Published online Mar 15, 2026. doi: 10.4239/wjd.v17.i3.119126
Revised: February 13, 2026
Accepted: March 9, 2026
Published online: March 15, 2026
Processing time: 52 Days and 2.3 Hours
Diabetic gastroparesis (DGP) is a common but long-underestimated gastro
Core Tip: Diabetic gastroparesis (DGP) has traditionally been regarded as a motility disorder characterized by delayed gastric emptying. Emerging evidence indicates that DGP is a systemic disorder driven by dysfunction of the brain-gut axis, involving the central nervous system, autonomic and enteric neural networks, immune-inflammatory responses, and gut microbiota dysbiosis. This review integrates these multilevel mechanisms into a unified brain-gut axis framework and highlights therapeutic strategies targeting neural regulation, inflammation, and microbiota, providing a conceptual basis for mechanism-guided and personalized management of DGP.
- Citation: Jiang WL, Hu XY, Zheng HX, Nan MH, Liang LZ, Wang TX, Li JL, Bai CC. Dysfunction of the brain-gut axis in diabetic gastroparesis: Underlying mechanisms and therapeutic strategies. World J Diabetes 2026; 17(3): 119126
- URL: https://www.wjgnet.com/1948-9358/full/v17/i3/119126.htm
- DOI: https://dx.doi.org/10.4239/wjd.v17.i3.119126
Diabetes has become one of the most rapidly growing and burdensome chronic metabolic diseases worldwide. As reported by the International Diabetes Federation, approximately 529 million individuals worldwide have diabetes, with over 90% diagnosed with type 2 diabetes mellitus (T2DM); this number is expected to rise to 1.3 billion by 2050[1]. In China, the number of individuals with diabetes has exceeded 140 million, exerting substantial medical and socioeconomic burdens[2]. In addition to cardiovascular, renal, and neurological complications, gastrointestinal motility disorders are also common among patients with diabetes, yet their clinical significance has long been underestimated[3,4]. The incidence of gastrointestinal dysfunction in individuals with diabetes is significantly higher than that in the general population, with the overall prevalence of dyspepsia being approximately 70% higher[5].
Diabetic gastroparesis (DGP) is a typical peripheral manifestation of diabetic autonomic neuropathy (DAN), characterized by delayed gastric emptying (DGE) and associated upper gastrointestinal symptoms without evidence of mechanical obstruction[6]. Common clinical manifestations include nausea, vomiting, early satiety, postprandial fullness, abdominal distension, and upper abdominal discomfort, which often occur as symptom clusters. Epidemiological data on DGP show substantial variability, with reported incidence rates ranging from 1% to 60%[7,8]. Studies based on United States electronic medical record databases indicate that the overall recorded prevalence of DGP is only 0.16%; however, DGP accounts for 57.4% of all recorded gastroparesis cases[9]. Among patients with type 1 diabetes mellitus and T2DM, the cumulative incidence of gastroparesis is approximately 4.8% and 1%, respectively, compared with approximately 0.1% in the non-diabetic population[10]. Diabetic gastrointestinal complications are characterized by a diffuse distribution and may involve multiple segments of the digestive tract, with esophageal involvement reported in more than 60% of cases, indicating that impairment of the autonomic and enteric nervous system (ENS) is systemic in nature[11,12].
Recent studies indicate that DGP and functional dyspepsia (FD) show considerable overlap in symptom phenotypes and several pathophysiological mechanisms, suggesting that they may represent distinct phenotypes within the same gastric neuromuscular disease spectrum[13]. Both FD and DGP exhibit impaired gastric regulation, increased visceral sensitivity, as well as abnormal central signal processing[14-16], and their symptoms are readily influenced by external factors, including psychosocial stress[17]. In addition, these two conditions share a common pathophysiological basis involving brain-gut axis dysfunction, characterized by low-grade inflammation and gut microbiota dysbiosis[18,19]. Although DGE is often used as an objective diagnostic indicator for DGP, the association between symptom severity and gastric motility abnormalities remains weak[20], suggesting that a motility-centered paradigm alone is insufficient to explain its complex clinical manifestations. A bibliometric cluster analysis revealed that “diabetic gastroparesis” and the “gut-brain-microbiota axis” frequently appeared within the same thematic clusters, suggesting a gradual shift in research focus from a traditional motility-disorder paradigm toward a brain-gut axis-oriented framework[21]. Therefore, reinterpreting the mechanisms of DGP through the brain-gut axis represents a promising approach to elucidate symptom heterogeneity and broaden potential therapeutic targets.
A complex neuroendocrine regulatory network linking the brain and gut, termed the brain-gut axis, maintains gastrointestinal motility and sensory homeostasis through multilayered integration of the central nervous system (CNS), autonomic nervous system (ANS), and ENS[22]. In addition, this system interacts dynamically with the immune, endocrine, and gut microbiota systems, forming a bidirectional regulatory network. Chronic hyperglycemia and associated metabolic stress disrupt the homeostatic coupling among gastric wall nerves, smooth muscle cells, and immune cells through mechanisms including oxidative stress, deposition of advanced glycation end products, and microvascular lesions, thereby leading to autonomic dysfunction and impaired gastric motility[3,23-25]. Overall, DGP should not be regarded as a single motility disorder, but rather as a syndrome characterized by multidimensional imbalance across neural, immune, and microecological systems driven by chronic hyperglycemia. Accordingly, this review systematically summarizes the pathological mechanisms and regulatory targets of DGP from the perspective of the brain-gut axis, with a particular focus on the interactions among dysglycemia, autonomic neuropathy, neuro-immune coupling, and gut microbiota, to provide insights into mechanistic interventions and precision therapeutic strategies.
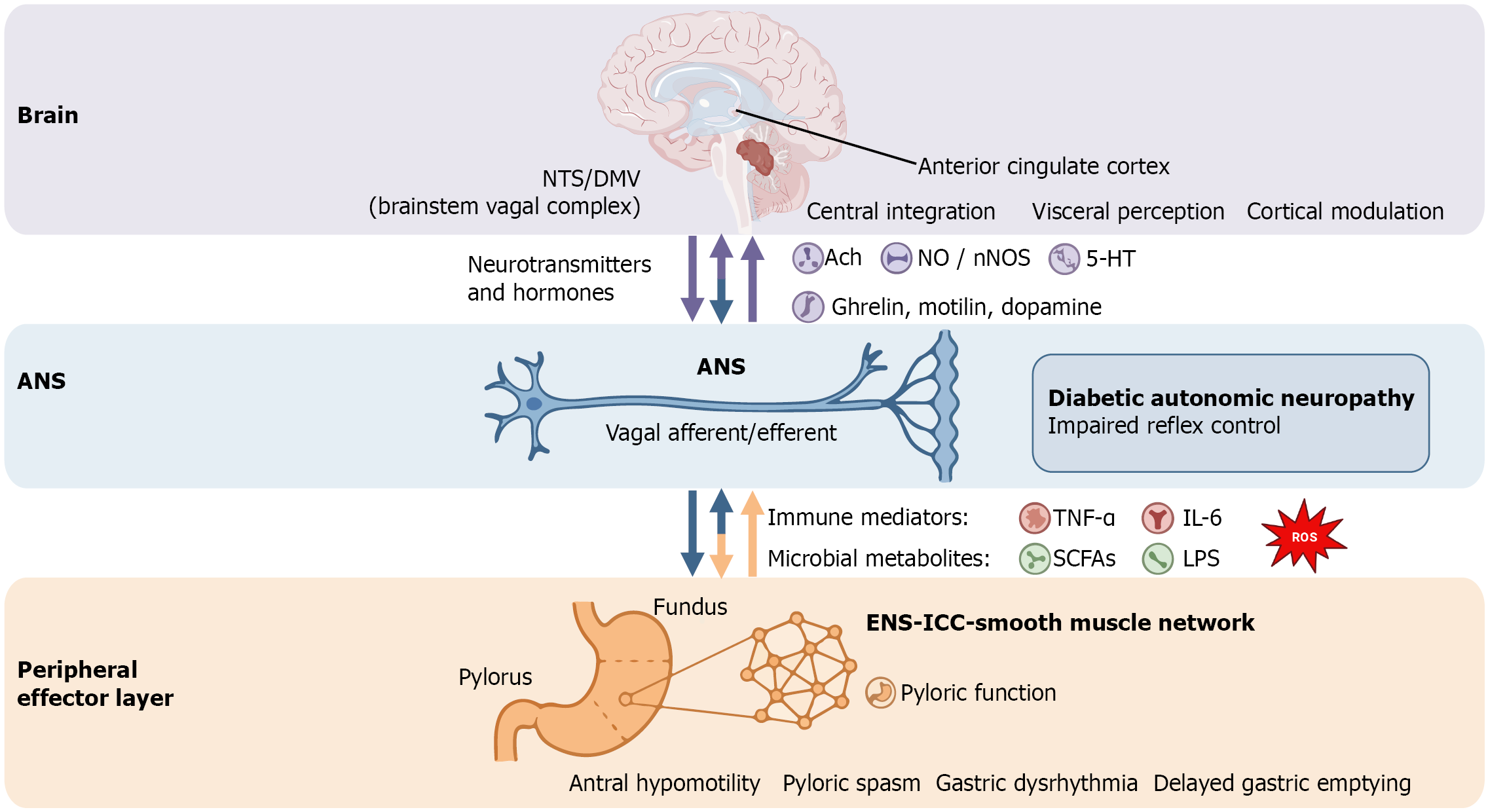
DGP can be considered a systemic pathological condition centered on multilevel dysfunction of the brain-gut axis. Accumulating evidence indicates that long-term hyperglycemia induces oxidative stress and chronic low-grade inflammation, which progressively impair the structural and functional integrity of the CNS, ANS, and ENS, thereby disrupting the coordinated regulatory network among enteric neurons, interstitial cells, and smooth muscle. Within this framework, the CNS functions as the primary hub for information integration and regulation, the ANS serves as a critical interface linking central control with peripheral effectors, and the ENS and its effector cells directly govern the execution of gastrointestinal motility and sensory functions. In addition, gut microbiota dysbiosis and immune-inflammatory abnormalities interact with neural injury through metabolic, neural, and humoral pathways, forming a positive feedback loop that amplifies pathological signaling and accelerates the development and progression of gastrointestinal dysfunction. Overall, the pathophysiological evolution of DGP represents a continuous process driven by metabolic disturbances, dysregulated neural control, and cumulative impairment of peripheral effectors, ultimately resulting in DGE and heterogeneous clinical manifestations (Figure 1). Based on this integrative framework, the following sections systematically delineate the specific roles and interrelationships of the individual components of the brain-gut axis in the pathogenesis of DGP.
Diabetes-related metabolic stress can impair the brainstem vagal complex, including the nucleus tractus solitarius and the dorsal motor nucleus of the vagus nerve, thereby weakening the integrated control of gastrointestinal motility. Neuroimaging studies have revealed structural and functional abnormalities in the brainstem, insula, and cingulate cortex, key regions involved in brain-gut regulation. These alterations are associated with gastroparesis symptom severity, indicating that changes in CNS plasticity contribute to symptom generation in DGP[26-30]. Animal studies further demonstrated that downregulation of neuregulin-1 in the dorsal motor nucleus of the vagus nerve leads to impaired cholinergic anti-inflammatory reflexes and reduced gastric motility, whereas neuregulin-1 overexpression activates α7nAChR signaling, restores gastric emptying, and attenuates local inflammation[31]. Collectively, these findings highlight the critical role of CNS plasticity in the pathogenesis of DGP. Clinical studies have shown that patients with diabetes are more prone to early satiety and nausea, whereas patients with idiopathic gastroparesis more frequently report abdominal pain[32,33]. Furthermore, the non-linear association between symptom severity and DGE suggests that disordered central sensory processing and signal integration may substantially contribute to symptom development in DGP[34-37]. Structural and functional abnormalities of the CNS not only disrupt vagal output but also alter central perception and regulation of gastric signals, thereby playing a pivotal role in brain-gut axis dysfunction in DGP.
Within the brain-gut axis regulatory network, the ANS serves as a critical interface linking the CNS and ENS. DAN represents a key pathological component of brain-gut axis imbalance, leading to disrupted coupling between central and peripheral signaling and constituting an important mechanistic basis of DGP. The vagus nerve is the primary neural mediator of gastric receptive relaxation, antral propulsion, and pyloric relaxation[38], with its activity jointly regulated by glycemic and hormonal signals. Acute hyperglycemia delays gastric emptying, whereas hypoglycemia accelerates gastric emptying via vagal reflexes[39,40]. Gastrointestinal hormones, including gastrin, secretin, and insulin, modulate gastric motility via both central and peripheral pathways. Insulin enhances gastric contractility via vagal nuclei, whereas gastrin primarily facilitates small intestinal peristalsis[41,42]. DAN affects both afferent and efferent pathways, resulting in impaired antral propulsion, limited pyloric relaxation, and visceral sensory dysfunction, which manifest clinically as early satiety, bloating, and epigastric abdominal discomfort[35,43,44]. These alterations are more pronounced in female patients and in individuals with longer disease duration and higher hemoglobin A1c levels[45].
The pathological manifestations of DAN include loss of sympathetic and vagal ganglion cells, reduction of interstitial cells of Cajal (ICC), and inflammatory infiltration of the enteric nerve plexus[13,20]. Prolonged hyperglycemia induces oxidative stress and microvascular ischemia through activation of the polyol and protein kinase C pathway, and accumulation of advanced glycation end products, thereby promoting neurodegeneration and ICC damage[46-48]. In addition, downregulation of neuronal nitric oxide synthase (nNOS) restricts smooth muscle relaxation, leading to pyloric spasm and DGE[49]. This peripheral neurodegeneration is frequently accompanied by systemic autonomic dysfunction. Studies have shown that DGE frequently coexists with cardiac denervation, retinopathy, and nephropathy[50-53]. Clinical and epidemiological investigations further support these observations: (1) Patients with gastroparesis often exhibit cardiovascular vagal nerve impairment[54]; (2) Approximately 60% of patients demonstrate DGE on gastric scintigraphy; and (3) Improved glycemic control is positively correlated with symptom relief[55]. Moreover, a study by AlOlaiwi et al[56] reported prevalence rates of 15.3% for cardiovascular autonomic neuropathy and 6.3% for gastroparesis symptoms, respectively, suggesting that DGP represents a peripheral phenotype of diabetic neural axis injury. Autonomic function tests further demonstrate attenuated cardiac vagal reflexes, reduced pancreatic polypeptide responses, and decreased heart rate variability in affected patients[57-61]. Overall, autonomic neuropathy – through impaired central integration and diminished vagal pathway function – disrupts the bidirectional coordination of brain-gut signaling, constituting a core pathological link in diabetic brain-gut axis imbalance and representing a key mechanistic basis for the development and progression of DGP[62-66].
Brain-gut axis dysfunction in DGP extends beyond structural neurodegeneration and involves dysregulation of multiple neurotransmitters and gastrointestinal hormones. Reduced excitatory mediators[67-70], including acetylcholine, substance P, and ghrelin, together with aberrant expression of inhibitory mediators such as nitric oxide (NO), cholecystokinin (CCK), vasoactive intestinal peptide, and monoamine neurotransmitters (serotonin, dopamine, and norepinephrine), disrupt the balance between gastric contraction and relaxation[71-73]. These neurotransmitters and hormones are subject to multilevel regulation by the CNS, ANS, and ENS and are further modulated by metabolic, psychological stress-related, and inflammatory factors[74-76]. Disruption of these regulatory processes impairs the integration and transmission of brain-gut signaling, thereby contributing to DGE and visceral sensory dysfunction.
Motilin is a key gastrointestinal hormone involved in the regulation of gastrointestinal motility. It primarily acts on the gastric body and antrum, thereby inducing antral contraction and pyloric relaxation and promoting gastric emptying during the interdigestive period[77]. In the fasting state, plasma motilin levels increase synchronously with phase III activity of the migratory motor complex, thereby triggering gastric peristalsis via cholinergic pathways[78]. In contrast, postprandial motilin levels decline, accompanied by relaxation of the gastric fundus to accommodate increased gastric volume. Exogenous motilin enhances proximal gastric tone and accelerates gastric emptying[79], and stimulates insulin secretion via neurohumoral mechanisms, suggesting a regulatory role in the gut-pancreas axis[80]. Motilin receptors are widely expressed in gastric smooth muscle cells and enteric nerve plexuses, rendering them important targets for the modulation of gastric motility. Accordingly, motilin receptor agonists, including erythromycin derivatives, have been shown to accelerate gastric emptying in patients with DGP, particularly in those with vagal nerve dysfunction[81,82].
Ghrelin is primarily produced by endocrine cells in the gastric fundus and body and belongs to the G protein-coupled receptor ligand family, exhibiting structural homology to kinins[83,84]. Ghrelin exerts dual effects on the central and peripheral nervous systems via activation of the growth hormone secretagogue receptor 1α, thereby promoting appetite, enhancing gastric contractions, and accelerating gastric emptying[85-87]. In healthy individuals, ghrelin levels exhibit phase-dependent oscillations in synchrony with migratory motor complex activity, thereby enhancing gastric motility during the interdigestive period[88]. In early-stage diabetes mellitus, circulating ghrelin levels are elevated and are associated with increased gastrointestinal motility and appetite, whereas ghrelin levels decline in advanced stages, potentially reflecting reduced gastric motility and DGE[89]. Clinical and experimental studies further suggest that exogenous ghrelin or its analogues may promote gastric emptying and relieve symptoms in DGP patients, highlighting its potential therapeutic value[90].
The 5-hydroxytryptamine (5-HT) is a key neurotransmitter in the brain-gut axis, with approximately 95% synthesized by enterochromaffin (EC) cells. Food intake and mechanical distension stimulate EC cells to secrete 5-HT, thereby activating 5-HT receptors on intrinsic and extrinsic neurons of the ENS and regulating intestinal peristalsis, secretion, and blood flow[91,92]. Among 5-HT receptor subtypes, 5-HT type 4 receptor (5-HT4) represent key therapeutic targets for gastric motility regulation, and their agonists have been shown to improve various functional gastrointestinal motility disorders, including constipation-predominant irritable bowel syndrome, FD, and DGP[93].
In addition to excitatory neurotransmitters, inhibitory hormones also play critical roles in gastric motility regulation. Somatostatin (SST) is mainly produced by D cells in the pyloric region and oxyntic mucosa of the stomach. Through paracrine signaling, SST inhibits gastrin secretion from G cells, histamine release from EC-like cells, and gastric acid secretion by parietal cells, thereby maintaining homeostasis between gastric acid secretion and motility[94,95]. SST and its receptors are widely distributed in the brain and gastrointestinal tract and regulate gastrointestinal motility as well as digestion and absorption through coordinated central-peripheral pathways[96]. In addition, CCK exerts inhibitory effects on gastric motility by activating intramuscular non-adrenergic and non-cholinergic inhibitory neurons. CCK1 receptor antagonists have been demonstrated to promote gastric emptying and exhibit potential therapeutic value in gastroparesis and gastroesophageal reflux disease[97].
As a key hub in the peripheral effector layer of the brain-gut axis, the ENS regulates gastrointestinal motility and secretion through bidirectional signal integration with the CNS[98,99]. The ENS consists primarily of the submucosal and myenteric plexuses. The submucosal plexus mainly regulates glandular secretion and mucosal activity, whereas the myenteric plexus is responsible for generating and coordinating peristaltic rhythms[100]. Both plexuses communicate with the CNS via sympathetic and parasympathetic pathways, forming an integrated neural network for gastrointestinal motility control. ICCs serve as pacemakers in the gastrointestinal tract by generating spontaneous slow-wave electrical activity and coordinating smooth muscle contraction, thereby providing the rhythmic basis for gastric motility[101]. Under physiological conditions, excitatory (cholinergic, substance P-positive) and inhibitory (nitrergic, purinergic) neurons function in concert to maintain the balance between gastric contraction and relaxation[43,102]. Dysfunction of either neuronal subtype disrupts gastric rhythmicity and results in DGE[41].
In diabetic patients, both ENS neurons and ICCs exhibit structural degeneration. Histological studies have demonstrated decreased nerve fiber density in the gastric wall of patients with DGP, accompanied by a reduction in nNOS-positive neurons, findings that are consistent with ICC loss and impaired gastric motility[103-105]. Pasricha et al[106] observed marked reductions in nerve fibers and myenteric neurons in full-thickness gastric wall biopsies, accompanied by smooth muscle fibrosis and ICC loss. Iwasaki et al[107] reported a marked reduction in ICCs within the antral circular muscle layer of patients with T2DM, accompanied by decreased nNOS and substance P expression, resulting in disordered gastric smooth muscle rhythmicity. Animal studies further support these observations, showing that in non-obese diabetic and streptozotocin-induced models, ICC reduction and DGE occur in parallel[41,102,108]. Mechanistically, downregulation of insulin/insulin-like growth factor 1 (IGF-1) signaling reduces stem cell factor secretion by smooth muscle cells, thereby impairing ICC maintenance and survival[109-111]. Furthermore, selective loss of nitrergic inhibitory neurons induces pyloric spasm and impaired gastric regulation, whereas insulin supplementation restores nNOS expression and improves gastric emptying[102,112]. Notably, Gangula et al[113] reported that tetrahydrobiopterin levels are significantly decreased in the gastric tissue of female diabetic rats, thereby impairing nNOS dimerization and NO bioavailability; tetrahydrobiopterin supplementation restored NO signaling and improved pyloric relaxation and gastric emptying.
Dysfunction of the ENS and ICCs directly leads to abnormal gastrointestinal motility. Decreased antral contractility and pyloric spasm are considered the principal motor abnormalities underlying DGE[53,112,114]. Alterations in neurotransmitter and neuropeptide signaling further exacerbate this process. Experimental studies have shown that levels of substance P and neuropeptide Y are significantly reduced in DGP animal models, whereas the neuropeptide Y receptor Y1 is not correspondingly upregulated, suggesting that neuropeptide depletion contributes to disease progression[115]. Furthermore, some patients continue to exhibit significant symptoms even in the presence of normal gastric emptying, indicating that ENS degeneration may promote symptom persistence by impairing sensory integration and central signal processing[116]. In summary, structural degeneration and signal imbalance within the ENS-ICC-smooth muscle network constitute a core mechanism of peripheral effector dysfunction in DGP and represent a key downstream manifestation of brain-gut axis dysfunction in diabetes.
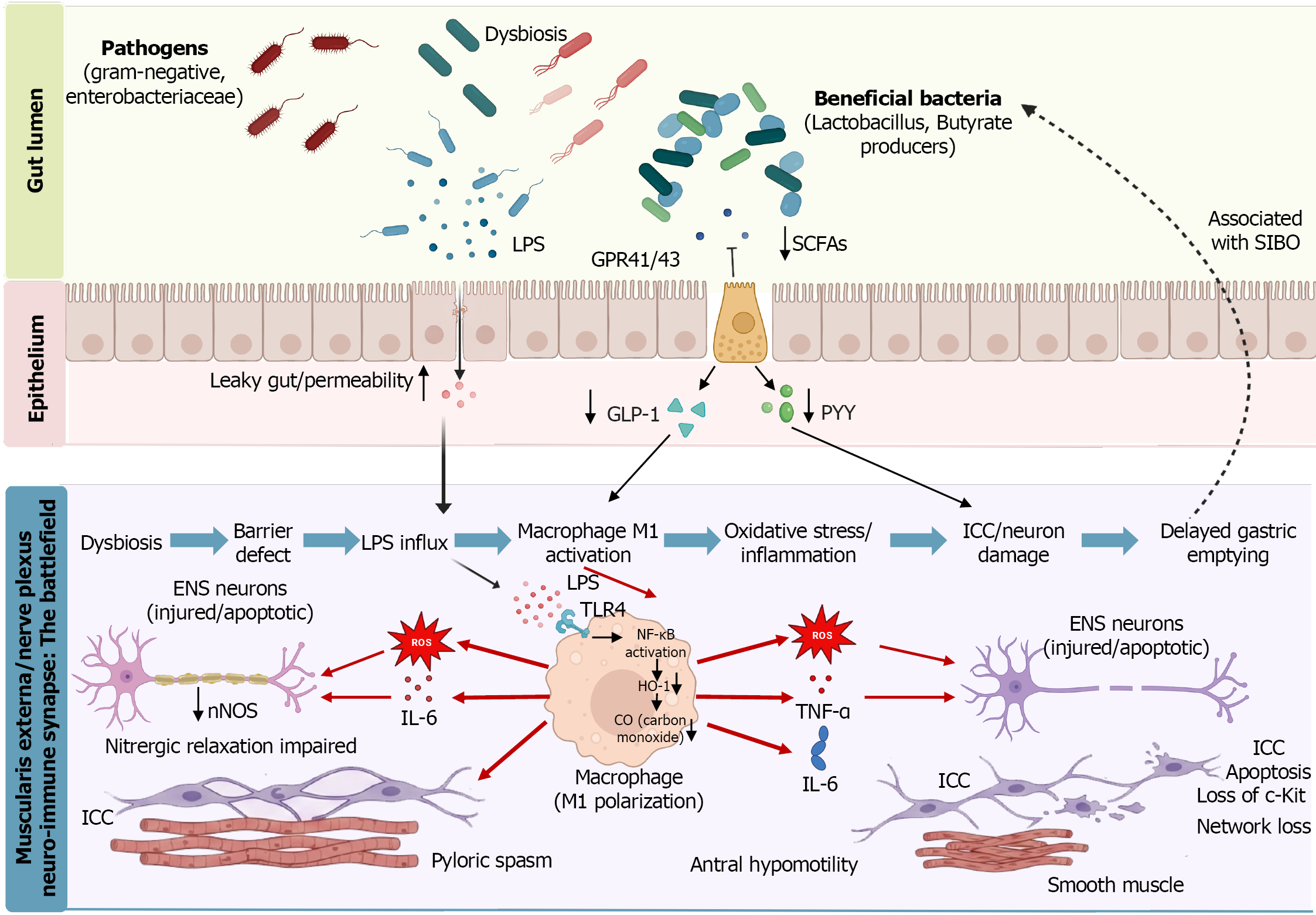
The gut microbiota, consisting of approximately one thousand bacterial species and trillions of microorganisms, plays a pivotal role in nutrient absorption, energy metabolism, and immune homeostasis[117]. In diabetes, the composition and function of the gut microbiota are markedly altered, marked by a reduction in beneficial taxa such as Lactobacillus and Bifidobacterium, along with an expansion of potentially pathogenic bacteria, including Gram-negative bacteria and members of the Enterobacteriaceae family[118,119]. This dysbiosis compromises intestinal barrier integrity, increases mucosal permeability and endotoxin translocation, and thereby promotes systemic inflammation, oxidative stress, and gastrointestinal motility disorders[120]. Diabetic neuropathy further exacerbates microbial dysbiosis, with DGE and small intestinal bacterial overgrowth forming a positive feedback loop between motility impairment and microbiota imbalance[121,122]. Epidemiological studies have reported that small intestinal bacterial overgrowth occurs in approximately 29% of diabetic patients, significantly higher than that in non-diabetic controls, and that approximately 45% of patients exhibit gastrointestinal complications on endoscopic evaluation[123,124]. Microbiome profiling has revealed a reduced Firmicu
Microbial metabolites are key mediators of brain-gut axis signaling, and alterations in their profiles directly influence neuroendocrine-immune interactions. Short-chain fatty acids enhance secretion of incretins, including glucagon-like peptide-1 (GLP-1) and peptide YY, via G protein-coupled receptors 41 and 43 activation[129,130]. Under dysbiotic conditions, butyrate production is reduced, leading to impaired ENS signaling and reduced vagal nerve sensitivity, thereby exacerbating neuropathic dysfunction[131]. Notably, butyrate enhances neuronal energy metabolism via the adenosine monophosphate-activated protein kinase-peroxisome proliferator-activated receptor γ coactivator-1α signaling pathway, thereby exerting neuroprotective effects[132,133]. Conversely, lipopolysaccharide derived from Gram-negative bacteria induces inflammatory responses and neuronal apoptosis via the toll-like receptor 4/nuclear factor-kappa B (NF-κB) pathway[134,135], whereas depletion of Akkermansia muciniphila compromises intestinal barrier integrity, further aggravating endotoxemia and neuroinflammation[136]. Collectively, these metabolic and inflammatory pathways constitute a metabolic-immune-neural coupling mechanism that drives peripheral pathophysiological processes in DGP.
The brain-gut axis mediates bidirectional communication between the gastrointestinal tract and the CNS, predominantly via the vagus nerve[137-139]. Diabetes-induced vagal degeneration, axonal demyelination, and reduced plasticity of the nucleus tractus solitarius impair vagal efferent signaling, resulting in DGE, postprandial fullness, and visceral sensory abnormalities[140,141]. Attenuated vagal function further reduces GLP-1 secretion, thereby weakening glycemic control and gastric motility regulation and forming a vicious cycle of metabolic imbalance and gastroparesis[142-144]. In addition, the gut microbiota can act on EC cells through microbiota-derived short-chain fatty acids, thereby promoting intestinal 5-HT production and modulating gastrointestinal neuroregulation and motility[145]. Grasset et al[122] further demonstrated that specific gut microbiota dysbiosis inhibits GLP-1-induced NO synthesis in enteric neurons, thereby inducing GLP-1 resistance and highlighting a key regulatory role of the gut microbiota in the pathogenesis of DGP through interference with ENS and endocrine signaling. In summary, gut microbiota dysbiosis constitutes a critical link in the peripheral pathophysiological processes of DGP through coordinated metabolic, immune, and neural signaling dysregulation. The key immune-microbiota-neural interactions and their associated molecular mechanisms are schematically illustrated in Figure 2.
Brain-gut axis disruption in DGP is closely linked to inflammatory and immune dysregulation. Chronic hyperglycemia induces oxidative stress and metabolic inflammation, thereby promoting activation of immune cells within the intestinal wall and disrupting neuromuscular homeostasis. Cipriani et al[146] demonstrated that muscularis macrophages are key mediators of DGE in diabetes. In macrophage-deficient mice, ICC damage failed to develop even under hyperglycemic conditions, whereas wild-type diabetic mice exhibited marked ICC network disruption accompanied by increased oxidative stress. Under diabetic conditions, oxidative stress and inflammatory responses can induce enteric neuronal apoptosis and reduce nNOS-positive neurons[147-149]. In contrast, activation of transient receptor potential vanilloid 1 channels in vagal afferent fibers promotes NO release and improves pyloric relaxation[150]. Collectively, these findings suggest a bidirectional interplay between peripheral immune activation and neural reflexes in the regulation of gastric motility.
Animal studies have further elucidated the role of immune polarization in the development of gastroparesis. Under physiological conditions, macrophages are predominantly of the M2 phenotype expressing heme oxygenase-1 (HO-1), thereby maintaining a local antioxidant microenvironment. In contrast, diabetic mice exhibit a predominance of M1-polarized macrophages that release pro-inflammatory mediators, including tumor necrosis factor-α and interleukin-6, leading to ICC apoptosis[151-153]. In non-obese diabetic mice, failure of HO-1 upregulation leads to ICC loss and DGE, whereas hemin-induced HO-1 expression reverses these changes[154]. In contrast, in patients with diabetic gastroenteropathy, hemin treatment does not significantly improve gastric emptying, suggesting the presence of irreversible structural damage in human disease[155]. In addition, Berntorp et al[156] demonstrated that diabetic patients exhibit elevated levels of gonadotropin-releasing hormone autoantibodies, particularly immunoglobulin G isotypes, which are closely associated with autonomic neuropathy, suggesting that immune responses may interfere with neuroendocrine signaling along the brain-gut axis. Consistently, the streptozotocin-induced diabetic model further confirmed that M1/M2 imbalance and reduced HO-1 activity weaken macrophage-mediated protection, leading to ICC loss and DGE[63,146,157]. Notably, Kashyap et al[158] demonstrated that carbon monoxide, a downstream product of HO-1, mimics its protective effects by reducing oxidative stress, restoring c-Kit expression, and improving gastric emptying.
Chronic inflammation and oxidative stress further exacerbate brain-gut axis dysfunction by inducing injury to the ENS and central regulatory pathways. Hyperglycemia-induced oxidative damage reduces ICC density, promotes neuronal degeneration, and impairs smooth muscle contractility, thereby compromising intrinsic gastrointestinal motility regulation[50,159]. In addition, inflammatory mediators and oxidative molecules generated during immune activation can influence central regulatory nuclei, including the nucleus tractus solitarius and the hypothalamus, via vagal afferent signaling and humoral pathways[160]. This process establishes a positive neuro-immune feedback loop, further amplifying gastric motility disturbances and ANS dysfunction. Overall, inflammation, immune dysregulation, and oxidative stress collectively constitute key pathological drivers of brain-gut axis imbalance, driving the progression of DGP from peripheral inflammatory responses toward central signaling dysregulation.
Pharmacological therapy remains the cornerstone of intervention for DGP, and its therapeutic efficacy can be conceptualized as multilevel modulation across the central, peripheral, and effector layers of the brain-gut axis. Current clinical practice and research primarily focus on three major classes of agents, including dopamine receptor antagonists, motilin receptor agonists, and ghrelin receptor agonists. Emerging studies are further exploring multitarget strategies involving intracellular signaling pathways, gut microbiota modulation, and smooth muscle function.
Dopamine receptor antagonists exert bidirectional brain-gut regulatory effects by modulating both central and peripheral dopaminergic signaling. Network meta-analyses have demonstrated that oral dopamine antagonists significantly improve gastroparesis symptoms compared with placebo[161]. Metoclopramide exerts both peripheral prokinetic and central antiemetic effects by enhancing lower esophageal sphincter tone, promoting antral and duodenal peristalsis, and reducing nausea and vomiting via blockade of D2 and 5-HT type 3 receptor within the medullary chemoreceptor trigger zone[162-165]. As the only Food and Drug Administration-approved pharmacological treatment for gastroparesis, its long-term use is limited by its ability to cross the blood-brain barrier, leading to extrapyramidal adverse effects[166,167]. Notably, novel nanoparticle-based delivery systems, such as bovine serum albumin-metoclopramide composite nanoparticles, offer a promising strategy for brain-gut axis-targeted drug delivery by reducing CNS toxicity while preserving therapeutic efficacy[168]. Domperidone, a peripheral D2 receptor antagonist, exhibits a favorable safety profile owing to its limited penetration of the CNS and effectively alleviates symptoms such as early satiety, nausea, and bloating[169-172], suggesting that its therapeutic effects are primarily mediated through peripheral neural pathways regulating gastric motility.
Motilin receptor agonists mimic endogenous motilin-mediated activation of gastric antral and pyloric neuromuscular circuits, thereby promoting phase III migratory motor complex activity, accelerating gastric emptying, and restoring peripheral rhythmicity of the brain-gut axis[173,174]. As a representative prokinetic, erythromycin exhibits short-term benefits by promoting gastric motility and alleviating relevant symptoms[175]. However, long-term use of erythromycin may increase the risk of cardiac arrhythmias. Azithromycin, with a longer half-life and a more favorable safety profile, has therefore been proposed as a potential alternative[176,177].
Ghrelin receptor agonists, by activating the brain-gut-pancreas signaling axis, coordinate gastric motility and systemic energy metabolism and have emerged as a focus of recent pharmacological research targeting the brain-gut axis. Representative agents include ulimorelin (TZP-101/102) and relamorelin (RM-131), both of which have demonstrated significant acceleration of solid gastric emptying and improvement of nausea and upper gastrointestinal symptoms in preclinical and clinical studies[178-181]. Phase II clinical trials of relamorelin further showed that subcutaneous administration significantly improved gastric emptying half-time and alleviated core gastroparesis symptoms[182].
Furthermore, 5-HT4 receptor agonists including mosapride and prucalopride, enhance acetylcholine release, thereby promoting antral propulsion and gastric emptying and representing an excitatory modulatory pathway within brain-gut axis chemical signaling. Animal studies have demonstrated that mosapride, either alone or in combination with metformin, improves gastric emptying and glycemic control in diabetic rat models[183]. Randomized controlled trials have further shown that prucalopride significantly accelerates solid gastric emptying, although symptomatic improvement appears limited[184]. In addition, the centrally acting neurokinin-1 receptor antagonist aprepitant alleviates nausea and vomiting and improves gastric regulatory function[185].
Beyond conventional prokinetic agents, targeted modulation of the gut microbiota represents an emerging dimension of brain-gut axis regulation. Studies in functional gastrointestinal disorders have demonstrated that gut microbiota dysbiosis and altered microbial metabolites disrupt brain-gut signaling and gastric motility by modulating ENS excitability, intestinal immune responses, and epithelial barrier integrity[186]. The herbal medicine Alpinia officinarum Hance extract markedly enhances gastric motility and mucosal architecture in DGP rat models, exerting multitarget brain-gut regulatory effects by upregulating the SCF/c-Kit signaling pathway and reshaping gut microbiota composition[187]. Similarly, the compound traditional Chinese medicine FoxiangSan improved gastric motility by modulation of gut microbiota structure and activation of the cAMP/PKA/5-HT4 signaling pathway, thereby supporting the concept of synergistic interactions among the gut microbiota, intracellular signaling pathways, and the brain-gut axis[188]. In addition, Salsola collina ethyl acetate extract promoted restoration of brain-gut signaling through upregulation of c-Kit and SCF expression and correction of abnormalities in multiple brain-gut peptides[72], whereas Amomum compactum volatile oil enhanced gastric emptying via activation of IGF-1/IGF-1R signaling[189]. Curcumin and berberine have been shown to reconstruct peripheral neural function within the brain-gut axis through inhibition of oxidative stress, modulation of NF-κB signaling, enhancement of cholinergic transmission, and repair of gastric nerve plexus structure, respectively[190,191]. Furthermore, plant-derived serotonin promotes gastric smooth muscle cell proliferation and inhibits apoptosis via the β2-adrenergic receptor/phosphatidylinositol 3-kinase/protein kinase B pathway, thereby improving the structural and functional integrity of the peripheral effector component of the brain-gut axis[192].
Overall, pharmacological therapy modulates brain-gut axis function across central-to-peripheral chemical signaling levels through regulation of a multilayered signaling network involving neurotransmitters, gastrointestinal hormones, gut microbiota, and smooth muscle-associated pathways. Future research should emphasize multitarget synergistic mechanisms and long-term safety, and explore integrated therapeutic strategies that combine pharmacological agents with neuromodulatory and metabolic interventions to achieve systemic remodeling of brain-gut axis function (Tables 1 and 2).
| Patient phenotype | Dominant pathophysiological features | Representative agents | Therapeutic focus |
| Central-dominant phenotype | Central sensory integration and regulatory dysfunction, with symptom severity disproportionate to DGE | Metoclopramide; aprepitant | Predominantly central modulation to alleviate nausea and vomiting and improve brain-gut signal processing |
| Peripheral neuro-effector-dominant phenotype | Enteric nervous system and interstitial cells of Cajal injury with impaired antral motility and pyloric dysfunction, associated with marked DGE | Domperidone; erythromycin; azithromycin; ulimorelin (TZP-101/102); relamorelin (RM-131); mosapride; prucalopride | Enhancement of peripheral neuromuscular function to restore gastric rhythmicity and accelerate gastric emptying |
| Inflammation-microbiota-associated phenotype | Low-grade inflammation, oxidative stress, and gut microbiota dysbiosis | Alpinia officinarum extract; FoxiangSan; Salsola collina ethyl acetate extract; amomum compactum volatile oil; curcumin; berberine; plant-derived serotonin | Modulation of inflammatory responses and gut microbiota to improve overall brain-gut axis homeostasis |
| Neuromodulation modality | Target population/stage | Main therapeutic effects | Proposed mechanisms |
| GES | Refractory DGP, particularly patients with severe nausea and vomiting | Marked reduction in vomiting frequency and overall symptom improvement; variable effects on gastric emptying | Activation of vagal afferent pathways, modulation of gastric electrical rhythms, promotion of ENS synaptic regeneration, and improvement of ICC function |
| GES combined with pyloroplasty | DGP with prominent pyloric dysfunction | Greater improvement in symptoms and gastric emptying compared with single intervention | Synergistic effects of neuromodulation and mechanical outflow relief, enhancing remodeling of peripheral effector components of the brain-gut axis |
| VNS/transcutaneous VNS | DGP with autonomic dysfunction or central regulatory abnormalities | Symptom improvement and partial acceleration of gastric emptying, with inconsistent results across randomized controlled trials | Enhancement of vagal efferent activity, modulation of central-peripheral brain-gut signaling, and activation of cholinergic anti-inflammatory reflexes |
| Intrapyloric botulinum toxin injection | DGP associated with pyloric spasm | Short-term improvement in gastric emptying and symptoms; limited long-term efficacy | Local inhibition of acetylcholine release and smooth muscle excitability, with limited capacity for sustained brain-gut axis remodeling |
| Low-energy shockwave | Experimental stage (preclinical DGP models) | Improvement of gastric contractile function and increased density of enteric neuromuscular plexuses | Promotion of axonal regeneration and enhancement of gastrointestinal neuromuscular structural integrity |
| EA/transcutaneous EA | Mild to moderate DGP or adjunctive therapy | Improvement of nausea, restoration of gastric slow-wave rhythm, and acceleration of gastric emptying | Regulation of autonomic balance via the nucleus tractus solitarius-vagal pathway, restoration of excitatory-inhibitory neurotransmitter balance in the ENS, and promotion of ICC protection and regeneration |
| Low-intensity pulsed ultrasound | Experimental stage (preclinical DGP models) | Acceleration of gastric emptying and improvement of ICC network integrity | Inhibition of the tumor necrosis factor-α/Ikappa B kinase beta/nuclear factor-kappa B inflammatory pathway and restoration of ENS-ICC coupling |
Neuromodulation therapy has emerged as a major focus in DGP research in recent years. By modulating neural signaling within the brain-gut axis, neuromodulatory interventions have the potential to improve gastric motility disturbances and ANS dysfunction.
Gastric electrical stimulation (GES) is currently the most widely applied neuromodulation technique. A multicenter prospective randomized crossover study by McCallum et al[193] demonstrated that, in patients with drug-refractory DGP, Enterra stimulation resulted in a 57% reduction in weekly vomiting frequency and a 67.8% improvement in overall symptoms at one year, accompanied by improvements in gastric emptying and quality of life. Additional studies have further confirmed that GES alleviates symptoms primarily through activation of vagal afferent fibers[194,195]. Animal studies have demonstrated that GES promotes enteric synaptic regeneration, upregulates synaptophysin, PGP9.5, 5-HT2B, nNOS, choline acetyltransferase, and glial cell line-derived neurotrophic factor, and thereby improves ICC function[196,197]. Vagus nerve stimulation (VNS) and its non-invasive modality (tVNS) represent key neuromodulatory approaches within the central-peripheral axis of the brain-gut system. Early studies suggested that tVNS could improve symptoms and accelerate gastric emptying[198]; however, results from subsequent multicenter randomized controlled trials have been inconsistent[199]. These findings indicate that peripheral vagal stimulation alone may be insufficient, and its therapeutic efficacy may be influenced by vagal nerve degeneration and central neural plasticity.
Pyloric dysfunction is regarded as a critical peripheral pathological component in DGP. Botulinum toxin transiently improves gastric emptying and associated symptoms by inhibiting acetylcholine release and excitatory neuromuscular transmission at nerve terminals[200]. However, randomized controlled trials have failed to demonstrate sustained efficacy, indicating that isolated local neural inhibition is insufficient to remodel brain-gut axis function[201]. Recent studies have demonstrated that GES combined with pyloroplasty significantly improves both symptoms and gastric emptying compared with pyloroplasty alone[202]. These findings suggest that neuro-mechanical dual-target interventions focusing on the pyloric component may represent a promising strategy for reconstruction of peripheral brain-gut axis function.
Physical energy-based neuromodulation techniques have also demonstrated therapeutic potential in DGP. Wu et al[203] demonstrated that low-energy shockwave therapy promotes axonal regeneration, increases the density of gastrointestinal myenteric fibers, and enhances gastric contractility. In addition, low-intensity pulsed ultrasound applied to the ST36 acupoint has been shown to improve gastric emptying in diabetic rat models, primarily through inhibition of the tumor necrosis factor-α/Ikappa B kinase beta/NF-κB inflammatory pathway and restoration of the ICC network[204]. Electroacupuncture (EA), as a non-invasive neuromodulatory approach, represents an important intervention targeting peripheral components of the brain-gut axis. Sarosiek et al[205] demonstrated, using combined EEG-EGG monitoring, that transcutaneous EA significantly reduces nausea scores and restores gastric slow-wave rhythm in patients with DGP, while modulating activity distribution in the prefrontal cortex, suggesting symptom improvement via a brain-prefrontal cortex-gastric electrical coupling mechanism. Animal studies have demonstrated that EA restores excitatory-inhibitory neurotransmitter balance within the ENS via the nucleus tractus solitarius-vagus nerve axis, upregulates nNOS and choline acetyltransferase expression in the gastric antrum, and increases ICC (c-Kit+) expression following ST36 stimulation, thereby improving gastric emptying[206-208]. Moreover, EA promotes ICC protection and regeneration by modulating autophagic flux and activating the SDF-1/CXCR4 and mSCF/c-Kit-ETV1 signaling pathways, thereby restoring ENS-ICC network integrity and enhancing gastric motility[209,210].
In summary, neuromodulation therapy improves gastric motility and symptom burden by modulating multiple hierarchical layers of the brain-gut axis, including the vagus nerve-ENS-ICC network. GES has established clinical efficacy, whereas non-invasive approaches such as tVNS, transcutaneous EA, EA, and low-energy shockwave offer emerging avenues for personalized neuromodulation. Future research should emphasize patient phenotypic stratification and stimulation parameter optimization to develop precision neuromodulation strategies that integrate central, peripheral, and gastric wall neural circuits.
Nutritional and metabolic disturbances are considered important contributors to brain-gut axis dysfunction in DGP. Ahmed et al[211] reported that 54.5% of DGP patients were found to have vitamin B12 deficiency, a prevalence significantly higher than that observed in the general diabetic population. Serum vitamin B12 levels are positively correlated with gastric emptying rates, and deficiency may result in peripheral myelin sheath damage and impaired vagal nerve conduction, suggesting that neurotrophic supplementation may help attenuate brain-gut axis degeneration. Patients with DGP frequently experience hypoglycemia and marked glycemic variability due to DGE, resulting in asynchronous insulin action and nutrient absorption[212]. Using continuous glucose monitoring, Betônico et al[213] demonstrated that semi-liquid, low-fat diets significantly reduce postprandial glycemic excursions and improve gastroparesis-related symptoms. Overall, vitamin supplementation, dietary texture optimization, and appropriate adjustment of insulin timing represent fundamental components of comprehensive DGP management by stabilizing metabolic homeostasis, enhancing gastric emptying, and improving neuroreflex coordination, thereby achieving bidirectional regulation of the brain-gut axis.
Future research should prioritize elucidating molecular mechanisms within the multilayered brain-gut axis and establishing biomarker systems that integrate neuroimaging, neurotransmitter profiling, and gut microbiota signatures to enable phenotypic stratification and precision intervention. Parallel efforts should focus on developing multitarget therapeutic strategies that combine neuromodulation, microbiota remodeling, and signaling pathway regulation. In addition, greater attention should be given to the potential roles of traditional Chinese medicine and natural bioactive compounds in brain-gut axis reconstruction and symptom control.
In summary, DGP is not merely a disorder of DGE, but rather a brain-gut axis-related syndrome arising from integrated dysfunction across central regulatory mechanisms, the autonomic and ENSs, the ICC network, pyloric function, immune inflammation, and gut microbiota under chronic hyperglycemia and metabolic stress. Diagnosis and therapeutic evaluation should therefore be based on standardized, multidimensional assessment frameworks, with four-hour solid-meal gastric emptying scintigraphy remaining a key objective measure[214]. Importantly, from a brain-gut axis perspective, clinical evaluation of DGP may benefit from integrating neural functional assessment, imaging of regulatory circuits, and gut microbiota-related indicators, rather than relying solely on gastric emptying measurements. Current management emphasizes comprehensive and individualized strategies, while available pharmacological options remain limited by long-term safety concerns[215]. Overall, DGP research is shifting from a motility-centered paradigm toward an integrated brain-gut axis model, in which multidisciplinary collaboration and mechanism-driven approaches are increasingly important for improving patient prognosis and quality of life.
| 1. | GBD 2021 Diabetes Collaborators. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2023;402:203-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3481] [Cited by in RCA: 2953] [Article Influence: 984.3] [Reference Citation Analysis (21)] |
| 2. | Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8030] [Cited by in RCA: 6225] [Article Influence: 1556.3] [Reference Citation Analysis (5)] |
| 3. | Bharucha AE, Kudva YC, Prichard DO. Diabetic Gastroparesis. Endocr Rev. 2019;40:1318-1352. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 165] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 4. | Zahid SA, Tated R, Mathew M, Rajkumar D, Karnik SB, Pramod Roy A, Jacob FP, Baskara Salian R, Razzaq W, Shivakumar D, Khawaja UA. Diabetic Gastroparesis and its Emerging Therapeutic Options: A Narrative Review of the Literature. Cureus. 2023;15:e44870. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 5. | Du YT, Rayner CK, Jones KL, Talley NJ, Horowitz M. Gastrointestinal Symptoms in Diabetes: Prevalence, Assessment, Pathogenesis, and Management. Diabetes Care. 2018;41:627-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 124] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 6. | Zhang H, You Q, Feng X, Qin Z. Emerging roles of traditional Chinese medicine in the treatment of diabetic gastroparesis. Front Nutr. 2025;12:1680943. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Choung RS, Locke GR 3rd, Schleck CD, Zinsmeister AR, Melton LJ 3rd, Talley NJ. Risk of gastroparesis in subjects with type 1 and 2 diabetes in the general population. Am J Gastroenterol. 2012;107:82-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 186] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 8. | Feldman M, Schiller LR. Disorders of gastrointestinal motility associated with diabetes mellitus. Ann Intern Med. 1983;98:378-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 413] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 9. | Ye Y, Yin Y, Huh SY, Almansa C, Bennett D, Camilleri M. Epidemiology, Etiology, and Treatment of Gastroparesis: Real-World Evidence From a Large US National Claims Database. Gastroenterology. 2022;162:109-121.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 92] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 10. | Camilleri M, Bharucha AE, Farrugia G. Epidemiology, mechanisms, and management of diabetic gastroparesis. Clin Gastroenterol Hepatol. 2011;9:5-12; quiz e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 227] [Cited by in RCA: 205] [Article Influence: 13.7] [Reference Citation Analysis (1)] |
| 11. | Chedid V, Brandler J, Vijayvargiya P, Park SY, Szarka LA, Camilleri M. Characterization of Upper Gastrointestinal Symptoms, Gastric Motor Functions, and Associations in Patients with Diabetes at a Referral Center. Am J Gastroenterol. 2019;114:143-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 12. | Iyer PG, Borah BJ, Heien HC, Das A, Cooper GS, Chak A. Association of Barrett's esophagus with type II Diabetes Mellitus: results from a large population-based case-control study. Clin Gastroenterol Hepatol. 2013;11:1108-1114.e5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 48] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 13. | Pasricha PJ, Grover M, Yates KP, Abell TL, Bernard CE, Koch KL, McCallum RW, Sarosiek I, Kuo B, Bulat R, Chen J, Shulman RJ, Lee L, Tonascia J, Miriel LA, Hamilton F, Farrugia G, Parkman HP; National Institute of Diabetes and Digestive and Kidney Diseases/National Institutes of Health Gastroparesis Clinical Research Consortium. Functional Dyspepsia and Gastroparesis in Tertiary Care are Interchangeable Syndromes With Common Clinical and Pathologic Features. Gastroenterology. 2021;160:2006-2017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 205] [Article Influence: 41.0] [Reference Citation Analysis (1)] |
| 14. | Stanghellini V, Chan FK, Hasler WL, Malagelada JR, Suzuki H, Tack J, Talley NJ. Gastroduodenal Disorders. Gastroenterology. 2016;150:1380-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1306] [Cited by in RCA: 1051] [Article Influence: 105.1] [Reference Citation Analysis (2)] |
| 15. | Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology. 2016;S0016-5085(16)00223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1904] [Cited by in RCA: 1536] [Article Influence: 153.6] [Reference Citation Analysis (6)] |
| 16. | Lee IS, Wang H, Chae Y, Preissl H, Enck P. Functional neuroimaging studies in functional dyspepsia patients: a systematic review. Neurogastroenterol Motil. 2016;28:793-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 17. | Van Oudenhove L, Aziz Q. The role of psychosocial factors and psychiatric disorders in functional dyspepsia. Nat Rev Gastroenterol Hepatol. 2013;10:158-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 146] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 18. | Wauters L, Talley NJ, Walker MM, Tack J, Vanuytsel T. Novel concepts in the pathophysiology and treatment of functional dyspepsia. Gut. 2020;69:591-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 19. | Bouhnik Y, Alain S, Attar A, Flourié B, Raskine L, Sanson-Le Pors MJ, Rambaud JC. Bacterial populations contaminating the upper gut in patients with small intestinal bacterial overgrowth syndrome. Am J Gastroenterol. 1999;94:1327-1331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 122] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 20. | Tack J, Van den Houte K, Carbone F. Gastroduodenal motility disorders. Curr Opin Gastroenterol. 2018;34:428-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 21. | Zhang T, Zhang B, Ma X, Zhang J, Wei Y, Wang F, Tang X. Research trends in the field of the gut-brain interaction: Functional dyspepsia in the spotlight - An integrated bibliometric and science mapping approach. Front Neurosci. 2023;17:1109510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 22. | Mayer EA, Nance K, Chen S. The Gut-Brain Axis. Annu Rev Med. 2022;73:439-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 670] [Cited by in RCA: 524] [Article Influence: 131.0] [Reference Citation Analysis (0)] |
| 23. | Bharucha AE, Batey-Schaefer B, Cleary PA, Murray JA, Cowie C, Lorenzi G, Driscoll M, Harth J, Larkin M, Christofi M, Bayless M, Wimmergren N, Herman W, Whitehouse F, Jones K, Kruger D, Martin C, Ziegler G, Zinsmeister AR, Nathan DM; Diabetes Control and Complications Trial–Epidemiology of Diabetes Interventions and Complications Research Group. Delayed Gastric Emptying Is Associated With Early and Long-term Hyperglycemia in Type 1 Diabetes Mellitus. Gastroenterology. 2015;149:330-339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 111] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 24. | Almogbel RA, Alhussan FA, Alnasser SA, Algeffari MA. Prevalence and risk factors of gastroparesis-related symptoms among patients with type 2 diabetes. Int J Health Sci (Qassim). 2016;10:397-404. [PubMed] |
| 25. | Halland M, Bharucha AE. Relationship Between Control of Glycemia and Gastric Emptying Disturbances in Diabetes Mellitus. Clin Gastroenterol Hepatol. 2016;14:929-936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 67] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 26. | Frøkjær JB, Andersen LW, Brock C, Simrén M, Ljungberg M, Søfteland E, Dimcevski G, Yavarian Y, Gregersen H, Drewes AM. Altered brain microstructure assessed by diffusion tensor imaging in patients with diabetes and gastrointestinal symptoms. Diabetes Care. 2013;36:662-668. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 27. | Frøkjær JB, Graversen C, Brock C, Khodayari-Rostamabad A, Olesen SS, Hansen TM, Søfteland E, Simrén M, Drewes AM. Integrity of central nervous function in diabetes mellitus assessed by resting state EEG frequency analysis and source localization. J Diabetes Complications. 2017;31:400-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Lelic D, Brock C, Simrén M, Frøkjaer JB, Søfteland E, Dimcevski G, Gregersen H, Drewes AM. The brain networks encoding visceral sensation in patients with gastrointestinal symptoms due to diabetic neuropathy. Neurogastroenterol Motil. 2014;26:46-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 29. | Brock C, Søfteland E, Gunterberg V, Frøkjær JB, Lelic D, Brock B, Dimcevski G, Gregersen H, Simrén M, Drewes AM. Diabetic autonomic neuropathy affects symptom generation and brain-gut axis. Diabetes Care. 2013;36:3698-3705. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 30. | Tesfaye S, Selvarajah D, Gandhi R, Greig M, Shillo P, Fang F, Wilkinson ID. Diabetic peripheral neuropathy may not be as its name suggests: evidence from magnetic resonance imaging. Pain. 2016;157 Suppl 1:S72-S80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 31. | Cui W, Ma Y, Zhang L, Zhang L, Yao Q, Zhang J, Cheng Y, Zeng W, Liu Q, Liu F, Liang C. Neuregulin 1 improved gastric motility and reduced gastric inflammation by activating the α7nAChR through the cholinergic anti-inflammatory pathway in diabetic rats. Toxicol Appl Pharmacol. 2025;495:117205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 32. | Concepción Zavaleta MJ, Gonzáles Yovera JG, Moreno Marreros DM, Rafael Robles LDP, Palomino Taype KR, Soto Gálvez KN, Arriola Torres LF, Coronado Arroyo JC, Concepción Urteaga LA. Diabetic gastroenteropathy: An underdiagnosed complication. World J Diabetes. 2021;12:794-809. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 33] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (4)] |
| 33. | Vijayvargiya P, Jameie-Oskooei S, Camilleri M, Chedid V, Erwin PJ, Murad MH. Association between delayed gastric emptying and upper gastrointestinal symptoms: a systematic review and meta-analysis. Gut. 2019;68:804-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 152] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 34. | Frøkjær JB, Due Andersen S, Ejskjær N, Funch-Jensen P, Arendt-Nielsen L, Gregersen H, Drewes AM. Gut sensations in diabetic autonomic neuropathy. Pain. 2007;131:320-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 35. | Brock C, Graversen C, Frøkjaer JB, Søfteland E, Valeriani M, Drewes AM. Peripheral and central nervous contribution to gastrointestinal symptoms in diabetic patients with autonomic neuropathy. Eur J Pain. 2013;17:820-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 36. | Gandhi RA, Marques JL, Selvarajah D, Emery CJ, Tesfaye S. Painful diabetic neuropathy is associated with greater autonomic dysfunction than painless diabetic neuropathy. Diabetes Care. 2010;33:1585-1590. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 37. | Kazamel M, Dyck PJ. Sensory manifestations of diabetic neuropathies: anatomical and clinical correlations. Prosthet Orthot Int. 2015;39:7-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 38. | Lu KH, Cao J, Oleson S, Ward MP, Phillips RJ, Powley TL, Liu Z. Vagus nerve stimulation promotes gastric emptying by increasing pyloric opening measured with magnetic resonance imaging. Neurogastroenterol Motil. 2018;30:e13380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 39. | Schvarcz E, Palmér M, Aman J, Horowitz M, Stridsberg M, Berne C. Physiological hyperglycemia slows gastric emptying in normal subjects and patients with insulin-dependent diabetes mellitus. Gastroenterology. 1997;113:60-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 289] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 40. | Siraj ES, Homko C, Wilson LA, May P, Rao AD, Calles J, Farrugia G, Hasler WL, Koch KL, Nguyen L, Snape WJ, Abell TL, Sarosiek I, McCallum RW, Pasricha PJ, Clarke J, Tonascia J, Hamilton F, Parkman HP. Islet Cell Associated Autoantibodies and C-Peptide Levels in Patients with Diabetes and Symptoms of Gastroparesis. Front Endocrinol (Lausanne). 2018;9:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 41. | Vanormelingen C, Tack J, Andrews CN. Diabetic gastroparesis. Br Med Bull. 2013;105:213-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 42. | Stephens JW. The incretin system in the management of type 2 diabetes mellitus. Clin Med (Lond). 2010;10:491-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 43. | Meldgaard T, Keller J, Olesen AE, Olesen SS, Krogh K, Borre M, Farmer A, Brock B, Brock C, Drewes AM. Pathophysiology and management of diabetic gastroenteropathy. Therap Adv Gastroenterol. 2019;12:1756284819852047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 44. | Nguyen LA, Snape WJ Jr. Clinical presentation and pathophysiology of gastroparesis. Gastroenterol Clin North Am. 2015;44:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 45. | Aleppo G, Calhoun P, Foster NC, Maahs DM, Shah VN, Miller KM; T1D Exchange Clinic Network. Reported gastroparesis in adults with type 1 diabetes (T1D) from the T1D Exchange clinic registry. J Diabetes Complications. 2017;31:1669-1673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 46. | Fregonesi CE, Miranda-Neto MH, Molinari SL, Zanoni JN. Quantitative study of the myenteric plexus of the stomach of rats with streptozotocin-induced diabetes. Arq Neuropsiquiatr. 2001;59:50-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 38] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 47. | Guy RJ, Dawson JL, Garrett JR, Laws JW, Thomas PK, Sharma AK, Watkins PJ. Diabetic gastroparesis from autonomic neuropathy: surgical considerations and changes in vagus nerve morphology. J Neurol Neurosurg Psychiatry. 1984;47:686-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 77] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 48. | Smith B. Neuropathology of the oesophagus in diabetes mellitus. J Neurol Neurosurg Psychiatry. 1974;37:1151-1154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 60] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 49. | Sullivan A, Temperley L, Ruban A. Pathophysiology, Aetiology and Treatment of Gastroparesis. Dig Dis Sci. 2020;65:1615-1631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 50. | Yarandi SS, Srinivasan S. Diabetic gastrointestinal motility disorders and the role of enteric nervous system: current status and future directions. Neurogastroenterol Motil. 2014;26:611-624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 156] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 51. | Koçkar MC, Kayahan IK, Bavbek N. Diabetic gastroparesis in association with autonomic neuropathy and microvasculopathy. Acta Med Okayama. 2002;56:237-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 52. | Bernardi L, Spallone V, Stevens M, Hilsted J, Frontoni S, Pop-Busui R, Ziegler D, Kempler P, Freeman R, Low P, Tesfaye S, Valensi P; Toronto Consensus Panel on Diabetic Neuropathy. Methods of investigation for cardiac autonomic dysfunction in human research studies. Diabetes Metab Res Rev. 2011;27:654-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 132] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 53. | Bharucha AE, Camilleri M, Low PA, Zinsmeister AR. Autonomic dysfunction in gastrointestinal motility disorders. Gut. 1993;34:397-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 107] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 54. | Freeman R. Diabetic autonomic neuropathy. Handb Clin Neurol. 2014;126:63-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 94] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 55. | Wifi MN, El-Sherbiny M, Mohamed RS, Kandeel A, Rizk SE. Clinical insights into diabetic gastroparesis: gastric scintigraphy-based diagnosis and treatment outcomes. BMC Gastroenterol. 2025;25:640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 56. | AlOlaiwi LA, AlHarbi TJ, Tourkmani AM. Prevalence of cardiovascular autonomic neuropathy and gastroparesis symptoms among patients with type 2 diabetes who attend a primary health care center. PLoS One. 2018;13:e0209500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 57. | Schwartz TW. Pancreatic polypeptide: a hormone under vagal control. Gastroenterology. 1983;85:1411-1425. [PubMed] [DOI] [Full Text] |
| 58. | Glasbrenner B, Bruckel J, Gritzmann R, Adler G. Cephalic phase of pancreatic polypeptide release: a valid test of autonomic neuropathy in diabetics? Diabetes Res Clin Pract. 1995;30:117-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 59. | Glasbrenner B, Dominguez-Munoz E, Riepl RL, Vetsi A, Malfertheiner P. Cholecystokinin and pancreatic polypeptide release in diabetic patients with and without autonomic neuropathy. Dig Dis Sci. 1995;40:406-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 60. | Kennedy FP, Go VL, Cryer PE, Bolli GB, Gerich JE. Subnormal pancreatic polypeptide and epinephrine responses to insulin-induced hypoglycemia identify patients with insulin-dependent diabetes mellitus predisposed to develop overt autonomic neuropathy. Ann Intern Med. 1988;108:54-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 61. | Buysschaert M, Donckier J, Dive A, Ketelslegers JM, Lambert AE. Gastric acid and pancreatic polypeptide responses to sham feeding are impaired in diabetic subjects with autonomic neuropathy. Diabetes. 1985;34:1181-1185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 52] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 62. | Kumar M, Chapman A, Javed S, Alam U, Malik RA, Azmi S. The Investigation and Treatment of Diabetic Gastroparesis. Clin Ther. 2018;40:850-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 63. | Borgoño CA, Zinman B. Insulins: past, present, and future. Endocrinol Metab Clin North Am. 2012;41:1-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 64. | Quianzon CC, Cheikh I. History of insulin. J Community Hosp Intern Med Perspect. 2012;2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 65. | Jalleh R, Marathe CS, Rayner CK, Jones KL, Horowitz M. Diabetic Gastroparesis and Glycaemic Control. Curr Diab Rep. 2019;19:153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 66. | Ohkuma T, Iwase M, Fujii H, Ide H, Komorita Y, Yoshinari M, Oku Y, Higashi T, Nakamura U, Kitazono T. Defecation frequency and glycemic control in patients with diabetes: The Fukuoka Diabetes Registry. J Diabetes Complications. 2021;35:107751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 67. | Gaddipati KV, Simonian HP, Kresge KM, Boden GH, Parkman HP. Abnormal ghrelin and pancreatic polypeptide responses in gastroparesis. Dig Dis Sci. 2006;51:1339-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 68. | Sanger GJ, Lee K. Hormones of the gut-brain axis as targets for the treatment of upper gastrointestinal disorders. Nat Rev Drug Discov. 2008;7:241-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 84] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 69. | Lan J, Wang K, Chen G, Cao G, Yang C. Effects of inulin and isomalto-oligosaccharide on diphenoxylate-induced constipation, gastrointestinal motility-related hormones, short-chain fatty acids, and the intestinal flora in rats. Food Funct. 2020;11:9216-9225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 70. | Johnson CD, Barlow-Anacker AJ, Pierre JF, Touw K, Erickson CS, Furness JB, Epstein ML, Gosain A. Deletion of choline acetyltransferase in enteric neurons results in postnatal intestinal dysmotility and dysbiosis. FASEB J. 2018;32:4744-4752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 71. | Van den Houte K, Scarpellini E, Verbeure W, Mori H, Schol J, Masuy I, Carbone F, Tack J. The Role of GI Peptides in Functional Dyspepsia and Gastroparesis: A Systematic Review. Front Psychiatry. 2020;11:172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 72. | Zhao X, Wang H, Zhang Z, Jin H, Gong Y. Effects of ethyl acetate extract of Salsola collina on brain-gut peptides and interstitial cells of gastric Cajal in rats with diabetic gastroparesis. Iran J Basic Med Sci. 2020;23:1218-1224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 73. | Chen W, Chen Q, Huang J, Shen X, Zhang L, Jiang G, Wu T, Wang F, Cheng X. Huanglian-banxia promotes gastric motility of diabetic rats by modulating brain-gut neurotransmitters through MAPK signaling pathway. Neurogastroenterol Motil. 2024;36:e14779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 74. | Wang B, Zeng KW, Hong ZF, Ti GX, Wang LY, Lu P, Liu Z. Banxia Xiexin Decoction () Treats Diabetic Gastroparesis through PLC-IP(3)-Ca(2+)/NO-cGMP-PKG Signal Pathway. Chin J Integr Med. 2020;26:833-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 75. | Rivera LR, Poole DP, Thacker M, Furness JB. The involvement of nitric oxide synthase neurons in enteric neuropathies. Neurogastroenterol Motil. 2011;23:980-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 141] [Article Influence: 9.4] [Reference Citation Analysis (12)] |
| 76. | Socała K, Doboszewska U, Szopa A, Serefko A, Włodarczyk M, Zielińska A, Poleszak E, Fichna J, Wlaź P. The role of microbiota-gut-brain axis in neuropsychiatric and neurological disorders. Pharmacol Res. 2021;172:105840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 701] [Cited by in RCA: 561] [Article Influence: 112.2] [Reference Citation Analysis (0)] |
| 77. | Cuomo R, Vandaele P, Coulie B, Peeters T, Depoortere I, Janssens J, Tack J. Influence of motilin on gastric fundus tone and on meal-induced satiety in man: role of cholinergic pathways. Am J Gastroenterol. 2006;101:804-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 78. | Kitazawa T, Kaiya H. Motilin Comparative Study: Structure, Distribution, Receptors, and Gastrointestinal Motility. Front Endocrinol (Lausanne). 2021;12:700884. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 79. | Tack J, Deloose E, Ang D, Scarpellini E, Vanuytsel T, Van Oudenhove L, Depoortere I. Motilin-induced gastric contractions signal hunger in man. Gut. 2016;65:214-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 80. | Singaram K, Gold-Smith FD, Petrov MS. Motilin: a panoply of communications between the gut, brain, and pancreas. Expert Rev Gastroenterol Hepatol. 2020;14:103-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 81. | Smieszek SP, Carlin JL, Xiao C, Birznieks G, Polymeropoulos CM, Polymeropoulos MH. Enrichment of Motilin Receptor Loss-of-Function Variants in Gastroparesis. Clin Transl Gastroenterol. 2022;13:e00474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 82. | Sanger GJ, Broad J, Callaghan B, Furness JB. Ghrelin and Motilin Control Systems in GI Physiology and Therapeutics. Handb Exp Pharmacol. 2017;239:379-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 83. | Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. 1999;402:656-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6602] [Cited by in RCA: 5840] [Article Influence: 216.3] [Reference Citation Analysis (0)] |
| 84. | Kitazawa T, Kaiya H. Regulation of Gastrointestinal Motility by Motilin and Ghrelin in Vertebrates. Front Endocrinol (Lausanne). 2019;10:278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 85. | Tack J, Verbeure W, Mori H, Schol J, Van den Houte K, Huang IH, Balsiger L, Broeders B, Colomier E, Scarpellini E, Carbone F. The gastrointestinal tract in hunger and satiety signalling. United European Gastroenterol J. 2021;9:727-734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 86. | Kim JN, Kim BJ. The Mechanism of Action of Ghrelin and Motilin in the Pacemaker Potentials of Interstitial Cells of Cajal from the Murine Small Intestine. Mol Cells. 2019;42:470-479. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 87. | Kojima M, Kangawa K. Ghrelin: structure and function. Physiol Rev. 2005;85:495-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1273] [Cited by in RCA: 1126] [Article Influence: 53.6] [Reference Citation Analysis (0)] |
| 88. | Mikami T, Ito K, Diaz-Tartera HO, Hellström PM, Mochiki E, Takemi S, Tanaka T, Tsuda S, Jogahara T, Sakata I, Sakai T. Study of termination of postprandial gastric contractions in humans, dogs and Suncus murinus: role of motilin- and ghrelin-induced strong contraction. Acta Physiol (Oxf). 2018;222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 89. | Chen CY, Fujimiya M, Laviano A, Chang FY, Lin HC, Lee SD. Modulation of ingestive behavior and gastrointestinal motility by ghrelin in diabetic animals and humans. J Chin Med Assoc. 2010;73:225-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 90. | Murray CD, Martin NM, Patterson M, Taylor SA, Ghatei MA, Kamm MA, Johnston C, Bloom SR, Emmanuel AV. Ghrelin enhances gastric emptying in diabetic gastroparesis: a double blind, placebo controlled, crossover study. Gut. 2005;54:1693-1698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 264] [Cited by in RCA: 241] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 91. | Linan-Rico A, Ochoa-Cortes F, Beyder A, Soghomonyan S, Zuleta-Alarcon A, Coppola V, Christofi FL. Mechanosensory Signaling in Enterochromaffin Cells and 5-HT Release: Potential Implications for Gut Inflammation. Front Neurosci. 2016;10:564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 90] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 92. | Mawe GM, Hoffman JM. Serotonin signalling in the gut--functions, dysfunctions and therapeutic targets. Nat Rev Gastroenterol Hepatol. 2013;10:473-486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 997] [Cited by in RCA: 852] [Article Influence: 65.5] [Reference Citation Analysis (2)] |
| 93. | Chedid V, Brandler J, Arndt K, Vijayvargiya P, Wang XJ, Burton D, Harmsen WS, Siegelman J, Chen C, Chen Y, Almansa C, Dukes G, Camilleri M. Randomised study: effects of the 5-HT(4) receptor agonist felcisetrag vs placebo on gut transit in patients with gastroparesis. Aliment Pharmacol Ther. 2021;53:1010-1020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 94. | Liguz-Lecznar M, Dobrzanski G, Kossut M. Somatostatin and Somatostatin-Containing Interneurons-From Plasticity to Pathology. Biomolecules. 2022;12:312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 95. | Schubert ML, Rehfeld JF. Gastric Peptides-Gastrin and Somatostatin. Compr Physiol. 2019;10:197-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 96. | Shamsi BH, Chatoo M, Xu XK, Xu X, Chen XQ. Versatile Functions of Somatostatin and Somatostatin Receptors in the Gastrointestinal System. Front Endocrinol (Lausanne). 2021;12:652363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 97. | Herranz R. Cholecystokinin antagonists: pharmacological and therapeutic potential. Med Res Rev. 2003;23:559-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 125] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 98. | Agirman G, Yu KB, Hsiao EY. Signaling inflammation across the gut-brain axis. Science. 2021;374:1087-1092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 701] [Cited by in RCA: 570] [Article Influence: 114.0] [Reference Citation Analysis (0)] |
| 99. | Martin CR, Osadchiy V, Kalani A, Mayer EA. The Brain-Gut-Microbiome Axis. Cell Mol Gastroenterol Hepatol. 2018;6:133-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1070] [Cited by in RCA: 897] [Article Influence: 112.1] [Reference Citation Analysis (0)] |
| 100. | Scheuermann DW, Stach W, Timmermans JP. Topography, architecture and structure of the plexus submucosus externus (Schabadasch) of the porcine small intestine in scanning electron microscopy. Acta Anat (Basel). 1987;129:105-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 101. | Horváth VJ, Putz Z, Izbéki F, Körei AE, Gerő L, Lengyel C, Kempler P, Várkonyi T. Diabetes-related dysfunction of the small intestine and the colon: focus on motility. Curr Diab Rep. 2015;15:94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 44] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 102. | Watkins CC, Sawa A, Jaffrey S, Blackshaw S, Barrow RK, Snyder SH, Ferris CD. Insulin restores neuronal nitric oxide synthase expression and function that is lost in diabetic gastropathy. J Clin Invest. 2000;106:373-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 171] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 103. | Baker C, Ahmed M, Cheng K, Arciero E, Bhave S, Ho WLN, Goldstein AM, Hotta R. Hypoganglionosis in the gastric antrum causes delayed gastric emptying. Neurogastroenterol Motil. 2020;32:e13766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 104. | Grover M, Bernard CE, Pasricha PJ, Lurken MS, Faussone-Pellegrini MS, Smyrk TC, Parkman HP, Abell TL, Snape WJ, Hasler WL, McCallum RW, Nguyen L, Koch KL, Calles J, Lee L, Tonascia J, Ünalp-Arida A, Hamilton FA, Farrugia G; NIDDK Gastroparesis Clinical Research Consortium (GpCRC). Clinical-histological associations in gastroparesis: results from the Gastroparesis Clinical Research Consortium. Neurogastroenterol Motil. 2012;24:531-539, e249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 149] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 105. | Angeli TR, Cheng LK, Du P, Wang TH, Bernard CE, Vannucchi MG, Faussone-Pellegrini MS, Lahr C, Vather R, Windsor JA, Farrugia G, Abell TL, O'Grady G. Loss of Interstitial Cells of Cajal and Patterns of Gastric Dysrhythmia in Patients With Chronic Unexplained Nausea and Vomiting. Gastroenterology. 2015;149:56-66.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 193] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 106. | Pasricha PJ, Pehlivanov ND, Gomez G, Vittal H, Lurken MS, Farrugia G. Changes in the gastric enteric nervous system and muscle: a case report on two patients with diabetic gastroparesis. BMC Gastroenterol. 2008;8:21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 66] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 107. | Iwasaki H, Kajimura M, Osawa S, Kanaoka S, Furuta T, Ikuma M, Hishida A. A deficiency of gastric interstitial cells of Cajal accompanied by decreased expression of neuronal nitric oxide synthase and substance P in patients with type 2 diabetes mellitus. J Gastroenterol. 2006;41:1076-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 148] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 108. | Gangula PR, Maner WL, Micci MA, Garfield RE, Pasricha PJ. Diabetes induces sex-dependent changes in neuronal nitric oxide synthase dimerization and function in the rat gastric antrum. Am J Physiol Gastrointest Liver Physiol. 2007;292:G725-G733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 86] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 109. | Horváth VJ, Vittal H, Lörincz A, Chen H, Almeida-Porada G, Redelman D, Ordög T. Reduced stem cell factor links smooth myopathy and loss of interstitial cells of cajal in murine diabetic gastroparesis. Gastroenterology. 2006;130:759-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 180] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 110. | Wood JD. Enteric neurophysiology. Am J Physiol. 1984;247:G585-G598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 111. | Li H, Cao W, Zhang XB, Zhang XX, Gu C, Gu LM, Pan CY, Tian YZ, Lu M. Atractylenolide1 alleviates gastroparesis in diabetic rats by activating the stem cell factor/ckit signaling pathway. Mol Med Rep. 2021;24:691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (6)] |
| 112. | Mearin F, Camilleri M, Malagelada JR. Pyloric dysfunction in diabetics with recurrent nausea and vomiting. Gastroenterology. 1986;90:1919-1925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 293] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 113. | Gangula PR, Mukhopadhyay S, Ravella K, Cai S, Channon KM, Garfield RE, Pasricha PJ. Tetrahydrobiopterin (BH4), a cofactor for nNOS, restores gastric emptying and nNOS expression in female diabetic rats. Am J Physiol Gastrointest Liver Physiol. 2010;298:G692-G699. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 114. | Feldman M, Corbett DB, Ramsey EJ, Walsh JH, Richardson CT. Abnormal gastric function in longstanding, insulin-dependent diabetic patients. Gastroenterology. 1979;77:12-17. [PubMed] [DOI] [Full Text] |
| 115. | Smiley R, McCallum R, Showkat Ali M. Decreased Level of Neuropeptide Y Is Associated With Gastroparesis in Male Diabetic Rats. Gastroenterology Res. 2020;13:246-252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 116. | Jehangir A, Abdallah RT, Parkman HP. Characterizing Abdominal Pain in Patients With Gastroparesis Into Neuropathic and Nociceptive Components. J Clin Gastroenterol. 2019;53:427-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 117. | Zhou B, Yuan Y, Zhang S, Guo C, Li X, Li G, Xiong W, Zeng Z. Intestinal Flora and Disease Mutually Shape the Regional Immune System in the Intestinal Tract. Front Immunol. 2020;11:575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 344] [Cited by in RCA: 285] [Article Influence: 47.5] [Reference Citation Analysis (0)] |
| 118. | Iatcu CO, Steen A, Covasa M. Gut Microbiota and Complications of Type-2 Diabetes. Nutrients. 2021;14:166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 365] [Cited by in RCA: 268] [Article Influence: 53.6] [Reference Citation Analysis (3)] |
| 119. | Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol. 2021;19:55-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3956] [Cited by in RCA: 3236] [Article Influence: 647.2] [Reference Citation Analysis (2)] |
| 120. | Caricilli AM, Saad MJ. The role of gut microbiota on insulin resistance. Nutrients. 2013;5:829-851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 196] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 121. | Kempler P, Várkonyi T, Körei AE, Horváth VJ. Gastrointestinal autonomic neuropathy in diabetes: the unattended borderline between diabetology and gastroenterology. Diabetologia. 2016;59:401-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 122. | Grasset E, Puel A, Charpentier J, Collet X, Christensen JE, Tercé F, Burcelin R. A Specific Gut Microbiota Dysbiosis of Type 2 Diabetic Mice Induces GLP-1 Resistance through an Enteric NO-Dependent and Gut-Brain Axis Mechanism. Cell Metab. 2017;25:1075-1090.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 199] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 123. | Varma R, Chakraborty S, Burton DD, Bailey KR, Bharucha AE. Small intestinal bacterial overgrowth in diabetic gastroenteropathy. Neurogastroenterol Motil. 2024;36:e14798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 124. | Feng X, Li XQ. The prevalence of small intestinal bacterial overgrowth in diabetes mellitus: a systematic review and meta-analysis. Aging (Albany NY). 2022;14:975-988. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 125. | Du Y, Neng Q, Li Y, Kang Y, Guo L, Huang X, Chen M, Yang F, Hong J, Zhou S, Zhao J, Yu F, Su H, Kong X. Gastrointestinal Autonomic Neuropathy Exacerbates Gut Microbiota Dysbiosis in Adult Patients With Type 2 Diabetes Mellitus. Front Cell Infect Microbiol. 2021;11:804733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 126. | Larsen N, Vogensen FK, van den Berg FW, Nielsen DS, Andreasen AS, Pedersen BK, Al-Soud WA, Sørensen SJ, Hansen LH, Jakobsen M. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS One. 2010;5:e9085. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2434] [Cited by in RCA: 2129] [Article Influence: 133.1] [Reference Citation Analysis (2)] |
| 127. | Salamon D, Sroka-Oleksiak A, Kapusta P, Szopa M, Mrozińska S, Ludwig-Słomczyńska AH, Wołkow PP, Bulanda M, Klupa T, Małecki MT, Gosiewski T. Characteristics of gut microbiota in adult patients with type 1 and type 2 diabetes based on nextgeneration sequencing of the 16S rRNA gene fragment. Pol Arch Intern Med. 2018;128:336-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 128. | Vallianou NG, Stratigou T, Tsagarakis S. Microbiome and diabetes: Where are we now? Diabetes Res Clin Pract. 2018;146:111-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 91] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 129. | Neag MA, Craciun AE, Inceu AI, Burlacu DE, Craciun CI, Buzoianu AD. Short-Chain Fatty Acids as Bacterial Enterocytes and Therapeutic Target in Diabetes Mellitus Type 2. Biomedicines. 2022;11:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 130. | R Muralitharan R, Zheng T, Dinakis E, Xie L, Barbaro-Wahl A, Jama HA, Nakai M, Paterson M, Leung KC, McArdle Z, Mirabito Colafella K, Johnson C, Qin W, Salimova E, Bitto NJ, Kaparakis-Liaskos M, Kaye DM, O'Donnell JA, Mackay CR, Marques FZ. Gut Microbiota Metabolites Sensed by Host GPR41/43 Protect Against Hypertension. Circ Res. 2025;136:e20-e33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 131. | Wu T, Zhang Y, Li W, Zhao Y, Long H, Muhindo EM, Liu R, Sui W, Li Q, Zhang M. Lactobacillus rhamnosus LRa05 Ameliorate Hyperglycemia through a Regulating Glucagon-Mediated Signaling Pathway and Gut Microbiota in Type 2 Diabetic Mice. J Agric Food Chem. 2021;69:8797-8806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 132. | Visconti A, Le Roy CI, Rosa F, Rossi N, Martin TC, Mohney RP, Li W, de Rinaldis E, Bell JT, Venter JC, Nelson KE, Spector TD, Falchi M. Interplay between the human gut microbiome and host metabolism. Nat Commun. 2019;10:4505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 655] [Cited by in RCA: 535] [Article Influence: 76.4] [Reference Citation Analysis (0)] |
| 133. | Yang J, Yang X, Wu G, Huang F, Shi X, Wei W, Zhang Y, Zhang H, Cheng L, Yu L, Shang J, Lv Y, Wang X, Zhai R, Li P, Cui B, Fang Y, Deng X, Tang S, Wang L, Yuan Q, Zhao L, Zhang F, Zhang C, Yuan H. Gut microbiota modulate distal symmetric polyneuropathy in patients with diabetes. Cell Metab. 2023;35:1548-1562.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 70] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 134. | Sanchez-Alcoholado L, Castellano-Castillo D, Jordán-Martínez L, Moreno-Indias I, Cardila-Cruz P, Elena D, Muñoz-Garcia AJ, Queipo-Ortuño MI, Jimenez-Navarro M. Role of Gut Microbiota on Cardio-Metabolic Parameters and Immunity in Coronary Artery Disease Patients with and without Type-2 Diabetes Mellitus. Front Microbiol. 2017;8:1936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 86] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 135. | Han Q, Wang J, Li W, Chen ZJ, Du Y. Androgen-induced gut dysbiosis disrupts glucolipid metabolism and endocrinal functions in polycystic ovary syndrome. Microbiome. 2021;9:101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 136. | Zhou Z, Sun B, Yu D, Zhu C. Gut Microbiota: An Important Player in Type 2 Diabetes Mellitus. Front Cell Infect Microbiol. 2022;12:834485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 197] [Article Influence: 49.3] [Reference Citation Analysis (3)] |
| 137. | Travagli RA, Anselmi L. Vagal neurocircuitry and its influence on gastric motility. Nat Rev Gastroenterol Hepatol. 2016;13:389-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 298] [Cited by in RCA: 241] [Article Influence: 24.1] [Reference Citation Analysis (1)] |
| 138. | Cryan JF, O'Riordan KJ, Cowan CSM, Sandhu KV, Bastiaanssen TFS, Boehme M, Codagnone MG, Cussotto S, Fulling C, Golubeva AV, Guzzetta KE, Jaggar M, Long-Smith CM, Lyte JM, Martin JA, Molinero-Perez A, Moloney G, Morelli E, Morillas E, O'Connor R, Cruz-Pereira JS, Peterson VL, Rea K, Ritz NL, Sherwin E, Spichak S, Teichman EM, van de Wouw M, Ventura-Silva AP, Wallace-Fitzsimons SE, Hyland N, Clarke G, Dinan TG. The Microbiota-Gut-Brain Axis. Physiol Rev. 2019;99:1877-2013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4131] [Cited by in RCA: 3400] [Article Influence: 485.7] [Reference Citation Analysis (1)] |
| 139. | Farmer AD, Pedersen AG, Brock B, Jakobsen PE, Karmisholt J, Mohammed SD, Scott SM, Drewes AM, Brock C. Type 1 diabetic patients with peripheral neuropathy have pan-enteric prolongation of gastrointestinal transit times and an altered caecal pH profile. Diabetologia. 2017;60:709-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 140. | Boychuk CR, Smith KC, Smith BN. Functional and molecular plasticity of γ and α1 GABA(A) receptor subunits in the dorsal motor nucleus of the vagus after experimentally induced diabetes. J Neurophysiol. 2017;118:2833-2841. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 141. | Guo C, Quobatari A, Shangguan Y, Hong S, Wiley JW. Diabetic autonomic neuropathy: evidence for apoptosis in situ in the rat. Neurogastroenterol Motil. 2004;16:335-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 55] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 142. | Krieger JP, Arnold M, Pettersen KG, Lossel P, Langhans W, Lee SJ. Knockdown of GLP-1 Receptors in Vagal Afferents Affects Normal Food Intake and Glycemia. Diabetes. 2016;65:34-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 211] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 143. | Labouesse MA, Stadlbauer U, Weber E, Arnold M, Langhans W, Pacheco-López G. Vagal afferents mediate early satiation and prevent flavour avoidance learning in response to intraperitoneally infused exendin-4. J Neuroendocrinol. 2012;24:1505-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 144. | Chandrasekharan B, Anitha M, Blatt R, Shahnavaz N, Kooby D, Staley C, Mwangi S, Jones DP, Sitaraman SV, Srinivasan S. Colonic motor dysfunction in human diabetes is associated with enteric neuronal loss and increased oxidative stress. Neurogastroenterol Motil. 2011;23:131-138, e26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 149] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 145. | Reigstad CS, Salmonson CE, Rainey JF 3rd, Szurszewski JH, Linden DR, Sonnenburg JL, Farrugia G, Kashyap PC. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015;29:1395-1403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1121] [Cited by in RCA: 978] [Article Influence: 88.9] [Reference Citation Analysis (0)] |
| 146. | Cipriani G, Gibbons SJ, Verhulst PJ, Choi KM, Eisenman ST, Hein SS, Ordog T, Linden DR, Szurszewski JH, Farrugia G. Diabetic Csf1(op/op) mice lacking macrophages are protected against the development of delayed gastric emptying. Cell Mol Gastroenterol Hepatol. 2016;2:40-47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 44] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 147. | Dillon BR, Ang L, Pop-Busui R. Spectrum of Diabetic Neuropathy: New Insights in Diagnosis and Treatment. Annu Rev Med. 2024;75:293-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 148. | Yu B, Sun M, Wang Z, Zhu B, Xue J, Yang W, Gao X, Zhi M, Cao J, Zhao J, Zhao X, Liu W, Wang F, Li T. Effects of Stimulating Local and Distal Acupoints on Diabetic Gastroparesis: A New Insight in Revealing Acupuncture Therapeutics. Am J Chin Med. 2021;49:1151-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (2)] |
| 149. | Anitha M, Vijay-Kumar M, Sitaraman SV, Gewirtz AT, Srinivasan S. Gut microbial products regulate murine gastrointestinal motility via Toll-like receptor 4 signaling. Gastroenterology. 2012;143:1006-16.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 348] [Cited by in RCA: 308] [Article Influence: 22.0] [Reference Citation Analysis (1)] |
| 150. | Xu S, Liang S, Pei Y, Wang R, Zhang Y, Xu Y, Huang B, Li H, Li J, Tan B, Cao H, Guo S. TRPV1 Dysfunction Impairs Gastric Nitrergic Neuromuscular Relaxation in High-Fat Diet-Induced Diabetic Gastroparesis Mice. Am J Pathol. 2023;193:548-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 151. | Hayashi Y, Toyomasu Y, Saravanaperumal SA, Bardsley MR, Smestad JA, Lorincz A, Eisenman ST, Cipriani G, Nelson Holte MH, Al Khazal FJ, Syed SA, Gajdos GB, Choi KM, Stoltz GJ, Miller KE, Kendrick ML, Rubin BP, Gibbons SJ, Bharucha AE, Linden DR, Maher LJ 3rd, Farrugia G, Ordog T. Hyperglycemia Increases Interstitial Cells of Cajal via MAPK1 and MAPK3 Signaling to ETV1 and KIT, Leading to Rapid Gastric Emptying. Gastroenterology. 2017;153:521-535.e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 152. | Choi KM, Kashyap PC, Dutta N, Stoltz GJ, Ordog T, Shea Donohue T, Bauer AJ, Linden DR, Szurszewski JH, Gibbons SJ, Farrugia G. CD206-positive M2 macrophages that express heme oxygenase-1 protect against diabetic gastroparesis in mice. Gastroenterology. 2010;138:2399-2409, 2409.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 186] [Article Influence: 11.6] [Reference Citation Analysis (1)] |
| 153. | Gibbons SJ, Verhulst PJ, Bharucha A, Farrugia G. Review article: carbon monoxide in gastrointestinal physiology and its potential in therapeutics. Aliment Pharmacol Ther. 2013;38:689-702. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 154. | Choi KM, Gibbons SJ, Nguyen TV, Stoltz GJ, Lurken MS, Ordog T, Szurszewski JH, Farrugia G. Heme oxygenase-1 protects interstitial cells of Cajal from oxidative stress and reverses diabetic gastroparesis. Gastroenterology. 2008;135:2055-2064, 2064.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 195] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 155. | Bharucha AE, Daley SL, Low PA, Gibbons SJ, Choi KM, Camilleri M, Saw JJ, Farrugia G, Zinsmeister AR. Effects of hemin on heme oxygenase-1, gastric emptying, and symptoms in diabetic gastroparesis. Neurogastroenterol Motil. 2016;28:1731-1740. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 156. | Berntorp K, Frid A, Alm R, Fredrikson GN, Sjöberg K, Ohlsson B. Antibodies against gonadotropin-releasing hormone (GnRH) in patients with diabetes mellitus is associated with lower body weight and autonomic neuropathy. BMC Res Notes. 2013;6:329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 157. | Cipriani G, Gibbons SJ, Miller KE, Yang DS, Terhaar ML, Eisenman ST, Ördög T, Linden DR, Gajdos GB, Szurszewski JH, Farrugia G. Change in Populations of Macrophages Promotes Development of Delayed Gastric Emptying in Mice. Gastroenterology. 2018;154:2122-2136.e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 158. | Kashyap PC, Choi KM, Dutta N, Linden DR, Szurszewski JH, Gibbons SJ, Farrugia G. Carbon monoxide reverses diabetic gastroparesis in NOD mice. Am J Physiol Gastrointest Liver Physiol. 2010;298:G1013-G1019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 159. | Stavely R, Ott LC, Rashidi N, Sakkal S, Nurgali K. The Oxidative Stress and Nervous Distress Connection in Gastrointestinal Disorders. Biomolecules. 2023;13:1586. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 160. | Huerta TS, Chen AC, Chaudhry S, Tynan A, Morgan T, Park K, Adamovich-Zeitlin R, Haider B, Li JH, Nagpal M, Zanos S, Pavlov VA, Brines M, Zanos TP, Chavan SS, Tracey KJ, Chang EH. Neural representation of cytokines by vagal sensory neurons. Nat Commun. 2025;16:3840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 161. | Ingrosso MR, Camilleri M, Tack J, Ianiro G, Black CJ, Ford AC. Efficacy and Safety of Drugs for Gastroparesis: Systematic Review and Network Meta-analysis. Gastroenterology. 2023;164:642-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 162. | Shakhatreh M, Jehangir A, Malik Z, Parkman HP. Metoclopramide for the treatment of diabetic gastroparesis. Expert Rev Gastroenterol Hepatol. 2019;13:711-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 163. | Lee A, Kuo B. Metoclopramide in the treatment of diabetic gastroparesis. Expert Rev Endocrinol Metab. 2010;5:653-662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 72] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 164. | Tonini M, Cipollina L, Poluzzi E, Crema F, Corazza GR, De Ponti F. Review article: clinical implications of enteric and central D2 receptor blockade by antidopaminergic gastrointestinal prokinetics. Aliment Pharmacol Ther. 2004;19:379-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 206] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 165. | Flake ZA, Linn BS, Hornecker JR. Practical selection of antiemetics in the ambulatory setting. Am Fam Physician. 2015;91:293-296. [PubMed] |
| 166. | Careyva B, Stello B. Diabetes Mellitus: Management of Gastrointestinal Complications. Am Fam Physician. 2016;94:980-986. [PubMed] |
| 167. | Emorinken A, Agbadaola OR. Metoclopramide-induced Acute Dystonia Misdiagnosed as an Epileptic Seizure in a Lupus Patient. J Epilepsy Res. 2021;11:156-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 168. | Abd Ellah NH, Ahmed EA, Abd-Ellatief RB, Ali MF, Zahran AM, Hetta HF. Metoclopramide nanoparticles modulate immune response in a diabetic rat model: association with regulatory T cells and proinflammatory cytokines. Int J Nanomedicine. 2019;14:2383-2395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 169. | Reddymasu SC, Soykan I, McCallum RW. Domperidone: review of pharmacology and clinical applications in gastroenterology. Am J Gastroenterol. 2007;102:2036-2045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 189] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 170. | Sanger GJ, Andrews PLR. A History of Drug Discovery for Treatment of Nausea and Vomiting and the Implications for Future Research. Front Pharmacol. 2018;9:913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 171. | Woods K, Gajendran M, Gonzalez Z, Bustamante-Bernal M, Sarosiek I, Espino K, Waterhouse N, Siddiqui T, McCallum R. Cardiac safety and clinical efficacy of high-dose domperidone for long-term treatment of gastroparesis symptoms. J Investig Med. 2022;70:1225-1232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 172. | Sarosiek I, Van Natta M, Parkman HP, Abell T, Koch KL, Kuo B, Shulman RJ, Farrugia G, Grover M, Hamilton FA, Pasricha PJ, Yates KP, Miriel L, Wilson L, Yamada G, Tonascia J, McCallum RW; National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) Gastroparesis Clinical Research Consortium (GpCRC). Effect of Domperidone Therapy on Gastroparesis Symptoms: Results of a Dynamic Cohort Study by NIDDK Gastroparesis Consortium. Clin Gastroenterol Hepatol. 2022;20:e452-e464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 173. | Kawamura T, Matsuura B, Miyake T, Abe M, Ikeda Y, Hiasa Y. Effects of Motilin Receptor Agonists and Ghrelin in Human motilin receptor Transgenic Mice. Int J Mol Sci. 2019;20:1521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 174. | Catnach SM, Fairclough PD. Erythromycin and the gut. Gut. 1992;33:397-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 78] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 175. | MacLaren R, Kiser TH, Fish DN, Wischmeyer PE. Erythromycin vs metoclopramide for facilitating gastric emptying and tolerance to intragastric nutrition in critically ill patients. JPEN J Parenter Enteral Nutr. 2008;32:412-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 65] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 176. | Potter TG, Snider KR. Azithromycin for the treatment of gastroparesis. Ann Pharmacother. 2013;47:411-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 177. | Chini P, Toskes PP, Waseem S, Hou W, McDonald R, Moshiree B. Effect of azithromycin on small bowel motility in patients with gastrointestinal dysmotility. Scand J Gastroenterol. 2012;47:422-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 178. | Hong SW, Chun J, Kim J, Lee J, Lee HJ, Chung H, Cho SJ, Im JP, Kim SG, Kim JS. Efficacy and Safety of Ghrelin Agonists in Patients with Diabetic Gastroparesis: A Systematic Review and Meta-Analysis. Gut Liver. 2020;14:589-600. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 179. | Petri M, Singh I, Baker C, Underkofler C, Rasouli N. Diabetic gastroparesis: An overview of pathogenesis, clinical presentation and novel therapies, with a focus on ghrelin receptor agonists. J Diabetes Complications. 2021;35:107733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 180. | James J, Mair S, Doll W, Sandefer E, Wurtman D, Maurer A, Deane AM, Harris MS. The effects of ulimorelin, a ghrelin agonist, on liquid gastric emptying and colonic transit in humans. Neurogastroenterol Motil. 2020;32:e13784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 181. | Shin A, Camilleri M, Busciglio I, Burton D, Smith SA, Vella A, Ryks M, Rhoten D, Zinsmeister AR. The ghrelin agonist RM-131 accelerates gastric emptying of solids and reduces symptoms in patients with type 1 diabetes mellitus. Clin Gastroenterol Hepatol. 2013;11:1453-1459.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 84] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 182. | Camilleri M, McCallum RW, Tack J, Spence SC, Gottesdiener K, Fiedorek FT. Efficacy and Safety of Relamorelin in Diabetics With Symptoms of Gastroparesis: A Randomized, Placebo-Controlled Study. Gastroenterology. 2017;153:1240-1250.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 112] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 183. | Akrab SNA, Al Gawhary NE, Shafik AN, Morcos GNB, Wissa MY. The role of mosapride and levosulpiride in gut function and glycemic control in diabetic rats. Arab J Gastroenterol. 2023;24:109-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 184. | Andrews CN, Woo M, Buresi M, Curley M, Gupta M, Tack J, Wilsack L, Nasser Y. Prucalopride in diabetic and connective tissue disease-related gastroparesis: Randomized placebo-controlled crossover pilot trial. Neurogastroenterol Motil. 2021;33:e13958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 185. | Grover M, Farrugia G, Stanghellini V. Gastroparesis: a turning point in understanding and treatment. Gut. 2019;68:2238-2250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 175] [Article Influence: 25.0] [Reference Citation Analysis (1)] |
| 186. | Wei L, Singh R, Ro S, Ghoshal UC. Gut microbiota dysbiosis in functional gastrointestinal disorders: Underpinning the symptoms and pathophysiology. JGH Open. 2021;5:976-987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 187. | Zheng X, Zhang Y, Tan Y, Li Y, Xue Q, Li H, Zhang X, Pan Y, Xu J, Zhang J. Alpinia officinarum Hance extract ameliorates diabetic gastroparesis by regulating SCF/c-kit signaling pathway and rebalancing gut microbiota. Fitoterapia. 2024;172:105730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 188. | Huang J, Song Y, Cheng S, Yang X. Mechanism of action of FoxiangSan in diabetic gastroparesis: Gut microbiota and cAMP/PKA pathway. Heliyon. 2024;10:e35558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 189. | Li H, Wang Y, Tian Y, Tian F, Xing Z, Wang Y, Yan M, Gong Y. Atractylodes chinensis volatile oil up-regulated IGF-1 to improve diabetic gastroparesis in rats. Iran J Basic Med Sci. 2022;25:520-526. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 190. | Jin QH, Shen HX, Wang H, Shou QY, Liu Q. Curcumin improves expression of SCF/c-kit through attenuating oxidative stress and NF-κB activation in gastric tissues of diabetic gastroparesis rats. Diabetol Metab Syndr. 2013;5:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 191. | Hou C, Liang H, Hao Z, Zhao D. Berberine ameliorates the neurological dysfunction of the gastric fundus by promoting calcium channels dependent release of ACh in STZ-induced diabetic rats. Saudi Pharm J. 2023;31:433-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 192. | An X, Long C, Deng X, Tang A, Xie J, Chen L, Wang Z. Higenamine inhibits apoptosis and maintains survival of gastric smooth muscle cells in diabetic gastroparesis rat model via activating the β2-AR/PI3K/AKT pathway. Biomed Pharmacother. 2017;95:1710-1717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 193. | McCallum RW, Snape W, Brody F, Wo J, Parkman HP, Nowak T. Gastric electrical stimulation with Enterra therapy improves symptoms from diabetic gastroparesis in a prospective study. Clin Gastroenterol Hepatol. 2010;8:947-54; quiz e116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 151] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 194. | Guerci B, Bourgeois C, Bresler L, Scherrer ML, Böhme P. Gastric electrical stimulation for the treatment of diabetic gastroparesis. Diabetes Metab. 2012;38:393-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 195. | Ward MP, Gupta A, Wo JM, Rajwa B, Furness JB, Powley TL, Nowak TV. An emerging method to noninvasively measure and identify vagal response markers to enable bioelectronic control of gastroparesis symptoms with gastric electrical stimulation. J Neurosci Methods. 2020;336:108631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 196. | Li C, Liu S, Guan Y, Qian W, du F, Hou X. Long pulse gastric electrical stimulation induces regeneration of myenteric plexus synaptic vesicles in diabetic rats. Neurogastroenterol Motil. 2010;22:453-461, e108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 197. | Chen Y, Zhang S, Li Y, Yan H, Ba Y, Wang X, Shi N, Liu C. Gastric Electrical Stimulation Increases the Proliferation of Interstitial Cells of Cajal and Alters the Enteric Nervous System in Diabetic Rats. Neuromodulation. 2022;25:1106-1114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 198. | Gottfried-Blackmore A, Adler EP, Fernandez-Becker N, Clarke J, Habtezion A, Nguyen L. Open-label pilot study: Non-invasive vagal nerve stimulation improves symptoms and gastric emptying in patients with idiopathic gastroparesis. Neurogastroenterol Motil. 2020;32:e13769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 199. | Kornum DS, Bertoli D, Kufaishi H, Wegeberg AM, Okdahl T, Mark EB, Høyer KL, Frøkjær JB, Brock B, Krogh K, Hansen CS, Knop FK, Brock C, Drewes AM. Transcutaneous vagal nerve stimulation for treating gastrointestinal symptoms in individuals with diabetes: a randomised, double-blind, sham-controlled, multicentre trial. Diabetologia. 2024;67:1122-1137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 200. | Bugeja L, Camilleri L, Schembri J. Pyloric Botulinum Toxin for Diabetic Gastroparesis: A Case of Sustained Glycaemic Improvement. Eur J Case Rep Intern Med. 2025;12:005495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 201. | Friedenberg FK, Palit A, Parkman HP, Hanlon A, Nelson DB. Botulinum toxin A for the treatment of delayed gastric emptying. Am J Gastroenterol. 2008;103:416-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 237] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 202. | Sarosiek I, Bashashati M, Davis BR, Espino K, Vasquez D, Torelli R, Bright T, Sarosiek J, Diaz JR, Padilla O, Dwivedi AK, McCallum RW. Combined Gastric Electrical Stimulation and Pyloroplasty in Gastroparesis: A Randomized Clinical Trial. JAMA Netw Open. 2025;8:e2546332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 203. | Wu KL, Chiu YC, Yao CC, Tsai CE, Hu ML, Kuo CM, Tai WC, Chuah SK, Hsiao CC. Effect of extracorporeal low-energy shock wave on diabetic gastroparesis in a rat model. J Gastroenterol Hepatol. 2019;34:720-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 204. | Han N, Jiang W, Li G, Lu L, Shan J, Feng L, Jin L. Low-intensity pulsed ultrasound at ST36 improves the gastric motility by TNF-α/IKKβ/NF-κB signaling pathway in diabetic rats. J Gastroenterol Hepatol. 2023;38:2018-2026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 205. | Sarosiek I, Song G, Sun Y, Sandoval H, Sands S, Chen J, McCallum RW. Central and Peripheral Effects of Transcutaneous Acupuncture Treatment for Nausea in Patients with Diabetic Gastroparesis. J Neurogastroenterol Motil. 2017;23:245-253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 206. | Zhang Y, Tang YW, Zhou J, Wei YR, Peng YT, Yan Z, Yue ZH. Electroacupuncture at ST36 ameliorates gastric dysmotility in rats with diabetic gastroparesis via the nucleus tractus solitarius-vagal axis. World J Gastroenterol. 2025;31:107395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 207. | Han X, Chen X, Wang X, Gong M, Lu M, Yu Z, Xu B, Yuan J. Electroacupuncture at ST36 Improve the Gastric Motility by Affecting Neurotransmitters in the Enteric Nervous System in Type 2 Diabetic Rats. Evid Based Complement Alternat Med. 2021;2021:6666323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (3)] |
| 208. | Tang YW, Zhang Y, Zhou J, Peng YT, Zi Y, Wei YR, Yue ZH. Electroacupuncture with different current intensities can improve gastrointestinal motility in diabetic gastroparesis via vagal and sympathetic pathways. World J Diabetes. 2025;16:107779. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (6)] |
| 209. | Wei X, Lin Y, Zhao D, Xiao X, Chen Q, Chen S, Peng Y. Electroacupuncture Relieves Suppression of Autophagy in Interstitial Cells of Cajal of Diabetic Gastroparesis Rats. Can J Gastroenterol Hepatol. 2020;2020:7920715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (6)] |
| 210. | Zhao J, An J, Liu S. Electroacupuncture at ST36 Increases Bone Marrow-Derived Interstitial Cells of Cajal via the SDF-1/CXCR4 and mSCF/Kit-ETV1 Pathways in the Stomach of Diabetic Mice. Evid Based Complement Alternat Med. 2018;2018:7878053. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 211. | Ahmed SS, El-Hafez HAA, Mohsen M, El-Baiomy AA, Elkhamisy ET, El-Eshmawy MM. Is vitamin B12 deficiency a risk factor for gastroparesis in patients with type 2 diabetes? Diabetol Metab Syndr. 2023;15:33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 212. | Abdelsayed N, Juarez A, Carter M. Severe Gastroparesis Leading to Hypoglycemia and Subsequent Seizures. Cureus. 2022;14:e30527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 213. | Betônico CC, Cobello AV, Santos-Bezerra DP, de A Leite AZ, Correa-Giannella ML, Nery M, Queiroz MS. Diet consistency modification improves postprandial glycemic and gastroparesis symptoms. J Diabetes Metab Disord. 2022;21:1661-1667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 214. | Staller K, Parkman HP, Greer KB, Leiman DA, Zhou MJ, Singh S, Camilleri M, Altayar O; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on Management of Gastroparesis. Gastroenterology. 2025;169:828-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 215. | Lacy BE, Tack J, Gyawali CP. AGA Clinical Practice Update on Management of Medically Refractory Gastroparesis: Expert Review. Clin Gastroenterol Hepatol. 2022;20:491-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (0)] |