Published online Mar 15, 2026. doi: 10.4239/wjd.v17.i3.116660
Revised: January 10, 2026
Accepted: February 5, 2026
Published online: March 15, 2026
Processing time: 115 Days and 13.4 Hours
Nesidioblastosis is a rare condition involving abnormal proliferation of pancreatic β-cell, leading to excessive insulin production and severe hypoglycemia. In con
A 23-year-old female presented with a protracted 13-year history of recurrent hypoglycemia refractory to medical management (dextrose and diazoxide). Labo
This case highlights the diagnostic challenge associated with nesidioblastosis especially in resource-limited settings to enable early intervention.
Core Tip: Adult-onset nesidioblastosis is an uncommon but important cause of endogenous hyperinsulinism that can mimic insulinoma and lead to years of misdiagnosis. This case illustrates how persistent hypoglycemia despite standard medical therapy should prompt consideration of rare etiologies and the use of advanced imaging, such as 68Ga-DOTATOC positron emission tomography/computed tomography, when conventional studies are non-localizing. Early recognition improves the likelihood of timely surgical management and prevents prolonged morbidity.
- Citation: Bavaria S, Chana A, Nurani KM, Kimang’a J. Recurrent hypoglycemia in a young female with nesidioblastosis: A case report and review of literature. World J Diabetes 2026; 17(3): 116660
- URL: https://www.wjgnet.com/1948-9358/full/v17/i3/116660.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i3.116660
Hypoglycemia, which is commonly defined by a plasma glucose concentration of below 3.9 mmol/L, is a clinically significant condition that warrants evaluation for an underlying cause[1]. Documentation of Whipple’s triad (blood glucose < 3.1 mmol/L, hypoglycemic symptoms, and resolution of symptoms with glucose intake) serves as a key indicator of true hypoglycemia, and is essential for the initial diagnostic workup. Hypoglycemia is most often seen in patients who have diabetes and are on management with meglitinides, sulfonylureas, or insulin, with patients who have type 1 diabetes being 3 times more likely to experience hypoglycemia than patients with type 2 diabetes[1,2].
In non-diabetic individuals, hypoglycemia is uncommon and can result from a wide range of etiologies, including medications, critical illness, alcohol use, counter-regulatory hormone deficiencies, functional/reactive hypoglycemia, or disorders causing endogenous hyperinsulinism, such as insulinoma or non–islet cell tumors producing insulin like growth factor[2]. Among adults, the most frequent cause of endogenous hyperinsulinemic hypoglycemia is an in
Nesidioblastosis, first described in neonates and infants, refers to diffuse proliferation or hyperplasia of pancreatic β-cells, often accompanied by nesidioblast-like structures reminiscent of fetal pancreatic tissue[5]. In adults, the condition is exceedingly rare, with only a few cases documented in medical literature and is reported to represent only about 0.5%-5% of cases of organic hyperinsulinemia[5].
A 23-year-old female, a non-diabetic nursing student, presented with a protracted history of recurrent, symptomatic hypoglycemic episodes, characterized by palpitations, sweating, chills, dizziness, blurred vision, generalized body weakness, and ultimately, loss of consciousness.
The initial hypoglycemic episode presented when she was ten years old weighing 15 kg. She was managed at Makueni County Referral Hospital with intravenous dextrose, which resolved her symptoms, and discharged with dietary advice. Notably, there was no history of diabetes or hypoglycemic medication use.
From 2012 to 2017, she attended the Makueni County Hospital outpatient clinic three times per week for follow-up. In 2017, oral dextrose was prescribed for home management, decreasing clinic visits, yet frequent hypoglycemic episodes persisted. In 2022, she presented to Makueni County Hospital with severe hypoglycemia leading to loss of consciousness. Persistent low blood glucose despite repeated intravenous dextrose prompted referral to Kenyatta National Hospital (KNH) for further evaluation, including fasting insulin and C-peptide assays, which were unavailable locally. At KNH, a two-week admission involved management with intravenous dextrose, diazoxide (60 mg twice daily), and hydrocor
Two weeks post-discharge, she returned to KNH outpatient clinic with severe bilateral lower limb edema and persistent hypoglycemia despite adherence to medication. She was readmitted for one month. Dextrose, diazoxide, and hydrocortisone were continued for two weeks but subsequently discontinued due to worsening edema, fatigue, and nausea. She remained on intravenous dextrose for an additional two weeks before discharge, with instructions for home blood glucose monitoring and monthly diabetic clinic follow-up. A 68Ga-DOTATOC positron emission tomography (PET)/CT scan was recommended in February 2023 for further evaluation but was deferred due to financial constraints. Throughout 2023 and 2024, she maintained clinic visits, utilizing oral and intravenous dextrose as required.
In the current admission in June 2025, she was readmitted to KNH due to escalating hypoglycemia, experiencing up to six daily episodes, including nocturnal events causing significant anxiety. During this hospitalization, frequent hypoglycemic events persisted, with documented instances of blood glucose levels rapidly declining from 10 mmol/L within minutes, leading to loss of consciousness. Repeat fasting insulin and C-peptide levels were both elevated, and the provisional diagnosis of insulinoma was maintained. While intravenous dextrose was maintained for glycemic management, the administration of octreotide and the newer drug, pasireotide, was indicated but deferred due to financial constraints. Her weight at this time was 43 kg. Additionally, in 2025, uncooked cornstarch was introduced to her diet for 3 months. However, intermittent hypoglycemic episodes persisted.
The patient reported a history of early childhood malnutrition, with the exact age unrecallable. Additionally, concomitant medical issues included anal fissures with hematochezia, severe anemia (hemoglobin 5.3 g/dL) requiring blood transfusion and managed with lactulose and anusol, and urinary incontinence treated with oxybutynin. She also reported dysmenorrhea and prolonged menstrual periods since menarche.
The patient has had no prior surgical procedures, no known food or drug allergies, and denies alcohol, or illicit drug use. There is no family history of diabetes mellitus, hypertension, or other chronic conditions.
She is a nursing student at Kenya medical training college Makueni. She is the first born in a family of 3 children. There is no family history of diabetes mellitus, similar illnesses, or other chronic conditions. Alcohol, tobacco and illicit drug use were denied.
Physical examination revealed a patient in good general and nutritional status. Vital signs: Blood pressure 128/70 mmHg, Pulse rate 86 per minute, SpO2 96% while on ambient air. No jaundice, pallor, cyanosis, clubbing, lymphadenopathy, or dehydration; capillary refill was normal. Abdominal examination: Normal fullness, movement with respiration, inverted umbilicus, no scars or distended veins on inspection. Palpation revealed mild suprapubic tenderness, no palpable masses, hepatomegaly, or splenomegaly. Auscultation showed normal bowel sounds. Central nervous system examination: Glasgow coma scale 15/15, no cranial nerve abnormalities, and no motor or sensory deficits. Respiratory and cardiovascular examinations were unremarkable.
During episodes of hypoglycemia, her blood glucose ranges from 1.6 mmol/L to 2.9 mmol/L requiring frequent resuscitation with intravenous dextrose. Her fasting C peptide level was 1.68 nmol/L (normal range 0.26-1.27 nmol/L) and her fasting insulin was 30.6 µIU/mL (normal range 2.6-24.9 µIU/mL) which are both elevated. Her full hemogram, liver function tests, urea, electrolytes and creatinine were all in the normal range. The thyroid function tests were unremarkable. Her morning cortisol level was 497.1 nmol/L (reference range is 133-507 nmol/L). Tumor markers (alpha-fetoprotein, carbohydrate antigen 19-9, carcinoembryonic antigen, cancer antigen 15-3, cancer antigen 125) were absent.
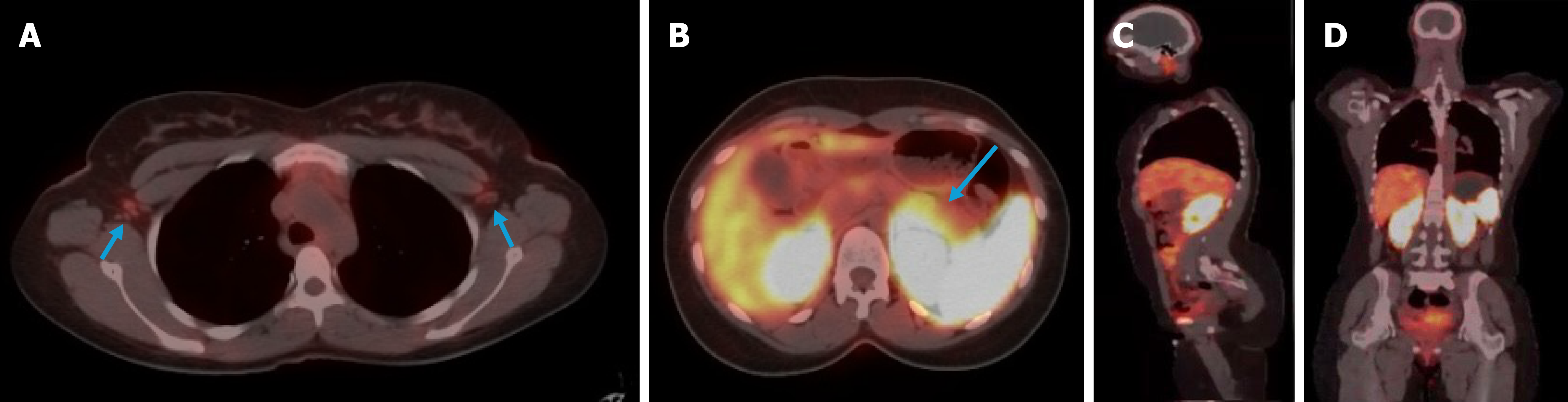
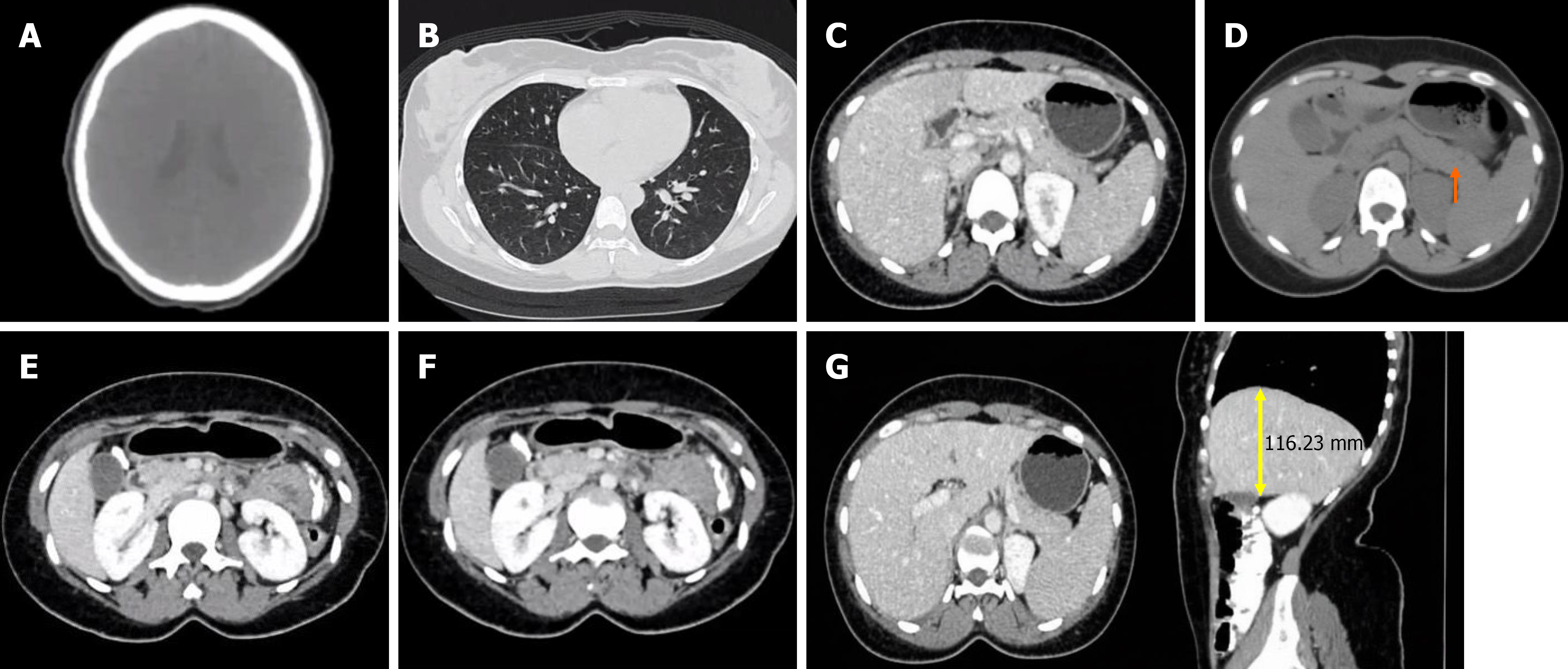
Following multiple delays due to cost and scanner unavailability, a 68Ga-DOTATOC PET/CT scan was performed at a private facility in August, 2025. 68Ga-DOTATOC PET/CT (August 29, 2025) showed diffuse, low-grade somatostatin-receptor expression throughout the distal body and tail of the pancreas, a pattern typical of β-cell hyperplasia rather than a discrete insulinoma (Figure 1). A month later, contrast-enhanced CT (October 1, 2025) confirmed relative enlargement of the body/tail compared with the head/neck. The remainder of the abdomen, including the gall-bladder, kidneys, stomach, bowel, and liver, appeared unremarkable (Figure 2). Following the PET/CT that confirmed a diagnosis of nesidioblastosis, the decision was made to forgo selective arterial calcium stimulation test to minimize the financial burden on the patient and avoid further invasive investigation.
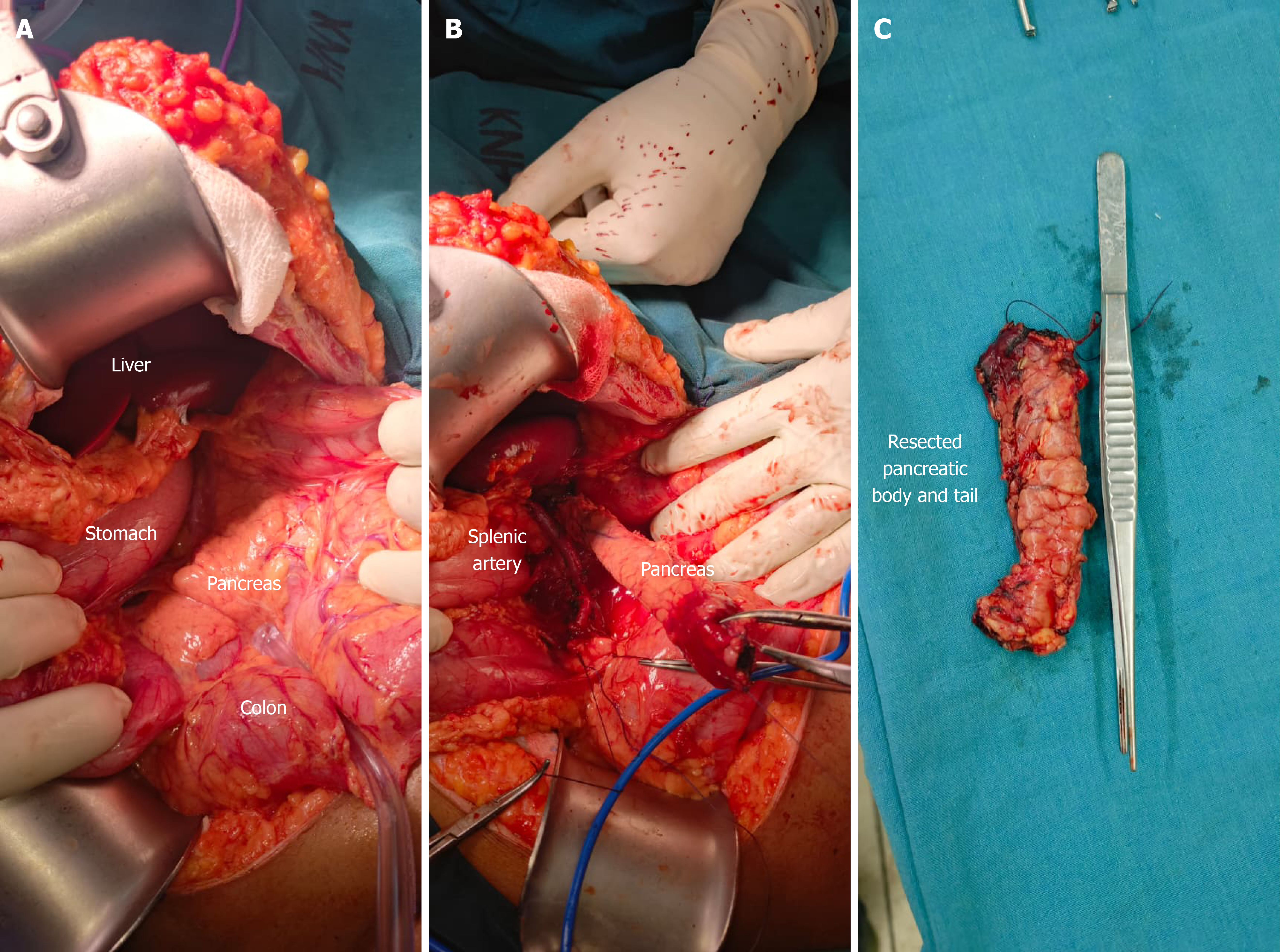
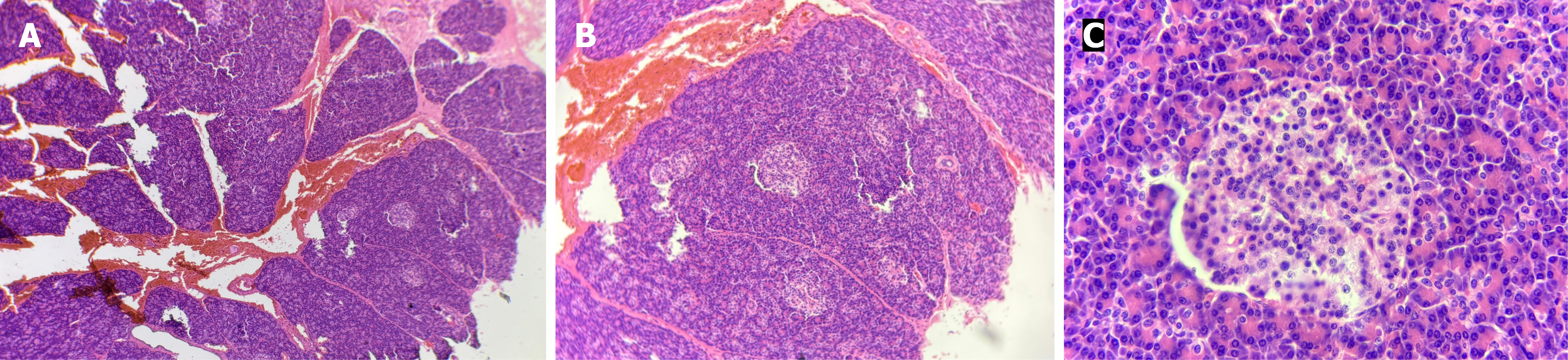
As medical management did not yield improvement, a distal pancreatectomy was scheduled. During the surgery, the abdomen, including the pancreas, was explored and found to be grossly normal with no masses. A point of transection was chosen distal to the superior mesenteric vein. The pancreas was gently elevated from the retroperitoneum, and was transected at the neck. The distal (body and tail of pancreas) was elevated and multiple small tributaries linking the splenic artery and vein to the pancreas were clipped and divided until the pancreas was free. Partial pancreatic resection was then performed and tissue sent for histopathology. Surgical and histopathology images are shown (Figures 3 and 4).
Based on the above clinical and histopathological findings, a diagnosis of nesidioblastosis was made.
The patient received both medical and surgical care. Medical management consisted of dextrose, diazoxide, hydrocortisone and uncooked cornstarch. However, as medical therapy was ineffective, the patient underwent a distal pancreatectomy.
Following surgery, the patient received intravenous dextrose for four days, during which her blood glucose levels remained within the normal range. Fasting blood glucose on postoperative days 2, 3, and 4 were 7.8 mmol/L, 7.8
In 1938, Laidlaw[6] introduced the term “nesidioblastosis” to describe the diffuse formation of new islets of Langerhans within the pancreas, representing a functional β-cell disorder. Nesidioblastosis, which is part of the NIPHS, is an uncommon phenomenon in the adult population, accounting for 0.5%-5% of all hyperinsulinemic hypoglycemia cases[5]. Table 1 summarizes published adult-onset case reports from the past five years[7-15].
| Ref. | Country | Case summary |
| Izumo et al[7], 2025 | Japan | A 37-year-old woman presented with Whipple’s triad and fasting hyperinsulinemic hypoglycemia. Imaging negative for insulinoma. SACI test localized hypersecretion to gastroduodenal artery. Underwent pancreatoduodenectomy, with histology confirming nesidioblastosis. Recurrence at 1 year required remnant pancreatectomy, resulting in long-term symptom resolution |
| Chen et al[8], 2022 | United States | An 83-year-old man presented with biochemical derangements suggestive of insulinoma. Preoperative cross-sectional imaging, Dotatate PET, and visceral angiogram all failed to definitively localize a neuroendocrine tumor. SACI showed a subtle increase in insulin secretion from the tail of the pancreas. Distal showing diffused islet hyperplasia consistent with a diagnosis of nesidioblastosis. Postoperative euglycaemia was achieved |
| Kim et al[9], 2022 | United States | Patient presented with worsening hypoglycemic symptoms for 1 year prior to presentation that eventually progressed to hypoglycemic seizures. The onset of this hypoglycemia was 5 years after Roux-en-Y gastric bypass surgery. Extensive neurological and metabolic evaluation excluded other causes. Subtotal pancreatectomy confirmed nesidioblastosis; postoperative management included acarbose, a competitive reversible inhibitor of pancreatic α-amylase and intestinal brush border α-glucosidases which slows carbohydrate absorption |
| Tu et al[10], 2023 | China | A 48-year-old male who suffered from repeated morning and fasting palpitations, sweating, and severe disturbance of consciousness for 5 years. His blood glucose was found to be as low as 1.79 mmol/L during an attack. However, abdominal computed tomography showed no abnormalities. Magnetic resonance imaging and endoscopic ultrasonography demonstrated a nodular mass in the head of the pancreas, combined with hyperinsulinemia and high serum C-peptide. The patient was diagnosed with insulinoma and underwent Beger surgery; however, the postoperative pathological results showed nesidioblastosis |
| Dieterle et al[11], 2023 | Germany | A 23-year-old man with long-standing presyncope and exercise-induced hypoglycemia. Fasting test showed endogenous hyperinsulinism. 68Ga-DOTA-Exendin-4 PET/CT demonstrated diffuse pancreatic uptake. Required subtotal then total pancreatectomy due to recurrence. A subtotal pancreatectomy was performed, and the diagnosis of diffuse, adult-onset nesidioblastosis was established histopathologically. After nine months, the symptoms recurred, making complete pancreatectomy necessary. Postoperative laboratory evaluation exhibited no residual endogenous C-peptide production |
| Lisboa et al[12], 2025 | Brazil | A 47-year-old male patient, diagnosed with Nesidioblastosis, in 2014, after frequent loss of consciousness and without association with fasting, was submitted to Frey's technique, after failure with other procedures. The method adopted proved to be effective for correcting the patient's endocrine problem, as well as without sequelae to the gastrointestinal tract |
| Said et al[13], 2025 | Egypt | A PET/CT male patient presented with recurrent attacks of hypoglycemia, neuroglycopenic symptoms, and weight loss, prompting evaluation for causes of hyperinsulinemic hypoglycemia. Imaging (Gallium Dotatate PET/CT, EUS) identified a pancreatic tail lesion, prompting distal pancreatectomy. Histopathology examination confirmed nesidioblastosis. Post-surgery, transient diabetes developed but hypoglycemia recurred at 4 months. Octreotide treatment failed and diazoxid treatment successfully resolved symptoms |
| Abdul-Hafez et al[14], 2025 | Palestine | A 55-year-old woman with a history of gastric sleeve and Roux-en-Y gastric bypass surgeries presented with a 15-year history of recurrent hypoglycemic episodes. Her symptoms persisted despite medical therapy with octreotide, acarbose, and nifedipine. Imaging ruled out insulinomas, raising suspicion of non-insulinoma pancreatogenous hypoglycemia syndrome. The patient underwent laparoscopic subtotal distal pancreatectomy. Histopathological examination confirmed nesidioblastosis, revealing irregular islet distribution and β-cell hypertrophy. Post-surgery, the patient achieved euglycemia without recurrence of hypoglycemic episodes during follow-up |
| Valentim et al[15], 2025 | Brazil | A 42-year-old non-diabetic man with a history of coronary artery disease and systemic arterial hypertension had persistent hypoglycemia associated with high insulin levels. Total pancreatectomy and splenectomy performed after multidisciplinary decision-making; histopathology confirmed nesidioblastosis |
Neonatal nesidioblastosis is most commonly caused by genetic mutations affecting β-cell ion channels, most commonly the ATP-sensitive potassium channels. Mutations in the ABCC8 or KCNJ11 genes impair proper β-cell membrane repolarization, leading to persistent depolarization and continuous insulin release, which manifests as severe neonatal hypoglycemia[16]. In contrast, adult-onset nesidioblastosis has an unclear etiology[7]. Proposed mechanisms include abnormal ductal–endocrine signaling, which alters communication between pancreatic ducts and islets, somatostatin deficiency leading to reduced inhibitory control of insulin secretion, and hormonal or autoimmune stimuli, such as gastrin excess or islet-stimulating immunoglobulins[17]. In some cases, particularly following gastric bypass surgery, it is proposed that an exaggerated GLP-1–mediated incretin response may overstimulate β-cells.
NIPHS has also been reported in conjunction with other diseases, such as Zollinger-Ellison syndrome, multiple endocrine neoplasia, β-cell adenomatosis, Von Hippel-Lindau disease, cystic fibrosis, and post-pancreatic transplant[5], suggesting a complex interplay of pathogenetic mechanisms. The most prevalent pathologic finding is the diffuse proliferation of islet cells budding from pancreatic ducts, forming ductulo-insular complexes[18]. In 2010, Bränström et al[19] suggested that β-cell hyperplasia and aberrant ATP-sensitive potassium channel (KATP) activity contribute to the inappropriate insulin secretion[19].
Histopathologically, nesidioblastosis can be classified into focal and diffuse forms[20]. Focal nesidioblastosis is characterized by localized β-cell hyperplasia confined to a discrete region of the pancreas, whereas diffuse nesidioblastosis involves widespread β-cell hyperplasia affecting most or all of the pancreatic parenchyma. While focal disease is more commonly described in neonates and infants, it is increasingly recognized in adults, whereas diffuse nesidioblastosis remains the predominant pattern reported in adult-onset disease[21]. This distinction is clinically important, as focal disease may be amenable to limited pancreatic resection, whereas diffuse disease often requires more extensive surgical or long-term medical management[22].
Hyperinsulinemic hypoglycemia presents with both adrenergic symptoms (e.g., tachycardia, tremors, sweating) and neuroglycopenic symptoms (e.g., altered consciousness, seizures, tunnel vision)[7,11,23,24]. A survey conducted at Mayo Clinic involving ten patients with NIPHS, aged 16-78 years, indicated that hypoglycemic symptoms characteristically manifest 2-4 hours after meals but are not observed after intermittent fasting[23,25,26]. The extensive differential diagnosis of hyperinsulinemic hypoglycemia often results in diagnostic delays, which significantly impair patients’ quality of life.
Key differential diagnoses thus include insulinoma, glucose dysregulation post-Roux-en-Y gastric bypass surgery, and dumping syndrome[23,24,27-30]. Table 2 shows the main differences between these conditions. In 2023, Dieterle et al[11] delineated a succinct diagnostic framework for patients presenting with hyperinsulinemic hypoglycemia, encompassing: (1) Confirming hypoglycemia; establishing the Whipple triad; excluding organ failure, sepsis, non-pancreatic tumors, factitious hypoglycemia, and hormone deficiencies; and (2) Performing an oral glucose tolerance test to differentiate between reactive hypoglycemia and endogenous hyperinsulinemic hypoglycemia[11].
| Feature | Nesidioblastosis | Insulinoma | Dumping syndrome |
| Pathophysiology | Diffuse or focal β-cell hyperplasia with inappropriate insulin secretion | Insulin-secreting pancreatic neuroendocrine tumor | Rapid gastric emptying causing exaggerated insulin response |
| Typical age | Neonates | Adults | Any age after gastric or bariatric surgery |
| Common risk factors | Post-bariatric surgery, idiopathic | MEN1, sporadic | Gastric surgery (RYGB, sleeve gastrectomy, gastrectomy) |
| Timing of hypoglycemia | Fasting and/or postprandial | Predominantly fasting | Postprandial (1-3 hours after meals) |
| Relationship to meals | Variable | Often relieved by eating | Occurs after meals, especially high-carbohydrate meals |
| Insulin during hypoglycemia | Inappropriately elevated | Inappropriately elevated | Elevated postprandially only |
| C-peptide | Elevated | Elevated | Normal to mildly elevated |
| Selective arterial calcium stimulation test | Diffuse or regional insulin response | Focal insulin step-up | Negative |
| Histopathology | Diffuse or focal islet hyperplasia, β-cell hypertrophy | Well-circumscribed neuroendocrine tumor | Normal pancreatic histology |
| Treatment | Dietary modification, diazoxide, octreotide. Subtotal or total pancreatectomy in refractory cases | Surgical enucleation or resection | Dietary modification |
| Risk of recurrence | High (especially after partial resection) | Low after complete excision | Symptoms may persist but improve with diet |
| Postoperative diabetes risk | High | Low to moderate | None |
In patients with NIPHS, imaging studies are often negative since the macroscopic appearance of the pancreas is usually normal[17]. Selective arterial calcium stimulation with hepatic venous sampling can help identify hyperinsulinemic regions[31]. Specialized imaging techniques such as DOTANOC PET/CT may demonstrate somatostatin receptor expression within the pancreas, consistent with β-cell hyperplasia[5,17,18]. Nonetheless, histopathology remains the gold standard, with major criteria including absence of insulinoma, β-cells with enlarged hyperchromatic nuclei and clear cytoplasm, normal islet architecture, and Ki-67 negativity; minor criteria include islet enlargement, lobulation, and macronucleoli[32]. It is however important to note that up to a third of patients diagnosed with nesidioblastosis do not meet these criteria or exhibit only minimal histologic changes[33].
Management of adult-onset nesidioblastosis remains challenging due to the absence of standardized treatment guidelines. Initial therapy focuses on acute correction of hypoglycemia with intravenous glucose, followed by medical suppression of insulin secretion using agents such as diazoxide, which binds to the SUR1 domain and opens K+ ATP channel to reduces insulin release by pancreatic β cells, and somatostatin analogs, including octreotide or pasireotide[20]. Additional therapies, such as verapamil, which blocks calcium-dependent release of insulin; corticosteroids, which combat hypoglycemia by decreasing peripheral utilization of glucose and increase gluconeogenesis; and acarbose which slows carbohydrate digestion, reducing rapid glucose absorption and preventing post-meal blood sugar drops[34]. Dietary interventions, such as frequent small meals or the use of uncooked cornstarch, have also been utilized to maintain glycemic stability[34].
Surgical intervention is indicated in patients with refractory hypoglycemia despite optimal medical therapy. Partial pancreatectomy (resection of 60%-80%) controls nesidioblastosis in 50% of patients without additional medication, especially in focal forms[35]. However, recurrence of hypoglycemia is common, and many patients may require subtotal or total pancreatectomy[35]. While total pancreatectomy may achieve definitive glycemic control, it is associated with significant comorbidities including insulin-dependent diabetes and exocrine insufficiency[15]. Medical treatment is thus preferred especially in cases of diffuse nesidioblastosis. Diazoxide remains the most frequently used medical treatment combined with dietetic measures[35]. Long-term outcomes remain poorly characterized, highlighting the need for individualized management strategies and additional studies to guide optimal treatment.
Our case illustrates several features that underscore the diagnostic and therapeutic complexity of endogenous hyperinsulinemic hypoglycemia, including an unusually early onset of symptoms, prolonged diagnostic delay, and limited access to advanced diagnostic modalities in a resource-constrained setting. Our patient presented with a history of recurrent hypoglycemia, beginning at 10 years of age and persisted for over a decade prior to diagnosis, thereby illustrating both the chronic nature and diagnostic complexity of this condition. Initially, biochemical analysis revealed elevated insulin and C-peptide levels, leading to a provisional diagnosis of insulinoma following the exclusion of exogenous insulin or sulfonylurea administration. Nevertheless, the persistence of symptoms despite therapeutic interventions with diazoxide and corticosteroids, coupled with the absence of any discernible focal pancreatic lesion across multiple imaging modalities (ultrasound, CT, and MRI), suggested an alternative pathology. A DOTANOC PET/CT scan subsequently demonstrated diffuse low-grade somatostatin receptor expression within the distal pancreatic body and tail, consistent with β-cell hyperplasia (nesidioblastosis) rather than a discrete insulin-secreting tumor[36]. While selective arterial calcium stimulation testing with hepatic venous sampling is often used to distinguish focal from diffuse disease, it is invasive, and not universally available, especially in low income settings. Recent reports describe PET-based imaging as a new noninvasive diagnostic tool for the localization of an endogenous source of hyperinsulinemic hypoglycemia[20].
To the best of our knowledge, this is the first documented case of adult-onset nesidioblastosis reported in Kenya. The case highlights the challenges faced in low- and middle-income countries, including limited access to specialized biochemical assays and advanced imaging modalities such as the local unavailability of advanced imaging like the 68Ga-DOTATOC PET/CT, which delayed confirmation for several years. Furthermore, the case demonstrates the overlap in clinical and biochemical findings between insulinoma and nesidioblastosis, underscoring the need for high clinical suspicion for persistent hyperinsulinemic hypoglycemia when imaging fails to localize a lesion.
In conclusion, there is need for heightened clinical suspicion of nesidioblastosis in patients with refractory hypoglycemia and non-localizing imaging. In addition, further studies are required to establish standardized diagnostic and therapeutic algorithms for this rare but debilitating condition.
| 1. | Mathew P, Thoppil D. Hypoglycemia. 2022 Dec 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 2. | Nakhleh A, Shehadeh N. Hypoglycemia in diabetes: An update on pathophysiology, treatment, and prevention. World J Diabetes. 2021;12:2036-2049. [PubMed] [DOI] [Full Text] |
| 3. | Yamusah N. Elusive early diagnosis of insulinomas. J Clin Transl Endocrinol: Case Rep. 2021;21:100087. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 4. | Dieterle MP, Husari A, Prozmann SN, Wiethoff H, Stenzinger A, Röhrich M, Pfeiffer U, Kießling WR, Engel H, Sourij H, Steinberg T, Tomakidi P, Kopf S, Szendroedi J. Diffuse, Adult-Onset Nesidioblastosis/Non-Insulinoma Pancreatogenous Hypoglycemia Syndrome (NIPHS): Review of the Literature of a Rare Cause of Hyperinsulinemic Hypoglycemia. Biomedicines. 2023;11:1732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 5. | Hong R, Choi DY, Lim SC. Hyperinsulinemic hypoglycemia due to diffuse nesidioblastosis in adults: a case report. World J Gastroenterol. 2008;14:140-142. [PubMed] [DOI] [Full Text] |
| 6. | Laidlaw GF. Nesidioblastoma, the islet tumor of the pancreas. Am J Pathol. 1938;14:125-134.5. [PubMed] |
| 7. | Izumo W, Higuchi R, Shiihara M, Uemura S, Yazawa T, Takano N, Ichihara A, Furukawa T, Nagashima Y, Yamamoto M, Honda G. Clinically Adult-Onset Nesidioblastosis with Repeated Severe Hypoglycemia, Successfully Treated by Two Times Pancreatectomies. A Rare Case Report. Clin Exp Gastroenterol. 2025;18:163-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 8. | Chen T, Rosen R, Grace W, Alden D. Case Report: A Case of Adult Nesidioblastosis. Hepato Pancreato Biliary. 2022;24:S328. [DOI] [Full Text] |
| 9. | Kim K, Greenspan JL, Mehrara S, Wynne D, Ennis E. Nesidioblastosis: an uncommon complication seen post Roux-en-Y gastric bypass. Endocrinol Diabetes Metab Case Rep. 2022;2022:22-0361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 10. | Tu K, Zhao LJ, Gu J. Adult focal β-cell nesidioblastosis: A case report. World J Clin Cases. 2023;11:150-156. [PubMed] [DOI] [Full Text] |
| 11. | Dieterle MP, Husari A, Prozmann SN, Wiethoff H, Stenzinger A, Röhrich M, Pfeiffer U, Kießling WR, Engel H, Sourij H, Steinberg T, Tomakidi P, Kopf S, Szendroedi J. An Uncommon Cause of Recurrent Presyncope, Dizziness, and Tachycardia: A Case Report of Diffuse, Adult-Onset Nesidioblastosis/Non-Insulinoma Pancreatogenous Hypoglycemia Syndrome (NIPHS). Biomedicines. 2023;11:1741. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 12. | Lisboa FF, Ferreira LFN, Dantas GDC, da Silva PAN. Case Report: Nesidioblastosis in Adult Patient. J Chronic Dis Prev Care. 2025;2:1-5. |
| 13. | Said M, Al sehemy L, al Baz T, el razky M, Omran S, Rashwan M, Hassan OA, Kassem AB, Hesham A, Sami MA, Mohamed DH, Hassaballah M. Adult-Onset Nesidioblastosis: A Challenge In Diagnosis and Management in Resource Limited Countries: A Case of Recurrent Hypoglycemia after Partial Pancreatectomy for Nesidioblastosis. J Case Rep Stud. 2025;13:104. |
| 14. | Abdul-Hafez HA, Rabi K, Sarama A, Melhem L, Abed W, Maree M. Nesidioblastosis post-bariatric surgery in an adult patient: a case report and review of literature. Ann Med Surg (Lond). 2025;87:3006-3011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 15. | Valentim CAS, Arimura VS, Pompei IM, Bonvechio D, Silva MS, Turrini FJ, Jorgino MSDM, Valentim AS. Diagnostic and Surgical Management of Nesidioblastosis in a 42-Year-Old Man with Refractory Hypoglycemia. Am J Case Rep. 2025;26:e945453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 16. | De Franco E, Saint-Martin C, Brusgaard K, Knight Johnson AE, Aguilar-Bryan L, Bowman P, Arnoux JB, Larsen AR, Sanyoura M, Greeley SAW, Calzada-León R, Harman B, Houghton JAL, Nishimura-Meguro E, Laver TW, Ellard S, Del Gaudio D, Christesen HT, Bellanné-Chantelot C, Flanagan SE. Update of variants identified in the pancreatic β-cell K(ATP) channel genes KCNJ11 and ABCC8 in individuals with congenital hyperinsulinism and diabetes. Hum Mutat. 2020;41:884-905. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 154] [Cited by in RCA: 125] [Article Influence: 20.8] [Reference Citation Analysis (1)] |
| 17. | Rinker RD, Friday K, Aydin F, Jaffe BM, Lambiase L. Adult nesidioblastosis: a case report and review of the literature. Dig Dis Sci. 1998;43:1784-1790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 18. | Toyomasu Y, Fukuchi M, Yoshida T, Tajima K, Osawa H, Motegi M, Iijima T, Nagashima K, Ishizaki M, Mochiki E, Kuwano H. Treatment of hyperinsulinemic hypoglycemia due to diffuse nesidioblastosis in adults: a case report. Am Surg. 2009;75:331-334. [PubMed] |
| 19. | Bränström R, Berglund E, Curman P, Forsberg L, Höög A, Grimelius L, Berggren PO, Mattsson P, Hellman P, Juntti-Berggren L. Electrical short-circuit in β-cells from a patient with non-insulinoma pancreatogenous hypoglycemic syndrome (NIPHS): a case report. J Med Case Rep. 2010;4:315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 20. | Demartin S, Goffette P, Christ E, Freitag MT, Maiter D, Maria Furnica R. Adult-onset nesidioblastosis: a challenging diagnosis revealed by glucagon-like-peptide-1 receptor imaging. Endocrinol Diabetes Metab Case Rep. 2022;2022:22-0325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 21. | Doi S, Yamada T, Kito Y, Obara S, Fujii Y, Nishimura T, Kato T, Nakayama H, Tsutsumi M, Okamura R. Adult-Onset Focal Nesidioblastosis With Nodular Formation Mimicking Insulinoma. J Endocr Soc. 2022;6:bvab185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 22. | Witteles RM, Straus II FH, Sugg SL, Koka MR, Costa EA, Kaplan EL. Adult-onset nesidioblastosis causing hypoglycemia: an important clinical entity and continuing treatment dilemma. Arch Surg. 2001;136:656-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 70] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 23. | Service FJ, Natt N, Thompson GB, Grant CS, van Heerden JA, Andrews JC, Lorenz E, Terzic A, Lloyd RV. Noninsulinoma pancreatogenous hypoglycemia: a novel syndrome of hyperinsulinemic hypoglycemia in adults independent of mutations in Kir6.2 and SUR1 genes. J Clin Endocrinol Metab. 1999;84:1582-1589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 50] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 24. | Won JG, Tseng HS, Yang AH, Tang KT, Jap TS, Lee CH, Lin HD, Burcus N, Pittenger G, Vinik A. Clinical features and morphological characterization of 10 patients with noninsulinoma pancreatogenous hypoglycaemia syndrome (NIPHS). Clin Endocrinol (Oxf). 2006;65:566-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 55] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 25. | Thompson GB, Service FJ, Andrews JC, Lloyd RV, Natt N, van Heerden JA, Grant CS. Noninsulinoma pancreatogenous hypoglycemia syndrome: an update in 10 surgically treated patients. Surgery. 2000;128:937-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 70] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 26. | Rumilla KM, Erickson LA, Service FJ, Vella A, Thompson GB, Grant CS, Lloyd RV. Hyperinsulinemic hypoglycemia with nesidioblastosis: histologic features and growth factor expression. Mod Pathol. 2009;22:239-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 54] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 27. | Albers N, Löhr M, Bogner U, Loy V, Klöppel G. Nesidioblastosis of the pancreas in an adult with persistent hyperinsulinemic hypoglycemia. Am J Clin Pathol. 1989;91:336-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 28. | Walmsley D, Matheson NA, Ewen S, Himsworth RL, Bevan JS. Nesidioblastosis in an elderly patient. Diabet Med. 1995;12:542-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 29. | Patti ME, McMahon G, Mun EC, Bitton A, Holst JJ, Goldsmith J, Hanto DW, Callery M, Arky R, Nose V, Bonner-Weir S, Goldfine AB. Severe hypoglycaemia post-gastric bypass requiring partial pancreatectomy: evidence for inappropriate insulin secretion and pancreatic islet hyperplasia. Diabetologia. 2005;48:2236-2240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 262] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 30. | Clancy TE, Moore FD Jr, Zinner MJ. Post-gastric bypass hyperinsulinism with nesidioblastosis: subtotal or total pancreatectomy may be needed to prevent recurrent hypoglycemia. J Gastrointest Surg. 2006;10:1116-1119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 98] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 31. | Thompson SM, Vella A, Thompson GB, Rumilla KM, Service FJ, Grant CS, Andrews JC. Selective Arterial Calcium Stimulation With Hepatic Venous Sampling Differentiates Insulinoma From Nesidioblastosis. J Clin Endocrinol Metab. 2015;100:4189-4197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 32. | Anlauf M, Wieben D, Perren A, Sipos B, Komminoth P, Raffel A, Kruse ML, Fottner C, Knoefel WT, Mönig H, Heitz PU, Klöppel G. Persistent hyperinsulinemic hypoglycemia in 15 adults with diffuse nesidioblastosis: diagnostic criteria, incidence, and characterization of beta-cell changes. Am J Surg Pathol. 2005;29:524-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 100] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 33. | McElroy MK, Lowy AM, Weidner N. Case report: focal nesidioblastosis ("nesidioblastoma") in an adult. Hum Pathol. 2010;41:447-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 34. | Schwetz V, Horvath K, Kump P, Lackner C, Perren A, Forrer F, Pieber TR, Treiber G, Sourij H, Mader JK. Successful Medical Treatment of Adult Nesidioblastosis With Pasireotide over 3 Years: A Case Report. Medicine (Baltimore). 2016;95:e3272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 35. | Baweja P. Adult-Onset Nesidioblastosis, A Diagnostic and Management Dilemma. J Diabetes Medicat Care. 2019;2. |
| 36. | Tasneem AA, Luck NH, Mubarak M. Pancreato-hepatobiliary neuroendocrine tumors diagnosed through endoscopic ultrasound: Clinical characteristics and factors associated with high-grade lesions. World J Gastrointest Endosc. 2025;17:105904. [PubMed] [DOI] [Full Text] |