Published online Feb 27, 2026. doi: 10.4240/wjgs.v18.i2.112776
Revised: October 27, 2025
Accepted: December 11, 2025
Published online: February 27, 2026
Processing time: 156 Days and 3 Hours
Endoscopic submucosal dissection (ESD) is applicable to early-stage gastrointes
To evaluated the efficacy and safety profile of ESD compared with conventional gastrectomy for managing early gastrointestinal lesions, emphasizing clinical outcomes and postoperative recovery.
A retrospective review was conducted on 206 patients diagnosed with early-stage gastrointestinal lesions at the Affiliated Hospital of North Sichuan Medical College between June 2021 and June 2024. Patients were divided into two groups: 98 underwent gastrectomy and 108 received ESD. Outcomes assessed included overall treatment efficacy, postoperative complications, serum tumor marker levels, gastrointestinal hormone concentrations, and mucosal barrier integrity indices.
The ESD group showed higher overall treatment efficacy (98.15% vs 92.86%; P > 0.05) and a significant lower complication rate (5.56% vs 13.27%). ESD was also associated with reduced intraoperative blood loss (58.36 mL vs 60.31 mL; P < 0.001) and shorter operative durations (69.21 minutes vs 69.86 minutes; P = 0.001). Gastrointestinal function recovered more rapidly following ESD, with an earlier return to oral intake (2.43 days vs 2.65 days; P = 0.006). Preoperatively, ESD patients exhibited lower thymidine kinase 1 and D-lactate levels, indicating enhanced preservation of mucosal integrity.
For early gastrointestinal lesions, ESD represents a safe and effective alternative to conventional gastrectomy. It enables precise lesion excision with fewer complications and faster postoperative recovery.
Core Tip: This study compared endoscopic submucosal dissection and conventional gastrectomy in 206 patients with early gastrointestinal lesions. The findings demonstrate endoscopic submucosal dissection significant clinical advantages, including reduced intraoperative bleeding, shorter operative time, and faster postoperative recovery. This study carries clinical implications, establishing endoscopic submucosal dissection as a preferred treatment option that can significantly reduce surgical trauma, shorten the perioperative process, and promote the rapid recovery of patients with early gastrointestinal lesions.
- Citation: Ling Y, Wang J, Xi CH, Liu J. Efficacy of endoscopic submucosal dissection in the treatment of early gastrointestinal lesions. World J Gastrointest Surg 2026; 18(2): 112776
- URL: https://www.wjgnet.com/1948-9366/full/v18/i2/112776.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v18.i2.112776
Endoscopic submucosal dissection (ESD) has emerged as a transformative approach for managing early gastrointestinal lesions[1]. Over the past two decades, the increasing prevalence of gastrointestinal malignancies has intensified research into more effective diagnostic and therapeutic strategies[2]. Among these, ESD - a minimally invasive procedure - offers high precision and reduced trauma compared with conventional gastrectomy[3]. Traditional surgical approaches often necessitate extensive resection, leading to substantial morbidity and delayed recovery[4]. These limitations underscore the need for innovative techniques that enhance safety and outcomes.
The primary advantage of ESD lies in its ability to achieve en bloc lesion excision while preserving surrounding healthy tissue, thereby minimizing postoperative complications[5]. Direct endoscopic visualization allows precise submucosal dissection and facilitates curative resection with negative margins[6,7]. This technique reduces tissue injury, preserves organ function, and accelerates recovery, making it particularly suitable for early-stage lesions requiring complete excision with minimal healthy tissue disruption[8].
In early gastric cancer management, ESD has revolutionized treatment paradigms by ensuring accurate mucosal resection and favorable short-term and long-term outcomes[9,10]. However, broader clinical adoption remains limited by the requirement of technical expertise and the need for specialized instrumentation[11]. With advancements in endo
This retrospective study included 206 patients with gastrointestinal lesions treated at the Affiliated Hospital of North Sichuan Medical College between June 2021 and June 2024. Based on the treatment modality, patients were categorized into two groups: Conventional surgery (gastrectomy; n = 98) and ESD (n = 108). Clinical outcomes, perioperative data, complications, and treatment efficacy were obtained from the hospital’s electronic medical record system, along with demographic baseline information.
This retrospective study was approved by the Medicine Ethics Committee of Affiliated Hospital of North Sichuan Medical College. Informed consent was waived because the research involved retrospective analysis of anonymized data and posed no risk to patients, in accordance with ethical guidelines for retrospective studies.
Inclusion criteria: (1) Patients in generally good health, without severe systemic disease or organ dysfunction, and capable of tolerating routine examinations and treatments; (2) Absence of coagulation disorders, severe gastrointestinal bleeding or perforation, cognitive impairments, or other congenital conditions; and (3) No prior clinical treatment for gastrointestinal disorders and fulfillment of surgical indications.
Exclusion criteria: (1) Contraindications to clinical examinations or participation in other clinical studies; and (2) Non
Upon admission, all patients underwent comprehensive clinical evaluation, including temperature, respiratory rate, and skin color assessment. Before surgery, both groups received standard preoperative management for gastrointestinal lesions, consisting of anti-inflammatory and antibacterial therapy, fasting, restricted water intake, electrolyte maintenance through fluid supplementation, and nutritional support. Postoperatively, patients received health education and were advised to follow a high-protein, high-fiber diet while avoiding smoking, alcohol, and high-fat foods.
Gastrectomy: Conventional open abdominal resection surgery was performed under general anesthesia with the patient in a supine position. A 5-cm incision was made at the intersection of the abdominal midline and the umbilicus. The skin and subcutaneous tissues were dissected layer by layer to expose the lesion and determine its location, shape, size, and distribution. The tumor was then partially or completely excised to ensure no residual tissue remained. The abdominal cavity was irrigated, a drainage tube was placed, and closure was achieved with layered absorbable sutures.
ESD: For ESD, the lesion’s outer margin was stained and marked approximately 3 mm from the edge to delineate the affected area. Using an endoscopic injection needle, a 1:10000 adrenaline-saline solution (National Medical Products Administration Standard H11020584; specification: 1 mL:1 mg) was injected into the submucosal layer at multiple sites (1.5 mL per point). The injection caused the lesion to protrude, facilitating visualization. A hook knife was used to incise the mucosa and dissect deep into the submucosal layer. Because the body gradually absorbs the adrenaline-saline, repeated injections were administered as necessary. Upon completion of the submucosal dissection, a metal titanium clip release device was employed. This clip was positioned perpendicularly to the blood vessel at the bleeding site, and the operator compressed the rod on both sides of the vessel to secure and detach the clip, effectively sealing the vessel and surrounding tissue. The number of titanium clips used depended on intraoperative findings. After hemostasis, the bleeding site was repeatedly irrigated with adrenaline-saline to ensure complete bleeding control.
Treatment outcomes were classified as follows: Significant effect - normal digestive function and bowel movements with intact mucosa confirmed via endoscopy; effective - improvement in clinical symptoms despite residual mucosal erosion; and ineffective - no symptomatic improvement. The total treatment effective rate was calculated as the sum of sig
A 3-mL fasting venous blood sample was obtained from each patient before surgery and 1 month postoperatively. Samples were centrifuged at 3500 rpm for 10 minutes, and serum was collected for quantification of carcinoembryonic antigen (CEA)[14], carbohydrate antigen 199 (CA199), and thymidine kinase 1 (TK1) using chemiluminescence assays.
Peripheral venous blood (5.0 mL) was collected from both groups preoperatively and on postoperative day 3 and day 7. Samples were centrifuged using a medical centrifuge with a 15 cm radius, set at 3000 rpm for 5 minutes, and the resulting serum was analyzed for gastrointestinal hormones - vasoactive intestinal peptide (VIP)[15], gastrin (GAS), and motilin (MTL) - as well as intestinal mucosal barrier makers: Diamine oxidase (DAO)[16], endotoxin (ET), nitric oxide (NO), and D-lactate (D-Lac). Two experienced technicians performed all measurements in duplicate using enzyme-linked immunosorbent assay. Serum indices from both groups were statistically compared across all three time points.
Data were standardized and cleaned before analysis to correct inconsistencies, entry errors, and missing values. Duplicates were removed, entry errors were corrected, and missing data were imputed using the k-nearest neighbors method via the Python impute library (version 3.6.0). The k-nearest neighbors algorithm employed a k-dimensional tree to identify nearest neighbors and computed their weighted averages for imputation.
Missing data were limited to < 5% to minimize bias. Sensitivity analyses considered both best-case and worst-case assumptions for loss to follow-up; consistent conclusions indicated negligible influence of missing data. Final analyses incorporated datasets with imputed values.
All statistical analyses were performed using SPSS version 29.0 (SPSS Inc., Chicago, IL, United States). Categorical data were expressed as n (%), and comparisons were made using the χ2 test: The basic formula was applied when total sample size was ≥ 40 and theoretical frequency T ≥ 5; Yates’ correction was used when 1 ≤ T < 5; and Fisher’s exact test was employed when sample size < 40 or T < 1. Continuous variables were tested for normality using the Shapiro-Wilk method. Normally distributed data were reported as mean ± SD and compared using the t-test; non-normally distributed data were expressed as median (25th percentile, 75th percentile) and analyzed using the Wilcoxon rank-sum test. A P < 0.05 was considered statistically significant.
A comparative analysis of baseline characteristics between the gastrectomy (n = 98) and ESD (n = 108) groups revealed no statistically significant differences across all measured parameters (Table 1). The mean ages were 47.30 ± 1.22 years and 47.12 ± 1.40 years, respectively (P = 0.633). Gender distribution was comparable, with males representing 59.18% in the gastrectomy group and 57.41% in the ESD group (P = 0.796). Body mass index was similarly matched at 25.42 ± 3.25 kg/m2 and 25.77 ± 3.36 kg/m2 for the gastrectomy and ESD groups, respectively (P = 0.445). Smoking and drinking histories were nearly identical (60.2% vs 60.19%, P = 0.998; 57.14% vs 57.41%, P = 0.969) in both groups. Employment status, disease duration, and the prevalence of hypertension and diabetes also showed no significant group differences (P > 0.05). Lesion locations - esophageal, gastric, or colonic - were similarly distributed (P = 0.723), and prior endoscopic procedures also showed no statistical significance (P = 0.190). Overall, the baseline characteristics were well matched between the treatment groups.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | t/χ2 | P value |
| Age (years) | 47.30 ± 1.22 | 47.12 ± 1.40 | 0.478 | 0.633 |
| Gender (male/female) | 58 (59.18) | 62 (57.41) | 0.067 | 0.796 |
| Body mass index (kg/m2) | 25.42 ± 3.25 | 25.77 ± 3.36 | 0.765 | 0.445 |
| Smoking history | 59 (60.2) | 65 (60.19) | 0 | 0.998 |
| Drinking history | 56 (57.14) | 62 (57.41) | 0.001 | 0.969 |
| Employment | 77 (78.57) | 89 (82.41) | 0.483 | 0.487 |
| Disease duration (weeks) | 3.00 ± 0.19 | 3.03 ± 0.17 | 1.007 | 0.315 |
| Hypertension | 17 (17.35) | 20 (18.52) | 0.048 | 0.827 |
| Diabetes | 26 (26.53) | 31 (28.7) | 0.121 | 0.728 |
| Location of lesion | 0.647 | 0.723 | ||
| Esophageal lesions | 32 (32.65) | 37 (34.26) | ||
| Gastric lesions | 31 (31.63) | 38 (35.19) | ||
| Colonic lesions | 35 (35.71) | 33 (30.56) | ||
| Previous endoscopic procedures | 3.325 | 0.19 | ||
| None | 50 (51.02) | 58 (53.7) | ||
| 1 or 2 | 33 (33.67) | 42 (38.89) | ||
| ≥ 3 | 15 (15.31) | 8 (7.41) |
The ESD group exhibited a slightly higher total effective rate (98.15%) than the gastrectomy group (92.86%; Table 2). In the gastrectomy group, 54 patients (55.10%) demonstrated a significant effect and 37 (37.76%) showed effectiveness, while 7 (14%) were classified as invalid. In the ESD group, 63 (58.33%) and 43 (39.81%) patients were classified as significantly effective and effective, respectively, with only 2 (1.85%) showing no improvement. The overall difference in total treatment effectiveness was not statistically significant (χ2 = 2.293, P > 0.05), indicating comparable effectiveness between the two approaches for early gastrointestinal lesions.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | χ2 | P value |
| Significant effect | 54 (55.10) | 63 (58.33) | ||
| Effective | 37 (37.76) | 43 (39.81) | ||
| Invalid | 7 (14) | 2 (1.85) | ||
| Total effective | 91 (92.86) | 106 (98.15) | 2.293 | > 0.05 |
The total complication rate was lower in the ESD group (5.56%) than in the gastrectomy group (13.27; Table 3). Gastrointestinal bloating occurred in 5 patients (5.10%) vs 3 (2.78%), infection in 3 (3.06%) vs 1 (0.93%), and gastrointes
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | χ2 | P value |
| Gastrointestinal bloating | 5 (5.1) | 3 (2.78) | ||
| Infected | 3 (3.06) | 1 (0.93) | ||
| Gastrointestinal perforation | 5 (5.1) | 2 (1.85) | ||
| Total | 13 (13.27) | 6 (5.56) | 3.648 | > 0.05 |
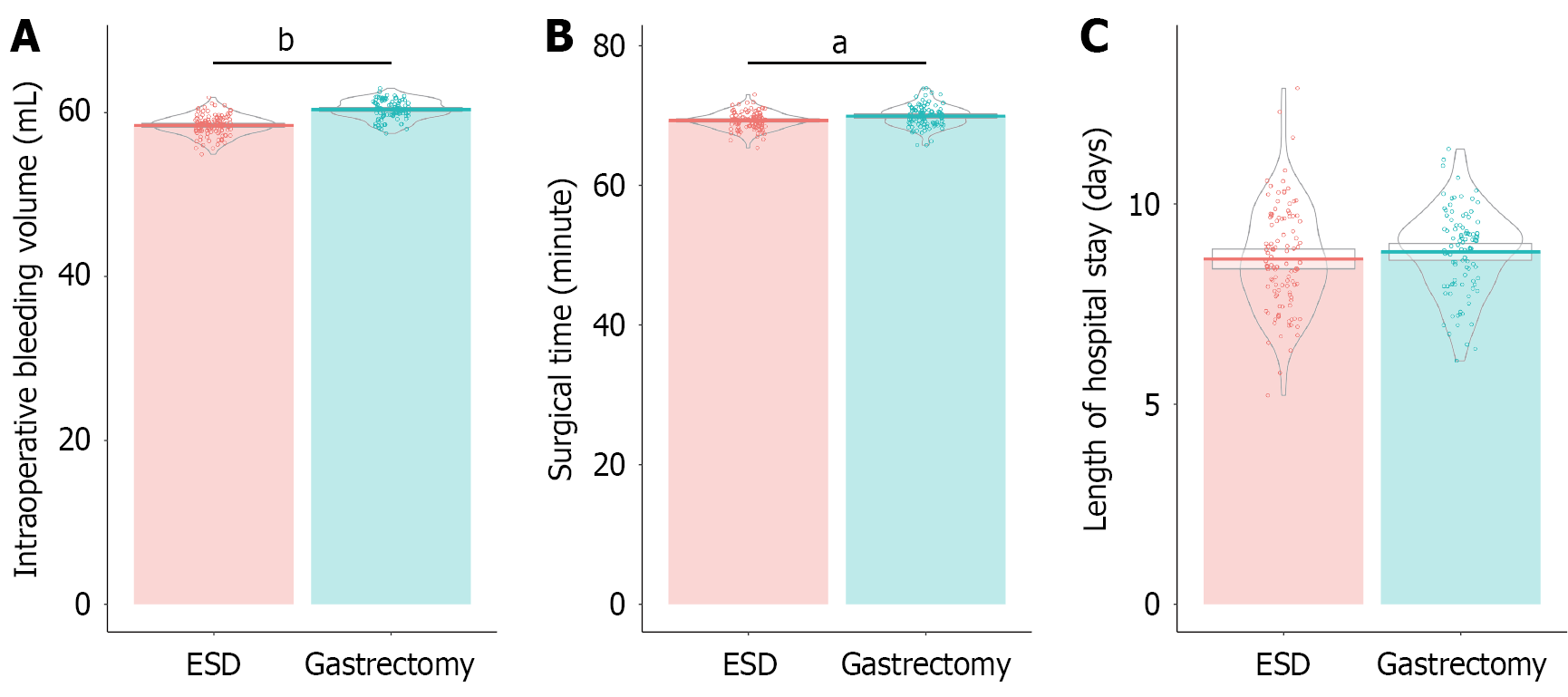
The ESD group demonstrated significantly reduced intraoperative blood loss (58.36 ± 1.25 mL) compared with the gastrectomy group (60.31 ± 1.15 mL; t = 11.583, P < 0.001; Figure 1). Mean surgical time was also significantly shorter for the ESD group (69.21 ± 1.24 minutes) than for the gastrectomy group (69.86 ± 1.56 minutes; t = 3.293, P = 0.001). The average hospital stays differed insignificantly (8.62 ± 1.31 days for ESD vs 8.81 ± 1.05 days for gastrectomy; P = 0.257). These results suggest that ESD offers notable intraoperative advantages in terms of reduced bleeding and shorter operative duration while maintaining similar hospital stay durations.
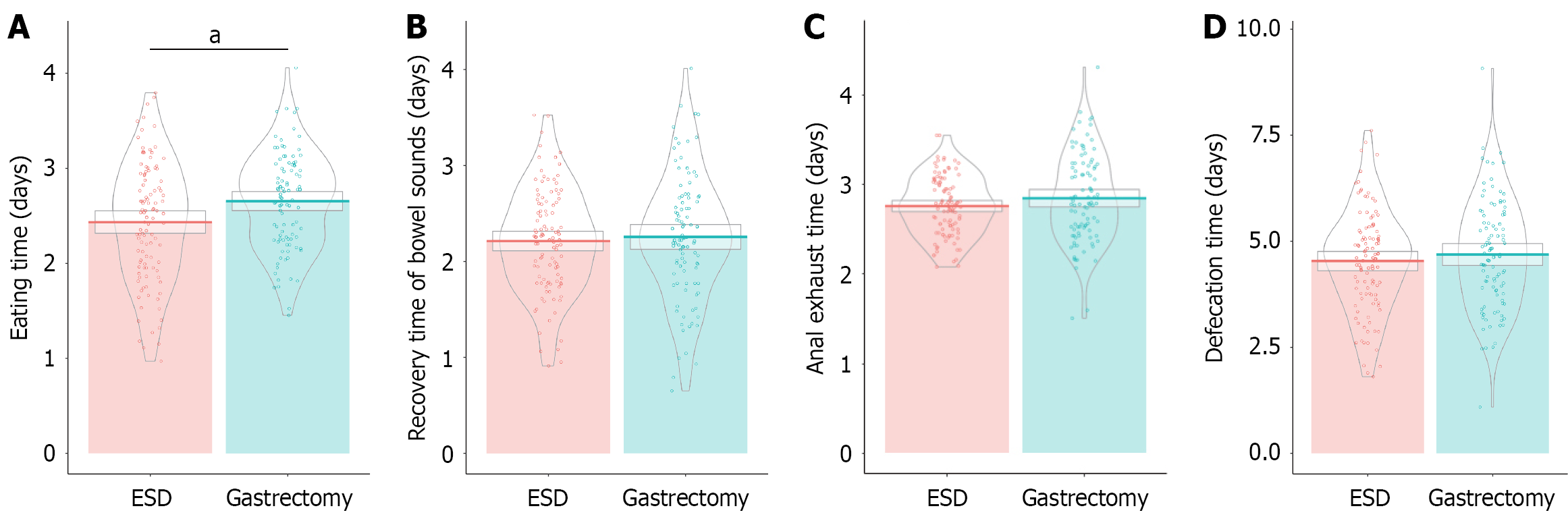
Patients in the ESD group resumed eating significantly earlier, averaging 2.43 ± 0.63 days compared with 2.65 ± 0.51 days in the gastrectomy group (t = 2.753, P = 0.006; Figure 2). Recovery time of bowel sounds was comparable between groups (2.21 ± 0.54 days for ESD vs 2.25 ± 0.66 days for gastrectomy, P = 0.588). Additionally, anal exhaust and defecation times did not show statistically significant differences, with anal exhaust occurring at 2.75 ± 0.33 days in the ESD group and 2.84 ± 0.49 days in the gastrectomy group (P = 0.133), whereas defecation times at 4.52 ± 1.21 days and 4.68 ± 1.30 days, respectively (P = 0.369). Overall, ESD enabled earlier food intake, with other postoperative recovery parameters showing no significant intergroup differences.
Postoperatively, the mean TK1 level was significantly lower in the ESD group (3.69 ± 1.21 pmol/L) than in the gastrectomy group (4.20 ± 1.61 pmol/L; t = 2.554, P = 0.011) (Table 4). However, CEA and CA199 showed no significant differences between groups. Mean CEA levels were 22.33 ± 3.56 μg/L for gastrectomy and 23.01 ± 3.98 μg/L for ESD (P = 0.201), whereas CA199 levels were 42.98 ± 5.55 kU/L and 42.49 ± 5.93 kU/L, respectively (P = 0.536). Thus, apart from the lower TK1 levels in the ESD group, the levels of other serum tumor markers were comparable between the groups preoperatively.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | t | P value |
| CEA (μg/L) | 22.33 ± 3.56 | 23.01 ± 3.98 | 1.284 | 0.201 |
| CA199 (kU/L) | 42.98 ± 5.55 | 42.49 ± 5.93 | 0.621 | 0.536 |
| TK1 (pmol/L) | 4.20 ± 1.61 | 3.69 ± 1.21 | 2.554 | 0.011 |
The postoperatively, the mean CA199 level was significantly lower in the gastrectomy group (22.01 ± 3.21 kU/L) than in the ESD group (23.32 ± 3.66 kU/L; t = 2.716, P = 0.007; Table 5). In contrast, CEA and TK1 Levels differed insignificantly between the groups, with mean CEA levels of 9.77 ± 1.63 μg/L for gastrectomy and 9.54 ± 1.69 μg/L for ESD (P = 0.319), and mean TK1 levels of 1.02 ± 0.25 pmol/L and 0.99 ± 0.15 pmol/L, respectively (P = 0.288). These findings indicate that the 1-month postoperative levels of CA199 differed significantly between groups, whereas those of CEA and TK1 did not.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | t | P value |
| CEA (μg/L) | 9.77 ± 1.63 | 9.54 ± 1.69 | 0.999 | 0.319 |
| CA199 (kU/L) | 22.01 ± 3.21 | 23.32 ± 3.66 | 2.716 | 0.007 |
| TK1 (pmol/L) | 1.02 ± 0.25 | 0.99 ± 0.15 | 1.065 | 0.288 |
Postoperatively, the mean level D-Lac was significantly lower in the ESD group (8.65 ± 1.17 μg/mL) than in the gastrectomy group (9.11 ± 1.32 μg/mL; t = 2.623, P = 0.009) (Table 6). Other makers - VIP, GAS, MTL, DAO, ET, and NO - showed no statistically significant differences. Mean VIP levels were 139.36 ± 16.13 ng/L for gastrectomy and 138.84 ± 15.33 ng/L for ESD (P = 0.815); GAS 16.78 ± 2.53 ng/L vs 16.97 ± 2.58 ng/L (P = 0.595); MTL 296.33 ± 32.01 ng/L vs 298.35 ± 30.84 ng/L (P = 0.645); DAO 1.33 ± 0.19 U/mL vs 1.36 ± 0.21 U/mL (P = 0.293); ET 0.21 ± 0.03 EU/mL vs 0.22 ± 0.05 EU/mL (P = 0.294); and NO 62.33 ± 7.68 μmol/L vs 63.02 ± 7.39 μmol/L (P = 0.514). These findings suggest that aside from the lower D-Lac level, preoperative gastrointestinal hormones and mucosal barrier markers were broadly similar between the two groups.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | t | P value |
| VIP (ng/L) | 139.36 ± 16.13 | 138.84 ± 15.33 | 0.235 | 0.815 |
| GAS (ng/L) | 16.78 ± 2.53 | 16.97 ± 2.58 | 0.533 | 0.595 |
| MTL (ng/L) | 296.33 ± 32.01 | 298.35 ± 30.84 | 0.461 | 0.645 |
| DAO (U/mL) | 1.33 ± 0.19 | 1.36 ± 0.21 | 1.055 | 0.293 |
| ET (EU/mL) | 0.21 ± 0.03 | 0.22 ± 0.05 | 1.053 | 0.294 |
| NO (μmol/L) | 62.33 ± 7.68 | 63.02 ± 7.39 | 0.654 | 0.514 |
| D-Lac (μg/mL) | 9.11 ± 1.32 | 8.65 ± 1.17 | 2.623 | 0.009 |
The postoperatively, the mean DAO level was significantly higher in the gastrectomy group (2.05 ± 0.33 U/mL) than in the ESD group (1.92 ± 0.26 U/mL; t = 3.249, P = 0.001) (Table 7). Similarly, the mean ET level was slightly higher in the ESD group (0.34 ± 0.04 EU/mL) than in the gastrectomy group (0.33 ± 0.05 EU/mL; t = 2.187, P = 0.030). The mean D-Lac level was notably higher in the gastrectomy group (12.56 ± 1.77 μg/mL) than in the ESD group (12.03 ± 1.36 μg/mL; t = 2.37, P = 0.019). No significant differences were found between groups for VIP, GAS, MTL, or NO. Specifically, VIP levels were 95.66 ± 9.11 ng/L vs 96.33 ± 13.25 ng/L (P = 0.673); GAS 13.66 ± 1.99 ng/L vs 13.88 ± 2.01 ng/L (P = 0.44); MTL 235.96 ± 26.17 ng/L vs 236.33 ± 28.71 ng/L (P = 0.922); and NO 79.26 ± 9.33 μmol/L vs 79.55 ± 8.21 μmol/L (P = 0.814). These results suggest that gastrectomy led to greater disruption to the intestinal mucosal barrier, as reflected by elevated DAO, ET, and D-Lac levels compared with ESD.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | t | P value |
| VIP (ng/L) | 95.66 ± 9.11 | 96.33 ± 13.25 | 0.423 | 0.673 |
| GAS (ng/L) | 13.66 ± 1.99 | 13.88 ± 2.01 | 0.773 | 0.44 |
| MTL (ng/L) | 235.96 ± 26.17 | 236.33 ± 28.71 | 0.098 | 0.922 |
| DAO (U/mL) | 2.05 ± 0.33 | 1.92 ± 0.26 | 3.249 | 0.001 |
| ET (EU/mL) | 0.33 ± 0.05 | 0.34 ± 0.04 | 2.187 | 0.030 |
| NO (μmol/L) | 79.26 ± 9.33 | 79.55 ± 8.21 | 0.235 | 0.814 |
| D-Lac (μg/mL) | 12.56 ± 1.77 | 12.03 ± 1.36 | 2.37 | 0.019 |
At 7 days postoperatively, the mean D-Lac level remained significantly higher in the gastrectomy group (11.63 ± 1.36 μg/mL) than in the ESD group (11.18 ± 1.31 μg/mL; t = 2.445, P = 0.015; Table 8). Other indices showed no significant intergroup differences: VIP levels were 103.55 ± 10.36 ng/L (gastrectomy) vs 103.53 ± 12.96 ng/L (ESD; P = 0.989); GAS 16.23 ± 2.25 ng/L vs 16.11 ± 2.63 ng/L (P = 0.746); MTL 258.93 ± 28.77 ng/L vs 257.00 ± 25.93 ng/L (P = 0.539); DAO 1.69 ± 0.22 U/mL vs 1.68 ± 0.12 U/mL (P = 0.752); ET 0.34 ± 0.04 EU/mL vs 0.33 ± 0.04 EU/mL (P = 0.388); and NO 75.69 ± 8.35 μmol/L vs 76.89 ± 8.33 μmol/L (P = 0.304). These findings indicate that, apart from the lower D-Lac levels in the ESD group, postoperative recovery of gastrointestinal hormones and mucosal barrier integrity was comparable between the two groups.
| Parameters | Gastrectomy (n = 98) | ESD (n = 108) | t | P value |
| VIP (ng/L) | 103.55 ± 10.36 | 103.53 ± 12.96 | 0.014 | 0.989 |
| GAS (ng/L) | 16.23 ± 2.25 | 16.11 ± 2.63 | 0.325 | 0.746 |
| MTL (ng/L) | 258.93 ± 28.77 | 257.00 ± 25.93 | 0.616 | 0.539 |
| DAO (U/mL) | 1.69 ± 0.22 | 1.68 ± 0.12 | 0.317 | 0.752 |
| ET (EU/mL) | 0.34 ± 0.04 | 0.33 ± 0.04 | 0.866 | 0.388 |
| NO (μmol/L) | 75.69 ± 8.35 | 76.89 ± 8.33 | 1.031 | 0.304 |
| D-Lac (μg/mL) | 11.63 ± 1.36 | 11.18 ± 1.31 | 2.445 | 0.015 |
This study compared the efficacy and safety of conventional gastrectomy and ESD in the treatment of early gastrointes
Another key finding was the favorable safety profile of ESD. The notably lower rates of postoperative complications - particularly gastrointestinal bloating and perforation - underscore its advantage in patient safety[20]. This benefits likely arise from the endoscopic approach, which avoids open incisions and reduce exposure to potential infection sources[20]. Moreover, the superior visualization and precise control afforded by endoscopic instruments allow meticulous hemo
The reduced intraoperative blood loss observed during ESD compared with gastrectomy results from the endoscopic approach’s inherent capacity to minimize vascular injury. Precise vessel control through adrenaline-saline injections and titanium clips ensures effective hemostasis during ESD[20], thereby reducing reliance on extensive intraoperative blood conservation strategies typically required in conventional open surgery. This reduced hemorrhage mitigates acute bleeding complications and lessens the likelihood of blood transfusions and its associated risks. The shorter operative duration of ESD reflects its procedural efficiency. By eliminating the need for extensive abdominal exploration characteristic of open gastrectomy[22], ESD reduces anesthesia time and perioperative risk while improving patient throughput without compromising clinical outcomes[23]. Consequently, patients experience faster functional recovery and earlier discharge. Accelerated gastrointestinal recovery following ESD supports existing evidence that minimally invasive techniques enhance physiological rebound[24]. Reduced systemic inflammation and localized tissue trauma with ESD, compared with gastrectomy, are major contributions[25]. Because ESD confines surgical impact to the target lesion, gastrointestinal motility and absorption are better preserved, allowing earlier oral intake and improved postoperative nutrition[26]. Lower preoperative TK1 levels among ESD candidates may reflect selection of less advanced disease[27]. However, as a proliferation marker[28], decreased TK1 level following ESD highlights the procedure’s effectiveness in suppressing tumor metabolic activity.
Analysis of gastrointestinal hormones and barrier indices showed similar VIP, GAS, and MTL levels between groups, but significantly lower D-Lac level in ESD patients, indicating superior preservation of intestinal barrier integrity[29]. As D-Lac reflects intestinal permeability and bacterial translocation[30], its reduction implies that ESD maintains mucosal integrity more effectively than gastrectomy. The normalization of DAO and ET levels further supports ESD’s ability to minimize systemic postoperative alterations[31], potentially reducing complications such as sepsis linked to gut barrier dysfunction.
This study has several limitations that may affect interpretation and generalizability. The sample size may be inadequate to detect significant intergroup differences, limiting statistical robustness. Potential selection bias exists, as factors such as lesion size, location, and comorbidities may have influenced the choice of ESD over conventional gastrectomy, affecting perceived efficacy and safety. The absence of recurrence data limits conclusion regarding long-term oncologic outcomes, and the short follow-up period precludes evaluation of recurrence and survival. Operator variability may also influence outcomes, underscoring the need for standardized procedural protocols. Additionally, the lower preoperative TK1 and D-Lac levels in patients with ESD may indicate intrinsic differences in disease burden or mucosal condition between the groups. Larger, randomized trials with extended follow-up are warranted to validate these findings across clinical settings.
In summary, this study demonstrates that ESD provides superior efficacy and safety compared with conventional gastrectomy for treating early-stage gastrointestinal lesions. It significantly reduces intraoperative bleeding and operative time during the same hospitalization, facilitating earlier postoperative oral intake. The minimally invasive nature of ESD, combined with preservation of normal organ function and favorable safety outcomes, supports its role as the preferred treatment option for eligible patients. These advantages - rapid recovery, shorter hospitalization, lower costs, and quicker social reintegration - underscores ESD’s clinical and socioeconomic value and define a clear direction for future research.
| 1. | Dickinson KJ, Dunkin BJ, Nguyen-Lee JJ, Ali AB, Zajac S. Task Deconstruction of Colonic Endoscopic Submucosal Dissection (cESD): An Expert Consensus. World J Surg. 2020;44:2401-2408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 2. | Li P, Ma B, Gong S, Zhang X, Li W. Endoscopic submucosal tunnel dissection for superficial esophageal neoplastic lesions: a meta-analysis. Surg Endosc. 2020;34:1214-1223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 3. | Zhang YR, Sun C, Cheng CL, Gao J, Zhang J, Chen J, Wang LW, Chen Y, Man XH, Shi XG, An W. Endoscopic submucosal dissection for proximal duodenal subepithelial lesions: a retrospective cohort study. Surg Endosc. 2022;36:6601-6608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Kamal F, Khan MA, Lee-Smith W, Sharma S, Imam Z, Jowhar D, Petryna E, Esswein J, Acharya A, Aziz M, Farooq U, Zafar U, Mcdonough S, Adler DG. Outcomes of Endoscopic Submucosal Dissection for Treatment of Superficial Pharyngeal Cancers: Systematic Review and Meta-Analysis. Dig Dis Sci. 2022;67:3518-3528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 5. | Ge PS, Aihara H. Advanced Endoscopic Resection Techniques: Endoscopic Submucosal Dissection and Endoscopic Full-Thickness Resection. Dig Dis Sci. 2022;67:1521-1538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 6. | Nabi Z, Ramchandani M, Asif S, Basha J, Chavan R, Darisetty S, Reddy N. Outcomes of Endoscopic Submucosal Dissection in Duodenal Neuroendocrine Tumors. J Gastrointest Surg. 2022;26:275-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 7. | Meng ZW, Bishay K, Vaska M, Ruan Y, Al-Haddad MA, Elhanafi SE, Qumseya BJ, Belletrutti PJ, Gill R, Debru E, Heitman SJ, Brenner DR, Forbes N. Endoscopic Submucosal Dissection Versus Surgery or Endoscopic Mucosal Resection for Metachronous Early Gastric Cancer: a Meta-analysis. J Gastrointest Surg. 2023;27:2628-2639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (3)] |
| 8. | Kobayashi Y, Nishikawa K, Akasaka T, Kato S, Hamakawa T, Yamamoto K, Kobayashi N, Kitakaze M, Maeda S, Uemura M, Miyake M, Hama N, Miyamoto A, Kato T, Miyazaki M, Nakamori S, Mita E, Sekimoto M, Mano M, Hirao M. Retrograde endoscopic submucosal dissection for early thoracic esophageal carcinoma. Clin J Gastroenterol. 2021;14:434-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Esaki M, Ihara E, Sumida Y, Fujii H, Takahashi S, Haraguchi K, Iwasa T, Somada S, Minoda Y, Ogino H, Tagawa K, Ogawa Y. Hybrid and Conventional Endoscopic Submucosal Dissection for Early Gastric Neoplasms: A Multi-Center Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2023;21:1810-1818.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 10. | Stéphane S, Timothée W, Jérémie A, Raphael O, Martin D, Emmanuelle P, Elodie L, Quentin D, Nikki C, Sonia B, Hugo L, Guillaume G, Romain L, Mathieu P, Sophie G, Jeremie J. Endoscopic submucosal dissection or piecemeal endoscopic mucosal resection for large superficial colorectal lesions: A cost effectiveness study. Clin Res Hepatol Gastroenterol. 2022;46:101969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 11. | Doumbe-Mandengue P, Pellat A, Belle A, Ali EA, Hallit R, Beuvon F, Terris B, Chaussade S, Coriat R, Barret M. Endoscopic submucosal dissection versus endoscopic mucosal resection for early esophageal adenocarcinoma. Clin Res Hepatol Gastroenterol. 2023;47:102138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 12. | Uppal DS, Wang AY. Traction-assisted endoscopic submucosal dissection in the esophagus: Should we all be flossing? Gastrointest Endosc. 2020;91:66-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 13. | Zhang T, Zhang T, Li C, Zhai X, Huo Q. Complementary and alternative therapies for precancerous lesions of gastric cancer: A protocol for a Bayesian network meta analysis. Medicine (Baltimore). 2021;100:e24249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Chen S, Huang K, Zou L, Chen L, Hu P. Diagnostic value of SHOX2, RASSF1A gene methylation combined with CEA level detection in malignant pleural effusion. BMC Pulm Med. 2023;23:160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 15. | Bdeir R, Al-Keilani MS, Khamaiseh K. Effects of the Neuropeptides Pituitary Adenylate Cyclase Activating Polypeptide and Vasoactive Intestinal Peptide in Male Fertility. Medicina (Kaunas). 2024;60:652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 16. | Dominguez-Romero JG, Jiménez-Rejano JJ, Ridao-Fernández C, Chamorro-Moriana G. Exercise-Based Muscle Development Programmes and Their Effectiveness in the Functional Recovery of Rotator Cuff Tendinopathy: A Systematic Review. Diagnostics (Basel). 2021;11:529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 17. | Bhatt A, Mehta NA. Stricture prevention after esophageal endoscopic submucosal dissection. Gastrointest Endosc. 2020;92:1187-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (3)] |
| 18. | Shimada S, Yabuuchi Y, Kawata N, Maeda Y, Yoshida M, Yamamoto Y, Minamide T, Shigeta K, Takada K, Kishida Y, Ito S, Imai K, Hotta K, Ishiwatari H, Matsubayashi H, Ono H. Endoscopic causes and characteristics of missed gastric cancers after endoscopic submucosal dissection. Gastrointest Endosc. 2023;98:735-743.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 19. | Bourke MJ. Endoscopic submucosal dissection: Will it always be a hyperspecialized skill? Gastrointest Endosc. 2024;100:140-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 20. | Nieuwenhuis EA, Pech O, Bergman JJGHM, Pouw RE. Role of Endoscopic Mucosal Resection and Endoscopic Submucosal Dissection in the Management of Barrett's Related Neoplasia. Gastrointest Endosc Clin N Am. 2021;31:171-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Hayat M, Azeem N, Bilal M. Colon Polypectomy with Endoscopic Submucosal Dissection and Endoscopic Full-Thickness Resection. Gastrointest Endosc Clin N Am. 2022;32:277-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (4)] |
| 22. | Simsek C, Aihara H. Training in Endoscopic Submucosal Dissection in the United States: The Current Paradigm. Gastrointest Endosc Clin N Am. 2023;33:41-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 23. | Al Ghamdi SS, Ngamruengphong S. Endoscopic Submucosal Dissection in the Stomach and Duodenum: Techniques, Indications, and Outcomes. Gastrointest Endosc Clin N Am. 2023;33:67-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 24. | Chiu PW, Zhou S, Dong Z. A Look into the Future of Endoscopic Submucosal Dissection and Third Space Endoscopy: The Role for Robotics and Other Innovation. Gastrointest Endosc Clin N Am. 2023;33:197-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 25. | Landin MD, Guerrón AD. Endoscopic Mucosal Resection and Endoscopic Submucosal Dissection. Surg Clin North Am. 2020;100:1069-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 26. | Draganov PV, Aihara H, Karasik MS, Ngamruengphong S, Aadam AA, Othman MO, Sharma N, Grimm IS, Rostom A, Elmunzer BJ, Jawaid SA, Westerveld D, Perbtani YB, Hoffman BJ, Schlachterman A, Siegel A, Coman RM, Wang AY, Yang D. Endoscopic Submucosal Dissection in North America: A Large Prospective Multicenter Study. Gastroenterology. 2021;160:2317-2327.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 151] [Article Influence: 30.2] [Reference Citation Analysis (1)] |
| 27. | Probst A. Colonic endoscopic submucosal dissection - increasing attraction with improved traction? Endoscopy. 2020;52:336-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 28. | Wang FG, Jiang Y, Liu C, Qi H. Comparison between Endoscopic Submucosal Dissection and Transanal Endoscopic Microsurgery in Early Rectal Neuroendocrine Tumor Patients: A Meta-Analysis. J Invest Surg. 2023;36:2278191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Esaki M, Ihara E, Gotoda T. Endoscopic instruments and techniques in endoscopic submucosal dissection for early gastric cancer. Expert Rev Gastroenterol Hepatol. 2021;15:1009-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 30. | Qiu J, Ouyang Q, Zhang Y, Xu J, Xie Y, Wei W, Pan X. Post-endoscopic submucosal dissection electrocoagulation syndrome: a clinical overview. Expert Rev Gastroenterol Hepatol. 2022;16:1079-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 31. | Chou CK, Tsai KF, Tseng CH, Lee CT, Yang KH, Chang MC, Hsu CW. Novel Colorectal Endoscopic Submucosal Dissection With Double-Endoscope and Snare-Based Traction. Dis Colon Rectum. 2022;65:936-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/