 56 females, mean age: 57.7 years), 99 (57 males and 42 females, mean age: 48.8 years), and 345 (180 males and 165 females, mean age: 57.8 years) in the EFR, STER, and LMIS groups, respectively. Notably, 19 cases in the LMIS group underwent robot-assisted surgery. Table 1 presents the basic patient characteristics across the three groups.
| EFR group (n = 98) | STER group (n = 99) | LMIS group (n = 345) | F/χ2 value | P value | |
| Age | 57.7 ± 9.5 | 48.8 ± 11.2 | 57.8 ± 12.1 | 24.72 | < 0.001 |
| Male | 42 (42.9) | 57 (57.6) | 180 (52.2) | 4.46 | 0.108 |
| Smoking history | 15(15.3) | 29 (29.3) | 90 (26.1) | 6.13 | 0.047 |
| Alcohol consumption history | 22 (22.4) | 34 (34.3) | 85 (24.6) | 4.56 | 0.103 |
| Cardiovascular disease | 38 (38.8) | 25 (25.3) | 119 (34.5) | 4.40 | 0.111 |
| Diabetes | 14 (14.3) | 6 (6.1) | 37 (10.7) | 3.58 | 0.167 |
| Fatty liver disease | 5 (5.1) | 15 (15.2) | 2 (0.6) | 35.78 | < 0.001 |
| Anticoagulants used and discontinued for > 7 days | 6 (6.1) | 3 (3.0) | 24 (7.1) | 2.16 | 0.340 |
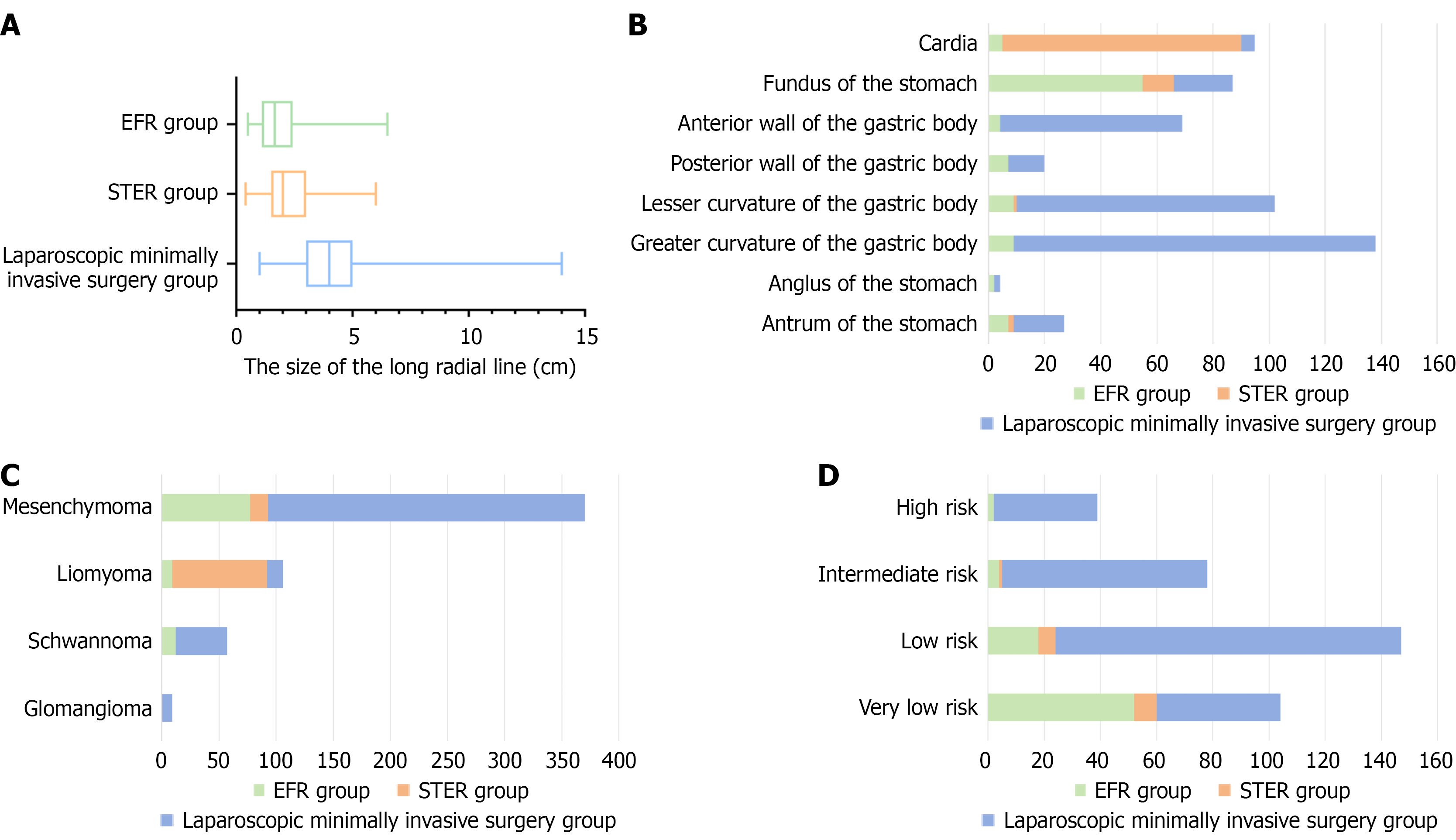
General situation of EGT-MP: The long-axis diameter of EGT-MP in the three groups followed a non-normal distribution (EFR: D = 0.173, P < 0.001; STER: D = 0.159, P < 0.001; LMIS: W = 0.925, P < 0.001). The long-axis diameter size of EGT-MP was compared among the three groups using the Kruskal-Wallis H test, and their differences were significant (H = 152.37, P < 0.001). Additionally, subsequent Bonferroni post-hoc pairwise comparisons indicated that the long-axis diameter of EGT-MP in the LMIS group was significantly greater than that in both the EFR group (adjusted P < 0.001) and the STER group (adjusted P < 0.001, Figure 1A). There was no statistically significant difference between the EFR groups and STER groups (adjusted P = 0.097). Figure 1B-D depicts the location, pathological classification of tumors, and National Institutes of Health risk category of GIST in the three groups, respectively.
Comparative analysis of surgical outcomes, health economics indicators, efficacy evaluation indicators, and post
| EFR group (n = 98) | STER group (n = 99) | LMIS group (n = 345) | H/χ2 value | P value | |
| Surgical indicators | |||||
| Operative times [M (Q1, Q3), minutes] | 44.5 (27.0, 78.0) | 59.5 (33.0, 91.0) | 87.0 (60.0, 115.0) | 73.33 | < 0.001 |
| Blood loss [M (Q1, Q3), mL] | 5.0 (3.0, 8.0) | 3.0 (2.0, 5.0) | 20.0 (10.0, 50.0) | 251.67 | < 0.001 |
| Health economics indicators | |||||
| Hospital stay [M (Q1, Q3), days] | 8.0 (7.0, 9.0) | 8.0 (7.0, 9.0) | 9.0 (7.0, 11.0) | 6.53 | 0.038 |
| Surgical costs [M (Q1, Q3), CNY] | 16753.4 (12914.0, 24603.0) | 13963.0 (9477.1, 16944.6) | 25127.8 (19532.8, 32571.5) | 139.62 | < 0.001 |
| Surgical costs [M (Q1, Q3), USD] | 2435.1 (1877.0, 3576.0) | 2029.5 (1377.4, 2462.9) | 3652.2 (2839.1, 4734.2) | 139.62 | < 0.001 |
| Efficacy evaluation indicators | |||||
| Overall postoperative adverse event incidence, n (%) | 34 (34.7) | 49 (49.5) | 2 (0.6) | 171.89 | < 0.001 |
| Postoperative abdominal pain incidence, n (%) | 13 (13.3) | 26 (26.3) | 1 (0.3) | 81.98 | < 0.001 |
| Postoperative fever incidence, n (%) | 28 (28.6) | 42 (42.4) | 0 (0.0) | 149.17 | < 0.001 |
| Postoperative hematemesis or melena incidence, n (%) | 2 (2.0) | 2 (2.0) | 0 (0.0) | 8.15 | 0.017 |
| Postoperative medical management | |||||
| Duration of nasogastric tube placement [M (Q1, Q3), days] | 0.0 (0.0, 4.0) | 0.0 (0.0, 0.0) | 1.0 (1.0, 3.0) | 89.92 | < 0.001 |
| Method of PPI administration, n (%) | 117.26 | < 0.001 | |||
| No | 1 (1.0) | 0 (0.0) | 10 (2.9) | ||
| Intravenous drip | 66 (67.3) | 81 (81.8) | 335 (97.1) | ||
| Intravenous pumping | 31 (31.6) | 18 (18.2) | 0 (0) | ||
| PPI use duration [M (Q1, Q3), days] | 6.0 (5.0, 7.0) | 6.0 (5.0, 7.0) | 4.0 (3.0, 5.0) | 148.95 | < 0.001 |
| Types of anti-infective agents, n (%) | 374.83 | < 0.001 | |||
| No | 1 (1.0) | 0 (0.0) | 3 (0.9) | ||
| Monotherapy with a first-line or second-line antimicrobial | 33 (33.7) | 18 (18.2) | 332 (96.2) | ||
| Combination therapy with metronidazole for anaerobic coverage | 47 (48.0) | 71 (71.7) | 0 (0.0) | ||
| Escalation to higher-level antimicrobials | 17 (17.3) | 10 (10.1) | 10 (2.9) | ||
| Duration of antimicrobial treatment [M (Q1, Q3), days] | 5.0 (4.0, 6.0) | 5.0 (4.0, 6.0) | 4.0 (3.0, 5.0) | 68.45 | < 0.001 |
| Time to resume oral intake [M (Q1, Q3), days] | 6.0 (5.0, 7.0) | 5.0 (4.0, 6.0) | 2.0 (1.0, 3.0) | 283.10 | < 0.001 |
| Adjusted P value | |||
| EFR group vs STER group | EFR group vs LMIS group | STER group vs LMIS group | |
| Surgical indicators | |||
| Operative times | 0.120 | < 0.001 | < 0.001 |
| Blood loss | 0.428 | < 0.001 | < 0.001 |
| Health economics indicators | |||
| Hospital stay | 0.355 | 1.000 | 0.032 |
| Surgical costs | < 0.001 | < 0.001 | < 0.001 |
| Postoperative medical management | |||
| Duration of nasogastric tube placement | < 0.001 | 0.547 | < 0.001 |
| PPI use duration | 0.306 | < 0.001 | < 0.001 |
| Duration of antimicrobial treatment | 0.219 | < 0.001 | < 0.001 |
| Time to resume oral intake | 0.307 | < 0.001 | < 0.001 |
Multiple linear regression analysis: Multivariable linear regression analyses were performed to identify independent predictors of the four primary outcomes (operative time, intraoperative blood loss, postoperative hospital stay, and total surgical costs), with adjustments made for age, alcohol consumption history, fatty liver disease, and the long-axis diameter of the tumor. The surgical approach (EFR, STER, or LMIS) served as the primary predictor of interest. The overall models were statistically significant for all outcomes (all P < 0.05). As summarized in Table 4, the endoscopic approaches (EFR and STER) were consistently associated with superior perioperative outcomes compared with LMIS. Specifically, both EFR and STER were independent predictors of significantly lower surgical costs. EFR was additionally associated with shorter operative times. STER was additionally associated with reduced blood loss and shorter hospital stay. Among the covariates, a larger tumor diameter was consistently associated with increased operative time, greater blood loss, and higher costs.
| Outcome | Model fit | Surgical approach (vs LMIS) | Significant covariates | ||||||||
| R2 | F | P value | B | 95%CI | P value | B | 95%CI | P value | |||
| Operative time | 0.22 | 25.39 | < 0.001 | EFR | -11.13 | -21.46 to -0.80 | 0.035 | Long-axis diameter of the tumor | 9.45 | 7.52-11.38 | < 0.001 |
| Blood loss | 0.17 | 18.41 | < 0.001 | STER | -12.14 | -22.11 to -2.18 | 0.017 | Tumor size | 6.25 | 4.45-8.05 | < 0.001 |
| Alcohol history | 9.57 | 1.87-17.18 | 0.015 | ||||||||
| Hospital stay | 0.02 | 2.17 | 0.045 | STER | -1.39 | -2.30 to -0.47 | 0.003 | None other | |||
| Surgical costs | 0.28 | 28.58 | < 0.001 | EFR | -6478.55 | -9021.72 to | < 0.001 | Long-axis diameter of the tumor | 106.52 | 25.92-187.12 | 0.010 |
| STER | -11616.91 | -14231.82 to | < 0.001 | Age | 725.34 | 219.70-1230.98 | 0.005 | ||||
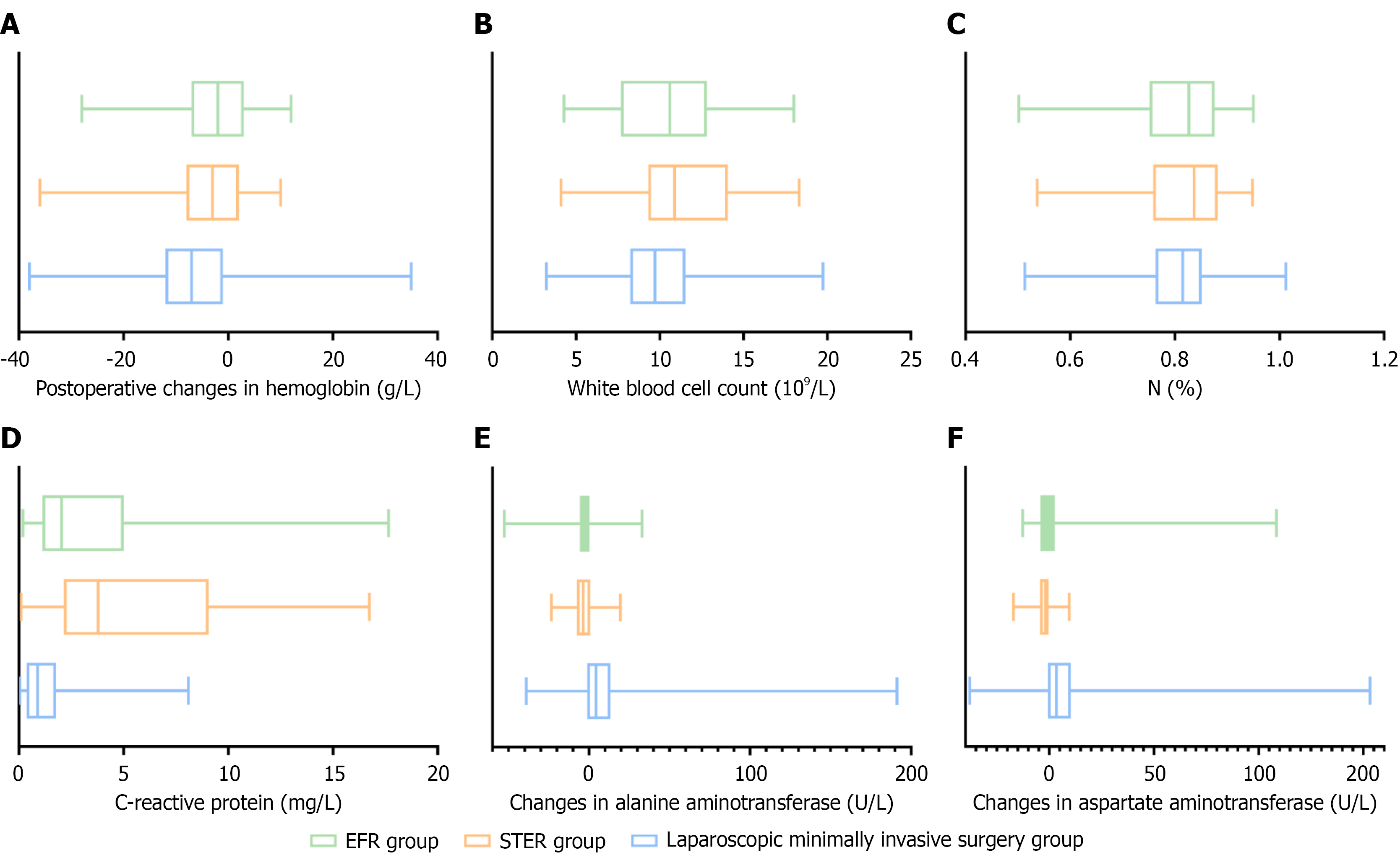
Comparative analysis of postoperative laboratory tests: Overall, 457 patients underwent postoperative routine blood tests, with 60, 52, and 345 in the EFR, STER, and LMIS groups, respectively. The changes in Hb levels from preoperative values, postoperative WBC, and postoperative CRP were compared among the three groups using the Kruskal-Wallis H test, and the differences were significant (H = 13.71, H = 10.50, and H = 80.86, with P = 0.001, P = 0.005, and P < 0.001, respectively). The difference in postoperative neutrophil levels was not statistically significant among the groups. Patients who underwent Lap MWR had a greater decrease in Hb levels from preoperative values (Figure 2) and lower postope
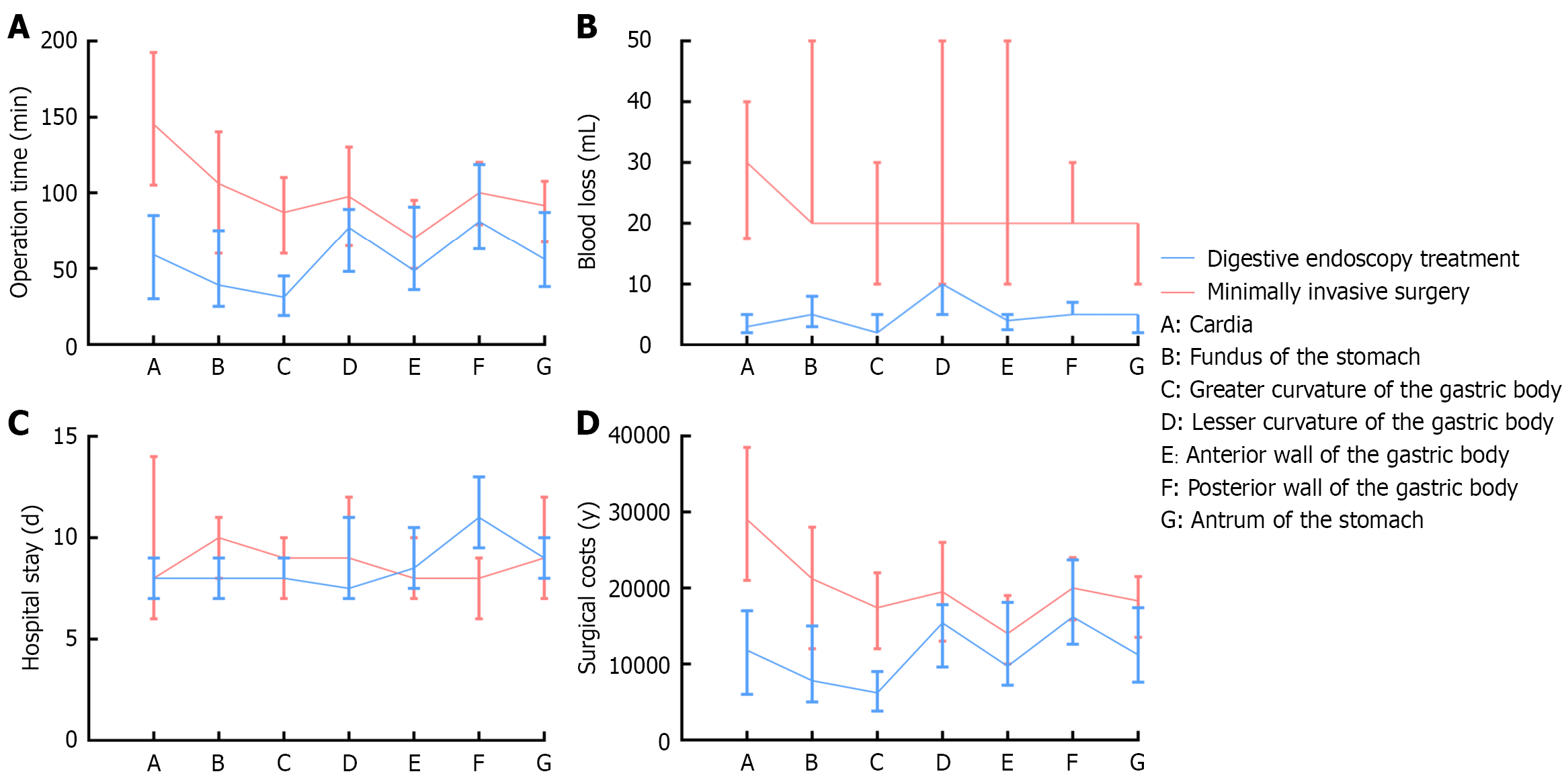
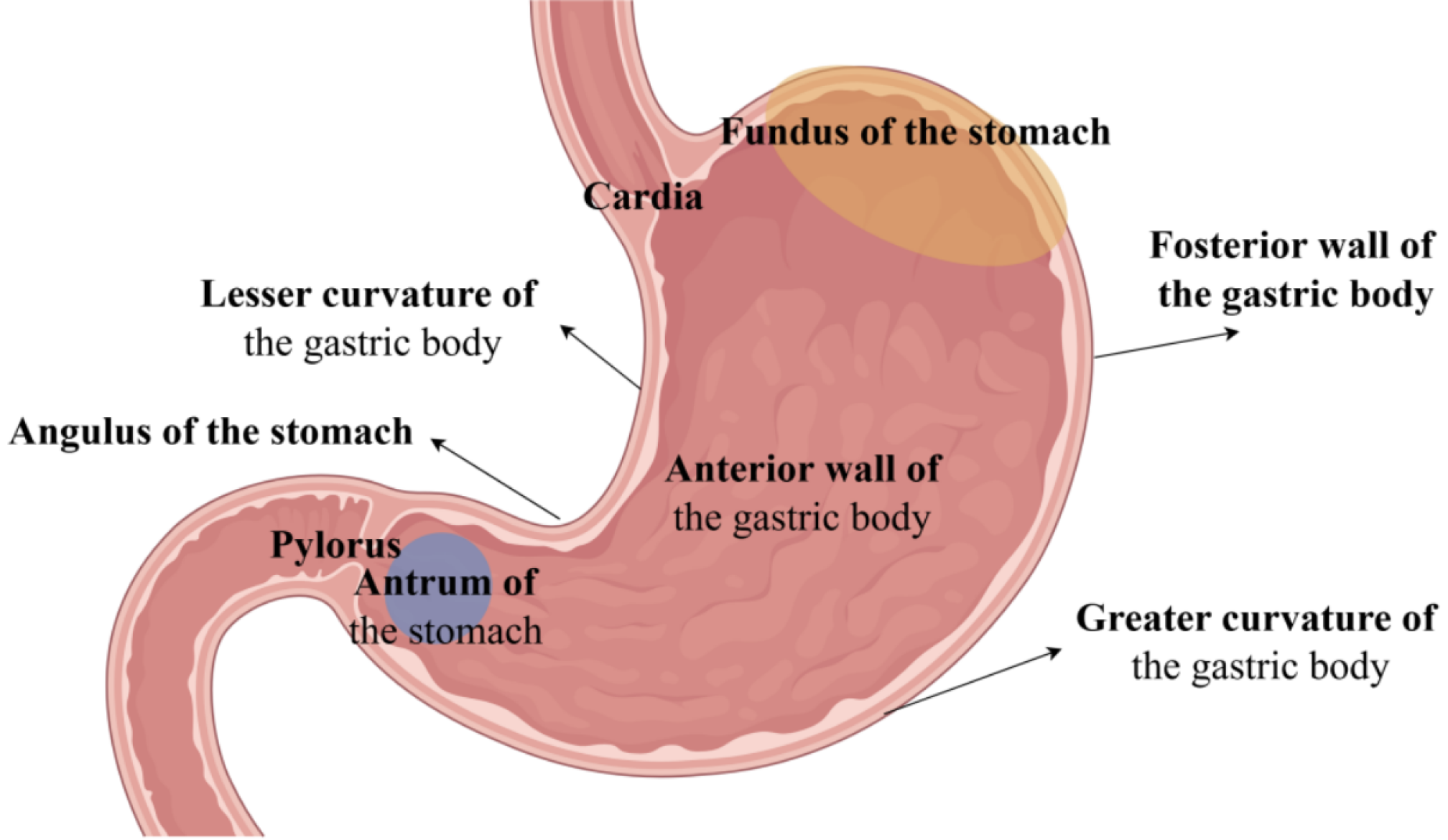
Comparative analysis of surgical and health economics indicators for EGT-MP at different locations: The surgical procedures for EGT-MP, specifically those located at the cardia, fundus of the stomach, and greater curvature of the gastric body, had shorter operative times when treated with DESMIS than with Lap MWR (U = 395.5, U = 1088.5, and U = 1003.0, with P = 0.004, P < 0.001, and P < 0.001, respectively, indicating significant differences). However, the differences for other locations were not statistically significant (Figure 3A). DESMIS for treating EGT-MP of the cardia, fundus of the stomach, greater curvature of the gastric body, lesser curvature of the gastric body, anterior wall of the gastric body, posterior wall of the gastric body, and antrum of the stomach resulted in less blood loss than Lap MWR (U = 413.0, U = 1254.5, U = 1081.0, U = 759.0, U = 240.0, U = 77.5, and U = 140.5, with P = 0.001, P < 0.001, P < 0.001, P = 0.001, P = 0.002, P = 0.008, and P = 0.001, respectively, indicating significant differences, Figure 3B). Additionally, DESMIS for treating EGT-MP of the posterior wall of the gastric body resulted in a shorter hospital stay than Lap MWR (U = 13.5, P = 0.008), with no statistically significant differences in hospital stay for other locations (Figure 3C). DESMIS for treating EGT-MP of the fundus of the stomach and greater curvature of the gastric body resulted in lower surgical costs than Lap MWR (U = 752.0 and U = 670.0, with P < 0.001 and P < 0.008, respectively); however, the differences for other locations were not statistically significant (Figure 3D).
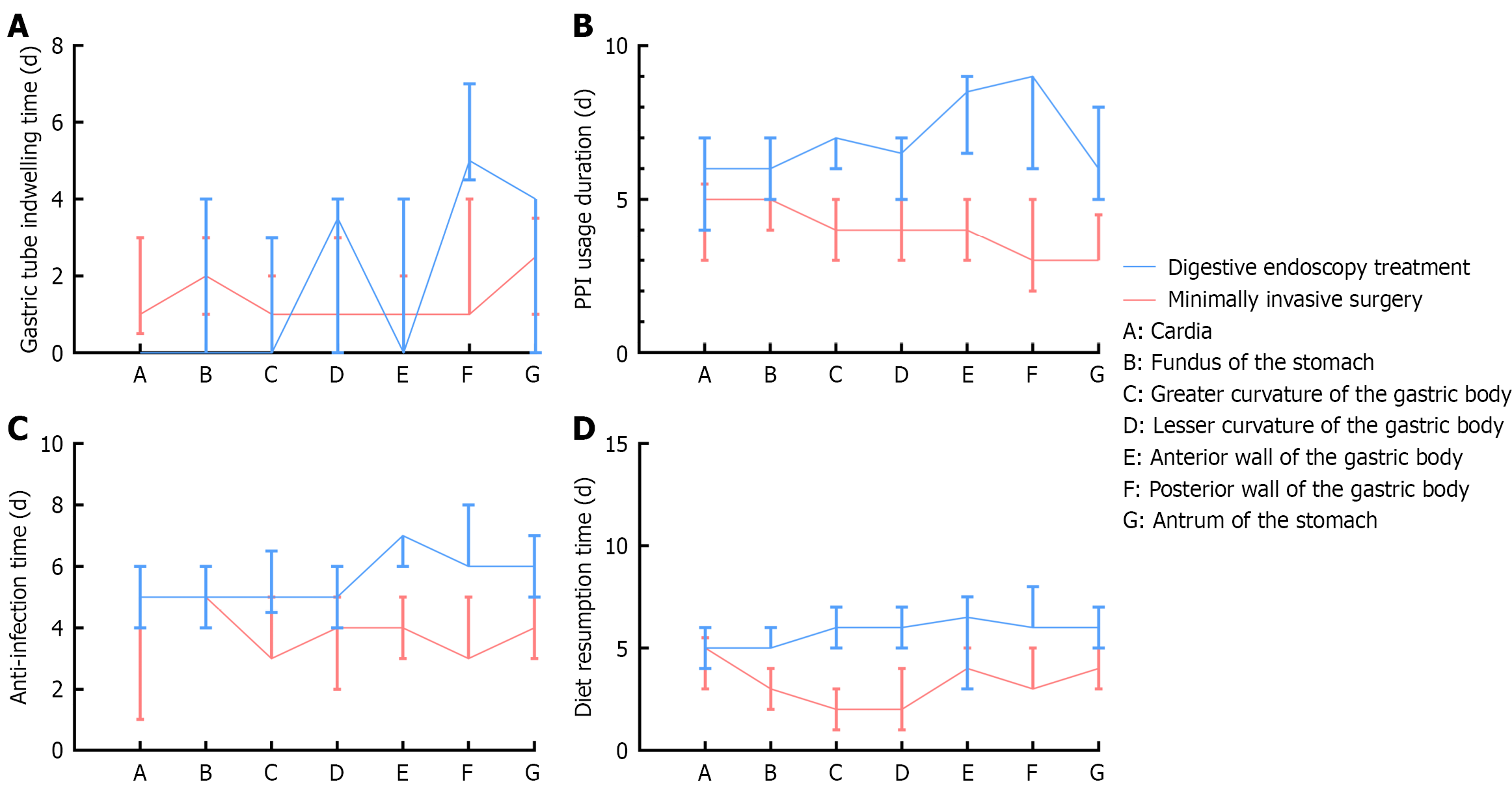
Comparative analysis of postoperative medical management for EGT-MP at different locations: The duration of nasogastric tube placement for gastric cardia tumors originating from the muscularis propria layer was shorter with DESMIS than with Lap MWR (U = 381.0, P < 0.001). Conversely, the duration of nasogastric tube placement for gastric cardia tumors originating from the muscularis propria layer of the posterior wall of the gastric body was longer with DESMIS than with Lap MWR (U = 16.0, P = 0.019). The differences were not statistically significant for other locations (Figure 4A). DESMIS for treating EGT-MP of the fundus of the stomach, greater curvature of the gastric body, lesser curvature of the gastric body, anterior wall of the gastric body, posterior wall of the gastric body, and antrum of the stomach resulted in a longer duration of PPI use than Lap MWR (U = 243.5, U = 162.0, U = 203.0, U = 20.5, U = 9.0, and U = 27.5, with P < 0.001, P < 0.001, P = 0.003, P = 0.002, P = 0.002, and P = 0.004, respectively, Figure 3B), with more frequent use of PPI infusion therapy (χ2 = 15.16, χ2 = 11.59, χ2 = 15.76, χ2 = 10.89, and χ2 = 7.97, with P = 0.001, P = 0.003, P < 0.001, P = 0.003, P = 0.004, respectively). The differences in the duration of PPI use for EGT-MP of the cardia were not statistically significant (Figure 4B), nor was the mode of PPI administration (P = 1.000). DESMIS for treating EGT-MP of the greater curvature of the gastric body, posterior wall of the gastric body, and antrum of the stomach resulted in a longer duration of antimicrobial therapy than Lap MWR (U = 244.5, U = 13.5, and U = 25.0, with P = 0.003, P = 0.008, and P = 0.003, respectively), although the differences for other locations were not statistically significant (Figure 4C). Moreover, DESMIS for treating EGT-MP at all locations had a higher proportion of patients being escalated to higher-level antibiotics (χ2 = 15.09, χ2 = 26.09, χ2 = 26.32, χ2 = 34.70, χ2 = 14.70, χ2 = 15.03, and χ2 = 21.37, with P = 0.001, P < 0.001, P < 0.001, P < 0.001, P = 0.001, P = 0.001, and P < 0.001, respectively). For EGT-MP of the fundus of the stomach, greater curvature of the gastric body, lesser curvature of the gastric body, anterior wall of the gastric body, posterior wall of the gastric body, and antrum of the stomach, DESMIS resulted in a longer time to resume oral intake than Lap MWR (U = 114.5, U = 66.5, U = 64.5, U = 7.5, U = 2.0, and U = 4.5, respectively, all P < 0.001). However, the difference in time to resume oral intake for EGT-MP of the cardia was not statistically significant (Figure 4D; represents Fisher’s exact test).
Recently, the concept of super minimally invasive surgery has been increasingly refined and well-established. Studies have shown that super minimally invasive surgery not only yields better outcomes but also lowers the incidence of postoperative adverse events, decreases patient trauma, and contributes to better long-term prognosis and patients’ quality of life[9]. Endoscopic resection for small EGT-MP, which has the advantages of minimal surgical trauma, rapid postoperative recovery, and low recurrence rates, can be considered when endoscopic technology is permissible, and patients opt for radical treatment[10-12]. It can be used for tissue diagnosis and may achieve a possible clinical cure[13]. Our study showed that DESMIS achieved surgical outcomes comparable to those of Lap MWR. No instances of tumor or tissue clip detachment into the abdominal cavity or intraoperative perforations occurred during the procedures. Postoperative pathology results showed clear resection margins, indicating that complete resection was achieved in all cases, with no recurrences during the 1-year follow-up. Notably, this result demonstrates the good safety and efficacy of DESMIS[14,15]. DESMIS can reduce operative time and length of hospital stay, decrease blood loss, and lower surgical costs compared to Lap MWR, thereby alleviating the economic burden on patients[16,17]. Studies have confirmed that endoscopic traction-assisted techniques can expand the surgical dissection field of view, reduce operative time, and decrease surgical risks[18]. Notably, the lower incidence of certain postoperative adverse events (e.g., pain, fever, bleeding) observed in the LMIS group may be partially attributable to the inherent technical challenges of achieving secure endoscopic closure of gastric defects, a recognized limitation supported by studies reporting non-negligible technical failure rates with dedicated resection devices[19]. While novel closure systems continue to evolve, the reliability of endoscopic methods for large defects requires further optimization, which can influence recovery profiles[20]. It is also important to note that a potential confounding factor in our study was the differential postoperative pain management protocol. Patients in the LMIS group routinely received patient-controlled analgesia pumps upon returning to the ward. This systematic difference in analgesic administration may have particularly influenced the assessment and reporting of postoperative pain.
However, our findings must be interpreted in the context of potential confounding. Although key variables, such as age and long-axis diameter of the tumor, were statistically adjusted for, their intrinsic link to surgical complexity and patient physiology warrants caution. As evidenced in our multivariate models, these factors exerted a substantial influence on operative time, blood loss, and cost, suggesting they may act not merely as confounders but as central mediating variables in the causal pathway. A specific methodological consideration arises from the model for hospital stay. Despite reaching statistical significance, this model exhibited low explanatory power (R2 = 0.02), indicating that the included variables account for only a minor fraction of the variance in length of stay. This suggests that other unmeasured or non-linear factors likely play a dominant role in determining postoperative hospitalization duration. In addition, the conclusion that DESMIS is associated with lower costs is based solely on an analysis of direct surgical expenses. This finding may not extend to total hospitalization expenditures, which could be influenced by confounding variables, such as increased postoperative antibiotic use or other ancillary costs.
This study found that DESMIS had a lesser impact on postoperative Hb level reduction than Lap MWR, indicating less bodily trauma and earlier patient recovery. A pathophysiological explanation for the transaminase increase in the LMIS group may lie in the effects of pneumoperitoneum. The sustained intra-abdominal pressure can cause compromised hepatic perfusion and transient regional ischemia, which may disrupt hepatocellular metabolism and manifest postoperatively as elevated serum transaminase levels. To ensure a more accurate interpretation of postoperative liver enzyme profiles (ALT and AST), patients with pre-existing fatty liver disease were excluded from this analysis. This exclusion criterion was implemented to mitigate the potential confounding influence of underlying hepatic steatosis on perioperative liver function tests, thereby strengthening the internal validity of the observed findings for the studied cohort. However, the results suggest that the rate of various adverse events is slightly higher post-DESMIS than post-Lap MWR. The higher incidence of abdominal pain and fever is considered to be related to the long-term use of analgesic pumps in patients post-Lap MWR and the weaker suturing effect in DESMIS, respectively. One of the significant factors currently affecting endoscopic surgery development is the limitation of suturing methods. Therefore, improving suturing techniques can make the surgery safer and more efficient[21]. This also leads to a longer duration of PPI acid suppression and antimicrobial therapies post-DESMIS, with a higher proportion of PPI infusion therapy usage and patients requiring escalation to higher-level antibiotic treatment. Consequently, DESMIS for treating EGT-MP has some advantages, not only achieving surgical outcomes comparable to those of Lap MWR but also yielding better health economics benefits.
The observed elevation of systemic inflammatory markers (e.g., CRP and WBC) following DESMIS merits careful interpretation. While the technical constraints of endoscopic defect closure represent a potential contributing factor, the primary driver is likely the physiological systemic inflammatory response to surgical trauma itself[22]. Irrespective of the approach, any surgical intervention induces tissue injury and activates a cascade of pro-inflammatory cytokines (e.g., interleukin-6), leading to measurable increases in CRP and WBC as part of the body’s normal healing process. Therefore, attributing these biochemical changes solely to the limitations of endoscopic suturing remains speculative without direct comparative evidence. The lesion’s protruding portion is thoroughly aspirated during Lap MWR, thereby preventing further damage to the surrounding and basal areas of the lesion. In contrast, DESMIS can damage the gastrointestinal mucosa, leading to the aggregation of inflammatory factors within the tissue, activation of related signaling pathways in the body, and the substantial release of inflammatory factors into the bloodstream, consequently resulting in significantly elevated levels of these inflammatory factors in the patient’s blood[23].
This study has some limitations. It is a single-center retrospective clinical study, which could create some biases. The interpretation of our findings must account for potential sources of bias. Significant systematic differences in key baseline characteristics - notably patient age, tumor long-axis diameter, and the distribution of underlying tumor pathologies (e.g., GIST, leiomyomas, schwannomas) with inherently heterogeneous biological behaviors and metastatic potentials between the surgical groups may have confounded the effect estimates. Furthermore, the substantial imbalance in sample sizes among the three comparison groups could compound these concerns, potentially affecting the stability of the com
The primary objective of this retrospective analysis was to conduct a comparative evaluation of DESMIS and Lap MWR for extraluminal gastric tumors. We aimed to establish evidence-based criteria to guide the optimal selection of surgical approaches. Ultimately, this study seeks to promote a paradigm shift from a department-driven triage model, where treatment is determined by the specialty clinic of initial consultation, to a lesion-driven, personalized therapeutic strategy. The goal is to ensure that each patient receives the most effective and least invasive intervention based on the intrinsic characteristics of their disease rather than the contingencies of their referral pathway. This study innovatively explores EGT-MP at different locations and reveals that Lap MWR and DESMIS appear to be more advantageous for tumors in the antrum and fundus of the stomach, respectively (Figure 5). However, to enhance the safety and efficacy of surgical treatment for EGT-MP by selecting a more appropriate surgical method preoperatively, a more detailed division of tumor locations still requires further validation with a substantial amount of medium-term to long-term statistical data. Consequently, with the advances of modern science and technology, the development of various instruments, and the maturation of endoscopic suturing techniques, DESMIS will become safer and more effective in treating EGT-MP.
The primary objective of this retrospective analysis was to conduct a comparative evaluation of DESMIS and Lap MWR for extraluminal gastric tumors. We aimed to establish evidence-based criteria to guide the optimal selection of surgical approaches. Ultimately, this study seeks to promote a paradigm shift from a department-driven triage model, where treatment is determined by the specialty clinic of initial consultation, to a lesion-driven, personalized therapeutic strategy. The goal is to ensure that each patient receives the most effective and least invasive intervention based on the intrinsic characteristics of their disease rather than the contingencies of their referral pathway. This study innovatively explores EGT-MP at different locations and reveals that Lap MWR and DESMIS are more advantageous for tumors in the antrum and fundus of the stomach, respectively (Figure 5). However, to enhance surgical treatment safety and efficacy for EGT-MP by selecting a more appropriate surgical method preoperatively, a more detailed division of tumor locations still requires further validation with a substantial amount of medium-term to long-term statistical data. Consequently, with the advances of modern science and technology, the development of various instruments, and the maturation of endoscopic suturing techniques, DESMIS will become safer and more effective for treating EGT-MP.
| 1. | Huang LY, Cui J, Liu YX, Wu CR, Yi DL. Endoscopic therapy for gastric stromal tumors originating from the muscularis propria. World J Gastroenterol. 2012;18:3465-3471. [PubMed] [DOI] [Full Text] |
| 2. | Nishida T, Goto O, Raut CP, Yahagi N. Diagnostic and treatment strategy for small gastrointestinal stromal tumors. Cancer. 2016;122:3110-3118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 119] [Article Influence: 11.9] [Reference Citation Analysis (1)] |
| 3. | Chen Y, Wang M, Zhao L, Chen H, Liu L, Wang X, Fan Z. The retrospective comparison between submucosal tunneling endoscopic resection and endoscopic submucosal excavation for managing esophageal submucosal tumors originating from the muscularis propria layer. Surg Endosc. 2020;34:417-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (3)] |
| 4. | Abdallah M, Suryawanshi G, McDonald N, Chandan S, Umar S, Azeem N, Bilal M. Endoscopic full-thickness resection for upper gastrointestinal tract lesions: a systematic review and meta-analysis. Surg Endosc. 2023;37:3293-3305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 5. | Shichijo S, Uedo N, Sawada A, Hirasawa K, Takeuchi H, Abe N, Miyaoka M, Yao K, Dobashi A, Sumiyama K, Ishida T, Morita Y, Ono H. Endoscopic full-thickness resection for gastric submucosal tumors: Japanese multicenter prospective study. Dig Endosc. 2024;36:811-821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 6. | Du C, Linghu E. Submucosal Tunneling Endoscopic Resection for the Treatment of Gastrointestinal Submucosal Tumors Originating from the Muscularis Propria Layer. J Gastrointest Surg. 2017;21:2100-2109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (2)] |
| 7. | Du Z, Ding W, Chen T. Suitability and efficacy of submucosal tunneling endoscopic resection for the treatment of giant leiomyoma in the middle and lower esophagus. Dis Esophagus. 2019;32:doz059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (4)] |
| 8. | Xiu H, Zhao CY, Liu FG, Sun XG, Sun H, Liu XS. Comparing about three types of endoscopic therapy methods for upper gastrointestinal submucosal tumors originating from the muscularis propria layer. Scand J Gastroenterol. 2019;54:1481-1486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 9. | Linghu EQ. New direction for surgery: Super minimally invasive surgery. World J Gastroenterol. 2024;30:1676-1679. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 1] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Schmidt A, Meier B, Caca K. Endoscopic full-thickness resection: Current status. World J Gastroenterol. 2015;21:9273-9285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 53] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (2)] |
| 11. | Cai MY, Martin Carreras-Presas F, Zhou PH. Endoscopic full-thickness resection for gastrointestinal submucosal tumors. Dig Endosc. 2018;30 Suppl 1:17-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 12. | Chen H, Li B, Li L, Vachaparambil CT, Lamm V, Chu Y, Xu M, Cai Q. Current Status of Endoscopic Resection of Gastric Subepithelial Tumors. Am J Gastroenterol. 2019;114:718-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 13. | El Chafic AH, Loren D, Siddiqui A, Mounzer R, Cosgrove N, Kowalski T. Comparison of FNA and fine-needle biopsy for EUS-guided sampling of suspected GI stromal tumors. Gastrointest Endosc. 2017;86:510-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 14. | Ponte Neto FL, de Moura DTH, Sagae VMT, Ribeiro IB, Mancini FC, Boghossian MB, McCarty TR, Miyajima NT, Ide E, Bernardo WM, de Moura EGH. Endoscopic resection of esophageal and gastric submucosal tumors from the muscularis propria layer: submucosal tunneling endoscopic resection versus endoscopic submucosal excavation: A systematic review and meta-analysis. Surg Endosc. 2021;35:6413-6426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 15. | Li DM, Ren LL, Jiang YP. Long-term Outcomes of Endoscopic Resection for Gastric Subepithelial Tumors. Surg Laparosc Endosc Percutan Tech. 2020;30:187-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 16. | Li S, Liang X, Zhang B, Tao X, Deng L. Novel endoscopic management for small gastric submucosal tumors: A single-center experience (with video). Dig Liver Dis. 2021;53:895-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 17. | Zimmer V, Bier B. Loop ligation-assisted endoscopic resection of a gastrointestinal stromal tumor in the gastric fundus. Dig Liver Dis. 2021;53:245-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 18. | Li X, He J, Zhu Z, Xiao Y, Jiang Y, Cheng Q, Hu Y. The value of single-channel endoscopic traction and kiss suture technique in closing wounds caused by endoscopic resection of gastrointestinal muscularis propria tumors. Surg Endosc. 2023;37:7709-7716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 19. | Gibiino G, Binda C, Papparella LG, Spada C, Andrisani G, Di Matteo FM, Gagliardi M, Maurano A, Sferrazza S, Azzolini F, Grande G, de Nucci G, Cesaro P, Aragona G, Cennamo V, Fusaroli P, Staiano T, Soriani P, Campanale M, Di Mitri R, Pugliese F, Anderloni A, Cucchetti A, Repici A, Fabbri C; “through thick and thin” study group. Technical failure during colorectal endoscopic full-thickness resection: the "through thick and thin" study. Endoscopy. 2024;56:831-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Takeshita N, Ho KY. Endoscopic Closure for Full-Thickness Gastrointestinal Defects: Available Applications and Emerging Innovations. Clin Endosc. 2016;49:438-443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Khan S, Ali FS, Ullah S, Huang X, Li H. Advancing endoscopic traction techniques in endoscopic submucosal dissection. Front Oncol. 2022;12:1059636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (13)] |
| 22. | Riedel B, Oughton C, Kehlet H, Dieleman JM. Taming Surgical Inflammation: should steroids be an essential component of microcirculatory care to reduce postoperative complications? ANZ J Surg. 2024;94:2096-2098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 23. | Slaats J, Ten Oever J, van de Veerdonk FL, Netea MG. IL-1β/IL-6/CRP and IL-18/ferritin: Distinct Inflammatory Programs in Infections. PLoS Pathog. 2016;12:e1005973. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 239] [Article Influence: 23.9] [Reference Citation Analysis (0)] |