Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.113476
Revised: November 17, 2025
Accepted: December 22, 2025
Published online: February 27, 2026
Processing time: 135 Days and 1.6 Hours
Previous studies have confirmed the association of type 2 diabetes mellitus with the occurrence of colorectal polyps and adenomas. However, the current research results concerning the extent and specific effect of type 2 diabetes mellitus on the occurrence and development of colorectal adenomas remain inconsistent.
To identify and investigate the association between type 2 diabetes mellitus and colorectal adenoma pathogenesis.
This retrospective case-control study reviewed all colonoscopies and definite pathology diagnoses performed in patients admitted to the Department of Gas
Type 2 diabetes mellitus was significantly associated with colorectal adenomas (odds ratio = 2.263, 95% confidence interval = 1.252-4.09, P = 0.007). Compared with the A + ND or NA + ND group, carcinoembryonic antigen and insulin-like growth factor-1 levels and TCF7L2, β-catenin, and Ki67 expre
The results of this small-scale retrospective cohort study indicate a significant association between type 2 diabetes mellitus and colorectal adenomas. The clinical sample experimental evidence in this study indicates that the increased activity and expression of Wnt/β-catenin/TCF7L2 is one of the mechanisms of type 2 diabetes mellitus and colorectal adenomas.
Core Tip: This small-scale retrospective cohort study confirmed that type 2 diabetes mellitus is a significant independent risk factor for colorectal adenomas. The results of this study indicate that Wnt/β-catenin/transcription factor 7 like 2 pathway activation is considered a common molecular pathological basis in the association of type 2 diabetes mellitus with colorectal adenoma occurrence and development. The results of this study support the inclusion of colonoscopy screening in the routine clinical management pathway for patients with type 2 diabetes mellitus.
- Citation: Gui SY, Tang RX, Huang X, Yang GQ, Yang J. Association between type 2 diabetes mellitus and colorectal adenoma: A retrospective study with insights into Wnt/β-Catenin/TCF7L2 pathway activation. World J Gastrointest Surg 2026; 18(2): 113476
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/113476.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.113476
Diabetes mellitus is a group of clinical metabolic disorders characterized by hyperglycemia. A state of chronic hyper
At present, the incidence of colorectal cancer (CRC) is increasing annually, accounting for the second to third of the malignant tumors in Western countries, second only to breast cancer and lung cancer, and is one of the most prevalent malignant tumors in clinical practice. In the United States, 140000 new cases of colon cancer and 50000 deaths are reported annually[2]. Compared with developed countries, developing countries reported a higher incidence of CRC. In China, the number of new CRC cases exceeds 250000, and death cases are approximately 140000 annually, accounting for 20% of all CRC cases globally[3]. Jiang et al[4] conducted a meta-analysis of 41 related group studies with a median follow-up of 10.5 years, and the results revealed that type 2 diabetes mellitus was associated with an increased CRC incidence (standardized rate ratio = 1.27). The occurrence of CRC is related to factors, including genetics, age, and sex. However, most CRCs are sporadic, and several studies have confirmed that CRC is closely associated with diet, overweight, and obesity. Western dietary patterns characterized by high amounts of meat, fat, and sweets are associated with an increased risk of CRC. In general, colorectal polyps, especially colorectal adenomas, are one of the major precancerous lesions of CRC. The vast majority of CRCs are developed due to the slow progression of colorectal adenomas. Investigating the risk factors of colorectal adenoma and treating colorectal adenoma early is the key to preventing CRC and reducing mortality.
At present, controversy regarding the research on type 2 diabetes mellitus and colorectal adenoma persists, and the biological connection between the two remains unclear. Budzynska et al[5] conducted a retrospective cohort study and revealed no significant association between diabetes mellitus and colorectal adenoma after controlling for confounding factors, including age, gender, and body mass index. Meanwhile, Soltani et al[6] found that the prevalence of combined diabetes mellitus and colorectal adenoma was significantly correlated in a study involving 693 patients. Yu et al[7] conducted a meta-analysis, comprising 17 related studies, and the results indicated that patients with type 2 diabetes mellitus had approximately 5% higher risk of colorectal adenoma than newly diagnosed patients, indicating that long-term type 2 diabetes mellitus is a risk factor for colorectal adenomas. Patients with long-term type 2 diabetes mellitus are speculated to be exposed to a longer period of hyperinsulinemia/insulin resistance and hyperglycemia, and such long-term hormonal or metabolic abnormalities increase the probability of intestinal epithelial atypia. Studies have revealed that the Wnt signaling pathway and the interaction between insulin and the Wnt pathway can decrease the phosphorylation of serine (Ser) 33/Ser 37 sites and increase the phosphorylation of Ser675 sites of β-catenin in patients with type 2 diabetes mellitus, thereby accumulating β-catenin in colorectal epithelium and promoting colorectal epithelial proliferation[8]. In animal models, hyperglycemia without insulin treatment still induced intestinal epithelial cell proliferation, which could be positively regulated via the Wnt/β-catenin pathway and was considered necessary for the nuclear localization of β-catenin[9]. Transcription factor 7 like 2 (TCF7 L2) is not only a key transcription factor in the Wnt pathway but also a genetic susceptibility locus for type 2 diabetes mellitus; thus, it is worthy of in-depth research in the context of adenoma pathogenesis[10].
The association between diabetes and CRC has been clearly established; however, the mechanism of their association with precancerous adenomas remains insufficiently studied. In particular, the role of the Wnt pathway in this process remains systematically unverified in clinical samples. In this study, the author aims to retrospectively analyze the clinical data of patients with type 2 diabetes mellitus and colorectal adenoma, group and select CRC pathological tissue sections of patients, and analyze the expression of key proteins, such as β-catenin and TCF7 L2, in the Wnt signaling pathway, to investigate the association between type 2 diabetes mellitus and the pathogenesis of colorectal adenoma with immunohistochemistry.
The clinical data of patients who underwent colorectal polypectomy in the Department of Gastroenterology, Nanshan People’s Hospital, from January 2015 to December 2017, and had pathological biopsy and report, were retrospectively analyzed. The adenoma group comprised patients with a pathological diagnosis of colorectal adenoma (including tubular adenoma, villous adenoma, and tubular villous adenoma). Two senior pathologists confirmed the pathological diagnosis in strict accordance with the World Health Organization digestive system tumor criteria in 2000, and graded them according to their pathological criteria. Exclusion criteria include patients with a history of CRC or pathological reports indicating colorectal malignancy.
The control group comprised patients who underwent colonoscopy during the same period, with matching age, sex, and length of hospital stay to those of the adenoma group, and with pathological replacement indicating no colorectal adenoma (including hyperplastic polyps or other nonspecific inflammation). All patients should have complete clinical data (including complete hospitalization history, clear pathological diagnosis, and corresponding paraffin-embedded samples). This study adhered to the ethical principles of the Declaration of Helsinki.
In the adenoma group, study group 1 (A + D) comprised 20 tubular adenoma cases with type 2 diabetes mellitus, whereas study group 2 (A + ND) included 20 tubular adenoma cases without type 2 diabetes mellitus, and the control group (NA + ND) involved 10 cases without diabetes mellitus and tubular adenoma. Fasting venous blood samples from all participants were collected and stored at -80 °C. Serum carcinoembryonic antigen (CEA) levels were measured using electrochemiluminescence immunoassay. Serum insulin-like growth factor-1 (IGF-1) levels were quantified with chemiluminescent immunoassay. All assays were performed strictly following the manufacturers’ protocols. The paraffin blocks of patients were borrowed from the department of pathology for immunohistochemistry.
Abcam supplied rabbit anti-TCF7 L2 polyclonal antibody and rabbit anti-β-catenin monoclonal antibody. Wuhan Boster Bioengineering Co., Ltd. (China), provided the anti-Ki-67 antibody.
The presence of Ki67, TCF7 L2, and β-catenin proteins were detected with immunohistochemistry. The conventional paraffin blocks were cut into 3.5-μm-thick slices, and histomorphological characteristics were observed under a light microscope. All pathological sections were stained employing the immunohistochemistry streptavidin-peroxidase method. The known positive sections were used as a positive control, and phosphate buffer solution was applied instead of primary antibody as a negative control. Diaminobenzidine was used as a substrate for detection of horse horseradish peroxidase-conjugated secondary antibody to identify the targeted protein in the sections, which were observed using a microscope. Both anti-TCF7 L2 and anti-β-catenin antibodies were used at a concentration of 1:500.
To minimize errors and ensure the consistency of data results, two senior pathologists from our hospital independently evaluated the immunohistochemical results of all pathological sections. These two pathologists were unaware of the patient’s group classification and clinical data. In case of differing opinions, they would negotiate to reach a consensus. The TCF7 L2 staining was observed in the nucleus, and the positive criterion considered was a clear background with a brown-yellow nucleus. β-catenin staining was mainly localized in the cell membrane of mucosal epithelium cells and was positively expressed in the cytoplasm or nucleus. Analysis software included Image-Pro Plus version 6.0 (Media Cybernetics, Inc., Rockville, MD, United States).
Immunohistochemical average optical (AO) analysis method: At least three visual fields (× 200) were randomly selected for each group of slices. When taking pictures, the field of view encompassed the tissue, and the background light was ensured to be consistent for each photo. Image-Pro Plus version 6.0 software was used to select images with the same color, and the same brown-yellow color was utilized as the unified standard to judge the positivity of all photos. The positive integrated optical density value and the pixel area of the tissue were analyzed for each photo, and the AO value was calculated. The higher the AO value, the higher the positive expression level (AO = integrated optical density/area)[11].
Quantitatively measured data were tested for normality and expressed as mean ± SD. Quantitatively enumerated data were expressed as a percentage (%) or constituent ratio. Pearson’s χ2 test was conducted for between-group comparisons of categorical variables. An independent t-test was used to compare normally distributed measurement data between two groups, and one-way ANOVA was used for comparisons among multiple groups. Binary logistic regression analysis was conducted to identify independent risk factors for colorectal adenomas. The model included the following covariates: Gender, age (years), and history of type 2 diabetes mellitus. The results are presented as adjusted odds ratios with their corresponding 95% confidence intervals. The variance inflation factor was used to assess the multicollinearity among independent variables, and all variance inflation factor values were below 2.0, indicating no significant multicollinearity. Statistical Package for the Social Sciences version 19.0 was used for all statistical analyses. A two-sided P value of < 0.05 indicated statistical significance.
The patients were categorized according to pathological and clinical diagnosis, comprising 315 cases and 103 cases in the adenoma and control groups, respectively. Table 1 shows the specific basic information. Univariate regression and then multivariate regression analyses were conducted for clinical data analyses. Type 2 diabetes mellitus is an independent risk factor for colorectal adenoma. After normalizing for factors such as sex and age, patients in the A + D group demonstrated a 2.263 times greater risk of colorectal adenoma than those in the A + ND group (Tables 2 and 3).
| Control group | Adenoma group | t/χ2 | P value | |
| Number | 103 | 315 | ||
| Gender | ||||
| Male | 55 (53.4) | 202 (64.1) | 3.773 | 0.052 |
| Female | 48 (46.6) | 113 (35.9) | ||
| Age (years) | 52.70 ± 14.35 | 54.97 ± 12.74 | -1.525 | 0.128 |
| History of diabetes | 16 (15.5) | 97 (30.8) | 9.163 | 0.002 |
| History of hypertension | 19 (18.4) | 66 (21.0) | 0.301 | 0.583 |
| Hyperuricemia | 1 (1.0) | 6 (1.9) | 0.040 | 0.842 |
| Hyperlipidemia | 5 (4.9) | 24 (7.6) | 0.919 | 0.338 |
| B | SE | Wald | OR | 95%CI | P value | |
| Gender | 0.445 | 0.230 | 3.745 | 1.560 | 0.994-2.448 | 0.053 |
| Age (years) | 0.013 | 0.009 | 2.309 | 1.013 | 0.996-1.03 | 0.129 |
| History of diabetes | 0.884 | 0.298 | 8.782 | 2.419 | 1.349-4.34 | 0.003 |
| History of hypertension | 0.159 | 0.289 | 0.300 | 1.172 | 0.665-2.066 | 0.584 |
| Hyperuricemia | 0.683 | 1.086 | 0.396 | 1.981 | 0.236-16.646 | 0.529 |
| Hyperlipidemia | 0.480 | 0.505 | 0.903 | 1.616 | 0.6-4.352 | 0.342 |
| B | SE | Wald | OR | 95%CI | P value | |
| Gender | 0.446 | 0.234 | 3.637 | 1.562 | 0.988-2.471 | 0.057 |
| Age (years) | 0.011 | 0.009 | 1.498 | 1.011 | 0.994-1.028 | 0.221 |
| History of diabetes | 0.817 | 0.302 | 7.312 | 2.263 | 1.252-4.09 | 0.007 |
Serum CEA and IGF-1 levels of each group of patients were measured. The analysis revealed higher CEA and IGF-1 levels in the A + D group than in the A + ND and NA + ND groups (P < 0.05; Table 4).
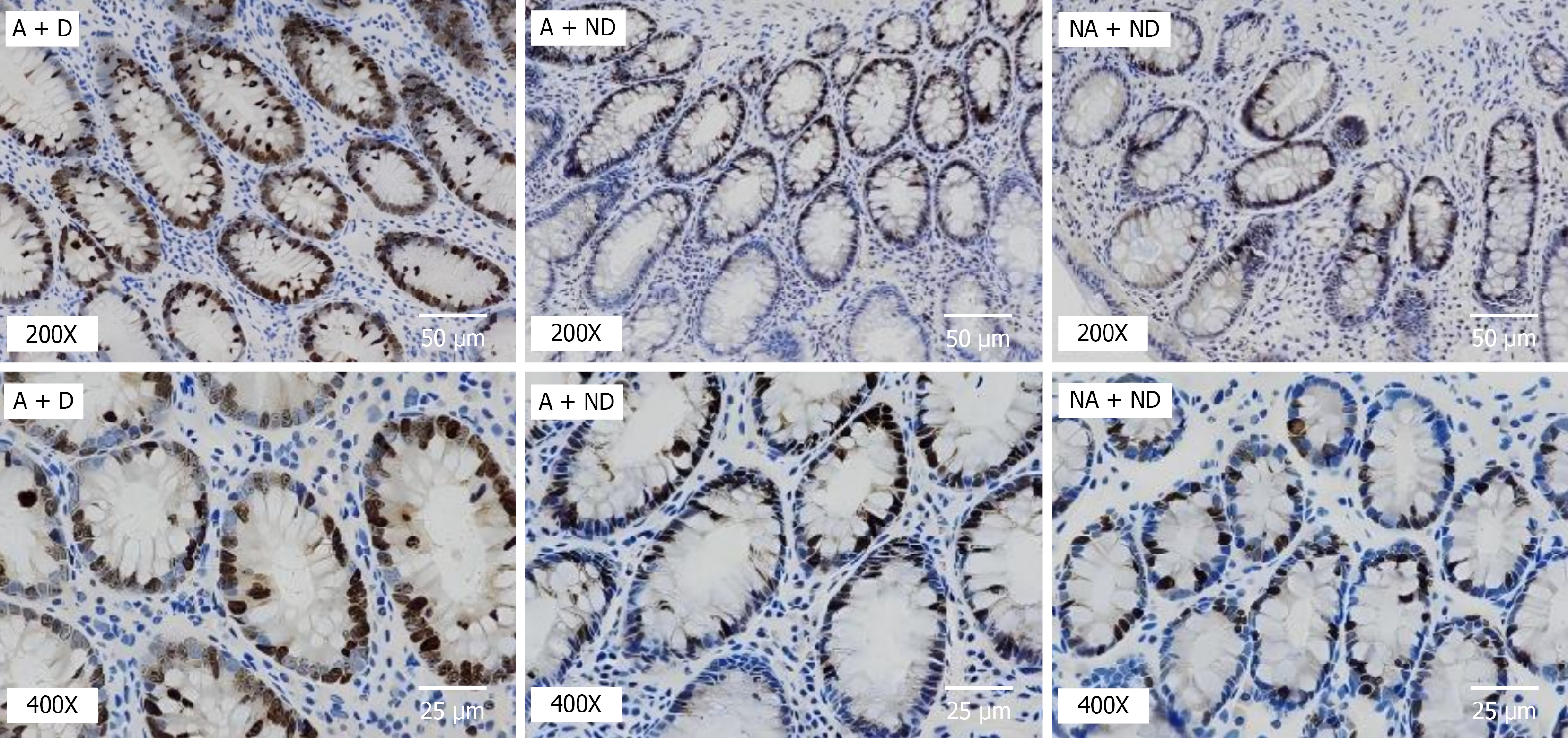
The cell proliferation rate of each group was evaluated with Ki67 staining, and positive Ki67 staining indicated proliferating cells (Figure 1). The intestinal epithelial cell proliferation rate in the A + D group was higher than that in the A + ND and NA + ND groups (P < 0.05; Table 5).
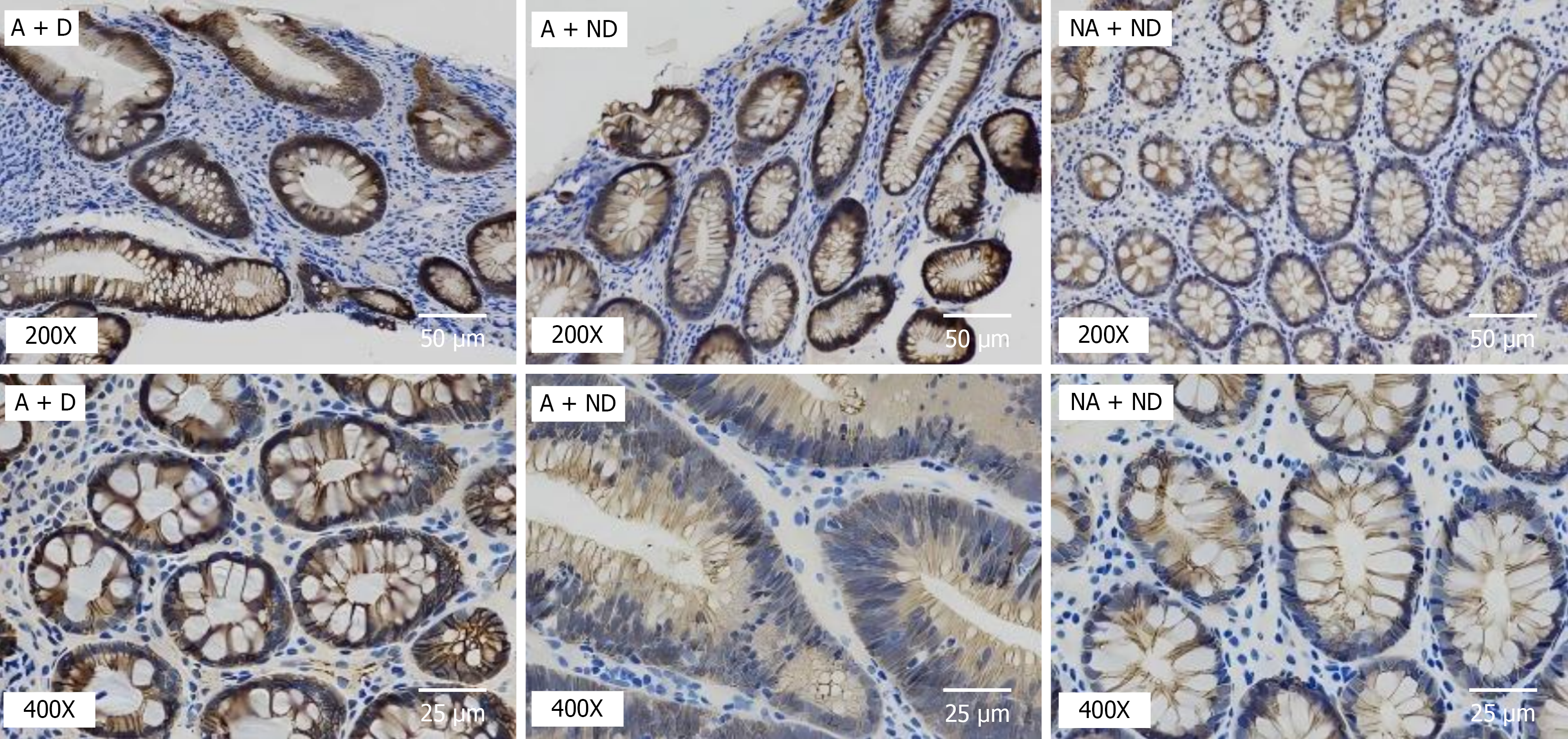
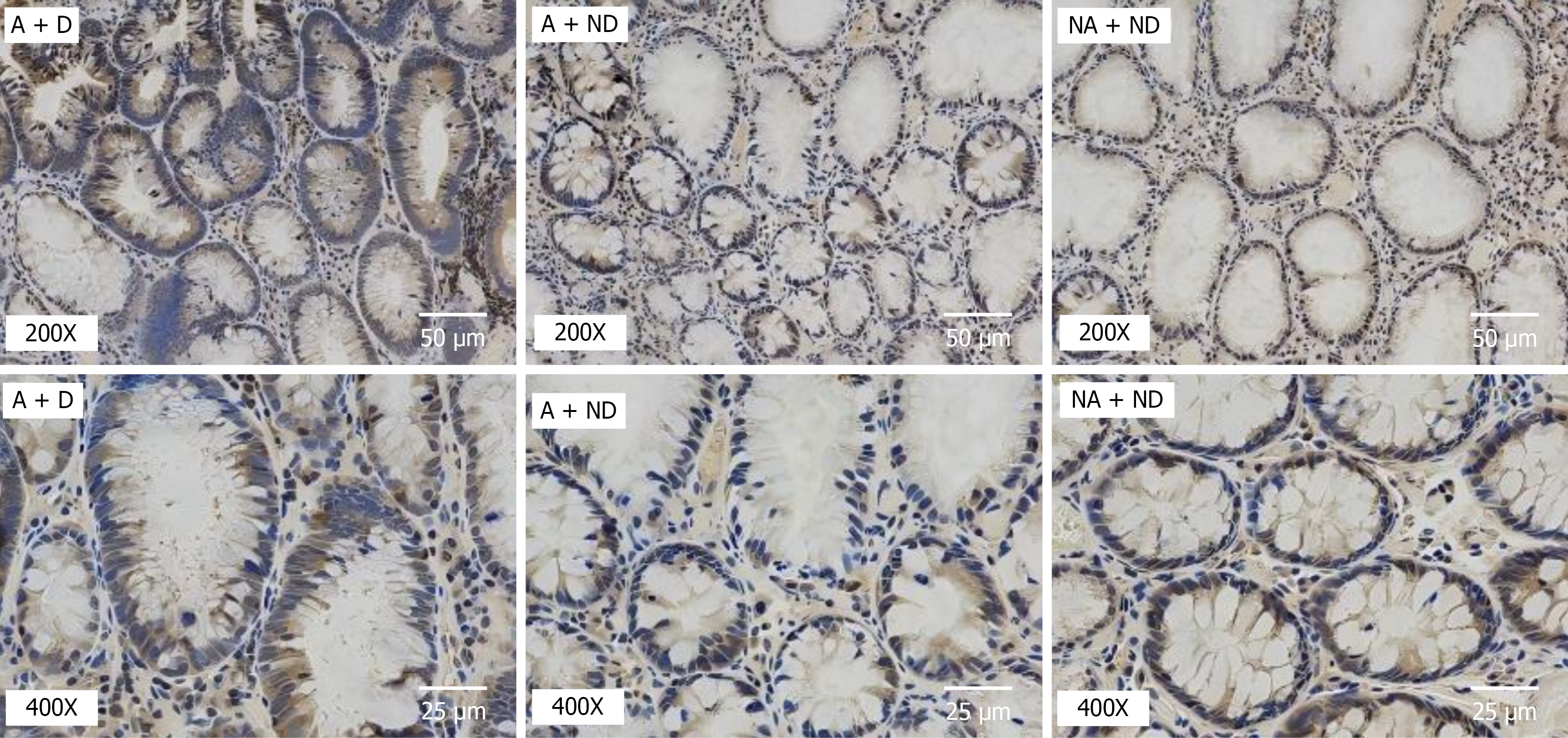
The abundance of β-catenin and TCF7 L2 protein in the intestine was evaluated using immunohistochemistry (Figures 2 and 3). The positive expression of the A + D group was significantly higher than that of the A + ND and NA + ND groups (P < 0.05; Table 5).
Type 2 diabetes mellitus and cancer are two of the biggest “chronic” killers of human health. With the change of environment and modern lifestyle, the incidence of type 2 diabetes mellitus and cancer is increasing annually worldwide. Current studies indicate that race, age, gender, obesity, diet, and smoking are common risk factors for these two diseases. A large number of epidemiological studies have revealed that type 2 diabetes mellitus is closely associated with the pathogenesis of various malignant tumors. Ben et al[12] conducted a meta-analysis of 35 group studies, and the results revealed that type 2 diabetes mellitus was associated with an increased risk of pancreatic cancer. Jiang et al[4] conducted a meta-analysis of 41 related group studies with a median follow-up of 10.5 years, and the results indicated the association of type 2 diabetes mellitus with an increased CRC incidence. Diabetes mellitus is associated with liver cancer, gastric cancer, leukemia, malignant lymphoma, breast cancer, and malignant melanoma to varying degrees[13-16]. Studies have indicated that type 2 diabetes mellitus is associated with increased cancer incidence, damaged DNA due to oxidative stress, activated tumor cell proliferation-related signaling pathways by inflammatory cytokines, inhibited anti-tumor immune mechanism, hyperglycemia caused by “glucose addiction” of tumor cells, and other theories. However, the mainstream view is that hyperinsulinemia caused by type 2 diabetes mellitus and its indirect effect on IGF-1 are important factors that promote tumorigenesis. However, the specific mechanism of the correlation between diabetes and cancer remains unclear and is the focus of current research and attention. This study focused on the correlation between type 2 diabetes mellitus and colorectal adenoma pathogenesis.
Epidemiological studies have confirmed that type 2 diabetes mellitus is an independent risk factor for CRC. Most CRCs develop from colorectal adenomas. Despite the clear association between diabetes and CRC, evidence of an association with colorectal adenoma in type 2 diabetes mellitus is limited. A retrospective study of patients who underwent polypectomy in the gastroenterology department from 2015 to 2017 revealed a higher colorectal adenoma incidence in patients with diabetes mellitus than in those with no diabetes, with a statistically significant difference. The state of hyperglycemia will increase the generation of free radicals in the body, causing severe oxidative stress and a significant increase in serum CEA levels[17]. The results of this study reveal a significantly higher CEA level in patients of the A + D group than in those of the A + ND and NA + ND groups. This phenomenon indicates that the state of hyperglycemia further exacerbates the CEA level in patients with colonic adenoma. Noteworthily, the change in serum IGF-1 level demonstrates this trend: The level is the highest in the A + D group, followed by the A + ND group, and the lowest in the NA + ND group. This result is statistically significant. On one hand, we observed higher IGF-1 Levels in patients in the A + D group, which is consistent with many previous studies; on the other hand, patients in the A + ND group still showed higher IGF-1 levels compared with those of the NA + ND group even in the non-diabetic state. These results indicate CEA and IGF-1 as important reference indicators for assessing the risk of colonic adenoma, especially in patients with diabetes mellitus. Further study on the factors related to colorectal adenoma formation and progression will provide more opportunities for early detection and prevention of CRC.
Wnt signaling not only plays a crucial role in regulating embryonic development, but also is involved in cell trafficking and apoptosis. The abnormality of the Wnt/β-catenin signaling pathway is associated with several human diseases, including tumorigenesis, osteoporosis, aging, diabetes, etc. TCF7 L2 is an important downstream transcription factor regulated by the Wnt signaling pathway and an important component of intracellular lymphoid enhanced binding factor/T cell-specific transcription factor. Wnt signaling pathway regulates TCF7 L2 expression in the nucleus through β-catenin, thereby controlling the transcription and expression of downstream target genes[18] and affecting cell proliferation and apoptosis. At present, many genetic studies have confirmed TCF7 L2 as the strongest predisposition gene for type 2 diabetes mellitus[19]. The Wnt pathway and its key factor TCF7 L2 are closely associated with the pathogenesis of type 2 diabetes mellitus, but the specific mechanism remains under research. TCF7 L2 is expressed in various tissues, including the pancreas and intestine. In in vitro studies, Wnt regulated the expression of target genes that were closely associated with the growth and function of islet β-cells by activating the transcriptional activity of downstream TCF7 L2. In vitro cell experiments revealed that TCF7 L2 expression increased and promoted islet cell proliferation and insulin release. The TCF7 L2 expression is increased in islet β-cells of patients with diabetes mellitus or risk gene carriers[20]. Later, the author established a type 2 diabetes mellitus rat model induced by a high-fat and high-glucose diet, and revealed an increased TCF7 L2 expression in rat islets, indicating that TCF7 L2 may exhibit a destructive effect on islet cells. TCF7 L2 and β-catenin are key functional target genes in the Wnt signaling pathway. The increased TCF7 L2 expression indicates the activation state of the Wnt pathway. The results indicate that the Wnt pathway in diabetic rat models may be active, whereas the Wnt pathway in normal control rats is in a relative resting state. The Wnt/β-catenin/TCF7 L2 pathway activation and the increased TCF7 L2 expression may be associated with type 2 diabetes mellitus.
Studies have demonstrated that the Wnt/β-catenin signaling pathway can be activated and play a key role in the genesis of various tumors, such as CRC[21,22], and also plays an important role in CRC invasion and metastasis[23]. TCF7 L2 and β-catenin are important components in the classical Wnt signaling pathway, and their detection results can reflect the changes in the Wnt/β-catenin signaling pathway. In this study, we investigated the abundance of β-catenin and TCF7 L2 proteins in intestinal epithelial cells of patients in the A + D group by conducting immunohistochemical analysis due to limited sampling. The presence of β-catenin and TCF7 L2 in intestinal epithelial cells of patients in the A + D group were significantly higher than those of patients in the A + ND and NA + ND groups. Their positive expression in the A + ND group was slightly higher than that of the NA + ND group, but the difference was not statistically significant (P > 0.05). Concurrently, we detected the presence of Ki67 in intestinal epithelial cells of all the three patient groups, which was consistent with that observed for β-catenin and TCF7 L2, indicating that the high incidence of tubular adenoma in patients with type 2 diabetes mellitus may be associated with the Wnt/β-catenin/TCF7 L2 signaling pathway activation in vivo. We speculate that the classical Wnt signaling pathway in vivo is induced and activated by genetic, environmental, obesity, inflammatory factors (interleukin-1β, tumor necrosis factor-α, and interferon-γ), etc. Further, the ubiquitin-proteasome system of β-catenin is inhibited; hence, the non-phosphorylated β-catenin accumulates in the cytosol and is transported to the nucleus. β-catenin interacts with nuclear transcription factor TCF7 L2, coregulates the expression of downstream target genes - such as c-myc, cyclin-dependent kinase 4, paired-like homeodomain transcription factor 2, and Cyclin D in the nucleus - and regulates cell proliferation and apoptosis. The expression of Wnt downstream target genes in islet cells may damage β-cells and induce and aggravate type 2 diabetes mellitus. Abnormal Wnt signal activation acts on and damages intestinal epithelial cells, which may be a crucial factor in inducing colorectal tubular adenoma. TCF7 L2 may be an important factor in the susceptibility of patients with type 2 diabetes mellitus to colorectal adenoma. Further, it is the common pathogenic basis of type 2 diabetes mellitus and colorectal adenoma. Colorectal adenoma is recognized as a crucial precancerous lesion of CRC, which gradually develops from normal mucosa to dysplasia (polyp adenoma) to CRC, but the mechanism remains unclear. To investigate the possible mechanism of colorectal adenoma in type 2 diabetes mellitus, discover new therapeutic targets, and provide a scientific basis for the early prevention and treatment of adenoma carcinogenesis.
Hyperinsulinemia and IGF-1 have been proven to mainly promote tumor formation by activating classic oncogenic pathways, such as phosphatidylinositol 3-kinase/protein kinase B[24]. However, our findings demonstrate not only a significant increase in serum IGF-1 levels but also an abnormally activated Wnt/β-catenin signaling pathway in patients with colonic adenomas who have type 2 diabetes. The presence of an interaction between these signaling axes deserves further exploration. However, this study is a retrospective analysis, and the sample size is relatively small; thus, certain limitations occur when conducting clinical sample experiments. First, the patient’s clinical data only includes basic information, including age and gender. This information ensures the comparability between groups, but more detailed information, such as body mass index (patient’s obesity status), hypoglycemic drug administration (especially metfor
This study confirmed that type 2 diabetes mellitus is an independent risk factor for colorectal adenomas in the retrospective group. Furthermore, this study provides preliminary histological evidence that indicates the abnormal Wnt/β-catenin/TCF7 L2 signaling pathway activation as a potential molecular bridge in the association of type 2 diabetes mellitus with colorectal adenoma occurrence and development.
| 1. | Trajkovic-Arsic M, Kalideris E, Siveke JT. The role of insulin and IGF system in pancreatic cancer. J Mol Endocrinol. 2013;50:R67-R74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 2. | Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9408] [Cited by in RCA: 10047] [Article Influence: 627.9] [Reference Citation Analysis (1)] |
| 3. | Winawer SJ, Zauber AG, Ho MN, O'Brien MJ, Gottlieb LS, Sternberg SS, Waye JD, Schapiro M, Bond JH, Panish JF. Prevention of colorectal cancer by colonoscopic polypectomy. The National Polyp Study Workgroup. N Engl J Med. 1993;329:1977-1981. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3521] [Cited by in RCA: 3125] [Article Influence: 94.7] [Reference Citation Analysis (4)] |
| 4. | Jiang Y, Ben Q, Shen H, Lu W, Zhang Y, Zhu J. Diabetes mellitus and incidence and mortality of colorectal cancer: a systematic review and meta-analysis of cohort studies. Eur J Epidemiol. 2011;26:863-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 156] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 5. | Budzynska K, Passerman D, White-Perkins D, Rees DA, Xu J, Lamerato L, Schooley S. Diabetes mellitus and hyperglycemia control on the risk of colorectal adenomatous polyps: a retrospective cohort study. BMC Fam Pract. 2018;19:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Soltani G, Poursheikhani A, Yassi M, Hayatbakhsh A, Kerachian M, Kerachian MA. Obesity, diabetes and the risk of colorectal adenoma and cancer. BMC Endocr Disord. 2019;19:113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 73] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 7. | Yu F, Guo Y, Wang H, Feng J, Jin Z, Chen Q, Liu Y, He J. Type 2 diabetes mellitus and risk of colorectal adenoma: a meta-analysis of observational studies. BMC Cancer. 2016;16:642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Li JY, Yu T, Xia ZS, Chen GC, Yuan YH, Zhong W, Zhao LN, Chen QK. Enhanced proliferation in colorectal epithelium of patients with type 2 diabetes correlates with β-catenin accumulation. J Diabetes Complications. 2014;28:689-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Chocarro-Calvo A, García-Martínez JM, Ardila-González S, De la Vieja A, García-Jiménez C. Glucose-induced β-catenin acetylation enhances Wnt signaling in cancer. Mol Cell. 2013;49:474-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 143] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 10. | Del Bosque-Plata L, Martínez-Martínez E, Espinoza-Camacho MÁ, Gragnoli C. The Role of TCF7L2 in Type 2 Diabetes. Diabetes. 2021;70:1220-1228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 120] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 11. | Matkowskyj KA, Schonfeld D, Benya RV. Quantitative immunohistochemistry by measuring cumulative signal strength using commercially available software photoshop and matlab. J Histochem Cytochem. 2000;48:303-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 115] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 12. | Ben Q, Xu M, Ning X, Liu J, Hong S, Huang W, Zhang H, Li Z. Diabetes mellitus and risk of pancreatic cancer: A meta-analysis of cohort studies. Eur J Cancer. 2011;47:1928-1937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 561] [Cited by in RCA: 493] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 13. | Shahid RK, Ahmed S, Le D, Yadav S. Diabetes and Cancer: Risk, Challenges, Management and Outcomes. Cancers (Basel). 2021;13:5735. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 121] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 14. | Wang C, Wang X, Gong G, Ben Q, Qiu W, Chen Y, Li G, Wang L. Increased risk of hepatocellular carcinoma in patients with diabetes mellitus: a systematic review and meta-analysis of cohort studies. Int J Cancer. 2012;130:1639-1648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 366] [Cited by in RCA: 330] [Article Influence: 23.6] [Reference Citation Analysis (2)] |
| 15. | Nowara E, Huszno J, Drosik-Rutowicz K, Nowara E, Maslyk B. The prospective estimation of blood serum free IGF-1, insulin concentration, and body mass index as a prognostic factor for breast cancer patients. J Clin Oncol. 2013;31:47. [DOI] [Full Text] |
| 16. | Autier P, Koechlin A, Robertson C, Bota M, Boniol M, Leroith D, Bolli G, Rosenstock J, Boyle P. Breast cancer and diabetes. J Clin Oncol. 2013;31:e12507. [DOI] [Full Text] |
| 17. | Ata N, Dal K, Kucukazman M, Yeniova AÖ, Karakaya S, Unsal O, Dagdeviren M, Akın KO, Baser S, Beyan E, Ertugrul DT. The effect of glycemic control on CEA, CA 19-9, amylase and lipase levels. Open Med (Wars). 2015;10:8-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Wachsmannova L, Stevurkova V, Ciernikova S. Changes in SNAI1 and VIM gene expression in Caco2 cells after cocultivation with bacteria from colorectal cancer biopsies. Neoplasma. 2019;66:271-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Bian J, Dannappel M, Wan C, Firestein R. Transcriptional Regulation of Wnt/β-Catenin Pathway in Colorectal Cancer. Cells. 2020;9:2125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 192] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 20. | Tang Q, Chen J, Di Z, Yuan W, Zhou Z, Liu Z, Han S, Liu Y, Ying G, Shu X, Di M. TM4SF1 promotes EMT and cancer stemness via the Wnt/β-catenin/SOX2 pathway in colorectal cancer. J Exp Clin Cancer Res. 2020;39:232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 267] [Article Influence: 44.5] [Reference Citation Analysis (1)] |
| 21. | Kanazawa A, Tsukada S, Kamiyama M, Yanagimoto T, Nakajima M, Maeda S. Wnt5b partially inhibits canonical Wnt/beta-catenin signaling pathway and promotes adipogenesis in 3T3-L1 preadipocytes. Biochem Biophys Res Commun. 2005;330:505-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 125] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 22. | Grant SF, Thorleifsson G, Reynisdottir I, Benediktsson R, Manolescu A, Sainz J, Helgason A, Stefansson H, Emilsson V, Helgadottir A, Styrkarsdottir U, Magnusson KP, Walters GB, Palsdottir E, Jonsdottir T, Gudmundsdottir T, Gylfason A, Saemundsdottir J, Wilensky RL, Reilly MP, Rader DJ, Bagger Y, Christiansen C, Gudnason V, Sigurdsson G, Thorsteinsdottir U, Gulcher JR, Kong A, Stefansson K. Variant of transcription factor 7-like 2 (TCF7L2) gene confers risk of type 2 diabetes. Nat Genet. 2006;38:320-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1802] [Cited by in RCA: 1569] [Article Influence: 78.5] [Reference Citation Analysis (2)] |
| 23. | Shu L, Sauter NS, Schulthess FT, Matveyenko AV, Oberholzer J, Maedler K. Transcription factor 7-like 2 regulates beta-cell survival and function in human pancreatic islets. Diabetes. 2008;57:645-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 196] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 24. | Zhao X, Huang F, Sun Y, Li L. Mechanisms of endurance and resistance exercise in type 2 diabetes mellitus: A Narrative review. Biochem Biophys Res Commun. 2025;761:151731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/