Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.116568
Revised: December 12, 2025
Accepted: February 6, 2026
Published online: May 15, 2026
Processing time: 181 Days and 15.4 Hours
Colorectal cancer (CRC) remains among the most common causes of cancer-related deaths worldwide. Ulcerative colitis and Crohn’s disease are inflammatory bowel diseases characterized by recurrent episodes of inflammation in the gastrointestinal tract, which increase the risk of developing CRC. One of the main pathways activated during the inflammatory process is sphingosine-1-phosphate (S1P). S1P activates the signal transducer and activator of transcription 3 (STAT3). Phosphorylated STAT3 inhibits apoptotic pathways, promoting tumorigenesis and, consequently, the development of CRC. Compounds with antioxidant potential, such as the hydroalcoholic extract of Butia capitata fruit pulp (HAEBC), may act to reduce oxidative stress, thereby decreasing inflammatory events.
To evaluate the anti-inflammatory and antinociceptive effects of HAEBC in an experimental colitis model and to investigate, in silico, the interaction of its phenolic compounds with molecular targets.
HAEBC was prepared using a modified protocol and administered in 10% dimethyl sulfoxide in saline. Male and female Swiss mice were used for pain tests induced by acetic acid, formalin, and carrageenan. Acute colitis was induced by intracolonic administration of acetic acid after pretreatment with HAEBC, mesalazine, or saline. Macroscopic lesions, histological changes, and visceral hyperalgesia (von Frey test) were evaluated. Statistical analysis was performed using one-way analysis of variance followed by Tukey’s test. In silico analyses explored potential anti-inflammatory targets of HAEBC compounds, including sphingosine kinase 1 (SPHK1), indoleamine 2,3-dioxygenase 1 (IDO1), and cyclooxygenase-2 (COX-2).
Pre-treatment with HAEBC at doses of 30 and 100 mg/kg significantly reduced contortions in mice (58% and 49%, respectively), with time-dependent effects. The extract inhibited both the neurogenic and inflammatory phases of the formalin test, decreasing paw flinching and licking time, indicating action on nociceptors and inflammatory mediators such as prostaglandins and cytokines. HAEBC at 100 mg/kg also reduced carrageenan-induced paw edema by 63%. In acute colitis, treated mice showed more than 75% inhibition of macroscopic lesions and preserved colon architecture, unlike mesalazine-treated animals. In the von Frey test, HAEBC at 300 mg/kg outperformed both the 10 mg/kg dose and mesalazine. Docking analyses indicated that gallic acid, p-hydroxybenzoic acid, and chlorogenic acid interact with SPHK1, IDO1, COX-2, and sphingosine 1-phosphate receptor 1, with chlorogenic acid forming the most stable complexes.
HAEBC exhibited anti-inflammatory and antinociceptive effects, preserving tissue integrity and reducing hyperalgesia. The proposed molecular interactions remain hypothetical. Implications for tumorigenesis are indirect and require validation using chronic models and molecular analyses.
Core Tip: The hydroalcoholic extract of Butia capitata showed antinociceptive activity in mice, in models of pain and/or hyperalgesia caused by acetic acid, formalin, and carrageenan, with an emphasis on reducing the histological damage and the pain symptoms caused by acetic acid-induced acute colitis. The in silico results indicate the potential of phenolic compounds of the extract to inhibit cellular pathways and receptors involved in inflammatory processes, as well as reduce the activation of pathways that promote genetic damage and tumorigenesis.
- Citation: Da Silva PB, Lopes-Bute L, Fischer J, Parcianello AP, Pires-Duarte R, Luiz RM, Brandalize APC, Melo FF, Wendel CF, Teixeira KN, Zarpelon-Schutz AC. Protective effects of Butia capitata extract against colitis-driven colorectal tumorigenesis: Insights into its anti-inflammatory and antinociceptive activities. World J Gastrointest Oncol 2026; 18(5): 116568
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/116568.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.116568
Colorectal cancer (CRC) is the third most commonly diagnosed type of cancer and the second most lethal neoplasm worldwide. Most cases are associated with modifiable lifestyle-related risk factors, such as diet, physical activity, smoking, alcohol consumption, and body weight[1]. The incidence of this disease is higher in developed countries, and the likelihood of developing it increases with age, with a cumulative lifetime risk of approximately 5%[2]. More than two-thirds of CRC cases are sporadic; the remaining cases are associated with familial clustering or hereditary syndromes, such as Lynch syndrome and familial adenomatous polyposis. Diagnosis is usually made by colonoscopy with biopsy in symptomatic patients or in individuals who meet screening criteria for this type of cancer[1].
Personal history provides an overview of CRC risk, including the presence of inflammatory bowel diseases (IBD). Ulcerative colitis (UC) and Crohn's disease are IBDs and present as recurrent episodes of inflammation in the gas
In addition, persistent inflammation leads to uncontrolled activation of pathways responsible for the production of cytokines, interleukins (IL), and growth factors. One of the main pathways activated during the inflammatory process is sphingosine-1-phosphate (S1P), generated by the phosphorylation of sphingosine mediated by tumor necrosis factor alpha (TNF-α). S1P activates nuclear factor-kappa B (NF-κB), inducing its nuclear translocation and leading to the production of IL-6. This cytokine, acting synergistically with S1P, activates the signal transducer and activator of transcription 3 (STAT3). Phosphorylated STAT3 translocates to the nucleus and inhibits apoptotic pathways, rendering the cell prone to tumorigenesis and, consequently, to the development of CRC[4,6].
The use of plants in the management of various diseases, including inflammatory conditions, dates back thousands of years. Numerous metabolites from natural sources have been studied to identify alternative therapeutic strategies or to develop new drugs for the treatment of diverse pathologies. Phenylpropanoids are among the metabolites investigated for their anti-inflammatory properties, as they can act on the NF-κB signaling pathway by inhibiting the transcription of cytokines, IL-β, IL-6, nitric oxide, and TNF-α[7]. Terpenes represent another class of plant metabolites with anti-inflammatory, antioxidant, and wound-healing properties, as well as triterpenes, which exhibit antitumor, anti-angiogenic, and antioxidant effects and can induce apoptosis, demonstrating their potential to act against malignant neoplasms[8].
Phenolic compounds, such as flavonoids and phenolic acids, are highly abundant secondary metabolites in plants, fruits, and seeds. In plants, flavonoids perform multiple functions, including acting as signaling molecules, UV filters, and reactive oxygen species (ROS) scavengers. In humans, these compounds are associated with a wide range of health benefits derived from their bioactive properties, including anti-inflammatory, anticancer, anti-aging, cardioprotective, neuroprotective, immunomodulatory, antidiabetic, antibacterial, antiparasitic, and antiviral activities[9]. Isoflavones appear to protect against inflammatory vascular diseases, and quercetin exhibits cardioprotective effects against myocardial injury and atheroprotective activity associated with reduced oxidative stress. During inflammatory processes, flavonoids can act as antioxidants by eliminating ROS or reducing free radical accumulation, as inhibitors of regulatory enzyme activity (e.g., protein kinases and phosphodiesterases), and as modulators of transcription factors involved in the control of inflammatory mediators and immune cell activity[10].
Recent studies have demonstrated high concentrations of vitamin C and phenolic compounds in fruits of the genus Butia[11-13], as well as significant levels of carotenoids, such as β-carotene, and tocopherol[14,15], which are antioxidant compounds capable of inhibiting or delaying the oxidation of oxidizable substrates. Phenolic compounds are secondary metabolites produced during normal plant development and are chemically defined by the presence of an aromatic ring with one or more hydroxyl substituents[16]. Among the phenolic compounds identified in the genus Butia, p-hydroxybenzoic acid, gallic acid, and ellagic acid stand out, presenting higher total levels than those observed in other palm species. The antioxidant activity of Butia fruit pulp is strongly correlated with its vitamin C and total phenolic content.
In this context, compounds with antioxidant potential, such as hydroalcoholic extract of Butia capitata (HAEBC), may reduce oxidative stress due to their ability to scavenge free radicals and donate electrons and hydrogen atoms, thereby limiting the perpetuation of inflammatory events and reducing the likelihood of genetic damage that may facilitate neoplasm development[7]. Therefore, the aim of this investigation is to assess the ability of HAEBC to attenuate nociception and improve inflammatory parameters in experimental colitis associated with the onset of CRC.
Butia capitata fruits were harvested in Marechal Cândido Rondon, Paraná, Brazil (coordinates 24°33′24.09″ S; 54°03′24.21″ W) in January 2022. Following sanitization with a 0.1% (v/v) hypochlorite solution, the seeds were manually removed from the fruits. The pulp was dried in a forced-air oven at 40 °C and ground in a knife mill to obtain the dried mass. HAEBC was prepared using a modified version of a previously published protocol[17]. An extraction solution composed of absolute ethanol and distilled water at a 4:1 (v/v) ratio was mixed with the dried mass at a 10:1 ratio. The mixture was stirred on a magnetic stirrer for five minutes, followed by sonication (two cycles of 5 minutes on/5 minutes off) and centrifugation. The resulting supernatant was filtered and evaporated on a hot plate at 60 °C. For in vivo experiments, HAEBC was dissolved in 10% (v/v) dimethyl sulfoxide in saline solution [0.9% (w/v) NaCl].
This study was approved by the Ethics Committee for the Use of Animals of the Setor de Ciências Biológicas at the Universidade Federal do Paraná, Paraná, Brazil (CEUA No. 1531/2023). All experiments were conducted in accordance with the regulations and principles established by the National Council for the Control of Animal Experimentation. Male and female Swiss mice (Mus musculus), weighing 25-30 g, were used in the experiments. The animals were housed in polypropylene cages at the animal facility of the Universidade Federal do Paraná-Toledo Campus. They had ad libitum access to standard rodent chow and chlorinated drinking water and were maintained under controlled temperature conditions (22 ± 2 °C) with a 12 hours light/dark cycle.
For the writhing test, female Swiss mice were divided into six groups (n = 8 per group) and fasted for 8 hours prior to the experiment. The nociceptive response was induced by intraperitoneal administration of 200 μL of 0.8% (v/v) acetic acid in saline (10 mL/kg). The mice were orally treated with different doses of HAEBC (standard administration volume of 150 μL at 10, 30, 100, and 300 mg/kg) 30 minutes before acetic acid injection. Each mouse was placed individually in glass cylinders, and the number of abdominal contortions was recorded. The intensity of the writhing response was expressed as the cumulative number of abdominal contortions over a total period of 20 minutes. The dose that proved most effective in this test (30 mg/kg) was subsequently used to treat the intervention group in the formalin test.
In the formalin test, female Swiss mice were divided into three groups (n = 8 per group), and 25 μL of a 1.5% (v/v) formalin solution was injected intraplantarly into the mouse paw 30 minutes after oral treatment with HAEBC (150 μL at 30 mg/kg). After formalin injection, the animals were placed individually in glass cylinders, and for 30 minutes, both the cumulative time (in seconds; nociception index) spent licking or biting the injected paw and the number of paw flinches were recorded.
To assess cutaneous hyperalgesia, the von Frey test was used in this study. Male Swiss mice were placed in acrylic boxes with a wire-mesh floor that allowed access to the animals’ hind paws for a period of 15-30 minutes before the beginning of the test. The animals were divided into five groups (n = 6 per group) and treated with HAEBC at four different doses (standard administration volume of 150 μL at 10, 30, 100, and 300 mg/kg). After 30 minutes, an intraplantar injection of carrageenan (25 μg/paw) was administered, and mechanical hyperalgesia was assessed at 0, 1, 3, and 5 hours.
The test consisted of inducing the paw withdrawal reflex using a portable force transducer (von Frey electronic aesthesiometer; Insight, Ribeirão Preto, SP, Brazil) equipped with a polypropylene tip with a contact area of 0.5 mm², which measured the nociceptive threshold and expressed the applied force in grams. The researcher positioned the transducer tip on the central region of the animal’s hind paw until paw withdrawal occurred, at which point the applied force was automatically recorded. Final values were expressed as the paw withdrawal threshold (Δ) in grams (g), calculated by subtracting the baseline value measured before treatment (T0) from the values obtained after treatment.
Male Swiss mice were divided into seven groups (n = 8 per group) and fasted for solid food for 24 hours. Colitis was induced by rectal administration of 200 μL of 7.5% (v/v) acetic acid in saline, and colitis induction was assessed 4 hours after acid administration. The mice were pretreated with HAEBC 4 and 10 hours before colitis induction and treated 2 hours after colitis induction (standard administration volume of 150 μL at 10, 30, 100, and 300 mg/kg). The positive control group was treated with mesalazine (30 mg/kg; 150 μL), and the negative control group received saline (150 μL). Four hours after colitis induction, abdominal mechanical hyperalgesia was assessed using the von Frey test, with an electronic aesthesiometer to determine the abdominal nociceptive threshold. At the end of the experiment, the animals were euthanized by isoflurane overdose followed by cervical dislocation.
The distal segment of the colon was collected and washed with saline solution to remove fecal residues, followed by macroscopic evaluation of lesion severity according to a previously described scoring index[18] (Table 1).
| Score | Criteria |
| 0 | No damage |
| 1 | Focal hyperemia without ulceration |
| 2 | Ulceration without hypertrophy of the intestinal wall |
| 3 | Ulceration with inflammation spot |
| 4 | Two or more sites of ulceration or inflammation |
| 5 | Large areas of inflammation and ulceration extending more than 1 cm |
| 6 | Large areas of tissue damage with an extension greater than 2 cm added 1 point for each additional 1 cm of extension |
Following macroscopic analysis, the collected material was fixed in 4% (v/v) buffered formaldehyde. Each sample was then divided into three fragments, processed, and individually embedded in paraffin. The paraffin blocks were sectioned at a thickness of 4 μm using a semi-motorized rotary microtome and mounted on glass slides. The slides were stained with hematoxylin–eosin and independently analyzed by two blinded researchers using an optical microscope. Tissue damage was classified as absent (0), mild (1), moderate (2), or severe (3).
To assess visceral mechanical hyperalgesia induced by acute colitis, mice were placed in boxes at controlled room temperature for at least 45 minutes prior to the beginning of the test. The test consisted of evaluating the animals’ withdrawal reflex in response to stimulation with a portable force transducer (von Frey electronic aesthesiometer; Insight, Ribeirão Preto, SP, Brazil). The transducer was equipped with a polypropylene tip with a contact area of 0.7 mm², which was applied to the lower abdomen up to the mid-abdominal region. The applied force was automatically recorded upon withdrawal of the transducer, and the mean of three measurements was considered the final value.
The primary outcomes measures for this study were abdominal contortions, nociception index, paw withdrawal threshold, abdominal mechanical hyperalgesia, macroscopic scoring index and histological damage. The secondary outcome was bioinformatic analysis.
The averages of the values acquired for each group of animals in each experiment were computed and stored in spreadsheets developed using Microsoft Excel software. GraphPrism software was used to analyze the data, and a one-way analysis of variance was employed to compare groups based on treatment dosage, followed by Tukey’s test for post-hoc analysis. Significance was established at P < 0.05.
The analysis of the interaction between the phenolic compound from Butia capitata and human cellular targets was conducted using the molecular docking technique with the rigid receptor/flexible ligand methodology[19] to extrapolate the results. The selected cellular targets were enzymes and receptors involved in the maintenance of chronic inflammatory processes in colitis and associated with the development of CRC-sphingosine kinase 1 (SPHK1), sphingosine 1-phosphate receptor 1 (S1PR1), indoleamine 2,3-dioxygenase 1 (IDO1), and cyclooxygenase-2 (COX-2)[4]. 3D structures of the cellular targets (receptors) were obtained from the protein data bank (PDB) (rcsb.org) and prepared for molecular docking using ViewerLite 5.0 (Accelrys Inc.) and AutoDock Tools (ADT). Binding sites in the cellular targets were identified using the CASTp active site tool (CASTp 3.0)[20]. Phenolic compounds from Butia capitata were selected based on literature reports describing their presence in the fruit[12,14,21,22]. The structures of the phenolic acids-gallic acid (CID 370), chlorogenic acid (CID 1794427), and p-hydroxybenzoic acid (CID 547)-were retrieved from the PubChem database (pubchem.ncbi.nlm.nih.gov). Molecular docking was performed using ADT 4[23] and AutoDock Vina v1.2.0[24]. Analysis of receptor–ligand interactions was carried out using BIOVIA Discovery Studio (Dassault Systèmes).
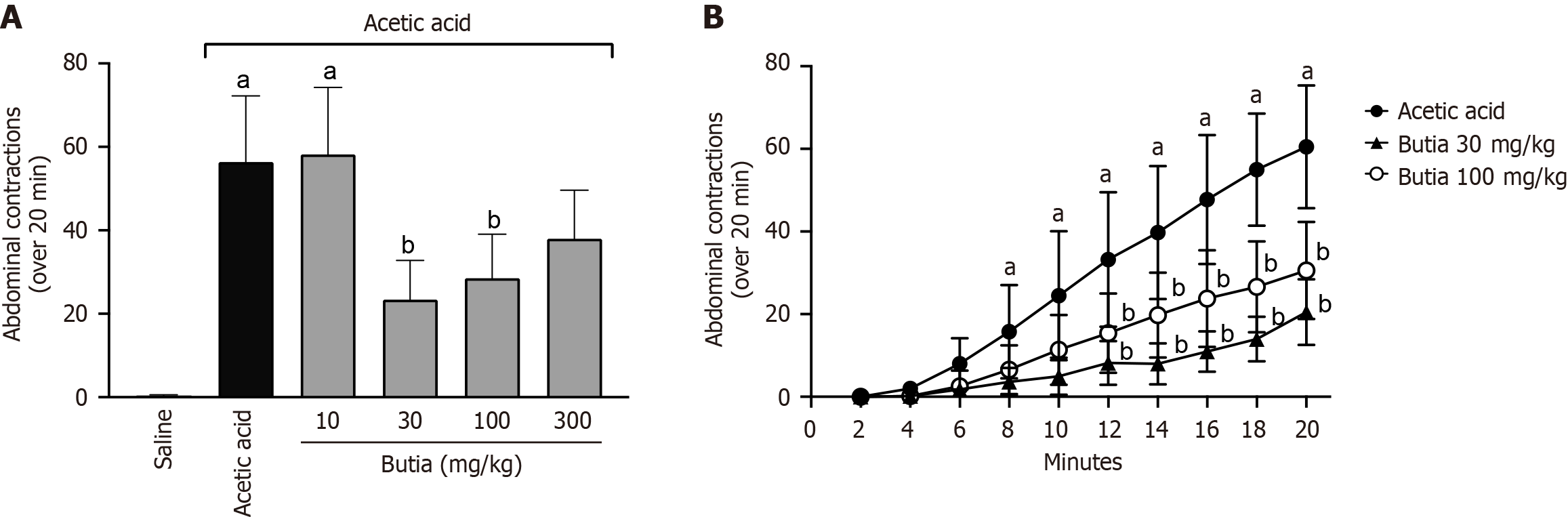
Writhing test indicated that HAEBC at 30 and 100 mg/kg significantly reduced the total number of writhing events occurring within 20 minutes compared with the acetic acid-treated group, with reductions of 58% and 49%, respectively. The control group (acetic acid–treated) showed an intense nociceptive response after administration of acetic acid, and the group that received HAEBC at 10 mg/kg showed a similar result, with no antinociceptive effect (Figure 1A). As shown in Figure 1B, which presents the cumulative number of abdominal writhing every 2 minutes (over 20 minutes), pretreatment with HAEBC at 30 and 100 mg/kg significantly reduced the number of writhing episodes in a time-dependent manner, with effects observed 12 minutes after administration.
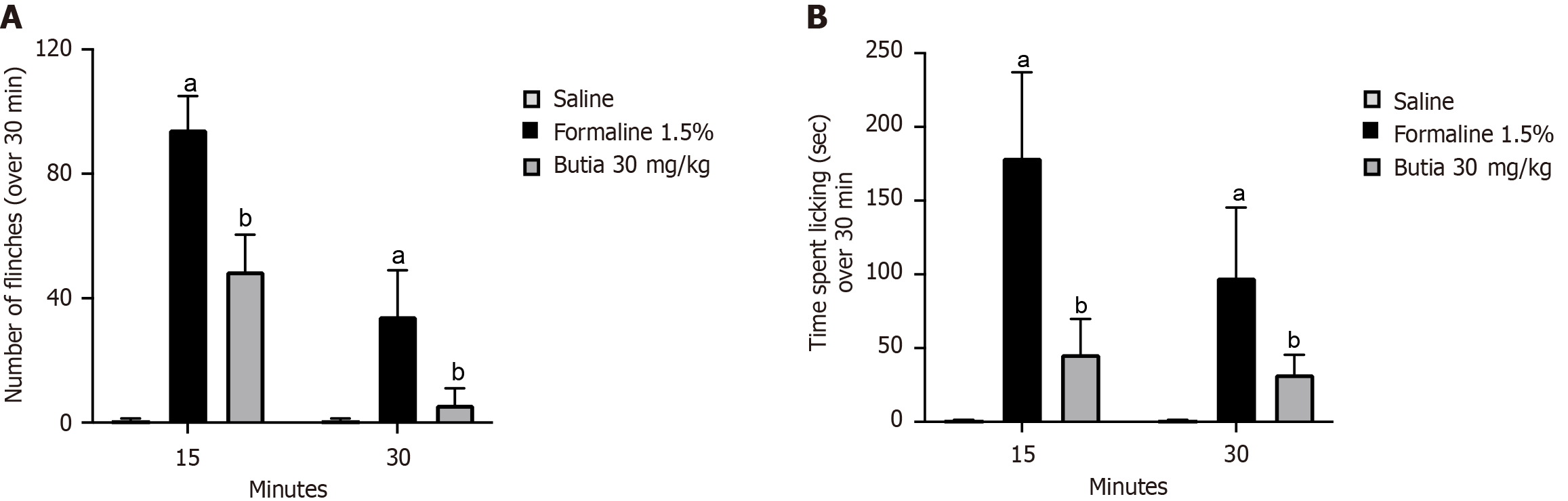
The HAEBC produced effects in mice treated with the 30 mg/kg dose, reducing paw contraction (Figure 2A) and the time spent licking the formalin-injected paw (Figure 2B). Mice treated with HAEBC showed a reduction in paw flinching of 48% in the initial phase and 84% in the late phase, as well as a reduction in licking time of 74% (initial phase) and 67% (late phase). The first phase of the formalin test is related to the direct effect of the substance on local nociceptors, whereas the second phase depends on the release of prostaglandins and cytokines. Therefore, it is assumed that the extract may act directly on nociception caused by receptor stimulation, as well as on inflammation resulting from the release of inflammatory mediators.
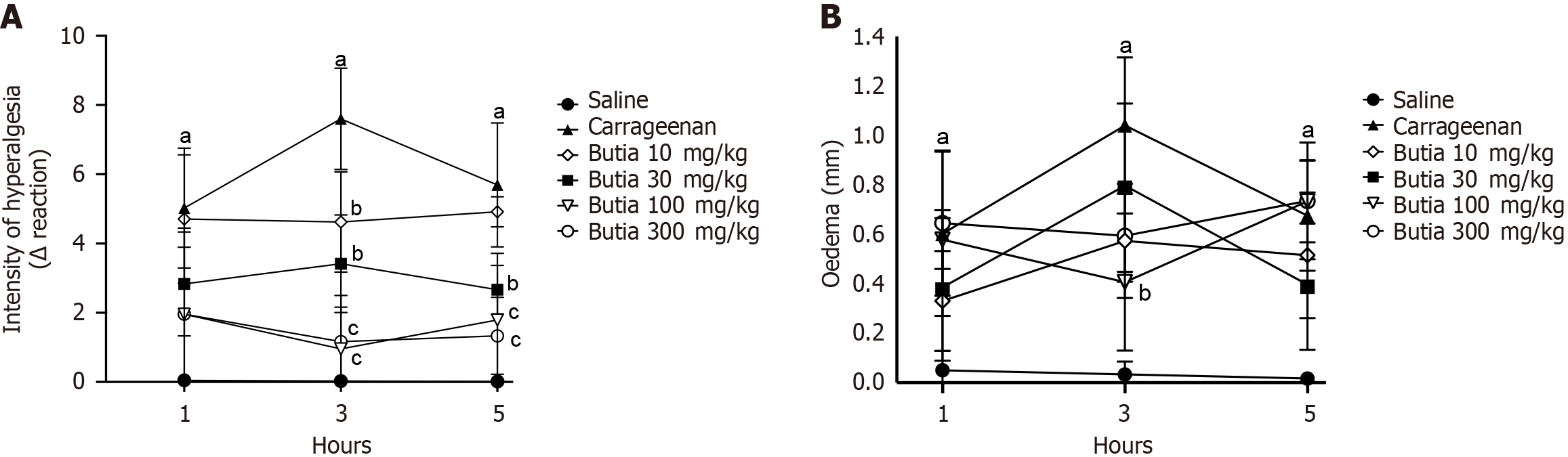
Administration of HAEBC produced a dose-dependent inhibition (10-300 mg/kg) of carrageenan-induced mechanical hyperalgesia (Figure 3A). None of the tested doses produced effects at 1 hour after carrageenan administration. At the 10 mg/kg dose, significant effects were observed only at 3 hours after the stimulus, reducing hyperalgesia by 39%. However, doses of 30, 100, and 300 mg/kg of HAEBC significantly inhibited carrageenan-induced hyperalgesia at 3 hours after the stimulus (45%, 87%, and 85% reduction, respectively) and at 5 hours after the stimulus (53%, 69%, and 77% reduction, respectively). In addition to attenuating inflammatory hyperalgesia, HAEBC at 100 mg/kg also suppressed carrageenan-induced paw edema at 3 hours post-stimulus (63% reduction), indicating a broader potential therapeutic applicability of HAEBC in inflammatory conditions (Figure 3B). The remaining doses did not demonstrate any anti-edematous effect during the assessed time points.
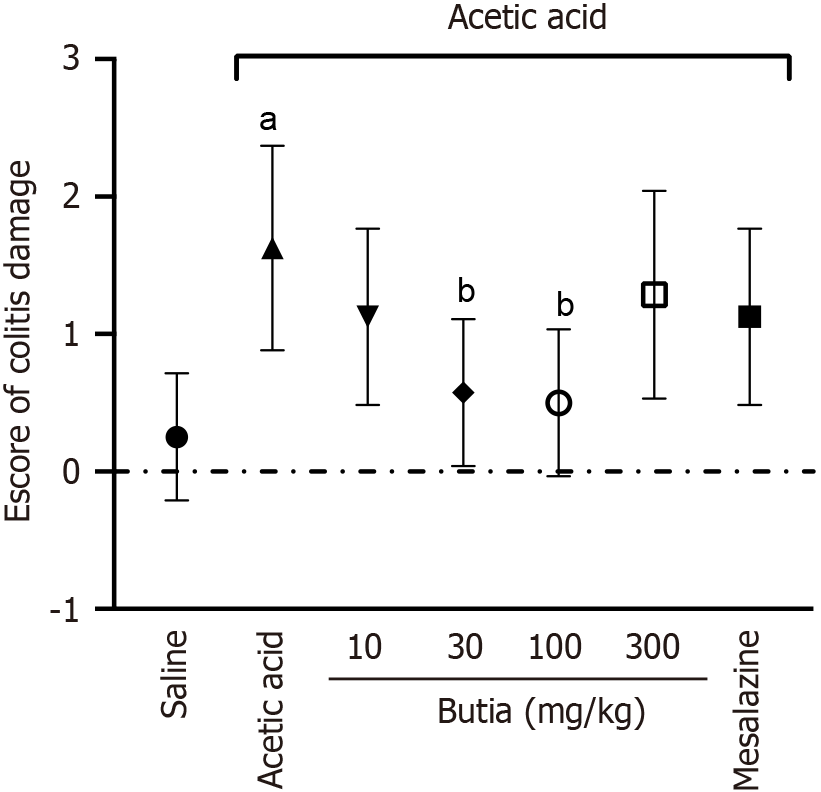
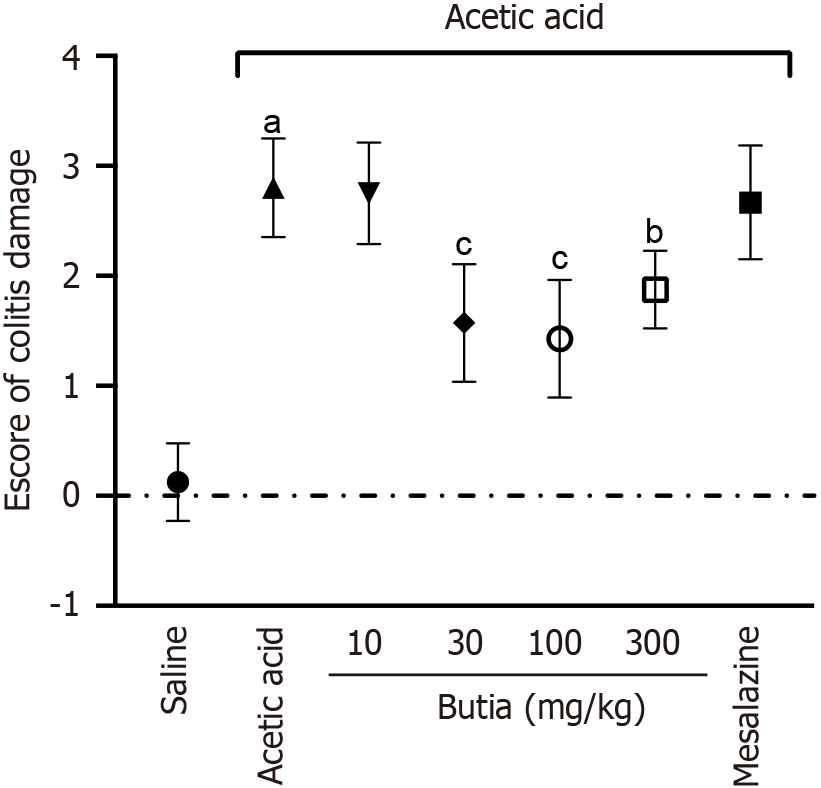
The intracolonic injection of acetic acid triggered an intense inflammatory reaction compared with the negative control group (saline) (Figure 4). Macroscopic lesion scores ranged from 0 to 3 (0 = no lesion; 3 = ulceration with inflammatory spots). Most control mice (no colitis induction) had no lesions (score 0), whereas most mice with induced colitis and without treatment showed a score of 2. Gross assessment of colonic segments indicated that mice treated with HAEBC at 30 and 100 mg/kg showed fewer lesions than untreated mice, with reductions of 76.6% and 81.8%, respectively, in macroscopic scores. Overall, the intermediate doses of HAEBC showed activity against the inflammatory process in the colon of animals with acute colitis. Mice treated with mesalazine presented intestinal lesions with scores of 1 or 2, suggesting that mesalazine was not effective in inhibiting this parameter and showed lower effectiveness compared with HAEBC.
Histological analysis graded the degree of inflammation and tissue damage from 0 to 3 (0 = no signs of inflammation; 3 = intense inflammation). The negative control group (saline) showed no signs of inflammation (score 0), and all untreated mice with induced colitis showed a score of 3. Regarding HAEBC, except for the lowest dose of 10 mg/kg, all other doses (30, 100, and 300 mg/kg) effectively reduced tissue injury and inflammation. Mice that received mesalazine presented colitis damage scores similar to untreated mice with induced colitis, indicating limited activity of this drug in reducing or preventing histological damage (Figure 5).
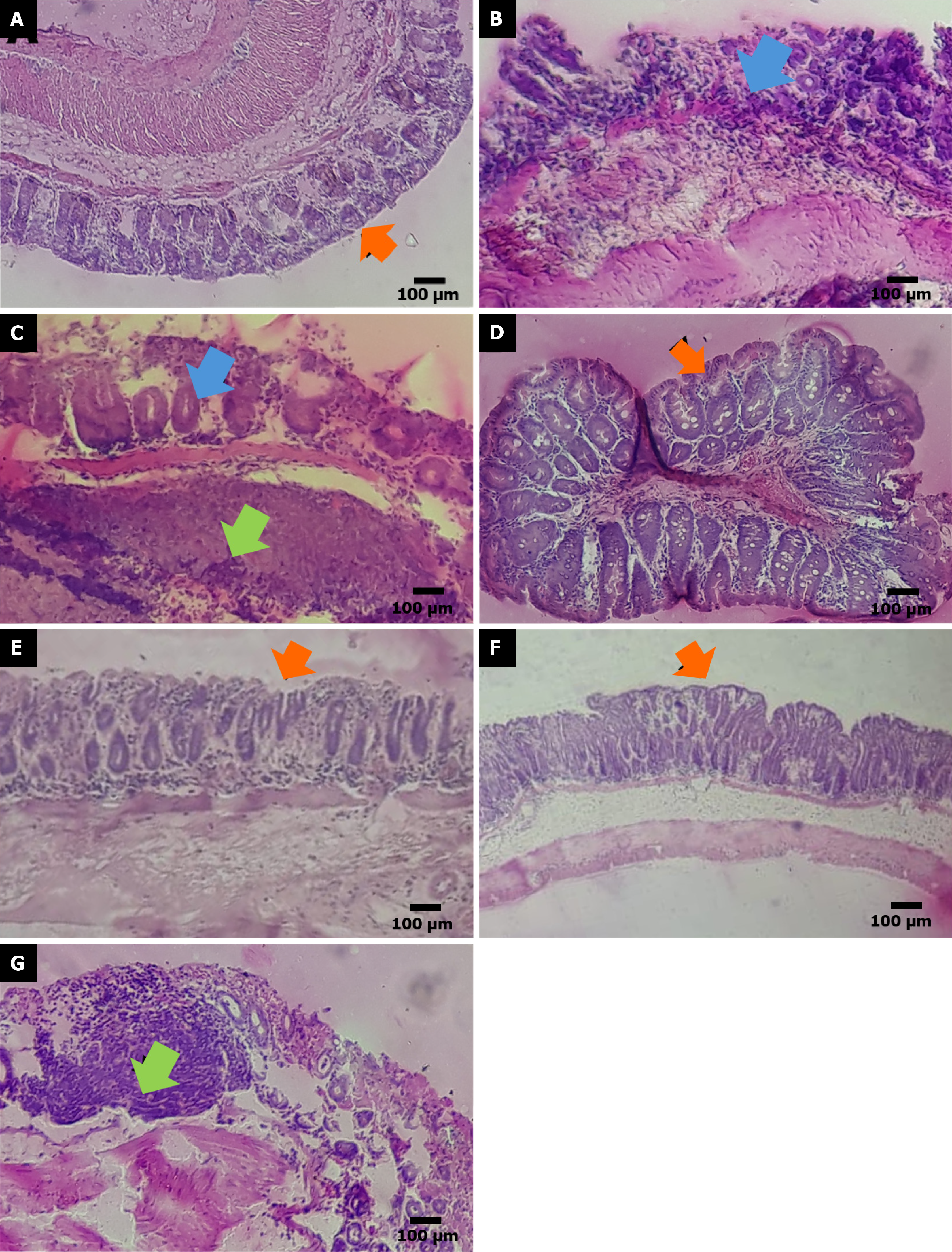
As illustrated in Figure 6, histological sections from saline-treated animals showed no signs of inflammatory cell infiltration, and the crypt and epithelial architecture were preserved (Figure 6A). In contrast, after acetic acid administration, there was marked distortion of crypts, loss of goblet cells, and the presence of inflammatory infiltrate (Figure 6B and C). HAEBC at 30, 100, and 300 mg/kg was effective in maintaining tissue architecture and preventing histological damage (Figures 6D-F). Treatment with mesalazine was not effective in reducing microscopic damage to the colon (Figure 6G).
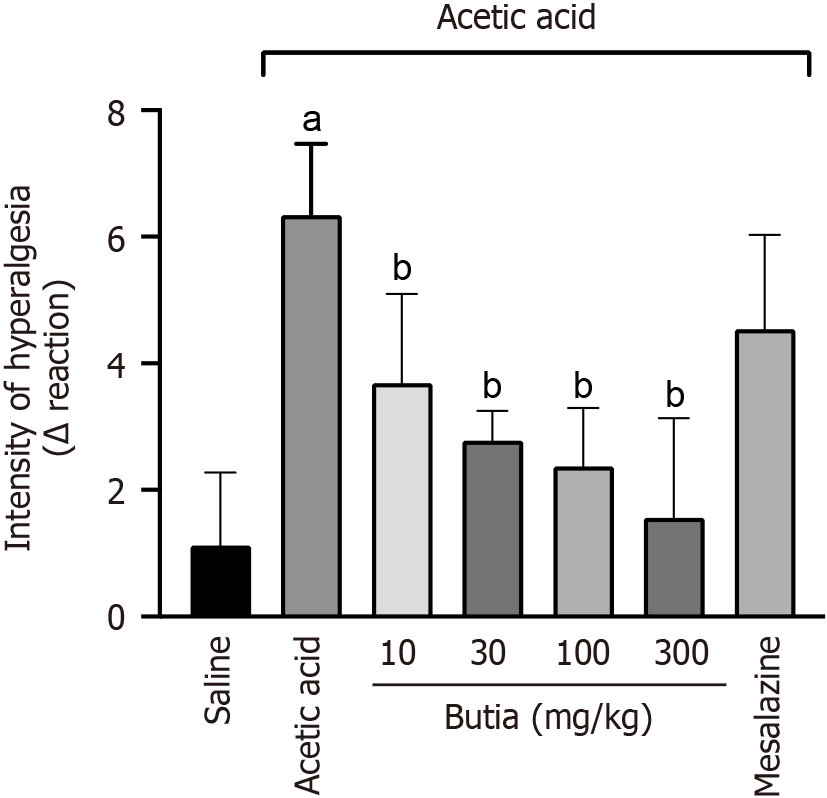
The outcomes of the von Frey test were expressed as the difference in force required to elicit a positive response before and after the induction of acute colitis by intracolonic injection of acetic acid. Mice treated with HAEBC at doses of 10, 30, 100, and 300 mg/kg exhibited significant differences in analgesic effects compared with untreated mice. Among the treatment groups, HAEBC at 300 mg/kg showed significantly superior effects compared with the 10 mg/kg dose and mesalazine. Treatment with mesalazine was not effective in inhibiting hyperalgesia (Figure 7).
Molecular docking simulations were performed using human, non-mutated 3D structures of SPHK1 (PDB 3VZB), S1PR1 (PDB 7TD3), IDO1 (PDB 5EK3), and COX-2 (PDB 5F1A). The results indicate that gallic acid, p-hydroxybenzoic acid, and chlorogenic acid form in silico complexes with all evaluated cellular targets. However, based on binding affinity energy, only chlorogenic acid appears to form stable complexes under physiological conditions, suggesting a potential role for this metabolite in the analyzed cellular pathways (Table 2).
| Receptors | Phenolic compounds | ||
| Gallic acid (kcal/mol) | Chlorogenic acid (kcal/mol) | p-hydroxybenzoic acid (kcal/mol) | |
| SPHK1 (PDB 3VZB) | -6.1 | -8.6 | -5.9 |
| S1PR1 (PDB 7TD3) | -5.9 | -8.7/-8.4 | -5.7 |
| IDO1 (PDB 5EK3) | -7.1 | -8.7 | -6.8 |
| COX-2 (PDB 5F1A) | -6.3 | -8.8 | -6.0 |
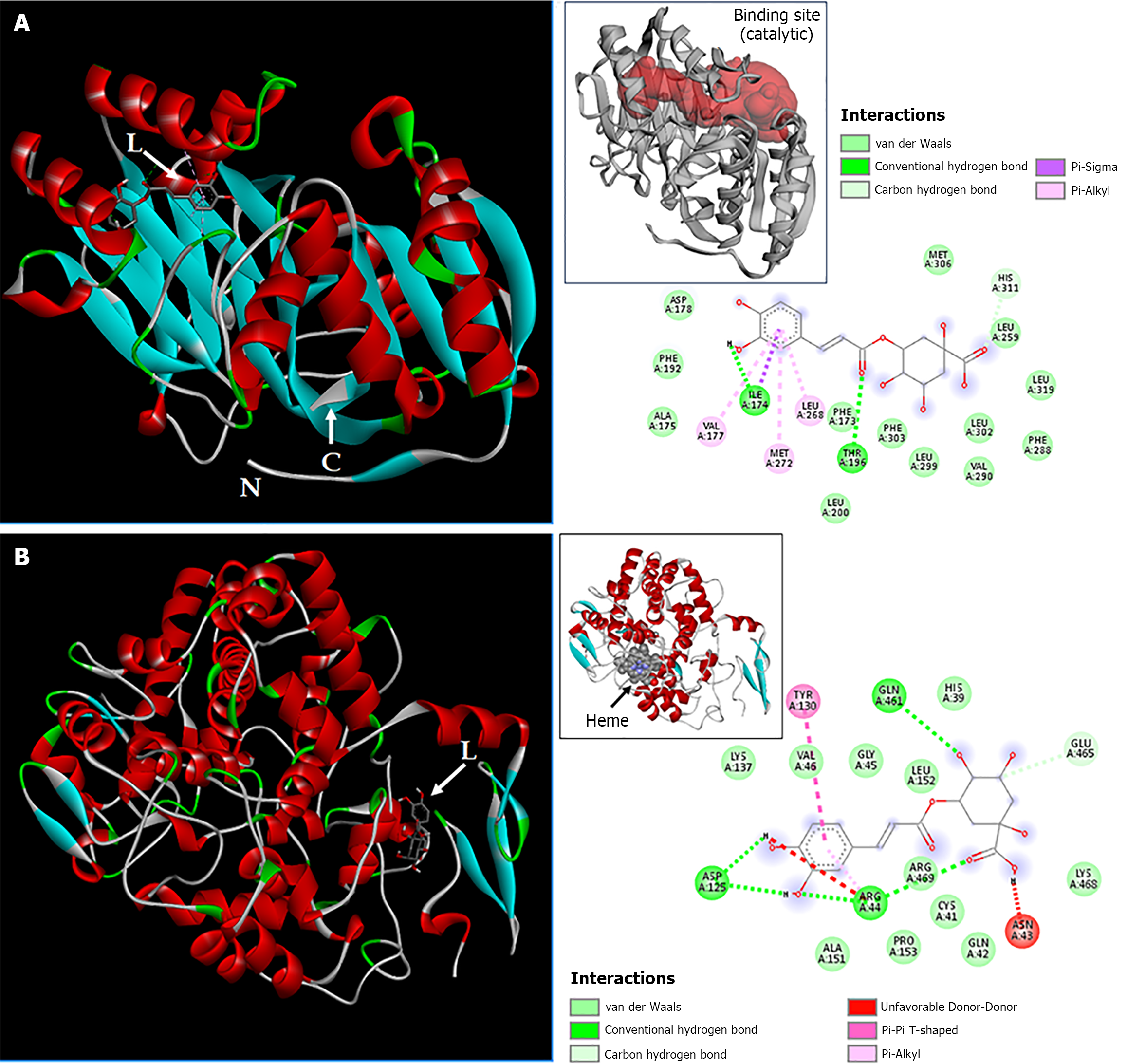
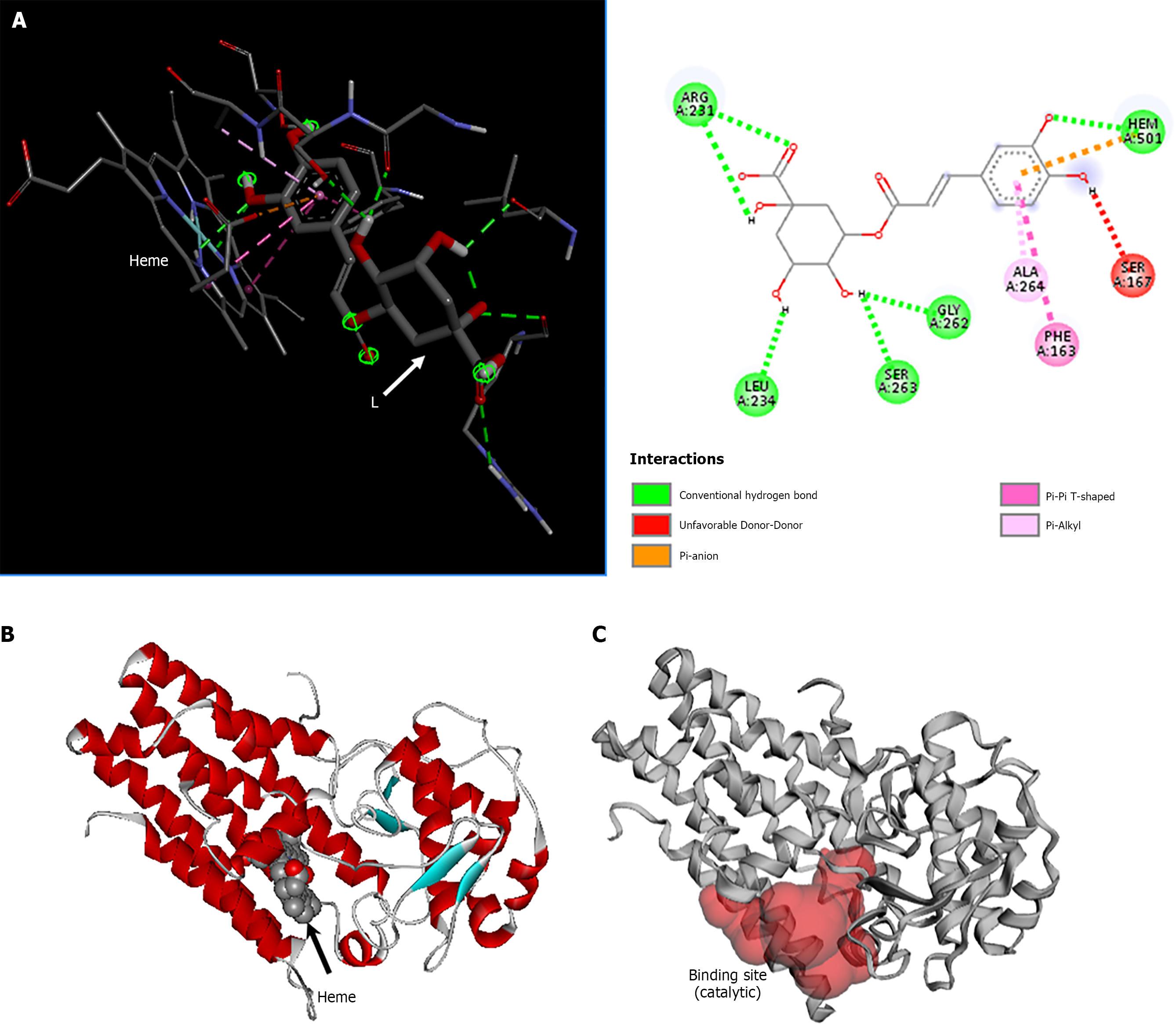
The SPHK1/chlorogenic acid complex is stabilized by 20 chemical interactions involving 19 SPHK1 residues and atoms of the ligand. SPHK1 is a homotrimeric enzyme, and the association of the three chains (A, B, and C) occurs through residues located in the central region of each chain. Chlorogenic acid binds to this central region within the binding site, which is possibly the catalytic site (Figure 8A).
COX-2 is a homodimeric enzyme (chains A and B) whose catalytic activity depends on the heme group. In silico, chlorogenic acid binds to COX-2 through residues near the heme group (21 interactions involving 17 residues) without directly interacting with the heme itself (Figure 8B).
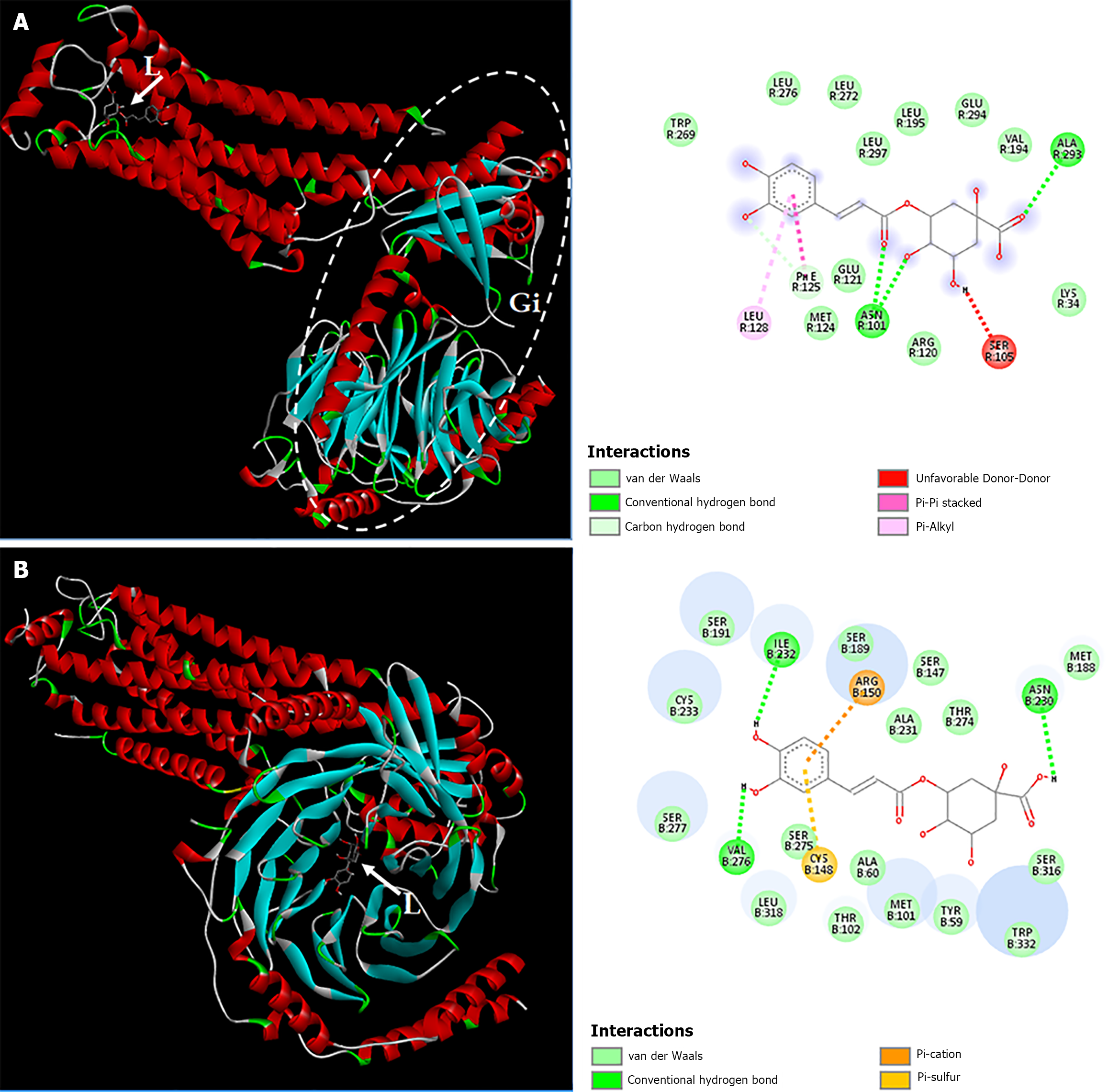
In the S1PR1/chlorogenic acid complex, chlorogenic acid bound both to the extracellular region of the seven-transmembrane receptor (7TM) (-8.7 kcal/mol; 18 interactions involving 17 residues of the 7TM domain) (Figure 9A) and to the β subunit of the Gi protein anchored to the 7TM on the cytoplasmic side (-8.4 kcal/mol; 21 interactions involving 21 residues of the Gi protein) (Figure 9B). IDO1 is a homodimeric heme-containing protein (chains A and B), with the heme group being essential for its catalytic activity. Molecular docking results indicate that chlorogenic acid binds to the catalytic site region of IDO1 through amino acid residues (eight interactions involving seven residues) and simultaneously interacts with the heme group (Figure 10).
All experiments related to pain manifestation (abdominal writhing test and formalin test) were conducted using female animals, considering that the larger sample size (eight animals per group) would not be influenced by additional variables associated with female samples. The remaining experiments were conducted using male Swiss mice.
The analgesic effects of substances against inflammatory pain can be assessed using the acetic acid–induced abdominal writhing model[25]. In this study, acetic acid was administered via intraperitoneal injection, inducing abdominal lesions in mice and leading to observable spasms or contortions. These contortions were subsequently quantified, allowing inference as to whether a reduction in the number of contortions occurred in groups treated with the tested substance. Nociception in this model is characterized by the presence of inflammatory mediators such as PGE2, PGF2α, PGI2, and other lipoxygenase products[26], along with the induction of TNF-α and IL-1β release. HAEBC proved to be effective in reducing abdominal writhing, with a significant decrease in the number of contortions from minute 12 onward at doses of 30 mg/kg and 100 mg/kg compared with the control group. These findings suggest that HAEBC, directly or indirectly, exhibits inhibitory properties on the release of pro-inflammatory mediators[27], thereby contributing to the antinociceptive process.
The formalin test evaluates antinociceptive and anti-inflammatory activity by analyzing two phases of the inflammatory process: The neurogenic and inflammatory phases[25]. During the neurogenic phase, formalin directly activates local nociceptors, occurring abruptly within the first 0-5 minutes after administration and extending until approximately 15 minutes, resulting in pain perception. In the inflammatory phase, pain arises from enhanced synaptic transmission in the spinal cord and from the release of local inflammatory mediators such as prostaglandins, serotonin, histamine, and bradykinin[28]. Studies indicate that the second phase of the formalin test results from a response to the first phase, during which central neurons become activated and sensitized due to peripheral inflammation, as well as from activation of primary afferent neurons[29]. HAEBC was effective in both phases, reducing the number of flinches in the neurogenic phase by 48.6% and producing a more pronounced reduction during the inflammatory phase, with an 83.9% decrease compared with the control group. These findings indicate that HAEBC exerts both central and peripheral antinociceptive effects, with greater efficacy observed during the second phase of the test[30].
To assess mechanical hyperalgesia, carrageenan was injected into the mouse paw, promoting acute and progressive edema proportional to the intensity of the inflammatory response. The von Frey test was subsequently applied to assess nociceptor sensitivity to mechanical stimuli[25]. HAEBC was effective in reducing the nociceptive response, except at the dose of 10 mg/kg. Doses of 30, 100, and 300 mg/kg produced statistically significant reductions in nociception, decreasing pain responses by 53.3%, 68.8%, and 76.8%, respectively. Paw edema was also significantly reduced following HAEBC administration; however, only the 100 mg/kg dose effectively reduced edema, with a decrease of 62.8%.
Carrageenan is a substance extracted from algae and composed of a complex group of polysaccharides, whereas formalin is an aqueous solution of formaldehyde (methanal), a volatile organic compound[31]. Both substances induce nociceptive responses and edema when injected into the mouse paw; however, their specific effects differ, making them suitable for distinct experimental purposes. Formalin stimulation induces paw flinching and licking behaviors, which are not observed following carrageenan injection. Formalin-induced edema develops slowly and is often dissociated from the peak behavioral response to intraplantar administration[32,33]. In contrast, carrageenan-induced edema and hyperalgesia develop more gradually and peak between 3 and 5 hours after injection[31], allowing for serial measurements and prolonged evaluation of treatment effects. Additionally, the absence of paw flinching facilitates edema measurement and performance of the von Frey test.
In the acute colitis model, acetic acid was administered rectally, inducing a pronounced inflammatory process characterized by macroscopic and histopathological alterations resulting from inflammatory mediators such as ROS, vasoactive amines, and eicosanoids[1]. A discrepancy was observed between macroscopic and histological findings. Macroscopic analysis revealed protective effects at doses of 30 and 100 mg/kg, whereas histological analysis demonstrated protective effects at doses of 30, 100, and 300 mg/kg. Microscopic evaluation showed that HAEBC preserved tissue architecture and reduced histological damage, as previously reported, demonstrating the extract’s efficacy in attenuating inflammatory processes.
Mesalazine did not reduce histological damage induced by acetic acid, which may be attributed to the fact that non-steroidal anti-inflammatory drugs is indicated for chronic intestinal disease exacerbations in coated tablet formulations rather than in solution form, as used in this study. Mesalazine inhibits inflammatory mediators in the intestinal mucosa, such as IL-1, IL-2, and TNF-α, suppresses the lipoxygenase pathway, and reduces NF-κB transcription[34,35]. Its therapeutic effect relies on topical action in the colonic mucosa, which is achieved through gastro-resistant formulations that prevent absorption in the small intestine and hepatic metabolism[35]. The absence of such a formulation may explain the lack of protective effects observed in this study.
Despite the relevance of these findings, several limitations must be acknowledged, including the absence of an optimized mesalazine formulation, the relatively small sample size, the lack of a chronic colitis model, and the absence of systemic cytokine quantification. These factors should be considered when interpreting the results.
Additionally, mesalazine is an aminosalicylate commonly prescribed for the treatment of IBD, which are chronic inflammatory conditions[36]. It is possible that the therapeutic effects of this drug were not observed due to the acute nature of the experimental model, and that a longer treatment period would be required to detect its activity.
Previous studies have demonstrated the antioxidant potential of Butia extracts. One study evaluated the antioxidant capacity of Butia eriospatha extract using Caenorhabditis elegans as an animal model[37], while another investigated extracts from Butia catarinensis and Butia eriospatha using the same model[17]. The former demonstrated that Butia extract, at all tested concentrations, significantly protected nematodes against oxidative damage induced by hydrogen peroxide. The latter study showed that ROS generation by the helminth’s metabolism was reduced compared with untreated animals.
Another investigation[38] evaluated the effects of Butia odorata pulp extract on oxidative stress, inflammatory, and metabolic parameters in Wistar rats subjected to tyloxapol-induced hyperlipidemia, revealing reductions in serum total cholesterol, triglycerides, and C-reactive protein levels in treated animals. Increased high-density lipoprotein concentrations and enhanced paraoxonase 1 activity were also observed. These findings support the conclusion that Butia odorata extract exhibits hypolipidemic and anti-inflammatory effects.
The role of ROS in inflammation and the importance of regulating their production in inflammatory diseases are well established[4,6]. The consistency between the present findings and previous studies strengthens the potential applicability of Butia extracts across a broad spectrum of diseases, whether through antioxidant mechanisms or modulation of pain and inflammation.
Computational analyses indicated that gallic acid, p-hydroxybenzoic acid, and chlorogenic acid interact with cellular targets of the SPHK1/S1P/S1PR1 and IDO1/kynurenine signaling pathways, as well as the COX-2/PGE2 axis. However, only chlorogenic acid formed stable complexes under physiological conditions, with binding affinity energies lower than -6.0 kcal/mol[39]. Despite its relatively low concentration in Butia capitata fruit compared with other phenolic acids[14], this stable interaction suggests that chlorogenic acid may modulate these signaling pathways.
The in silico results indicate that chlorogenic acid interacts with two targets of the SPHK1/S1P/S1PR1 axis. Binding to SPHK1 Likely inhibits its catalytic activity, as interaction occurs within the catalytic region, thereby reducing or blocking S1P formation. Chlorogenic acid may also bind to S1PR1 either at the extracellular 7TM domain or at the cytoplasmic Gi protein coupled to the receptor. In both scenarios, S1PR1 activation is impaired-either due to the absence of S1P binding or interruption of signal transduction via Gi protein inactivation. Simultaneous binding to both sites may further enhance pathway blockade. Thus, chlorogenic acid may exert a dual inhibitory effect on the SPHK1/S1P/S1PR1 signaling pathway.
Persistent activation of the SPHK1/S1P/S1PR1 pathway promotes sustained STAT3 activation[40], which inhibits apoptosis and drives tumorigenesis by modulating the expression of apoptotic regulators[41,42]. Inhibition of this pathway may therefore represent a strategy to limit cell differentiation processes associated with chronic inflammation in UC and the development of CRC. Notably, inhibitors targeting this pathway have demonstrated promising results in phase II and III clinical trials for severe UC[43].
COX-2 is an inducible isoform expressed in inflamed tissues and is responsible for synthesizing inflammatory mediators such as prostaglandins and leukotrienes. Structurally, COX-2 functions as a homodimer, with each subunit comprising an epidermal growth factor–like domain, a membrane-binding domain, and a catalytic domain[44]. In silico analyses indicate that chlorogenic acid binds to the catalytic domain of COX-2. Although it does not directly interact with the prosthetic heme group, binding to adjacent residues may induce conformational changes that impair substrate access, thereby negatively modulating COX-2 activity and disrupting the COX-2/PGE2 axis.
PGE2 is a lipid mediator synthesized from arachidonic acid by COX enzymes and acts through four receptor subtypes (EP1–EP4), mediating pyrexia, pain, and inflammation[45]. In the gastrointestinal tract, PGE2 plays key physiological roles, including maintenance of mucosal integrity. However, experimental and clinical evidence indicates that PGE2 also contributes to cancer initiation and progression, particularly in CRC[46]. Non-steroidal anti-inflammatory drugs such as ibuprofen and selective COX-2 inhibitors like celecoxib have been shown to reduce CRC risk, and aspirin use has been associated with a 40%-50% reduction in CRC-related mortality[47].
IDO1 expression has been detected in several cancers, including colorectal, lung, breast, oesophageal, gastric, and melanoma tumors[48], and is associated with poor prognosis[49,50]. IDO1 expression is induced by pro-inflammatory cytokines such as TNF-α and IFN-γ. By converting tryptophan into kynurenine, IDO1 suppresses local T-cell proliferation[51,52]. In tumor cells, kynurenine accelerates the cell cycle, favoring tumor proliferation[53]. In vitro, IDO1 promotes cell proliferation through activation of the Wnt/β-catenin pathway, leading to cytoplasmic accumulation and subsequent nuclear translocation of β-catenin[48]. Dysregulation of this pathway is closely associated with the development of multiple cancer types[54].
Computational simulations demonstrated that chlorogenic acid stably binds to the prosthetic group and residues of IDO1, likely inhibiting its enzymatic activity. Consequently, pathways involved in cancer development and progression, including CRC, may be attenuated.
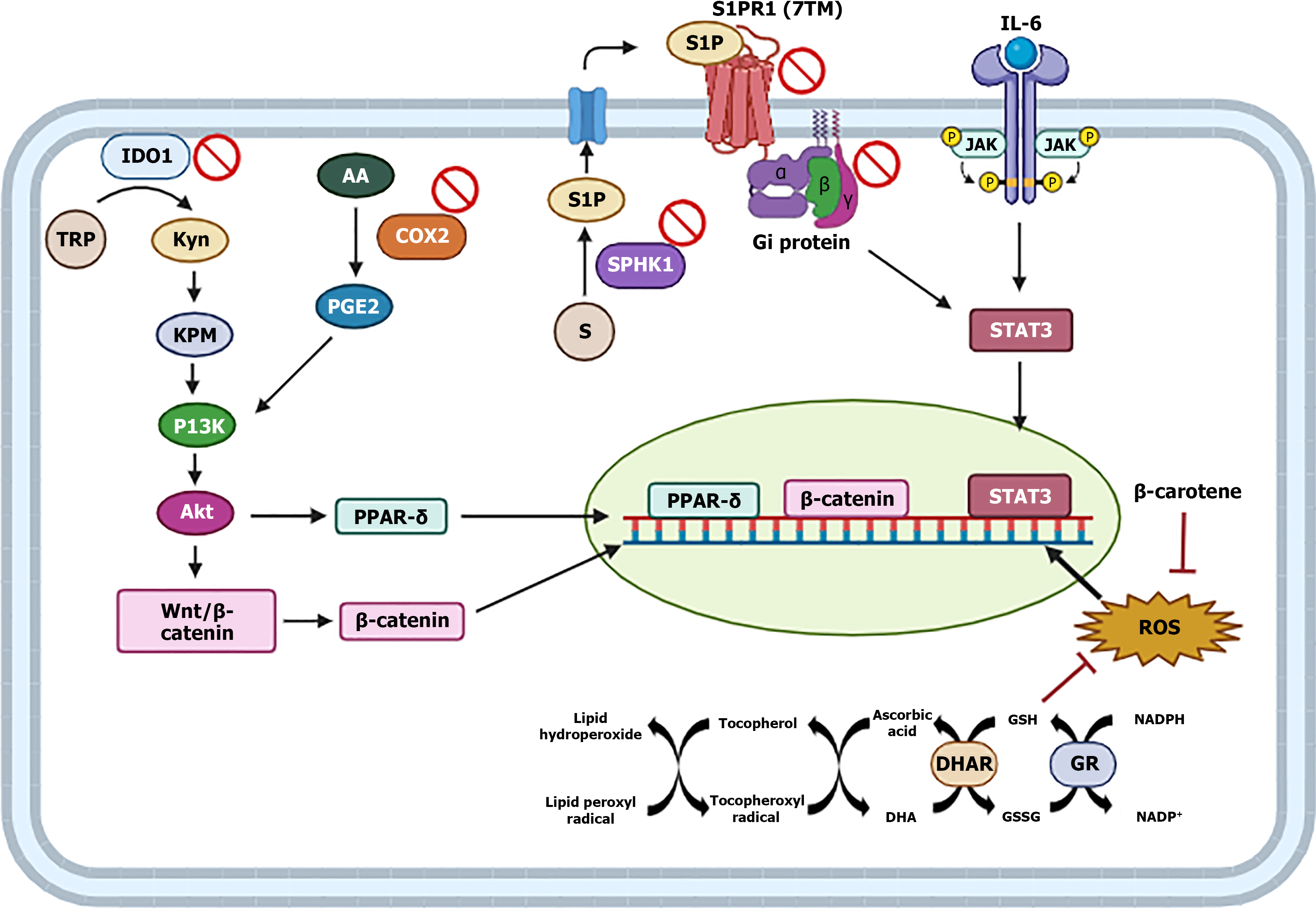
By integrating literature data on the phytochemical composition and bioactive properties of Butia capitata fruit pulp with the in silico findings of this study, it is possible to suggest that secondary metabolites and antioxidant molecules act synergistically to inhibit metabolic pathways associated with the initiation and maintenance of colitis-related inflammation, mitigate DNA damage that favors tumorigenesis, and suppress metabolic pathways that promote tumor proliferation (Figure 11).
It is important to emphasize that although molecular docking analyses provide insights into potential molecular targets, this study did not include in vivo or in vitro mechanistic assays to confirm modulation of the SPHK1/S1P/S1PR1 axis, the IDO1/kynurenine pathway, or COX-2/PGE2 signaling in colon tissue. No measurements of inflammatory mediators (e.g., TNF-α, IL-1β), myeloperoxidase activity, COX-2 expression, or STAT3 phosphorylation were performed. Therefore, the mechanistic interpretations proposed here should be considered hypothesis-generating rather than experimentally validated. Future studies employing chronic colitis models and molecular analyses will be essential to determine whether phenolic compounds from Butia capitata modulate these pathways in vivo.
In this preclinical study, HAEBC demonstrated anti-inflammatory and antinociceptive effects in models of acute pain and acetic acid–induced colitis, with preservation of tissue architecture and attenuation of inflammatory hyperalgesia. Our findings therefore relate specifically to colitis-induced injury, inflammation, and nociceptive modulation. Although the docking analysis suggests potential interactions between chlorogenic acid and inflammatory targets such as SPHK1, IDO1, and COX-2, these mechanistic insights remain hypothetical, as no molecular assays were conducted to confirm pathway modulation in vivo. Consequently, any inference regarding tumorigenesis refers only to mechanistic implications supported by existing literature on the inflammation–tumorigenesis axis, rather than to direct experimental evidence. In this context, the term “protective effects” denotes modulation of biological processes associated with early tumor-promoting conditions, not morphological tumor outcomes. Further studies using chronic colitis models and molecular or immunological analyses will be necessary to validate these proposed mechanisms and deepen under
The authors acknowledge the Universidade Federal do Paraná-Campus Toledo for the institutional and logistical support provided during the writing of the manuscript and the execution of the experiments.
| 1. | Sayers EW, Bolton EE, Brister JR, Canese K, Chan J, Comeau DC, Connor R, Funk K, Kelly C, Kim S, Madej T, Marchler-Bauer A, Lanczycki C, Lathrop S, Lu Z, Thibaud-Nissen F, Murphy T, Phan L, Skripchenko Y, Tse T, Wang J, Williams R, Trawick BW, Pruitt KD, Sherry ST. Database resources of the national center for biotechnology information. Nucleic Acids Res. 2022;50:D20-D26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2178] [Cited by in RCA: 1865] [Article Influence: 466.3] [Reference Citation Analysis (0)] |
| 2. | Campos FGCM, Figueiredo MN, Monteiro M, Nahas SC, Cecconello I. Incidence of colorectal cancer in young patients. Rev Col Bras Cir. 2017;44:208-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (2)] |
| 3. | McDowell C, Farooq U, Haseeb M. Inflammatory Bowel Disease. 2023 Aug 4. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025. [PubMed] |
| 4. | Li W, Zhao T, Wu D, Li J, Wang M, Sun Y, Hou S. Colorectal Cancer in Ulcerative Colitis: Mechanisms, Surveillance and Chemoprevention. Curr Oncol. 2022;29:6091-6114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 67] [Reference Citation Analysis (8)] |
| 5. | Hussain T, Tan B, Yin Y, Blachier F, Tossou MC, Rahu N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid Med Cell Longev. 2016;2016:7432797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 764] [Cited by in RCA: 1396] [Article Influence: 139.6] [Reference Citation Analysis (4)] |
| 6. | Frigerio S, Lartey DA, D'Haens GR, Grootjans J. The Role of the Immune System in IBD-Associated Colorectal Cancer: From Pro to Anti-Tumorigenic Mechanisms. Int J Mol Sci. 2021;22:12739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 7. | Barboza JN, da Silva Maia Bezerra Filho C, Silva RO, Medeiros JVR, de Sousa DP. An Overview on the Anti-inflammatory Potential and Antioxidant Profile of Eugenol. Oxid Med Cell Longev. 2018;2018:3957262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 176] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 8. | Paduch R, Kandefer-Szerszen M. Antitumor and Antiviral Activity of Pentacyclic Triterpenes. Mini-Rev Org Chem. 2014;11:262-268. [RCA] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 9. | Dias MC, Pinto DCGA, Silva AMS. Plant Flavonoids: Chemical Characteristics and Biological Activity. Molecules. 2021;26:5377. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 731] [Article Influence: 146.2] [Reference Citation Analysis (0)] |
| 10. | Maleki SJ, Crespo JF, Cabanillas B. Anti-inflammatory effects of flavonoids. Food Chem. 2019;299:125124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 762] [Article Influence: 108.9] [Reference Citation Analysis (1)] |
| 11. | Barbosa JR, de Carvalho Junior RN. Polysaccharides obtained from natural edible sources and their role in modulating the immune system: Biologically active potential that can be exploited against COVID-19. Trends Food Sci Technol. 2021;108:223-235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 112] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 12. | Faria JP, Almeida F, Silva LCRD, Vieira RF, Agostini-Costa TDS. Caracterização da polpa do coquinho-azedo (Butia capitata var capitata). Rev Bras Frutic. 2008;30:827-829. [DOI] [Full Text] |
| 13. | Helida MM, Telma MB, Juliana S, Herick FDJ, D`iemesson STM, Moacir P. Evaluating chemical composition of Butia capitata pulp among various populations and locations using multivariate analysis. Afr J Biotechnol. 2017;16:1902-1910. [DOI] [Full Text] |
| 14. | Tassoult M, Kati DE, Bachir-Bey M, Benouadah A, Rodriguez-Gutiérrez G. Valorization of date palm biodiversity: physico‐chemical composition, phenolic profile, antioxidant activity, and sensory evaluation of date pastes. Food Meas. 2021;15:2601-2612. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Barbosa MCDA, Rosa QDS, Cardoso LDM, Gomides AFDF, Barbosa LCDA, Sant’anna HMP, Pinheiro SS, Peluzio MDCG, Teixeira RDBL, Valente MAS. Composition proximate, bioactive compounds and antioxidant capacity of Butia capitata. Food Sci Technol. 2021. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 16. | Naczk M, Shahidi F. Extraction and analysis of phenolics in food. J Chromatogr A. 2004;1054:95-111. [PubMed] [DOI] [Full Text] |
| 17. | Camboim Rockett F, de Oliveira Schmidt H, Schmidt L, Rodrigues E, Tischer B, Ruffo de Oliveira V, Lima da Silva V, Rossini Augusti P, Hickmann Flôres S, Rios A. Phenolic compounds and antioxidant activity in vitro and in vivo of Butia and Opuntia fruits. Food Res Int. 2020;137:109740. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 18. | Wallace JL, MacNaughton WK, Morris GP, Beck PL. Inhibition of leukotriene synthesis markedly accelerates healing in a rat model of inflammatory bowel disease. Gastroenterology. 1989;96:29-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 304] [Article Influence: 8.2] [Reference Citation Analysis (1)] |
| 19. | Lengauer T, Rarey M. Computational methods for biomolecular docking. Curr Opin Struct Biol. 1996;6:402-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 613] [Cited by in RCA: 396] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 20. | Tian W, Chen C, Lei X, Zhao J, Liang J. CASTp 3.0: computed atlas of surface topography of proteins. Nucleic Acids Res. 2018;46:W363-W367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1318] [Cited by in RCA: 1539] [Article Influence: 192.4] [Reference Citation Analysis (0)] |
| 21. | Genovese M, Da Silva Pinto M, De Souza Schmidt Gonçalves A, Lajolo F. Bioactive Compounds and Antioxidant Capacity of Exotic Fruits and Commercial Frozen Pulps from Brazil. Food Sci Technol Int. 2008;14:207-214. [DOI] [Full Text] |
| 22. | Pereira NRL, Lopes B, Fagundes IV, de Moraes FM, Morisso FDP, Parma GOC, Zepon KM, Magnago RF. Bio-packaging based on cellulose acetate from banana pseudostem and containing Butia catarinensis extracts. Int J Biol Macromol. 2022;194:32-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 23. | Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J Comput Chem. 2009;30:2785-2791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21989] [Cited by in RCA: 17235] [Article Influence: 1013.8] [Reference Citation Analysis (0)] |
| 24. | Eberhardt J, Santos-Martins D, Tillack AF, Forli S. AutoDock Vina 1.2.0: New Docking Methods, Expanded Force Field, and Python Bindings. J Chem Inf Model. 2021;61:3891-3898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5851] [Cited by in RCA: 4203] [Article Influence: 840.6] [Reference Citation Analysis (4)] |
| 25. | Almeida Junior SD. In vivo methods for the evaluation of anti-inflammatory and antinoceptive potential. Braz J Pain. 2019;2. [DOI] [Full Text] |
| 26. | Parveen Z, Deng Y, Saeed MK, Dai R, Ahamad W, Yu YH. Antiinflammatory and analgesic activities of Thesium chinense Turcz extracts and its major flavonoids, kaempferol and kaempferol-3-O-glucoside. Yakugaku Zasshi. 2007;127:1275-1279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 27. | Moreira BO, de Carvalho AL, Alves CQ, Morbeck LLB, Cruz MP, Yatsuda R, David JP, David JM. Evaluation of anti-inflammatory, antinociceptive and biological activities of Cenostigma macrophyllum standardized extracts and determination and quantification of the main metabolites. RSC Adv. 2019;9:41256-41268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 28. | Batista E, Trindade H, Lira S, Muller J, Silva L, Batista M. Atividades antinociceptiva e antiinflamatória do extrato etanólico de Luehea divaricata. Rev Bras Plantas Med. 2016;18:433-441. [DOI] [Full Text] |
| 29. | Araújo FL, Melo CT, Rocha NF, Moura BA, Leite CP, Amaral JF, Barbosa-Filho JM, Gutierrez SJ, Vasconcelos SM, Viana GS, de Sousa FC. Antinociceptive effects of (O-methyl)-N-benzoyl tyramine (riparin I) from Aniba riparia (Nees) Mez (Lauraceae) in mice. Naunyn Schmiedebergs Arch Pharmacol. 2009;380:337-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Pereira MC, Steffens RS, Jablonski A, Hertz PF, Rios ADO, Vizzotto M, Flôres SH. Characterization, bioactive compounds and antioxidant potential of three Brazilian fruits. J Food Compos Anal. 2013;29:19-24. [RCA] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 31. | Morris CJ. Carrageenan-induced paw edema in the rat and mouse. Methods Mol Biol. 2003;225:115-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 271] [Article Influence: 11.8] [Reference Citation Analysis (2)] |
| 32. | Wheeler-Aceto H, Porreca F, Cowan A. The rat paw formalin test: comparison of noxious agents. Pain. 1990;40:229-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 247] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 33. | Rivot JP, Montagne-Clavel J, Besson JM. Subcutaneous formalin and intraplantar carrageenan increase nitric oxide release as measured by in vivo voltammetry in the spinal cord. Eur J Pain. 2002;6:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 34. | Iacucci M, de Silva S, Ghosh S. Mesalazine in inflammatory bowel disease: a trendy topic once again? Can J Gastroenterol. 2010;24:127-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 82] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 35. | Nidhi, Rashid M, Kaur V, Hallan SS, Sharma S, Mishra N. Microparticles as controlled drug delivery carrier for the treatment of ulcerative colitis: A brief review. Saudi Pharm J. 2016;24:458-472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 92] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 36. | Veloso PM, Machado R, Nobre C. Mesalazine and inflammatory bowel disease - From well-established therapies to progress beyond the state of the art. Eur J Pharm Biopharm. 2021;167:89-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 37. | Tambara AL, da Silveira ÉC, Soares ATG, Salgueiro WG, Rodrigues CF, Boldori JR, de Ávila DS, Denardin CC. Butiá fruit extract (Butia eriospatha) protects against oxidative damage and increases lifespan on Caenorhabditis elegans. J Food Biochem. 2020;44:e13139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 38. | Ramos VP, da Silva PG, Oliveira PS, Bona NP, Soares MSP, Cardoso JS, Hoffmann JF, Chaves FC, Schneider A, Spanevello RM, Lencina CL, Stefanello FM, Tavares RG. Hypolipidemic and anti-inflammatory properties of phenolic rich Butia odorata fruit extract: potential involvement of paraoxonase activity. Biomarkers. 2020;25:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 39. | Pantsar T, Poso A. Binding Affinity via Docking: Fact and Fiction. Molecules. 2018;23:1899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 559] [Cited by in RCA: 383] [Article Influence: 47.9] [Reference Citation Analysis (0)] |
| 40. | Lee H, Deng J, Kujawski M, Yang C, Liu Y, Herrmann A, Kortylewski M, Horne D, Somlo G, Forman S, Jove R, Yu H. STAT3-induced S1PR1 expression is crucial for persistent STAT3 activation in tumors. Nat Med. 2010;16:1421-1428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 322] [Cited by in RCA: 336] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 41. | Taniguchi K, Karin M. IL-6 and related cytokines as the critical lynchpins between inflammation and cancer. Semin Immunol. 2014;26:54-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 583] [Cited by in RCA: 539] [Article Influence: 44.9] [Reference Citation Analysis (7)] |
| 42. | Saadatdoust Z, Pandurangan AK, Ananda Sadagopan SK, Mohd Esa N, Ismail A, Mustafa MR. Dietary cocoa inhibits colitis associated cancer: a crucial involvement of the IL-6/STAT3 pathway. J Nutr Biochem. 2015;26:1547-1558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 43. | Wang J, Goren I, Yang B, Lin S, Li J, Elias M, Fiocchi C, Rieder F. Review article: the sphingosine 1 phosphate/sphingosine 1 phosphate receptor axis - a unique therapeutic target in inflammatory bowel disease. Aliment Pharmacol Ther. 2022;55:277-291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 44. | Alexanian A, Sorokin A. Cyclooxygenase 2: protein-protein interactions and posttranslational modifications. Physiol Genomics. 2017;49:667-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 62] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 45. | Kawahara K, Hohjoh H, Inazumi T, Tsuchiya S, Sugimoto Y. Prostaglandin E2-induced inflammation: Relevance of prostaglandin E receptors. Biochim Biophys Acta. 2015;1851:414-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 345] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 46. | Chen XY, Wang ZC, Li H, Cheng XX, Sun Y, Wang XW, Wu ML, Liu J. Nuclear translocations of beta-catenin and TCF4 in gastric cancers correlate with lymph node metastasis but probably not with CD44 expression. Hum Pathol. 2005;36:1294-1301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 47. | Benson AB 3rd. Epidemiology, disease progression, and economic burden of colorectal cancer. J Manag Care Pharm. 2007;13:S5-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 82] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 48. | Li F, Zhang R, Li S, Liu J. IDO1: An important immunotherapy target in cancer treatment. Int Immunopharmacol. 2017;47:70-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 122] [Article Influence: 13.6] [Reference Citation Analysis (2)] |
| 49. | Carvajal-Hausdorf DE, Mani N, Velcheti V, Schalper KA, Rimm DL. Objective measurement and clinical significance of IDO1 protein in hormone receptor-positive breast cancer. J Immunother Cancer. 2017;5:81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 50. | Ferdinande L, Decaestecker C, Verset L, Mathieu A, Moles Lopez X, Negulescu AM, Van Maerken T, Salmon I, Cuvelier CA, Demetter P. Clinicopathological significance of indoleamine 2,3-dioxygenase 1 expression in colorectal cancer. Br J Cancer. 2012;106:141-147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 113] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 51. | Munn DH, Shafizadeh E, Attwood JT, Bondarev I, Pashine A, Mellor AL. Inhibition of T cell proliferation by macrophage tryptophan catabolism. J Exp Med. 1999;189:1363-1372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1312] [Cited by in RCA: 1247] [Article Influence: 46.2] [Reference Citation Analysis (0)] |
| 52. | Santhanam S, Alvarado DM, Ciorba MA. Therapeutic targeting of inflammation and tryptophan metabolism in colon and gastrointestinal cancer. Transl Res. 2016;167:67-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 77] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 53. | Thaker AI, Rao MS, Bishnupuri KS, Kerr TA, Foster L, Marinshaw JM, Newberry RD, Stenson WF, Ciorba MA. IDO1 metabolites activate β-catenin signaling to promote cancer cell proliferation and colon tumorigenesis in mice. Gastroenterology. 2013;145:416-25.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 162] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 54. | Chiarini F, Paganelli F, Martelli AM, Evangelisti C. The Role Played by Wnt/β-Catenin Signaling Pathway in Acute Lymphoblastic Leukemia. Int J Mol Sci. 2020;21:1098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |