Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.117580
Revised: January 12, 2026
Accepted: February 24, 2026
Published online: May 15, 2026
Processing time: 155 Days and 0.6 Hours
Primary gastric adenosquamous carcinoma (PGASC) is a rare malignancy with a poor prognosis. The current clinical understanding of PGASC has been derived from Western populations and may not accurately reflect the disease characteristics of Eastern populations. Furthermore, the efficacy of standard adjuvant chemotherapy for the treatment of PGASC has not been determined.
To explore the clinicopathological features, prognosis, and chemotherapy efficacy of PGASC in a Chinese cohort.
This retrospective analysis of 108 patients with PGASC was conducted from March 2007 to December 2021 at six medical centers in China. The data from these patients was compared with the data from 468 randomly selected patients with pathologically diagnosed gastric adenocarcinoma (GAC) from the same time period at Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine. The characteristics, prognosis, and chemotherapy efficacy were investigated by propensity score matching.
Patients with PGASC had higher rates of tumors located in the cardia (44.4% vs 22.9%), lymphovascular invasion (55.6% vs 35.5%), and perineural invasion (50.0% vs 32.7%) than patients with GAC. Immunohistochemical analysis revealed that the PGASC group had a significantly higher proportion of cells, with at least 10% expressing pro
PGASC has different characteristics than GAC, including a more aggressive presentation, a poorer prognosis, and higher programmed cell death-ligand 1 expression.
Core Tip: This multicenter retrospective study represents the largest Eastern cohort of patients with primary gastric ade
- Citation: Chen YW, Dong ZY, Wang XX, Cao Y, Lin JX, Chen H, Zhang HY, Xiao YB, Li GX, Li ZR, Chen L, Xia X, Zhang ZZ. Characteristics and prognosis of primary gastric adenosquamous carcinoma: A multicenter retrospective study based on propensity score matching. World J Gastrointest Oncol 2026; 18(5): 117580
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/117580.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.117580
Adenosquamous carcinoma (ASC) is a malignant epithelial tumor that can occur in a multitude of organs and tissues, having both typical glandular and squamous components. Primary gastric ASC (PGASC) originates in the gastric mucosa. It appears that patients diagnosed with PGASC respond poorly to the current recommended therapy[1,2]; however, due to the low incidence of PGASC and the majority of clinical experiences reported in case reports and small sample studies, this anecdotal evidence requires further clarification.
PGASC is highly malignant and has a poor prognosis. Specifically, PGASC has higher rates of invasion and metastasis, larger tumor sizes, and poor differentiation. Previous studies have shown that patients with PGASC have a higher rate of tumors located in the cardia (65.1% vs 46.0%, P < 0.001). Higher tumor-node-metastasis (TNM) stages are also present at diagnosis (stage II/III/IV 87.0% vs stage I 78.0%) and a lower rate of differentiation (poorly differentiated/undifferentiated 80.8% vs differentiated 60.3%). However, data from the surveillance, epidemiology, and end results (SEER) database may have certain flaws such as a higher rate of data lost[1]. The SEER database also only includes data from Western populations, thereby limiting the generalizability to Eastern populations. Therefore, systematic studies with large sample sizes that include Eastern populations are necessary to understand the characteristics, prognosis, and chemotherapy efficacy in PGASC[2,3].
In this study, data of patients with PGASC from six medical centers in China were retrospectively analyzed. Propensity score matching (PSM) was used to compare patients with PGASC and patients with gastric adenocarcinoma (GAC), and the characteristics, prognosis, and chemotherapy efficacy of PGASC were analyzed.
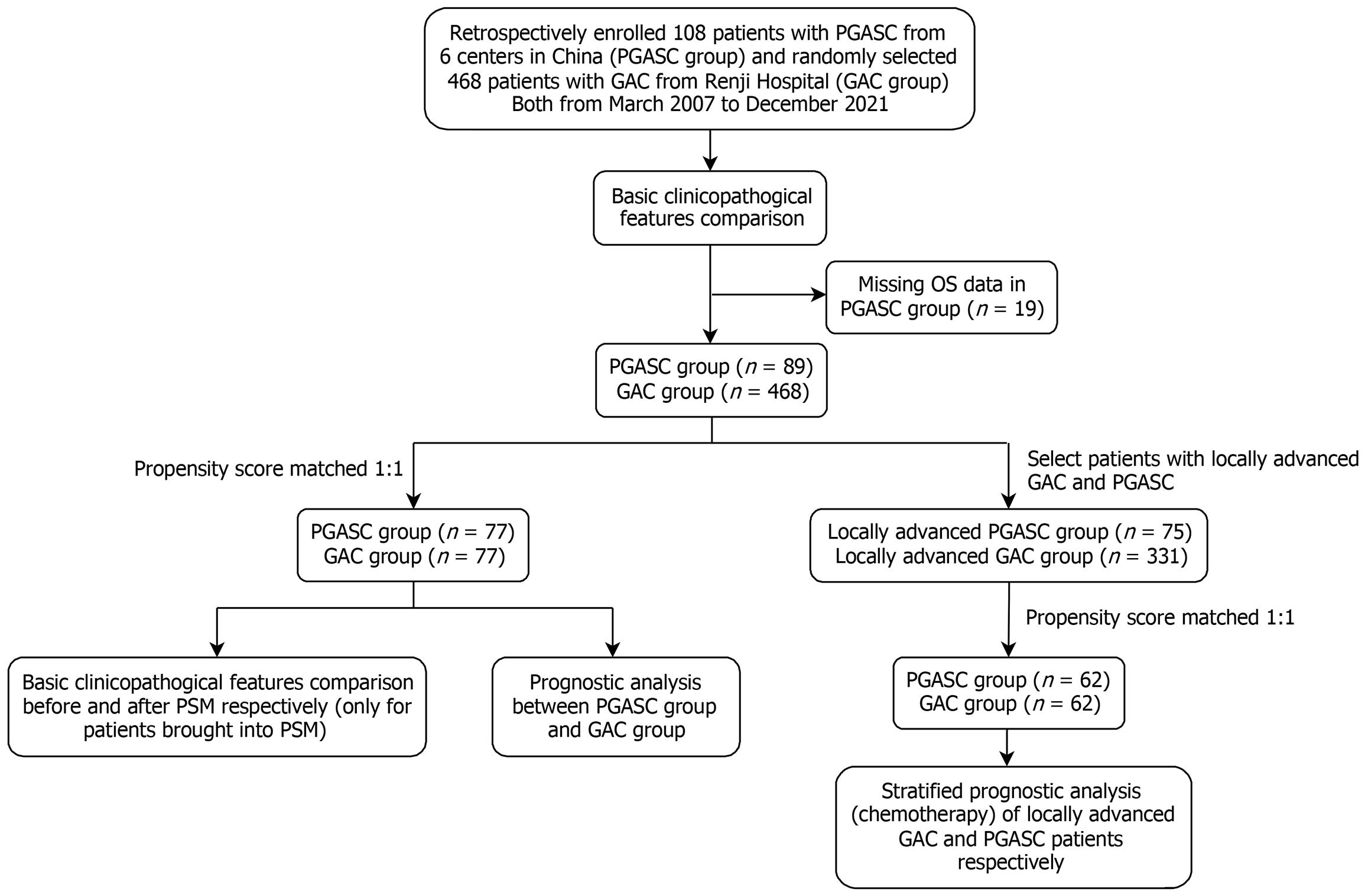
This study was approved by the Ethics Committee of Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine (No. KY2022-084-B). This study was also registered at the Chinese Clinical Trial Registry under the number No. ChiCTR2200062463. The need for informed consent was waived because of the retrospective nature of this study. This study adhered to the Declaration of Helsinki[4]. It followed the reporting and guidelines in propensity score analysis, which is modified from the strengthening the reporting of observational studies in epidemiology statement[5,6]. A flowchart of the study protocol is shown in Figure 1.
Data from 108 patients with PGASC that had been diagnosed pathologically were collected from six centers in China [Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine (n = 31); Chinese People’s Liberation Army General Hospital (n = 30); The First Affiliated Hospital, Nanchang University (n = 24); Nanfang Hospital, Southern Medical University (n = 10); Fujian Medical University Union Hospital (n = 10); and The First People’s Hospital of Kashgar (n = 3)]. Patients diagnosed between March 2007 and December 2021 were retrospectively enrolled.
Pathological examination served as the gold standard for PGASC diagnosis and was defined as gastric carcinoma containing both squamous cell carcinoma and adenocarcinoma components with each component accounting for no less than 25% of the tumor[3,7,8]. A control group of 468 patients with GAC diagnosed during the same period was recruited from the gastric cancer database of Renji Hospital. To ensure comparability and minimize selection bias, the GAC group was generated using simple random sampling from the pool of eligible patients.
The exclusion criteria for the PGASC and GAC groups were as follows: (1) Remnant gastric carcinoma; (2) Pregnancy or breastfeeding; (3) History of other malignant diseases within 5 years; and (4) Concurrent surgical treatment for other diseases. Additionally, patients with ASC originating in the lower esophagus invading the stomach were excluded from the PGASC group.
The Japanese gastric cancer treatment guidelines were utilized to determine the appropriate surgical approach. The resected tissues were pathologically examined by two experienced pathologists. Tumor staging was performed according to the American Joint Committee on Cancer TNM staging system.
Postoperative follow-up included physical examination, imaging examination, and esophagogastroduodenoscopy. All patients received follow-up care once every 3-6 months during the first 2 years and once every 6-12 months during the following 3 years. Overall survival (OS) was defined as the period from the date of surgery to the date of the last follow-up or death. In the PGASC group the OS of 19 patients was unknown because the patients were lost to follow-up. In accordance with standard statistical practice, these data were classified as missing at random. Multiple imputation was not performed because the missing OS values would adversely affect the PSM, potentially introducing bias and baseline data were unaffected by the exclusion of these data (Table 1).
| Characteristic | GAC (n = 468) | PGASC1 (n = 108) | P value |
| Age in years (≤ 60/> 60) | 163/305 | 38/70 | 0.940 |
| Sex (female/male) | 152/316 | 23/85 | 0.020 |
| Surgical resection (incomplete resection/complete resection) | 275/193 | 64/44 | 0.920 |
| Primary site (non-cardia/cardia) | 361/107 | 60/48 | < 0.001 |
| pT stage2 (pT1/pT2/pT3/pT4) | 79/51/31/307 | 2/5/35/66 | < 0.001 |
| pN stage2 (pN0/pN1/pN2/pN3) | 163/76/99/130 | 21/27/26/34 | 0.010 |
| pM stage2 (pM0/pM1) | 434/34 | 92/16 | 0.010 |
| Lymphovascular invasion (VI-/VI+) | 302/166 | 48/60 | < 0.001 |
| Perineural invasion (PI-/PI+) | 315/153 | 54/54 | 0.001 |
| Chemotherapy (no/yes) | 155/313 | 53/55 | < 0.01 |
PSM was used to reduce bias in the comparison of the PGASC and GAC groups. With a 0.1 caliper width, 1:1 nearest-neighbor matching was carried out. There was no replacement sampling. We enrolled the following eight covariables to compare the prognosis between the PGASC and GAC groups: Age, sex, resection range, primary site, pT stage, pN stage, pM stage and chemotherapy induction. Furthermore, six covariables including age, sex, resection range, primary site, pT stage, and pN stage were enrolled for the comparison of chemotherapy efficacy in the GAC and PGASC groups.
Survival analysis and PSM were performed using the R 4.2.2 software (R Foundation for Statistical Computing) by R package “survminer” and “MatchIt”. Other results were analyzed by SPSS 22.0 statistical software (IBM Corp., Armonk, NY, United States). The χ2 test and Fisher’s exact test were used for the comparison of categorical variables. The Kaplan-Meier method was used to calculate time-specific survival probabilities. The log-rank test was used to compare the 3-year and 5-year OS rates. Further analysis was carried out on the efficacy of adjuvant chemotherapy in patients with locally advanced PGASC. A time-dependent Cox regression was used when the survival curves were crossed, indicating that the log-rank test was unsuitable. P values < 0.05 were considered statistically significant.
Baseline data of the PGASC and GAC groups are shown in Table 1. There was a higher proportion of males in the PGASC group (78.7% vs 67.5%, P = 0.02). The lesion was significantly more likely to be located in the cardia of the PGASC group than in the GAC group (44.4% vs 22.9%, P < 0.001). The PGASC group had a higher proportion of patients with deeper tumor invasion (pT2-4) compared with the GAC group (98.1% vs 83.1%, P < 0.001) according to the American Joint Committee on Cancer 8th edition TNM staging system. The PGASC group was more likely to present with deeper invasion (proportion of pT3 and pT4: 93.5% vs 72.2%, P < 0.001), more lymph node metastasis (proportion of pN +: 80.6% vs 65.2%, P < 0.010), and more distant metastasis (proportion of M1: 14.8% vs 7.3%, P < 0.050). Compared with the GAC group, the proportion of lymphovascular invasion (35.5% vs 55.6%, P < 0.001) and perineural invasion (32.7% vs 50.0%, P = 0.001) were significantly higher in the PGASC group.
Immunohistochemical (IHC) tests revealed that the proportion of patients with Ki-67 expression higher than 50% was significantly lower in the PGASC group than in the GAC group (54.3% vs 66.0%, P < 0.050) (Table 2). Significantly more patients with PGASC who completed the programmed cell death-ligand 1 (PD-L1) IHC test (n = 38) had at least 10% more PD-L1 expression than the GAC group (13.2% vs 2.8%, P < 0.001). There was no significant difference in the expression of human epidermal growth factor receptor 2 (P = 0.070) or P53 (P = 0.780) between the two groups. All unknown data were excluded during the comparison of IHC results. We also performed sensitivity analyses by excluding patients in the GAC and PGASC groups from March 2007 to December 2011 and found that the survival results did not change (Supplementary Figure 1).
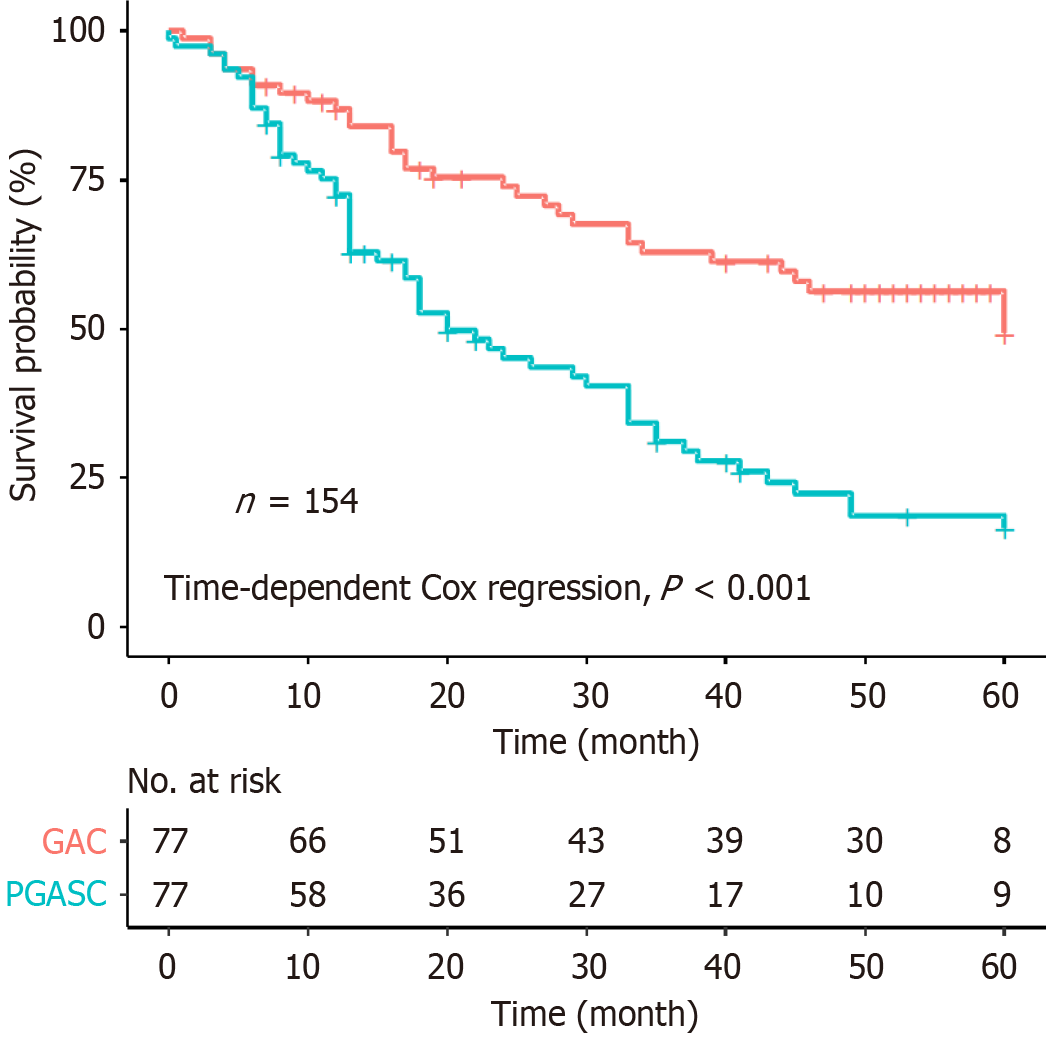
No significant differences were detected when the entire PGASC cohort was compared with the PGASC cohort with available OS data (Supplementary Table 1). After the exclusion of the patients without OS data, 77 patients with PGASC and 77 patients with GAC were selected for 1:1 PSM. The propensity score distribution showed a good match of the two groups (Supplementary Figure 2), and no statistical significance was found in the baseline characteristics of the matched covariables between the two groups (Table 3). Due to the inevitable loss of partial IHC data in the PGASC group, the distribution of IHC covariables was imbalanced, resulting in variability in PD-L1 and Ki-67 expression. The results of prognostic analysis showed that the OS of the PGASC group was significantly worse than that of the GAC group (3-year OS: 31.1% vs 62.9%, P < 0.001; 5-year OS: 16.6% vs 49.3%, P < 0.001). The results are shown in Figure 2.
| Characteristics | Before PSM | After PSM | ||||
| GAC (n = 468) | PGASC1 (n = 89) | P value | GAC (n = 77) | PGASC (n = 77) | P value | |
| Age, years (≤ 60/> 60) | 163/305 | 31/58 | > 0.990 | 26/51 | 27/50 | 0.870 |
| Sex (female/male) | 152/316 | 17/72 | 0.010 | 19/58 | 17/60 | 0.700 |
| Surgical resection (incomplete resection/complete resection) | 275/193 | 58/31 | 0.260 | 44/33 | 46/31 | 0.740 |
| Primary site (non-cardia/cardia) | 361/107 | 52/37 | < 0.001 | 49/28 | 43/34 | 0.320 |
| pT stage2 (pT1/pT2/pT3/pT4) | 79/51/31/307 | 2/5/21/61 | < 0.001 | 2/3/7/65 | 2/5/19/51 | 0.050 |
| pN stage2 (pN0/pN1/pN2/pN3) | 163/76/99/130 | 16/20/22/31 | 0.020 | 19/12/12/34 | 16/20/16/25 | 0.240 |
| pM stage2 (pM0/pM1) | 434/34 | 76/13 | 0.020 | 64/13 | 65/12 | 0.830 |
| Chemotherapy (no/yes) | 155/313 | 39/50 | 0.050 | 29/48 | 28/49 | 0.870 |
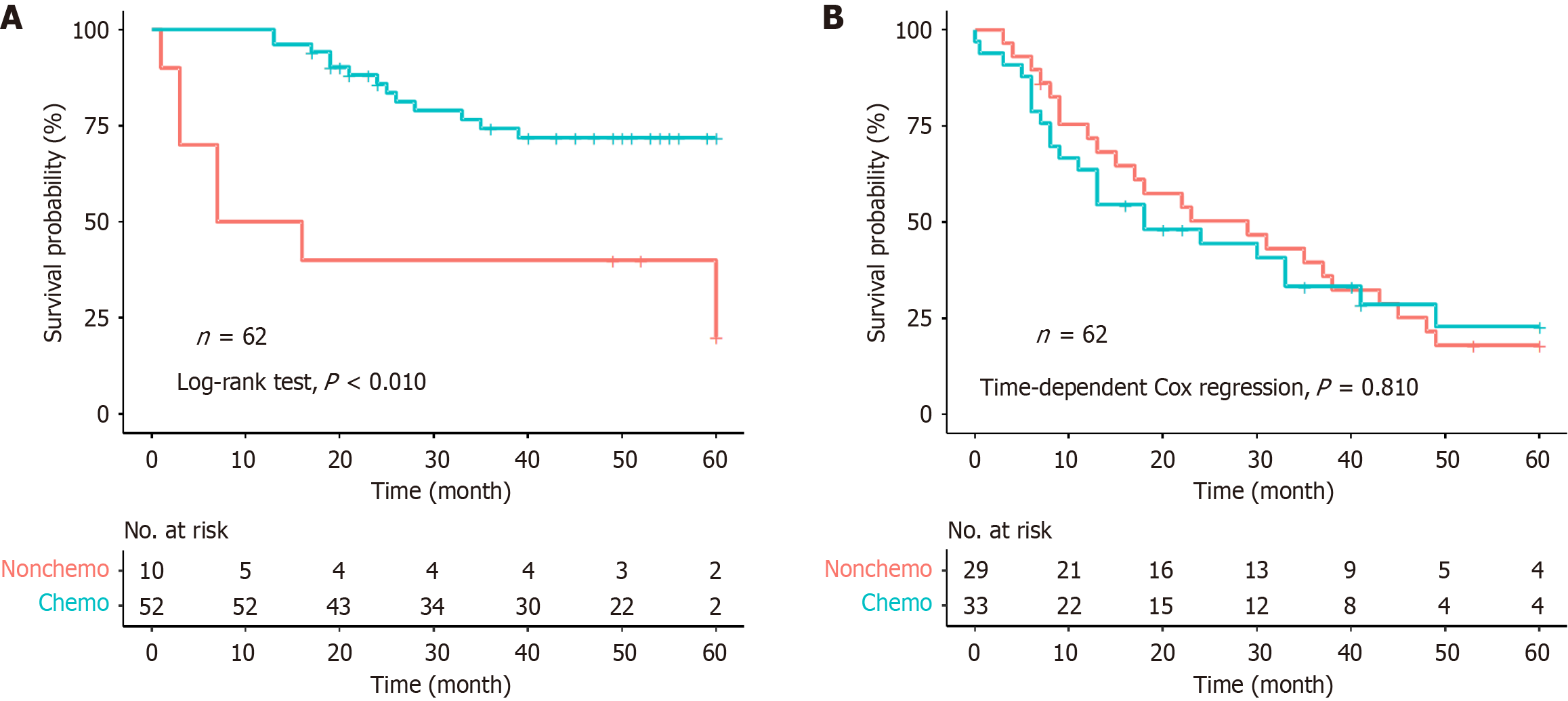
Locally advanced gastric cancer was defined as gastric cancer in TNM stage II and III. Further analysis was conducted according to whether adjuvant chemotherapy was given after surgery. After 1:1 PSM on patients with locally advanced disease, 62 patients with GAC and 62 patients with PGASC were selected. The covariables of patients with PGASC remained even after PSM (Supplementary Table 2 and Supplementary Figure 3). Among the patients with GAC, the OS of patients who received adjuvant chemotherapy after surgery was significantly better than those who did not receive adjuvant chemotherapy after surgery (3-year OS: 74.3% vs 40.0%, P < 0.010; 5-year OS: 71.9% vs 20.0%, P < 0.010). Among the patients with PGASC, there was no significant difference in OS in patients who received adjuvant chemotherapy or in those who did not (3-year OS: 39.5% vs 33.3%, P = 0.810; 5-year OS: 18.0% vs 22.8%, P = 0.810) (Figure 3).
This multicenter study represents the largest PGASC study undertaken in an Eastern population to date. Currently, only three studies from the SEER database include data on patients with PGASC[1,9,10]. However, these data came from Western populations and may not reflect the characteristics of Eastern populations because the differences in gastric cancer between Western and Eastern populations has already been established[11,12]. Chen et al[3] found that 53.8% of their patients had tumors localized to the upper third of the stomach and 92.3% of patients were diagnosed at the T3-T4 stage. Our results confirmed these findings, showing 44.4% of patients with tumors located in upper third of the stomach and 93.5% of patients diagnosed at the pT3-T4 stage. The SEER database showed that 90.3% of patients had tumors located in the middle and lower third of the stomach and 46.9% of patients diagnosed at the T3-T4 stage[1]. Also, a higher rate of unknown data is inevitable in a public database.
Generally, PGASC accounts for 0.3%-0.8% of all gastric cancers[1]. PGASC accounted for about 0.6% of gastric cancer cases in the SEER database from 1988 to 2016[9]. The six centers that participated in study are representative of the Chinese in terms of geographical distribution and number of surgical cases. Although this study enrolled the largest number of Eastern participants, the incidence of PGASC was small with only 108 total patients across all medical centers and over a period of almost 15 years. At the Renji Hospital affiliated to Shanghai Jiao Tong University School of Medicine, PGASC accounted for 0.66% (31/4680) of patients with gastric cancer.
The male to female ratio was 3.7:1, which was consistent with other studies[3,13]. Compared with patients with GAC, PGASC most likely occurred in the cardia of the stomach (44.4%), remaining consistent with other studies based in Chinese populations[3] but inconsistent to the SEER database[1,9]. This discrepancy is likely due to genetic or diet/environmental differences between Eastern and Western populations[1,9]. Because over half of the tumors were located in areas other than the cardia, it is assumed that the squamous component did not originate from the esophagus[3]. Only 2 of the 108 PGASC cases were diagnosed as early gastric cancer, yielding similar findings to other studies in which most cases are diagnosed at an advanced stage[1,9,14]. A higher proportion of perineural invasion and lymphovascular invasion were found in PGASC. Yoshida et al[15] reported a case of PGASC with biopsy results that showed that the tumor only invaded the submucosa, and no lymphovascular invasion was observed. However, regional lymph node invasion and liver metastases had occurred. Collectively, these results revealed that PGASC has a high malignancy, poor biological behavior, and rapid progression.
The preliminary prognostic analysis of this study revealed that PGASC may be the key factor directly leading to a poor prognosis in patients. The prognosis of patients with PGASC and GAC were compared in this study, and PSM was performed to balance covariables. PSM decreases data bias, leading to more reliable results. Previous studies may have significant bias due to the varying distribution of covariables[1]. A higher proportion of Ki-67 expression was seen in the GAC group, potentially indicating a worse prognosis[16]. However, a higher proportion of PD-L1 expression was seen in the PGASC group, also potentially indicating a worse prognosis[17,18]. However, no studies have yet reported on the use of immunotherapy in patients with PGASC. We plan to expand the scale of our PGASC research and focus on the PD-L1-related immune microenvironment in the future.
Prognostic analysis showed that the 3-year OS and 5-year OS of patients with PGASC were significantly worse than those in patients with GAC. These results are consistent with the results of previous studies[1,3,9]. Notably, there were 19 patients with PGASC without OS data. This lack of data did not appear to have any effect on the outcome of our results (Table 1).
Currently, there is no standard treatment for PGASC, and GAC treatment protocols are applied to PGASC. In this study 33 patients with locally advanced PGASC and complete OS data received adjuvant chemotherapy. Postoperative adjuvant chemotherapy did not lead to a survival benefit. It is possible that PGASC is resistant to the current GAC chemotherapy strategy, and it is worth considering the formulation of a treatment strategy for locally advanced PGASC. Comprehensive treatment reports are limited to a few case reports[19] and need to be further expanded.
The poor prognosis of PGASC can be potentially attributed to the following: (1) PGASC shows malignant biological behavior of both squamous and adenomatous components, and the tumor heterogeneity is relatively higher[1,8,20]; (2) PGASC has stronger invasion and metastasis ability and is more prone to serosal invasion, lymph node metastasis, and distant metastasis[3,21]; and (3) PGASC is resistant to the standard chemotherapy protocol (platinum combined with fluorouracil)[22].
The potential mechanisms underlying the chemorefractory nature of PGASC remain to be fully elucidated, but tumor heterogeneity and the immune microenvironment may play critical roles. ASC contains two distinct histological components, and the squamous component may exhibit intrinsic resistance to standard 5-fluorouracil and platinum-based regimens typically used for adenocarcinoma. Furthermore, our study revealed a significantly higher proportion of PD-L1 expression in PGASC compared with GAC. High PD-L1 expression is often associated with an immunosuppressive microenvironment that promotes tumor immune evasion and has been linked to chemotherapy resistance in various malignancies.
Given the limited efficacy of chemotherapy and elevated PD-L1 expression (nearly 20% in this study), immune checkpoint inhibitors may be a promising alternative treatment strategy. Instead of relying solely on traditional first-line recommended chemotherapy (e.g., SOX, XELOX, FOLFOX), future clinical practice should investigate the use of immunotherapy for the treatment of PGASC, potentially guided by routine PD-L1 testing.
There are some limitations of this study that should be acknowledged: (1) Although this research included the largest PGASC cohort, we were still unable to carry out multilevel and multiangle research; (2) The included subjects were all patients undergoing surgical treatment. Therefore, the total milieu of PGASC may not have been revealed; (3) GAC data for the control group came from a single center; (4) There was an uneven distribution of the number of cases enrolled by each of the six centers; (5) The inevitable data loss of a retrospective study, particularly regarding IHC markers like PD-L1 and P53, may have led to imbalanced data. Due to the long duration and retrospective nature of sample collection, we could not retrieve tissue blocks for all patients. To ensure the reliability of our results, we performed analyses strictly on the subset of patients with complete IHC data rather than using imputation methods; (6) The patients in the GAC group were randomly selected by a 1:10 ratio, potentially influencing the efficacy of PSM. However, we did achieve successful matching; and (7) The survival analysis of patients who received chemotherapy only showed that PGASC may be resistant to the current chemotherapy strategy.
We demonstrated that PGASC has a high malignancy, low sensitivity to traditional chemotherapy, poor prognosis, and special histological characteristics in the largest cohort of patients with PGASC in an Eastern region. Our data showed that a considerable proportion of patients with PGASC may benefit from immunotherapy. Additional studies are required to confirm this hypothesis.
| 1. | Ge Y, Lin L, Ma X, Luo D, Shi L, Jiang M, Fan H, He Y, Yang L, Xu Z. Adenosquamous Carcinoma of the Stomach: A Population-based Study from the SEER Database. J Cancer. 2019;10:5705-5713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 2. | Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, Washington KM, Carneiro F, Cree IA; WHO Classification of Tumours Editorial Board. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76:182-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3203] [Cited by in RCA: 2885] [Article Influence: 480.8] [Reference Citation Analysis (5)] |
| 3. | Chen H, Shen C, Yin R, Yin Y, Chen J, Han L, Zhang B, Chen Z, Chen J. Clinicopathological characteristics, diagnosis, treatment, and outcomes of primary gastric adenosquamous carcinoma. World J Surg Oncol. 2015;13:136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 4. | World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310:2191-2194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24525] [Cited by in RCA: 20920] [Article Influence: 1609.2] [Reference Citation Analysis (16)] |
| 5. | von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014;12:1495-1499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8394] [Cited by in RCA: 7656] [Article Influence: 638.0] [Reference Citation Analysis (10)] |
| 6. | Yao XI, Wang X, Speicher PJ, Hwang ES, Cheng P, Harpole DH, Berry MF, Schrag D, Pang HH. Reporting and Guidelines in Propensity Score Analysis: A Systematic Review of Cancer and Cancer Surgical Studies. J Natl Cancer Inst. 2017;109:djw323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 320] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 7. | Shirahige A, Suzuki H, Oda I, Sekiguchi M, Mori G, Abe S, Nonaka S, Yoshinaga S, Sekine S, Kushima R, Saito Y, Fukagawa T, Katai H. Fatal submucosal invasive gastric adenosquamous carcinoma detected at surveillance after gastric endoscopic submucosal dissection. World J Gastroenterol. 2015;21:4385-4390. [PubMed] [DOI] [Full Text] |
| 8. | Chen YY, Li AF, Huang KH, Lan YT, Chen MH, Chao Y, Lo SS, Wu CW, Shyr YM, Fang WL. Adenosquamous carcinoma of the stomach and review of the literature. Pathol Oncol Res. 2015;21:547-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Li HS, Liu X, Zhang MY, Cheng K, Chen Y, Zhou YW, Liu JY. Clinicopathologic characteristics, survival, and treatments for gastric adenosquamous carcinoma: a population-based study. Curr Oncol. 2020;27:e527-e536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 10. | Chu YX, Gong HY, Hu QY, Song QB. Adenosquamous carcinoma may have an inferior prognosis to signet ring cell carcinoma in patients with stages I and II gastric cancer. World J Gastrointest Oncol. 2020;12:101-112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 11. | Strong VE, Song KY, Park CH, Jacks LM, Gonen M, Shah M, Coit DG, Brennan MF. Comparison of gastric cancer survival following R0 resection in the United States and Korea using an internationally validated nomogram. Ann Surg. 2010;251:640-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 279] [Article Influence: 17.4] [Reference Citation Analysis (4)] |
| 12. | Strong VE, Wu AW, Selby LV, Gonen M, Hsu M, Song KY, Park CH, Coit DG, Ji JF, Brennan MF. Differences in gastric cancer survival between the U.S. and China. J Surg Oncol. 2015;112:31-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 152] [Article Influence: 13.8] [Reference Citation Analysis (1)] |
| 13. | Li HS, Chen Y, Zhang MY, Cheng K, Zhou YW, Liu JY. Increased proportion of the squamous cell carcinoma component is associated with worse survival in resected gastric adenosquamous carcinoma: A STROBE compliant cohort study. Medicine (Baltimore). 2020;99:e21980. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Ren W, Yu J, Zhang ZM, Song YK, Li YH, Wang L. Missed diagnosis of early gastric cancer or high-grade intraepithelial neoplasia. World J Gastroenterol. 2013;19:2092-2096. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 51] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (4)] |
| 15. | Yoshida K, Manabe T, Tsunoda T, Kimoto M, Tadaoka Y, Shimizu M. Early gastric cancer of adenosquamous carcinoma type: report of a case and review of literature. Jpn J Clin Oncol. 1996;26:252-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 16. | Ko GH, Go SI, Lee WS, Lee JH, Jeong SH, Lee YJ, Hong SC, Ha WS. Prognostic impact of Ki-67 in patients with gastric cancer-the importance of depth of invasion and histologic differentiation. Medicine (Baltimore). 2017;96:e7181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 17. | Zhang Y, Yang Y, Chen Y, Lin W, Chen X, Liu J, Huang Y, Wang H, Teng L. PD-L1: Biological mechanism, function, and immunotherapy in gastric cancer. Front Immunol. 2022;13:1060497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 18. | Zurlo IV, Schino M, Strippoli A, Calegari MA, Cocomazzi A, Cassano A, Pozzo C, Di Salvatore M, Ricci R, Barone C, Bria E, Tortora G, Larocca LM, Basso M, Martini M. Predictive value of NLR, TILs (CD4+/CD8+) and PD-L1 expression for prognosis and response to preoperative chemotherapy in gastric cancer. Cancer Immunol Immunother. 2022;71:45-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 19. | Kadowaki S, Yatabe Y, Nitta S, Ito Y, Muro K. Durable response of human epidermal growth factor receptor-2-positive gastric adenosquamous carcinoma to trastuzumab-based chemotherapy. Case Rep Oncol. 2014;7:210-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 20. | Akce M, Jiang R, Alese OB, Shaib WL, Wu C, Behera M, El-Rayes BF. Gastric squamous cell carcinoma and gastric adenosquamous carcinoma, clinical features and outcomes of rare clinical entities: a National Cancer Database (NCDB) analysis. J Gastrointest Oncol. 2019;10:85-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Moro K, Nagahashi M, Naito T, Nagai Y, Katada T, Minagawa M, Hasegawa J, Tani T, Shimakage N, Usuda H, Gabriel E, Kawaguchi T, Takabe K, Wakai T. Gastric adenosquamous carcinoma producing granulocyte-colony stimulating factor: a case of a rare malignancy. Surg Case Rep. 2017;3:67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 22. | Saito S, Hosoya Y, Morishima K, Ui T, Haruta H, Kurashina K, Meguro Y, Zuiki T, Sata N, Fujii H, Matsubara D, Niki T, Lefor AT, Yasuda Y. A clinicopathological and immunohistochemical study of gastric cancer with squamous cell carcinoma components: a clinically aggressive tumor. J Dig Dis. 2012;13:407-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |