Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.113730
Revised: November 17, 2025
Accepted: January 4, 2026
Published online: February 27, 2026
Processing time: 125 Days and 3.3 Hours
Postoperative gastrointestinal recovery is a critical factor influencing patient rehabilitation.
To investigate the effects of conventional care compared with the failure mode and effects analysis (FMEA) protocol on postoperative recovery in patients under
A retrospective cohort study analyzed 225 patients with colorectal cancer (conventional care group: n = 122; FMEA group: n = 103) treated between June 2021 and June 2024. Outcomes included gastrointestinal recovery (time to flatus/bowel movement and diet tolerance), functional capacity (6-minute walk distance test and sit-to-stand test), complications, quality of life (General Comfort Question
The FMEA group demonstrated significantly accelerated recovery: Shorter hos
FMEA-based nursing significantly enhances postoperative functional recovery, reduces complications, and improves quality of life in patients undergoing extended colectomy.
Core Tip: This study introduces the failure mode and effects analysis nursing model into the postoperative care of patients undergoing laparoscopic extended colectomy for colorectal cancer. By systematically identifying and proactively mitigating potential risks throughout the perioperative pathway, this innovative approach was found to significantly accelerate gas
- Citation: Yang QQ, Wu ZK. Failure mode and effects analysis nursing model improves recovery and quality of life post-laparoscopic extended radical colectomy for colorectal cancer. World J Gastrointest Surg 2026; 18(2): 113730
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/113730.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.113730
Colorectal cancer (CRC) is a leading cause of cancer-related mortality globally, characterized by malignant proliferation in the colon/rectum[1,2]. Clinical manifestations include altered bowel habits, hematochezia, and weight loss[3]. Patho
Postoperative gastrointestinal dysfunction is a common complication following surgical procedures, leading to prolonged hospital stays and increased morbidity[8]. Advances in perioperative care have introduced various protocols aimed at optimizing recovery. The current enhanced recovery after surgery (ERAS) protocols prioritizes early mo
Failure mode and effects analysis (FMEA) is a proactive method used to identify potential failures in processes, assess their impact, and implement preventive measures. Originally developed in engineering, FMEA has been adapted for use in healthcare to enhance patient safety and improve clinical outcomes[10]. In the context of surgical care, FMEA can be applied to identify and mitigate risks throughout the perioperative period, from preoperative assessment to post
Previous studies have shown that FMEA-based interventions can lead to significant improvements in patient safety and care quality[11]. For example, FMEA has been successfully used to reduce medication errors, improve handover processes, and enhance compliance with best practices[12]. However, evidence on the application of FMEA in the context of postoperative recovery is limited, particularly in comparison with conventional care (CC) protocols. This study aims to evaluate the impact of the FMEA protocol on postoperative recovery, focusing on gastrointestinal function, exercise recovery, and quality of life. By integrating detailed risk assessments and targeted interventions, this protocol seeks to provide a more personalized and effective approach to patient care. The primary objective is to demonstrate improved recovery outcomes and patient satisfaction, thereby setting a new standard for postoperative care.
This study selected 225 patients who underwent laparoscopic extended colectomy for CRC at Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College from June 2021 to June 2024 as research subjects. All patients met the inclusion criteria. Patients were divided into two groups based on the care model received after admission: The CC group and the FMEA group. Among them, 122 patients received CC, while 103 patients received care interventions based on FMEA.
This research was approved by Ethics Committee of Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College. Since the study involved the use of anonymized patient data and posed no potential risks or negative impacts on patient care, the requirement for informed consent was waived for this retrospective analysis. This exemption was granted by Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College’s Ethics Committee in compliance with relevant regulatory and ethical standards.
Inclusion criteria: (1) Diagnosed with CRC and underwent laparoscopic extended colectomy for CRC[13]; (2) Aged between 18 years and 75 years; (3) No severe cardiopulmonary insufficiency or other systemic diseases (such as severe diabetes or liver or kidney failure); and (4) Complete data.
Exclusion criteria: (1) Extensive metastasis or advanced cancer; (2) Severe cardiopulmonary insufficiency that cannot tolerate general anesthesia; (3) Extensive intra-abdominal adhesions or other conditions unsuitable for laparoscopic procedures; (4) Other malignancies within the past 5 years; (5) Severe mental or cognitive impairments that prevent understanding of the study content or cooperation in follow-up and evaluation; (6) Underwent other major surgeries within the past 6 months; and (7) Incomplete data.
The CC group received routine clinical care protocols from the time of patient admission. This model focuses on reactive management of issues that have already arisen, centered around the execution of established clinical pathways. These care measures included, but were not limited to, routine preoperative assessments, surgical preparations, postoperative care, and follow-up after discharge. Healthcare providers followed the hospital’s existing standard operating procedures. All care activities were documented in the patients’ electronic medical record system to ensure data integrity and traceability.
The FMEA group received a structured, prospective care protocol built on the FMEA model. The core purpose of applying the FMEA model in this study was to systematically identify and anticipate potential nursing weaknesses and risk factors throughout the entire perioperative process, thereby implementing targeted preventive interventions aimed at fundamentally reducing complications, accelerating functional recovery, and improving patients’ quality of life. A multidisciplinary team was formed before the implementation of the protocol to achieve this goal. The team systematically mapped out the entire care pathway from admission to discharge and conducted prospective risk analyses for each step (such as preoperative psychological counseling, pain management strategies, and early mobilization). They identified “what could go wrong” (failure modes), analyzed “the causes and consequences of these failures” (effect analysis), and accordingly developed and integrated specific optimization measures into the care plan. This approach fundamentally differs from traditional care due to its proactive prevention and high degree of personalization. For example, while traditional care provides analgesics when a patient complains of pain, the FMEA model preemptively assesses the risk that patients may avoid coughing or getting out of bed due to fear of pain, thus developing and strictly enforcing a preventive analgesia plan on the basis of non-pharmacological measures and scheduled medication. Nurses actively assist and supervise patients in early mobilization. Therefore, FMEA care is not simply a collection of isolated measures but a systematic, continuous quality improvement model that integrates risk warnings and proactive inter
Baseline data and demographic information were obtained from the medical record system. Demographic information, such as gender, age, and body mass index, was retrieved for each patient. Baseline data, including tumor staging assessed using the tumor-node-metastasis staging system, were extracted from the medical records[14]. Information regarding the surgical procedure and its location were obtained from the surgical reports.
Functional recovery data were systematically collected from the hospital’s electronic medical record system. Parameters, such as surgical duration, hospital stay duration, time to first flatus, time to first bowel movement, and time to initiation of food intake, were retrieved to assess the recovery of gastrointestinal function in patients. Additionally, the time to resumption of normal activity was documented. The patients were evaluated using two standardized functional tests: The 6-minute walk distance (6MWD) test and the sit-to-stand test. These assessments were conducted at postoperative weeks 4 and weeks 8.
The 6MWD test was conducted in accordance with the protocol published by the American Thoracic Society to evaluate functional capacity[15]. The 6MWD test is a submaximal exercise endurance test widely used to assess functional exercise capacity in individuals with cardiopulmonary diseases, post-surgical patients, and elderly populations. Its primary purpose is to evaluate an individual’s overall exercise endurance and functional status over a specific period, with results highly correlated with the ability to perform activities of daily living (ADL). The test is conducted on a quiet, flat corridor that is 30 m long. Before the test, patients are informed that the goal is to “walk as far as possible within 6 minutes” and that they can adjust their speed or rest in accordance with their fatigue level. At the end of the 6 minutes, the total walking distance is recorded in meters. A longer distance indicates better cardiopulmonary function and exercise endurance.
The sit-to-stand test is a simple and effective tool used to assess lower limb muscle strength, endurance, and functional activity capacity, particularly suitable for elderly and postoperative patients[16]. This test effectively reflects the patient’s ability to perform daily activities such as transitioning from sitting to standing. During the test, the patient sits on a chair without armrests, with a height of approximately 43 cm, maintaining an upright back and feet flat on the floor. Upon hearing the “start” command, the patient completes as many full cycles as possible of moving from a seated position to a fully standing position and then returning to a seated position within 60 seconds. The number of complete cycles per
Quality of life was assessed at 4 weeks postoperatively. The General Comfort Questionnaire by Kolcaba[17] was used to evaluate patient comfort, encompassing four domains: Physical comfort (six items), psychological comfort (six items), environmental comfort (six items), and social comfort (five items). Each item is rated on a scale from 1 (strongly disagree) to 5 (strongly agree), with total scores ranging from 23 to 115. A total score below 60 indicates significant discomfort, a score between 60 and 90 suggests mild discomfort, and a score above 90 indicates high comfort.
The Barthel index was employed to assess individuals’ ADL by using a quantitative scoring system[18]. It evaluates 10 domains: Eating, bathing, grooming, dressing, bowel control, bladder control, toileting, transferring from bed to chair, walking on a level surface, and climbing stairs. Scores for each domain range from 0 (complete dependence) to the highest score for independent completion, with a total score ranging from 0 to 100. The total score reflects the patient’s self-care ability level, where higher scores indicate greater independence and lower scores indicate a greater need for assistance and support. Scores between 21 and 40, 41 and 60, 61 and 90, and 91 and above indicate severe dependence, moderate dependence, mild dependence, and full independence, respectively.
The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 questionnaire was used to assess quality of life, focusing on functional scales and symptom scales[19]. This questionnaire includes five functional scales (physical, role, emotional, cognitive, and social functioning), and three symptom scales (fatigue, nausea and vomiting, and pain). The functional scales consist of 14 items, with each question scored from 1 (not at all) to 4 (very much), where higher scores are better. The symptom scales include six items, each rated from 1 (not at all) to 4 (very much), where higher scores indicate less severe symptoms and impact on quality of life.
Patient satisfaction was assessed using the Service Quality Questionnaire, which encompasses five dimensions: Tangibles, reliability, responsiveness, assurance, and empathy[20]. Each dimension is rated on a scale from 1 (very poor) to 7 (very good). A total score above 30 indicates very satisfied patients, scores between 15 and 30 indicate basically satisfied patients, and scores below 15 indicate dissatisfied patients.
This study had no missing data. Data analysis was performed using SPSS (version 29.0) statistical software (SPSS Inc., Chicago, IL, United States). Categorical variables were expressed as n (%) and analyzed using χ2 tests and basic formulas. Continuous variables were tested for normal distribution by using the Shapiro-Wilk method. For continuous variables that followed a normal distribution, they were reported as mean ± SD and analyzed using t-tests adjusted for variance. A two-tailed P < 0.05 was considered statistically significant.
In comparing the baseline data and demographic information between the CC group (n = 122) and the FMEA group (n = 103), no significant differences were observed across all indicators (Table 1). Specifically, no significant differences were found in age (47.82 ± 3.74 vs 48.29 ± 3.58, P = 0.344), gender distribution (female: 26.23% vs 28.16%, male: 73.77% vs 71.84%, P = 0.746), body mass index (22.18 ± 2.21 vs 22.23 ± 2.13, P = 0.864), smoking history (40.16% vs 37.86%, P = 0.725), alcohol history (45.9% vs 45.63%, P = 0.968), educational attainment (P = 0.919), residence location (urban: 68.03% vs 65.05%, rural: 31.97% vs 34.95%, P = 0.636), tumor-node-metastasis tumor stage (P = 0.977), resection site (P = 0.883), extent of resection (P = 0.962), hypertension prevalence (22.13% vs 27.18%, P = 0.380), cardiovascular disease (9.84% vs 10.68%, P = 0.835), diabetes mellitus (10.66% vs 13.59%, P = 0.499), hemoglobin level (13.17 ± 1.76 vs 13.33 ± 1.85, P = 0.522), and blood glucose level (96.34 ± 6.53 vs 95.47 ± 6.18, P = 0.309).
| Indicator | CC group (n = 122) | FMEA group (n = 103) | t/χ2 | P value |
| Age | 47.82 ± 3.74 | 48.29 ± 3.58 | 0.947 | 0.344 |
| Gender | 0.105 | 0.746 | ||
| Female | 32 (26.23) | 29 (28.16) | ||
| Male | 90 (73.77) | 74 (71.84) | ||
| BMI | 22.18 ± 2.21 | 22.23 ± 2.13 | 0.171 | 0.864 |
| Smoking history | 49 (40.16) | 39 (37.86) | 0.124 | 0.725 |
| Alcohol history | 56 (45.9) | 47 (45.63) | 0.002 | 0.968 |
| Educational attainment | 0.169 | 0.919 | ||
| Elementary school and below | 27 (22.13) | 25 (24.27) | ||
| Middle school | 62 (50.82) | 50 (48.54) | ||
| University and above | 33 (27.05) | 28 (27.18) | ||
| Residence location | 0.224 | 0.636 | ||
| Urban | 83 (68.03) | 67 (65.05) | ||
| Rural | 39 (31.97) | 36 (34.95) | ||
| TNM tumor stage | 0.047 | 0.977 | ||
| I | 45 (36.89) | 37 (35.92) | ||
| II | 41 (33.61) | 36 (34.95) | ||
| III | 36 (29.51) | 30 (29.13) | ||
| Resection site | 0.249 | 0.883 | ||
| Right hemicolectomy | 63 (51.64) | 55 (53.40) | ||
| Left hemicolectomy | 37 (30.33) | 32 (31.07) | ||
| Sigmoidectomy | 22 (18.03) | 16 (15.53) | ||
| Extent of resection | 0.002 | 0.962 | ||
| Standard radical resection | 98 (80.33) | 83 (80.58) | ||
| Extended radical resection | 24 (19.67) | 20 (19.42) | ||
| Hypertension | 27 (22.13) | 28 (27.18) | 0.772 | 0.380 |
| Cardiovascular disease | 12 (9.84) | 11 (10.68) | 0.043 | 0.835 |
| Diabetes mellitus | 13 (10.66) | 14 (13.59) | 0.456 | 0.499 |
| Hemoglobin level | 13.17 ± 1.76 | 13.33 ± 1.85 | 0.641 | 0.522 |
| Blood glucose level | 96.34 ± 6.53 | 95.47 ± 6.18 | 1.019 | 0.309 |
Several significant differences were observed upon comparing postoperative gastrointestinal function recovery between the CC group and the FMEA group (Table 2). Surgical duration did not differ significantly between the two groups, with the CC and FMEA groups having mean durations of 149.67 ± 15.23 minutes and 147.49 ± 13.77 minutes, respectively (P = 0.265). However, notable differences were found in other key indicators. The length of hospital stay was significantly shorter in the FMEA group than in the CC group (4.49 ± 0.38 days vs 4.66 ± 0.42 days, P = 0.002). The time to first flatus was significantly shorter in the FMEA group (18.58 ± 1.86 hours vs 19.09 ± 1.93 hours, P = 0.045). Similarly, the time to first bowel movement was shorter in the FMEA group (48.16 ± 3.68 hours vs 49.35 ± 3.84 hours, P = 0.019). Additionally, the FMEA group tolerated a liquid diet sooner than the CC group (1.75 ± 0.35 days vs 1.86 ± 0.37 days, P = 0.030) and reached tolerance for solid diets more quickly (3.78 ± 0.41 days vs 3.95 ± 0.46 days, P = 0.004). The time to recovery of bowel sounds was significantly shorter in the FMEA group (23.93 ± 2.66 hours vs 25.07 ± 2.78 hours, P = 0.002).
| Indicator | CC group (n = 122) | FMEA group (n = 103) | t | P value |
| Surgical duration (mins) | 149.67 ± 15.23 | 147.49 ± 13.77 | 1.118 | 0.265 |
| Length of hospital stay (days) | 4.66 ± 0.42 | 4.49 ± 0.38 | 3.202 | 0.002 |
| Time to first flatus (hours) | 19.09 ± 1.93 | 18.58 ± 1.86 | 2.014 | 0.045 |
| Time to first bowel movement (hours) | 49.35 ± 3.84 | 48.16 ± 3.68 | 2.369 | 0.019 |
| Time to liquid diet tolerance (days) | 1.86 ± 0.37 | 1.75 ± 0.35 | 2.178 | 0.030 |
| Time to solid diet tolerance (days) | 3.95 ± 0.46 | 3.78 ± 0.41 | 2.913 | 0.004 |
| Time to bowel sound recovery (hours) | 25.07 ± 2.78 | 23.93 ± 2.66 | 3.120 | 0.002 |
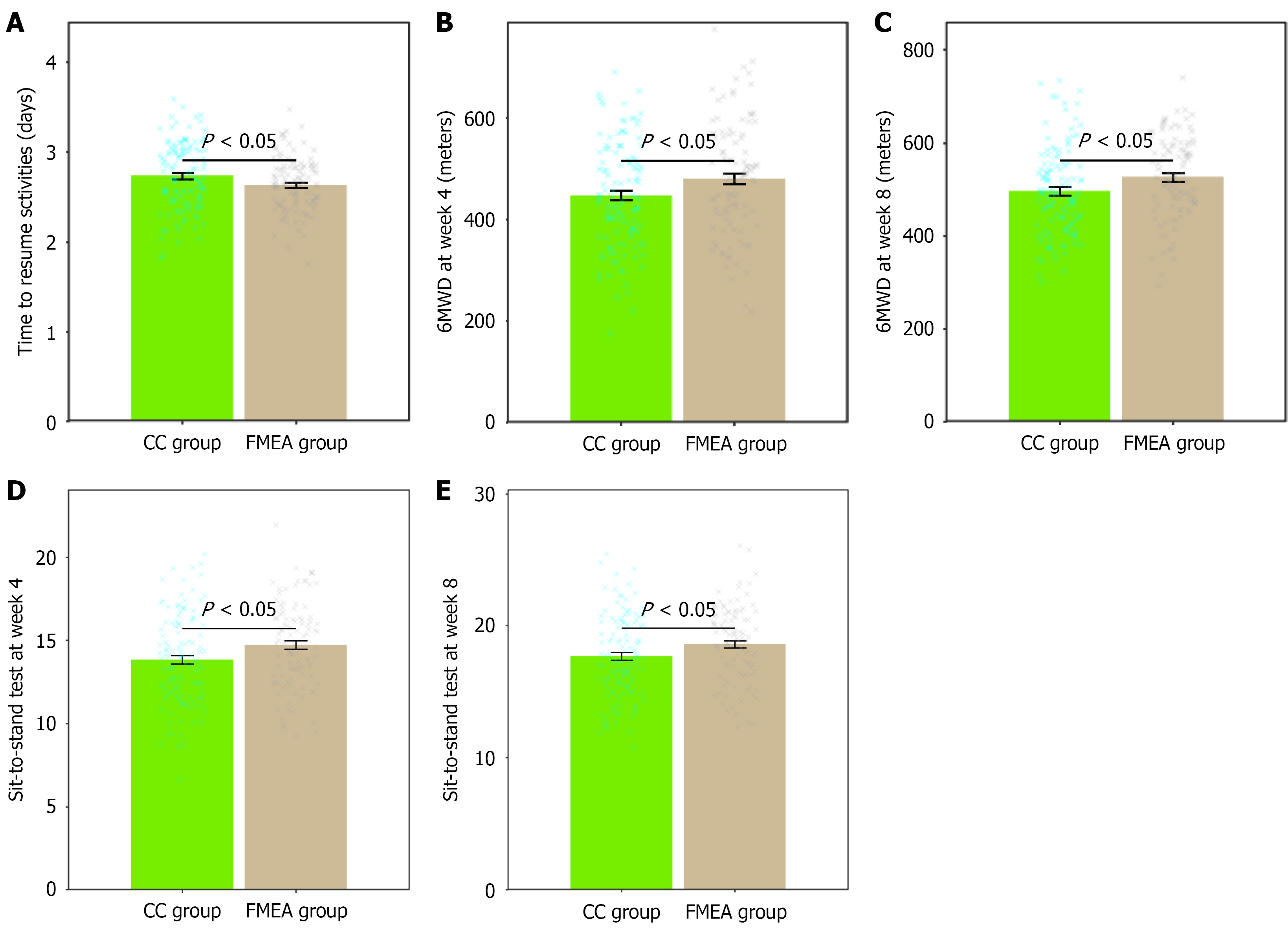
In evaluating postoperative exercise recovery between the CC group and the FMEA group, significant differences were observed in several key indicators (Figure 1). The time to resume normal activities was slightly shorter in the FMEA group than in the CC group (2.63 ± 0.31 days vs 2.73 ± 0.38 days, P = 0.023). At 4 weeks post-operation, the 6MWD was significantly greater in the FMEA group (480.12 ± 111.9 m) than in the CC group (446.72 ± 105.12 m, P = 0.022). This trend continued at 8 weeks, with the FMEA group again showing a higher 6MWD (526.16 ± 88.71 m vs 495.29 ± 94.82 m, P = 0.013). The sit-to-stand test performance favored the FMEA group. At 4 weeks, the FMEA group demonstrated better results (14.72 ± 2.59 repetitions) than the CC group (13.83 ± 2.72 repetitions, P = 0.013). Similarly, at 8 weeks, the FMEA group showed improved performance (18.58 ± 2.90 repetitions vs 17.69 ± 3.12 repetitions, P = 0.029).
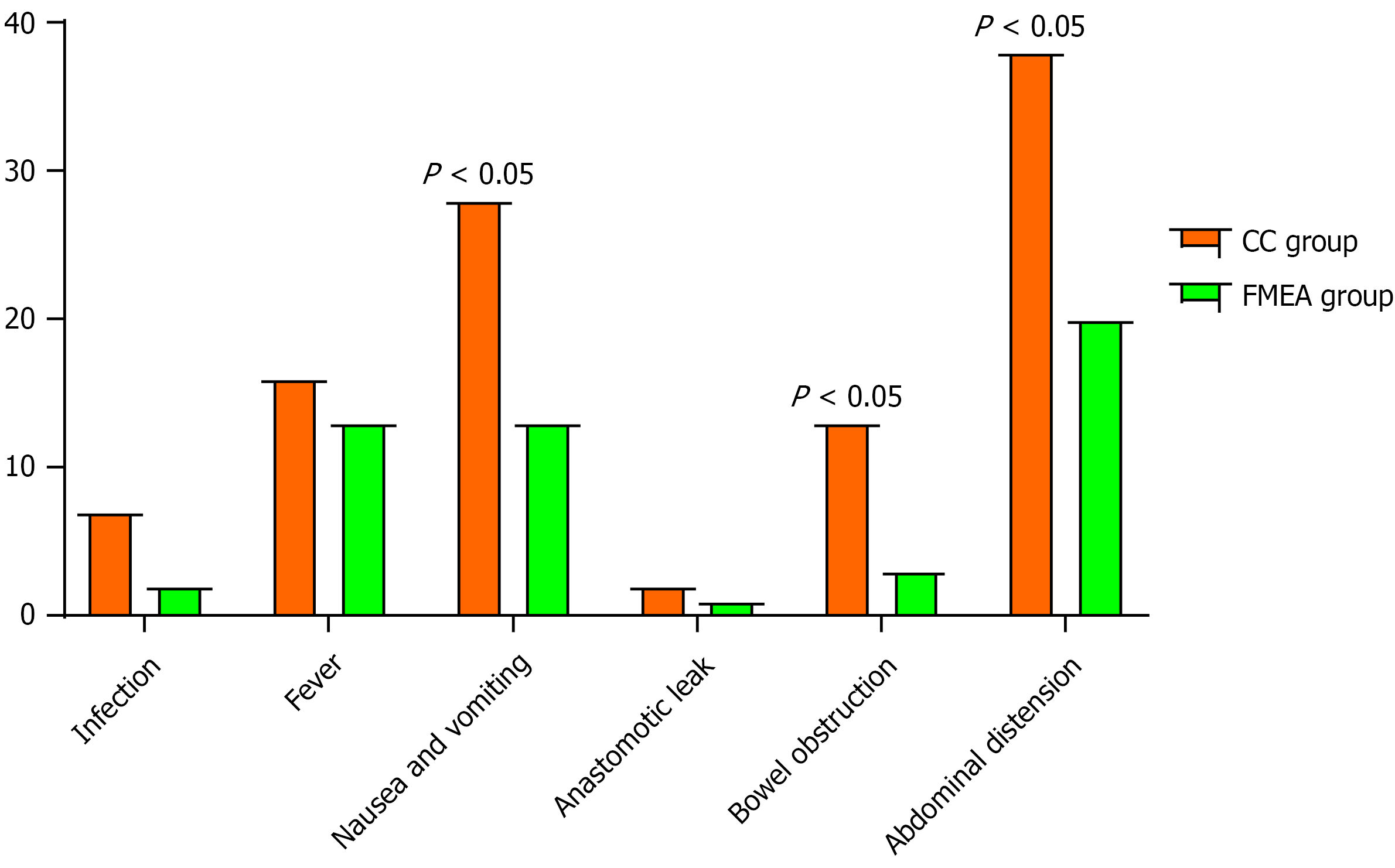
The postoperative complication occurrence between the CC group and the FMEA group showed several significant differences (Figure 2). For infections, no significant difference was found between the two groups, with rates of 5.74% in the CC group and 1.94% in the FMEA group (P = 0.269). Similarly, fever incidence was comparable, with 13.11% in the CC group and 12.62% in the FMEA group (P = 0.912). However, notable differences were found in other complications. The incidence of nausea and vomiting was significantly lower in the FMEA group than in the CC group (12.62% vs 22.95%, P = 0.046). Additionally, bowel obstruction occurred less frequently in the FMEA group (2.91% vs 10.66%, P = 0.024). Abdominal distension was less common in the FMEA group (19.42% vs 31.15%, P = 0.045). No significant difference was observed in the rate of anastomotic leak between the two groups (1.64% vs 0.97%, P = 1.000).
Evaluation of the preoperative comfort between the CC group and the FMEA group demonstrated no significant differences across various dimensions of comfort (Table 3). Specifically, the physiological comfort scores were similar between the CC group (15.27 ± 2.53) and the FMEA group (15.34 ± 2.42, P = 0.818). Psychological comfort showed no significant difference, with the CC group scoring 18.21 ± 2.13 and the FMEA group scoring 18.66 ± 2.08 (P = 0.113). Similarly, the social comfort scores did not differ significantly between the two groups, with the CC group at 19.59 ± 2.46 and the FMEA group at 19.71 ± 2.39 (P = 0.734). The environmental comfort scores were nearly identical, with the CC group scoring 21.39 ± 2.59 and the FMEA group scoring 21.34 ± 2.41 (P = 0.867). The total comfort scores for both groups were comparable, with the CC group at 74.47 ± 5.80 and the FMEA group at 75.05 ± 4.66 (P = 0.415).
| Indicator | CC group (n = 122) | FMEA group (n = 103) | t | P value |
| Physiological comfort | 15.27 ± 2.53 | 15.34 ± 2.42 | 0.231 | 0.818 |
| Psychological comfort | 18.21 ± 2.13 | 18.66 ± 2.08 | 1.590 | 0.113 |
| Social comfort | 19.59 ± 2.46 | 19.71 ± 2.39 | 0.340 | 0.734 |
| Environmental comfort | 21.39 ± 2.59 | 21.34 ± 2.41 | 0.168 | 0.867 |
| Total score | 74.47 ± 5.80 | 75.05 ± 4.66 | 0.816 | 0.415 |
Evaluation of postoperative comfort between the two groups showed significant differences across several dimensions of comfort (Table 4). The FMEA group reported significantly higher scores in physiological comfort (22.26 ± 2.31 vs 21.37 ± 2.42, P = 0.006). Similarly, psychological comfort was higher in the FMEA group (24.29 ± 2.59 vs 23.56 ± 2.75, P = 0.043). The social comfort scores were higher for the FMEA group than for the CC group (21.74 ± 2.07 vs 21.09 ± 2.16, P = 0.023). However, no significant difference was noted in the environmental comfort scores between the two groups (23.37 ± 2.41 vs 23.24 ± 2.37, P = 0.685). The total comfort score, which aggregates all dimensions, was significantly higher in the FMEA group than in the CC group (91.66 ± 4.71 vs 89.27 ± 4.87, P < 0.001). These results indicate that the FMEA group experienced better overall postoperative comfort across multiple dimensions, including physiological, psychological, and social aspects.
| Indicator | CC group (n = 122) | FMEA group (n = 103) | t | P value |
| Physiological comfort | 21.37 ± 2.42 | 22.26 ± 2.31 | 2.793 | 0.006 |
| Psychological comfort | 23.56 ± 2.75 | 24.29 ± 2.59 | 2.034 | 0.043 |
| Social comfort | 21.09 ± 2.16 | 21.74 ± 2.07 | 2.285 | 0.023 |
| Environmental comfort | 23.24 ± 2.37 | 23.37 ± 2.41 | 0.406 | 0.685 |
| Total score | 89.27 ± 4.87 | 91.66 ± 4.71 | 3.723 | < 0.001 |
Assessment of ADL before and after treatment between the CC group and the FMEA group revealed that the baseline ADL scores prior to any intervention were similar in both groups (Table 5). Specifically, the CC and FMEA groups had mean scores of 55.97 ± 3.92 and 55.88 ± 3.81, respectively, showing no significant difference (P = 0.863). However, the post-treatment ADL scores revealed notable differences. After treatment, the FMEA group demonstrated a significantly higher ADL score than the CC group (67.93 ± 3.19 vs 66.71 ± 3.35, P = 0.006). This finding indicates that the FMEA group experienced greater improvement in their ability to perform daily activities following the procedure.
| Indicator | CC group (n = 122) | FMEA group (n = 103) | t | P value |
| Before treatment | 55.97 ± 3.92 | 55.88 ± 3.81 | 0.173 | 0.863 |
| After treatment | 66.71 ± 3.35 | 67.93 ± 3.19 | 2.775 | 0.006 |
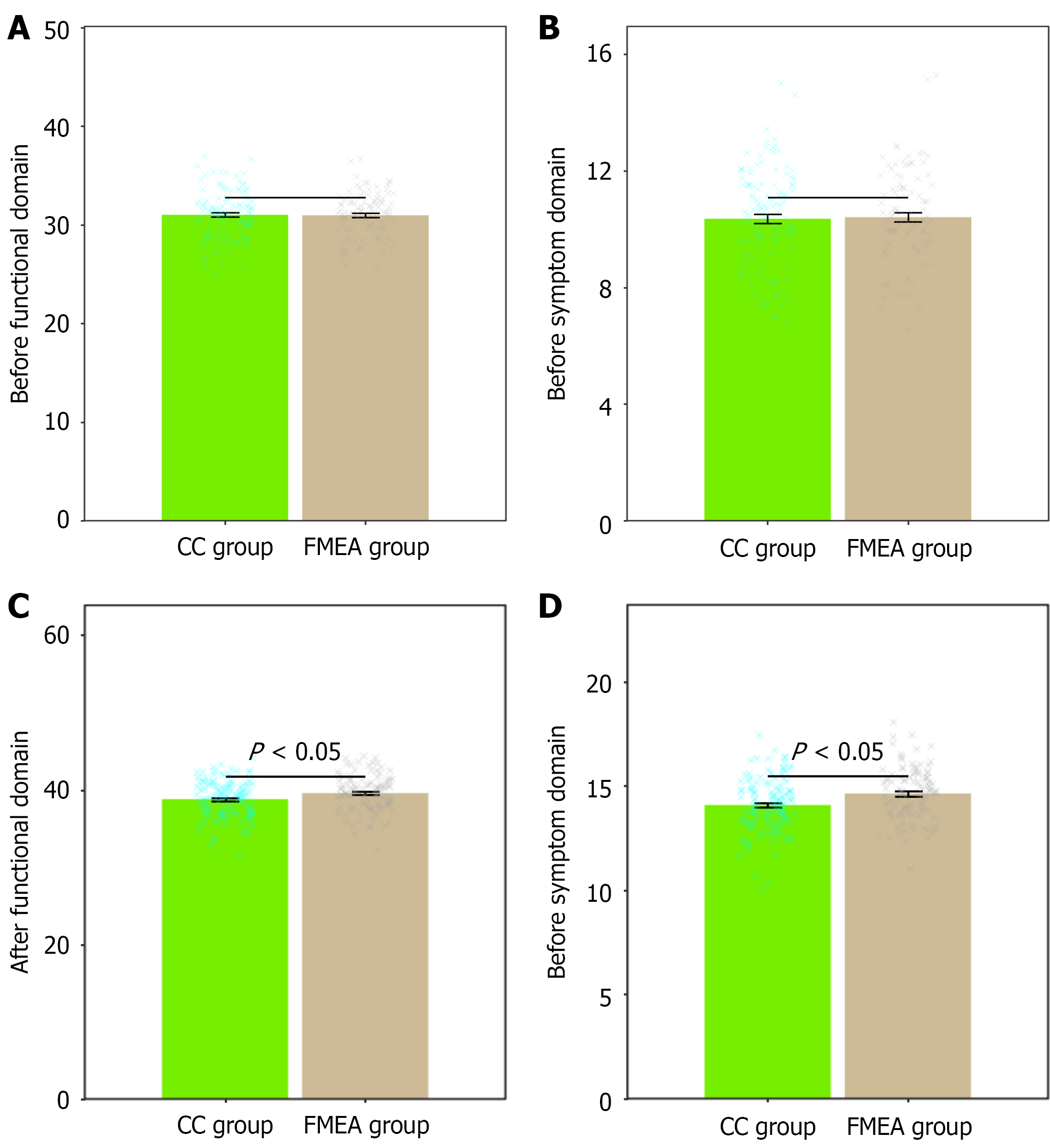
Functional and symptom domains were assessed before and after treatment between the two groups (Figure 3). At baseline, no significant differences were found in the quality of life scores between the two groups. For the functional domain, the CC and FMEA groups had mean scores of 31.02 ± 2.46 and 30.97 ± 2.28, respectively (P = 0.874). Similarly, for the symptom domain, the CC group scored 10.36 ± 1.68, and the FMEA group scored 10.41 ± 1.59 (P = 0.805). These results indicate that both groups started with comparable baseline quality of life scores. Post-treatment assessments revealed significant differences between the two groups. In the functional domain, the FMEA group showed a higher score than the CC group (39.56 ± 2.37 vs 38.78 ± 2.42, P = 0.015). In the symptom domain, the FMEA group also demon
In assessing patient care satisfaction between the CC group and the FMEA group, significant differences were observed in the levels of satisfaction reported by patients (Table 6). For patients reporting being “very satisfied”, a significantly higher proportion was noted in the FMEA group (61.17%) than in the CC group (47.54%, P = 0.041). In terms of “generally satisfied” responses, the proportion was slightly lower in the FMEA group (34.95%) than in the CC group (45.08%), but this difference did not reach statistical significance (P = 0.123). Additionally, fewer patients in the FMEA group reported being “dissatisfied” (3.88%) than in the CC group (7.38%), although this difference was also not statistically significant (P = 0.263).
| Indicator | CC group (n = 122) | FMEA group (n = 103) | χ2 | P value |
| Very satisfied | 58 (47.54) | 63 (61.17) | 4.170 | 0.041 |
| Generally satisfied | 55 (45.08) | 36 (34.95) | 2.380 | 0.123 |
| Dissatisfied | 9 (7.38) | 4 (3.88) | 1.252 | 0.263 |
Compared with traditional care, an FMEA-based care plan for patients undergoing laparoscopic extended radical colectomy improves postoperative recovery by accelerating the return of basic physiological functions, reducing compli
The accelerated gastrointestinal recovery in the FMEA group is a key finding, marked by early return of bowel sounds, flatus, and defecation, and shortened time to tolerate liquid and solid diets. By planning the postoperative care pathway and using FMEA to identify key risk factors, the care team can proactively intervene against factors, such as inadequate pain control leading to opioid overuse, poor fluid management, delayed mobilization, and lack of standardized chewing gum protocols, before complications arise, thereby mitigating intestinal motility disorders[21]. Early passage of gastroin
In terms of functional recovery assessment, the results of the 6MWD test and the sit-to-stand test provide objective evidence of the benefits of the FMEA protocol. The 6MWD test is a standard method for assessing submaximal exercise endurance, directly reflecting the patient’s ability to perform daily physical activities[27]. The increase in walking distance in the FMEA group indicates enhanced cardiopulmonary function and overall physical recovery, enabling patients to better perform daily activities such as prolonged walking. The sit-to-stand test specifically assesses lower limb muscle strength, balance, and the basic action of transitioning from sitting to standing, which is closely related to key life activities such as toileting and transferring[28]. Improvements in this test in the FMEA group demonstrate enhanced fundamental motor functions and independent living abilities of the patients. The improvement can be attributed to several factors. First, faster recovery of gastrointestinal function and reduced complications like nausea and abdominal distension enable patients to engage more fully in early activities. Second, the FMEA process addresses barriers to rehabilitation, such as fear of pain or wound complications, by enhancing education, support, and pain management[29]. The FMEA protocol promotes sustained functional recovery by optimizing each step from bed rest to ambulation. The present study extends previous research, showing that FMEA not only prevents errors but also actively enhances patient outcomes, including physical rehabilitation[30]. While FMEA has been used to improve physical therapy safety, this study demonstrates its direct benefits on patient-level functional indicators.
The FMEA cohort had lower incidences of complications like nausea, vomiting, ileus, and abdominal distension than the CC cohort. For nausea and vomiting, FMEA involves reviewing perioperative drug administration, anesthesia techniques, and dietary protocols to develop enhanced preventive strategies[31]. This reduces risks from improper opioid use and mechanical factors like inadequate bowel preparation or delayed mobilization[32]. Notably, the reduction in ileus is evident because it can be influenced by surgical techniques and postoperative care, including early activity and careful opioid use, which prevent adhesions and obstruction[33]. These findings align with FMEA’s success in reducing adverse events in other high-risk clinical processes such as chemotherapy and blood transfusion[34].
The FMEA model enhances patient comfort through systematic risk assessment across physical, psychological, social, and environmental dimensions. Improved symptom control and accelerated gastrointestinal recovery reduce pain, and clear risk communication and proactive guidance lower anxiety and boost a sense of control[35]. Family involvement in educational sessions strengthens support networks, and better bedside ergonomics, along with timely equipment checks, create a safer care environment. These improvements align with early oncology care research, showing that comprehensive risk assessment can enhance patient comfort[36]. Within the FMEA framework, ADL and overall quality of life improve as functional independence reduces psychological burden and increases health satisfaction[37]. Fewer complications also mean less need for additional interventions, thus preserving physical and mental health. FMEA offers greater improvements in ADL and quality of life than ERAS protocols.
Patient satisfaction was higher in the FMEA group due to a faster, smoother recovery process and more comprehensive support, which improved functional independence. The FMEA approach emphasizes process optimization and failure prevention, offering a coherent, predictable, and patient-centered care experience with fewer delays, clearer com
Despite these encouraging findings, several limitations affect the interpretation of the results. The retrospective and single-center design introduces the possibility of selection bias and limits the generalizability of the results. The inherent limitations of a retrospective review mean that unmeasured confounding factors may influence the outcomes. The relatively short follow-up period captured the immediate and early recovery phases but did not provide information on the long-term sustainability of the observed benefits, such as sustained functional advantages or long-term complication rates. A formal cost-effectiveness analysis of the FMEA protocol implementation was not conducted, which could be valuable for hospital administrators.
Future research should aim to validate these results in multicenter, prospective randomized trials with long-term follow-up to assess the durability of the benefits. Additionally, investigating the specific components of the FMEA protocol that have the most impact could help refine and streamline interventions. Finally, integrating a cost-effectiveness analysis could determine whether the resource investment required for comprehensive failure mode assessment can yield favorable economic returns for the health system.
The FMEA nursing model significantly enhances functional recovery, reduces complications, and improves quality of life in patients with CRC undergoing laparoscopic extended colectomy. Its implementation represents a clinically valuable strategy for optimizing postoperative rehabilitation within enhanced recovery protocols.
| 1. | Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66:683-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3978] [Cited by in RCA: 3562] [Article Influence: 395.8] [Reference Citation Analysis (8)] |
| 2. | Hossain MS, Karuniawati H, Jairoun AA, Urbi Z, Ooi J, John A, Lim YC, Kibria KMK, Mohiuddin AKM, Ming LC, Goh KW, Hadi MA. Colorectal Cancer: A Review of Carcinogenesis, Global Epidemiology, Current Challenges, Risk Factors, Preventive and Treatment Strategies. Cancers (Basel). 2022;14:1732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 744] [Cited by in RCA: 572] [Article Influence: 143.0] [Reference Citation Analysis (0)] |
| 3. | Lavikainen LI, Guyatt GH, Sallinen VJ, Karanicolas PJ, Couban RJ, Singh T, Lee Y, Elberkennou J, Aaltonen R, Ahopelto K, Beilmann-Lehtonen I, Blanker MH, Cárdenas JL, Cartwright R, Craigie S, Devereaux PJ, Garcia-Perdomo HA, Ge FZ, Gomaa HA, Halme ALE, Haukka J, Karjalainen PK, Kilpeläinen TP, Kivelä AJ, Lampela H, Mattila AK, Najafabadi BT, Nykänen TP, Pandanaboyana S, Pourjamal N, Ratnayake CBB, Raudasoja A, Vernooij RWM, Violette PD, Wang Y, Xiao Y, Yao L, Tikkinen KAO; ROTBIGGS Investigators. Systematic Reviews and Meta-analyses of the Procedure-specific Risks of Thrombosis and Bleeding in General Abdominal, Colorectal, Upper Gastrointestinal, and Hepatopancreatobiliary Surgery. Ann Surg. 2024;279:213-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 4. | Kim DS, Risca VI, Reynolds DL, Chappell J, Rubin AJ, Jung N, Donohue LKH, Lopez-Pajares V, Kathiria A, Shi M, Zhao Z, Deep H, Sharmin M, Rao D, Lin S, Chang HY, Snyder MP, Greenleaf WJ, Kundaje A, Khavari PA. The dynamic, combinatorial cis-regulatory lexicon of epidermal differentiation. Nat Genet. 2021;53:1564-1576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 5. | Małczak P, Mizera M, Torbicz G, Witowski J, Major P, Pisarska M, Wysocki M, Strzałka M, Budzyński A, Pędziwiatr M. Is the laparoscopic approach for rectal cancer superior to open surgery? A systematic review and meta-analysis on short-term surgical outcomes. Wideochir Inne Tech Maloinwazyjne. 2018;13:129-140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Vilsan J, Maddineni SA, Ahsan N, Mathew M, Chilakuri N, Yadav N, Munoz EJ, Nadeem MA, Abbas K, Razzaq W, Abdin ZU, Ahmed M. Open, Laparoscopic, and Robotic Approaches to Treat Colorectal Cancer: A Comprehensive Review of Literature. Cureus. 2023;15:e38956. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 7. | Song JH, Shin Y, Lee KH, Kim JY, Kim JS. Correlation between inflammatory markers and enhanced recovery after surgery (ERAS) failure in laparoscopic colectomy. Surg Today. 2025;55:1353-1360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 8. | Chung SL, Li W, Wang Q, Qiu X, Tang Y, Hu S, Wu H, Jin Z. Clinical study of electroacupuncture on the recovery of gastrointestinal dysfunction after laparoscopic surgery for gastrointestinal cancer - study protocol for a randomized controlled trial. BMC Complement Med Ther. 2024;24:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Jain SN, Lamture Y, Krishna M. Enhanced Recovery After Surgery: Exploring the Advances and Strategies. Cureus. 2023;15:e47237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 10. | Sölkner L, Georg D, Wolff U, Renner A, Widder J, Heilemann G. Enhancing clinical safety in radiation oncology: A data-driven approach to risk management. Z Med Phys. 2025;35:408-415. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Mora A, Ayala L, Bielza R, Ataúlfo González F, Villegas A. Improving safety in blood transfusion using failure mode and effect analysis. Transfusion. 2019;59:516-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 12. | Ilkic J, Milovanovic M, Marinkovic V. Prospective systematic risk analysis of the digital technology use within pharmaceutical care. J Am Pharm Assoc (2003). 2024;64:102081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 13. | Diagnosis And Treatment Guidelines For Colorectal Cancer Working Group CSOCOC. Chinese Society of Clinical Oncology (CSCO) diagnosis and treatment guidelines for colorectal cancer 2018 (English version). Chin J Cancer Res. 2019;31:117-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 157] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 14. | Ratto C, Ricci R. Potential pitfalls concerning colorectal cancer classification in the seventh edition of the AJCC Cancer Staging Manual. Dis Colon Rectum. 2011;54:e232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166:111-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9595] [Cited by in RCA: 8516] [Article Influence: 354.8] [Reference Citation Analysis (1)] |
| 16. | Guralnik JM, Simonsick EM, Ferrucci L, Glynn RJ, Berkman LF, Blazer DG, Scherr PA, Wallace RB. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85-M94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7796] [Cited by in RCA: 7046] [Article Influence: 220.2] [Reference Citation Analysis (1)] |
| 17. | Kolcaba KY. Holistic comfort: operationalizing the construct as a nurse-sensitive outcome. ANS Adv Nurs Sci. 1992;15:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 135] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Mahoney FI, Barthel DW. Functional evaluation: The Barthel index. Md State Med J. 1965;14:61-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 804] [Cited by in RCA: 433] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 19. | Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman SB, de Haes JC. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12937] [Cited by in RCA: 11806] [Article Influence: 357.8] [Reference Citation Analysis (3)] |
| 20. | Shaikh BT, Mobeen N, Azam SI, Rabbani F. Using SERVQUAL for assessing and improving patient satisfaction at a rural health facility in Pakistan. East Mediterr Health J. 2008;14:447-456. [PubMed] |
| 21. | Iskander O. An outline of the management and prevention of postoperative ileus: A review. Medicine (Baltimore). 2024;103:e38177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Yu S, Kerolus K, Jin Z, Bajrami S, Denoya P, Bergese SD. Multidisciplinary Postoperative Ileus Management: A Narrative Review. Medicina (Kaunas). 2025;61:1344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 23. | Canzan F, Longhini J, Caliaro A, Cavada ML, Mezzalira E, Paiella S, Ambrosi E. The effect of early oral postoperative feeding on the recovery of intestinal motility after gastrointestinal surgery: a systematic review and meta-analysis of randomized clinical trials. Front Nutr. 2024;11:1369141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 24. | Frankel L, Maurente D, Ardeljan AD, Divesh M, Rashid AM, Takabe K, Rashid OM. Improving Gastrointestinal Cancer Care by Enhanced Recovery Protocol Implementation. World J Oncol. 2023;14:135-144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 25. | Zhou Y, Yin ZH, Sun MS, Wang YY, Yang C, Li SH, Liang FR, Liu F. Global research trends in postoperative ileus from 2011 to 2023: A scientometric study. World J Gastrointest Surg. 2024;16:3020-3031. [PubMed] [DOI] [Full Text] |
| 26. | Vecchia M, Sacchi P, Marvulli LN, Ragazzoni L, Muzzi A, Polo L, Bruno R, Salio F. Healthcare Application of Failure Mode and Effect Analysis (FMEA): Is There Room in the Infectious Disease Setting? A Scoping Review. Healthcare (Basel). 2025;13:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Wu Y, Ren B, Li J, Chi L, Li P, Wu J. Physical performance on the early 6-minute walk test in coronary artery bypass grafting patients during inpatient cardiac rehabilitation. Front Cardiovasc Med. 2025;12:1580442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Kronberger C, Mousavi RA, Ermolaev N, Willixhofer R, Rassoulpour N, Kaya MB, Poledniczek M, Eslami M, Krall C, Litschauer B, Grzeda MT, McKenna I, Badr Eslam R. One-minute sit-to-stand test cut-offs for risk stratification based on six-minute walk test thresholds in pulmonary hypertension. Eur J Prev Cardiol. 2025;32:1517-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 29. | Yang Y. Impact of FMEA-Based Nursing on Postoperative Condition of Appendicitis Patients. Altern Ther Health Med. 2025;31:343-347. [PubMed] |
| 30. | Wu C, Tian Q, Wang H, Yang W, Liu A, Tang J, Nie X, Pu R, Huang Q. Failure mode and effects analysis-based strategies for controlling multidrug-resistant organism infections in cancer patients. Sci Rep. 2024;14:28564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 31. | Abrishami R, Golestani K, Farhang Ranjbar M, Ghasemie Abarghouie MH, Ghadami A. A survey on the effects of patient safety training programs based on SBAR and FMEA techniques on the level of self-efficacy and observance of patient safety culture in Iran hospital, Shiraz in 2022-2023. J Educ Health Promot. 2024;13:66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 32. | Yu L, Dong Y, Shi S, Liu X, Wang M, Jiang G. Analysis of postoperative nausea and vomiting in patients with lung cancer undergoing thoracoscopic surgery under general anesthesia and its influencing factors: a observational study. BMC Surg. 2024;24:316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 33. | Reichert M, Willis F, Post S, Schneider M, Vilz T, Willis M, Hecker A. Pharmacologic prevention and therapy of postoperative paralytic ileus after gastrointestinal cancer surgery: systematic review and meta-analysis. Int J Surg. 2024;110:4329-4341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 34. | Joly-Mischlich T, Maltais S, Tétu A, Delorme MN, Boilard B, Pavic M. Application of the Failure Mode and Effects Analysis (FMEA) to identify vulnerabilities and opportunities for improvement prior to implementing a computerized prescription order entry (CPOE) system in a university hospital oncology clinic. J Oncol Pharm Pract. 2023;29:88-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Chen Q, Chen S, Zhang L. Impact of FMEA-based nursing intervention combined with early rehabilitation training on the prognosis of mechanically ventilated patients in cardiac intensive care units. Medicine (Baltimore). 2025;104:e41735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 36. | Berumen KS, Sánchez-Navarro A, Velázquez A, Solano M, Anaya F, Rosales G, Díaz A, Jazer J, Zamudio K, Suder S, Quintero M, González-Martínez A, García LA. Multispecialty sessions model for comprehensive care and decision-making in cancer patients. Ecancermedicalscience. 2025;19:1830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Kim LH, Juneja BR, Viscariello NN. A method for empirically validating FMEA RPN scores in a radiation oncology clinic using physics QC data. J Appl Clin Med Phys. 2024;25:e14391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 38. | Micheletta F, Ferrara M, Bertozzi G, Volonnino G, Nasso M, La Russa R. Proactive Risk Assessment through Failure Mode and Effect Analysis (FMEA) for Perioperative Management Model of Oral Anticoagulant Therapy: A Pilot Project. Int J Environ Res Public Health. 2022;19:16430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Kornek D, Lotter M, Szkitsak J, Dürrbeck C, Karius A, Ott OJ, Brandl C, Bert C. Improving the safety of radiotherapy treatment processes via incident-driven FMEA feedback loops. J Appl Clin Med Phys. 2024;25:e14455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 40. | LATAM collaborative colorectal surgery consortium; Viola Malet M. Short-term surgical outcomes of rectal adenocarcinoma surgical treatment in Latin America: a multicenter, retrospective assessment in 49 centers from 12 countries. Int J Colorectal Dis. 2024;39:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/