Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.117765
Revised: December 26, 2025
Accepted: January 26, 2026
Published online: April 15, 2026
Processing time: 114 Days and 3.5 Hours
Hematological parameters are important markers that can provide insight into the pathogenesis and progression of pancreatic cancer (PC). According to the li
Core Tip: Hematological parameters are important markers that can provide insight into the pathogenesis and progression of pancreatic cancer (PC). According to the literature, there are many hematological parameters in the pathogenesis of PC. However, this review article only summarized a few parameters, such as red blood cells, red cell distribution width, mean corpuscular volume, mean corpuscular haemoglobin concentration, platelets, mean platelet volume, white blood cells and their types, such as neutrophils, lymphocytes, monocytes, eosinophils and basophils, and their ratios in PC.
- Citation: Jamil T, Anis K, Rehman A, Iqbal S, Pervaiz S, Irfan Latif M, Hafeez R, Khurshid H, Rafaqat S. Role of hematological parameters in pancreatic cancer: A literature review. World J Gastrointest Oncol 2026; 18(4): 117765
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/117765.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.117765
Pancreatic cancer (PC) arises when cells in the pancreas, a glandular organ behind the stomach, begin to multiply out of control and form a mass. These malignant cells might spread to other areas of the body. There are several recognized forms of PC. About 90% of cases are of the most prevalent kind, pancreatic adenocarcinoma, which is frequently referred to as “pancreatic cancer” exclusively. The portion of the pancreas that produces digestive enzymes is where these adenocarcinomas begin. These cells can also give rise to several other cancer forms, which together account for most non-adenocarcinomas. Neuroendocrine tumors (NETs), which originate from the pancreatic cells that produce hormones, account for 1%-2% of instances of PC[1].
PC is a serious health issue. The incidence of PC is expected to rise globally, although the rate of increase differs by country, with some showing a dropping or slowing trend in incidence[2]. Pancreatic ductal adenocarcinoma (PDAC) is the fourth most deadly kind of cancer, accounting for 90%-95% of all PC cases reported globally[3]. PDAC accounts for around 90% of PCs[4]. PC is linked to unusually high death rates in several Asia-Pacific nations, including South Korea, Japan, Kazakhstan, New Zealand, Australia, and Armenia. On the other hand, PC mortality is increasing in other countries, such as China, indicating that peak death rates could be seen in the upcoming years[5].
The prevalence of PC is predicted to increase globally by 2040, with women predicted to be more vulnerable[6]. According to GLOBOCAN 2020 figures, PC incidence and mortality rates were predicted to rise significantly in the upcoming years[7]. According to the latest GLOBOCAN figures, PC is presently the sixth most common cause of cancer-related death globally and has one of the lowest survival rates of any malignancy[8,9].
DNA abnormalities in pancreatic cells, frequently in the ducts, cause uncontrolled proliferation and the formation of masses (tumors) that penetrate tissue and have the potential to spread. This process entails intricate interactions with the stroma, or surrounding tumor microenvironment (TME), which promotes tumor invasion, development, and resistance to treatment. Genetic predispositions, environmental variables (such as smoking, diabetes, and pancreatitis), and particular biochemical pathways are important drivers of pathogenesis, resulting in aggressive illness and sometimes delayed detection because of sluggish early development and resistance. Dysplastic abnormalities in the epithelia classified as low- and high-grade with a build-up of molecular modifications cause PDAC to develop. Sustaining PDAC is largely dependent on constitutive activation of mitogen-activated protein kinase (MAPK), which is facilitated by reduction of dual specificity phosphatase 6. Different molecules that act as effectors to maintain PDAC are induced by active MAPK. The growth and survival of PDAC cells are significantly aided by downstream Aurora kinase A and SON large protein located in the nucleus, specifically nuclear speckles, that binds to RNA and DNA. Additionally, active MAPK stimulates microRNAs that control PDAC cell growth. Familial PC kindreds have a variety of germline mutations thought to raise the risk of PC. Dilated ducts bordered with papillary neoplastic epithelia of different forms and atypia grades make up intraductal papillary mucinous neoplasms (IPMNs). Four kinds of IPMN papillae are distinguished based on clinicopathological characteristics, such as patient prognosis[10,11].
PC can be classified as either endocrine or exocrine. Rare endocrine cancers originate in cells that produce hormones. The cells that comprise the pancreatic ducts or the glands that produce enzymes are the source of exocrine pancreatic lesions. Among these is the relatively uncommon tumor known as acinar cell carcinoma. Mucinous cystic neoplasms, IPMNs, and benign or less malignant pancreatic intraepithelial neoplasms (PanINs) are also uncommon. Each of the three might go through a phase of malignant change. PDAC is the most prevalent exocrine neoplasm, accounting for 90%-95% of all instances of PC; around 10% of these cases have a family history[12].
Major risk factors for tumor formation include a family history of cancer, diabetes, smoking, and chronic pancreatitis (CP). Additionally, it was shown that strong alcohol use and obesity were linked to a higher risk. Back discomfort, weight loss, jaundice, nausea, and the beginning of diabetes are nonspecific signs of PC[13]. Measurements of blood components that show physiological health and can be used to identify illnesses are known as hematological parameters. The three primary blood cell types are derived from stem cells in the bone marrow: Red blood cells (RBCs, erythrocytes), which carry oxygen; white blood cells (WBCs, leukocytes), which fight infection; and platelets (thrombocytes), which aid in blood clotting. With distinct immunological roles, WBCs may be further classified into lymphocytes, monocytes, neutrophils, eosinophils, and basophils.
RBC count, hemoglobin, hematocrit, WBC count, and platelet count are important metrics. The neutrophil-to-lymphocyte ratio (NLR), the red cell distribution width (RDW), and RBC indices [such as mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular haemoglobin concentration (MCHC)] are additional crucial measurements. Hematological and immunological parameter reference ranges are commonly used in clinical practice to assess health and disease situations. The reference ranges might potentially be important indicators for assessing how a disease is progressing or how well a therapy is working. These characteristics can vary according to age, gender, ethnicity, environment, and genetic background[14-16].
Hematological parameters actively contribute to the pathogenesis of PC by driving inflammation, immune suppression, and hypercoagulability, all of which promote tumor progression. Elevated neutrophil counts stimulate chronic inflammation and release cytokines and neutrophil extracellular traps that enhance genetic instability, tumor invasion, and metastatic potential, while a concurrent reduction in lymphocytes weakens cytotoxic T-cell and natural killer (NK)-cell-mediated anti-tumor immunity. Increased platelet levels further support tumor growth by releasing pro-angiogenic and pro-invasive mediators and by shielding circulating tumor cells from immune detection, thereby facilitating metastasis. High monocyte counts contribute to the recruitment of tumor-associated macrophages (TAM), which promote matrix degradation, stromal fibrosis, and immunosuppression. However, literature reported that to forecast the prognosis of cancer with the help of certain recent blood test findings. Counts of WBCs, neutrophils, lymphocytes, granulocytes, NLRs, and platelet-to-lymphocyte ratios (PLRs) are some of these findings. These metrics show the patient’s level of inflammation, which may be important at various points in the tumor’s development. Initiation, promotion, invasion, and metastasis are among the phases[17,18].
The tumor’s microenvironment contains both innate (such as neutrophils, mast cells, myeloid-derived suppressor cells, dendritic cells, NK cells, and macrophages) and adaptive (such as T and B lymphocytes) immune cells. Within the tumor’s microenvironment, these immune cells take part in inflammatory reactions. It is yet unclear how inflammation affects cancer. On the one hand, inflammation could increase the growth of tumors[19]. In contrast, it promotes the cross-presentation of tumor antigens, which in turn triggers immune responses against the tumor[20]. Tumorigenesis has been linked to RBC abnormalities. However, the relationship between the risk of cancer and red cell indices, which represent the condition of RBCs, is yet unknown. As possible biomarkers for cancer risk prediction, MCV, MCH, and RDW are clinically significant. To clarify the underlying processes behind these relationships, more research is required[21].
Hematological parameters are widely used in clinical practice because they provide quick, inexpensive, and valuable information about a patient’s physiological status, disease activity, and treatment response. Complete blood count (CBC) derived indices such as hemoglobin, hematocrit, total leukocyte count, differential leukocyte count, platelet count, RDW, MCV, MCH, and MCHC help clinicians assess anemia, infection, inflammation, nutritional status, bone marrow function, and systemic stress. Parameters like NLR, PLR, and lymphocyte-to-monocyte ratio (LMR) are increasingly used as biomarkers of systemic inflammation and prognosis in various conditions, including cancers, cardiovascular diseases, and autoimmune disorders. Coagulation-related hematological markers aid in diagnosing thrombotic or bleeding disorders, which are common complications in malignancies and chronic inflammatory states.
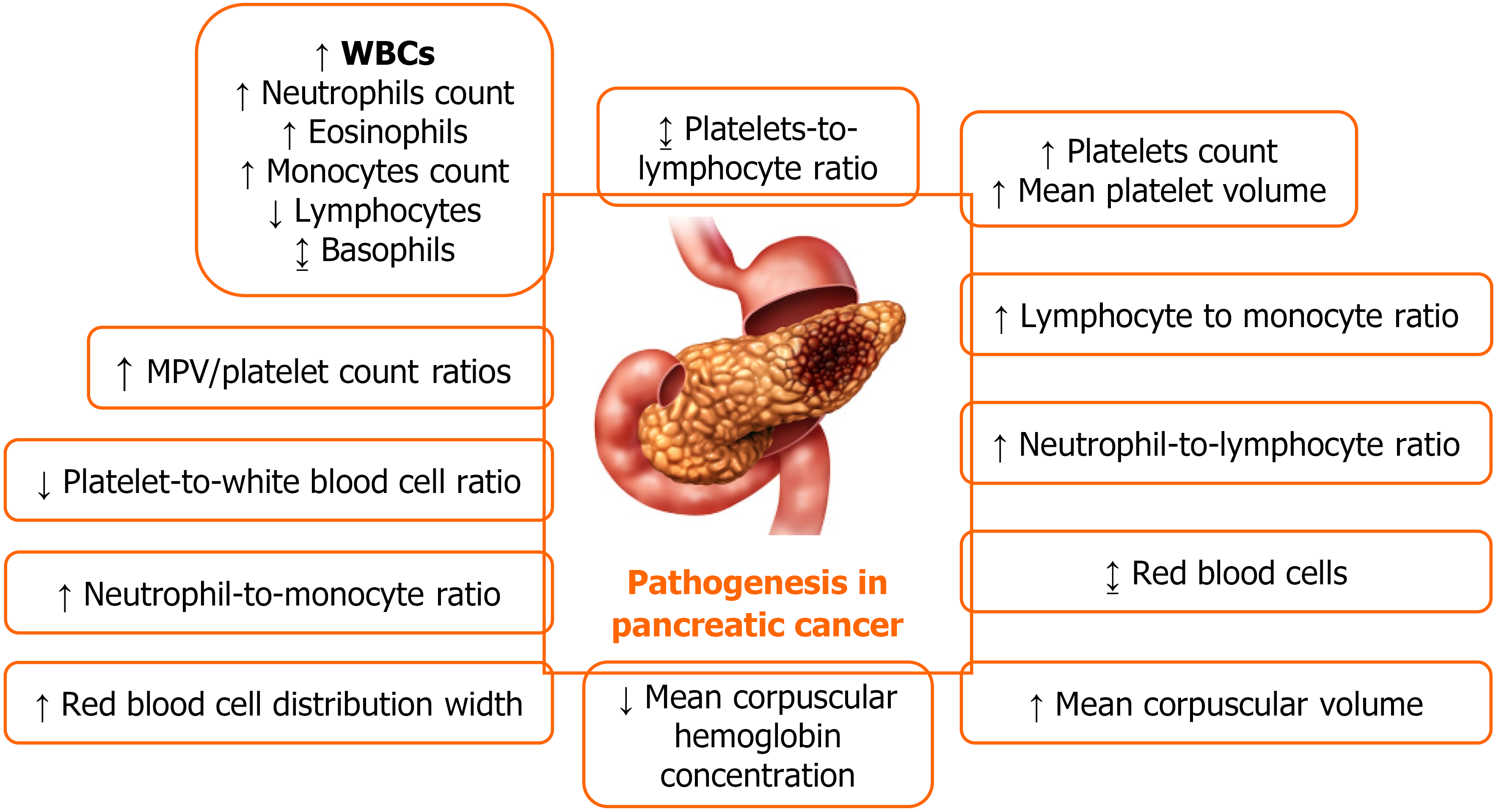
Overall, hematological parameters are essential tools in routine clinical practice because they support diagnosis, monitor disease progression, guide treatment decisions, and provide prognostic information across a wide range of medical conditions. According to the literature, there are many hematological parameters in the pathogenesis of PC. However, this review article only summarised a few parameters, such as RBCs, RDW, MCV, MCHC, platelets, mean platelet volume (MPV), WBCs and their types, such as neutrophils, lymphocytes, monocytes, eosinophils and basophils, and their ratios in PC.
According to the literature, there are many hematological parameters in the pathogenesis of PC. However, this review article only summarized a few parameters, such as RBCs, RDW, MCV, MCHC, platelets, MPV, WBCs and its types such as neutrophils, lymphocytes, monocytes, eosinophils and basophils, ratios including NLR, monocyte-to-lymphocyte ratio (MLR), and PLR, LMR, MPV/platelet count ratio, neutrophil-to-monocyte ratio (NMR) and platelet-to-WBC ratio (PWR) and MPV/platelet ratio (MPR) in PC as explained in Figure 1 and Table 1.
| Hematological parameters | Pathogenesis of PC |
| RBCs | PDAC is associated with the excessive aggregation of RBCs |
| RDW | Advanced tumor stages were linked to higher RDW levels. There is a correlation between RDW and the length of survival for PC patients |
| Platelets | Platelets have been identified as important participants in cancer, thrombosis, and hemostasis. Platelets can promote carcinogenesis and metastasis through a wide range of interplay between cancer cells and platelets. Pancreatic tumors often cause thrombosis by speeding up platelet aggregation. Platelets were once assumed to be solely involved in hemostasis, but they are now known to be important in the development of cancer and inflammation. Platelets contribute to the formation of an immunosuppressive milieu that encourages fibrotic remodeling, tumor initiation, development, metastasis, and immune evasion by releasing cytokines, growth factors, and pro-angiogenic mediators. There are several functional subtypes of neutrophils and macrophages that have both pro- and anti-tumorigenic properties |
| MPV | Activated platelets play a key role in tumor growth and tumor metastases. Mean platelet volume is a platelet index and is altered in patients with malignancies. There was no significant correlation seen between increased MPV and CA19-9, tumor location, or tumor size |
| WBCs | Patients with diabetes who had PC had greater levels of macrophage infiltration in lymph node metastases than in original tumors. The prognosis of PC was not substantially affected by type 2 diabetes or intra-tumoral leukocyte (macrophage, neutrophil, or eosinophil) infiltration alone. High macrophage or neutrophil tumor-infiltration, however, was linked to a markedly worse overall survival rate in cancer patients with type 2 diabetes |
| Neutrophils | Neutrophils are involved in acute inflammation. In general, neutrophils are drawn into the tumor microenvironment and have an impact on inflammation and tumor growth. Neutrophils, a vital component of the tumor microenvironment which are involved in angiogenesis, metastasis, progression, and immunosuppression in PC. Numerous biological processes that attract neutrophils to tumor lesions and stimulate carcinogenesis have been discovered. Neutrophils play an important part in the microenvironment of PC and influence tumor formation and development. Through a variety of processes, such as angiogenesis, immune suppression, immune evasion, and epithelial-mesenchymal transition, neutrophils aid in the liver metastasis of PC. The repercussions of neutrophil and macrophage activity aid in the development, hypoxia, fibrosis, and remodeling of tumors. Nuclear factor erythroid 2 is a crucial transcription factor that controls the phenotypic polarization of neutrophils. Transforming growth factor β, which is produced by metastatic tumors, activates the SMAD3 pathway in neutrophils, causing nuclear factor erythroid 2-driven polarization. By binding to the promoter of peptidylarginine deiminase 4, nuclear factor erythroid 2 stimulates its transcription, which results in the production of neutrophil extracellular traps near the invasive front. Neutrophil extracellular traps are one of them, and they are important for tumor metastasis |
| Lymphocytes | B lymphocytes are heavily infiltrated in pancreatic adenocarcinoma |
| Monocytes | It reveals two unique tumor-associated monocyte and macrophage populations that represent systemic immunological alterations in individuals with pancreatic ductal adenocarcinoma. PC-derived extracellular vesicles carry specific cargo that, upon uptake, enhance monocyte recruitment to tumor microenvironment and their subsequent differentiation to tumor-associated macrophages. High monocyte counts were a poor prognostic indicator in PC and that monocytes may directly trigger the epithelial-mesenchymal transition process in PC cells by upregulating the expression of Snail via the nuclear factor-κB signaling pathway |
| Eosinophils | Eosinophil accumulation and degranulation contribute to PC and fibrosis. Hypereosinophilia is a sign of malignancy in pancreatic adenocarcinoma and other solid tumor and hematological malignancies, in addition to prodromal symptoms. NOD-like receptor family pyrin domain containing 3-regulated interleukin-18, which is produced by accumulated macrophages in a murine model of PC, stimulates eosinophilic inflammation-mediated accumulation of periductal mucin and collagen, leading to the development of acinar-to-ductal metaplasia, pancreatic intraepithelial neoplasia, and intraductal papillary mucinous neoplasm |
| Basophils | Basophils can stimulate the formation of inflammation-driven skin tumors and are found in the immunological landscape of PC and human lung adenocarcinoma. Basophils play a useful role in the development of tumors. While basophil activation was triggered by T-cell-derived interleukin 3, basophil migration into tumor-draining lymph nodes depended in part on the production of chemokine C-C motif chemokine ligand 7/monocyte chemoattractant protein 3 by “alternatively activated” monocytes |
To conduct this literature review, multiple databases such as Google Scholar, PubMed, and Science Direct were utilized. The search process was concluded on December 10, 2025. Various keywords, including hematological para
The predictive use of hematological parameters and carbohydrate antigen 19-9 (CA19-9) in the assessment of inoperability by comparing resectable and unresectable patients over the laboratory data of PC patients was assessed. There was a significant difference between the groups when comparing the patients’ NLR, mass size, C-reactive protein/Lymphocyte ratios (CRP/L), C-reactive protein (CRP), and CA19-9 values. A statistical difference was found between patients in group 2 with superior mesenteric artery and superior mesenteric vein invasion (n = 26) and those in group I in terms of age, NLR, mass size, platelet/platelet distribution volume ratio (P/PDW), platelet/MPV, C-reactive protein, CRP/L, platelet distribution volume, and CA19-9 values. As a result, the likelihood of resectable surgery was found to be strongly influenced by NLR, CRP, CRP/L, CA19-9 levels, and tumor mass[22].
Nutrition and systemic inflammation are key factors in the spread of cancer. The haemoglobin, albumin, lymphocytes, and platelets that make up the combination index of haemoglobin, albumin, and platelets (HALP) are thought to be a unique way to measure systemic inflammation and nutritional status. HALP and the survival of patients with PC after radical resection, however, have not been examined in any research. High tumor-node-metastasis (TNM) stage, poor tumor differentiation, lymph node metastases, female patients, and tumor placement in the pancreatic head were all substantially correlated with low levels of HALP. For patients with resected pancreatic adenocarcinoma, low levels of HALP were linked to short survival and early recurrence for recurrence-free survival (RFS). Regardless of tumor site or sex, a low level of HALP was a risk factor for early recurrence and poor survival. For patients with resected PC, low levels of HALP may be a major risk factor for both overall survival (OS) and RFS[23].
To investigate the association between the prognosis of patients receiving radical resection for PDAC and their baseline clinical features and hematological markers, and to offer resources for classifying the patients’ clinical risks. The median OS (mOS) following surgery was 17.0 months [95% confidence interval (CI): 15.0-19.0]. Patients in the research had survival rates of 60.6%, 33.4%, 19.1%, 12.7%, and 9.6% at 1, 2, 3, 4, and 5 years, respectively. Numerous variables independently influence surgical survival in PDAC patients, according to the multivariate Cox proportional hazards model study. Tumor location [hazard ratio (HR) = 1.574, 95%CI: 1.233-2.011], tumor cell differentiation degree (HR = 0.687, 95%CI: 0.542-0.870), neural invasion (HR = 0.686, 95%CI: 0.538-0.876), TNM staging (HR = 1.572, 95%CI: 1.252-1.974), postoperative adjuvant therapy (HR = 1.799, 95%CI: 1.390-2.328), and elevated serum CA19-9 levels before surgery are some of these factors. Tumors in the head of the pancreas, moderate and high degrees of differentiation, absence of local neurovascular invasion, TNM stage I, postoperative adjuvant therapy, no prior alcohol use, and preoperative serum CA19-9 levels of less than or equal to 37 U/mL are all strongly linked to a better prognosis in PDAC patients[24].
Systematic inflammatory response has been intimately linked to carcinogenesis. WBC and platelet indices have been shown in several studies to be associated with malignancies as a prognostic factor. To examine the function of WBC and PLT indicators in the prediction of disease stage and differential diagnosis of pancreatic lesions was observed. Tumor marker levels, WBC and PLT indices, and possible correlations with specific types of pancreatic disease and clinicopathological characteristics were explored. It included 131 patients (69 males) with a mean age of 66.6 ± 10.7 years who had treatment for pancreatic lesions, including 23 cystic neoplasms, 12 NETs, and 94 adenocarcinomas. In comparison to pancreatic cystic neoplasms, adenocarcinomas showed considerably higher PLRs and significantly lower lymphocyte counts and lymphocyte to monocyte ratios. In comparison to operable disease, non-operable adenocarcinomas showed a lower LMR and a higher monocyte count. In comparison to non-metastatic adenocarcinomas, patients with metastatic cancer had reduced lymphocyte percentage and LMR and greater neutrophil, monocyte count and neutrophil to lymphocyte ratio. Lymphocyte count and LMR can be used to forecast the disease’s metastatic state and differentiate adenocarcinomas from pancreatic cystic neoplasms[25].
RBCs are the most prevalent type of blood cells. They are produced in the bone marrow, carry oxygen from the lungs to the body using hemoglobin, which gives blood its red color, and return carbon dioxide. They are biconcave, lack a nucleus (in mammals) for more space, and last roughly 120 days before being recycled. Blood tests are used to measure RBC levels for diseases like anemia. Also, RDW is a blood test component of a CBC that measures the variation in the size (volume) of RBCs. A normal RDW indicates uniform cell size, which helps doctors diagnose blood disorders by comparing RDW with MCV. A high RDW indicates cells vary a lot in size, which can signal underlying issues like iron deficiency or other anemias[26,27].
The RBC distribution is a test measure of erythrocyte variation and volume level that demonstrates heterogeneity and has been shown in the literature to be a reliable method for determining survival in cardiovascular illnesses. Investigating the association between the RDW level and postoperative morbidity and its phases in patients with PC was assessed. It found that low blood albumin levels and CA19-9 levels were positively correlated, as were illness stages and RDW levels. However, there was no statistically significant variation in postoperative morbidity[28].
The hemoglobin-to-RDW ratio (HRR) has been linked to the prognosis of certain malignant cancers. Nevertheless, the association between HRR and the prognosis of PDAC is still unknown. Zhou et al’s study[29] determined the prognostic importance of HRR in patients with PDAC. Age, hemoglobin levels, RDW, tumor location, surgical intervention, and postoperative chemotherapy showed significant differences between the two groups (P < 0.05). Notably, the disease-free survival (DFS) and OS rates were lower in the low HRR sample (P = 0.002 for both). Male gender, surrounding tissue invasion, TNM stages III/IV, non-O blood types, low HRR, and absence of postoperative chemotherapy were associated with poor DFS and OS outcomes, according to univariate analysis. Low HRR was also identified by multivariate analysis as an independent predictor of poor DFS and OS outcomes (HR = 1.520, 95%CI: 1.028-2.247, P = 0.036; HR = 1.537, 95%CI: 1.034-2.284, P = 0.034, respectively). The potential usefulness of HRR as a prognostic biomarker for this group indicates that lower HRR is associated with worse DFS and OS in PDAC patients.
The host’s immune system may be adversely affected by perioperative RBC transfusion (RBCT). Another study looked at how perioperative RBCT affected patients with PDAC in terms of long-term survival. Age, preoperative haemoglobin levels, levels of the CA19-9, maximal tumor size, tumor staging, operating time, intraoperative blood loss, major vascular resection, and the percentage of pancreaticoduodenectomies done were all significantly different between the two groups. The OS and RFS of patients who had RBCTs were considerably lower than those who did not. Patients with PDAC who had surgery with the intent of curing their condition had a worse long-term survival rate when perioperative RBCT was used[30]. Blood-based biomarkers and abnormalities in RBC aggregation linked to PC were evaluated. It showed that blood-based biomarkers for distinguishing between PDAC and inflammatory pancreatic tumors varied significantly. Additionally, it verified the link between PDAC and excessive RBC aggregation[31].
To predict the prognosis of PC patients, another study evaluated the RDW value. The high-RDW group had a lower immune-nutritional state than the low-RDW group. The prognosis was likewise worse for the patients in the high-RDW group. An independent risk factor for PC may be the RDW, according to risk factor analysis. An independent predictor of postoperative survival in PC patients, the RDW is linked to the immune-nutritional state[32].
The absence of accurate and affordable prognostic factors makes it difficult to predict the clinical course of PC. In some cancers, RDW is a measure of the variation in the size of circulating RBCs, has been shown to have prognostic relevance. Literature demonstrating its applicability to PC is few. Another study linked RDW with survival following PC surgery and investigated the relationship between RDW and tumor stage in patients with PC who were admitted to a tertiary care hospital in South India. 254 patients with PC who had surgery at a tertiary facility between 2002 and 2015 had their prospectively gathered digital medical information retrospectively analyzed. Advanced tumor stages were linked to higher RDW levels. High RDW values were seen in 84.2% of patients with stage 3 cancer and 92.3% of patients with stage 4 cancer. Reduced survival was substantially correlated with high levels. Patients with normal levels had a mean survival time of 83 months, but those with higher values had a much shorter mean survival time of 72 months. In PC, there seems to be a strong correlation between RDW and tumor stage. Additionally, there is a correlation between RDW and the length of survival for individuals with PC. Therefore, it might help forecast the clinical course of PC[33].
Another study used a national Korean database to examine perioperative patterns of RBCT and associated factors in PC surgery. It evaluated data from the National Health Insurance Service between 2012 and 2020, which included patients with newly diagnosed PC who were at least 20 years old and had pancreatic surgery within a year after the diagnosis. Receiving at least one unit of allogenic RBCs from one week before surgery until hospital discharge was considered a perioperative RBCT. 18% of the 10473 patients had RBCTs during surgery[34].
The transfusion rate increased to 19.9% in 2020 after falling from 20.1% in 2012 to 12.7% in 2015. The likelihood of requiring RBCTs increased with each 10-year increase in age [odds ratio (OR) = 1.30; 95%CI: 1.24-1.37], female sex (OR = 1.16; 95%CI: 1.05-1.29), and being in the lowest income quartile as opposed to the highest (OR = 1.29; 95%CI: 1.11-1.49). Additionally, a higher risk was independently associated with a higher Charlson comorbidity index. Total pancreatectomy showed greater chances (OR = 1.91; 95%CI: 1.56-2.35) than pancreaticoduodenectomy, but distal pancreatectomy had lower odds. Additionally, the transfusion likelihood was greater in general hospitals than in tertiary institutions (OR = 1.38; 95%CI: 1.22-1.56). Korea should emphasize greater adoption of multidisciplinary blood management over prolonged dependence on transfusion, given increased RBCT rates among low-income patients and restricted National Health Insurance Service coverage for novel transfusion-sparing techniques[34].
In patients with advanced pancreatic NETs (PNETs), there is a dearth of information on easily accessible indicators that allow for quick risk assessment and individualized treatment. In patients with PNETs, another study investigated the relationship between characteristics linked to RBCs and prognostic indicators. RBC mass level < 3.9 m/μL and a 6% and 9% relative reduction in hemoglobin and hematocrit levels, respectively, were linked to an increased risk of disease progression. Patients with disease progression had lower hemoglobin, RBC mass values, and hematocrit at the most recent evaluation. In a similar vein, during the final follow-up, patients who passed away during the study period had lower hemoglobin, RBC mass levels, and hematocrit[35].
An 11% decrease in hemoglobin levels was observed, suggesting an increased risk of death. A higher probability of illness progression was independently linked to negative hemoglobin and hematocrit dynamics. Patients with PNETs have a bad prognosis when their RBC mass, hemoglobin, and/or hematocrit levels fall. In this patient population, it is recommended to use these characteristics as supplementary follow-up prognostic indicators to radiologic imaging[35].
The effectiveness of L-asparaginase (L-asp) depends on how sensitive tumor cells are to asparagine deprivation because they lack asparagine synthethase (ASNS). However, serious toxicities, including allergic reactions, coagulation problems, and pancreatic insufficiency, make its usage difficult. ASNS deficiency has been linked to potential sensitivity to L-asp treatment in several tumor cell lines, including PC. However, the literature reports tentative use of L-asp in solid tumors, particularly PC, but all attempts have failed because of toxicity thus far. In leukemic patients, a novel formulation of L-asp loaded into RBC (GRASPA) has recently demonstrated a very encouraging safety profile. The first phase of I clinical trial using GRASPA in PC patients is called GRASPANC. Finding the cell-based formulation’s dose limiting toxicity was investigated[36].
Secondary goals were to evaluate GRASPA’s overall safety profile and activity (measured by serum asparagine depletion). Patients with non-resectable, relapsed PC were treated with a single injection of GRASPA at escalating doses (25, 50, 100, or 150 IU/kg) in a cohort of three. Up to the GRASPA maximum dosage (150 IU/kg) scheduled for the trial, no limiting toxicity was discovered, and the dose limiting toxicity was not met. There was no indication of any particular unanticipated non-limiting toxicity. Furthermore, it was demonstrated that GRASPA was efficient for depleting serum asparagine at all dosages, with a trend toward prolonged action with increasing dose. The dose of 150 IU/kg of GRASPA is safe and seems appropriate for additional clinical research in PC patients. Patients should be chosen based on whether or not their tumor cells express ASNS in order to maximize the potential efficacy of this novel treatment[36].
A common blood test, known as a CBC, determines the MCV. RBCs’ average size is measured by MCV. It uses RBCs to transport oxygen. The cells are subsequently powered by this oxygen. The RBCs’ size and other properties tell you how well they can carry oxygen. There is increasing evidence that MCV is a predictive factor for certain cancers. Another study looked at the predictive significance of pre-therapeutic MCV in patients with PDAC who had either upfront resection or resection following neoadjuvant therapy (NT). The median MCV values are used as a cut-off point to differentiate between high and low MCV levels. According to multivariate analysis, there was a significant negative correlation between OS and high MCV before and after NT. Additionally, the tumor response to NT was linked to the median MCV value from before to after NT, which increased considerably and was connected with tumor response[37].
The MCHC, a crucial component of a CBC used to diagnose various forms of anemia and blood disorders, measures the average concentration of hemoglobin (an oxygen-carrying protein) within a given volume of RBCs, indicating how densely packed the hemoglobin is inside each cell. To determine the significance of MCHC on OS in cases of resectable PC was designed. A significant contributing factor to the development and spread of malignant tumors is the systemic inflammatory response. MCHC is thought to have an impact on the prognosis of several cancers, such as head and neck malignancies and lung cancers. Nevertheless, the relationship between these and PC has not been well investigated. The OS was considerably better for resectable PC patients with reduced preoperative MCHC, according to the survival curve (P = 0.015). MCHC (HR = 1.266; 95%CI: 0.895-1.792; P = 0.028) was found to be an independent prognostic factor for patients with resectable PC, according to multivariate analysis[38].
Small blood cells called platelets serve a variety of physiological functions, the most well-researched of which is thrombosis activation. They play a critical role in preserving sufficient blood volume in individuals with vascular damage by activating the coagulation cascade and clotting. By sticking to the vascular endothelium, clumping together with other platelets, and starting the coagulation cascade, platelets preserve hemostasis by creating a fibrin mesh that successfully stops large blood loss. Inflammation, tissue development, and immunological response all depend on platelets. These processes are mediated by the release of various growth factors, immunoglobulin G, and complement system components from the alpha and dense granules[39].
Platelets have been identified as important participants in cancer, thrombosis, and hemostasis. Platelets can promote carcinogenesis and metastasis through a wide range of interplay between cancer cells and platelets, according to preclinical and clinical studies. Around the world, PC is a terrible illness with a high morbidity and death rate. Even if the clinical diagnosis of PC and platelets is explained, the underlying pathological mechanism and routes of this interaction are still being thoroughly investigated. It was evident from the data that their intricate interaction was caused by a variety of underlying processes. Pancreatic tumors often cause thrombosis by speeding up platelet aggregation. Additionally, platelets’ extracellular vesicles (EVs) facilitate communication within the neoplastic milieu and demonstrate how these interactions propel the course of illness. Potential diagnostic and treatment approaches for the advancement of PC may be offered by a deeper comprehension of tumor and platelet interplay, which is founded on organoids and translational treatments[40].
One of the most aggressive and deadly types of cancer is PDAC, which has a highly desmoplastic TME. CP is a major risk factor. Persistent inflammation that promotes genomic instability, acinar-ductal metaplasia, and the development of PanIN has a significant impact on the progression of CP to PDAC. Immune cells and other extracellular matrix components can influence this stage of development. This comprises innate immune system cells that either promote or inhibit tumor growth, such as NK cells, macrophages, dendritic cells, mast cells, neutrophils, and myeloid-derived suppressor cells[41].
On the one hand, by releasing growth factors and cytokines, innate immune cells can initiate inflammatory reactions that promote tumor cell invasion, proliferation, and metastasis. However, they can also trigger immune surveillance systems, which can stop the growth of tumors. For instance, active dendritic cells are essential for a successful anti-tumor immune response, and NK cells are cytotoxic innate lymphoid cells that may destroy tumor cells. Mast cells and myeloid-derived suppressor cells, on the other hand, promote a pro-tumorigenic TME, which is further maintained by platelets. Platelets were once assumed to be solely involved in hemostasis, but they are now known to be important in the development of cancer and inflammation. Platelets contribute to the formation of an immunosuppressive milieu that encourages fibrotic remodeling, tumor initiation, development, metastasis, and immune evasion by releasing cytokines, growth factors, and pro-angiogenic mediators[41]. There are several functional subtypes of neutrophils and macrophages that have both pro- and anti-tumorigenic properties. Developing novel therapeutic strategies that can use the immune system and possibly the coagulation system to target and eradicate tumors, providing hope for better patient outcomes, requires an understanding of the intricate relationships between innate immune cells, platelets, early precursor lesions, and PDAC cells[41].
Another study assessed how well platelet counts predict the prognosis of individuals with PC. Patients with high platelet counts were predicted to have poor OS following therapy, as evidenced by the pooled HR of 1.51. Those who underwent surgical resection had a higher predictive value of platelet levels, according to subgroup analysis, followed by those who received palliative care. A valuable prognostic indicator for PC may be platelet counts. It is expected that patients with high platelet counts would not survive[42].
To sum up, in recent years, incredible strides have been achieved in the knowledge of platelet-mediated signaling pathways. Studying platelets offers new perspectives on how they function and serves as the foundation for creating tailored treatments that might enhance patient outcomes. While how PC cells evade NK cells and cause distant metastases has been somewhat identified, it is still unknown if PC cells use the same strategies to evade other immune cells. A possible strategy for PC therapy might be to enhance immune cell surveillance and killing capacity against cancer cells. It is important to highlight that therapeutic effectiveness has not yet been assessed for antiplatelet treatment in combination with radiation or chemotherapy and platelet-related nanotechnology in vitro and animal models. To identify critical areas in the pathophysiology and therapy of platelet-induced PC, platelets and PC organoid co-culture models are useful[43].
Moreover, scientific and rational methods should be prioritized to guarantee patient safety, considering the high frequency of peptic ulcers and gastrointestinal bleeding brought on by antiplatelet medications. The improvement of patient treatment will be firmly based on a deeper comprehension of platelet-mediated signaling networks. Individuals with PC may benefit from combination treatments that include immunotherapy, chemotherapy, and nanotechnology[43].
Finding early-stage diagnostic biomarkers is crucial since PC is a disease with a high death rate because it is discovered at an advanced stage. Since platelets are known to have a bidirectional interaction with tumor cells, they are an excellent option for the liquid biopsies that are now being investigated. It was discovered that co-culturing platelets with PC cells enhanced the ability of BXCP3 cells to proliferate and migrate, enhanced clonogenicity, and resulted in increased expression of Nanog, Sox2, and octamer-binding transcription factor 4[44].
Given that platelets can transmit microRNAs horizontally, it also identified the differences in microRNAs expression between platelets from a small group of PC patients and healthy individuals. Platelets from cancer patients and healthy persons showed distinct variations in the expression of several microRNAs. Additionally, upon examining microRNAs from the platelets of pancreatic juice and blood obtained from each cancer patient, it discovered intriguing variations between the platelets derived from pancreatic juice and blood, indicating the existence of distinct subpopulations of platelets in cancer patients that require additional investigation[44].
As part of a typical CBC, MPV determines the average size of your platelets, which are small blood cells essential for clotting. MPV helps diagnose bleeding disorders, bone marrow disorders, inflammatory diseases, and heart disease by showing how effectively your platelets are functioning. Larger platelets often indicate they’re younger and more active, while smaller ones signal older platelets. The majority of PC patients have many liver metastases when they are first diagnosed. Both tumor development and metastasis are significantly influenced by activated platelets. Patients with cancer have changed MPV, a platelet indicator. Examining whether MPV can accurately forecast mortality in PC patients with synchronous liver metastases was investigated. A retrospective collection of clinicopathological data was done to assess the correlations between MPV levels and clinical indicators. There was no significant correlation seen between increased MPV and CA19-9, tumor location, or tumor size. Patients with MPV > 8.7 fL had a substantially worse OS rate than those with MPV ≤ 8.7 fL, according to the Kaplan-Meier analysis (log-rank P < 0.001). According to the multivariable Cox proportional hazards model, MPV is a separate poor prognostic factor for OS. To sum up, in PC patients with synchronous liver metastases, higher MPV is linked to a worse survival result[45]. A lower MPV indicates a worse prognosis for some types of cancer. It is unknown, therefore, how important it is as a predictive marker for PC. It may be possible to predict long-term results for PC patients by classifying them according to MPV and CA19-9[46].
Another study observed the general features of PC patients and deep vein thrombosis (DVT) and assessed the connection between survival, DVT, and MPV. There were 28 females (36.4%) and 49 males (63.6%). Nine patients (11.7%) had malignant epithelial tumors, while sixty-eight patients (88.3%) had adenocarcinomas. At diagnosis, 36 (46.7%) had metastases to the liver. Of the patients, twenty (26%) were deceased, twenty-six (33.8%) were living, and thirty-one (40.2%) had an unknown status. Only 14 individuals (18.1%) experienced DVT. MPV levels were normal in 42 (54.5%) patients, above normal in 28 (36.4%) patients, and below normal in 7 (9.1%) patients. Gender, tumor location, treatment, and survival rates did not differ statistically significantly. DVT, gender, location, histological subtype, metastasis, stage, and whether the patient had received chemotherapy treatment did not significantly differ from one another. Additionally, there was a significant difference between liver metastasis and DVT, but not between MPV and DVT. PC patients were predicted by their age, stage, metastases, and DVT. More research should be done on cases of PC with liver metastases since these individuals are more likely to develop thrombosis[47].
The current study aims to assess the diagnostic utility of MPV in PNETs and pancreatic adenocarcinomas retrospectively[48]. The demographics and clinical data of 92 patients who were hospitalized between March 2007 and December 2009 for pancreatic adenocarcinoma (n = 76) and PNET (n = 16) were examined retrospectively. While 37 patients (48.7%) had an incurable pancreatic adenocarcinoma, 39 patients (51.3%) had a resectable one. Nine patients (56.3%) had a non-functional PNET, 6 patients (37.5%) had an insulinoma, and the remaining one patient had a gastrinoma. For pancreatic adenocarcinomas, the mean age was 59.3 ± 10.5, whereas for PNETs, it was 45.1 ± 10.6. Patients with pancreatic adenocarcinomas were substantially older at diagnosis than those with PNET (P < 0.001). Patients with pancreatic adenocarcinoma had substantially lower preoperative mean hemoglobin levels than those with PNET (12.4 ± 1.8 g/dL vs 13.7 ± 2.2 g/dL, P < 0.013)[48].
Patients with pancreatic adenocarcinomas had preoperative median MPV values of 8.6 fL (6.6-13.5) compared to 7.8 fL (7.2-9.4) in patients with PNET (P < 0.014). The difference between pancreatic adenocarcinomas and non-functional PNETs was the primary source of a significant variation in MPV levels in subgroup analysis (P = 0.017). Using receiver operating characteristic (ROC) analysis, the MPV level cut-off value for PNET detection was determined to be -7.8 fL [sensitivity: 66.7%, specificity: 75.9%, area under the curve: 0.734 (0.587-0.880); P = 0.022]. Age (OR = 0.068, 95%CI: 0.012-0.398), CA19-9 (OR = 0.039, 95%CI: 0.006-0.263), MPV (OR = 0.595, 95%CI: 0.243-1.458), and hemoglobin (OR = 1.317, 95%CI: 0.831-2.086) were determined as independent predictive factors for PNETs in the differential diagnosis of pancreatic adenocarcinomas. In order to differentiate PNETs from pancreatic adenocarcinomas, age, CA19-9, MPV, and hemoglobin levels are useful diagnostic markers[48]. PC risk has been increased by type 2 diabetes mellitus (T2DM). The most prevalent neoplastic condition that starts in the pancreas is called PDAC. There is growing evidence that tumor and T2DM are significantly influenced by platelet activation[49].
A measure of active platelets, MPV, is changed in a number of malignancies. Another study assessed MPV’s prognostic significance in T2DM patients with resectable PDAC. MPV levels were much greater in PDAC patients with T2DM than in those without the condition. Additionally, MPV was strongly linked to the distinction between T2DM and non-T2DM. Additionally, diabetic individuals with low MPV levels had a worse 5-year OS than those with high MPV levels, according to Kaplan-Meier analysis. MPV was an independent predictive factor for OS in T2DM patients, according to multivariate analyses. However, patients lacking T2DM did not exhibit the independent predictive effect of MPV[49].
Leukocytes, also known as WBCs, are vital immune system cells produced in the bone marrow that fight infections, illnesses, and foreign invaders like bacteria and viruses. They travel through blood and tissues to where they are needed. The main types of WBCs are neutrophils, lymphocytes (B and T cells), monocytes, eosinophils, and basophils, each of which has a specific role in defense. A WBC count counts these cells to assess general health.
To determine the predictive importance of WBC counts in patients receiving first-line treatment with gemcitabine (GEM) and nab-paclitaxel for locally progressed or metastatic PC was observed[50]. From July 2014 to March 2019, all patients received first-line nab-paclitaxel and GEM treatment for locally advanced or metastatic pancreatic adenocarcinoma at a single referral centre (the Second Department of Oncology, “Agios Savvas” Anticancer Hospital). All patients at the “Agios Savvas” Anticancer Hospital had their blood levels of WBCs, neutrophils, lymphocytes, and monocytes tested centrally during the final two days before the beginning of the first cycle of chemotherapy. The study comprised 19 (48.7%) male and 20 (51.3%) female patients with a median age of 65 years (range, 46-79). One in twenty (51.3%), two in two (5.1%), and seventeen (43.6%) patients had zero Eastern Cooperative Oncology Group performance status (ECOG-PS). Thirty patients (76.9%) were at stage IV[50].
The median blood values for WBC, neutrophils, lymphocytes, and monocytes were 7.8 × 103/μL (range: 3.8-17.5), 5.1 × 103/μL (range: 2.0-14.2), 1.8 × 103/μL (range: 0.4-4.9), and 0.6 × 103/μL (range: 0.1-1.8). The median WBC levels were 7.7 × 103/μL for patients with stage III and 7.6 × 103/μL for patients with stage IV disease (P = 0.658); the median neutrophils blood levels were 4.9 × 103/μL for patients with stage III and 5.0 × 103/μL for patients with stage IV disease (P = 0.979); the median lymphocytes blood levels were 1.9 × 103/μL (range, 0.4-4.9) and 1.5 × 103/μL for patients with stage IV disease (P = 0.696). Twenty-four (61.5%) experienced progressive illness after a median follow-up of 13.2 months (range, 0.9-28.4 months). 7.1 months (95%CI: 2.9-11.2) was the median progression-free survival (PFS). The median PFS for stage III patients was 9.2 months (95%CI: 0.4-18.0), while the median PFS for stage IV patients was 6.0 months (95%CI: 3.6-8.5). This difference was not statistically significant (P = 0.901)[50].
Additionally, the median PFS for patients with ECOG-PS 0 was 9.2 months (95%CI: 4.6-13.8), which was not statistically different from the median PFS for patients with ECOG-PS 1-2, which was 5.1 months (95%CI: 3.0-7.2; P = 0.606). Prognosis was linked to WBC levels above the median (HR = 0.23, 95%CI: 0.08-0.62, P = 0.004). Multivariate examination of prognostic variables verified their independent prognostic value (HR = 0.15, 95%CI: 0.04-0.57, P = 0.005 for WBC levels and HR = 3.00, 95%CI: 1.11-8.12, P = 0.030 for ECOG-PS). On the other hand, blood levels of neutrophils, lymphocytes, and monocytes were not predictive[50].
Another study assessed the usefulness of utilizing blood test data to predict the prognosis of PC. The pretreatment values of WBC > 5.8 × 109/L, granulocyte ≥ 3.7 × 109/L, neutrophil ≥ 3.9 × 109/L, lymphocyte < 1.4 × 109/L, and NLR ≥ 2.8 were significantly correlated with a worse OS rate. WBC > 5.8 × 109/L and granulocyte ≥ 3.7 × 109/L were shown to be independent predictive variables for OS in PC patients. Granulocyte count and WBC pretreatment values were independent variables with a poor prognostic capacity for PC[51].
Patients with PC continue to have a very short life expectancy. Although there are still significant gaps regarding their combined impacts on disease outcome, T2DM and inflammatory state may have an impact on the illness’s course[52]. Investigating the clinical importance of the tumor immune microenvironment on the prognosis of PC in connection to T2DM status was conducted. In a retrospective cohort of patients with resectable PC in Sweden, tumor-infiltrating macrophages, neutrophils, and eosinophils were examined in relation to patient and tumor characteristics, T2DM status, and OS in primary pancreatic tumors and paired lymph node metastases. Twenty-two (27.2%) of the eighty individuals with PC had T2DM. Compared to individuals without diabetes, patients with diabetic PC showed a substantially greater systemic high WBC count. Patients with diabetes who had PC had greater levels of macrophage infiltration in lymph node metastases than in original tumors[52].
The prognosis of PC was not substantially affected by T2DM or intra-tumoral leukocyte (macrophage, neutrophil, or eosinophil) infiltration alone. High macrophage or neutrophil tumor-infiltration, however, was linked to a markedly worse OS rate in cancer patients with T2D (adjusted HR = 7.2; 95%CI: 1.5-35.0 and HR = 5.4, 95%CI: 1.1-26.3, respectively). These findings show links between T2DM and increased inflammatory processes, which have important consequences for survival in T2DM patients with PC. Additional prognostic tools and better treatment approaches for particular patient subgroups may be found through validation in larger independent patient cohorts[52].
Neutrophils: Neutrophils are the most prevalent type of WBC and serve as the immune system’s first responders to infection and injury by engulfing and destroying pathogens (phagocytosis), releasing enzymes, and signaling other immune cells. Although levels can increase (neutrophilic leukocytosis) or decrease (neutropenia) with different conditions. They are essential for fighting bacteria, fungi, and viruses and promoting tissue repair. To defend against infection and inflammatory assaults, the innate immune system relies heavily on neutrophils, the most prevalent immune cells in the blood. The surface markers CD71 and CD117 were used to identify an early homogenous neutrophil progenitor subset in human bone marrow, according to recent research[53].
One type of cancer that affects the digestive tract and has a high global death rate is PC. Neutrophils are traditionally believed to be involved in acute inflammation. On the other hand, their significance in malignant disorders has not received as much attention. In general, neutrophils are drawn into the TME and have an impact on inflammation and tumor growth. Neutrophils, a vital component of the TME which are involved in angiogenesis, metastasis, progression, and immunosuppression in PC. Furthermore, neutrophils may be a novel therapeutic target for cancer treatment. Antitumor effects may be produced by cytokine, chemokine, and neutrophil extracellular trap inhibitors[54].
PDAC is a cancer with a low survival rate and a bad prognosis. PDAC is known for its aggressiveness, which is attributed to its fibroinflammatory TME, which is rich in fibroblasts and other immune cells. Important immune cells that infiltrate the PDAC microenvironment include neutrophils. Numerous biological processes that attract neutrophils to tumor lesions and stimulate carcinogenesis have been discovered by recent research. Another study provides an overview of the existing knowledge about the interactions of tumor cells, neutrophils, and other elements in the PDAC TME. Additionally covered are neutrophil prognosis and treatment implications in PDAC[55].
Neutrophils play an important part in the microenvironment of PC, and recent research has demonstrated their substantial influence on tumor formation and development. The development and outcome of PC are significantly influenced by neutrophil infiltration. Through a variety of processes, such as angiogenesis, immune suppression, immune evasion, and epithelial-mesenchymal transition (EMT), neutrophils aid in the liver metastasis of PC. Targeting neutrophils, therefore, shows promise as a key treatment approach to prevent liver metastases of PC[56]. The innate immune system’s essential components, neutrophils, are crucial to the PDAC immunological milieu. Neutrophils contribute to the formation and spread of tumors in PDAC and are linked to a poor prognosis[57].
Individuals with locally advanced PC (LAPC) may have a baseline neutrophil count that predicts their OS. It examined the prognostic significance of both baseline neutrophilia (neutrophil count > 7 g/L) and raised or growing neutrophilia as either neutrophilia or increased absolute neutrophil count (ANC) following induction chemotherapy in comparison to baseline for OS, PFS, and local control (LC)[58].
Ninety-eight patients (38%) with blood counts available after induction treatment had increased or growing neutrophil counts, which were linked to worse OS in both univariate (median 14.4 months vs 17.9 months; P = 0.001) and multivariate analysis (P = 0.004). Chemoradiation therapy’s reduced benefit on LC was likewise predicted by an elevated or rising neutrophil count. One-year LC was 80% in the chemoradiation arm and 54% in the chemotherapy arm in 126 patients when the neutrophil count was neither high nor rising during investigation. A large series of patients with LAPC showed lower OS among those with baseline neutrophilia and an elevated ANC. A powerful predictive LC biomarker for the effectiveness of chemoradiation treatment, the numbers were also an independent prognostic predictor. When choosing patients who may benefit from chemoradiation treatment following induction chemotherapy, a neutrophil count measurement might be helpful[58].
The concept that interactions with tumor-associated neutrophils (TAN) sustain the microenvironment is introduced by focusing on the spatial organization, polarity/function, recruitment, and origin of TAM. The repercussions of neutrophil and macrophage activity aid in the development, hypoxia, fibrosis, and remodeling of tumors. To overcome the obstacles posed by immune infiltration in pancreatic adenocarcinoma, it concludes by outlining a suite of therapeutic targets, including those meant to overcome TAM and TAN recruitment and function[59].
Liver metastases from PC are a major contributing cause to poor prognoses. The specifics of adaptive immunological remodeling in liver metastasis are still unclear, particularly with regard to neutrophils’ function. Here, the results of spatial transcriptomics paired with single-cell sequencing showed that liver metastases have greater levels of immunosuppression and more aggressive transcriptional traits than the original tumor. Specifically found in the invasive front of the metastatic lesions, neutrophils S100 calcium-binding protein A12 cells were identified as the crucial pro-metastatic cluster. Nuclear factor erythroid 2 (NFE2) is a crucial transcription factor that controls the phenotypic polarization of neutrophils. Transforming growth factor β, which is produced by metastatic tumors, activates the SMAD3 pathway in neutrophils, causing NFE2-driven polarization. By binding to the promoter of peptidylarginine deiminase 4, NFE2 stimulates its transcription, which results in the production of neutrophil extracellular traps near the invasive front. All of the findings point to NFE2-driven neutrophil polarization as a possible anti-metastatic treatment target[60].
The five-year survival rate for PDAC, a very aggressive malignant tumor of the digestive system, is about 12%. The long-term survival rate of PDAC patients is significantly impacted by liver metastasis, and the effectiveness of existing therapeutic approaches has been inadequate. Investigating the causes behind PDAC metastasis is crucial. A variety of cellular components make up the TME. Among these, neutrophils’ function in TME has garnered significant interest. In TME, neutrophils are polarized into TANs and then stimulated to develop into tumor-promoting (N2) and anti-tumor (N1) subtypes. Through a variety of strategies, including immunosuppression, angiogenesis, and DNA damage, neutrophils can promote tumorigenesis and metastasis. Neutrophil extracellular traps are one of them, and they are important for tumor metastasis. The prognosis of patients is correlated with several subgroups of neutrophils in TME. Neutrophils in liver metastases from PC have distinct features. TANs appear to be interesting targets for the therapy of PC, according to recent research. This study highlights many therapeutic approaches that target TANs, including polarization, neutrophil chemotaxis, and the development of neutrophil extracellular traps[61].
One of the most aggressive tumors is PDAC and chemoresistance makes therapy extremely difficult. Although neutrophil extracellular traps have become important components of the TME, it is still unknown how they affect chemoresistance. Using patient tumor samples, in vitro experiments with GEM-treated PDAC cells, and in vivo mice models, it examined the role of neutrophil extracellular traps in PDAC chemoresistance. With and without the C-X-C motif chemokine receptor (CXCR) 1/2 inhibitor navarixin, it assessed cytokine production, neutrophil extracellular trap formation, and tumor response to GEM. Patients with PDAC who do not respond well to treatment have substantial accumulations of neutrophil extracellular traps in their tumors[62].
Pro-inflammatory cytokines like interleukin-8 (IL-8) are secreted by PDAC cells treated with GEM. By activating CXCR 1/2 on neutrophils, IL-8 facilitates the development of chemotherapy-induced neutrophil extracellular traps (chemoNETosis). Crucially, navarixin therapy considerably decreased tumor growth in vivo, restored sensitivity to GEM, and prevented chemoNETosis. Chemoresistance in PDAC is influenced by neutrophil extracellular traps, and IL-8-mediated chemoNETosis is essential. GEM’s effectiveness is increased by blocking CXCR 1/2-mediated NET formation. In order to overcome chemoresistance in PDAC, this method might be a potential treatment option. These findings encourage more clinical research on anti- neutrophil extracellular traps treatments[62].
LYMPHOCYTES: Lymphocytes are key WBCs in your immune system. Lymphocytes are essential for combating infections (viruses, bacteria) and aberrant cells (cancer). The three major kinds of lymphocytes are B cells (which produce antibodies), T cells (which kill infected cells and coordinate the immune response), and NK cells (innate immunity). PC cells can both evade immunosurveillance and cause immunosuppression in patients. Reduced survival and an advanced stage of PC are linked to low circulating lymphocyte counts. Compared to lymphocytes expressed as an absolute value (LAB > 1500 n°/mm3) or lymphocytes >22%, which is the lowest limit of normal values in laboratory, blood lymphocytes (L) expressed as a percentage of total white blood cells could more accurately predict chemotolerance (n° of tolerated cycles), survival time, and body weight (BW). According to the study, chemotolerance (r = +0.50, P = 0.001), BW (r = +0.35, P = 0.027), and survival (Cox model, using age, sex, BW, serum creatinine, bilirubin, and lymphocytes as covariates) were all better predicted by l ≥ 29.7% than by LAB > 1500 or l > 22%. Chemotolerance and survival did not significantly associated with BW. According to the study’s preliminary findings, l ≥ 29.7% predicts chemotolerance, survival time, and nutritional status better than LAB > 1500 or l > 22%. Given the correlation between BW and L%, a potential influence of dietary status on chemotherapy and survival appears to be lymphocyte-mediated[63].
Both tumor stage and total lymphocyte count (TLC) were revealed to be significant when using Cox regression analysis. The primary immunologic alteration in advanced PC is the decrease in blood total lymphocytes. These patients’ longevity is mostly determined by the tumor stage, but it is also influenced by the number of lymphocytes in circulation, indicating that the immune system is crucial for pancreatic adenocarcinoma immunosurveillance and immunoediting[64].
Tissue size, differentiation, involvement of the resection margin, and lymph node metastases are known prognostic markers for resected PDAC. An investigation into whether preoperative lymphocyte count is a prognostic sign in PDAC patients was conducted. The univariate analysis of the 44 patients revealed the following predictors of poor survival: Lymph node status [node-positive (+ve) 10.3 (5.4-20.9) months vs node-negative (-ve) 14.2 (10.9-31.4) months; P = 0.038], posterior resection margin invasion [margin +ve 7.0 (5.1-15.0) months vs margin -ve 13.1 (10.0-28.3) months; P = 0.025], and lymphocyte count below the reference range [< 1.5 × 109/L, 8.8 (7.0-13.1) months vs ≥ 1.5 × 109/L, 14.3 (7.0-28.3) months; P = 0.029]. Posterior margin invasion and low preoperative lymphocyte count remained significant on multivariate analysis. There was no significant predictive factor associated with the preoperative NLR[65].
B lymphocytes are heavily infiltrated in pancreatic adenocarcinoma, but until recently, the importance of these immune cells in tumor growth was shockingly disregarded. The information that is now available from other solid tumors suggests that B lymphocytes and neoplastic cells do not always interact in a stimulatory or inhibitory manner. Certain subsets of B lymphocytes can secrete growth factors for neoplastic cells and immunosuppressive cytokines, which promote escape from immunosurveillance and cancer progression, even though presentation of tumor antigens to T cells and production of antitumor immunoglobulins may intuitively suggest a prominent tumor suppressive activity. Determining the involvement of B-lymphocytes in this deadly disease becomes crucial to developing new treatment approaches since many of these processes may potentially be linked to the development of PDAC, and immune-modulation of B-cell activity is now achievable at various levels[66]. In both LAPC and resected PC, radiation-induced lymphopenia is linked to a poor prognosis. However, there hasn’t been much research done on how lymphocyte recovery affects the prognosis of PC. It assessed 497 individuals with LAPC receiving concurrent chemoradiotherapy (CCRT)[67].
Up to the 12-month follow-up visit, patients’ CBCs were taken prior to, during, and following CCRT. Based on the development and recovery from acute severe lymphopenia (ASL), patients were divided into three groups: No ASL (group A, n = 198), recovery from ASL (group B, n = 141), and no recovery from ASL (group C, n = 89). Cox regression analysis was used to identify prognostic markers for both OS and PFS. The 2-year OS and PFS rates in groups A, B, and C were 40.4%, 31.9%, and 14.6%, respectively, and 23.7%, 18.4%, and 10.1%, respectively. Group C had poor OS and PFS (P < 0.001), whereas groups A and B had equivalent OS and PFS. Superior OS (HR = 0.42, 95%CI: 0.32-0.55, P < 0.001) and PFS (HR = 0.53, 95%CI: 0.41-0.70, P < 0.001) were linked to recovery from ASL. Development and recovery from ASL were independently predicted by the target volume and baseline lymphocyte numbers. In patients receiving CCRT for LAPC, recovery from ASL was linked to better OS and PFS. Both the onset of treatment-related ASL and its recovery were linked to baseline lymphocyte counts and target volume[67].
Pancreatic abnormalities are linked to high rates of morbidity and death worldwide and produce a wide range of clinical conditions, primarily PC and acute and CP. Unfortunately, there are no targeted therapies for pancreatic illness, and its pathophysiology is still unknown. T cells are essential to multicellular organisms’ adaptive immune systems. Local abnormalities in T-cell subsets in inflammatory and tumor settings, as well as the circulation, have been noted during the development of pancreatic disease. Moreover, it has been demonstrated that T cell-targeting medicines can stop pancreatic disorders from progressing naturally. The clinical significance of T-cell changes as a possible outcome predictor, the underlying processes, and the current state of immunotherapy targeting T cells in pancreatitis and neoplasms have all been covered. The groundbreaking discoveries compiled the significant ramifications for novel medication development as well as the potential use of immunotherapy for pancreatitis and PC[68].
There are few effective therapies for PC, and the prognosis is dismal. The significant heterogeneity of cancer cells inside the tumor contributes to therapeutic failure. T cells use the unique identification of cancer-associated antigens to target and eliminate cancer cells. Deep sequencing was used to characterize T cells from the original tumor and blood of sixteen PC patients. As controls, T cells from the blood of eight more healthy participants were examined. It discovered no discernible variations in the T cell receptor repertoires between patients and healthy controls by examining the complementarity determining region 3 (CDR3) gene sequence. CDR3 types and lengths were comparable between groups. Nonetheless, based on the level of CDR3 overlap within the tumor sample group, two patient clusters were found. Furthermore, compared to patient and healthy control blood samples, clonotypes with low frequencies were much more prevalent in primary pancreatic tumors. The T cell receptor repertoires of pancreatic malignancies in matched blood samples and primary tumors have never been described before. The findings suggest that several forms of PC have potentially significant immunological traits in common[69].
MONOCYTES: Monocytes are the biggest kind of WBC which are produced in the bone marrow and serve as vital innate immune system defenders by scouring the body for pathogens, dead cells, and debris. They help fight infections, remove damaged cells, and initiate adaptive immune responses when they develop into dendritic cells and macrophages (large eaters) in tissues. Among the most deadly cancers, PDAC is often regarded as having the worst prognosis. Immunotherapy has essentially transformed the treatment of cancer, yet it seems to have little use in PDAC. Recently, monocytes have been suggested as possible targets of such immunotherapeutic regimens due to their protumoral activities. However, there is currently little data about monocytes’ involvement in PDAC. Monocyte counts were higher in PDAC patients than in healthy controls. Furthermore, PDAC G3 was linked to greater levels of monocytes than PDAC G2, and patients with perineural infiltration showed a larger percentage of monocytes than those with non-infiltrating tumors. It was shown that patients with monocyte levels above 5% had an 8.9-fold higher probability of developing a G3 and perineural infiltrated PDAC, which would lead to a worse survival rate than patients with levels below 5%. In monocytes, PDAC patients also displayed lower levels of programmed death-ligand 1 and higher expressions of CD86 and CD11c in comparison to healthy people. Last but not least, there was a favorable correlation between monocyte counts and tumor necrosis factor-α and IL-6 levels in PDAC patients’ plasma[70].
PDAC patients initially had higher peripheral blood monocyte (PBM) levels. The mean PBM count (× 109/L) was higher (0.73 vs 0.59; P < 0.001), and monocytosis was more common in PDAC patients at diagnosis than in controls (23% vs 8%; P < 0.001). The mean PBM counts in PDAC patients during the 6 months before diagnosis were substantially higher over the 2-year pre-diagnostic period. The median survival for PDAC subjects with monocytosis upon diagnosis was considerably shorter. At diagnosis, monocytosis is more common in PDAC patients than in controls, and it is linked to a worse median survival. A minority of individuals had elevated PBM counts six months before being diagnosed with PDAC[71].
Although the local microenvironment is reprogrammed in conjunction with PDAC, less is known about the alterations in distant locales. After implanting biomaterial scaffolds, which serve as a synthetic premetastatic niche into immunocompetent tumor-bearing and control mice, it discovered a distinct tumor-specific gene expression profile was discovered that includes elevated expression of C1qa, C1qb, Trem2, and Chil3. Through single-cell RNA sequencing, these genes were linked to two different populations of macrophages in the scaffolds: One with high levels of C1qa, C1qb, and Trem2, and the other with high levels of Chil3, Ly6c2, and Plac8. Compared to macrophages in the normal pancreas, TAM in mice exhibited higher expression of these genes in the respective populations. The expression of C1qa, C1qb, and Trem2 is increased in human macrophages in primary tumors and liver metastases, according to the analysis of single-cell RNA sequencing from patient samples. The same gene profile was much more abundant in monocytes, according to single-cell sequencing study of patient blood. Collectively, it reveals two unique tumor-associated monocyte and macrophage populations that represent systemic immunological alterations in individuals with PDAC[72].
To determine how the systemic inflammatory state in PC patients influences PBMs’ intracellular signaling and how this influences the monocytes’ differentiation into macrophages were reported. Twenty healthy controls and fifty willing PC patients had their monocytes extracted. Using flow cytometry, the phosphorylation status of the signaling molecules from both suitably activated and unstimulated monocytes was evaluated. Patients had decreased phosphorylation levels of activated nuclear factor-κB (NF-κB; P = 0.004) and signal transducer and activator of transcription (STAT) 5 (P = 0.006) compared to controls, but unstimulated STAT2, STAT3, and STAT6 were greater (P < 0.05). Proinflammatory (M1) marker CD86 was expressed more often in co-cultured macrophages obtained from controls than from patients (P = 0.029). Constitutive phosphorylation and reduced responsiveness to stimuli have been observed in circulating monocytes from PC patients, suggesting abnormal activation and immunological suppression. The patient-derived monocytes developed into macrophages with lower levels of the M1 macrophage marker CD86 when co-cultured with cancer cells, indicating weakened anti-tumor characteristics. The findings emphasize the necessity of treating tumor-associated immune abnormalities globally while treating PC[73].
The most prevalent immune cell group in the pancreatic TME is TAMs, which significantly contribute to the TME’s immunosuppressive characteristics. Instead of organ-specific resident macrophages, circulating monocytes are thought to be the main source of TAMs. All cell types produce membrane-bound nanoparticles called EVs that contain bioactive chemicals[74]. The importance of EVs in immune regulation has grown in recent years. To investigate the function of EVs produced from PC cell-derived EVs (cEV) in the recruitment of monocytes to TME and their development into the TAM phenotype was observed. Size exclusion chromatography was used to separate EVs from the pancreatic patient-derived xenograft cell line (PDX188), one PC cell line (Mia PaCa-2), and a non-tumorigenic pancreatic epithelial cell line (hTERT-HPNE)[74].
Using cryo-electron microscopy, nanoparticle tracking analysis, and immunoblot analysis, the EVs were described in accordance with International Society for Extracellular Vesicles recommendations. Multi-omics analysis, flow cytometry, western blot, and cytokine profiling were used in co-culture studies using the human monocyte cell line THP-1 and primary monocytes generated from peripheral blood mononuclear cells to examine the impact of cEVs on monocyte phenotypic differentiation and chemotaxis. By injecting PDAC tumor-bearing mice, the in vivo impact of both normal and cancer-derived EVs on monocyte recruitment was examined. According to proteomics studies, the cargo of cEVs was enriched with proteins linked to myeloid cell activation, but not of EVs produced from HPNE. Furthermore, compared to HPNE-EV-treated monocytes, cEV-treated THP-1 monocytes exhibited noticeably higher amounts of surface CD14, according to flow cytometry data, indicating that cEVs probably promote monocyte differentiation. According to chemotaxis results, THP-1 monocytes treated with cEVs migrated faster in the direction of monocyte chemoattractant protein-1 (C-C motif chemokine ligand 2), which is consistent with in vivo findings that tumor-bearing animals injected with cEVs had more macrophages in their tumors[74].
Western blot and cytokine profile analysis demonstrated that monocytes treated with cEVs released higher levels of anti-inflammatory cytokines, including IL-10 and IL-4 and verified the production of anti-inflammatory proteins like STAT3. A recent discovery that EVs originating from PC carry a particular cargo that, following absorption, improves monocyte recruitment to TME and their subsequent differentiation to TAMs. Research on the fundamental processes is still in progress[74].
Although monocytes and macrophages have antitumor potential, clinicopathological data indicate that they may both promote and inhibit tumor development. This study sought to ascertain how monocytes affected the development of human PC-4 in severe combined immunodeficient (SCID) mice. Human PC-4 cells were coengrafted into SCID mice in different ratios with freshly isolated human monocytes or CD14+ cells from cocultures with tumor cells. Angiogenesis and tumor growth were assessed. Tumor growth was shown to be enhanced at high ratios of monocytes to cancer cells and inhibited at low ratios[75].
Large-scale intratumoral applications of monocytes also accelerated the development of tumors. Tumor development was accelerated by deactivating monocytes by a brief pre-exposure to tumor cells in vitro before engraftment. Tumor-induced angiogenesis was increased by the use of monocytes in high quantities and deactivated monocytes in small amounts. In SCID mice, monocytes may both promote and inhibit the formation of human tumors. The final result of monocyte-tumor interactions depends on both the quantity of monocytes and their level of deactivation[75].
One of the deadliest cancers, PDAC has a very bad prognosis. Giving a thorough grasp of PDAC pathophysiology in light of the host immune response was observed. Using immunohistochemical labeling, flow cytometry, cytokine/chemokine concentration measurements, and gene expression analysis, it investigated the PDAC tissues, sera, and peripheral blood cells of PDAC patients. Macrophages, particularly CD33+ CD163+ M2 macrophages and CD4+ T cells that concurrently express programmed cell death-1 (PD-1), penetrated the PDAC tissues. PDAC patients’ sera had significantly higher levels of IL-6, IL-7, IL-15, monocyte chemotactic protein-1, and interferon-γ-inducible protein-1. There was a difference in the gene expression profile of CD4+ T cells and CD14+ monocytes between PDAC patients and healthy volunteers, and these differences were associated with active inflammation. PD-1 was markedly elevated in PDAC patients’ peripheral blood CD4+ T cells. Accordingly, PDAC patients’ peripheral blood cells had a higher frequency of CD4+ PD-1+ T cells, which was associated with treatment resistance. In conclusion, inflammatory conditions were prevalent in PDAC patients’ peripheral blood cells and PDAC tissue, with monocytes/macrophages and CD4+ T cells having an impact on the clinical prognosis[76].
Eosinophils: The immune system depends on eosinophils, a type of WBC (granulocyte) that mainly fights parasitic infections and regulates allergic reactions, asthma, and inflammation by releasing granule-contained enzymes. However, if their numbers rise too high, they can harm tissue (eosinophilia). They are important markers in blood testing and are typically low in blood, but their concentrations rise with allergies, infections, or some types of cancer. According to several studies, PC patients and CP both had an increase in eosinophils. The importance of eosinophilic pancreatitis, however, is not well known or investigated. Eosinophil accumulation and degranulation contribute to PC and fibrosis. Human pancreatic tissue biopsy samples were utilized for hematoxylin and eosin, anti-major basic protein, anti-tryptase, anti-IgE, and Mason’s trichrome staining. The samples included CP (n = 3), malignant (n = 4), non-malignant (n = 3), and normal (n = 3). The presence of anti-major basic protein stained extracellular granules in both malignant PC and non-malignant human pancreatic tissues indicates the existence of induced eosinophils and degranulated eosinophils. Non-malignant and malignant pancreatic tissue sections showed similar numbers of eosinophils; however, the sections varied in the presence of extracellular granules and degranulated eosinophils. In addition, compared to non-malignant human pancreatic patients, induced mast cells and tissue-specific IgE-positive cells were found in the tissue sections of patients with malignant pancreatitis. The degranulation of mast cells and eosinophils, which may result in a greater buildup of collagen in malignant human pancreatic tissue samples as opposed to non-malignant ones, depends on tissue-specific IgE induction. In the tissue sections of individuals with malignant PC, a high quantity of extracellular granules stained with anti-tryptase was seen. The activation and degranulation of mast cells in tissues have been linked to both IgE and eosinophil major basic proteins. When considered together, research indicates that the accumulation and degranulation of mast cells and eosinophils play a crucial role in the pathophysiology of pancreatitis, which can result in the development of pancreatic fibrosis and cancer[77].
Hypereosinophilia is a sign of malignancy in pancreatic adenocarcinoma and other solid tumor and hematological malignancies, in addition to prodromal symptoms. However, solid organ tumors seldom cause peripheral hypereosinophilia; just four cases of pancreatic adenocarcinoma have been documented so far. It describes a man in his early 50s who had advanced pancreatic adenocarcinoma and severe peripheral hypereosinophilia. This patient’s short 6-week time from diagnosis to death highlights the need to take cancer into account when making a differential diagnosis for a patient who has severe peripheral hypereosinophilia of unknown cause[78].
A strong fibroinflammatory response at the initial tumor site, which is mostly made up of infiltrating immune cells, and survival throughout transit to develop in the secondary site, enables metastasis in PDAC. In comparison to the normal pancreas, it discovered more eosinophils were discovered in the main tumor reactive stroma of both human and animal PDAC. Eosinophils are innate immune cells that have been linked to either pro- or anti-cancer activities in a variety of solid tumors. They are well recognized for their capacity to regulate parasite responses. Using a resectable model of PDAC metastasis, it investigated the function of eosinophils during metastatic dissemination using the ΔdblGata mice, which lack eosinophils. This metastasis paradigm was created to enable consistent lung metastasis seeding in about half of the mice from a temporary subcutaneous tumor that is then surgically removed and harvested to assess metastasis at four weeks. In resectable model of PDAC metastasis, it was observed that animals missing eosinophils had more lung metastases but no change in the development of IV-injected PDAC tumor cells, indicating eosinophil control at the main tumor site. Single-cell sequencing of subcutaneous cancers reveals changes in the tumor cells’ EMT states but no change in the immune cell compartment. In eosinophil knockout mice, staining verifies a lower level of E-cadherin with unaltered vimentin and zeb1, indicating a partial EMT condition. It implies that by regulating the tumor cell state, eosinophils in PDAC prevent the metastatic process[79].
In asthma, chronic sinusitis, and vasculitis, the total eosinophil count in peripheral blood is a type 2 inflammatory measure that is recognized to be crucial. Their involvement in cancer is still unknown despite their dubious antiproliferative activity. In patients with PDAC, another study characterized the association between baseline blood eosinophil and OS. It conducted a retrospective analysis of data from 137 adult patients treated surgically for PDAC between 2012 and 2019[80].
Individuals who did not have intraoperative metastases or a history of systemic steroid usage were included. Based on eosinophil (≥ 0.1 G/L and < 0.1 G/L), patients were divided into two groups. Cox proportional hazards regression models were used to examine survival outcomes. The mOS values by eosinophil and PDAC stage were as follows: 14.5 months for the eosinophil ≥ 0.1 G/L group in stage I-III, 8.0 months for the eosinophil < 0.1 G/L group in stage I-III, 7.0 months for the eosinophil ≥ 0.1 G/L group in stage IV, and 5.0 months for the eosinophil < 0.1 G/L group in stage IV. In both the univariate and multivariate Cox regression, eosinophil < 0.1 G/L (vs ≥ 0.1 G/L) was an independent predictor of OS (HR = 1.48, P = 0.035 and HR = 1.57, P = 0.021)[80].
According to reports, the pathophysiology of CP is facilitated by accumulating macrophages in the pancreas. Cytokines released by macrophages have recently been linked to the promotion of pancreatic acinar-to-ductal metaplasia (ADM). To determine how the NOD-like receptor family pyrin domain containing 3 (NLRP3)-IL-18-eosinophil mechanistic pathway, which is triggered by accumulating macrophages, promotes several aspects of PC in CP was reported. NLRP3-regulated IL-18, which is produced by accumulated macrophages in a murine model of PC, stimulates eosinophilic inflammation-mediated accumulation of periductal mucin and collagen, leading to the development of ADM, PanINs, and IPMN. Most significantly, it demonstrates enhanced malignant traits with decreased oncogene levels in a mouse model of CP that is both IL-18 gene-deficient and anti-IL-18 neutralized. Finally, human biopsies confirmed that NLRP3-IL-18-induced eosinophils build up close to the ducts, demonstrating the production of PanINs in PC. When considered collectively, it shows how IL-18-induced eosinophilia contributes to the emergence of PC phenotypes such as ADM, PanINs, and ductal cell differentiation in inflammation-induced CP[81].
Basophils: More than 140 years ago, Paul Ehrlich discovered basophils in human peripheral blood. Less than 1% of leukocytes in peripheral blood are human basophils. Basophils have also been reported in mice, guinea pigs, rabbits, and monkeys throughout the past few decades. Human and mouse basophils have many characteristics, but they also differ in a number of immunological ways. Currently, there are a number of mouse strains with severe constitutive or inducible basophil deficits that may be used to demonstrate the distinct functions of these cells in vivo. None of these mice, however, is totally and exclusively lacking in basophils. Thus, it is still to be determined if these findings apply to people. The tendency of basophils to move toward the site of inflammation has long been known. According to recent findings, tissue-resident basophils support macrophage M2 polarization locally and aid in lung growth. Furthermore, there is growing evidence that lung-resident basophils vary from circulating basophils in their phenotype. Human and mouse basophils that have been activated produce limited and unique cytokine profiles[82].
Many canonical (like vascular endothelial growth factors and angiopoietin 1) and non-canonical (such cysteinyl leukotriene C4) angiogenic agents are produced by human basophils. Extracellular DNA traps released by activated human and mouse basophils may have a variety of impacts on cancer. Patients with JAK2V617F-positive polycythemia vera have been shown to exhibit hyperresponsiveness of basophils. Basophils can stimulate the formation of inflammation-driven skin tumors and are found in the immunological landscape of PC and human lung adenocarcinoma. The limited investigations that have been done so far employing various basophil-deficient mouse models have yielded insightful findings regarding the functions of these cells in carcinogenesis[82].
Basophils are powerful immune effector cells, even though they make up a relatively tiny percentage of circulating blood leukocytes. Strong inflammatory mediators, including histamine, granzyme B, and cytokines, are stored in dense cytoplasmic granules, ready to be produced in response to a variety of immunological stimuli, and the basophil cell surface expresses the high-affinity receptor for IgE. In allergic disorders and type 1 hypersensitivity, basophils are essential for evoking powerful effector activities. In addition to allergies, basophils can be drawn to tissues in response to viral, bacterial, and parasite infections as well as chronic and autoimmune inflammation. Basophils have not gotten much attention in cancer, although their activation states and functions can be affected by T helper 2 (Th2)-biased inflammatory signals, which are also recognized characteristics of several tumor forms. Here, the existence of basophils and their functional importance in the TME and circulation of cancer patients was observed. The gene expression analysis using publicly accessible information to investigate basophil signatures and correlations with clinical outcomes in a number of cancer types. Additionally, utilizing tests like the basophil activation test, it evaluates how basophils might be used to predict responsiveness to cancer therapies and track patients’ desensitization to oncology medications[83].
Patients with PDAC who have lymphoid infiltrates, primarily Th2 cells, have a poor prognosis. In this context, IL-4 signaling has been proposed to stabilize the Th2 phenotype; however, the cellular source of IL-4 in PDAC is unknown. Here, it demonstrates the enrichment of IL-4-expressing basophils in the tumor-draining lymph nodes (TDLN) of PDAC patients. The Th2/Th1 cell ratio in tumors was substantially associated with basophils found in TDLNs, and these basophils functioned as an independent predictive biomarker of patient survival following surgery. Research in PC animal models verified that basophils play a useful role in the development of tumors. While basophil activation was triggered by T-cell-derived IL-3, basophil migration into TDLN depended in part on the production of chemokine C-C motif chemokine ligand 7/monocyte chemoattractant protein 3 by “alternatively activated” monocytes. The findings shed light on a crucial aspect of the immunological milieu of PC by demonstrating how basophils recruited and activated in TDLNs under the impact of the TME control tumor-promoting Th2 inflammation in PDAC[84].
In recent decades, researchers have disregarded basophils, the rarest granulocytes in peripheral circulation and redundant cousins of mast cells. Their critical functions in parasite infections and allergic illnesses have been demonstrated by earlier research. Interestingly, since activated basophils produce and release a range of cytokines and chemokines in response to malignancies, new research has even shown that basophils may be linked to the development of cancer. The question of whether basophils serve as tumor-promoting or tumor-protecting elements is still up for dispute; the answer may vary depending on the microenvironment and tumor biology[85].
This review article focused on the NLR, MLR, and PLR, LMR, MPV/platelet count ratio, NMR and PWR, MPR pathogenesis in PC. In cases of PC, a high neutrophil-lymphocyte ratio is associated with poor OS. Another study to find out how different hematologic markers, such as NLR in particular, relate to distant metastases, which are a prevalent occurrence in PC[86]. It investigated how the presence of baseline distant metastases and OS related to the ANC, absolute lymphocyte count, absolute eosinophilic count, absolute monocytic count (AMC), NLR, MLR, and PLR. To determine the NLR optimal cutoff value suggestive of its correlation with distant metastases, a ROC curve analysis was plotted[86].
Patients with a high NLR (≥ 3.3) at presentation had more baseline distant metastases than patients with a low NLR (< 3.3) in both univariate and multivariate analyses. In contrast to patients with lower baseline distant metastases, patients with high baseline ANC (≥ 5500/μL), AMC (≥ 600/μL), and MLR (≥ 0.3) had more baseline distant metastases[86]. A lower OS was linked to higher ANC, NLR, MLR, and PLR and lower absolute lymphocyte count. Several hematologic markers, including ANC, NLR, AMC, and MLR, are linked to baseline distant metastases and poor prognoses in PC. It is not known if these immunological phenomena may be used to identify patients who are more likely to develop distant metastases[86].
Prognostic nutritional index (PNI), modified Glasgow prognostic score (GPS), NLR, PLR, LMR and other systemic inflammation-based scores have recently been proposed as prognostic factors for several cancers. To ascertain the impact of dietary status and systemic inflammation-based ratings on the prognosis of patients undergoing chemotherapy for incurable PC was reported[87]. Multivariate analysis revealed that LMR < 3.4 and PNI < 43 independently predicted a poor prognosis in patients undergoing chemotherapy for incurable PC. LMR and PNI alterations before and after treatment showed that individuals whose LMR dropped from ≥ 3.4 to < 3.4 had substantially poorer OS rates than those who maintained LMR ≥ 3.4. Likewise, patients whose PNI declined had far worse OS rates than those who maintained PNI ≥ 43. In patients undergoing treatment for incurable PC, LMR < 3.4 and PNI < 43 are independent indicators of poor prognosis[87].
Another study evaluated the potential of three inflammatory biomarkers in peripheral blood as a prognostic indicator for all patients who had surgery for PC and as a predictor of chemotherapy response in patients with prior illness treated with neoadjuvant chemotherapy. Using retrospective data, it found that the mOS was worse for patients with a NLR (> 5) at diagnosis than for those with ratios ≤ 5 at 13 and 32.4 months, respectively[88]. It was able to observe a modest link between a larger PLR and a greater amount of residual tumor in the histological material in patients undergoing neoadjuvant chemotherapy. Larger prospective studies are required to verify these findings, although it is not unexpected that immune markers might be helpful as possible biomarkers given the dynamic link between the immune system and PC[88].
It should be noted that the pathophysiology and development of cancer depend heavily on the recognition of inflammation. The NLR and the PLR were analyzed with the OS of patients with PDAC who underwent resection either after or without neoadjuvant chemotherapy/chemoradiation. As prognostic indicators for patients having surgery for PDAC, another study aimed to determine the importance of NLR and PLR. According to the univariate analysis, a lower OS was linked to both high NLR and high PLR. The multivariate analysis showed that a lower OS was independently predicted by a high NLR but not by a high PLR. Three-year OS rates for patients who were split into three groups (group 1: Normal both NLR and PLR, group 2: High NLR or high PLR, and group 3: High both NLR and PLR) were 48%, 32% and 7% respectively[89].
Another study examined the predictive significance of the inflammatory markers PLR and NLR in PC patients. 2.0 to 5.0 and 150 to 200 were regarded as high NLR and PLR levels, respectively. The estimated rates of high PLR and NLR, using a random-effects model, were 0.490 and 0.379, respectively. Patients who had surgery and those with lower tumor stages were more likely to have high NLRs. Poor survival rates were significantly associated with high NLR and PLR. In patients receiving chemoradiotherapy, the NLR and PLR showed little predictive value[90].
One early indicator of platelet activation is an elevated MPV. In some malignancies, the MPV and MPV/platelet count ratio have been shown to have diagnostic and prognostic significance. To assess the preoperative MPV/platelet count ratio’s predictive value in PC was observed. The study involved a retrospective analysis of 124 individuals with PC. The patient’s clinicopathological characteristics, prognosis, and preoperative hematological parameters were all reviewed and evaluated. The PC group had considerably higher MPV and MPV/platelet count ratios[91]. Patients with benign pancreatic tumors or healthy controls showed somewhat higher platelet counts than those with PC. The group with a high MPV/platelet count ratio had a considerably lower OS than the other groups, according to univariate analysis (median survival time: 7.0 months vs 13.0 months, log-rank, P < 0.001). A high MPV/platelet count ratio was found to be an independent adverse predictor of OS by multivariate analysis (HR = 3.726, 95%CI: 2.018-6.880, P < 0.001)[91].
The correlation between tumors and the NLR as a predictor of outcome has been documented. Another study assessed the NLR’s predictive significance in PC. As a prognostic factor, the NLR has been linked to tumors in several studies. To examine the prognostic role of the NLR, a meta-analysis was conducted using 95%CI and the HR. A higher NLR was predicted to result in a shorter OS for patients who received therapy, according to the pooled HR of 1.587. An increased NLR may be a predictor of survival for PC patients[92].
To determine the prognostic importance of postoperative immune status alterations in resectable PC, as indicated by the NLR and TLC. After the procedure, their immunologic condition declined for three to four days before improving. OS rates were much lower in the group with low TLC (< 1.66 × 109/L and < 1.62 × 109/L, respectively) and high NLR (> 2.535 and > 3.21, respectively) at 1 month and 6 months after surgery[93]. Increased NLR at postoperative 1 month and 6 months and decreased TLC at postoperative 1 month were significant prognostic factors in multiple regression analysis. Following curative pancreatectomy, immune status changes, such as reduced TLC and raised NLR at postoperative 1 month and 6 months, are useful prognostic indicators for pancreatic adenocarcinoma patients[93].
The prognosis and responsiveness to therapies for PDAC are dismal, even with the latest therapeutic advancements. The lymphocyte to monocyte ratio, one of the most studied prognostic indicators, is receiving more attention in the literature, primarily in relation to hematological malignancies, breast cancer, bladder cancer, colorectal cancer, non-small cell lung cancer, and resected pancreatic adenocarcinoma[94]. A greater LMR in these circumstances enables the identification of a patient subgroup with a better prognosis. In order to determine a cut-off value capable of identifying a subset of patients with a better prognosis and perhaps sensitive to alternative treatment approaches, another study sought to assess the role of the LMR as a prognostic factor in patients affected by metastatic PDAC[94].
There were 123 (54%) men and a median age of 68 years old (range 39-84 years old). The LMR cutoff value was 4. Of the patients, 156 (68.4%) had an LMR < 4 and 72 (31.6%) had an LMR > 4. In terms of mOS, patients with a ratio > 4 differed statistically significantly from those with a ratio < 4 (23 months vs 11 months, P < 0.0001). Patients with a value greater than or equal to 4 had a different first-line median PFS (11 months vs 6 months, P = 0.005), indicating that the first group of patients responded better to therapy. Lastly, LMR > 4 is an independent predictive factor for OS, according to multivariate analysis (P = 0.0005). Additionally, these results point to a subset of patients who would benefit most from a targeted or combination immunotherapy treatment, suggesting the active participation of the immune response in limiting disease progression[94].
The prognosis for PC is dismal. While there are a number of possible indicators for prognosis prediction, it is difficult to predict treatment response. One interesting prognostic indicator is the LMR. Clinical decision-making that incorporates LMR can assist in identifying patients who might need closer observation or more aggressive therapy. In order to forecast bad outcomes and choose the best therapeutic approaches, to evaluate the impact of LMR as an inexpensive, readily available, and trustworthy biomarker on the results of patients receiving chemotherapy for incurable cases was evaluated[95].
The mean lymphocyte and monocyte counts before and after a month of treatment did not differ statistically significantly. Fifty percent of the patients had a full response, and sixteen percent exhibited signs of illness progression. PFS and OS averaged 19.74 and 17.24 months, respectively. Compared to patients with low LMR1 (10.39 months and 10.66 months), individuals with high LMR1 had substantially greater mean OS and PFS (24 months). The mean OS and PFS of patients with low LMR2 and those with high LMR2 differed statistically significantly[95]. Patients with high LMR2 had more complete and partial responses, whereas patients with low LMR2 had more disease progression; this difference was statistically significant. PC patients who had higher pretreatment LMR had better OS, PFS, and chemotherapeutic response[95].
The predictive significance of the NLR in PC has been investigated in a number of studies; however, the findings have been inconsistent. Another study aimed to provide an overview of NLR’s prognostic significance in PC. According to the data (HR = 1.81, 95%CI: 1.59-2.05, P < 0.00001; I2 = 82%), individuals with low NLR may have a longer OS than those with high NLR. Subgroup studies of OS based on the analytic model, ethnicity, therapy, sample size, and cut-off value revealed similar outcomes. Furthermore, in PC, low NLR was substantially linked to a longer DFS than high NLR (HR = 1.66, 95%CI: 1.17-2.35, P = 0.005; I2 = 67%). Additionally, patients with low NLR had substantially smaller tumors (P = 0.0007), better differentiation (P = 0.003), earlier stages (P = 0.02), and lower levels of CA19-9 (P = 0.007)[96].
The possibility of using the LMR as a predictor of the prognosis of patients with resectable PC was investigated. The thorough analysis’s findings show that LMR has substantial prognostic value and is a reliable predictor of OS (HR = 0.55, 95%CI: 0.44-0.69, I2 = 79%, P < 0.00001)[97]. PDAC (HR = 0.73, 95%CI: 0.57-0.93, I2 = 46%, P = 0.01), pancreatic neuroendocrine neoplasms (HR = 0.81, 95%CI: 0.66-0.99, P = 0.04), and other subtypes (HR = 0.40, 95%CI: 0.22-0.72, I2 = 89%, P < 0.00001), but not pancreatic head cancer (HR = 0.46, 95%CI: 0.16-1.13, I2 = 59%, P = 0.12). Asia (HR = 0.62, 95%CI: 0.47-0.76, I2 = 68%, P < 0.0001), Europe (HR = 0.78, 95%CI: 0.67-0.91, I2 = 0%, P = 0.002), and the Americas (HR = 0.14, 95%CI: 0.08-0.24, I2 = 0%, P < 0.0001). Notably, prognostic significance was shown by both LMR cut-off values less than three (HR = 0.47, 95%CI: 0.32-0.69, I2 = 85%, P = 0.0001) and larger than or equal to three (HR = 0.62, 95%CI: 0.47-0.82, I2 = 67%, P = 0.0009)[97]. While LMR did not show predictive significance for RFS (HR = 0.35, 95%CI: 0.09-1.32, I2 = 95%, P = 0.12), the sensitivity analysis for OS validated LMR’s substantial predictive value. LMR was shown to have predictive significance in both subgroups with Newcastle-Ottawa Scale values of ≥ 7 (HR = 0.66, 95%CI: 0.54-0.80, I2 = 53%, P = 0.04) and < 7 (HR = 0.41, 95%CI: 0.23-0.72, I2 = 89%, P < 0.00001)[97].
PC is mostly initiated and progresses through the tumor inflammatory microenvironment. PC patients’ prognosis is correlated with both preoperative PBMs and the LMR. It is unclear how monocytes directly affect PC cells. There was an adverse relationship between a high monocyte count and the 5-year OS and lymph node status in PC. When PC cells were cocultured with THP-1 monocytes, their cellular shape changed and their cell motility increased. The THP-1 monocytes released several proinflammatory cytokines, including IL-1α and tumor necrosis factor-α, which triggered the NF-κB signaling pathway, upregulating Snail and favoring PC cell EMT. The number of PBMs and the expression level of Snail were highly connected, and the 5-year OS was significantly correlated with the level of Snail. It showed that high monocyte counts were a poor prognostic indicator in PC and that monocytes may directly trigger the EMT process in PC cells by upregulating the expression of Snail via the NF-κB signaling pathway[98].
Prognostic markers based on inflammation have been created to forecast the prognosis of PC patients. For patients with incurable PC, including those who were diagnosed without a treatment indication, prognostic indicators have not yet been developed. The PNI, CRP albumin ratio, PLR, NLR, GPS, and modified GPS were all assessed. 3.74 was found to be the NLR cut-off value. Low and high NLR groups had 6-month OS rates of 75.5% and 18.8%, respectively. Prognostic factors in the univariate analysis included advanced age, metastatic PC, no treatment, worse ECOG-PS, high GPS, high modified GPS, high NLR, high PLR, high CRP/albumin ratio, and low PNI. The results of the multivariate analysis showed that OS was independently linked with metastatic PC, no therapy, poorer ECOG-PS, and increased NLR. In patients with incurable PC, it was found that a high NLR may be a stand-alone predictor of a poor prognosis[99].
It has been discovered that the NLR is substantially correlated with the survival of PC. Nevertheless, the relationship between neutrophil count, lymphocyte count, and PC survival was not covered in any preceding research. To examine the impact of neutrophil and lymphocyte counts at the time of illness diagnosis on PC patients’ OS. Restricted cubic spline and the multivariate Cox model were utilized to determine the relationships between OS, neutrophil count, and lymphocyte count of PC. The HR for PC patients with lymphocyte counts below 1.5 × 109/L was 1.82, indicating a strong correlation between OS and a lower lymphocyte count at diagnosis. While OS was not linked to unusually high baseline neutrophil levels of PC, restricted cubic spline discovered that PC patients with baseline neutrophil counts at the cutoff point (7.0 × 109/L) had significantly worse survival. The number of neutrophils and lymphocytes upon diagnosis, particularly the lymphocyte count, may be predictive of PC survival. There should be more discussion of the therapeutic relevance of medications that promote lymphocytes and restrict neutrophils in PC patients[100].
The oncologic prognosis of individuals with PDAC is significantly influenced by immunologic parameters such as the NLR and the PLR. It is predicted that the TLC at the diagnostic stage, which represents host immunity, will affect the oncologic prognosis in left-sided PDAC. At the time of diagnosis, the mean value (standard deviation) of the TLC for left-sided PDAC was 1.8 ± 0.7 103/μL. 1.7 was the cut-off number that most strongly indicated substantial differences in long-term oncologic outcomes. A preoperative lymphocyte count of less than 1.7 was linked to a lower survival rate and an early recurrence[101]. Individuals with a preoperative TLC greater than 1.7 had higher platelet counts, neutrophil counts, monocyte counts and WBC counts. More intriguingly, though, individuals with a TLC below 1.7 had substantially greater NLR and PLR. A negative correlation was seen between the preoperative TLC and the LMR. The only factor shown to differ substantially was age. The two groups did not vary in any clinicopathological criteria, which are often linked to tumor aggressiveness[101].
PLR’s predictive usefulness in PC is still unclear, despite new reports linking PLR to the prognosis of PC. Based on the pooled analysis, a poor OS was predicted by a higher PLR. PLR had a significant prognostic role on OS in the following subgroups: Asian patients treated with chemotherapy and mixed methods, pathological subtype of pancreatic adenocarcinoma, and a cut-off value of PLR ≥ 160. Patients with PC have a worse OS rate when their PLR is increased[102].
Recent research indicates that several inflammatory indicators may have a predictive role in people with PC that is curable. Even though meta-analyses and systematic reviews have examined the significance of different blood indicators, there is ongoing debate over the preoperative PLR’s predictive relevance. According to the investigation, there was no relationship between high PLR and lower OS[103]. A subgroup analysis was conducted due to the considerable heterogeneity among the chosen studies: Low OS was not linked to high PLR in patients who were Caucasian, had a pancreaticoduodenectomy, or had a preoperative PLR greater than 150. However, among Asian patients and those with a preoperative PLR < 150, higher OS was linked to low PLR[103].
The cut-off value of the PLR was established using the receiver operating characteristic curve. For the Kaplan-Meier analysis, the mOS was 19.7 months for PDAC patients with a PLR of 123 or less, while 13.7 months for those with a PLR of 123 or above (P = 0.014). According to Li et al[104], PLR pretreatment may be a prognostic indicator for patients with metastatic PDAC.
A frequent precursor lesion to pancreatic adenocarcinoma is PanIN. There are no established criteria to measure the number of patients with PanIN who are at risk of developing PDAC. The PLR is a measure of inflammation that has been linked to OS in PC patients and other invasive cancers. More severe illnesses in pancreatic neoplasms have been connected to preoperative sarcopenia. Patients with PDAC linked to PanIN had substantially higher PLR. In patients with PLR over 110, invasive carcinomas were much more common, according to the multivariate analysis. Individuals with greater PLR were twice as likely to die during the recovery phase. The highest likelihood of PanIN-associated PDAC was found in patients with sarcopenia, preoperative jaundice, and increased PLR. According to Wagner et al[105], PLR is a predictive indicator that may be used independently to determine whether PanIN-associated invasive cancer is present.
Analyses of OS subgroups based on analytic methodology, ethnicity, sample size, and cut-off value showed similar findings. Low PLR was found to be protective against PC based on additional analyses based on the adjusted potential confounders, which included CA19-9, NLR, modified GPS, albumin, CRP, ECOG, stage, tumor size, nodal involvement, tumor differentiation, margin status, age, and gender. Moreover, in PC, low PLR was substantially linked to a longer PFS than high PLR. PLR is a promising predictive biomarker for PC, and it was discovered that high PLR is an adverse predictor of OS and PFS in patients with the disease[106].
The LMR has been identified in recent studies as a useful predictor of PC prognosis. LMR’s predictive significance for PC is still debatable. Notably, it found that elevated pretreatment LMR predicted improved DFS/RFS/time to progression and OS in PC patients. Through subgroup analyses, it also demonstrated that independent of research design, treatment technique, variable type, ethnicity, LMR cut-off value, and illness stages I-IV and III-IV, high pretreatment LMR was substantially linked with the favourable OS. PC patients who have a high pretreatment LMR also had superior OS and DFS/RFS/time to progression. For individuals with PC, it may thus be a unique prognostic biomarker[107].
An essential factor in the growth of malignancies is inflammation. The predictive significance of a number of serum-based indicators and ratios in PC has been studied. Nevertheless, little research has been done on the predictive significance of the NMR and PWR for PC patients. NMR and PWR had ideal cutoff values of 48 and 6, respectively. According to univariate analysis, individuals with PC who had NMR > 48 and PWR ≤ 6 had a lower survival time than those who had NMR ≤ 48 and PWR > 6 (P < 0.001). NMR (HR = 9.095; 95%CI: 3.64-22.72; P < 0.001) and PWR (HR = 8.230; 95%CI: 3.32-20.43; P < 0.001) showed significant correlations with OS in Cox univariate and multivariate models. NMR and PWR might be new and useful inflammatory prognostic indicators for PC patients[108].
Making the right clinical distinction between PC and mass-forming CP (MFCP) is crucial to choosing the best course of treatment and enhancing patient outcomes. Research indicates that PC and MFCP have distinct inflammatory statuses. Because MPV and platelet count alter under aberrant inflammatory circumstances brought on by tumors, the MPR, a platelet-related inflammation measure, is useful in the diagnosis and prognosis of several tumors[91].
Therefore, another study examined the clinical use of MPR in differentiating MFCP from PC. Based on their pathological diagnosis, the included patients were split into two groups: PC (n = 383) and MFCP (n = 39). MFCP and PC patients were matched using propensity score matching based on age and CA19-9. It examined the differences in MPR between the groups. The best cut-off value was 0.045 with a 69.2% sensitivity and 68.0% accuracy, while the area under the ROC curve between MPR and PC incidence was 0.728 (95%CI: 0.652-0.805). MPRs in the MFCP and PC groups were 0.04 (0.04, 0.06) and 0.06 (0.04, 0.07) for all included patients, respectively (P = 0.005). MPRs in the MFCP and PC groups were 0.04 (0.03, 0.06) and 0.06 (0.05, 0.08) in patients with comparable propensity scores, respectively (P = 0.005)[91].
MPR and CA19-9 were verified as independent risk indicators in differentiating PC by multiple logistic regression in all included patients and matched individuals. When CA19-9 and MPR are combined, the sensitivity and accuracy of PC diagnosis can rise to 93.2% and 89.5%, respectively. MPR may be used as a possible signal to differentiate between MFCP and PC, as it is much greater in PC patients than in MFCP patients. Combining it with CA19-9 can increase its ability to make differential diagnoses[91].
The onset and spread of PC are linked to inflammation. Both locally, anti-inflammatory macrophages and systemically, elevated levels of certain cytokines that modulate inflammation are linked to a poor prognosis in PC. It is yet unknown how systemic inflammation specifically affects circulating monocytes and macrophage polarization[73]. Patients with PC may have a poor prognosis because of weakened host immunity. Currently, there are no reliable prognostic markers for PC, which is linked to high mortality rates and few viable treatments[51].
RDW levels can be used as a marker to indicate the stages of PC in individuals who have been diagnosed[28]. The prognosis of patients with PC is being predicted by a novel noninvasive biomarker. Certain tumor types have been found to have a predictive value for red-cell volume distribution width, a descriptive measure of erythrocyte variance[32]. In PDAC patients, the preoperative lymphocyte count is a major predictive factor[65]. It suggested monocytes as a new prognostic indicator. To further understand the function of monocytes in PDAC and explore their potential as therapeutic targets, more research is required[70].
There is increasing evidence that MCV is a predictive factor for certain cancers[37]. For patients with resectable neoadjuvant-treated PDAC, high MCV is an independent unfavourable prognostic factor that might be a helpful sign to assist doctors in providing individualized prognostication[37]. MCHC is thought to have an impact on the prognosis of several cancers, such as head and neck malignancies and lung cancers[38]. In resectable PC, low MCHC was positively linked with OS, which may be utilized to forecast the prognosis[38].
Numerous studies have documented the correlation between platelet counts and PC as a prognostic factor[42]. A valuable prognostic indicator for PC may be platelet counts[42]. MPV is a separate poor prognostic factor for OS. To sum up, in PC patients with synchronous liver metastases, higher MPV is linked to a worse survival result[45]. In PDAC patients with T2DM, MPV is an independent predictor of poor survival. To validate MPV’s function in PDAC, prospective research is needed[49]. Granulocyte count and WBC pretreatment values were independent variables with a poor prognostic capacity for PC[51]. In patients with advanced pancreatic adenocarcinoma receiving first-line nab-paclitaxel-GEM treatment, the current retrospective research showed the independent prognostic relevance of WBC levels[50].
Neutrophils contribute to the formation and spread of tumors in PDAC and are linked to a poor prognosis. To improve the prognosis for PDAC, immunotherapy targeting neutrophils may be made possible by their complexity, diversity, and adaptability, as well as their intricate interactions with other immune cells and the extracellular matrix[57]. Inflammatory conditions were prevalent in PDAC patients’ peripheral blood cells and PDAC tissue, with monocytes/macrophages and CD4+ T cells having an impact on the clinical prognosis[76]. Peripheral eosinophilia may be an independent predictor. Type 2 inflammation may be the component directly or indirectly extending the longevity of patients with PDAC[80].
In patients undergoing treatment for incurable PC, LMR < 3.4 and PNI < 43 are independent indicators of poor prognosis. Clinicians may find people at high risk for a poor prognosis with the use of LMR and PNI[87]. In patients who have a PDAC resection with or without neoadjuvant chemotherapy or chemoradiation, the pre-treatment NLR is shown to be an independent unfavourable prognostic indicator and is thought to be better than the PLR[89]. Overall, it demonstrated that inflammatory indicators might be used to predict the prognosis of PC patients. Regardless of the patient’s state, the NLR is a better metric for prognostic prediction[90]. NLR is a promising predictive biomarker for PC, and it was found that reduced NLR was a positive predictor of OS and DFS in patients with the disease[96]. In patients with incurable PC, it was found that a high NLR may be a stand-alone predictor of a poor prognosis[99].
All things considered, preoperative total lymphocytes at the diagnostic stage are straightforward and a reliable indicator of prognosis for left-sided PC[101]. In patients with resectable PC, the PLR is not a valid indicator of OS. Nonetheless, low PLR may indicate improved DFS[103]. PLR pretreatment may be a prognostic indicator for patients with metastatic PDAC[102,104]. According to Wagner et al[105], PLR is a predictive indicator that may be used independently to determine whether PanIN-associated invasive cancer is present. PLR is a promising predictive biomarker for PC, and it was discovered that high PLR is an adverse predictor of OS and PFS in patients with the disease[106]. In individuals with PC, a poor prognosis was independently linked to elevated NMR (> 48) and decreased PWR (< 6)[108]. For individuals with PC, a high MPV/platelet count ratio is strongly associated with a poor prognosis[91]. The LMR may be a significant predictive factor in patients with metastatic PDAC[94]. Furthermore, it was discovered that pretreatment LMR could serve as a potential predictor of PC patients’ prognosis[95]. LMR may be a useful predictor of OS in patients with resectable PC, despite the observed heterogeneity and potential biases in the included trials[97].
Hematological and inflammation-based indices derived from routine CBCs are increasingly used in clinical practice due to their low cost, accessibility, and prognostic value. NLR is frequently utilized as an indicator of immunological dysregulation and systemic inflammation. Elevated NLR is clinically linked to poor prognosis, advanced disease stage, treatment resistance, decreased survival in many cancers, and bad outcome illnesses. It is an indicator of neutrophil-driven tumor-promoting inflammation and lymphocyte suppression. PLR is a measure of inflammatory load and platelet activity. Increased PLR has been used therapeutically for risk assessment, prognosis evaluation, and monitoring of inflammatory and thrombotic diseases. It is associated with tumor development, angiogenesis, and metastatic potential in cancer.
The relationship between TAM activity and the anticancer immune response is reflected in LMR. Low LMR is a valuable prognostic and predictive biomarker since it is clinically linked to immune suppression, aggressive disease activity, and poor survival outcomes in cancer patients. CBCs frequently indicate RDW, which is a measure of erythrocyte size fluctuation. Elevated RDW is clinically linked to oxidative stress, chronic inflammation, nutritional deficiencies, and poor prognosis in cancer and critical illness, which helps predict outcomes and measure the severity of diseases. Standard erythrocyte indicators used in ordinary clinical practice to assess anemia subtypes include MCV and MCHC.
Changes in MCV and MCHC are becoming more widely acknowledged in malignancies and chronic inflammatory conditions. These changes may indicate bone marrow malfunction, nutritional inadequacies, or treatment-related side effects, which can aid in the diagnosis and planning of supportive therapy. The HALP score is a composite assessment that incorporates immunological response, inflammatory response, and nutritional status. Low HALP scores are utilized for prognostic classification and therapeutic decision-making in PC patients and are clinically linked to poor performance status, advanced tumor stage, and adverse survival outcomes. An developing biomarker that represents the combined effects of inflammation and anemia is called HRR. Beyond conventional hematological measures, a low HRR has been clinically associated with poor prognosis, increased illness severity, and poor consequences disorders. Together, these indicators are being used more and more in risk assessment, prognosis assessment, therapy response tracking, and individualized clinical decision-making, especially in PC.
This article concluded that RBCs, RDW, MCV, MCHC, platelets, MPV, WBCs and its types such as neutrophils, lymphocytes, monocytes, eosinophils, and basophils, ratios including NLR, MLR, and PLR, LMR, MPV/platelet count ratio, NMR and PWR, MPR play significant role in the pathogenesis of PC as explained in Figure 1 and Table 1. These parameters also serve as valuable, easily accessible clinical markers in PC. Together, they reflect the complex interplay between tumor biology, systemic inflammation, nutritional status, and immune function. Alterations in RBC indices often indicate cancer-related anemia and metabolic disturbances, while elevated platelet counts and MPV highlight tumor-driven platelet activation that supports cancer progression and metastasis. Similarly, changes in WBC counts, especially shifts in neutrophils and lymphocytes, provide important insights into the patient’s inflammatory state and prognosis. Because these hematological parameters are inexpensive, routinely available, and highly informative, they play a crucial role in assessing disease severity, monitoring treatment response, and predicting outcomes in PC patients.
| 1. | Collisson EA, Bailey P, Chang DK, Biankin AV. Molecular subtypes of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2019;16:207-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 776] [Cited by in RCA: 658] [Article Influence: 94.0] [Reference Citation Analysis (3)] |
| 2. | Shou S, Liu R, He J, Jiang X, Liu F, Li Y, Zhang X, En G, Pu Z, Hua B, Pang B, Zhang X. Current and projected incidence rates of pancreatic cancer in 43 countries: an analysis of the Cancer Incidence in Five Continents database. BMJ Open Gastroenterol. 2025;12:e001544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 3. | Pergamo M, Miller G. Myeloid-derived suppressor cells and their role in pancreatic cancer. Cancer Gene Ther. 2017;24:100-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 91] [Article Influence: 10.1] [Reference Citation Analysis (7)] |
| 4. | Stoffel EM, Brand RE, Goggins M. Pancreatic Cancer: Changing Epidemiology and New Approaches to Risk Assessment, Early Detection, and Prevention. Gastroenterology. 2023;164:752-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 404] [Cited by in RCA: 363] [Article Influence: 121.0] [Reference Citation Analysis (7)] |
| 5. | Huang J, Lucero-Prisno DE 3rd, Zhang L, Xu W, Wong SH, Ng SC, Wong MCS. Updated epidemiology of gastrointestinal cancers in East Asia. Nat Rev Gastroenterol Hepatol. 2023;20:271-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 192] [Article Influence: 64.0] [Reference Citation Analysis (4)] |
| 6. | Hesami Z, Olfatifar M, Sadeghi A, Zali MR, Mohammadi-Yeganeh S, Habibi MA, Ghadir MR, Houri H. Global Trend in Pancreatic Cancer Prevalence Rates Through 2040: An Illness-Death Modeling Study. Cancer Med. 2024;13:e70318. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (2)] |
| 7. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69605] [Article Influence: 13921.0] [Reference Citation Analysis (45)] |
| 8. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14765] [Article Influence: 7382.5] [Reference Citation Analysis (21)] |
| 9. | Filho AM, Laversanne M, Ferlay J, Colombet M, Piñeros M, Znaor A, Parkin DM, Soerjomataram I, Bray F. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide. Int J Cancer. 2025;156:1336-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 615] [Cited by in RCA: 476] [Article Influence: 476.0] [Reference Citation Analysis (2)] |
| 10. | Furukawa T. Mechanisms of development and progression of pancreatic neoplasms. Pathol Int. 2022;72:529-540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 11. | Dedushi K, Kabashi S, Mucaj S, Hasbahta G, Ramadani N, Hoxhaj A. Imaging Characteristics and Prevalence of Pancreatic Carcinoma in Kosovo During 2011-2015 - Diagnostic Method as Choice. Acta Inform Med. 2016;24:162-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 12. | Hidalgo M, Cascinu S, Kleeff J, Labianca R, Löhr JM, Neoptolemos J, Real FX, Van Laethem JL, Heinemann V. Addressing the challenges of pancreatic cancer: future directions for improving outcomes. Pancreatology. 2015;15:8-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 380] [Article Influence: 34.5] [Reference Citation Analysis (0)] |
| 13. | Costache MI, Costache CA, Dumitrescu CI, Tica AA, Popescu M, Baluta EA, Anghel AC, Saftoiu A, Dumitrescu D. Which is the Best Imaging Method in Pancreatic Adenocarcinoma Diagnosis and Staging - CT, MRI or EUS? Curr Health Sci J. 2017;43:132-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 29] [Reference Citation Analysis (1)] |
| 14. | Lugada ES, Mermin J, Kaharuza F, Ulvestad E, Were W, Langeland N, Asjo B, Malamba S, Downing R. Population-based hematologic and immunologic reference values for a healthy Ugandan population. Clin Diagn Lab Immunol. 2004;11:29-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 209] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 15. | Rafaqat S, Rafaqat S. Role of hematological parameters in pathogenesis of diabetes mellitus: A review of the literature. World J Hematol. 2023;10:25-41. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 3] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (7)] |
| 16. | Rafaqat S, Zafar A, Tahir H, Khurshid H, Rafaqat S. Pathogenesis of hematological parameters in hypertension. Acta Haematol Pol. 2023;54:389-398. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 17. | Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140:883-899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8980] [Cited by in RCA: 8395] [Article Influence: 524.7] [Reference Citation Analysis (12)] |
| 18. | Diakos CI, Charles KA, McMillan DC, Clarke SJ. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 2014;15:e493-e503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1819] [Cited by in RCA: 1747] [Article Influence: 145.6] [Reference Citation Analysis (4)] |
| 19. | Ammirante M, Luo JL, Grivennikov S, Nedospasov S, Karin M. B-cell-derived lymphotoxin promotes castration-resistant prostate cancer. Nature. 2010;464:302-305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 533] [Cited by in RCA: 495] [Article Influence: 30.9] [Reference Citation Analysis (1)] |
| 20. | Zitvogel L, Apetoh L, Ghiringhelli F, Kroemer G. Immunological aspects of cancer chemotherapy. Nat Rev Immunol. 2008;8:59-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1440] [Cited by in RCA: 1254] [Article Influence: 69.7] [Reference Citation Analysis (1)] |
| 21. | Fu C, Cheng X, Shen Y, Liu X, Huang J, Chen W, He L, Mao Y, Zhao W, Deng Y, Hang D. Red cell indices as predictors of cancer risk: findings from a large prospective cohort study. BMC Cancer. 2025;25:1342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Durak D, Alkurt EG, Topcu R, Kendirci M, Şahiner İT. The significance of haematological parameters and CA 19-9 in assessing vascular invasion and inoperability in pancreatic cancer. J Med Palliat Care. 2022;3:81-86. [DOI] [Full Text] |
| 23. | Xu SS, Li S, Xu HX, Li H, Wu CT, Wang WQ, Gao HL, Jiang W, Zhang WH, Li TJ, Ni QX, Liu L, Yu XJ. Haemoglobin, albumin, lymphocyte and platelet predicts postoperative survival in pancreatic cancer. World J Gastroenterol. 2020;26:828-838. [PubMed] [DOI] [Full Text] |
| 24. | Zeng L, Li S, Yue P, Yi C. [The Value of Clinical Characteristics and Hematological Parameters for Prognostic Assessment of Pancreatic Cancer Patients Undergoing Radical Resection]. Sichuan Da Xue Xue Bao Yi Xue Ban. 2024;55:708-716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 25. | Machairas N, Kostakis I, Papaconstantinou D, Paspala A, Koliakos N, Ntomi V, Zavras N, Misiakos E, Patapis P, Machairas A. White blood cell and platelet indices as indicators of pancreatic tumor type and disease stage. HPB. 2018;20:S551. [DOI] [Full Text] |
| 26. | Yeh CF, Chen KF, Ye JJ, Huang CT. Derivation of a clinical prediction rule for bloodstream infection mortality of patients visiting the emergency department based on predisposition, infection, response, and organ dysfunction concept. J Microbiol Immunol Infect. 2014;47:469-477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 27. | Niu T, Wang Y, Lu L, Li J, Cheng T, Dai Y. The value of preoperative RDW for post-pancreatectomy haemorrhage and surgical prognosis in patients with pancreatic cancer: a retrospective study. BMC Cancer. 2025;25:437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 28. | Yilmaz A, Malya F, Ozturk G, Citgez B, Ozdenkaya Y, Ersavas C, Agan A, Senturk H, Karatepe O. Effect of pre-operative red blood cell distribution on cancer stage and morbidity rate in patients with pancreatic cancer. Int J Clin Exp Med. 2014;7:3072-3075. [PubMed] |
| 29. | Zhou G, Yang L, Lu Y, Lu G. Prognostic value of hemoglobin to red blood cell distribution width ratio in pancreatic ductal adenocarcinoma: a retrospective study. BMC Gastroenterol. 2024;24:288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 30. | Abe T, Amano H, Hanada K, Minami T, Yonehara S, Hattori M, Kobayashi T, Fukuda T, Nakahara M, Ohdan H, Noriyuki T. Perioperative Red Blood Cell Transfusion Is Associated with Poor Long-term Survival in Pancreatic Adenocarcinoma. Anticancer Res. 2017;37:5863-5870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 31. | Wiewiora M, Jopek J, Świętochowska E, Sławomir G, Piecuch J, Gąska M, Piecuch J. Blood-based protein biomarkers and red blood cell aggregation in pancreatic cancer. Clin Hemorheol Microcirc. 2023;85:371-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 32. | Dang C, Wang M, Qin T, Qin R. Clinical importance of preoperative red-cell volume distribution width as a prognostic marker in patients undergoing radical surgery for pancreatic cancer. Surg Today. 2022;52:465-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 33. | Pradeep A, Rameshan A, Krishna Kumar AN, George LS. Association between red cell distribution width and tumour stage in patients with resected cancer of the head of the pancreas. Int J Community Med Public Health. 2020;7:2778. [DOI] [Full Text] |
| 34. | Hong S, Jung YK, Kim S, Yoon J, Choi D, Park B. Decreased use of red blood cell transfusion and associated factors for pancreatic cancer surgery. Ann Hepatobiliary Pancreat Surg. 2025;29:334-342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Halperin R, Ahron-Hananel G, Badarna M, Greidinger D, Uri I, Percik R, Tirosh A. Plasma Hemoglobin and Red Blood Cell Mass Levels as Dynamic Prognostic Markers for Progression and Survival in Pancreatic Neuroendocrine Tumors. Horm Metab Res. 2021;53:810-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 36. | Godfrin Y, Liens D, Andre T, Adenis A, Joly F. L-asparaginase loaded red blood cells (RBC) in pancreatic cancer: A phase I clinical study. J Clin Oncol. 2012;30:e14650. [DOI] [Full Text] |
| 37. | Jomrich G, Gruber M, Gruber ES, Mühlbacher J, Radosavljevic S, Wilfing L, Winkler D, Prager G, Reiterer C, Kabon B, Haslacher H, Sahora K, Schindl M. Prognostic significance of mean corpuscular volume in patients with pancreatic ductal adenocarcinoma and multimodal treatment. J Visc Surg. 2024;161:99-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 38. | Qu X, Zhang T, Ma H, Sui P, Du J. Lower mean corpuscular hemoglobin concentration is associated with unfavorable prognosis of resected lung cancer. Future Oncol. 2014;10:2149-2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 39. | Jurk K, Kehrel BE. Platelets: physiology and biochemistry. Semin Thromb Hemost. 2005;31:381-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 321] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 40. | Mai S, Inkielewicz-Stepniak I. Pancreatic Cancer and Platelets Crosstalk: A Potential Biomarker and Target. Front Cell Dev Biol. 2021;9:749689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 41. | Blümke J, Schameitat M, Verma A, Limbecker C, Arlt E, Kessler SM, Kielstein H, Krug S, Bazwinsky-Wutschke I, Haemmerle M. Innate Immunity and Platelets: Unveiling Their Role in Chronic Pancreatitis and Pancreatic Cancer. Cancers (Basel). 2025;17:1689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 42. | Chen S, Na N, Jian Z. Pretreatment platelet count as a prognostic factor in patients with pancreatic cancer: a systematic review and meta-analysis. Onco Targets Ther. 2018;11:59-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 43. | Chen Z, Wei X, Dong S, Han F, He R, Zhou W. Challenges and Opportunities Associated With Platelets in Pancreatic Cancer. Front Oncol. 2022;12:850485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 44. | Díaz-Blancas JY, Dominguez-Rosado I, Chan-Nuñez C, Melendez-Zajgla J, Maldonado V. Pancreatic Cancer Cells Induce MicroRNA Deregulation in Platelets. Int J Mol Sci. 2022;23:11438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 45. | Yin JB, Wang X, Zhang X, Liu L, Wang RT. Mean platelet volume predicts survival in pancreatic cancer patients with synchronous liver metastases. Sci Rep. 2018;8:6014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (2)] |
| 46. | Yagyu T, Saito H, Sakamoto T, Uchinaka E, Morimoto M, Hanaki T, Watanabe J, Matsunaga T, Yamamoto M, Tokuyasu N, Honjo S, Fujiwara Y. Decreased mean platelet volume predicts poor prognosis in patients with pancreatic cancer. BMC Surg. 2021;21:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 47. | Afsar CU, Gunaldi M, Kum P, Sahin B, Erkisi M, Kara IO, Paydas S, Duman BB, Ercolak V, Karaca F, Uyeturk U, Guner SI. Pancreatic carcinoma, thrombosis and mean platelet volume: single center experience from the southeast region of Turkey. Asian Pac J Cancer Prev. 2014;15:9143-9146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 48. | Karaman K, Bostanci EB, Aksoy E, Kurt M, Celep B, Ulas M, Dalgic T, Surmelioglu A, Hayran M, Akoglu M. The predictive value of mean platelet volume in differential diagnosis of non-functional pancreatic neuroendocrine tumors from pancreatic adenocarcinomas. Eur J Intern Med. 2011;22:e95-e98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 49. | Yin JB, Li N, Cui MM, Wang X, Wang RT. Reduced mean platelet volume levels predict shorter survival in patients with resectable pancreatic ductal adenocarcinoma and type 2 diabetes. BMC Gastroenterol. 2020;20:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 50. | Ioannou A, Demiri S, Papaxoinis G, Sarris E, Klouva E, Baxevanos P, Garefalakis G, Pitsillos N, Kyriakidou A, Mavrogenis I, Goumas G, Gouveris P, Kourakos P, Trfonopoulos D, Koumakis G. P-223 Prognostic role of inflammatory biomarkers in patients with advanced pancreatic adenocarcinoma undergoing first-line chemotherapy. Ann Oncol. 2020;31:S163. [RCA] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 51. | Feng L, Gu S, Wang P, Chen H, Chen Z, Meng Z, Liu L. White Blood Cell and Granulocyte Counts Are Independent Predictive Factors for Prognosis of Advanced Pancreatic Caner. Gastroenterol Res Pract. 2018;2018:8096234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 52. | Karnevi E, Sasor A, Hilmersson KS, Ansari D, Erjefält JS, Rosendahl AH, Andersson R. Intratumoural leukocyte infiltration is a prognostic indicator among pancreatic cancer patients with type 2 diabetes. Pancreatology. 2018;18:85-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 53. | Dinh HQ, Eggert T, Meyer MA, Zhu YP, Olingy CE, Llewellyn R, Wu R, Hedrick CC. Coexpression of CD71 and CD117 Identifies an Early Unipotent Neutrophil Progenitor Population in Human Bone Marrow. Immunity. 2020;53:319-334.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 105] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 54. | Jiang W, Li X, Xiang C, Zhou W. Neutrophils in pancreatic cancer: Potential therapeutic targets. Front Oncol. 2022;12:1025805. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 55. | Jin L, Kim HS, Shi J. Neutrophil in the Pancreatic Tumor Microenvironment. Biomolecules. 2021;11:1170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (2)] |
| 56. | Guo M, Sheng W, Yuan X, Wang X. Neutrophils as promising therapeutic targets in pancreatic cancer liver metastasis. Int Immunopharmacol. 2024;140:112888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 57. | Jin Y, Christenson ES, Zheng L, Li K. Neutrophils in pancreatic ductal adenocarcinoma: bridging preclinical insights to clinical prospects for improved therapeutic strategies. Expert Rev Clin Immunol. 2024;20:945-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 58. | Schernberg A, Vernerey D, Goldstein D, Van Laethem JL, Glimelius B, van Houtte P, Bonnetain F, Louvet C, Hammel P, Huguet F. Predictive Value of Neutrophils Count for Local Tumor Control After Chemoradiotherapy in Patients With Locally Advanced Pancreatic Carcinoma. Int J Radiat Oncol Biol Phys. 2021;110:1022-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 59. | Pratt HG, Steinberger KJ, Mihalik NE, Ott S, Whalley T, Szomolay B, Boone BA, Eubank TD. Macrophage and Neutrophil Interactions in the Pancreatic Tumor Microenvironment Drive the Pathogenesis of Pancreatic Cancer. Cancers (Basel). 2021;14:194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 60. | Xu W, Liu J, Liu Q, Xu J, Zhou L, Liang Z, Huang H, Huang B, Xiao GG, Guo J. NFE2-driven neutrophil polarization promotes pancreatic cancer liver metastasis progression. Cell Rep. 2025;44:115226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 61. | Zhu Y, Zhang F, Ma Y, Deng Y, Yang Y, Tian X. Research progress of pancreatic cancer-associated neutrophils. J Pancreatol. 2025;. [DOI] [Full Text] |
| 62. | Nogi S, Kagawa S, Taniguchi A, Yagi T, Kanaya N, Kakiuchi Y, Yasui K, Fuji T, Kono Y, Kikuchi S, Takagi K, Kuroda S, Teraishi F, Tazawa H, Fujiwara T. Gemcitabine-induced neutrophil extracellular traps via interleukin-8-CXCR1/2 pathway promote chemoresistance in pancreatic cancer. Br J Cancer. 2025;133:1640-1651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 63. | Aquilani R, Brugnatelli S, Maestri R, Boschi F, Filippi B, Perrone L, Barbieri A, Buonocore D, Dossena M, Verri M. Peripheral Blood Lymphocyte Percentage May Predict Chemotolerance and Survival in Patients with Advanced Pancreatic Cancer. Association between Adaptive Immunity and Nutritional State. Curr Oncol. 2021;28:3280-3296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 64. | Fogar P, Sperti C, Basso D, Sanzari MC, Greco E, Davoli C, Navaglia F, Zambon CF, Pasquali C, Venza E, Pedrazzoli S, Plebani M. Decreased total lymphocyte counts in pancreatic cancer: an index of adverse outcome. Pancreas. 2006;32:22-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 195] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 65. | Clark EJ, Connor S, Taylor MA, Madhavan KK, Garden OJ, Parks RW. Preoperative lymphocyte count as a prognostic factor in resected pancreatic ductal adenocarcinoma. HPB (Oxford). 2007;9:456-460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 112] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 66. | Minici C, Testoni S, Della-Torre E. B-Lymphocytes in the Pathophysiology of Pancreatic Adenocarcinoma. Front Immunol. 2022;13:867902. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 67. | Lee BM, Byun HK, Seong J. Significance of lymphocyte recovery from treatment-related lymphopenia in locally advanced pancreatic cancer. Radiother Oncol. 2020;151:82-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 68. | Zhou Q, Tao X, Xia S, Guo F, Pan C, Xiang H, Shang D. T Lymphocytes: A Promising Immunotherapeutic Target for Pancreatitis and Pancreatic Cancer? Front Oncol. 2020;10:382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 69. | Bai X, Zhang Q, Wu S, Zhang X, Wang M, He F, Wei T, Yang J, Lou Y, Cai Z, Liang T. Characteristics of Tumor Infiltrating Lymphocyte and Circulating Lymphocyte Repertoires in Pancreatic Cancer by the Sequencing of T Cell Receptors. Sci Rep. 2015;5:13664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 70. | Hansen FJ, David P, Akram M, Knoedler S, Mittelstädt A, Merkel S, Podolska MJ, Swierzy I, Roßdeutsch L, Klösch B, Kouhestani D, Anthuber A, Bénard A, Brunner M, Krautz C, Grützmann R, Weber GF. Circulating Monocytes Serve as Novel Prognostic Biomarker in Pancreatic Ductal Adenocarcinoma Patients. Cancers (Basel). 2023;15:363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 71. | de la Fuente J, Sharma A, Chari S, Majumder S. Peripheral blood monocyte counts are elevated in the pre-diagnostic phase of pancreatic cancer: A population based study. Pancreatology. 2019;19:1043-1048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 72. | Kemp SB, Steele NG, Carpenter ES, Donahue KL, Bushnell GG, Morris AH, The S, Orbach SM, Sirihorachai VR, Nwosu ZC, Espinoza C, Lima F, Brown K, Girgis AA, Gunchick V, Zhang Y, Lyssiotis CA, Frankel TL, Bednar F, Rao A, Sahai V, Shea LD, Crawford HC, Pasca di Magliano M. Pancreatic cancer is marked by complement-high blood monocytes and tumor-associated macrophages. Life Sci Alliance. 2021;4:e202000935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 73. | Juusola M, Kuuliala K, Kuuliala A, Mustonen H, Vähä-Koskela M, Puolakkainen P, Seppänen H. Pancreatic cancer is associated with aberrant monocyte function and successive differentiation into macrophages with inferior anti-tumour characteristics. Pancreatology. 2021;21:397-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 74. | Cheema AK, Zhang YR, Singh B, Bose P, Kandhavelu J, Deol S, Jayatilake M, Wang S. Abstract A044: Pancreatic cancer cell-derived EVs facilitate monocyte recruitment to the tumor site. Cancer Res. 2025;85:A044. [DOI] [Full Text] |
| 75. | Mytar B, Baj-Krzyworzeka M, Majka M, Stankiewicz D, Zembala M. Human monocytes both enhance and inhibit the growth of human pancreatic cancer in SCID mice. Anticancer Res. 2008;28:187-192. [PubMed] |
| 76. | Komura T, Sakai Y, Harada K, Kawaguchi K, Takabatake H, Kitagawa H, Wada T, Honda M, Ohta T, Nakanuma Y, Kaneko S. Inflammatory features of pancreatic cancer highlighted by monocytes/macrophages and CD4+ T cells with clinical impact. Cancer Sci. 2015;106:672-686. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 77. | Manohar M, Verma AK, Venkateshaiah SU, Mishra A. Significance of Eosinophils in Promoting Pancreatic malignancy. J Gastroenterol Pancreatol Liver Disord. 2017;5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 78. | Cohen EM, Russeth TE, Thati N. Hypereosinophilia as a presenting sign of advanced pancreatic adenocarcinoma: a rare, severe presentation. BMJ Case Rep. 2023;16:e256235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 79. | Hoffman MT, Lenehan PJ, Bertocchi A, Wang J, Ali L, Qiang L, Uchida A, Vayrynen S, Costa AD, Dougan M, Heckler M, Nowak J, Wolpin BW, Dougan SK. Abstract B065: Eosinophils alter metastatic spread in pancreatic cancer. Cancer Res. 2024;84:B065. [DOI] [Full Text] |
| 80. | Ciesielski W, Kupryś-Lipińska I, Kumor-Kisielewska A, Grząsiak O, Borodacz J, Niedźwiecki S, Hogendorf P, Durczyński A, Strzelczyk J, Majos A. Peripheral Eosinophil Count May Be the Prognostic Factor for Overall Survival in Patients with Pancreatic Ductal Adenocarcinoma Undergoing Surgical Treatment. Biomedicines. 2024;12:2596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 81. | Kandikattu HK, Manohar M, Verma AK, Kumar S, Yadavalli CS, Upparahalli Venkateshaiah S, Mishra A. Macrophages-induced IL-18-mediated eosinophilia promotes characteristics of pancreatic malignancy. Life Sci Alliance. 2021;4:e202000979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 82. | Marone G, Schroeder JT, Mattei F, Loffredo S, Gambardella AR, Poto R, de Paulis A, Schiavoni G, Varricchi G. Is There a Role for Basophils in Cancer? Front Immunol. 2020;11:2103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 83. | Chauhan J, Stavraka C, Grandits M, Palhares LCGF, Josephs DH, Lacy KE, Spicer J, Bax HJ, Karagiannis SN. Clinical and Translational Significance of Basophils in Patients with Cancer. Cells. 2022;11:438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 84. | De Monte L, Wörmann S, Brunetto E, Heltai S, Magliacane G, Reni M, Paganoni AM, Recalde H, Mondino A, Falconi M, Aleotti F, Balzano G, Algül H, Doglioni C, Protti MP. Basophil Recruitment into Tumor-Draining Lymph Nodes Correlates with Th2 Inflammation and Reduced Survival in Pancreatic Cancer Patients. Cancer Res. 2016;76:1792-1803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 131] [Article Influence: 13.1] [Reference Citation Analysis (1)] |
| 85. | Zhang J, Yin H, Chen Q, Zhao G, Lou W, Wu W, Pu N. Basophils as a potential therapeutic target in cancer. J Zhejiang Univ Sci B. 2021;22:971-984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 86. | Abu-Shawer O, Abu-Shawer M, Shurman A, Lattouf A, Haimour A, Hamdan O, Mansour R, Altamimi T, Al-Hussaini M. The clinical value of peripheral immune cell counts in pancreatic cancer. PLoS One. 2020;15:e0232043. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 87. | Shimizu T, Taniguchi K, Asakuma M, Tomioka A, Inoue Y, Komeda K, Hirokawa F, Uchiyama K. Lymphocyte-to-Monocyte Ratio and Prognostic Nutritional Index Predict Poor Prognosis in Patients on Chemotherapy for Unresectable Pancreatic Cancer. Anticancer Res. 2019;39:2169-2176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 88. | Maloney S, Pavlakis N, Itchins M, Arena J, Mittal A, Hudson A, Colvin E, Sahni S, Diakos C, Chan D, Gill AJ, Samra J, Clarke SJ. The Prognostic and Predictive Role of the Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Lymphocyte-to-Monocyte Ratio (LMR) as Biomarkers in Resected Pancreatic Cancer. J Clin Med. 2023;12:1989. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 89. | Giakoustidis A, Neofytou K, Costa Neves M, Giakoustidis D, Louri E, Cunningham D, Mudan S. Identifying the role of neutrophil-to-lymphocyte ratio and platelets-to-lymphocyte ratio as prognostic markers in patients undergoing resection of pancreatic ductal adenocarcinoma. Ann Hepatobiliary Pancreat Surg. 2018;22:197-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 90. | Oh D, Pyo JS, Son BK. Prognostic Roles of Inflammatory Markers in Pancreatic Cancer: Comparison between the Neutrophil-to-Lymphocyte Ratio and Platelet-to-Lymphocyte Ratio. Gastroenterol Res Pract. 2018;2018:9745601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 91. | Gong X, Zhang L, Yang D, Xiao X, Wei L, Wu L, Xu J, Pan S. Mean platelet volume/platelet count ratio is associated with prognosis in patients with pancreatic cancer. Int J Clin Exp Med. 2016;9:16379-16386. |
| 92. | Cheng H, Long F, Jaiswar M, Yang L, Wang C, Zhou Z. Prognostic role of the neutrophil-to-lymphocyte ratio in pancreatic cancer: a meta-analysis. Sci Rep. 2015;5:11026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 93. | Kim EY, Hong TH. Changes in total lymphocyte count and neutrophil-to-lymphocyte ratio after curative pancreatectomy in patients with pancreas adenocarcinoma and their prognostic role. J Surg Oncol. 2019;120:1102-1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 94. | Pretta A, Spanu D, Giampieri R, Lai E, Cimbro E, Pecci F, Balconi F, Lupi A, Pozzari M, Murgia S, Bardanzellu F, Maccioni A, Contu F, Persano M, Donisi C, Valeria P, Puzzoni M, Ziranu P, Berardi R, Scartozzi M. Lymphocyte to monocyte ratio in metastatic pancreatic ductal adenocarcinoma as a prognostic factor and its potential role in identifying a subset of patients with a favorable response to therapy. J Clin Oncol. 2022;40:4153. [DOI] [Full Text] |
| 95. | Hegazy H, Tawfik N, Arafat W. 379P The role of lymphocyte to monocyte ratio as a prognostic index to predict poor prognosis in patients with unresectable pancreatic cancer treated with chemotherapy. Ann Oncol. 2024;35:S153. [DOI] [Full Text] |
| 96. | Zhou Y, Wei Q, Fan J, Cheng S, Ding W, Hua Z. Prognostic role of the neutrophil-to-lymphocyte ratio in pancreatic cancer: A meta-analysis containing 8252 patients. Clin Chim Acta. 2018;479:181-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 120] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 97. | Li H, Peng S, An R, Du N, Wu H, Zhen X, Gao Y, Li Z, Min J. The prognostic role of lymphocyte-to-monocyte ratio in patients with resectable pancreatic cancer: a systematic review and meta-analysis. PeerJ. 2024;12:e17585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 98. | Chen S, Huang F, He C, Li J, Chen S, Li Y, Chen Y, Lian G, Huang K. Peripheral blood monocytes predict clinical prognosis and support tumor invasiveness through NF-κB-dependent upregulation of Snail in pancreatic cancer. Transl Cancer Res. 2021;10:4773-4785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 99. | Iwai N, Okuda T, Sakagami J, Harada T, Ohara T, Taniguchi M, Sakai H, Oka K, Hara T, Tsuji T, Komaki T, Kagawa K, Yasuda H, Naito Y, Itoh Y. Neutrophil to lymphocyte ratio predicts prognosis in unresectable pancreatic cancer. Sci Rep. 2020;10:18758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 100. | Xiao Y, Xie Z, Shao Z, Chen W, Xie H, Qin G, Zhao N. Neutrophil and lymphocyte counts at diagnosis are associated with overall survival of pancreatic cancer: A retrospective cohort study. Medicine (Baltimore). 2016;95:e5024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 101. | Rho SY, Hwang HK, Chong JU, Yoon DS, Lee WJ, Kang CM. Association of preoperative total lymphocyte count with prognosis in resected left-sided pancreatic cancer. ANZ J Surg. 2019;89:503-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 102. | Li W, Tao L, Lu M, Xiu D. Prognostic role of platelet to lymphocyte ratio in pancreatic cancers: A meta-analysis including 3028 patients. Medicine (Baltimore). 2018;97:e9616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 103. | Riauka R, Ignatavicius P, Barauskas G. Preoperative Platelet to Lymphocyte Ratio as a Prognostic Factor for Resectable Pancreatic Cancer: A Systematic Review and Meta-Analysis. Dig Surg. 2020;37:447-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 104. | Li W, Chen Y, Wang X, Shi Y, Dai G, Li X. Pretreatment platelet to lymphocyte ratio is predictive of overall survival in metastatic pancreatic ductal adenocarcinoma. Transl Cancer Res. 2019;8:17-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 105. | Wagner D, Haybaeck J, Wienerroither V, Bajric T, Tomberger A, Schemmer P, Mischinger HJ, Kornprat P. Platelet to Lymphocyte Ratio Correlates With Carcinoma Progression in Pancreatic Intra Epithelial Neoplasia. Anticancer Res. 2022;42:1413-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 106. | Zhou Y, Cheng S, Fathy AH, Qian H, Zhao Y. Prognostic value of platelet-to-lymphocyte ratio in pancreatic cancer: a comprehensive meta-analysis of 17 cohort studies. Onco Targets Ther. 2018;11:1899-1908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (2)] |
| 107. | Lin S, Fang Y, Mo Z, Lin Y, Ji C, Jian Z. Prognostic value of lymphocyte to monocyte ratio in pancreatic cancer: a systematic review and meta-analysis including 3338 patients. World J Surg Oncol. 2020;18:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 108. | Tang F, Dai P, Wei Q, Gan K, Wang Z, Chen H, Li T, Lv M, Deng M, Luo G. The Neutrophil-to-Monocyte Ratio and Platelet-to-White Blood Cell Ratio Represent Novel Prognostic Markers in Patients with Pancreatic Cancer. Gastroenterol Res Pract. 2021;2021:6693028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |