Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.113698
Revised: October 6, 2025
Accepted: December 15, 2025
Published online: February 27, 2026
Processing time: 178 Days and 19.2 Hours
Pancreaticoduodenectomy (PD) is currently the preferred surgical procedure to remove duodenal papillary adenoma. It offers a low recurrence rate, but a high rate of postoperative complications.
To determine the efficacy and safety of endoscopic papillectomy (EP) for duode
This retrospective case-control study included 102 patients who received treat
After propensity score matching for sex, year, and tumor size, 37 patients each were assigned to the PD and EP groups. All patients underwent successful re
EP may be effective and safe for the removal of duodenal papillary adenoma in a minimally invasive manner, and can offer superior long-term survival compared with conventional PD.
Core Tip: Endoscopic papillectomy (EP) is a minimally invasive, effective, and safe treatment for duodenal papillary neoplasms, offering comparable recurrence rates and superior long-term survival benefits to pancreaticoduodenectomy (PD). While PD has a low recurrence rate, its high postoperative complications limit its use. This study found similar recurrence rates between EP and PD, with fewer complications in the endoscopic group. EP is a viable alternative to PD for treating duodenal papillary neoplasms.
- Citation: Xu L, Chen SJ, Wang P, Mao L, Zou XP, Wang L, Wang Y. Safety and efficacy of endoscopic papillectomy for duodenal papillary adenomas: A single-center retrospective study. World J Gastrointest Surg 2026; 18(2): 113698
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/113698.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.113698
Duodenal papillary adenoma is a rare condition, with an incidence of 0.3%–5%, and is usually discovered incidentally during upper gastrointestinal endoscopy[1,2]. While most ampullary lesions are non-neoplastic, duodenal papillary adenoma is prone to malignant transformation. Estimates show that up to 40% of villous adenomas of the duodenum may become malignant[3-5]. To prevent malignant transformation and improve prognosis, early recognition and surgical or endoscopic removal of duodenal papillary adenomas is critical.
Currently, there is no clear consensus regarding the choice of resection method for duodenal papillary adenomas; treatment is based on the experience of individual physicians. Considering its cancerous potential, surgical excision rather than endoscopic surveillance is recommended, such as pancreaticoduodenectomy (PD)[6] and transduodenal ampullectomy (TDA)[7], which are both invasive procedures. However, given the complex anatomy and poor blood supply of the ampulla, these surgical procedures remain challenging and are associated with high risks of perioperative and postoperative complications. Specifically, PD is associated with a low recurrence rate but a relatively high mortality[8], while TDA is commonly accompanied by recurrence[6]. Therefore, regular endoscopic surveillance is required after local excision of duodenal papillary adenomas after PD and TDA.
Recent advances have improved the safety and efficacy of endoscopic resection for gastrointestinal lesions[9]. Endoscopic resection is superior to conventional surgical procedures and is characterized by minimal invasion and shorter hospital stays[10,11]. Although this approach preserves the integrity of the digestive tract, the safety and efficacy of endoscopic resection for duodenal papillary adenomas remain to be validated in clinical settings.
Currently, long-term follow-up data from large cohorts of patients with duodenal papillary adenoma treated by endoscopic papillectomy (EP) are limited. This single-center retrospective study compared the safety and efficacy of EP and PD in treating papillary adenomas to provide a reference for clinical decision-making.
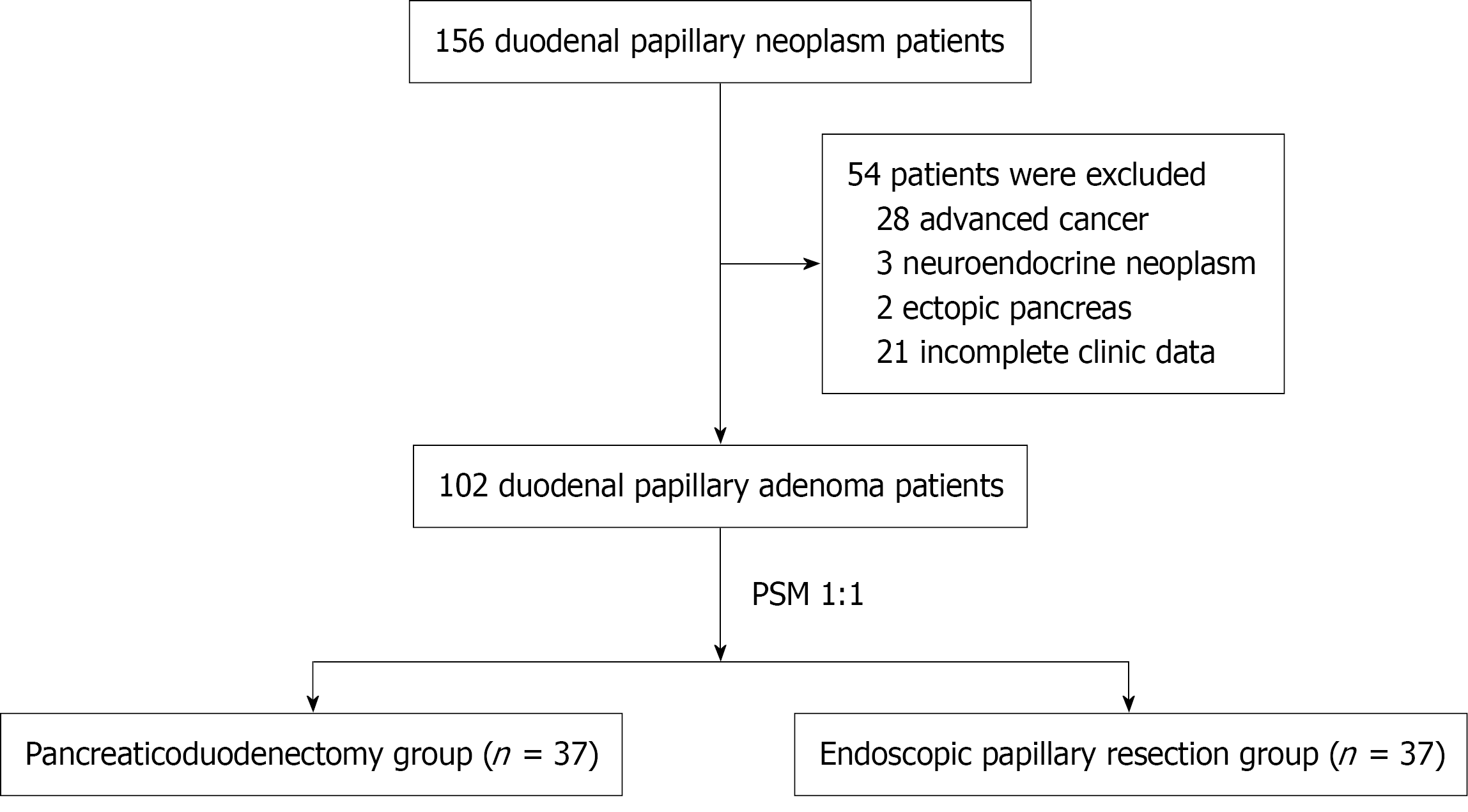
From January 2010 to December 2024, 156 patients with duodenal papillary adenoma treated at Drum Tower Hospital were initially recruited. Patients with biopsy findings indicative of malignancy or those deemed to have advanced cancer based on endoscopic evaluation were excluded. Five patients were further excluded owing to postoperative pathological confirmation of neuroendocrine tumors or ectopic pancreas. An additional 21 patients were excluded owing to insuffi
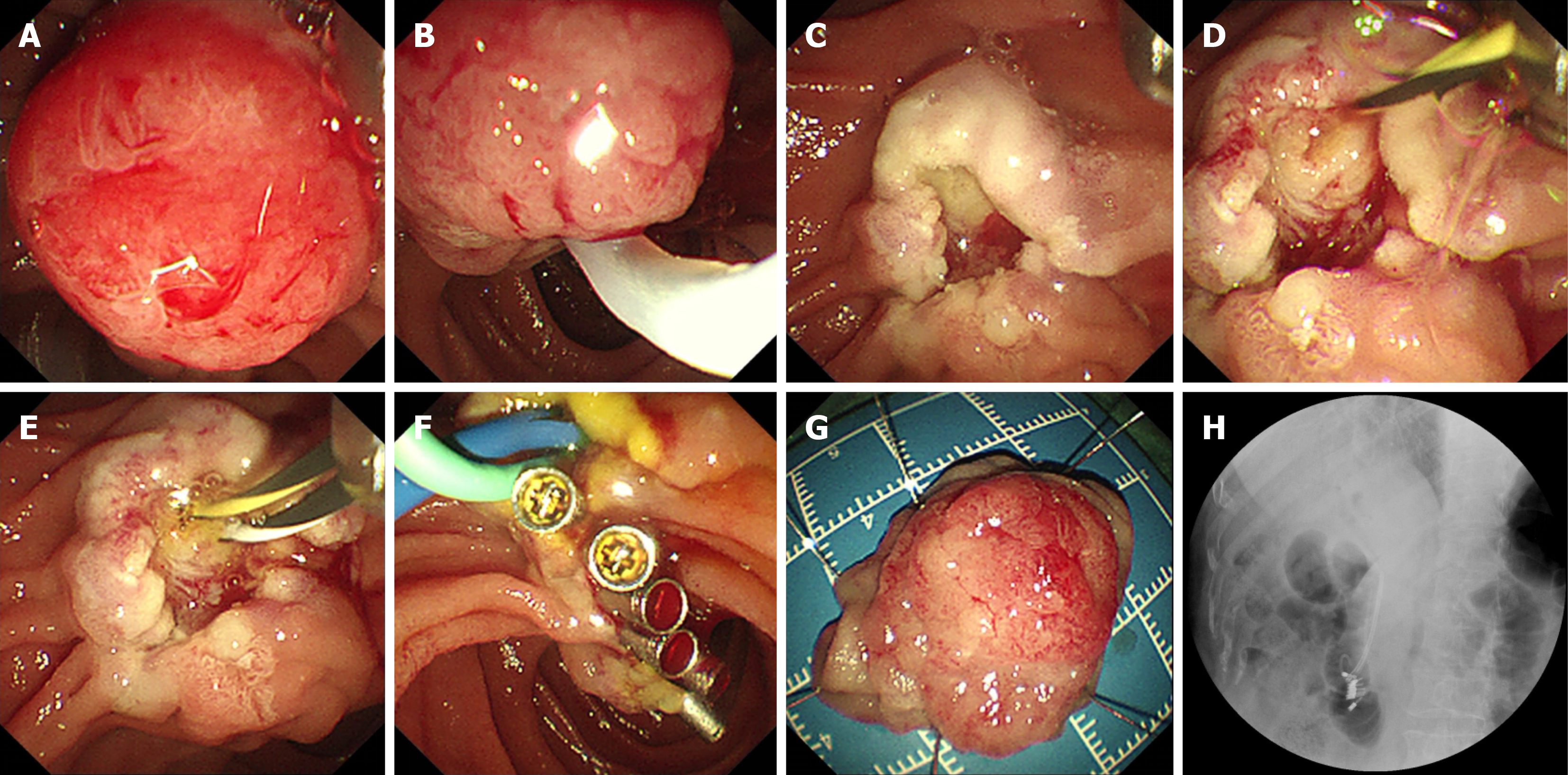
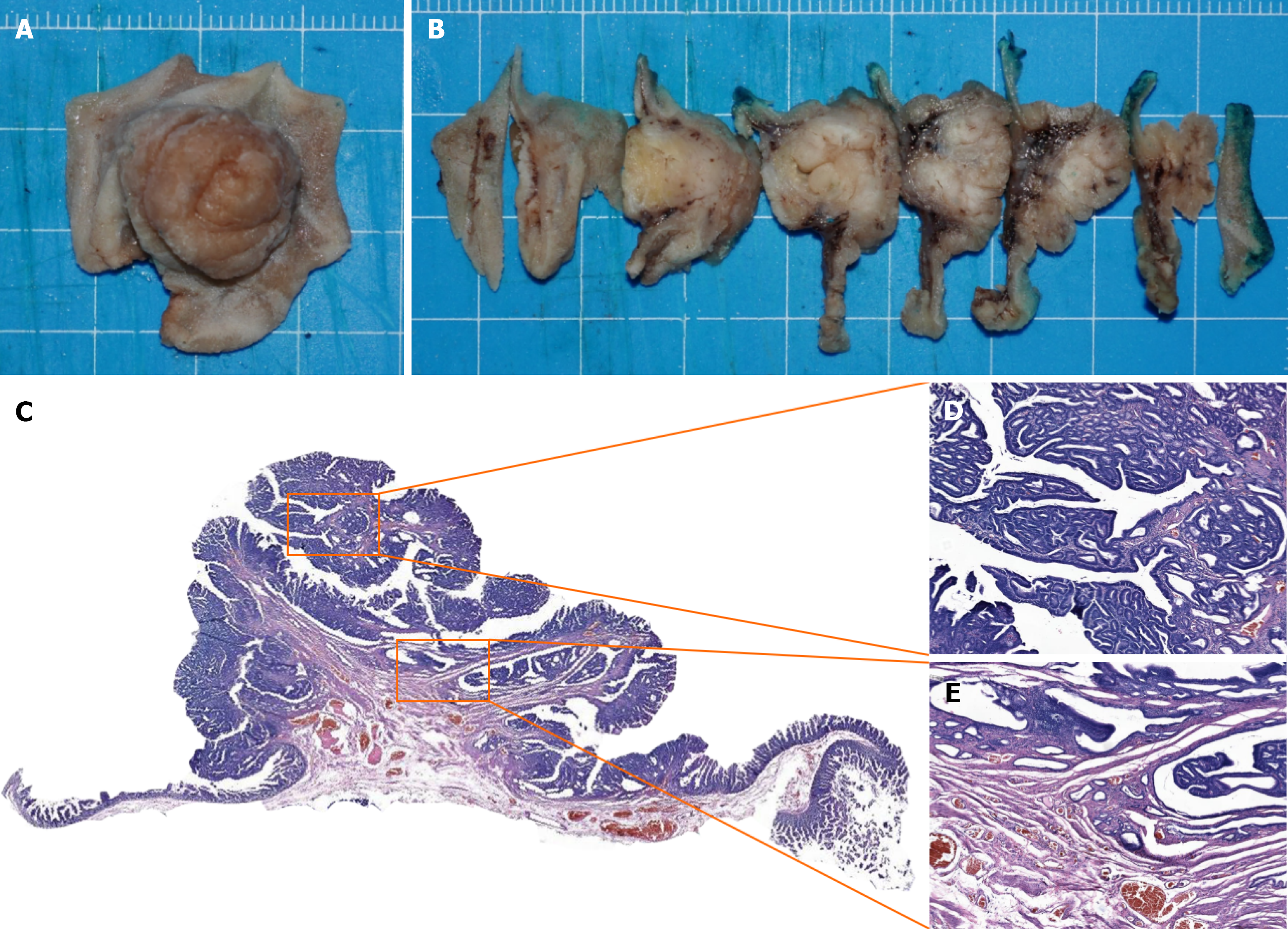
EP was performed by five experienced endoscopists using an Olympus TJF-Q260 endoscope. Other equipment and accessories used were a Micro-Tech snare, titanium clips, and an ERBE Erbotom ICC 200 Electrosurgical Unit. The EP procedure was performed in a manner similar to endoscopic mucosal resection for colonic polyps. The procedure started with submucosal injection of a solution containing 100 mL of saline and 1 mL of methylene blue around the lesion to elevate the duodenal papillary neoplasm and create a safety cushion. After adequate lifting, the lesion was resected using a snare. Any bleeding was controlled using hot biopsy forceps and bipolar electrocoagulation. Mucosal defects were partially closed with titanium clips to prevent postoperative bleeding and perforation. Finally, a plastic stent was inserted into the pancreatic duct to prevent the development of pancreatitis (Figure 2). Some patients had plastic stents implanted in the bile duct to prevent biliary stricture. The resected specimens were fixed in formalin and sectioned at intervals of 2–3 mm (Figure 3).
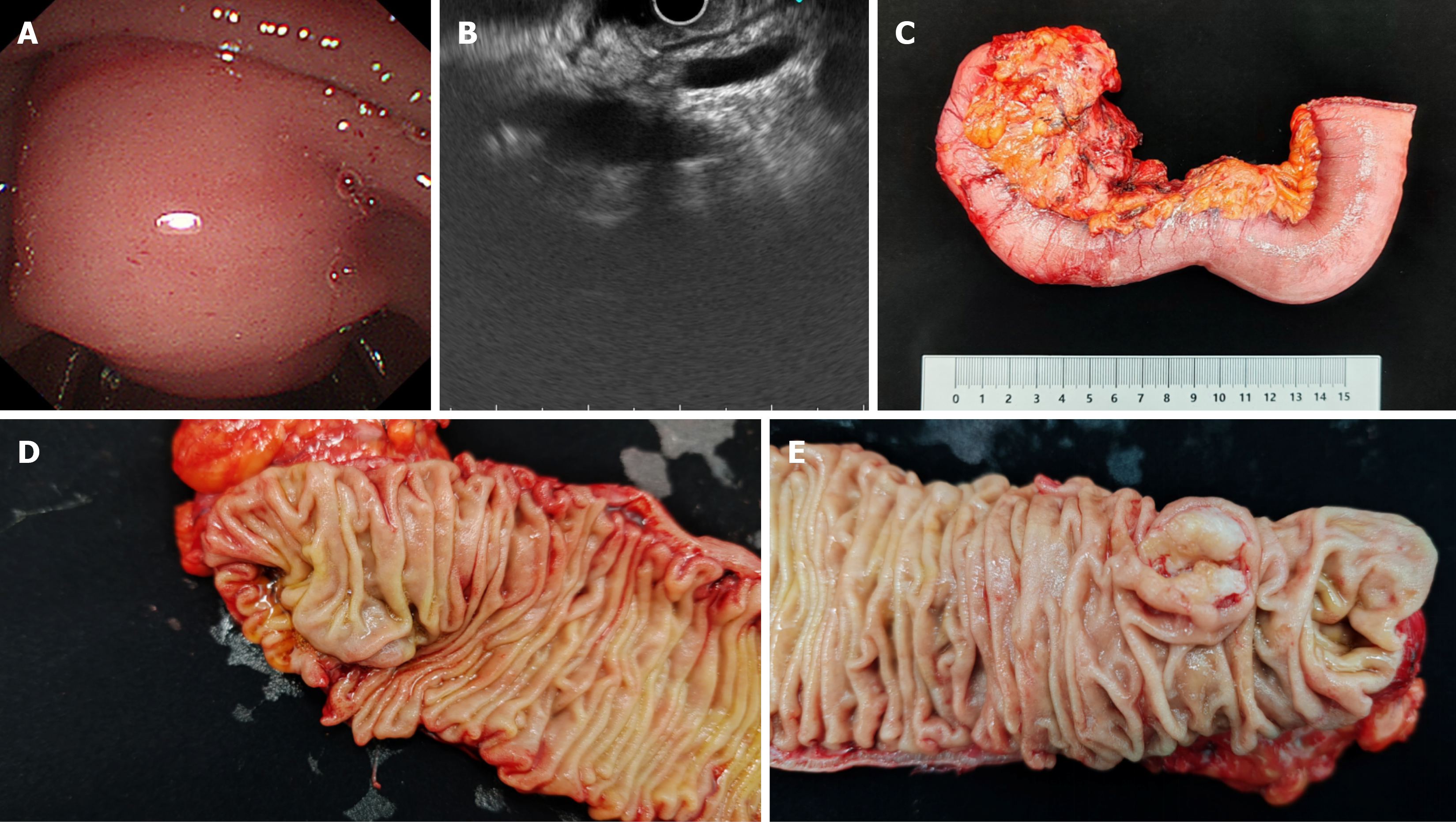
PD was performed by three experienced pancreatic surgeons. The head of the pancreas, gallbladder, duodenum, a portion of the stomach, and surrounding lymph nodes were removed. The remaining pancreas, gastric body, and common bile duct were reconnected with the jejunum. Anatomical sampling was conducted on the postoperative specimens (Figure 4).
Perioperative perforation was a serious complication of EP. It was identified by the presence of excessive duodenal tissues, a kidney shadow on X-ray imaging, or radiological evidence of intraperitoneal free gas. Hemorrhage was defined as bleeding associated with melena, unstable blood pressure, or a decrease in hemoglobin of > 2 g/dL. Successful resection was endoscopically confirmed by the complete removal of adenomatous tissue. Surveillance endoscopy was performed at 3, 6, 12, and 24 months postoperatively to monitor the ampullary wound. Patients with malignant lesions also underwent abdominal computed tomography (CT) at 3, 6, 12, and 24 months after surgery.
Clinical data were analyzed using SPSS version 25.0 (IBM, Armonk, NY, USA). Continuous variables were expressed as mean ± SD. Differences between groups were assessed using the t-test or Fisher’s exact test, as appropriate. Categorical variables were compared using the χ2 test. A two-sided P < 0.05 was considered statistically significant. To minimize confounding bias, propensity score matching (PSM) was used to analyze the data from the two groups. The patients were matched at a 1:1 ratio according to age, sex, and tumor size. All statistical analyses were performed using R software, version 4.1.2 (R Project for Statistical Computing).
A total of 102 eligible patients with a single lesion in the duodenal papilla were enrolled. After PSM, 74 patients were divided into the PD and EP groups (Figure 1). The serum levels of total bilirubin and direct bilirubin in the PD group (26.59 ± 33.75 mmol/L and 16.88 ± 26.77 mmol/L) were significantly higher than those in the EP group (12.08 ± 5.75 mmol/L and 3.71 ± 1.58 mmol/L). No significant differences were observed in sex, age, preoperative blood routine examination, preoperative coagulation function, and renal functions between the PD and EP groups. No significant differences were observed between the two groups with respect to past medical history or prior use of anticoagulant medications (Table 1).
| PD (n = 37) | EP (n = 37) | t/χ2 | P value | |
| Age (year) | 61.35 ± 8.11 | 62.83 ± 11.32 | -0.649 | 0.518 |
| Gender | ||||
| Male | 17 (45.95) | 25 (67.75) | 3.52 | 0.06 |
| Female | 20 (54.05) | 12 (32.43) | ||
| Tumor size (cm) | 1.94 ± 0.77 | 1.94 ± 0.85 | 0.123 | 0.481 |
| Previous abdominal surgery | ||||
| No | 25 (67.57) | 28 (75.68) | 0.598 | 0.439 |
| Yes | 12 (32.43) | 9 (24.32) | ||
| Hypertension | ||||
| No | 21 (56.76) | 24 (64.86) | 0.51 | 0.475 |
| Yes | 16 (43.24) | 13 (35.14) | ||
| Diabetes | ||||
| No | 34 (91.89) | 32 (86.49) | 0.561 | 0.454 |
| Yes | 3 (8.11) | 5 (13.51) | ||
| Coronary heart disease | ||||
| No | 36 (97.30) | 35 (94.59) | 0.347 | 0.556 |
| Yes | 1 (2.70) | 2 (5.41) | ||
| Anticoagulant | ||||
| No | 36 (97.30) | 36 (97.30) | 0.01 | 0.99 |
| Yes | 1 (2.70) | 1 (2.70) | ||
| CBC | ||||
| Hemameba | 6.14 ± 2.34 | 5.90 ± 1.48 | 0.534 | 0.595 |
| Hemoglobin | 124.70 ± 26.34 | 134.14 ± 19.14 | -1.762 | 0.082 |
| Blood platelet | 212.76 ± 65.93 | 187.14 ± 60.09 | 1.747 | 0.085 |
| Blood coagulation | ||||
| PT | 11.93 ± 1.10 | 12.92 ± 2.44 | -2.239 | 0.58 |
| APTT | 26.96 ± 3.64 | 27.68 ± 3.95 | -0.811 | 0.42 |
| Biochemical function | ||||
| Total bilirubin | 26.59 ± 33.75 | 12.08 ± 5.75 | 2.578 | 0.012 |
| Direct bilirubin | 16.88 ± 26.77 | 3.71 ± 1.58 | 2.987 | 0.004 |
| Albumin | 39.70 ± 4.289 | 40.00 ± 2.45 | -0.37 | 0.713 |
| Urea nitrogen | 4.61 ± 1.66 | 5.20 ± 1.42 | -1.659 | 0.102 |
| Creatinine | 59.00 ± 12.43 | 68.03 ± 18.48 | -2.465 | 0.058 |
| Tumor marker | ||||
| CA19-9 | 21.32 ± 27.53 | 13.92 ± 10.55 | 1.526 | 0.131 |
The surgical outcomes are shown in Table 2. The en bloc resection rate was 100% in each group. None of the patients who underwent endoscopic resection required surgical intervention owing to complications. Postoperative pathological findings revealed that 28 cases exhibited adenoma combined with carcinoma in situ, including 17 in the PD group and 11 in the EP group. No significant difference was observed in the pathological findings between the two groups (P > 0.05). To prevent postoperative stenosis, biliary stents were placed in 7 patients in the EP group and only 1 patient in the PD group (P < 0.05). To prevent postoperative pancreatitis and pancreatic duct stenosis, 75% of patients who underwent PD and 81% of patients who underwent EP had pancreatic duct stents placed, with no significant difference between the groups (P > 0.05; Table 2).
| PD (n = 37) | EP (n = 37) | χ2 | P value | |
| Pathology | ||||
| Adenoma | 20 (54.05) | 26 (70.27) | 2.157 | 0.141 |
| Carcinoma in situ | 17 (45.95) | 11 (29.73) | ||
| Biliary stent placement | ||||
| No | 36 (97.30) | 30 (81.08) | 5.045 | 0.025 |
| Yes | 1 (2.70) | 7 (18.92) | ||
| Pancreatic stent placement | ||||
| No | 9 (24.32) | 7 (18.92) | 0.319 | 0.572 |
| Yes | 28 (75.68) | 30 (81.08) | ||
| Complications | ||||
| Hemorrhage | ||||
| No | 31 (83.78) | 33 (89.19) | 0.463 | 0.496 |
| Yes | 6 (16.22) | 4 (10.81) | ||
| Perforation | ||||
| No | 35 (94.59) | 36 (97.30) | 0.347 | 0.556 |
| Yes | 2 (5.41) | 1 (2.70) | ||
| Biliary fistula | ||||
| No | 36 (97.30) | 37 (100.00) | 1.014 | 0.314 |
| Yes | 1 (2.70) | 0 (0.00) | ||
| Pancreatic fistula | ||||
| No | 32 (86.49) | 37 (100.00) | 5.362 | 0.021 |
| Yes | 5 (13.51) | 0 (0.00) | ||
| Cholangitis | ||||
| No | 35 (94.59) | 34 (91.89) | 0.214 | 0.643 |
| Yes | 2 (5.41) | 3 (8.11) | ||
| Pancreatitis | ||||
| No | 34 (91.89) | 34 (91.89) | 0.001 | 0.999 |
| Yes | 3 (8.11) | 3 (8.11) | ||
| Abdominal infection | ||||
| No | 29 (78.38) | 36 (97.30) | 6.198 | 0.013 |
| Yes | 8 (21.62) | 1 (2.70) | ||
| Mortality | ||||
| No | 35 (94.59) | 37 (100.00) | 2.056 | 0.152 |
| Yes | 2 (5.41) | 0 (0.00) |
Five cases of pancreatic fistula were observed in the PD group, whereas no occurrence was noted in the EP group (P < 0.05; Table 2). There were 8 cases of abdominal cavity infection in the PD group, but only 1 in the EP group, showing a significant difference (P < 0.05). Two patients in the PD group died from surgery-related complications, whereas no mortality was observed in the EP group (P = 0.152). No significant differences were observed in complications, including bleeding, perforation, cholangitis, biliary fistula, and pancreatitis, between the two groups (all P > 0.05).
Elevated serum amylase levels within 24 h postoperatively, prolonged activated partial thromboplastin time before surgery, and PD were associated with an increased risk of postoperative complications (Table 3). Follow-up data were available for 35 patients in the EP group, with a median follow-up of 24 months and a maximum of 60 months. Seven patients in the EP group experienced local recurrence; 2 underwent surgical treatment, while the remaining cases were managed with endoscopic radiofrequency ablation. The median follow-up duration for patients in the PD group was 26 months. Four patients were lost to follow-up. Among the remaining patients, 2 died from surgical complications, 1 from ovarian cancer, and 1 from cerebral infarction; in addition, 2 experienced recurrence at the choledochojejunostomy site during follow-up, and 1 patient developed distant metastasis (Table 4).
| Non-complication (n = 49) | Complication (n = 25) | t/χ2 | P value | |
| Age (year) | 62.63 ± 10.10 | 61.04 ± 9.33 | 0.658 | 0.513 |
| Gender | ||||
| Male | 27 (55.10) | 15 (60.0) | 0.162 | 0.687 |
| Female | 22 (44.90) | 10 (40.0) | ||
| Tumor diameter (cm) | 1.95 ± 0.80 | 1.93 ± 0.83 | 0.124 | 0.902 |
| Previous abdominal surgery | ||||
| No | 34 (69.39) | 19 (76.0) | 0.356 | 0.551 |
| Yes | 15 (30.61) | 6 (24.0) | ||
| Hypertension | ||||
| No | 31 (63.27) | 14 (56.0) | 0.367 | 0.545 |
| Yes | 18 (36.73) | 11 (44.0) | ||
| Diabetes | ||||
| No | 43 (87.76) | 23 (92.0) | 0.309 | 0.578 |
| Yes | 6 (12.24) | 2 (8.0) | ||
| Coronary heart disease | ||||
| No | 46 (93.88) | 25 (100.0) | 1.595 | 0.207 |
| Yes | 3 (6.12) | 0 | ||
| CBC (24 h postoperatively) | ||||
| Hemameba | 8.60 ± 3.44 | 10.81 ± 4.51 | -2.354 | 0.021 |
| Hemoglobin | 127.96 ± 18.12 | 124.28 ± 15.65 | 0.863 | 0.391 |
| Blood coagulation | ||||
| PT | 12.53 ± 2.12 | 12.21 ± 1.57 | 0.685 | 0.496 |
| APTT | 26.63 ± 2.99 | 28.66 ± 4.78 | -2.244 | 0.028 |
| Pathology | ||||
| Adenoma | 25 (51.02) | 12 (48.00) | 0.06 | 0.806 |
| Carcinoma in situ | 24 (48.98) | 13 (52.00) | ||
| Operation methods | ||||
| PD | 20 (40.82) | 17 (68.00) | 4.893 | 0.027 |
| EP | 29 (59.18) | 8 (32.00) | ||
| PD (n = 32) | EP (n = 35) | χ2 | P value | |
| Patients without recurrence | 29 | 28 | 2.773 | 0.096 |
| Dead patients | 4 | 0 | ||
| Living patients | 25 | 28 | ||
| Patients with recurrence | 3 | 7 | 2.628 | 1.000 |
| Radiofrequency ablation | 0 | 5 | ||
| Surgical treatment | 0 | 2 | ||
| Observation | 3 | 0 |
Duodenal papillary neoplasms are a heterogeneous group of lesions that originate from the biliary, duodenal, and pancreatic epithelial cells of the ampulla of Vater and are usually detected during an endoscopic examination of the upper gastrointestinal tract. Among all its subtypes, duodenal papillary adenoma is most commonly observed. Ampul
PD involves resection of the head of the pancreas, duodenum, and part of the stomach. However, as a technically demanding procedure, PD is associated with a high risk of postoperative complications, such as intraperitoneal hemor
TDA is a less invasive alternative for the excision of papillary neoplasms that does not require the removal of surrounding organs of the lesion[14], thus offering a faster postoperative recovery, shorter hospital length of stay, and higher long-term success rate (83.6%)[7]. However, owing to the high recurrence rate following TDA and the persistence of complications such as bile fistula and intra-abdominal infection, this approach has not gained widespread acceptance in clinical practice. Consequently, most surgeons continue to prefer PD for the management of lesions located in the ampulla of Vater.
Endoscopic interventions are emerging modalities to avoid the need for surgery[15-17]. EP is an endoscopic inter
In patients with appropriate indications, EP can minimize the risks of local recurrence and metastasis. Papillary adenoma is a benign disease, and thus is the best indication for endoscopic resection. However, papillary lesions with intraductal extension usually recur. We therefore recommend endoscopic ultrasound to exclude submucosal invasion, along with computed tomography or magnetic resonance imaging to assess regional lymph nodes in patients with suspected malignant lesions[24,25]. A previous study demonstrated that endoscopic ultrasound has a specificity of 88% and a negative predictive value of 90% for evaluating the infiltration depth of duodenal adenomas[26]. Although preoperative imaging plays a critical role in determining the indications for EP, postoperative pathological analysis remains necessary. We suggest processing EP specimens using endoscopic submucosal dissection to ensure accurate histopathological assessment. If intravascular tumor thrombus or submucosal invasion is identified, surgical intervention is warranted[27].
Several limitations of this study should be acknowledged. First, this was a single-center retrospective study, which may be subject to selection bias. Second, duodenal papillary adenoma is a rare condition, and the relatively small sample size may have influenced the reliability of our findings. Further large-scale multicenter randomized controlled trials are required to validate the findings of this research.
EP is safe and effective for treating duodenal papillary adenoma, offering a lower recurrence rate and better long-term prognosis than standard PD. In cases of post-EP recurrence, repeat endoscopic treatment or surgical intervention may be considered as appropriate therapeutic options.
The authors thank the members of the Department of Gastroenterology, Drum Tower Hospital, Nanjing University School of Medicine, for their valuable contributions to the discussion and critical review of the manuscript.
| 1. | Kővári B, Kim BH, Lauwers GY. The pathology of gastric and duodenal polyps: current concepts. Histopathology. 2021;78:106-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (2)] |
| 2. | Mortensen MB, Fenger C. [Polyps in the stomach and duodenum]. Ugeskr Laeger. 2008;170:25-30. [PubMed] |
| 3. | Giehl-Brown E, Weitz J, Distler M. [Ampullary Carcinoma-prognostic and Therapeutical Contrast to Pancreatic Ductal Adenocarcinoma]. Zentralbl Chir. 2022;147:160-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 4. | Singh A, Siddiqui UD, Konda VJ, Whitcomb E, Hart J, Xiao SY, Ruiz MG, Koons A, Waxman I. Safety and efficacy of EMR for sporadic, nonampullary duodenal adenomas: a single U.S. center experience (with video). Gastrointest Endosc. 2016;84:700-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Mo W, Li J, Dai Y, Chen J, Xu X. A duodenal ampullary tumor with malignant transformation of papillary polyps: a case report and literature review. J Int Med Res. 2021;49:3000605211053230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 6. | Song J, Liu H, Li Z, Yang C, Sun Y, Wang C. Long-term prognosis of surgical treatment for early ampullary cancers and implications for local ampullectomy. BMC Surg. 2015;15:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 7. | Schneider L, Contin P, Fritz S, Strobel O, Büchler MW, Hackert T. Surgical ampullectomy: an underestimated operation in the era of endoscopy. HPB (Oxford). 2016;18:65-71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 8. | Martin JA, Haber GB. Ampullary adenoma: clinical manifestations, diagnosis, and treatment. Gastrointest Endosc Clin N Am. 2003;13:649-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 64] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 9. | Basford PJ, George R, Nixon E, Chaudhuri T, Mead R, Bhandari P. Endoscopic resection of sporadic duodenal adenomas: comparison of endoscopic mucosal resection (EMR) with hybrid endoscopic submucosal dissection (ESD) techniques and the risks of late delayed bleeding. Surg Endosc. 2014;28:1594-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 10. | Klein A, Nayyar D, Bahin FF, Qi Z, Lee E, Williams SJ, Byth K, Bourke MJ. Endoscopic mucosal resection of large and giant lateral spreading lesions of the duodenum: success, adverse events, and long-term outcomes. Gastrointest Endosc. 2016;84:688-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 75] [Article Influence: 7.5] [Reference Citation Analysis (2)] |
| 11. | Rajkomar K, Kweon M, Khan I, Frankish P, Rodgers M, Koea JB. Endoscopic assessment and management of sporadic duodenal adenomas: The results of single centre multidisciplinary management. World J Gastrointest Endosc. 2017;9:196-203. [PubMed] [DOI] [Full Text] |
| 12. | Kapoor VK. Complications of pancreato-duodenectomy. Rozhl Chir. 2016;95:53-59. [PubMed] |
| 13. | Zubair AB, Khan Sherwani IAR, Ahmad M, Tahir MA, Khalil MI, Bukhari MM, Sabir M, Bhatti AA, Afzal N, Kaneez M. The Spectrum of Postoperative Complications and Outcomes After Pancreaticoduodenectomy: A Retrospective Outlook From a Developing Country. Cureus. 2022;14:e22218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Mathur A, Paul H, Ross S, Luberice K, Hernandez J, Vice M, Rosemurgy AS. Transduodenal ampullectomy for ampullary adenomas: a safe and effective procedure with long-term salutary outcomes. Am Surg. 2014;80:185-190. [PubMed] |
| 15. | Will U, Müller AK, Fueldner F, Wanzar I, Meyer F. Endoscopic papillectomy: data of a prospective observational study. World J Gastroenterol. 2013;19:4316-4324. [PubMed] [DOI] [Full Text] |
| 16. | Matsui T, Matsubayashi H, Hotta K, Sasaki K, Ito H, Ono H. A case of carcinoma in an adenoma of the duodenal minor papilla successfully treated with endoscopic mucosal resection. Endosc Int Open. 2016;4:E252-E254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Ye LP, Mao XL, Zheng HH, Zhang Y, Shen LY, Zhou XB, Zhu LH. Safety of endoscopic resection for duodenal subepithelial lesions with wound closure using clips and an endoloop: an analysis of 68 cases. Surg Endosc. 2017;31:1070-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Lü S, Jiang M, Liu F, Tang H, Yang Y, Zhang W, Zhang M, Jin Z, Li Z. Endoscopic papillectomy of benign papillary tumors: A single-center experience. Medicine (Baltimore). 2020;99:e20414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Patel R, Varadarajulu S, Wilcox CM. Endoscopic ampullectomy: techniques and outcomes. J Clin Gastroenterol. 2012;46:8-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 20. | Kim HK, Lo SK. Endoscopic approach to the patient with benign or malignant ampullary lesions. Gastrointest Endosc Clin N Am. 2013;23:347-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Moon JH, Choi HJ, Lee YN. Current status of endoscopic papillectomy for ampullary tumors. Gut Liver. 2014;8:598-604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Cho SH, Oh D, Song TJ, Park DH, Seo DW, Lee SK, Kim MH, Lee SS. Long-term Outcomes of Endoscopic Intraductal Radiofrequency Ablation for Ampullary Adenoma with Intraductal Extension after Endoscopic Snare Papillectomy. Gut Liver. 2023;17:638-646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 23. | Landim DL, de Moura DTH, Hirsch BS, de Oliveira GHP, Veras MO, Nunes FG, Cavassola PRP, Bernardo WM, Mahmood S, de Moura EGH. Radiofrequency ablation for ampullary neoplasia with intraductal extension after endoscopic papillectomy: Systematic review and meta-analysis. Endosc Int Open. 2024;12:E440-E447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 24. | Alvarez-Sánchez MV, Napoleon B. EUS and ampullary adenoma: Why? Endosc Int Open. 2016;4:E1319-E1321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 25. | Rejeski JJ, Kundu S, Hauser M, Conway JD, Evans JA, Pawa R, Mishra G. Characteristic endoscopic ultrasound findings of ampullary lesions that predict the need for surgical excision or endoscopic ampullectomy. Endosc Ultrasound. 2016;5:184-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 26. | Azih LC, Broussard BL, Phadnis MA, Heslin MJ, Eloubeidi MA, Varadarajulu S, Arnoletti JP. Endoscopic ultrasound evaluation in the surgical treatment of duodenal and peri-ampullary adenomas. World J Gastroenterol. 2013;19:511-515. [PubMed] [DOI] [Full Text] |
| 27. | Klein A, Tutticci N, Bourke MJ. Endoscopic resection of advanced and laterally spreading duodenal papillary tumors. Dig Endosc. 2016;28:121-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/