Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.116640
Revised: January 20, 2026
Accepted: March 2, 2026
Published online: May 15, 2026
Processing time: 149 Days and 1 Hours
The intensification of global aging has led to an increasing burden of digestive system tumors in the elderly. The comprehensive geriatric assessment (CGA) provides important basis for developing individualized treatment plans by systematically assessing factors such as patient function, nutrition, and comor
To examine the value of the CGA in treatment decision-making and survival prognosis among elderly patients with digestive system tumors.
This retrospective study analyzed the clinical data of 210 elderly patients with digestive system tumors admitted between April 2022 and July 2025. Of these, 108 patients who received traditional diagnostic and therapeutic decisions (April 2022 to December 2023) constituted the control group, whereas 102 patients who received CGA-guided diagnostic and therapeutic decisions (January 2024 to July 2025) comprised the observation group. The initial treatment strategy, intensity, safety, and tolerance, as well as progression-free survival (PFS), were compared between the two groups.
Fewer patients in the observation group received standard treatment and standard-intensity chemotherapy compared with the control group, whereas more received reduced-intensity chemotherapy. The overall incidence of adverse reactions (≥ grade 3) was 28.4% in the observation group, compared with 45.4% in the control group. Treatment delay and discontinuation rates were lower in the observation group (20.6% and 11.8%, respectively) than in the control group (32.4% and 24.1%, respectively). The treatment completion rate was higher in the observation group (88.2%) than in the control group (75.9%). The median PFS was longer in the observation group (11.5 months, 95% confidence interval: 10.2-12.8) than in the control group (9.1 months, 95% confidence interval: 8.0-10.2). All differences were statistically significant (P < 0.05). Multivariate Cox regression analysis showed that a CGA-assessed “frail” status was an independent risk factor affecting patient PFS (P < 0.05).
CGA-guided diagnostic and therapeutic strategies facilitate precise, individualized decision-making for elderly patients with digestive system tumors. By tailoring treatment selection and intensity to overall health status, this approach optimizes the balance between efficacy and safety and is associated with improved PFS, supporting its clinical utility.
Core Tip: Comprehensive geriatric assessment plays a pivotal role in the clinical management of elderly patients with digestive system tumors. By systematically evaluating their physical function, cognitive status, nutritional condition and social support, comprehensive geriatric assessment precisely guides the formulation of personalized treatment regimens. Notably, this evidence-based approach effectively mitigates treatment-related toxicities, enhances patients’ treatment tolerance, and extends their progression-free survival, thus exerting remarkable clinical benefits and practical value in geriatric oncology practice.
- Citation: Zhu L, Liu XF. Comprehensive geriatric assessment guiding treatment and survival in elderly digestive tumor patients: A retrospective study. World J Gastrointest Oncol 2026; 18(5): 116640
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/116640.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.116640
With the accelerating trend of global population aging, the incidence of malignant tumors shows a significant positive correlation with age, making advanced age the strongest risk factor[1,2]. Digestive system tumors, such as esophageal, gastric, colorectal, liver, and pancreatic cancers, are among the most common malignancies in China, ranking high in incidence and mortality rates, particularly burdening the elderly population[3,4]. Traditional oncology decision-making primarily relies on factors such as tumor-node-metastasis (TNM) stage, pathological type, and molecular markers of the tumor. However, it often overlooks the impact of geriatric syndromes, such as the patient's overall functional status, comorbidities, cognition, psychological state, and social support. This oversight can lead to overly conservative ap
Recently, comprehensive geriatric assessment (CGA), as a multidimensional tool, has gradually shown unique value in diagnosing and treating elderly patients with cancer[7,8]. By systematically analyzing information regarding the patient’s physical function, mental health, social support, and comorbidities, CGA can comprehensively reflect the overall health status of elderly patients. Thus, in this retrospective study we analyzed the clinical data of 210 elderly patients with digestive system tumors admitted to our hospital between April 2022 and July 2025. The study aimed to examine the role of CGA in treatment decision-making and survival prognosis, thereby providing a scientific basis for optimizing individualized treatment.
This study retrospectively analyzed clinical data of 210 elderly patients with digestive system tumors admitted to our hospital from April 2022 to July 2025. Of these patients, 108 admitted from April 2022 to December 2023 who received traditional oncology decision-making comprised the control group, whereas 102 patients admitted from January 2024 to July 2025 who received CGA-guided oncology decision-making constituted the observation group. The study protocol was approved by the hospital ethics committee.
Inclusion criteria were as follows: (1) Age ≥ 65 years; (2) Diagnosis of a digestive system malignant tumor by histopa
Exclusion criteria were as follows: (1) Concurrent primary malignant tumors in other organ systems; (2) Substantially incomplete clinical data; (3) Inability to cooperate with relevant assessments due to severe mental illness or cognitive impairment; (4) Expected survival of less than 3 months (terminal stage); and (5) Loss to follow-up.
Baseline demographic and clinical characteristics were balanced between the groups. No statistically significant differences were observed in age, sex distribution, tumor type, or ECOG performance status (all P > 0.05), indicating good comparability between the cohorts (Table 1).
| Group | n | Age (year), mean ± SD | Gender | Tumor type | ECOG grade (point) | |||||
| Male | Female | Colorectal cancer | Gastric cancer | Esophageal cancer | Liver cancer | Pancreatic cancer | ||||
| Observation group | 102 | 76.5 ± 4.8 | 59 (57.8) | 43 (42.2) | 46 (45.1) | 28 (27.5) | 10 (9.8) | 12 (11.1) | 6 (5.9) | 1.2 ± 0.5 |
| Control group | 108 | 77.1 ± 5.2 | 65 (60.2) | 43 (39.8) | 48 (44.4) | 32 (29.6) | 12 (11.1) | 10 (9.8) | 6 (5.6) | 1.1 ± 0.4 |
| χ2/t value | - | 0.851 | 0.102 | 1.234 | 1.666 | |||||
| P value | - | 0.396 | 0.749 | 0.872 | 0.107 | |||||
Control group: Patients in the control group were treated based on traditional oncology decision-making: (1) After admission, the attending physician collected medical history, performed physical examination, and completed imaging studies (computed tomography, magnetic resonance imaging, positron emission tomography-computed tomography) and laboratory tests to diagnose the tumor and determine its stage; (2) A multidisciplinary team, comprising medical oncologists, surgeons, and radiation oncologists, discussed the case. Based on comprehensive consideration of tumor factors (TNM stage, pathological type, differentiation degree, molecular markers like Kirsten rat sarcoma viral oncogene homolog/neuroblastoma rat sarcoma viral oncogene homolog/B-Raf proto-oncogene, serine/threonine kinase, human epidermal growth factor receptor 2, microsatellite instability) and general patient factors (age, ECOG score, routine blood test results, liver and kidney function, etc.), a treatment plan - such as surgery, chemotherapy, radiotherapy, targeted therapy, or immunotherapy - was formulated; and (3) During treatment, the plan was adjusted promptly based on the patient’s tolerance and short-term efficacy. In case of severe adverse reactions or disease progression, the treatment strategy was re-evaluated and adjusted.
Observation group: Patients in the observation group underwent treatment guided by CGA-based oncology decision-making: (1) After admission, in addition to routine examinations, a unified CGA was performed by trained geriatricians and oncology nurses, covering eight domains: Comorbidities, functional status, nutritional status, cognitive function, psychological state, frailty, polypharmacy, and social support; (2) According to the CGA results, patients were cate
| CGA assessment dimensions | Assessment tool/method | Abnormality threshold | General interventions |
| Comorbidities | CCI | CCI ≥ 2 | Refer to geriatrics or relevant specialty; optimize comorbidity management |
| Functional status | Activities of Daily Living scale | Total score < 100 | Refer to rehabilitation medicine; devise a rehab plan; arrange home-care services |
| Nutritional status | MNA-SF | ≤ 11 points | Refer to clinical nutrition; provide dietary counseling, oral nutritional supplements or mandatory nutritional support |
| Cognitive function | MMSE | Illiterate ≤ 17; primary-school ≤ 20; middle-school ≤ 24 | Enhance communication with primary caregiver; remind and supervise medication; schedule cognitive training |
| Psychological status | GDS-15 | ≥ 5 points | Refer to psychiatry/psychology; provide psychological counseling; strengthen family emotional support |
| Frailty status | CFS | Grade ≥ 4 | Use as core input for MDT decisions; initiate comprehensive nutrition, rehabilitation and supportive care |
| Polypharmacy | Medication list review | Concurrent use of ≥ 5 drugs | Clinical pharmacist or geriatrician conducts medication reconciliation; discontinue non-essential or inappropriate drugs |
| Social support | Structured interview | Living alone/no stable caregiver/financial hardship | Social-worker intervention; assist with subsidy applications; link to community support services |
| CGA grade | Target | Core treatment strategy | Specific example |
| Health | Cure | Standard neoadjuvant/adjuvant chemotherapy + radical surgery | Perioperative chemotherapy: Standard FLOT regimen (doxorubicin + oxaliplatin + fluorouracil/Leucovorin) is administered.rmed |
| Surgery: Radical gastrectomy (D2 standard) is perfo | |||
| Postoperative care: Complete all planned adjuvant chemotherapy cycles | |||
| Fragile | Control and function equally important | Optimize the standard protocol to reduce treatment-related risks and ensure successful completion of treatment | Pretreatment support: Initiate intensive nutritional support prior to chemotherapy and administer G-CSF prophylactically |
| Perioperative chemotherapy: Employ the less toxic SOX (tegafur + oxaliplatin) or XELOX (capecitabine + oxaliplatin) combination regimens, with initial doses reduced to 80% of standard levels | |||
| Surgery: Perform radical resection, followed by adjustment of adjuvant chemotherapy based on postoperative recovery | |||
| Caducity | Quality of life | Avoid high-intensity radical treatment and focus on controlling tumor-related symptoms, maintaining the feeding channel and quality of life | Nonsurgical local treatment: Palliative radiotherapy to control bleeding or pain |
| Systemic treatment: Monotherapy (e.g., tegafur) or targeted therapy (e.g., trastuzumab for HER2-positive patients) | |||
| Core measures: Enhanced nutritional support; professional pain and symptom management; if obstruction exists, perform gastrojejunostomy or place intestinal stent |
Follow-up: All patients were followed up regularly until the cutoff date of October 15, 2025.
Differences in initial treatment strategy, treatment intensity, safety, and tolerability, as well as survival prognosis indicators, were compared between the two groups.
Initial treatment strategy: The initial treatment modalities received by patients in both groups were recorded and categorized primarily into standard therapy (curative surgery + adjuvant chemotherapy), palliative therapy (palliative chemotherapy/targeted therapy/immunotherapy), or supportive care only.
Treatment intensity: Treatment intensity was evaluated using the relative dose intensity (RDI) of chemotherapeutic drugs, calculated as RDI = (actual dose/planned dose) × 100%. RDI ≥ 85% was defined as standard intensity, 60%-85% as reduced intensity, and < 60% as low intensity.
Treatment safety: According to the Common Terminology Criteria for Adverse Events version 5.0, the occurrence of all chemotherapy-related adverse reactions of grade ≥ 3 (including nausea or vomiting, diarrhea, myelosuppression, liver or kidney dysfunction, neurotoxicity, etc.) during treatment was recorded for groups.
Treatment tolerability: The treatment delay (chemotherapy delayed > 7 days due to toxicity), discontinuation (per
Survival prognosis indicator: Progression-free survival (PFS) was used for evaluation, and the impact of CGA classification on PFS was analyzed.
Data were analyzed using SPSS software (version 26.0). Data conforming to a normal distribution according to the Shapiro-Wilk test were expressed as mean ± SD and compared between groups using the t-test. Count data were expressed as n (%) and compared between groups using the χ2 or Fisher’s exact test. Survival curves were plotted using the Kaplan-Meier method and compared using the Log-rank test. Multivariate survival analysis was performed using the Cox proportional hazards regression model. A P value < 0.05 was considered statistically significant.
Significant differences were observed between the two groups regarding initial treatment strategies and chemotherapy intensity (Table 4). The proportion of patients receiving standard treatment was significantly lower in the observation group compared with the control group (35.3% vs 48.1%, P = 0.035), whereas a higher proportion of patients in the observation group received palliative therapy (46.1% vs 36.1%). Regarding treatment intensity, fewer patients in the observation group received standard-intensity chemotherapy compared with the control group (41.2% vs 60.2%, P = 0.006), whereas the proportion of patients receiving reduced-intensity chemotherapy was significantly higher in the observation group (47.1% vs 29.6%, P = 0.008) compared with the control group. No significant difference was found in the proportion of patients receiving low-intensity chemotherapy between the two groups (P = 0.709). These findings indicate that CGA-guided decision-making was associated with more frequent adjustment of treatment intensity while maintaining individualized treatment strategies (Table 4).
| Group | n | Initial treatment strategy | Treatment intensity | ||||
| Standard treatment | Palliative therapy | Supportive care only | Normal intensity | Reduced intensity | Low intensity | ||
| Observation group | 102 | 35 (35.3) | 48 (46.1) | 19 (18.6) | 42 (41.2) | 48 (47.1) | 12 (11.8) |
| Control group | 108 | 53 (48.1) | 38 (36.1) | 17 (15.7) | 65 (60.2) | 32 (29.6) | 11 (10.2) |
| χ2/t value | - | 4.427 | 2.686 | 0.332 | 7.699 | 6.941 | 0.139 |
| P value | - | 0.035 | 0.101 | 0.564 | 0.006 | 0.008 | 0.709 |
The overall incidence of grade ≥ 3 treatment-related adverse events was significantly lower in the observation group (28.4%) than in the control group (45.4%; P = 0.009; Table 5). Specifically, the observation group demonstrated reduced rates of severe neutropenia (11.8% vs 23.1%), thrombocytopenia (7.8% vs 13.9%), diarrhea (4.9% vs 10.2%), and neurotoxicity (3.9% vs 7.4%) compared with the control group. Although individual adverse events varied in frequency, the overall reduction in severe toxicity was statistically significant, indicating improvement in safety profiles under CGA-guided management.
| Group | n | Neutropenia | Thrombocytopenia | Diarrhea | Neurotoxicity | Overall grade ≥ 3 adverse events |
| Observation group | 102 | 12 (11.8) | 8 (7.8) | 5 (4.9) | 4 (3.9) | 29 (28.4) |
| Control group | 108 | 25 (23.1) | 15 (13.9) | 11 (10.2) | 8 (7.4) | 49 (45.4) |
| χ2/t value | - | - | - | - | - | 6.834 |
| P value | - | - | - | - | - | 0.009 |
Rates of treatment delay (20.6%) and discontinuation (11.8%) were lower in the observation group than in the control group (32.4% and 24.1%, respectively), whereas the completion rate was higher (88.2% vs 75.9%). The differences were statistically significant (P < 0.05; Table 6).
| Group | n | Treatment delay | Treatment discontinuation | Treatment completion |
| Observation group | 102 | 21 (20.6) | 12 (11.8) | 90 (88.2) |
| Control group | 108 | 35 (32.4) | 26 (24.1) | 82 (75.9) |
| χ2/t value | - | 3.945 | 5.678 | 5.512 |
| P value | - | 0.047 | 0.017 | 0.019 |
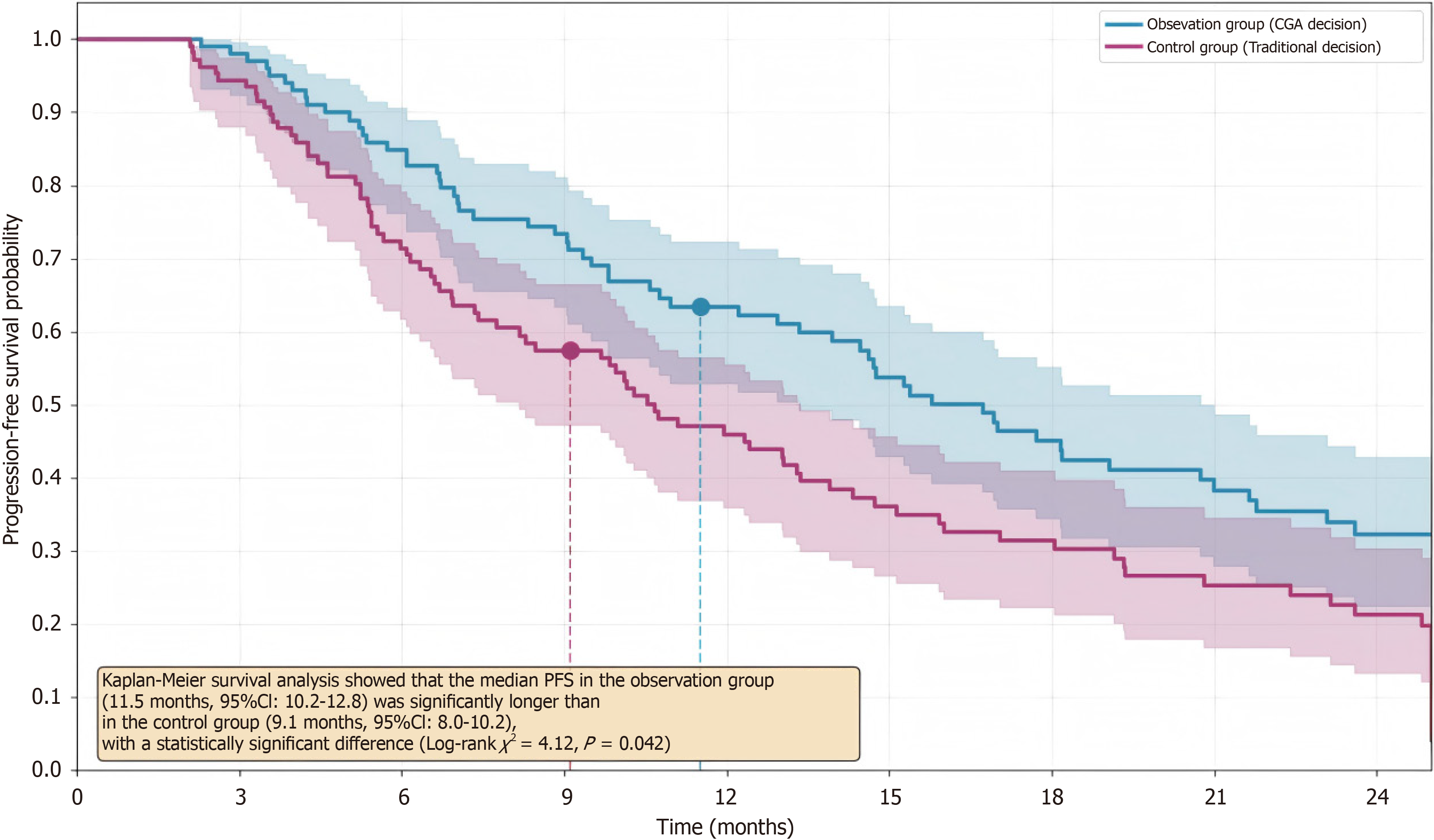
The median PFS was significantly longer in the observation group (11.5 months, 95% confidence interval: 10.2-12.8) than in the control group (9.1 months, 95% confidence interval: 8.0-10.2). The difference was statistically significant (Log-rank χ2 = 4.12, P = 0.042; Figure 1).
Multivariate Cox regression, adjusted for age, ECOG score, and TNM stage, showed that a CGA-assessed “vulnerable” status was an independent risk factor affecting patient PFS (P < 0.05; Table 7).
| Variable | HR | 95%CI | P value |
| Age (per five-year increase) | 1.32 | 1.05-1.66 | 0.018 |
| ECOG score (≥ 2 vs < 2) | 1.87 | 1.24-2.82 | 0.003 |
| TNM stage (stage IV vs I-III) | 2.45 | 1.65-3.64 | < 0.001 |
| Comprehensive geriatric assessment category (ref = control group) | - | - | 0.028 |
| Fit | 0.65 | 0.42-1.01 | 0.055 |
| Vulnerable | 0.71 | 0.50-0.99 | 0.046 |
| Frail | 1.05 | 0.62-1.78 | 0.856 |
Elderly patients with cancer represent a highly heterogeneous population in whom physiological age often diverges from chronological age. The prevalence of geriatric syndromes such as comorbidities, cognitive impairment, functional decline, malnutrition, polypharmacy, and psychosocial and social support issues make diagnostic and therapeutic decision-making for this group highly challenging[9,10]. CGA is a multidimensional, interdisciplinary tool that provides clinicians with precise “profiles” of elderly patients with cancer by evaluating multiple domains, including physical function, nutritional status, cognitive and psychological function, and social support[11,12]. Studies have shown that the application of CGA in breast, lung, urological cancers, as well as other cancers, can predict treatment complications, toxicities, and survival outcomes, and guide treatment adjustments[13,14]. Recent updates from the International Society of Geriatric Oncology strongly recommend CGA as a standard component of oncologic assessment for elderly people[15,16]. Accordingly, our institution began implementing CGA-guided oncology decision-making for elderly patients with digestive system tumors in January 2024, achieving relatively ideal results.
Our findings showed that, compared with the control group, the observation group had a significantly lower proportion of patients receiving standard therapy and standard-intensity chemotherapy (P < 0.05), and a higher proportion receiving reduced-intensity chemotherapy (P < 0.05). Traditional oncology decision-making - based primarily on biological indicators such as TNM stage and pathological type - has inherent limitations in elderly patients whose physiological reserve is diminished and comorbidities are complex. Such approaches may overestimate tolerance in “seemingly healthy” elderly patients, leading to overtreatment and severe toxicity[17,18], while underestimating the potential for active treatment in “vulnerable” patients whose condition could improve with appropriate intervention, resulting in undertreatment[19,20].
CGA systematically evaluates multiple domains - comorbidities, function, nutrition, cognition, psychology, frailty, medication use, and social support - to refine the distinction between physiological age and health status, categorizing patients into “fit”, “vulnerable”, and “frail”[21,22]. Standard or near-standard regimens can still be considered in patients identified as “fit” despite advanced age, enabling curative or optimal disease control while avoiding unnecessary undertreatment based on age alone[23,24]. Conversely, “vulnerable” patients possess limited physiological reserve and face high risks from unmodified standard therapy. For these individuals, the CGA-guided strategy in this study employed reduced-intensity chemotherapy and enhanced supportive care, successfully balancing tumor control with maintenance of function and quality of life. This aligns with the findings of Bonneau et al[25], who reported that CGA altered nearly one-third of treatment decisions in elderly prostate cancer patients, most often by reducing treatment intensity or increasing supportive care. For frail patients, our decision framework recommended avoiding high-intensity curative treatments and instead emphasizing comfort and quality of life - reflecting the geriatric oncology principle that “sometimes, no treatment is the best treatment”. For those with very limited life expectancy, transitioning to a palliative care model focused on symptom relief and psychosocial support represents a humane and patient-centered approach.
In addition, the incidence of grade ≥ 3 adverse events, treatment delays, and treatment discontinuations was lower in the observation group than in the control group (P < 0.05), while the treatment completion rate was higher (P < 0.05). Elderly patients with digestive system tumors frequently experience polypharmacy, malnutrition, and mood disorders, all of which impair tolerance to systemic therapy. The CGA-guided model enables early identification of risk factors such as frailty, nutritional deficiency, and functional dependence, allowing clinicians to anticipate vulnerability and implement targeted interventions before initiating treatment. By integrating anti-cancer therapy with concurrent geriatric interventions, this model improves patients’ overall physiological and psychological resilience, reduces complications, and facilitates completion of planned therapy[26]. A systematic review by Disalvo et al[27] similarly concluded that CGA enhances treatment completion rates and reduces toxicity in elderly patients receiving systemic anti-cancer therapy, consistent with our findings.
The median PFS was also significantly longer in the observation group than in the control group (P < 0.05), and a CGA-assessed vulnerable status emerged as an independent risk factor for PFS (P < 0.05). Although treatment intensity appeared “milder” in the observation group, their median PFS was longer. This seemingly paradoxical result de
This study has limitations. The follow-up duration was relatively short, and the sample size was small, which may introduce selection and information bias. Future large-scale, multicenter, prospective randomized controlled trials are warranted to comprehensively validate the long-term efficacy and safety of CGA-guided oncology decision-making for elderly patients with digestive system tumors.
The application of CGA-based diagnosis and treatment decisions in elderly patients with digestive system tumors helps guide the formulation of more individualized treatment plans. This approach effectively reduces treatment toxicity, improves treatment tolerance, and prolongs PFS, while ensuring efficacy; hence, is worthy of application.
| 1. | Giger AW, Ditzel HM, Ewertz M, Ditzel H, Jørgensen TL, Pfeiffer P, Lund C, Ryg J. Effect of comprehensive geriatric assessment on hospitalizations in older adults with frailty initiating curatively intended oncologic treatment: The PROGNOSIS-RCT study. J Geriatr Oncol. 2024;15:101821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 2. | Hu Q, Yang X. Impact of Comprehensive Geriatric Assessment on Treatment Strategies and Complications in Older Adults With Colorectal Cancer Considering Surgery. J Surg Oncol. 2025;131:100-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Hosoi T, Yamana H, Matsumoto S, Matsui H, Fushimi K, Akishita M, Yasunaga H, Ogawa S. Implementation status of comprehensive geriatric assessment among older inpatients: A nationwide retrospective study. Geriatr Gerontol Int. 2024;24:904-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 4. | Teraishi F, Shoji R, Kondo Y, Kagawa S, Tamura R, Matsuoka Y, Morimatsu H, Mitsuhashi T, Fujiwara T. Comprehensive geriatric assessment as an indicator of postoperative recovery in older patients with colorectal cancer. J Geriatr Oncol. 2024;15:101837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 5. | François E, Magné N, Boulahssass R, Ronchin P, Huguenet V, De Lavigerie B, Nouhaud E, Cattenoz C, Martel-Lafay I, Terret C, Artru P, Clavere P, Tchalla A, Vendrely V, Duc S, Boige V, De Sousa Carvalho N, Gal J, De Bari B. Short course radiotherapy versus radiochemotherapy for locally advanced rectal cancers in the elderly (UNICANCER PRODIGE 42/GERICO 12 study): Quality of life and comprehensive geriatric assessment. Radiother Oncol. 2024;193:110144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 6. | Nishijima TF, Shimokawa M, Esaki T, Morita M, Toh Y, Muss HB. Comprehensive geriatric assessment: Valuation and patient preferences in older Japanese adults with cancer. J Am Geriatr Soc. 2023;71:259-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Banh Chong T, Grosclaude P, Chabrillac E, Lozano S, Vergez S, Mourey L, Dupret-Bories A. Outcomes of comprehensive geriatric assessment and surgical management in head and neck cancers of the elderly: an observational study. Eur Arch Otorhinolaryngol. 2023;280:329-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 8. | Tarchand GR, Morrison V, Klein MA, Watkins E. Use of Comprehensive Geriatric Assessment in Oncology Patients to Guide Treatment Decisions and Predict Chemotherapy Toxicity. Fed Pract. 2021;38:S22-S28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 9. | Katayama H, Mizusawa J, Fukuda H, Nakamura S, Nakamura K, Saijo N, Yokoyama A, Ohe Y, Shinkai T, Nakagawa K, Abe T, Mitsuoka S, Okamoto H, Yamamoto N, Yoshioka H, Ando M, Tamura T, Takeda K. Prognostic impact of geriatric assessment in elderly patients with non-small cell lung cancer: an integrated analysis of two randomized phase III trials (JCOG1115-A). Jpn J Clin Oncol. 2021;51:685-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 10. | Pecoraro A, Testa GD, Marandino L, Albiges L, Bex A, Capitanio U, Cappiello I, Masieri L, Mir C, Roupret M, Serni S, Ungar A, Rivasi G, Campi R. Frailty and Renal Cell Carcinoma: Integration of Comprehensive Geriatric Assessment into Shared Decision-making. Eur Urol Oncol. 2025;8:190-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 11. | Chun YJ, Kim KJ, Lee JY, Choi JW, Kim TH, Kim HR, Cho BC, Hong MH, Kim CO. The prognostic value of comprehensive geriatric assessment on the management of older patients with small cell lung cancer. Korean J Intern Med. 2023;38:254-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 12. | Penning Y, El Asmar A, Moreau M, Raspé J, Dal Lago L, Pepersack T, Donckier V, Liberale G. Evaluation of the Comprehensive Geriatric Assessment (CGA) tool as a predictor of postoperative complications following major oncological abdominal surgery in geriatric patients. PLoS One. 2022;17:e0264790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 13. | Normann M, Ekerstad N, Angenete E, Prytz M. Effect of comprehensive geriatric assessment for frail elderly patients operated for colorectal cancer-the colorectal cancer frailty study: study protocol for a randomized, controlled, multicentre trial. Trials. 2022;23:948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 14. | Pierantoni F, Basso U, Maruzzo M, Lamberti E, Bimbatti D, Tierno G, Bergo E, Brunello A, Zagonel V. Comprehensive geriatric assessment is an independent prognostic factor in older patients with metastatic renal cell cancer treated with first-line Sunitinib or Pazopanib: a single center experience. J Geriatr Oncol. 2021;12:290-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 15. | Mahmoud AM, Biello F, Maggiora PM, Bruna R, Burrafato G, Cappelli M, Varughese F, Martini V, Platini F, Deambrogi C, Patriarca A, Nicolosi M, Vachanaram AR, Pisani C, Ferrara E, Catania E, Azzolina D, Barone-Adesi F, Krengli M, Gaidano G, Gennari A. A randomized clinical study on the impact of Comprehensive Geriatric Assessment (CGA) based interventions on the quality of life of elderly, frail, onco-hematologic patients candidate to anticancer therapy: protocol of the ONCO-Aging study. BMC Geriatr. 2021;21:320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Liu WY, Jin J, Tang Y, Li N, Tang Y, Wang J, Cheng YJ, Yang L, Fang H, Lu NN, Qi SN, Chen B, Wang SL, Song YW, Liu YP, Li YX, Liu Z, Zhou HT, Liang JW, Pei W, Wang XS, Zhang HZ, Zhou ZX. Safety and efficacy of preoperative chemoradiotherapy in fit older patients with intermediate or locally advanced rectal cancer evaluated by comprehensive geriatric assessment: A planned interim analysis of a multicenter, phase II trial. J Geriatr Oncol. 2021;12:572-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Sentí S, Gené C, Troya J, Pacho C, Nuñez R, Parrales M, Jimenez I, Fernandez-Llamazares J, Julian JF, Parés D. Comprehensive geriatric assessment: Influence on clinical results after colorectal surgery in advanced age patients. Gastroenterol Hepatol. 2021;44:472-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 18. | Miraoui D, Ghomari-Bezzar S, Yekrou D. P-280 Impact of comprehensive geriatric assessment in elderly patients with colorectal cancer. Ann Oncol. 2023;34:S114. [DOI] [Full Text] |
| 19. | Chahal M, Thiessen B, Mariano C. Treatment of Older Adult Patients with Glioblastoma: Moving towards the Inclusion of a Comprehensive Geriatric Assessment for Guiding Management. Curr Oncol. 2022;29:360-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 20. | Åhlund K, Larsson LG, Ekerstad N, Normann M, Prytz M, Johnsson A. Experiences of participating in a preoperative comprehensive geriatric assessment and care intervention among frail older adults before colorectal cancer resection surgery. BMC Geriatr. 2025;25:310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 21. | Versteeg KS, Looijaard SMLM, Slee-Valentijn MS, Verheul HMW, Maier AB, Konings IRHM. Predicting outcome in older patients with cancer: Comprehensive geriatric assessment and clinical judgment. J Geriatr Oncol. 2021;12:49-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Yang YX, Tang Y, Li N, Tang Y, Cheng YJ, Yang L, Fang H, Lu NN, Qi SN, Chen B, Wang SL, Song YW, Liu YP, Li YX, Liu Z, Liang JW, Zhang HZ, Zhou HT, Wang J, Liu WY, Jin J. Comprehensive geriatric assessment guided radiotherapy in elderly patients with locally advanced rectal cancer-exploratory results on nonoperative cohort of a multicenter prospective study. Ther Adv Med Oncol. 2024;16:17588359241296386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 23. | Sourdet S, Brechemier D, Steinmeyer Z, Gerard S, Balardy L. Impact of the comprehensive geriatric assessment on treatment decision in geriatric oncology. BMC Cancer. 2020;20:384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 24. | Ritchie T, Sivarajan S, Penney N, Kumar B. Understanding and Applying the Comprehensive Geriatric Assessment as a Frailty Index in Patients Undergoing Esophagectomy. Ann Surg Oncol. 2024;31:3493-3494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 25. | Bonneau M, Steinmeyer Z, Morisseau M, Lozano S, Barbe P, Chauvet C, Brechemier D, Mourey L, Balardy L. Impact of comprehensive geriatric assessment on treatment decisions in older prostate cancer patients. BMC Cancer. 2025;25:642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 26. | Liuu E, Hu C, Valero S, Brunet T, Jamet A, Bureau ML, Pilotto A, Saulnier PJ, Paccalin M. Comprehensive geriatric assessment in older patients with cancer: an external validation of the multidimensional prognostic index in a French prospective cohort study. BMC Geriatr. 2020;20:295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 27. | Disalvo D, Moth E, Soo WK, Garcia MV, Blinman P, Steer C, Amgarth-Duff I, Power J, Phillips J, Agar M. The effect of comprehensive geriatric assessment on care received, treatment completion, toxicity, cancer-related and geriatric assessment outcomes, and quality of life for older adults receiving systemic anti-cancer treatment: A systematic review. J Geriatr Oncol. 2023;14:101585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 33] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 28. | Yamashita K, Yamasaki M, Makino T, Tanaka K, Saito T, Yamamoto K, Takahashi T, Kurokawa Y, Yasunobe Y, Akasaka H, Rakugi H, Nakajima K, Eguchi H, Doki Y. Preoperative Comprehensive Geriatric Assessment Predicts Postoperative Risk in Older Patients with Esophageal Cancer. Ann Surg Oncol. 2023;30:901-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 29. | Itami T, Yamamoto K, Kurokawa Y, Saito T, Takahashi T, Momose K, Yamashita K, Tanaka K, Makino T, Yasunobe Y, Akasaka H, Fujimoto T, Yamamoto K, Nakajima K, Eguchi H, Doki Y. Assessing the Risk of Postoperative Delirium Through Comprehensive Geriatric Assessment and Eastern Cooperative Oncology Group Performance Status of Elderly Patients With Gastric Cancer. Ann Surg Oncol. 2024;31:9039-9047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |