Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.116776
Revised: January 27, 2026
Accepted: March 30, 2026
Published online: May 15, 2026
Processing time: 172 Days and 18.4 Hours
Pancreatoblastoma is a rare malignant epithelial tumor of the pancreas, predominantly affecting children. However, the prognosis is significantly poorer in adults, who typically present with liver metastasis. The management of adult pancreatoblastoma with liver metastases poses a significant challenge, and the median survival remains poor.
A 49-year-old male was incidentally found to have a pancreatic body/tail mass and liver metastasis during routine health screening. He underwent distal pancreatectomy, splenectomy, and hepatectomy, with pathology confirming pancreatoblastoma. Over a 7-year follow-up, the patient experienced multiple liver recur
This case outlines the positron emission tomography/computed tomography presentation of and therapeutic strategies for pancreatoblastoma with liver metastases.
Core Tip: We report the 7-year follow-up of an adult pancreatoblastoma patient with liver metastases, emphasizing the importance of multimodal management, including repeated surgeries and targeted therapy. The case highlights the diagnostic utility of positron emission tomography/computed tomography and identifies a novel fibroblast growth factor receptor 1-synaptonemal complex protein 1 gene rearrangement, suggesting a potential target for future therapeutic strategies.
- Citation: Tang SG, Wei XL, Ye RJ, Xu HQ, Shang SH, Han YX, Wu LW, Xia WZ. Successful seven-year management of recurrent adult pancreatoblastoma with liver metastasis: A case report. World J Gastrointest Oncol 2026; 18(5): 116776
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/116776.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.116776
Pancreatoblastoma is a rare malignant tumor of the pancreas with an estimated annual incidence of 0.004 per 100000 individuals. Pancreatoblastoma may be associated with genetic syndromes such as Beckwith-Wiedemann syndrome or familial adenomatous polyposis[1-3]. Although it predominantly occurs in pediatric patients[4], approximately one-third of reported cases involve adults[5,6]. Adult patients exhibit a poor prognosis, with a median survival of only 17 months post-diagnosis[7]. Approximately 17%-35% of patients present with metastases at diagnosis, most commonly involving the liver[3,8]. The symptoms of pancreatoblastoma are nonspecific, including abdominal pain, abdominal mass, diarrhea, and nausea[4,9]. Some laboratory values may increase, including alpha-fetoprotein, α1-antitrypsin, and lactate dehydrogenase, which are important auxiliary diagnostic clues[9]. However, not all cases show an increase in these values[4]. Clinically, pancreatoblastoma is diagnosed through a combination of patient symptoms, physical signs, laboratory tests, and imaging examinations. Nevertheless, histopathological examination remains the gold standard for diagnosing pancreatoblastoma, characterized by features of epithelial differentiation (acinar, glandular, and trabecular architectures) and the presence of squamous corpuscles[10]. Current treatment options include surgical resection, chemotherapy, ra
A 49-year-old male was admitted following incidental detection of pancreatic body/tail and liver masses 7 years ago.
The patient was incidentally found to have a pancreatic mass during routine health screening and was asymptomatic, with no reports of abdominal pain, nausea, vomiting, or jaundice. Magnetic resonance imaging (MRI) demonstrated a mass in the pancreatic body and tail, and a metastatic liver mass in the left lobe.
Physical examination revealed no abnormalities.
Tumor markers and liver function parameters were within normal ranges, as follows: (1) Carcinoembryonic antigen: 0.86 ng/mL (0.00-5.00 ng/mL); (2) Alpha-fetoprotein: 1.91 ng/mL (0.00-9.00 ng/mL); (3) Carbohydrate antigen 125: 7.85 U/mL (0.00-35.00 U/mL); (4) Carbohydrate antigen 199: 7.82 U/mL (0.00-27.00 U/mL); (5) Total bilirubin: 17.5 μmol/L (7.0-19.0 μmol/L); (6) Albumin: 42.5 g/L (40.0-55.0 g/L); (7) Aspartate aminotransferase: 17 U/L (15-40 U/L); and (8) Alanine aminotransferase: 14 U/L (9-50 U/L).
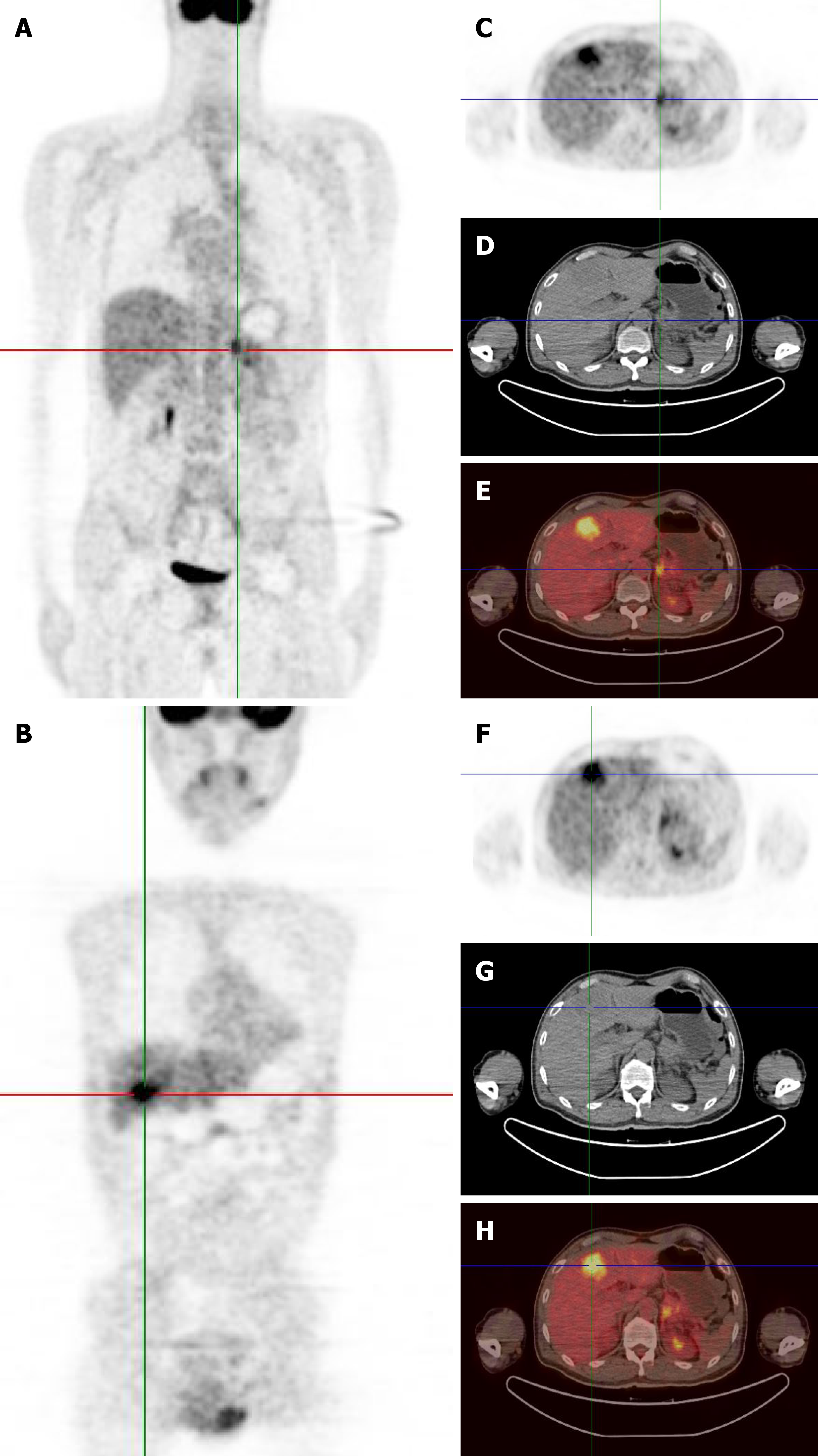
Preoperative 18F-FDG PET/CT maximum intensity projection images revealed intense uptake of 18F-fluorodeoxyglucose (18F-FDG) in the pancreatic body and tail (Figure 1A) and liver segment 4 (Figure 1B). The positron emission tomography (PET), CT, and fused PET/CT images showed a contrast-enhanced round mass of 4.4 cm × 3.4 cm in the pancreatic body and tail, encircling the splenic artery and vein (Figure 1C-E). Additionally, the PET, CT, and fused PET/CT images revealed a liver metastasis of 4.7 cm × 4.2 cm in segment 4 (Figure 1F-H).
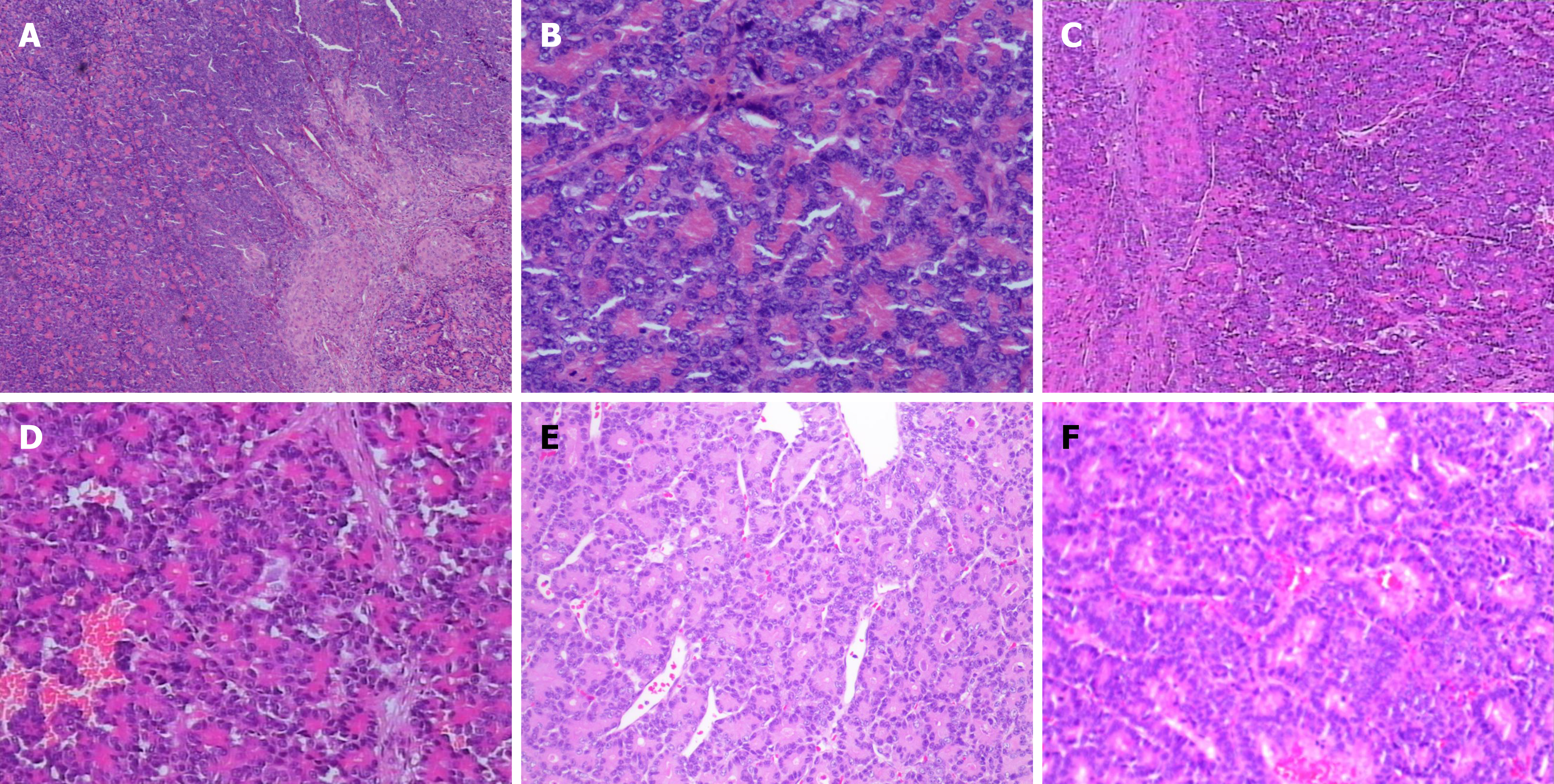
The initial diagnosis was pancreatic cancer with liver metastasis. Postoperative pathology (Figure 2A and B) initially indicated a highly proliferative neuroendocrine tumor of the pancreas with liver metastasis. Intriguingly, immunohistochemistry confirmed the diagnosis as pancreatoblastoma with liver metastasis. Immunophenotype and special staining results were as follows: (1) Liver tumor: Trypsin, partially positive (++); chymotrypsin, partially positive (++); synaptophysin, mostly positive (+++); Ki67, approximately 30% positive; CD56, a small subset of cells were positive (++); CK7, only a minority of cells were positive (+); β-catenin, nuclear positivity; and (2) Pancreatic tumor: Trypsin, partially posi
The patient underwent distal pancreatectomy, splenectomy, cholecystectomy, and hepatectomy. Given the propensity for recurrence, the patient received four cycles of chemotherapy (S-1 + oxaliplatin), initiated 2 months postoperatively.
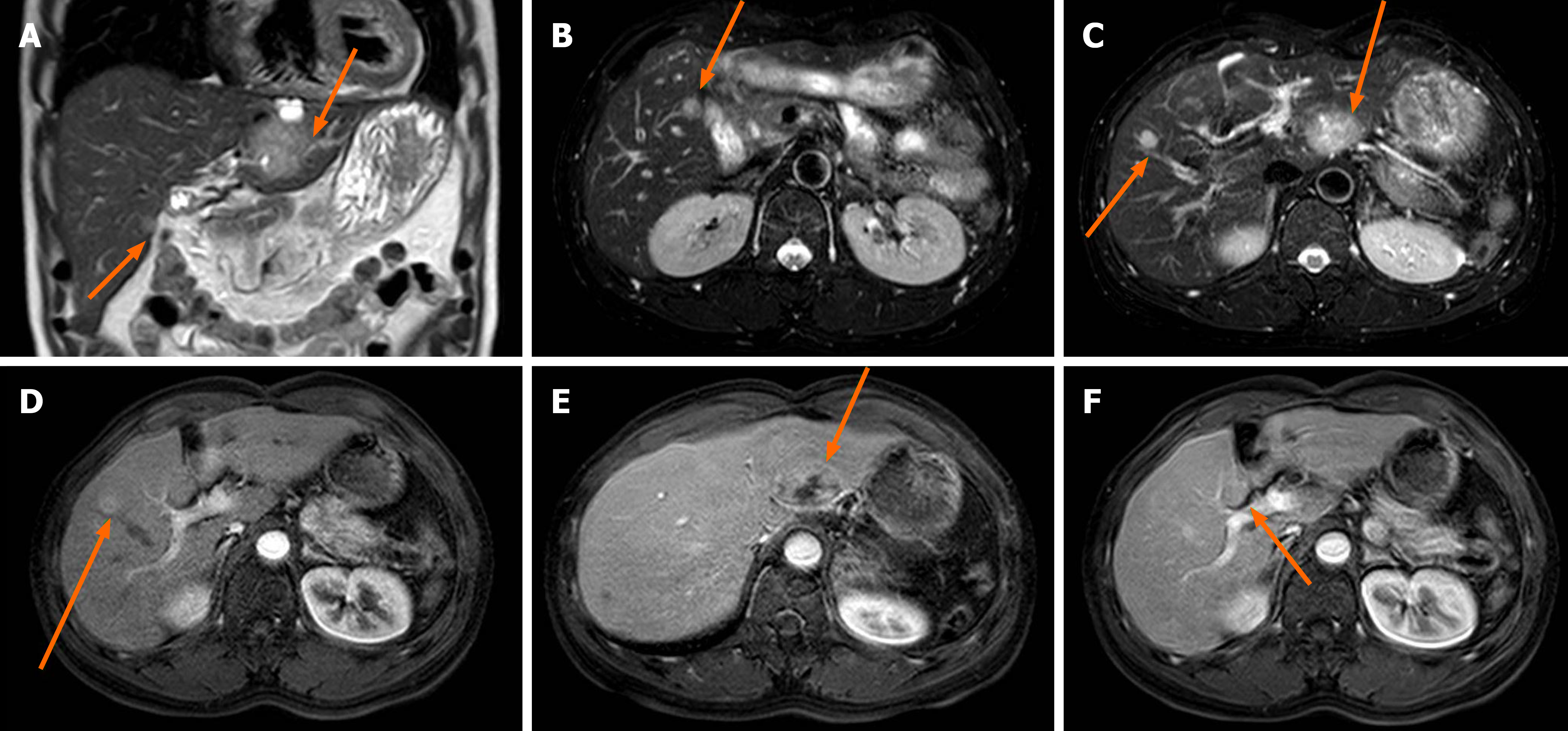
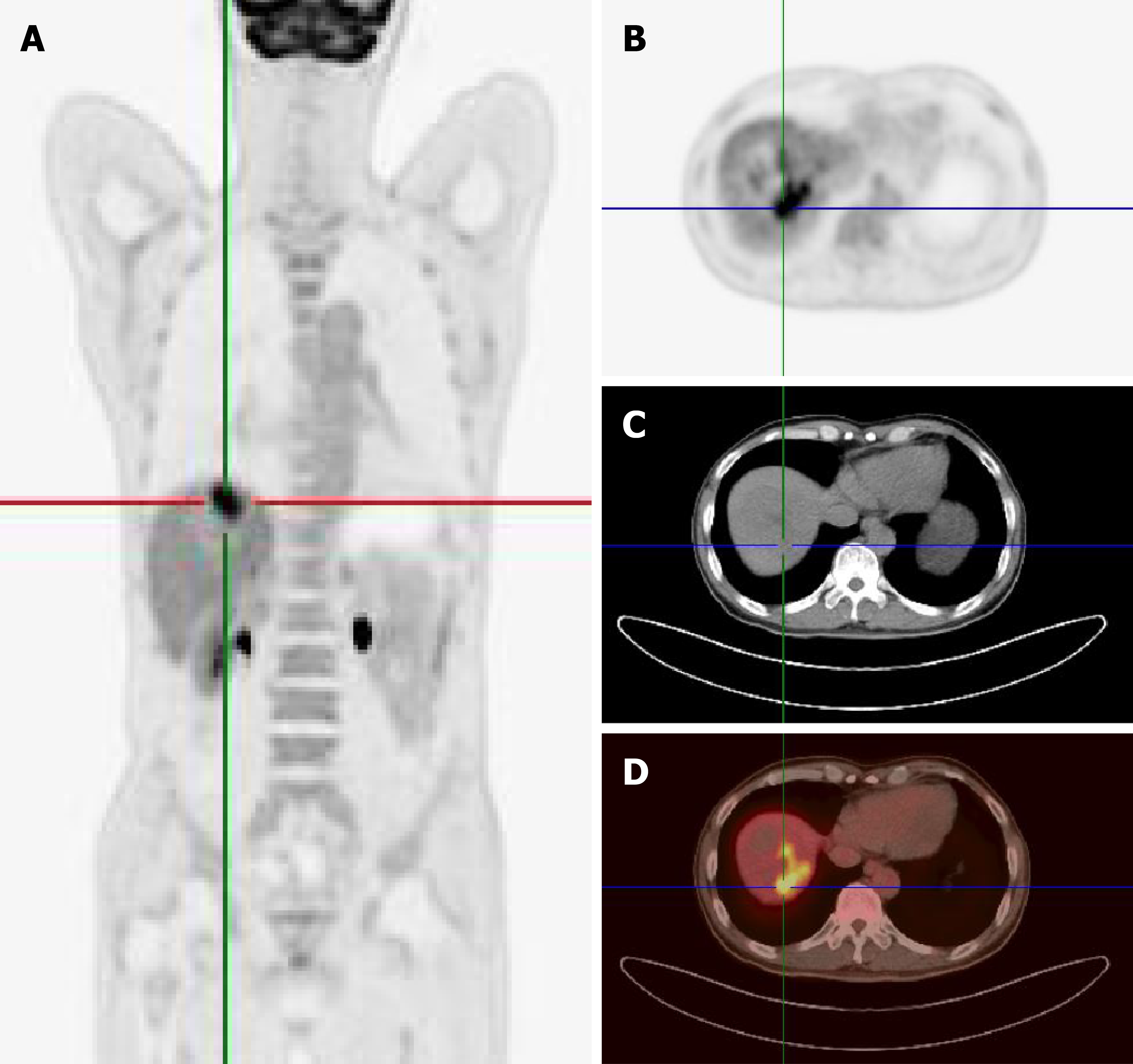
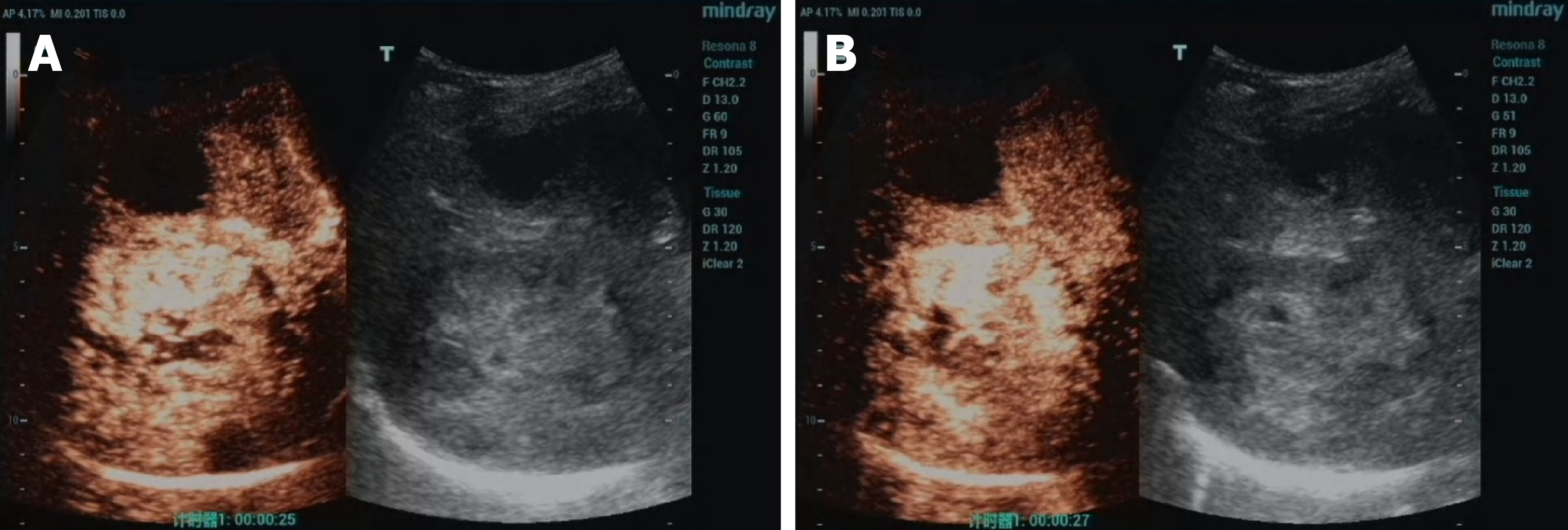
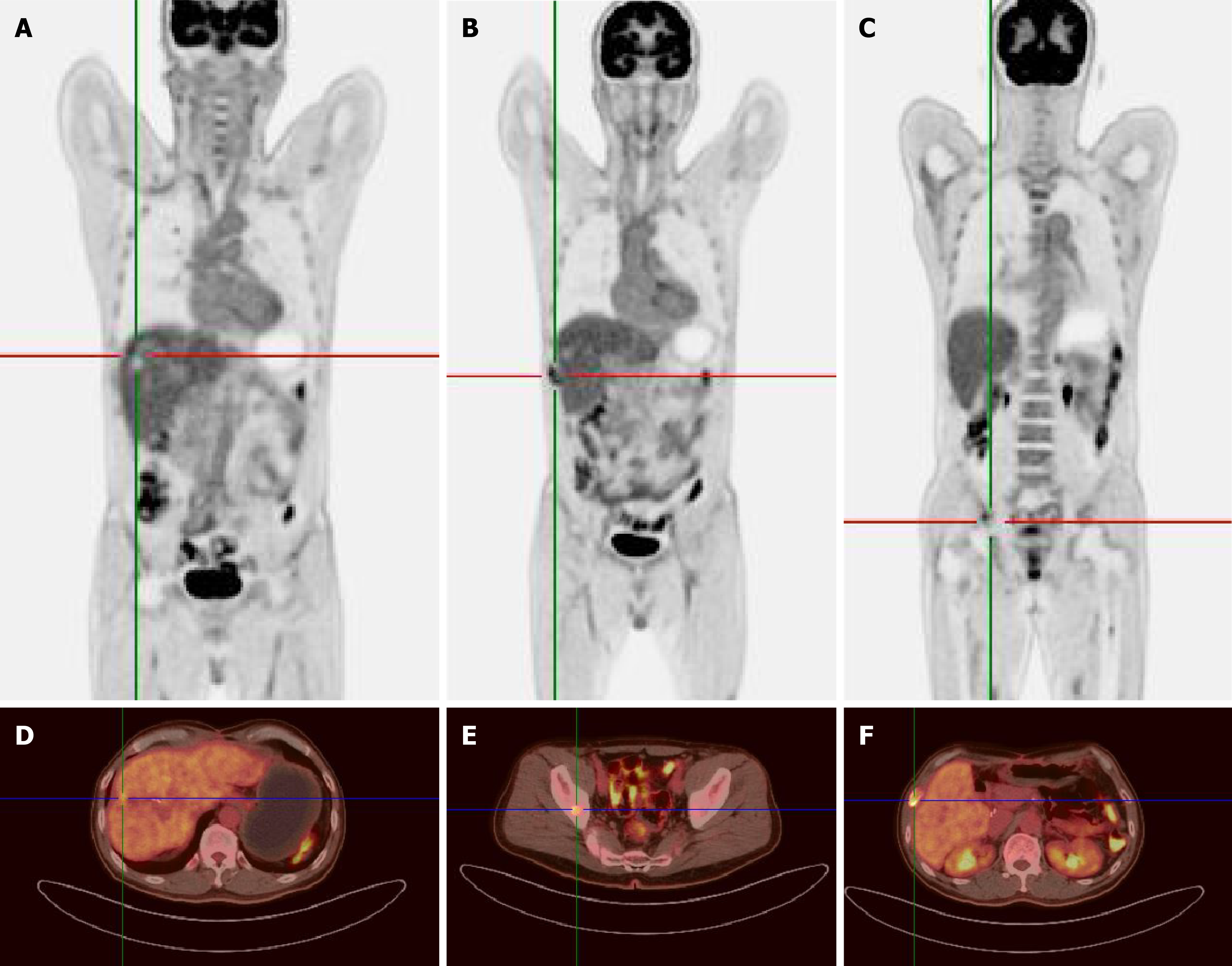
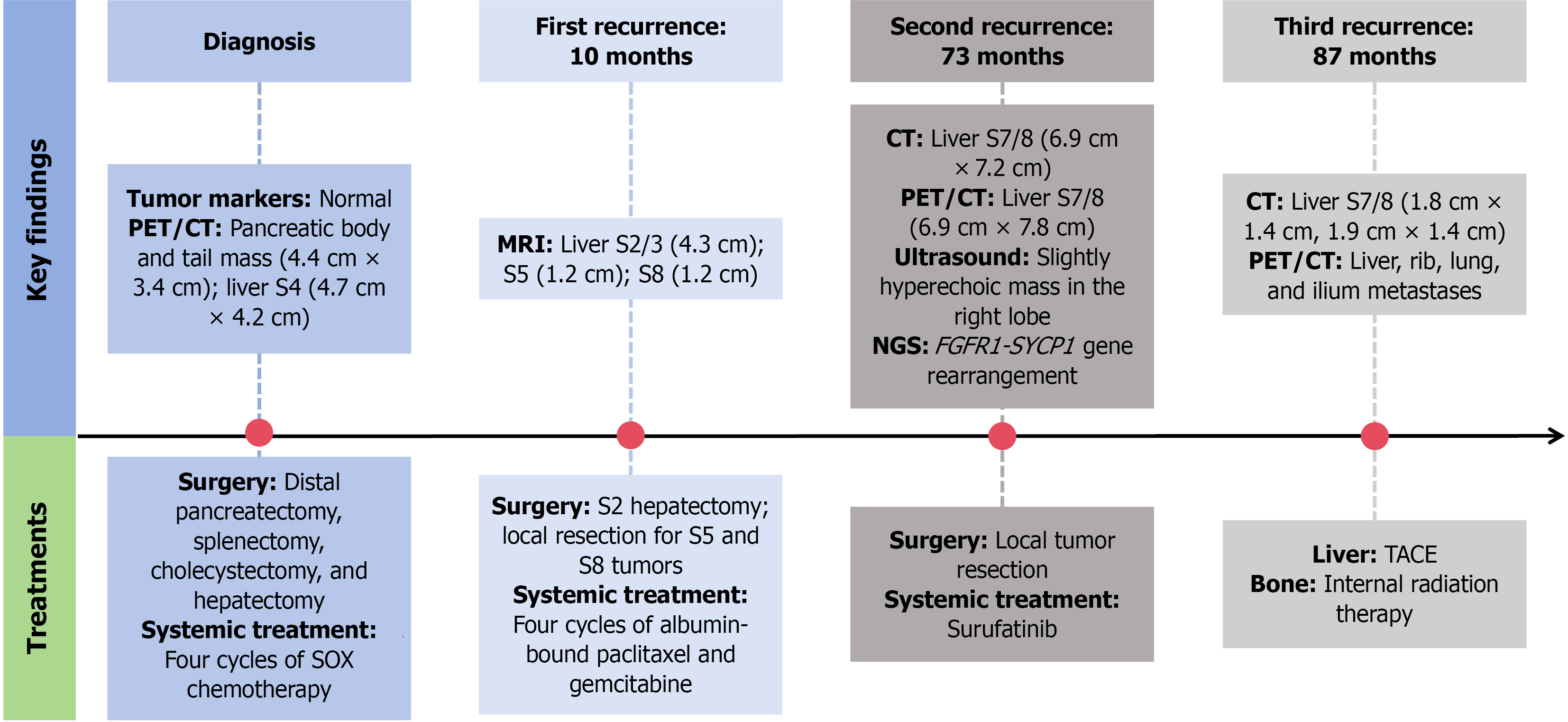
During follow-up, the patient experienced multiple recurrences and remains under active surveillance. Ten months postoperatively, abdominal MRI revealed suspected tumor recurrence in liver segment 2/3, segment 5, and segment 8, with diameters of 4.3 cm, 1.2 cm, and 1.2 cm, respectively (Figure 3). Consequently, the patient underwent segment 2 hepatectomy and local resection for segment 5 and segment 8 tumors, which were pathologically confirmed as liver metastases from pancreatoblastoma (Figure 2C and D). Considering the rapid recurrence, the multidisciplinary team recommended four cycles of albumin-bound paclitaxel and gemcitabine, administered every 3 weeks. However, a second tumor recurrence was detected 73 months after the operation. Abdominal CT revealed a 6.9 cm × 7.2 cm liver mass in segment 7/8 (S7/8). Subsequent 18F-FDG PET/CT for staging showed uptake of 18F-FDG with standardized uptake value maximum of 8.4 in the liver, with no discernible avid lesions elsewhere (Figure 4A). The PET, CT, and fused PET/CT images confirmed a 6.9 cm × 7.8 cm round mass in liver S7/8, suspected to be liver metastasis (Figure 4B-D). Contrast-enhanced ultrasound of the liver revealed a slightly hyperechoic mass in the right lobe, demonstrating heterogeneous hyperenhancement in the arterial phase with washout commencing in the portal phase, progressing to mild washout in the delayed phase and marked washout in the Kupffer phase (Figure 5). Local tumor resection confirmed liver metastasis from pancreatoblastoma with microvascular invasion (Figure 2E and F). Next-generation sequencing of the recurrent liver metastatic lesion revealed FGFR1 and SYCP1 gene rearrangement (80%). The patient then received surufatinib, a tyrosine kinase inhibitor targeting VEGFR1-3 and FGFR1. At 87 months after the initial operation, routine abdominal CT revealed two liver masses in S7/8, measuring 1.8 cm × 1.4 cm and 1.9 cm × 1.4 cm, suggesting a third recurrence. The maximum intensity projection (Figure 6A-C) showed uptake of 18F-FDG in the liver, rib, and ilium. Fused PET/CT (Figure 6D-F) images demonstrated multiple hepatic metastases and metastases in the right ninth rib, both lungs, and the right ilium. After deliberation by the multidisciplinary team, hepatic and bone metastases were treated by transarterial chemoembolization and internal radiation therapy, respectively. The patient is currently undergoing regular hospitalizations for internal radiation therapy. The treatment flowchart for the whole follow-up period is presented in Figure 7.
Pancreatoblastoma, though more common in children, carries a particularly poor prognosis in adults, with 45% suc
Contrary to Zhang et al[2], who reported that pancreatoblastoma typically exhibited minimal uptake on 18F-FDG PET/CT, our case demonstrated intense 18F-FDG uptake in suspicious tumor lesions throughout follow-up. This discrepancy may be attributed to tumor heterogeneity, underscoring the variable metabolic behavior of pancreatoblastoma. Furthermore, compared with conventional imaging modalities such as CT and MRI, PET/CT offers superior sensitivity in detecting metastases, making it valuable for monitoring disease progression in pancreatoblastoma. Nevertheless, the high cost of PET/CT remains a significant barrier to its widespread clinical implementation.
Radical resection remains the optimal therapeutic approach for pancreatoblastoma, with resection of metastases con
This rare case of recurrent adult pancreatoblastoma with liver metastasis supports a multimodal treatment strategy incorporating surgery, chemotherapy, and targeted therapy. It demonstrates the diagnostic and monitoring value of 18F-FDG PET/CT and highlights the importance of investigating the prognostic and therapeutic implications of recurrent genetic alterations, particularly in the FGFR pathway, for developing novel targeted therapies.
| 1. | Omiyale AO. Adult pancreatoblastoma: Current concepts in pathology. World J Gastroenterol. 2021;27:4172-4181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 2. | Zhang X, Ni SJ, Wang XH, Huang D, Tang W. Adult pancreatoblastoma: clinical features and Imaging findings. Sci Rep. 2020;10:11285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 3. | Hammer ST, Owens SR. Pancreatoblastoma: a rare, adult pancreatic tumor with many faces. Arch Pathol Lab Med. 2013;137:1224-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 4. | Bien E, Godzinski J, Dall'igna P, Defachelles AS, Stachowicz-Stencel T, Orbach D, Bisogno G, Cecchetto G, Warmann S, Ellerkamp V, Brennan B, Balcerska A, Rapala M, Brecht I, Schneider D, Ferrari A. Pancreatoblastoma: a report from the European cooperative study group for paediatric rare tumours (EXPeRT). Eur J Cancer. 2011;47:2347-2352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 70] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 5. | Klimstra DS. Nonductal neoplasms of the pancreas. Mod Pathol. 2007;20 Suppl 1:S94-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 130] [Article Influence: 6.8] [Reference Citation Analysis (3)] |
| 6. | Thompson ED, Wood LD. Pancreatic Neoplasms With Acinar Differentiation: A Review of Pathologic and Molecular Features. Arch Pathol Lab Med. 2020;144:808-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 7. | Elghawy O, Wang JS, Whitehair RM, Grosh W, Kindwall-Keller TL. Successful treatment of metastatic pancreatoblastoma in an adult with autologous hematopoietic cell transplant. Pancreatology. 2021;21:188-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 8. | Reissig TM, Uhrig S, Jost PJ, Luchini C, Vicentini C, Liffers ST, Allgäuer M, Adsay V, Scarpa A, Lawlor RT, Fröhling S, Stenzinger A, Klöppel G, Schildhaus HU, Siveke JT. MCL1 as putative target in pancreatoblastoma. Virchows Arch. 2022;481:265-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Montemarano H, Lonergan GJ, Bulas DI, Selby DM. Pancreatoblastoma: imaging findings in 10 patients and review of the literature. Radiology. 2000;214:476-482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 66] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Liu T, Zhao T, Shi C, Chen L. Pancreatoblastoma in children: Clinical management and literature review. Transl Oncol. 2022;18:101359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 11. | Berger AK, Mughal SS, Allgäuer M, Springfeld C, Hackert T, Weber TF, Naumann P, Hutter B, Horak P, Jahn A, Schröck E, Haag GM, Apostolidis L, Jäger D, Stenzinger A, Fröhling S, Glimm H, Heining C. Metastatic adult pancreatoblastoma: Multimodal treatment and molecular characterization of a very rare disease. Pancreatology. 2020;20:425-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 12. | Yuan J, Guo Y, Li Y. Diagnosis, treatment, and prognosis of adult pancreatoblastoma. Cancer Med. 2024;13:e70132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 13. | Reid MD, Bhattarai S, Graham RP, Pehlivanoglu B, Sigel CS, Shi J, Saqi A, Shirazi M, Xue Y, Basturk O, Adsay V. Pancreatoblastoma: Cytologic and histologic analysis of 12 adult cases reveals helpful criteria in their diagnosis and distinction from common mimics. Cancer Cytopathol. 2019;127:708-719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 14. | Remo A, Negro S, Bao RQ, d'Angelo E, Alaggio R, Crivellari G, Mammi I, Intini R, Bergamo F, Fassan M, Agostini M, Vitellaro M, Pucciarelli S, Urso EDL. Association between Pancreatoblastoma and Familial Adenomatous Polyposis: Review of the Literature with an Additional Case. Genes (Basel). 2023;15:44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |