Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.115427
Revised: December 5, 2025
Accepted: January 6, 2026
Published online: February 27, 2026
Processing time: 133 Days and 6.5 Hours
Colonic perforation is a surgical emergency with high mortality due to rapid progression to septic shock. Early identification of high-risk patients is critical for improving outcomes, yet existing predictive tools are often complex and lack clinical practicality.
To identify the risk factors for postoperative mortality in patients with colonic perforation and develop and validate a predictive model.
A retrospective analysis was conducted on patients who underwent surgery for colonic perforation at the Department of Critical Medicine and General Surgery, Anqing Municipal Hospital, between January 2020 and July 2025. Patients were selected on the basis of inclusion and exclusion criteria and were classified into two groups according to postoperative outcomes: Death and survival. General demographics, laboratory results, and imaging data were collected and compared between the two groups. Univariate analysis was performed initially, followed by multivariate logistic regression analysis for variables with significant differences in the univariate analysis. A predictive model for postoperative mortality was constructed on the basis of the multivariate results. Internal validation was con
A total of 134 patients were included in the study, with 21 patients in the postoperative death group and 113 in the survival group, yielding a mortality rate of 15.6%. Moreover, no significant differences were found between the two groups concerning sex, history of hypertension, diabetes, cerebrovascular sequelae, cardiac history, hae
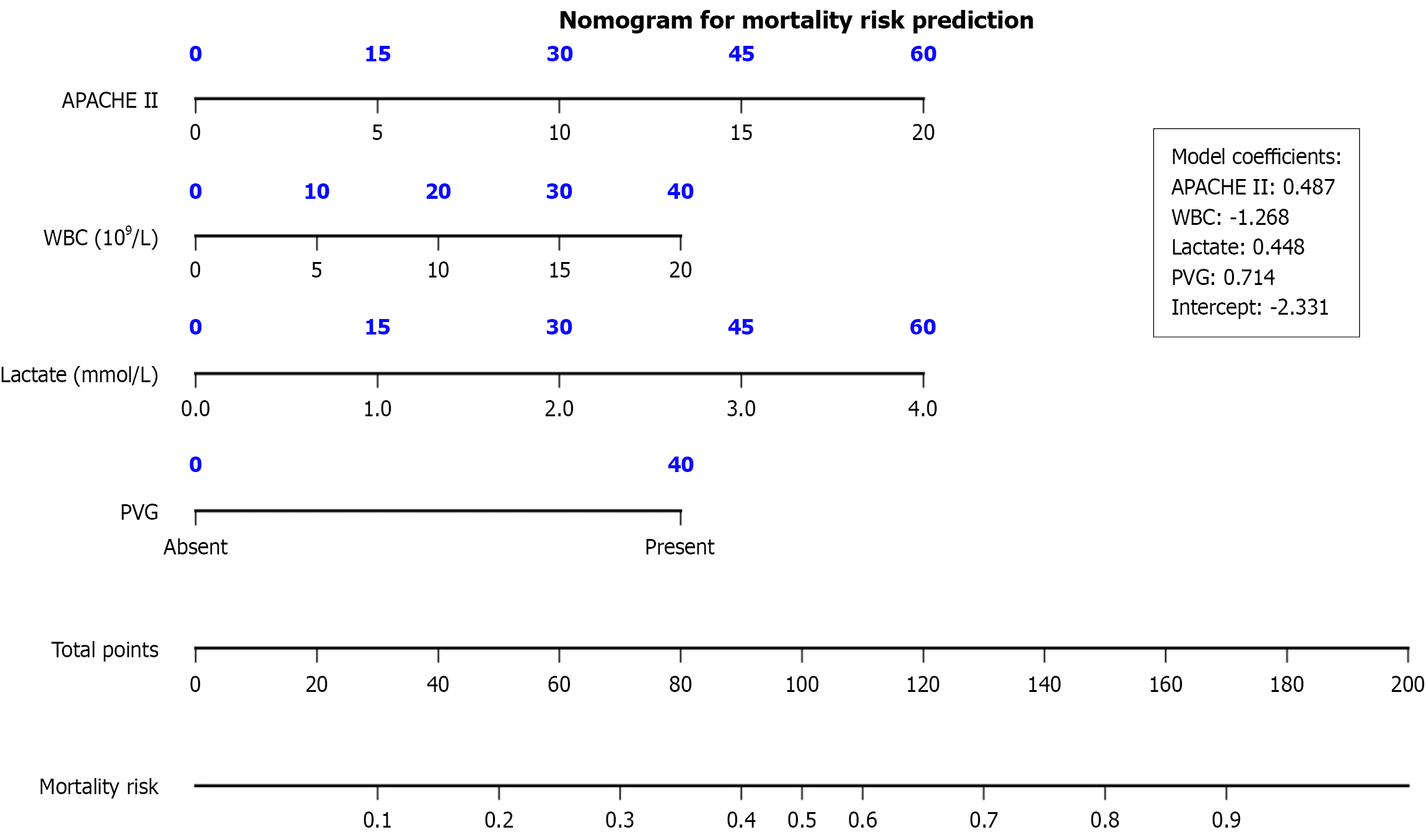
The predictive model for postoperative mortality in patients with colonic perforation, which is based on four indicators (APACHE II score, lactate level, WBC count, and PVG presence), demonstrates strong predictive performance. The dual cut-off pathway (0.020 high-sensitivity screen + 0.121 resource-efficient confirmation) markedly reduced intensive care unit resource use while maintaining high sensitivity (95.2%). This investigation offers a replicable decision-making tool that can be integrated into information systems for future prospective studies.
Core Tip: A nomogram model incorporating the Acute Physiology and Chronic Health Evaluation II score, lactate level, white blood cell count, and presence of portal venous gas was developed and internally validated to predict mortality after colonic perforation surgery (area under the curve = 0.852). The dual-threshold “0.020 high-sensitivity screen + 0.121 resource-efficient confirmation” funnel strategy reduced the need for high-level intervention by 53% in this cohort while maintaining high sensitivity (95.2%). This visual tool offers a practical, system-compatible decision aid for early risk stratification and intensive care unit resource optimization, although external validation in multicentre, prospective studies is needed before clinical implementation.
- Citation: Xu XJ, Zhang HD, Cheng CJ, Zhang YM, Zhang Q. Risk factor analysis and nomogram model construction for mortality in patients following colonic perforation surgery. World J Gastrointest Surg 2026; 18(2): 115427
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/115427.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.115427
Colonic perforation is a surgical condition that is associated with severe complications, and its incidence has been increasing[1]. Typically, colonic perforation manifests as sudden and severe abdominal pain, and subsequent leakage of intestinal contents into the peritoneal cavity leads to rapid development of intra-abdominal infection, which can progress swiftly to septic shock and, in severe cases, result in death[2,3]. The most common aetiologies of colonic perforation include tumour perforation, diverticulitis, chronic constipation, and iatrogenic injury[4]. Colonic perforation is diagnosed through abdominal physical examination and imaging studies, with surgical exploration being the primary treatment option. Surgical approaches include open exploration or laparoscopic assessment, with bowel resection or repair performed on the basis of intraoperative findings, and the creation of a stoma when deemed necessary[5,6]. Therefore, colonic perforation is considered a serious intra-abdominal infectious condition with a high mortality rate, making the identification of risk factors for postoperative death following colonic perforation critical. Timely and effective inter
A retrospective analysis of the medical records of 134 patients with colonic perforation who underwent surgical treatment at the Department of Critical Medicine and the Department of General Surgery at Anqing Municipal Hospital between January 2020 and July 2025 was performed. The inclusion criteria were as follows: (1) Patients with intraoperatively confirmed colonic perforation; (2) Patients older than 18; and (3) Patients with complete clinical data. The exclusion criteria included the following: (1) Patients who did not undergo surgical treatment; (2) Patients with concurrent infections in other organs; (3) Patients with end-stage tumours; (4) Patients who experienced accidental deaths due to other causes; and (5) Patients whose clinical data were incomplete.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Declaration of Helsinki 1964 and later versions. This study was approved by the Ethics Committee of Anqing Municipal Hospital under ethical approval number Medical Ethics Review (2025) No. 173. Informed consent was obtained from all patients enrolled in the study.
Clinical data: Clinical data for patients with colonic perforation were obtained from the medical records database of Anqing Municipal Hospital and included the following: (1) General information: Sex, age, comorbidities (e.g., diabetes mellitus, hypertension, cerebrovascular sequelae, and cardiac diseases), American Society of Anaesthesiologists (ASA) classification, and Acute Physiology and Chronic Health Evaluation II (APACHE II) score; (2) Perioperative laboratory data: Preoperative peripheral blood white blood cell (WBC) count, haemoglobin level, platelet count, albumin level, serum total bilirubin level, serum creatinine level, lactate level, C-reactive protein (CRP) level, and procalcitonin level; (3) Perioperative imaging data: Results of abdominal computed tomography examination [presence of intraperitoneal free gas, intraperitoneal free fluid, and portal venous gas (PVG)]; and (4) Perioperative clinical data: Time from onset to surgery, perforation site, cause of perforation, operation time, and amount of intraoperative blood loss.
Outcome determination: (1) Patients were split into the death and survival groups on the basis of 30-day postoperative survival outcomes; (2) Cardiac diseases included myocardial infarction and chronic heart failure; and (3) Intraperitoneal free fluid was defined as fluid depth exceeding 2 cm in the hepatorenal recess, paracolic gutters, or pelvis.
Missing data handling: The extent of missing data was assessed for all the candidate variables. Variables with a high percentage of missingness (> 15%) were excluded from the analysis. For the remaining variables, the percentage of missing data was very low (< 2% for any individual variable). Given the minimal and sporadic nature of the missing values, a complete-case analysis was deemed the most appropriate method. Consequently, only patients with complete data for all variables included in the final univariate and multivariate analyses were retained in the study cohort.
Statistical analyses were conducted utilizing SPSS 27.0 and R software (version 4.3.1). Continuous variables following a normal distribution are denoted as the mean ± SD, and intergroup comparisons were performed using t tests. For nonnormally distributed continuous variables, data are presented as medians (interquartile ranges), and group com
A total of 134 patients with colonic perforation were included in the study; these patients were divided into a death group (n = 21) and a survival group (n = 113) on the basis of survival outcomes, resulting in a mortality rate of 15.6%. Moreover, no statistically significant differences were detected between the groups in terms of sex, history of hypertension, diabetes, cerebrovascular sequelae, cardiac disease history, haemoglobin level, albumin level, free intraperitoneal gas presence, free intraperitoneal fluid presence, perforation site, cause of perforation, operation time, intraoperative blood loss volume or surgical approach. Statistically significant differences in age, ASA classification, APACHE II score, preoperative peri
| Variable | Mortality group (n = 21) | Survival group (n = 113) | t/χ2/Z | P value |
| Demographics | ||||
| Female | 8 (38.1) | 53 (46.9) | χ2 = 0.53 | 0.46 |
| Age (year) | 82.00 (67-88) | 71.00 (25-89) | Z = 4.27 | < 0.001 |
| Comorbidities | ||||
| Hypertension | 10 (47.6) | 51 (45.1) | χ2 = 0.04 | 0.83 |
| Diabetes | 9 (42.9) | 32 (28.3) | χ2 = 1.77 | 0.18 |
| Cerebrovascular disease | 7 (33.3) | 29 (25.7) | χ2 = 0.52 | 0.47 |
| Heart disease | 5 (23.8) | 29 (25.7) | χ2 = 0.03 | 0.85 |
| Disease severity | ||||
| ASA classification | ||||
| II | 7 (33.3) | 91 (80.5) | ||
| III | 12 (57.1) | 22 (19.5) | χ2 = 20.57 | < 0.001 |
| IV | 2 (9.5) | 0 (0.0) | ||
| APACHE II score | 8.95 ± 2.61 | 10.52 ± 3.80 | t = 2.318 | 0.022 |
| Laboratory findings | ||||
| WBC (× 109/L) | 7.52 (3.33-12.32) | 9.93 (4.63-14.58) | Z = 3.73 | < 0.001 |
| Haemoglobin (g/L) | 104.37 ± 10.63 | 100.24 ± 13.23 | t = 1.38 | 0.169 |
| Platelet (× 109/L) | 83.00 (44-112) | 197.00 (96-354) | Z = 4.68 | < 0.001 |
| Albumin (g/L) | 30.8 ± 4.2 | 30.4 ± 2.3 | t = 1.24 | 0.2176 |
| Total bilirubin (μmol/L) | 18.80 (14.00-28.80) | 14.50 (7.60-28.80) | Z = 3.89 | < 0.001 |
| Serum creatinine (μmol/L) | 128.00 (88-207) | 95.00 (63-180) | Z = 3.88 | < 0.001 |
| Lactate (mmol/L) | 1.90 (0.60-4.00) | 1.70 (0.70-2.80) | Z = 2.07 | 0.038 |
| CRP (mg/L) | 247.00 (159-322) | 156.00 (98-256) | Z = 4.12 | < 0.001 |
| PCT (μg/L) | 45.50 (18.30-69.90) | 15.60 (3.50-35.10) | Z = 4.18 | < 0.001 |
| Imaging findings | ||||
| Pneumoperitoneum | 15 (71.4) | 77 (68.1) | χ2 = 0.09 | 0.76 |
| Ascites | 12 (57.1) | 57 (50.4) | χ2 = 0.32 | 0.57 |
| Portal venous gas | 4 (19.0) | 0 (0.0) | - | < 0.0011 |
| Perforation details | ||||
| Onset to surgery > 24 hours | 12 (57.1) | 36 (31.9) | χ2 = 5.21 | 0.022 |
| Perforation site | ||||
| Ascending colon | 2 (9.5) | 15 (13.3) | ||
| Transverse colon | 1 (4.8) | 3 (2.7) | χ2 = 0.63 | 0.96 |
| Descending colon | 1 (4.8) | 7 (6.2) | ||
| Sigmoid colon | 17 (81.0) | 88 (77.9) | ||
| Perforation aetiology | ||||
| Tumour | 4 (19.0) | 23 (20.4) | ||
| Diverticulum | 6 (28.6) | 30 (26.5) | ||
| Hernia | 3 (14.3) | 17 (15.0) | χ2 = 0.31 | 0.99 |
| Chronic constipation | 2 (9.5) | 14 (12.4) | ||
| Foreign body | 2 (9.5) | 9 (8.0) | ||
| Iatrogenic | 2 (9.5) | 10 (8.8) | ||
| Spontaneous | 2 (9.5) | 10 (8.8) | ||
| Surgical data | ||||
| Operative time (minutes) | 178.57 ± 15.48 | 178.76 ± 18.23 | t = -0.04 | 0.96 |
| Intraoperative blood loss (mL) | 38.57 ± 9.15 | 37.96 ± 9.96 | t = 0.25 | 0.8 |
| Surgical approach | ||||
| Primary anastomosis | 2 (9.5) | 15 (13.3) | ||
| Hartmann's procedure | 17 (81.0) | 88 (77.9) | χ2 = 0.224 | 0.894 |
| Simple repair | 2 (9.5) | 10 (8.8) |
Variables found to be significant in the univariate analysis were incorporated into the multivariate analysis, with collinearity assessed (all VIFs < 5). The results revealed that the APACHE II score, lactate level, and PVG presence were independent risk factors, while an elevated WBC count served as a protective factor (Table 2).
| Variable | β coefficient | SE | χ² | P value | OR (95%CI) | VIF |
| APACHE II | 0.214 | 0.092 | 5.41 | 0.020 | 1.24 (1.03-1.48) | 1.02 |
| WBC | -0.382 | 0.112 | 11.62 | < 0.001 | 0.68 (0.55-0.85) | 1.18 |
| Lactate | 0.876 | 0.298 | 8.64 | 0.003 | 2.40 (1.34-4.31) | 1.04 |
| PVG | 3.012 | 1.214 | 6.15 | 0.013 | 20.32 (1.89-218.45) | 1.05 |
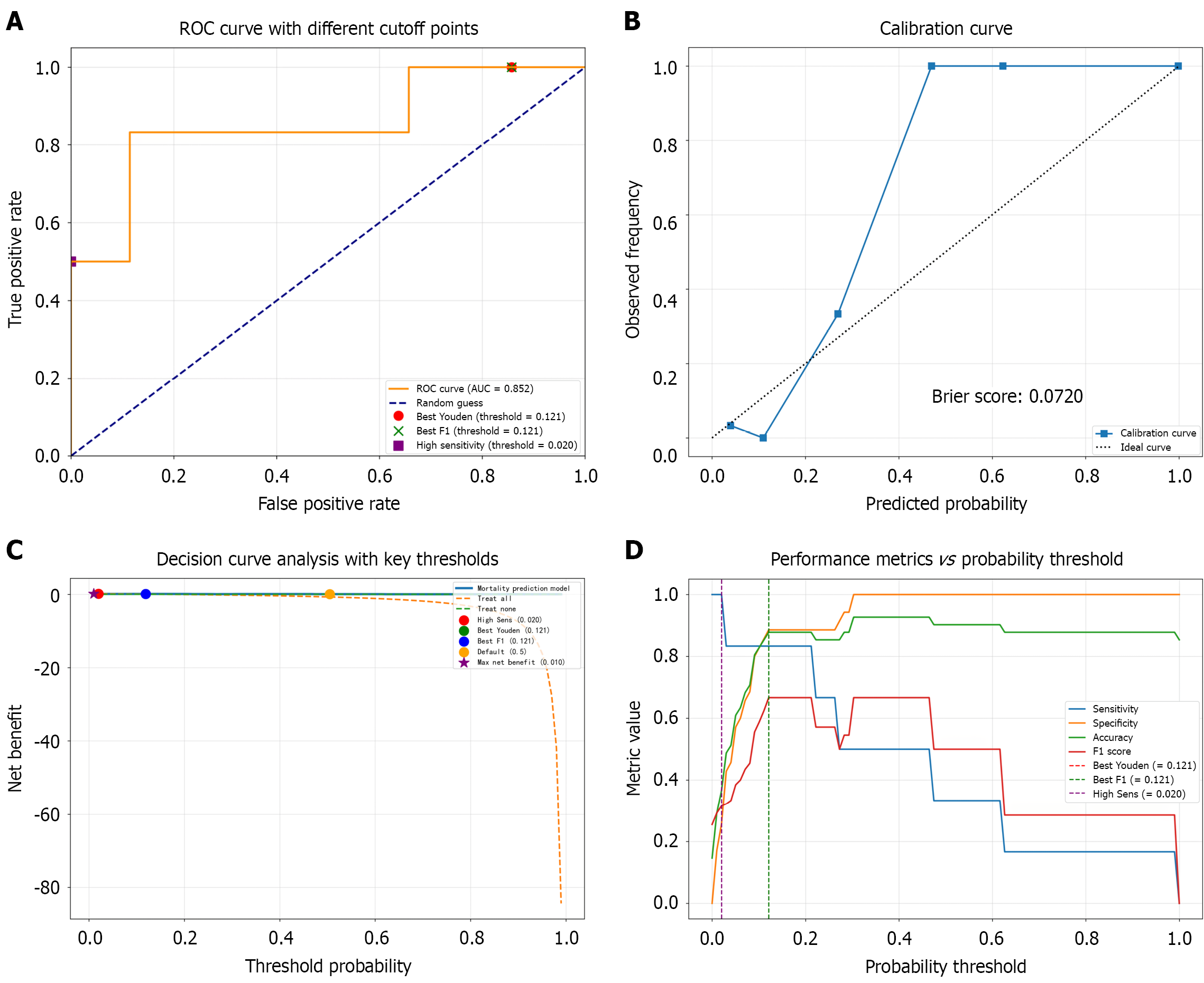
The four independent variables (APACHE II score, lactate level, PVG presence, and WBC count), which were identified via multivariate logistic regression, were incorporated into the R language rms package to develop a visual nomogram (Figure 1). Internal validation was performed using the bootstrap method with 1000 repeated samples. Discrimination: The area under the curve (AUC) value was 0.852 [95% confidence interval (CI): 0.791-0.913] (Figure 2A). Calibration: The calibration curve exhibited a slope close to 1 (0.98) and an intercept near 0 (-0.02). The Brier score was 0.072. To account for overoptimism, bootstrap-corrected calibration estimates were obtained from 1000 resamples: The optimism-corrected slope was 0.95, the intercept was -0.03, and the Brier score was 0.075 (Figure 2B). Clinical utility: DCA demonstrated that within the 1%-40% threshold range, the model’s net benefit consistently surpassed that of both the “treat all” and “treat none” strategies (Figure 2C). Threshold sensitivity analysis revealed that both the Youden index and the F1 score reached their peak values at a threshold of 0.121 (Youden: 0.719; F1: 0.667), while at a threshold of 0.020, the sensitivity approached 100%, with the specificity reduced to 25%, representing the “zero missed diagnosis” entry threshold (Figure 2D).
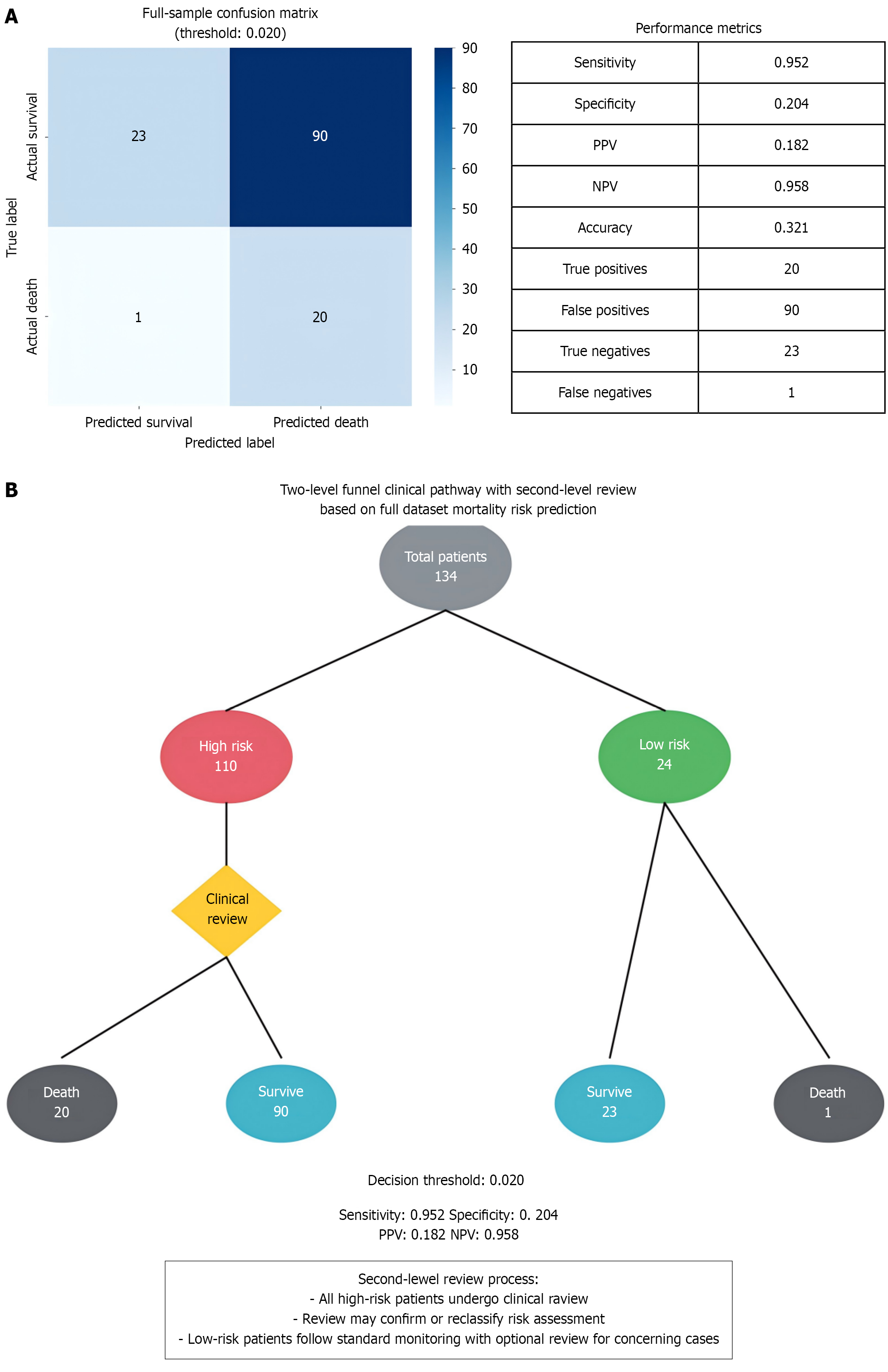
The strategy of employing a “0.020 high-sensitivity red line + 0.121 resource-efficient screening” was implemented. The confusion matrix for the first cut-off (≥ 0.020) revealed a sensitivity of 95.2% (20/21), a specificity of 20.4%, and a negative predictive value of 95.8%. A review of the second cut-off (≥ 0.121) demonstrated a sensitivity of 83.3% and a specificity of 88.6%, representing the optimal balance point (Figure 3A). The full-sample pathway diagram indicated that 92 of 134 cases (68.7%) were flagged as high risk by the first tier. After the second-tier review, only 17 cases continued to require high-level intervention, while 75 cases were downgraded to standard monitoring, resulting in a 53% reduction in intensive care unit admissions (Figure 3B).
The patient’s APACHE II score, WBC count, lactate level, and the presence or absence of a PVG were entered into the corresponding points axis at the top. These values were then summed to determine the total score, which was used to determine the total score axis, allowing for the individualized mortality risk of the patient to be calculated, thereby facilitating real-time bedside assessment.
Acute colonic perforation represents a prevalent surgical condition within the spectrum of acute abdominal disorders. Common aetiological factors include tumour-related factors, diverticulitis, chronic constipation, incarcerated inguinal hernia, and iatrogenic procedures. Neoplastic perforation can result from tumour-induced erosion of the normal bowel wall or from closed-loop obstruction caused by the tumour[9]. Spontaneous colonic perforation refers to perforation occurring in the absence of organic lesions or external trauma, most frequently affecting the antimesenteric border of the sigmoid colon and the rectosigmoid junction. This accounts for approximately 65% of cases, with chronic constipation serving as a primary precipitating factor[1]. Iatrogenic perforations are typically associated with endoscopic procedures. Despite relatively high bowel cleanliness in these patients, the onset is often insidious and frequently overlooked, leading to delays in treatment[10]. While the fluid content of the colonic lumen is relatively low, chemical irritation is less pro
This study revealed that the APACHE II score is the most robust single predictor of patient mortality risk, with an AUC value of 0.8055 when applied independently, thereby demonstrating exceptional discriminative ability. These results align with findings from numerous prior studies, further validating the use of the APACHE II score as the gold standard tool for assessment in critical care medicine[12]. The APACHE II score serves as an objective measure for the comprehensive evaluation of disease severity by integrating factors such as age, acute physiological parameters, and chronic health conditions. Its superior predictive performance derives from its multisystem, multidimensional evaluation framework, which captures the overall pathophysiological status, an aspect often missed by individual physiological indicators. In the multivariate logistic regression model of this study, the APACHE II score exhibited a significant positive predictive value [odds ratio (OR) = 1.24, 95%CI: 1.03-1.48], indicating a corresponding increase in patient mortality risk with higher scores, which is consistent with both its design and clinical relevance. Notably, although the APACHE II score alone demonstrated substantial predictive value, its performance was further enhanced when it was combined with additional indicators such as the lactate level and WBC count (the AUC value increased from 0.8055 to 0.852), empha
Numerous studies have established that the lactate level acts as an independent prognostic predictor of mortality in patients with sepsis[14]. Elevated blood lactate concentrations are considered early indicators of multiple organ failure and have demonstrated superior accuracy over sequential organ failure assessment in predicting sepsis-related mortality[15]. Sepsis-3 guidelines designate serum lactate levels exceeding 2 mmol/L as the primary criterion for the clinical diagnosis of septic shock[16]. Lactate concentrations above 4 mmol/L have been reported to have 96% specificity for predicting in-hospital mortality in nonhypotensive patients[17]. In this study, an elevated lactate level (OR = 2.40; 95%CI: 1.34-4.31) was identified as a significant risk factor for postoperative mortality in patients with colonic perforation, which aligns with findings from previous studies[18].
WBCs are integral components of the human immune system and play pivotal roles in defending against infections and inflammation. Upon infection, cytokines such as granulocyte colony-stimulating factor, interleukin-1, and tumor necrosis factor-α stimulate the proliferation of bone marrow haematopoietic stem cells, increase the release of neutrophils into the bloodstream, and increase peripheral blood WBC counts[19]. However, in the case of severe bacterial infections, endotoxins impair the ability of the bone marrow to release neutrophils, while excessive consumption of WBCs occurs in conjunction with inadequate bone marrow compensation, resulting in reduced WBC counts, which serve as a prognostic marker for poor outcomes[20]. The findings of this study indicated that an elevated WBC count (OR = 0.68, 95%CI: 0.55-0.85) was a protective factor against postoperative mortality in patients with colonic perforation. A lower WBC count was associated with an increased risk of postoperative mortality in these patients. A preoperative reduction in the WBC count reflects a suppressed immune state and poor overall health, indicating impaired immune function, which is correlated with adverse postoperative outcomes.
The theoretical mechanisms underlying the occurrence of PVG include both mechanical and bacterial theories. Intestinal mucosal injury, bowel wall necrosis, or increased intraluminal gas pressure, which results in mucosal tears, allow gas to enter the portal venous system through the bowel wall. The bacterial theory posits that, in the presence of microbial infection, gas produced within the intestine escapes through the intestinal mucosa into the submucosal layer and blood vessels, subsequently entering the portal venous system[21]. Koizumi et al[22] analysed 1590 PVG cases from the Japanese national inpatient database and identified aetiologies such as intestinal ischaemia, gastrointestinal obstruc
The final model developed in this study achieved an AUC value of 0.852 (95%CI: 0.791-0.913) for patients with colonic perforation, markedly outperforming both the APACHE II score alone and traditional lactate cut-off values. This suggests that the integration of APACHE II, WBC, lactate, and PVG data enhances the performance of the model in predicting mortality[25]. The calibration curve exhibited a Brier score of 0.072 and a slope approaching 1, indicating high con
The translation of this dual-cut-off model into clinical practice requires a structured protocol. We propose a two-stage implementation: First, upon patient admission, the nomogram score is calculated. Those with a risk probability ≥ 0.020 are immediately flagged for 'high-risk' status, triggering intensified initial monitoring (e.g., closer vital sign observation, senior surgical review, and point-of-care lactate testing). Second, within a short timeframe (e.g., 2-4 hours), this high-risk group undergoes a comprehensive reassessment. This reassessment incorporates the refined model prediction (using the ≥ 0.121 cut-off) alongside critical clinical judgement, response to initial resuscitation, and resource availability to make the final decision regarding intensive care unit (ICU) admission. This funnel approach leverages the model's speed for initial triage while ensuring that final, high-stakes decisions are augmented by clinical expertise.
This study has several limitations that should be acknowledged. First, the sample size, particularly the number of mortality events (n = 21), is limited. This results in a suboptimal event-per-variable ratio, which increases the risk of model overfitting and may affect the stability of the coefficient estimates. Although we employed bootstrap internal validation and maintained a parsimonious model to mitigate these concerns, the wide CI for PVG presence as a predictive factor of mortality is a direct manifestation of this statistical constraint.
The model was developed and validated using a single-center, retrospective dataset. The lack of external validation in an independent cohort limits definitive conclusions about its generalizability to other populations or healthcare settings. Therefore, future multicenter, prospective studies are essential for external validation prior to any clinical application. Finally, a formal cost-effectiveness analysis of the dual-cutoff pathway, while important for future implementation research, was beyond the scope of this study.
A postoperative mortality prediction model for patients with colonic perforation, developed using four key indicators, APACHE II score, lactate level, WBC count, and PVG presence, demonstrated robust predictive performance. In this cohort, the dual-cut-off pathway, consisting of a “0.020 zero-missed-diagnosis threshold” and a “0.121 resource-efficient screening” cut-off, allowed for a reduction in ICU resource utilization of more than 50%. This approach offers a repli
| 1. | Bielecki K, Kamiński P, Klukowski M. Large bowel perforation: morbidity and mortality. Tech Coloproctol. 2002;6:177-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 80] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 2. | Pisano M, Zorcolo L, Merli C, Cimbanassi S, Poiasina E, Ceresoli M, Agresta F, Allievi N, Bellanova G, Coccolini F, Coy C, Fugazzola P, Martinez CA, Montori G, Paolillo C, Penachim TJ, Pereira B, Reis T, Restivo A, Rezende-Neto J, Sartelli M, Valentino M, Abu-Zidan FM, Ashkenazi I, Bala M, Chiara O, De' Angelis N, Deidda S, De Simone B, Di Saverio S, Finotti E, Kenji I, Moore E, Wexner S, Biffl W, Coimbra R, Guttadauro A, Leppäniemi A, Maier R, Magnone S, Mefire AC, Peitzmann A, Sakakushev B, Sugrue M, Viale P, Weber D, Kashuk J, Fraga GP, Kluger I, Catena F, Ansaloni L. 2017 WSES guidelines on colon and rectal cancer emergencies: obstruction and perforation. World J Emerg Surg. 2018;13:36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 296] [Cited by in RCA: 219] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 3. | Sartelli M, Weber DG, Kluger Y, Ansaloni L, Coccolini F, Abu-Zidan F, Augustin G, Ben-Ishay O, Biffl WL, Bouliaris K, Catena R, Ceresoli M, Chiara O, Chiarugi M, Coimbra R, Cortese F, Cui Y, Damaskos D, De' Angelis GL, Delibegovic S, Demetrashvili Z, De Simone B, Di Marzo F, Di Saverio S, Duane TM, Faro MP, Fraga GP, Gkiokas G, Gomes CA, Hardcastle TC, Hecker A, Karamarkovic A, Kashuk J, Khokha V, Kirkpatrick AW, Kok KYY, Inaba K, Isik A, Labricciosa FM, Latifi R, Leppäniemi A, Litvin A, Mazuski JE, Maier RV, Marwah S, McFarlane M, Moore EE, Moore FA, Negoi I, Pagani L, Rasa K, Rubio-Perez I, Sakakushev B, Sato N, Sganga G, Siquini W, Tarasconi A, Tolonen M, Ulrych J, Zachariah SK, Catena F. 2020 update of the WSES guidelines for the management of acute colonic diverticulitis in the emergency setting. World J Emerg Surg. 2020;15:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 236] [Article Influence: 39.3] [Reference Citation Analysis (0)] |
| 4. | Lee DB, Shin S, Yang CS. Patient outcomes and prognostic factors associated with colonic perforation surgery: a retrospective study. J Yeungnam Med Sci. 2022;39:133-140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 5. | Joo Y, Lee Y, Yoo T, Kim J, Park I, Gwak G, Cho H, Yang K, Kim K, Bae BN. Prognostic Factors and Management for Left Colonic Perforation: Can Hartmann's Procedure Be Preventable? Ann Coloproctol. 2020;36:178-185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Tsuchiya A, Yasunaga H, Tsutsumi Y, Matsui H, Fushimi K. Mortality and Morbidity After Hartmann's Procedure Versus Primary Anastomosis Without a Diverting Stoma for Colorectal Perforation: A Nationwide Observational Study. World J Surg. 2018;42:866-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Kudou K, Kajiwara S, Motomura T, Yukaya T, Nakanoko T, Kuroda Y, Okamoto M, Koga T, Yamashita YI. Risk Factors of Postoperative Complication and Hospital Mortality after Colorectal Perforation Surgery. J Anus Rectum Colon. 2024;8:118-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 8. | Han EC, Ryoo SB, Park BK, Park JW, Lee SY, Oh HK, Ha HK, Choe EK, Moon SH, Jeong SY, Park KJ. Surgical outcomes and prognostic factors of emergency surgery for colonic perforation: would fecal contamination increase morbidity and mortality? Int J Colorectal Dis. 2015;30:1495-1504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 9. | Vogel JD, Felder SI, Bhama AR, Hawkins AT, Langenfeld SJ, Shaffer VO, Thorsen AJ, Weiser MR, Chang GJ, Lightner AL, Feingold DL, Paquette IM. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Colon Cancer. Dis Colon Rectum. 2022;65:148-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 251] [Article Influence: 62.8] [Reference Citation Analysis (1)] |
| 10. | Lee J, Lee YJ, Seo JW, Kim ES, Kim SK, Jung MK, Heo J, Lee HS, Lee JS, Jang BI, Kim KO, Cho KB, Kim EY, Kim DJ, Chung YJ; Daegu-Gyeongbuk Gastrointestinal Study Group. Incidence of colonoscopy-related perforation and risk factors for poor outcomes: 3-year results from a prospective, multicenter registry (with videos). Surg Endosc. 2023;37:5865-5874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 11. | Brunner M, Gärtner L, Weiß A, Weber K, Denz A, Krautz C, Weber GF, Grützmann R. Risk Factors for Postoperative Major Morbidity, Anastomotic Leakage, Re-Surgery and Mortality in Patients with Colonic Perforation. J Clin Med. 2024;13:5220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 12. | Huang J, Xuan D, Li X, Ma L, Zhou Y, Zou H. The value of APACHE II in predicting mortality after paraquat poisoning in Chinese and Korean population: A systematic review and meta-analysis. Medicine (Baltimore). 2017;96:e6838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 13. | Tong-Minh K, Welten I, Endeman H, Hagenaars T, Ramakers C, Gommers D, van Gorp E, van der Does Y. Predicting mortality in adult patients with sepsis in the emergency department by using combinations of biomarkers and clinical scoring systems: a systematic review. BMC Emerg Med. 2021;21:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 14. | Ryoo SM, Lee J, Lee YS, Lee JH, Lim KS, Huh JW, Hong SB, Lim CM, Koh Y, Kim WY. Lactate Level Versus Lactate Clearance for Predicting Mortality in Patients With Septic Shock Defined by Sepsis-3. Crit Care Med. 2018;46:e489-e495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 166] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 15. | Vincent JL, Quintairos E Silva A, Couto L Jr, Taccone FS. The value of blood lactate kinetics in critically ill patients: a systematic review. Crit Care. 2016;20:257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 381] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 16. | Yang WS, Kang HD, Jung SK, Lee YJ, Oh SH, Kim YJ, Sohn CH, Kim WY. A mortality analysis of septic shock, vasoplegic shock and cryptic shock classified by the third international consensus definitions (Sepsis-3). Clin Respir J. 2020;14:857-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 17. | Mikkelsen ME, Miltiades AN, Gaieski DF, Goyal M, Fuchs BD, Shah CV, Bellamy SL, Christie JD. Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock. Crit Care Med. 2009;37:1670-1677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 601] [Cited by in RCA: 658] [Article Influence: 38.7] [Reference Citation Analysis (0)] |
| 18. | Jansen TC, van Bommel J, Schoonderbeek FJ, Sleeswijk Visser SJ, van der Klooster JM, Lima AP, Willemsen SP, Bakker J; LACTATE study group. Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial. Am J Respir Crit Care Med. 2010;182:752-761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 739] [Cited by in RCA: 639] [Article Influence: 39.9] [Reference Citation Analysis (0)] |
| 19. | Teng TS, Ji AL, Ji XY, Li YZ. Neutrophils and Immunity: From Bactericidal Action to Being Conquered. J Immunol Res. 2017;2017:9671604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 158] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 20. | Urrechaga E. Reviewing the value of leukocytes cell population data (CPD) in the management of sepsis. Ann Transl Med. 2020;8:953. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 21. | Liebman PR, Patten MT, Manny J, Benfield JR, Hechtman HB. Hepatic--portal venous gas in adults: etiology, pathophysiology and clinical significance. Ann Surg. 1978;187:281-287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 422] [Cited by in RCA: 312] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 22. | Koizumi C, Michihata N, Matsui H, Fushimi K, Yasunaga H. In-Hospital Mortality for Hepatic Portal Venous Gas: Analysis of 1590 Patients Using a Japanese National Inpatient Database. World J Surg. 2018;42:816-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Sauerbrei W, Perperoglou A, Schmid M, Abrahamowicz M, Becher H, Binder H, Dunkler D, Harrell FE Jr, Royston P, Heinze G; for TG2 of the STRATOS initiative. State of the art in selection of variables and functional forms in multivariable analysis-outstanding issues. Diagn Progn Res. 2020;4:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 171] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 24. | Nelson AL, Millington TM, Sahani D, Chung RT, Bauer C, Hertl M, Warshaw AL, Conrad C. Hepatic portal venous gas: the ABCs of management. Arch Surg. 2009;144:575-81; discussion 581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 142] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 25. | Steyerberg EW, Vickers AJ, Cook NR, Gerds T, Gonen M, Obuchowski N, Pencina MJ, Kattan MW. Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology. 2010;21:128-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4068] [Cited by in RCA: 3637] [Article Influence: 227.3] [Reference Citation Analysis (5)] |
| 26. | Vickers AJ, Elkin EB. Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making. 2006;26:565-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4224] [Cited by in RCA: 3980] [Article Influence: 199.0] [Reference Citation Analysis (4)] |
| 27. | Van Calster B, McLernon DJ, van Smeden M, Wynants L, Steyerberg EW; Topic Group ‘Evaluating diagnostic tests and prediction models’ of the STRATOS initiative. Calibration: the Achilles heel of predictive analytics. BMC Med. 2019;17:230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1457] [Cited by in RCA: 1220] [Article Influence: 174.3] [Reference Citation Analysis (0)] |
| 28. | Leisman DE, Harhay MO, Lederer DJ, Abramson M, Adjei AA, Bakker J, Ballas ZK, Barreiro E, Bell SC, Bellomo R, Bernstein JA, Branson RD, Brusasco V, Chalmers JD, Chokroverty S, Citerio G, Collop NA, Cooke CR, Crapo JD, Donaldson G, Fitzgerald DA, Grainger E, Hale L, Herth FJ, Kochanek PM, Marks G, Moorman JR, Ost DE, Schatz M, Sheikh A, Smyth AR, Stewart I, Stewart PW, Swenson ER, Szymusiak R, Teboul JL, Vincent JL, Wedzicha JA, Maslove DM. Development and Reporting of Prediction Models: Guidance for Authors From Editors of Respiratory, Sleep, and Critical Care Journals. Crit Care Med. 2020;48:623-633. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 212] [Article Influence: 42.4] [Reference Citation Analysis (0)] |
| 29. | Wynants L, van Smeden M, McLernon DJ, Timmerman D, Steyerberg EW, Van Calster B; Topic Group ‘Evaluating diagnostic tests and prediction models’ of the STRATOS initiative. Three myths about risk thresholds for prediction models. BMC Med. 2019;17:192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 127] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/