Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.117815
Revised: January 27, 2026
Accepted: February 26, 2026
Published online: April 15, 2026
Processing time: 112 Days and 23.7 Hours
Chronic atrophic gastritis is a common gastric disorder frequently accompanied by precancerous lesions, including intestinal metaplasia and dysplasia. Progre
To determine dynamic changes in HIF-1α/BNIP3 signaling and investigate mito
Eighty male Sprague-Dawley rats were randomly assigned to normal and model groups. A PLGC model was established using a comprehensive induction method. Eight rats per group were sacrificed at weeks 4, 10, 16, 22, and 28. Gastric histopathology was evaluated by hematoxylin and eosin staining. Immunofluorescence assessed HIF-1α and BNIP3 expression in gastric antrum tissue. Quantitative real-time polymerase chain reaction and western blotting were used to assess the expression of mitophagy-related and glycolysis-related genes and proteins.
As time progressed, gastric mucosa in the model group gradually developed inflammation, atrophy, intestinal metaplasia, and dysplasia. In gastric antrum tissue, HIF-1α protein and mRNA expression continuously increased. The expression of mitophagy-related markers, including BNIP3, Beclin1, and microtubule-associated protein 1 light chain 3, initially increased but subsequently declined, whereas sequestosome 1 expression showed an opposite trend. Regarding metabolic alterations, cytochrome C oxidase subunit 4 expression exhibited a mild decrease from week 16, while hexokinase 2 and lactate dehydrogenase A protein and mRNA expression significantly increased from week 22 onward.
HIF-1α/BNIP3 signaling dynamically regulates mitophagy and glycolysis during PLGC progression; appropriate HIF-1α is protective, whereas sustained overexpression induces autophagy dysfunction and metabolic imbalance.
Core Tip: In the hypoxic microenvironment of gastric precancerogenesis, the transcription factor hypoxia-inducible factor 1-alpha (HIF-1α) is a master regulator of both mitophagy and glycolysis. Our study reveals that sustained overexpression of HIF-1α during the progression of precancerous gastric lesions drives autophagic failure via the BCL2/adenovirus E1B 19 kDa interacting protein 3 pathway. This defective autophagy is tightly linked to a subsequent metabolic switch, characterized by the late-stage activation of glycolysis. This work identifies the dysregulation of the HIF-1α/BCL2/adenovirus E1B 19 kDa interacting protein 3 axis as a key event linking failed mitochondrial quality control to the pro-tumorigenic glycolytic reprogramming in the stomach.
- Citation: Zhang XF, Lv GH, An SJ, Yang XR, Guo WZ, Yu CY, Zhu CH. Dysregulation of the HIF-1α/BNIP3 axis links defective mitophagy to glycolytic reprogramming in a rat model of gastric precancerogenesis. World J Gastrointest Oncol 2026; 18(4): 117815
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/117815.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.117815
Gastric cancer is one of the most common malignancies globally, ranking fifth in both incidence and mortality[1]. The incidence of gastric cancer is positively correlated with age, typically beginning to rise after the age of 40 and peaking in individuals over 75[2]. While advancements in modern medical technology and the implementation of public health strategies have led to a decline in its incidence, the total number of diagnosed cases remains substantial, with an alarming trend towards affecting younger populations[3]. Conventional treatments for gastric cancer include surgery, ra
Gastric cancer progresses through several stages, and its histological evolution from normal gastric mucosa to carcinoma is classically described by the Correa cascade[5]. The progression usually starts with chronic non-atrophic gastritis, commonly triggered by factors like Helicobacter pylori infection, which may subsequently progress to chronic atrophic gastritis, marked by the loss of native gastric glands. Amidst persistent injury and repair, the gastric epithelium is replaced by intestinal-type epithelium, a process known as intestinal metaplasia. This can further develop into dysplasia, which features both cellular and architectural atypia. High-grade dysplasia is considered the penultimate step before cancer, carrying an extremely high risk of malignant transformation to invasive adenocarcinoma.
Precancerous lesions of gastric cancer (PLGC) are considered the critical stage just before the development of gastric cancer. According to the World Health Organization, PLGC is characterized by intestinal metaplasia and dysplasia within the context of chronic atrophic gastritis[6]. Epidemiological studies have reported that the annual progression rate to gastric cancer in patients with atrophic gastritis varies globally between 0.1% and 0.25%, though this number can exceed 1.5% in certain Asian regions[7]. The risk of malignant transformation associated with PLGC not only poses a severe threat to patients' physical health but also imposes a significant psychological burden. Therefore, an in-depth investigation into the mechanisms underlying PLGC formation is of great clinical significance for improving the prevention, control, early diagnosis, and therapeutic outcomes of gastric cancer.
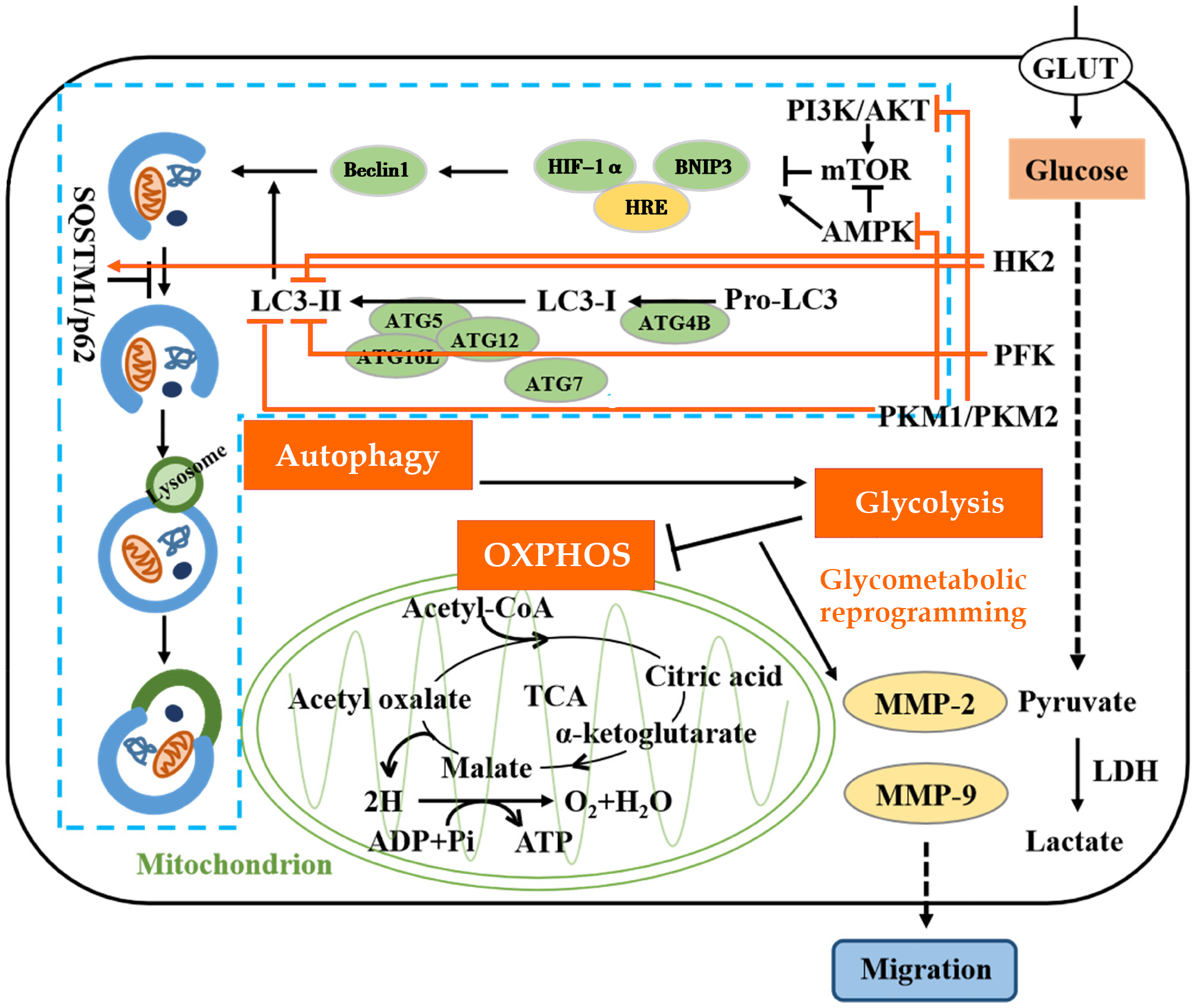
Persistent chronic inflammation is a core driving force throughout the Correa cascade[8]. It not only directly damages epithelial cells but, more critically, reshapes the gastric mucosal microenvironment. Inflammation leads to disordered local vasculature and tissue edema, resulting in an insufficient oxygen supply and the formation of a hypoxic microenvironment[9]. Under such hypoxic conditions, hypoxia-inducible factor 1-alpha (HIF-1α) is among the first transcription factors to be activated, essential for the induction of both mitophagy and glycolysis[10].
BCL2/adenovirus E1B 19 kDa interacting protein 3 (BNIP3) is a pro-apoptotic protein found on the outer mitochondrial membrane, playing a crucial role in mitophagy by facilitating the selective removal of damaged or excess mitochondria[11]. Mechanistically, BNIP3 competitively binds to the anti-apoptotic protein Bcl-2, thereby disrupting the Bcl-2/Beclin1 complex. This liberates Beclin1 to initiate autophagy. Under hypoxia, BNIP3 also interacts with microtubule-associated protein 1 light chain 3 (LC3) and the scaffold protein sequestosome 1 (P62/SQSTM1) to facilitate selective mitophagy[12]. The regulation of BNIP3 by HIF-1α appears to be a dual process: Under physiological or early stress conditions, HIF-1α upregulates BNIP3 to activate protective mitophagy and clear reactive oxygen species (ROS). However, under sustained and severe hypoxic stress, the overexpression of HIF-1α may paradoxically inhibit BNIP3 activation, leading to autophagic dysfunction[13]. Corroborating this, numerous studies have reported that BNIP3 expression is significantly downregulated in the majority of pancreatic, liver, and gastric cancer cases[14,15].
During tumor initiation and progression, glycolysis is frequently aberrantly enhanced in the form of the Warburg effect, which represents a hallmark metabolic feature of cancer and has been demonstrated to be present as early as the PLGC stage, while mitochondrial autophagy dysfunction is considered a critical initiating event for this abnormal glycolytic activation[16,17]. When the BNIP3-mediated mitochondrial clearance pathway is impaired, damaged mitochondria cannot be efficiently eliminated, leading to excessive accumulation of ROS. Elevated ROS levels directly impair the activity of cytochrome C oxidase subunit 4 (COX4), thereby causing severe disruption of oxidative phosphorylation[18]. To compensate for the resulting deficiency in adenosine triphosphate (ATP) production and to sustain cellular survival and proliferation, cancer cells are forced to undergo metabolic reprogramming and shift toward glycolysis, a less efficient yet more rapid energy-producing pathway. Glycolysis is initiated by hexokinase (HK), which can preferentially utilize mitochondria-derived ATP through its interaction with the voltage-dependent anion channel[19,20]. Notably, this adaptive metabolic shift is also directly regulated by HIF-1α. Hypoxia responsive element binding sites are widely distributed within the promoter regions of genes encoding glycolytic enzymes[21]. Consequently, under hypoxic conditions, prolonged activation of HIF-1α not only inhibits BNIP3-mediated mitophagy but also selectively upregulates glycolysis-associated genes, thereby establishing a detrimental regulatory loop that collectively drives malignant progression (Figure 1).
Although accumulating evidence indicates a close association between autophagy and glycolysis in gastric cancer, it remains unclear how the HIF-1α/BNIP3 signaling pathway precisely coordinates the interplay between these two metabolic processes during the dynamic transition of PLGC from adaptive inflammation to malignant proliferation, and whether disruption of this balance represents a critical driving event in the progression of precancerous lesions. Therefore, this study aimed to establish a rat PLGC model and systematically evaluate the dynamic alterations of key molecules within the HIF-1α/BNIP3 signaling pathway, with the objective of elucidating the mechanistic crosstalk between mitophagy and glycolytic metabolism and identifying potential therapeutic strategies to delay or suppress PLGC progression.
The 1-methyl-3-nitro-1-nitrosoguanidine (No. M105583) was purchased from Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). A Total RNA Extraction Kit (No. AK801) and a Reverse Transcription Kit (No. AK3002) were procured from Takara Biomedical Technology Co., Ltd. (Dalian, Liaoning Province, China). ChamQ™ SYBR® qPCR Master Mix (No. AK7803) was obtained from Vazyme Biotech Co., Ltd. (Nanjing, Jiangsu Province, China). The Minute™ Animal Cell/Tissue Total Protein Extraction Kit (No. SD-001) was from Invent Biotechnologies, Inc. (Beijing, China), and the BCA Protein Assay Kit (No. AR0146) was from Boster Biological Technology Co., Ltd. (Wuhan, Hubei Province, China).
The following primary antibodies were all purchased from Bioss Antibodies Inc. (Beijing, China): (1) HIF-1α rabbit monoclonal antibody (No. bsm-62534R); (2) BNIP3 rabbit monoclonal antibody (No. bsm-61440R); (3) Beclin1 rabbit monoclonal antibody (No. bsm-61085R); (4) LC3 II rabbit monoclonal antibody (No. bsm-60842R); (5) P62 rabbit monoclonal antibody (No. bsm-60709R); (6) COX4 rabbit monoclonal antibody (No. bsm-52750R); (7) HK2 rabbit monoclonal antibody (No. bs-3993R); (8) Lactate dehydrogenase A (LDHA) rabbit monoclonal antibody (No. bsm-62875R); and (9) GAPDH rabbit monoclonal antibody (No. bsm-52262R). The horseradish peroxidase-conjugated goat anti-rabbit IgG secondary antibody (No. bs-0295G-HRP) was also sourced from Bioss Antibodies Inc.
Quick Block™ Protein-Free Western Blocking Buffer (No. P0240) was purchased from Beyotime Biotechnology (Shanghai, China). The Hematoxylin and Eosin Staining Kit (No. G1005) was purchased from Servicebio Technology Co., Ltd. (Wuhan, Hubei Province, China).
Eighty male Sprague-Dawley rats (specific-pathogen-free grade), aged 3-4 weeks and weighing 100-130 g, were purchased from Liaoning Changsheng Biotechnology Co., Ltd. [Animal Production License No. SCXK (Liao) 2020-0001]. All animals were housed in the specific-pathogen-free animal facility at the Research Center of the Second Affiliated Hospital of Liaoning University of Traditional Chinese Medicine. The rats were maintained under a 12 hours/12 hours light/dark cycle, and all experimental procedures adhered to the principles of the 3Rs (replacement, reduction, and refinement). This animal study was reviewed and approved by the Ethics Committee of Second Affiliated Hospital of Liaoning University of Traditional Chinese Medicine (No. LZYY250203).
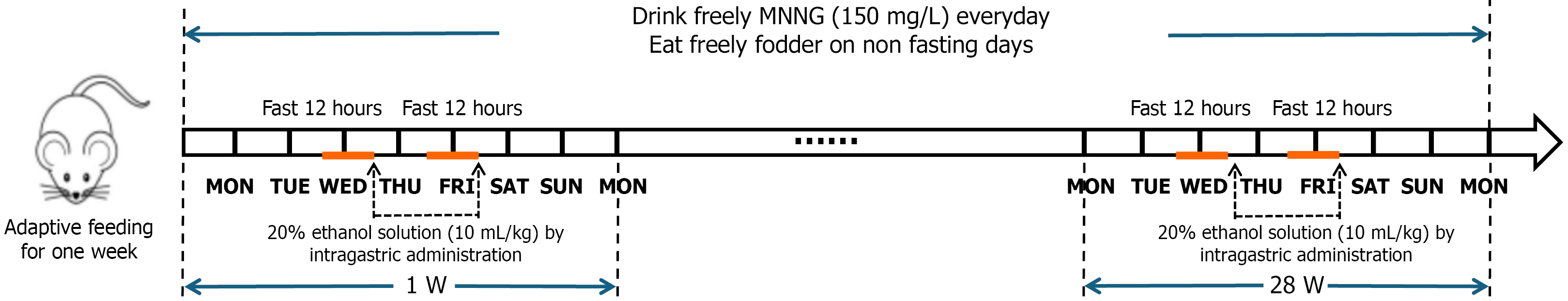
The PLGC model was established following the method described by Liu et al[22]. After a 7-day acclimatization period, the 80 male Sprague-Dawley rats were randomly divided into two groups (n = 40 per group): The normal group and the model group. Rats in the model group were given free access to a 150 mg/L solution of 1-methyl-3-nitro-1-nitrosoguanidine in their drinking water. Additionally, every Wednesday and Friday, after a 12-hour fast, they were administered 2 mL of a 20% ethanol solution via oral gavage. At the same time, rats in the control group received an equal volume of normal saline via oral gavage. This protocol was maintained for 28 weeks.
The day after the initiation of the modeling protocol was designated as day 1 of the experiment. At five time points – the end of weeks 4, 10, 16, 22, and 28 – eight rats were selected from each group for sample collection. Prior to sacrifice, the selected rats were fasted for 24 hours with free access to water. They were then anesthetized by an intraperitoneal injection of 2% sodium pentobarbital. The entire stomach was promptly excised, opened along the greater curvature, and its contents were thoroughly rinsed with normal saline. The gastric mucosa was first examined for gross pathological changes. Subsequently, a tissue sample of approximately 1 cm³ was harvested from the lesser curvature, extending from the gastric body to the antrum, and fixed in 4% paraformaldehyde for subsequent histopathological analysis and immunofluorescence staining. The leftover gastric antrum tissue was carefully excised, rapidly frozen, and preserved at -80 °C for later gene and protein expression analyses (Figure 2).
Gastric antrum tissues, previously fixed in 4% paraformaldehyde, were retrieved and processed through sequential steps of dehydration in a graded ethanol series, then cleared in xylene, and embedded in paraffin. The paraffin-embedded blocks were then sectioned, stained with hematoxylin and eosin, and mounted with coverslips. Histopathological alterations in the gastric mucosa, such as inflammation, atrophy, intestinal metaplasia, and dysplasia, were examined using a light microscope. The severity of dysplasia was evaluated according to the Padova International Classification[23].
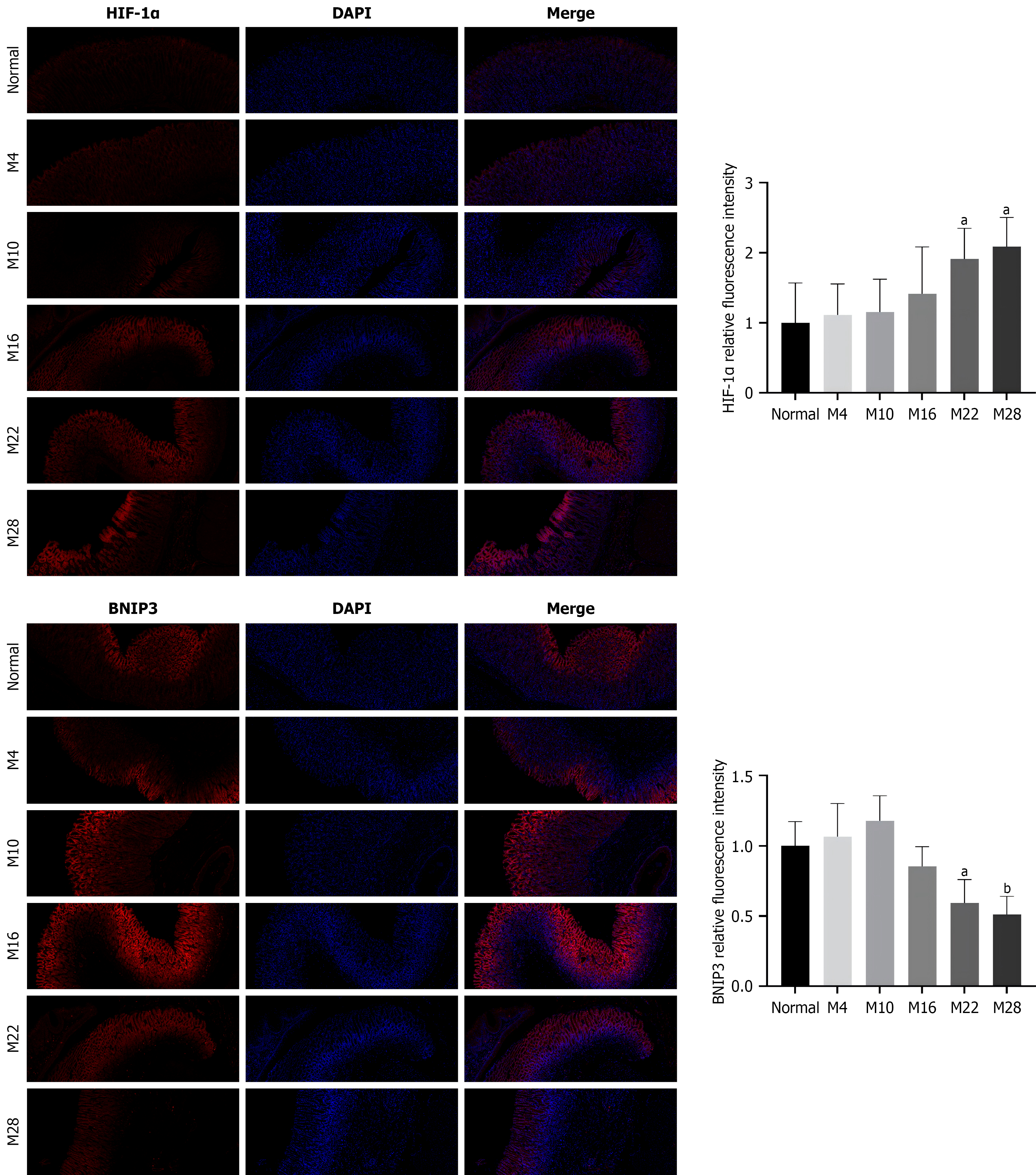
Paraffin-embedded sections of the gastric antrum tissue were deparaffinized and subjected to antigen retrieval. After being washed with phosphate buffered saline (PBS), the sections were blocked for 30 minutes at room temperature and then incubated overnight at 4 °C with primary antibodies against HIF-1α (1:200) and BNIP3 (1:200). The following day, the sections were washed with PBS and incubated with a fluorescent secondary antibody for 50 minutes, protected from light. After further washes with PBS, the nuclei were counterstained with DAPI for 10 minutes. Finally, the sections were mounted, and images were captured for analysis.
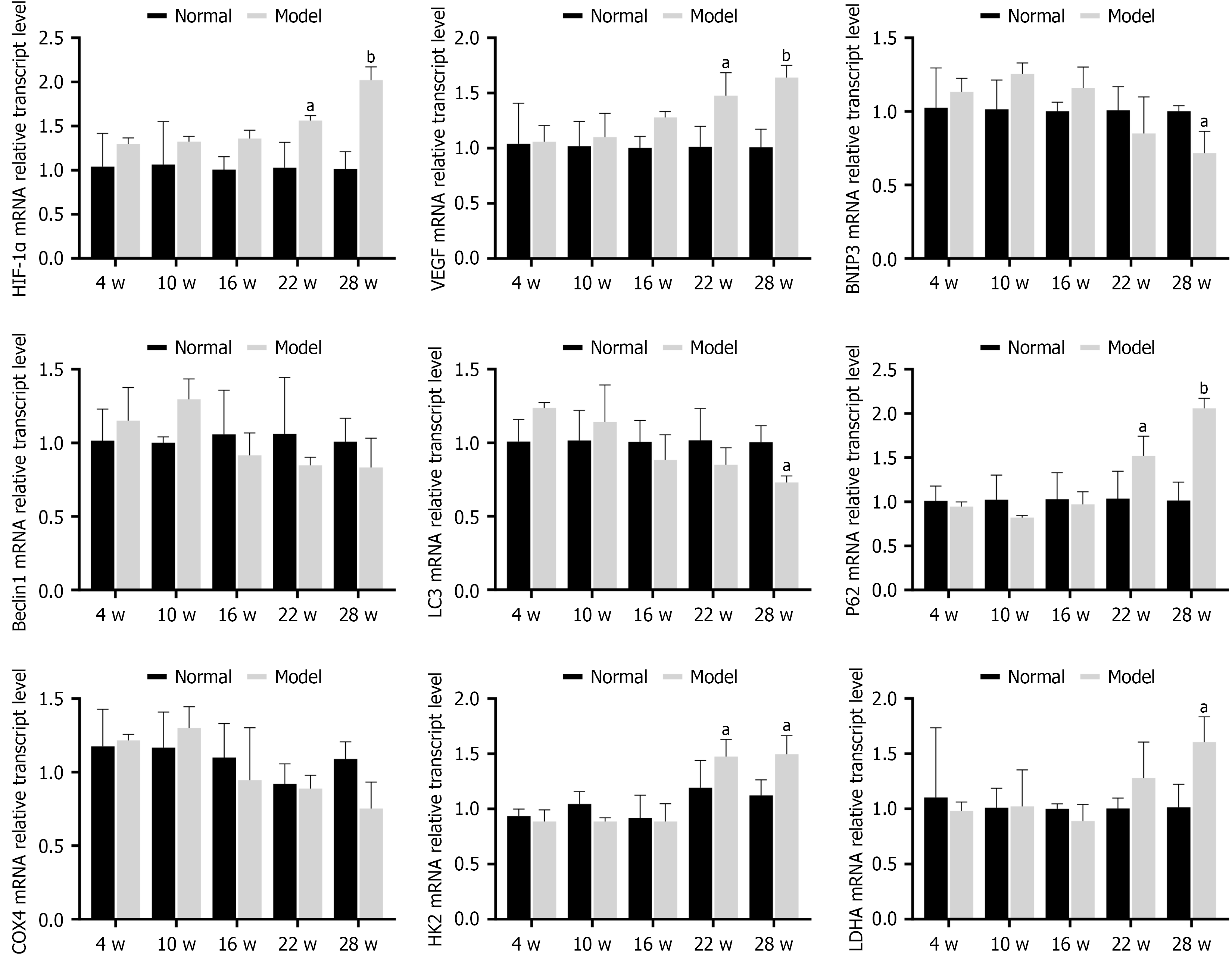
Total RNA was extracted from gastric antrum tissues using a suitable lysis buffer, and RNA concentration was measured. The extracted RNA was then reverse-transcribed into cDNA using a commercial kit according to the manufacturer’s instructions. The quantitative real-time polymerase chain reaction amplification was performed under the following thermal cycling conditions: (1) An initial denaturation at 95 °C for 3 minutes; (2) Followed by 40 cycles of denaturation at 95 °C for 5 seconds; and (3) Annealing/extension at 60 °C for 30 seconds. β-actin was used as the internal reference gene, and the relative mRNA expression levels were calculated. The primers for gene expression analysis were synthesized by Takara (Dalian, Liaoning Province, China), and the corresponding sequences are provided in Table 1.
| Gene name | Sequence | Size (bp) |
| HIF-1α | Forward: ACCGTGCCCCTACTATGTCG | 197 |
| Reverse: GCCTTGTATGGGAGCATTAACTT | ||
| VEGF | Forward: GCACTGGACCCTGGCTTTACT | 102 |
| Reverse: AACTTCACCACTTCATGGGCTTT | ||
| BNIP3 | Forward: CTTCAGCAATGGGAATGGGAG | 160 |
| Reverse: GGTATCTTGTGGTGTCTGGGAGC | ||
| Beclin1 | Forward: AGGAGTTGCCGTTGTACTGTTCT | 178 |
| Reverse: GTGTCTTCAATCTTGCCTTTCTCC | ||
| LC3 | Forward: TTGGTCAAGATCATCCGGCG | 175 |
| Reverse: AGCCGAAGGTTTCTTGGGAG | ||
| P62 | Forward: GCTATTACAGCCAGAGTCAAGGG | 105 |
| Reverse: TGGTCCCATTCCAGTCATCTT | ||
| COX4 | Forward: CATCTTATGTTGATCGGCGTG | 238 |
| Reverse: GCAGTGAAGCCGATGAAGAACA | ||
| HK2 | Forward: CCCATTTTGCCAAGCGTCTC | 137 |
| Reverse: TTGGTCAGCCAGACGGTAAG | ||
| LDHA | Forward: ATGAGCTTGCCCTTGTTGATGT | 214 |
| Reverse: AAGATGTTCACGTTTCGCTGG | ||
| β-actin | Forward: TGCTATGTTGCCCTAGACTTCG | 240 |
| Reverse: GTTGGCATAGAGGTCTTTACGG |
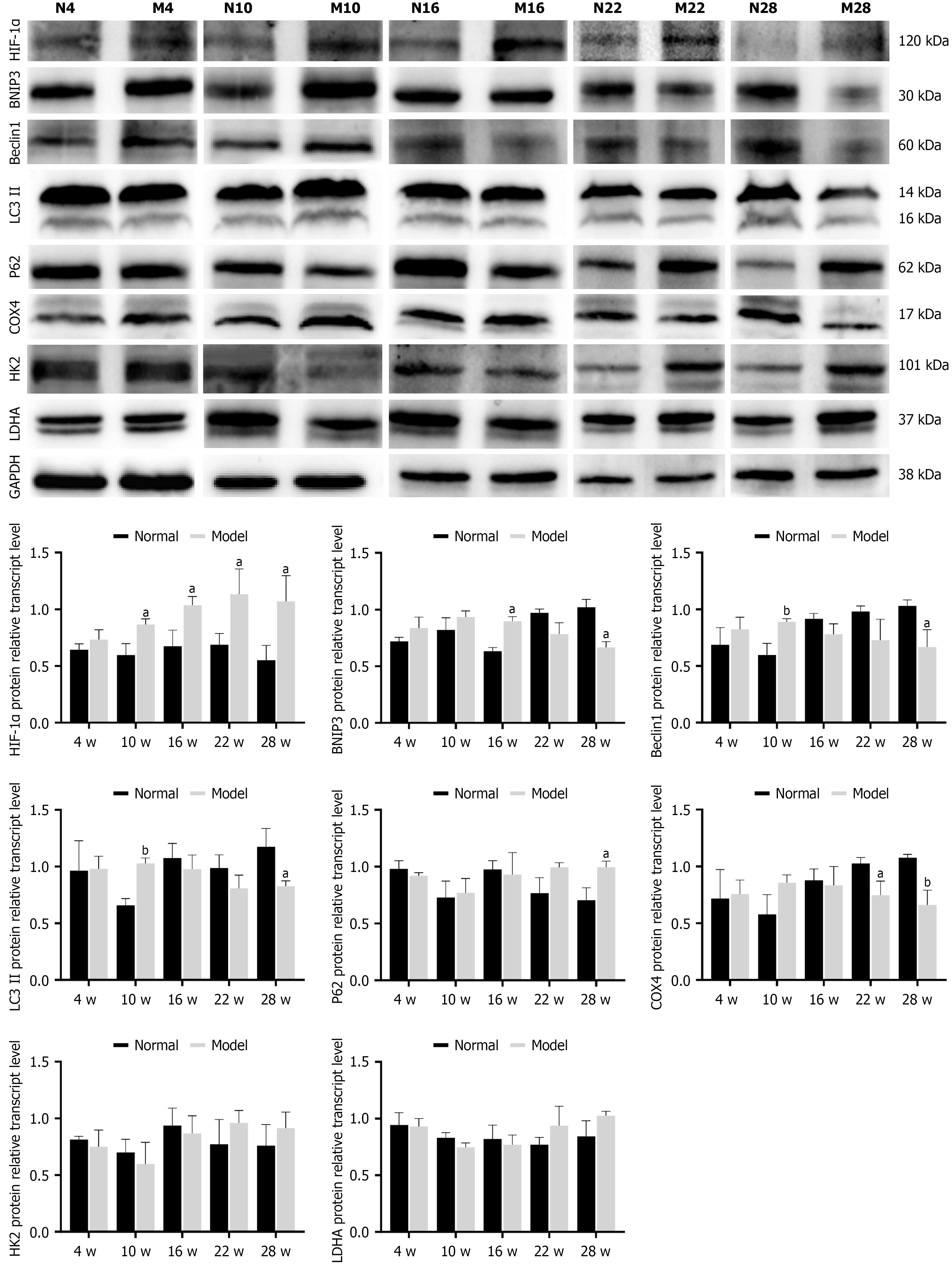
Total protein was extracted from rat gastric antrum tissues using a total protein extraction kit, and the protein concentration was determined using the BCA assay. Equal amounts of protein from each sample were denatured with loading buffer, separated by SDS-polyacrylamide gel electrophoresis, and subsequently transferred to polyvinylidene difluoride membranes using a wet transfer system. The membranes were then blocked with a protein-free Western blocking buffer for 15 minutes at room temperature.
Subsequently, the membranes were incubated overnight at 4 °C with the following primary antibodies: (1) GAPDH (1:10000); (2) HIF-1α (1:1000); (3) BNIP3 (1:1000); (4) Beclin1 (1:1000); (5) LC3 II (1:1000); (6) P62 (1:2500); (7) COX4 (1:1000); (8) HK2 (1:1000); and (9) LDHA (1:1000). On the following day, the membranes were rinsed with 1 × TBST three times, with each wash lasting 10 minutes. They were then incubated with a horseradish peroxidase-conjugated goat anti-rabbit IgG secondary antibody (1:10000) for 90 minutes at room temperature.
Following the final washing steps, protein bands were detected using an enhanced chemiluminescence substrate. The chemiluminescent signals were captured and imaged using a gel imaging system (chemiluminescence detector) with automatic exposure. Band intensities were quantified using ImageJ software, and the expression levels of target proteins were normalized to GAPDH as the internal loading control. The quantification of protein bands was performed using samples from three biologically independent animals per group. The representative blots shown are from one of three independent experiments with similar results.
All statistical analyses were performed using GraphPad Prism software (version 8.0). Quantitative data are expressed as the mean ± SD. Differences between two groups were analyzed using an independent-samples t-test. When more than two groups were compared, one-way analysis of variance was applied, with Tukey’s post hoc test used for pairwise comparisons. A P < 0.05 was considered statistically significant. In all analyses, ‘n’ refers to the number of independent biological replicates, which corresponds to the number of individual animals used for each experiment.
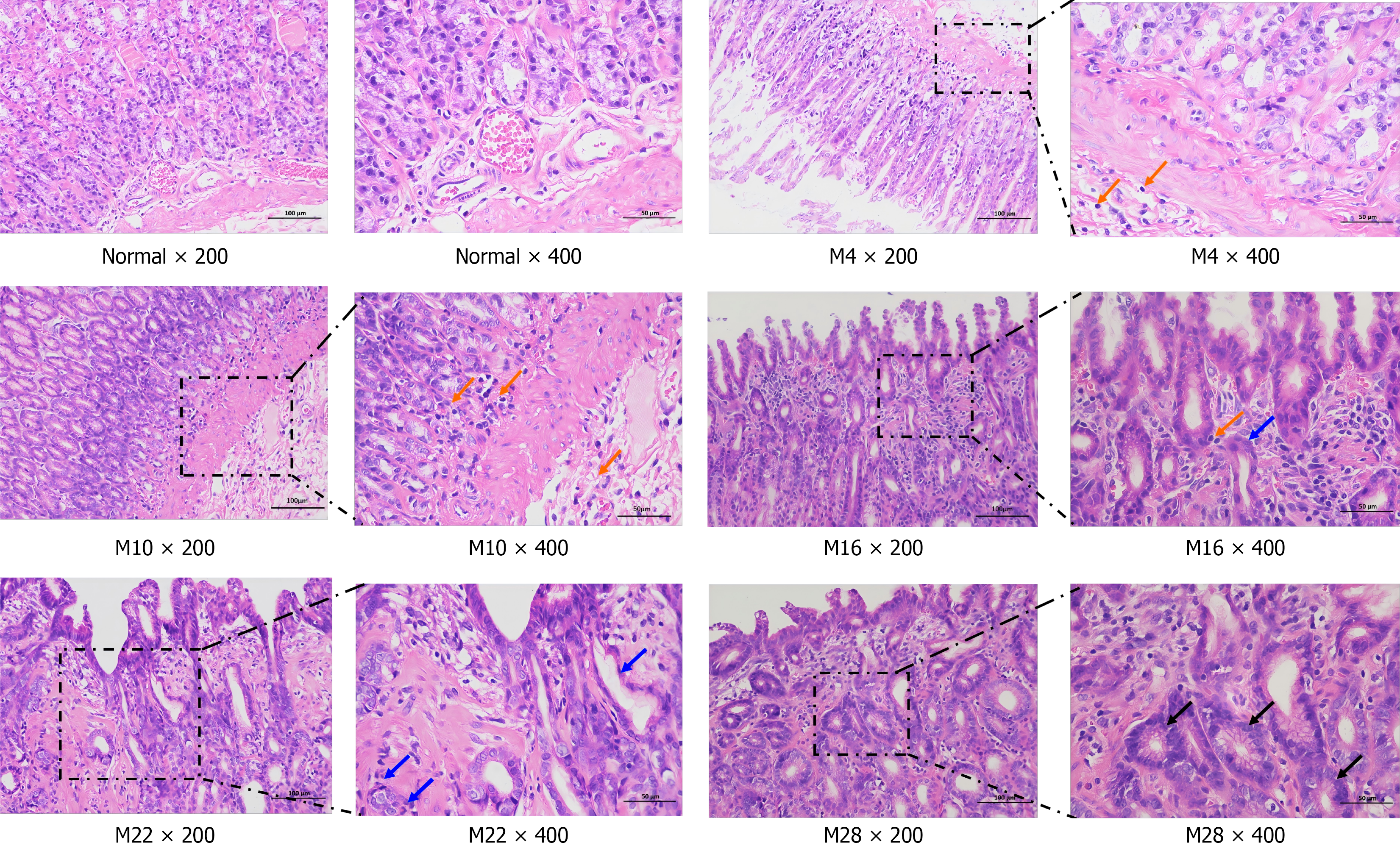
Histopathological analysis of hematoxylin and eosin-stained sections (Figure 3) revealed distinct differences between the groups. In the normal group, the gastric mucosa exhibited a well-organized architecture with intact gastric foveolae and clearly defined layers of lamina propria glands. The glands were regularly arranged, and the cells showed no atypia, featuring round or oval, basally located nuclei. No significant inflammatory cell infiltration was observed. In contrast, the model group displayed progressive pathological changes over the 28-week period. At week 4, an increased infiltration of chronic inflammatory cells, such as lymphocytes and plasma cells, was observed within the lamina propria (orange arrow). At week 10, the foveolar epithelial cells began to show atypia, characterized by variations in cell shape and size and a loss of basal polarity. The extent and density of the inflammatory infiltration were further increased. At week 16, the gastric mucosa began to exhibit atrophy, evidenced by a mild reduction in the number of proper gastric glands. Some glands became disordered and irregularly shaped, with a small amount of stromal connective tissue proliferation. Notably, the appearance of occasional goblet cells (blue arrow) indicated the onset of intestinal metaplasia. At week 22, gastric mucosal atrophy became more pronounced, with severe glandular disarray and loss of polarity. A few glands showed cystic dilation, and the nuclei became rounded, consistent with low-grade dysplasia. At week 28, the gastric mucosal tissue displayed marked inflammatory cell infiltration. The glandular disorganization was severe, and the cells exhibited features of high-grade dysplasia, including nuclear hyperchromasia, enlarged nuclei, pronounced atypia, a complete loss of polarity, and the presence of mitotic figures (black arrow) (Figure 3).
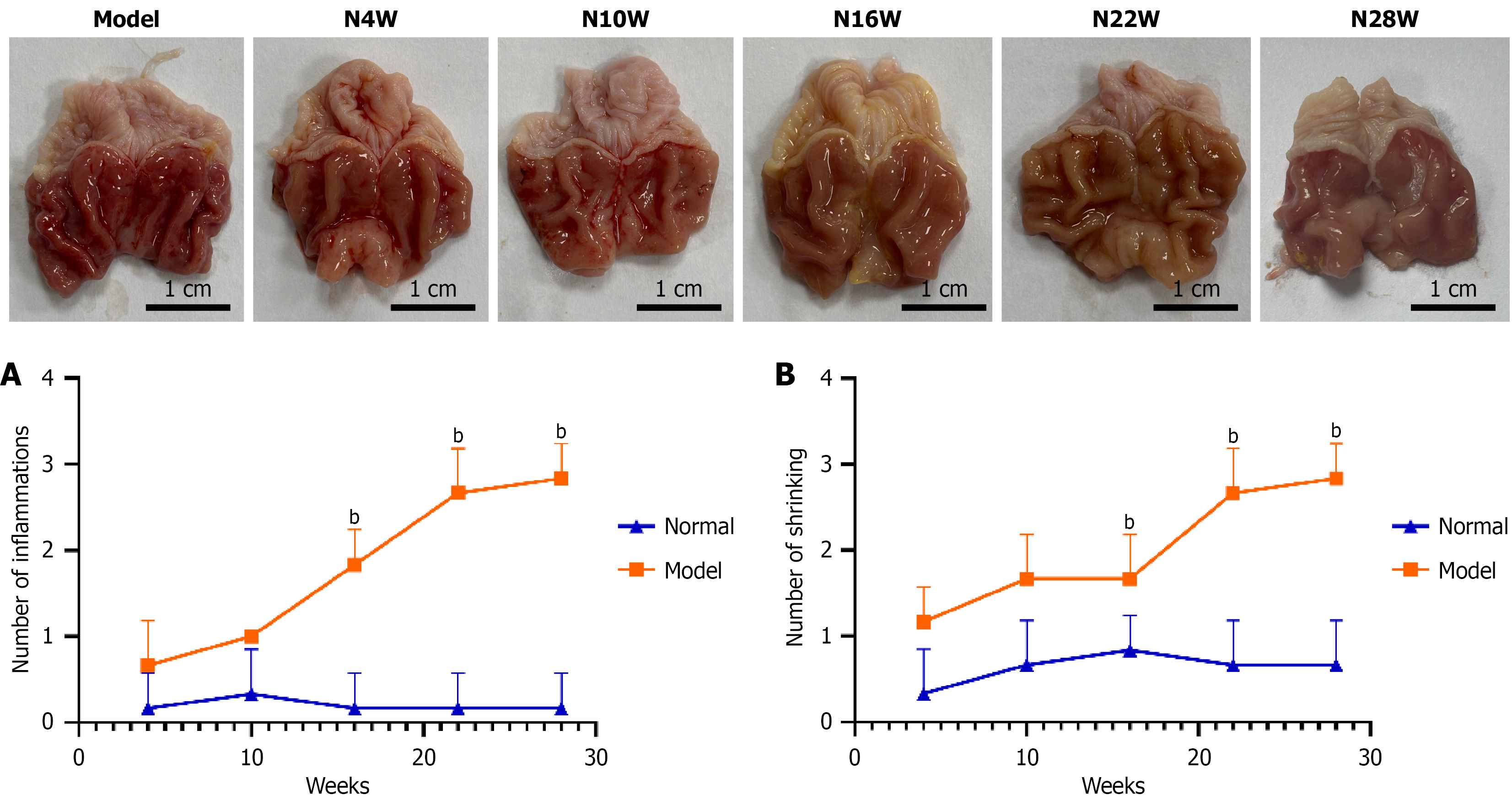
A semi-quantitative analysis of the pathological scores further detailed these observations. From weeks 4 to 10, the gastric mucosa of rats in the model group exhibited mild to moderate inflammation. Although the inflammation scores were slightly higher than those of the normal group, the difference was not statistically significant (P > 0.05). However, from weeks 16 to weeks 28, nearly all gastric mucosal samples from the model group displayed mild to moderate inflammation, with some cases progressing to severe inflammation. During this later period, the difference in inflammation scores between the model and normal groups became highly significant (P < 0.01; Figure 4A). Mild atrophy began to appear in the gastric mucosa between weeks 4 and weeks 10, but this was not significantly different from the normal group (P > 0.05). From week 16 onwards, the pathological changes advanced significantly, with nearly all specimens from the model group showing mild to moderate atrophy and the emergence of dysplasia. These changes were statistically significant when compared to the normal group (P < 0.01; Figure 4B).
Compared to the normal group, the expression of HIF-1α protein in the gastric antrum of rats in the model group showed a progressive increase throughout the experimental period. This increase became statistically significant during the later stages of the experiment, specifically at weeks 22 and 28 (P < 0.05). In contrast, the expression of BNIP3 protein in the model group initially exhibited an upward trend during the early to mid-stages of the experiment; however, this change was not statistically significant (P > 0.05). Subsequently, a marked decrease in BNIP3 expression was observed in the later stages, at weeks 22 and 28. This downregulation was statistically significant (P < 0.05 and P < 0.01, respectively; Figure 5).
In the gastric antrum of rats in the model group, the mRNA expression of both HIF-1α and its target gene vascular endothelial growth factor exhibited a progressive upward trend throughout the modeling period. This increase became statistically significant at week 22 (P < 0.05) and highly significant at week 28 (P < 0.01). The mRNA expression of the autophagy-related genes BNIP3, Beclin1, and LC3 initially increased in the early experimental stages compared to the normal group, but subsequently began to decrease during the mid-to-late stages. By week 28, the downregulation of BNIP3 and LC3 mRNA was statistically significant (P < 0.05) when compared to the normal group. Conversely, P62 mRNA expression showed a downward trend in the early-to-mid stages but was significantly upregulated in the later stages at weeks 22 and 28 (P < 0.05 and P < 0.01, respectively).
Regarding glycolysis-related genes, the mRNA expression of COX4 in the model group showed a slight upward trend in the early stages, followed by a decrease in the mid-to-late stages; however, these changes were not statistically significant (P > 0.05). Furthermore, the mRNA expression of the glycolytic enzymes HK2 and LDHA was markedly elevated in the later stages of the experiment (weeks 22 and 28) compared to the normal group, and these differences were statistically significant (P < 0.05; Figure 6).
The relative protein expression of HIF-1α in the gastric antrum of rats in the model group was markedly elevated at all observed time points compared to the normal group, with this increase becoming statistically significant from week 10 onwards (P < 0.05). For the autophagy-related proteins, the relative expression of BNIP3, Beclin1, and LC3II showed an upward trend during the early-to-mid stages of the experiment, and these increases were statistically significant (P < 0.05, P < 0.01). However, starting from week 22, the expression of all three proteins exhibited a significant downward trend (P < 0.05).
Regarding the glycolysis-related proteins, the relative expression of COX4 protein showed a slight upward trend in the early stages, which further increased in the mid-to-late stages, becoming statistically significant at weeks 22 and 28 (P < 0.05, P < 0.01, respectively). The relative protein expression of HK2 and LDHA trended lower than the normal group during the early-to-mid stages. However, their expression began to increase from week 22 onwards, although this upward trend was not statistically significant (P > 0.05; Figure 7).
PLGC is mainly characterized by chronic atrophic gastritis accompanied by varying degrees of intestinal metaplasia and dysplasia, representing a long-term, multistage, and hypoxia-associated pathological process preceding gastric cancer that is driven by multiple factors, including Helicobacter pylori infection, inflammation, and genetic alterations[24-27]. During PLGC progression, gastric mucosal cells must activate a series of quality control mechanisms to cope with substantial cellular stress, among which mitophagy plays a particularly critical role[28]. Under physiological conditions, autophagy is maintained at a basal level to regulate the balance between proliferation and apoptosis in gastric mucosal cells, thereby preserving epithelial homeostasis[29]. However, under persistent inflammatory stimulation, autophagy can be induced to alleviate inflammation and facilitate the clearance of harmful metabolic byproducts. Notably, studies using an autophagy-deficient model harboring Kirsten rat sarcoma viral oncogene homolog-driven Atg4B mutations have shown that acute autophagy inhibition can markedly induce tumor regression, suggesting that Kirsten rat sarcoma viral oncogene homolog mutations may represent a window of vulnerability for autophagic dysregulation[30]. Therefore, understanding the evolution of PLGC from the perspective of cellular autophagy is of critical importance. Although current evidence indicates dysregulated autophagy activation or suppression during the progression from chronic gastritis to gastric cancer, the precise patterns and underlying mechanisms of autophagy during this process remain unclear. In the present study, we propose that autophagy may be initially activated and subsequently suppressed during this pathological transition.
The initiation and execution of cellular autophagy constitute a tightly regulated multistep process. Within the hypoxic microenvironment of the gastric mucosa, the transcription factor HIF-1α is first activated and subsequently triggers autophagy. Under normoxic conditions, HIF-1α is hydroxylated by prolyl hydroxylases and then recognized by the von Hippel-Lindau E3 ubiquitin ligase complex, leading to its ubiquitination and proteasomal degradation; however, hypoxia, oxidative stress, or inflammatory stimuli can impair this degradation process. Consequently, stabilized and highly active HIF-1α translocates into the nucleus, where it heterodimerizes with HIF-1β to form the HIF-1α/HIF-1β complex, which participates in the regulation of multiple biological processes, including glucose metabolism, angiogenesis, autophagy, and apoptosis[31,32]. BNIP3, a key autophagy receptor localized on the outer mitochondrial membrane, is directly regulated by HIF-1α and competitively binds to the anti-apoptotic protein Bcl-2 through its BH3 domain, thereby disrupting the inhibitory Bcl-2–Beclin1 complex[33]. The released Beclin1 subsequently activates the downstream class III phosphatidylinositol 3-kinase complex, initiating the autophagic process. Formation of the autophagosomal membrane represents a central event in autophagy, with lipidation of LC3 I serving as a critical hallmark[34]. Upon autophagic signaling, LC3 I is converted into the membrane-bound form LC3 II, which is recruited and incor
In this study, we found that with the progressive increase in HIF-1α gene and protein expression, the gene and protein levels of the autophagy-related molecules BNIP3, Beclin1, and LC3 II exhibited an upward trend at weeks 4, 10, and 16, but declined markedly at weeks 22 and 28, whereas both gene and protein expression of P62 were significantly elevated during the late experimental stages. Integrating these autophagy-related alterations, we reasonably speculate that during the early phase of chronic inflammation in the gastric mucosa, HIF-1α promotes BNIP3 expression under hypoxic conditions and preserves mitochondrial function by limiting excessive ROS generation. In addition, early activation of the HIF-1α signaling pathway may facilitate an adaptive autophagic response to counteract inflammation and oxidative stress. However, sustained activation of autophagy subsequently disrupts the physiological balance between cellular proliferation and apoptosis, leading to autophagic attack on gastric epithelial cells while simultaneously suppressing caspase-mediated apoptotic pathways, thereby inhibiting programmed cell death. This imbalance between autophagy and apoptosis results in selective cell proliferation, which may constitute an initial event driving malignant trans
During PLGC progression, excessive accumulation of ROS induced by autophagic dysfunction represents one of the critical drivers of the metabolic shift from oxidative phosphorylation to glycolysis. Activation of glycolysis enables abnormally proliferating cells to enhance the uptake of nutrients such as glucose, thereby further satisfying their metabolic demands for growth and proliferation. Previous studies have shown that during the transdifferentiation of chief cells into spasmolytic polypeptide-expressing metaplasia cells, the central energy-sensing regulator mechanistic target of rapamycin complex 1 initially exhibits reduced activity and is subsequently reactivated[37]. Characterizing the dynamic alterations of glycolysis is therefore essential for understanding the metabolic transition point at which gastric mucosal cells shift from adaptive responses to malignant proliferation during the progression from chronic gastritis to gastric cancer. In the present study, we propose that glycolytic activity displays a sequential dynamic pattern characterized by initial suppression followed by subsequent activation during this pathological evolution.
Metabolic reprogramming of glucose metabolism is closely associated with overexpression of HIF-1α[38]. As a key enzyme of the respiratory chain complex, COX4 accounts for approximately 90% of mitochondrial oxygen consumption. Under hypoxic conditions, activation of HIF-1α promotes the metabolic shift from oxidative phosphorylation to glycolysis by suppressing COX4 expression, thereby facilitating cellular adaptation to the hypoxic microenvironment[39]. In addition, HIF-1α systematically enhances glycolytic flux by transcriptionally upregulating a series of key genes, forming a coordinated regulatory network that spans multiple core steps from substrate uptake to end-product generation. At the initial stage, HIF-1α induces the expression of glucose transporters GLUT1 and GLUT4, thereby increasing glucose uptake. Following cytoplasmic entry, HK, particularly the HK2 isoform, catalyzes the first rate-limiting step by converting glucose into glucose-6-phosphate. Subsequently, in the second major regulatory step, phosphofructokinase-1 converts fructose-6-phosphate into fructose-1,6-bisphosphate, and its activity is tightly regulated by the potent allosteric activator fructose-2,6-bisphosphate, which is synthesized by the bifunctional enzyme PFKFB3. At the terminal stage of glycolysis, pyruvate kinase, especially the pyruvate kinase muscle isozyme M2 isoform, catalyzes the conversion of phosphoenolpyruvate to pyruvate. Finally, LDHA reduces pyruvate to lactate, which is subsequently exported out of the cell via monocarboxylate transporter 4[19]. These glycolysis-related genes and proteins are consistently upregulated in gastric cancer[20]. In the present study, we focused on the dynamic changes in the expression levels of COX4, HK2, and LDHA.
Rapid proliferation of dysplastic cells through mitosis requires substantial amounts of nucleotides, amino acids, lipids, and other biosynthetic substrates, for which glycolysis is essential. Unlike oxidative phosphorylation, glycolytic metabolism generates ATP by directly reducing pyruvate to lactate, thereby ensuring rapid glucose utilization to compensate for the high energy demands of proliferating cells[40,41]. In the present study, we observed that protein levels of the key glycolytic enzymes HK2 and LDHA in PLGC rats exhibited a declining trend at weeks 4, 10, and 16, followed by a marked increase at weeks 22 and 28. This pattern may be attributable to intense inflammatory responses during the early transition from chronic gastritis to gastric cancer, which induce DNA damage and oxidative stress; to cope with such injury, cells may downregulate glycolysis to reduce metabolic burden, resulting in early glycolytic suppression. Previous studies have suggested that parietal cell atrophy and malignant proliferative changes in chief cells may occur concomitantly, and as gastric parietal cells undergo atrophy, chief cells may enter an initial stage of malignant proliferation, leading to a transient reduction in energy demand[42]. However, with progressive inhibition of autophagy, cells subsequently enter a phase of malignant proliferation characterized by excessive ROS accumulation, which impairs oxidative phosphorylation and ultimately triggers activation of the glycolytic pathway.
We further observed that glycolytic activation during the late stage of PLGC was highly temporally coincident with autophagy inhibition, strongly suggesting that suppression of autophagy may represent a critical upstream event triggering activation of glycolytic metabolism. We propose that when autophagic function is impaired, damaged mitochondria accumulate, leading to functional disruption of the mitochondrial respiratory chain, particularly through impairment of the key enzyme COX4, thereby resulting in oxidative phosphorylation dysfunction[43]. To compensate for the ensuing ATP deficiency and to meet the substantial energy demands required for rapid malignant cell proliferation, cells are forced to undergo glucose metabolic reprogramming and shift toward glycolysis as a rapid energy-producing pathway. Overexpressed HIF-1α exacerbates autophagic dysfunction by suppressing BNIP3 activity, while simultaneously acting as the central transcriptional activator of glycolysis. Through its hypoxia responsive element-binding domain, HIF-1α robustly drives the transcription of a series of glycolytic enzymes, including HK2 and LDHA. Consequently, during the late stage of PLGC, a vicious cycle characterized by HIF-1α-driven autophagy inhibition and glycolytic activation is established, collectively propelling gastric mucosal lesions toward irreversible malignant transformation.
This study has several limitations. First, although a strong association between autophagy and glycolysis was identified, the causal relationship between these processes was not directly validated through in vitro cell models or in vivo interventional experiments. Future studies should therefore employ functional approaches, such as genetic knockout[44] or pharmacological modulation[45], to determine whether inhibition of HIF-1α or activation of autophagy can effectively reverse glycolytic reprogramming and delay PLGC progression. Second, the regulation of this complex process may involve the coordinated participation of additional signaling pathways, such as the phosphatidylinositol 3-kinase/protein kinase B/mechanistic target of rapamycin pathway, and elucidating the crosstalk among these pathways will represent an important direction for future investigation.
Our study delineates a critical molecular switch during the progression of precancerous gastric lesions in rats, orchestrated by the HIF-1α/BNIP3 signaling pathway. We demonstrate that sustained HIF-1α overexpression drives a transition from an early, protective state characterized by activated mitophagy, to a late-stage, pro-tumorigenic state defined by two concurrent events: Autophagic dysfunction and a robust activation of glycolytic metabolism. This dynamic interplay, where autophagic failure appears to precede and potentially trigger glycolytic reprogramming, identifies a key metabolic vulnerability and highlights the HIF-1α axis as a promising therapeutic target to intercept the malignant transformation of gastric precancerous lesions.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14765] [Article Influence: 7382.5] [Reference Citation Analysis (21)] |
| 2. | Li Y, Ren N, Zhang B, Yang C, Li A, Li X, Lei Z, Fei L, Fan S, Zhang J. Gastric cancer incidence trends in China and Japan from 1990 to 2019: Disentangling age-period-cohort patterns. Cancer. 2023;129:98-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 3. | Wong MCS, Huang J, Chan PSF, Choi P, Lao XQ, Chan SM, Teoh A, Liang P. Global Incidence and Mortality of Gastric Cancer, 1980-2018. JAMA Netw Open. 2021;4:e2118457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 328] [Cited by in RCA: 289] [Article Influence: 57.8] [Reference Citation Analysis (6)] |
| 4. | Ajani JA, D'Amico TA, Bentrem DJ, Chao J, Cooke D, Corvera C, Das P, Enzinger PC, Enzler T, Fanta P, Farjah F, Gerdes H, Gibson MK, Hochwald S, Hofstetter WL, Ilson DH, Keswani RN, Kim S, Kleinberg LR, Klempner SJ, Lacy J, Ly QP, Matkowskyj KA, McNamara M, Mulcahy MF, Outlaw D, Park H, Perry KA, Pimiento J, Poultsides GA, Reznik S, Roses RE, Strong VE, Su S, Wang HL, Wiesner G, Willett CG, Yakoub D, Yoon H, McMillian N, Pluchino LA. Gastric Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022;20:167-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1229] [Article Influence: 307.3] [Reference Citation Analysis (5)] |
| 5. | Correa P. Human gastric carcinogenesis: a multistep and multifactorial process--First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992;52:6735-6740. [PubMed] |
| 6. | Pimentel-Nunes P, Libânio D, Marcos-Pinto R, Areia M, Leja M, Esposito G, Garrido M, Kikuste I, Megraud F, Matysiak-Budnik T, Annibale B, Dumonceau JM, Barros R, Fléjou JF, Carneiro F, van Hooft JE, Kuipers EJ, Dinis-Ribeiro M. Management of epithelial precancerous conditions and lesions in the stomach (MAPS II): European Society of Gastrointestinal Endoscopy (ESGE), European Helicobacter and Microbiota Study Group (EHMSG), European Society of Pathology (ESP), and Sociedade Portuguesa de Endoscopia Digestiva (SPED) guideline update 2019. Endoscopy. 2019;51:365-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 813] [Cited by in RCA: 744] [Article Influence: 106.3] [Reference Citation Analysis (2)] |
| 7. | Akbari M, Tabrizi R, Kardeh S, Lankarani KB. Gastric cancer in patients with gastric atrophy and intestinal metaplasia: A systematic review and meta-analysis. PLoS One. 2019;14:e0219865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 8. | Zhou J, Li J, Chen J, Lan X, Ai Y, Liu P, Peng J, Pan X, Zhang Y, Zhang H, Li X, Wen Y, Jiang W, Zeng J. Decoding inflammatory mediators in the Correa's cascade: From chronic gastritis to carcinogenesis and targeted therapies. Int Immunopharmacol. 2025;162:115191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 9. | Almanzar G, Alarcon JC, Garzon R, Navarro AM, Ondo-Méndez A, Prelog M. Hypoxia and activation of hypoxia inducible factor alpha as influencers of inflammatory helper T cells in autoimmune disease - a link between cancer and autoimmunity. Front Immunol. 2025;16:1633845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Dong F, Yin H, Zheng Z. Hypoxia-Inducible Factor-1α Regulates BNIP3-Dependent Mitophagy and Mediates Metabolic Reprogramming Through Histone Lysine Lactylation Modification to Affect Glioma Proliferation and Invasion. J Biochem Mol Toxicol. 2025;39:e70069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 11. | Field JT, Gordon JW. BNIP3 and Nix: Atypical regulators of cell fate. Biochim Biophys Acta Mol Cell Res. 2022;1869:119325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 12. | Pan Y, Wu G, Chen M, Lu X, Shen M, Li H, Liu H. Lactate Promotes Hypoxic Granulosa Cells' Autophagy by Activating the HIF-1α/BNIP3/Beclin-1 Signaling Axis. Genes (Basel). 2024;16:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Gao A, Jiang J, Xie F, Chen L. Bnip3 in mitophagy: Novel insights and potential therapeutic target for diseases of secondary mitochondrial dysfunction. Clin Chim Acta. 2020;506:72-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 130] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 14. | Berardi DE, Bock-Hughes A, Terry AR, Drake LE, Bozek G, Macleod KF. Lipid droplet turnover at the lysosome inhibits growth of hepatocellular carcinoma in a BNIP3-dependent manner. Sci Adv. 2022;8:eabo2510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 15. | Chen C, Guan S, Zhuang Y, Xie M, Huang Q, Li X, Yang C, Jian J. miRNA-214-3p targets BNIP3 to affect autophagy and thus drive gastric cancer progression. J Toxicol Sci. 2025;50:235-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Fernández-Coto DL, Gil J, Hernández A, Herrera-Goepfert R, Castro-Romero I, Hernández-Márquez E, Arenas-Linares AS, Calderon-Sosa VT, Sanchez-Aleman MÁ, Mendez-Tenorio A, Encarnación-Guevara S, Ayala G. Quantitative proteomics reveals proteins involved in the progression from non-cancerous lesions to gastric cancer. J Proteomics. 2018;186:15-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 17. | Vaupel P, Multhoff G. Revisiting the Warburg effect: historical dogma versus current understanding. J Physiol. 2021;599:1745-1757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 713] [Cited by in RCA: 611] [Article Influence: 122.2] [Reference Citation Analysis (0)] |
| 18. | Liang S, Dong S, Liu W, Wang M, Tian S, Ai Y, Wang H. Accumulated ROS Activates HIF-1α-Induced Glycolysis and Exerts a Protective Effect on Sensory Hair Cells Against Noise-Induced Damage. Front Mol Biosci. 2021;8:806650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 19. | Shao M, Zhang J, Zhang J, Shi H, Zhang Y, Ji R, Mao F, Qian H, Xu W, Zhang X. SALL4 promotes gastric cancer progression via hexokinase II mediated glycolysis. Cancer Cell Int. 2020;20:188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 20. | Feng Y, Xiong Y, Qiao T, Li X, Jia L, Han Y. Lactate dehydrogenase A: A key player in carcinogenesis and potential target in cancer therapy. Cancer Med. 2018;7:6124-6136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 565] [Cited by in RCA: 511] [Article Influence: 63.9] [Reference Citation Analysis (1)] |
| 21. | DeMichele E, Buret AG, Taylor CT. Hypoxia-inducible factor-driven glycolytic adaptations in host-microbe interactions. Pflugers Arch. 2024;476:1353-1368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Liu S, Ji H, Zhang T, Huang J, Yin X, Zhang J, Wang P, Wang F, Tang X. Modified Zuojin pill alleviates gastric precancerous lesions by inhibiting glycolysis through the HIF-1α pathway. Phytomedicine. 2025;136:156255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 23. | Robinson MJ. Gastric dysplasia: the Pavoda International Classification. Am J Surg Pathol. 2001;25:694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 24. | Gao MJ, Li SB, Zhu XJ, Zhang LF, Chen M, Shi YQ. Analysis of Risk Factors for Gastric Cancer and Precancerous Lesions: A Case-Control Study. J Dig Dis. 2024;25:674-684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 25. | Zhang S, Shen Y, Liu H, Zhu D, Fang J, Pan H, Liu W. Inflammatory microenvironment in gastric premalignant lesions: implication and application. Front Immunol. 2023;14:1297101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 26. | Lopes C, Almeida TC, Pimentel-Nunes P, Dinis-Ribeiro M, Pereira C. Linking dysbiosis to precancerous stomach through inflammation: Deeper than and beyond imaging. Front Immunol. 2023;14:1134785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 27. | Li X, Li C, Zhang W, Wang Y, Qian P, Huang H. Inflammation and aging: signaling pathways and intervention therapies. Signal Transduct Target Ther. 2023;8:239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1128] [Cited by in RCA: 931] [Article Influence: 310.3] [Reference Citation Analysis (1)] |
| 28. | Yamamoto K, Iwadate D, Naito E, Tateishi K, Fujishiro M. Autophagy as a critical driver of metabolic adaptation, therapeutic resistance, and immune evasion of cancer. Curr Opin Biotechnol. 2023;84:103012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Lu SY, Guo S, Chai SB, Yang JQ, Yue Y, Li H, Sun PM, Zhang T, Sun HW, Zhou JL, Yang JW, Yang HM, Li ZP, Cui Y. Autophagy in Gastric Mucosa: The Dual Role and Potential Therapeutic Target. Biomed Res Int. 2021;2021:2648065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Yang A, Herter-Sprie G, Zhang H, Lin EY, Biancur D, Wang X, Deng J, Hai J, Yang S, Wong KK, Kimmelman AC. Autophagy Sustains Pancreatic Cancer Growth through Both Cell-Autonomous and Nonautonomous Mechanisms. Cancer Discov. 2018;8:276-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 279] [Article Influence: 34.9] [Reference Citation Analysis (1)] |
| 31. | Zhang J, Yao M, Xia S, Zeng F, Liu Q. Systematic and comprehensive insights into HIF-1 stabilization under normoxic conditions: implications for cellular adaptation and therapeutic strategies in cancer. Cell Mol Biol Lett. 2025;30:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 76] [Article Influence: 76.0] [Reference Citation Analysis (0)] |
| 32. | Cui Q, Sun Q, Iniguez AB, Li X, Du M, Zhu MJ. Purple Potato Extract Suppresses Hypoxia-Induced Metabolic Reprogramming and Inhibits HIF-1α Signaling in Caco-2 Cells. Nutrients. 2025;17:2079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 33. | Huang L, Wang L, Yuan D, Xu Y, Wang Y, Yao K, Zhong X, Liu Q, Jia K, Lei L, Wang H, Liu D. Overexpression of BNIP3 in renal carcinoma cells can promote apoptosis of renal carcinoma cells through HIF-1α-BNIP3-mediated autophagy. Front Oncol. 2025;15:1614378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 34. | Zhang F, Ma C. Kaempferol suppresses human gastric cancer SNU-216 cell proliferation, promotes cell autophagy, but has no influence on cell apoptosis. Braz J Med Biol Res. 2019;52:e7843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 35. | Galluzzi L, Green DR. Autophagy-Independent Functions of the Autophagy Machinery. Cell. 2019;177:1682-1699. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 786] [Cited by in RCA: 713] [Article Influence: 101.9] [Reference Citation Analysis (6)] |
| 36. | He Y, Zhang X, Zhang X, Fu B, Xing J, Fu R, Lv J, Guo M, Huo X, Liu X, Lu J, Cao L, Du X, Ge Z, Chen Z, Lu X, Li C. Hypoxia exacerbates the malignant transformation of gastric epithelial cells induced by long-term H. pylori infection. Microbiol Spectr. 2024;12:e0031124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 37. | Willet SG, Lewis MA, Miao ZF, Liu D, Radyk MD, Cunningham RL, Burclaff J, Sibbel G, Lo HG, Blanc V, Davidson NO, Wang ZN, Mills JC. Regenerative proliferation of differentiated cells by mTORC1-dependent paligenosis. EMBO J. 2018;37:e98311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 159] [Article Influence: 19.9] [Reference Citation Analysis (1)] |
| 38. | Mazurakova A, Koklesova L, Csizmár SH, Samec M, Brockmueller A, Šudomová M, Biringer K, Kudela E, Pec M, Samuel SM, Kassayova M, Hassan STS, Smejkal K, Shakibaei M, Büsselberg D, Saso L, Kubatka P, Golubnitschaja O. Significance of flavonoids targeting PI3K/Akt/HIF-1α signaling pathway in therapy-resistant cancer cells - A potential contribution to the predictive, preventive, and personalized medicine. J Adv Res. 2024;55:103-118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 50] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 39. | Tao H, Zhu P, Xia W, Chu M, Chen K, Wang Q, Gu Y, Lu X, Bai J, Geng D. The Emerging Role of the Mitochondrial Respiratory Chain in Skeletal Aging. Aging Dis. 2024;15:1784-1812. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 40. | Ganapathy-Kanniappan S. Linking tumor glycolysis and immune evasion in cancer: Emerging concepts and therapeutic opportunities. Biochim Biophys Acta Rev Cancer. 2017;1868:212-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 97] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 41. | Kocianova E, Piatrikova V, Golias T. Revisiting the Warburg Effect with Focus on Lactate. Cancers (Basel). 2022;14:6028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 80] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 42. | Miao ZF, Sun JX, Adkins-Threats M, Pang MJ, Zhao JH, Wang X, Tang KW, Wang ZN, Mills JC. DDIT4 Licenses Only Healthy Cells to Proliferate During Injury-induced Metaplasia. Gastroenterology. 2021;160:260-271.e10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 43. | Trachsel-Moncho L, Veroni C, Mathai BJ, Lapao A, Singh S, Asp NT, Schultz SW, Pankiv S, Simonsen A. SNX10 functions as a modulator of piecemeal mitophagy and mitochondrial bioenergetics. J Cell Biol. 2025;224:e202404009. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 44. | Bao YY, Zhong JT, Shen LF, Dai LB, Zhou SH, Fan J, Yao HT, Lu ZJ. Effect of Glut-1 and HIF-1α double knockout by CRISPR/CAS9 on radiosensitivity in laryngeal carcinoma via the PI3K/Akt/mTOR pathway. J Cell Mol Med. 2022;26:2881-2894. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 45. | Ye Y, Wang Y, Xu Q, Liu J, Yang Z, Wuren T, Ge RL. In vitro study: HIF-1α-dependent glycolysis enhances NETosis in hypoxic conditions. Front Immunol. 2025;16:1583587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |