Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.113525
Revised: October 24, 2025
Accepted: December 8, 2025
Published online: February 27, 2026
Processing time: 167 Days and 2.6 Hours
Elderly patients undergoing laparoscopic cholecystectomy (LC) are at increased risk for postoperative complications.
To investigate the impact of dexmedetomidine (DEX) preconditioning on post
A retrospective analysis was conducted involving elderly patients (aged ≥ 60 years), who underwent LC between May 2023 and April 2024. Patients were categorized into two groups based on DEX administration protocols: (1) DEX preconditioning anesthesia (DEX-PA) group; and (2) DEX conventional anesthesia group. Postoperative assessments included pain (Visual Analog Scale), sedation (Ramsay Sedation Scale), oxidative stress and inflammation at 6 hours, recovery from anesthesia, gastrointestinal function recovery, cognitive scores (mini-mental state examination), and incidence of adverse events.
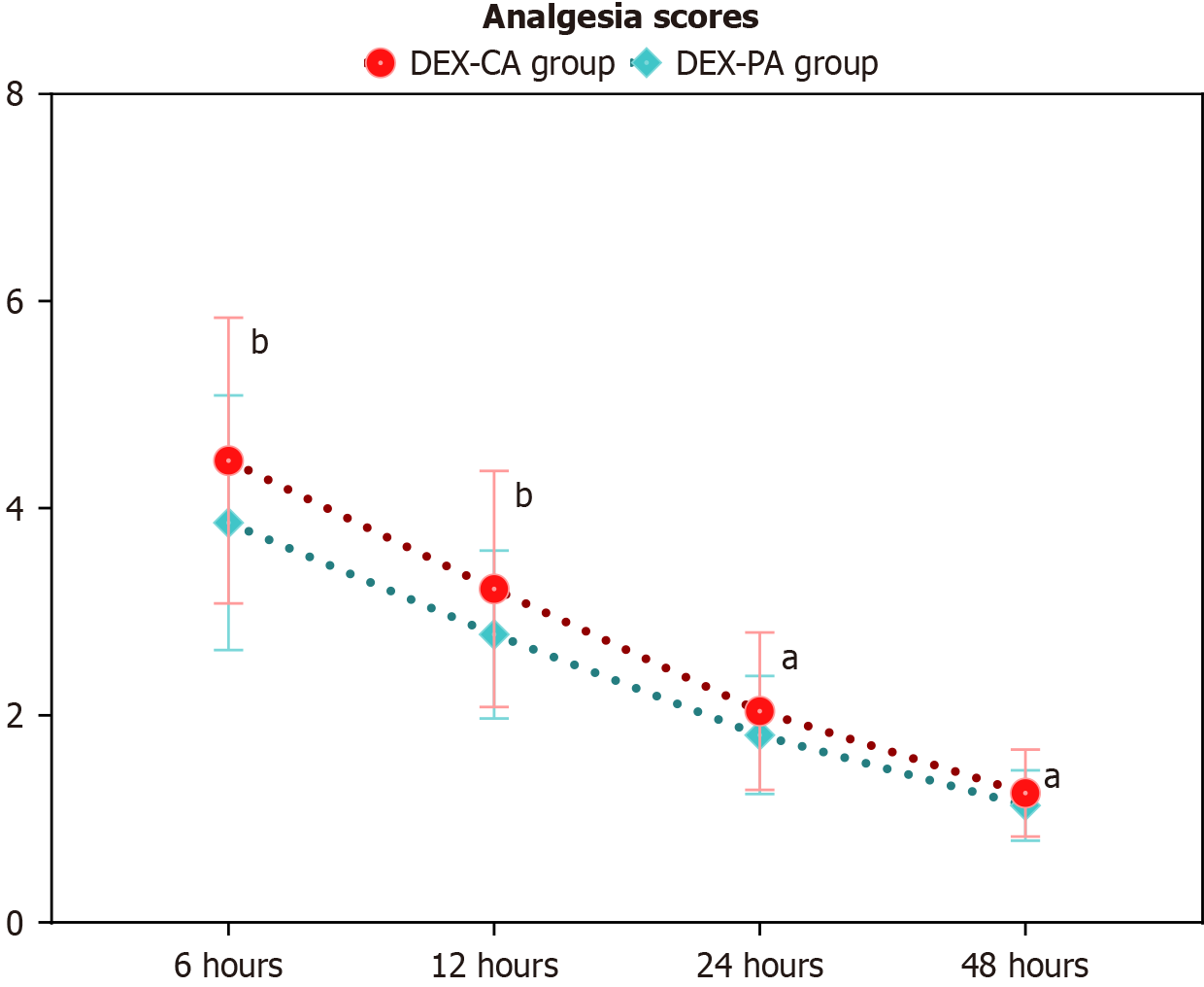
Among 182 patients (DEX-PA = 87, DEX conventional anesthesia = 95), the DEX-PA group showed significantly lower Visual Analog Scale scores (6 hours: 3.86 ± 1.23 vs 4.46 ± 1.38, P = 0.002), reduced oxidative stress (malondialdehyde: 3.17 ± 0.56 μmol/L vs 3.39 ± 0.61 μmol/L, P = 0.013) and inflammation (C-reactive protein: 5.24 ± 1.35 mg/L vs 5.77 ± 1.62 mg/L, P = 0.018), shorter awakening times (14.87 ± 2.67 minutes vs 15.75 ± 2.82 minutes, P = 0.032), shorter stays in the post-anesthesia care unit (58.24 ± 10.85 minutes vs 62.38 ± 12.47 minutes, P = 0.018), accelerated gastrointestinal recovery (time to first defecation: 36.82 ± 7.63 hours vs 39.95 ± 8.24 hours, P = 0.009), better cognitive performance at 6 hours (25.53 ± 1.87 vs 24.72 ± 2.15, P = 0.008), and fewer adverse events (agitation: 4.60% vs 14.74%, P = 0.022).
DEX preconditioning prior to anesthesia induction significantly enhances postoperative analgesia, reduces oxi
Core Tip: This study demonstrates that administering a loading dose of dexmedetomidine (DEX) as a pre-treatment strategy before anesthesia induction offers notable benefits for elderly patients undergoing laparoscopic cholecystectomy. Compared to the routine use of DEX solely during surgery, pre-treatment with DEX enhanced postoperative analgesia, reduced oxidative stress and inflammation, accelerated recovery of gastrointestinal and cognitive functions, and lowered the incidence of agitation without increasing intraoperative hemodynamic risks. These findings highlight the critical importance of the timing of DEX administration and suggest a promising optimized anesthesia approach to promote recovery in elderly surgical patients.
- Citation: Chen M, Chen FC, Pan Q, Liu ZH. Impact of dexmedetomidine preconditioning on analgesic and postoperative recovery in elderly patients undergoing laparoscopic cholecystectomy. World J Gastrointest Surg 2026; 18(2): 113525
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/113525.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.113525
Gallstones and gallbladder polyps are common biliary diseases, particularly prevalent among older adults[1]. Gallstones develop as solid particles from cholesterol, bilirubin, or a mixture of both within the gallbladder, leading to symptoms such as biliary colic, acute cholecystitis, pancreatitis, or cholangitis[2]. This condition arises due to bile supersaturation with cholesterol, which promotes crystal formation[3]. Although gallbladder polyps are often asymptomatic, they require monitoring or removal due to their potential for malignant transformation[4]. For symptomatic patients, laparoscopic cholecystectomy (LC) remains the definitive treatment and the gold standard surgical approach[5]. Despite its minimally invasive nature, LC can still induce significant surgical stress, inflammation, and pain, especially in elderly patients, who often exhibit reduced physiological reserves, heightened pain sensitivity, altered drug metabolism, increased risk of postoperative cognitive dysfunction (POCD), and prolonged recovery times[6,7]. Therefore, optimizing perioperative management, particularly anesthesia and analgesia, is crucial to alleviate these challenges and facilitate faster recovery in this growing patient population[8]. This study aims to evaluate whether dexmedetomidine (DEX) pre-treatment offers benefits superior to conventional anesthesia (CA) in improving pain control, reducing surgical stress, and accelerating postoperative recovery in elderly patients undergoing LC.
DEX is a highly selective α2-adrenergic receptor agonist that has garnered significant interest in anesthesiology due to its unique pharmacological properties[9]. It offers dose-dependent sedation, analgesia, anti-anxiety effects, and sympathetic relaxation without causing significant respiratory depression. Beyond these direct effects, preclinical and some clinical evidence suggest that DEX may offer organ-protective benefits by modulating inflammatory cascades, reducing oxidative stress, and inhibiting neuroendocrine stress responses triggered by surgery and anesthesia[10]. Activation of α2-adrenergic receptors in the central nervous system and peripheral tissues is associated with reduced release of pro-inflammatory cytokines and catecholamines, potentially mitigating systemic inflammatory response syndrome associated with major surgeries[11]. Furthermore, its neuroprotective potential may be linked to decreased excitotoxicity and inhibition of inflammatory signaling within the central nervous system, mechanisms closely tied to the prevention of POCD in elderly patients[12]. While DEX is commonly used as an adjunct during anesthesia maintenance, administering it as a pre-treatment prior to surgical injury represents a strategic approach aimed at actively regulating these stress and inflammation pathways from the outset[13].
Therefore, this retrospective study aims to investigate the specific effects of administering DEX as a pre-treatment prior to anesthesia induction on pain relief, postoperative recovery parameters, and the incidence of adverse events in elderly patients undergoing elective LC. By introducing this innovative anesthesia approach, we aim to offer a promising alternative for patients undergoing cholecystectomy that may considerably improve clinical outcomes. These findings could contribute to refining perioperative protocols, enhancing pain management, accelerating recovery, reducing complications, and ultimately improving postoperative care quality for elderly surgical patients.
This retrospective study included 182 elderly patients who underwent LC at the First People's Hospital of Linping District Hangzhou City, between May 2023 and April 2024. Inclusion Criteria: (1) Definitive diagnosis of gallstones[14] or gallbladder polyps[15] confirmed by abdominal ultrasound; (2) Age ≥ 60 years, regardless of gender; (3) American Society of Anesthesiologists (ASA)[16] classification I or II; and (4) Complete medical records without missing data. Exclusion Criteria: (1) Use of anesthetic sedatives or analgesics within three months prior to surgery; (2) History of abdominal surgery or presence of severe abdominal infection; (3) Neurological dysfunction or severe mental disorders; and (4) Respiratory diseases or severe cardiac, hepatic, or renal dysfunction.
Based on the DEX administration protocols, the 182 eligible patients were divided into two groups: (1) The DEX-CA group; and (2) The DEX preconditioning anesthesia (DEX-PA) group. Patients in the DEX-CA group (n = 95) received the standard LC anesthesia protocol, whereas those in the DEX-PA group (n = 87) received anesthesia in addition to the CA protocol.
All procedures in this study complied with the ethical guidelines set by the First People's Hospital of Linping District Hangzhou City, as well as national ethics committee for human research, and were conducted in accordance with the Declaration of Helsinki. Furthermore, the study received approval from the Ethics Committee of the First People's Hospital of Linping District Hangzhou City. Considering that the data utilized in this study comprised anonymized patient information and posed no potential risk or impact on patient care, the ethics committee waived the requirement for informed consent for this retrospective study.
Preoperative preparation: Both groups of patients fasted for 8 hours and abstained from drinking for 4 hours prior to surgery. Upon entering the operating room, intravenous access was immediately established, followed by radial artery puncture and catheterization. Vital signs, including mean arterial pressure, electrocardiogram, heart rate, and oxygen saturation, were continuously monitored using a multi-parameter monitor (Infinity® Gamma XL, Dräger, Germany) to maintain stable circulatory and respiratory function during anesthesia.
Preconditioning anesthesia: Ten minutes prior to anesthesia induction, patients in the DEX-PA group received an intravenous injection of DEX hydrochloride at a dose of 0.5 μg/kg. Patients in the DEX-CA group did not receive any pre-treatment medications during this stage.
Anesthesia induction: Both groups of patients received intravenous anesthesia induction with midazolam (0.03 mg/kg), propofol (2 mg/kg), sufentanil (0.4 μg/kg), and cisatracurium besylate (0.2 mg/kg). These agents were administered sequentially via intravenous injection. Three minutes after drug administration, endotracheal intubation was performed, followed by mechanical ventilation. Ventilation parameters were set as follows: (1) Oxygen flow rate of 2 L/minute; (2) Tidal volume of 7 mL/kg; (3) Fraction of inspired oxygen at 60%; (4) Respiratory rate of 12 breaths/minute; (5) Inspiratory-to-expiratory ratio of 1:1.5; and (6) End-tidal carbon dioxide concentration maintained between 35 mmHg and 40 mmHg to ensure stable intraoperative respiratory function.
Anesthesia maintenance: Both groups received maintenance anesthesia via intravenous infusion of propofol at a rate of 4-6 mg/kg/hour, remifentanil at 0.3-0.5 μg/kg/minute, and DEX hydrochloride at 0.5 μg/kg/hour. During surgery, cisatracurium besylate was intermittently administered intravenously at 0.1 mg/kg as needed for muscle relaxation. Anesthesia depth was adjusted using bispectral index monitoring, with bispectral index value maintained between 40 and 60. All anesthesia agents were immediately discontinued upon completion of the surgery.
LC procedure: Patients were positioned supine with the head elevated approximately 15°-20° and tilted 10°-15° to the left. The abdominal skin was routinely disinfected, from the nipple line superiorly, to the pubic symphysis inferiorly, and laterally to the midaxillary lines, and sterile drapes were applied. A 10 mm incision was made at the lower edge of the umbilicus, through which a Veress needle was inserted to establish pneumoperitoneum, maintaining pressure between 12 mmHg and 14 mmHg. A 10 mm trocar was then introduced at the same site to serve as the observation port. After exploring the abdominal cavity, two additional 5 mm trocars were placed: One in the subxiphoid region and another 2 cm below the right midclavicular line at the costal margin, serving as operation ports.
Adhesions around the gallbladder were carefully dissected to expose Calot’s triangle. Once the anatomical relation
Postoperative management: At the end of surgery, patients received an intravenous injection of tropisetron (5 mg) and parecoxib sodium (40 mg). Once the patients regained spontaneous breathing and were fully awake, the endotracheal tube was removed, and they were transferred to the anesthesia care unit for further observation.
Analgesic and sedative effect: Pain and sedation levels in both patient groups were assessed at 6 hours, 12 hours, 24 hours, and 48 hours postoperatively using the visual analog scale and the Ramsay sedation scale. The Visual Analog Scale scores range from 0 to 10, with higher scores indicating more severe pain. The intraclass correlation coefficient for test-retest reliability of this scale ranges from 0.97 to 0.99[17]. The Ramsay scores range from 1 to 6, with higher scores indicating deeper sedation. The Cronbach’s alpha for this scale is 0.83[18].
Postoperative recovery status: (1) Oxidative stress and stress response markers: Blood samples (3 mL) were collected from both patient groups preoperatively and 6 hours postoperatively. The samples were centrifuged at 3000 rpm for 10 minutes, and the supernatant was retained for analysis. Enzyme-linked immunosorbent assay was used to measure levels of malondialdehyde (MDA), superoxide dismutase (SOD), cortisol (Cor), tumor necrosis factor-alpha (TNF-α), and C-reactive protein (CRP). Total antioxidant capacity (TAC) was determined using a micro-method; (2) Recovery of anesthesia and gastrointestinal function. The following parameters were recorded: Time to awakening (from discontinuation of anesthesia to the patient’s response to verbal stimuli), time to extubating (from discontinuation of anesthesia to removal of the endotracheal tube), duration of stay in the anesthesia care unit, time to recovery of bowel sounds (first audible bowel sounds postoperatively), time to first exhaust, and time to first defecation; (3) Cognitive function recovery: Cognitive function was assessed using the mini-mental state examination (MMSE) at preoperative, 6-hour, 12-hour, 24-hour, and 48-hour postoperative intervals. The MMSE has a maximum score of 30 points, with higher scores indicating better cognitive function. The Cronbach’s alpha for this scale is 0.61[19]; and (4) Adverse reaction: Intraoperative and postoperative adverse reactions were observed and compared between the two patient groups. Intraoperative complications, including hypotension and bradycardia, were closely monitored. Postoperative adverse reactions, such as nausea and vomiting, bradycardia, hypotension, and agitation, were also assessed.
All statistical analyses in this research were performed using Statistical Package for the Social Sciences software (version 29.0; developed by SPSS Inc., based in Chicago, IL, United States). Continuous variables were evaluated for normality using the Shapiro-Wilk test and presented as mean ± SD. Group differences were analyzed using independent samples t-test. Categorical variables were expressed as n (%) and compared between groups using χ2 test.
In the comparison of demographic characteristics between the DEX-CA and DEX-PA groups, no significant differences were observed in age, body mass index, gender distribution, educational background, smoking history, drinking history, as well as the prevalence of hypertension, diabetes, coronary heart disease, hyperlipidemia, and ASA grading (all P values > 0.05; Table 1). However, a trend toward significance in disease type distribution was observed between the two groups (P = 0.091), with the DEX-CA group exhibiting a higher proportion of patients with gallstones and a lower proportion with gallbladder polyps than the DEX-PA group.
| Parameter | DEX conventional anesthesia group | DEX preconditioning anesthesia group | t/χ2 | P value |
| Age (years) | 71.38 ± 5.26 | 71.41 ± 5.42 | 0.031 | 0.975 |
| Gender | 0.316 | 0.574 | ||
| Male | 52 (54.74) | 44 (50.57) | ||
| Female | 43 (45.26) | 43 (49.43) | ||
| Body mass index (kg/m2) | 24.37 ± 1.27 | 24.43 ± 1.32 | 0.337 | 0.736 |
| Educational background | 0.356 | 0.837 | ||
| Junior high or below | 38 (40.00) | 32 (36.78) | ||
| High school | 30 (31.58) | 31 (35.63) | ||
| Tertiary and above | 27 (28.42) | 24 (27.59) | ||
| Smoking history | 22 (23.16) | 20 (22.99) | 0.001 | 0.978 |
| Drinking history | 18 (18.95) | 15 (17.24) | 0.089 | 0.765 |
| Complication | ||||
| Hypertension | 21 (22.11) | 23 (26.44) | 0.465 | 0.495 |
| Diabetes | 10 (10.53) | 12 (13.79) | 0.456 | 0.499 |
| Coronary heart disease | 11 (11.58) | 14 (16.09) | 0.781 | 0.377 |
| Hyperlipidemia | 17 (17.89) | 19 (21.84) | 0.445 | 0.505 |
| American Society of Anesthesiologists grading | 0.173 | 0.677 | ||
| I grade | 65 (68.42) | 57 (65.52) | ||
| II grade | 30 (31.58) | 30 (34.48) | ||
| Disease type | 2.854 | 0.091 | ||
| Gallstone | 81 (85.26) | 81 (93.10) | ||
| Gallbladder polyps | 14 (14.74) | 6 (6.90) |
In terms of surgery features, no significant differences were observed between the DEX-CA and DEX-PA groups in operative duration, anesthesia duration, intraoperative bleeding volume, fluid replacement volume, or in the doses of anesthesia inducing drugs including midazolam, propofol, sufentanil, and atracurium (all P > 0.05; Table 2). Similarly, no significant differences were found in the doses of anesthesia maintenance drugs, such as propofol, remifentanil, and DEX, or in the frequency of atracurium administration.
| Parameter | DEX conventional anesthesia group | DEX preconditioning anesthesia group | t/χ2 | P value |
| Operative duration (minutes) | 82.86 ± 12.43 | 82.25 ± 12.27 | 0.334 | 0.739 |
| Anesthesia duration (minutes) | 89.62 ± 13.84 | 89.84 ± 13.43 | 0.107 | 0.915 |
| Intraoperative bleeding volume (mL) | 14.44 ± 1.92 | 14.86 ± 2.24 | 1.351 | 0.178 |
| Fluid replacement volume (mL) | 746.25 ± 48.43 | 758.24 ± 50.91 | 1.628 | 0.105 |
| Dose of anesthesia inducing drugs | ||||
| Midazolam (mg) | 2.12 ± 0.35 | 2.08 ± 0.32 | 0.711 | 0.478 |
| Propofol (mg) | 135.52 ± 20.25 | 132.75 ± 19.83 | 0.932 | 0.353 |
| Sufentanil (μg) | 24.38 ± 3.72 | 23.92 ± 3.69 | 0.847 | 0.398 |
| Atracurium (mg) | 12.25 ± 1.87 | 12.16 ± 1.78 | 0.344 | 0.731 |
| Dose of anesthesia maintenance drugs | ||||
| Propofol (mg) | 452.38 ± 67.21 | 443.92 ± 65.84 | 0.856 | 0.393 |
| Remifentanil (μg) | 1286.54 ± 214.36 | 1262.47 ± 208.92 | 0.766 | 0.445 |
| DEX (μg) | 42.18 ± 6.32 | 41.75 ± 6.18 | 0.459 | 0.647 |
| Times of atracurium is added | 25 (26.32) | 20 (22.99) | 0.270 | 0.603 |
Significant differences in postoperative analgesia scores were observed between the DEX-CA and DEX-PA groups at several time points (Figure 1). At 6 hours postoperatively, the DEX-CA group reported higher scores than the DEX-PA group (P = 0.002). This pattern persisted at 12 hours (P = 0.003) and 24 hours (P = 0.024) postoperatively, with the DEX-CA group consistently showing higher scores. At 48 hours postoperatively, the difference remained statistically significant (P = 0.033), indicating that the higher analgesia scores in the DEX-CA group persisted even though the magnitude of the difference had decreased.
Postoperative sedation scores did not differ significantly between the DEX-CA and DEX-PA groups at any of the measured time points (Table 3). Scores were comparable between the two groups at 6 hours, 12 hours, 24 hours, and 48 hours postoperatively (all P > 0.05).
| Parameter | DEX conventional anesthesia group (n = 95) | DEX preconditioning anesthesia group (n = 87) | t value | P value |
| Postoperative 6 hours | 4.23 ± 0.47 | 4.26 ± 0.35 | 0.440 | 0.660 |
| Postoperative 12 hours | 3.97 ± 0.39 | 4.01 ± 0.32 | 0.847 | 0.398 |
| Postoperative 24 hours | 3.52 ± 0.45 | 3.66 ± 0.68 | 1.652 | 0.101 |
| Postoperative 48 hours | 3.07 ± 0.26 | 3.14 ± 0.33 | 1.470 | 0.143 |
In the comparison of oxidative stress and stress response markers between the DEX-CA and DEX-PA groups, no significant differences were observed preoperatively in levels of MDA, SOD activity, TAC, Cor, TNF-α, or CRP (all P > 0.05; Table 4). However, significant differences emerged at 6 hours postoperatively. The DEX-CA group exhibited higher MDA levels than the DEX-PA group (P = 0.013), indicating greater oxidative stress in the DEX-CA group. Conversely, SOD activity was lower in the DEX-CA group than in the DEX-PA group (P = 0.014), suggesting reduced antioxidant capacity. Similarly, TAC levels were lower in the DEX-CA group (P = 0.018), reinforcing this trend. Regarding stress response markers, Cor levels were higher in the DEX-CA group than the DEX-PA group (P = 0.032). In addition, TNF-α levels were significantly elevated in the DEX-CA group relative to the DEX-PA group (P = 0.016). Lastly, CRP levels were also higher in the DEX-CA group (P = 0.018).
| Parameter | DEX conventional anesthesia group (n = 95) | DEX preconditioning anesthesia group (n = 87) | t value | P value |
| Oxidative stress markers | ||||
| Malondialdehyde (μmol/L) | ||||
| Preoperative | 2.31 ± 0.43 | 2.23 ± 0.35 | 1.465 | 0.145 |
| Postoperative 6 hours | 3.39 ± 0.61 | 3.17 ± 0.56 | 2.520 | 0.013 |
| Superoxide dismutase (U/mL) | ||||
| Preoperative | 73.41 ± 7.85 | 72.04 ± 7.62 | 1.193 | 0.234 |
| Postoperative 6 hours | 88.32 ± 8.46 | 91.14 ± 6.79 | 2.494 | 0.014 |
| Total antioxidant capacity (mmol ascorbic acid Eq/L) | ||||
| Preoperative | 16.23 ± 2.26 | 16.79 ± 2.95 | 1.420 | 0.157 |
| Postoperative 6 hours | 13.59 ± 2.33 | 14.47 ± 2.61 | 2.391 | 0.018 |
| Stress response markers | ||||
| Cortisol (µg/dL) | ||||
| Preoperative | 12.62 ± 1.47 | 12.94 ± 1.08 | 1.663 | 0.098 |
| Postoperative 6 hours | 15.02 ± 2.35 | 14.32 ± 2.01 | 2.164 | 0.032 |
| Tumor necrosis factor-alpha (pg/mL) | ||||
| Preoperative | 1.83 ± 0.31 | 1.86 ± 0.37 | 0.577 | 0.565 |
| Postoperative 6 hours | 3.47 ± 0.98 | 3.15 ± 0.76 | 2.429 | 0.016 |
| C-reactive protein (mg/L) | ||||
| Preoperative | 1.54 ± 0.32 | 1.49 ± 0.43 | 0.874 | 0.384 |
| Postoperative 6 hours | 5.77 ± 1.62 | 5.24 ± 1.35 | 2.388 | 0.018 |
Regarding recovery from anesthesia, the awakening time was significantly shorter in the DEX-PA group than the DEX-CA group (P = 0.032), indicating a more rapid return to consciousness in patients receiving DEX-PA (Table 5). However, no significant difference in extubating time was found between the two groups (P > 0.05). The duration of stay in the anesthesia care unit was also significantly shorter for the DEX-PA group (P = 0.018). For gastrointestinal function recovery, significant differences were observed in all measured parameters. Bowel sounds recovery time was shorter in the DEX-PA group (P = 0.001), as were the first exhaust time (P = 0.003) and the first defecation time (P = 0.009). These results indicate that patients in the DEX-PA group experienced a faster recovery of gastrointestinal function following surgery.
| Parameter | DEX conventional anesthesia group (n = 95) | DEX preconditioning anesthesia group (n = 87) | t value | P value |
| Anesthesia recovery | ||||
| Awakening time (minutes) | 15.75 ± 2.82 | 14.87 ± 2.67 | 2.164 | 0.032 |
| Extubating time (minutes) | 17.44 ± 3.51 | 17.46 ± 3.63 | 0.039 | 0.969 |
| Duration of stay in the anesthesia care unit (minutes) | 62.38 ± 12.47 | 58.24 ± 10.85 | 2.377 | 0.018 |
| Gastrointestinal function recovery | ||||
| Bowel sounds recovery time (hours) | 7.52 ± 1.85 | 6.73 ± 1.42 | 3.259 | 0.001 |
| First exhaust time (hours) | 20.47 ± 4.32 | 18.63 ± 3.75 | 3.052 | 0.003 |
| First defecation time (hours) | 39.95 ± 8.24 | 36.82 ± 7.63 | 2.653 | 0.009 |
Cognitive function scores showed no significant differences between the DEX-CA and DEX-PA groups preoperatively (P = 0.691; Table 6). However, significant differences were noted at 6 hours and 12 hours postoperatively. At 6 hours postoperatively, the DEX-CA group had lower scores than the DEX-PA group (P = 0.008), indicating poorer cognitive function. This difference persisted at 12 hours postoperatively (P = 0.025). However, by 24 hours postoperatively, no significant difference in cognitive function scores was observed between the two groups (P > 0.05), a trend that continued at 48 hours postoperatively (P > 0.05).
| Parameter | DEX conventional anesthesia group (n = 95) | DEX preconditioning anesthesia group (n = 87) | t value | P value |
| Preoperative | 27.35 ± 1.24 | 27.42 ± 1.18 | 0.398 | 0.691 |
| Postoperative 6 hours | 24.72 ± 2.15 | 25.53 ± 1.87 | 2.683 | 0.008 |
| Postoperative 12 hours | 25.63 ± 1.96 | 26.24 ± 1.65 | 2.254 | 0.025 |
| Postoperative 24 hours | 26.85 ± 1.47 | 26.92 ± 1.32 | 0.296 | 0.767 |
| Postoperative 48 hours | 26.93 ± 1.25 | 27.15 ± 1.18 | 1.236 | 0.218 |
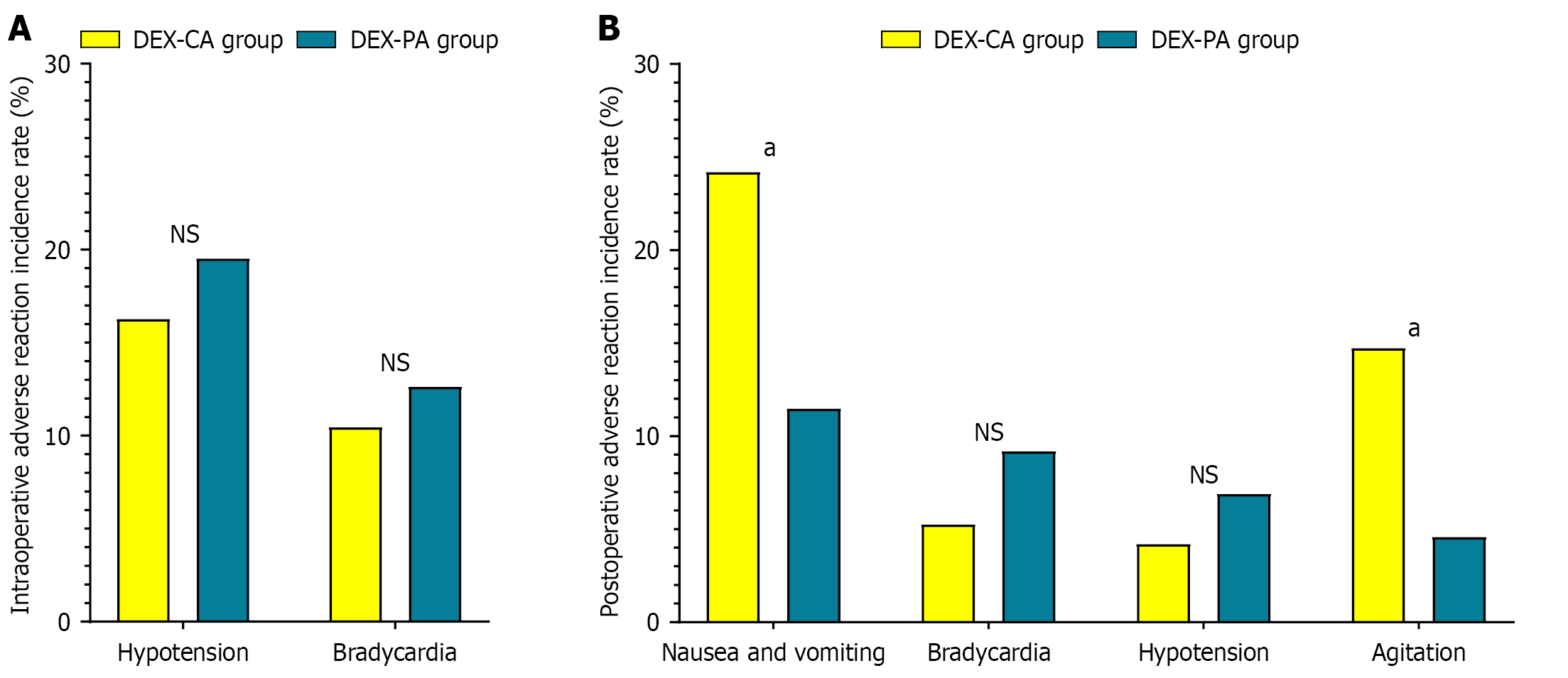
When comparing intraoperative complications between the DEX-CA and DEX-PA groups, no significant differences were observed. Specifically, no significant differences in the incidence of intraoperative hypotension (P = 0.576) and brady
This retrospective study demonstrated that administering DEX prior to anesthesia induction may offer greater benefits than CA alone in elderly patients undergoing LC. Findings across multiple domains of postoperative recovery suggest that early administration of DEX may help modulate the body’s response to surgical stress, contributing to more favorable recovery outcomes in this patient population.
The pain scores recorded at all postoperative time points in the DEX-PA group underscored its effective and sustained analgesic effect. This finding aligns with the established analgesic properties of DEX, mediated through central α2-adrenergic receptor activation in the locus coeruleus and spinal cord, inhibiting the release of nociceptive neurotransmitters[20]. Notably, although both groups received continuous infusion of DEX during surgery, the superior per
Biochemical analysis revealed notable differences in oxidative stress and systemic inflammatory response markers. The DEX-PA group demonstrated lower levels of MDA, a marker of lipid peroxidation and oxidative damage, along with higher levels of SOD and TAC. These findings suggest that DEX pre-treatment effectively reduced oxidative stress in the early postoperative period. In addition, the DEX-PA group exhibited reduced levels of Cor, TNF-α, and CRP, indicating a dampened surgical stress and inflammation. These results align with growing clinical and preclinical evidence supporting the anti-inflammatory and antioxidant properties of DEX[27]. Previous studies have shown that elevated oxidative stress correlate with poorer postoperative outcomes, including prolonged recovery times and increased complication risks[28]. Activation of central α2-adrenergic receptors by DEX can inhibit sympathetic outflow, reducing catecholamine surges and subsequent pro-inflammatory cytokine release[29]. Moreover, DEX may directly modulate inflammatory signaling pathways, such as nuclear factor kappa B inhibition, and enhance endogenous antioxidant defenses[30]. Our findings reinforce the role of DEX in reducing inflammatory markers, highlight the effectiveness of the pre-treatment strategy in elderly patients undergoing LC.
Accelerated recovery was a notable feature of the DEX-PA group. Patients in this group experienced shorter awa
Cognitive function assessments indicated that the DEX-PA group exhibited a brief but clear protective effect during the critical early recovery phase. Higher cognitive scores at 6 hours and 12 hours postoperatively suggested that DEX pre-treatment may provide early neuroprotection against POCD, which is particularly important for older adults. These findings are consistent with proposed neuroprotection mechanisms of DEX reported in previous studies, including reducing central inflammation, mitigating excitotoxicity, lowering cerebral metabolic rate, and improving cerebral perfusion stability[34]. The absence of these differences beyond 24 hours implied that pre-treatment primarily mitigates acute neuroinflammatory responses triggered by surgery and anesthesia, rather than preventing long-term dysfunction. The MMSE tool may have lacked the sensitivity to capture subtle or prolonged cognitive changes in this specific context. Nonetheless, our findings emphasized the effects of a pre-treatment approach during the early stages, adding nuanced insight into its potential role in mitigating immediate postoperative cognitive decline.
Safety data further supported the advantages of the DEX-PA group. Lower rates of nausea and vomiting aligned with improved pain relief and reduced opioid requirements, given that opioids are a primary trigger to postoperative nausea and vomiting[35]. In addition, reduced agitation rates indicated a smoother emergence and recovery process associated with DEX. Importantly, no notable differences in hemodynamic adverse events, such as bradycardia and hypotension, were observed between the DEX-CA and DEX-PA groups during surgery and postoperatively, which is reassuring. Although DEX is known to produce dose-dependent hemodynamic effects, the specific doses and timing used in this study, coupled with vigilant monitoring protocols, appeared to be well-tolerated in elderly patients with ASA I and II status[36,37]. This favorable safety profile, alongside the observed clinical benefits, further enhanced the potential clinical utility of this approach.
Several limitations of this study merit consideration. As a single-center retrospective analysis, the results might not generalize to broader populations or different healthcare settings. Although we conducted rigorous statistical ad
Future research should focus on conducting prospective, randomized, double-blind, multicenter controlled trials to address these limitations. This approach is essential for confirming the efficacy and safety of DEX pre-treatment in this population. Such studies should use more sensitive cognitive assessment tools, closely monitor opioid consumption, and extend follow-up times to observe long-term recovery and complications. Delving into potential mechanisms is also crucial. Exploring the optimal dosing regimen for pre-treatment and comparing it with other regional or multimodal analgesia strategies would provide valuable insights. Future studies could incorporate more objective indicators, such as postoperative changes in serum gastrointestinal hormone levels, to more accurately assess gastrointestinal function recovery. In addition, assessing economic impacts, particularly the cost-effectiveness associated with potentially shor
In elderly patients undergoing LC, PA with DEX administered prior to induction was associated with reduced oxidative stress, attenuated systemic inflammation, and improved postoperative pain control. This approach also helped facilitated faster recovery of gastrointestinal function and cognitive performance. Compared with CA approaches, administering DEX at this critical time enhanced clinical outcomes and safety, offering a promising direction for optimizing perioperative results in this vulnerable population.
| 1. | Li ZZ, Guan LJ, Ouyang R, Chen ZX, Ouyang GQ, Jiang HX. Global, regional, and national burden of gallbladder and biliary diseases from 1990 to 2019. World J Gastrointest Surg. 2023;15:2564-2578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 2. | BK B, Adhikari A, Gyawali G. In-vitro Dissolution Study of Gallstone with Medicinal Plant Extracts. BIBECHANA. 2023;20:175-182. [DOI] [Full Text] |
| 3. | Dahiya DS, Facciorusso A, Chandan S, Sohail AH, Gangwani MK, Franchellucci G, Mangiavillano B. Understanding evidence-based clinical practice guidelines for cholelithiasis 2021. Hepatobiliary Surg Nutr. 2024;13:352-355. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 4. | Inzunza M, Irarrazaval MJ, Pozo P, Pimentel F, Crovari F, Ibañez L. Gallbladder polyps: Correlation and agreement between ultrasonographic and histopathological findings in a population with high incidence of gallbladder cancer. Arq Bras Cir Dig. 2023;36:e1732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 5. | Gerosa M, Chiarelli M, Maggioni D, Cioffi U, Guttadauro A. Acute biliary pancreatitis: the current role of endoscopic and minimally invasive surgical procedures. Ann Ital Chir. 2023;94:36-44. [PubMed] |
| 6. | Fu Y, Wei Q, Wang Z, Zhao Q, Shi W. Effects of dexmedetomidine on postoperative pain and early cognitive impairment in older male patients undergoing laparoscopic cholecystectomy. Exp Ther Med. 2024;27:189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 7. | Zhang L, Qiu Y, Zhang ZF, Zhao YF, Ding YM. Current perspectives on postoperative cognitive dysfunction in geriatric patients: insights from clinical practice. Front Med (Lausanne). 2024;11:1466681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 8. | Tippireddy S, Ghatol D. Anesthetic Management for Enhanced Recovery After Major Surgery (ERAS). 2023 Jan 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 9. | Hou M, Chen F, He Y, Tan Z, Han X, Shi Y, Xu Y, Leng Y. Dexmedetomidine against intestinal ischemia/reperfusion injury: A systematic review and meta-analysis of preclinical studies. Eur J Pharmacol. 2023;959:176090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 10. | He G, Ni H, Wang K, Gao H, Li Y, Gu J, Ni X, Wang Z, Bao Y. Dexmedetomidine attenuates the neuroinflammation and cognitive dysfunction in aged mice by targeting the SNHG14/miR340/NFκB axis. Biomed Rep. 2023;19:100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 11. | Aboulata AA, Shatla IM. Physiology and immunology of the adrenergic anti-inflammatory pathway. Egypt J Immunol. 2024;31:46-57. [PubMed] |
| 12. | Bu HM, Zhao M, Ma HM, Tian XP. Application value of dexmedetomidine in anesthesia for elderly patients undergoing radical colon cancer surgery. World J Gastrointest Surg. 2024;16:2671-2678. [PubMed] [DOI] [Full Text] |
| 13. | Koh HJ, Joo J. The Role of Cytokines in Perioperative Neurocognitive Disorders: A Review in the Context of Anesthetic Care. Biomedicines. 2025;13:506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 14. | Gutt C, Schläfer S, Lammert F. The Treatment of Gallstone Disease. Dtsch Arztebl Int. 2020;117:148-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 93] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 15. | McCain RS, Diamond A, Jones C, Coleman HG. Current practices and future prospects for the management of gallbladder polyps: A topical review. World J Gastroenterol. 2018;24:2844-2852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 83] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (7)] |
| 16. | Irlbeck T, Zwißler B, Bauer A. [ASA classification : Transition in the course of time and depiction in the literature]. Anaesthesist. 2017;66:5-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 17. | Williamson A, Hoggart B. Pain: a review of three commonly used pain rating scales. J Clin Nurs. 2005;14:798-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2353] [Cited by in RCA: 2009] [Article Influence: 95.7] [Reference Citation Analysis (0)] |
| 18. | Rasheed AM, Amirah MF, Abdallah M, P J P, Issa M, Alharthy A. Ramsay Sedation Scale and Richmond Agitation Sedation Scale: A Cross-sectional Study. Dimens Crit Care Nurs. 2019;38:90-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 19. | Costa D, Severo M, Fraga S, Barros H. Mini-Cog and Mini-Mental State Examination: agreement in a cross-sectional study with an elderly sample. Dement Geriatr Cogn Disord. 2012;33:118-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 20. | Elkenany S, Elsaeed MM, Abdelmageed NA, Badr ME. Effect of Intrathecal Fentanyl and Dexmedetomidine On Rebound Pain following Orthopedic Surgery: A prospective Randomized Comparative Study. Int J Med Arts. 2024;6:5092-5098. [DOI] [Full Text] |
| 21. | Weerink MAS, Barends CRM, Muskiet ERR, Reyntjens KMEM, Knotnerus FH, Oostra M, van Bocxlaer JFP, Struys MMRF, Colin PJ. Pharmacodynamic Interaction of Remifentanil and Dexmedetomidine on Depth of Sedation and Tolerance of Laryngoscopy. Anesthesiology. 2019;131:1004-1017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 22. | Li Y, Qi L, Wang Z, Wang W, Zhang L, Yang L, Liu C, Zhong W, Wang X. Pharmacokinetics, pharmacodynamics and bioavailability of dexmedetomidine nasal spray in healthy Chinese adults: A phase I clinical trial. Front Pharmacol. 2024;15:1488462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 23. | Yang Y, Zhang L, Yao W. Anesthetic Effects of Dexmedetomidine Combined with Nalbuphine in Patients Undergoing Laparoscopic Cholecystectomy and its Impact on Nutritional Status. Ann Ital Chir. 2024;95:832-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 24. | Yu DH, Shen X, Lai L, Chen YJ, Liu K, Shen QH. Application of Dexmedetomidine as an Opioid Substitute in Opioid-Free Anesthesia: A Systematic Review and Meta-analysis. Pain Physician. 2023;26:E635-E649. [PubMed] |
| 25. | Afrati SR, Lianou I, Kaspiris A, Marougklianis V, Kotanidou A, Pneumaticos SG. Analgesic and Neuroprotective Roles of Dexmedetomidine in Spine Surgery: A Systematic Review. Diseases. 2025;13:212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 26. | Hailu S, Abbabu S, Seifu A, Gorde N, Besha A. Effectiveness of different doses of dexmedetomidine on intraoperative haemodynamic profiles and postoperative pain in patients undergoing abdominal surgery at Dilla University Referral Hospital, Ethiopia, 2024: a double-blind randomized controlled trial. Ann Med Surg (Lond). 2024;86:4495-4504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 27. | Pöyhiä R, Nieminen T, Tuompo VWT, Parikka H. Effects of Dexmedetomidine on Basic Cardiac Electrophysiology in Adults; a Descriptive Review and a Prospective Case Study. Pharmaceuticals (Basel). 2022;15:1372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 28. | Wang T, Zhao W, Ma L, Wu J, Ma X, Liu L, Cao J, Lou J, Mi W, Zhang C. Higher fraction of inspired oxygen during anesthesia increase the risk of postoperative pulmonary complications in patients undergoing non-cardiothoracic surgery: a retrospective cohort study. Front Physiol. 2024;15:1471454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 29. | Anselmo M, Tahsin CT, Bigalke JA. The role of central α(2)-adrenergic activity in sympathetic outflow: a potential intervention to ease the mind (and body)? J Physiol. 2024;602:5991-5993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 30. | Zhou Y, Du X, Wang Q, Xiao S, Zhi J, Gao H, Yang D. Dexmedetomidine Protects against Airway Inflammation and Airway Remodeling in a Murine Model of Chronic Asthma through TLR4/NF-κB Signaling Pathway. Mediators Inflamm. 2023;2023:3695469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 31. | Singhal A, Taksande K. Role of Adjuvants in Enhancing the Efficacy and Duration of Anesthesia Blocks: A Comprehensive Review. Cureus. 2024;16:e69880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 32. | Huang SS, Song FX, Yang SZ, Hu S, Zhao LY, Wang SQ, Wu Q, Liu X, Qi F. Impact of intravenous dexmedetomidine on postoperative bowel movement recovery after laparoscopic nephrectomy: A consort-prospective, randomized, controlled trial. World J Clin Cases. 2021;9:7762-7771. [PubMed] [DOI] [Full Text] |
| 33. | Garcia-Alonso I, Velasco-Oraa X, Cearra I, Iturrizaga Correcher S, Mar Medina C, Alonso-Varona A, García Ruiz de Gordejuela A, Ruiz-Montesinos I, Herrero de la Parte B. Prophylactic Treatment of Intestinal Ischemia-Reperfusion Injury Reduces Mucosal Damage and Improves Intestinal Absorption. J Inflamm Res. 2023;16:4141-4152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 34. | Yuan HX, Zhang LN, Li G, Qiao L. Brain protective effect of dexmedetomidine vs propofol for sedation during prolonged mechanical ventilation in non-brain injured patients. World J Psychiatry. 2024;14:370-379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 35. | Koo BW. Achieving relief from nausea and vomiting: from intraoperative to postoperative management. Korean J Anesthesiol. 2024;77:573-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 36. | Tian LJ, Yao YT, Yuan S, Dai Z. Effect of Dexmedetomidine on Maintaining Perioperative Hemodynamic Stability in Elderly Patients: A Systematic Review and Meta-analysis. Chin Med Sci J. 2023;38:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 37. | Liu H, Gao M, Zheng Y, Sun C, Lu Q, Shao D. Effects of dexmedetomidine at different dosages on perioperative haemodynamics and postoperative recovery quality in elderly patients undergoing hip replacement surgery under general anaesthesia: a randomized controlled trial. Trials. 2023;24:386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/