Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.117512
Revised: January 16, 2026
Accepted: February 10, 2026
Published online: May 15, 2026
Processing time: 156 Days and 18.8 Hours
Hepatocellular carcinoma (HCC) remains a major complication in patients with chronic hepatitis C (CHC). Practical, low-cost tools that identify untreated patients at high cancer risk are needed for surveillance and treatment prioritization. Noni
To evaluate whether baseline and longitudinal trajectories of noninvasive biomarkers, particularly the FIB-4 index, predict subsequent HCC in treatment-naïve CHC.
We performed a single-center retrospective cohort study of adults with CHC first evaluated between 1999 and 2015. Patients with other major liver diseases or prior HCC were excluded. Demographics, virology, labs and imaging were collected at baseline and year 3. FIB-4 categories were < 1.45, 1.45-3.25, and > 3.25; 3-year dynamics were assessed by category transition and absolute change (ΔFIB-4). Time-to-event analyses (Kaplan-Meier, Cox models) estimated associations with incident HCC.
Over a median 9.4-year follow-up, 29.9% of patients developed HCC. Baseline FIB-4 > 3.25 significantly predicted HCC [adjusted hazard ratio (aHR) = 2.72; P < 0.001]. At year 3, AFP ≥ 20 ng/mL was independent predictor (aHR = 6.74; P < 0.001). Notably, longitudinal analysis demonstrated that a 3-year absolute increase in FIB-4 (ΔFIB-4 ≥ 1.0) was an independent risk factor (aHR = 2.67; P < 0.001). Stratification by trajectory revealed that the increase group (ΔFIB-4 ≥ +1.0) exhibited the highest 15-year cumulative HCC incidence of 72.7%.
High baseline and rising FIB-4 scores strongly predict HCC. Integrating longitudinal FIB-4 monitoring with AFP assessment is essential for risk stratification and prognosis in CHC.
Core Tip: This retrospective cohort study evaluated 271 treatment-naïve patients with chronic hepatitis C to determine whether baseline and 3-year trajectories of the fibrosis-4 (FIB-4) index and other biomarkers predict long-term hepatocellular carcinoma (HCC) risk. A baseline FIB-4 value > 3.25 strongly predicted HCC independently, while a 3-year ΔFIB-4 ≥ 1 helped risk stratification. Persistently low FIB-4 values identified a group with negligible HCC incidence. Periodic, low-cost monitoring of FIB-4 and alpha-fetoprotein may offer a practical approach to guide surveillance and treatment prioritization in resource-limited settings.
- Citation: Li YL, Tung HD, Chuang TW, Cheng CT, Pang MG, Chen JJ, Zhong KM, Huang TY, Lee PL. Baseline and longitudinal biomarkers predict hepatocellular carcinoma in chronic hepatitis C: A cohort study. World J Gastrointest Oncol 2026; 18(5): 117512
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/117512.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.117512
Hepatitis C virus (HCV) infection remains a significant public health issue globally due to its progression to severe liver complications, including decompensated liver cirrhosis and hepatocellular carcinoma (HCC)[1]. While direct-acting antivirals (DAAs) have revolutionized HCV management with cure rates exceeding 95% across diverse populations[2], the World Health Organization’s goal of eliminating viral hepatitis by 2030 faces substantial hurdles[3]. A significant disparity persists between treatment eligibility and actual initiation, particularly in real-world settings[4]. Global esti
This gap in the “cascade of care” is multifactorial. System-level barriers include economic constraints, limited heal
Serum alpha-fetoprotein (AFP) remains the most commonly used biomarker for HCC screening; however, its specificity is limited, especially in distinguishing HCC from benign liver conditions[11]. The fibrosis-4 (FIB-4) index has emerged as a non-invasive, cost-effective surrogate marker for assessing liver fibrosis severity and stratifying HCC risk in chronic liver disease patients, including those infected with HCV[12]. Specifically, chronic hepatitis C (CHC) patients with a baseline FIB-4 score ≥ 3.25 exhibit a significantly elevated risk of developing HCC, highlighting its potential as a robust risk stratification tool[13]. Furthermore, longitudinal changes in FIB-4 scores have recently gained attention as potential predictors of HCC development in patients who achieved sustained virologic response (SVR) with DAA[14]. However, the prognostic value of dynamic FIB-4 changes in untreated patients remains understudied. In the absence of viral eradication, ongoing inflammation and fibrosis remodeling may manifest as distinct longitudinal biomarker patterns that static scores fail to detect.
Therefore, this study aimed to evaluate the prognostic utility of baseline and longitudinal FIB-4 dynamics in a long-term cohort of treatment-naïve CHC patients. We hypothesized that longitudinal changes in FIB-4 scores (ΔFIB-4) would provide superior risk stratification for HCC compared to static baseline measurements alone. Our findings aim to bridge the knowledge gap in the natural history of HCV and offer evidence for optimizing surveillance strategies in settings where universal DAA coverage is not yet fully realized.
A retrospective analysis was conducted on a cohort of treatment-naïve patients with CHC at Chi Mei Hospital, Liouying from 1999 to 2015. These patients underwent longitudinal follow-up at the Department of Hepatology. Patients were excluded if they had coexisting severe liver diseases such as hepatitis B co-infection, autoimmune hepatitis, alcoholic liver disease, prior HCC history, follow-up less than 3 years without development of HCC and incomplete treatment information. This study was conducted and performed in accordance to the ethical principles for medical research involving human subjects of the Declaration of Helsinki, updated in 2013. It was approved by the Institutional Review Board of the Chi Mei Hospital, Liouying (Approval No. 11412-L02) Patient consent was waived due to the retrospective nature of the study and data anonymization.
The index date was defined as the earliest date of tested positive anti-HCV, and follow-up continued until the first occurrence of HCC, death, loss to follow-up, or the study end date. Data were collected including demographic in
The primary end-point was incident HCC, confirmed either histologically or by characteristic arterial-phase enhan
Baseline characteristics were presented as median (interquartile range) and n (%). The FIB-4 scores were categorized into three groups: < 1.45, 1.45-3.25, and > 3.25 for analysis[16]. For the variables either at baseline or 3-year time point contributing to HCC development, we firstly use univariable Cox regression models to identify possible correlation factors which were shown in hazard ratio (HR) and 95% confidence intervals (95%CIs). Then, clinically relevant variables (including age, sex, diabetes, hypertension, and hyperlipidemia) and those with P < 0.1 in univariable analysis were included in the multivariable Cox proportional hazards regression models which were shown in adjusted HR (aHR) and 95%CIs. Also, we conducted the Kaplan-Meier method to estimate the cumulative incidence of HCC by baseline and 3-year FIB-4 score along with the Log-Rank test for significance comparison.
For the dynamic change of biochemical parameters during follow up, we focused on the 3-year trajectory of the FIB-4 score compared with baseline. Trajectories were defined based on transitions between these FIB-4 categories. An improved trajectory was defined as a shift from > 3.25 to either 1.45-3.25 or < 1.45, or from 1.45-3.25 to < 1.45. A worsened trajectory was defined as a transition from < 1.45 to either 1.45-3.25 or > 3.25, or from 1.45-3.25 to > 3.25. Patients who remained within the same category over the 3-year period were classified as stable. The heterogeneity between each FIB-4 trajectory group was assessed using the Pearson χ2 test.
Absolute change in FIB4 (ΔFIB-4) was defined as the 3-year value minus the baseline value and was analyzed as a continuous variable to preserve information using landmark Cox regression models. To derive an empirical cutpoint for ΔFIB-4, we conducted a grid search across candidate thresholds. Based on this datadriven approach, patients were then further grouped into three categories based on ΔFIB-4: Decrease (ΔFIB-4 ≤ -1.0), stable (-1.0 < ΔFIB-4 < +1.0), and increase (ΔFIB-4 ≥ +1.0) to further evaluate whether improvement in FIB-4 was associated with a lower subsequent HCC risk. The heterogeneity between each FIB-4 trajectory group was assessed using the χ2 test and HR was estimated using Cox regression models. Then, the Kaplan-Meier method was conducted to analyze cumulative incidence of HCC by these three categories and compared using the log-rank test. All statistical analyses were performed with a two-tailed significance threshold of P < 0.05.
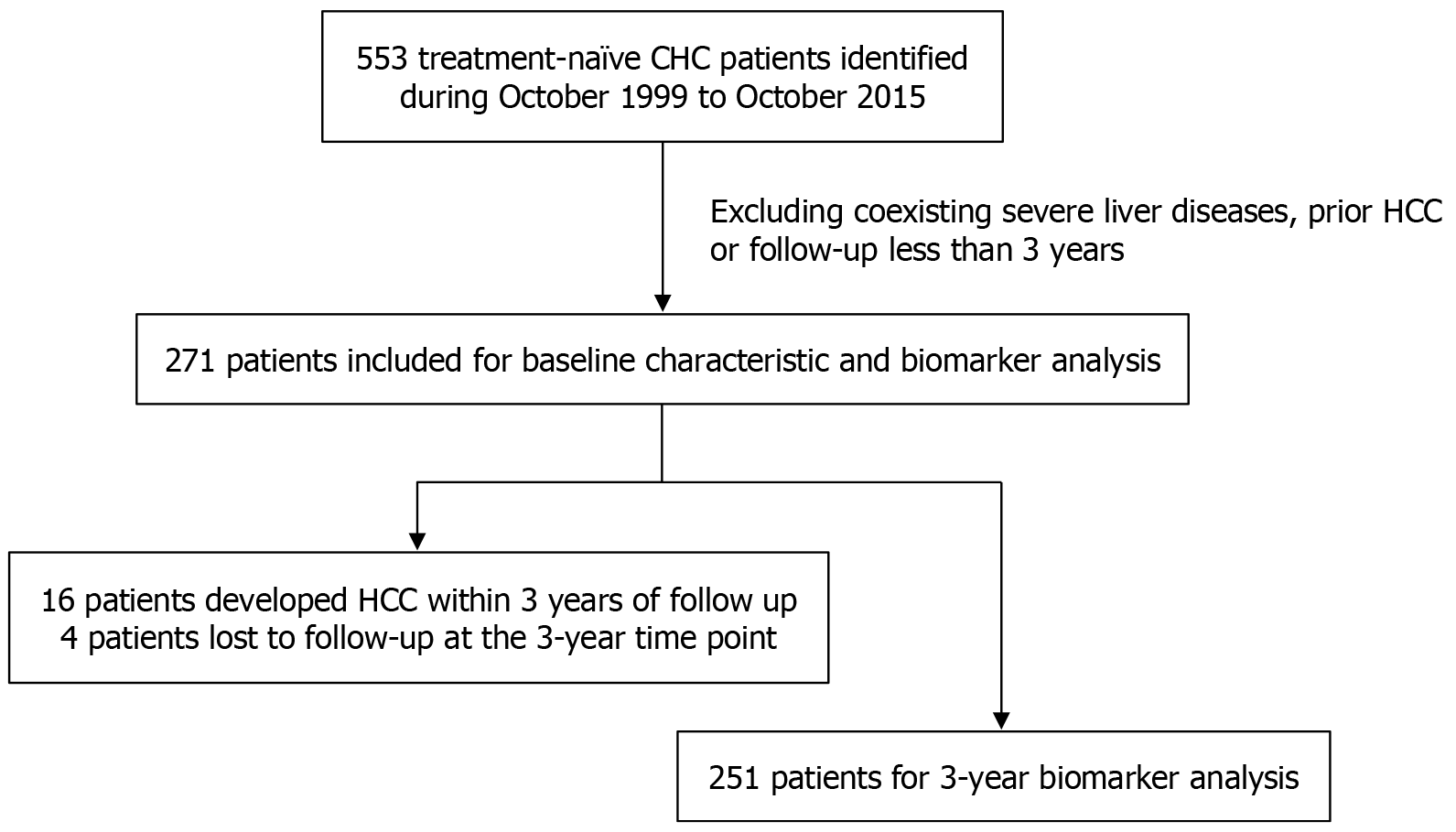
From 1999 to 2015, a total of 553 CHC patients were identified as treatment-naïve patients at Chi Mei Hospital, Liouying. After excluding those meeting exclusion criteria, 271 patients were included in the analysis with a median age of 66.0 (58.0-73.0) years. The 156 patients (57.6%) were female and 76 patients (28.0%) had baseline liver cirrhosis. Other basic characteristics are shown in Table 1. After a median 9.4 years of follow up, 81 patients (29.9%) developed HCC with an interval of 7.45 years. BCLC stages were distributed as: Stage 0: 8 (9.9%), A: 43 (53.1%), B: 10 (12.3%), C: 12 (14.8%), and D: 8 (9.9%). The 16 patients developed HCC within 3 years of follow up and 4 patients lost to follow up at the 3-year time point; therefore, 251 patients were eligible for 3-year biomarkers analysis (Figure 1).
| Parameter | Value |
| Demographics | |
| Age (years, n = 271) | 66.0 (58.0-73.0) |
| Sex (n = 271) | |
| Male | 115 (42.4) |
| Female | 156 (57.6) |
| Follow-up time (years, n = 271) | 9.4 (8.7-10.1) |
| Virological profile | |
| HCV viral load (IU/mL, n = 190) | 941202 (64914-2861279) |
| HCV genotype (n = 136) | |
| Type 1 | 49 (36.0) |
| Type 2 | 48 (35.3) |
| Type 6 | 23 (16.9) |
| Other (mixed) | 16 (11.8) |
| Comorbidities | |
| Hypertension (n = 271) | 139 (51.3) |
| Diabetes mellitus (n = 271) | 86 (31.7) |
| Dyslipidemia (n = 271) | 31 (11.4) |
| Disease severity | |
| Cirrhosis (n = 270) | 76 (28.0) |
| Baseline FIB-4 index (n = 263) | |
| < 1.45 | 35 (13.3) |
| 1.45-3.25 | 93 (35.4) |
| > 3.25 | 135 (51.3) |
In baseline univariable Cox models, older age, higher baseline fibrosis burden and tumor marker levels were associated with subsequent HCC. Compared to patients with baseline FIB-4 1.45-3.25, those with FIB-4 > 3.25 had an increased hazard of HCC (HR = 3.13, 95%CI: 1.84-5.31; P < 0.001), whereas FIB-4 < 1.45 was associated with a lower hazard (HR = 0.12, 95%CI: 0.02-0.89; P = 0.039). Per 1-year increase in age (HR = 1.03, 95%CI: 1.00-1.05; P = 0.033), baseline AFP ≥ 20 ng/mL (HR = 3.31, 95%CI: 1.85-5.92; P < 0.001) and total bilirubin ≥ 1 mg/dL (HR = 3.37, 95%CI: 1.85-6.15; P < 0.001) were also significant predictors.
When these variables were entered into a multivariable model, male sex (aHR = 1.74, 95%CI: 1.05-2.86; P = 0.030), baseline FIB-4 > 3.25 (aHR = 2.72 vs 1.45-3.25, 95%CI: 1.51-4.90; P < 0.001), and AFP ≥ 20 ng/mL (aHR = 2.79, 95%CI: 1.50-5.19; P = 0.001) remained independently associated with increased HCC risk. The comparison of FIB-4 < 1.45 vs 1.45-3.25 was not estimable in this multivariable complete-case dataset due to sparse events in the < 1.45 group after exclusion for missing covariates (Table 2).
| Variable | Univariate | Multivariate | ||
| HR (95%CI) | P value | aHR (95%CI) | P value | |
| Demographics | ||||
| Sex (male) | 1.51 (0.97-2.33) | 0.067 | 1.74 (1.05-2.86) | 0.03 |
| Age (per 1-year increase) | 1.03 (1.00-1.05) | 0.033 | 1.01 (0.98-1.04) | 0.559 |
| Biomarkers | ||||
| HCV RNA > 6 × 106 (IU/mL) | 0.78 (0.28-2.16) | 0.634 | - | - |
| AFP ≥ 20 (ng/mL) | 3.31 (1.85-5.92) | < 0.001 | 2.79 (1.50-5.19) | 0.001 |
| Cr ≥ 1 (mg/dL) | 1.49 (0.87-2.55) | 0.142 | - | - |
| T-Bil ≥ 1 (mg/dL) | 3.37 (1.85-6.15) | < 0.001 | 2.06 (0.89-4.75) | 0.091 |
| PT ≥ 12 (second) | 1.22 (0.56-2.67) | 0.621 | 1.36 (0.48-3.83) | 0.564 |
| Alb < 3.5 (g/dL) | 1.77 (0.80-3.95) | 0.16 | 1.22 (0.38-3.96) | 0.739 |
| Disease severity | ||||
| FIB-4 score | ||||
| 1.45-3.25 | Reference | |||
| < 1.45 | 0.12 (0.02-0.89) | 0.039 | Not estimable1 | |
| > 3.25 | 3.13 (1.84-5.31) | < 0.001 | 2.72 (1.51-4.90) | < 0.001 |
| Comorbidities | ||||
| Diabetes Mellitus | 1.53 (0.98-2.39) | 0.061 | 1.43 (0.84-2.43) | 0.184 |
| Hypertension | 0.90 (0.58-1.40) | 0.646 | 0.62 (0.37-1.06) | 0.079 |
| Dyslipidemia | 0.77 (0.37-1.59) | 0.475 | 0.71 (0.27-1.87) | 0.493 |
For the longitudinal 3-year biomarkers, a sub-analysis was conducted in 251 patients who remained under observation at the 3-year time point. In univariable Cox models, third-year AFP ≥ 20 ng/mL was strongly associated with subsequent HCC (HR = 9.27, 95%CI: 4.80-17.87; P < 0.001). Third-year FIB-4 > 3.25 was also associated with higher risk (HR = 2.75, 95%CI: 1.54-4.93; P = 0.001). Third-year total bilirubin ≥ 1 mg/dL (HR = 2.92, 95%CI: 1.38-6.20; P = 0.005) and creatinine ≥ 1 mg/dL (HR = 1.81, 95%CI: 1.00-3.27; P = 0.049) showed a significant association. Sex and age at year 3 were not statistically significant in univariable landmark analyses.
In the multivariable model, third-year AFP ≥ 20 ng/mL remained independently associated with increased HCC risk (aHR = 6.74, 95%CI: 2.48-18.33; P < 0.001). Although the FIB-4 score > 3.25 showed a trend toward increased risk (aHR = 2.30, 95%CI: 0.75-7.03; P = 0.143), it did not reach statistical significance in the adjusted model. And the third-year FIB-4 < 1.45 was not estimable due to zero events in the category within the complete-case multivariable dataset (Table 3).
| Variable | Univariate | Multivariate | ||
| HR (95%CI) | P value | aHR (95%CI) | P value | |
| Demographics | ||||
| Sex (male) | 1.32 (0.81-2.15) | 0.27 | 1.72 (0.64-4.66) | 0.282 |
| Age (per 1-year increase) | 1.02 (1.00-1.05) | 0.098 | 1.01 (0.96-1.06) | 0.78 |
| Biomarkers | ||||
| AFP ≥ 20 (ng/mL) | 9.27 (4.80-17.87) | < 0.001 | 6.74 (2.48-18.33) | < 0.001 |
| Cr ≥ 1 (mg/dL) | 1.81 (1.00-3.27) | 0.049 | 0.92 (0.35-2.39) | 0.859 |
| T-Bil ≥ 1 (mg/dL) | 2.92 (1.38-6.20) | 0.005 | 2.57 (0.96-6.88) | 0.06 |
| PT ≥ 12 (second) | 1.76 (0.69-4.47) | 0.233 | - | - |
| Alb < 3.5 (g/dL) | 1.70 (0.70-4.16) | 0.243 | - | - |
| Disease severity | ||||
| FIB-4 score | ||||
| 1.45-3.25 | Reference | |||
| < 1.45 | 0.14 (0.02-1.06) | 0.057 | Not estimable1 | |
| > 3.25 | 2.75 (1.54-4.93) | 0.001 | 2.30 (0.75-7.03) | 0.143 |
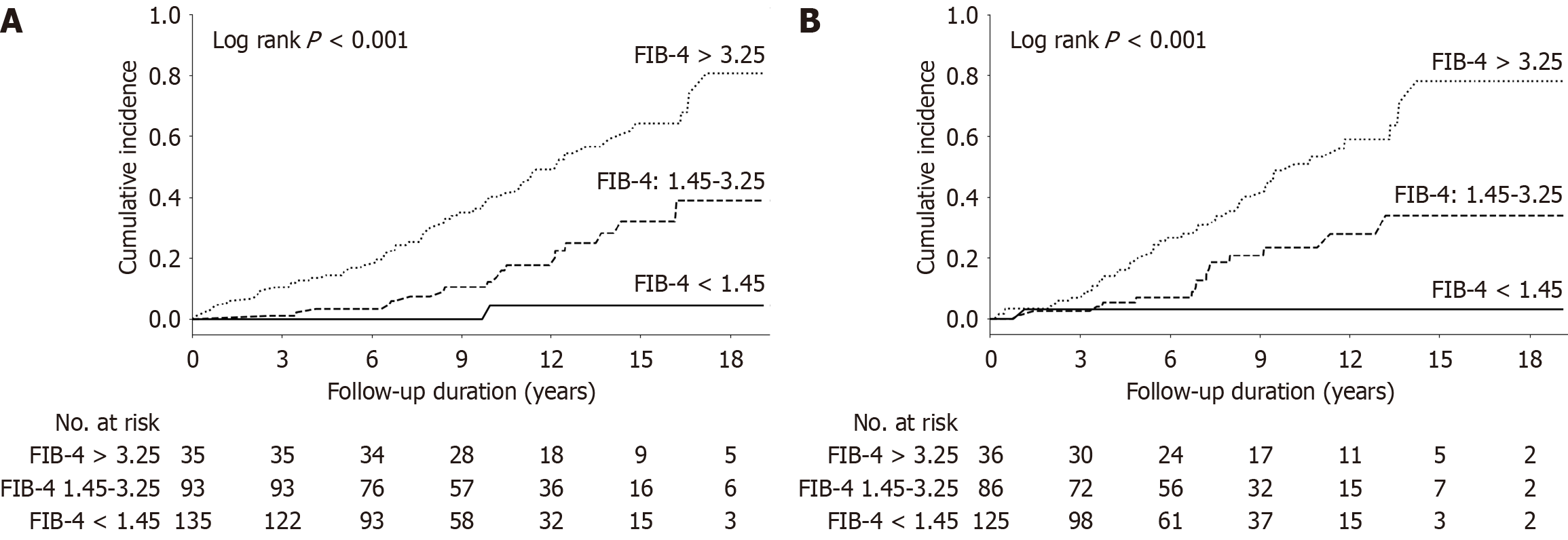
The cumulative incidence of HCC stratified according to different FIB-4 scores either at baseline or 3-year time point was illustrated in Figure 2. At baseline, the 5-, 10-, and 15-year cumulative risk of HCC in patients with FIB-4 index < 1.45 was 0%, 4.3%, and 4.3%, respectively, while they were 3.2%, 12.2%, and 31.9% in patients with FIB-4 index 1.45-3.25, and 14.2%, 40.2%, and 64.2% in those with FIB-4 index > 3.25, respectively. Using the 3-year FIB-4 index stratification, the corresponding 5-, 10-, and 15-year cumulative incidence was 3.0%, 3.0%, and 3.0% for FIB-4 index < 1.45; 7.0%, 23.4%, and 33.9% for FIB-4 index 1.45-3.25; and 19.6%, 48.8%, and 78.1% for FIB-4 > 3.25. Both log-rank tests showed clinical significance with P < 0.001.
For the impact of longitudinal fibrosis progression on HCC risk, patients were stratified into three groups based on their 3-year FIB-4 score trajectory: Improved (n = 39), stable (n = 159), and worsened (n = 45). The respective incidences of HCC were 20.5%, 28.9%, and 20.0%. The χ2 test for trend across the three groups was P = 0.376, indicating HCC across the three trajectory groups having no difference. When further subdividing the stable group into three FIB-4 strata: Stable FIB-4 < 1.45, stable FIB-4 1.45-3.25, and stable FIB-4 > 3.25, resulting in five comparison groups overall. The incidences of HCC were 0% for stable FIB4 < 1.45, 15.2% for stable FIB4 1.45-3.25, and 42.4% for stable FIB4 > 3.25. The P value of five-level Pearson χ2 test across these groups was P < 0.001. Using stable FIB-4 1.45-3.25 as the reference, patients with FIB-4 remaining > 3.25 had 4.1-fold increased risk of HCC (95%CI: 1.66-10.13, P = 0.002). Whereas, FIB-4 staying < 1.45 appeared protective but this did not reach statistical significance (HR = 0.12, 95%CI: 0.01-2.25, P = 0.157). Improved and worsened trajectories did not differ significantly from the reference group (Table 4).
| HCC cases (n) | Incidence (%) | HR (95%CI) | P value | |
| Stable 1.45-3.25 | 7/46 | 15.2 | Reference | - |
| Stable < 1.45 | 0/21 | 0 | 0.12 (0.01-2.25) | 0.157 |
| Stable > 3.25 | 39/92 | 42.4 | 4.10 (1.66-10.13) | 0.002 |
| Improved | 8/39 | 20.5 | 1.44 (0.47-4.40) | 0.525 |
| Worsened | 9/45 | 20 | 1.39 (0.47-4.13) | 0.55 |
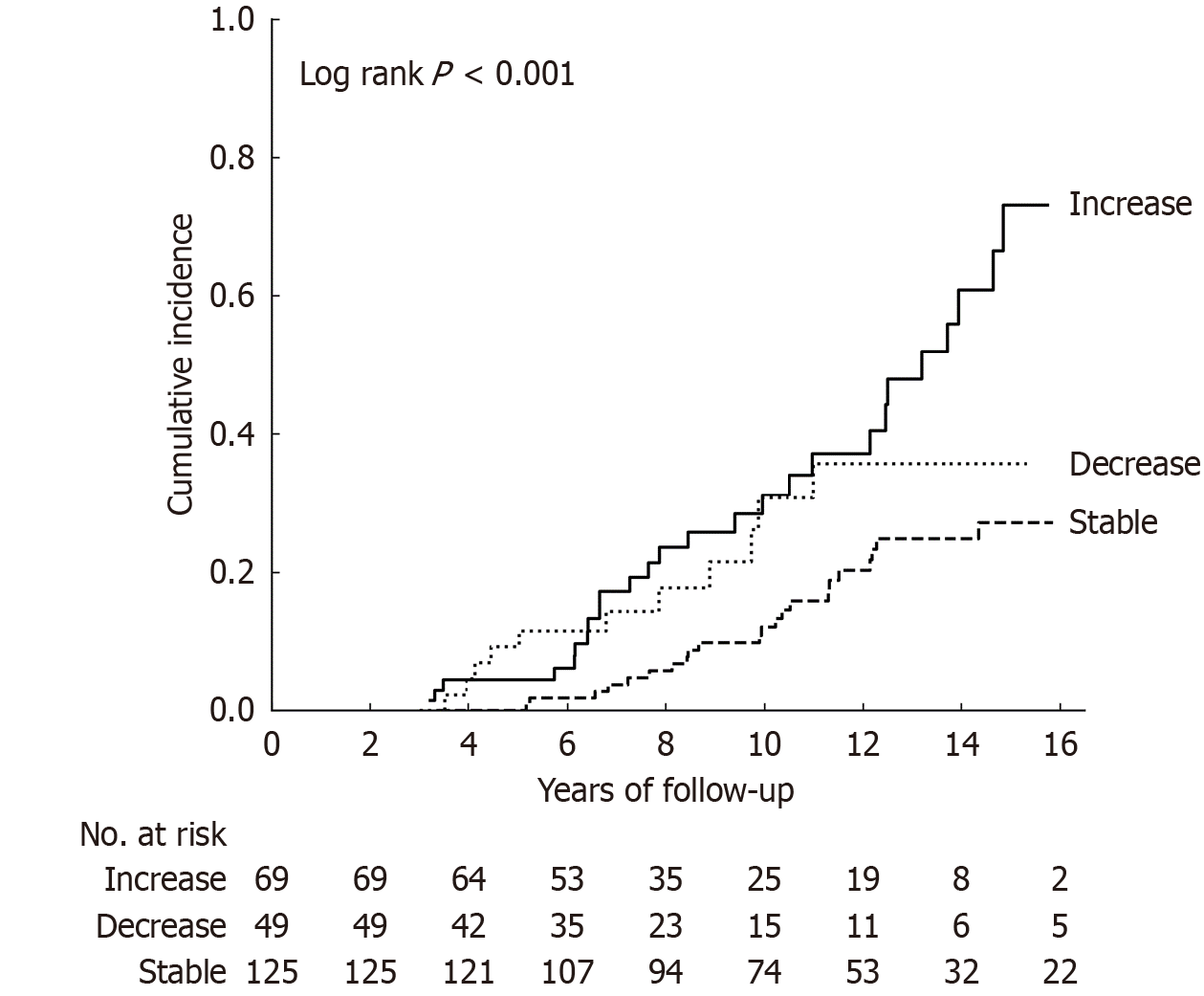
In the absolute change of value in FIB-4 score, using landmark Cox proportional hazards model for events occurring after year 3, each 1-point increase in ΔFIB-4 was associated with a significantly higher risk of subsequent HCC de
In this retrospective cohort study of 271 treatment-naïve CHC patients, both baseline FIB-4 burden and AFP level were strongly associated with subsequent HCC, and these signals remained robust when reassessed at year 3 using a landmark design. Although year3 FIB4 categories alone did not significantly predict incident HCC, analyses of FIB4 trajectories demonstrated that patients with worsening fibrosis over three years had a substantially higher risk of HCC compared with those whose FIB4 scores remained stable. These findings underscore the importance of not only initial biomarker and baseline characteristic assessment but also continuous monitoring for identifying high risk patients. Accordingly, patients presented with a high baseline FIB-4 index or evidence of worsening FIB-4 scores during follow-up warrant intensified surveillance for HCC.
In the modern era, DAAs have been approved worldwide for the treatment of HCV to improve clinical outcomes and reduce transmission[17]. A notable characteristic of our cohort is the treatment-naïve patients managed under long-term follow-up. This observation must be interpreted within the historical context of the study data collection period (1999-2015), which predates the era of universal DAA reimbursement in Taiwan[18]. Many patients in our cohort declined treatment due to advanced age or fear of severe side effects, opting instead for a “watchful waiting” strategy. These patient-level factors align with well-documented barriers to HCV therapy[9,10]. Consequently, this study captures a unique “natural history” cohort unconfounded by antiviral therapy, offering valuable insights into fibrosis progression and HCC risk that are increasingly difficult to observe in the modern DAA era.
Consistent with established literature identifying older age, male sex, metabolic comorbidities, and coinfections as key risks of hepatocarcinogenesis[19,20], our multivariate analysis confirmed that male sex acts as an independent predictor of HCC. However, the most potent driver of risk in our cohort was liver fibrosis burden. The strong association between a baseline FIB-4 > 3.25 and HCC development likely reflects a dual mechanism: It serves not only as a surrogate for advanced fibrosis/cirrhosis but also indicates a biologically permissive hepatic microenvironment that favors carcinogenesis[21]. Our findings align with prior studies linking elevated FIB-4 to HCC risk and liver-related mortality, extending these observations to a treatment-naïve natural history cohort[13,22-24]. Our results demonstrate a clear, stepwise gradient in long-term outcomes, with 5-, 10-, and 15-year cumulative HCC incidence progressively increasing across the < 1.45, 1.45-3.25, and > 3.25 tiers. Notably, patients with a baseline FIB-4 > 3.25 exhibited a 15-year cumulative incidence that was 15-fold higher than those with FIB-4 < 1.45 (which remained low at approximately 4%). Complementing the fibrosis-driven risk, an AFP threshold of 20 ng/mL is widely used in surveillance algorithms and has been shown to be associated with HCC risk[25]; this threshold remained a significant predictor of HCC in our analysis. This distinct stratification supports a risk-adapted surveillance strategy: Patients with baseline FIB-4 below 1.45 may be candidates for less-intensive monitoring, whereas those with FIB-4 > 3.25 or elevated AFP warrant rigorous follow-up.
Longitudinal monitoring of albumin, AFP, FIB-4 index, and liver stiffness provides better risk prediction than baseline-only measurements in patients with HCV who achieved SVR with DAA[14,26]. These investigations have employed landmark intervals ranging from one to three years for biomarkers follow-up and HCC risk assessment[14,27]. In our 3-year landmark analysis, the vast majority of participants had complete data at the threeyear timepoint, preserving statistical power; moreover, the median interval from enrolment to HCC diagnosis was 7.45 years, making year 3 a clinically relevant intermediate point for pre-HCC biomarker review. We excluded patients with less than 3 years of follow-up who remained HCC-free; this exclusion criterion was methodologically necessary to ensure valid data points for calculating longitudinal dynamic changes (ΔFIB-4), aligning with established methodologies in previous study focusing on HCC risk stratification in treatment-naïve CHC patients[13]. Indeed, on univariate analysis a three-year FIB-4 > 3.25 was associated with a significantly higher HCC risk. After multivariable adjustment, only AFP ≥ 20 ng/mL retained independent significance. Even so, the cumulative incidence gradient across FIB-4 strata persisted to 15 years (78% in > 3.25 vs 3% in < 1.45), aligning with the concept that the duration of advanced fibrosis and cirrhosis are domi
The variation of FIB-4 score from baseline is also informative and can serve as valuable indicators for stratifying HCC risk in patients with CHC receiving treatment of DAAs[29]. A prospective study conducted in South Korea reported that CHC patients, regardless of treatment status, who exhibited a ΔFIB-4 index/year ≥ 0.5, were at an increased risk of cir
The longitudinal fibrosis dynamics by category transitions dividing into five groups confirmed that persistently high FIB-4, consistent with advanced fibrosis, dominates subsequent cancer risk. In contrast, those who remained FIB-4 < 1.45 over three years experienced no HCC events. These findings indicate that serial assessments of the FIB4 index may serve as a prognostic marker for identifying a highrisk cohort HCC, who could benefit from more intensive surveillance strategies[33]. Whereas the FIB-4 index remaining < 1.45 over three years identifies a low-risk subset suitable for less intensive surveillance intervals. Taken together, the magnitude of change of the FIB-4 index and between group change are both informative for risk assessment of HCC. This correlation is explained by fibrosis progression, which, even in the absence of overt clinical cirrhosis, involves excessive accumulation of extracellular matrix proteins causing chronic liver injury, thereby inducing oncogenic changes in the hepatic microenvironment[21].
This study demonstrates that serial changes in FIB-4, along with longitudinal biomarker follow-up, provide prognostic information independent of baseline fibrosis stage. The long follow-up in a treatment-naïve cohort captured incident HCC events and therefore reflects the natural history of CHC before exposure to antiviral therapy. From a clinical standpoint, our data support a risk-adapted approach: (1) Patients with FIB-4 persistently > 3.25 and/or ΔFIB-4 ≥ 1.0 over three years should be prioritized for antiviral therapy (where applicable) and intensified HCC surveillance; (2) Those persistently < 1.45 appear to have very low risk and may be suitable for less-intensive surveillance intervals; and (3) Individuals in the mid-tier (1.45-3.25) warrant continued monitoring given their non-trivial long-term risk. Because FIB-4 and AFP are low-cost and widely available[34], this strategy is particularly attractive in settings with limited DAA up
There are limitations that should be noted. First, we acknowledge the potential for selection bias and immortal time bias inherent in excluding patients with less than 3 years of follow-up who remained HCC-free. However, as noted, this was necessary to construct the dynamic model. Therefore, our findings should be interpreted as reflecting risk profiles specifically among patients who survive the initial 3-year natural history phase. Second, the patient cohort was derived from a single center, which may introduce regional selection bias. Third, the optimal cut-off values for longitudinal ΔFIB-4 were identified using a data-driven approach within the same study cohort. Given the exploratory nature of this study and the rarity of long-term untreated cohorts, our primary aim was to demonstrate the concept that dynamic changes matter, rather than to establish a definitive clinical threshold. Therefore, the specific ΔFIB-4 cut-offs proposed here should be regarded as preliminary. Further validation in independent, larger, and multi-center cohorts is essential to confirm their generalizability and clinical applicability before routine implementation.
In conclusion, our findings demonstrate that the longitudinal assessment of non-invasive fibrosis scores, specifically the dynamic worsening of FIB-4, combined with AFP offers additional prognostic value to baseline measures alone. This accessible and cost-effective strategy is particularly valuable in resource-limited settings or regions where universal DAA coverage remains restricted. By identifying treatment-naïve patients with worsening fibrosis trajectories, clinicians can better prioritize scarce resources for intensified surveillance and therapeutic intervention, ultimately optimizing long-term outcomes in this vulnerable population.
| 1. | Westbrook RH, Dusheiko G. Natural history of hepatitis C. J Hepatol. 2014;61:S58-S68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 696] [Cited by in RCA: 626] [Article Influence: 52.2] [Reference Citation Analysis (1)] |
| 2. | Asselah T, Marcellin P, Schinazi RF. Treatment of hepatitis C virus infection with direct-acting antiviral agents: 100% cure? Liver Int. 2018;38 Suppl 1:7-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 140] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 3. | Hutin YJ, Bulterys M, Hirnschall GO. How far are we from viral hepatitis elimination service coverage targets? J Int AIDS Soc. 2018;21 Suppl 2:e25050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 4. | Cui F, Blach S, Manzengo Mingiedi C, Gonzalez MA, Sabry Alaama A, Mozalevskis A, Séguy N, Rewari BB, Chan PL, Le LV, Doherty M, Luhmann N, Easterbrook P, Dirac M, de Martel C, Nayagam S, Hallett TB, Vickerman P, Razavi H, Lesi O, Low-Beer D. Global reporting of progress towards elimination of hepatitis B and hepatitis C. Lancet Gastroenterol Hepatol. 2023;8:332-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 228] [Article Influence: 76.0] [Reference Citation Analysis (5)] |
| 5. | Stroffolini T, Stroffolini G. Prevalence and Modes of Transmission of Hepatitis C Virus Infection: A Historical Worldwide Review. Viruses. 2024;16:1115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 6. | Polaris Observatory HCV Collaborators. Global change in hepatitis C virus prevalence and cascade of care between 2015 and 2020: a modelling study. Lancet Gastroenterol Hepatol. 2022;7:396-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 497] [Cited by in RCA: 446] [Article Influence: 111.5] [Reference Citation Analysis (5)] |
| 7. | Marshall AD, Willing AR, Kairouz A, Cunningham EB, Wheeler A, O'Brien N, Perera V, Ward JW, Hiebert L, Degenhardt L, Hajarizadeh B, Colledge S, Hickman M, Jawad D, Lazarus JV, Matthews GV, Scheibe A, Vickerman P, Dore GJ, Grebely J; Global HCV and HIV Treatment Restrictions Group. Direct-acting antiviral therapies for hepatitis C infection: global registration, reimbursement, and restrictions. Lancet Gastroenterol Hepatol. 2024;9:366-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 52] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 8. | Hiebert-Suwondo L, Manning J, Tohme RA, Buti M, Kondili LA, Spearman CW, Hajarizadeh B, Turnier V, Lazarus JV, Grebely J, Dore GJ, Waked I, Ward JW; National Hepatitis Elimination Profile Collaborators. A 2024 global report on national policies, programmes, and progress towards hepatitis C elimination: findings from 33 hepatitis elimination profiles. Lancet Gastroenterol Hepatol. 2025;10:685-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 9. | McGowan CE, Fried MW. Barriers to hepatitis C treatment. Liver Int. 2012;32 Suppl 1:151-156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 141] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 10. | Yang X, Xu Z, Mei Y, Lin G, Wei J, Shao X, Luo Q, Zhao Z, Lin C, Zhang X. Barriers to hepatitis C virus treatment in Guangdong Province. Ann Palliat Med. 2019;8:640-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Cabral LKD, Grisetti L, Pratama MY, Tiribelli C, Pascut D. Biomarkers for the Detection and Management of Hepatocellular Carcinoma in Patients Treated with Direct-Acting Antivirals. Cancers (Basel). 2022;14:2700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Xu XL, Jiang LS, Wu CS, Pan LY, Lou ZQ, Peng CT, Dong Y, Ruan B. The role of fibrosis index FIB-4 in predicting liver fibrosis stage and clinical prognosis: A diagnostic or screening tool? J Formos Med Assoc. 2022;121:454-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 13. | Chang SH, Su TH, Ling ZM, Lee MH, Liu CJ, Chen PJ, Yang HC, Liu CH, Chen CL, Tseng TC, Chen CH, Lee HS, Chen CJ, Kao JH. Fibrosis-4 index stratifies risks of hepatocellular carcinoma in patients with chronic hepatitis C. J Formos Med Assoc. 2024;123:1154-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 14. | Kramer JR, Cao Y, Li L, Smith D, Chhatwal J, El-Serag HB, Kanwal F. Longitudinal Associations of Risk Factors and Hepatocellular Carcinoma in Patients With Cured Hepatitis C Virus Infection. Am J Gastroenterol. 2022;117:1834-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 15. | Forner A, Reig ME, de Lope CR, Bruix J. Current strategy for staging and treatment: the BCLC update and future prospects. Semin Liver Dis. 2010;30:61-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 843] [Article Influence: 52.7] [Reference Citation Analysis (7)] |
| 16. | Albhaisi S, Sun J, Sanyal AJ. Fibrosis-4 index is associated with the risk of hepatocellular carcinoma in patients with cirrhosis and nonalcoholic steatohepatitis. Front Oncol. 2023;13:1198871. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 17. | Venkatesh R, Huang AS, Gurmessa K, Hsu EB. Understanding Barriers to Hepatitis C Antiviral Treatment in Low-Middle-Income Countries. Healthcare (Basel). 2024;13:43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 18. | Yen YH, Chen CH, Hung CH, Wang JH, Lu SN, Kee KM, Hu TH. Active hepatocellular carcinoma is an independent risk factor of direct-acting antiviral treatment failure: A retrospective study with prospectively collected data. PLoS One. 2019;14:e0222605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Wang J, Qiu K, Zhou S, Gan Y, Jiang K, Wang D, Wang H. Risk factors for hepatocellular carcinoma: an umbrella review of systematic review and meta-analysis. Ann Med. 2025;57:2455539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 25] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 20. | Ioannou GN, Green P, Lowy E, Mun EJ, Berry K. Differences in hepatocellular carcinoma risk, predictors and trends over time according to etiology of cirrhosis. PLoS One. 2018;13:e0204412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 85] [Article Influence: 10.6] [Reference Citation Analysis (4)] |
| 21. | Dhar D, Baglieri J, Kisseleva T, Brenner DA. Mechanisms of liver fibrosis and its role in liver cancer. Exp Biol Med (Maywood). 2020;245:96-108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 328] [Article Influence: 54.7] [Reference Citation Analysis (0)] |
| 22. | Ogawa E, Chien N, Kam L, Yeo YH, Ji F, Huang DQ, Cheung R, Nguyen MH. Association of Direct-Acting Antiviral Therapy With Liver and Nonliver Complications and Long-term Mortality in Patients With Chronic Hepatitis C. JAMA Intern Med. 2023;183:97-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 75] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 23. | Li X, Xu H, Gao P. Fibrosis Index Based on 4 Factors (FIB-4) Predicts Liver Cirrhosis and Hepatocellular Carcinoma in Chronic Hepatitis C Virus (HCV) Patients. Med Sci Monit. 2019;25:7243-7250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 24. | Ghoneim S, Butt MU, Trujillo S, Asaad I. FIB-4 Regression With Direct-Acting Antiviral Therapy in Patients With Hepatitis C Infection: A Safety-Net Hospital Experience. Front Med (Lausanne). 2020;7:359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | El-Serag HB, Kanwal F, Davila JA, Kramer J, Richardson P. A new laboratory-based algorithm to predict development of hepatocellular carcinoma in patients with hepatitis C and cirrhosis. Gastroenterology. 2014;146:1249-55.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 149] [Article Influence: 12.4] [Reference Citation Analysis (4)] |
| 26. | Azzi J, Dorival C, Cagnot C, Fontaine H, Lusivika-Nzinga C, Leroy V, De Ledinghen V, Tran A, Zoulim F, Alric L, Gournay J, Bronowicki JP, Decaens T, Riachi G, Mikhail N, Soliman R, Shiha G, Pol S, Carrat F, Ganne-Carrié N; ANRS-AFEF Hepather Study group. Prediction of hepatocellular carcinoma in Hepatitis C patients with advanced fibrosis after sustained virologic response. Clin Res Hepatol Gastroenterol. 2022;46:101923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Ioannou GN, Green PK, Beste LA, Mun EJ, Kerr KF, Berry K. Development of models estimating the risk of hepatocellular carcinoma after antiviral treatment for hepatitis C. J Hepatol. 2018;69:1088-1098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 134] [Article Influence: 16.8] [Reference Citation Analysis (4)] |
| 28. | Saviano A, Tripon S, Baumert TF. FIB-4 score and hepatocellular carcinoma risk after hepatitis C virus cure: time to revise surveillance? Hepatobiliary Surg Nutr. 2020;9:661-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Wang HW, Zeng YS, Huang CF, Chen CY, Kuo HT, Tseng KC, Mo LR, Cheng PN, Tai CM, Hung CH, Lo CC, Chen CH, Lee PL, Yang CC, Chen CT, Lin CY, Hsieh TY, Chong LW, Lin CL, Hu JT, Yang SS, Kao JH, Liu CJ, Chuang WL, Huang JF, Yeh ML, Dai CY, Huang YH, Lin HC, Bair MJ, Wang SJ, Huang CW, Tsai MC, Wang CC, Su WW, Lin CW, Lin CL, Chu CH, Yu ML, Peng CY; TACR investigators. Posttreatment FIB-4 score change predicts hepatocellular carcinoma in chronic hepatitis C patients: Findings from the Taiwan hepatitis C registry program. J Formos Med Assoc. 2025;S0929-6646(25)00056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 30. | Ok KS, Jeong SH, Jang ES, Kim YS, Lee YJ, Kim IH, Cho SB, Bae SH, Lee HC. The clinical outcomes of chronic hepatitis C in South Korea: A prospective, multicenter cohort study. Medicine (Baltimore). 2016;95:e4594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 31. | Tamaki N, Kurosaki M, Tanaka K, Suzuki Y, Hoshioka Y, Kato T, Yasui Y, Hosokawa T, Ueda K, Tsuchiya K, Nakanishi H, Itakura J, Asahina Y, Izumi N. Noninvasive estimation of fibrosis progression overtime using the FIB-4 index in chronic hepatitis C. J Viral Hepat. 2013;20:72-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 32. | Zou Y, Yue M, Jia L, Wang Y, Chen H, Wang Y, Zhang M, Feng Y, Yu R, Yang S, Huang P. Repeated Measurement of FIB-4 to Predict Long-Term Risk of HCC Development Up to 10 Years After SVR. J Hepatocell Carcinoma. 2022;9:1433-1443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Tamaki N, Kurosaki M, Yasui Y, Mori N, Tsuji K, Hasebe C, Joko K, Akahane T, Furuta K, Kobashi H, Kimura H, Yagisawa H, Marusawa H, Kondo M, Kojima Y, Yoshida H, Uchida Y, Loomba R, Izumi N. Change in Fibrosis 4 Index as Predictor of High Risk of Incident Hepatocellular Carcinoma After Eradication of Hepatitis C Virus. Clin Infect Dis. 2021;73:e3349-e3354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 34. | Marsh TL, Johnston JM, Homan C, Townshend-Bulson LJ, Kim NJ, VoPham T, Li X, He Q, McMahon BJ, Ioannou GN, Feng Z. HCC surveillance in hepatitis C: A longitudinal algorithm improves alpha-fetoprotein screening. Hepatol Commun. 2025;9:e0719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |